Abstract
Various lithium recovery technologies have been developed as securing lithium resources has become increasingly important. Among these technologies, the electrochemical lithium recovery (ELR) system is a rapid and eco-friendly extraction method that has been studied recently. In this paper, an ELR system using a spinel-type LiMn2O4 (LMO) is briefly reviewed. As LMO electrodes have high Li+ selectivity and stability compared to other lithium battery cathodes, they have been widely used as positive electrodes for the ELR system. This paper summarizes the system proposal, LMO electrode modification, system analysis, and industrial applications. Perspectives of the ELR technology are presented considering the progress of the research.
1. Introduction
With the recent rapid growth of the electric vehicle industry, the lithium battery industry has grown at the same time [1]. However, there have been concerns about an undersupply of lithium as a resource, and a possible scenario for the shortage in the mid-2020s was reported [2,3]. Therefore, it has been argued that there is a need for a rapid extraction technology that can recover lithium from various sources [4]. The currently commercialized lithium recovery technology is lime soda evaporation, which is suitable for the mass production of lithium from brine sources. Unfortunately, it is a time-consuming process (12–18 months) with a low recovery rate and causes industrial pollution [5]. Thus, electrochemical lithium recovery (ELR) systems have been studied to solve these problems [6,7,8].
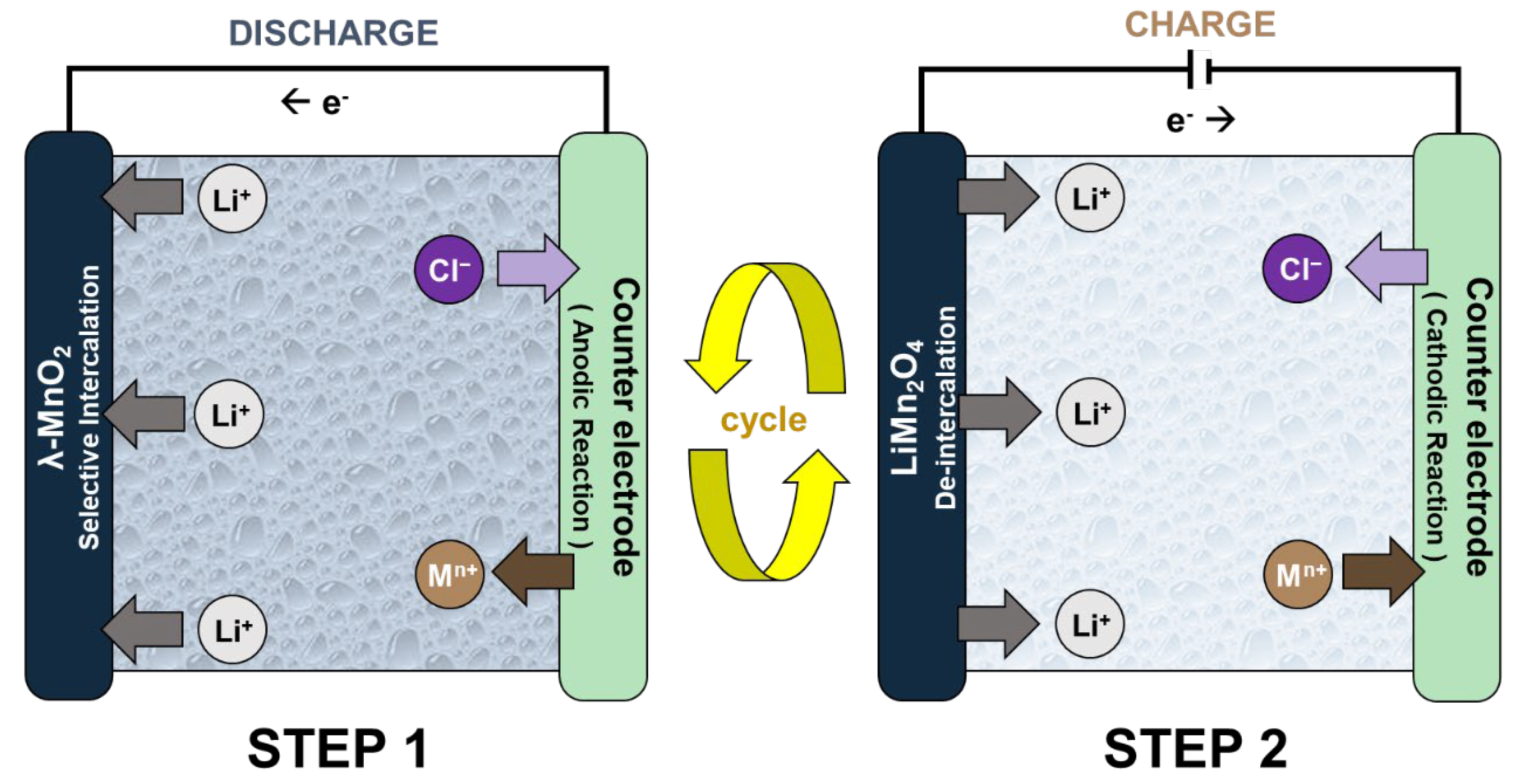
The ELR system is an electrochemical process that separates and concentrates lithium-ion (Li+) by using lithium selective electrodes [9]. Figure 1 shows the operational principle of the ELR system using the spinel LiMn2O4 electrode. Generally, the system has an electrochemistry cell structure that consists of the working electrode (lithium-ion battery cathode) and the counter electrode. The system operates by repeated conduction of the following two steps. In Step 1, the Li+ brine water source is inserted between the electrodes so that the system cell spontaneously discharges. This results in Li+ intercalation into the lithium-ion battery cathode and the oxidation reaction on the counter electrode. After Step 1, the exhausted brine source is replaced by the recovery solution. When the charging current is applied (Step 2), Li+ de-intercalates from the LiMn2O4 electrode as the reduction reaction occurs on the counter electrode. Li+ then diffuses into the recovery solution, which concentrates the product. These two steps are conducted repeatedly until a Li+ recovery solution with high concentration and purity is produced.
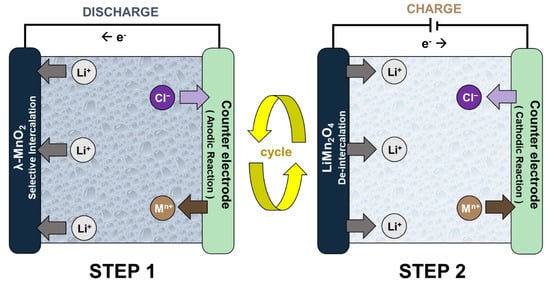
Figure 1.
The operational principle of the electrochemical lithium recovery (ELR) systems.
The representative cathode electrodes used for the ELR system are olivine LFP (LiFePO4) and spinel LMO (LiMn2O4 or λ-MnO2) [5,10]. Both have sufficient reactivity with Li+ and proper potential windows for lithium recovery. Yet compared to the LFP electrode, the LMO electrode shows much higher stability for repeated cycles with higher selectivity for Li+, especially in the high Mg2+ concentration solution [11]. Accordingly, the LMO based ELR system has been studied more intensively than LFP and the LMO is considered a suitable material.
LiMn2O4 has a spinel structure which is corresponding with the Fd3m space group [12]. Spinel structure consists of three-dimensional pores that cause the steric effect. These small pores, which transport only Li+, enable selective recovery of Li+ [5,13]. LiMn2O4 has tetrahedral 8a sites for the Li+, octahedral 16d sites for the Mn3+ and Mn4+, and face-centered cubic (FCC) 32e sites for the oxygen anions [14]. Intercalation phenomena in the ELR system can be described by the redox mechanism [15,16]. At the electrochemical reduction step, the valence of the manganese cations is changed from +4 to +3 and Li+ intercalates in the tetrahedral 8a sites. Contrary to this step, at the electrochemical oxidation step, the valence of the manganese cations is changed from +3 to +4 and Li+ de-intercalates [17].
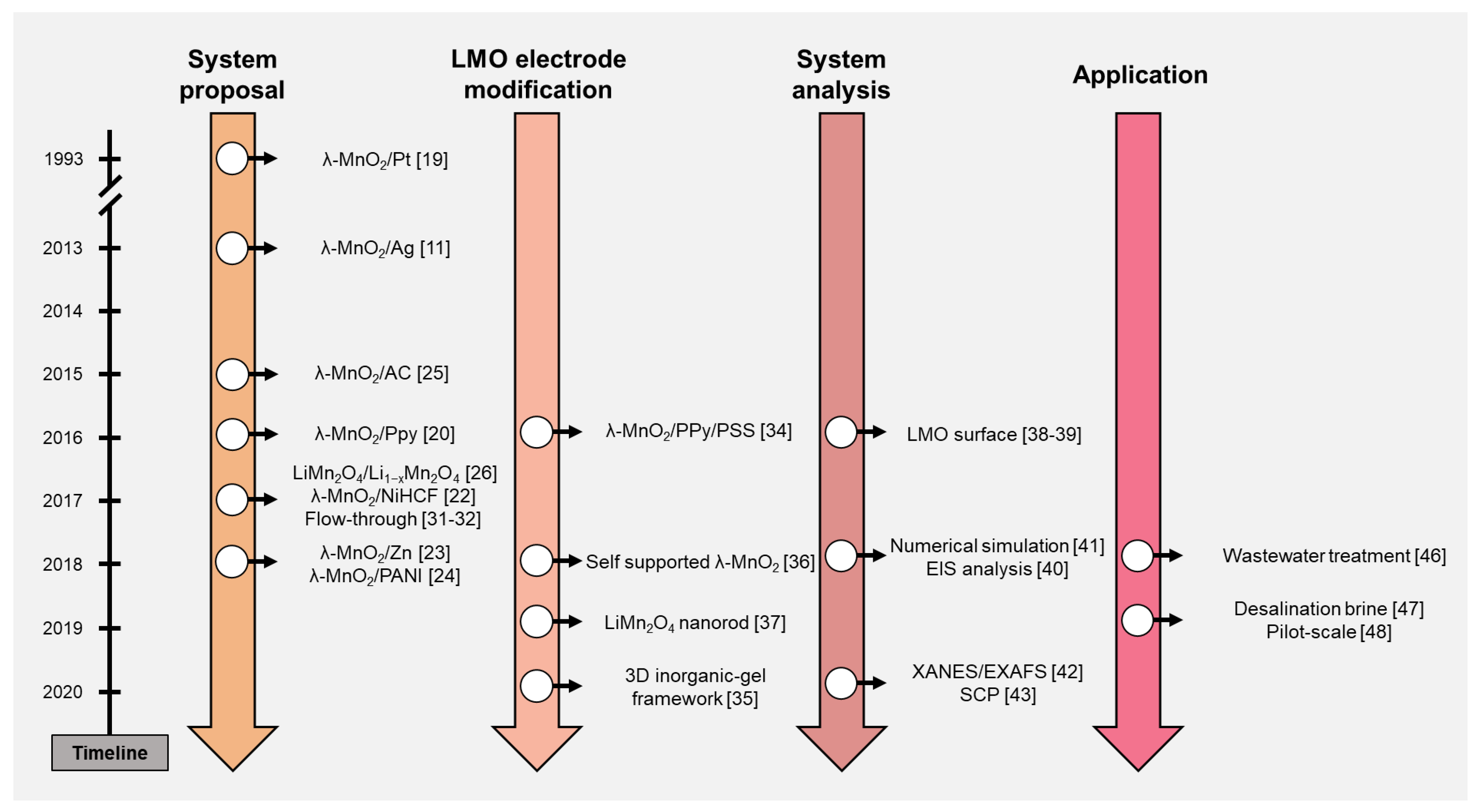
ELR systems using the LMO electrode are reviewed in this study. The timeline in Figure 2 briefly summarizes the contents of this paper. The ELR research using the LMO electrode can be classified and discussed by four categories: system proposal, LMO electrode modification, system analysis, and application. After the discussion of the literature, ELR system performance was organized and discussed further. Based on the trend and development process of the ELR system, we suggested the perspectives of this research.
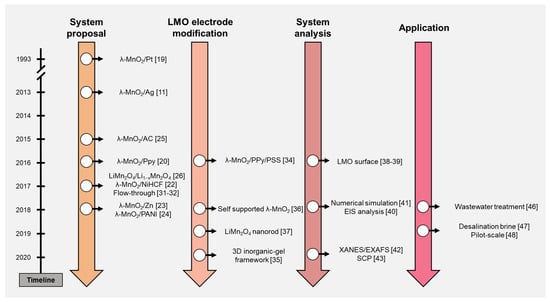
Figure 2.
Timeline of the electrochemical lithium recovery (ELR) system research.
2. System Proposal
2.1. The System with the Hydrolysis (λ-MnO2/Pt System)
The first electrochemical lithium recovery (ELR) system was the λ-MnO2/Pt system proposed by Kanoh et al. in 1993 [18,19], which is a three-electrode cell consisting of a λ-MnO2 electrode (working), Pt wire electrode (counter), and Calomel electrode (reference). Li+ was selectively recovered by the forward sweep method (1.0 to 0.2 V, scan rate: 0.1 mV s−1) of cyclic voltammetry. The lithium capturing reaction is as follows:
Kanoh et al. investigated the lithium recovery performance of the λ-MnO2/Pt system in aqueous solutions of various metal chlorides. From the results of these lithium recovery experiments, they found that the system showed remarkable Li+ selectivity when the electrolyte contained more than 10 mM of Li+ while insufficient performance was reported when the electrolyte contained less than 0.1 mM of Li+. Expectedly, this system showed low energy efficiency due to the hydrolysis occurring at the Pt electrode surface. They also reported that alkaline earth metal ions had stronger inhibitory effects on lithium recovery than alkaline metal ions due to the electrostatic interaction and complexation ability on the oxide surface. For a system application, lithium was recovered from the geothermal water and the uptake (similar to the capacity) was 11 mg g−1. The first ELR system was well-defined by their pioneering work. Nevertheless, this system was needed to be refined because of the pH variation and high energy consumption that occurred by the hydrolysis.
2.2. Asymmetric Battery System
For a high energy efficient and reversible process without the pH variation, Lee et al. reported the first battery-type ELR system consisting of a λ-MnO2 electrode (positive electrode) and silver electrode (Ag, negative electrode) [11]. The reversible lithium capturing/releasing reaction is as follows:
The system was operated under constant-current mode. By repeating the two steps (capturing and releasing), Li+ in the source solution was selectively transported to the recovery solution. In this study, an additional lithium recovery test was carried out from the simulated brine (similar to the Salar de Atacama) and the results showed that lithium was selectively recovered with efficient energy consumption (specific energy consumption: 1.0 Wh mol−1). The research of the battery-type ELR system using the LMO was completed based on this research. Certain follow-up studies have focused on developing alternatives to the silver electrode due to its high cost. Materials such as polypyrrole (PPy), nickel hexacyanoferrate (NiHCF), zinc, and polyaniline (PANI) were suggested as the alternatives. These materials showed enough electrical capacity, good reversibility, high stability, and high current efficiency to the targeted counter-reaction.
In the study conducted by Marchini et al., PPy, the reversible chloride capturing electrode, was applied to the ELR system [20]. The PPy electrode captured and released Cl− with high reversibility and stability in the natural brine. The reversible chloride capturing reaction is as follows:
The system operated under 1 V of cell voltage for 200 cycles, and Li+ was recovered from the natural brine solution (Salar de Olaroz) and the Faradaic efficiency was around 50%. The specific energy consumption was 5 (recovery solution: 25 mM LiCl) and 9.4 (recovery solution: brine) Wh mol−1. Although the Li+ concentration is considerably high (180 mM), the Faradaic efficiency is relatively low and the energy consumption is relatively high. This is because the LMO/PPy battery cell was operated under high current (~10 C). For a fair comparison with other ELR systems, this cell should be operated under a lower current (~1 C). They also compared the suitability of PPy, carbon felt, and Pt as the counter electrode. As the PPy electrode showed few variations in electric potential, ELR system with PPy counter electrode had the lowest energy consumption compared to other electrodes. They concluded that PPy is the most suitable alternative to the silver electrode.
An NiHCF is a Prussian blue analogue material that selectively intercalates K+ and Na+. The reversible K+ and Na+ capturing reaction is as follows:
As the affinity of the NiHCF electrode for Li+ is low, it can be used as the counter electrode of the ELR system. Trócoli et al. suggested the λ-MnO2/NiHCF ELR system [21,22]. Similar to the λ-MnO2/Ag system, the λ-MnO2/NiHCF system showed high selectivity towards Li+ (1.63, 57.6, and 1633 to K+, Na+, and Mg2+, respectively) and relatively low energy consumption (3.6 Wh mol−1). The recovery solution, however, produced by using this system contained a high concentration of K+ and Li+ purity is low (70 to 80%). An additional separation process could be required for highly pure lithium products.
Kim et al. proposed zinc as a counter electrode in the ELR system due to its low cost, high stability, and large capacity [23]. The ELR cell was divided equally into two compartments with an anion exchange membrane placed between the compartments. The λ-MnO2 electrode and zinc foil electrode were placed in each compartment and used as positive and negative electrodes, respectively. When lithium recovery occurs at the λ-MnO2 electrode, the zinc electrode was oxidized and converted into zinc ions, which attracted chloride ions of the λ-MnO2 compartment by the charge compensation. This reaction is the reversible reaction. Zinc, thus, can be used as a negative electrode for the ELR system. The overall reaction is as follows:
The specific energy consumption for the lithium recovery from simulated Atacama brine was 6.3 Wh mol−1 and the capacity retention was 73% after 100 cycles (1 mA cm−2). This study showed the possibility of using metal electrode usage in the ELR system.
Zhao et al. reported the LixMn2O4/PANI cell for lithium extraction from the simulated Taijinair lake brine [24]. The PANI electrode reversibly reacts with chloride ion on the principle of p-doping/de-doping. This reversible reaction is as follows:
The PANI is an affordable and eco-friendly polymer material. Thus, it is suitable as a silver electrode alternative. The LixMn2O4/PANI system shows high current efficiency (95%) with good cyclability (70.8% after 200 cycles). The energy consumption during the process was 3.95 Wh mol−1 on average. They insisted that this system showed relatively good performance comparing to other ELR systems using the silver alternative electrodes. This result should be further verified because the specific energy consumption considerably increased with the number of cycles (3.13 Wh mol−1 at the first cycle and 5.08 Wh mol−1 at the fourth cycle).
2.3. Hybrid Supercapacitor System
A hybrid supercapacitor system was also suggested by Kim et al. [25]. In this system, an activated carbon (AC) electrode is used instead of the silver electrode, because of the economic feasibility and system stability. A reversible counter reaction occurred when the AC electrode adsorbed and desorbed the dissolved ions like an electric double-layer capacitor. An anion exchange membrane was placed in front of the AC electrode to prevent the cation adsorption into the electrode. The flow-type reactor was designed to make it easier for the actual chemical processes. The capacity retention was 97% until the 50th cycle of the process with almost 100% coulombic efficiency. The cycling performance that represents the λ-MnO2/AC hybrid supercapacitor system had excellent long-term stability.
2.4. Symmetric Rocking Chair Battery-Liked System
A symmetric rocking chair battery-like system is a system using LiMn2O4 as a positive electrode and Li1−xMn2O4 as a negative electrode. Two LMO electrodes are capturing and releasing Li+ simultaneously in the continuous lithium extraction process. Zhao et al. firstly proposed a symmetric rocking chair ELR system using two LMO electrodes [26]. In this research, the effects of the potential and temperature on the performance factors (extraction capacity, separation coefficient, current efficiency, and specific energy consumption) were investigated and the optimized conditions (0.6 V, 25 °C) were presented according to comprehensive evaluations. Furthermore, the effect of coexisting ions on the recovery performance was evaluated (Mg2+ > Na+ > Ca2+ > K+), which was explained by the difference in ionic radii and ionic valence. The specific energy consumption of this system was relatively high (16–19 Wh mol−1) since this system operated under the constant-voltage mode [27]. In the follow-up studies, a rocking chair battery-like system using LiMn2O4 has been operated under the constant-current mode, and the specific energy consumption is much lower (under 3 Wh mol−1) [28,29,30].
2.5. Flow-Through Type Reactor
The Li+ concentration of the brine source is relatively lower than that of the battery electrolyte, therefore, the concentration polarization is influential to the recovery performance. A flow-through type reactor is one of the solutions for decreasing the negative effect of the concentration polarization (further discussed in Section 4.4). Palagonia et al. operated the flow-through type reactor and analyzed the concentration polarization [31]. The authors compared the lithium recovery performance (specific capacity and rate capability) with and without flow rate and proved that convection mass transport decreased the negative effect of the concentration polarization. When the brine from Salar de Atacama was used as a lithium source, the specific capacity with the flow was 5 times higher than without flow. In their follow-up parameter study, the effect of the current density and mass loading on the recovery performance was also examined [32] by using the optimized flow-through reactor and obtained the 100 mM LiCl recovery solution (purity: 94%) from the 1 mM LiCl + 100 mM NaCl source water. By evaluating the economic feasibility of the process with the pumping energy, it was calculated to be commercially valuable when the source water contained more than 1 mM of Li+. It is verified that the flow-through type reactor is an efficient recovery method even for the low Li+ concentration brine. Subsequent studies should focus on controlling the convection mass transport to further improve performance.
3. LMO Electrode Modification to Enhance the Li+ Recovery Performance
The most widely used methods for the LMO synthesis are the solid-phase combustion method and the hydrothermal method, which are simple and economical [5,33]. The pristine LMO fabricated by these methods has high selectivity to Li+, but its rate capability, long-term stability, and energy efficiency need to be improved. For this, strategies to refine the LMO electrodes have been proposed.
The first strategy is a polymer film coating to prevent capacity loss and improve capacity. Du et al. fabricated the λ-MnO2/PPy/PSS electrode [34]. The λ-MnO2 nanorod electrode was fabricated by hydrothermal synthesis, and polypyrrole (PPy)/polystyrene sulfonate (PSS) films were covered by the unipolar pulse electrodeposition (UPED). PSS doped PPy acted as a cation exchanger, allowing λ-MnO2/PPy/PSS electrodes to have higher Li+ selectivity than the pristine λ-MnO2 electrode. A high lithium recovery capacity of 35.2 mg g−1 was shown using this electrode, which is near the theoretical value (38.3 mg g−1). Assuming the Faradaic efficiency is 100%, the capacity of the LMO electrode is 135 mAh g−1, which is the highest value among the ELR studies, as shown in Table A1. The authors elucidate that the nanorod morphology provided the high surface area with the enhancement of the ionic conductivity of the electrode structure by a polymer film. Further verification, however, is needed because the operating voltage range is slightly out of the potential window where the redox reaction of LMO occurs [7]. Recently, LMO covered by a three-dimensional nanostructured hybrid inorganic-gel framework is developed as an electrode for the electrochemical lithium extraction [35]. PPy and Al2O3 are used as the framework to prevent capacity loss caused by the dissolution of manganese. The synthesis process is as follows. First, the LMO was fabricated by the solid combustion method. Then, Al2O3 was coated on the LMO particle by using the sol-gel method. Last, PPy was coated on the Al2O3/LMO by the oxidative polymerization and PPy/Al2O3/LMO was fabricated. The PPy/Al2O3/LMO electrode showed enhanced cyclability (91.66% after 30 cycles at the 1C rate) compared to the pristine LMO electrode (77.70% in the same condition). The lithium recovery performance of the continuous hybrid capacitor system using this electrode showed a high purity of the product (97.37%, highest value among the ELR studies) with efficient energy consumption (1.41 Wh mol−1).
Another strategy is to modify the physical properties of the LMO electrode. A self-supported λ-MnO2 film electrode was developed for the ELR system by Xu et al. [36]. MnO2 film on the platinum plate was fabricated by the cathodic deposition, which is followed by the aerobic oxidation and lithiation step (hydrothermal method). As this electrode was fabricated without any binder material, the electrode had high electrical conductivity and high ion transfer efficiency. The ELR system using this electrode showed higher rate capability (100 mAh g−1 at the current density of 50 mA g−1 in 30 mM Li+ aqueous solution) and better long-term cyclability (91% of the capacity remained after 100 cycles) than the λ-MnO2 powder electrode (80 mAh g−1 of capacity and 76% of the capacity remained at the same conditions). Xie et al. has reported a LiMn2O4 nanorod electrode applied to the ELR system. LiMn2O4 nanorod was fabricated by a continuous process of the hydrothermal method, grounding, and sintering [37]. The average diameter and length of the nanorod are 500 nm and 4–7 μm, respectively (the pristine LMO is micron-sized and spherical-like). Due to the large surface area of the nanosized electrode, the charge transport reaction at the LMO electrode accelerated and Li+ was selectively recovered in a highly energy-efficient way (1.76 Wh mol−1). The limited cycling performance of the electrode (37.4% of the capacity retention after 200 cycles) is required to be improved for the practical application.
4. System Analysis
Understanding the mechanism and behavior of the ELR system is important for further development. Some research utilizing electroanalysis, X-ray measurement, and numerical simulation have been reported. In recent years, selective concentration polarization was analyzed.
4.1. Electroanalysis
Marchini et al. have studied surface chemistry and ion exchange behavior of the LMO electrode through electroanalysis [38]. The cyclic voltammetry (CV) and galvanostatic intermittent titration technique (GITT) measurements were used to show how the Li+ was stably and reversibly intercalated/de-intercalated in the natural brine (Salar de Olaroz). The diffusivity of Li+ in the nanocrystalline structure was measured as 10−10 cm2 s−1 using the chronoamperometry method. The ratio of Mn/O at the surface and initial MnIV/MnIII of the crystal was measured as 1:2 and 1:1, respectively, by the photoelectron spectroscopy. The content of MnIII was depleted when the potential exceeded 1.1 V (vs. Ag/AgCl) and dominant when the potential was less than 0.4 V (vs. Ag/AgCl). The same research group investigated LMO surface composition and crystal structure affected by the electrode potential in the brine [39]. They found that the intercalation/de-intercalation reactions of the LMO consist of 2 steps involving two cubic phase transitions in an aqueous brine solution resembling the behavior in organic electrolytes. They found that the oxide surface of LMO adsorbed the sodium ion, but the intercalation of sodium ion into the crystal structure did not occur. They also analyzed the interface of LMO in the LiCl and natural brine solution by using electrochemical impedance spectroscopy (EIS) with a modified Randles equivalent electrical circuit model [40]. Unlike with the organic solvent, a solid-electrolyte interface (SEI) was not created on the LMO electrode in the aqueous brine solution, which results in a single semicircle in the Nyquist plot. It was also found that the Rct value was at the minimum for a specific state-of-charge value (0.25 and 0.75) when the intercalation site was half-filled with Li+ at each phase. The overall behavior of Li+ intercalation in aqueous brine solution was precisely analyzed by this research group. By using this result, the operation condition for the efficient lithium recovery can be provided with analytical evidence.
4.2. Numerical Simulation
Romero et al. numerically simulated the LMO/PPy and LMO/AC systems in the one-dimensional domain using COMSOL Multiphysics software [41]. This is the first study that analyzed the ELR system behavior by using computational modeling and simulation. The model was constructed based on the pseudo-2D model (P2D) widely used in lithium-ion battery research. The concentration profiles of Li+, Na+, K+, Mg2+, B2O72−, cell voltage, and electrolyte potential with the constant-current operation were calculated. Joo et al. demonstrated the flow-type λ-MnO2/LiMn2O4 ELR system through experimentation and simulation [28]. This is the first study to propose the symmetric rocking chair battery-liked ELR system as the flow-type reactor and conduct a two-dimensional simulation to investigate the hydrodynamic phenomena. The spatiotemporal concentration distribution of Li+ in the flow channel with the spacer mesh and energy consumption under various current condition were calculated. Additionally, Romero et al. tested and numerically simulated a 3D packed bed reactor consists of a porous petroleum coke electrode covered with LiMn2O4 [29]. They found that the degree of de-lithiation of the Li1−xMn2O4 electrode determines the amount of recovered Li+. The effect of flow rate on the electrolyte concentration distribution was also investigated and it was concluded that a specific value of flow rate is needed for the uniform concentration distribution in the flow channel. The authors proposed a direction of designing the scaled-up ELR system such as increasing the volume fraction of LMO and decreasing electrode particle size and contact resistance between electrode particles.
4.3. X-ray Measurement
Kim et al. analyzed the physicochemical behavior of the LMO electrode at various concentrations and current density conditions [42]. From the X-ray absorption near edge structure (XANES) and extended X-ray absorption fine structure (EXAFS) spectra, the structural transition of the LMO electrode after the recovery process was investigated. The λ-MnO2 electrode was found to have three chemical states: depletion, transition, and saturated regions. The authors presented that the depletion state is dominant when the concentration of Li+ is low or the current density is high. To reduce this negative effect, they suggested the ball-milling method for decreasing the particle size of LMO and improving the capacity of the system. This result justified why small-sized electrode particles are essential for the high-performance ELR system.
4.4. Selective Concentration Polarization
The negative effect of the concentration polarization on the system performance was analyzed by Guo et al. [43]. Contrary to the lithium-ion battery, the electrolyte of the ELR system contains a low concentration of Li+ and a high concentration of coexisting ions like Na+ and Mg2+ [44,45]. Therefore, the LMO electrode of the ELR system is vulnerable to the concentration polarization and exhibits low rate capability and high energy consumption. They named this unique phenomenon found only in the ELR system as selective concentration polarization (SCP), which is caused by the depletion of Li+ and accumulation of coexisting ion in the boundary layer. Mg2+ is an ion that strongly induces SCP more than other cations whose strength of SCP is followed by Na+, Ca2+, and K+, in that order. They also proved that stirring can reduce the negative effect of SCP by the experiment. This study identified the reason for the performance degradation in the ELR system, which is distinguished from the battery system. This suggests the development direction of a flow-type ELR system which has been studied recently.
5. Application
Lithium can be recovered from various sources, such as wastewater and desalination brine. For this, it is necessary to design the ELR systems considering the special characteristic of the source water.
5.1. Lithium Recovery with the Wastewater Treatment
Kim et al. suggested the λ-MnO2/BDD bi-functional system to recover Li+ from the wastewater discharged from the actual battery recycling plant [46]. The wastewater contained a relatively high concentration of Li+ (270 mM) but also contained lots of organic pollutants that should be treated. To decompose dissolved organics, a boron-doped diamond (BDD) electrode was adopted as an oxidant-generating electrode (counter). Using the system, a high purity recovery solution (98.6 mol% of Li+) was produced and the dissolved organic carbon (DOC) of the wastewater was reduced to 65%. This study is meaningful in that the range of electrolytes that can be applied to the ELR system has been increased through the development of a bi-functional cell system.
5.2. Lithium Recovery from the Desalination Concentrate
A λ-MnO2/Ag system that recovers lithium from the desalination brine was developed and evaluated by Kim et al. [47]. The desalination brine contained extremely low concentrations of Li+ (0.063 mM), which makes it impossible to manufacture the product with high Li+ concentration and purity through a one-stage ELR system. Accordingly, the research team invented a two consecutive recovery process that recovers Li+ once again from the Li+ recovered solution. The two consecutive recovery processes produced a recovery solution with high purity (up to 99.0%) and high Li+ concentration (190 mM). The authors compared the lithium recovery performance of the adsorption and ELR process and confirmed that the ELR system is much faster at capturing and releasing steps. As a follow-up study, Joo et al. designed and demonstrated the pilot-scale λ-MnO2/Ag system (as shown in Figure 3) to recover lithium from the actual desalination concentrate [48]. As the actual desalination concentrate retained only 0.035 mM of Li+, the two consecutive ELR process was designed to obtain the recovery product of high Li+ concentration (62 mM) and high purity (88 mol% of Li+). These studies have shown that the ELR system can be applied in actual practice even from a brine source at extremely low Li+ concentrations.

Figure 3.
A portion of the pilot-scale ELR system: primary recovery process using λ-MnO2/Ag electrode stack.
6. Summary of the System Performance and Perspectives
ELR systems using LMO electrodes have been intensively studied since 2013. The research trend in this field is gradually shifting from quantitative research to qualitative research. In other words, improving the performance of the ELR system is a major concern in the present and the future. In this section, the system performance of the literature is summarized (as shown in Appendix A Table A1) and the perspectives are presented.
6.1. System Proposal and Operation
In recent years, the number of studies on the flow-type ELR system is increasing. This trend is different from the past when the batch-type or semi batch-type systems were actively investigated. Compared with the batch-type reactor, the flow-type reactors have the advantages of reducing SCP and producing a uniform quality of Li+ solution. This feature is advantageous for applying the ELR system to practical usage. Future ELR system research is expected to be conducted for the optimization and design of the flow-type system.
To evaluate the newly developed system, it is necessary to compare it with the system that previously developed. However, it is not possible to directly compare the performance of each system because the current density and electrolyte conditions are different. These parameters have a great effect on performance. Thus, for a fair comparison, a definite standard for the solution composition and operation mode is required to evaluate the system objectively [49].
Most ELR systems were operated under the constant-current (CC) mode and showed energy consumption of less than 10 Wh mol−1 as shown in Table A1. This value is lower than the energy consumption of the ELR system operated under constant-voltage (CV) mode (over 10 Wh mol−1). CC mode consumes less energy than CV mode because of lower resistive dissipation in the operation [27]. However, CC mode consumes more time than CV mode. Considering the benefits and drawbacks of CC and CV mode, an appropriate combination of operating mode is a promising strategy for rapid and energy-efficient processes.
6.2. LMO Electrode Modification
As mentioned in previous sections, one of the main problems of the LMO is that only the surface of the LMO particle is utilized at the low Li+ concentration sources, so that the total capacity of the LMO is limited. To solve this problem, a small-sized LMO particle to increase the surface-to-volume ratio is required to be developed, but little research has been reported for the ELR system (only nanorod). Therefore, various types of nanosized LMOs need to be investigated to increase the capacity and rate capability of the LMO electrode used in the low Li+ concentration sources.
Another problem is that the capacity of the LMO decreases with the number of cycles due to the manganese dissolution. To solve this problem, it is necessary to develop the protection method for the electrode particle to prevent manganese dissolution. Only two coating methods (λ-MnO2/PPy/PSS and PPy/Al2O3/LMO) were reported in the previous literature, and these electrodes showed high capacity and cyclability during the process. Various types of coating methods should be examined to increase the stability of the LMO electrode during the long-term process.
6.3. System Analysis
The behavior of LMO electrodes used in the ELR system is different from the behavior of LMO electrodes used in the battery system. Most of the solutions used for ELR research were electrolytes containing equivalent moles of salt or simulated brine, which normally contains 30–300 mM of Li+. This value is considerably low compared to lithium-ion battery electrolyte (typically, 1 M) [50]. Meanwhile, the solutions used for ELR studies contain a lot of impurities such as Mg2+, Na+, and Ca2+. Recent studies have begun to analyze the behavior of the ELR system such as SCP, and more future research is required to fully understand the special phenomena that occur only in the ELR system, not lithium-ion battery systems.
7. Conclusions
In this study, ELR systems using the LMO electrode were reviewed. The λ-MnO2/Pt system, λ-MnO2/Ag battery system, λ-MnO2/AC hybrid supercapacitor, LiMn2O4/Li1−xMn2O4 rocking chair battery-liked, and the flow-type system were proposed as representative electrode systems. Activated carbon, polypyrrole, nickel hexacyanoferrate, zinc, and polyaniline were suggested as alternatives for the silver electrode. LMO modification research was conducted in addition to developing λ-MnO2/PPy/PSS, PPy/Al2O3/LMO, self-supported λ-MnO2, and LiMn2O4 nanorod electrodes. To understand the behavior of the LMO, the surface and crystal of the LMO were analyzed by electrochemical and X-ray measurements. Interest in the hydrodynamics of the ELR system has grown recently, and the computational simulation and the concept of the concentration polarization have been used to analyze the system with the convection mass transport. For a practical demonstration of the process, ELR systems were applied to the wastewater treatment of the battery recycling plant and desalination plant. Due to the high Li+ selectivity and stability of the LMO electrode, the ELR system can be applied to various lithium resources with a high concentration of impurities such as salt brine, wastewater, geothermal water, desalination brine, and even seawater. ELR technology will be a promising extraction method in the green energy society of the future.
Author Contributions
Conceptualization, J.Y., and J.L.; methodology, J.J.; formal analysis, J.J.; investigation, J.J.; resources, J.J.; writing—original draft preparation, J.J.; writing—review and editing, J.L. and J.Y.; visualization, J.J.; supervision, J.L.; project administration, J.L.; funding acquisition, J.L. All authors have read and agreed to the published version of the manuscript
Funding
This work was supported by the Hongik University new faculty research support fund.
Conflicts of Interest
The authors declare no conflict of interest.
Appendix A

Table A1.
Performance of the ELR system using LiMn2O4 (LMO) electrode.
Table A1.
Performance of the ELR system using LiMn2O4 (LMO) electrode.
| LMO | Mass Loading (mg cm−2) | Electrode Dimension (cm × cm or cm2) | Counter | Solution Composition | Operation Mode | Capacity (mAh gLMO−1) | Energy Consumption (Wh molLi−1) | Ref. |
|---|---|---|---|---|---|---|---|---|
| λ-MnO2 | - | 1.0 × 1.0 | Pt | Geothermal (Li+: 0.74 mM) | Forward sweep | 85 | - | [19] |
| λ-MnO2 | - | 3.0 × 3.0 | Ag | Simulated brine: Salar de Atacama (Li+: 210 mM) | CC mode (0.5 mA cm−2) | - | 1.0 | [11] |
| λ-MnO2 | - | - | AC | 30 mM LiCl, NaCl, KCl, MgCl2, and CaCl2 | CC mode (0.5 mA cm−2) | - | 4.2 | [25] |
| λ-MnO2 | 0.52 |
Plane: 3.0 × 2.0 Total: 56.0 cm2 | PPy | Brine (Salar de Olaroz) (Li+: 180 mM) | CC mode (2.5 mA cm−2) | - | 9.4 | [20] |
| λ-MnO2 /PPy/PSS | - |
5 × 5 (porous) | Pt | 30 mM LiCl and NaCl | CV mode | 135 | - | [34] |
| LMO | - | 2 cm2 | NiHCF | Simulated brine: Salar de Atacama (Li+: 42 mM) | CC mode (1 C) | 100 | 3.6 | [22] |
| LiMn2O4 | - | 3.5 × 3.0 | Li1−xMn2O4 | Simulated brine (Li+: 21 mM) and concentrated seawater (Li+: 32 mM) | CV mode (0.6 V) | 77–97 | 17.89–18.67 | [26] |
| λ-MnO2 | 28.6 | 8.75 cm2 | BDD | Wastewater: battery recycling plant (Li+: 276.5 mM) | CC mode (2.9 mA cm−2) | 88 | 8.71 | [46] |
| LiMn2O4 | - | 2.5 × 4.0 | Zn | Simulated brine: Salar de Atacama (Li+: 210 mM) | CC mode (0.5 mA cm−2) | - | 6.3 | [23] |
| Self-supported λ-MnO2 | - | 3.0 × 3.0 | Ag | 30 mM LiCl, NaCl, KCl, MgCl2, and CaCl2 | CC mode (50 mA g−1) | 100 | 4.14 | [36] |
| LiMn2O4 | 25–35 | 2.0 × 2.0 | PANI | Simulated brine: Taijinair (Li+: 64 mM) | CC mode (0.5 mA cm−2) | ~90 | 3.95 | [24] |
| LiMn2O4 nanorod | 5 | - | NiHCF | Simulated brine: Taijinair (Li+: 31.7 mM) | CC mode (1 C) | 105 | 1.76 | [37] |
| λ-MnO2 | - | Diameter: 5 cm | Ag | Desalination brine (Li+: 0.063 mM) | CC mode (0.01 mA cm−2) | - | 17.2 (1st step) | [47] |
| λ-MnO2 | 5 | 2.0 × 2.0 | LiMn2O4 | 30 mM LiCl | CC mode (0.0625 mA cm−2) | 128 | 0.56 | [28] |
| PPy/Al2O3/LMO | - | 1 cm2 | AC | Simulated diluted brine (Li+: 23.48 mM) | CC mode (0.75 mA) | ~50 | 1.41 | [35] |
| Li1−xMn2O4 | 0.76 | 15.5 × 4.5 (packed bed) | LiMn2O4 | Simulated brine: Hombre Muerto (Li+: 190 mM) | CC mode (1.43 mA cm−2) | ~100 | 2.76 | [29] |
References
- Martin, G.; Rentsch, L.; Höck, M.; Bertau, M. Lithium market research—Global supply, future demand and price development. Energy Storage Mater. 2017, 6, 171–179. [Google Scholar] [CrossRef]
- Olivetti, E.A.; Ceder, G.; Gaustad, G.G.; Fu, X. Lithium-Ion Battery Supply Chain Considerations: Analysis of Potential Bottlenecks in Critical Metals. Joule 2017, 1, 229–243. [Google Scholar] [CrossRef]
- Wanger, T.C. The Lithium future-resources, recycling, and the environment. Conserv. Lett. 2011, 4, 202–206. [Google Scholar] [CrossRef]
- Calvo, E.J. Electrochemical methods for sustainable recovery of lithium from natural brines and battery recycling. Curr. Opin. Electrochem. 2019, 15, 102–108. [Google Scholar] [CrossRef]
- Xu, X.; Chen, Y.; Wan, P.; Gasem, K.; Wang, K.; He, T.; Adidharma, H.; Fan, M. Extraction of lithium with functionalized lithium ion-sieves. Prog. Mater. Sci. 2016, 84, 276–313. [Google Scholar] [CrossRef]
- Yoon, H.; Lee, J.; Kim, S.; Yoon, J. Review of concepts and applications of electrochemical ion separation (EIONS) process. Sep. Purif. Technol. 2019, 215, 190–207. [Google Scholar] [CrossRef]
- Battistel, A.; Palagonia, M.S.; Brogioli, D.; La Mantia, F.; Trócoli, R. Electrochemical Methods for Lithium Recovery: A Comprehensive and Critical Review. Adv. Mater. 2020, 32. [Google Scholar] [CrossRef] [PubMed]
- Srimuk, P.; Su, X.; Yoon, J.; Aurbach, D.; Presser, V. Charge-transfer materials for electrochemical water desalination, ion separation and the recovery of elements. Nat. Rev. Mater. 2020, 5, 517–538. [Google Scholar] [CrossRef]
- Pasta, M.; Battistel, A.; La Mantia, F. Batteries for lithium recovery from brines. Energy Environ. Sci. 2012, 5, 9487. [Google Scholar] [CrossRef]
- Xu, P.; Hong, J.; Qian, X.; Xu, Z.; Xia, H.; Tao, X.; Xu, Z.; Ni, Q.Q. Materials for lithium recovery from salt lake brine. J. Mater. Sci. 2020. [Google Scholar] [CrossRef]
- Lee, J.; Yu, S.-H.; Kim, C.; Sung, Y.-E.; Yoon, J. Highly selective lithium recovery from brine using a λ-MnO2–Ag battery. Phys. Chem. Chem. Phys. 2013, 15, 7690. [Google Scholar] [CrossRef] [PubMed]
- Ooi, K.; Miyai, Y.; Katoh, S.; Maeda, H.; Abe, M. Topotactic Li+ Insertion to λ-MnO2 in the Aqueous Phase. Langmuir 1989, 5, 150–157. [Google Scholar] [CrossRef]
- Ooi, K.; Miyai, Y.; Katoh, S.; Maeda, H.; Abe, M. Lithium-ion Insertion/Extraction Reaction with λ-MnO2 in the Aqueous Phase. Chem. Lett. 1988, 17, 989–992. [Google Scholar] [CrossRef]
- Greedan, J.E.; Raju, N.P.; Wills, A.S.; Morin, C.; Shaw, S.M.; Reimers, J.N. Structure and Magnetism in λ-MnO2. Geometric Frustration in a Defect Spinel. Chem. Mater. 1998, 10, 3058–3067. [Google Scholar] [CrossRef]
- Ooi, K.; Miyai, Y.; Sakakihara, J. Mechanism of Li Insertion in Spinel-Type Manganese Oxide. Redox and Ion-Exchange Reactions. Langmuir 1991, 7, 1167–1171. [Google Scholar] [CrossRef]
- Feng, Q.; Miyai, Y.; Kanoh, H.; Ooi, K. Li+ Extraction/Insertion with Spinel-Type Lithium Manganese Oxides. Characterization of Redox-Type and Ion-Exchange-Type Sites. Langmuir 1992, 8, 1861–1867. [Google Scholar] [CrossRef]
- Liu, W.; Kowal, K.; Farrington, G.C. Mechanism of the Electrochemical Insertion of Lithium into LiMn2O4 Spinels. J. Electrochem. Soc. 1998, 145, 459–465. [Google Scholar] [CrossRef]
- Kanoh, H.; Ooi, K.; Miyai, Y.; Katoh, S. Selective Electroinsertion of Lithium Ions into a Pt/λ-MnO2 Electrode in the Aqueous Phase. Langmuir 1991, 7, 1841–1842. [Google Scholar] [CrossRef]
- Kanoh, H.; Ooi, K.; Miyai, Y.; Katoh, S. Electrochemical recovery of lithium ions in the aqueous phase. Sep. Sci. Technol. 1993, 28, 643–651. [Google Scholar] [CrossRef]
- Missoni, L.L.; Marchini, F.; del Pozo, M.; Calvo, E.J. A LiMn2O4 -Polypyrrole System for the Extraction of LiCl from Natural Brine. J. Electrochem. Soc. 2016, 163, A1898–A1902. [Google Scholar] [CrossRef]
- Trócoli, R.; Battistel, A.; La Mantia, F. Nickel Hexacyanoferrate as Suitable Alternative to Ag for Electrochemical Lithium Recovery. ChemSusChem 2015, 8, 2514–2519. [Google Scholar] [CrossRef] [PubMed]
- Trócoli, R.; Erinmwingbovo, C.; La Mantia, F. Optimized Lithium Recovery from Brines by using an Electrochemical Ion-Pumping Process Based on λ-MnO2 and Nickel Hexacyanoferrate. ChemElectroChem 2017, 4, 143–149. [Google Scholar] [CrossRef]
- Kim, S.; Lee, J.; Kim, S.; Kim, S.; Yoon, J. Electrochemical Lithium Recovery with a LiMn2O4–Zinc Battery System using Zinc as a Negative Electrode. Energy Technol. 2018, 6, 340–344. [Google Scholar] [CrossRef]
- Zhao, A.; Liu, J.; Ai, X.; Yang, H.; Cao, Y. Highly Selective and Pollution-Free Electrochemical Extraction of Lithium by a Polyaniline/LixMn2O4 Cell. ChemSusChem 2019, 12, 1361–1367. [Google Scholar] [CrossRef]
- Kim, S.; Lee, J.; Kang, J.S.; Jo, K.; Kim, S.; Sung, Y.E.; Yoon, J. Lithium recovery from brine using a λ-MnO2/activated carbon hybrid supercapacitor system. Chemosphere 2015, 125, 50–56. [Google Scholar] [CrossRef]
- Zhao, M.Y.; Ji, Z.Y.; Zhang, Y.G.; Guo, Z.Y.; Zhao, Y.Y.; Liu, J.; Yuan, J.S. Study on lithium extraction from brines based on LiMn2O4/Li1−xMn2O4 by electrochemical method. Electrochim. Acta 2017, 252, 350–361. [Google Scholar] [CrossRef]
- Qu, Y.; Campbell, P.G.; Gu, L.; Knipe, J.M.; Dzenitis, E.; Santiago, J.G.; Stadermann, M. Energy consumption analysis of constant voltage and constant current operations in capacitive deionization. Desalination 2016, 400, 18–24. [Google Scholar] [CrossRef]
- Joo, H.; Jung, S.Y.; Kim, S.; Ahn, K.H.; Ryoo, W.S.; Yoon, J. Application of a Flow-Type Electrochemical Lithium Recovery System with λ-MnO2/LiMn2O4: Experiment and Simulation. ACS Sustain. Chem. Eng. 2020. [Google Scholar] [CrossRef]
- Romero, V.C.E.; Putrino, D.S.; Tagliazucchi, M.; Flexer, V.; Calvo, E.J. Electrochemical Flow Reactor for Selective Extraction of Lithium Chloride from Natural Brines. J. Electrochem. Soc. 2020, 167, 120522. [Google Scholar] [CrossRef]
- Marchini, F.; Williams, F.J.; Calvo, E.J. Sustainable Selective Extraction of Lithium Chloride from Natural Brine Using a Li1-xMn2O4 Ion Pump. J. Electrochem. Soc. 2018, 165, A3292–A3298. [Google Scholar] [CrossRef]
- Palagonia, M.S.; Brogioli, D.; Mantia, F.L. Influence of Hydrodynamics on the Lithium Recovery Efficiency in an Electrochemical Ion Pumping Separation Process. J. Electrochem. Soc. 2017, 164, E586–E595. [Google Scholar] [CrossRef]
- Palagonia, M.S.; Brogioli, D.; La Mantia, F. Effect of Current Density and Mass Loading on the Performance of a Flow-Through Electrodes Cell for Lithium Recovery. J. Electrochem. Soc. 2019, 166, E286–E292. [Google Scholar] [CrossRef]
- Xiao, J.L.; Sun, S.Y.; Wang, J.; Li, P.; Yu, J.G. Synthesis and adsorption properties of Li1.6Mn1.6O4 spinel. Ind. Eng. Chem. Res. 2013, 52, 11967–11973. [Google Scholar] [CrossRef]
- Du, X.; Guan, G.; Li, X.; Jagadale, A.D.; Ma, X.; Wang, Z.; Hao, X.; Abudula, A. A novel electroactive λ-MnO2/PPy/PSS core-shell nanorod coated electrode for selective recovery of lithium ions at low concentration. J. Mater. Chem. A 2016, 4, 13989–13996. [Google Scholar] [CrossRef]
- Zhao, X.; Jiao, Y.; Xue, P.; Feng, M.; Wang, Y.; Sha, Z. Efficient Lithium Extraction from Brine Using a Three-Dimensional Nanostructured Hybrid Inorganic-Gel Framework Electrode. ACS Sustain. Chem. Eng. 2020, 8, 4827–4837. [Google Scholar] [CrossRef]
- Xu, X.; Zhou, Y.; Feng, Z.; Kahn, N.U.; Haq Khan, Z.U.; Tang, Y.; Sun, Y.; Wan, P.; Chen, Y.; Fan, M. A Self-Supported λ-MnO2 Film Electrode used for Electrochemical Lithium Recovery from Brines. Chempluschem 2018, 83, 521–528. [Google Scholar] [CrossRef]
- Xie, N.; Li, Y.; Lu, Y.; Gong, J.; Hu, X. Electrochemically Controlled Reversible Lithium Capture and Release Enabled by LiMn2O4 Nanorods. ChemElectroChem 2019, 1–8. [Google Scholar] [CrossRef]
- Marchini, F.; Rubi, D.; Del Pozo, M.; Williams, F.J.; Calvo, E.J. Surface Chemistry and Lithium-Ion Exchange in LiMn2O4for the Electrochemical Selective Extraction of LiCl from Natural Salt Lake Brines. J. Phys. Chem. C 2016, 120, 15875–15883. [Google Scholar] [CrossRef]
- Marchini, F.; Calvo, E.J.; Williams, F.J. Effect of the electrode potential on the surface composition and crystal structure of LiMn2O4 in aqueous solutions. Electrochim. Acta 2018, 269, 706–713. [Google Scholar] [CrossRef]
- Marchini, F.; Williams, F.J.; Calvo, E.J. Electrochemical impedance spectroscopy study of the LixMn2O4 interface with natural brine. J. Electroanal. Chem. 2018, 819, 428–434. [Google Scholar] [CrossRef]
- Romero, V.C.E.; Tagliazucchi, M.; Flexer, V.; Calvo, E.J. Sustainable Electrochemical Extraction of Lithium from Natural Brine for Renewable Energy Storage. J. Electrochem. Soc. 2018, 165, A2294–A2302. [Google Scholar] [CrossRef]
- Kim, S.; Kang, J.S.; Joo, H.; Sung, Y.E.; Yoon, J. Understanding the Behaviors of λ-MnO2 in Electrochemical Lithium Recovery: Key Limiting Factors and a Route to the Enhanced Performance. Environ. Sci. Technol. 2020, 54, 9044–9051. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.-Y.; Ji, Z.-Y.; Chen, H.-Y.; Liu, J.; Zhao, Y.-Y.; Li, F.; Yuan, J.-S. Effect of Impurity Ions in the Electrosorption Lithium Extraction Process: Generation and Restriction of “Selective Concentration Polarization”. ACS Sustain. Chem. Eng. 2020, 4. [Google Scholar] [CrossRef]
- Zhao, Z.; Si, X.; Liu, X.; He, L.; Liang, X. Li extraction from high Mg/Li ratio brine with LiFePO4/FePO4 as electrode materials. Hydrometallurgy 2013, 133, 75–83. [Google Scholar] [CrossRef]
- Pramanik, B.K.; Asif, M.B.; Roychand, R.; Shu, L.; Jegatheesan, V.; Bhuiyan, M.; Hai, F.I. Lithium recovery from salt-lake brine: Impact of competing cations, pretreatment and preconcentration. Chemosphere 2020, 260, 127623. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Kim, J.; Kim, S.; Lee, J.; Yoon, J. Electrochemical lithium recovery and organic pollutant removal from industrial wastewater of a battery recycling plant. Environ. Sci. Water Res. Technol. 2018, 4, 175–182. [Google Scholar] [CrossRef]
- Kim, S.; Joo, H.; Moon, T.; Kim, S.; Yoon, J. Environmental Science Processes & Impacts Rapid and selective lithium recovery from desalination brine using an electrochemical. Environ. Sci. Process. Impacts 2019, 667–676. [Google Scholar] [CrossRef]
- Joo, H.; Kim, S.; Kim, S.; Choi, M.; Kim, S.-H.; Yoon, J. Pilot-scale demonstration of an electrochemical system for lithium recovery from the desalination concentrate. Environ. Sci. Water Res. Technol. 2019. [Google Scholar] [CrossRef]
- Hawks, S.A.; Ramachandran, A.; Porada, S.; Campbell, P.G.; Suss, M.E.; Biesheuvel, P.M.; Santiago, J.G.; Stadermann, M. Performance metrics for the objective assessment of capacitive deionization systems. Water Res. 2019, 152, 126–137. [Google Scholar] [CrossRef]
- Ravikumar, B.; Mynam, M.; Rai, B. Effect of Salt Concentration on Properties of Lithium Ion Battery Electrolytes: A Molecular Dynamics Study. J. Phys. Chem. C 2018, 122, 8173–8181. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).