Current Progress in Lipidomics of Marine Invertebrates
Abstract
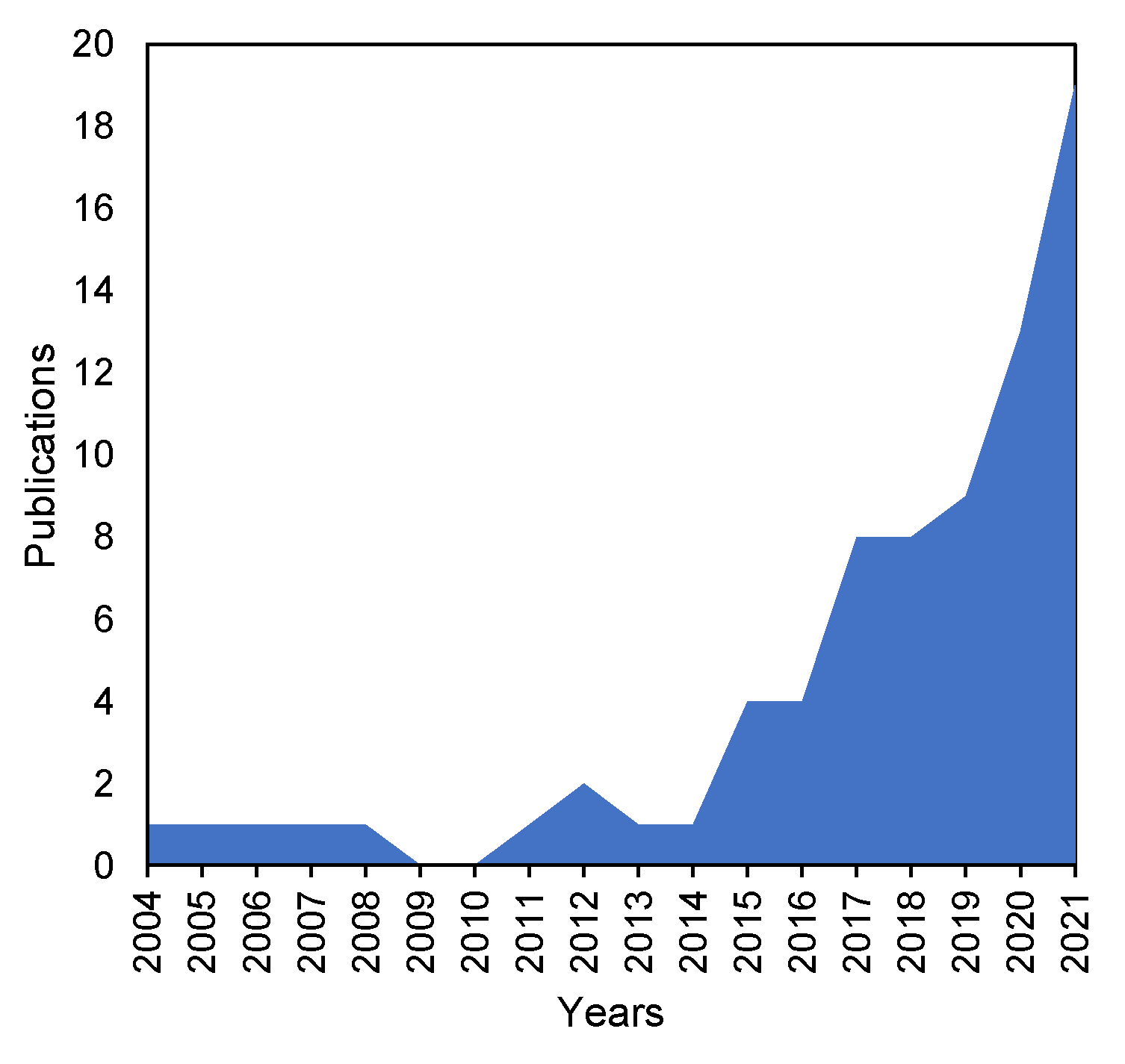
1. Introduction
2. Research Objects
3. Lipidomic Data
3.1. Mollusca
3.2. Cnidaria
3.3. Crustacea
3.4. Echinodermata
3.5. Porifera
4. Major Applications
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
Note about Andrey B. Imbs
References
- Ruppert, E.E.; Fox, R.S.; Barnes, R.D. Invertebrate Zoology: A Functional Evolutionary Approach, 7th ed.; Cengage Learning: New York, NY, USA, 2003; p. 1008. [Google Scholar]
- Wood, P. Lipidomics; Springer: New York, NY, USA, 2017; pp. 1–248. [Google Scholar]
- Han, X.; Gross, R.W. Global analyses of cellular lipidomes directly from crude extracts of biological samples by ESI mass spectrometry: A bridge to lipidomics. J. Lipid Res. 2003, 44, 1071–1079. [Google Scholar] [CrossRef]
- Sun, C.; Zhao, Y.Y.; Curtis, J.M. The direct determination of double bond positions in lipid mixtures by liquid chromatography/in-line ozonolysis/mass spectrometry. Anal. Chim. Acta 2013, 762, 68–75. [Google Scholar] [CrossRef]
- Pham, H.T.; Maccarone, A.T.; Thomas, M.C.; Campbell, J.L.; Mitchell, T.W.; Blanksby, S.J. Structural characterization of glycerophospholipids by combinations of ozone- and collision-induced dissociation mass spectrometry: The next step towards “top-down” lipidomics. Analyst 2014, 139, 204–214. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.Y.; Wang, W.X. A lipidomic approach to understand copper resilience in oyster Crassostrea hongkongensis. Aquat. Toxicol. 2018, 204, 160–170. [Google Scholar] [CrossRef]
- Chen, Q.S.; Wang, X.C.; Cong, P.X.; Liu, Y.J.; Wang, Y.M.; Xu, J.; Xue, C.H. Mechanism of phospholipid hydrolysis for oyster Crassostrea plicatula phospholipids during storage using shotgun lipidomics. Lipids 2017, 52, 1045–1058. [Google Scholar] [CrossRef]
- Le, T.T.T.; Tran, Q.T.; Velansky, P.; Dam, T.D.; Bach, L.G.; Pham, L.Q. Lipid composition and molecular species of phospholipid in oyster Crassostrea lugubris (Sowerby, 1871) from Lang Co Beach, Hue Province, Vietnam. Food Sci. Nutr. 2021, 9, 4199–4210. [Google Scholar]
- Chen, S.; Belikova, N.A.; Subbaiah, P.V. Structural elucidation of molecular species of pacific oyster ether amino phospholipids by normal-phase liquid chromatography/negative-ion electrospray ionization and quadrupole/multiple-stage linear ion-trap mass spectrometry. Anal. Chim. Acta 2012, 735, 76–89. [Google Scholar] [CrossRef][Green Version]
- Liu, Y.F.; Wu, Z.X.; Zhang, J.; Liu, Y.X.; Liu, Z.Y.; Xie, H.K.; Rakariyatham, K.; Zhou, D.Y. Seasonal variation of lipid profile of oyster Crassostrea talienwhanensis from the Yellow Sea area. J. Aquat. Food Prod. Technol. 2020, 29, 360–372. [Google Scholar] [CrossRef]
- Wang, J.; Liao, J.; Wang, H.H.; Zhu, X.F.; Li, L.Q.; Lu, W.B.; Song, G.S.; Shen, Q. Quantitative and comparative study of plasmalogen molecular species in six edible shellfishes by hydrophilic interaction chromatography mass spectrometry. Food Chem. 2021, 334, 127558. [Google Scholar] [CrossRef]
- Boselli, E.; Pacetti, D.; Lucci, P.; Frega, N.G. Characterization of phospholipid molecular species in the edible parts of bony fish and shellfish. J. Agric. Food Chem. 2012, 60, 3234–3245. [Google Scholar] [CrossRef]
- Donato, P.; Micalizzi, G.; Oteri, M.; Rigano, F.; Sciarrone, D.; Dugo, P.; Mondello, L. Comprehensive lipid profiling in the Mediterranean mussel (Mytilus galloprovincialis) using hyphenated and multidimensional chromatography techniques coupled to mass spectrometry detection. Anal. Bioanal. Chem. 2018, 410, 3297–3313. [Google Scholar] [CrossRef] [PubMed]
- Facchini, L.; Losito, I.; Cataldi, T.R.I.; Palmisano, F. Ceramide lipids in alive and thermally stressed mussels: An investigation by hydrophilic interaction liquid chromatography-electrospray ionization Fourier transform mass spectrometry. J. Mass Spectrom. 2016, 51, 768–781. [Google Scholar] [CrossRef]
- Facchini, L.; Losito, I.; Cataldi, T.R.I.; Palmisano, F. Seasonal variations in the profile of main phospholipids in Mytilus galloprovincialis mussels: A study by hydrophilic interaction liquid chromatography-electrospray ionization Fourier transform mass spectrometry. J. Mass Spectrom. 2018, 53, 1–20. [Google Scholar] [CrossRef]
- Facchini, L.; Losito, I.; Cianci, C.; Cataldi, T.R.I.; Palmisano, F. Structural characterization and profiling of lyso-phospholipids in fresh and in thermally stressed mussels by hydrophilic interaction liquid chromatography—electrospray ionization—Fourier transform mass spectrometry. Electrophoresis 2016, 37, 1823–1838. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.Y.; Liu, Y.X.; Zhou, Z.; Zhou, D.Y.; Du, M.; Zhu, B.W.; Qin, L. Improving lipidomic coverage using UPLC-ESI-Q-TOF-MS for marine shellfish by optimizing the mobile phase and resuspension solvents. J. Agric. Food Chem. 2019, 67, 8677–8688. [Google Scholar] [CrossRef] [PubMed]
- Yin, F.W.; Zhou, D.Y.; Zhao, Q.; Liu, Z.Y.; Hu, X.P.; Liu, Y.F.; Song, L.; Zhou, X.; Qin, L.; Zhu, B.W.; et al. Identification of glycerophospholipid molecular species of mussel (Mytilus edulis) lipids by high-performance liquid chromatography-electrospray ionization-tandem mass spectrometry. Food Chem. 2016, 213, 344–351. [Google Scholar] [CrossRef]
- Chen, J.N.; Huang, X.H.; Zheng, J.; Sun, Y.H.; Dong, X.P.; Zhou, D.Y.; Zhu, B.W.; Qin, L. Comprehensive metabolomic and lipidomic profiling of the seasonal variation of blue mussels (Mytilus edulis L.): Free amino acids, 5′-nucleotides, and lipids. LWT-Food Sci. Technol. 2021, 149, 111835. [Google Scholar] [CrossRef]
- Laudicella, V.A.; Beveridge, C.; Carboni, S.; Franco, S.C.; Doherty, M.K.; Long, N.; Mitchell, E.; Stanley, M.S.; Whitfield, P.D.; Hughes, A.D. Lipidomics analysis of juveniles’ blue mussels (Mytilus edulis L. 1758), a key economic and ecological species. PLoS ONE 2020, 15, e0223031. [Google Scholar] [CrossRef]
- Song, G.S.; Zhu, Q.C.; Li, L.Q.; Zheng, Z.X.; Zhao, Q.L.; Feng, J.L.; Zhang, X.D.; Wang, P.Y.; Chen, K.; Shen, Q. Lipidomics phenotyping of clam (Corbicula fluminea) through graphene/ fibrous silica nanohybrids based solid-phase extraction and HILIC-MS analysis. Food Chem. 2021, 354, 129565. [Google Scholar] [CrossRef]
- Zhang, M.N.; Song, G.S.; Zhu, Q.C.; Zhao, Q.L.; Zhang, X.D.; Hu, X.J.; Feng, J.L.; Wang, P.Y.; Shen, Q.; Wang, H.X. Compositional study of plasmalogens in clam (Corbicula fluminea) by TiO2/ KCC-1 extraction, enzymatic purification, and lipidomics analysis. J. Food Comp. Anal. 2021, 101, 103966. [Google Scholar] [CrossRef]
- Liu, Z.Y.; Zhou, D.Y.; Zhao, Q.; Yin, F.W.; Hu, X.P.; Song, L.; Qin, L.; Zhang, J.R.; Zhu, B.W.; Shahidi, F. Characterization of glycerophospholipid molecular species in six species of edible clams by high-performance liquid chromatography-electrospray ionization-tandem mass spectrometry. Food Chem. 2017, 219, 419–427. [Google Scholar] [CrossRef] [PubMed]
- Tran, Q.T.; Le, T.T.T.; Pham, M.Q.; Do, T.L.; Vu, M.H.; Nguyen, D.C.; Bach, L.G.; Bui, L.M.; Pham, Q.L. Fatty acid, lipid classes and phospholipid molecular species composition of the marine clam Meretrix lyrata (Sowerby 1851) from Cua Lo Beach, Nghe An Province, Vietnam. Molecules 2019, 24, 895. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Zhang, Y.Y.; Gao, J.X.; Ma, L.X.; Huang, X.H.; Zheng, J.; Dong, X.P.; Qin, L. Metabolomic approaches to analyze the seasonal variations of amino acid, 5 ‘-Nucleotide, and lipid profile of clam (Ruditapes philippinarum). LWT—Food Sci. Technol. 2021, 148, 111709. [Google Scholar] [CrossRef]
- Wu, Z.X.; Zhou, D.Y.; Zhao, M.T.; Xie, H.K.; Liu, Z.Y.; Liu, Y.F.; Liu, Y.X.; Shahidi, F. Lipid profile and glycerophospholipid molecular species in two species of edible razor clams Sinonovacula constricta and Solen gouldi. Lipids 2019, 54, 347–356. [Google Scholar] [CrossRef]
- Zhang, Y.Y.; Qin, L.; Liu, Y.X.; Zhou, D.Y.; Xu, X.B.; Du, M.; Zhu, B.W.; Thornton, M. Evaluation of lipid profile in different tissues of Japanese abalone Haliotis discus hannai Ino with UPLC-ESI-Q-TOF-MS-based lipidomic study. Food Chem. 2018, 265, 49–56. [Google Scholar] [CrossRef]
- Gilabert, A.; Geraudie, P.; Jaumot, J.; Porte, C. Partial characterization of the lipidome of the cold-water scallop, Chlamys islandica. Environ. Sci. Pollut. Res. 2020, 27, 1475–1484. [Google Scholar] [CrossRef] [PubMed]
- Imbs, A.B.; Chernyshev, A.V. Tracing of lipid markers of soft corals in a polar lipidome of the nudibranch mollusk Tritonia tetraquetra from the Sea of Okhotsk. Polar Biol. 2019, 42, 245–256. [Google Scholar] [CrossRef]
- Imbs, A.B.; Grigorchuk, V.P. Lipidomic study of the influence of dietary fatty acids on structural lipids of cold-water nudibranch molluscs. Sci. Rep. 2019, 9, 20013. [Google Scholar] [CrossRef]
- Rey, F.; Melo, T.; Cartaxana, P.; Calado, R.; Domingues, P.; Cruz, S.; Domingues, M.R.M. Coping with starvation: Contrasting lipidomic dynamics in the cells of two sacoglossan sea slugs incorporating stolen plastids from the same macroalga. Integr. Comp. Biol. 2020, 60, 43–56. [Google Scholar] [CrossRef] [PubMed]
- Rybin, V.G.; Imbs, A.B.; Demidkova, D.A.; Ermolenko, E.V. Identification of molecular species of monoalkyldiacylglycerol from the squid Berryteuthis magister using liquid chromatography–APCI high-resolution mass spectrometry. Chem. Phys. Lipids 2017, 202, 55–61. [Google Scholar] [CrossRef]
- Li, P.H.; Zhang, M.Q.; Xie, D.X.; Zhang, X.M.; Zhang, S.S.; Gao, F.Y.; Wang, Y.L.; Hsiao, C.D.; Li, X.B.; Liu, K.C. Characterization and bioactivities of phospholipids from squid viscera and gonads using ultra-performance liquid chromatography-Q-exactive orbitrap/mass spectrometry-based lipidomics and zebrafish models. Food Funct. 2021, 12, 7986–7996. [Google Scholar] [CrossRef]
- Li, X.B.; He, Q.X.; Hou, H.R.; Zhang, S.S.; Zhang, X.M.; Zhang, Y.; Wang, X.M.; Han, L.W.; Liu, K.C. Targeted lipidomics profiling of marine phospholipids from different resources by UPLC-Q-Exactive Orbitrap/MS approach. J. Chromatogr. B—Anal. Tech. Biomed. Life Sci. 2018, 1096, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Bosh, T.V.; Long, P.Q. A comparison of the composition of wax ester molecular species of different coral groups (Subclasses Hexacorallia and Octocorallia). Russ. J. Mar. Biol. 2017, 43, 471–478. [Google Scholar] [CrossRef]
- Ermolenko, E.V.; Sikorskaya, T.V. Lipidome of the reef-building coral Acropora cerealis: Changes under thermal stress. Biochem. Syst. Ecol. 2021, 97, 104276. [Google Scholar] [CrossRef]
- Henry, J.A.; Khattri, R.B.; Guingab-Cagmat, J.; Merritt, M.E.; Garrett, T.J.; Patterson, J.T.; Lohr, K.E. Intraspecific variation in polar and nonpolar metabolite profiles of a threatened Caribbean coral. Metabolomics 2021, 17, 60. [Google Scholar] [CrossRef]
- Sogin, E.M.; Putnam, H.M.; Anderson, P.E.; Gates, R.D. Metabolomic signatures of increases in temperature and ocean acidification from the reef-building coral, Pocillopora damicornis. Metabolomics 2016, 12, 71. [Google Scholar] [CrossRef]
- Tang, C.H.; Lin, C.Y.; Lee, S.H.; Wang, W.H. Membrane lipid profiles of coral responded to zinc oxide nanoparticle-induced perturbations on the cellular membrane. Aquat. Toxicol. 2017, 187, 72–81. [Google Scholar] [CrossRef]
- Tang, C.H.; Shi, S.H.; Lin, C.Y.; Li, H.H.; Wang, W.H. Using lipidomic methodology to characterize coral response to herbicide contamination and develop an early biomonitoring model. Sci. Total Environ. 2019, 648, 1275–1283. [Google Scholar] [CrossRef] [PubMed]
- Imbs, A.B.; Dang, L.T.P. The molecular species of phospholipids of the cold-water soft coral Gersemia rubiformis (Ehrenberg, 1834) (Alcyonacea, Nephtheidae). Russ. J. Mar. Biol. 2017, 43, 239–244. [Google Scholar] [CrossRef]
- Imbs, A.B.; Dang, L.P.T.; Rybin, V.G.; Nguyen, N.T.; Pham, L.Q. Distribution of very-long-chain fatty acids between molecular species of different phospholipid classes of two soft corals. Biochem. Anal. Biochem. 2015, 4, 205. [Google Scholar]
- Sikorskaya, T.V.; Ermolenko, E.V.; Imbs, A.B. Effect of experimental thermal stress on lipidomes of the soft coral Sinularia sp. and its symbiotic dinoflagellates. J. Exp. Mar. Biol. Ecol. 2020, 524, 151295. [Google Scholar] [CrossRef]
- Sikorskaya, T.V.; Ermolenko, E.V.; Boroda, A.V.; Ginanova, T.T. Physiological processes and lipidome dynamics in the soft coral Sinularia heterospiculata under experimental bleaching. Comp. Biochem. Physiol. B 2021, 255, 110609. [Google Scholar] [CrossRef] [PubMed]
- Sikorskaya, T.V.; Imbs, A.B. Study of total lipidome of the Sinularia siaesensis soft coral. Russ. J. Bioorg. Chem. 2018, 44, 712–723. [Google Scholar] [CrossRef]
- Imbs, A.B.; Dang, L.P.T.; Rybin, V.G.; Svetashev, V.I. Fatty acid, lipid class, and phospholipid molecular species composition of the soft coral Xenia sp. (Nha Trang Bay, the South China Sea, Vietnam). Lipids 2015, 50, 575–589. [Google Scholar] [CrossRef]
- Imbs, A.B.; Dang, L.P.T.; Nguyen, K.B. Comparative lipidomic analysis of phospholipids of hydrocorals and corals from tropical and cold-water regions. PLoS ONE 2019, 14, e0215759. [Google Scholar] [CrossRef] [PubMed]
- Imbs, A.B.; Ermolenko, E.V.; Grigorchuk, V.P.; Dang, L.T.P. Seasonal variation in the lipidome of two species of Millepora hydrocorals from Vietnam coastal waters (the South China Sea). Coral Reefs 2021, 40, 719–734. [Google Scholar] [CrossRef]
- Sikorskaya, T.V. Composition of triacylglycerides and monoalkyldiacylglycerides of the hydrocoral Millepora platyphylla. Chem. Nat. Comp. 2021, 57, 803–805. [Google Scholar] [CrossRef]
- Sikorskaya, T.V. Investigation of the total lipidoma from a zoantharia Palythoa sp. Chem. Nat. Comp. 2020, 56, 44–49. [Google Scholar] [CrossRef]
- Hou, Q.; Huang, Y.T.; Jiang, L.H.; Zhong, K.; Huang, Y.N.; Gao, H.; Bu, Q. Evaluation of lipid profiles in three species of ascidians using UPLC-ESI-Q-TOF-MS-based lipidomic study. Food Res. Int. 2021, 146, 110454. [Google Scholar] [CrossRef]
- Zhu, S.; Ye, M.W.; Xu, J.L.; Guo, C.Y.; Zheng, H.K.; Hu, J.B.; Chen, J.J.; Wang, Y.J.; Xu, S.L.; Yan, X.J. Lipid profile in different parts of edible jellyfish Rhopilema esculentum. J. Agric. Food Chem. 2015, 63, 8283–8291. [Google Scholar] [CrossRef]
- de Souza, L.M.; Iacomini, M.; Gorin, P.A.J.; Sari, R.S.; Haddad, M.A.; Sassaki, G.L. Glyco- and sphingophosphonolipids from the medusa Phyllorhiza punctata: NMR and ESI-MS/MS fingerprints. Chem. Phys. Lipids 2007, 145, 85–96. [Google Scholar] [CrossRef]
- Garrett, T.A.; Hwang, J.; Schmeitzel, J.L.; Schwarz, J. Lipidomics of Aiptasia pallida and Symbiodinium: A model system for investigating the molecular basis of coral symbiosis. FASEB J. 2011, 25, 9382. [Google Scholar] [CrossRef]
- Garrett, T.A.; Schmeitzel, J.L.; Klein, J.A.; Hwang, J.J.; Schwarz, J.A. Comparative lipid profiling of the cnidarian Aiptasia pallida and its dinoflagellate symbiont. PLoS ONE 2013, 8, e57975. [Google Scholar] [CrossRef] [PubMed]
- Trigg, S.A.; McElhany, P.; Maher, M.; Perez, D.; Busch, D.S.; Nichols, K.M. Uncovering mechanisms of global ocean change effects on the Dungeness crab (Cancer magister) through metabolomics analysis. Sci. Rep. 2019, 9, 10717. [Google Scholar] [CrossRef] [PubMed]
- Rey, F.; Alves, E.; Domingues, P.; Domingues, M.R.M.; Calado, R. A lipidomic perspective on the embryogenesis of two commercially important crabs, Carcinus maenas and Necora puber. Bull. Mar. Sci. 2018, 94, 1395–1411. [Google Scholar] [CrossRef]
- Rey, F.; Alves, E.; Melo, T.; Domingues, P.; Queiroga, H.; Rosa, R.; Domingues, M.R.M.; Calado, R. Unravelling polar lipids dynamics during embryonic development of two sympatric brachyuran crabs (Carcinus maenas and Necora puber) using lipidomics. Sci. Rep. 2015, 5, 14549. [Google Scholar] [CrossRef]
- Ding, Z.F. Lipid metabolism disorders contribute to the pathogenesis of Hepatospora eriocheir in the crab Eriocheir sinensis. J. Fish Dis. 2021, 44, 305–313. [Google Scholar] [CrossRef]
- Xu, C.; Ma, Q.Q.; Li, E.C.; Liu, Y.; Wang, X.D.; Du, Z.Y.; Qin, J.G.; Chen, L.Q. Response of lipid molecular structure to dietary lipid type in Chinese mitten crab Eriocheir sinensis: A deep lipidomics analysis. Aquacult. Rep. 2021, 19, 100596. [Google Scholar] [CrossRef]
- Lu, T.; Shen, Y.; Cui, G.X.; Yin, F.W.; Yu, Z.L.; Zhou, D.Y. Detailed analysis of lipids in edible viscera and muscles of cooked crabs Portunus trituberculatus and Portunus pelagicus. J. Aquat. Food Prod. Technol. 2020, 29, 391–406. [Google Scholar] [CrossRef]
- Yuan, Y.; Xu, F.M.; Jin, M.; Wang, X.X.; Hu, X.Y.; Zhao, M.M.; Cheng, X.; Luo, J.X.; Jiao, L.F.; Betancor, M.B.; et al. Untargeted lipidomics reveals metabolic responses to different dietary n-3 PUFA in juvenile swimming crab (Portunus trituberculatus). Food Chem. 2021, 354, 129570. [Google Scholar] [CrossRef]
- Huang, M.X.; Dong, Y.F.; Zhang, Y.; Chen, Q.S.; Xie, J.; Xu, C.; Zhao, Q.; Li, E.C. Growth and lipidomic responses of juvenile pacific white shrimp Litopenaeus vannamei to low salinity. Front. Physiol. 2019, 10, 1087. [Google Scholar] [CrossRef] [PubMed]
- Lavarias, S.; Dreon, M.S.; Pollero, R.J.; Heras, H. Changes in phosphatidylcholine molecular species in the shrimp Macrobrachium borellii in response to a water-soluble fraction of petroleum. Lipids 2005, 40, 487–494. [Google Scholar] [CrossRef] [PubMed]
- Shillito, B.; Desurmont, C.; Barthelemy, D.; Farabos, D.; Despres, G.; Ravaux, J.; Zbinden, M.; Lamaziere, A. Lipidome variations of deep-sea vent shrimps according to acclimation pressure: A homeoviscous response? Deep-Sea Res. Part I—Oceanogr. Res. Pap. 2020, 161, 103285. [Google Scholar] [CrossRef]
- Miniadis-Meimaroglou, S.; Kora, L.; Sinanogiou, V.J. Isolation and identification of phospholipid molecular species in a wild marine shrimp Penaeus kerathurus muscle and cephalothorax. Chem. Phys. Lipids 2008, 152, 104–112. [Google Scholar] [CrossRef] [PubMed]
- Miniadis-Meimaroglou, S.; Tsapes, K.; Sinanoglou, V.; Kapsalis, A. Main phospholipid classes and their molecular species in shrimp and lobster cephalothorax. Chem. Phys. Lipids 2004, 130, 45. [Google Scholar]
- Yu, Z.L.; Li, D.Y.; Yin, F.W.; Zhao, Q.; Liu, Z.Y.; Song, L.; Zhou, D.Y.; Wang, T. Lipid Profiles in By-Products and Muscles of Three Shrimp Species (Penaeus monodon, Penaeus vannamei, and Penaeus chinensis). Eur. J. Lipid Sci. Tech. 2020, 122, 1900309. [Google Scholar] [CrossRef]
- Garofalaki, T.F.; Miniadis-Meimaroglou, S.; Sinanoglou, V.J. Main phospholipids and their fatty acid composition in muscle and cephalothorax of the edible Mediterranean crustacean Palinurus vulgaris (spiny lobster). Chem. Phys. Lipids 2006, 140, 55–65. [Google Scholar] [CrossRef] [PubMed]
- Hansen, B.H.; Sorensen, L.; Storseth, T.R.; Altin, D.; Gonzalez, S.V.; Skancke, J.; Ronsberg, M.U.; Nordtug, T. The use of PAH, metabolite and lipid profiling to assess exposure and effects of produced water discharges on pelagic copepods. Sci. Total Environ. 2020, 714, 136674. [Google Scholar] [CrossRef]
- Kostetsky, E.Y.; Sanina, N.M.; Velansky, P.V. The thermotropic behavior and major molecular species composition of the phospholipids of echinoderms. Russ. J. Mar. Biol. 2014, 40, 131–139. [Google Scholar] [CrossRef]
- Omran, N.E.; Salem, H.K.; Eissa, S.H.; Kabbash, A.M.; Kandeil, M.A.; Salem, M.A. Chemotaxonomic study of the most abundant Egyptian sea-cucumbers using ultra-performance liquid chromatography (UPLC) coupled to high-resolution mass spectrometry (HRMS). Chemoecology 2020, 30, 35–48. [Google Scholar] [CrossRef]
- Wang, X.C.; Cong, P.X.; Chen, Q.S.; Li, Z.J.; Xu, J.; Xue, C.H. Characterizing the phospholipid composition of six edible sea cucumbers by NPLC-Triple TOF-MS/MS. J. Food Comp. Anal. 2020, 94, 103626. [Google Scholar] [CrossRef]
- Gold, D.A.; O’Reilly, S.S.; Watson, J.; Degnan, B.M.; Degnan, S.M.; Kromer, J.O.; Summons, R.E. Lipidomics of the sea sponge Amphimedon queenslandica and implication for biomarker geochemistry. Geobiology 2017, 15, 836–843. [Google Scholar] [CrossRef]
- Rosset, S.; Koster, G.; Brandsma, J.; Hunt, A.N.; Postle, A.D.; D’Angelo, C. Lipidome analysis of Symbiodiniaceae reveals possible mechanisms of heat stress tolerance in reef coral symbionts. Coral Reefs 2019, 38, 1241–1253. [Google Scholar] [CrossRef]
- Imbs, A.B.; Rybin, V.G.; Kharlamenko, V.I.; Dang, L.P.T.; Nguyen, N.T.; Pham, K.M.; Pham, L.Q. Polyunsaturated molecular species of galactolipids: Markers of zooxanthellae in a symbiotic association of the soft coral Capnella sp. (Anthozoa: Alcyonacea). Russ. J. Mar. Biol. 2015, 41, 461–467. [Google Scholar] [CrossRef]
- Rosset, S.L.; Oakley, C.A.; Ferrier-Pages, C.; Suggett, D.J.; Weis, V.M.; Davy, S.K. The molecular language of the cnidarian-dinoflagellate symbiosis. Trends Microbiol. 2021, 29, 320–333. [Google Scholar] [CrossRef]
- Schmeitzel, J.L.; Klein, J.; Smith, M.; Schwarz, J.; Garrett, T.A. Comparative lipidomic analysis of the symbiosis between Aiptasia pallida and Symbiodinium. FASEB J. 2012, 26, 7891. [Google Scholar] [CrossRef]
- Sikorskaya, T.V.; Efimova, K.V.; Imbs, A.B. Lipidomes of phylogenetically different symbiotic dinoflagellates of corals. Phytochemistry 2021, 181, 112579. [Google Scholar] [CrossRef]
- Imbs, A.B.; Latyshev, N.A.; Dautova, T.N.; Latypov, Y.Y. Distribution of lipids and fatty acids in corals by their taxonomic position and presence of zooxanthellae. Mar. Ecol. Prog. Ser. 2010, 409, 65–75. [Google Scholar] [CrossRef]
- Svetashev, V.I. Fatty acids of the medusae Aurelia aurita (Linnaeus, 1758) and Rhopilema esculentum (Kishinouye, 1891): The presence of families of polyenoic acids with 24 and 26 carbon atoms. Russ. J. Mar. Biol. 2019, 45, 113–117. [Google Scholar] [CrossRef]
- Vysotskii, M.V.; Svetashev, V.I. Identification, isolation and characterization of tetracosapolyenoic acids in lipids of marine coelenterates. Biochim. Biophys. Acta 1991, 1083, 161–165. [Google Scholar] [CrossRef]
- Imbs, A.B.; Demidkova, D.A.; Dautova, T.N. Lipids and fatty acids of cold-water soft corals and hydrocorals: A comparison with tropical species and implications for coral nutrition. Mar. Biol. 2016, 163, 202. [Google Scholar] [CrossRef]
- Bishop, D.G.; Kenrick, J.R. Fatty acid composition of symbiotic zooxanthellae in relation to their hosts. Lipids 1980, 15, 799–804. [Google Scholar] [CrossRef]
- Imbs, A.B.; Ermolenko, E.V.; Grigorchuk, V.P.; Dang, L.P.T. A lipidomic approach to the study of biodiversity of symbiotic dinoflagellates in Millepora hydrocorals from Vietnam coral reefs. Russ. J. Mar. Biol. 2021, 47, 312–317. [Google Scholar] [CrossRef]
- Latyshev, N.A.; Kasyanov, S.P.; Kharlamenko, V.I.; Svetashev, V.I. Lipids and of fatty acids of edible crabs of the north-western Pacific. Food Chem. 2009, 116, 657–661. [Google Scholar] [CrossRef]
- Folch, J.; Lees, M.; Sloane Stanley, G.H. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [CrossRef]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef] [PubMed]
| Phylum or Subphylum | Animal or Tissues | Species Name | Total Molecular Species Identified | Reference |
|---|---|---|---|---|
| Mollusca | oyster, digestive glands | Crassostrea hongkongensis | 1234 TL * | [6] |
| oyster | C. plicatula | 94 PL | [7] | |
| oyster | C. lugubris | 90 PL | [8] | |
| oyster | C. sikamea | 18 PS, 32 PE | [9] | |
| oyster | C. talienwhanensis | 290 PL | [10] | |
| oyster | C. gigas (=Ostrea gigas) | 59 PL, 18 PS, 32 PE | [9,11] | |
| oyster, edible foot | Ostrea edulis | 62 PL | [12] | |
| mussel | Mytilus galloprovincialis | 185 PC, 131 PE | [13,14,15,16] | |
| mussel, edible foot | M. galloprovincialis | 29 PE, 4 PI, 5 PS, 15 PC | [12] | |
| mussel | M. edulis | 230 PL, 413 TL | [11,17,18,19] | |
| juvenile blue mussels | M. edulis | 343 TL | [20] | |
| clam | Corbicula fluminea | 35 PL | [21,22] | |
| clam | Cyclina sinensis | 435 PL | [23] | |
| clam | Mactra chinensis | 453 PL | [23] | |
| clam | M. veneriformis | 468 PL | [23] | |
| clam | Meretrix lyrata | 96 PL | [24] | |
| clams | M. meretrix | 443 PL | [23] | |
| clam | Ruditapes philippinarum | 427 PL | [17,23,25] | |
| clam | Saxidomus purpurata | 444 PL | [23] | |
| clam | Sinonovacula constricta | 238 PL | [11,26] | |
| clam | Solen gouldi | 235 PL | [26] | |
| clam | Venerupis variegata | 19 PlsGPL | [11] | |
| clam, edible foot | Venus gallina | 88 PL | [12] | |
| abalone | Haliotis rubra | 19 PlsGPL | [11] | |
| abalone | H. discus hannai | 125 TL | [17,27] | |
| scallop, digestive glands | Chlamys islandica | 224 TL | [28] | |
| scallop | Patinopecten yessoensis | 125 TL | [17] | |
| scallop | Placopecta magellanicus | 19 PlsGPL | [11] | |
| nudibranch | Tritonia tetraquetra | 90 PL | [29,30] | |
| nudibranch | Dendronotus sp. | 90 PL | [30] | |
| nudibranch | D. robustus | 90 PL | [30] | |
| sacoglossan sea slug | Elysia viridis | 322 PL, 47 GL | [31] | |
| sacoglossan sea slug | Placida dendritica | 318 PL, 14 GL | [31] | |
| squid, digestive glands | Berryteuthis magister | 90 DAGE | [32] | |
| squid, viscera and gonads | - | 1223 PL | [33,34] | |
| Cnidaria | reef-building coral | Acropora acuminate | 83 WE | [35] |
| reef-building coral | A. cerealis | 79 TL | [36] | |
| reef-building coral | A. cervicornis | 922 non-polar metabolites | [37] | |
| reef-building coral | A. acuminata | 83 WE | [35] | |
| reef-building coral | A. cytherea | 83 WE | [35] | |
| reef-building coral | Pocillopora damicornis | 450 TL | [38] | |
| reef-building coral | Seriatopora caliendrum | 22 PC | [39,40] | |
| reef-building coral | Turbinaria peltata | 83 WE | [35] | |
| soft coral | Mopsella spinosa | 83 WE | [35] | |
| soft coral | Menella flora | 83 WE | [35] | |
| soft coral | M. praelonga | 83 WE | [35] | |
| soft coral | Gersemia rubiformis | 68 GPL | [41] | |
| soft coral | Capnella sp. | 32 GPL | [42] | |
| soft coral | Cladiella laciniosa | 83 WE | [35] | |
| soft coral | Sinularia sp. | 170 TL | [43] | |
| soft coral | S. brassica | 83 WE | [35] | |
| soft coral | S. heterospiculata | 170 TL | [44] | |
| soft corals | S. macropodia | 32 GPL | [42] | |
| soft coral | S. robusta | 83 WE | [35] | |
| soft coral | S. siaesensis | 144 TL | [45] | |
| soft coral | Xenia sp. | 32 PL | [46] | |
| hydrocoral | Millepora dichotoma | 152 PL | [47,48] | |
| hydrocoral | M. platyphylla | 179 TL | [47,48,49] | |
| hydrocoral | Allopora steinegeri | 127 PL | [47] | |
| zoantharian | Palythoa sp. | 145 TL | [50] | |
| ascidian | Ciona intestinalis | 245 TL | [51] | |
| ascidian | Halocynthia roretzi | 245 TL | [51] | |
| ascidian | Styela clava | 245 TL | [51] | |
| jellyfish | Rhopilema esculentum | 75 PL, 12 TG | [52] | |
| jellyfish | Phyllorhiza punctata | 12 PL | [53] | |
| sea anemone | Aiptasia pallida | 109 PL | [54,55] | |
| Crustacea | crab | Cancer magister | 195 TL | [56] |
| crab embryos | Carcinus maenas | 98 PL, 16 TG | [57,58] | |
| crab hepatopancreas | Eriocheir sinensis | 67 TL | [59,60] | |
| crab embryos | Necora puber | 98 PL, 16 TG | [57,58] | |
| crab, viscera and muscles | Portunus trituberculatus | 250 PL | [61] | |
| crab, viscera and muscles | Portunus pelagicus | 250 PL | [61] | |
| crab hepatopancreas | P. trituberculatus | 541 TG, 313 PC, 153 Cer, 147 PE | [62] | |
| juvenile shrimp | Litopenaeus vannamei | 196 TL | [63] | |
| shrimp | Macrobrachium borellii | 15 PC | [64] | |
| shrimp head | - | [34] | ||
| shrimp | Mirocaris fortunata | 44 PE, 44 PC, 15 SM | [65] | |
| shrimp, muscle and cephalothorax | Penaeus kerathurus | FA of PL | [66,67] | |
| shrimp, head and body carapace | P. monodon, P. vannamei, P. chinensis | 200 PL | [68] | |
| lobster, muscle and cephalothorax | Palinurus vulgaris | 40 PE and PC | [69] | |
| copepods | Calanus sp. | 99 WE, 233 TG * | [70] | |
| Echinodermata | sea cucumber | Apostichopus japonicus | 29 PE, 26 PC | [71] |
| sea cucumber | Actinopyga mauritiana | 45 TG, 15 FFA | [72] | |
| sea cucumber | Bohadschia marmorata | 45 TG, 295 PL | [72,73] | |
| sea cucumber | Cucumaria frondosa japonica | 295 PL | [71,73] | |
| sea cucumber | Eupentacta frau datrix | 29 PE, 26 PC | [71] | |
| sea cucumber | Isostichopus fuscus | 295 PL | [73] | |
| sea cucumber | Holothuria atra | 45 TG, 15 FFA | [72] | |
| sea cucumber | H. edulis | 45 TG, 15 FFA | [72] | |
| sea cucumber | H. leucospilota | 45 TG, 15 FFA | [72] | |
| sea cucumber | H. mexicana | 295 PL | [73] | |
| sea cucumber | H. polii | 45 TG | [72,73] | |
| sea cucumber | Parastichopus califormicus | 295 PL | [73] | |
| starfish | Distolasterias nipon | 29 PE, 26 PC | [71] | |
| starfish | Asterias amurensis | 29 PE, 26 PC | [71] | |
| sea urchin | Strongylocentrotus intermedius | 29 PE, 26 PC | [71] | |
| Porifera | sea sponge | Amphimedon queenslandica | 37 FA | [74] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Imbs, A.B.; Ermolenko, E.V.; Grigorchuk, V.P.; Sikorskaya, T.V.; Velansky, P.V. Current Progress in Lipidomics of Marine Invertebrates. Mar. Drugs 2021, 19, 660. https://doi.org/10.3390/md19120660
Imbs AB, Ermolenko EV, Grigorchuk VP, Sikorskaya TV, Velansky PV. Current Progress in Lipidomics of Marine Invertebrates. Marine Drugs. 2021; 19(12):660. https://doi.org/10.3390/md19120660
Chicago/Turabian StyleImbs, Andrey B., Ekaterina V. Ermolenko, Valeria P. Grigorchuk, Tatiana V. Sikorskaya, and Peter V. Velansky. 2021. "Current Progress in Lipidomics of Marine Invertebrates" Marine Drugs 19, no. 12: 660. https://doi.org/10.3390/md19120660
APA StyleImbs, A. B., Ermolenko, E. V., Grigorchuk, V. P., Sikorskaya, T. V., & Velansky, P. V. (2021). Current Progress in Lipidomics of Marine Invertebrates. Marine Drugs, 19(12), 660. https://doi.org/10.3390/md19120660






