In Vitro Anti-Prostate Cancer Activity of Two Ebselen Analogues
Abstract
1. Introduction
2. Materials and Methods
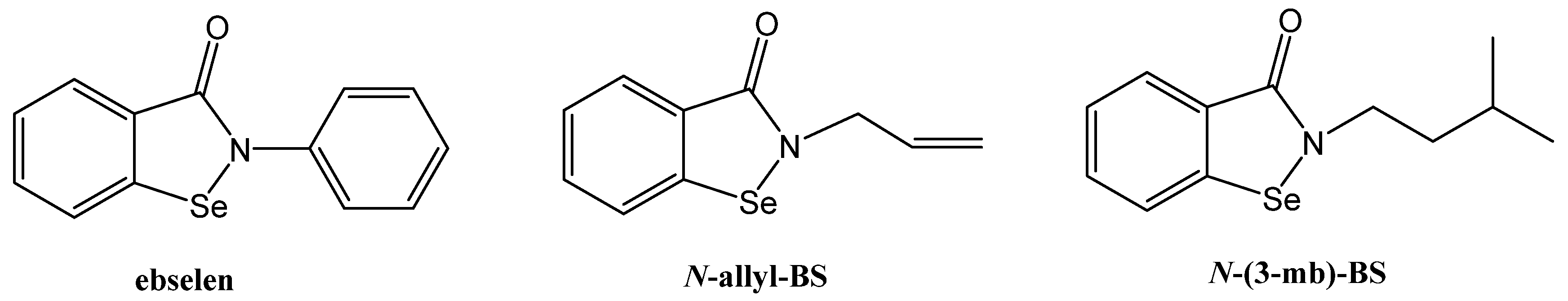
2.1. Synthesis of N-allyl-1,2-benzisoselenazol-3(2H)-one (N-allyl-BS) and N-(3-methylbutyl)-1,2-benzisoselenazol-3(2H)-one (N-(3-mb)-BS)
2.2. Reagents
2.3. Cell Line and Cell Culture
2.4. SRB Cell Viability Assay
2.5. Western Blot Analysis
2.6. Cell Death Assay
2.7. Cell Cycle Assay
2.8. Cell Viability Assay
2.9. Detection of Intracellular ROS
2.10. Comet Assay
2.11. Analysis of the Comet Assay Data
2.12. Statistical Analysis
3. Results
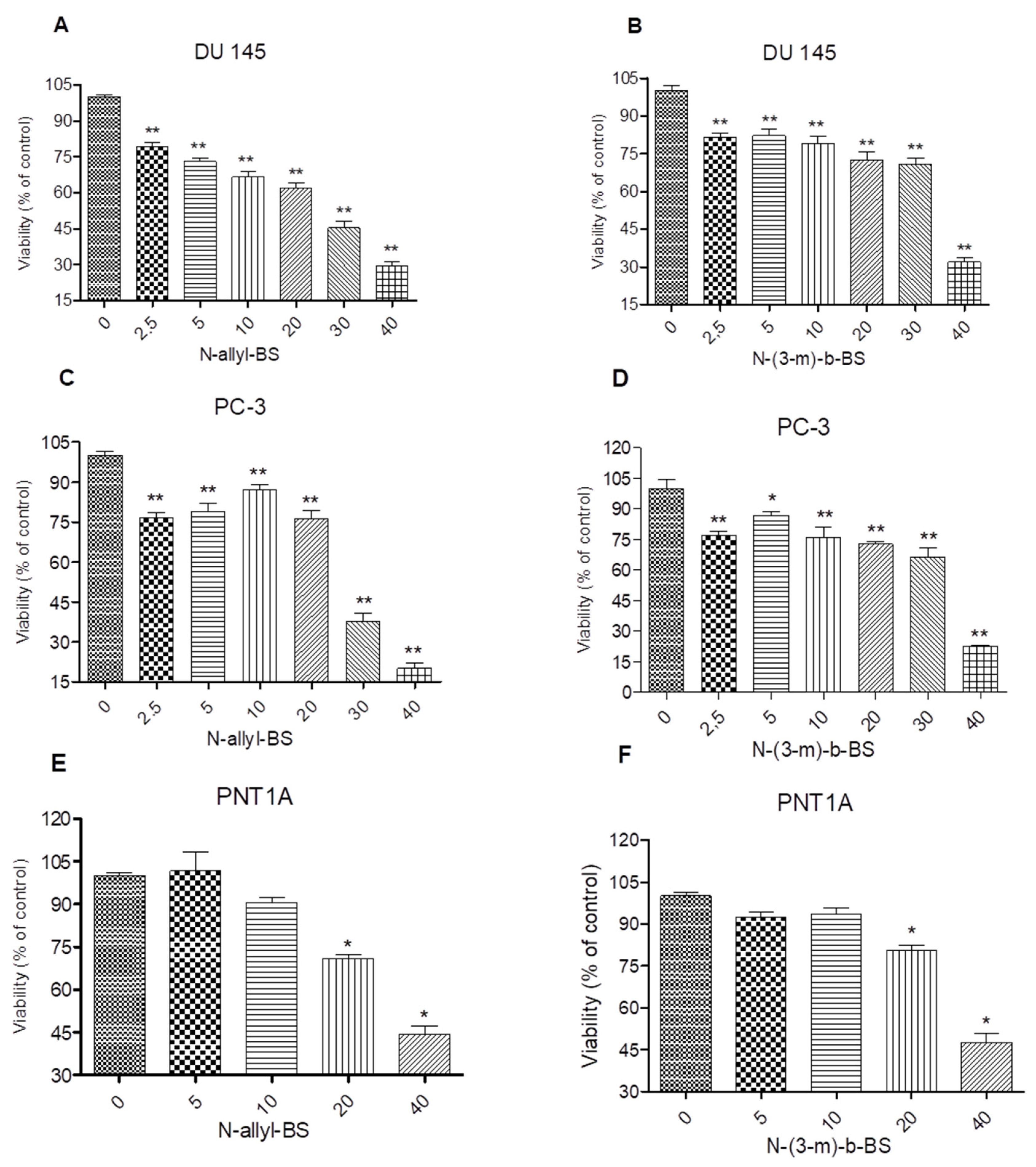
3.1. N-Allyl-BS and N-(3-mb)-BS Inhibit the Viability of DU145 and PC-3 Prostate Cancer Cell
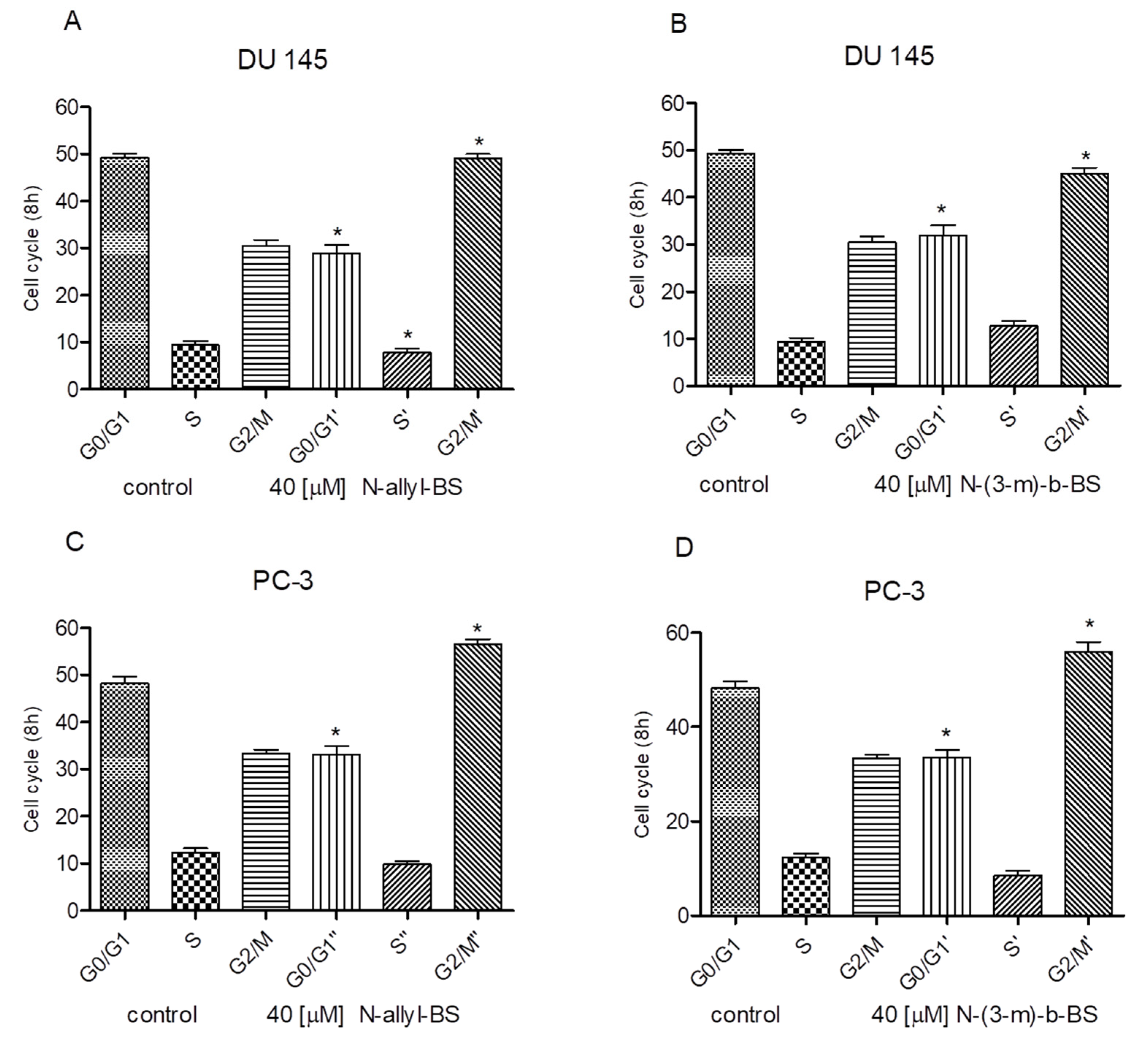
3.2. N-allyl-BS and N-(3-mb)-BS Induce G2/M Cell Cycle Arrest in Prostate Cancer Cells
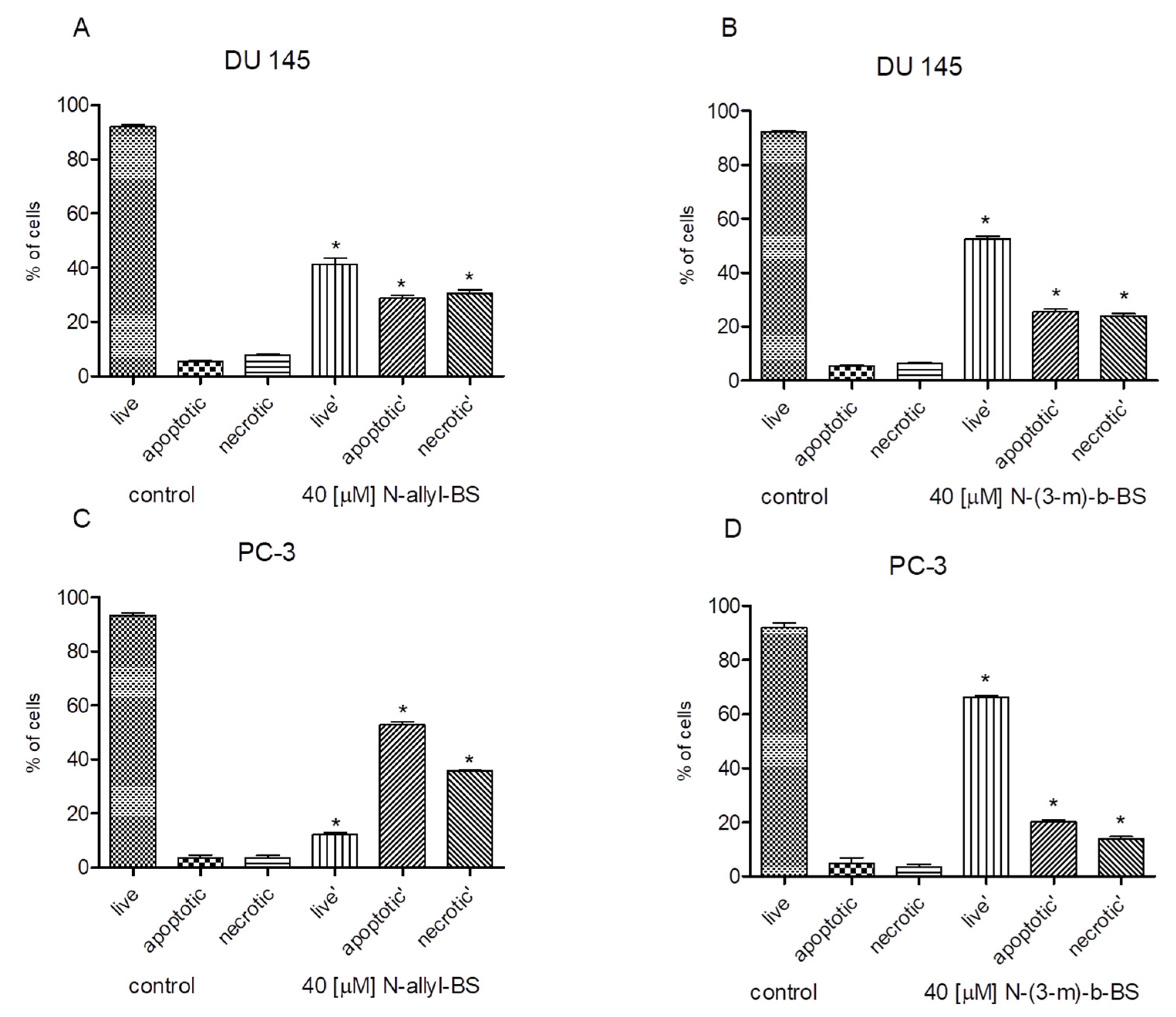
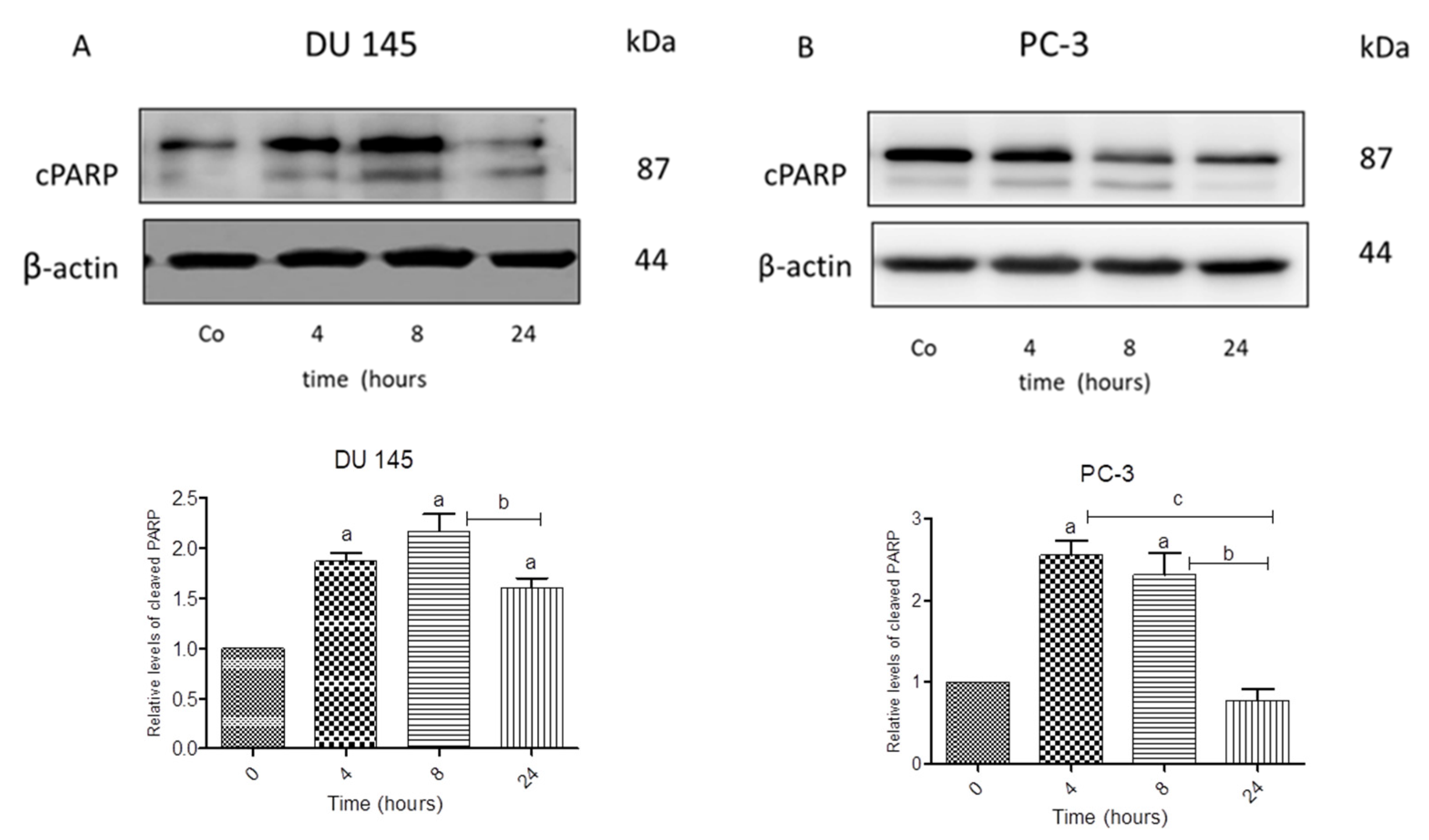
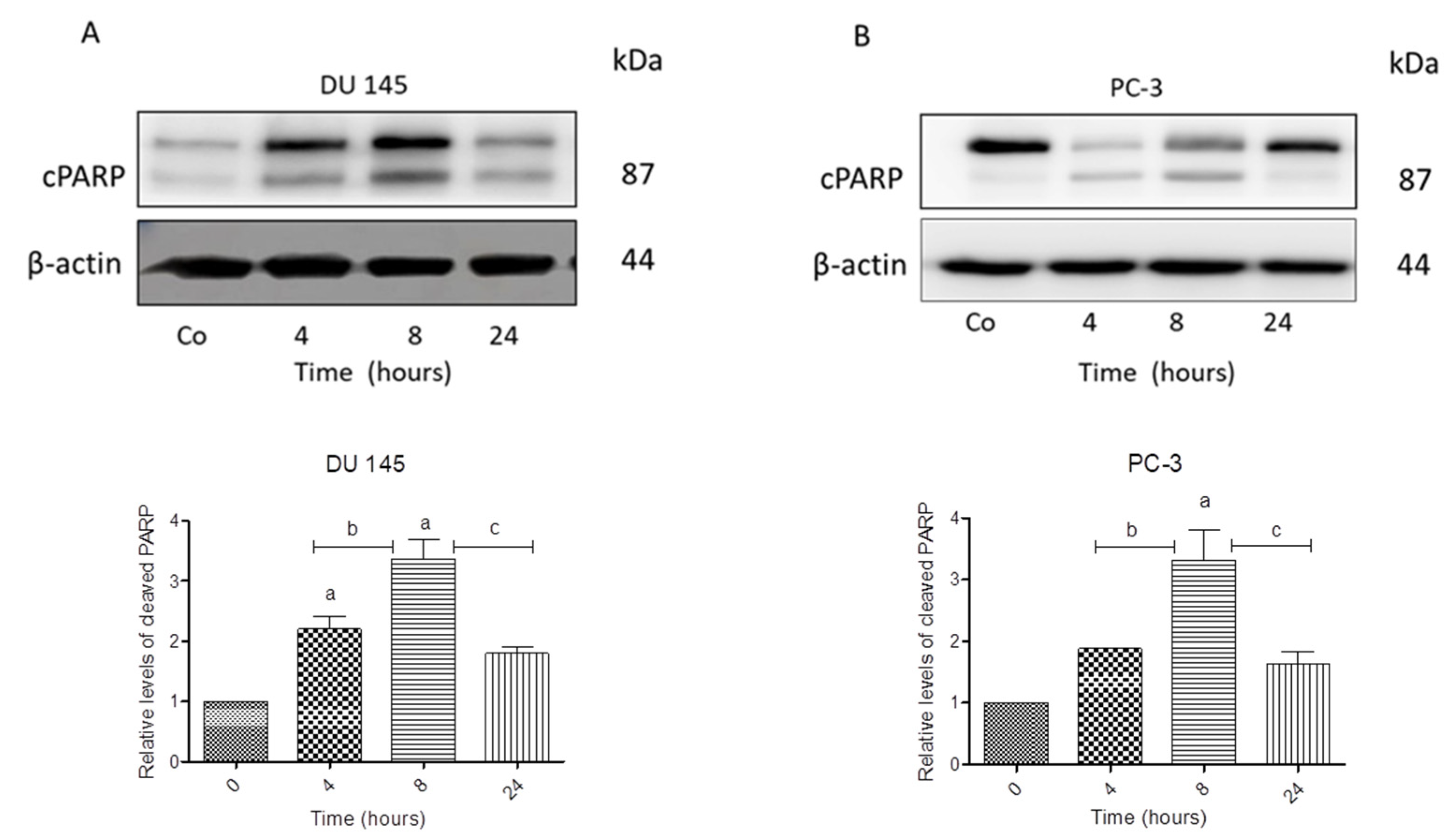
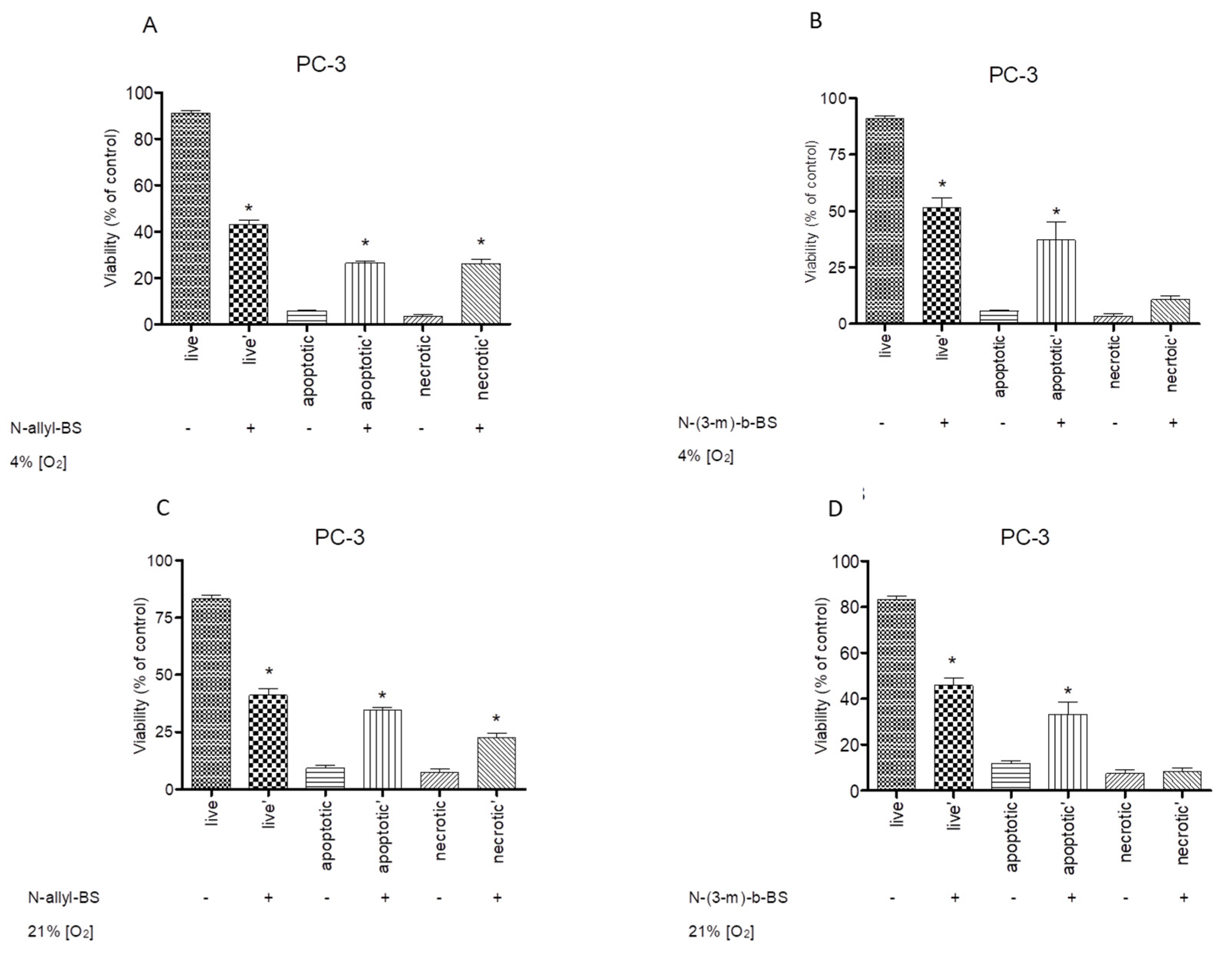
3.3. N-allyl-BS and N-(3-mb)-BS Induce Apoptosis and Necrosis of Cancer Cells
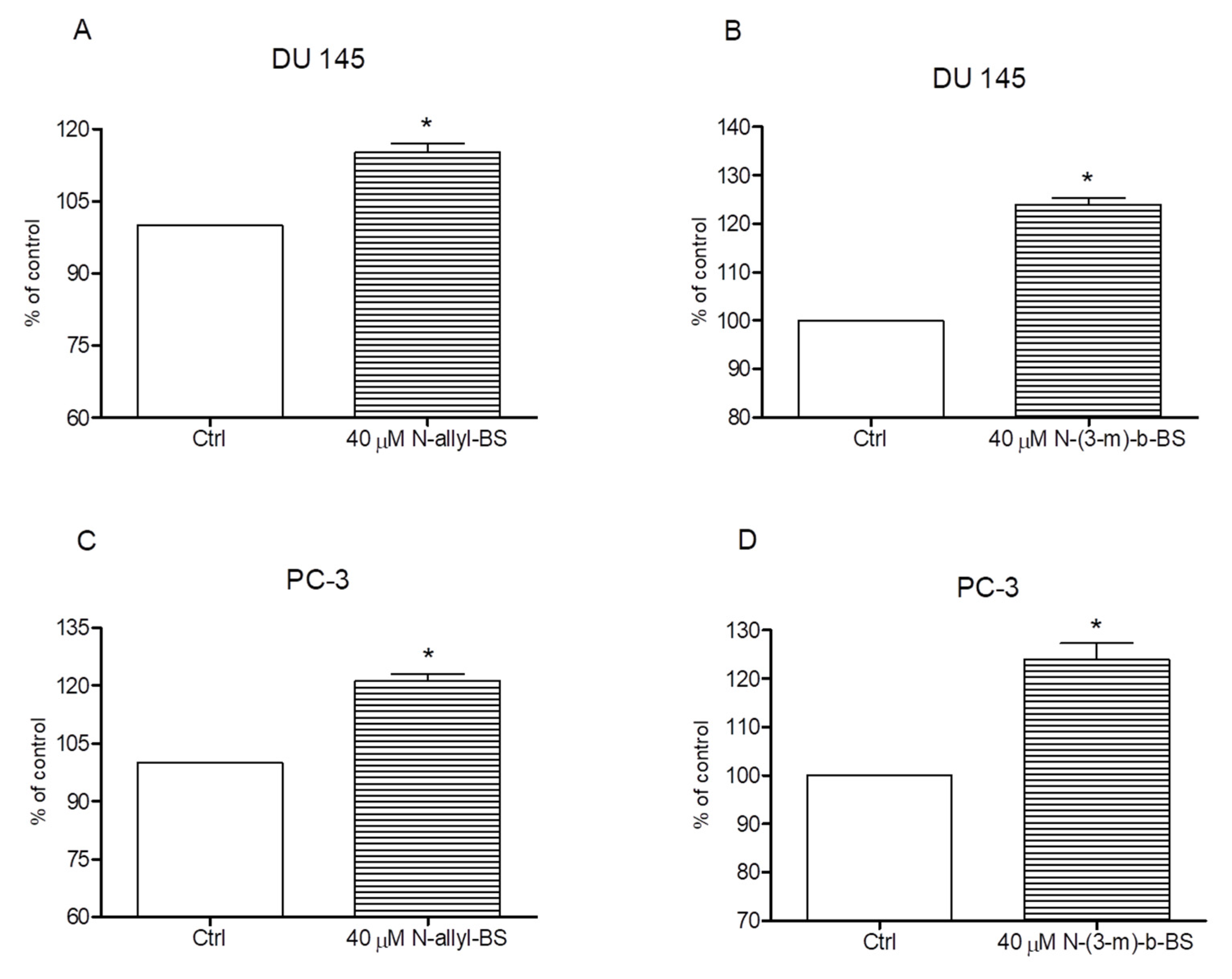
3.4. N-Allyl-BS and N-(3-mb)-BS Respectively Enhance ROS Generation in Cancer Cells
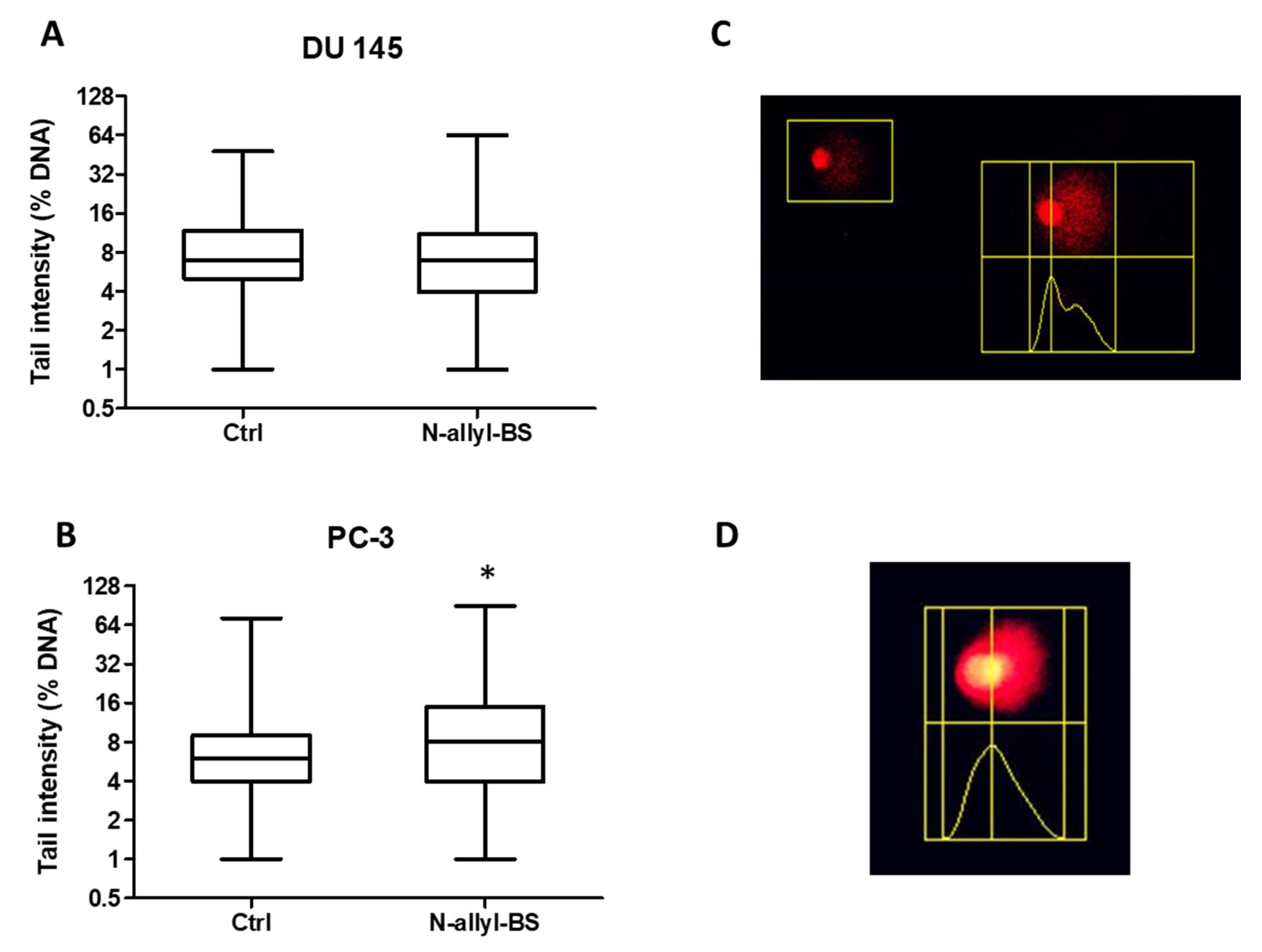
3.5. N-allyl-BS Induces DNA Damage
3.6. The Cytotoxic Activity of N-Allyl-BS and N-(3-mb)-BS are Similar under Hypoxic and Normoxic Conditions
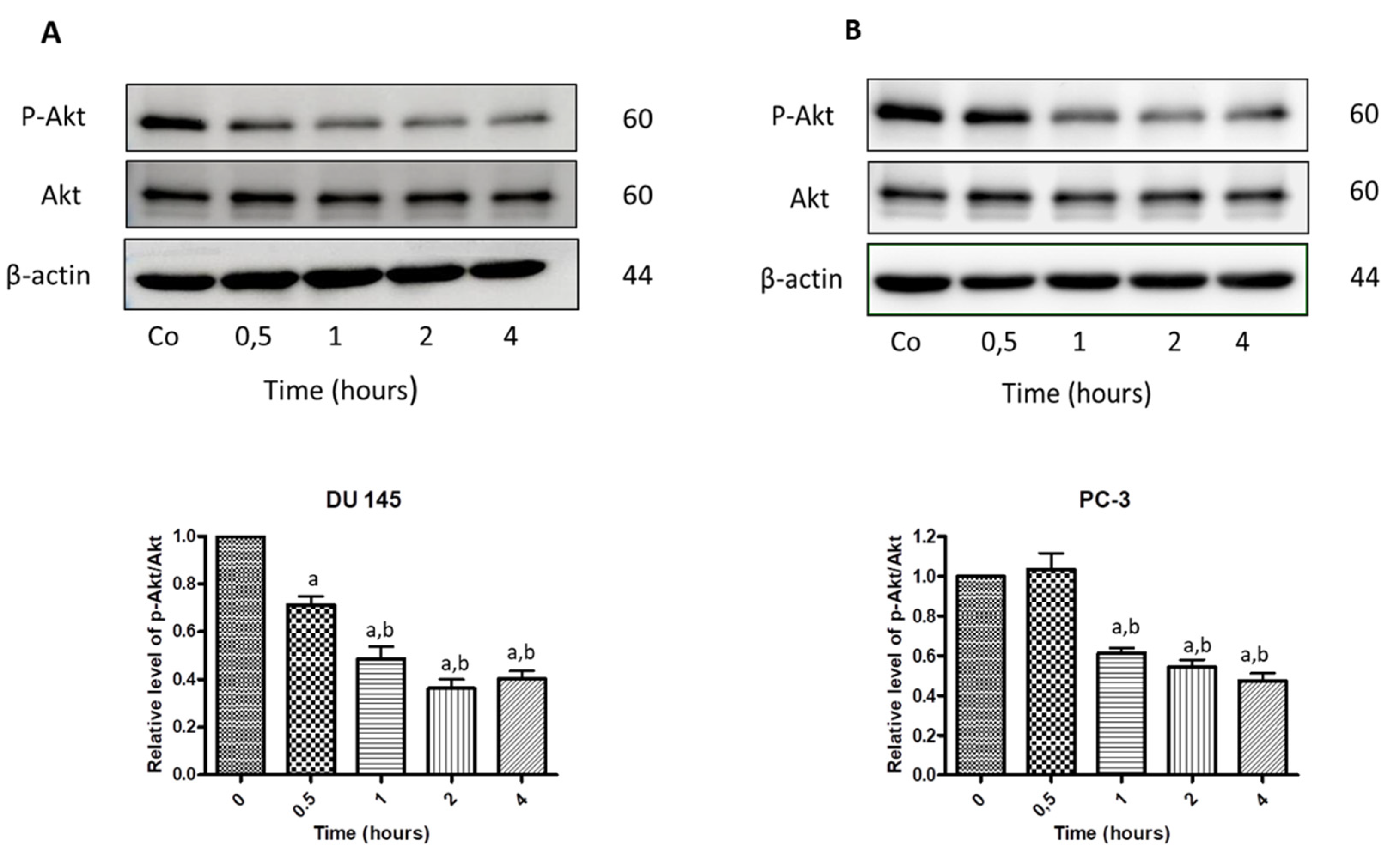
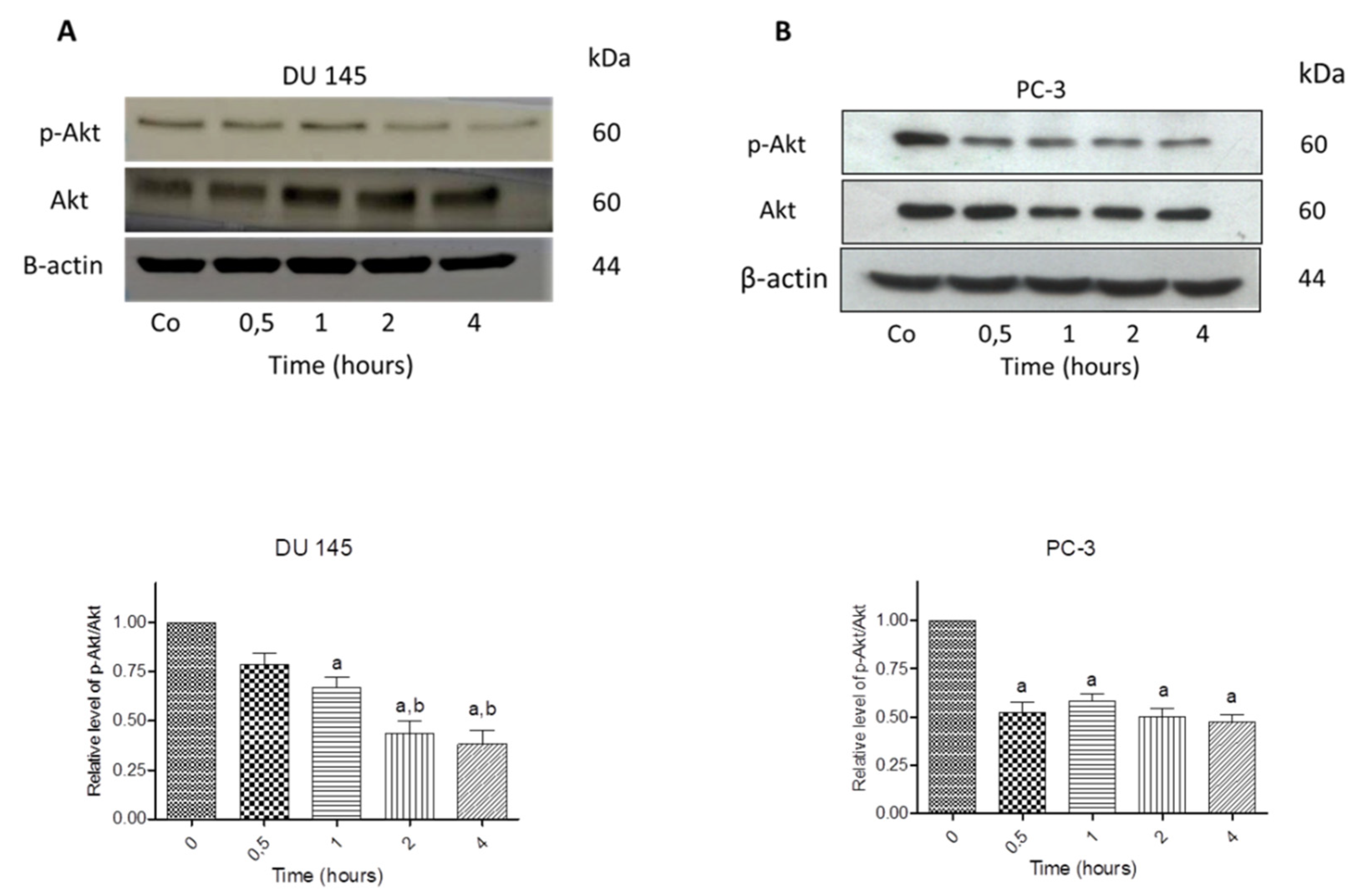
3.7. N-Allyl-BS and N-(3-mb)-BS Reduce Akt Phosphorylation
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: Globocan estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Venkateswaran, V.; Klotz, L.H.; Fleshner, N.E. Selenium modulation of cell proliferation and cell cycle biomarkers in human prostate carcinoma cell lines. Cancer Res. 2002, 62, 2540–2545. [Google Scholar] [PubMed]
- Sundaram, N.; Pahwa, A.K.; Ard, M.D.; Lin, N.; Perkins, E.; Bowles, A.P. Selenium causes growth inhibition and apoptosis in human brain tumor cell lines. J. Neurooncol. 2000, 46, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.D.; Jiang, C.; Malewicz, B.; Dong, Y.; Young, C.Y.; Kang, K.S.; Lee, Y.S.; Ip, C.; Lu, J. Methyl selenium metabolites decrease prostate-specific antigen expression by inducing protein degradation and suppressing androgen-stimulated transcription. Mol. Cancer Ther. 2004, 3, 605–611. [Google Scholar] [PubMed]
- Zhao, H.; Whitfield, M.L.; Xu, T.; Botstein, D.; Brooks, J.D. Diverse effects of methylseleninic acid on the transcriptional program of human prostate cancer cells. Mol. Biol. Cell 2004, 15, 506–519. [Google Scholar] [CrossRef] [PubMed]
- Chun, J.Y.; Nadiminty, N.; Lee, S.O.; Onate, S.A.; Lou, W.; Gao, A.C. Mechanisms of selenium down-regulation of androgen receptor signaling in prostate cancer. Mol. Cancer Ther. 2006, 5, 913–918. [Google Scholar] [CrossRef]
- Pacula, A.J.; Kaczor, K.B.; Wojtowicz, A.; Antosiewicz, J.; Janecka, A.; Dlugosz, A.; Janecki, T.; Scianowski, J. New glutathione peroxidase mimetics-insights into antioxidant and cytotoxic activity. Bioorg. Med. Chem. 2017, 25, 126–131. [Google Scholar] [CrossRef]
- Pacula, A.J.; Kaczor, K.B.; Antosiewicz, J.; Janecka, A.; Dlugosz, A.; Janecki, T.; Wojtczak, A.; Scianowski, J. New chiral ebselen analogues with antioxidant and cytotoxic potential. Molecules 2017, 22, 492. [Google Scholar] [CrossRef]
- Brunet, A.; Bonni, A.; Zigmond, M.J.; Lin, M.Z.; Juo, P.; Hu, L.S.; Anderson, M.J.; Arden, K.C.; Blenis, J.; Greenberg, M.E. Akt promotes cell survival by phosphorylating and inhibiting a forkhead transcription factor. Cell 1999, 96, 857–868. [Google Scholar] [CrossRef]
- Song, G.; Ouyang, G.; Bao, S. The activation of akt/pkb signaling pathway and cell survival. J. Cell Mol. Med. 2005, 9, 59–71. [Google Scholar] [CrossRef]
- Choi, B.H.; Xie, S.; Dai, W. Pten is a negative regulator of mitotic checkpoint complex during the cell cycle. Exp. Hematol. Oncol. 2017, 6, 19. [Google Scholar] [CrossRef] [PubMed]
- Pacula, A.J.; Scianowski, J.; Aleksandrzak, K.B. Highly efficient synthesis and antioxidant capacity of n-substituted benzisoselenazol-3(2h)-ones. RSC Adv. 2014, 4, 48959–48962. [Google Scholar] [CrossRef]
- Tiano, L.; Padella, L.; Santoro, L.; Carnevali, P.; Principi, F.; Bruge, F.; Gabrielli, O.; Littarru, G.P. Prolonged coenzyme q10 treatment in down syndrome patients: Effect on DNA oxidation. Neurobiol. Aging 2012, 33, 626.e1–626.e8. [Google Scholar] [CrossRef] [PubMed]
- Borkowska, A.; Sielicka-Dudzin, A.; Herman-Antosiewicz, A.; Wozniak, M.; Fedeli, D.; Falcioni, G.; Antosiewicz, J. Diallyl trisulfide-induced prostate cancer cell death is associated with akt/pkb dephosphorylation mediated by p-p66shc. Eur. J. Nutr. 2012, 51, 817–825. [Google Scholar] [CrossRef] [PubMed]
- DeGraffenried, L.A.; Fulcher, L.; Friedrichs, W.E.; Grunwald, V.; Ray, R.B.; Hidalgo, M. Reduced pten expression in breast cancer cells confers susceptibility to inhibitors of the pi3 kinase/akt pathway. Ann. Oncol. 2004, 15, 1510–1516. [Google Scholar] [CrossRef]
- Piscitello, D.; Varshney, D.; Lilla, S.; Vizioli, M.G.; Reid, C.; Gorbunova, V.; Seluanov, A.; Gillespie, D.A.; Adams, P.D. Akt overactivation can suppress DNA repair via p70s6 kinase-dependent downregulation of mre11. Oncogene 2018, 37, 427–438. [Google Scholar] [CrossRef]
- Al-Ani, A.; Toms, D.; Kondro, D.; Thundathil, J.; Yu, Y.; Ungrin, M. Oxygenation in cell culture: Critical parameters for reproducibility are routinely not reported. PLoS ONE 2018, 13, e0204269. [Google Scholar] [CrossRef]
- Miller, R.K.; Genbacev, O.; Turner, M.A.; Aplin, J.D.; Caniggia, I.; Huppertz, B. Human placental explants in culture: Approaches and assessments. Placenta 2005, 26, 439–448. [Google Scholar] [CrossRef]
- Zhang, K.; Zhao, T.; Huang, X.; He, Y.; Zhou, Y.; Wu, L.; Wu, K.; Fan, M.; Zhu, L. Dissolved oxygen concentration in the medium during cell culture: Defects and improvements. Cell Biol. Int. 2016, 40, 354–360. [Google Scholar] [CrossRef]
- Halliwell, B. Oxidative stress in cell culture: An under-appreciated problem? FEBS Lett. 2003, 540, 3–6. [Google Scholar] [CrossRef]
- Santofimia-Castano, P.; Garcia-Sanchez, L.; Ruy, D.C.; Fernandez-Bermejo, M.; Salido, G.M.; Gonzalez, A. The seleno-organic compound ebselen impairs mitochondrial physiology and induces cell death in ar42j cells. Toxicol. Lett. 2014, 229, 465–473. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Zhou, L.; Du, J.; Li, M.; Qian, C.; Cheng, Y.; Peng, Y.; Xie, J.; Wang, D. Induction of apoptosis in human multiple myeloma cell lines by ebselen via enhancing the endogenous reactive oxygen species production. Biomed. Res. Int. 2014, 2014, 696107. [Google Scholar] [CrossRef] [PubMed]
- Xie, Q.; Zhou, Y.; Lan, G.; Yang, L.; Zheng, W.; Liang, Y.; Chen, T. Sensitization of cancer cells to radiation by selenadiazole derivatives by regulation of ros-mediated DNA damage and erk and akt pathways. Biochem. Biophys. Res. Commun. 2014, 449, 88–93. [Google Scholar] [CrossRef] [PubMed]
- Morales, J.; Li, L.; Fattah, F.J.; Dong, Y.; Bey, E.A.; Patel, M.; Gao, J.; Boothman, D.A. Review of poly (adp-ribose) polymerase (parp) mechanisms of action and rationale for targeting in cancer and other diseases. Crit. Rev. Eukaryot. Gene Expr. 2014, 24, 15–28. [Google Scholar] [CrossRef]
- Nicotera, P.; Leist, M.; Ferrando-May, E. Intracellular atp, a switch in the decision between apoptosis and necrosis. Toxicol. Lett. 1998, 102–103, 139–142. [Google Scholar] [CrossRef]
- Whitacre, C.M.; Zborowska, E.; Willson, J.K.; Berger, N.A. Detection of poly(adp-ribose) polymerase cleavage in response to treatment with topoisomerase i inhibitors: A potential surrogate end point to assess treatment effectiveness. Clin. Cancer Res. 1999, 5, 665–672. [Google Scholar]
- Rovere-Querini, P.; Capobianco, A.; Scaffidi, P.; Valentinis, B.; Catalanotti, F.; Giazzon, M.; Dumitriu, I.E.; Muller, S.; Iannacone, M.; Traversari, C.; et al. Hmgb1 is an endogenous immune adjuvant released by necrotic cells. EMBO Rep. 2004, 5, 825–830. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaczor-Keller, K.B.; Pawlik, A.; Scianowski, J.; Pacuła, A.; Obieziurska, M.; Marcheggiani, F.; Cirilli, I.; Tiano, L.; Antosiewicz, J. In Vitro Anti-Prostate Cancer Activity of Two Ebselen Analogues. Pharmaceuticals 2020, 13, 47. https://doi.org/10.3390/ph13030047
Kaczor-Keller KB, Pawlik A, Scianowski J, Pacuła A, Obieziurska M, Marcheggiani F, Cirilli I, Tiano L, Antosiewicz J. In Vitro Anti-Prostate Cancer Activity of Two Ebselen Analogues. Pharmaceuticals. 2020; 13(3):47. https://doi.org/10.3390/ph13030047
Chicago/Turabian StyleKaczor-Keller, Katarzyna B., Anna Pawlik, Jacek Scianowski, Agata Pacuła, Magdalena Obieziurska, Fabio Marcheggiani, Ilenia Cirilli, Luca Tiano, and Jedrzej Antosiewicz. 2020. "In Vitro Anti-Prostate Cancer Activity of Two Ebselen Analogues" Pharmaceuticals 13, no. 3: 47. https://doi.org/10.3390/ph13030047
APA StyleKaczor-Keller, K. B., Pawlik, A., Scianowski, J., Pacuła, A., Obieziurska, M., Marcheggiani, F., Cirilli, I., Tiano, L., & Antosiewicz, J. (2020). In Vitro Anti-Prostate Cancer Activity of Two Ebselen Analogues. Pharmaceuticals, 13(3), 47. https://doi.org/10.3390/ph13030047







