Molecular Markers in Melanoma Progression: A Study on the Expression of miRNA Gene Subtypes in Tumoral vs. Benign Nevi
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Sample Collection
2.3. miRNA Purification and Analysis
2.4. Data Analysis
2.5. Statistical Analysis
3. Results
4. Discussion
4.1. Literature Findings
4.2. Study Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Weiss, S.A.; Wolchok, J.D.; Sznol, M. Immunotherapy of Melanoma: Facts and Hopes. Clin. Cancer Res. 2019, 25, 5191–5201. [Google Scholar] [CrossRef] [PubMed]
- Kirkwood, J.M.; Ibrahim, J.G.; Sosman, J.A.; Sondak, V.K.; Agarwala, S.S.; Ernstoff, M.S.; Rao, U. High-dose interferon alfa-2b significantly prolongs relapse-free and overall survival compared with the GM2-KLH/QS-21 vaccine in patients with resected stage IIB-III melanoma: Results of intergroup trial E1694/S9512/C509801. J. Clin. Oncol. 2001, 19, 2370–2380. [Google Scholar] [CrossRef] [PubMed]
- Kirkwood, J.M.; Strawderman, M.H.; Ernstoff, M.S.; Smith, T.J.; Borden, E.C.; Blum, R.H. Interferon alfa-2b adjuvant therapy of high-risk resected cutaneous melanoma: The Eastern Cooperative Oncology Group Trial EST 1684. J. Clin. Oncol. 1996, 14, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Coit, D.G.; Andtbacka, R.; Bichakjian, C.K.; Dilawari, R.A.; Dimaio, D.; Guild, V.; Halpern, A.C.; Hodi, F.S.; Kashani-Sabet, M.; Lange, J.R.; et al. Melanoma. J. Natl. Compr. Cancer Netw. 2009, 7, 250–275. [Google Scholar] [CrossRef] [PubMed]
- Hodi, F.S.; O’Day, S.J.; McDermott, D.F.; Weber, R.W.; Sosman, J.A.; Haanen, J.B.; Gonzalez, R.; Robert, C.; Schadendorf, D.; Hassel, J.C.; et al. Improved survival with ipilimumab in patients with metastatic melanoma. N. Engl. J. Med. 2010, 363, 711–723. [Google Scholar] [CrossRef] [PubMed]
- Mansh, M. Ipilimumab and cancer immunotherapy: A new hope for advanced stage melanoma. Yale J. Biol. Med. 2011, 84, 381–389. [Google Scholar] [PubMed]
- Keung, E.Z.; Gershenwald, J.E. The eighth edition American Joint Committee on Cancer (AJCC) melanoma staging system: Implications for melanoma treatment and care. Expert Rev. Anticancer Ther. 2018, 18, 775–784. [Google Scholar] [CrossRef] [PubMed]
- Shain, A.H.; Bastian, B.C. From melanocytes to melanomas. Nat. Rev. Cancer 2016, 16, 345–358. [Google Scholar] [CrossRef] [PubMed]
- Rebecca, V.W.; Somasundaram, R.; Herlyn, M. Pre-clinical modeling of cutaneous melanoma. Nat. Commun. 2020, 11, 2858. [Google Scholar] [CrossRef]
- Dimitriou, F.; Krattinger, R.; Ramelyte, E.; Barysch, M.J.; Micaletto, S.; Dummer, R.; Goldinger, S.M. The World of Melanoma: Epidemiologic, Genetic, and Anatomic Differences of Melanoma across the Globe. Curr. Oncol. Rep. 2018, 20, 87. [Google Scholar] [CrossRef]
- Pitt, J.M.; Vétizou, M.; Daillère, R.; Roberti, M.P.; Yamazaki, T.; Routy, B.; Lepage, P.; Boneca, I.G.; Chamaillard, M.; Kroemer, G.; et al. Resistance Mechanisms to Immune-Checkpoint Blockade in Cancer: Tumor-Intrinsic and—Extrinsic Factors. Immunity 2016, 44, 1255–1269. [Google Scholar] [CrossRef]
- Akbani, R.; Akdemir, K.C.; Aksoy, B.A.; Albert, M.; Ally, A.; Amin, S.B.; Arachchi, H.; Arora, A.; Auman, J.T.; Ayala, B.; et al. Genomic Classification of Cutaneous Melanoma. Cell 2015, 161, 1681–1696. [Google Scholar] [CrossRef] [PubMed]
- Lauss, M.; Nsengimana, J.; Staaf, J.; Newton-Bishop, J.; Jönsson, G. Consensus of Melanoma Gene Expression Subtypes Converges on Biological Entities. J. Investig. Dermatol. 2016, 136, 2502–2505. [Google Scholar] [CrossRef] [PubMed]
- Bhalla, S.; Kaur, H.; Dhall, A.; Raghava, G.P.S. Prediction and Analysis of Skin Cancer Progression using Genomics Profiles of Patients. Sci. Rep. 2019, 9, 15790. [Google Scholar] [CrossRef] [PubMed]
- Marie, K.L.; Sassano, A.; Yang, H.H.; Michalowski, A.M.; Michael, H.T.; Guo, T.; Tsai, Y.C.; Weissman, A.M.; Lee, M.P.; Jenkins, L.M.; et al. Melanoblast transcriptome analysis reveals pathways promoting melanoma metastasis. Nat. Commun. 2020, 11, 333. [Google Scholar] [CrossRef] [PubMed]
- Varrone, F.; Caputo, E. The miRNAs Role in Melanoma and in Its Resistance to Therapy. Int. J. Mol. Sci. 2020, 21, 878. [Google Scholar] [CrossRef]
- Denning, M.F. Specifying protein kinase C functions in melanoma. Pigment Cell Melanoma Res. 2012, 25, 466–476. [Google Scholar] [CrossRef] [PubMed]
- Ghafouri-Fard, S.; Gholipour, M.; Taheri, M. MicroRNA Signature in Melanoma: Biomarkers and Therapeutic Targets. Front. Oncol. 2021, 11, 608987. [Google Scholar] [CrossRef]
- Gencia, I.; Baderca, F.; Avram, S.; Gogulescu, A.; Marcu, A.; Seclaman, E.; Marian, C.; Solovan, C. A preliminary study of microRNA expression in different types of primary melanoma. Bosn. J. Basic Med. Sci. 2020, 20, 197–208. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chan, E.; Patel, R.; Nallur, S.; Ratner, E.; Bacchiocchi, A.; Hoyt, K.; Szpakowski, S.; Godshalk, S.; Ariyan, S.; Sznol, M.; et al. MicroRNA signatures differentiate melanoma subtypes. Cell Cycle 2011, 10, 1845–1852. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Rother, J.; Jones, D. Molecular markers of tumor progression in melanoma. Curr. Genom. 2009, 10, 231–239. [Google Scholar] [CrossRef] [PubMed] [PubMed Central][Green Version]
- Deacon, D.C.; Smith, E.A.; Judson-Torres, R.L. Molecular Biomarkers for Melanoma Screening, Diagnosis and Prognosis: Current State and Future Prospects. Front. Med. 2021, 8, 642380. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Timis, T.; Bergthorsson, J.T.; Greiff, V.; Cenariu, M.; Cenariu, D. Pathology and Molecular Biology of Melanoma. Curr. Issues Mol. Biol. 2023, 45, 352. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Neagu, M.; Constantin, C.; Cretoiu, S.M.; Zurac, S. miRNAs in the Diagnosis and Prognosis of Skin Cancer. Front. Cell Dev. Biol. 2020, 8, 71. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Van Laar, R.; Lincoln, M.; Van Laar, B. Development and validation of a plasma-based melanoma biomarker suitable for clinical use. Br. J. Cancer 2018, 118, 857–866. [Google Scholar] [CrossRef] [PubMed]
- Fawzy, M.S.; Ibrahiem, A.T.; Bayomy, N.A.; Makhdoom, A.K.; Alanazi, K.S.; Alanazi, A.M.; Mukhlef, A.M.; Toraih, E.A. MicroRNA-155 and Disease-Related Immunohistochemical Parameters in Cutaneous Melanoma. Diagnostics 2023, 13, 1205. [Google Scholar] [CrossRef] [PubMed]
- Reuland, S.N.; Smith, S.M.; Bemis, L.T.; Goldstein, N.B.; Almeida, A.R.; Partyka, K.A.; Marquez, V.E.; Zhang, Q.; Norris, D.A.; Shellman, Y.G. MicroRNA-26a is strongly downregulated in melanoma and induces cell death through repression of silencer of death domains (SODD). J. Investig Dermatol. 2013, 133, 1286–1293. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Giles, K.M.; Brown, R.A.; Ganda, C.; Podgorny, M.J.; Candy, P.A.; Wintle, L.C.; Richardson, K.L.; Kalinowski, F.C.; Stuart, L.M.; Epis, M.R.; et al. microRNA-7-5p inhibits melanoma cell proliferation and metastasis by suppressing RelA/NF-κB. Oncotarget 2016, 7, 31663–31680. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Jenike, A.E.; Halushka, M.K. miR-21: A non-specific biomarker of all maladies. Biomark. Res. 2021, 9, 18. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Eom, K.; Kim, J.; Bang, H.; Wang, H.-Y.; Ahn, S.; Kim, G.; Jang, H.; Kim, S.; Lee, D.; et al. MiR-9, miR-21, and miR-155 as potential biomarkers for HPV positive and negative cervical cancer. BMC Cancer 2017, 17, 658. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Wang, J.; Zhang, H.; Fu, B.; Zhang, Y.; Jia, Q.; Wang, Y. Diagnostic value of circulating miR-155 for breast cancer: A meta-analysis. Front. Oncol. 2024, 14, 1374674. [Google Scholar] [CrossRef] [PubMed]
- De Palma, F.D.E.; Raia, V.; Kroemer, G.; Maiuri, M.C. The Multifaceted Roles of MicroRNAs in Cystic Fibrosis. Diagnostics 2020, 10, 1102. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ciuca, I.M.; Pop, L.L.; Rogobete, A.F.; Onet, D.I.; Guta-Almajan, B.; Popa, Z.; Horhat, F.G. Genetic Expression in Cystic Fibrosis Related Bone Disease. An Observational, Transversal, Cross-Sectional Study. Clin. Lab. 2016, 62, 1725–1730. [Google Scholar] [CrossRef] [PubMed]
- Saikia, B.J.; Bhardwaj, J.; Paul, S.; Sharma, S.; Neog, A.; Paul, S.R.; Bk, B. Understanding the roles and regulation of mitochondrial microRNAs (MitomiRs) in neurodegenerative diseases: Current status and advances. Mech. Ageing Dev. 2023, 213, 111838. [Google Scholar] [CrossRef] [PubMed]
- Bînă, A.M.; Aburel, O.M.; Avram, V.F.; Lelcu, T.; Lința, A.V.; Chiriac, D.V.; Mocanu, A.G.; Bernad, E.; Borza, C.; Craina, M.L.; et al. Impairment of mitochondrial respiration in platelets and placentas: A pilot study in preeclamptic pregnancies. Mol. Cell. Biochem. 2022, 477, 1987–2000. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wu, N.; Sun, H.; Sun, Q.; Zhang, F.; Ma, L.; Hu, Y.; Cong, X. Circulating microRNAs as diagnostic biomarkers for melanoma: A systematic review and meta-analysis. BMC Cancer 2023, 23, 414. [Google Scholar] [CrossRef] [PubMed]
- Poniewierska-Baran, A.; Słuczanowska-Głąbowska, S.; Małkowska, P.; Sierawska, O.; Zadroga, Ł.; Pawlik, A.; Niedźwiedzka-Rystwej, P. Role of miRNA in Melanoma Development and Progression. Int. J. Mol. Sci. 2023, 24, 201. [Google Scholar] [CrossRef]
- Roccuzzo, G.; Bongiovanni, E.; Tonella, L.; Pala, V.; Marchisio, S.; Ricci, A.; Senetta, R.; Bertero, L.; Ribero, S.; Berrino, E.; et al. Emerging prognostic biomarkers in advanced cutaneous melanoma: A literature update. Expert Rev. Mol. Diagn. 2024, 24, 49–66. [Google Scholar] [CrossRef] [PubMed]
| Variables | Malignant Melanoma (n = 45) | Benign Nevi (n = 45) | p-Value |
|---|---|---|---|
| Age (mean ± SD) | 52.4 ± 11.0 | 50.2 ± 9.2 | 0.306 |
| Gender | 0.289 | ||
| Male | 27 (60.0%) | 22 (48.9%) | |
| Female | 18 (40.0%) | 23 (51.1%) | |
| Pigmentation | <0.001 | ||
| Heavy | 29 (64.4%) | 12 (26.7%) | |
| Light/None | 16 (35.6%) | 33 (73.3%) | |
| Presence of ulceration | <0.001 | ||
| Yes | 24 (53.3%) | 6 (13.3%) | |
| No | 21 (46.7%) | 39 (86.7%) | |
| Presence of necrosis | <0.001 | ||
| Yes | 19 (42.2%) | 2 (4.4%) | |
| No | 26 (57.8%) | 43 (95.6%) | |
| Location | 0.323 | ||
| Occipital region | 12 (26.7%) | 9 (20.0%) | |
| Scapular region | 10 (22.2%) | 11 (24.4%) | |
| Subscapular region | 13 (28.9%) | 8 (17.8%) | |
| Lumber region | 10 (22.2%) | 17 (37.8%) |
| Variables | Malignant Melanoma (n = 45) | Benign Nevi (n = 45) | p-Value |
|---|---|---|---|
| Breslow thickness (mean ± SD, mm) | 2.0 ± 1.4 | N/A | - |
| Tumor infiltration depth (mean ± SD, mm) | 2.0 ± 1.1 | 0.3 ± 0.1 | <0.001 |
| Clark level | - | ||
| I–II | 6 (13.3%) | N/A | |
| III–IV | 22 (48.9%) | N/A | |
| V | 17 (37.8%) | N/A | |
| Mitotic rate | - | ||
| High | 18 (40%) | N/A | |
| Low | 27 (60%) | N/A | |
| Immunohistochemical markers (positive) | |||
| S100 | 30 (66.7%) | 14 (31.1%) | <0.001 |
| HMB45 | 26 (57.8%) | 19 (42.2%) | 0.140 |
| Resection margins | - | ||
| Excised with safety limits | 41 (91.1%) | N/A | |
| Lateral resection margins present | 4 (8.9%) | N/A | |
| Tumor appearance | 0.540 | ||
| Nodular | 23 (51.1%) | 18 (40.0%) | |
| Polypoid | 12 (26.7%) | 16 (35.6%) | |
| Epithelioid | 10 (22.2%) | 11 (24.4%) | |
| Vascular involvement | - | ||
| Presence of tumor emboli in blood vessels | 7 (15.6%) | N/A | |
| Absence of tumor emboli | 38 (84.4%) | N/A | |
| Inflammatory response | <0.001 | ||
| Present | 34 (75.6%) | 13 (28.9%) | |
| Absent | 11 (24.4%) | 32 (71.1%) | |
| Immune cell infiltrate type | 0.114 | ||
| Lymphocytic | 22 (64.7%) | 6 (46.2%) | |
| Plasma cell | 8 (23.5%) | 2 (15.4%) | |
| Mixed | 4 (11.8%) | 5 (38.5%) |
| Gene | Fold Change (M/C) | Malignant Melanoma (n = 45) | Benign Nevi (n = 45) | p-Value |
|---|---|---|---|---|
| hsa-miR-133b | 1.12 | 34.82 ± 2.81 | 31.17 ± 2.64 | <0.001 |
| hsa-miR-335-5p | 1.09 | 34.15 ± 2.03 | 31.29 ± 3.15 | <0.001 |
| hsa-miR-200a-3p | 1.10 | 34.07 ± 1.99 | 30.96 ± 2.61 | <0.001 |
| hsa-miR-885-5p | 1.10 | 35.31 ± 2.64 | 32.05 ± 2.22 | <0.001 |
| hsa-miR-20b-5p | 1.09 | 31.87 ± 2.06 | 29.21 ± 2.43 | <0.001 |
| hsa-miR-7-1-3p | 1.04 | 34.75 ± 2.87 | 33.39 ± 2.29 | 0.014 |
| hsa-miR-301a-3p | 1.04 | 33.60 ± 2.43 | 32.30 ± 2.61 | 0.016 |
| hsa-let-7b-3p | 1.04 | 32.99 ± 2.71 | 31.63 ± 2.14 | 0.009 |
| hsa-miR-148b-3p | 1.06 | 30.78 ± 2.02 | 29.17 ± 2.29 | <0.001 |
| hsa-miR-584-5p | 1.07 | 30.94 ± 2.97 | 28.73 ± 2.31 | <0.001 |
| Gene | Fold Change (M/C) | Malignant Melanoma (n = 45) | Benign Nevi (n = 45) | p-Value |
|---|---|---|---|---|
| hsa-miR-451a | 0.90 | 31.28 ± 2.06 | 34.65 ± 2.27 | <0.001 |
| hsa-miR-29c-3p | 0.97 | 31.21 ± 2.78 | 32.28 ± 2.95 | 0.080 |
| hsa-miR-29b-3p | 0.92 | 31.11 ± 2.94 | 33.93 ± 2.97 | <0.001 |
| hsa-miR-29a-3p | 0.95 | 29.48 ± 2.89 | 31.00 ± 2.81 | 0.013 |
| hsa-miR-361-5p | 0.93 | 30.25 ± 2.61 | 32.57 ± 2.39 | <0.001 |
| hsa-miR-18a-5p | 0.95 | 31.34 ± 2.92 | 32.96 ± 2.10 | 0.003 |
| hsa-miR-495-3p | 0.93 | 28.19 ± 2.09 | 30.23 ± 2.68 | <0.001 |
| hsa-miR-590-5p | 0.94 | 30.94 ± 2.20 | 33.04 ± 2.44 | <0.001 |
| hsa-miR-215-5p | 0.96 | 29.48 ± 2.05 | 30.85 ± 2.12 | 0.002 |
| hsa-miR-127-3p | 0.90 | 27.39 ± 2.33 | 30.33 ± 2.54 | <0.001 |
| miRNA Gene Subtype | Breslow Index (ρ) | p-Value | Mitotic Rate (ρ) | p-Value | Vascular Involvement (ρ) | p-Value |
|---|---|---|---|---|---|---|
| hsa-miR-133b | 0.351 | 0.036 | 0.256 | 0.059 | 0.126 | 0.182 |
| hsa-miR-335-5p | 0.401 | <0.001 | 0.313 | 0.014 | 0.247 | 0.052 |
| hsa-miR-200a-3p | 0.464 | 0.015 | 0.331 | 0.006 | −0.038 | 0.616 |
| hsa-miR-885-5p | 0.197 | 0.418 | 0.219 | 0.033 | 0.173 | 0.225 |
| hsa-miR-20b-5p | 0.388 | 0.001 | 0.482 | <0.001 | 0.278 | 0.045 |
| hsa-miR-7-1-3p | −0.214 | 0.099 | 0.025 | 0.427 | −0.350 | 0.212 |
| hsa-miR-301a-3p | −0.384 | 0.001 | −0.377 | 0.007 | 0.059 | 0.621 |
| hsa-let-7b-3p | 0.132 | 0.326 | −0.170 | 0.149 | 0.275 | 0.038 |
| hsa-miR-148b-3p | 0.202 | 0.080 | −0.026 | 0.713 | 0.074 | 0.411 |
| hsa-miR-584-5p | 0.316 | <0.001 | 0.198 | 0.047 | 0.056 | 0.304 |
| hsa-miR-451a | −0.359 | 0.005 | −0.139 | 0.306 | −0.261 | 0.014 |
| hsa-miR-29c-3p | 0.240 | 0.059 | 0.150 | 0.081 | 0.326 | 0.008 |
| hsa-miR-29b-3p | 0.565 | <0.001 | 0.279 | 0.006 | 0.322 | 0.046 |
| hsa-miR-29a-3p | −0.173 | 0.147 | 0.079 | 0.341 | 0.129 | 0.094 |
| hsa-miR-361-5p | −0.236 | 0.248 | 0.196 | 0.035 | 0.242 | 0.138 |
| hsa-miR-18a-5p | −0.333 | 0.040 | 0.244 | 0.036 | −0.012 | 0.514 |
| hsa-miR-495-3p | 0.192 | 0.715 | −0.023 | 0.439 | 0.045 | 0.447 |
| hsa-miR-590-5p | 0.050 | 0.614 | 0.108 | 0.054 | −0.145 | 0.189 |
| hsa-miR-215-5p | −0.136 | 0.134 | −0.091 | 0.318 | −0.249 | 0.045 |
| hsa-miR-127-3p | −0.418 | <0.001 | −0.349 | 0.006 | −0.384 | 0.027 |
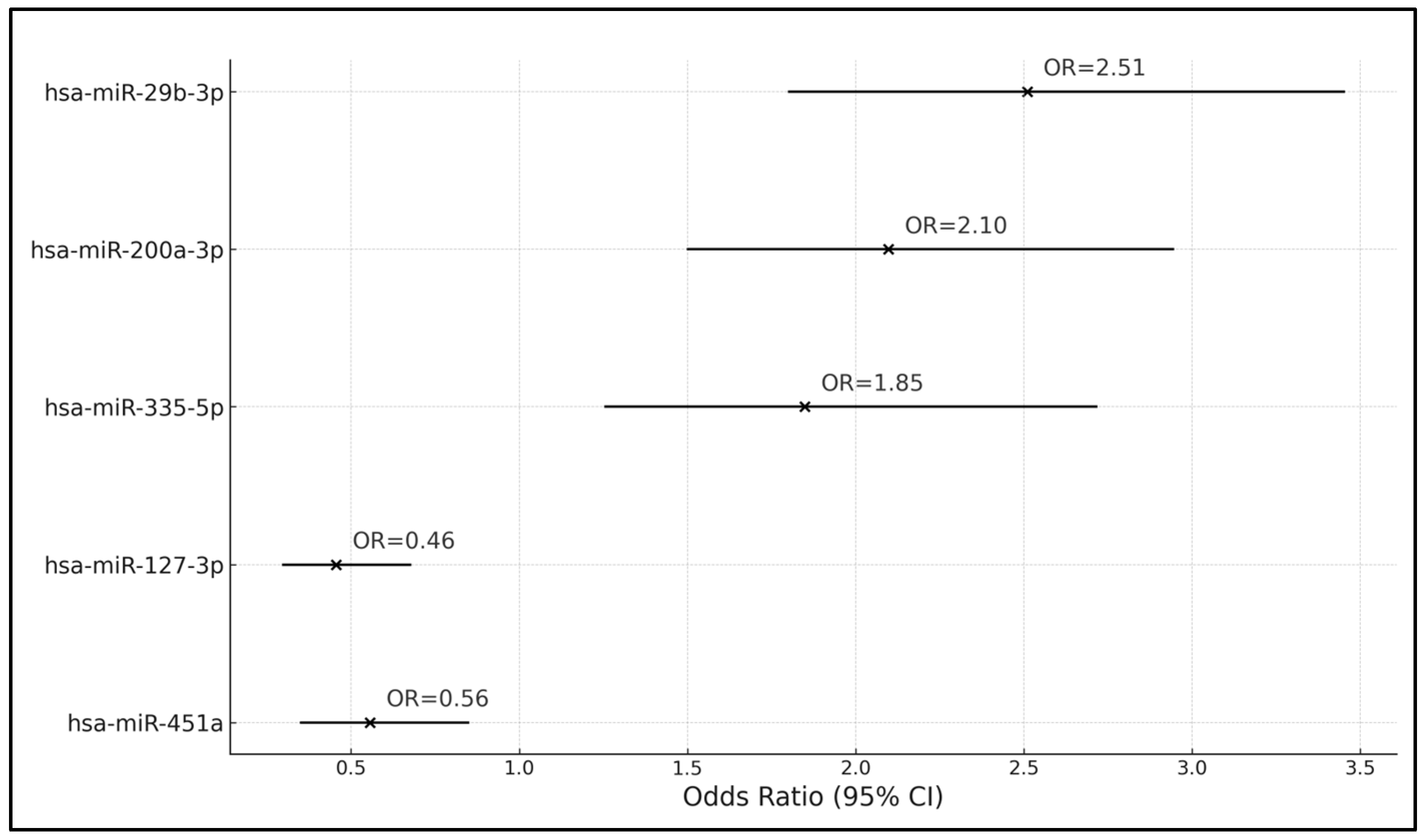
| Gene Type | Coefficient (β) | SE | OR | 95% CI | p-Value |
|---|---|---|---|---|---|
| hsa-miR-29b-3p | 0.916 | 0.213 | 2.510 | 1.803–3.451 | <0.001 |
| hsa-miR-200a-3p | 0.741 | 0.189 | 2.098 | 1.501–2.943 | 0.003 |
| hsa-miR-335-5p | 0.615 | 0.221 | 1.849 | 1.256–2.715 | 0.002 |
| hsa-miR-127-3p | −0.784 | 0.245 | 0.456 | 0.298–0.675 | <0.001 |
| hsa-miR-451a | −0.587 | 0.220 | 0.556 | 0.351–0.848 | 0.008 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Prodan, M.; Costescu, S.; Elagez, A.; Laitin, S.M.D.; Bloanca, V.; Crainiceanu, Z.; Seclaman, E.; Toma, A.-O.; Fericean, R.M.; Puenea, G.; et al. Molecular Markers in Melanoma Progression: A Study on the Expression of miRNA Gene Subtypes in Tumoral vs. Benign Nevi. Curr. Oncol. 2024, 31, 2881-2894. https://doi.org/10.3390/curroncol31050220
Prodan M, Costescu S, Elagez A, Laitin SMD, Bloanca V, Crainiceanu Z, Seclaman E, Toma A-O, Fericean RM, Puenea G, et al. Molecular Markers in Melanoma Progression: A Study on the Expression of miRNA Gene Subtypes in Tumoral vs. Benign Nevi. Current Oncology. 2024; 31(5):2881-2894. https://doi.org/10.3390/curroncol31050220
Chicago/Turabian StyleProdan, Mihaela, Sergiu Costescu, Ahmed Elagez, Sorina Maria Denisa Laitin, Vlad Bloanca, Zorin Crainiceanu, Edward Seclaman, Ana-Olivia Toma, Roxana Manuela Fericean, George Puenea, and et al. 2024. "Molecular Markers in Melanoma Progression: A Study on the Expression of miRNA Gene Subtypes in Tumoral vs. Benign Nevi" Current Oncology 31, no. 5: 2881-2894. https://doi.org/10.3390/curroncol31050220
APA StyleProdan, M., Costescu, S., Elagez, A., Laitin, S. M. D., Bloanca, V., Crainiceanu, Z., Seclaman, E., Toma, A.-O., Fericean, R. M., Puenea, G., & Cozma, G. V. (2024). Molecular Markers in Melanoma Progression: A Study on the Expression of miRNA Gene Subtypes in Tumoral vs. Benign Nevi. Current Oncology, 31(5), 2881-2894. https://doi.org/10.3390/curroncol31050220





