Fly Ash Waste Recycling by Pt/TiO2 Incorporation for Industrial Dye Removal
Abstract
1. Introduction
2. Materials and Methods
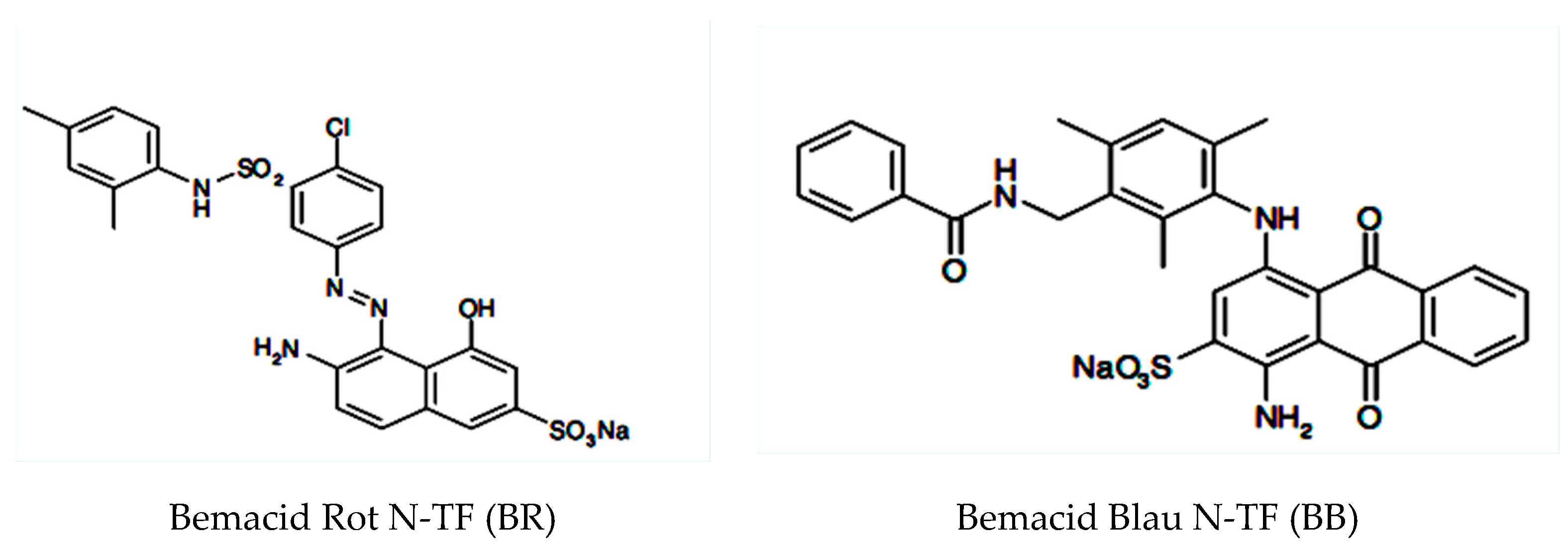
2.1. Materials
2.2. Materials Preparation Procedures
2.2.1. Preparation of Pt-Cys NPs
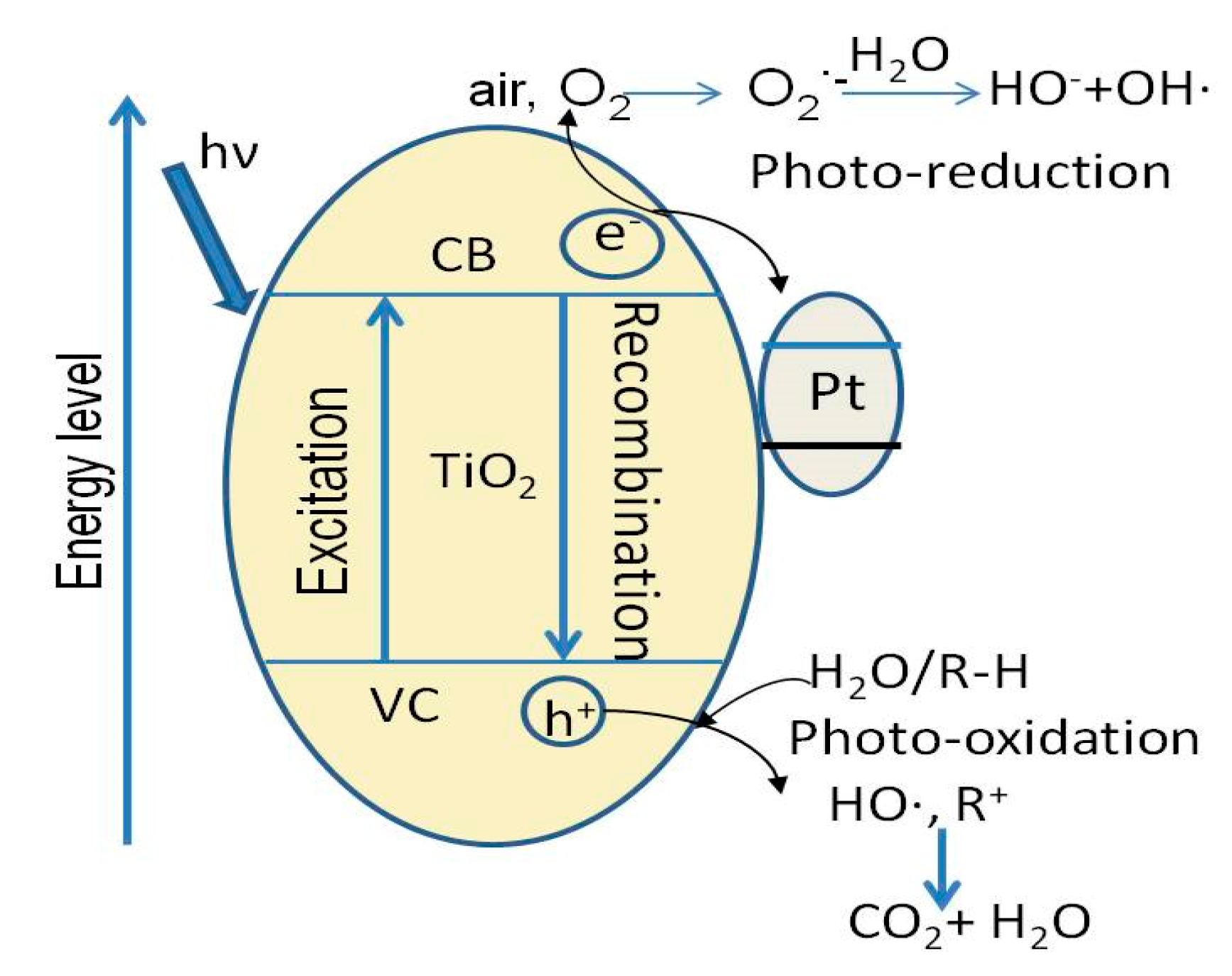
2.2.2. Preparation of Photocatalytic Adsorbent Materials
2.3. Structure and Surface Characterization
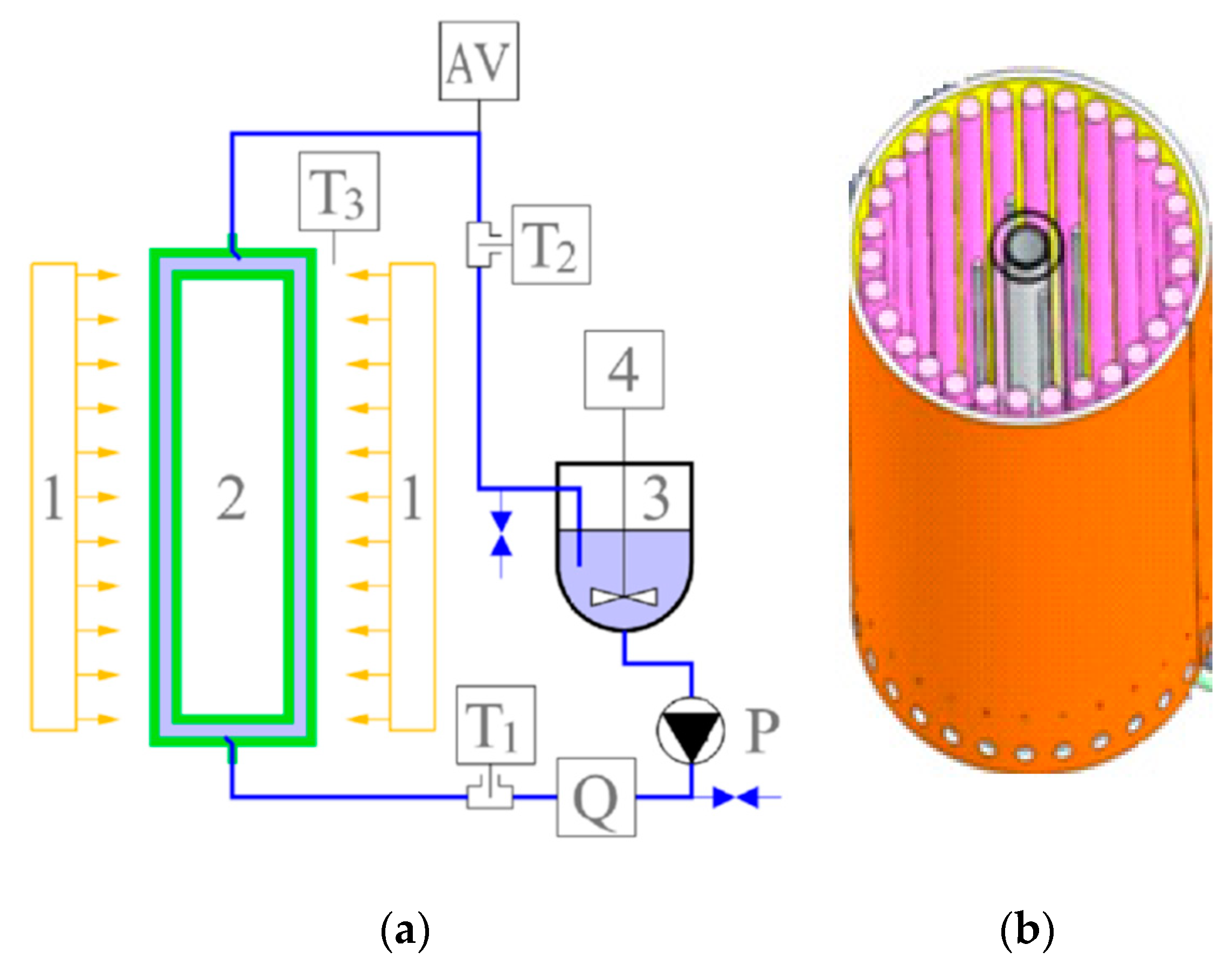
2.4. Tandem Adsorption and Photodegradation Experiments
3. Results and Discussion
3.1. Characterization of the Composite
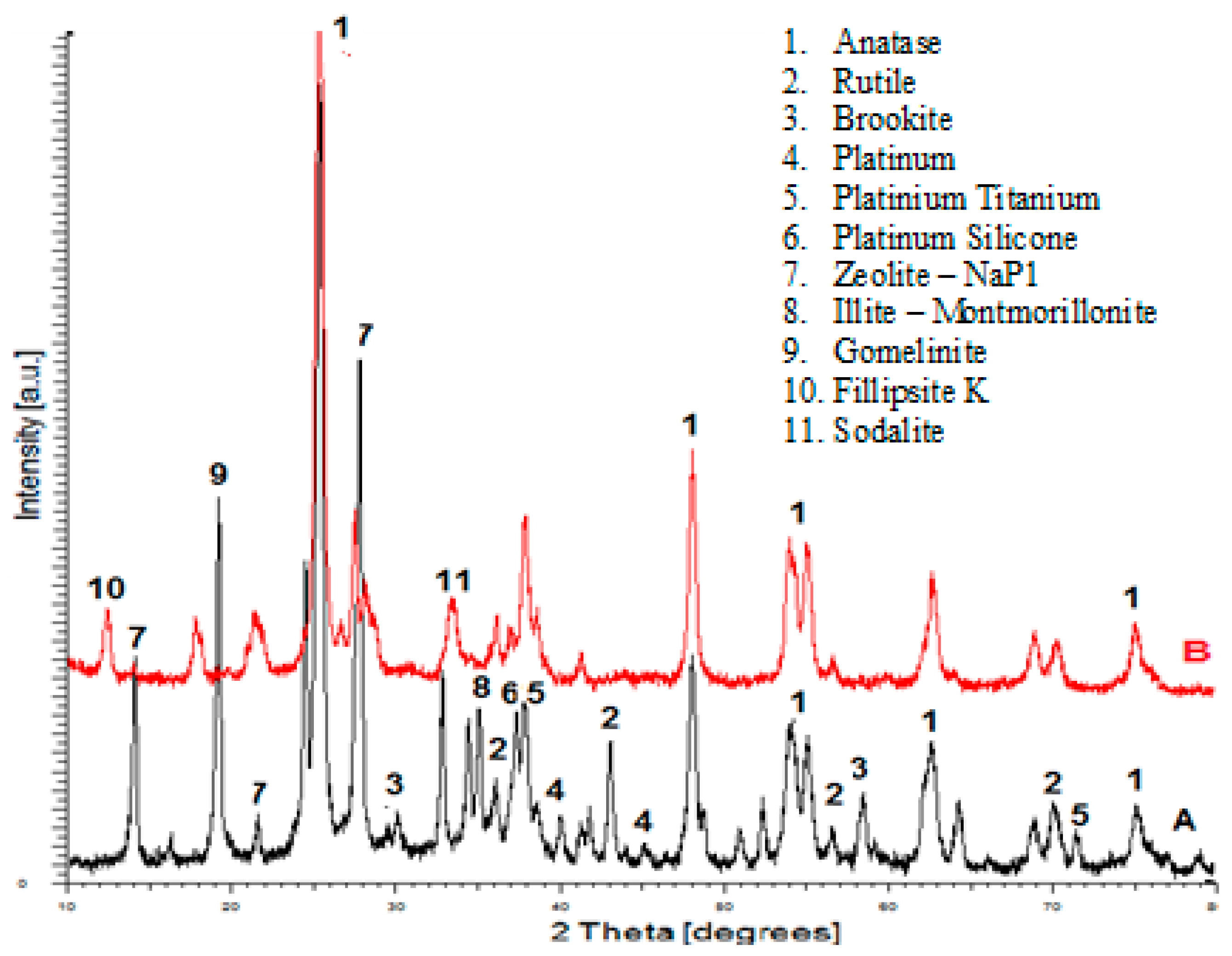
3.1.1. Crystalline Structure
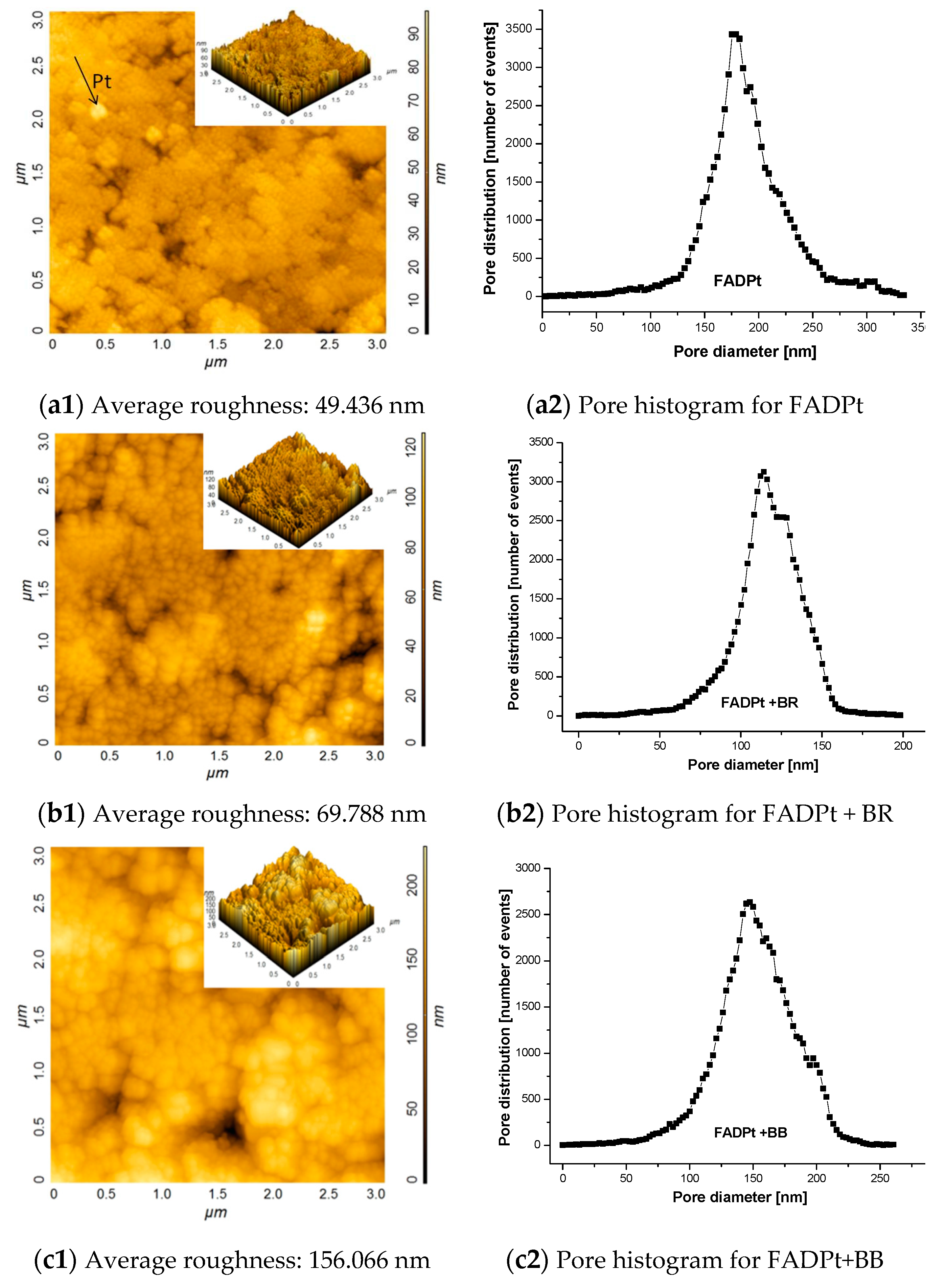
3.1.2. Surface Morphology
3.1.3. UV–Vis Diffuse Reflectance Spectra
3.1.4. X-ray Photoelectron Spectroscopy
3.1.5. FT-IR Analysis
3.2. Tandem Adsorption and Photodegradation on FADPt
3.2.1. Dye Adsorption on FADPt
3.2.2. The Tandem Adsorption and Photocatalysis Processes
3.2.3. The Effect of Concentration of the H2O2 on the Photocatalytic Process
3.3. Kinetics Modeling of the Adsorption Processes
3.4. Kinetics Modeling of the Photocatalysis Processes
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Safavi, A.; Momeni, S. Highly efficient degradation of azo dyes by palladium/hydroxiapatite/Fe3O4 nanocatalyst. J. Hazard. Mater. 2012, 201, 125–131. [Google Scholar] [CrossRef]
- Semeraro, P.; Rizzi, V.; Fini, P.; Matera, S.; Cosma, P.; Franco, E.; García, R.; Ferrándiz, M.; Núñez, E.; Gabaldón, J.A.; et al. Interaction between industrial textile dyes and cyclodextrins. Dyes Pigment. 2015, 119, 84–94. [Google Scholar] [CrossRef]
- Rizzi, V.; D’Agostino, F.; Gubitosa, J.; Fini, P.; Petrella, A.; Agostiano, A.; Semeraro, P.; Cosma, P. An Alternative Use of Olive Pomace as a Wide-Ranging Bioremediation Strategy to Adsorb and Recover Disperse Orange and Disperse Red Industrial Dyes from Wastewater. Separations 2017, 4, 29. [Google Scholar] [CrossRef]
- Luo, J.; Zhang, H.; Yang, J. Hydrothermal synthesis of sodalite on alkali-activated coal fly ashfor removal of lead ions. Procedia Environ. Sci. 2016, 31, 605–614. [Google Scholar] [CrossRef]
- Visa, M.; Pricop, F.; Duta, A. Sustainable treatment of wastewaters resulted in textile dyeing industry. J. Clean Techn. Environ. Policy 2012, 13, 855–861. [Google Scholar] [CrossRef]
- Malakootian, M.; Almasi, A.; Hossaini, H. Pb and Co removal from paint industries effluents using wood ash. Environ. Sci. Technol. 2008, 5, 217–222. [Google Scholar] [CrossRef]
- Patel, H.; Vashi, R.T. Treatment of textile wastewater by adsorption and coagulation. E J. Chem. 2010, 7, 1468–1476. [Google Scholar] [CrossRef]
- Çınar, Ö.; Yaşar, S.; Kertmen, M.; Demiröz, K.; Kitis, M. Effect of cycle time on biodegradation of azo dye in sequencing batch reactor. Process Saf. Environ. 2008, 86, 455–460. [Google Scholar] [CrossRef]
- Shaw, C.B.; Carliell, C.M.; Wheatley, A.D. Anaerobic/aerobic treatment of coloured textile effluents using sequencing batch reactors. Water Res. 2002, 36, 1193–2001. [Google Scholar] [CrossRef]
- Tantak, N.P.; Chaudhari, S. Degradation of azo dyes sequential treatment. J. Hazard. Mater. B 2006, 136, 698–705. [Google Scholar] [CrossRef]
- Jing, G.; Luan, M.; Chen, T. Progress of catalytic wet air oxidation technology. Arab. J. Chem. 2016, 9, S1208–S1213. [Google Scholar] [CrossRef]
- Visa, M.; Duta, A. Methyl-orange and cadmium simultaneous removal using fly ash and photo-Fenton systems. J. Hazard. Mater. 2013, 244, 773–779. [Google Scholar] [CrossRef]
- Azizi, A.; Alavi Moghaddam, M.R.; Maknoon, R.; Kowsari, E. Innovative combined technique for high concentration of azo dye AR18 wastewater treatment using modified SBR and enhanced Fenton process as post treatment. Process Saf. Environ. 2015, 95, 255–264. [Google Scholar] [CrossRef]
- Rogozea, E.A.; Petcu, A.R.; Olteanu, N.L.; Lazar, C.A.; Cadar, D.; Mihaly, M. Tandem adsorption-photodegradation activity induced by light on NiO-ZnO p–n couple modified silica nanomaterials. Mater. Sci. Semicond. Process. 2017, 57, 1–11. [Google Scholar] [CrossRef]
- Olteanu, N.L.; Rogozea, E.A.; Petcu, A.R.; Lazar, C.A.; Meghea, A.; Mihaly, M. “One-pot” synthesis of Au-ZnO-SiO2 nanostructures for sunlight photodegradation. J. Mol. Catal. A-Chem. 2016, 414, 148–159. [Google Scholar] [CrossRef]
- Rogozea, E.A.; Olteanu, N.L.; Petcu, A.R.; Lazar, C.A.; Meghea, A.; Mihaly, M. Extension of optical properties of ZnO/SiO2 materials induced by incorporation of Au or NiO nanoparticles. Opt. Mater. 2016, 56, 45–48. [Google Scholar] [CrossRef]
- Sun, P.; Xue, R.; Zhang, W.; Zada, I.; Liu, Q.; Gu, J.; Su, H.; Zhang, Z.; Zhang, J.; Zhang, D. Photocatalyst of organic pollutants decomposition: TiO2/glass fiber cloth composites. Catal. Today 2016, 274, 2–7. [Google Scholar] [CrossRef]
- Zang, Q.; Zheng, D.D.; Xu, L.S.; Chang, C.T. Photocatalytic conversion of terephthalic acid preparation wastewater to hydrogen by graphene-modified TiO2. Catal. Today 2016, 274, 8–14. [Google Scholar] [CrossRef]
- Legrand, A.; Moissette, A.; Hureau, M.; Casale, S.; Massiani, P.; Vezin, H.; Mamede, A.S.; Batonneau-Gener, I. Electron transfers in a TiO2-containing MOR zeolite: Synthesis of the nanoassemblies and application using a probe chromophore molecule. Phys. Chem. Chem. Phys. 2014, 26, 13145–13155. [Google Scholar] [CrossRef]
- Andronic, L.; Cazan, C.; Enesca, A.; Visa, M. TiO2–active carbon composites for wastewater photocatalysis. J. Sol-Gel Sci. Technol. 2014, 71, 396–405. [Google Scholar] [CrossRef]
- Andronic, L.; Hristache, B.; Enesca, A.; Visa, M.; Duta, A. Studies on titanium oxide catalyst doped with heavy metals (cadmium, copper and nickel). Environ. Engin. Manag. J. 2009, 8, 747–751. [Google Scholar] [CrossRef]
- Chen, Y.H.; Chen, L.L.; Shang, N.C. Photocatalytic degradation of dimethyl phthalate in an aqueous solution with Pt-doped TiO2-coated magnetic PMMA microspheres. J. Hazard. Mater. 2009, 172, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Mohapatra, S.K.; Kondamudi, N.; Banerjee, S.; Misra, M. Functionalization of self-organized TiO2 nanotubes with Pd nanoparticles for photocatalytic decomposition of dyes under solar light illumination. Langmuir 2008, 24, 11276–11281. [Google Scholar] [CrossRef] [PubMed]
- Vaiano, V.; Iervolino, G.; Sannino, D.; Murcia, J.J.; Hidalgo, M.C.; Ciambelli, P.; Navio, J.A. Photocatalytic removal of patent blue V dye on Au-TiO2 and Pt-TiO2 catalysts. Appl. Catal. B-Environ. 2016, 188, 134–146. [Google Scholar] [CrossRef]
- Visa, M.; Andronic, L.; Enesca, A. Behavior of the new composites obtained from fly ash and titanium dioxide in removing of the pollutants from wastewater. J. Appl. Surf. Sci. 2016, 388, 359–36912. [Google Scholar] [CrossRef]
- Wang, H.; Niu, J.; Long, X.; He, Y. Sonophotocatalytic degradation of methyl orange by nano-sized Ag/TiO2 particles in aqueous solutions. Ultrason. Sonochem J. 2008, 15, 386–392. [Google Scholar] [CrossRef]
- Visa, M.; Duta, A. TiO2/fly ash novel substrate for simultaneous removal of heavy metals and surfactants. Chem. Engin. J. 2013, 223, 860–868. [Google Scholar] [CrossRef]
- Gasca-Tirado, J.R.; Manzano-Ramirez, A.; Villaseñor-Mora, C.; Muñiz-Villarreal, M.S.; Zaldivar-Cadena, A.A.; Rubio-Ávalos, J.C.; Borráse, V.A.; Mendoza, R.N. Incorporation of photoactive TiO2 in an aluminosilicate inorganic polymer by ion-exchange. Microp. Mesopor. Mat. 2012, 153, 282–287. [Google Scholar] [CrossRef]
- Ramme, B.W.; Tharaniyil, M.P. Coal Combustion Products Utilization Handbook, 2nd ed.; Manufactured in the U.S. of America: Milwaukee, WI, USA, 2004; pp. 15–18. [Google Scholar]
- Mihaly, M.; Fleancu, M.C.; Olteanu, N.L.; Bojin, D.; Meghea, A.; Enachescu, M. Synthesis of gold nanoparticles by microemulsion assisted photoreduction method. Comptes R. Chim. 2012, 15, 1012–1021. [Google Scholar] [CrossRef]
- Visa, M.; Duta, A.; Visa, I.; Moldovan, M.; Neagoe, M. Continuous Flow Tubular Reactor for Simultaneous Photocatalysis and Suspension Adsorption Processes. Patent 201600536, 28 April 2017. [Google Scholar]
- Zhang, N.; Liu, S.; Xu, Y.J. Recent progress on metal core@semiconductor shell nano-composites as a promising type of photocatalyst. Nanoscale 2012, 4, 2227–2238. [Google Scholar] [CrossRef]
- Owens, D.K.; Wendt, R.C. Estimation of the surface free energy of polymers. J. Appl. Polym. Sci. 1969, 13, 1741–1747. [Google Scholar] [CrossRef]
- Otero, J.A.; Mazarrasa, O.; Villasante, J.; Silva, V.; Pradanos, P.; Calvo, J.I.; Hernandez, A. Three independent ways to obtain information on pore size distributions of nanofiltration membranes. J. Membr. Sci. 2008, 309, 17–27. [Google Scholar] [CrossRef]
- Ökte, A.N.; Karamanis, D.; Tuncel, D. Dual functionality of TiO2–flyash nanocomposites: Water vapor adsorption and photocatalysis. Catal. Today 2014, 230, 205–213. [Google Scholar] [CrossRef]
- Zhang, B.; Wang, D.; Hou, Y.; Yang, S.; Yang, X.H.; Zhong, J.H.; Liu, J.; Wang, H.F.; Hu, P.; Zhao, H.J.; et al. Facet-dependent catalytic activity of platinum nanocrystals for triiodide reduction in dye-sensitized solar cells. Sci. Rep. 2013, 3, 1836. [Google Scholar] [CrossRef]
- Jason, M.A.; Morris, W.H.; Lukehart, C.M. Synthesis of shaped Pt nanoparticles using common anions or small molecules as shape-directing agents: Observation of a strong halide or pseudo-halide effect. J. Mater. Chem. A. 2015, 3, 2012–2018. [Google Scholar] [CrossRef]
- Devi, L.G.; Kumar, S.G. Exploring the critical dependence of adsorption of various dyes on the degradation rate using Ln3+-TiO2 surface under UV/solar light. Appl. Surf. Sci. 2012, 261, 137–146. [Google Scholar] [CrossRef]
- Visa, M.; Isac, L.; Duta, A. Fly ash adsorbents for multi-cation wastewater treatment. Appl. Surf. Sci. 2012, 17, 6345–6352. [Google Scholar] [CrossRef]
- Duta, A.; Visa, M. Simultaneous removal of two industrial dyes by adsorption and photocatalysis on a fly-ash-TiO2 composite. Photochem. Photobiol. A-Chem. 2015, 306, 21–30. [Google Scholar] [CrossRef]
- Bruno, T.J.; Svoronos, P.D.N. CRC Handbook of Fundamental Spectroscopic Correlation Charts, 1st ed.; CRC Press: Boca Raton, FL, USA, 2006. [Google Scholar] [CrossRef]
- Stem, N.; De Souza, M.L.; Araujo de Faria, D.L.; Dos Santos Filho, S.G. Formation of Ti(III) and Ti(IV) states in Ti3O5 nano- and microfibers obtained from hydrothermal annealing of C-doped TiO2 on Si. Thin Solid Films. 2014, 558, 67–74. [Google Scholar] [CrossRef]
- Gözmen, B.; Kayan, B.; Gizir, A.M.; Hesenov, A. Oxidative degradations of reactive blue 4 dye by different advanced oxidation methods. J. Hazard. Mater. 2009, 168, 129–136. [Google Scholar] [CrossRef]
- Ohtani, B.; Prieto-Mahaney, O.O.; Li, D.; Abe, R. What is Degussa (Evonik) P25? Crystalline composition analysis, reconstruction from isolated pure particles and photocatalytic activity test. J. Photochem. Photobiol. A 2010, 216, 179–182. [Google Scholar] [CrossRef]
- El-Mekkawi, D.; Galal, H.R. Removal of a synthetic dye “Direct Fast Blue B2RL” via adsorption and photocatalytic degradation using low cost rutile and Degussa P25 titanium dioxide. J. Hydro-Environ. Res. 2013, 7, 219–226. [Google Scholar] [CrossRef]
- Najdanovic, S.; Mitrovic, J.; Zarubica, A.; Bojic, A. The effectof operational parameters on decolourisation of textile and comparison efficiencies of the UV/H2O2, Fenton and photo-Fenton processes: A Review. Facta Univ. Phys. Chem. Tecnol. 2017, 15, 23–34. [Google Scholar] [CrossRef]
- Muruganandham, M.; Swaminathan, M. Solar driven decolourisation ofreactive yellow 14 by advanced oxidation processes in heterogeneous and homogeneous media. Dye. Pigm. 2007, 72, 137–143. [Google Scholar] [CrossRef]
- Chu, W.; Choy, W.K.; So, T.Y. The effect of solution pH and peroxide in theTiO2-induced photocatalysis of chlorinated aniline. J. Hazard. Mater. 2007, 141, 86–91. [Google Scholar] [CrossRef] [PubMed]
- Ho, Y.S.; Mckay, G. The kinetic of sorption of basic dyes from aqueous solution by sphagnum Moss peat. J. Chem. Eng. 1998, 76, 822–827. [Google Scholar] [CrossRef]
- Guettaı, N.; Amar, H.A. Photocatalytic oxidation of methyl orange in presence of titanium dioxide in aqueous suspension. Part II: Kinetics study. Desalination 2005, 185, 439–448. [Google Scholar] [CrossRef]


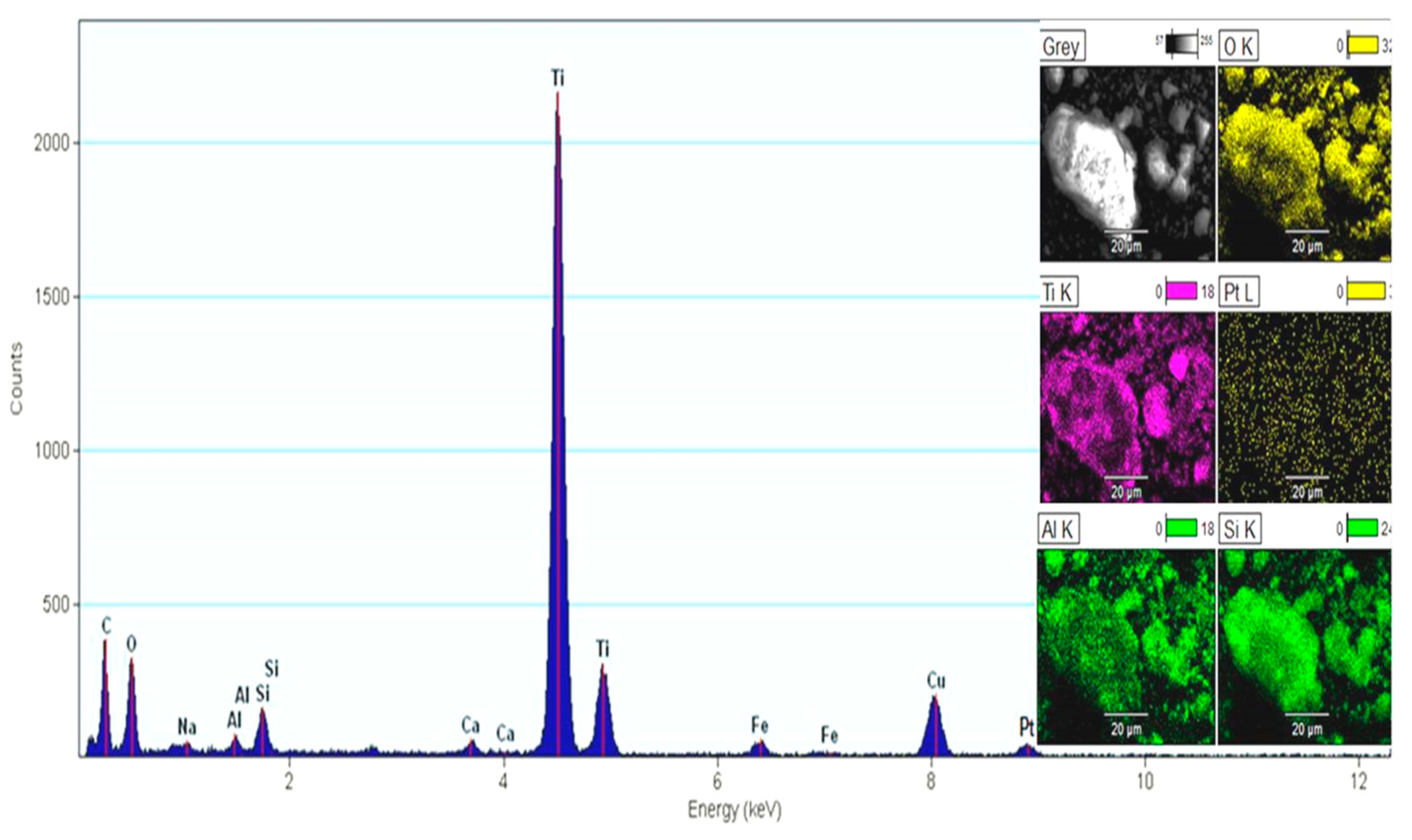

| Components of FADPt | Anatase | Rutile | Brookite | Pt (NPs) | Zeolite NaP1 | Other |
|---|---|---|---|---|---|---|
| Composition (%) | 43.99 | 1.81 | 1.79 | 0.88 | 17.14 | 35.27 |
| Sample | SBET (m2/g) | VMicropores (cm3/g) | DAverage pores (nm) | Surface Energy | Eg (eV) | |
|---|---|---|---|---|---|---|
| Polar (mN/m) | Dispersive (mN/m) | |||||
| FA | 6.14 | 0.0042 | 27.2 | 58.43 | 5.54 | 2.45 |
| FADPt | 40 | 0.17 | 35 | 88.90 | 1.42 | 2.12 |
| Sample | Area (µm2) | Volume (µm2·nm) |
|---|---|---|
| FADPt/grain | 0.049 | 3.269 |
| FADPt loaded with BR/grain | 0.872 | 6.946 |
| FADPt loaded with BB/grain | 0.916 | 18.621 |
| Element Line | Element Wt. before (%) | Atom before (%) | Element Wt. (%) Substrate Loaded with BR | Atom (%) Substrate Loaded with BR | Element Wt. (%) Substrate Loaded with BB | Atom (%) Substrate Loaded with BB |
|---|---|---|---|---|---|---|
| C K | 1.94 | 4.84 | 60.44 | 62.90 | 2.15 | 5.61 |
| N K | 0 | 0 | 18.18 | 17.54 | 11.38 | 16.39 |
| O K | 18.89 | 35.38 | 14.2 | 12.1 | 47.07 | 58.38 |
| Na K | 3.01 | 3.92 | 0.35 | 0.23 | 2.42 | 2.12 |
| Mg K | 0.99 | 1.32 | 0.08 | 0.05 | 0.11 | 0.09 |
| Al K | 2.61 | 2.9 | 0.77 | 0.38 | 4.18 | 2.13 |
| Si K | 12.81 | 13.62 | 0.85 | 4.41 | 4.92 | 3.54 |
| S K | 0 | 0 | 0.01 | 0.3 | 0.11 | 0.07 |
| Ca K | 22.9 | 17.13 | 0.32 | 0.41 | 1.58 | 0.8 |
| Ti K | 32.36 | 20.2 | 4.75 | 1.47 | 25.98 | 10.86 |
| Pt M | 4.49 | 0.69 | 0.05 | 0.21 | 0.1 | 0.01 |
| Total | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 |
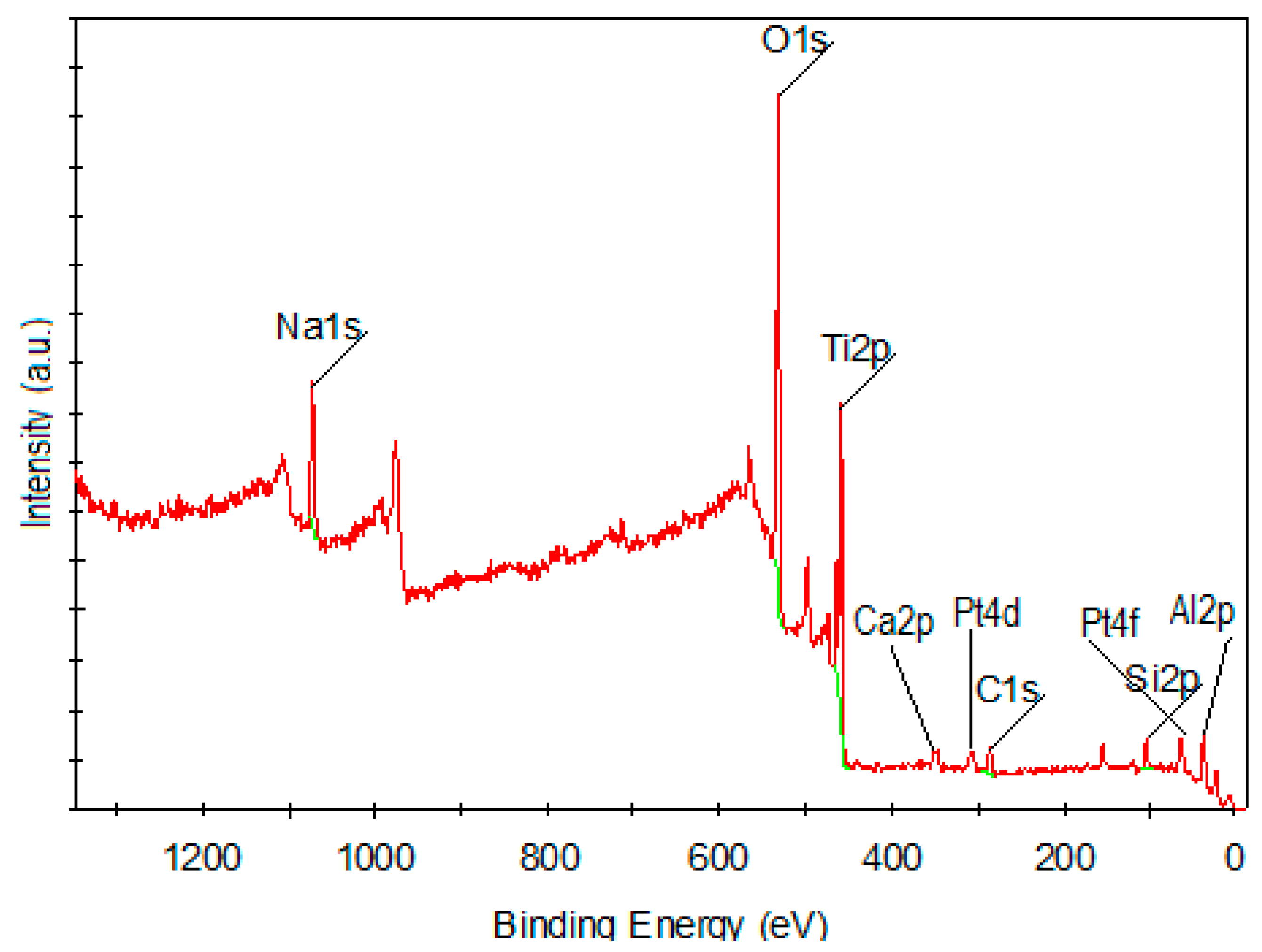
| Element | Binding Energy (eV) | Atomic % |
|---|---|---|
| O1s | 531 | 52.07 |
| Ti2p | 459.1 | 13.24 |
| Na1s | 1072.77 | 9.79 |
| Si2p | 103.18 | 8.58 |
| C1s | 286.03 | 8.64 |
| Ca2p | 347.91 | 1.81 |
| Al2p | 75.11 | 4.01 |
| Pt4d | 315.08 | 1.45 |
| Pt4f | 75.08 | 0.40 |
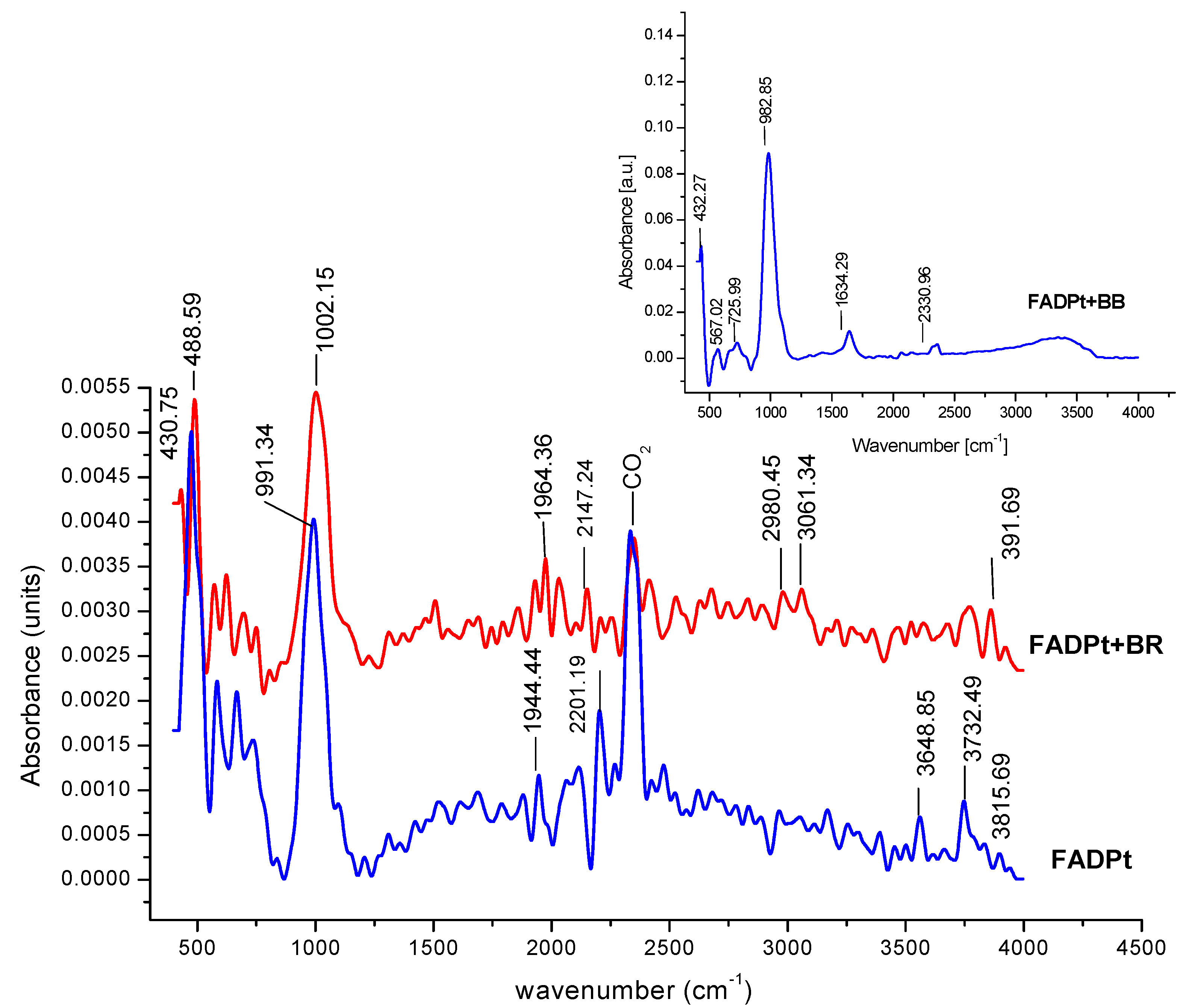
| Characteristics Groups | FADPt (cm−1) | FADPt + BB (cm−1) | FADPt + BR (cm−1) |
|---|---|---|---|
| Si–OH groups | 3815 | - | 3931 |
| Si–(OH)Al hydroxyl group stretching | 3730 | - | 3755 |
| OH groups bridging hydroxyls in zeolite cages to the same Al–OH–Si | 3648 | - | 3661; 3509 |
| Linear carbonyl Pt4+, Pt–CO, [Pt3CO)6]2− Pt/Al2O3—(2093 cm−1); CO adsorbed on Pt atoms Pt2+–CO: 2155, 2141 cm−1 | 2176 2195 | - | 2246; 2184; 2111 |
| 2093 2069 2155 | - | 2054 | |
| Weak bands Pt–(CO)–Pt | 1918 | 1873 | |
| Water molecules | 1615 | 1642 | 1678 |
| Si–Al–O; Al–O asymmetric stretch Ti–O–Si | 990 1002 | 982 992 | 997 971 |
| O–Ti–O from rutile | 425 | - | 447 |
| Ti–O–Ti bridging vibration | 811 | 730 | 788,740 |
| Si–O bond of the zeolite structure | 689 | 662 | 684 |
| HO–Pt–OH stretch vibration | 577 | 561 | 582 |
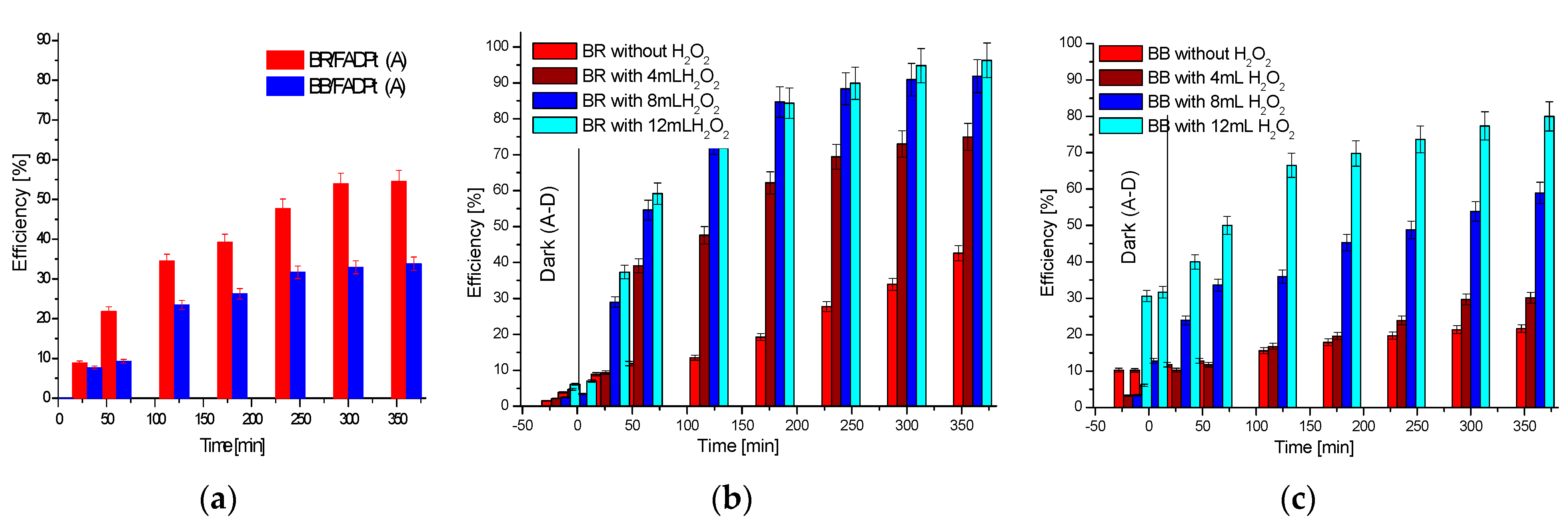
| BR—Removal (%) | BB—Removal (%) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| VH2O2 (30%) (mL) | VH2O2 (30%) (mL) | |||||||||
| Time (min) | 4 | 8 | 12 | 8 − 4 | 12 − 8 | 4 | 8 | 12 | 8 − 4 | 12 − 8 |
| 120 | 47.59 | 73.76 | 74.50 | 26.16 | 0.74 | 16.80 | 36.01 | 66.54 | 19.21 | 30.53 |
| 180 | 62.14 | 84.65 | 84.33 | 22.51 | −0.32 | 19.63 | 45.32 | 69.77 | 25.69 | 24.44 |
| 240 | 69.43 | 88.39 | 89.92 | 18.96 | 1.53 | 23.94 | 48.80 | 73.65 | 24.85 | 24.85 |
| 300 | 72.98 | 90.89 | 93.89 | 17.91 | 3.00 | 29.74 | 53.87 | 77.37 | 24.13 | 23.49 |
| 360 | 74.96 | 91.88 | 94.04 | 16.92 | 2.15 | 30.16 | 58.94 | 79.99 | 28.77 | 21.05 |
| Pollutant (Dye) | Pseudo-Second-Order | ||
|---|---|---|---|
| k2 (g·mg−1·min−1) | qe (mg/g) | R2 | |
| BR (A) | 2.532 | 8.024 | 0.849 |
| BB (A) | 4.385 | 5.204 | 0.929 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Visa, M.; Cosnita, M.; Moldovan, M.; Marin, C.A.; Mihaly, M. Fly Ash Waste Recycling by Pt/TiO2 Incorporation for Industrial Dye Removal. Int. J. Environ. Res. Public Health 2021, 18, 3887. https://doi.org/10.3390/ijerph18083887
Visa M, Cosnita M, Moldovan M, Marin CA, Mihaly M. Fly Ash Waste Recycling by Pt/TiO2 Incorporation for Industrial Dye Removal. International Journal of Environmental Research and Public Health. 2021; 18(8):3887. https://doi.org/10.3390/ijerph18083887
Chicago/Turabian StyleVisa, Maria, Mihaela Cosnita, Macedon Moldovan, Cosmina Andreea Marin, and Maria Mihaly. 2021. "Fly Ash Waste Recycling by Pt/TiO2 Incorporation for Industrial Dye Removal" International Journal of Environmental Research and Public Health 18, no. 8: 3887. https://doi.org/10.3390/ijerph18083887
APA StyleVisa, M., Cosnita, M., Moldovan, M., Marin, C. A., & Mihaly, M. (2021). Fly Ash Waste Recycling by Pt/TiO2 Incorporation for Industrial Dye Removal. International Journal of Environmental Research and Public Health, 18(8), 3887. https://doi.org/10.3390/ijerph18083887









