Chemical Synthesis of Marine-Derived Sulfoglycolipids, a New Class of Molecular Adjuvants
Abstract
:1. Introduction
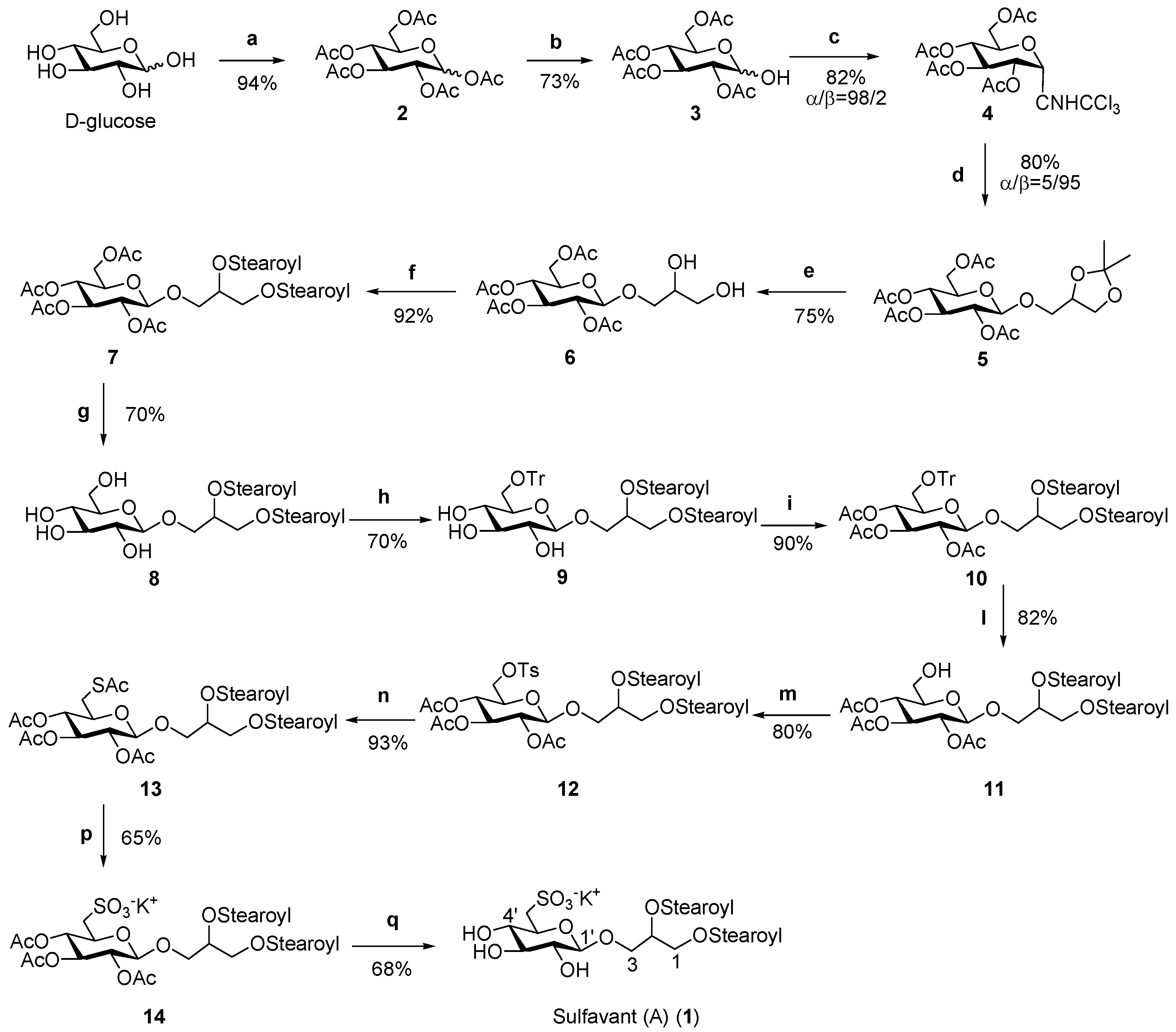
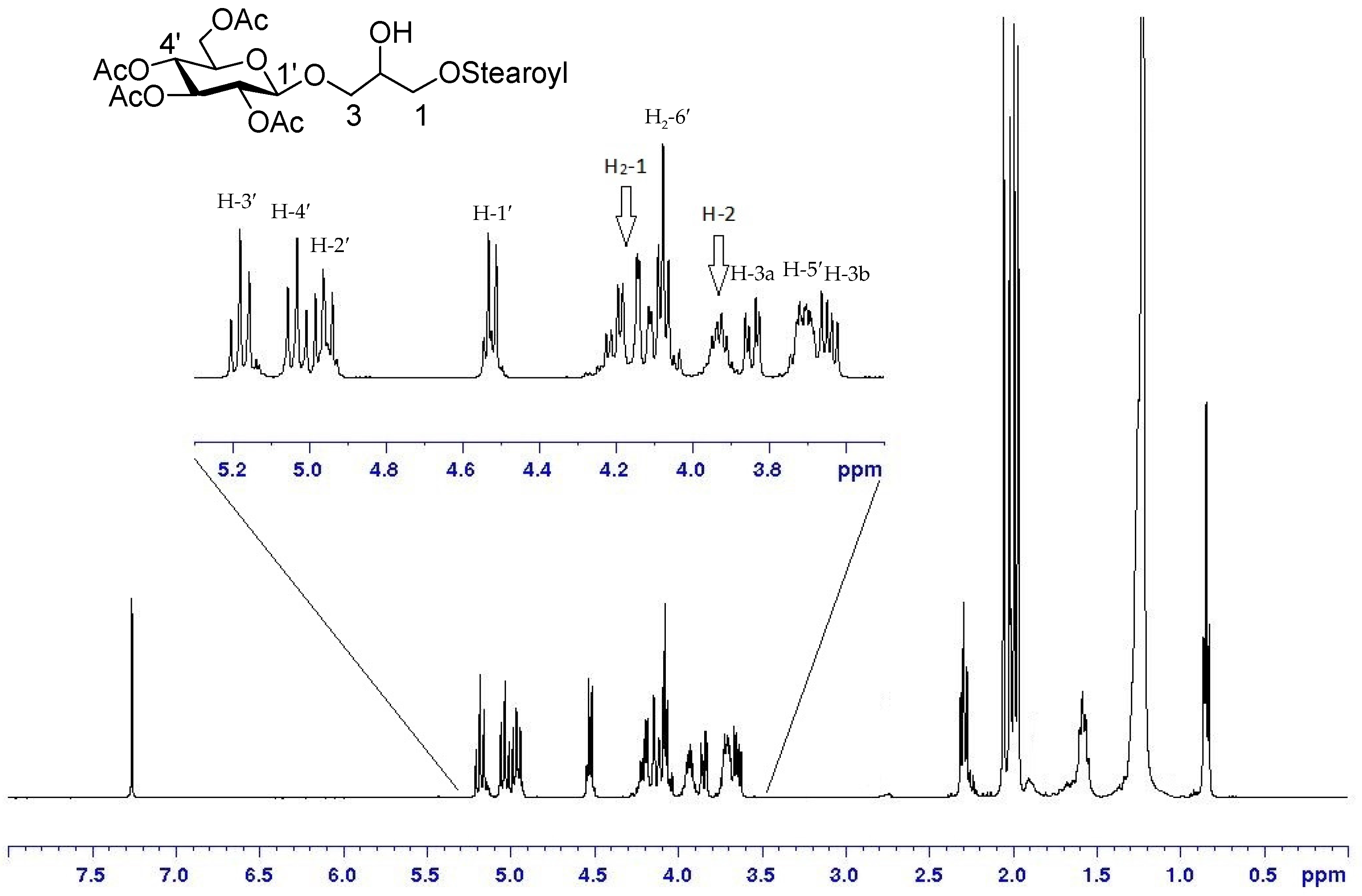
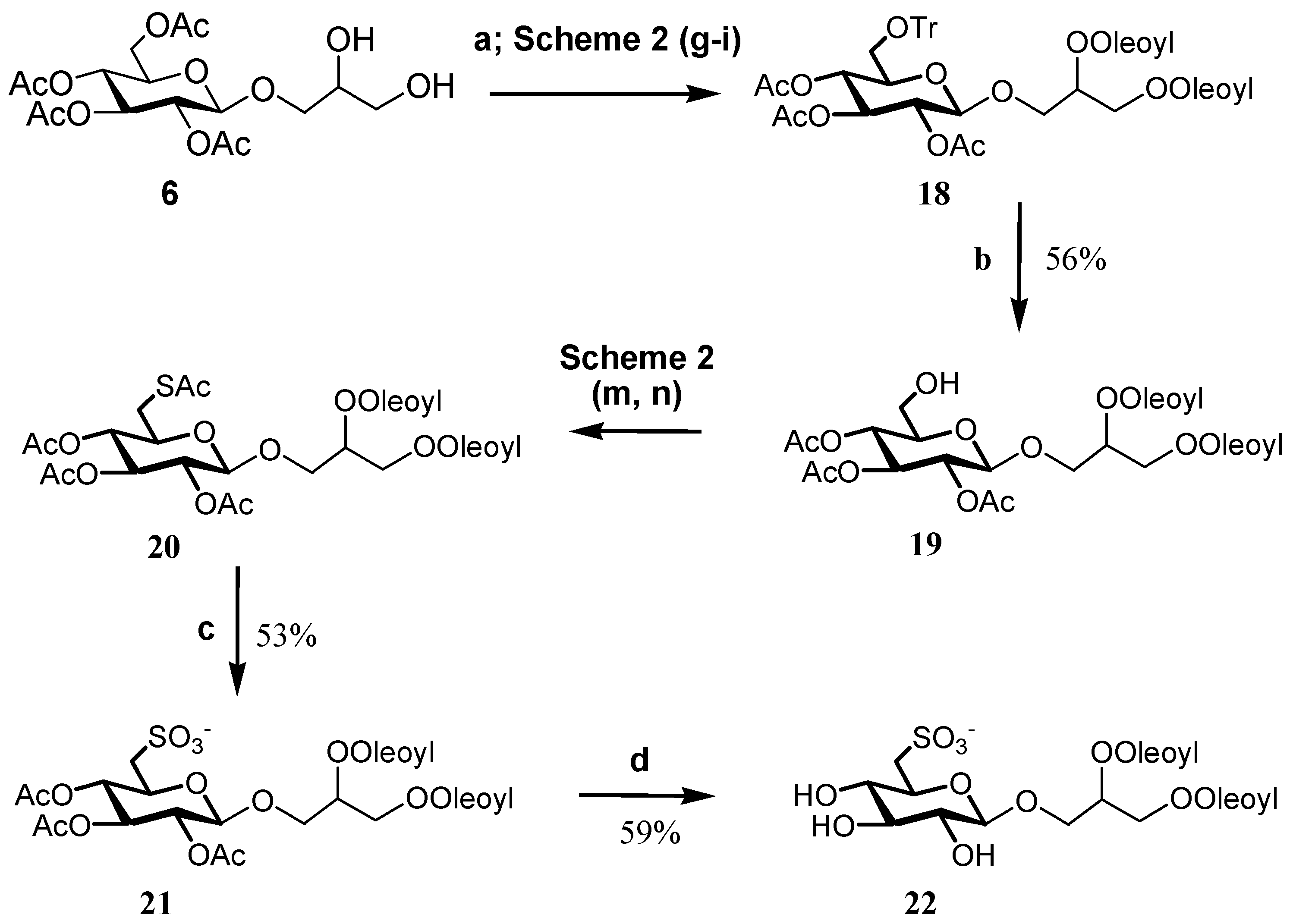
2. Results and Discussion
3. Materials and Methods
Preparation and Characterization of Synthetic Molecules
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Di Pasquale, A.; Preiss, S.; Da Silva, F.T.; Garcon, N. Vaccine Adjuvants: Form 1920 to 2015 and Beyond. Vaccines 2015, 3, 320–343. [Google Scholar] [CrossRef] [PubMed]
- Rappuoli, R.; Mandl, C.W.; Black, S.; De Gregorio, E. Vaccines for the twenthy-first century society. Nat. Rev. Immunol. 2011, 11, 865–872. [Google Scholar] [PubMed]
- Kastenmuller, W.; Kastenmuller, K.; Kurts, C.; Seder, R.A. Dendritic cell-targeted vaccines-hope or hype? Nat. Rev. Immunol. 2014, 14, 705–711. [Google Scholar] [CrossRef] [PubMed]
- Manzo, E.; Cutignano, A.; Pagano, D.; Gallo, C.; Barra, G.; Nuzzo, G.; Sansone, C.; Ianora, A.; Urbanek, K.; Fenoglio, D.; et al. A new marine-derived sulfoglycolipid triggers dendritic cell activation and immune adjuvant response. Sci. Rep. Nat. 2017, in press. [Google Scholar] [CrossRef] [PubMed]
- Sahara, H.; Ishikawa, M.; Takahashi, N.; Ohtani, S.; Sato, N.; Gasa, S.; Akino, T.; Kikuchi, K. In vivo anti-tumour effectof 3′-sulphonoquinovosyl 1′-monoacylglyceride isolated from sea urchin (Strongylocentrotus intermedius) intestine. Br. J. Cancer 1997, 75, 324–332. [Google Scholar] [CrossRef] [PubMed]
- Golik, J.; Dickey, J.K.; Todderud, G.; Lee, D.; Alford, J.; Huang, S.; Klohr, S.; Eustice, D.; Aruffo, A.; Agler, M.L. Isolation and Structure Determination of Sulfonoquinovoyl Dipalmitoyl Glyceride, a P-selectin Receptor Inhibitor from the Alga Dictyochloris fragrans. J. Nat. Prod. 1997, 60, 387–389. [Google Scholar] [CrossRef] [PubMed]
- Loya, S.; Reshef, V.; Mizrachi, E.; Silberstein, C.; Rachamim, Y.; Carmeli, S.; Hizi, A. The Inhibition of the Reverse Transcriptase of HIV-1 by the Natural Sulfoglycolipids from Cyanobacteria: Contribution of Different Moieties to Their High Potency. J. Nat. Prod. 1998, 61, 891–895. [Google Scholar] [CrossRef] [PubMed]
- Gustafson, R.; Cardelina, J.H.; Fuller, R.W.; Weislow, O.S.; Kiser, R.F.; Snader, K.M.; Patterson, G.M.L.; Boyd, M.R. AIDS-Antiviral Sulfolipids From Cyanobacteria (Blue-Green Algae). J. Nat. Cancer Inst. 1989, 81, 1254–1258. [Google Scholar] [CrossRef] [PubMed]
- Murakami, C.; Yamazaki, T.; Hanashima, S.; Takahashi, S.; Ohta, K.; Yoshida, H.; Sugawara, F.; Sakaguchi, K.; Mizushina, Y. Structure-function relationship of synthetic sulfoquinovosyl-acylglycerols as mammalian DNA polymerase inhibitors. Arch. Biochem. Biophys. 2002, 403, 229–236. [Google Scholar] [CrossRef]
- Matsumoto, Y.; Sahara, H.; Fujita, T.; Shimozawa, K.; Takenouchi, M.; Torigoe, T.; Hanashima, S.; Yamazaki, T.; Takahashi, S.; Sugawara, F.; et al. An immunosuppressive effect by synthetic sulfonolipids deduced from sulfoquinovosyl diacylglycerols of sea urchin. Transplantation 2002, 74, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, Y.; Sahara, H.; Fujita, T.; Hanashima, S.; Yamazaki, T.; Takahashi, S.; Sugawara, F.; Mizushina, Y.; Ohta, K.; Takahashi, N.; et al. A novel immunosuppressive agent, SQDG, derived from sea urchin. Transplant. Proc. 2000, 32, 2051–2053. [Google Scholar] [CrossRef]
- Ohta, K.; Mizushina, Y.; Yamazaki, T.; Hanashina, S.; Sugawara, F.; Sakaguchi, K. Specific interaction between an Oligosaccharide on the Tumour Cell Surface and the Novel Antitumour Agents, Sulfoquinovosylacylglycerols. Biochem. Biophys. Res. Commun. 2001, 288, 893–900. [Google Scholar] [CrossRef] [PubMed]
- Reshef, V.; Mizrachi, E.; Maretzki, T.; Silberstein, C.; Loya, S.; Hizi, A.; Carmeli, S. New Acylated Sulfoglycolipids and Digalactolipids and Related Known Glycolipids from Cyanobacteria with a Potential to Inhibit the Reverse Transcriptase of HIV-1. J. Nat. Prod. 1997, 60, 1251–1260. [Google Scholar] [CrossRef] [PubMed]
- Hanashima, S.; Mizushina, Y.; Yamazaki, T.; Ohta, K.; Takahashi, S.; Sahara, H.; Sagakuchi, K.; Sugawara, F. Synthesis of sulfoquinovosylacylglycerols, inhibitors of Eukaryotic DNA Polymerase α and β. Bioorg. Med. Chem. 2001, 9, 367–376. [Google Scholar] [CrossRef]
- Hanashima, S.; Mizushina, Y.; Yamazaki, T.; Ohta, K.; Takahashi, S.; Koshino, H.; Sahara, H.; Sagakuchi, K.; Sugawara, F. Structural determination of sulfoquinovosyldiacylglycerol by chiral syntheses. Tetrahedron Lett. 2000, 41, 4403–4407. [Google Scholar] [CrossRef]
- Mizushina, Y.; Maeda, N.; Kawasaki, M.; Ichikawa, H.; Murakami, C.; Takemura, M.; Xu, X.; Sugawara, F.; Fukumori, Y.; Yoshida, H.; Sakaguchi, K. Inhibitory action of emulsified sulfoquinovosyl acylglycerol on mammalian DNA polymerases. Lipids 2003, 38, 1065–1074. [Google Scholar] [CrossRef] [PubMed]
- Hanashima, S.; Mizushina, Y.; Ohta, K.; Yamazaki, T.; Sugawara, F.; Sakaguchi, K. Structure-Activity Relationship of a ovel Group of Mammalian DNA Polymerase Inhibitors, Synthetic Sulfoquinovosylacylglycerols. Jpn. J. Cancer Res. 2000, 91, 1073–1083. [Google Scholar] [CrossRef] [PubMed]
- Mizushina, Y.; Watanabe, I.; Ohta, K.; Takemura, M.; Sahara, H.; Takahashi, N.; Gasa, S.; Sugawara, F.; Matsukage, A.; Yoshida, S.; et al. Studies on inhibitors of mammalian DNA polymerase α and β: Sulfolipids from a pteridophyte, Athyrium niponicum. Biochem. Pharmacol. 1998, 55, 537–541. [Google Scholar] [CrossRef]
- Ohta, K.; Hanashima, S.; Mizushina, Y.; Yamazaki, T.; Saneyoshi, M.; Sugawara, F.; Sakaguchi, K. Studies on a novel DNA polymerase inhibitor group, synthetic sulfoquinovosylacylglycerol: inhibitory action on cell proliferation. Mutat. Res. 2000, 467, 139–152. [Google Scholar] [CrossRef]
- Mizushina, Y.; Xu, X.; Asahara, H.; Takemura, M.; Yamaguchi, T.; Kuroda, K.; Linn, S.; Yoshida, H.; Koiwai, O.; Saneyoshi, M.; et al. A sulfoquinovosyl diacylglycerol is a DNA polymerase epsilon inhibotor. Biochem. J. 2003, 370, 299–305. [Google Scholar] [CrossRef] [PubMed]
- Gigg, R.; Penglis, A.A.E.; Conant, R. Synthesis of 3-O-(6-deoxy-6-sulfo-α-d-glucopyranosyl)-1,2-di-O-hexadecanoyl-l-glycerol, ‘sulphoquinovosyl diglyceride’. J. Chem. Soc. 1980, 2490–2493. [Google Scholar]
- Yamazaki, T.; Aoki, S.; Ohta, K.; Hyuma, S.; Sakaguchi, K.; Sugawara, F. Synthesis of an immunosuppressant SQAG9 and the determination of the binding peptide by T7 phage display. Bioorg. Med. Chem. Lett. 2004, 4343–4346. [Google Scholar] [CrossRef] [PubMed]
- Manzo, E.; Ciavatta, M.L.; Pagano, D.; Fontana, A. An efficient and verstaile chemical synthesis of bioactive glyco-glycerolipids. Tetrahedron Lett. 2012, 53, 879–881. [Google Scholar] [CrossRef]
- Pagano, D.; Cutignano, A.; Manzo, E.; Tinto, F.; Fontana, A. Glycolipids synthesis: Improved hydrazinolysis conditions for preparation of 1,2-polyunsaturated fatty acyl-β-monogalactosyl-glycerols. Carbohydr. Res. 2016, 424, 21–23. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, R.R.; Michel, J. Facile synthesis of α- and β-O-Glycosil Imidates; Preparation of Glycosides and Disaccharides. Angew. Chem. Int. Ed. Engl. 1980, 19, 731–732. [Google Scholar] [CrossRef]
- Schmidt, R.R. New Methods for the Synthesis of Glycosides and Oligosaccharides—Are There Alternatives to the Koenigs-Knorr Method? [New Synthetic Methods (56)]. Angew. Chem. Int. Ed. 1986, 25, 212–235. [Google Scholar] [CrossRef]
- Vijayasaradhi, S.; Singh, J.; Aidhen, I.S. An Efficient, Selective Hydrolysis of Terminal Isopropylidene Acetal Protection by Zn(NO3)26H2O in Acetonitrile. Synlett 2000, 110–112. [Google Scholar]
- Wahlstrom, J.L.; Ronald, R.C. Detritylation of Ethers using Iodine-Alcohol Reagents: An Acid Catalytized Reaction. J. Org. Chem. 1998, 63, 6021–6022. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Bolanos, J.G.; Morales, J.; Garcia, S.; Dianez, M.J.; Estrada, M.D.; Lopez-Castro, A.; Perez, S. Synthesis and cristal structure of methyl 2-amino-2,6-dideoxy-α-d-glucopiranoside-6-sulfonic acid. Carbohydr. Res. 1993, 248, 1–14. [Google Scholar] [CrossRef]
- Chen, J.; Profit, A.A.; Prestwich, G.D. Synthesis of Photoactivatable 1,2-O-Diacyl-sn-glycerol Derivatives of 1-l-Phosphatidyl-d-myo-inositol 4,5-Bisphosphate (PtdInsP2) and 3,4,5-Trisphosphate (PtdInsP3). J. Org. Chem. 1996, 61, 6305–6312. [Google Scholar] [CrossRef] [PubMed]
- Dosuna-Rodríguez, I.D.; Gaigneaux, E.M. Glycerol acetylation catalysed by ion exchange resins. Catal. Today 2012, 195, 14–21. [Google Scholar] [CrossRef]
- Pruvost, R.; Boulanger, J.; Léger, B.; Ponchel, A.; Monflier, E.; Ibert, M.; Mortreux, A.; Sauthie, M. Biphasic Palladium-Catalyzed Hydroesterification in aPolyol Phase: Selective Synthesis of Derived Monoesters. ChemSusChem 2015, 8, 2133–2137. [Google Scholar] [CrossRef] [PubMed]



| Position | δC | δH, m, J (Hz) |
|---|---|---|
| 1′ | 103.4 | 4.34/4.32 (each for 1H), d, 7.8 |
| 2′ | 73.6 | 3.26, m |
| 3′ | 76.5 | 3.42, m |
| 4′ | 73.1 | 3.14, m |
| 5′ | 72.5 | 3.79–3.75, m |
| 6′a | 53.8 | 3.32, m |
| 6′b | 53.8 | 2.98, m |
| 1a | 63.3 | 4.47, m |
| 1b | 63.3 | 4.19, m |
| 2 | 70.5 | 5.29, m |
| 3a | 67.8 | 4.13–4.03, m |
| 3b | 67.8 | 3.79–3.75, m |
| Fatty acyls | ||
| α-CH2 | 34.4 | 2.43–2.35, overlapped |
| β-CH2 | 25.1 | 1.69–1.54, overlapped |
| CH2 (chain) | 30.4–29.6 | 1.32–1.25, overlapped |
| CH3 | 14.1 | 0.94, overlapped |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manzo, E.; Fioretto, L.; Pagano, D.; Nuzzo, G.; Gallo, C.; De Palma, R.; Fontana, A. Chemical Synthesis of Marine-Derived Sulfoglycolipids, a New Class of Molecular Adjuvants. Mar. Drugs 2017, 15, 288. https://doi.org/10.3390/md15090288
Manzo E, Fioretto L, Pagano D, Nuzzo G, Gallo C, De Palma R, Fontana A. Chemical Synthesis of Marine-Derived Sulfoglycolipids, a New Class of Molecular Adjuvants. Marine Drugs. 2017; 15(9):288. https://doi.org/10.3390/md15090288
Chicago/Turabian StyleManzo, Emiliano, Laura Fioretto, Dario Pagano, Genoveffa Nuzzo, Carmela Gallo, Raffaele De Palma, and Angelo Fontana. 2017. "Chemical Synthesis of Marine-Derived Sulfoglycolipids, a New Class of Molecular Adjuvants" Marine Drugs 15, no. 9: 288. https://doi.org/10.3390/md15090288
APA StyleManzo, E., Fioretto, L., Pagano, D., Nuzzo, G., Gallo, C., De Palma, R., & Fontana, A. (2017). Chemical Synthesis of Marine-Derived Sulfoglycolipids, a New Class of Molecular Adjuvants. Marine Drugs, 15(9), 288. https://doi.org/10.3390/md15090288








