Interplay of Gut Microbiota in Polycystic Ovarian Syndrome: Role of Gut Microbiota, Mechanistic Pathways and Potential Treatment Strategies
Abstract
1. Introduction
2. Gut Microbiota and the Endocrine System
3. Role of Gut Microbiota in PCOS
3.1. Bacteria Involved in PCOS
3.1.1. Firmicutes
3.1.2. Actinobacteria
3.1.3. Proteobacteria
3.1.4. Bacteroidetes
3.2. Fungi Involved in PCOS
3.3. Virus Involved in PCOS
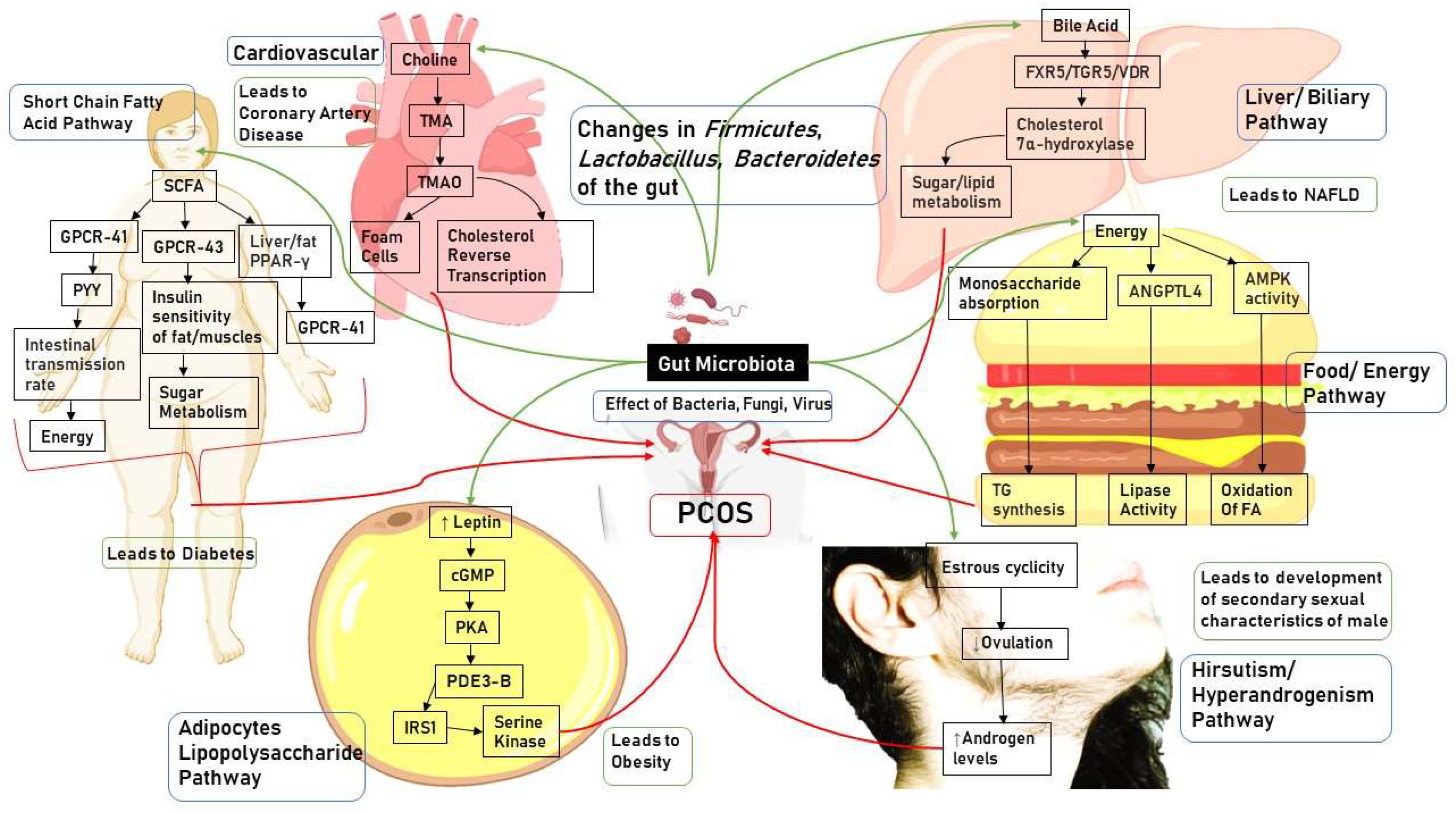
4. Mechanistic Pathways in PCOS
4.1. Correlation between Gut Microbiota and Hyperandrogenism
4.2. Correlation between Gut Microbiota and Energy Absorption
4.3. Correlation between Gut Microbiota and Short Chain Fatty Acid Metabolism
4.4. Correlation between Gut Microbiota and Lipopolysaccharide (LPS) Metabolism
4.5. Correlation between Gut Microbiota and Choline Pathway
4.6. Correlation between Gut Microbiota and Bile Acid Pathway
4.7. Correlation between Gut Microbiota and Intestinal Permeability
4.8. Correlation between Gut-Brain Axis
5. Potential Treatment Strategies for PCOS
5.1. Fecal Microbiota Transplant (FMT)
5.2. Prebiotics
5.3. Probiotics
5.4. Polyphenols
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Witchel, S.F.; Oberfield, S.E.; Pena, A.S. Polycystic Ovary Syndrome: Pathophysiology, Presentation, and Treatment with Emphasis on Adolescent Girls. J. Endocr. Soc. 2019, 3, 1545–1573. [Google Scholar] [CrossRef] [PubMed]
- Wolf, W.M.; Wattick, R.A.; Kinkade, O.N.; Olfert, M.D. Geographical Prevalence of Polycystic Ovary Syndrome as Determined by Region and Race/Ethnicity. Int. J. Env. Res. Public Health 2018, 15. [Google Scholar] [CrossRef] [PubMed]
- Abdalla, M.A.; Deshmukh, H.; Atkin, S.; Sathyapalan, T. A review of therapeutic options for managing the metabolic aspects of polycystic ovary syndrome. Adv. Endocrinol Metab 2020, 11, 2042018820938305. [Google Scholar] [CrossRef] [PubMed]
- Thursby, E.; Juge, N. Introduction to the human gut microbiota. Biochem. J. 2017, 474, 1823–1836. [Google Scholar] [CrossRef] [PubMed]
- Parada Venegas, D.; De la Fuente, M.K.; Landskron, G.; Gonzalez, M.J.; Quera, R.; Dijkstra, G.; Harmsen, H.J.M.; Faber, K.N.; Hermoso, M.A. Short Chain Fatty Acids (SCFAs)-Mediated Gut Epithelial and Immune Regulation and Its Relevance for Inflammatory Bowel Diseases. Front. Immunol. 2019, 10, 277. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Li, X.; Yang, F.; Zhao, R.; Pan, X.; Liang, J.; Tian, L.; Li, X.; Liu, L.; Xing, Y.; et al. Gut Microbiota-Dependent Marker TMAO in Promoting Cardiovascular Disease: Inflammation Mechanism, Clinical Prognostic, and Potential as a Therapeutic Target. Front Pharm. 2019, 10, 1360. [Google Scholar] [CrossRef]
- Rowland, I.; Gibson, G.; Heinken, A.; Scott, K.; Swann, J.; Thiele, I.; Tuohy, K. Gut microbiota functions: Metabolism of nutrients and other food components. Eur. J. Nutr. 2018, 57, 1–24. [Google Scholar] [CrossRef]
- Carding, S.; Verbeke, K.; Vipond, D.T.; Corfe, B.M.; Owen, L.J. Dysbiosis of the gut microbiota in disease. Microb Ecol. Health Dis. 2015, 26, 26191. [Google Scholar] [CrossRef]
- Corrie, L.; Gulati, M.; Vishwas, S.; Kapoor, B.; Singh, S.K.; Awasthi, A.; Khursheed, R. Combination therapy of curcumin and fecal microbiota transplant: Potential treatment of polycystic ovarian syndrome. Med. Hypotheses 2021, 154, 110644. [Google Scholar] [CrossRef]
- Fukui, H.; Xu, X.; Miwa, H. Role of Gut Microbiota-Gut Hormone Axis in the Pathophysiology of Functional Gastrointestinal Disorders. J. Neurogastroenterol Motil 2018, 24, 367–386. [Google Scholar] [CrossRef]
- Zhang, G.; Meredith, T.C.; Kahne, D.; Kahne, D. On the essentiality of lipopolysaccharide to Gram-negative bacteria. Curr. Opin. Microbiol. 2013, 16, 779–785. [Google Scholar] [PubMed]
- Kho, Z.Y.; Lal, S.K. The Human Gut Microbiome—A Potential Controller of Wellness and Disease. Front. Microbiol 2018, 9, 1835. [Google Scholar] [CrossRef] [PubMed]
- He, F.F.; Li, Y.A.-O. Role of gut microbiota in the development of insulin resistance and the mechanism underlying polycystic ovary syndrome: A review. J. Ovarian Res. 2020, 13, 73–76. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, J.A.; Blaser, M.J.; Caporaso, J.G.; Jansson, J.K.; Lynch, S.V.; Knight, R. Current understanding of the human microbiome. Nat. Med. 2018, 24, 392–400. [Google Scholar] [CrossRef]
- Clarke, G.; Stilling, R.M.; Kennedy, P.J.; Stanton, C.; Cryan, J.F.; Dinan, T.G. Minireview: Gut microbiota: The neglected endocrine organ. Mol. Endocrinol 2014, 28, 1221–1238. [Google Scholar] [CrossRef]
- Gomez, A.; Luckey, D.; Taneja, V. The gut microbiome in autoimmunity: Sex matters. Clin. Immunol. 2015, 159, 154–162. [Google Scholar] [CrossRef]
- Kim, Y.S.; Unno, T.; Kim, B.Y.; Park, M.S. Sex Differences in Gut Microbiota. World J. Mens Health 2020, 38, 48–60. [Google Scholar] [CrossRef]
- He, S.; Li, H.; Yu, Z.; Zhang, F.; Liang, S.; Liu, H.; Chen, H.; Lu, M. The Gut Microbiome and Sex Hormone-Related Diseases. Front. Microbiol 2021, 12, 711137. [Google Scholar] [CrossRef]
- Santos-Marcos, J.A.; Rangel-Zuniga, O.A.; Jimenez-Lucena, R.; Quintana-Navarro, G.M.; Garcia-Carpintero, S.; Malagon, M.M.; Landa, B.B.; Tena-Sempere, M.; Perez-Martinez, P.; Lopez-Miranda, J.; et al. Influence of gender and menopausal status on gut microbiota. Maturitas 2018, 116, 43–53. [Google Scholar] [CrossRef]
- Mayneris-Perxachs, J.; Arnoriaga-Rodriguez, M.; Luque-Cordoba, D.; Priego-Capote, F.; Perez-Brocal, V.; Moya, A.; Burokas, A.; Maldonado, R.; Fernandez-Real, J.M. Gut microbiota steroid sexual dimorphism and its impact on gonadal steroids: Influences of obesity and menopausal status. Microbiome 2020, 8, 136. [Google Scholar] [CrossRef]
- Flores, R.; Shi, J.; Fuhrman, B.; Xu, X.; Veenstra, T.D.; Gail, M.H.; Gajer, P.; Ravel, J.; Goedert, J.J. Fecal microbial determinants of fecal and systemic estrogens and estrogen metabolites: A cross-sectional study. J. Transl. Med. 2012, 10, 253. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.H.; Park, Y.H.; Sim, M.; Kim, S.A.; Joung, H.; Shin, D.M. Serum level of sex steroid hormone is associated with diversity and profiles of human gut microbiome. Res. Microbiol 2019, 170, 192–201. [Google Scholar] [CrossRef] [PubMed]
- Mihajlovic, J.; Leutner, M.; Hausmann, B.; Kohl, G.; Schwarz, J.; Rover, H.; Stimakovits, N.; Wolf, P.; Maruszczak, K.; Bastian, M.; et al. Combined hormonal contraceptives are associated with minor changes in composition and diversity in gut microbiota of healthy women. Env. Microbiol 2021, 23, 3037–3047. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; Powell, J.E.; Steele, M.I.; Dietrich, C.; Moran, N.A. Honeybee gut microbiota promotes host weight gain via bacterial metabolism and hormonal signaling. Proc. Natl. Acad. Sci. USA 2017, 114, 4775–4780. [Google Scholar] [CrossRef]
- Song, J.; Zhou, N.; Ma, W.; Gu, X.; Chen, B.; Zeng, Y.; Yang, L.; Zhou, M. Modulation of gut microbiota by chlorogenic acid pretreatment on rats with adrenocorticotropic hormone induced depression-like behavior. Food Funct. 2019, 10, 2947–2957. [Google Scholar] [CrossRef]
- Li, J.W.; Fang, B.; Pang, G.F.; Zhang, M.; Ren, F.Z. Age- and diet-specific effects of chronic exposure to chlorpyrifos on hormones, inflammation and gut microbiota in rats. Pestic Biochem. Physiol 2019, 159, 68–79. [Google Scholar] [CrossRef]
- Lee, J.; Kim, C.H.; Jang, H.A.; Kim, J.K.; Kotaki, T.; Shinoda, T.; Shinada, T.; Yoo, J.W.; Lee, B.L. Burkholderia gut symbiont modulates titer of specific juvenile hormone in the bean bug Riptortus pedestris. Dev. Comp Immunol. 2019, 99, 103399. [Google Scholar] [CrossRef]
- Coklo, M.; Maslov, D.R.; Kraljevic Pavelic, S. Modulation of gut microbiota in healthy rats after exposure to nutritional supplements. Gut Microbes 2020, 12, 1–28. [Google Scholar] [CrossRef]
- Boyanova, L. Stress hormone epinephrine (adrenaline) and norepinephrine (noradrenaline) effects on the anaerobic bacteria. Anaerobe 2017, 44, 13–19. [Google Scholar] [CrossRef]
- Knezevic, J.; Starchl, C.; Tmava Berisha, A.; Amrein, K. Thyroid-Gut-Axis: How Does the Microbiota Influence Thyroid Function? Nutrients 2020, 12. [Google Scholar] [CrossRef]
- He, M.; Gao, J.; Wu, J.; Zhou, Y.; Fu, H.; Ke, S.; Yang, H.; Chen, C.; Huang, L. Host Gender and Androgen Levels Regulate Gut Bacterial Taxa in Pigs Leading to Sex-Biased Serum Metabolite Profiles. Front. Microbiol 2019, 10, 1359. [Google Scholar] [CrossRef] [PubMed]
- Sherman, S.B.; Sarsour, N.; Salehi, M.; Schroering, A.; Mell, B.; Joe, B.; Hill, J.W. Prenatal androgen exposure causes hypertension and gut microbiota dysbiosis. Gut Microbes 2018, 9, 400–421. [Google Scholar] [CrossRef]
- Yadav, H.; Lee, J.H.; Lloyd, J.; Walter, P.; Rane, S.G. Beneficial metabolic effects of a probiotic via butyrate-induced GLP-1 hormone secretion. J. Biol. Chem. 2013, 288, 25088–25097. [Google Scholar] [CrossRef] [PubMed]
- Messaoudi, M.; Lalonde, R.; Violle, N.; Javelot, H.; Desor, D.; Nejdi, A.; Bisson, J.F.; Rougeot, C.; Pichelin, M.; Cazaubiel, M.; et al. Assessment of psychotropic-like properties of a probiotic formulation (Lactobacillus helveticus R0052 and Bifidobacterium longum R0175) in rats and human subjects. Br. J. Nutr. 2011, 105, 755–764. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Bao, Y.; Zhou, X.; Zheng, L. Polycystic ovary syndrome and mitochondrial dysfunction. Reprod Biol. Endocrinol 2019, 17, 67. [Google Scholar] [CrossRef]
- Corrie, L.; Gulati, M.; Singh, S.K.; Kapoor, B.; Khursheed, R.; Awasthi, A.; Vishwas, S.; Chellappan, D.K.; Gupta, G.; Jha, N.K.; et al. Recent updates on animal models for understanding the etiopathogenesis of polycystic ovarian syndrome. Life Sci. 2021, 280, 119753. [Google Scholar] [CrossRef]
- Rizk, M.G.; Thackray, V.G. Intersection of Polycystic Ovary Syndrome and the Gut Microbiome. J. Endocr. Soc. 2021, 5, bvaa177. [Google Scholar] [CrossRef]
- Bradford, L.L.; Ravel, J. The vaginal mycobiome: A poorly understood contributor to women’s health and diseases. Virulence 2017, 8, 342–351. [Google Scholar]
- Minot, S.; Sinha, R.; Chen, J.; Li, H.; Keilbaugh, S.A.; Wu, G.D.; Lewis, J.D.; Bushman, F.D. The human gut virome: Inter-individual variation and dynamic response to diet. Genome Res. 2011, 21, 1616–1625. [Google Scholar] [CrossRef]
- King, C.H.; Desai, H.; Sylvetsky, A.C.; LoTempio, J.; Ayanyan, S.; Carrie, J.; Crandall, K.A.; Fochtman, B.C.; Gasparyan, L.; Gulzar, N.; et al. Baseline human gut microbiota profile in healthy people and standard reporting template. PLoS ONE 2019, 14, e0206484. [Google Scholar] [CrossRef]
- Rinninella, E.; Raoul, P.; Cintoni, M.; Franceschi, F.; Miggiano, G.A.D.; Gasbarrini, A.; Mele, M.C. What is the Healthy Gut Microbiota Composition? A Changing Ecosystem across Age, Environment, Diet, and Diseases. Microorganisms 2019, 7. [Google Scholar] [CrossRef]
- Magne, F.; Gotteland, M.; Gauthier, L.; Zazueta, A.; Pesoa, S.; Navarrete, P.; Balamurugan, R. The Firmicutes/Bacteroidetes Ratio: A Relevant Marker of Gut Dysbiosis in Obese Patients? Nutrients 2020, 12. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Zhang, C.; Shi, Y.; Zhang, F.; Li, L.; Wang, X.; Ling, Y.; Fu, H.; Dong, W.; Shen, J.; et al. Dysbiosis of Gut Microbiota Associated with Clinical Parameters in Polycystic Ovary Syndrome. Front. Microbiol 2017, 8, 324. [Google Scholar] [CrossRef]
- Kelley, S.T.; Skarra, D.V.; Rivera, A.J.; Thackray, V.G. The Gut Microbiome Is Altered in a Letrozole-Induced Mouse Model of Polycystic Ovary Syndrome. PLoS ONE 2016, 11, e0146509. [Google Scholar] [CrossRef] [PubMed]
- Insenser, M.; Murri, M.; Del Campo, R.; Martinez-Garcia, M.A.; Fernandez-Duran, E.; Escobar-Morreale, H.F. Gut Microbiota and the Polycystic Ovary Syndrome: Influence of Sex, Sex Hormones, and Obesity. J. Clin. Endocrinol Metab 2018, 103, 2552–2562. [Google Scholar] [CrossRef]
- Cao, S.-Y.; Zhao, C.-N.; Xu, X.-Y.; Tang, G.-Y.; Corke, H.; Gan, R.-Y.; Li, H.-B. Dietary plants, gut microbiota, and obesity: Effects and mechanisms. Trends Food Sci. Technol. 2019, 92, 194–204. [Google Scholar] [CrossRef]
- Le Roy, C.I.; Štšepetova, J.; Sepp, E.; Songisepp, E.; Claus, S.P.; Mikelsaar, M. New insights into the impact of Lactobacillus population on host-bacteria metabolic interplay. Oncotarget 2015, 6, 30545–30556. [Google Scholar] [CrossRef]
- He, Y.; Wang, Q.; Li, X.; Wang, G.; Zhao, J.; Zhang, H.; Chen, W. Lactic acid bacteria alleviate polycystic ovarian syndrome by regulating sex hormone related gut microbiota. Food Funct. 2020, 11, 5192–5204. [Google Scholar] [CrossRef]
- Saez-Lara, M.J.; Robles-Sanchez, C.; Ruiz-Ojeda, F.J.; Plaza-Diaz, J.; Gil, A. Effects of Probiotics and Synbiotics on Obesity, Insulin Resistance Syndrome, Type 2 Diabetes and Non-Alcoholic Fatty Liver Disease: A Review of Human Clinical Trials. Int. J. Mol. Sci. 2016, 17. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Qi, Y.; Yang, X.; Zhao, L.; Wen, S.; Liu, Y.; Tang, L. Association between Polycystic Ovary Syndrome and Gut Microbiota. PLoS ONE 2016, 11, e0153196. [Google Scholar] [CrossRef] [PubMed]
- Mazidi, M.; Rezaie, P.; Ferns, G.A.; Vatanparast, H. Impact of Probiotic Administration on Serum C-Reactive Protein Concentrations: Systematic Review and Meta-Analysis of Randomized Control Trials. Nutrients 2017, 9. [Google Scholar] [CrossRef] [PubMed]
- He, B.; Hoang, T.K.; Tian, X.; Taylor, C.M.; Blanchard, E.; Luo, M.; Bhattacharjee, M.B.; Freeborn, J.; Park, S.; Couturier, J.; et al. Lactobacillus reuteri Reduces the Severity of Experimental Autoimmune Encephalomyelitis in Mice by Modulating Gut Microbiota. Front. Immunol. 2019, 10, 385. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.H.; Chen, Y.H.; Cheng, K.C.; Chien, T.Y.; Chan, C.H.; Tsao, S.P.; Huang, H.Y. Antiobesity effect of Lactobacillus reuteri 263 associated with energy metabolism remodeling of white adipose tissue in high-energy-diet-fed rats. J. Nutr. Biochem 2018, 54, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Laitinen, K.; Poussa, T.; Isolauri, E.; Nutrition, A.M.I.; Intestinal Microbiota, G. Probiotics and dietary counselling contribute to glucose regulation during and after pregnancy: A randomised controlled trial. Br. J. Nutr. 2009, 101, 1679–1687. [Google Scholar] [CrossRef] [PubMed]
- Andreasen, A.S.; Larsen, N.; Pedersen-Skovsgaard, T.; Berg, R.M.; Moller, K.; Svendsen, K.D.; Jakobsen, M.; Pedersen, B.K. Effects of Lactobacillus acidophilus NCFM on insulin sensitivity and the systemic inflammatory response in human subjects. Br. J. Nutr. 2010, 104, 1831–1838. [Google Scholar] [CrossRef]
- Kim, S.Y.; Shin, J.S.; Chung, K.S.; Han, H.S.; Lee, H.H.; Lee, J.H.; Kim, S.Y.; Ji, Y.W.; Ha, Y.; Kang, J.; et al. Immunostimulatory Effects of Live Lactobacillus sakei K040706 on the CYP-Induced Immunosuppression Mouse Model. Nutrients 2020, 12. [Google Scholar] [CrossRef]
- Chung, H.J.; Yu, J.G.; Lee, I.A.; Liu, M.J.; Shen, Y.F.; Sharma, S.P.; Jamal, M.A.; Yoo, J.H.; Kim, H.J.; Hong, S.T. Intestinal removal of free fatty acids from hosts by Lactobacilli for the treatment of obesity. FEBS Open Bio 2016, 6, 64–76. [Google Scholar] [CrossRef]
- Kim, J.; Yun, J.M.; Kim, M.K.; Kwon, O.; Cho, B. Lactobacillus gasseri BNR17 Supplementation Reduces the Visceral Fat Accumulation and Waist Circumference in Obese Adults: A Randomized, Double-Blind, Placebo-Controlled Trial. J. Med. Food 2018, 21, 454–461. [Google Scholar] [CrossRef]
- Nasri, K.; Jamilian, M.; Rahmani, E.; Bahmani, F.; Tajabadi-Ebrahimi, M.; Asemi, Z. The effects of synbiotic supplementation on hormonal status, biomarkers of inflammation and oxidative stress in subjects with polycystic ovary syndrome: A randomized, double-blind, placebo-controlled trial. BMC Endocr Disord 2018, 18, 21. [Google Scholar] [CrossRef]
- Jamilian, M.; Mansury, S.; Bahmani, F.; Heidar, Z.; Amirani, E.; Asemi, Z. The effects of probiotic and selenium co-supplementation on parameters of mental health, hormonal profiles, and biomarkers of inflammation and oxidative stress in women with polycystic ovary syndrome. J. Ovarian Res. 2018, 11, 80. [Google Scholar] [CrossRef] [PubMed]
- Shabani, A.; Noshadian, M.; Jamilian, M.; Chamani, M.; Mohammadi, S.; Asemi, Z. The effects of a novel combination of selenium and probiotic on weight loss, glycemic control and markers of cardio-metabolic risk in women with polycystic ovary syndrome. J. Funct. Foods 2018, 46, 329–334. [Google Scholar] [CrossRef]
- Valenlia, K.B.; Morshedi, M.; Saghafi-Asl, M.; Shahabi, P.; Abbasi, M.M. Beneficial impacts of Lactobacillus plantarum and inulin on hypothalamic levels of insulin, leptin, and oxidative markers in diabetic rats. J. Funct. Foods 2018, 46, 529–537. [Google Scholar] [CrossRef]
- Ooi, L.G.; Liong, M.T. Cholesterol-lowering effects of probiotics and prebiotics: A review of in vivo and in vitro findings. Int. J. Mol. Sci. 2010, 11, 2499–2522. [Google Scholar] [CrossRef] [PubMed]
- Binda, C.; Lopetuso, L.R.; Rizzatti, G.; Gibiino, G.; Cennamo, V.; Gasbarrini, A. Actinobacteria: A relevant minority for the maintenance of gut homeostasis. Dig. Liver Dis. 2018, 50, 421–428. [Google Scholar] [CrossRef] [PubMed]
- Shivlata, L.; Satyanarayana, T. Thermophilic and alkaliphilic Actinobacteria: Biology and potential applications. Front. Microbiol 2015, 6, 1014. [Google Scholar] [CrossRef] [PubMed]
- Arboleya, S.; Watkins, C.; Stanton, C.; Ross, R.P. Gut Bifidobacteria Populations in Human Health and Aging. Front. Microbiol 2016, 7, 1204. [Google Scholar] [CrossRef] [PubMed]
- Ventura, M.; Canchaya, C.; Tauch, A.; Chandra, G.; Fitzgerald, G.F.; Chater, K.F.; van Sinderen, D. Genomics of Actinobacteria: Tracing the evolutionary history of an ancient phylum. Microbiol Mol. Biol. Rev. 2007, 71, 495–548. [Google Scholar] [CrossRef]
- Gago, G.; Diacovich, L.; Arabolaza, A.; Tsai, S.C.; Gramajo, H. Fatty acid biosynthesis in actinomycetes. FEMS Microbiol Rev. 2011, 35, 475–497. [Google Scholar] [CrossRef]
- Stenman, L.K.; Waget, A.; Garret, C.; Klopp, P.; Burcelin, R.; Lahtinen, S. Potential probiotic Bifidobacterium animalis ssp. lactis 420 prevents weight gain and glucose intolerance in diet-induced obese mice. Benef Microbes 2014, 5, 437–445. [Google Scholar] [CrossRef]
- Moya-Perez, A.; Neef, A.; Sanz, Y. Bifidobacterium pseudocatenulatum CECT 7765 Reduces Obesity-Associated Inflammation by Restoring the Lymphocyte-Macrophage Balance and Gut Microbiota Structure in High-Fat Diet-Fed Mice. PLoS ONE 2015, 10, e0126976. [Google Scholar] [CrossRef] [PubMed]
- Underwood, M.A.; German, J.B.; Lebrilla, C.B.; Mills, D.A. Bifidobacterium longum subspecies infantis: Champion colonizer of the infant gut. Pediatr Res. 2015, 77, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Groeger, D.; O’Mahony, L.; Murphy, E.F.; Bourke, J.F.; Dinan, T.G.; Kiely, B.; Shanahan, F.; Quigley, E.M. Bifidobacterium infantis 35624 modulates host inflammatory processes beyond the gut. Gut Microbes 2013, 4, 325–339. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Xie, J.P.; Deng, K.; Li, X.; Yuan, Y.; Xuan, Q.; Xie, J.; He, X.M.; Wang, Q.; Li, J.J.; et al. Prophylactic Effects of Bifidobacterium adolescentis on Anxiety and Depression-Like Phenotypes After Chronic Stress: A Role of the Gut Microbiota-Inflammation Axis. Front. Behav. Neurosci. 2019, 13, 126. [Google Scholar] [CrossRef]
- Moya-Perez, A.; Romo-Vaquero, M.; Tomas-Barberan, F.; Sanz, Y.; Garcia-Conesa, M.T. Hepatic molecular responses to Bifidobacterium pseudocatenulatum CECT 7765 in a mouse model of diet-induced obesity. Nutr. Metab Cardiovasc Dis. 2014, 24, 57–64. [Google Scholar] [CrossRef]
- Li, Z.; Jin, H.; Oh, S.Y.; Ji, G.E. Anti-obese effects of two Lactobacilli and two Bifidobacteria on ICR mice fed on a high fat diet. Biochem. Biophys Res. Commun. 2016, 480, 222–227. [Google Scholar] [CrossRef]
- Ray, M.; Hor, P.K.; Ojha, D.; Soren, J.P.; Singh, S.N.; Mondal, K.C. Bifidobacteria and its rice fermented products on diet induced obese mice: Analysis of physical status, serum profile and gene expressions. Benef Microbes 2018, 9, 441–452. [Google Scholar] [CrossRef]
- Minami, J.; Kondo, S.; Yanagisawa, N.; Odamaki, T.; Xiao, J.Z.; Abe, F.; Nakajima, S.; Hamamoto, Y.; Saitoh, S.; Shimoda, T. Oral administration of Bifidobacterium breve B-3 modifies metabolic functions in adults with obese tendencies in a randomised controlled trial. J. Nutr. Sci. 2015, 4, e17. [Google Scholar] [CrossRef]
- Zhang, J.; Sun, Z.; Jiang, S.; Bai, X.; Ma, C.; Peng, Q.; Chen, K.; Chang, H.; Fang, T.; Zhang, H. Probiotic Bifidobacterium lactis V9 Regulates the Secretion of Sex Hormones in Polycystic Ovary Syndrome Patients through the Gut-Brain Axis. mSystems 2019, 4. [Google Scholar] [CrossRef]
- Rizzatti, G.; Lopetuso, L.R.; Gibiino, G.; Binda, C.; Gasbarrini, A. Proteobacteria: A Common Factor in Human Diseases. Biomed. Res. Int. 2017, 2017, 9351507. [Google Scholar] [CrossRef]
- Amabebe, E.; Anumba, D.O.C. Female Gut and Genital Tract Microbiota-Induced Crosstalk and Differential Effects of Short-Chain Fatty Acids on Immune Sequelae. Front. Immunol. 2020, 11, 2184. [Google Scholar] [CrossRef] [PubMed]
- Savini, V.; Catavitello, C.; Talia, M.; Manna, A.; Pompetti, F.; Favaro, M.; Fontana, C.; Febbo, F.; Balbinot, A.; Di Berardino, F.; et al. Multidrug-resistant Escherichia fergusonii: A case of acute cystitis. J. Clin. Microbiol. 2008, 46, 1551–1552. [Google Scholar] [CrossRef] [PubMed]
- Flores-Mireles, A.; Hreha, T.N.; Hunstad, D.A. Pathophysiology, Treatment, and Prevention of Catheter-Associated Urinary Tract Infection. Top Spinal Cord Inj. Rehabil 2019, 25, 228–240. [Google Scholar] [CrossRef] [PubMed]
- Gantois, I.; Ducatelle, R.; Pasmans, F.; Haesebrouck, F.; Gast, R.; Humphrey, T.J.; Van Immerseel, F. Mechanisms of egg contamination by Salmonella Enteritidis. FEMS Microbiol Rev. 2009, 33, 718–738. [Google Scholar] [CrossRef]
- Matsumoto, Y.; Miwa, S.; Zhang, Y.; Hiroshima, Y.; Yano, S.; Uehara, F.; Yamamoto, M.; Toneri, M.; Bouvet, M.; Matsubara, H.; et al. Efficacy of tumor-targeting Salmonella typhimurium A1-R on nude mouse models of metastatic and disseminated human ovarian cancer. J. Cell Biochem 2014, 115, 1996–2003. [Google Scholar] [CrossRef] [PubMed]
- Yavasoglu, I.; Kucuk, M.; Cildag, B.; Arslan, E.; Gok, M.; Kafkas, S. A novel association between polycystic ovary syndrome and Helicobacter pylori. Am. J. Med. Sci. 2009, 338, 174–177. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Liu, W.; Zheng, B. Complete genome sequencing of Comamonas kerstersii 8943, a causative agent for peritonitis. Sci. Data 2018, 5, 180222. [Google Scholar] [CrossRef]
- Almuzara, M.; Cittadini, R.; Estraviz, M.L.; Ellis, A.; Vay, C. First report of Comamonas kerstersii causing urinary tract infection. New Microbes New Infect 2018, 24, 4–7. [Google Scholar] [CrossRef]
- Chu, W.; Han, Q.; Xu, J.; Wang, J.; Sun, Y.; Li, W.; Chen, Z.J.; Du, Y. Metagenomic analysis identified microbiome alterations and pathological association between intestinal microbiota and polycystic ovary syndrome. Fertil Steril 2020, 113, 1286–1298.e1284. [Google Scholar] [CrossRef]
- Mattock, E.; Blocker, A.J. How Do the Virulence Factors of Shigella Work Together to Cause Disease? Front. Cell Infect Microbiol 2017, 7, 64. [Google Scholar] [CrossRef]
- Wang, T.; Sha, L.; Li, Y.; Zhu, L.; Wang, Z.; Li, K.; Lu, H.; Bao, T.; Guo, L.; Zhang, X.; et al. Dietary alpha-Linolenic Acid-Rich Flaxseed Oil Exerts Beneficial Effects on Polycystic Ovary Syndrome Through Sex Steroid Hormones-Microbiota-Inflammation Axis in Rats. Front. Endocrinol 2020, 11, 284. [Google Scholar] [CrossRef]
- Gibiino, G.; Lopetuso, L.R.; Scaldaferri, F.; Rizzatti, G.; Binda, C.; Gasbarrini, A. Exploring Bacteroidetes: Metabolic key points and immunological tricks of our gut commensals. Dig. Liver Dis. 2018, 50, 635–639. [Google Scholar] [CrossRef] [PubMed]
- Johnson, E.L.; Heaver, S.L.; Walters, W.A.; Ley, R.E. Microbiome and metabolic disease: Revisiting the bacterial phylum Bacteroidetes. J. Mol. Med. 2017, 95, 1–8. [Google Scholar] [CrossRef]
- Lindheim, L.; Bashir, M.; Munzker, J.; Trummer, C.; Zachhuber, V.; Leber, B.; Horvath, A.; Pieber, T.R.; Gorkiewicz, G.; Stadlbauer, V.; et al. Alterations in Gut Microbiome Composition and Barrier Function Are Associated with Reproductive and Metabolic Defects in Women with Polycystic Ovary Syndrome (PCOS): A Pilot Study. PLoS ONE 2017, 12, e0168390. [Google Scholar] [CrossRef] [PubMed]
- Arroyo, P.; Ho, B.S.; Sau, L.; Kelley, S.T.; Thackray, V.G. Letrozole treatment of pubertal female mice results in activational effects on reproduction, metabolism and the gut microbiome. PLoS ONE 2019, 14, e0223274. [Google Scholar] [CrossRef]
- Dugas, L.R.; Bernabe, B.P.; Priyadarshini, M.; Fei, N.; Park, S.J.; Brown, L.; Plange-Rhule, J.; Nelson, D.; Toh, E.C.; Gao, X.; et al. Decreased microbial co-occurrence network stability and SCFA receptor level correlates with obesity in African-origin women. Sci. Rep. 2018, 8, 17135. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Yu, J.; Liang, C.; Li, S.; Wen, X.; Li, Y. Characterization on gut microbiome of PCOS rats and its further design by shifts in high-fat diet and dihydrotestosterone induction in PCOS rats. Bioprocess Biosyst Eng. 2021, 44, 953–964. [Google Scholar] [CrossRef] [PubMed]
- Underhill, D.M.; Iliev, I.D. The mycobiota: Interactions between commensal fungi and the host immune system. Nat. Rev. Immunol. 2014, 14, 405–416. [Google Scholar] [CrossRef] [PubMed]
- Huffnagle, G.B.; Noverr, M.C. The emerging world of the fungal microbiome. Trends Microbiol 2013, 21, 334–341. [Google Scholar] [CrossRef] [PubMed]
- Hamad, I.; Sokhna, C.; Raoult, D.; Bittar, F. Molecular detection of eukaryotes in a single human stool sample from Senegal. PLoS ONE 2012, 7, e40888. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudi, E.; Mozhgani, S.H.; Sharifinejad, N. The role of mycobiota-genotype association in inflammatory bowel diseases: A narrative review. Gut Pathog 2021, 13, 31. [Google Scholar] [CrossRef] [PubMed]
- Iliev, I.D.; Funari, V.A.; Taylor, K.D.; Nguyen, Q.; Reyes, C.N.; Strom, S.P.; Brown, J.; Becker, C.A.; Fleshner, P.R.; Dubinsky, M.; et al. Interactions between commensal fungi and the C-type lectin receptor Dectin-1 influence colitis. Science 2012, 336, 1314–1317. [Google Scholar] [CrossRef]
- Mar Rodriguez, M.; Perez, D.; Javier Chaves, F.; Esteve, E.; Marin-Garcia, P.; Xifra, G.; Vendrell, J.; Jove, M.; Pamplona, R.; Ricart, W.; et al. Obesity changes the human gut mycobiome. Sci. Rep. 2015, 5, 14600. [Google Scholar] [CrossRef]
- Ott, S.J.; Kuhbacher, T.; Musfeldt, M.; Rosenstiel, P.; Hellmig, S.; Rehman, A.; Drews, O.; Weichert, W.; Timmis, K.N.; Schreiber, S. Fungi and inflammatory bowel diseases: Alterations of composition and diversity. Scand J. Gastroenterol 2008, 43, 831–841. [Google Scholar] [CrossRef]
- Gosiewski, T. Quantitative evaluation of fungi of the genus Candida in the feces of adult patients with type 1 and 2 diabetes—A pilot study. Gut Pathog. 2014, 6. [Google Scholar] [CrossRef]
- Cui, L.; Morris, A.; Ghedin, E. The human mycobiome in health and disease. Genome Med. 2013, 5. [Google Scholar] [CrossRef]
- Scarpellini, E.; Ianiro, G.; Attili, F.; Bassanelli, C.; De Santis, A.; Gasbarrini, A. The human gut microbiota and virome: Potential therapeutic implications. Dig Liver Dis 2015, 47, 1007–1012. [Google Scholar] [CrossRef] [PubMed]
- Kramná, L.; Kolarova, K.; Oikarinen, S.; Pursiheimo, J.P.; Ilonen, J.; Simell, O.; Knip, M.; Veijola, R.; Hyöty, H.; Cinek, O. Gut virome sequencing in children with early islet autoimmunity. Diabetes Care 2015, 38. [Google Scholar] [CrossRef]
- Foca, A.; Liberto, M.C.; Quirino, A.; Marascio, N.; Zicca, E.; Pavia, G. Gut inflammation and immunity: What is the role of the human gut virome? Mediat. Inflamm 2015, 2015, 326032. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.Y.; Kim, M.S.; Kim, E.; Cheon, J.H.; Lee, Y.S.; Kim, Y.; Lee, S.H.; Seo, S.U.; Shin, S.H.; Choi, S.S.; et al. Enteric Viruses Ameliorate Gut Inflammation via Toll-like Receptor 3 and Toll-like Receptor 7-Mediated Interferon-beta Production. Immunity 2016, 44, 889–900. [Google Scholar] [CrossRef] [PubMed]
- Corrie, L.; Gulati, M.; Awasthi, A.; Vishwas, S.; Kaur, J.; Khursheed, R.; Kumar, R.; Kumar, A.; Imran, M.; Chellappan, D.K.; et al. Polysaccharide, fecal microbiota, and curcumin-based novel oral colon-targeted solid self-nanoemulsifying delivery system: Formulation, characterization, and in-vitro anticancer evaluation. Mater. Today Chem. 2022, 26, 101165. [Google Scholar] [CrossRef]
- Tremellen, K.; Pearce, K. Dysbiosis of Gut Microbiota (DOGMA)--a novel theory for the development of Polycystic Ovarian Syndrome. Med Hypotheses 2012, 79, 104–112. [Google Scholar] [CrossRef]
- Qi, X.; Yun, C.; Sun, L.; Xia, J.; Wu, Q.; Wang, Y.; Wang, L.; Zhang, Y.; Liang, X.; Wang, L.; et al. Gut microbiota-bile acid-interleukin-22 axis orchestrates polycystic ovary syndrome. Nat. Med. 2019, 25, 1225–1233. [Google Scholar] [CrossRef]
- Thackray, V.G. Sex, Microbes, and Polycystic Ovary Syndrome. Trends Endocrinol Metab. 2019, 30, 54–65. [Google Scholar] [CrossRef] [PubMed]
- Vom Steeg, L.G.; Klein, S.L. Sex Steroids Mediate Bidirectional Interactions Between Hosts and Microbes. Horm Behav. 2017, 88, 45–51. [Google Scholar] [CrossRef]
- Zhou, L.; Ni, Z.; Cheng, W.; Yu, J.; Sun, S.; Zhai, D.; Yu, C.; Cai, Z. Characteristic gut microbiota and predicted metabolic functions in women with PCOS. Endocr Connect 2020, 9, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Zeng, B.; Lai, Z.; Sun, L.; Zhang, Z.; Yang, J.; Li, Z.; Lin, J.; Zhang, Z. Structural and functional profiles of the gut microbial community in polycystic ovary syndrome with insulin resistance (IR-PCOS): A pilot study. Res. Microbiol 2019, 170, 43–52. [Google Scholar] [CrossRef]
- Torres, P.J.; Skarra, D.V.; Ho, B.S.; Sau, L.; Anvar, A.R.; Kelley, S.T.; Thackray, V.G. Letrozole treatment of adult female mice results in a similar reproductive phenotype but distinct changes in metabolism and the gut microbiome compared to pubertal mice. BMC Microbiol 2019, 19, 57. [Google Scholar] [CrossRef]
- Flint, H.J.; Scott, K.P.; Duncan, S.H.; Louis, P.; Forano, E. Microbial degradation of complex carbohydrates in the gut. Gut Microbes 2012, 3, 289–306. [Google Scholar] [CrossRef]
- Kocełak, A.Z.-G.P.; Zahorska-Markiewicz, B.; Aptekorz, M.; Zientara, M.; Martirosian, G.; Chudek, J.; Olszanecka-Glinianowicz, M. Resting energy expenditure and gut microbiota in obese and normal weight subjects. Eur. Rev. Med. Pharacol. Sci. 2013, 17, 2816–2821. [Google Scholar]
- Turnbaugh, P.J.; Ridaura, V.K.; Faith, J.J.; Rey, F.E.; Knight, R.; Gordon, J.I. The effect of diet on the human gut microbiome: A metagenomic analysis in humanized gnotobiotic mice. Sci. Transl. Med. 2009, 1, 6ra14. [Google Scholar] [CrossRef] [PubMed]
- Dadachanji, R.; Shaikh, N.; Mukherjee, S. Genetic Variants Associated with Hyperandrogenemia in PCOS Pathophysiology. Genet Res. Int. 2018, 2018, 7624932. [Google Scholar] [CrossRef]
- Torres, P.J.; Siakowska, M.; Banaszewska, B.; Pawelczyk, L.; Duleba, A.J.; Kelley, S.T.; Thackray, V.G. Gut Microbial Diversity in Women With Polycystic Ovary Syndrome Correlates With Hyperandrogenism. J. Clin. Endocrinol Metab 2018, 103, 1502–1511. [Google Scholar] [CrossRef]
- Silva, Y.P.; Bernardi, A.; Frozza, R.L. The Role of Short-Chain Fatty Acids From Gut Microbiota in Gut-Brain Communication. Front. Endocrinol 2020, 11, 25. [Google Scholar] [CrossRef]
- den Besten, G.; Lange, K.; Havinga, R.; van Dijk, T.H.; Gerding, A.; van Eunen, K.; Muller, M.; Groen, A.K.; Hooiveld, G.J.; Bakker, B.M.; et al. Gut-derived short-chain fatty acids are vividly assimilated into host carbohydrates and lipids. Am. J. Physiol. Gastrointest Liver Physiol 2013, 305, G900–G910. [Google Scholar] [CrossRef] [PubMed]
- Saad, M.J.; Santos, A.; Prada, P.O. Linking Gut Microbiota and Inflammation to Obesity and Insulin Resistance. Physiol 2016, 31, 283–293. [Google Scholar] [CrossRef]
- Lotta, L.A.; Scott, R.A.; Sharp, S.J.; Burgess, S.; Luan, J.; Tillin, T.; Schmidt, A.F.; Imamura, F.; Stewart, I.D.; Perry, J.R.; et al. Genetic Predisposition to an Impaired Metabolism of the Branched-Chain Amino Acids and Risk of Type 2 Diabetes: A Mendelian Randomisation Analysis. PLoS Med. 2016, 13, e1002179. [Google Scholar] [CrossRef] [PubMed]
- Neis, E.P.; Dejong, C.H.; Rensen, S.S. The role of microbial amino acid metabolism in host metabolism. Nutrients 2015, 7, 2930–2946. [Google Scholar] [CrossRef]
- Pedersen, H.K.; Gudmundsdottir, V.; Nielsen, H.B.; Hyotylainen, T.; Nielsen, T.; Jensen, B.A.; Forslund, K.; Hildebrand, F.; Prifti, E.; Falony, G.; et al. Human gut microbes impact host serum metabolome and insulin sensitivity. Nature 2016, 535, 376–381. [Google Scholar] [CrossRef] [PubMed]
- Engelstoft, M.S.; Schwartz, T.W. Opposite Regulation of Ghrelin and Glucagon-like Peptide-1 by Metabolite G-Protein-Coupled Receptors. Trends Endocrinol Metab 2016, 27, 665–675. [Google Scholar] [CrossRef] [PubMed]
- Novelle, M.G.; Vazquez, M.J.; Martinello, K.D.; Sanchez-Garrido, M.A.; Tena-Sempere, M.; Dieguez, C. Neonatal events, such as androgenization and postnatal overfeeding, modify the response to ghrelin. Sci. Rep. 2014, 4, 4855. [Google Scholar] [CrossRef] [PubMed]
- Gao, T.; Wu, L.; Chang, F.; Cao, G. Low circulating ghrelin levels in women with polycystic ovary syndrome: A systematic review and meta-analysis. Endocr. J. 2016, 63, 93–100. [Google Scholar] [CrossRef]
- Lin, T.L.; Shu, C.C.; Chen, Y.M.; Lu, J.J.; Wu, T.S.; Lai, W.F.; Tzeng, C.M.; Lai, H.C.; Lu, C.C. Like Cures Like: Pharmacological Activity of Anti-Inflammatory Lipopolysaccharides From Gut Microbiome. Front. Pharm. 2020, 11, 554. [Google Scholar] [CrossRef] [PubMed]
- Cani, P.D.; Amar, J.; Iglesias, M.A.; Poggi, M.; Knauf, C.; Bastelica, D.; Neyrinck, A.M.; Fava, F.; Tuohy, K.M.; Chabo, C.; et al. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes 2007, 56, 1761–1772. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Ou, H.; Wu, H.; Wu, P.; Mo, Z. Role of microRNA in the Pathogenesis of Polycystic Ovary Syndrome. DNA Cell Biol. 2019, 38, 754–762. [Google Scholar] [CrossRef] [PubMed]
- Yadav, S.; Gupta, M. A clinic biochemical study of status of fasting serum insulin and lipid profile in PCOS patients and to determine correlation between BMI and HOMA index in PCOS patients. Indian J. Obstet. Gynecol. Res. 2021, 8, 305–309. [Google Scholar] [CrossRef]
- Su, C.; Chen, M.; Huang, H.; Lin, J. Testosterone enhances lipopolysaccharide-induced interleukin-6 and macrophage chemotactic protein-1 expression by activating the extracellular signal-regulated kinase 1/2/nuclear factor-kappaB signalling pathways in 3T3-L1 adipocytes. Mol. Med. Rep. 2015, 12, 696–704. [Google Scholar] [CrossRef]
- Huang, J.; Liu, L.; Chen, C.; Gao, Y. PCOS without hyperandrogenism is associated with higher plasma Trimethylamine N-oxide levels. BMC Endocr Disord 2020, 20, 3. [Google Scholar] [CrossRef] [PubMed]
- Wild, R.A.; Carmina, E.; Diamanti-Kandarakis, E.; Dokras, A.; Escobar-Morreale, H.F.; Futterweit, W.; Lobo, R.; Norman, R.J.; Talbott, E.; Dumesic, D.A. Assessment of cardiovascular risk and prevention of cardiovascular disease in women with the polycystic ovary syndrome: A consensus statement by the Androgen Excess and Polycystic Ovary Syndrome (AE-PCOS) Society. J. Clin. Endocrinol Metab 2010, 95, 2038–2049. [Google Scholar] [CrossRef]
- Craciun, S.; Balskus, E.P. Microbial conversion of choline to trimethylamine requires a glycyl radical enzyme. Proc. Natl. Acad. Sci. USA 2012, 109, 21307–21312. [Google Scholar] [CrossRef]
- Zhu, Y.; Jameson, E.; Crosatti, M.; Schäfer, H.; Rajakumar, K.; Bugg, T.D.; Chen, Y. Carnitine metabolism to trimethylamine by an unusual Rieske-type oxygenase from human microbiota. Proc. Natl. Acad. Sci. USA 2014, 111, 4268–4273. [Google Scholar] [CrossRef]
- Chen, J.; Wang, R.; Li, X.F.; Wang, R.L. Bifidobacterium adolescentis supplementation ameliorates visceral fat accumulation and insulin sensitivity in an experimental model of the metabolic syndrome. Br. J. Nutr. 2012, 107, 1429–1434. [Google Scholar] [CrossRef] [PubMed]
- Koeth, R.A.; Wang, Z.; Levison, B.S.; Buffa, J.A.; Org, E.; Sheehy, B.T.; Britt, E.B.; Fu, X.; Wu, Y.; Li, L.; et al. Intestinal microbiota metabolism of L-carnitine, a nutrient in red meat, promotes atherosclerosis. Nat. Med. 2013, 19, 576–585. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Pérez, O.; Cruz-Ramón, V.; Chinchilla-López, P.; Méndez-Sánchez, N. The Role of the Gut Microbiota in Bile Acid Metabolism. Ann. Hepatol. 2017, 16, S21–S26. [Google Scholar] [CrossRef]
- Chiang, J.Y.L.; Ferrell, J.M. Up to date on cholesterol 7 alpha-hydroxylase (CYP7A1) in bile acid synthesis. Liver Res. 2020, 4, 47–63. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.J.; Choi, Y.M. Dyslipidemia in women with polycystic ovary syndrome. Obs. Gynecol Sci. 2013, 56, 137–142. [Google Scholar] [CrossRef]
- Shindo, K.; Machida, M.; Fukumura, M.; Koide, K.; Yamazaki, R. Omeprazole induces altered bile acid metabolism. Gut 1999, 42, 266–271. [Google Scholar] [CrossRef]
- Fujisaka, S.; Ussar, S.; Clish, C.; Devkota, S.; Dreyfuss, J.M.; Sakaguchi, M.; Soto, M.; Konishi, M.; Softic, S.; Altindis, E.; et al. Antibiotic effects on gut microbiota and metabolism are host dependent. J. Clin. Invest 2016, 126, 4430–4443. [Google Scholar] [CrossRef]
- Damms-Machado, A.; Louis, S.; Schnitzer, A.; Volynets, V.; Rings, A.; Basrai, M.; Bischoff, S.C. Gut permeability is related to body weight, fatty liver disease, and insulin resistance in obese individuals undergoing weight reduction. Am. J. Clin. Nutr. 2017, 105, 127–135. [Google Scholar] [CrossRef]
- Teixeira, T.F.; Souza, N.C.; Chiarello, P.G.; Franceschini, S.C.; Bressan, J.; Ferreira, C.L.; Peluzio Mdo, C. Intestinal permeability parameters in obese patients are correlated with metabolic syndrome risk factors. Clin. Nutr. 2012, 31, 735–740. [Google Scholar] [CrossRef]
- Luther, J.; Garber, J.J.; Khalili, H.; Dave, M.; Bale, S.S.; Jindal, R.; Motola, D.L.; Luther, S.; Bohr, S.; Jeoung, S.W.; et al. Hepatic Injury in Nonalcoholic Steatohepatitis Contributes to Altered Intestinal Permeability. Cell Mol. Gastroenterol Hepatol 2015, 1, 222–232. [Google Scholar] [CrossRef]
- Devriese, S.; Van den Bossche, L.; Van Welden, S.; Holvoet, T.; Pinheiro, I.; Hindryckx, P.; De Vos, M.; Laukens, D. T84 monolayers are superior to Caco-2 as a model system of colonocytes. Histochem Cell Biol. 2017, 148, 85–93. [Google Scholar] [CrossRef]
- Lam, Y.Y.; Ha, C.W.; Campbell, C.R.; Mitchell, A.J.; Dinudom, A.; Oscarsson, J.; Cook, D.I.; Hunt, N.H.; Caterson, I.D.; Holmes, A.J.; et al. Increased gut permeability and microbiota change associate with mesenteric fat inflammation and metabolic dysfunction in diet-induced obese mice. PLoS ONE 2012, 7, e34233. [Google Scholar] [CrossRef] [PubMed]
- Muccioli, G.G.; Naslain, D.; Backhed, F.; Reigstad, C.S.; Lambert, D.M.; Delzenne, N.M.; Cani, P.D. The endocannabinoid system links gut microbiota to adipogenesis. Mol. Syst. Biol. 2010, 6, 392. [Google Scholar] [CrossRef]
- Simren, M.; Barbara, G.; Flint, H.J.; Spiegel, B.M.; Spiller, R.C.; Vanner, S.; Verdu, E.F.; Whorwell, P.J.; Zoetendal, E.G.; Rome Foundation, C. Intestinal microbiota in functional bowel disorders: A Rome foundation report. Gut 2013, 62, 159–176. [Google Scholar] [CrossRef]
- Aydin, K.; Arusoglu, G.; Koksal, G.; Cinar, N.; Aksoy, D.Y.; Yildiz, B.O. Fasting and post-prandial glucagon like peptide 1 and oral contraception in polycystic ovary syndrome. Clin. Endocrinol 2014, 81, 588–592. [Google Scholar] [CrossRef]
- Sun, L.; Ji, C.; Jin, L.; Bi, Y.; Feng, W.; Li, P.; Shen, S.; Zhu, D. Effects of Exenatide on Metabolic Changes, Sexual Hormones, Inflammatory Cytokines, Adipokines, and Weight Change in a DHEA-Treated Rat Model. Reprod Sci. 2016, 23, 1242–1249. [Google Scholar] [CrossRef]
- Chaudhari, A.P.; Mazumdar, K.; Mehta, P.D. Anxiety, Depression, and Quality of Life in Women with Polycystic Ovarian Syndrome. Indian J. Psychol Med. 2018, 40, 239–246. [Google Scholar] [CrossRef]
- Chaudhari, N.K.; Nampoothiri, L.P. Neurotransmitter alteration in a testosterone propionate-induced polycystic ovarian syndrome rat model. Horm Mol. Biol. Clin. Investig. 2017, 29, 71–77. [Google Scholar] [CrossRef]
- Riaz, T.; Sollid, L.M.; Olsen, I.; de Souza, G.A. Quantitative Proteomics of Gut-Derived Th1 and Th1/Th17 Clones Reveal the Presence of CD28+ NKG2D- Th1 Cytotoxic CD4+ T cells. Mol. Cell. Proteom. 2016, 15, 1007–1016. [Google Scholar] [CrossRef]
- Qin, L.; Xu, W.; Li, X.; Meng, W.; Hu, L.; Luo, Z.; Wang, Y.; Luo, S.; Li, S. Differential Expression Profile of Immunological Cytokines in Local Ovary in Patients with Polycystic Ovarian Syndrome: Analysis by Flow Cytometry. Eur. J. Obs. Gynecol Reprod Biol. 2016, 197, 136–141. [Google Scholar] [CrossRef]
- Kim, C.H.; Park, J.; Kim, M. Gut microbiota-derived short-chain Fatty acids, T cells, and inflammation. Immune Netw. 2014, 14, 277–288. [Google Scholar] [CrossRef]
- Melo, A.S.; Ferriani, R.A.; Navarro, P.A. Treatment of infertility in women with polycystic ovary syndrome: Approach to clinical practice. Clinics 2015, 70, 765–769. [Google Scholar] [CrossRef] [PubMed]
- Sahu, M.; Rout, N.R. Comparative study of clomiphene citrate versus letrozole as first-line ovulation induction drug in infertile polycystic ovary syndrome women. Int. J. Reprod. Contracept. Obstet. Gynecol. 2020, 9, 2948. [Google Scholar] [CrossRef]
- Marshall, J.C.; Dunaif, A. Should all women with PCOS be treated for insulin resistance? Fertil Steril 2012, 97, 18–22. [Google Scholar] [CrossRef] [PubMed]
- Sawant, S.; Bhide, P. Fertility Treatment Options for Women With Polycystic Ovary Syndrome. Clin. Med. Insights Reprod Health 2019, 13, 1179558119890867. [Google Scholar] [CrossRef]
- Mathur, R.; Levin, O.F.; Azziz, R. Use of ethinylestradiol/drospirenone combination in patients with the polycystic ovary syndrome. Ther. Clin. Risk Manag. 2008, 4, 487–492. [Google Scholar] [CrossRef]
- Gulati, M.; Singh, S.K.; Corrie, L.; Kaur, I.P.; Chandwani, L. Delivery routes for faecal microbiota transplants: Available, anticipated and aspired. Pharm. Res 2020, 159, 104954. [Google Scholar] [CrossRef]
- Torres, P.J.; Ho, B.S.; Arroyo, P.; Sau, L.; Chen, A.; Kelley, S.T.; Thackray, V.G. Exposure to a Healthy Gut Microbiome Protects Against Reproductive and Metabolic Dysregulation in a PCOS Mouse Model. Endocrinology 2019, 160, 1193–1204. [Google Scholar] [CrossRef]
- Corrie, L.; Gulati, M.; Awasthi, A.; Vishwas, S.; Kaur, J.; Khursheed, R.; Porwal, O.; Alam, A.; Parveen, S.R.; Singh, H.; et al. Harnessing the dual role of polysaccharides in treating gastrointestinal diseases: As therapeutics and polymers for drug delivery. Chem Biol Interact 2022, 368, 110238. [Google Scholar] [CrossRef]
- Davani-Davari, D.; Negahdaripour, M.; Karimzadeh, I.; Seifan, M.; Mohkam, M.; Masoumi, S.J.; Berenjian, A.; Ghasemi, Y. Prebiotics: Definition, Types, Sources, Mechanisms, and Clinical Applications. Foods 2019, 8. [Google Scholar] [CrossRef]
- Nilsson, A.C.; Ostman, E.M.; Holst, J.J.; Björck, I.M. Including indigestible carbohydrates in the evening meal of healthy subjects improves glucose tolerance, lowers inflammatory markers, and increases satiety after a subsequent standardized breakfast. J. Nutr. 2008, 138, 732–739. [Google Scholar] [CrossRef]
- Urias-Silvas, J.E.; Cani, P.D.; Delmée, E.; Neyrinck, A.; López, M.G.; Delzenne, N.M. Physiological effects of dietary fructans extracted from Agave tequilana Gto and Dasylirion spp. Br. J. Nutr. 2008, 99, 254–261. [Google Scholar] [CrossRef]
- Fazel Torshizi, F.; Chamani, M.; Khodaei, H.R.; Sadeghi, A.A.; Hejazi, S.H.; Majidzadeh Heravi, R. Therapeutic effects of organic zinc on reproductive hormones, insulin resistance and mTOR expression, as a novel component, in a rat model of Polycystic ovary syndrome. Iran J. Basic Med. Sci. 2020, 23, 36–45. [Google Scholar] [CrossRef]
- Xue, J.; Li, X.; Liu, P.; Li, K.; Sha, L.; Yang, X.; Zhu, L.; Wang, Z.; Dong, Y.; Zhang, L.; et al. Inulin and metformin ameliorate polycystic ovary syndrome via anti-inflammation and modulating gut microbiota in mice. Endocr J. 2019, 66, 859–870. [Google Scholar] [CrossRef]
- Report of a Joint FAO/WHO Expert Consultation on Evaluation of Health and Nutritional Properties of Probiotics in Food including Powder Milk with Live Lactic Acid Bacteria. Available online: https://www.fao.org/3/a0512e/a0512e.pdf (accessed on 3 December 2022).
- Zhang, F.; Ma, T.; Cui, P.; Tamadon, A.; He, S.; Huo, C.; Yierfulati, G.; Xu, X.; Hu, W.; Li, X.; et al. Diversity of the Gut Microbiota in Dihydrotestosterone-Induced PCOS Rats and the Pharmacologic Effects of Diane-35, Probiotics, and Berberine. Front. Microbiol. 2019, 10, 175. [Google Scholar] [CrossRef]
- Karamali, M.; Eghbalpour, S.; Rajabi, S.; Jamilian, M.; Bahmani, F.; TajabadiEbrahimi, M.; Keneshlou, F.; Mirhashemi, S.M.; Chamani, M.; Gelougerdi, S.H.; et al. Effects of Probiotic Supplementation on Hormonal Profiles, Biomarkers of Inflammation and Oxidative Stress in Women With Polycystic Ovary Syndrome: A Randomized, DoubleBlind, Placebo-Controlled Trial. Arch. Iran Med. 2018, 21, 1–7. [Google Scholar]
- Ejtahed, H.S.; Mohtadi-Nia, J.; Homayouni-Rad, A.; Niafar, M.; Asghari-Jafarabadi, M.; Mofid, V.; Akbarian-Moghari, A. Effect of probiotic yogurt containing Lactobacillus acidophilus and Bifidobacterium lactis on lipid profile in individuals with type 2 diabetes mellitus. J. Dairy Sci. 2011, 94, 3288–3294. [Google Scholar] [CrossRef]
- Ejtahed, H.S.; Mohtadi-Nia, J.; Homayouni-Rad, A.; Niafar, M.; Asghari-Jafarabadi, M.; Mofid, V. Probiotic yogurt improves antioxidant status in type 2 diabetic patients. Nutrition 2012, 28, 539–543. [Google Scholar] [CrossRef]
- Asemi, Z.; Zare, Z.; Shakeri, H.; Sabihi, S.S.; Esmaillzadeh, A. Effect of multispecies probiotic supplements on metabolic profiles, hs-CRP, and oxidative stress in patients with type 2 diabetes. Ann. Nutr. Metab 2013, 63, 1–9. [Google Scholar] [CrossRef]
- Mazloom, Z.; Yousefinejad, A.; Dabbaghmanesh, M.H. Effect of probiotics on lipid profile, glycemic control, insulin action, oxidative stress, and inflammatory markers in patients with type 2 diabetes: A clinical trial. Iran J. Med. Sci. 2013, 38, 38–43. [Google Scholar]
- Tajadadi-Ebrahimi, M.; Bahmani, F.; Shakeri, H.; Hadaegh, H.; Hijijafari, M.; Abedi, F.; Asemi, Z. Effects of daily consumption of synbiotic bread on insulin metabolism and serum high-sensitivity C-reactive protein among diabetic patients: A double-blind, randomized, controlled clinical trial. Ann. Nutr. Metab 2014, 65, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Mohamadshahi, M.; Veissi, M.; Haidari, F.; Javid, A.Z.; Mohammadi, F.; Shirbeigi, E. Effects of probiotic yogurt consumption on lipid profile in type 2 diabetic patients: A randomized controlled clinical trial. J. Res. Med. Sci. 2014, 19, 531–536. [Google Scholar] [PubMed]
- Hove, K.D.; Brons, C.; Faerch, K.; Lund, S.S.; Rossing, P.; Vaag, A. Effects of 12 weeks of treatment with fermented milk on blood pressure, glucose metabolism and markers of cardiovascular risk in patients with type 2 diabetes: A randomised double-blind placebo-controlled study. Eur. J. Endocrinol 2015, 172, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Firouzi, S.; Majid, H.A.; Ismail, A.; Kamaruddin, N.A.; Barakatun-Nisak, M.Y. Effect of multi-strain probiotics (multi-strain microbial cell preparation) on glycemic control and other diabetes-related outcomes in people with type 2 diabetes: A randomized controlled trial. Eur. J. Nutr. 2017, 56, 1535–1550. [Google Scholar] [CrossRef]
- Corrie, L.; Kaur, J.; Awasthi, A.; Vishwas, S.; Gulati, M.; Saini, S.; Kumar, B.; Pandey, N.K.; Gupta, G.; Dureja, H.; et al. Multivariate Data Analysis and Central Composite Design-Oriented Optimization of Solid Carriers for Formulation of Curcumin-Loaded Solid SNEDDS: Dissolution and Bioavailability Assessment. Pharmaceutics 2022, 14. [Google Scholar] [CrossRef]
- Kumar Singh, A.; Cabral, C.; Kumar, R.; Ganguly, R.; Kumar Rana, H.; Gupta, A.; Rosaria Lauro, M.; Carbone, C.; Reis, F.; Pandey, A.K. Beneficial Effects of Dietary Polyphenols on Gut Microbiota and Strategies to Improve Delivery Efficiency. Nutrients 2019, 11. [Google Scholar] [CrossRef]
- Ghowsi, M.; Khazali, H.; Sisakhtnezhad, S. The effect of resveratrol on oxidative stress in the liver and serum of a rat model of polycystic ovary syndrome: An experimental study. Int. J. Reprod Biomed 2018, 16, 149. [Google Scholar] [CrossRef]
- Jahan, S.; Abid, A.; Khalid, S.; Afsar, T.; Qurat Ul, A.; Shaheen, G.; Almajwal, A.; Razak, S. Therapeutic potentials of Quercetin in management of polycystic ovarian syndrome using Letrozole induced rat model: A histological and a biochemical study. J. Ovarian Res. 2018, 11, 26. [Google Scholar] [CrossRef]
- Reddy, P.S.; Begum, N.; Mutha, S.; Bakshi, V. Beneficial effect of Curcumin in Letrozole induced polycystic ovary syndrome. Asian Pac. J. Reprod. 2016, 5, 116–122. [Google Scholar] [CrossRef]
- Masumoto, S.; Terao, A.; Yamamoto, Y.; Mukai, T.; Miura, T.; Shoji, T. Non-absorbable apple procyanidins prevent obesity associated with gut microbial and metabolomic changes. Sci. Rep. 2016, 6, 31208. [Google Scholar] [CrossRef]
- Wang, S.; Li, Q.; Zang, Y.; Zhao, Y.; Liu, N.; Wang, Y.; Xu, X.; Liu, L.; Mei, Q. Apple Polysaccharide inhibits microbial dysbiosis and chronic inflammation and modulates gut permeability in HFD-fed rats. Int. J. Biol. Macromol. 2017, 99, 282–292. [Google Scholar] [CrossRef]
- Qiao, Y.; Sun, J.; Xia, S.; Tang, X.; Shi, Y.; Le, G. Effects of resveratrol on gut microbiota and fat storage in a mouse model with high-fat-induced obesity. Food Funct 2014, 5, 1241–1249. [Google Scholar] [CrossRef]
- Lee, S.; Keirsey, K.I.; Kirkland, R.; Grunewald, Z.I.; Fischer, J.G.; de La Serre, C.B. Blueberry Supplementation Influences the Gut Microbiota, Inflammation, and Insulin Resistance in High-Fat-Diet-Fed Rats. J. Nutr. 2018, 148, 209–219. [Google Scholar] [CrossRef]
- Pan, P.; Lam, V.; Salzman, N.; Huang, Y.W.; Yu, J.; Zhang, J.; Wang, L.S. Black Raspberries and Their Anthocyanin and Fiber Fractions Alter the Composition and Diversity of Gut Microbiota in F-344 Rats. Nutr. Cancer 2017, 69, 943–951. [Google Scholar] [CrossRef]
- Prior, R.L.; Wilkes, S.; Rogers, T.; Khanal, R.C.; Wu, X.; Hager, T.J.; Hager, A.; Howard, L. Dietary black raspberry anthocyanins do not alter development of obesity in mice fed an obesogenic high-fat diet. J. Agric Food Chem. 2010, 58, 3977–3983. [Google Scholar] [CrossRef]
- Tung, Y.C.; Lin, Y.H.; Chen, H.J.; Chou, S.C.; Cheng, A.C.; Kalyanam, N.; Ho, C.T.; Pan, M.H. Piceatannol Exerts Anti-Obesity Effects in C57BL/6 Mice through Modulating Adipogenic Proteins and Gut Microbiota. Molecules 2016, 21. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Lin, K.; Li, K.; Deng, X.; Li, C. Reshaped fecal gut microbiota composition by the intake of high molecular weight persimmon tannin in normal and high-cholesterol diet-fed rats. Food Funct 2018, 9, 541–551. [Google Scholar] [CrossRef]
- Shen, W.; Wolf, P.G.; Carbonero, F.; Zhong, W.; Reid, T.; Gaskins, H.R.; McIntosh, M.K. Intestinal and systemic inflammatory responses are positively associated with sulfidogenic bacteria abundance in high-fat-fed male C57BL/6J mice. J. Nutr. 2014, 144, 1181–1187. [Google Scholar] [CrossRef]
- Wang, J.; Ghosh, S.S.; Ghosh, S. Curcumin improves intestinal barrier function: Modulation of intracellular signaling, and organization of tight junctions. Am. J. Physiol Cell Physiol 2017, 312, C438–C445. [Google Scholar] [CrossRef] [PubMed]
- Song, J.X.; Ren, H.; Gao, Y.F.; Lee, C.Y.; Li, S.F.; Zhang, F.; Li, L.; Chen, H. Dietary Capsaicin Improves Glucose Homeostasis and Alters the Gut Microbiota in Obese Diabetic ob/ob Mice. Front. Physiol 2017, 8, 602. [Google Scholar] [CrossRef] [PubMed]
- Romo-Vaquero, M.; Selma, M.V.; Larrosa, M.; Obiol, M.; Garcia-Villalba, R.; Gonzalez-Barrio, R.; Issaly, N.; Flanagan, J.; Roller, M.; Tomas-Barberan, F.A.; et al. A rosemary extract rich in carnosic acid selectively modulates caecum microbiota and inhibits beta-glucosidase activity, altering fiber and short chain fatty acids fecal excretion in lean and obese female rats. PLoS ONE 2014, 9, e94687. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Honda, K. Towards the development of defined microbial therapeutics. Int. Immunol. 2021. [Google Scholar] [CrossRef] [PubMed]
| Hormone | Model | Finding | Microbiota studied | Reference |
|---|---|---|---|---|
| Insulin | Honey Bees | Changes in insulin signaling reduces gut pH and gustatory response | Gilliamella apicola and Lactobacillus spp. | [24] |
| Testosterone, estradiol | Human | Correlation of diversity of microbiota with sex hormones | Acinetobacter, Dorea, Ruminococcus, and Megamonas, Bacteroidetes and Firmicutes | [22] |
| Adrenocorticotropic | Wistar rats | Role of gut microbiota in depression | Desulfovibrionales, Desulfovibrio, Klebsiella, Burkholderiales, and Bifidobacterium | [25] |
| Ghrelin | Wistar rats | Short-chain acid-producing bacteria and its effects on gut microbiota | Turicibacter, Brevibacterium, Parasutterella, and Oscillibacter | [26] |
| Juvenile hormone III | Riptortus pedestris | A more significant number of eggs produced as a result of Burkholderia gut symbiont | Burkholderia | [27] |
| Adrenocorticotrophic hormone | Wistar rats | Changes in the microbiome diversity | Akkermansia and Lactobacillus | [28] |
| Epinephrine and Norepinephrine | Bacterial growth | Stress hormones can affect the growth of anaerobic bacteria in the gut | Fusobacterium nucleatum, Prevotella, Porhyromonas spp., Tanerella forsythia, and Propionibacterium acnes | [29] |
| Thyroxine | Mice | Reduced diversity of some bacterial species | Bacteria belonging to families Lactobacillaceae and Bifidobacteriaceae | [30] |
| Androgen | Pigs | Influence of host gender on gut microbiota and sex-biased bacterial taxa | Bacteria belonging to families Veillonellaceae, Roseburia, Bulleidia, and Escherichia | [31] |
| Androgen | Wistar rats | Prenatal androgen was associated with the abundance of gut microbiota | Akkermansia, Bacteroides, Lactobacillus, Clostridium, Nocardiaceae, and Clostridiaceae | [32] |
| Glucagon-like peptide 1 | Mice | Intestinal microbiota increases GLP-1 levels | L. paracasei, L. bulgaricus, and Streptococcus thermophilus | [33] |
| Corticosterone, adrenocorticosterone | Probiotics in rats and humans | Bacteria reduce levels of stress hormones | L. helveticus and B. longum | [34] |
| Synbiotic Treatment | Model | Outcome | Reference |
|---|---|---|---|
| L. bifidum, L. acidophilus, L. casei and inulin | Clinical study | No change in hirsutism | [59] |
| Lactobacillus, Bifidobacterium, and Selenium | Clinical study | Improved testosterone and hirsutism | [60] |
| L. bifidum, L. acidophilus, L. casei and inulin | Clinical study | Improved insulin sensitivity | [59] |
| L. acidophilus, L. reuteri, L. fermentum, B. bifidum and selenium | Clinical study | Increased insulin sensitivity | [61] |
| L. plantarum and inulin | Wistar rats | Decrease hyperglycemia, IR, hyperlipidemia, and ameliorate oxidative stress | [62] |
| L. acidophilus, L. casei, L. rhamnosus and inulin | Clinical study | Reduced low-density lipoprotein (LDL) and increased high-density lipoprotein (HDL) | [63] |
| Probiotic Used/Dose | Design | Source and Duration | Number of Participants | Outcome | Reference |
|---|---|---|---|---|---|
| L. acidophilus La5 (4.14 × 106 CFU/g), Bifidobacterium lactis Bb12 (3.61 × 106 CFU/g) | Double-blind, placebo | Yogurt, 6 weeks | 64 | ↓ Cholesterol, ↓ LDL-C | [178] |
| L. acidophilus La5 (7.23–1.85 × 106 CFU/g), Bifidobacterium lactis Bb12 (6.04–1.79 × 106 CFU/g) | Double-blind placebo | Yogurt, 6 weeks | 64 | ↓ Fasting glucose, ↓serum malondialdehyde concentration, ↑ erythrocyte superoxide dismutase, ↑ glutathione peroxidase | [179] |
| L. acidophilus (2 × 109 CFU) L. casei (7 × 109 CFU) L. rhamnosus (1.5 × 109 CFU) L. bulgaricus (2 × 108 CFU), B. breve (2 × 1010 CFU), B. longum (7 × 109 CFU), S. thermophilus (1.5 × 109 CFU) | Single blind, placebo, parallel | Capsule, 8 weeks | 54 | ↑ Serum insulin, ↑ LDL-C, ↑ GSH levels, ↓ serum hs-CRP | [180] |
| L. acidophilus, L. bulgaricus, L. bifidum, L. casei, L. sporogenes (1 × 108 CFU) | Single-blind, placebo, parallel | Capsule, 6 weeks | 34 | ↑ HDL-C, ↓ Insulin, ↓ MDA, ↓ IL-6 | [181] |
| L. sporogenes (1 × 108 CFU) | Double-blind, placebo, parallel | Bread, 8 weeks | 81 | ↓ Serum insulin levels, ↓ HOMA-IR scores, ↓ HOMA-B, ↑ inflammation markers | [182] |
| L. acidophilus, B. lactis (3.7 × 106 CFU/mg) | Double-blind, placebo, parallel | Yogurt, 8 weeks | 44 | ↓LDL-C/HDL-C ratio, ↓ triglycerides | [183] |
| L. actobacillus, L. helveticus | Double-blind, placebo control | Yogurt, 12 weeks | 41 | ↓ Blood glucose concentration, ↓serum glucose | [184] |
| Lactobacillus species | Double-blind, placebo control, parallel | Sachet, 12 weeks | 136 | ↓HbA1c, ↓ triglycerides, ↓ insulin resistance | [185] |
| Source | Polyphenol | Animal Used, Number of Treatment Controls | Changes in Gut Microbiota | Changes in Metabolic Parameters | Reference |
|---|---|---|---|---|---|
| Apple (Pyrus Malus) | Procyanidins | C57BL/6J mice, n = 10 | ↓The ratio of Firmicutes to Bacteroidetes, ↑ Akkermansia, Bacteroidetes and Lactobacillus | ↓Pro-inflammatory factors TNF-α, IL-1β, MCP-1, and chemokine ligand 1, metabolic endotoxemia | [191,192] |
| Grapes (Vitis Vinifera) | Resveratrol | Male Kunming mice, n = 8 | ↑ Bacteroidetes to Firmicutes ratio, Lactobacillus, and Bifidobacterium, ↓ Enterococcus faecalis | ↓Weight gain and visceral adipose weight | [193] |
| Berries | Anthocyanins | SD rats, n = 8 Wistar, n = 8 C57BL/6J mice, n = 8 | ↑ Akkermansia and Desulfovibrio, Faecalibacterium, Gammaproteobacteria | ↓TNF-α and IL-1β levels, | [194,195,196] |
| Citrus fruits | Piceatannol | C57BL/6J mice, n = 8 | ↑ Prevotella | ↓ Lipid droplets, perilipin 1 protein, and sterol regulatory element-binding protein 1 | [197] |
| Japanese Persimmon (Diospyros kaki) | Tannin | SD rats, n = 6 | ↑ Bifidobacterium and Lactobacillus, ↓ Firmicutes, Escherichia coli, and Enterococcus | ↓Serum lipids and cholesterol | [198] |
| Turmeric (Curcuma Longa) | Curcumin | C57BL/6J mice, n = 6 | ↑ Prevotella, Bacteroidaceae, and Rikenella | ↑Expression of tight junction proteins, gut permeability, ↓NF-κB | [199,200] |
| Chilli (Capsicum frutescens) | Capsaicin | ob/ob mice, n = 5 | ↑ Firmicutes to Bacteroidetes Ratio, ↓ Bacteroides and Parabacteroides | ↑Fecal butyrate and plasma total glucagon-like peptide-1 (GLP-1) levels, and ↓total ghrelin, TNF-α, IL-1 β, and IL-6 levels | [201] |
| Rosemary (Salvia rosmarinus) | Carnosic acid | Zucker obese rats, n = 10 | ↑ Blautia coccoides and Prevotella | ↓ Bodyweight | [202] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Corrie, L.; Awasthi, A.; Kaur, J.; Vishwas, S.; Gulati, M.; Kaur, I.P.; Gupta, G.; Kommineni, N.; Dua, K.; Singh, S.K. Interplay of Gut Microbiota in Polycystic Ovarian Syndrome: Role of Gut Microbiota, Mechanistic Pathways and Potential Treatment Strategies. Pharmaceuticals 2023, 16, 197. https://doi.org/10.3390/ph16020197
Corrie L, Awasthi A, Kaur J, Vishwas S, Gulati M, Kaur IP, Gupta G, Kommineni N, Dua K, Singh SK. Interplay of Gut Microbiota in Polycystic Ovarian Syndrome: Role of Gut Microbiota, Mechanistic Pathways and Potential Treatment Strategies. Pharmaceuticals. 2023; 16(2):197. https://doi.org/10.3390/ph16020197
Chicago/Turabian StyleCorrie, Leander, Ankit Awasthi, Jaskiran Kaur, Sukriti Vishwas, Monica Gulati, Indu Pal Kaur, Gaurav Gupta, Nagavendra Kommineni, Kamal Dua, and Sachin Kumar Singh. 2023. "Interplay of Gut Microbiota in Polycystic Ovarian Syndrome: Role of Gut Microbiota, Mechanistic Pathways and Potential Treatment Strategies" Pharmaceuticals 16, no. 2: 197. https://doi.org/10.3390/ph16020197
APA StyleCorrie, L., Awasthi, A., Kaur, J., Vishwas, S., Gulati, M., Kaur, I. P., Gupta, G., Kommineni, N., Dua, K., & Singh, S. K. (2023). Interplay of Gut Microbiota in Polycystic Ovarian Syndrome: Role of Gut Microbiota, Mechanistic Pathways and Potential Treatment Strategies. Pharmaceuticals, 16(2), 197. https://doi.org/10.3390/ph16020197










