Development and Preliminary Characterization of Polyester-Urethane Microparticles Used in Curcumin Drug Delivery System for Oropharyngeal Cancer
Abstract
1. Introduction
2. Materials and Methods
2.1. Design and Ethical Considerations
2.2. The Reagents
2.3. Chemical Synthesis
2.4. Sample Calibration
2.5. Sample Analysis and Statistical Analysis
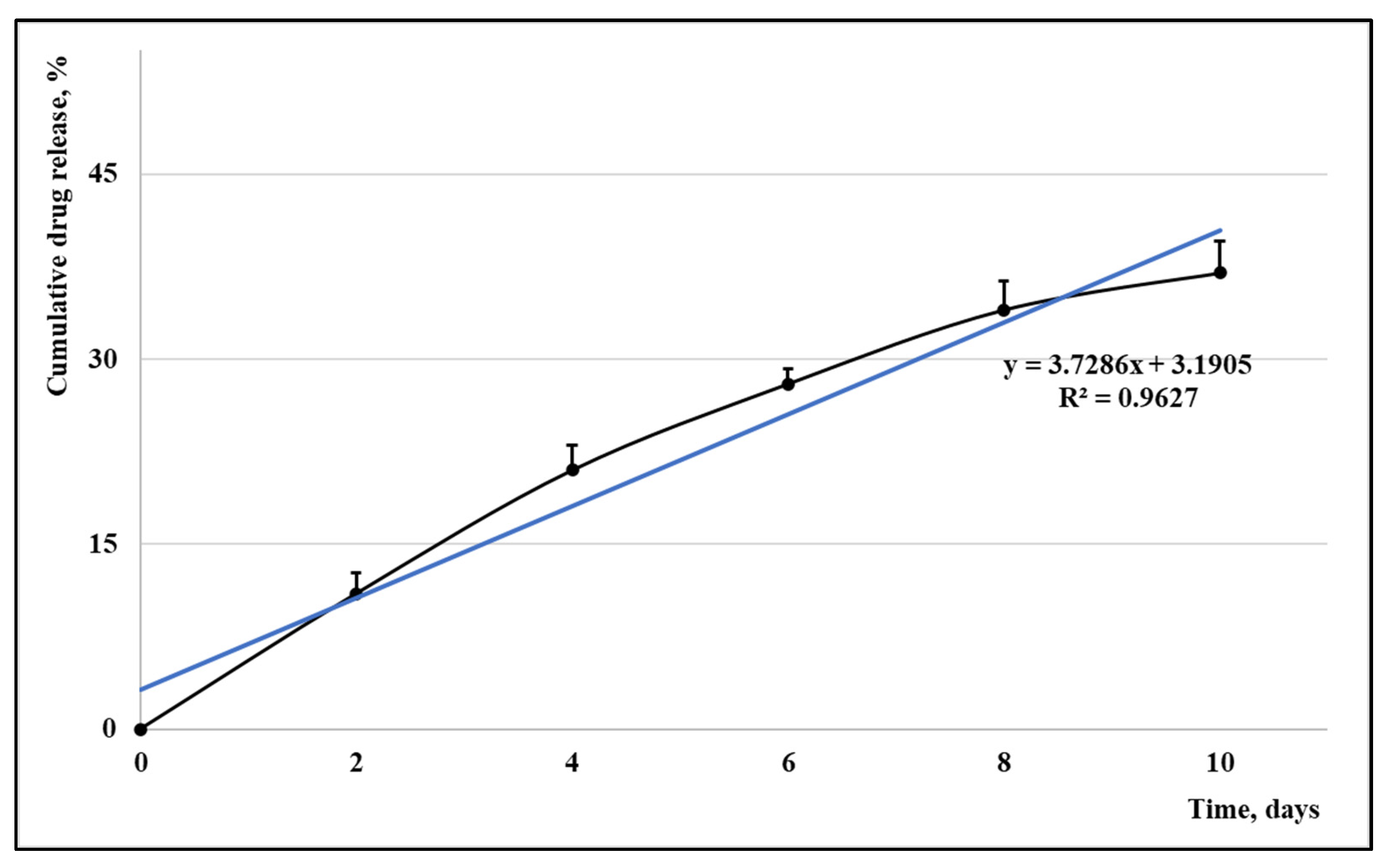
3. Results
3.1. Characterization of the Encapsulated Active Agent
3.2. Characterization of the Zetasizer
3.3. In Vivo Characterization of Samples
4. Discussion
4.1. Supporting Literature
4.2. Study Limitations and Future Perspectives
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Borcan, F.; Mioc, M.; Ghiulai, R.; Pinzaru, I.; Dehelean, C.A.; Soica, C.M. A Study on the Behavior of a Polyurethane Drug Carrier in Various pH Media. J. Med. Life 2020, 13, 195–199. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Xu, D.; Liu, C.; Guan, Y.; Zhang, J.; Su, Y.; Zhao, L.; Meng, F.; Luo, J. Biodegradable pH-sensitive polyurethane micelles with different polyethylene glycol (PEG) locations for anti-cancer drug carrier applications. RSC Adv. 2016, 6, 97684–97693. [Google Scholar] [CrossRef]
- Warnakulasuriya, S. Global epidemiology of oral and oropharyngeal cancer. Oral Oncol. 2009, 45, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Alqahtani, W.S.; Almufareh, N.A.; Al-Johani, H.A.; Alotaibi, R.K.; Juliana, C.I.; Aljarba, N.H.; Alqahtani, A.S.; Almarshedy, B.; Elasbali, A.M.; Ahmed, H.G.; et al. Oral and Oropharyngeal Cancers and Possible Risk Factors Across Gulf Cooperation Council Countries: A Systematic Review. World J. Oncol. 2020, 11, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Cohan, D.M.; Popat, S.; Kaplan, S.E.; Rigual, N.; Loree, T.; Hicks, W.L. Oropharyngeal cancer: Current understanding and management. Curr. Opin. Otolaryngol. Head Neck Surg. 2009, 17, 88–94. [Google Scholar] [CrossRef]
- Lynch, K.L.; Twesten, J.E.; Stern, A.L.; Augustson, E.M. Level of Alcohol Consumption and Successful Smoking Cessation. Nicotine Tob. Res. 2018, 21, 1058–1064. [Google Scholar] [CrossRef]
- Huang, C.-C.; Hsiao, J.-R.; Lee, W.-T.; Lee, Y.-C.; Ou, C.-Y.; Chang, C.-C.; Lu, Y.-C.; Huang, J.-S.; Wong, T.-Y.; Chen, K.-C.; et al. Investigating the Association between Alcohol and Risk of Head and Neck Cancer in Taiwan. Sci. Rep. 2017, 7, 9701. [Google Scholar] [CrossRef]
- Elrefaey, S.; Massaro, M.A.; Chiocca, S.; Chiesa, F.; Ansarin, M. HPV in oropharyngeal cancer: The basics to know in clinical prac-tice. Acta Otorhinolaryngol. Ital. 2014, 34, 299–309. [Google Scholar]
- Lechner, M.; Liu, J.; Masterson, L.; Fenton, T.R. HPV-associated oropharyngeal cancer: Epidemiology, molecular biology and clinical management. Nat. Rev. Clin. Oncol. 2022, 19, 306–327. [Google Scholar] [CrossRef]
- Zoi, V.; Galani, V.; Lianos, G.D.; Voulgaris, S.; Kyritsis, A.P.; Alexiou, G.A. The Role of Curcumin in Cancer Treatment. Biomedicines 2021, 9, 1086. [Google Scholar] [CrossRef]
- Rahaman, M.; Rakib, A.; Mitra, S.; Tareq, A.; Emran, T.; Shahid-Ud-Daula, A.; Amin, M.; Simal-Gandara, J. The Genus Curcuma and Inflammation: Overview of the Pharmacological Perspectives. Plants 2020, 10, 63. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Rad, J.; El Rayess, Y.; Rizk, A.A.; Sadaka, C.; Zgheib, R.; Zam, W.; Sestito, S.; Rapposelli, S.; Neffe-Skocińska, K.; Zielińska, D.; et al. Turmeric and Its Major Compound Curcumin on Health: Bioactive Effects and Safety Profiles for Food, Pharmaceutical, Biotechnological and Medicinal Applications. Front. Pharmacol. 2020, 11, 01021. [Google Scholar] [CrossRef] [PubMed]
- Maiti, P.; Dunbar, G.L. Use of Curcumin, a Natural Polyphenol for Targeting Molecular Pathways in Treating Age-Related Neurodegenerative Diseases. Int. J. Mol. Sci. 2018, 19, 1637. [Google Scholar] [CrossRef]
- Hewlings, S.J.; Kalman, D.S. Curcumin: A Review of Its Effects on Human Health. Foods 2017, 6, 92. [Google Scholar] [CrossRef] [PubMed]
- Keihanian, F.; Saeidinia, A.; Bagheri, R.K.; Johnston, T.P.; Sahebkar, A. Curcumin, hemostasis, thrombosis, and coagulation. J. Cell Physiol. 2017, 233, 4497–4511. [Google Scholar] [CrossRef]
- Valokola, M.G.; Karimi, G.; Razavi, B.M.; Kianfar, M.; Jafarian, A.H.; Jaafari, M.R.; Imenshahidi, M. The protective activity of nanomicelle curcumin in bisphenol A-induced cardiotoxicity following subacute exposure in rats. Environ. Toxicol. 2018, 34, 319–329. [Google Scholar] [CrossRef]
- Heghes, A.; Soica, C.M.; Ardelean, S.; Ambrus, R.; Muntean, D.; Galuscan, A.; Dragos, D.; Ionescu, D.; Borcan, F. Influence of emulsifi-ers on the characteristics of polyurethane structures used as drug carrier. Chem. Cent. J. 2013, 7, 66. [Google Scholar] [CrossRef]
- Danciu, C.; Borcan, F.; Șoica, C.; Zupko, I.; Csanyi, E.; Ambrus, R.; Muntean, D.; Sass, C.; Antal, D.; Toma, C.; et al. Polyurethane Microstructures-a Good or Bad in vitro Partner for the Isoflavone Genistein? Nat. Prod. Commun. 2015, 10, 951–954. [Google Scholar] [CrossRef]
- Borcan, L.-C.; Dudas, Z.; Len, A.; Fuzi, J.; Borcan, F.; Tomescu, M.C. Synthesis and characterization of a polyurethane carrier used for a prolonged transmembrane transfer of a chili pepper extract. Int. J. Nanomed. 2018, 13, 7155–7166. [Google Scholar] [CrossRef]
- Borcan, F.; Chirita-Emandi, A.; Andreescu, N.I.; Borcan, L.-C.; Albulescu, R.C.; Puiu, M.; Tomescu, M.C. Synthesis and preliminary characterization of polyurethane nanoparticles with ginger extract as a possible cardiovascular protector. Int. J. Nanomed. 2019, 14, 3691–3703. [Google Scholar] [CrossRef]
- Gallardo, V.; Morales, M.E.; Ruiz, M.A.; Delgado, A.V. An experimental investigation of the stability of ethylcellulose latex: Cor-relation between zeta potential and sedimentation. Eur. J. Pharm. Sci. 2005, 26, 170–175. [Google Scholar] [CrossRef] [PubMed]
- Gurita, V.G.; Pavel, I.Z.; Borcan, F.; Moaca, A.; Danciu, C.; Diaconeasa, Z. Toxicological Evaluation of Some Essential Oils Obtained from Se-lected Romania Lamiaceae Species in Complex with Hydroxypropyl—Gamma-cyclodextrin. Rev. Chim. Buchar. 2019, 70, 3703–3707. [Google Scholar] [CrossRef]
- Cimmino, I.; Fiory, F.; Perruolo, G.; Miele, C.; Beguinot, F.; Formisano, P.; Oriente, F. Potential Mechanisms of Bisphenol A (BPA) Contributing to Human Disease. Int. J. Mol. Sci. 2020, 21, 5761. [Google Scholar] [CrossRef] [PubMed]
- Saura, M.; Marquez, S.; Reventun, P.; Olea-Herrero, N.; Arenas, M.I.; Moreno-Gómez-Toledano, R.; Gómez-Parrizas, M.; Muñóz-Moreno, C.; González-Santander, M.; Zaragoza, C.; et al. Oral administration of bisphenol A induces high blood pressure through angiotensin II/CaMKII-dependent uncoupling of eNOS. FASEB J. 2014, 28, 4719–4728. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Wang, W.E.; Li, M.; Ren, H.; Chen, C.; Wang, J.; Yang, J.; Zeng, C. Curcumin Exerts its Anti-hypertensive Effect by Down-regulating the AT1 Receptor in Vascular Smooth Muscle Cells. Sci. Rep. 2016, 6, 25579. [Google Scholar] [CrossRef]
- Kia, S.J.; Basirat, M.; Saedi, H.S.; Arab, S.A. Effects of nanomicelle curcumin capsules on prevention and treatment of oral mucosits in patients under chemotherapy with or without head and neck radiotherapy: A randomized clinical trial. BMC Complement. Med. Ther. 2021, 21, 232. [Google Scholar] [CrossRef]
- Zikaki, K.; Aggeli, I.-K.; Gaitanaki, C.; Beis, I. Curcumin induces the apoptotic intrinsic pathway via upregulation of reactive oxygen species and JNKs in H9c2 cardiac myoblasts. Apoptosis 2014, 19, 958–974. [Google Scholar] [CrossRef]
- Li, X.; Xie, W.; Xie, C.; Huang, C.; Zhu, J.; Liang, Z.; Deng, F.; Zhu, M.; Zhu, W.; Wu, R.; et al. Curcumin modulates miR-19/PTEN/AKT/p53 axis to suppress bisphenol A-induced MCF-7 breast cancer cell proliferation. Phytother. Res. 2014, 28, 1553–1560. [Google Scholar] [CrossRef]
- Geng, S.; Wang, S.; Zhu, W.; Xie, C.; Li, X.; Wu, J.; Zhu, J.; Jiang, Y.; Yang, X.; Li, Y.; et al. Curcumin attenuates BPA-induced insulin resistance in HepG2 cells through suppression of JNK/p38 pathways. Toxicol. Lett. 2017, 272, 75–83. [Google Scholar] [CrossRef]
- De Leo, V.; Casiello, M.; Deluca, G.; Cotugno, P.; Catucci, L.; Nacci, A.; Fusco, C.; D’Accolti, L. Concerning Synthesis of New Biobased Polycarbonates with Curcumin in Replacement of Bisphenol A and Recycled Diphenyl Carbonate as Example of Circular Economy. Polymers 2021, 13, 361. [Google Scholar] [CrossRef]
- Tátraaljai, D.; Kirschweng, B.; Kovács, J.; Földes, E.; Pukánszky, B. Processing stabilisation of PE with a natural antioxidant, curcumin. Eur. Polym. J. 2013, 49, 1196–1203. [Google Scholar] [CrossRef]
- Zheng, Y.; Jia, R.; Li, J.; Tian, X.; Qian, Y. Curcumin- and resveratrol-co-loaded nanoparticles in synergistic treatment of hepatocellular carcinoma. J. Nanobiotechnol. 2022, 20, 339. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, D.; Dash, P.; Ramadass, B.; Mangaraj, M. Nanocurcumin in Oral Squamous Cancer Cells and Its Efficacy as a Chemo-Adjuvant. Cureus 2022, 14, e24678. [Google Scholar] [CrossRef]
- Vicente-Martínez, Y.; Caravaca, M.; Soto-Meca, A. Determination of Very Low Concentration of Bisphenol A in Toys and Baby Pacifiers Using Dispersive Liquid–Liquid Microextraction by In Situ Ionic Liquid Formation and High-Performance Liquid Chromatography. Pharmaceuticals 2020, 13, 301. [Google Scholar] [CrossRef] [PubMed]



| Sample | Hydroxylic Comp. (mL/50 mL) | Curcumin (mg/50 mL) | Organic Comp. (mL/50 mL) | ||||
|---|---|---|---|---|---|---|---|
| BD | PC | PEG | Tween | IPDI | HMDI | ||
| PU | 0.40 | 0.60 | 2.15 | 1.50 | 0.00 | 2.15 | 2.85 |
| PU_Cc | 0.40 | 0.60 | 2.15 | 1.50 | 5.50 | 2.15 | 2.85 |
| Sample | Size of Structures (nm) | Zeta Potential (mV) | |
|---|---|---|---|
| Mean ± SD | IPDI | ||
| PU | 215 ± 11 | 0.4 | +22.91 |
| PU_Cc | 271 ± 19 | 0.6 | +23.74 |
| Variables | PU (n = 8) | PU_Cc (n = 8) | p-Value |
|---|---|---|---|
| Gender | 0.614 | ||
| Men | 5 (62.5%) | 4 (50.0%) | |
| Women | 3 (37.5%) | 4 (50.0%) | |
| Age (mean ± SD) | 57.6 ± 12.2 | 55.1 ± 10.9 | 0.672 |
| Etiology | 0.521 | ||
| HPV-related | 6 (75.0%) | 7 (87.5%) | |
| Non-HPV | 2 (25.0%) | 1 (12.5%) | |
| TNM Staging | 1 | ||
| Stage II | 4 (50.0%) | 4 (50.0%) | |
| Stage III | 4 (50.0%) | 4 (50.0%) | |
| Self-reported side effects | 0.583 | ||
| 0 | 6 (75.0%) | 7 (87.5%) | |
| 1 | 1 (12.5%) | 1 (12.5%) | |
| ≥2 | 1 (12.5%) | 0 (0.0%) |
| Variables (Mean ± SE) | PU (n = 8) | PU_Cc (n = 8) | p-Value |
|---|---|---|---|
| TEWL | |||
| Time 0 | 1.9 ± 0.1 | 2.1 ± 0.2 | 0.386 |
| Time 12 | 2.0 ± 0.2 | 2.4 ± 0.2 | 0.179 |
| Time 24 | 2.2 ± 0.1 | 2.5 ± 0.2 | 0.201 |
| Time 48 | 2.2 ± 0.1 | 2.6 ± 0.1 | 0.013 |
| Erythema | |||
| Time 0 | 15.1 ± 3.2 | 20.2 ± 2.4 | 0.223 |
| Time 12 | 21.0 ± 1.9 | 24.3 ± 3.1 | 0.379 |
| Time 24 | 20.3 ± 2.7 | 20.5 ± 2.9 | 0.960 |
| Time 48 | 23.4 ± 2.0 | 25.1 ± 2.2 | 0.576 |
| Skin hydration | |||
| Time 0 | −1.9 ± 0.5 | −2.2 ± 0.7 | 0.732 |
| Time 12 | −3.0 ± 0.3 | −2.6 ± 0.5 | 0.503 |
| Time 24 | −3.1 ± 0.2 | −2.2 ± 0.4 | 0.063 |
| Time 48 | −3.0 ± 0.1 | −2.8 ± 0.1 | 0.179 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chioreanu, A.; Mot, I.C.; Horhat, D.I.; Balica, N.C.; Sarau, C.A.; Morar, R.; Domuta, E.M.; Dumitru, C.; Negrean, R.A.; Bumbu, B.A.; et al. Development and Preliminary Characterization of Polyester-Urethane Microparticles Used in Curcumin Drug Delivery System for Oropharyngeal Cancer. Medicina 2022, 58, 1689. https://doi.org/10.3390/medicina58111689
Chioreanu A, Mot IC, Horhat DI, Balica NC, Sarau CA, Morar R, Domuta EM, Dumitru C, Negrean RA, Bumbu BA, et al. Development and Preliminary Characterization of Polyester-Urethane Microparticles Used in Curcumin Drug Delivery System for Oropharyngeal Cancer. Medicina. 2022; 58(11):1689. https://doi.org/10.3390/medicina58111689
Chicago/Turabian StyleChioreanu, Alexandru, Ion Cristian Mot, Delia Ioana Horhat, Nicolae Constantin Balica, Cristian Andrei Sarau, Raluca Morar, Eugenia Maria Domuta, Catalin Dumitru, Rodica Anamaria Negrean, Bogdan Andrei Bumbu, and et al. 2022. "Development and Preliminary Characterization of Polyester-Urethane Microparticles Used in Curcumin Drug Delivery System for Oropharyngeal Cancer" Medicina 58, no. 11: 1689. https://doi.org/10.3390/medicina58111689
APA StyleChioreanu, A., Mot, I. C., Horhat, D. I., Balica, N. C., Sarau, C. A., Morar, R., Domuta, E. M., Dumitru, C., Negrean, R. A., Bumbu, B. A., Ravulapalli, M., Alambaram, S., Akshay, R., & Pricop, M. (2022). Development and Preliminary Characterization of Polyester-Urethane Microparticles Used in Curcumin Drug Delivery System for Oropharyngeal Cancer. Medicina, 58(11), 1689. https://doi.org/10.3390/medicina58111689








