Anticoagulant Activity of Sulfated Ulvan Isolated from the Green Macroalga Ulva rigida
Abstract
1. Introduction
2. Results and Discussion
2.1. Extraction, Purification, and Sulfation of Ulvan from U. rigida
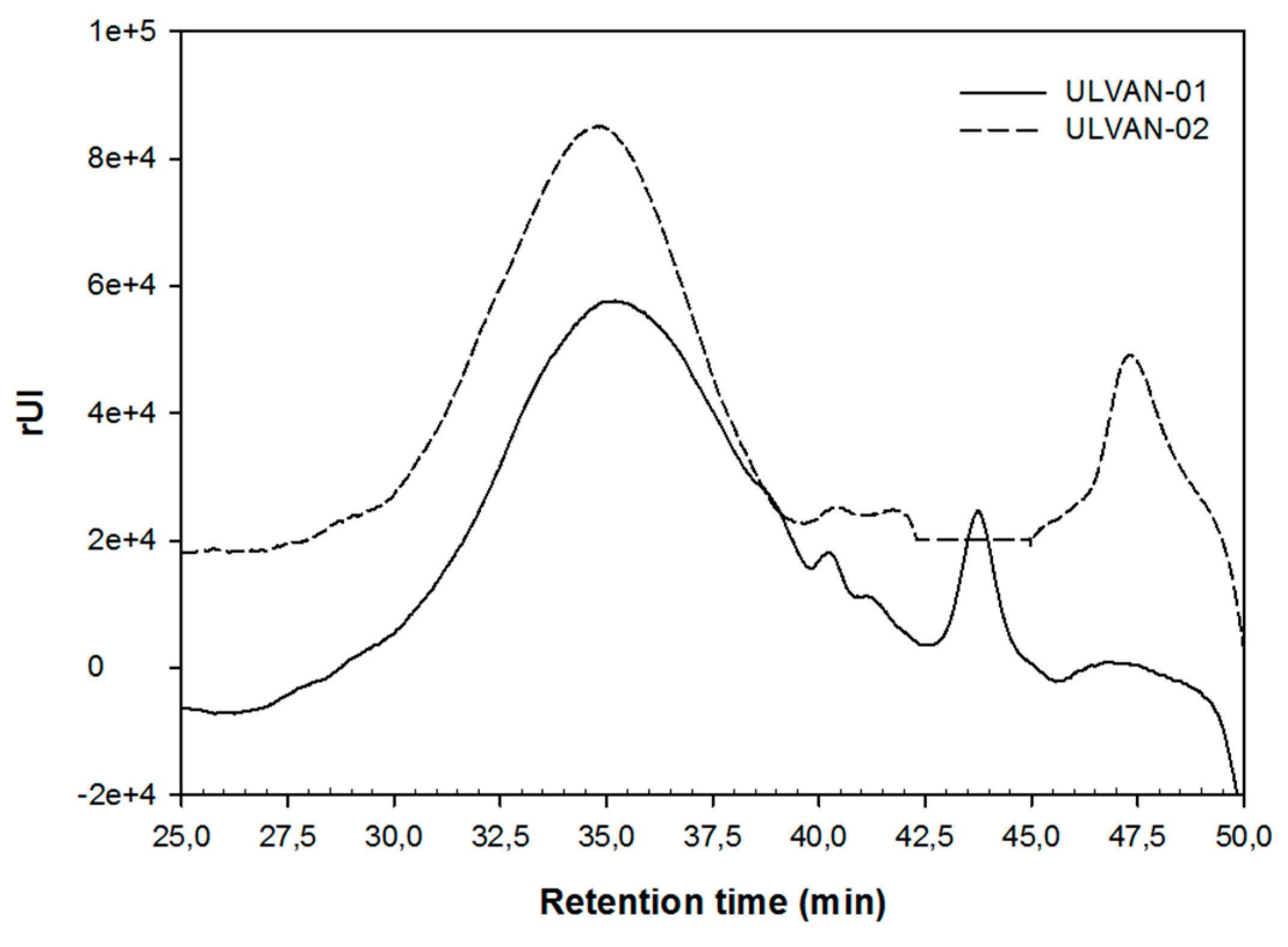
2.2. Chemical Characterization of the ULVAN-01 and ULVAN-02 Fractions
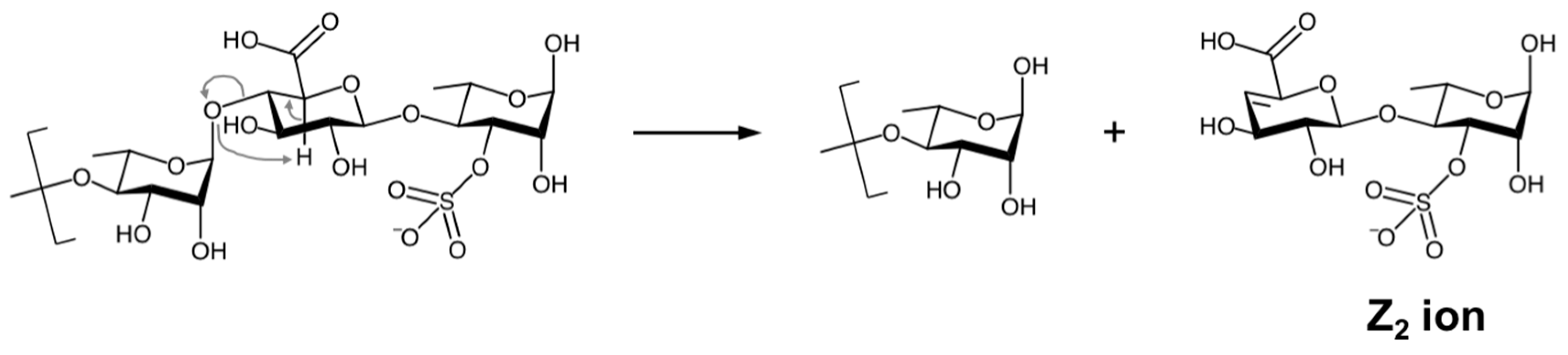
2.3. Structural Characterization of the ULVAN-01 and ULVAN-02 Fractions
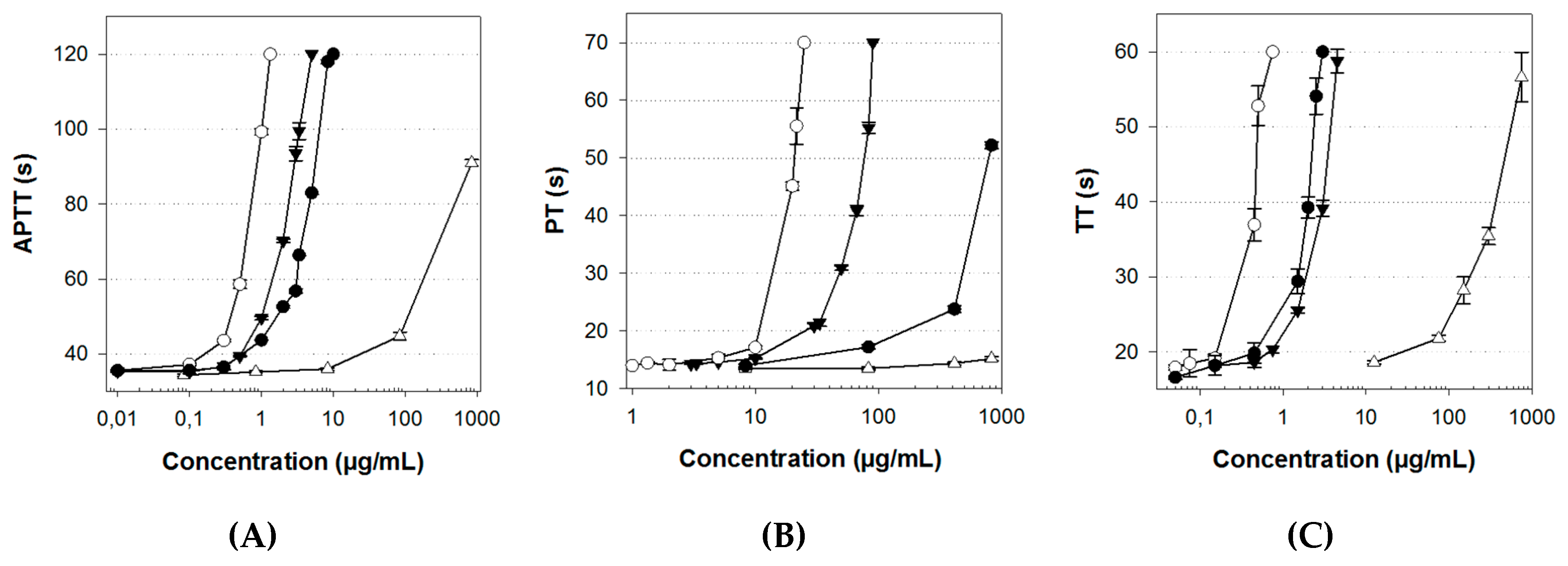
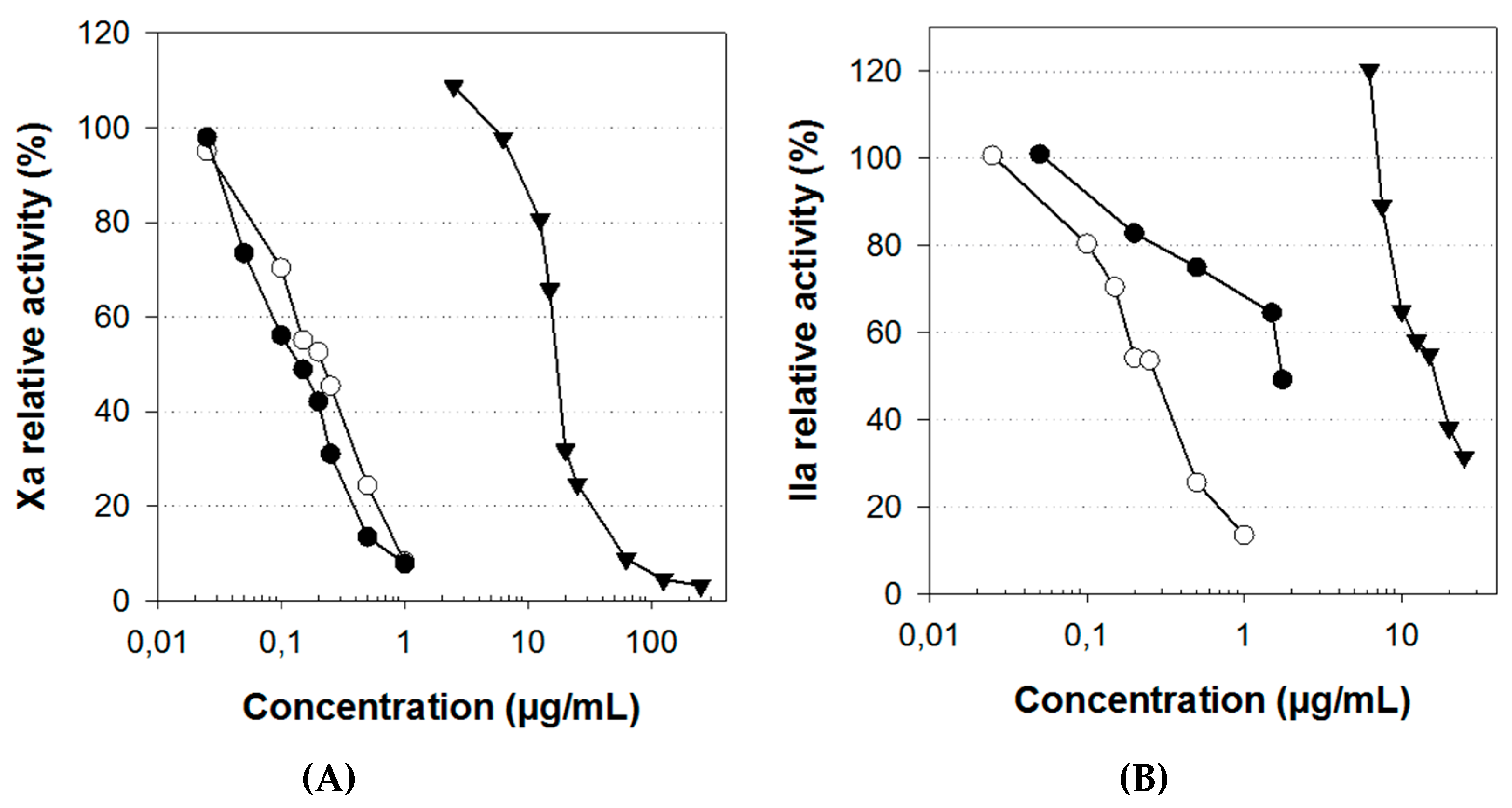
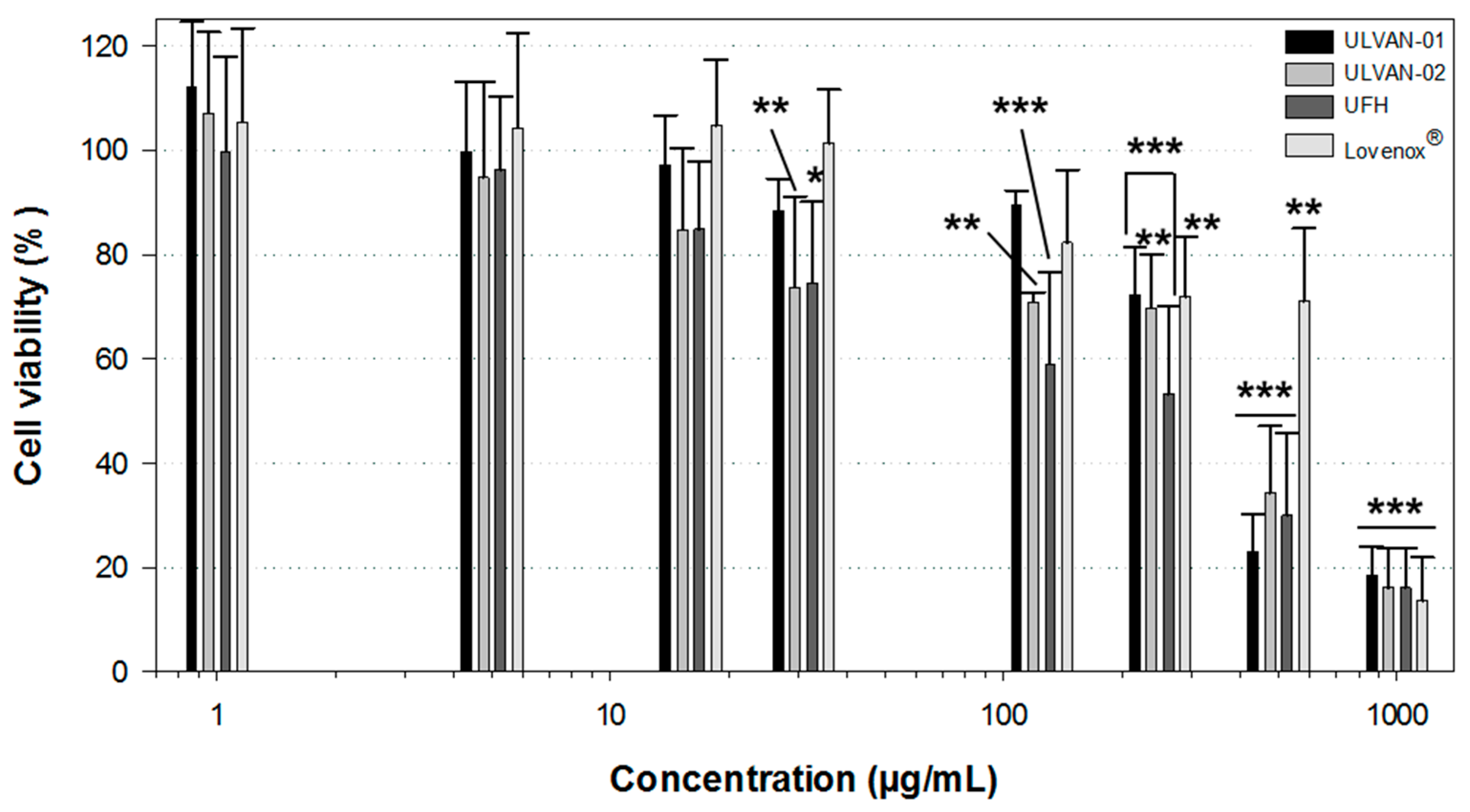
2.4. Anticoagulant Activity
2.5. Effect of ULVAN-01 and ULVAN-02 on Cell Viability
3. Materials and Methods
3.1. Materials
3.2. Methods
3.2.1. Extraction and Purification Process
3.2.2. Sulfation Procedure
3.2.3. Chemical Composition
3.2.4. Analysis of the Fractions by High Performance Size Exclusion Chromatography
- Mn = (Σ Ni × Mi)/Σ Ni
- Mw = (Σ Ni × Mi2)/(Σ Ni × Mi)
- I = Mw/Mn
3.2.5. Nuclear Magnetic Resonance (NMR) and Ultra High Performance Liquid Chromatography-High Resolution Mass Spectrometry (UHPLC-HRMS)
3.2.6. Clotting Time Assays
3.2.7. Assays of Antithrombin-Mediated Inhibition of Factors Xa and IIa
3.2.8. Evaluation of Cell Viability
3.2.9. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- McHugh, D.J. A Guide to the Seaweed Industry; FAO Fisheries Technical Paper; Food and Agriculture Organization of the United Nations: Rome, Italy, 2003; ISBN 978-92-5-104958-7. [Google Scholar]
- Calumpong, H.P.; West, J.; Martin, G. Seaweeds. In The First Global Integrated Marine Assessment: World Ocean Assessment I; United Nations, Ed.; Cambridge University Press: Cambridge, UK, 2017; ISBN 978-1-316-51001-8. [Google Scholar]
- Ferdouse, F.; Lovestad Holdt, S.; Smith, R.; Murua, P.; Yang, Z. The Global Status of Seaweed Production, Trade and Utilization; FAO Globefish Research Programme; Food and Agriculture Organization of the United Nations: Rome, Italy, 2018; ISBN 978-92-5-130870-7. [Google Scholar]
- Pomponi, S.A. The bioprocess–technological potential of the sea. J. Biotechnol. 1999, 70, 5–13. [Google Scholar] [CrossRef]
- Sasi, N.; Kriston, B. Current status of global cultivated seaweed production and markets. World Aquac. 2014, 45, 32–37. [Google Scholar]
- Granert, C.; Raud, J.; Xie, X.; Lindquist, L.; Lindbom, L. Inhibition of leukocyte rolling with polysaccharide fucoidin prevents pleocytosis in experimental meningitis in the rabbit. J. Clin. Investig. 1994, 93, 929–936. [Google Scholar] [CrossRef] [PubMed]
- Jiao, G.; Yu, G.; Zhang, J.; Ewart, H. Chemical Structures and Bioactivities of Sulfated Polysaccharides from Marine Algae. Mar. Drugs 2011, 9, 196–223. [Google Scholar] [CrossRef]
- Mohamed, S.; Hashim, S.N.; Rahman, H.A. Seaweeds: A sustainable functional food for complementary and alternative therapy. Trends Food Sci. Technol. 2012, 23, 83–96. [Google Scholar] [CrossRef]
- Rupérez, P.; Ahrazem, O.; Leal, J.A. Potential antioxidant capacity of sulfated polysaccharides from the edible marine brown seaweed Fucus vesiculosus. J. Agric. Food Chem. 2002, 50, 840–845. [Google Scholar] [CrossRef] [PubMed]
- Rocha de Souza, M.C.; Marques, C.T.; Guerra Dore, C.M.; Ferreira da Silva, F.R.; Oliveira Rocha, H.A.; Leite, E.L. Antioxidant activities of sulfated polysaccharides from brown and red seaweeds. J. Appl. Phycol. 2007, 19, 153–160. [Google Scholar] [CrossRef]
- Noda, H.; Amano, H.; Arashima, K.; Nisizawa, K. Antitumor activity of marine algae. Hydrobiologia 1990, 204, 577–584. [Google Scholar] [CrossRef]
- Bourgougnon, N.; Roussakis, C.; Kornprobst, J.-M.; Lahaye, M. Effects in vitro of sulfated polysaccharide from Schizymenia dubyi (Rhodophyta, Gigartinales) on a non-small-cell bronchopulmonary carcinoma line (NSCLC-N6). Cancer Lett. 1994, 85, 87–92. [Google Scholar] [CrossRef]
- Zorofchian Moghadamtousi, S.; Karimian, H.; Khanabdali, R.; Razavi, M.; Firoozinia, M.; Zandi, K.; Abdul Kadir, H. Anticancer and Antitumor Potential of Fucoidan and Fucoxanthin, Two Main Metabolites Isolated from Brown Algae. Sci. World J. 2014, 2014, 1–10. [Google Scholar] [CrossRef]
- Damonte, E.B.; Matulewicz, M.C.; Cerezo, A.S. Sulfated seaweed polysaccharides as antiviral agents. Curr. Med. Chem. 2004, 11, 2399–2419. [Google Scholar] [CrossRef] [PubMed]
- Pujol, C.A.; Ray, S.; Ray, B.; Damonte, E.B. Antiviral activity against dengue virus of diverse classes of algal sulfated polysaccharides. Int. J. Biol. Macromol. 2012, 51, 412–416. [Google Scholar] [CrossRef] [PubMed]
- Witvrouw, M.; De Clercq, E. Sulfated polysaccharides extracted from sea algae as potential antiviral drugs. Gen. Pharmacol. 1997, 29, 497–511. [Google Scholar] [CrossRef]
- Jin, W.; Zhang, Q.; Wang, J.; Zhang, W. A comparative study of the anticoagulant activities of eleven fucoidans. Carbohydr. Polym. 2013, 91, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Pereira, M.S.; Mulloy, B.; Mourão, P.A.S. Structure and anticoagulant activity of sulfated fucans. Comparison between the regular, repetitive, and linear fucans from echinoderms with the more heterogeneous and branched polymers from brown algae. J. Biol. Chem. 1999, 274, 7656–7667. [Google Scholar] [CrossRef]
- Carlucci, M.J.; Pujol, C.A.; Ciancia, M.; Noseda, M.D.; Matulewicz, M.C.; Damonte, E.B.; Cerezo, A.S. Antiherpetic and anticoagulant properties of carrageenans from the red seaweed Gigartina skottsbergii and their cyclized derivatives: correlation between structure and biological activity. Int. J. Biol. Macromol. 1997, 20, 97–105. [Google Scholar] [CrossRef]
- Chevolot, L.; Mulloy, B.; Ratiskol, J.; Foucault, A.; Colliec-Jouault, S. A disaccharide repeat unit is the major structure in fucoidans from two species of brown algae. Carbohydr. Res. 2001, 330, 529–535. [Google Scholar] [CrossRef]
- Percival, E.; McDowell, R.H. Chemistry and Enzymology of Marine Algal Polysaccharides; Academic Press: London, UK; New York, NY, USA, 1967; ISBN 9780125506502. [Google Scholar]
- Lahaye, M.; Robic, A. Structure and functional properties of ulvan, a polysaccharide from green seaweeds. Biomacromolecules 2007, 8, 1765–1774. [Google Scholar] [CrossRef]
- Sommers, C.D.; Ye, H.; Kolinski, R.E.; Nasr, M.; Buhse, L.F.; Al-Hakim, A.; Keire, D.A. Characterization of currently marketed heparin products: analysis of molecular weight and heparinase-I digest patterns. Anal. Bioanal. Chem. 2011, 401, 2445–2454. [Google Scholar] [CrossRef]
- Mulloy, B.; Hogwood, J.; Gray, E. Assays and reference materials for current and future applications of heparins. Biologicals 2010, 38, 459–466. [Google Scholar] [CrossRef]
- Warkentin, T.E.; Levine, M.N.; Hirsh, J.; Horsewood, P.; Roberts, R.S.; Gent, M.; Kelton, J.G. Heparin-Induced Thrombocytopenia in Patients Treated with Low-Molecular-Weight Heparin or Unfractionated Heparin. N. Engl. J. Med. 1995, 332, 1330–1336. [Google Scholar] [CrossRef]
- Da Silva, M.S.; Sobel, M. Anticoagulants: To bleed or not to bleed, that is the question. Semin. Vasc. Surg. 2002, 15, 256–267. [Google Scholar] [CrossRef]
- Emanuele, R.M.; Fareed, J. The effect of molecular weight on the bioavailability of heparin. Thromb. Res. 1987, 48, 591–596. [Google Scholar] [CrossRef]
- Hao, L.; Zhang, Q.; Yu, T.; Cheng, Y.; Ji, S. Antagonistic effects of ultra-low-molecular-weight heparin on Aβ25–35-induced apoptosis in cultured rat cortical neurons. Brain Res. 2011, 1368, 1–10. [Google Scholar] [CrossRef]
- Sattari, M.; Lowenthal, D.T. Novel oral anticoagulants in development: Dabigatran, Rivaroxaban, and Apixaban. Am. J. Ther. 2011, 18, 332–338. [Google Scholar] [CrossRef] [PubMed]
- van der Wall, S.J.; Klok, F.A.; den Exter, P.L.; Barrios, D.; Morillo, R.; Cannegieter, S.C.; Jimenez, D.; Huisman, M.V. Continuation of low-molecular-weight heparin treatment for cancer-related venous thromboembolism: a prospective cohort study in daily clinical practice. J. Thromb. Haemost. 2017, 15, 74–79. [Google Scholar] [CrossRef]
- Jurd, K.M.; Rogers, D.J.; Blunden, G.; McLellan, D.S. Anticoagulant properties of sulphated polysaccharides and a proteoglycan from Codium fragile ssp. atlanticum. J. Appl. Phycol. 1995, 7, 339–345. [Google Scholar] [CrossRef]
- Siddhanta, A.K.; Shanmugam, M.; Mody, K.H.; Goswami, A.M.; Ramavat, B.K. Sulphated polysaccharides of Codium dwarkense Boergs. from the west coast of India: chemical composition and blood anticoagulant activity. Int. J. Biol. Macromol. 1999, 26, 151–154. [Google Scholar] [CrossRef]
- Matsubara, K.; Matsuura, Y.; Bacic, A.; Liao, M.-L.; Hori, K.; Miyazawa, K. Anticoagulant properties of a sulfated galactan preparation from a marine green alga, Codium cylindricum. Int. J. Biol. Macromol. 2001, 28, 395–399. [Google Scholar] [CrossRef]
- Shanmugam, M.; Mody, K.H.; Siddhanta, A.K. Blood anticoagulant sulphated polysaccharides of the marine green algae Codium dwarkense (Boergs.) and C. tomentosum (Huds.) Stackh. Indian J. Exp. Biol. 2001, 39, 365–370. [Google Scholar]
- Hayakawa, Y.; Hayashi, T.; Lee, J.-B.; Srisomporn, P.; Maeda, M.; Ozawa, T.; Sakuragawa, N. Inhibition of thrombin by sulfated polysaccharides isolated from green algae. Biochim. Biophys. Acta BBA - Protein Struct. Mol. Enzymol. 2000, 1543, 86–94. [Google Scholar] [CrossRef]
- Ciancia, M.; Quintana, I.; Vizcargüénaga, M.I.; Kasulin, L.; de Dios, A.; Estevez, J.M.; Cerezo, A.S. Polysaccharides from the green seaweeds Codium fragile and C. vermilara with controversial effects on hemostasis. Int. J. Biol. Macromol. 2007, 41, 641–649. [Google Scholar] [CrossRef]
- Maeda, M.; Uehara, T.; Harada, N.; Sekiguchi, M.; Hiraoka, A. Heparinoid-active sulphated polysaccharides from Monostroma nitidum and their distribution in the chlorophyta. Phytochemistry 1991, 30, 3611–3614. [Google Scholar] [CrossRef]
- Mao, W.; Li, H.; Li, Y.; Zhang, H.; Qi, X.; Sun, H.; Chen, Y.; Guo, S. Chemical characteristic and anticoagulant activity of the sulfated polysaccharide isolated from Monostroma latissimum (Chlorophyta). Int. J. Biol. Macromol. 2009, 44, 70–74. [Google Scholar] [CrossRef]
- Mao, W.-J.; Fang, F.; Li, H.-Y.; Qi, X.-H.; Sun, H.-H.; Chen, Y.; Guo, S.-D. Heparinoid-active two sulfated polysaccharides isolated from marine green algae Monostroma nitidum. Carbohydr. Polym. 2008, 74, 834–839. [Google Scholar] [CrossRef]
- Zhang, H.; Mao, W.; Fang, F.; Li, H.; Sun, H.; Chen, Y.; Qi, X. Chemical characteristics and anticoagulant activities of a sulfated polysaccharide and its fragments from Monostroma latissimum. Carbohydr. Polym. 2008, 71, 428–434. [Google Scholar] [CrossRef]
- Li, H.; Mao, W.; Zhang, X.; Qi, X.; Chen, Y.; Chen, Y.; Xu, J.; Zhao, C.; Hou, Y.; Yang, Y.; et al. Structural characterization of an anticoagulant-active sulfated polysaccharide isolated from green alga Monostroma latissimum. Carbohydr. Polym. 2011, 85, 394–400. [Google Scholar] [CrossRef]
- Li, H.; Mao, W.; Hou, Y.; Gao, Y.; Qi, X.; Zhao, C.; Chen, Y.; Chen, Y.; Li, N.; Wang, C. Preparation, structure and anticoagulant activity of a low molecular weight fraction produced by mild acid hydrolysis of sulfated rhamnan from Monostroma latissimum. Bioresour. Technol. 2012, 114, 414–418. [Google Scholar] [CrossRef] [PubMed]
- Mao, W.; Zang, X.; Li, Y.; Zhang, H. Sulfated polysaccharides from marine green algae Ulva conglobata and their anticoagulant activity. J. Appl. Phycol. 2006, 18, 9–14. [Google Scholar] [CrossRef]
- Minh Thu, Q.T. Effect of sulfation on the structure and anticoagulant activity of ulvan extracted from green seaweed Ulva reticulata. Vietnam J. Sci. Technol. 2018, 54, 373. [Google Scholar] [CrossRef]
- Barros Gomes Camara, R.; Silva Costa, L.; Pereira Fidelis, G.; Duarte Barreto Nobre, L.T.; Dantas-Santos, N.; Lima Cordeiro, S.; Santana Santos Pereira Costa, M.; Guimaraes Alves, L.; Oliveira Rocha, H.A. Heterofucans from the Brown Seaweed Canistrocarpus cervicornis with Anticoagulant and Antioxidant Activities. Mar. Drugs 2011, 9, 124–138. [Google Scholar] [CrossRef]
- Li, B.; Lu, F.; Wei, X.; Zhao, R. Fucoidan: Structure and Bioactivity. Molecules 2008, 13, 1671–1695. [Google Scholar] [CrossRef] [PubMed]
- Mestechkina, N.M.; Shcherbukhin, V.D. Sulfated polysaccharides and their anticoagulant activity: A review. Appl. Biochem. Microbiol. 2010, 46, 267–273. [Google Scholar] [CrossRef]
- Adrien, A.; Bonnet, A.; Dufour, D.; Baudouin, S.; Maugard, T.; Bridiau, N. Pilot production of ulvans from Ulva sp. and their effects on hyaluronan and collagen production in cultured dermal fibroblasts. Carbohydr. Polym. 2017, 157, 1306–1314. [Google Scholar] [CrossRef] [PubMed]
- Lahaye, M.; Alvarez-Cabal Cimadevilla, E.; Kuhlenkamp, R.; Quemener, B.; Lognoné, V.; Dion, P. Chemical composition and 13C NMR spectroscopic characterisation of ulvans from Ulva (Ulvales, Chlorophyta). J. Appl. Phycol. 1999, 11, 1. [Google Scholar] [CrossRef]
- Nishino, T.; Aizu, Y.; Nagumo, T. Antithrombin activity of a fucan sulfate from the brown seaweed Ecklonia kurome. Thromb. Res. 1991, 62, 765–773. [Google Scholar] [CrossRef]
- Costa, M.S.S.P.; Costa, L.S.; Cordeiro, S.L.; Almeida-Lima, J.; Dantas-Santos, N.; Magalhães, K.D.; Sabry, D.A.; Albuquerque, I.R.L.; Pereira, M.R.; Leite, E.L.; et al. Evaluating the possible anticoagulant and antioxidant effects of sulfated polysaccharides from the tropical green alga Caulerpa cupressoides var. flabellata. J. Appl. Phycol. 2012, 24, 1159–1167. [Google Scholar] [CrossRef]
- Ceroni, A.; Dell, A.; Haslam, S.M. The GlycanBuilder: a fast, intuitive and flexible software tool for building and displaying glycan structures. Source Code Biol. Med. 2007, 2, 3. [Google Scholar] [CrossRef]
- Saad, O.M.; Leary, J.A. Delineating mechanisms of dissociation for isomeric heparin disaccharides using isotope labeling and ion trap tandem mass spectrometry. J. Am. Soc. Mass Spectrom. 2004, 15, 1274–1286. [Google Scholar] [CrossRef] [PubMed]
- Qi, X.; Mao, W.; Gao, Y.; Chen, Y.; Chen, Y.; Zhao, C.; Li, N.; Wang, C.; Yan, M.; Lin, C.; et al. Chemical characteristic of an anticoagulant-active sulfated polysaccharide from Enteromorpha clathrata. Carbohydr. Polym. 2012, 90, 1804–1810. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, Z.; Yao, Z.; Zhao, M.; Qi, H. Sulfation, anticoagulant and antioxidant activities of polysaccharide from green algae Enteromorpha linza. Int. J. Biol. Macromol. 2013, 58, 225–230. [Google Scholar] [CrossRef]
- Rodrigues, J.A.G.; Queiroz, I.N.L.D.; Quinderé, A.L.G.; Vairo, B.C.; Mourão, P.A.D.S.; Benevides, N.M.B. An antithrombin-dependent sulfated polysaccharide isolated from the green alga Caulerpa cupressoides has in vivo anti- and prothrombotic effects. Ciênc. Rural 2011, 41, 634–639. [Google Scholar] [CrossRef]
- Li, N.; Mao, W.; Yan, M.; Liu, X.; Xia, Z.; Wang, S.; Xiao, B.; Chen, C.; Zhang, L.; Cao, S. Structural characterization and anticoagulant activity of a sulfated polysaccharide from the green alga Codium divaricatum. Carbohydr. Polym. 2015, 121, 175–182. [Google Scholar] [CrossRef]
- Majdoub, H.; Mansour, M.B.; Chaubet, F.; Roudesli, M.S.; Maaroufi, R.M. Anticoagulant activity of a sulfated polysaccharide from the green alga Arthrospira platensis. Biochim. Biophys. Acta BBA - Gen. Subj. 2009, 1790, 1377–1381. [Google Scholar] [CrossRef]
- Synytsya, A.; Choi, D.J.; Pohl, R.; Na, Y.S.; Capek, P.; Lattová, E.; Taubner, T.; Choi, J.W.; Lee, C.W.; Park, J.K.; et al. Structural Features and Anti-coagulant Activity of the Sulphated Polysaccharide SPS-CF from a Green Alga Capsosiphon fulvescens. Mar. Biotechnol. 2015, 17, 718–735. [Google Scholar] [CrossRef] [PubMed]
- Melo, F.R.; Pereira, M.S.; Foguel, D.; Mourão, P.A.S. Antithrombin-mediated Anticoagulant Activity of Sulfated Polysaccharides: Different mechanisms for heparin and sulfated galactans. J. Biol. Chem. 2004, 279, 20824–20835. [Google Scholar] [CrossRef]
- Mourão, P.A.S. Use of sulfated fucans as anticoagulant and antithrombotic agents: future perspectives. Curr. Pharm. Des. 2004, 10, 967–981. [Google Scholar] [CrossRef]
- Becker, C.F.; Guimarães, J.A.; Mourão, P.A.S.; Verli, H. Conformation of sulfated galactan and sulfated fucan in aqueous solutions: Implications to their anticoagulant activities. J. Mol. Graph. Model. 2007, 26, 391–399. [Google Scholar] [CrossRef]
- De Araújo, C.A.; Noseda, M.D.; Cipriani, T.R.; Gonçalves, A.G.; Duarte, M.E.R.; Ducatti, D.R.B. Selective sulfation of carrageenans and the influence of sulfate regiochemistry on anticoagulant properties. Carbohydr. Polym. 2013, 91, 483–491. [Google Scholar] [CrossRef]
- Ménard, R.; Alban, S.; de Ruffray, P.; Jamois, F.; Franz, G.; Fritig, B.; Yvin, J.-C.; Kauffmann, S. Beta-1,3 glucan sulfate, but not beta-1,3 glucan, induces the salicylic acid signaling pathway in tobacco and Arabidopsis. Plant Cell 2004, 16, 3020–3032. [Google Scholar] [CrossRef] [PubMed]
- Dubois, M.; Gilles, K.; Hamilton, J.K.; Rebers, P.A.; Smith, F. A Colorimetric Method for the Determination of Sugars. Nature 1951, 168, 167. [Google Scholar] [CrossRef]
- Bitter, T.; Muir, H.M. A modified uronic acid carbazole reaction. Anal. Biochem. 1962, 4, 330–334. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Jaques, L.B.; Balueux, R.E.; Dietrich, C.P.; Kavanagh, L.W. A microelectrophoresis method for heparin. Can. J. Physiol. Pharmacol. 1968, 46, 351–360. [Google Scholar] [CrossRef] [PubMed]
- Gao, G.; Jiao, Q.; Ding, Y.; Chen, L. Study on quantitative assay of chondroitin sulfate with a spectrophotometric method of azure A. Guang Pu Xue Yu Guang Pu Fen Xi 2003, 23, 600–602. [Google Scholar]
- Singleton, V.L.; Rossi, J.A. Colorimetry of Total Phenolics with Phosphomolybdic-Phosphotungstic Acid Reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Ye, F.; Kuang, Y.; Chen, S.; Zhang, C.; Chen, Y.; Xing, X.-H. Characteristics of low molecular weight heparin production by an ultrafiltration membrane bioreactor using maltose binding protein fused heparinase I. Biochem. Eng. J. 2009, 46, 193–198. [Google Scholar] [CrossRef]
- Miller, G.L. Use of Dinitrosalicylic Acid Reagent for Determination of Reducing Sugar. Anal. Chem. 1959, 31, 426–428. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]

| ULVAN-01 | ULVAN-02 | ||
|---|---|---|---|
| Neutral sugars | % (w/wdry fraction) | 41 | 33 |
| Uronic acids | 34 | 31 | |
| Sulfates on the polysaccharide backbone (sulfate to uronic acid molar ratio) | 11 (0.65) | 20 (1.30) | |
| Proteins | 4 | <1 | |
| Polyphenols | 0 | 0 | |
| Lipids | 0 | 0 | |
| Ashes | 23 | nd (d) | |
| Monosaccharide composition (% molar ratio) | Glc/Gal | 12.2 | nd (d) |
| Xyl | 8.0 | nd (d) | |
| Rha | 42.6 | nd (d) | |
| GlcN | 6.9 | nd (d) | |
| GlcA/IdoA | 30.3 | nd (d) | |
| Mn (a) (kDa) | 31.3 | 39.0 | |
| Mw (b) (kDa) | 56.7 | 55.3 | |
| I (c) | 1.8 | 1.4 | |
| Oligosaccharide Sequence | Ion (m/z) | Ionization | Molecular Formula | Symbolic Representation of Oligosaccharide Fragments (a) (CFG Nomenclature (b))  Rha, Rha,  GlcA (or IdoA) (c) GlcA (or IdoA) (c)  Glc (or Gal) (c), Glc (or Gal) (c),  Xyl XylS = Sulfate Group | ||
|---|---|---|---|---|---|---|
| Experimental Value | Predicted Value | Resolution (ppm) | ||||
| 1 | 1561.3492 | 1561.3516 | 1.54 | [M − H]− | C54H81O50S |  |
| 2 | 1561.3492 | 1561.3516 | 1.54 | [M − H]− | C54H81O50S | 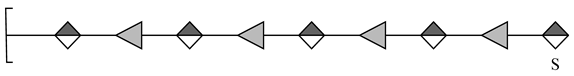 |
| 3 | 1723.4083 | 1723.4045 | 2.20 | [M − H]− | C60H91O55S | 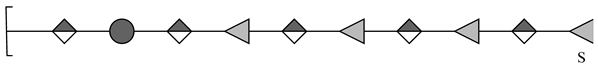 |
| 4 | 1839.4554 | 1839.4518 | 1.95 | [M − H]− | C65H99O58S |  |
| 5 | 401.0385 | 401.0390 | 1.25 | [M − H − H2O]− | C12H17O13S | 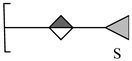 |
| 6 | 502.9772 | 502.9777 | 0.99 | [M − 2H − H2O + Na]− | C12H16O16S2Na | 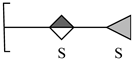 |
| 7 | 604.9165 | 604.9165 | 0.00 | [M − 3H − H2O + 2Na]− | C12H15O19S3Na2 | 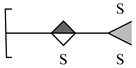 |
| 8 | 706.8561 | 706.8553 | 1.13 | [M − 4H − H2O + 3Na]− | C12H14O22S4Na3 | 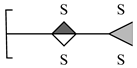 |
| - | Concentration (µg/mL) | |||
|---|---|---|---|---|
| Unfractionated Heparin (UFH) | Lovenox® | ULVAN-01 | ULVAN-02 | |
| Activated partial thromboplastin time (APTT)(a) | 0.75 | 4.7 | 500 | 2.4 |
| Prothrombin time (PT)(a) | 14.25 | 480 | inactive | 45 |
| Thrombin time (TT)(a) | 0.45 | 1.82 | 117 | 2.62 |
| Anti-Xa(b) | 0.2 | 0.125 | inactive | 17.5 |
| Anti-IIa(b) | 0.25 | 1.75 | inactive | 18 |
| Monosaccharide Composition | Oligosaccharide Sequences | |
|---|---|---|
| UHPLC Parameters | ||
| Column | “Acquity UPLC BEH Amide” (Waters) (2.1 × 500 mm, 1.7 µm), maintained at 30 °C | |
| Flow rate (µL/min) | 0–15 min, 130–200; 15–18 min, 200; 18–30 min, 200–130; 30–37 min, 200 | 200 |
| Gradient: water (A)/acetonitrile (B) + 0.015% (v/v) ammoniac | 0–15 min, 90–55% B; 15–17 min, 55% B; 17–18 min, 55–90% B; 18–37 min, 90% B | 0–3 min, 100% B; 3–3.1 min 100–70% B; 3.1–10 min, 70–50% B; 10–10.5 min, 50–45% B; 10.5–18 min, 45–100% B |
| Injection | 5 µL (7 °C) | |
| MS Parameters | ||
| Mode | ESI+ (centroid) | ESI− (centroid) |
| Source temperature | 120 °C | |
| Desolvation temperature | 250 °C | 500 °C |
| Gas flow rate of the cone | 50 L/h | |
| Desolvation gas flow rate | 800 L/h | |
| Capillary voltage | 2.5 kV | |
| Sampling cone voltage | 35 V | 130 V |
| Source compensation | 80 V | |
| Acquisition mass range | 50–1200 m/z | 50–2100 m/z (down-regulated according to the fragmented ion with a scan time of 0.15 s) |
| Lock-mass | Leucine Enkephalin (MW = 555.62 Da, 1 ng/µL) | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Adrien, A.; Bonnet, A.; Dufour, D.; Baudouin, S.; Maugard, T.; Bridiau, N. Anticoagulant Activity of Sulfated Ulvan Isolated from the Green Macroalga Ulva rigida. Mar. Drugs 2019, 17, 291. https://doi.org/10.3390/md17050291
Adrien A, Bonnet A, Dufour D, Baudouin S, Maugard T, Bridiau N. Anticoagulant Activity of Sulfated Ulvan Isolated from the Green Macroalga Ulva rigida. Marine Drugs. 2019; 17(5):291. https://doi.org/10.3390/md17050291
Chicago/Turabian StyleAdrien, Amandine, Antoine Bonnet, Delphine Dufour, Stanislas Baudouin, Thierry Maugard, and Nicolas Bridiau. 2019. "Anticoagulant Activity of Sulfated Ulvan Isolated from the Green Macroalga Ulva rigida" Marine Drugs 17, no. 5: 291. https://doi.org/10.3390/md17050291
APA StyleAdrien, A., Bonnet, A., Dufour, D., Baudouin, S., Maugard, T., & Bridiau, N. (2019). Anticoagulant Activity of Sulfated Ulvan Isolated from the Green Macroalga Ulva rigida. Marine Drugs, 17(5), 291. https://doi.org/10.3390/md17050291






