Current State and Future Prospects for Electrochemical Energy Storage and Conversion Systems
Abstract
1. Introduction
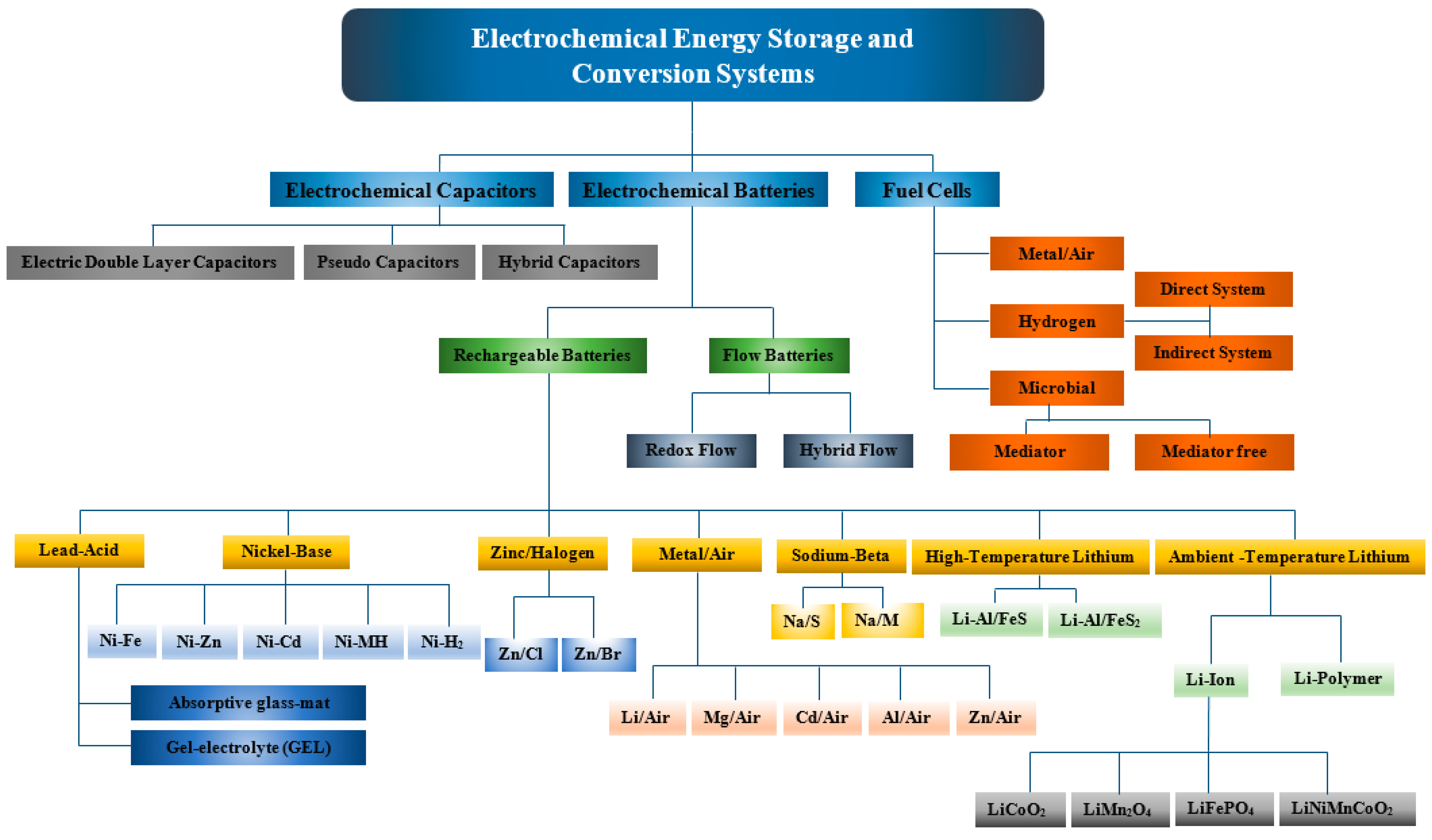
2. Electrochemical Energy Storage and Conversion Systems
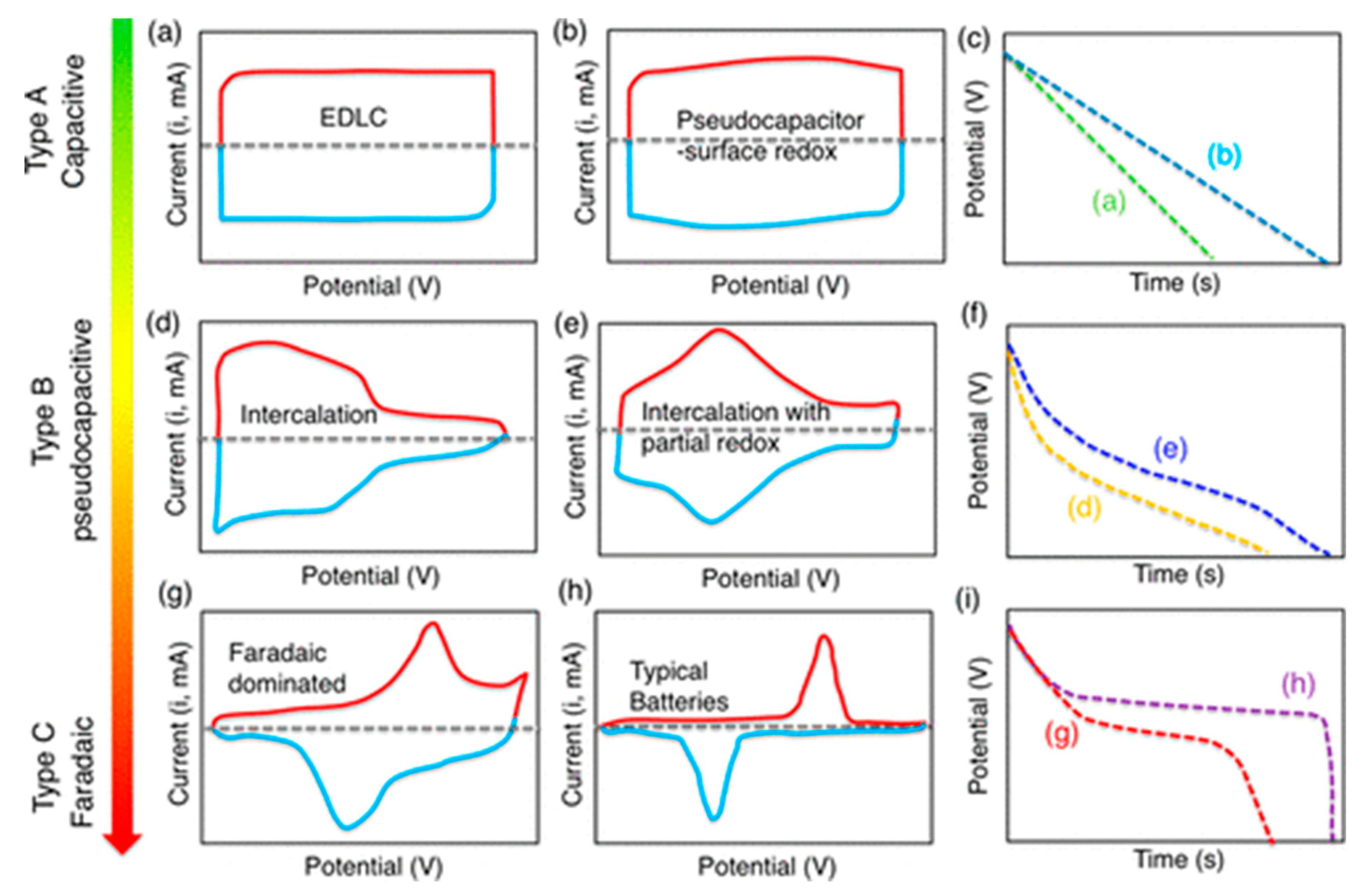
2.1. Electrochemical Capacitors
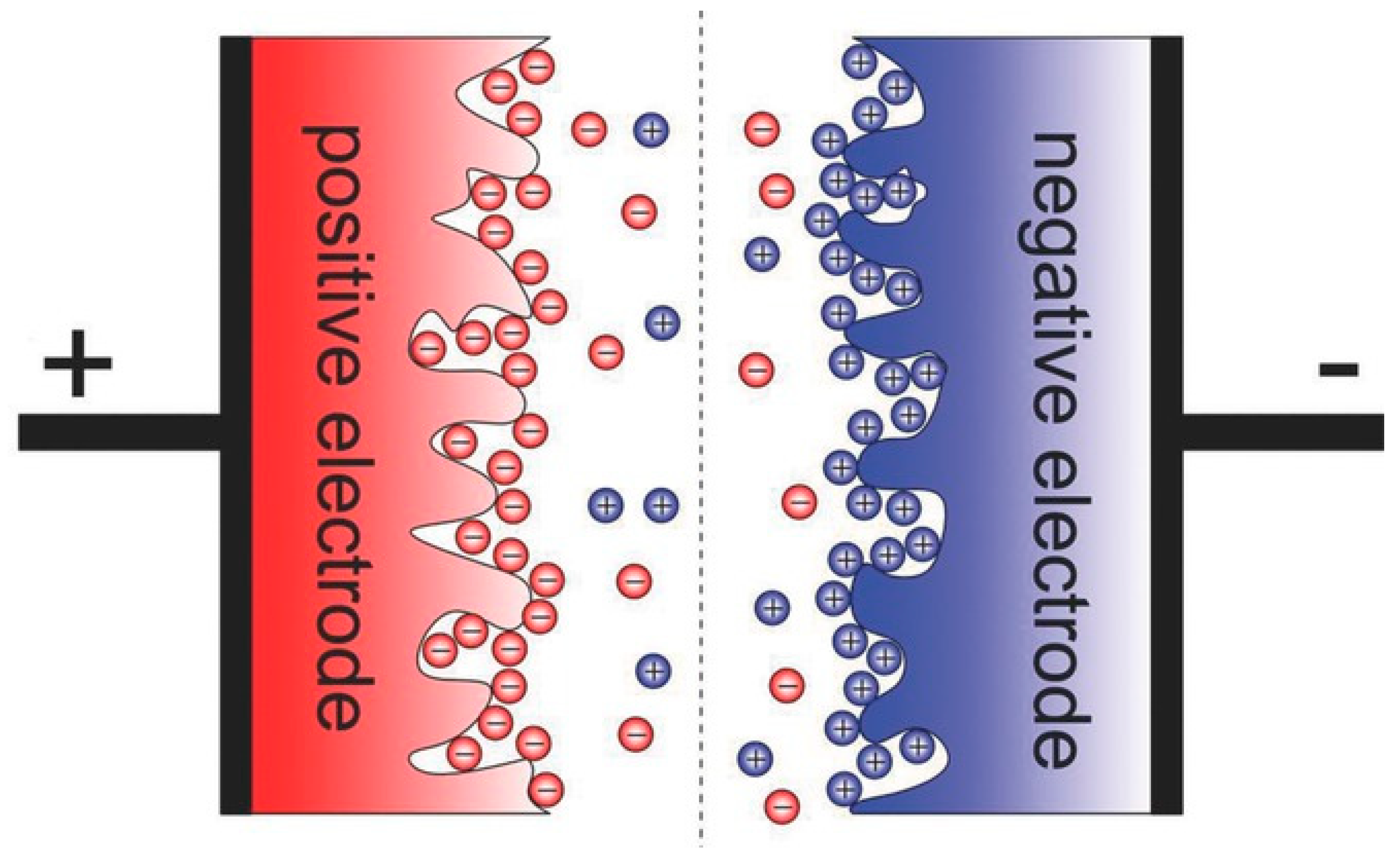
2.1.1. Electric Double Layer Capacitors (EDLCs)
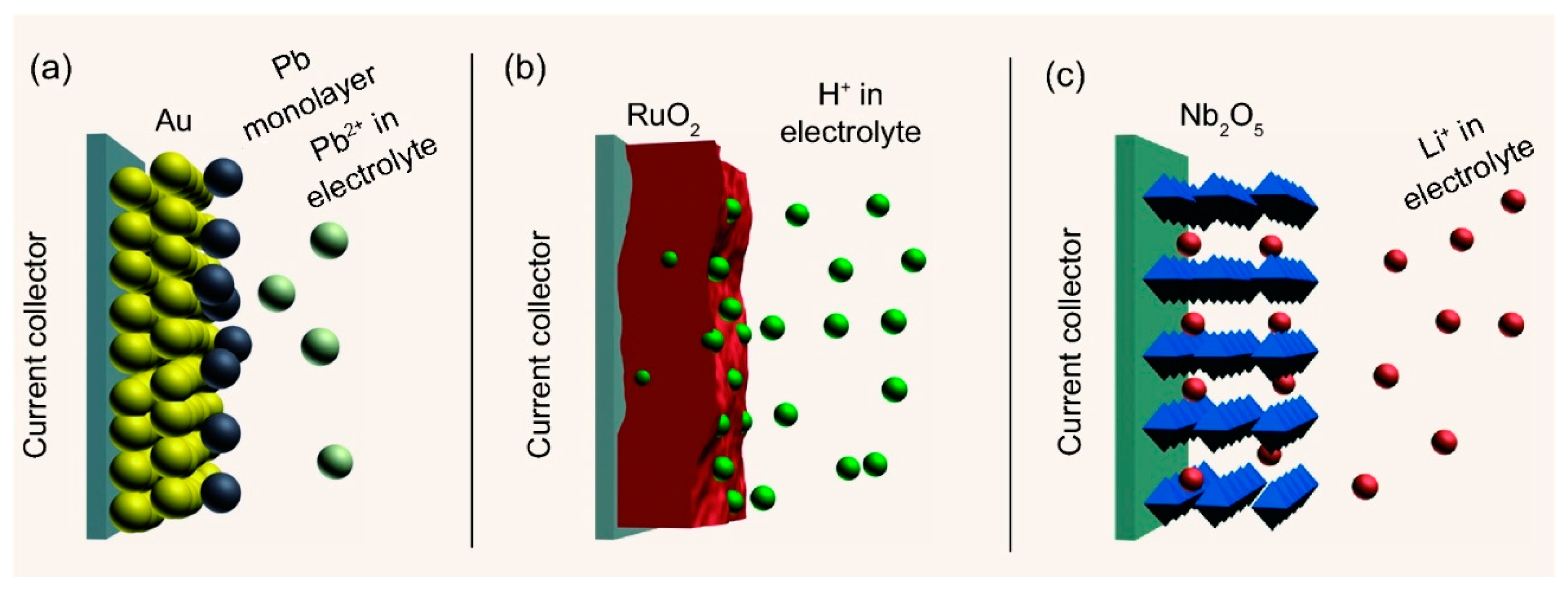
2.1.2. Pseudo-Capacitors (PCs)
2.1.3. Hybrid-Capacitors (HCs)
3. Comparison of Different Types of ECs
- Synthesis of novel and better-quality materials (both electrode and electrolyte) to enhance performance and reduce cost.
- Development of hierarchical porous microstructures in order to maintain high power densities while improving the energy densities and avoiding the creation of inaccessible specific surface area.
- Improved understanding of energy storage mechanisms due to interfacial reactions at the electrode/electrode interface.
- Improvements to the interfacial interactions between electrolyte ions and electroactive material through surface modification.
3.1. Electrochemical Batteries
3.1.1. Rechargeable Batteries
Lithium-Ion Batteries (Li-ion)
Lead-Acid Batteries (Pb-A)
Nickel-Cadmium Batteries (Ni-Cd)
Nickel-Metal Hybrid Batteries (Ni-MH)
3.1.2. Flow Batteries
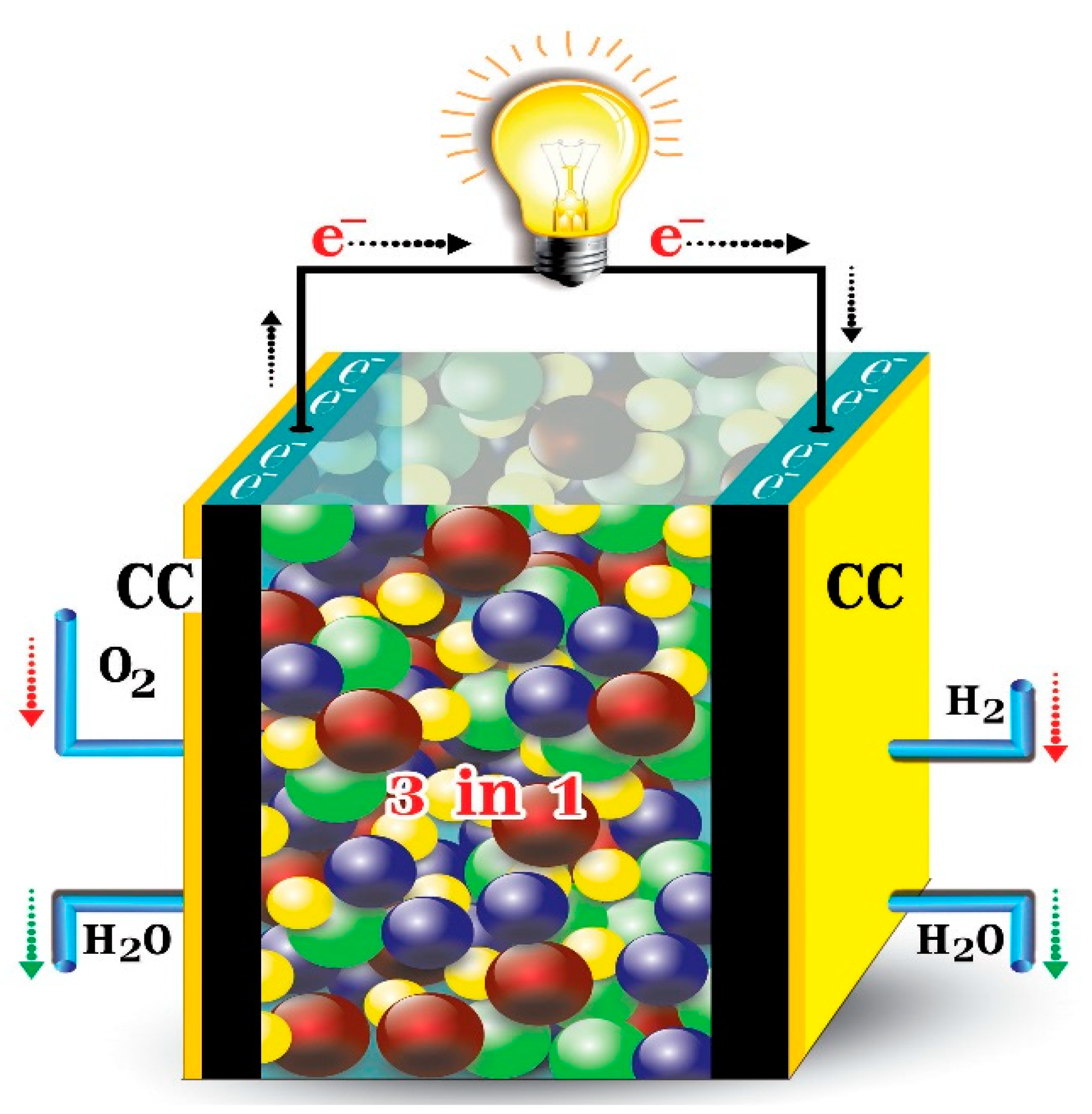
3.2. Fuel Cells
- Solid oxide fuel cell (SOFC)
- Proton exchange membrane or polymer electrolyte membrane fuel cell (PEMFC). These can be further subdivided into standard PEMFC (operating below 100 ℃) or high temperature PEMFC or HT-PEMFC (operating in temperature ranges of up to 200 ℃).
- Molten carbonate fuel cell (MCFC)
- Alkaline fuel cell (AFC)
- Phosphoric acid fuel cell (PAFC)
- Direct methanol fuel cell (DMFC)
- Single layered fuel cell (SLFC)
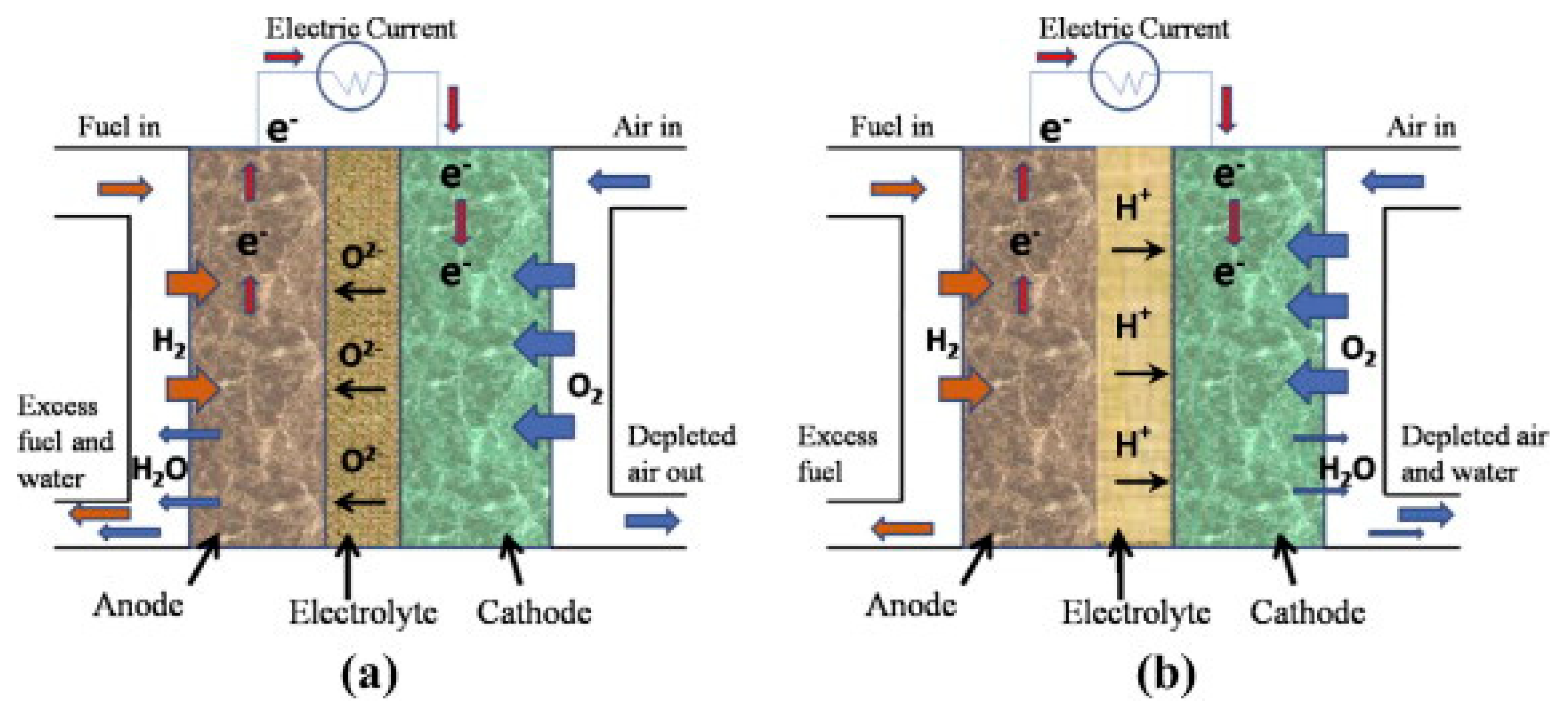
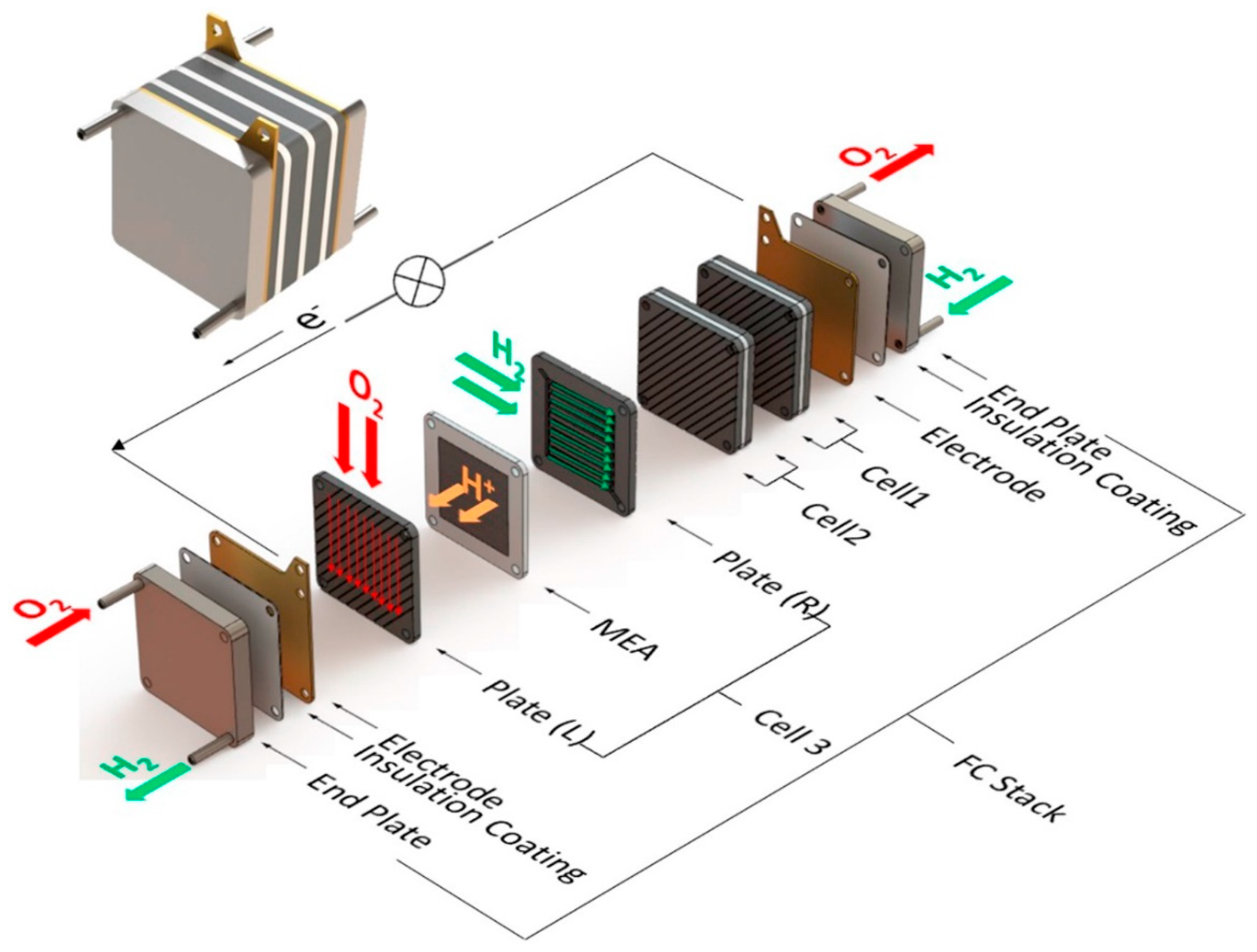
3.2.1. Solid Oxide Fuel Cell (SOFC)
| Fuel Cell Type | Operating Temperature (℃) | Output (kW) | Efficiency (%) | Applications | Advantages | Disadvantages |
|---|---|---|---|---|---|---|
| Alkaline | 900–100 | 10–100 | 60 | (i) Military/Space | (i) Faster cathode reaction in alkaline electrolyte (ii) Variety of catalysts used | (i) Costly removal of CO2 |
| Molten Carbonate | 600–700 | 1–1000 | ≥40 | (i) Electric utility (ii) Distributed generation (large scale) | (i) High efficiency (ii) Fuel flexibility (iii) Variety of catalysts used | (i) Complex electrolyte management (ii) High operating temperature (iii) Slow start up |
| Polymer electrolyte membrane | 50–100 | 1–250 | 53–58 | (i) Backup power (ii) Portable power (iii) Transportation (iv) Distributed generation (small scale) | (i) Solid electrolyte: reduced electrolyte and corrosion issues (ii) Quick start up (iii) Low temperature | (i) Expensive catalyst requirement (ii) Highly sensitive to fuel impurities (iii) Low temperature (Heat wastage) |
| Direct methanol | 60–200 | 0.001–100 | 40 | (i) Portable devices | (i) Reduced cost due to the absence of fuel reformer | (i) Methanol high toxic and flammable (ii) Low efficiency |
| Phosphoric acid | 150–200 | 50–1000 | 40 | (i) Distributed generation | (i) Higher overall efficiencies (ii) Increased impurity tolerance | (i) Platinum catalysts required (ii) Low power output (iii) Large size |
| Solid Oxide | 600–1000 | 1–3000 | 35–45 | (i) Electric utility (ii) Auxiliary power (iii) Distributed generation (large scale) | (i) High efficiency (ii) Fuel flexibility (iii) Suitable for CHP (iv) Hybrid GT/Cycle (v) Variety of catalysts used | (i) High temperature enhances cell breakdown and corrosion (ii) Slow start up |
| Single layered | 550–750 | 97–98 | (i) Transportations (ii) Portable devices | (i) High efficiency (ii) Low cost (iii) Low emissions | (i) Un-tested technology |
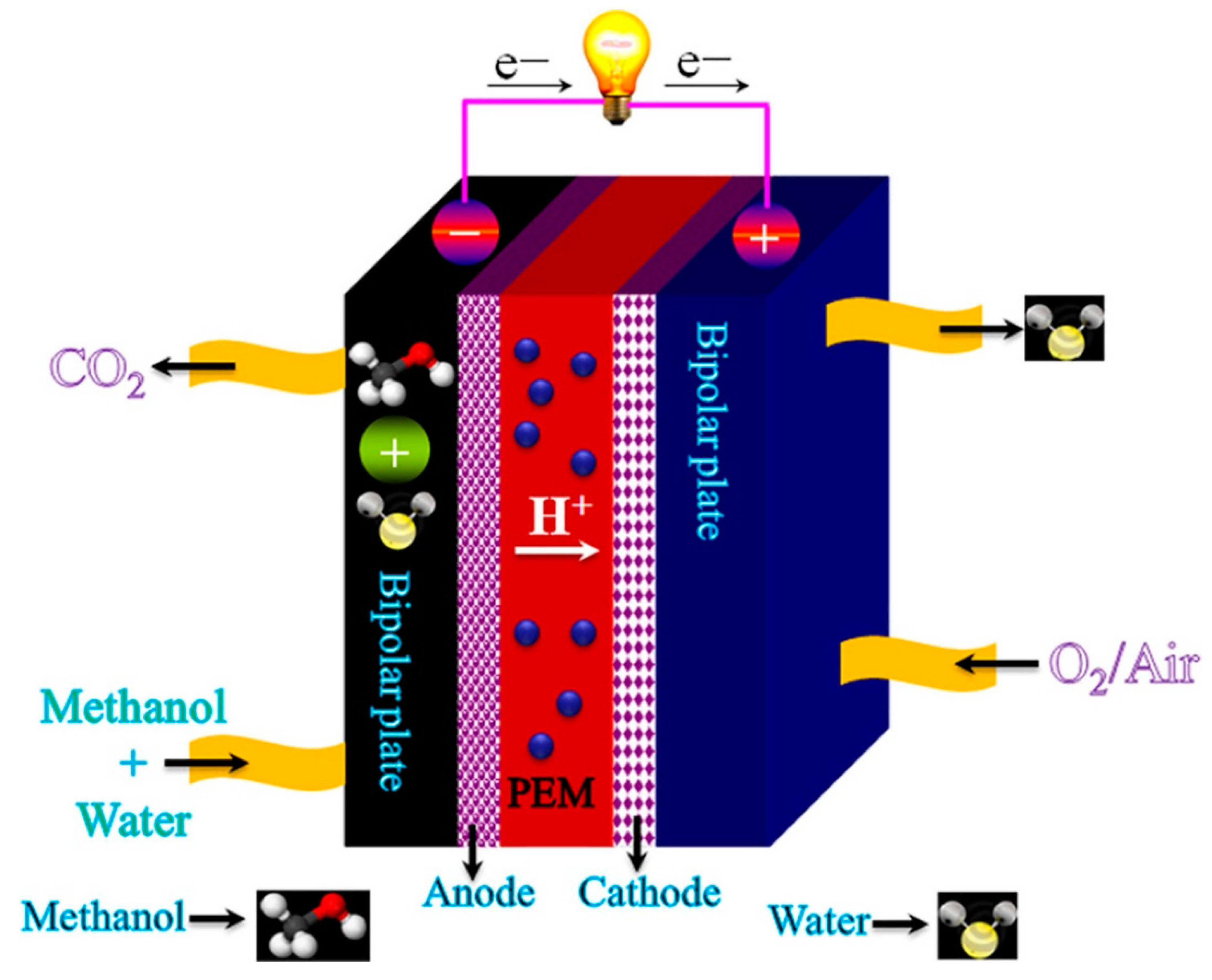
3.2.2. Direct Methanol Fuel Cell (DMFC)
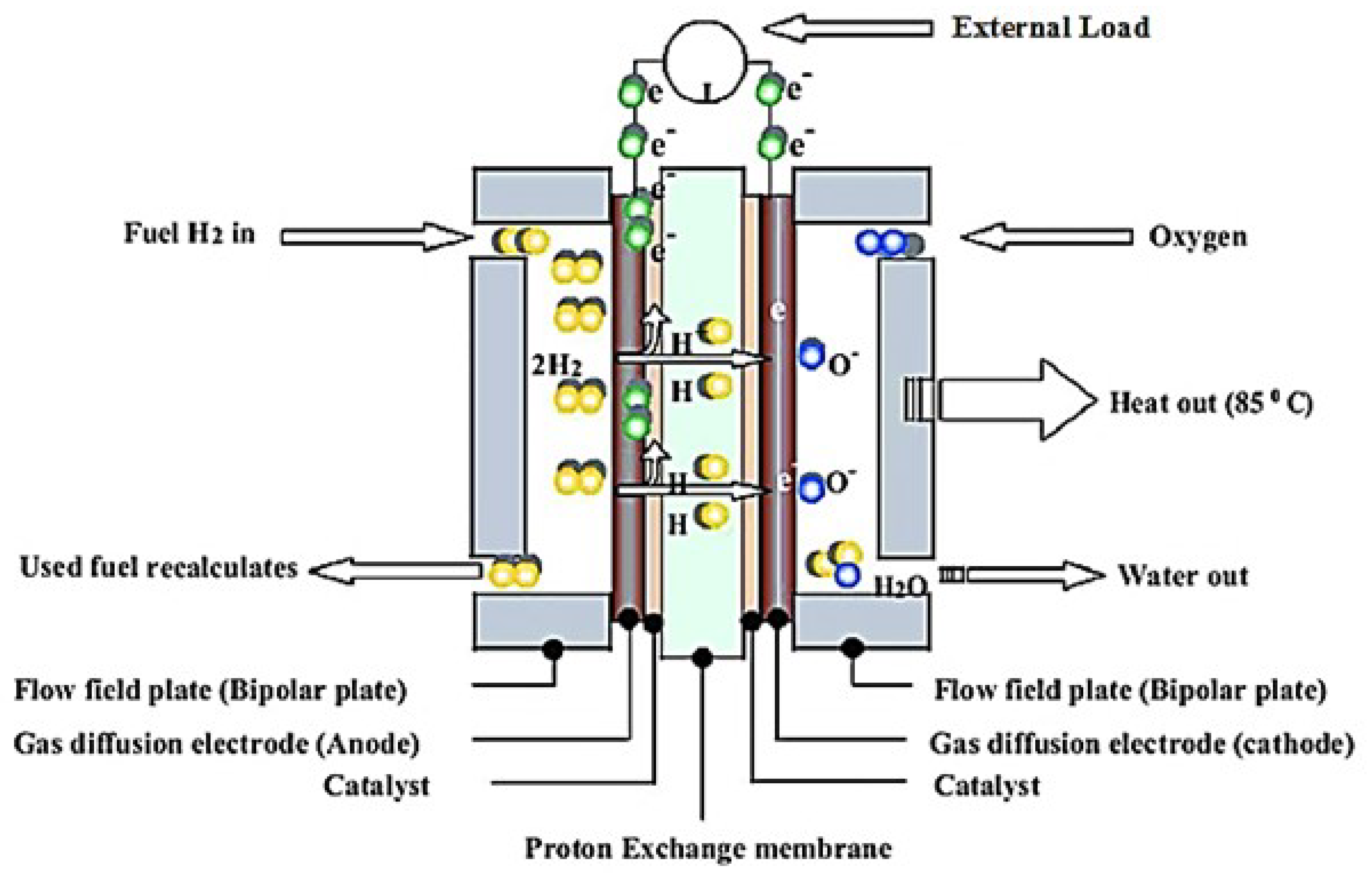
3.2.3. Polymer Electrolyte Membrane Fuel Cell (PEMFC)
3.2.4. Single Layer Fuel Cell (SLFC)
4. Comparison of Different Electrochemical Energy Storage and Conversion Systems
5. Computational Modelling Applications in Electrochemical Energy Storage and Conversion Systems
6. Conclusions and Future Prospects
Author Contributions
Funding
Conflicts of Interest
References
- Shabbir, I.; Mirzaeian, M. Feasibility analysis of different cogeneration systems for a paper mill to improve its energy efficiency. Int. J. Hydrogen Energy 2016, 41, 16535–16548. [Google Scholar] [CrossRef]
- Kutscher, C.F. Principles of Sustainable Energy Systems, 3rd ed.; Informa UK Limited: Colchester, UK, 2018. [Google Scholar]
- Dehghani-Sanij, A.; Dehghani, S.; Naterer, G.; Muzychka, Y. Marine icing phenomena on vessels and offshore structures: Prediction and analysis. Ocean Eng. 2017, 143, 1–23. [Google Scholar] [CrossRef]
- Khajuria, A.; Ravindranath, N. Climate Change Vulnerability Assessment: Approaches DPSIR Framework and Vulnerability Index. J. Earth Sci. Clim. Chang. 2012, 3. [Google Scholar] [CrossRef]
- Guney, M.S.; Tepe, Y. Classification and assessment of energy storage systems. Renew. Sustain. Energy Rev. 2017, 75, 1187–1197. [Google Scholar] [CrossRef]
- Bird, L.; Lew, D.; Milligan, M.; Carlini, E.M.; Estanqueiro, A.; Flynn, D.; Gomez-Lazaro, E.; Holttinen, H.; Menemenlis, N.; Orths, A.; et al. Wind and solar energy curtailment: A review of international experience. Renew. Sustain. Energy Rev. 2016, 65, 577–586. [Google Scholar] [CrossRef]
- Lu, L.; Han, X.; Li, J.; Hua, J.; Ouyang, M. A review on the key issues for lithium-ion battery management in electric vehicles. J. Power Sour. 2013, 226, 272–288. [Google Scholar] [CrossRef]
- Mernagh, T.P. A Review of Australian Salt Lakes and Assessment of Their Potential for Strategic Resources. Geosci. Aust. Record 2013/39; Geoscience Australia: Canberra, Australia, 2013; p. 8. [Google Scholar]
- Wanger, T.C. The Lithium future-resources, recycling, and the environment. Conserv. Lett. 2011, 4, 202–206. [Google Scholar] [CrossRef]
- Dawouda, H.D.; Altahtamounia, T.M.; Zaghoa, M.M.; Bensalahb, N. A brief overview of flexible CNT/PANI super capacitors. Mater. Sci. Nanotechnol. 2017, 1, 23–36. [Google Scholar] [CrossRef]
- Watanabe, M.; Tryk, D.A. Fuel Cells: An Overview with Emphasis on Polymer Electrolyte Fuel Cells. In Electrochemical Science for a Sustainable Society; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2017; pp. 51–94. [Google Scholar]
- Ferreira, H.L.; Garde, R.; Fulli, G.; Kling, W.; Lopes, J.P. Characterisation of electrical energy storage technologies. Energy 2013, 53, 288–298. [Google Scholar] [CrossRef]
- Mirzaeian, M.; Abbas, Q.; Ogwu, A.; Hall, P.; Goldin, M.; Mirzaeian, M.; Jirandehi, H.F. Electrode and electrolyte materials for electrochemical capacitors. Int. J. Hydrogen Energy 2017, 42, 25565–25587. [Google Scholar] [CrossRef]
- Kötz, R.; Carlen, M. Principles and applications of electrochemical capacitors. Electrochim. Acta 2000, 45, 2483–2498. [Google Scholar] [CrossRef]
- Conway, B.E. Transition from “Supercapacitor” to “Battery” Behavior in Electrochemical Energy Storage. J. Electrochem. Soc. 1991, 138, 1539–1548. [Google Scholar] [CrossRef]
- Conway, B.; Birss, V.; Wojtowicz, J. The role and utilization of pseudocapacitance for energy storage by supercapacitors. J. Power Sour. 1997, 66, 1–14. [Google Scholar] [CrossRef]
- Gogotsi, Y.; Penner, R.M. Energy Storage in Nanomaterials—Capacitive, Pseudocapacitive, or Battery-like? ACS Nano 2018, 12, 2081–2083. [Google Scholar] [CrossRef]
- Dhibar, S. Electrochemical behaviour of graphene and carbon nanotubes based hybrid polymer composites. In Hybrid Polymer Composite Materials; Elsevier BV: Amsterdam, The Netherlands, 2017; pp. 211–248. [Google Scholar]
- Béguin, F.; Presser, V.; Balducci, A.; Frackowiak, E. Carbons and Electrolytes for Advanced Supercapacitors. Adv. Mater. 2014, 26, 2219–2251. [Google Scholar] [CrossRef]
- Kesavan, T.; Partheeban, T.; Vivekanantha, M.; Kundu, M.; Maduraiveeran, G.; Sasidharan, M. Hierarchical nanoporous activated carbon as potential electrode materials for high performance electrochemical supercapacitor. Microporous Mesoporous Mater. 2019, 274, 236–244. [Google Scholar] [CrossRef]
- Wang, Y.; Chang, Z.; Qian, M.; Zhang, Z.; Lin, J.; Huang, F. Enhanced specific capacitance by a new dual redox-active electrolyte in activated carbon-based supercapacitors. Carbon 2019, 143, 300–308. [Google Scholar] [CrossRef]
- Liu, L.; Zhao, H.; Lei, Y. Nanoarchitectured Current Collectors: Review on Nanoarchitectured Current Collectors for Pseudocapacitors (Small Methods 8/2019). Small Methods 2019, 3, 1800341. [Google Scholar] [CrossRef]
- Abbas, Q.; Raza, R.; Shabbir, I.; Olabi, A. Heteroatom doped high porosity carbon nanomaterials as electrodes for energy storage in electrochemical capacitors: A review. J. Sci. Adv. Mater. Devices 2019, 4, 341–352. [Google Scholar] [CrossRef]
- Wang, J.; Dong, S.; Ding, B.; Wang, Y.; Hao, X.; Dou, H.; Xia, Y.; Zhang, X. Pseudocapacitive materials for electrochemical capacitors: From rational synthesis to capacitance optimization. Natl. Sci. Rev. 2016, 4, 71–90. [Google Scholar] [CrossRef]
- Zhang, K.; Zhang, L.L.; Zhao, X.; Wu, J. Graphene/polyaniline nanofiber composites as supercapacitor electrodes. Chem. Mater. 2010, 22, 1392–1401. [Google Scholar] [CrossRef]
- Dettlaff, A.; Wilamowska, M. Electrochemical synthesis and characterization of nanocomposites based on poly(3,4-ethylenedioxythiophene) and functionalized carbon nanotubes. Synth. Met. 2016, 212, 31–43. [Google Scholar] [CrossRef]
- Majumdara, D.; Maiyalagan, T.; Jiang, Z. Recent Progress in Ruthenium Oxide-Based Composites for Supercapacitor Applications. ChemElectroChem 2019, 6, 4343–4372. [Google Scholar] [CrossRef]
- Kate, R.S.; Khalate, S.A.; Deokate, R.J. Overview of nanostructured metal oxides and pure nickel oxide (NiO) electrodes for supercapacitors: A review. J. Alloys Compd. 2018, 734, 89–111. [Google Scholar] [CrossRef]
- Mei, J.; Liao, T.; Ayoko, G.A.; Bell, J.; Sun, Z. Cobalt oxide-based nanoarchitectures for electrochemical energy applications. Prog. Mater. Sci. 2019, 103, 596–677. [Google Scholar] [CrossRef]
- Xu, C.; Kang, F.; Li, B.; Du, H. Recent progress on manganese dioxide based supercapacitors. J. Mater. Res. 2010, 25, 1421–1432. [Google Scholar] [CrossRef]
- Lu, M. Supercapacitors: Materials, Systems, and Applications; John Wiley & Sons: Hoboken, NJ, USA, 2013. [Google Scholar]
- Shinde, P.A.; Seo, Y.; Lee, S.; Kim, H.; Pham, Q.N.; Won, Y.; Jun, S.C. Layered manganese metal-organic framework with high specific and areal capacitance for hybrid supercapacitors. Chem. Eng. J. 2020, 387, 122982. [Google Scholar] [CrossRef]
- Muzaffar, A.; Ahamed, M.B.; Deshmukh, K.; Thirumalai, J. A review on recent advances in hybrid supercapacitors: Design, fabrication and applications. Renew. Sustain. Energy Rev. 2019, 101, 123–145. [Google Scholar] [CrossRef]
- Halper, M.S.; Ellenbogen, J.C. Supercapacitors: A Brief Overview; MITRE Nanosystems Group: McLean, VA, USA, 2006. [Google Scholar]
- Yu, P.; Zeng, Y.; Zeng, Y.; Dong, H.; Hu, H.; Liu, Y.; Zheng, M.; Xiao, Y.; Lu, X.; Liang, Y. Achieving high-energy-density and ultra-stable zinc-ion hybrid supercapacitors by engineering hierarchical porous carbon architecture. Electrochim. Acta 2019, 327, 134999. [Google Scholar] [CrossRef]
- Wei, D.; Zhang, Y.; Zhu, X.; Fan, M.; Wang, Y. CNT/Co3S4@NiCo LDH ternary nanocomposites as battery-type electrode materials for hybrid supercapacitors. J. Alloys Compd. 2020, 824, 153937. [Google Scholar] [CrossRef]
- Xu, B.; Feng, W.; Chen, S.; Zhou, Z.; Cao, G.; Yang, Y. High-capacitance carbon electrode prepared by PVDC carbonization for aqueous EDLCs. Electrochim. Acta 2009, 54, 2185–2189. [Google Scholar] [CrossRef]
- Augustyn, V.; Simon, P.; Dunn, B. Pseudocapacitive oxide materials for high-rate electrochemical energy storage. Energy Environ. Sci. 2014, 7, 1597–1614. [Google Scholar] [CrossRef]
- Senthilkumar, B.; Khan, Z.; Park, S.; Kim, K.; Ko, H.; Kim, Y. Highly porous graphitic carbon and Ni2P2O7 for a high performance aqueous hybrid supercapacitor. J. Mater. Chem. A 2015, 3, 21553–21561. [Google Scholar] [CrossRef]
- Wei, J.-S.; Wan, S.; Zhang, P.; Ding, H.; Chen, X.-B.; Xiong, H.-M.; Gao, S.; Wei, X. Preparation of porous carbon electrodes from semen cassiae for high-performance electric double-layer capacitors. New J. Chem. 2018, 42, 6763–6769. [Google Scholar] [CrossRef]
- Wang, G.; Oswald, S.; Löffler, M.; Müllen, K.; Feng, X. Beyond Activated Carbon: Graphite-Cathode-Derived Li-Ion Pseudocapacitors with High Energy and High Power Densities. Adv. Mater. 2019, 31, e1807712. [Google Scholar] [CrossRef]
- Ahn, W.; Lee, D.U.; Li, G.; Feng, K.; Wang, X.; Yu, A.; Lui, G.; Chen, Z. Highly Oriented Graphene Sponge Electrode for Ultra High Energy Density Lithium Ion Hybrid Capacitors. ACS Appl. Mater. Interfaces 2016, 8, 25297–25305. [Google Scholar] [CrossRef] [PubMed]
- Mostazo-López, M.J.; Ruiz-Rosas, R.; Castro-Muñiz, A.; Nishihara, H.; Kyotani, T.; Morallón, E.; Cazorla-Amorós, D. Ultraporous nitrogen-doped zeolite-templated carbon for high power density aqueous-based supercapacitors. Carbon 2018, 129, 510–519. [Google Scholar] [CrossRef]
- Gao, H.; Li, J.; Miller, J.R.; Outlaw, R.A.; Butler, S.; Lian, K. Solid-state electric double layer capacitors for ac line-filtering. Energy Storage Mater. 2016, 4, 66–70. [Google Scholar] [CrossRef]
- Liu, Q.; Ji, S.; Yang, J.; Wang, H.; Pollet, B.G.; Wang, R. Enhanced Cycleability of Amorphous MnO2 by Covering on α-MnO2 Needles in an Electrochemical Capacitor. Materials 2017, 10, 988. [Google Scholar] [CrossRef] [PubMed]
- Attias, R.; Hana, O.; Sharon, D.; Malka, D.; Hirshberg, D.; Luski, S.; Aurbach, D. Solid state synthesis of Li0.33MnO2 as positive electrode material for highly stable 2V aqueous hybrid supercapacitors. Electrochim. Acta 2017, 254, 155–164. [Google Scholar] [CrossRef]
- Wang, D.; Geng, Z.; Li, B.; Zhang, C. High performance electrode materials for electric double-layer capacitors based on biomass-derived activated carbons. Electrochim. Acta 2015, 173, 377–384. [Google Scholar] [CrossRef]
- Senthilkumar, S.T.; Selvan, R.K.; Ponpandian, N.; Melo, J.S.; Lee, Y.S. Improved performance of electric double layer capacitor using redox additive (VO2+/VO2+) aqueous electrolyte. J. Mater. Chem. A 2013, 1, 7913–7919. [Google Scholar] [CrossRef]
- Yang, H.-Z.; Zou, J.-P. Controllable preparation of hierarchical NiO hollow microspheres with high pseudo-capacitance. Trans. Nonferrous Met. Soc. China 2018, 28, 1808–1818. [Google Scholar] [CrossRef]
- Sohn, J.I.; Ahn, H.-J. Surface modification of RuO2 nanoparticles–carbon nanofiber composites for electrochemical capacitors. J. Electroanal. Chem. 2015, 744, 32–36. [Google Scholar] [CrossRef]
- Cheng, Z.; Tan, G.; Qiu, Y.; Guo, B.; Cheng, F.; Fan, H. High performance electrochemical capacitors based on MnO2/activated-carbon-paper. J. Mater. Chem C 2015, 3, 6166–6171. [Google Scholar] [CrossRef]
- Wang, P.; Wang, R.; Lang, J.; Zhang, X.; Chen, Z.; Yan, X. Porous niobium nitride as a capacitive anode material for advanced Li-ion hybrid capacitors with superior cycling stability. J. Mater. Chem. A 2016, 4, 9760–9766. [Google Scholar] [CrossRef]
- Zeng, Z.; Wang, D.; Zhu, J.; Xiao, F.; Li, Y. NiCo2S4 nanoparticles//activated balsam pear pulp for asymmetric hybrid capacitors. CrystEngComm 2016, 18, 2363–2374. [Google Scholar] [CrossRef]
- Linden, D. Handbook of Batteries and Fuel Cells; McGraw-Hill Book Co.: New York, NY, USA, 1984; p. 1075. [Google Scholar]
- Ponrouch, A.; Bitenc, J.; Dominko, R.; Lindahl, N.; Johansson, P.; Palacin, M. Multivalent rechargeable batteries. Energy Storage Mater. 2019, 20, 253–262. [Google Scholar] [CrossRef]
- Bradbury, K. Energy Storage Technology Review; Duke University: Durham, NC, USA, 2010; pp. 1–34. [Google Scholar]
- Liang, Y.; Zhao, C.; Yuan, H.; Chen, Y.; Zhang, W.; Huang, J.; Yu, D.; Liu, Y.; Titirici, M.; Chueh, Y.; et al. A review of rechargeable batteries for portable electronic devices. InfoMat 2019, 1, 6–32. [Google Scholar] [CrossRef]
- Chen, R.; Li, L.; Fan, E.; Xue, Q.; Bian, Y.; Wu, F.; Chen, R. Toward sustainable and systematic recycling of spent rechargeable batteries. Chem. Soc. Rev. 2018, 47, 7239–7302. [Google Scholar] [CrossRef]
- Dunlop, J.P. Batteries and Charge Control in Stand-Alone Photovoltaic Systems: Fundamentals and Application; FSEC-CR-1292-01; Florida Solar Energy Center/University of Central Florida: Cocoa, FL, USA, 1997. [Google Scholar]
- Chen, H.; Cong, T.N.; Yang, W.; Tan, C.; Li, Y.; Ding, Y. Progress in electrical energy storage system: A critical review. Prog. Nat. Sci. 2009, 19, 291–312. [Google Scholar] [CrossRef]
- Díaz-González, F.; Sumper, A.; Gomis-Bellmunt, O.; Villafáfila-Robles, R. A review of energy storage technologies for wind power applications. Renew. Sustain. Energy Rev. 2012, 16, 2154–2171. [Google Scholar] [CrossRef]
- Yuan, T.; Tan, Z.; Ma, C.; Yang, J.; Ma, Z.; Zheng, S. Challenges of Spinel Li4Ti5O12for Lithium-Ion Battery Industrial Applications. Adv. Energy Mater. 2017, 7, 1601625. [Google Scholar] [CrossRef]
- Ren, W.-F.; Zhou, Y.; Li, J.-T.; Huang, L.; Sun, S.-G. Si anode for next-generation lithium-ion battery. Curr. Opin. Electrochem. 2019, 18, 46–54. [Google Scholar] [CrossRef]
- Pistoia, G. Lithium-Ion Batteries: Advances and Applications; Elsevier: Amsterdam, The Netherlands, 2013; p. 664. [Google Scholar]
- Wu, Y.-P.; Rahm, E.; Holze, R. Carbon anode materials for lithium ion batteries. J. Power Sour. 2003, 114, 228–236. [Google Scholar] [CrossRef]
- Shen, X.; Tian, Z.; Fan, R.; Shao, L.; Zhang, D.; Cao, G.; Kou, L.; Bai, Y. Research progress on silicon/carbon composite anode materials for lithium-ion battery. J. Energy Chem. 2018, 27, 1067–1090. [Google Scholar] [CrossRef]
- Mahmood, N.; Tang, T.; Hou, Y. Nanostructured Anode Materials for Lithium Ion Batteries: Progress, Challenge and Perspective. Adv. Energy Mater. 2016, 6, 1600374. [Google Scholar] [CrossRef]
- Zuo, D.; Tian, G.; Li, X.; Chen, D.; Shu, K. Recent progress in surface coating of cathode materials for lithium ion secondary batteries. J. Alloys Compd. 2017, 706, 24–40. [Google Scholar] [CrossRef]
- Schipper, F.; Nayak, P.K.; Erickson, E.M.; Amalraj, S.F.; Srur-Lavi, O.; Penki, T.R.; Talianker, M.; Grinblat, J.; Sclar, H.; Breuer, O.; et al. Study of Cathode Materials for Lithium-Ion Batteries: Recent Progress and New Challenges. Inorganics 2017, 5, 32. [Google Scholar] [CrossRef]
- Montanino, M.; Moreno, M.; Carewska, M.; Maresca, G.; Simonetti, E.; Presti, R.L.; Alessandrini, F.; Appetecchi, G. Mixed organic compound-ionic liquid electrolytes for lithium battery electrolyte systems. J. Power Sour. 2014, 269, 608–615. [Google Scholar] [CrossRef]
- Eshetu, G.G.; Judez, X.; Li, C.; Martinez-Ibañez, M.; Sánchez-Diez, E.; Rodriguez-Martinez, L.M.; Zhang, H.; Armand, M. CHAPTER 4. Solid Electrolytes for Lithium Metal and Future Lithium-ion Batteries. In Future Lithium-ion Batteries; Royal Society of Chemistry (RSC): London, UK, 2019; pp. 72–101. [Google Scholar]
- Arya, A.; Sharma, A.L. Polymer electrolytes for lithium ion batteries: A critical study. Ionics 2017, 23, 497–540. [Google Scholar] [CrossRef]
- Sullivan, J.; Gaines, L. Status of life cycle inventories for batteries. Energy Convers. Manag. 2012, 58, 134–148. [Google Scholar] [CrossRef]
- Wang, G.; Li, F.; Liu, D.; Zheng, D.; Abeggien, C.J.; Luo, Y.; Yang, X.-Q.; Ding, T.; Qu, D. High performance lithium-ion and lithium—Sulfur batteries using prelithiated phosphorus/carbon composite anode. Energy Storage Mater. 2020, 24, 147–152. [Google Scholar] [CrossRef]
- Pavlov, D. Lead-Acid Batteries: Science and Technology; Elsevier BV: Amsterdam, The Netherlands, 2011. [Google Scholar]
- Ballantyne, A.D.; Hallett, J.P.; Riley, D.J.; Shah, N.; Payne, D.J. Lead acid battery recycling for the twenty-first century. R. Soc. Open Sci. 2018, 5, 171368. [Google Scholar] [CrossRef]
- May, G.J.; Davidson, A.J.; Monahov, B. Lead batteries for utility energy storage: A review. J. Energy Storage 2018, 15, 145–157. [Google Scholar] [CrossRef]
- Rand, D.; Holden, L.; May, G.; Newnham, R.; Peters, K. Valve-regulated lead/acid batteries. J. Power Sour. 1996, 59, 191–197. [Google Scholar] [CrossRef]
- Gaines, L.; Sullivan, J.; Burnham, A.; Belharouak, I. Life-cycle analysis for lithium-ion battery production and recycling. In Proceedings of the Transportation Research Board 90th Annual Meeting, Washington, DC, USA, 23–27 January 2011; Available online: https://www.researchgate.net/profile/Linda_Gaines/publication/265158823_Paper_No._11-3891_LifeCycle_Analysis_for_LithiumIon_Battery_Production_and_Recycling/links/547336180cf216f8cfaeb58a.pdf (accessed on 17 October 2020).
- Sullivan, J.; Gaines, L. A Review of Battery Life-Cycle Analysis: State of Knowledge and Critical Needs; Argonne National Lab. (ANL): Argonne, IL, USA, 2010. Available online: https://publications.anl.gov/anlpubs/2010/11/68455.pdf (accessed on 17 October 2020).
- Otto, J. Lead Acid Type Storage Battery. Google Patents US3257237A, 21 June 1966. [Google Scholar]
- Mcclelland, D.H.; Devitt, J.L. Maintenance-Free Type Lead Acid. Google Patents US4166155A, 8 August 1979. [Google Scholar]
- Beaudin, M.; Zareipour, H.; Schellenberglabe, A.; Rosehart, W. Energy storage for mitigating the variability of renewable electricity sources: An updated review. Energy Sustain. Dev. 2010, 14, 302–314. [Google Scholar] [CrossRef]
- Hadjipaschalis, I.; Poullikkas, A.; Efthimiou, V. Overview of current and future energy storage technologies for electric power applications. Renew. Sustain. Energy Rev. 2009, 13, 1513–1522. [Google Scholar] [CrossRef]
- Kondoh, J.; Ishii, I.; Yamaguchi, H.; Murata, A.; Otani, K.; Sakuta, K.; Higuchi, N.; Sekine, S.; Kamimoto, M. Electrical energy storage systems for energy networks. Energy Convers. Manag. 2000, 41, 1863–1874. [Google Scholar] [CrossRef]
- Linden, D.; Reddy, T.B. Handbook of Batteries, 3rd ed.; McGraw-Hill: New York, NY, USA, 2002. [Google Scholar]
- McKeon, B.B.; Furukawa, J.; Fenstermacher, S. Advanced Lead-Acid Batteries and the Development of Grid-Scale Energy Storage Systems. Proc. IEEE 2014, 102, 951–963. [Google Scholar] [CrossRef]
- Farret, F.A.; Simoes, M.G. Integration of Alternative Sources of Energy; John Wiley & Sons: Hoboken, NJ, USA, 2006. [Google Scholar]
- Rastler, D. Electricity Energy Storage Technology Options: A White Paper Primer on Applications, Costs, and Options; Electric Power Research Institute (EPRI): Palo Alto, CA, USA, 2010; Available online: http://large.stanford.edu/courses/2012/ph240/doshay1/docs/EPRI.pdf (accessed on 17 October 2020).
- Hage, D.S. Reference Module in Chemistry, Molecular Sciences and Chemical Engineering; Elsevier Inc.: Amsterdam, The Netherlands, 2013. [Google Scholar]
- Zelinsky, M.; Fetcenko, M.; Kusay, J.; Koch, J. Storage-integrated PV systems using advanced NiMH battery technology. In Proceedings of the Fifth International Renewable Energy Storage Conference (IRES 2010), Berlin, Germany, 22–24 November 2010. [Google Scholar]
- Poullikkas, A. A comparative overview of large-scale battery systems for electricity storage. Renew. Sustain. Energy Rev. 2013, 27, 778–788. [Google Scholar] [CrossRef]
- Rydh, C.J.; Karlström, M. Life cycle inventory of recycling portable nickel–cadmium batteries. Resour. Conserv. Recycl. 2002, 34, 289–309. [Google Scholar] [CrossRef]
- Bruce, P.G. Energy storage beyond the horizon: Rechargeable lithium batteries. Solid State Ion. 2008, 179, 752–760. [Google Scholar] [CrossRef]
- Wakihara, M. Recent developments in lithium ion batteries. Mater. Sci. Eng. R Rep. 2001, 33, 109–134. [Google Scholar] [CrossRef]
- Avril, S.; Arnaud, G.; Florentin, A.; Vinard, M. Multi-objective optimization of batteries and hydrogen storage technologies for remote photovoltaic systems. Energy 2010, 35, 5300–5308. [Google Scholar] [CrossRef]
- Hung, Y.-Y.; Yin, L.-T.; Wang, J.-W.; Wang, C.-T.; Tsai, C.-H.; Kuo, Y.-M. Recycling of spent nickel-cadmium battery using a thermal separation process. Env. Prog. Sustain. Energy 2017, 37, 645–654. [Google Scholar] [CrossRef]
- Assefi, M.; Maroufi, S.; Mayyas, M.; Sahajwalla, V. Recycling of Ni-Cd batteries by selective isolation and hydrothermal synthesis of porous NiO nanocuboid. J. Environ. Chem. Eng. 2018, 6, 4671–4675. [Google Scholar] [CrossRef]
- Zelinsky, M.A.; Koch, J.; Young, K.-H. Performance Comparison of Rechargeable Batteries for Stationary Applications (Ni/MH vs. Ni–Cd and VRLA). Batteries 2017, 4, 1. [Google Scholar] [CrossRef]
- Moeller, K.-C. Overview of battery systems. In Lithium-Ion Batteries: Basics and Applications; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2018; pp. 3–10. [Google Scholar]
- Williams, J.B. Portable Power: Batteries. In The Electric Century; Springer Science and Business Media LLC: New York, NY, USA, 2017; pp. 84–92. [Google Scholar]
- Yazami, R.; Deschamps, M. The 13th International Seminar on Primary and Secondary Battery Technology and Application; Florida Educational Seminars: Boca Raton, FL, USA, 4–7 March 1996; p. 19960304. [Google Scholar]
- Dhar, S.; Ovshínsky, S.; Gifford, P.; Corrigan, D.; Fetcenko, M.; Venkatesan, S. Nickel/metal hydride technology for consumer and electric vehicle batteries—A review and up-date. J. Power Sour. 1997, 65, 1–7. [Google Scholar] [CrossRef]
- Rantik, M. Life Cycle Assessment of Five Batteries for Electric Vehicles under Different Charging Regimes, Technical Report for, Life Cycle Assessment of Five Batteries for Electric Vehicles under Different Charging Regimes; U.S. Department of Energy: Washington, DC, USA, 1999; ISSN 1401-1271. TRN: SE0007008.
- Fetcenko, M.; Ovshinsky, S.; Reichman, B.; Young, K.; Fierro, C.; Koch, J.; Zallen, A.; Mays, W.; Ouchi, T. Recent advances in NiMH battery technology. J. Power Sour. 2007, 165, 544–551. [Google Scholar] [CrossRef]
- Notten, P.H. Nickel-metal hydride batteries: From concept to characteristics. Hem. Ind. 2000, 54, 102–115. [Google Scholar]
- Qiao, S.; Hu, M.; Fu, C.; Qin, D.; Zhou, A.; Wang, P.; Lin, F. Experimental Study on Storage and Maintenance Method of Ni-MH Battery Modules for Hybrid Electric Vehicles. Appl. Sci. 2019, 9, 1742. [Google Scholar] [CrossRef]
- Noga, M.; Juda, Z. The application of nimh batteries in a light-duty electric vehicle. Czas. Tech. 2019, 1, 197–222. [Google Scholar] [CrossRef]
- Budde-Meiwes, H.; Drillkens, J.; Lunz, B.; Muennix, J.; Rothgang, S.; Kowal, J.; Sauer, D.U. A review of current automotive battery technology and future prospects. Proc. Inst. Mech. Eng. Part. D J. Automob. Eng. 2013, 227, 761–776. [Google Scholar] [CrossRef]
- El Kharbachi, A.; Zavorotynska, O.; Latroche, M.; Cuevas, F.; Yartys, V.; Fichtner, M. Exploits, advances and challenges benefiting beyond Li-ion battery technologies. J. Alloys Compd. 2020, 817, 153261. [Google Scholar] [CrossRef]
- Landi, B.J.; Ganter, M.J.; Cress, C.D.; Dileo, R.A.; Raffaelle, R.P. Carbon nanotubes for lithium ion batteries. Energy Environ. Sci. 2009, 2, 638–654. [Google Scholar] [CrossRef]
- Zubi, G.; Dufo-López, R.; Carvalho, M.; Pasaoglu, G. The lithium-ion battery: State of the art and future perspectives. Renew. Sustain. Energy Rev. 2018, 89, 292–308. [Google Scholar] [CrossRef]
- Wang, W.; Luo, Q.; Li, B.; Wei, X.; Li, L.; Yang, Z. Recent Progress in Redox Flow Battery Research and Development. Adv. Funct. Mater. 2013, 23, 970–986. [Google Scholar] [CrossRef]
- Li, L.; Kim, S.; Wang, W.; Vijayakumar, M.; Nie, Z.; Chen, B.; Zhang, J.; Xia, G.; Hu, J.; Graff, G.L.; et al. A Stable Vanadium Redox-Flow Battery with High Energy Density for Large-Scale Energy Storage. Adv. Energy Mater. 2011, 1, 394–400. [Google Scholar] [CrossRef]
- Gandomi, Y.A.; Aaron, D.S.; Houser, J.R.; Daugherty, M.C.; Clement, J.T.; Pezeshki, A.M.; Ertugrul, T.Y.; Moseley, D.P.; Mench, M.M. Critical Review—Experimental Diagnostics and Material Characterization Techniques Used on Redox Flow Batteries. J. Electrochem. Soc. 2018, 165, A970–A1010. [Google Scholar] [CrossRef]
- Ye, R.; Henkensmeier, D.; Yoon, S.J.; Huang, Z.; Kim, D.K.; Chang, Z.; Kim, S.; Chen, R. Redox Flow Batteries for Energy Storage: A Technology Review. J. Electrochem. Energy Convers. Storage 2017, 15, 010801. [Google Scholar] [CrossRef]
- Xu, Q.; Ji, Y.; Qin, L.; Leung, P.; Qiao, F.; Li, Y.; Su, H. Evaluation of redox flow batteries goes beyond round-trip efficiency: A technical review. J. Energy Storage 2018, 16, 108–115. [Google Scholar] [CrossRef]
- McCrabb, H.; Snyder, S.; Taylor, E.J. An Electrochemical Etching Process for Flow Battery Structures to Improve Performance and Reduce Manufacturing Cost. ECS Meet. Abstr. 2014. [Google Scholar] [CrossRef]
- Nguyen, T.; Savinell, R.F. Flow batteries. Elec. Soc. Interf. 2010, 19, 54. [Google Scholar] [CrossRef]
- Perry, M.L.; Fuller, T.F. A Historical Perspective of Fuel Cell Technology in the 20th Century. J. Electrochem. Soc. 2002, 149, S59–S67. [Google Scholar] [CrossRef]
- Sharaf, O.Z.; Orhan, M.F. An overview of fuel cell technology: Fundamentals and applications. Renew. Sustain. Energy Rev. 2014, 32, 810–853. [Google Scholar] [CrossRef]
- Hoed, R.V.D.; Vergragt, P.J. Institutional change in the automotive industry. In The Business of Sustainable Mobility; Informa UK Limited: Colchester, UK, 2017; pp. 61–79. [Google Scholar]
- Raza, R.; Akram, N.; Javed, M.S.; Rafique, A.; Ullah, K.; Ali, A.; Saleem, M.; Ahmed, R. Fuel cell technology for sustainable development in Pakistan—An over-view. Renew. Sustain. Energy Rev. 2016, 53, 450–461. [Google Scholar] [CrossRef]
- Alaswad, A.; Baroutaji, A.; Achour, H.; Carton, J.; Al Makky, A.; Olabi, A. Developments in fuel cell technologies in the transport sector. Int. J. Hydrogen Energy 2016, 41, 16499–16508. [Google Scholar] [CrossRef]
- Xu, Q.; Zhang, F.; Xu, L.; Leung, P.; Yang, C.; Li, H. The applications and prospect of fuel cells in medical field: A review. Renew. Sustain. Energy Rev. 2017, 67, 574–580. [Google Scholar] [CrossRef]
- Das, V.; Padmanaban, S.; Venkitusamy, K.; Selvamuthukumaran, R.; Blaabjerg, F.; Siano, P. Recent advances and challenges of fuel cell based power system architectures and control—A review. Renew. Sustain. Energy Rev. 2017, 73, 10–18. [Google Scholar] [CrossRef]
- Mekhilef, S.; Saidur, R.; Safari, A. Comparative study of different fuel cell technologies. Renew. Sustain. Energy Rev. 2012, 16, 981–989. [Google Scholar] [CrossRef]
- Dong, X.; Tian, L.; Li, J.; Zhao, Y.; Tian, Y.; Li, Y. Single layer fuel cell based on a composite of Ce0.8Sm0.2O2−δ–Na2CO3 and a mixed ionic and electronic conductor Sr2Fe1.5Mo0.5O6−δ. J. Power Sour. 2014, 249, 270–276. [Google Scholar] [CrossRef]
- Nymand, M.; Andersen, M.A.E. A new approach to high efficiency in isolated boost converters for high-power low-voltage fuel cell applications. In Proceedings of the 2008 13th International Power Electronics and Motion Control Conference, Poznan, Poland, 1–3 September 2008; Institute of Electrical and Electronics Engineers (IEEE): Piscataway Township, NJ, USA, 2008; pp. 127–131. [Google Scholar]
- Wang, W.; Su, C.; Wu, Y.; Ran, R.; Shao, Z. Progress in Solid Oxide Fuel Cells with Nickel-Based Anodes Operating on Methane and Related Fuels. Chem. Rev. 2013, 113, 8104–8151. [Google Scholar] [CrossRef]
- Song, X.; Dong, X.; Li, M.; Wang, H. Effects of adding alumina to the nickel-zirconia anode materials for solid oxide fuel cells and a two-step sintering method for half-cells. J. Power Sour. 2016, 308, 58–64. [Google Scholar] [CrossRef]
- Da Silva, F.S.; De Souza, T.M. Novel materials for solid oxide fuel cell technologies: A literature review. Int. J. Hydrogen Energy 2017, 42, 26020–26036. [Google Scholar] [CrossRef]
- Alnegren, P.; Sattari, M.; Froitzheim, J.; Svensson, J.-E. Degradation of ferritic stainless steels under conditions used for solid oxide fuel cells and electrolyzers at varying oxygen pressures. Corros. Sci. 2016, 110, 200–212. [Google Scholar] [CrossRef]
- Fan, L.; Zhu, B.; Su, P.-C.; He, C. Nanomaterials and technologies for low temperature solid oxide fuel cells: Recent advances, challenges and opportunities. Nano Energy 2018, 45, 148–176. [Google Scholar] [CrossRef]
- Steele, B.C.H.; Heinzel, A. Materials for fuel-cell technologies. Mater. Sustain. Energy 2010, 224–231. [Google Scholar] [CrossRef]
- Mahato, N.; Banerjee, A.; Gupta, A.; Omar, S.; Balani, K. Progress in material selection for solid oxide fuel cell technology: A review. Prog. Mater. Sci. 2015, 72, 141–337. [Google Scholar] [CrossRef]
- Irvine, J.; Tao, S. Fuel Electrodes for Solid Oxide Fuel Cells. Lubr. Addit. 2005, 8, 739–765. [Google Scholar] [CrossRef]
- Bae, K.; Jang, D.Y.; Choi, H.J.; Kim, D.; Hong, J.; Kim, B.-K.; Lee, J.-H.; Son, J.-W.; Shim, J.H. Demonstrating the potential of yttrium-doped barium zirconate electrolyte for high-performance fuel cells. Nat. Commun. 2017, 8, 14553. [Google Scholar] [CrossRef] [PubMed]
- Shaheen, K.; Shah, Z.; Gulab, H.; Hanif, M.B.; Faisal, S.; Suo, H. Metal oxide nanocomposites as anode and cathode for low temperature solid oxide fuel cell. Solid State Sci. 2020, 102, 106162. [Google Scholar] [CrossRef]
- Goor, M.; Menkin, S.; Peled, E. High power direct methanol fuel cell for mobility and portable applications. Int. J. Hydrogen Energy 2019, 44, 3138–3143. [Google Scholar] [CrossRef]
- Das, S.; Dutta, K.; Nessim, G.D.; Kader, M.A. Direct Methanol Fuel Cell Technology. In Direct Methanol Fuel Cell Technology; Elsevier BV: Amsterdam, The Netherlands, 2020; pp. 1–12. [Google Scholar]
- Xia, Z.; Zhang, X.; Sun, H.; Wang, S.; Sun, G. Recent advances in multi-scale design and construction of materials for direct methanol fuel cells. Nano Energy 2019, 65, 104048. [Google Scholar] [CrossRef]
- Tiwari, J.N.; Tiwari, R.N.; Singh, G.; Kim, K.S. Recent progress in the development of anode and cathode catalysts for direct methanol fuel cells. Nano Energy 2013, 2, 553–578. [Google Scholar] [CrossRef]
- Vielstich, W.; Lamm, A.; Gasteiger, H.A. Handbook of Fuel Cells: Fundamentals Technology and Applications; Wiley: New York, NY, USA, 2003; p. 2690. [Google Scholar]
- Behret, H. Karl Kordesch und Günter Simader: Fuel Cells and Their Applications, VCH, Weinheim, 1996. ISBN 3-527-28579-2, DM 248,-. Ber. Der Bunsenges. Für Phys. Chem. 1996, 100, 1922. [Google Scholar] [CrossRef]
- Jung, G.-B.; Tu, C.-H.; Chi, P.-H.; Su, A.; Weng, F.-B.; Lin, Y.-T.; Chiang, Y.-C.; Lee, C.-Y.; Yan, W.-M. Investigations of flow field designs in direct methanol fuel cell. J. Solid State Electrochem. 2008, 13, 1455–1465. [Google Scholar] [CrossRef]
- Yan, X.; Wu, R.; Xu, J.; Luo, Z.; Zhao, T. A monolayer graphene—Nafion sandwich membrane for direct methanol fuel cells. J. Power Sour. 2016, 311, 188–194. [Google Scholar] [CrossRef]
- Ong, B.; Kamarudin, S.; Basri, S. Direct liquid fuel cells: A review. Int. J. Hydrogen Energy 2017, 42, 10142–10157. [Google Scholar] [CrossRef]
- Gong, L.; Yang, Z.; Li, K.; Xing, W.; Liu, C.; Ge, J. Recent development of methanol electrooxidation catalysts for direct methanol fuel cell. J. Energy Chem. 2018, 27, 1618–1628. [Google Scholar] [CrossRef]
- Kakati, B.K.; Mohan, V. Development of Low-Cost Advanced Composite Bipolar Plate for Proton Exchange Membrane Fuel Cell? Fuel Cells 2008, 8, 45–51. [Google Scholar] [CrossRef]
- Authayanun, S.; Im-Orb, K.; Arpornwichanop, A. A review of the development of high temperature proton exchange membrane fuel cells. Chin. J. Catal. 2015, 36, 473–483. [Google Scholar] [CrossRef]
- Cropper, M.A.; Geiger, S.; Jollie, D.M. Fuel cells: A survey of current developments. J. Power Sour. 2004, 131, 57–61. [Google Scholar] [CrossRef]
- Jiao, K.; Li, X. Water transport in polymer electrolyte membrane fuel cells. Prog. Energy Combust. Sci. 2011, 37, 221–291. [Google Scholar] [CrossRef]
- Verhage, A.J.; Coolegem, J.F.; Mulder, M.J.; Yildirim, M.H.; De Bruijn, F.A. 30,000 h operation of a 70 kW stationary PEM fuel cell system using hydrogen from a chlorine factory. Int. J. Hydrogen Energy 2013, 38, 4714–4724. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, K.S.; Mishler, J.; Cho, S.C.; Adroher, X.C. A review of polymer electrolyte membrane fuel cells: Technology, applications, and needs on fundamental research. Appl. Energy 2011, 88, 981–1007. [Google Scholar] [CrossRef]
- Gasteiger, H.; Panels, J.; Yan, S. Dependence of PEM fuel cell performance on catalyst loading. J. Power Sour. 2004, 127, 162–171. [Google Scholar] [CrossRef]
- Akimoto, Y.; Okajima, K. Semi-Empirical Equation of PEMFC Considering Operation Temperature. Energy Technol. Policy 2014, 1, 91–96. [Google Scholar] [CrossRef]
- Sutharssan, T.; Montalvao, D.; Chen, Y.K.; Wang, W.-C.; Pisac, C.; Elemara, H. A review on prognostics and health monitoring of proton exchange membrane fuel cell. Renew. Sustain. Energy Rev. 2017, 75, 440–450. [Google Scholar] [CrossRef]
- Borup, R.; Davey, J.; Wood, D.; Garzon, F.; Inbody, M.; Guidry, D. VII. I.3 PEM fuel cell durability. 2005. Available online: https://www.hydrogen.energy.gov/pdfs/progress05/vii_i_3_borup.pdf (accessed on 17 October 2020).
- Wargo, E.; Dennison, C.; Kumbur, E. Durability of Polymer Electrolyte Fuel Cells. Polym. Electrolyte Fuel Cell Degrad. 2012, 1–14. [Google Scholar] [CrossRef]
- Moreno, N.G.; Molina, M.C.; Gervasio, D.F.; Robles, J.F.P. Approaches to polymer electrolyte membrane fuel cells (PEMFCs) and their cost. Renew. Sustain. Energy Rev. 2015, 52, 897–906. [Google Scholar] [CrossRef]
- Zhu, B.; Raza, R.; Abbas, G.; Singh, M. An Electrolyte-Free Fuel Cell Constructed from One Homogenous Layer with Mixed Conductivity. Adv. Funct. Mater. 2011, 21, 2465–2469. [Google Scholar] [CrossRef]
- Zhu, B.; Lund, P.; Raza, R.; Patakangas, J.; Huang, Q.-A.; Fan, L.; Singh, M. A new energy conversion technology based on nano-redox and nano-device processes. Nano Energy 2013, 2, 1179–1185. [Google Scholar] [CrossRef]
- Zhu, B.; Raza, R.; Qin, H.; Fan, L. Single-component and three-component fuel cells. J. Power Sour. 2011, 196, 6362–6365. [Google Scholar] [CrossRef]
- Wang, C.; Nehrir, M.H. Distributed Generation Applications of Fuel Cells. In Proceedings of the 2006 Power Systems Conference: Advanced Metering, Protection, Control, Communication, and Distributed Resources, Clemson, SC, USA, 14–17 March 2006; Institute of Electrical and Electronics Engineers (IEEE): Piscataway Township, NJ, USA, 2006; pp. 244–248. [Google Scholar]
- Ozawa, K. Lithium Ion Rechargeable Batteries: Materials, Technology, and New Applications; John Wiley & Sons: Hoboken, NJ, USA, 2012; pp. 1–329. ISBN 978-3-527.31983-1. [Google Scholar]
- Conway, B.E. Electrochemical Supercapacitors; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 1999; pp. 1–698. [Google Scholar]
- Bohlen, O.; Kowal, J.; Sauer, D.U. Ageing behaviour of electrochemical double layer capacitors. J. Power Sour. 2007, 173, 626–632. [Google Scholar] [CrossRef]
- Simon, P.; Brousse, T.; Favier, F. Supercapacitors Based on Carbon or Pseudocapacitive Materials; Wiley: New York, NY, USA, 2017. [Google Scholar]
- Miller, J.R.; Simon, P. Materials Science: Electrochemical Capacitors for Energy Management. Science 2008, 321, 651–652. [Google Scholar] [CrossRef]
- Ricketts, B.; Ton-That, C. Self-discharge of carbon-based supercapacitors with organic electrolytes. J. Power Sour. 2000, 89, 64–69. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhao, Y.; An, W.; Xing, L.; Gao, Y.; Liu, J. Heteroelement Y-doped α-Ni(OH)2 nanosheets with excellent pseudocapacitive performance. J. Mater. Chem. A 2017, 5, 10039–10047. [Google Scholar] [CrossRef]
- Sasaki, M.; Araki, S.; Miyata, T.; Kawaji, T. Development of capacitor hybrid system for urban buses. JSAE Rev. 2002, 23, 451–457. [Google Scholar] [CrossRef]
- Martín, J.S.; Zamora, I.; Martín, J.S.; Aperribay, V.; Eguia, P. Energy storage technologies for electric applications. Renew. Energy Power Qual. J. 2011, 593–598. [Google Scholar] [CrossRef]
- Rydh, C.J.; Sandén, B.A. Energy analysis of batteries in photovoltaic systems. Part II: Energy return factors and overall battery efficiencies. Energy Convers. Manag. 2005, 46, 1980–2000. [Google Scholar] [CrossRef]
- Author, N. Review of electrical energy storage technologies and systems and of their potential for the UK. EA Technol. 2004, 1, 34. [Google Scholar]
- Baker, J. New technology and possible advances in energy storage. Energy Policy 2008, 36, 4368–4373. [Google Scholar] [CrossRef]
- McDowall, J. Integrating energy storage with wind power in weak electricity grids. J. Power Sour. 2006, 162, 959–964. [Google Scholar] [CrossRef]
- Ren, Z.; Yu, J.; Li, Y.; Zhi, C. Tunable Free-Standing Ultrathin Porous Nickel Film for High Performance Flexible Nickel-Metal Hydride Batteries. Adv. Energy Mater. 2018, 8, 1702467. [Google Scholar] [CrossRef]
- Kong, L.; Li, X.; Liao, X.; Young, K.-H. A BCC-C14 alloy suitable for EV application of Ni/MH battery. Int. J. Hydrogen Energy 2019, 44, 29338–29343. [Google Scholar] [CrossRef]
- Meng, T.; Young, K.-H.; Hu, C.; Reichman, B. Effects of Alkaline Pre-Etching to Metal Hydride Alloys. Batteries 2017, 3, 30. [Google Scholar] [CrossRef]
- Shen, Y.; Noréus, D.; Starborg, S. Increasing NiMH battery cycle life with oxygen. Int. J. Hydrogen Energy 2018, 43, 18626–18631. [Google Scholar] [CrossRef]
- Idowu, I.A. Feasibility Study on the Use of Recycled NIMH Batteries for Emergency Power during a Natural Disaster; ProQuest Dissertations Publishing LLC: Morgan State University, MD, USA, 2018. [Google Scholar]
- Barote, L.; Weissbach, R.; Teodorescu, R.; Marinescu, C.; Cîrstea, M. Stand-alone wind system with Vanadium Redox Battery energy storage. In Proceedings of the 2008 11th International Conference on Optimization of Electrical and Electronic Equipment, Brasov, Romania, 22–24 May 2008; Institute of Electrical and Electronics Engineers (IEEE): Berlin/Heidelberg, 2008; pp. 407–412. [Google Scholar]
- De León, C.P.; Ferrer Ángel, F.; García, J.G.; Szánto, D.; Walsh, F.C. Redox flow cells for energy conversion. J. Power Sour. 2006, 160, 716–732. [Google Scholar] [CrossRef]
- Amodeo, S.J.; Chiacchiarini, H.; Solsona, J.; Busada, C.A. High-performance sensorless nonlinear power control of a flywheel energy storage system. Energy Convers. Manag. 2009, 50, 1722–1729. [Google Scholar] [CrossRef]
- Pistoia, G. Electric and Hybrid. Vehicles; Elsevier BV: Amsterdam, The Netherlands, 2010. [Google Scholar]
- Li, P. Energy storage is the core of renewable technologies. IEEE Nanotechnol. Mag. 2008, 2, 13–18. [Google Scholar] [CrossRef]
- Yang, Z.; Zhang, J.; Kintner-Meyer, M.C.W.; Lu, X.; Choi, D.; Lemmon, J.P.; Liu, J. Electrochemical Energy Storage for Green Grid. Chem. Rev. 2011, 111, 3577–3613. [Google Scholar] [CrossRef]
- Zhang, S.; Guo, W.; Yang, F.; Zheng, P.; Qiao, R.; Li, Z. Recent Progress in Polysulfide Redox-Flow Batteries. Batter. Supercaps 2019, 2, 627–637. [Google Scholar] [CrossRef]
- Zhang, C.; Ding, Y.; Zhang, L.; Wang, X.; Zhao, Y.; Zhang, X.; Yu, G. A Sustainable Redox-Flow Battery with an Aluminum-Based, Deep-Eutectic-Solvent Anolyte. Angew. Chem. Int. Ed. 2017, 56, 7454–7459. [Google Scholar] [CrossRef]
- Cheng, Y.; Zhang, H.; Lai, Q.; Li, X.; Shi, D.; Zhang, L. A high power density single flow zinc–nickel battery with three-dimensional porous negative electrode. J. Power Sour. 2013, 241, 196–202. [Google Scholar] [CrossRef]
- Zhang, L.; Lai, Q.; Zhang, J.; Zhang, H. A High-Energy-Density Redox Flow Battery based on Zinc/Polyhalide Chemistry. ChemSusChem 2012, 5, 867–869. [Google Scholar] [CrossRef]
- Yang, Y.; Zheng, G.; Cui, Y. A Membrane-Free Lithium/Polysulfide Semi-Liquid Battery for Large-Scale Energy Storage. ECS Meet. Abstr. 2013, 6, 1552–1558. [Google Scholar] [CrossRef]
- Li, Z.; Weng, G.; Zou, Q.; Cong, G.; Lu, Y.-C. A high-energy and low-cost polysulfide/iodide redox flow battery. Nano Energy 2016, 30, 283–292. [Google Scholar] [CrossRef]
- Mah, J.C.; Muchtar, A.; Somalu, M.R.; Ghazali, M.J. Metallic interconnects for solid oxide fuel cell: A review on protective coating and deposition techniques. Int. J. Hydrogen Energy 2017, 42, 9219–9229. [Google Scholar] [CrossRef]
- Cable, T.L.; Sofie, S.W. A symmetrical, planar SOFC design for NASA’s high specific power density requirements. J. Power Sour. 2007, 174, 221–227. [Google Scholar] [CrossRef]
- Yang, Z. Recent advances in metallic interconnects for solid oxide fuel cells. Int. Mater. Rev. 2008, 53, 39–54. [Google Scholar] [CrossRef]
- Huang, Y.-H.; Dass, R.I.; Xing, Z.-L.; Goodenough, J.B. Double Perovskites as Anode Materials for Solid-Oxide Fuel Cells. Chemin 2006, 37, 254–257. [Google Scholar] [CrossRef]
- Chou, Y.; Stevenson, J. Long-term thermal cycling of Phlogopite mica-based compressive seals for solid oxide fuel cells. J. Power Sour. 2005, 140, 340–345. [Google Scholar] [CrossRef]
- Guida, D.; Minutillo, M. Design methodology for a PEM fuel cell power system in a more electrical aircraft. Appl. Energy 2017, 192, 446–456. [Google Scholar] [CrossRef]
- Gang, B.G.; Kwon, S. All-in-one portable electric power plant using proton exchange membrane fuel cells for mobile applications. Int. J. Hydrogen Energy 2018, 43, 6331–6339. [Google Scholar] [CrossRef]
- Farnes, J.; Bokach, D.; Hoopen, S.T.; Skåtun, K.; Geneste, X.; Vik, A.; Schautz, M. Optimized High Temperature PEM Fuel Cell & High Pressure PEM Electrolyser for Regenerative Fuel Cell Systems in GEO Telecommunication Satellites. In Proceedings of the E3S Web of Conferences, EDP Sciences, Porto Palace Thessaloniki, Greece, 3–7 October 2017; Volume 16, p. 10004. [Google Scholar]
- Cano, Z.P.; Banhamd, D.; Ye, S.; Hintennach, A.; Lu, J.; Fowler, M.; Chen, Z. Batteries and fuel cells for emerging electric vehicle markets. Nat. Energy 2018, 3, 279–289. [Google Scholar] [CrossRef]
- Barbir, F. Efficiency and economics of proton exchange membrane (PEM) fuel cells. Int. J. Hydrogen Energy 1997, 22, 1027–1037. [Google Scholar] [CrossRef]
- Shaneeth, M.; Basu, S.; Aravamuthan, S.; Suddhasatwa, B. PEM fuel cell cathode catalyst layer durability: An electrochemical spectroscopic investigation. Chem. Eng. Sci. 2016, 154, 72–80. [Google Scholar] [CrossRef]
- Yamaguchi, T.; Zhou, H.; Nakazawa, S.; Hara, N. An Extremely Low Methanol Crossover and Highly Durable Aromatic Pore-Filling Electrolyte Membrane for Direct Methanol Fuel Cells. Adv. Mater. 2007, 19, 592–596. [Google Scholar] [CrossRef]
- Kimiaie, N.; Wedlich, K.; Hehemann, M.; Lambertz, R.; Müller, M.; Korte, C.; Stolten, D. Results of a 20,000 h lifetime test of a 7 kW direct methanol fuel cell (DMFC) hybrid system—Degradation of the DMFC stack and the energy storage. Energy Env. Sci. 2014, 7, 3013–3025. [Google Scholar] [CrossRef]
- Müller, M.; Kimiaie, N.; Glüsen, A. Direct methanol fuel cell systems for backup power—Influence of the standby procedure on the lifetime. Int. J. Hydrogen Energy 2014, 39, 21739–21745. [Google Scholar] [CrossRef]
- Haldorai, Y.; Arreaga-Salas, D.; Rak, C.S.; Huh, Y.S.; Han, Y.-K.; Voit, W. Platinized titanium nitride/graphene ternary hybrids for direct methanol fuel cells and titanium nitride/graphene composites for high performance supercapacitors. Electrochim. Acta 2016, 220, 465–474. [Google Scholar] [CrossRef]
- Pawar, S.M.; Kim, J.; Inamdar, A.I.; Woo, H.; Jo, Y.; Pawar, B.S.; Cho, S.; Kim, H.; Im, H. Multi-functional reactively-sputtered copper oxide electrodes for supercapacitor and electro-catalyst in direct methanol fuel cell applications. Sci. Rep. 2016, 6, 21310. [Google Scholar] [CrossRef]
- Grigoriev, A.S. A Hybrid Power Plant Based on Renewables and Electrochemical Energy Storage and Generation Systems for Decentralized Electricity Supply of the Northern Territories. Int. J. Electrochem. Sci. 2018, 13, 1822–1830. [Google Scholar] [CrossRef]
- Zhan, C.; Lian, C.; Zhang, Y.; Thompson, M.W.; Xie, Y.; Wu, J.; Kent, P.R.C.; Cummings, P.T.; Jiang, D.-E.; Wesolowski, D.J. Computational Insights into Materials and Interfaces for Capacitive Energy Storage. Adv. Sci. 2017, 4, 1700059. [Google Scholar] [CrossRef]
- Grazioli, D.; Magri, M.; Salvadori, A.A. Computational modeling of Li-ion batteries. Comput. Mech. 2016, 58, 889–909. [Google Scholar] [CrossRef]
- Salvadori, A.; Grazioli, D. Computer simulation for battery design and lifetime prediction. In Advances in Battery Technologies for Electric Vehicles Scrosati; Garche, B.J., Tillmetz, W., Eds.; Woodhead Publishing: Cambridge, UK, 2015; pp. 417–442. [Google Scholar]
- Franco, A.A. Multiscale modelling and numerical simulation of rechargeable lithium ion batteries: Concepts, methods and challenges. RSC Adv. 2013, 3, 13027–13058. [Google Scholar] [CrossRef]
- Feng, G.; Qiao, R.; Huang, J.; Sumpter, B.G.; Meunier, V. Computational Modelling of Carbon Nanostructures for Energy Storage Applications. In Proceedings of the 10th IEEE international Conference on Nanotechnology joint Symposium Nano Korea, Kintex, Korea, 17–20 August 2010; pp. 100–104. [Google Scholar]
- Chen, Y.K.; Enearu, O.; Montalvão, D.; Sutharssan, T. A Review of Computational Fluid Dynamics Simulations on PEFC Performance. J. Appl. Mech. Eng. 2016, 5. [Google Scholar] [CrossRef]
- Springer, T.E.; Wilson, M.S.; Gottesfeld, S. Modeling and Experimental Diagnostics in Polymer Electrolyte Fuel Cells. J. Electrochem. Soc. 2019, 140, 3513–3526. [Google Scholar] [CrossRef]
- Zhang, L.; Huc, X.; Wang, Z.; Sun, F.; Dorrell, D.G. A review of supercapacitor modelling, estimation, and applications: A control/management perspective. Renew. Sust. Energ. Rev. 2018, 81, 1868–1878. [Google Scholar] [CrossRef]
- Bououchma, Z.; Sabor, J.; Aitbouh, H. New electrical model of supercapacitors for electric hybrid vehicle applications. Mater. Today: Proc. 2019, 13, 688–697. [Google Scholar] [CrossRef]
- Newman, J.; Thomas-Alyea, K.E. Electrochemical Systems; John Wiley & Sons: Hoboken, NJ, USA, 2012. [Google Scholar]
- Lin, C.; Popov, B.N.; Ploehn, H.J. Modeling the Effects of Electrode Composition and Pore Structure on the Performance of Electrochemical Capacitors. J. Electrochem. Soc. 2002, 149, A167–A175. [Google Scholar] [CrossRef][Green Version]
- Ers, H.; Lembinen, M.; Misin, M.; Seitsonen, A.P.; Fedorov, M.V.; Ivaništšev, V.B. Graphene-Ionic Liquid Interfacial Potential Drop from Density Functional Theory-Based Molecular Dynamics Simulations. J. Phys. Chem. C 2020, 124, 19548–19555. [Google Scholar] [CrossRef]
- Wasalathilake, K.C.; Ayoko, G.A.; Yan, C. Effects of heteroatom doping on the performance of graphene in sodium-ion batteries: A density functional theory investigation. Carbon 2018, 140, 276–285. [Google Scholar] [CrossRef]
- Case, D.; McSloy, A.J.; Sharpe, R.; Yeandel, S.R.; Bartlett, T.; Cookson, J.; Dashjav, E.; Tietz, F.; Kumar, C.M.N.; Goddard, P. Structure and ion transport of lithium-rich Li1+xAlxTi2− x (PO4) 3 with 0.3< x< 0.5: A combined computational and experimental study. Solid State Ionics 2020, 346, 115192. [Google Scholar]
- Dashjav, E.; Tietz, F. Neutron Diffraction Analysis of NASICON-type Li1+xAlxTi2-xP3O12. Z. Für Anorg. Allg. Chem. 2014, 640, 3070–3073. [Google Scholar] [CrossRef]
- Jeon, D.H. Computational fluid dynamics simulation of anode-supported solid oxide fuel cells with implementing complete overpotential model. Energy 2019, 188, 116050. [Google Scholar] [CrossRef]
- Zhang, S.S. Status, Opportunities, and Challenges of Electrochemical Energy Storage. Front. Energy Res. 2013, 1, 1. [Google Scholar] [CrossRef]
- Kim, B.K.; Sy, S.; Yu, A.; Zhang, J. Electrochemical Supercapacitors for Energy Storage and Conversion; Wiley: Hoboken, NJ, USA, 2015; pp. 1–25. [Google Scholar]
- Marques, F.M. Grand Challenges in Fuel Cells: Materials Issues at all Length Scales. Front. Energy Res. 2013, 1, 1. [Google Scholar] [CrossRef]
- Tarascon, J.-M. Key challenges in future Li-battery research. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2010, 368, 3227–3241. [Google Scholar] [CrossRef]

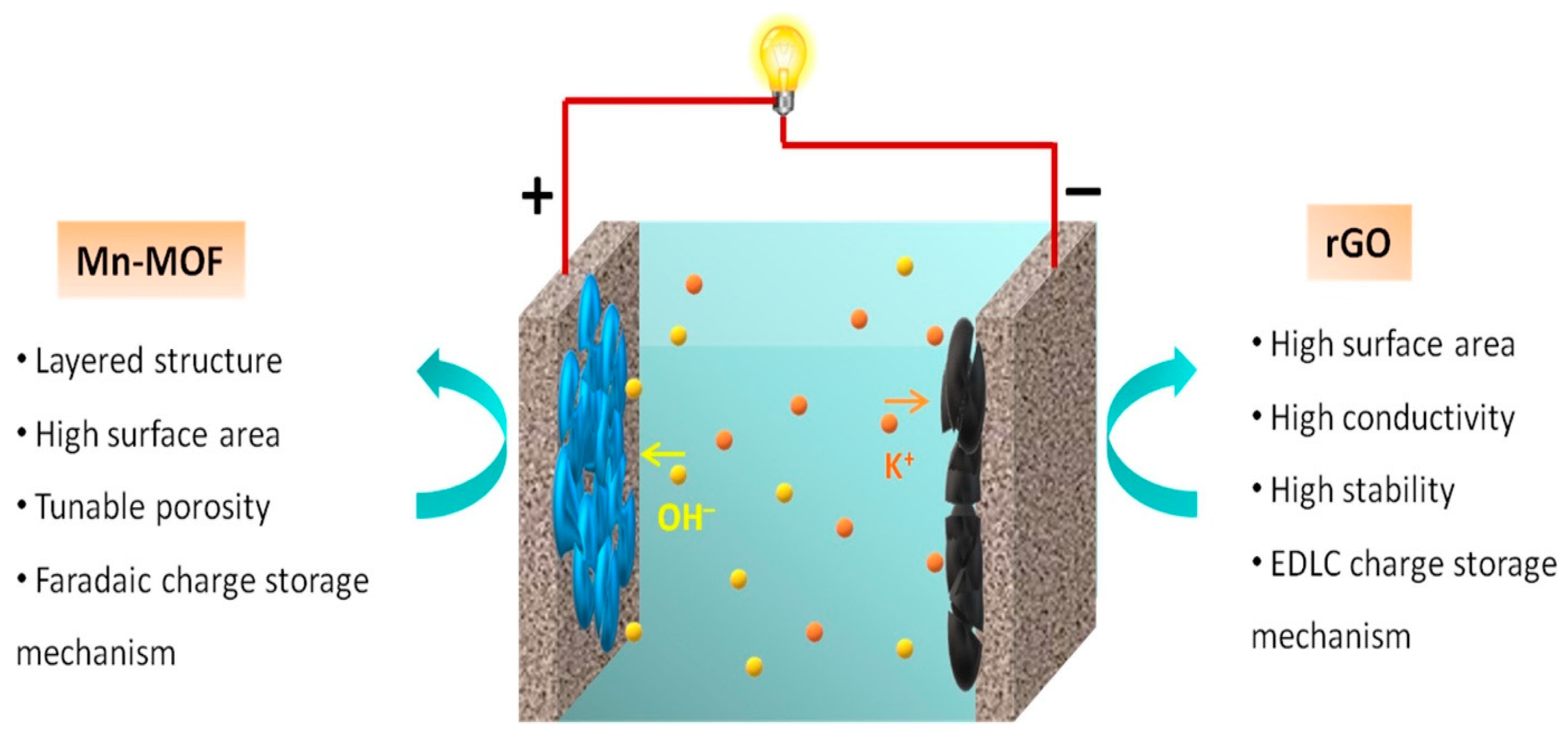
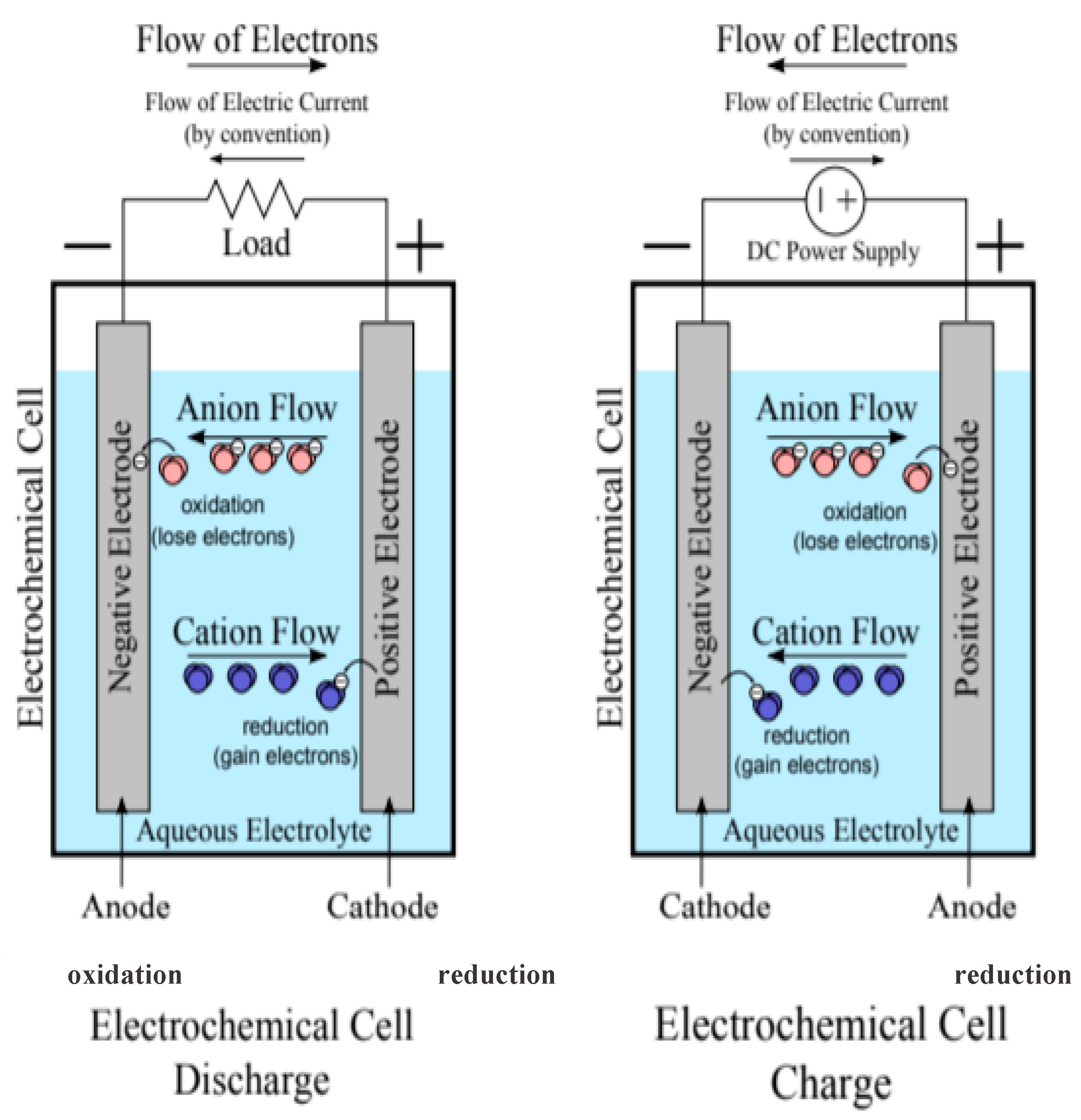
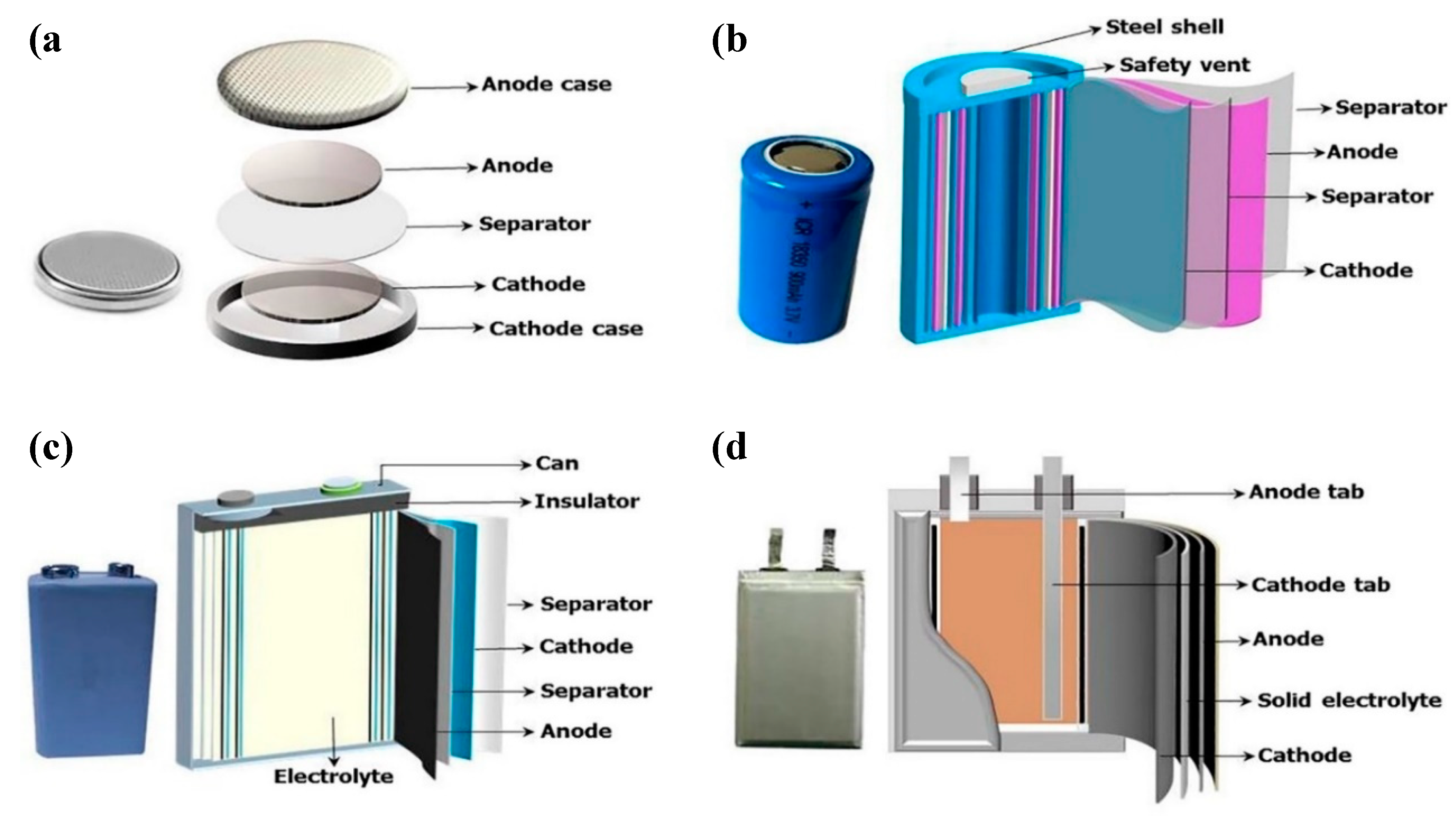

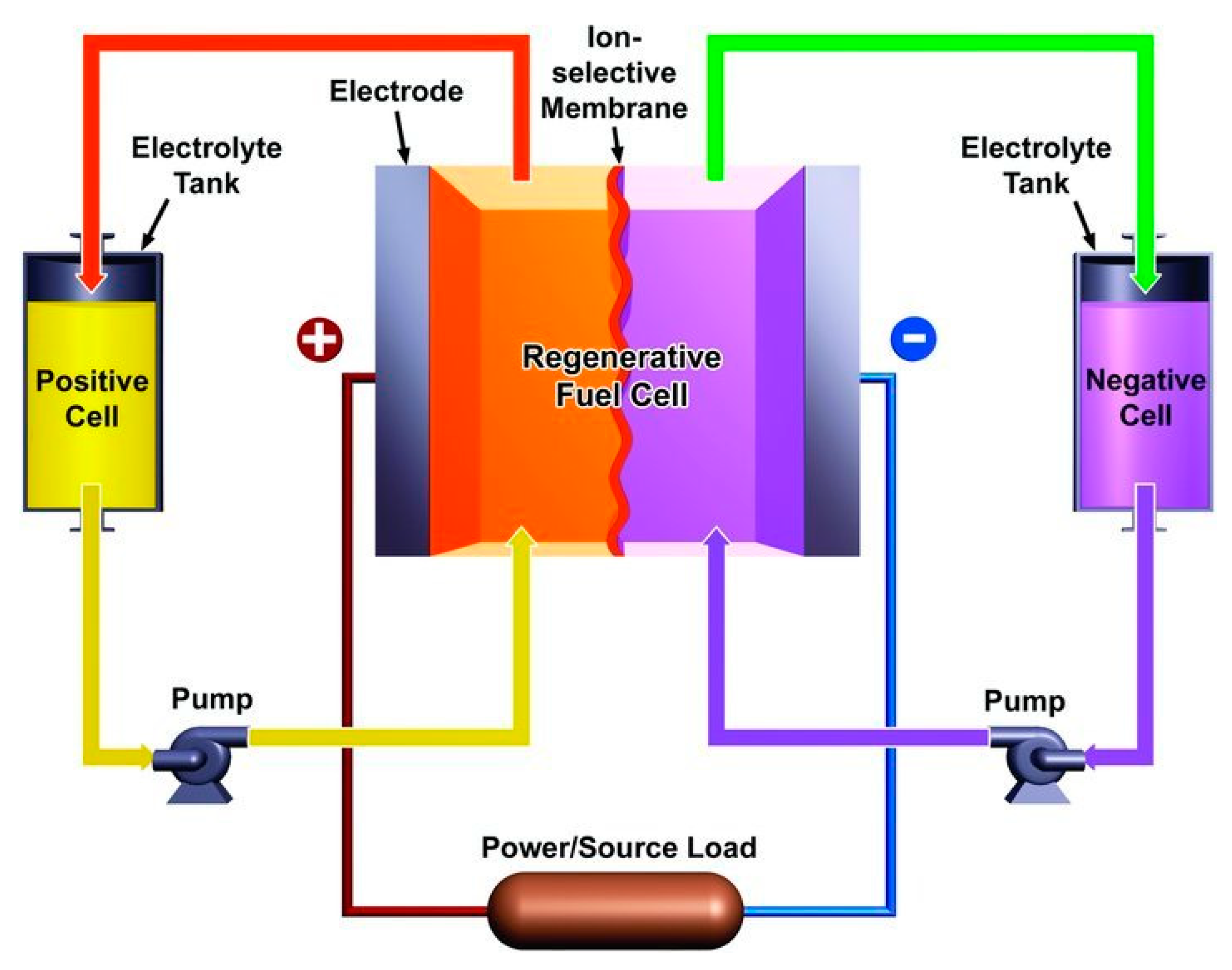
| Electric Double Layer Capacitors (EDLCs) | Pseudo-Capacitors (PCs) | Hybrid Capacitor (HCs) | |
|---|---|---|---|
| Electrode material | Carbon-based materials: activated carbon, graphene, carbon nanotubes and polymeric carbons | MOs: MnO2, NiO and RuO2. CPs: PANI, PT and PPY | Combination of EDL material and pseudo-capacitive material |
| Specific capacitance (F g−1) | 200–300 [37] | 200–1340 [38] | 50–1893 [39] |
| Energy density (Wh kg−1) | 6.8–12 [40] | 167–223 [41] | 132–231 [42] |
| Power density (kW kg−1) | 98,000–165 [40,43] | 21,000–220 [41] | 2800–57 [42] |
| Cyclability (cycles) | 100,000 [44] | 5000 [45] | 12,000 [46] |
| Capacitance retention (%) | 60–100 [47,48] | 52–96 [49,50,51] | 80–95 [52,53] |
| Charge storage mechanism | Physical | Chemical | physical + chemical |
| Battery Type | Chemical Reaction at Anodes and Cathodes | Voltage (V) |
|---|---|---|
| Lead-acid | 2.0 | |
| Lithium-ion | 3.7 | |
| Nickel-cadmium | 1.0–1.3 | |
| Nickel-metal hybrid | 1.0–1.3 | |
| Component | Typical Material | PHEV 20° | PHEV 40° | EV | Laptop |
|---|---|---|---|---|---|
| Cathode | LiCoCO2, LiMn2O4 LiNiO2 | 36 | 40 | 33 | 25 |
| Anode | Graphite (LiC6) Li4Ti5O12, Cu | 31 | 29 | 21 | 20 |
| Electrolyte b | ECM c Propylene carbonate LiPF6, LiBF4, LiClO4 | 11 | 12 | 7 | 12 |
| Separator | PP | 2 | 1 | 4 | 3 |
| Case | Steel, PE/PP, Al, Cu, Fe | 20 | 17 | 19 | 40 |
| Others a | Cu, PE/PP, Steel, Fe | 16 |
| Material (%) | Industrial [79] | EV battery [80] |
|---|---|---|
| Lead | 25 | 69 * |
| Lead oxide | 35 | |
| Polypropylene | 10 | 4 |
| Sulphuric acid | 10 | 11 |
| Water | 16 | 18 |
| Glass | 2 | 4 |
| Antimony | 1 |
| Material (%) | Automotive [79] | Portable Electronic [93] |
|---|---|---|
| Cd | 25 | 16.4 |
| Ni powder | 20 | 20.5 |
| Ni (OH)2 | 17 | 8.1 |
| KOH | 5 | 3.5 |
| Plastic | 3 | 3.7 |
| Steel & copper | 16 | 39 |
| Water | 12 | 8 |
| Others | 2 |
| Material | NiMH-AB2 a Percentage | NiMH-AB5 b Percentage |
|---|---|---|
| Ni | 12 | 15 |
| Ni(OH)2 | 12 | 15 |
| M: Ni, Ti, V, Zr | 13 | |
| M: La, Nd, Pr, Ce | 8 | |
| KOH | 3 | 3 |
| PP | 5 | 5 |
| Steel | 44 | 44 |
| Others | 11 | 10 |
| Battery Type | Advantages | Disadvantages |
|---|---|---|
| Lead-acid | (i) Mature technology (ii) Word wide production (iii) Low material cost (iv) No memory effect (v) Low self-discharge rate (vi) Relatively low capital cost | (i) Short cycle life (ii) Modest energy/power density (iii) Long charging time (iv) Safety issues (gas discharge) (v) Temperature sensitive output (vi) Poor reliability |
| Lithium-ion (Li-ion) | (i) Long cycle life (ii) High round trip efficiency (iii) Global R&D efforts (iv) Relatively fast charging (v) Highly reliable (vi) Low discharge rates (vii) Excellent energy/power density | (i) High capital cost (ii) Safety issues (Thermal runaway) (iii) Material bottle concerns (iv) Poor recovery/recycling (v) Advanced battery management systems required |
| Nickel metal hydride (NiMH) | (i) Modest initial cost (ii) Acceptable energy/power density (iii) Modest round-trip efficiency (iv) Highly reliable (v) Excellent safety record (vi) Relatively fast charging (vii) Eco-friendly materials (viii) Low operational maintenance | (i) Higher self-discharge rate (ii) Memory effect (iii) Relatively short cycle life (iv) Poor recovery/recycling |
| Nickel-cadmium (Ni-Cd) | (i) Comparatively low capital cost (ii) Highly reliable (iii) Mature technology (iv) Superb safety record (v) Wide operating temperatures (vi) Relatively fast recharge (vii) Excellent cycle life (viii) Low operational maintenance | (i) Modest energy/power density (ii) Memory effect (iii) Relatively poor round trip efficiency (iv) Reliance on hazardous cadmium |
| Technology | Specific Energy (Wh kg−1) | Specific Power (Wkg−1) | Life-Time (Years) | Cycle-Ability (Cycles) | Cyclic Efficiency (%) | Daily Discharge (%) |
|---|---|---|---|---|---|---|
| Different operating parameters of electrochemical capacitors | ||||||
| EDLCs | 6.8–12 [40] | 65–10,200 [41] | <30 [168,169] | Up to 500,000 [170] | 60–100 [44] | ~25 [171] |
| PCs | 23–67 [41] | 21,000–220 [41] | 5–9 [172] | Up to 5000 [44] | 52–96 [49,51] | |
| HCs | 132–231 [42] | 2800–57 [42] | <10 [173] | 12,000 [46] | 80–95 [52,53] | |
| Different operating parameters of Batteries | ||||||
| Lead-acid | 25–50 [174] | 10–400 [60] | 5–15 [84] | 200–1800 [61] | 63–90 [83] | 0.1–0.3 [60] |
| Li-ion | 75–200 [60] | 500–2000 [84] | 14–16 [175] | Up to 20,000 [176] | 75–90 [89] | 1–5 [61] |
| NiCd | 45–80 [177] | 150–300 [60] | 10–20 [60] | Up to 3500 [178] | 60–83 [83] | 0.2–0.6 [60] |
| NiMH | 145–152 [179,180] | 390–2000 [179,181] | <15 [182] | 40,000 [182] | 88–98 [179] | ~1 [183] |
| VRB | 10–30 [60] | 166 [184] | 5–20 [185] | ≤12,000 [60] | 75–85 [60,186] | Very low [61] |
| ZnBr | 30–80 [60,187] | 45–100 [187,188] | ~10 [175] | ≤2000 [60] | 66–80 [83] | Small [189] |
| PSB | 97–165 [190,191] | 77–83 [192,193] | 10–15 [60] | ≤2000 [194] | 93–95 [195] | ~Zero [185] |
| Different operating parameters of Fuel cells | ||||||
| SOFC | 800–1000 [196] | 200–1000 [197] | ~4.5 [198] | 50–1000 [199,200] | 35–45 [127] | |
| PEMFC | 500–1000 [201,202] | 90–1000 [203,204] | ~5 [205] | Up to 9000 [206] | 53–58 [127] | |
| DMFC | Up to 1500 [207] | ~1000 [208] | ~10 [209] | 300–10,000 [210,211] | ~40 [127] | ~Zero [212] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abbas, Q.; Mirzaeian, M.; Hunt, M.R.C.; Hall, P.; Raza, R. Current State and Future Prospects for Electrochemical Energy Storage and Conversion Systems. Energies 2020, 13, 5847. https://doi.org/10.3390/en13215847
Abbas Q, Mirzaeian M, Hunt MRC, Hall P, Raza R. Current State and Future Prospects for Electrochemical Energy Storage and Conversion Systems. Energies. 2020; 13(21):5847. https://doi.org/10.3390/en13215847
Chicago/Turabian StyleAbbas, Qaisar, Mojtaba Mirzaeian, Michael R.C. Hunt, Peter Hall, and Rizwan Raza. 2020. "Current State and Future Prospects for Electrochemical Energy Storage and Conversion Systems" Energies 13, no. 21: 5847. https://doi.org/10.3390/en13215847
APA StyleAbbas, Q., Mirzaeian, M., Hunt, M. R. C., Hall, P., & Raza, R. (2020). Current State and Future Prospects for Electrochemical Energy Storage and Conversion Systems. Energies, 13(21), 5847. https://doi.org/10.3390/en13215847








