Bioh2, Heat and Power from Palm Empty Fruit Bunch via Pyrolysis-Autothermal Reforming: Plant Simulation, Experiments, and CO2 Mitigation
Abstract
:1. Introduction
2. Methods
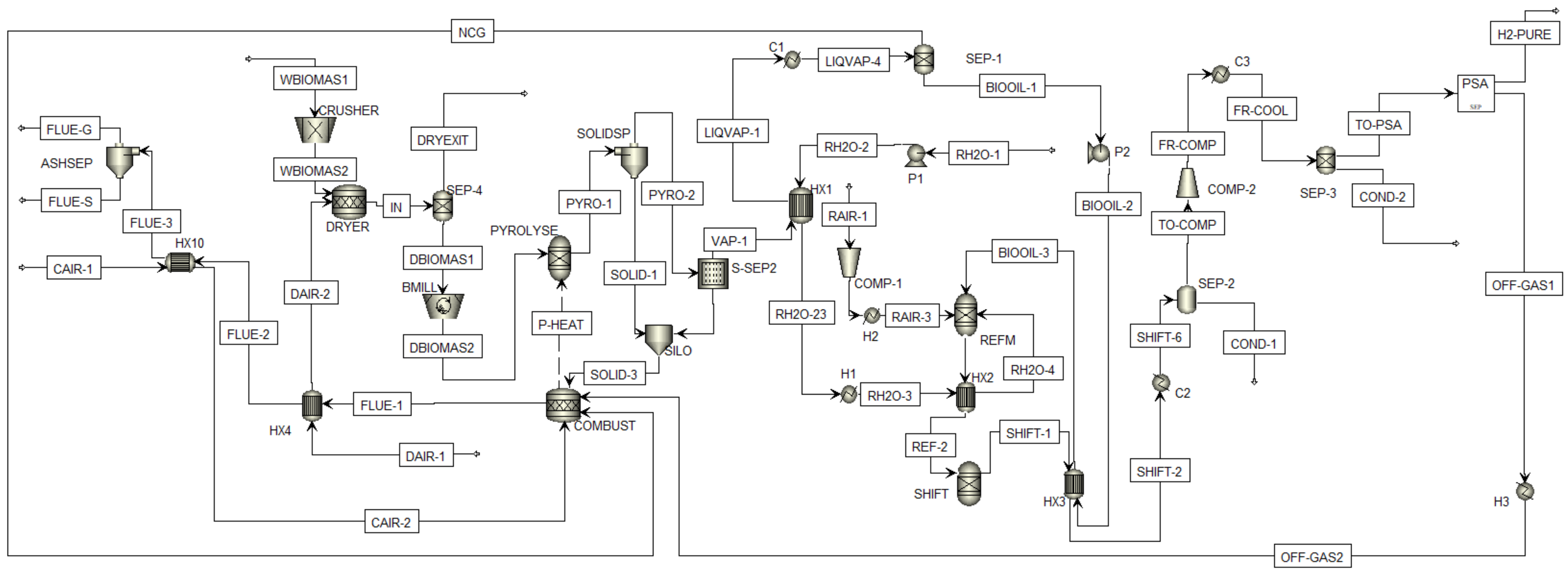
2.1. Process Description
2.2. Process Design in Aspen Plus
2.3. Thermodynamic Method
2.4. Biomass Specification
2.5. Aspen Plus Blocks Specification
2.6. Equations
2.7. Experimental
3. Results
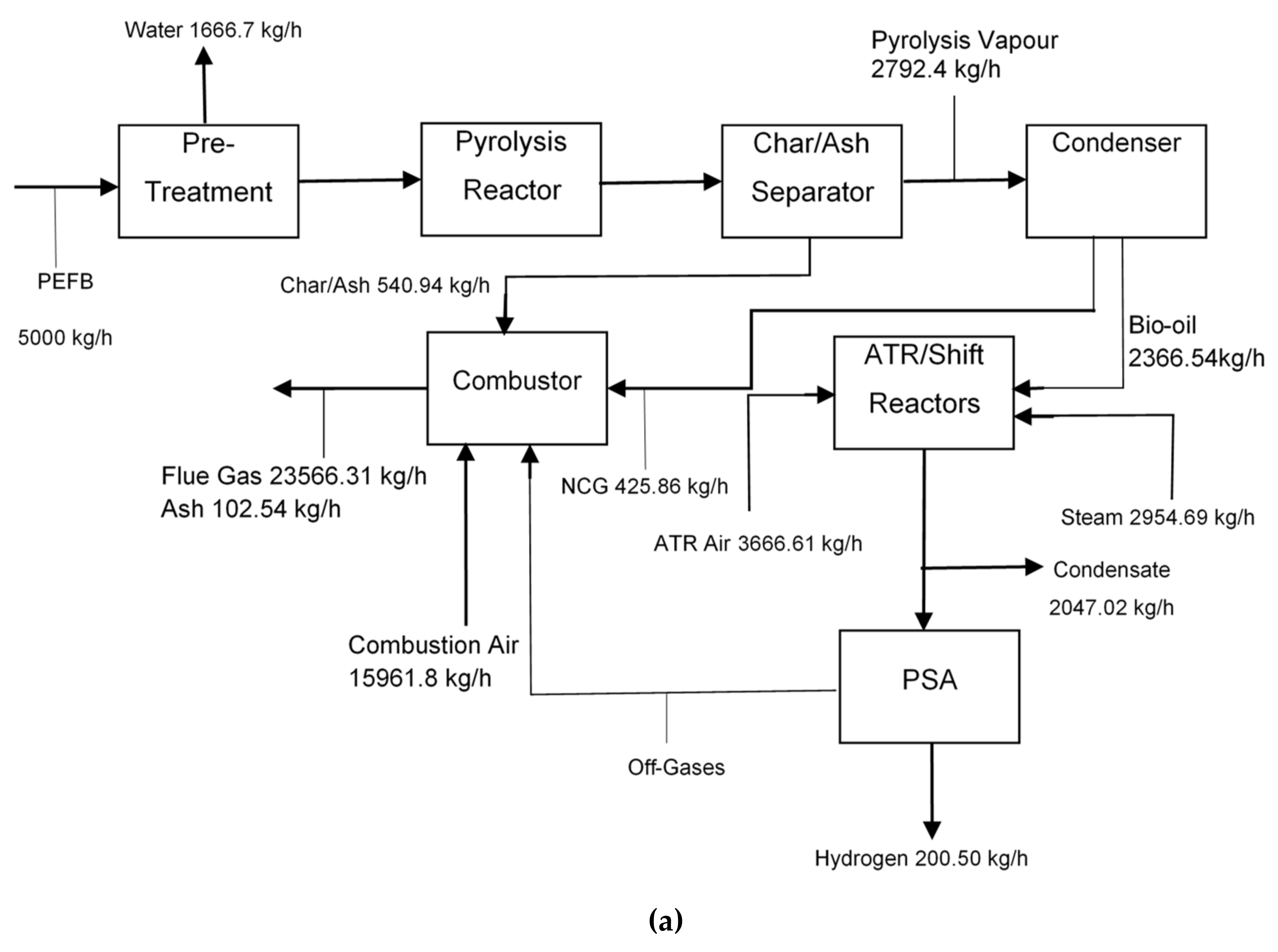
3.1. Process and Plant Modelling
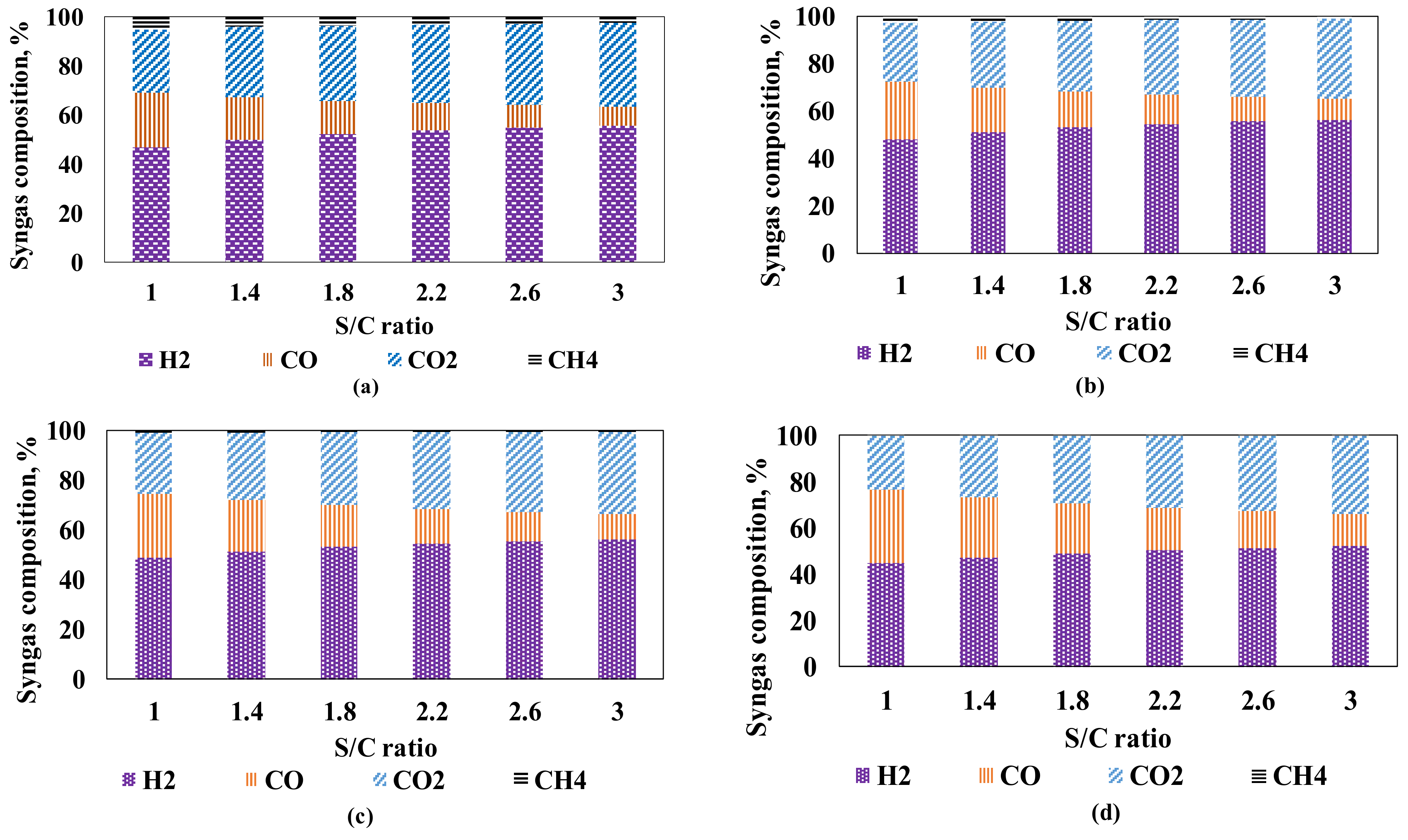
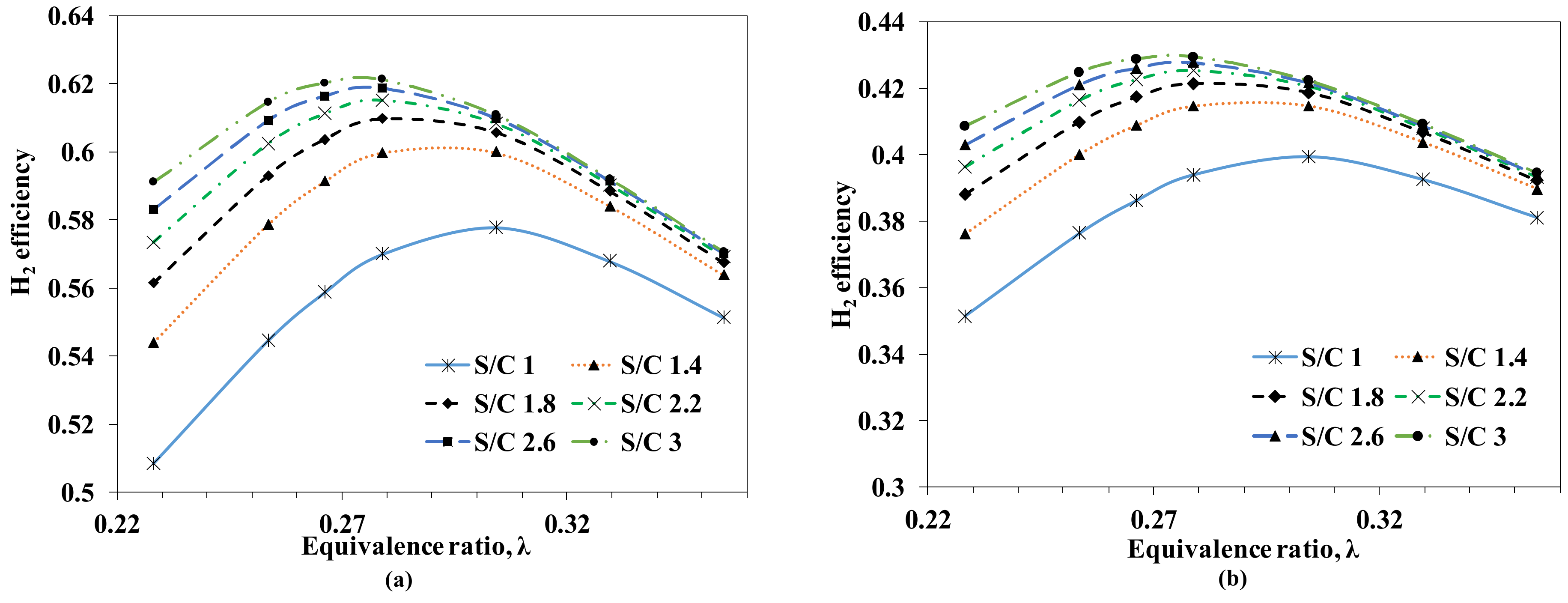
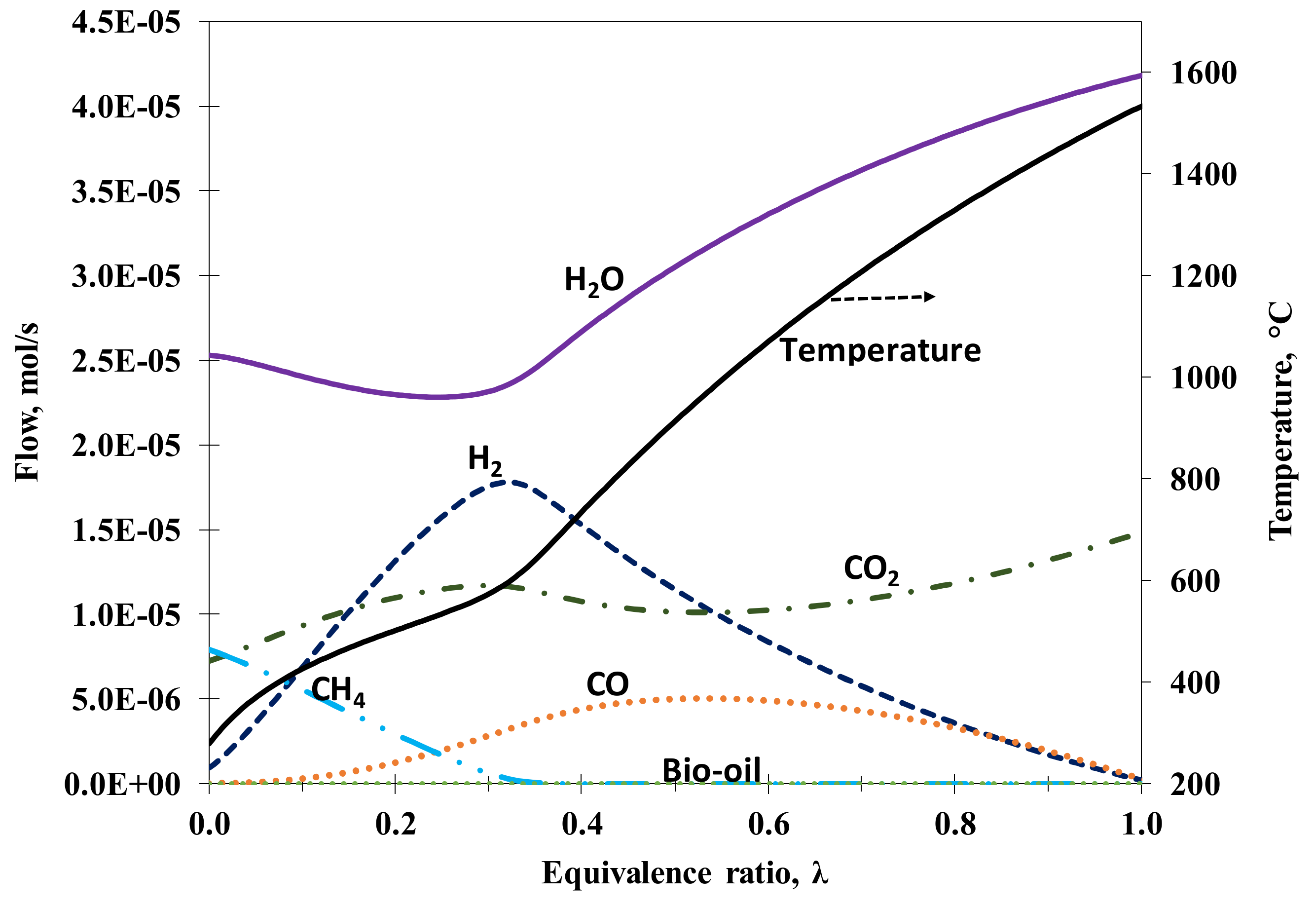
3.1.1. Influence of Steam and Air Feeds at the Autothermal Reformer
Full Plant’s Hydrogen Yield
Dry Gas Composition at the Autothermal Reformer (Syngas Composition)
Hydrogen (Thermal) Efficiency
3.1.2. Overall Process Plant Efficiency
3.2. Model Sensitivity Analyses
3.2.1. Pyrolysis Product Distribution
3.2.2. Pressure
3.2.3. Oxygen Feed to ATR from an Air Separation Unit (ASU)
3.3. CO2 Emission Savings
3.4. Experiments of Bio-Oil ATR
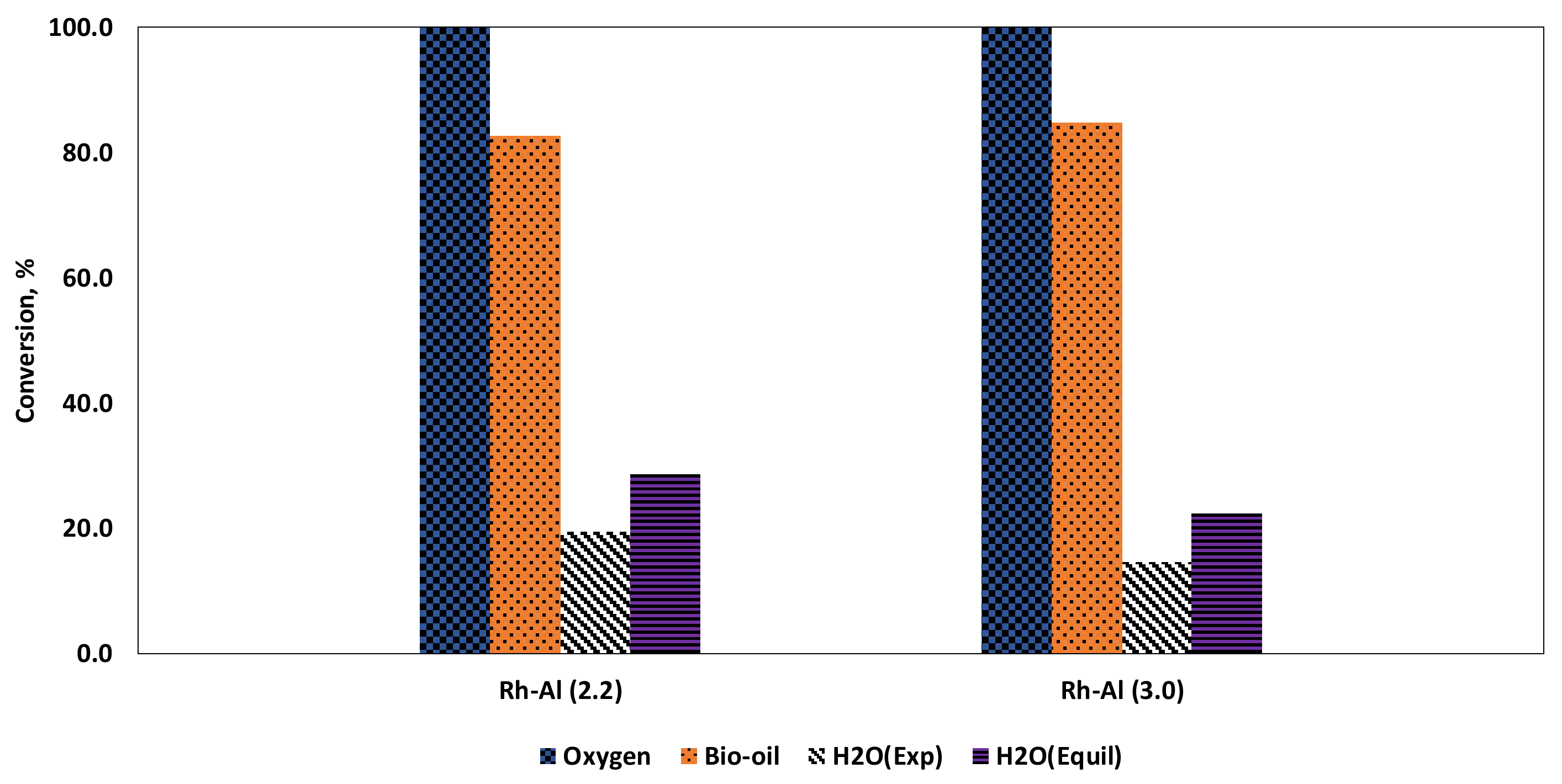
3.4.1. Feed Flows Conversion
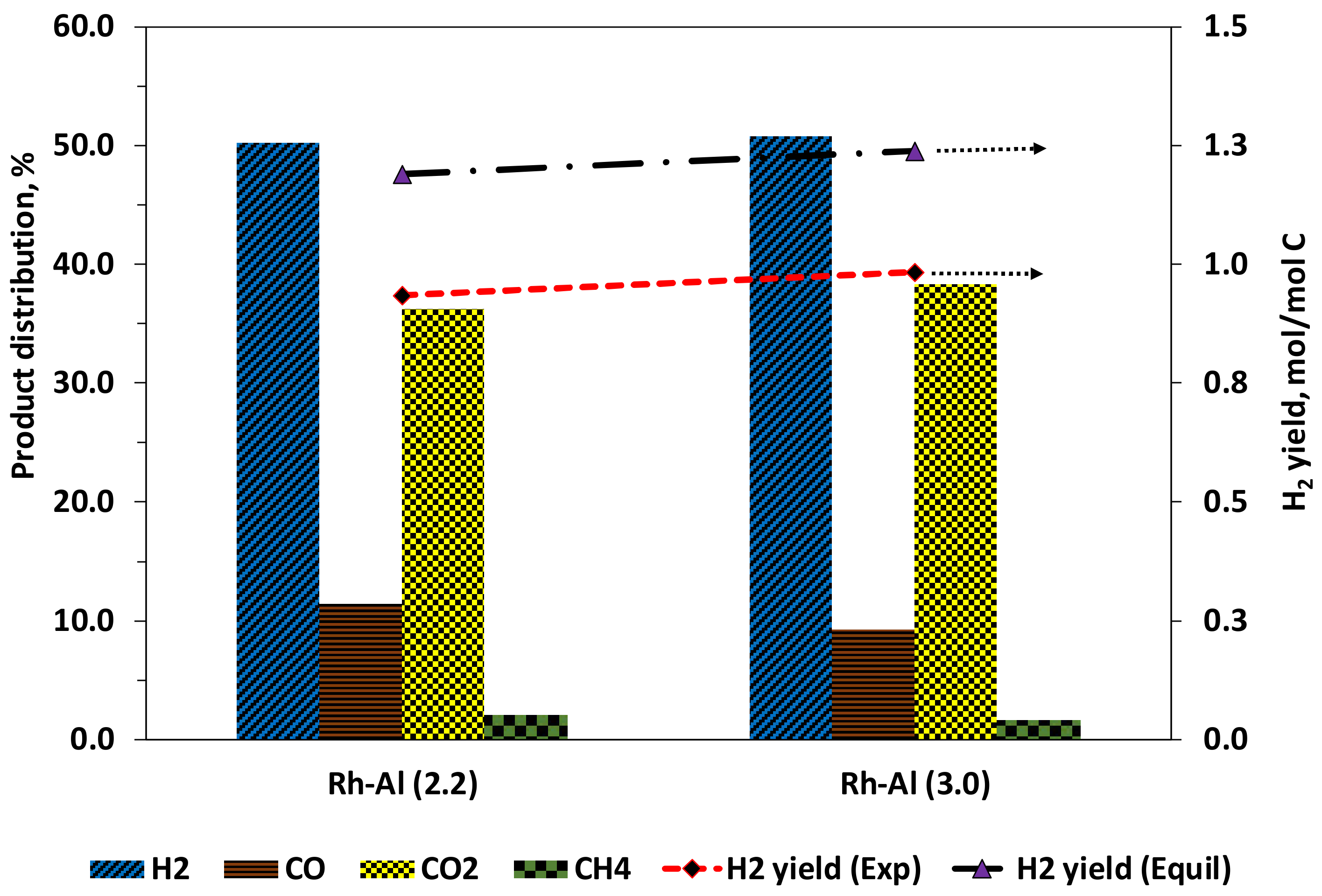
3.4.2. Product Distribution and Selectivity
3.4.3. Optimisation of Bio-Oil Conversion by Increased Air Feed
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kırtay, E. Recent advances in production of hydrogen from biomass. Energy Convers. Manag. 2011, 52, 1778–1789. [Google Scholar] [CrossRef]
- Balat, H.; Kırtay, E. Hydrogen from biomass—Present scenario and future prospects. Int. J. Hydrogen Energy 2010, 35, 7416–7426. [Google Scholar] [CrossRef]
- Demirbas, M.F. Biofuels from algae for sustainable development. Appl. Energy 2011, 88, 3473–3480. [Google Scholar] [CrossRef]
- Chattanathan, S.A.; Adhikari, S.; Abdoulmoumine, N. A review on current status of hydrogen production from bio-oil. Renew. Sustain. Energy Rev. 2012, 16, 2366–2372. [Google Scholar] [CrossRef]
- REN21. REN12 Renewables 2020 Global Status Report; REN21 Secretariat: Paris, France, 2020. [Google Scholar]
- Chang, A.C.; Chang, H.F.; Lin, F.J.; Lin, K.H.; Chen, C.H. Biomass gasification for hydrogen production. Int. J. Hydrogen Energy 2011, 36, 14252–14260. [Google Scholar] [CrossRef]
- Bridgwater, A.V.; Meier, D.; Radlein, D. An overview of fast pyrolysis of biomass. Org. Geochem. 1999, 30, 1479–1493. [Google Scholar] [CrossRef]
- Czernik, S.; Evans, R.; French, R. Hydrogen from biomass-production by steam reforming of biomass pyrolysis oil. Catal. Today 2007, 129, 265–268. [Google Scholar] [CrossRef]
- Martin, S.; Albrecht, F.G.; Van der Veer, P.; Lieftink, D.; Dietrich, R.U. Evaluation of on-site hydrogen generation via steam reforming of biodiesel: Process optimization and heat integration. Int. J. Hydrogen Energy 2016, 41, 6640–6652. [Google Scholar] [CrossRef]
- Holladay, J.D.; Hu, J.; King, D.I.; Wang, Y. An overview of hydrogen production technologies. Catal. Today 2009, 139, 244–260. [Google Scholar] [CrossRef]
- Tande, L.N.; Dupont, V. Autothermal reforming of palm empty fruit bunch bio-oil: Thermodynamic modelling. AIMS Energy 2016, 4, 68–92. [Google Scholar] [CrossRef]
- Arandia, A.; Remiro, A.; Oar-Arteta Gonzalez, L.; Bilbao, J.; Gayubo, A.G. Reaction conditions effect and pathways in the oxidative steam reforming of raw bio-oil on a Rh/CeO2-ZrO2 catalyst in a fluidized bed reactor. Int. J. Hydrogen Energy 2017, 42, 29175–29185. [Google Scholar] [CrossRef]
- Remiro, A.; Arandia, A.; Bilbao, J.; Gayubo, A.G. Comparison of Ni Based and Rh Based Catalyst Performance in the Oxidative Steam Reforming of Raw Bio-Oil. Energy Fuels 2017, 31, 7147–7156. [Google Scholar] [CrossRef]
- Acevedo-Paez, J.C.; Duran, J.M.; Posso, F.; Arenas, E. Hydrogen production from palm kernel shell: Kinetic modeling and simulation. Int. J. Hydrogen Energy 2020, 45, 25689–25697. [Google Scholar] [CrossRef]
- Ishaq, H.; Dincer, I. A new energy system based on biomass gasification for hydrogen and power production. Energy Rep. 2020, 6, 771–781. [Google Scholar] [CrossRef]
- Yun, H.A.H.; Ramirez-Solis, S.; Dupont, V. Bio-CH4 from palm empty fruit bunch via pyrolysis-direct methanation: Full plant model and experiments with bio-oil surrogate. J. Clean. Prod. 2020, 244, 118737. [Google Scholar] [CrossRef]
- Doherty, W.; Reynolds, A.; Kennedy, D. Computer simulation of a biomass gasification-solid oxide fuel cell power system using Aspen Plus. Energy 2010, 35, 4545–4555. [Google Scholar] [CrossRef] [Green Version]
- Dufour, A.; Authier, O.; Mauviel, G.; Corriou, J.P.; Verdier, G.; Abdelouahed, L. Detailed Modeling of Biomass Gasification in Dual Fluidized Bed Reactors under Aspen Plus. Energy Fuels 2012, 26, 3840–3855. [Google Scholar]
- Onarheim, K.; Solantausta, Y.; Lehto, J. Process Simulation Development of Fast Pyrolysis of Wood Using Aspen Plus. Energy Fuels 2015, 29, 205–217. [Google Scholar]
- Ward, J.; Rasul, M.G.; Bhuiya, M.M.K. Energy Recovery from Biomass by Fast Pyrolysis. Procedia Eng. 2014, 90, 669–674. [Google Scholar] [CrossRef] [Green Version]
- Doherty, W.; Reynolds, A.; Kennedy, D. Aspen Plus Simulation of Biomass Gasification in a Steam Blown Dual Fluidised Bed, Book Chapter in Materials and Processes for Energy: Communicating Current Research and Technological Developments; Méndez-Vilas, A., Ed.; Formatex Research Centre: Badajoz, Spain, 2013. [Google Scholar]
- Erlach, B.; Harder, B.; Tsatsaronis, G. Combined hydrothermal carbonization and gasification of biomass with carbon capture. Energy 2012, 45, 329–338. [Google Scholar] [CrossRef]
- Gautam, N.; Chaurasia, A. Study on kinetics and bio-oil production from rice husk, rice straw, bamboo, sugarcane bagasse and neem bark in a fixed-bed pyrolysis process. Energy 2020, 190, 116434. [Google Scholar] [CrossRef]
- Ighalo, J.O.; Adeniyi, A.G. Modelling of thermochemical energy recovery processes for switchgrass (Panicum virgatum). Indian Chem. Eng. 2020, 63, 240–251. [Google Scholar] [CrossRef]
- Mohammed, I.Y.; Abakr, Y.A.; Mokaya, R. Integrated biomass thermochemical conversion for clean energy production: Process design and economic analysis. J. Environ. Chem. Eng. 2019, 7, 103093. [Google Scholar] [CrossRef]
- Wang, X.Y.; Panahi, A.; Qi, H.X.; Zhai, M.; Dong, P.; Levendis, Y.A. Product Compositions from Sequential Biomass Pyrolysis and Gasification of Its Char Residue. J. Energy Eng. 2020, 146, 04020049. [Google Scholar] [CrossRef]
- Bridgwater, A.V. Review of fast pyrolysis of biomass and product upgrading. Biomass Bioenergy 2012, 38, 68–94. [Google Scholar] [CrossRef]
- Sarkar, S.; Kumar, A. Large-scale biohydrogen production from bio-oil. Bioresour. Technol. 2010, 101, 7350–7361. [Google Scholar] [CrossRef] [PubMed]
- Halvorsen, B.M.; Adhikari, U.; Eikeland, M.S. Gasification of Biomass for Production of Syngas for Biofuel. In Proceedings of the 56th Conference on Simulation and Modelling, Linköping, Sweden, 7–9 October 2015. [Google Scholar]
- Omar, R.; Idris, A.; Yunus, R.; Khalid, K.; Aida Isma, M.I. Characterization of empty fruit bunch for microwave-assisted pyrolysis. Fuel 2011, 90, 1536–1544. [Google Scholar] [CrossRef]
- Chang, S.H. An overview of empty fruit bunch from oil palm as feedstock for bio-oil production. Biomass Bioenergy 2014, 62, 174–181. [Google Scholar] [CrossRef]
- Abdullah, N.; Gerhauser, H. Bio-oil derived from empty fruit bunches. Fuel 2008, 87, 2606–2613. [Google Scholar] [CrossRef] [Green Version]
- Abdullah, N.; Sulaiman, F.; Gerhauser, H. Characterisation of Oil Palm Empty Fruit Bunches for Fuel Application. J. Phys. Sci. 2011, 22, 1–24. [Google Scholar]
- Khor, K.; Lim, K.; Zainal, Z. Characterization of bio-oil: A by-product from slow pyrolysis of oil palm empty fruit bunches. Am. J. Appl. Sci. 2009, 6, 1647–1652. [Google Scholar] [CrossRef] [Green Version]
- Dupont, V.; Yun, H.A.H.; White, R.; Tande, L.N. High methane conversion efficiency by low temperature steam reforming of bio-feedstock. In Proceedings of the REGATEC 2017 4th International Conference on renewable Energy gas Technology, Verona, Italy, 22–23 May 2017. [Google Scholar]
- Pimenidou, P.; Dupont, V. Characterisation of palm empty fruit bunch (PEFB) and pinewood bio-oils and kinetics of their thermal degradation. Bioresour. Technol. 2012, 109, 198–205. [Google Scholar] [CrossRef] [Green Version]
- Quaak, P.; Knoef, H.; Stassen, H. Energy from biomass: A review of combustion and gasification technologies. In Energy Series; Paper, W.B.T., Ed.; World Bank: Washington, DC, USA, 1999. [Google Scholar]
- Lind, F.; Heyne, S.; Johnson, F. What Is the Efficiency of a Biorefinery? 2012, pp. 59–71. Available online: https://publications.lib.chalmers.se/records/fulltext/local_162671.pdf (accessed on 26 July 2021).
- Martin, S.; Kraaij, G.; Ascher, T.; Wails, D.; Wörner, A. An experimental investigation of biodiesel steam reforming. Int. J. Hydrogen Energy 2015, 40, 95–105. [Google Scholar] [CrossRef] [Green Version]
- Rostrup-Nielsen, J.R.; Sehested, J.; Nørskov, J.K. Hydrogen and Synthesis Gas. by steam and CO2 reforming. Adv. Catal. 2002, 47, 65–139. [Google Scholar] [CrossRef]
- Aasberg-Petersen, K.; Dybkjær, I.; Ovesen, C.V.; Schjødt, N.C.; Sehested, J.; Thomsen, S.G. Natural gas to synthesis gas—Catalysts and catalytic processes. J. Nat. Gas Sci. Eng. 2011, 3, 423–459. [Google Scholar] [CrossRef]
- Wilhelm, D.J.; Simbeck, D.R.; Karp, A.D.; Dickenson, R.L. Syngas production for gas-to-liquids applications: Technologies, issues and outlook. Fuel Process. Technol. 2001, 71, 139–148. [Google Scholar] [CrossRef]
- Martin, S.; Wörner, A. On-board reforming of biodiesel and bioethanol for high temperature PEM fuel cells: Comparison of autothermal reforming and steam reforming. J. Power Sources 2011, 196, 3163–3171. [Google Scholar] [CrossRef]
- Kemp, I.C. Reducing Dryer Energy Use by Process Integration and Pinch Analysis. Dry. Technol. 2005, 23, 2089–2104. [Google Scholar] [CrossRef]
- Peryoga, Y.; Dewi Solikhah, M.; Agus Raksodewanto, A. Production Cost Assessment of Palm Empty Fruit Bunch Conversion to Bio-oil via Fast Pyrolysis. Int. J. Adv. Sci. Eng. Inf. 2014, 4, 6–12. [Google Scholar] [CrossRef] [Green Version]
- Spliethoff, H.; Hein, K.R.G. Effect of co-combustion of biomass on emissions in pulverized fuel furnaces. Fuel Process. Technol. 1998, 54, 189–205. [Google Scholar] [CrossRef]
- Demirbas, A. Biomass resource facilities and biomass conversion processing for fuels and chemicals. Energy Convers. Manag. 2001, 42, 1357–1378. [Google Scholar] [CrossRef]
- Lange, J.P. Methanol synthesis: A short review of technology improvements. Catal. Today 2001, 64, 3–8. [Google Scholar] [CrossRef]
- Banaszkiewicz, T.; Chorowski, M.; Gizicki, W. Comparative analysis of cryogenic and PTSA technologies for systems of oxygen production. AIP Conf. Proc. 2014, 1573, 1373–1378. [Google Scholar]
- Katikaneni, S.P.; Al-Muhaish, F.; Harale, A.; Pham, T.V. On-site hydrogen production from transportation fuels: An overview and techno-economic assessment. Int. J. Hydrogen Energy 2014, 39, 4331–4350. [Google Scholar] [CrossRef]
- Ewan, B.; Allen, R. A figure of merit assessment of the routes to hydrogen. Int. J. Hydrogen Energy 2005, 30, 809–819. [Google Scholar] [CrossRef]
- Spath, P.L.; Mann, M.K. Life cycle assessment of hydrogen production via natural gas steam reforming. In National Technical Information Service; National Renewable Energy Laboratory, Ed.; US Department of Energy: Golden, CO, USA, 2000. [Google Scholar]
- Granovskii, M.; Dincer, I.; Rosen, M. Greenhouse gas emissions reduction by use of wind and solar energies for hydrogen and electricity production: Economic factors. Int. J. Hydrogen Energy 2007, 32, 927–931. [Google Scholar] [CrossRef]
- Voldsund, M.; Jordal, K.; Anantharaman, R. Hydrogen production with CO2 capture. Int. J. Hydrogen Energy 2016, 41, 4969–4992. [Google Scholar] [CrossRef]
- Rioche, C.; Kulkarni, S.; Meunier, F.C.; Breen, J.P.; Burch, R. Steam reforming of model compounds and fast pyrolysis bio-oil on supported noble metal catalysts. Appl. Catal. B. Environ. 2005, 61, 130–139. [Google Scholar] [CrossRef]
- Aupretre, F.; Descorme, C.; Duprez, D. Bio-ethanol catalytic steam reforming over supported metal catalysts. Catal. Commun. 2002, 3, 63–267. [Google Scholar] [CrossRef]
- Cavallaro, S.; Chiodo, V.; Vita, A.; Freni, S. Hydrogen production by auto-thermal reforming of ethanol on Rh/Al2O3 catalyst. J. Power Sources 2003, 123, 10–16. [Google Scholar] [CrossRef]



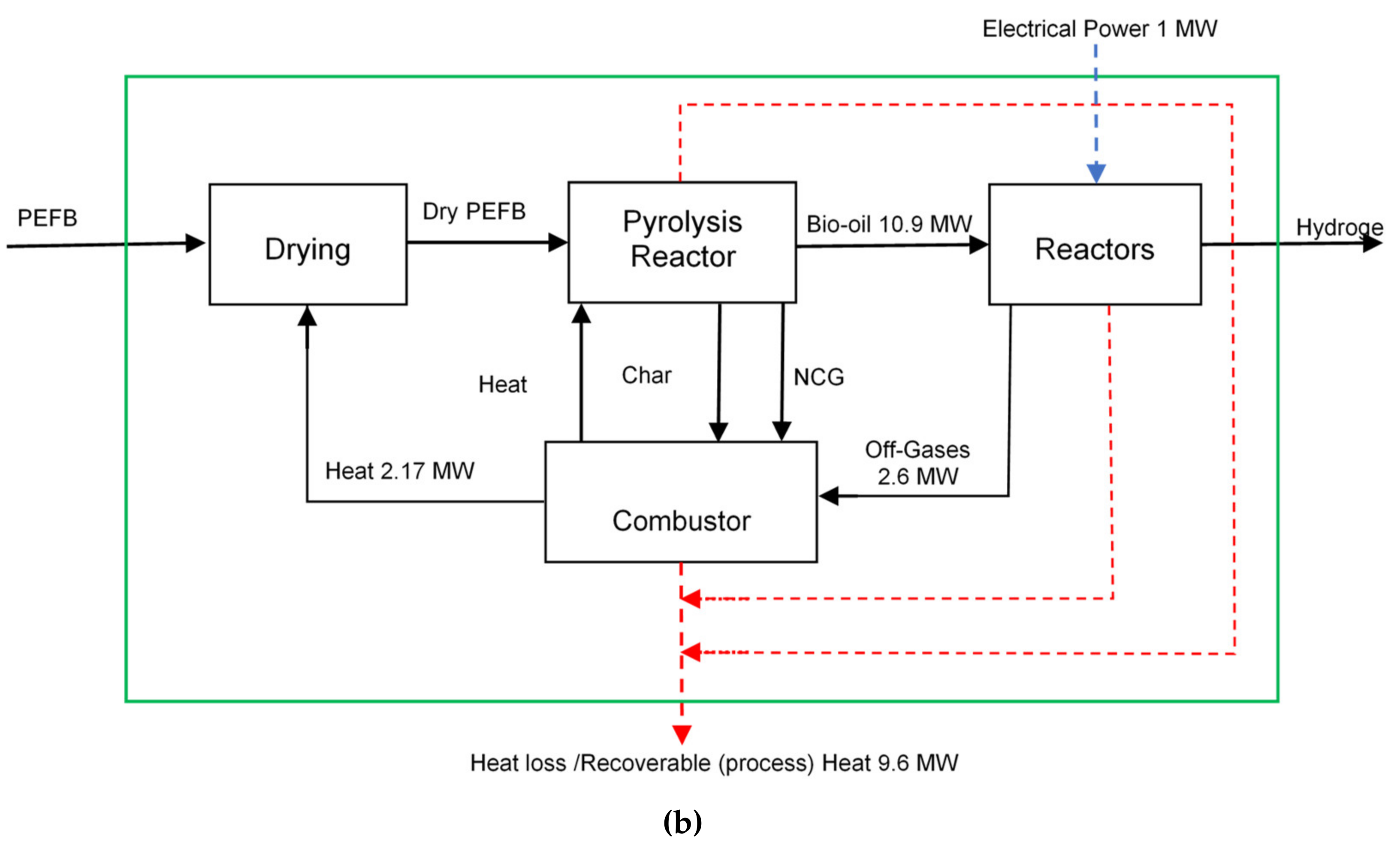

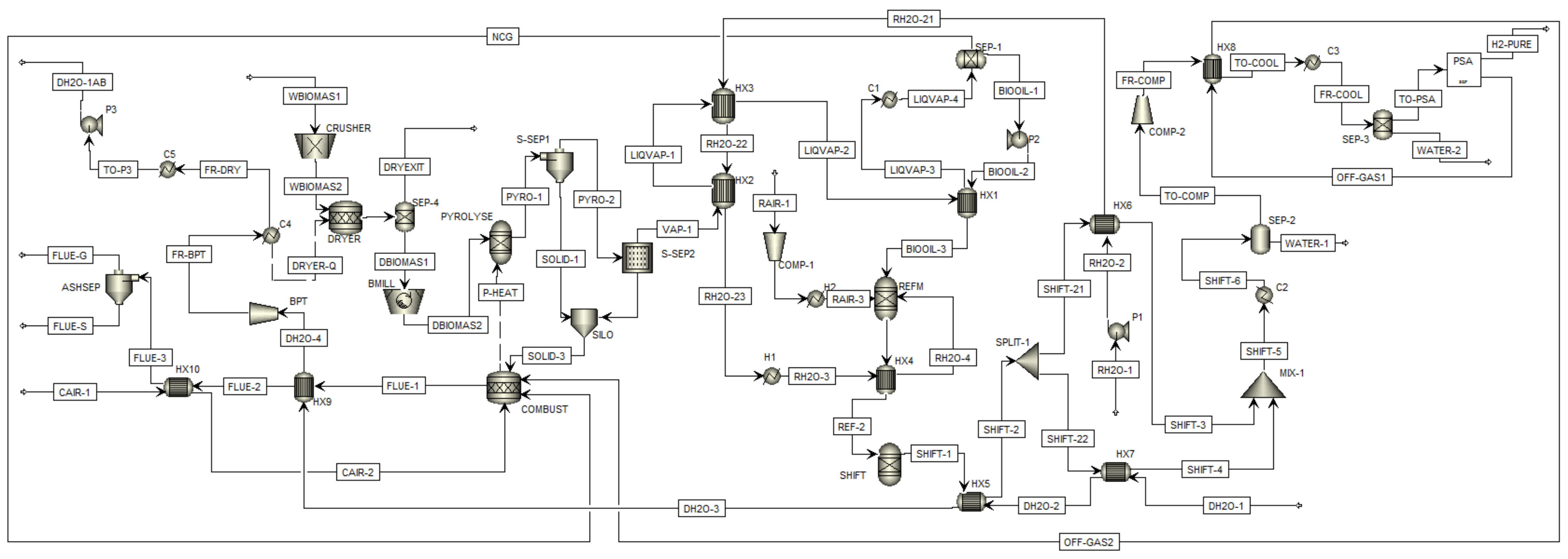

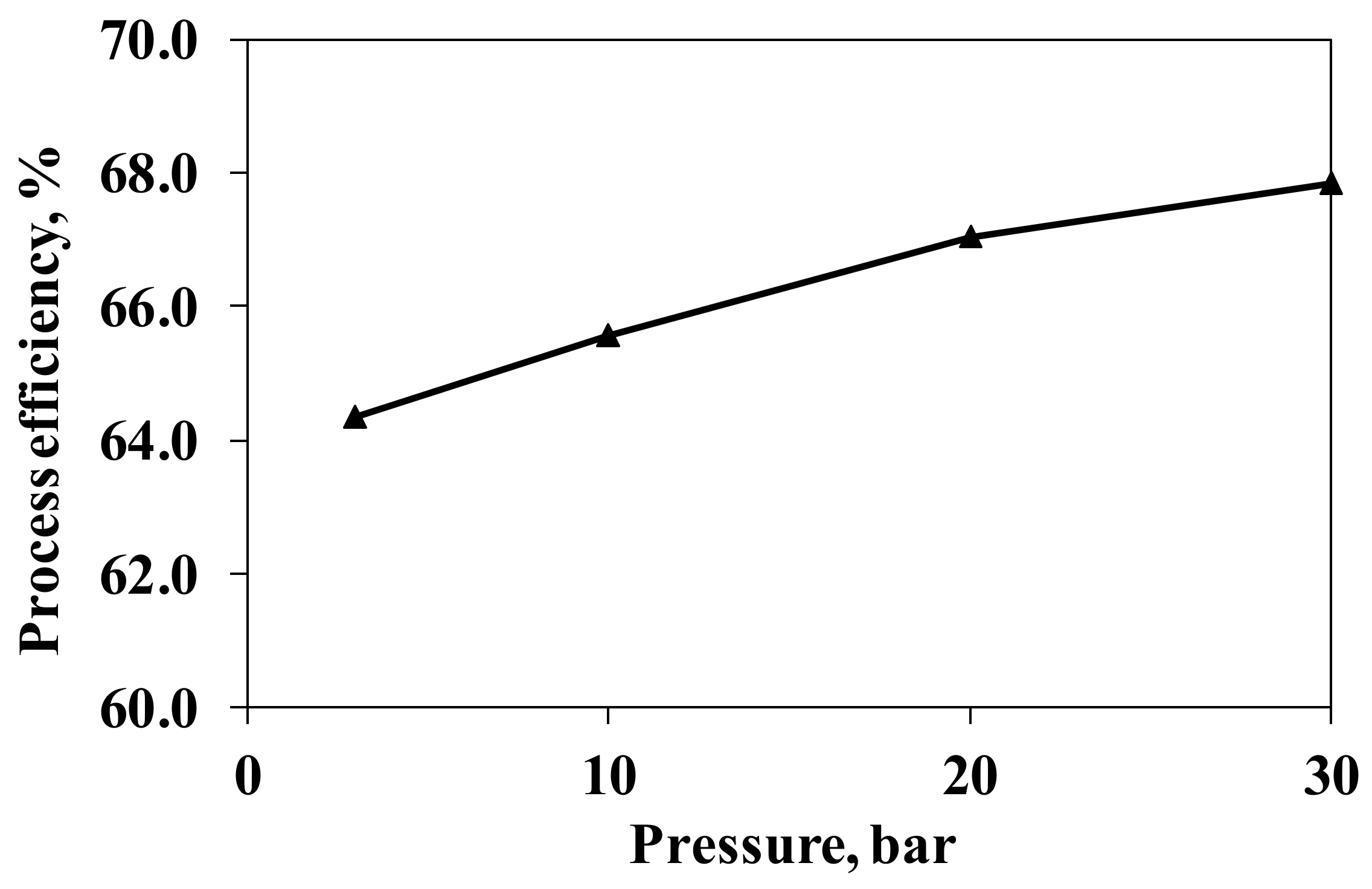
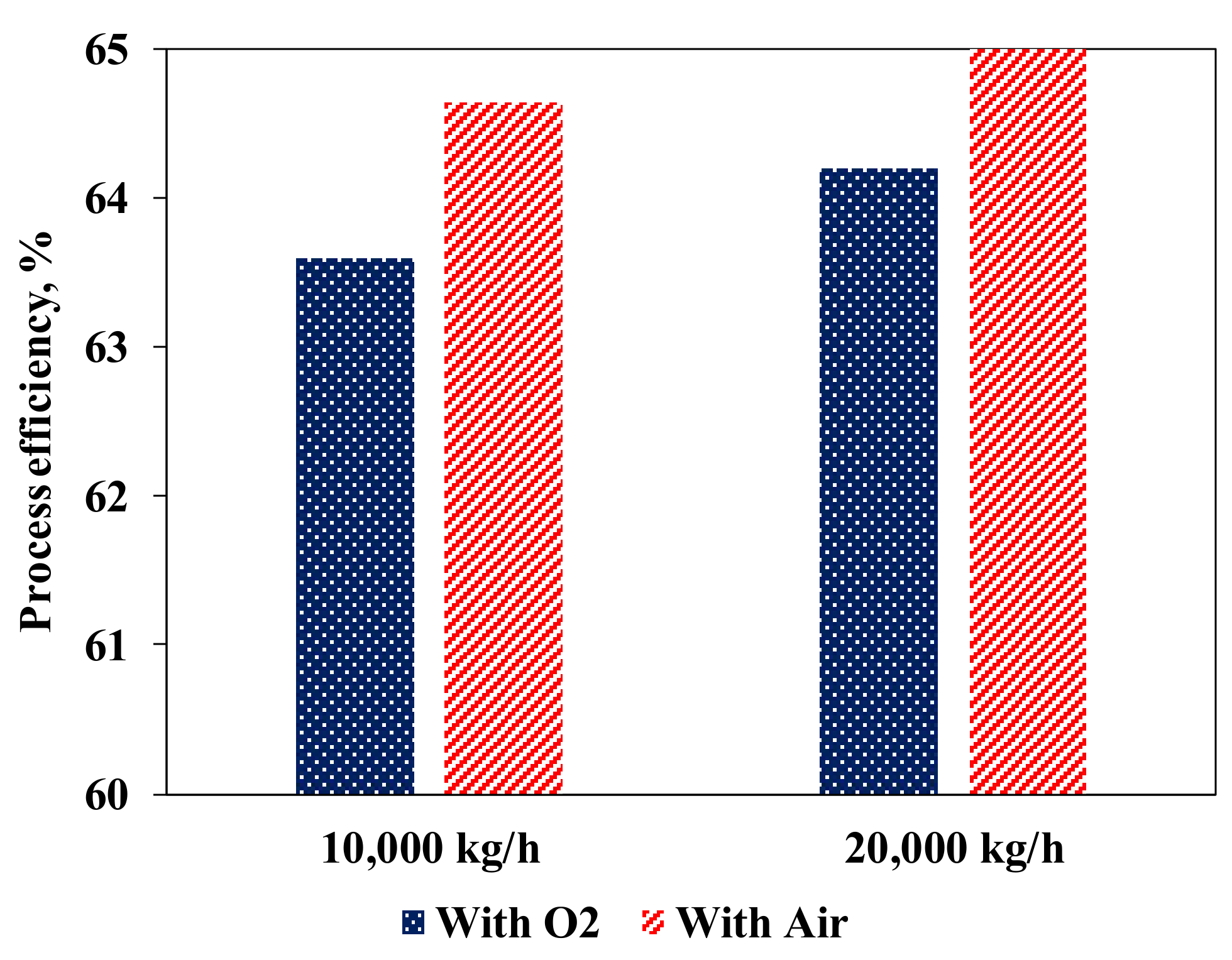
| Reaction | Reaction | Description |
|---|---|---|
| R1 | CnHmOk + (n–k)/2 O2 → nCO + m/2 H2 | Partial oxidation |
| R2 | CnHmOk + (n + m/4–k/2)O2 → nCO2 + m/2 H2O | Complete oxidation |
| R3 | CnHmOk + (n – k) H2O ⇆ nCO + (2n+m–2k)/2 H2 | Steam reforming |
| R4 | CnHmOk → kCO + m/2 H2 + (n – k) C | Decomposition |
| R5 | CO + H2O ⇆ CO2 + H2 | Water gas shift |
| R6 | 2CO ⇆ CO2 + C | Boudouard reaction |
| R7 | C + 2H2 ⇆ CH4 | Methanation of carbon |
| R8 | C + H2O ⇆CO + H2 | Carbon gasification 1 |
| R9 | C + 0.5O2 → CO | Carbon gasification 2 |
| R10 | CH4 + H2O ⇆ CO + 3H2 | Methane steam reforming |
| R11 | CO + 0.5O2 → CO2 | Carbon monoxide oxidation |
| R12 | H2 + 0.5O2 → H2O | Hydrogen oxidation |
| Ultimate | Wt% | Proximate | Wt% |
|---|---|---|---|
| C | 51.7 | Moisture | 40.0 |
| H | 5.9 | Fixed carbon (FC) 1 | 13.3 |
| O | 42.4 | Volatile matter (VM) 1 | 83.4 |
| Cl | 0.0 | Ash 1 | 3.3 |
| S | 0.0 | ||
| N | 0.0 |
| Unit | Block ID | Type | Specifications |
|---|---|---|---|
| Crusher | CRUSHER | Crusher | Used to reduce initial PEFB size from 400 mm to about 15 mm; specified using a particle size distribution (PSD). |
| Dryer | DRYER | RStoic | Air drying at 1 bar and 101 °C. PEFB’s moisture reduced from 40 to 10%. Drying reaction: Biomass(wet)→0.0555084H2O |
| Mill | BMILL | Crusher | Used to further reduce the size of the dried PEFB from 15 mm to 2 mm. |
| Pyrolysis reactor | PYROLYSE | RYield | Uses product yield distribution data. Operated at 500 °C and 1 bar. Reaction heat supplied by the heat stream, P-HEAT, from the combustor. |
| Solids removal | SOLIDSP1 & S-SEP2 | Cyclone and ESP | 99% solid removal |
| Bio-oil recovery | SEP-1 | Sep | Cooling and condensation to 40 °C at 1 bar. |
| Reformer | REFM | RGibbs | Calculates equilibrium composition of the reformer by minimisation of Gibbs free energy. Operated at 3 bar under adiabatic conditions. |
| Shift reactor | SHIFT | RGibbs | 3 bar and 250 °C (isothermal). CH4 set as inert. |
| PSA | PSA | Sep | 75% H2 recovery at 10 bar (base case). |
| Combustor | COMBUST | RStoic | 1100 °C and 1 bar with sufficient excess air to match the combustor’s enthalpy to the P-HEAT stream used for pyrolysis (fired heat exchanger). |
| Ash removal | ASHSEP | Cyclone | 99% ash removal. |
| Coolers | C1, C2 and C3 | Heater | Cool inputs streams to 30, 40 and 30 °C, respectively |
| Heaters | H1, H2 and H3 | Heater | Supply heating duties of 436 kW, 11 kW and 257 kW resp. (for the base case). |
| Heat exchangers | HX1-HX5 | HeatX | Minimum temperature of approach set at 20 °C. |
| Phase separators | SEP-2, 3 and 4 | Sep | Two phase flash adiabatic separators at input stream temperature and pressure. Actual output stream composition specified in some cases. |
| Compressors | COMP-1 and 2 | Compr | Increase the pressure of air and syngas to 3 and 10 bar, respectively. |
| Pumps | P1 and P2 | Pump | Increase bio-oil and water pressure, respectively, to reformer value. |
| Phase | Yield (wt.%) | Phase | Yield (wt.%) |
|---|---|---|---|
| Solid (char and ash) | 15 | Liquid (bio-oil) | 70 |
| Gas | 12 | Ash | 3 |
| Bio-Oil Compound | Mass Fraction | Bio-Oil Compound | Mass Fraction |
| Acetic acid | 0.055 | Levoglucosan | 0.027 |
| Formaldehyde | 0.060 | Palmitic acid | 0.005 |
| Acetaldehyde | 0.005 | H2 | 0.006 |
| 2-butanol | 0.005 | CH4 | 0.003 |
| Furfural | 0.098 | CO | 0.063 |
| Phenol | 0.005 | CO2 | 0.052 |
| Creosol | 0.022 | C (char) | 0.123 |
| Guaiacol | 0.087 | Ash | 0.031 |
| Catechol | 0.180 | H2O | 0.172 |
| Bio-Oil Compound | Feed (mol s−1) | Carbon in Feed (mol s−1) | % C in Feed |
|---|---|---|---|
| Formaldehyde | 1.07 × 10−6 | 1.18 × 10−6 | 7.80 |
| Acetaldehyde | 6.97 × 10−8 | 1.54 × 10−7 | 1.02 |
| 2-butanone | 4.36 × 10−8 | 1.92 × 10−7 | 1.27 |
| Acetic acid | 4.88 × 10−7 | 1.08 × 10−6 | 7.14 |
| Water | 5.14 × 10−6 | 0 | 0.00 |
| Furfural | 5.49 × 10−7 | 3.02 × 10−6 | 19.97 |
| Phenol | 3.49 × 10−8 | 2.30 × 10−7 | 1.52 |
| Creosol | 8.71 × 10−8 | 7.68 × 10−7 | 5.08 |
| Guaiacol | 2.53 × 10−7 | 1.95 × 10−6 | 12.89 |
| Catechol | 8.80 × 10−7 | 5.82 × 10−6 | 38.48 |
| Palmitic acid | 8.71 × 10−9 | 1.54 × 10−7 | 1.02 |
| Levoglucosan | 8.71 × 10−8 | 5.76 × 10−7 | 3.81 |
| Total | 8.71 × 10−6 | 1.51 × 10−5 | 100 |
| Unit or Process | Value | Notes/Reference(s) |
|---|---|---|
| Crushing and milling | 120 kW | Peryoga, et al. [45] and Spliethoff and Hein [46] |
| Drying (with steam at 101 °C) | 1.86 MW | For a 70 % efficient stream heated dryer |
| Pyrolysis | 2.055 MW | Corresponds to a heat requirement of 2219 kJ/kg. This heat requirement is satisfied completely by the combustor’s enthalpy |
| Compression | 682 kW | Value includes the energy required by a blower or compressor for combustion air transport (not included in Figure 9) |
| Pumping | 14.3 kW | This includes cooling water circulation with a calculated flow of 315 m3/h and pump to recirculate water for CHP unit |
| Electrostatic precipitator | 4.8 kW | 1 kWh per 1000 m3 of treated gas |
| Phase | Case 1 | Case 2 | Case 3 | Case 4 * | Case 5 |
|---|---|---|---|---|---|
| Liquid | 50 | 55 | 60 | 70 | 75 |
| Char | 25 | 20 | 15 | 15 | 12 |
| Gas | 20 | 20 | 22 | 12 | 10 |
| Ash | 5 | 5 | 3 | 3 | 3 |
| Selectivity | Carbon (mg/g(cat).h) | |||||||
|---|---|---|---|---|---|---|---|---|
| Catalyst | S/C | H2 | CO | CO2 | CH4 | H2/CO | CO2/CO | |
| Rh-Al | 2.2 | 96.0 | 22.9 | 72.9 | 4.2 | 4.4 | 3.2 | 65.4 |
| Equilibrium | 2.2 | 98.2 | 20.8 | 77.1 | 2.2 | 5.7 | 3.7 | 0.0 |
| Rh-Al | 3 | 96.8 | 18.8 | 77.8 | 3.4 | 5.5 | 4.1 | 71.6 |
| Equilibrium | 3 | 98.6 | 14.0 | 84.2 | 1.8 | 8.8 | 6.0 | 0.0 |
| Conversion, % | Dry Gas Composition, Mol% | Syngas | F-T Module | H2 Yield, mol H/mol C | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| S/C | λ * | T (°C) | Bio-oil | H2O | H2 | CO | CO2 | CH4 | H2/CO | M | |
| 2.2 | 0.318 1 | 593 | 83.7 | 20.4 | 50.3 | 11.4 | 36.2 | 2.1 | 4.42 | 0.296 | 0.93 |
| 0.35 2 | 641 | 91.7 | 16.3 | 48.8 | 16.1 | 34.5 | 0.6 | 3.04 | 0.283 | 0.89 | |
| 0.366 3 | 670 | 95.2 | 16.5 | 48.7 | 14.6 | 36.4 | 0.3 | 3.34 | 0.241 | 0.91 | |
| 3 | 0.3351 1 | 572 | 87.1 | 14.8 | 50.8 | 9.2 | 38.3 | 1.7 | 5.49 | 0.263 | 0.98 |
| 0.3682 2 | 620 | 93.7 | 17.0 | 52.7 | 6.7 | 40.1 | 0.4 | 7.82 | 0.269 | 1.02 | |
| 0.3853 3 | 647 | 95.3 | 11.7 | 50.6 | 9.4 | 39.8 | 0.2 | 5.36 | 0.219 | 0.89 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tande, L.N.; Resendiz-Mora, E.; Dupont, V. Bioh2, Heat and Power from Palm Empty Fruit Bunch via Pyrolysis-Autothermal Reforming: Plant Simulation, Experiments, and CO2 Mitigation. Energies 2021, 14, 4767. https://doi.org/10.3390/en14164767
Tande LN, Resendiz-Mora E, Dupont V. Bioh2, Heat and Power from Palm Empty Fruit Bunch via Pyrolysis-Autothermal Reforming: Plant Simulation, Experiments, and CO2 Mitigation. Energies. 2021; 14(16):4767. https://doi.org/10.3390/en14164767
Chicago/Turabian StyleTande, Lifita N., Erik Resendiz-Mora, and Valerie Dupont. 2021. "Bioh2, Heat and Power from Palm Empty Fruit Bunch via Pyrolysis-Autothermal Reforming: Plant Simulation, Experiments, and CO2 Mitigation" Energies 14, no. 16: 4767. https://doi.org/10.3390/en14164767
APA StyleTande, L. N., Resendiz-Mora, E., & Dupont, V. (2021). Bioh2, Heat and Power from Palm Empty Fruit Bunch via Pyrolysis-Autothermal Reforming: Plant Simulation, Experiments, and CO2 Mitigation. Energies, 14(16), 4767. https://doi.org/10.3390/en14164767






