Effect of Tocilizumab in Hospitalized Patients with Severe COVID-19 Pneumonia: A Case-Control Cohort Study
Abstract
1. Introduction
2. Results
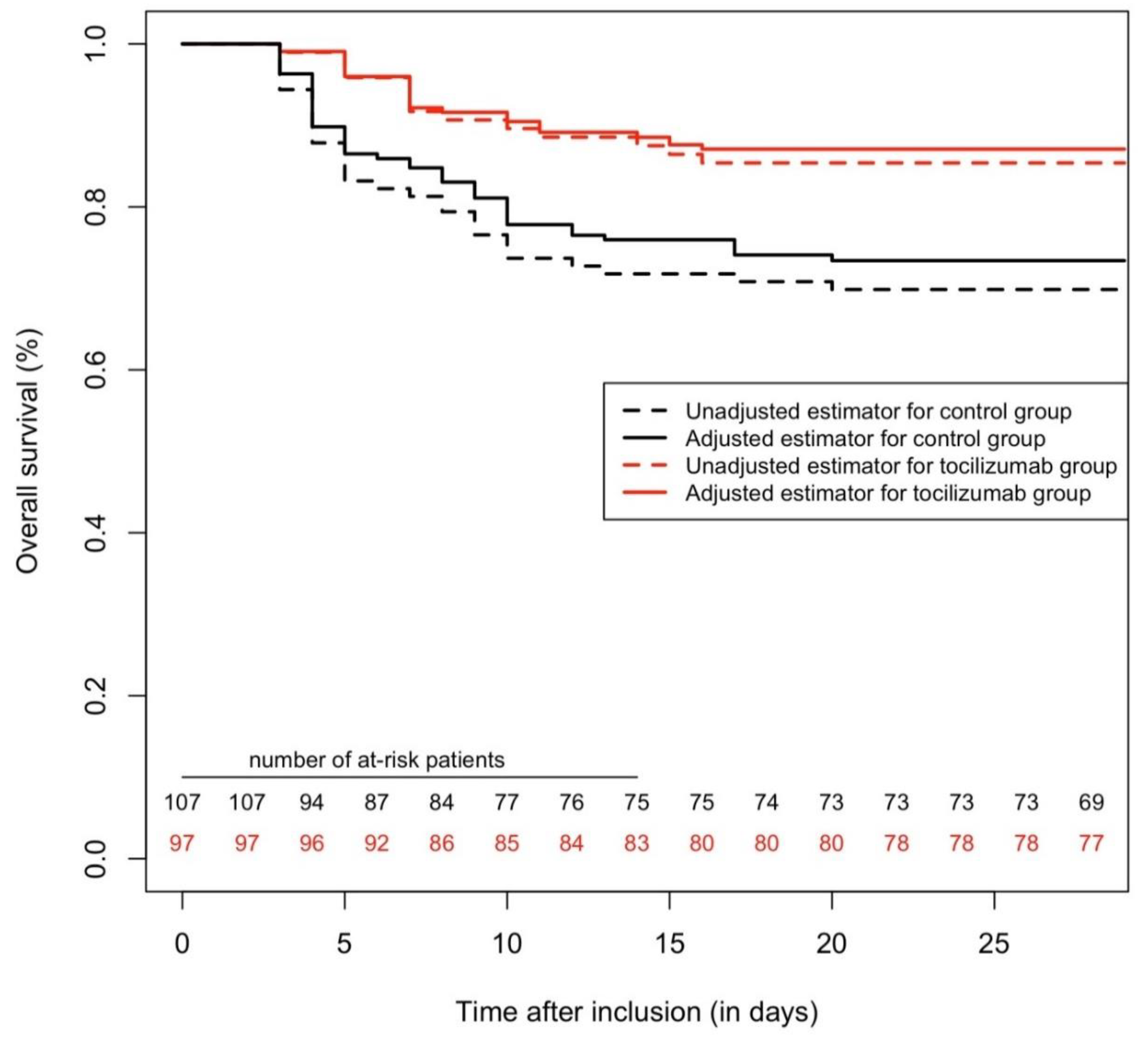
2.1. Propensity-Score Matched Cohort
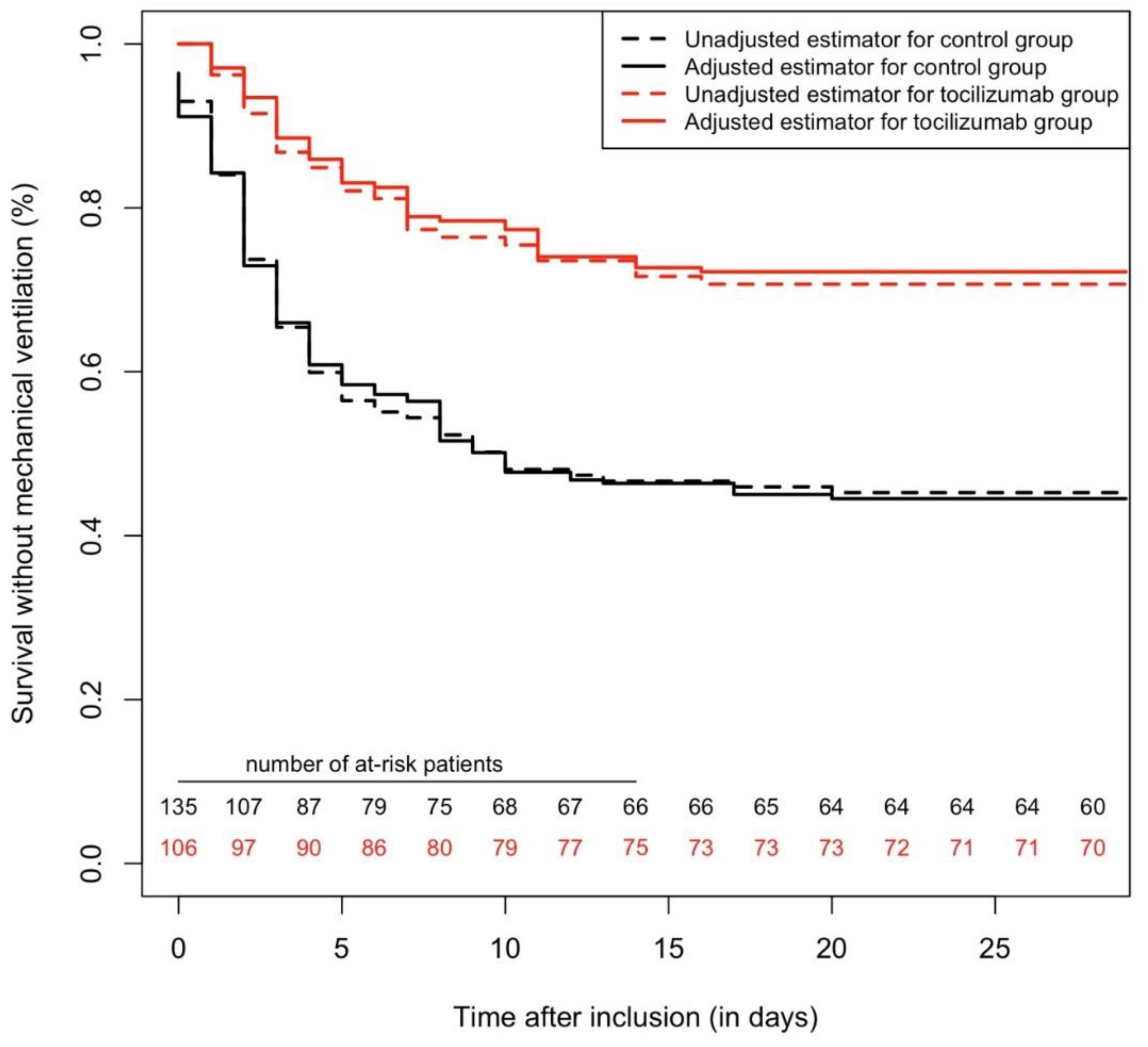
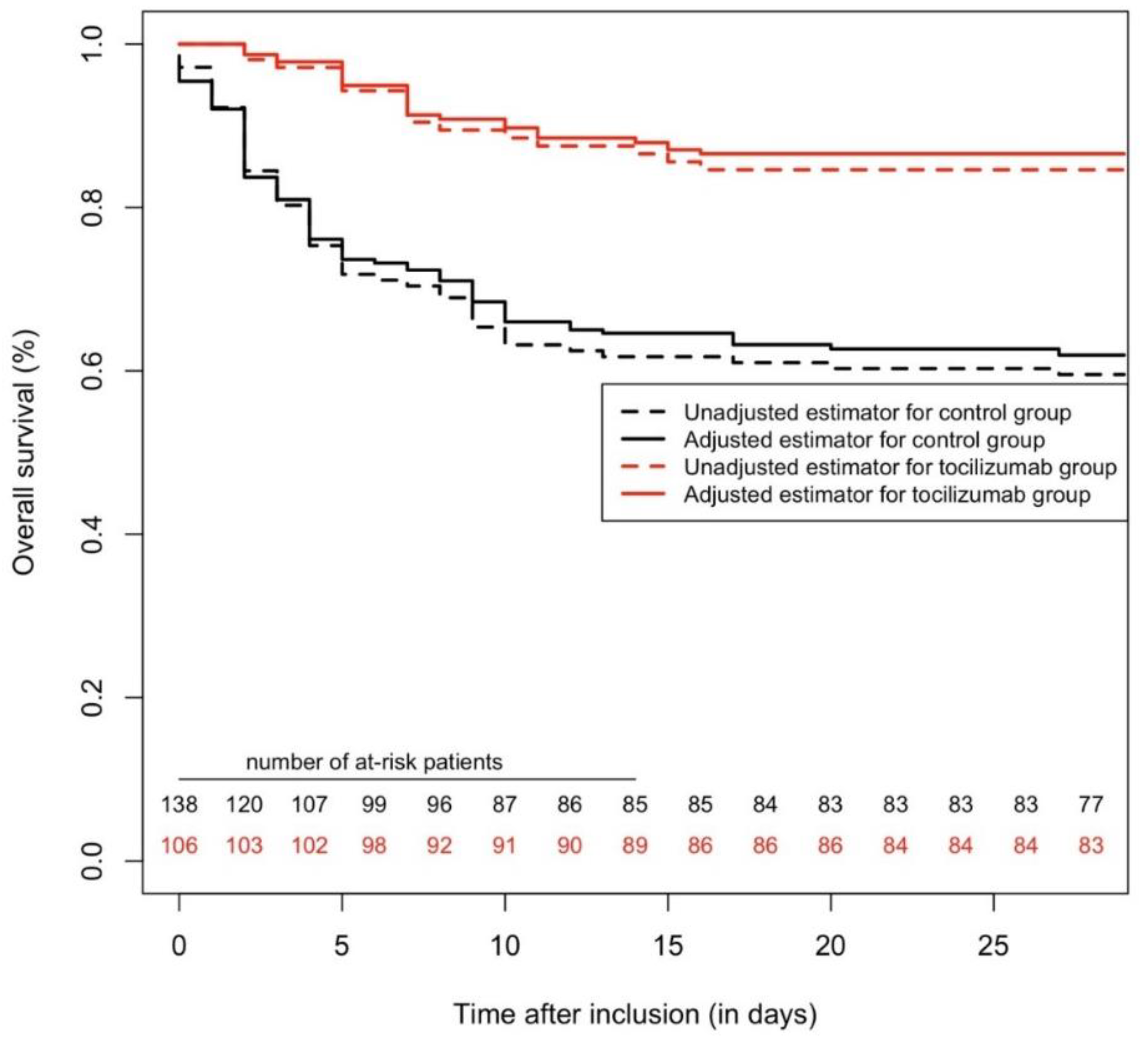
2.2. Overall Cohort (Cox Multivariable and IPSW Analyses)
2.3. All-Cause Mortality (28 Days Maximum Follow-Up)
2.4. Sensitivity Analyses
3. Discussion
4. Materials and Methods
4.1. Study Population
4.2. Study Design
4.3. Treatment Intervention
4.4. Study Variables
4.5. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Spiteri, G.; Fielding, J.; Diercke, M.; Campese, C.; Enouf, V.; Gaymard, A.; Bella, A.; Sognamiglio, P.; Sierra Moros, M.J.; Riutort, A.N.; et al. First Cases of Coronavirus Disease 2019 (COVID-19) in the WHO European Region, 24 January to 21 February 2020. Eurosurveillance 2020, 25. [Google Scholar] [CrossRef] [PubMed]
- Grasselli, G.; Zangrillo, A.; Zanella, A.; Antonelli, M.; Cabrini, L.; Castelli, A.; Cereda, D.; Coluccello, A.; Foti, G.; Fumagalli, R.; et al. Baseline Characteristics and Outcomes of 1591 Patients Infected With SARS-CoV-2 Admitted to ICUs of the Lombardy Region, Italy. JAMA 2020, 323, 1574. [Google Scholar] [CrossRef] [PubMed]
- Mehta, P.; McAuley, D.F.; Brown, M.; Sanchez, E.; Tattersall, R.S.; Manson, J.J. COVID-19: Consider Cytokine Storm Syndromes and Immunosuppression. Lancet 2020, 395, 1033–1034. [Google Scholar] [CrossRef]
- Le, R.Q.; Li, L.; Yuan, W.; Shord, S.S.; Nie, L.; Habtemariam, B.A.; Przepiorka, D.; Farrell, A.T.; Pazdur, R. FDA Approval Summary: Tocilizumab for Treatment of Chimeric Antigen Receptor T Cell-Induced Severe or Life-Threatening Cytokine Release Syndrome. Oncologist 2018, 23, 943–947. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Li, M.; Zhou, Z.; Guan, X.; Xiang, Y. Can We Use Interleukin-6 (IL-6) Blockade for Coronavirus Disease 2019 (COVID-19)-Induced Cytokine Release Syndrome (CRS)? J. Autoimmun. 2020, 111, 102452. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Wu, Z.; Li, J.-W.; Zhao, H.; Wang, G.-Q. Cytokine Release Syndrome in Severe COVID-19: Interleukin-6 Receptor Antagonist Tocilizumab May Be the Key to Reduce Mortality. Int. J. Antimicrob. Agents 2020, 55, 105954. [Google Scholar] [CrossRef] [PubMed]
- Richardson, S.; Hirsch, J.S.; Narasimhan, M.; Crawford, J.M.; McGinn, T.; Davidson, K.W.; the Northwell COVID-19 Research Consortium; Barnaby, D.P.; Becker, L.B.; Chelico, J.D.; et al. Presenting Characteristics, Comorbidities, and Outcomes Among 5700 Patients Hospitalized With COVID-19 in the New York City Area. JAMA 2020, 323, 2052. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Liu, Y.; Zhou, L.; Chen, E.; Liu, P.; Pan, X.; Lu, Y. Epidemiological Assessment of Imported Coronavirus Disease 2019 (COVID-19) Cases in the Most Affected City Outside of Hubei Province, Wenzhou, China. JAMA Netw. Open 2020, 3, e206785. [Google Scholar] [CrossRef] [PubMed]
- Huet, T.; Beaussier, H.; Voisin, O.; Jouveshomme, S.; Dauriat, G.; Lazareth, I.; Sacco, E.; Naccache, J.-M.; Bézie, Y.; Laplanche, S.; et al. Anakinra for Severe Forms of COVID-19: A Cohort Study. Lancet Rheumatol. 2020, 2, e393–e400. [Google Scholar] [CrossRef]
- The RECOVERY Collaborative Group. Dexamethasone in Hospitalized Patients with Covid-19—Preliminary Report. N. Engl. J. Med. 2020. [Google Scholar] [CrossRef]
- Horby, P.; Lim, W.S.; Emberson, J.; Mafham, M.; Bell, J.; Linsell, L.; Staplin, N.; Brightling, C.; Ustianowski, A.; Elmahi, E.; et al. Effect of Dexamethasone in Hospitalized Patients with COVID-19: Preliminary Report. Infect. Dis. 2020. [Google Scholar] [CrossRef]
- Toniati, P.; Piva, S.; Cattalini, M.; Garrafa, E.; Regola, F.; Castelli, F.; Franceschini, F.; Airò, P.; Bazzani, C.; Beindorf, E.-A.; et al. Tocilizumab for the Treatment of Severe COVID-19 Pneumonia with Hyperinflammatory Syndrome and Acute Respiratory Failure: A Single Center Study of 100 Patients in Brescia, Italy. Autoimmun. Rev. 2020, 19, 102568. [Google Scholar] [CrossRef] [PubMed]
- Guaraldi, G.; Meschiari, M.; Cozzi-Lepri, A.; Milic, J.; Tonelli, R.; Menozzi, M.; Franceschini, E.; Cuomo, G.; Orlando, G.; Borghi, V.; et al. Tocilizumab in Patients with Severe COVID-19: A Retrospective Cohort Study. Lancet Rheumatol. 2020, 2, e474–e484. [Google Scholar] [CrossRef]
- CORIMUNO-19-Tocilizumab Trial-TOCI (CORIMUNO-TOCI). 2020. Available online: https://clinicaltrials.gov/ct2/show/NCT04331808201202317 (accessed on 15 October 2020).
- A Study to Evaluate the Safety and Efficacy of Tocilizumab in Patients With Severe COVID-19 Pneumonia (COVACTA). 2020. Available online: https://clinicaltrials.gov/ct2/show/NCT04320615 (accessed on 15 October 2020).
- Chiu, Y.; Ostor, A.J.K.; Hammond, A.; Sokoll, K.; Anderson, M.; Buch, M.; Ehrenstein, M.R.; Gordon, P.; Steer, S.; Bruce, I.N. Access to the next Wave of Biologic Therapies (Abatacept and Tocilizumab) for the Treatment of Rheumatoid Arthritis in England and Wales: Addressing Treatment Outside the Current NICE Guidance. Clin. Rheumatol. 2012, 31, 1005–1012. [Google Scholar] [CrossRef] [PubMed]
- Abdallah, H.; Hsu, J.C.; Lu, P.; Fettner, S.; Zhang, X.; Douglass, W.; Bao, M.; Rowell, L.; Burmester, G.R.; Kivitz, A. Pharmacokinetic and Pharmacodynamic Analysis of Subcutaneous Tocilizumab in Patients With Rheumatoid Arthritis From 2 Randomized, Controlled Trials: SUMMACTA and BREVACTA: Pharmacokinetic and Pharmacodynamic Analysis of Subcutaneous. J. Clin. Pharmacol. 2017, 57, 459–468. [Google Scholar] [CrossRef] [PubMed]
- Lunceford, J.K.; Davidian, M. Stratification and Weighting via the Propensity Score in Estimation of Causal Treatment Effects: A Comparative Study. Stat. Med. 2004, 23, 2937–2960. [Google Scholar] [CrossRef] [PubMed]
- Austin, P.C.; Stuart, E.A. The Performance of Inverse Probability of Treatment Weighting and Full Matching on the Propensity Score in the Presence of Model Misspecification When Estimating the Effect of Treatment on Survival Outcomes. Stat. Methods Med. Res. 2017, 26, 1654–1670. [Google Scholar] [CrossRef] [PubMed]
- Bilan, N.; Dastranji, A.; Ghalehgolab Behbahani, A. Comparison of the Spo2 /Fio2 Ratio and the Pao 2 /Fio 2 Ratio in Patients With Acute Lung Injury or Acute Respiratory Distress Syndrome. J. Cardiovasc. Thorac. Res. 2015, 7, 28–31. [Google Scholar] [CrossRef] [PubMed]
- Rubin, G.D.; Ryerson, C.J.; Haramati, L.B.; Sverzellati, N.; Kanne, J.P.; Raoof, S.; Schluger, N.W.; Volpi, A.; Yim, J.-J.; Martin, I.B.K.; et al. The Role of Chest Imaging in Patient Management During the COVID-19 Pandemic. Chest 2020, 158, 106–116. [Google Scholar] [CrossRef] [PubMed]

| Control Group (n = 140) | Tocilizumab Group (n = 106) | p-Value | Matched Control (n = 84) | Matched Tocilizumab Group (n = 84) | p-Value | |
|---|---|---|---|---|---|---|
| Clinical Features, No (%) | ||||||
| Age, mean ± Standard deviation (SD), years | 70.1 ± 16.5 | 64.3 ± 13.0 | 0.003 * | 64.4 ± 16.9 | 64.8 ± 12.8 | 0.88 |
| Female | 59 (42.1%) | 36 (34.0%) | 0.19 † | 35 (41.7%) | 29 (34.5%) | 0.24 † |
| Full engagement | 72 (51.4%) | 73 (68.9%) | 0.006 † | 56 (66.7%) | 58 (69%) | 0.74 † |
| Diabetes | 53 (37.9%) | 48 (45.3%) | 0.24 † | 31 (36.9%) | 35 (41.7%) | 0.53 † |
| Insulin treatment | 23 (16.4%) | 17 (16.0%) | 0.93 † | 12 (14.3%) | 9 (10.7%) | 0.48 † |
| Obesity (BMI > 30 kg/m2) | 40 (28.6%) | 34 (32.1%) | 0.55 † | 26 (31.0%) | 27 (32.1%) | 0.87 † |
| Hypertension | 79 (56.4%) | 64 (60.4%) | 0.53 † | 47 (56.0%) | 47 (56.0%) | 1 † |
| ACEI treatment | 21 (15.0%) | 16 (15.1%) | 0.98 † | 11 (13.1%) | 12 (14.3%) | 0.82 † |
| ARB treatment | 22 (15.7%) | 28 (26.4%) | 0.039 † | 14 (16.7%) | 20 (23.8%) | 0.25 † |
| History of cardiovascular disease, stroke, peripheral artery disease, heart failure | 43 (30.7%) | 25 (23.6%) | 0.22 † | 21 (25.0%) | 21 (25.0%) | 0.1 † |
| Smoker (active or past) | 29 (20.7%) | 33 (31.1%) | 0.06 † | 21 (25.0%) | 23 (27.4%) | 0.73 † |
| History of COPD, asthma, emphysema, fibrosis | 22 (15.7%) | 17 (16.0%) | 0.369 † | 12 (14.3%) | 15 (17.9%) | 0.53 † |
| eGFR < 60 mL/min/1.73 m2 | 24 (17.1%) | 17 (16.0%) | 0.82 † | 13 (15.5%) | 12 (14.3%) | 0.83 † |
| Solid organ transplantation | 3 (2.1%) | 0 (0.0%) | 0.26 ‡ | 2 (2.4%) | 0 (0.0%) | 0.5 ‡ |
| HIV | 1 (0.7%) | 1 (0.9%) | 1 ‡ | 1 (1.2%) | 0 (0.0%) | 1 ‡ |
| Immunosuppressant drugs | 12 (8.6%) | 5 (4.7%) | 0.24 ‡ | 4 (4.8%) | 4 (4.8%) | 1 ‡ |
| Long-term oral corticosteroids | 13 (9.3%) | 5 (4.7%) | 0.17 ‡ | 6 (7.1%) | 4 (4.8%) | 0.75 ‡ |
| Malignancy (active) | 17 (12.1%) | 6 (5.7%) | 0.08 † | 7 (8.3%) | 5 (6.0%) | 0.55 ‡ |
| Treatments after Admission | ||||||
| No.(%) under antibiotics | 134 (95.7%) | 106 (100.0%) | 0.04 ‡ | 84 (100.0%) | 84 (100.0%) | NA |
| Betalactamin | 129 (92.1%) | 105 (99.1%) | 0.13 ‡ | 81 (96.4%) | 83 (98.8%) | 0.62 ‡ |
| Macrolide | 98 (70.0%) | 93 (87.7%) | 0.001 † | 64 (76.2%) | 75 (89.3%) | 0.025 † |
| Others | 21 (15.0%) | 7 (6.6%) | 0.04 † | 15 (17.9%) | 6 (7.1%) | 0.036 † |
| No.(%) under antiviral therapy | 106 (75.7%) | 88 (83.0%) | 0.16 † | 68 (81.0%) | 68 (81.0%) | 1 ‡ |
| Hydroxychloroquine | 100 (71.4%) | 88 (83.0%) | 0.034 † | 62 (73.8%) | 68 (81.0%) | 0.27 † |
| Lopinavir/ritonavir | 8 (5.7%) | 1 (0.9%) | 0.08 ‡ | 8 (9.5%) | 1 (1.2%) | 0.03 ‡ |
| Immunosuppressants and/or corticosteroids | 47 (33.6%) | 43 (40.6%) | 0.26 † | 24 (28.6%) | 26 (31.0%) | 0.74 † |
| Baricitinib | 18 (12.9%) | 1 (0.9%) | 0.001 ‡ | 0 (0.0%) | 1 (1.2%) | 1 ‡ |
| Characteristics at Hospital Admission | ||||||
| Delay between first symptoms and admission, means, days | 5.4 ± 8.8 | 4.8 ± 15.3 | 0.74 * | 5.2 ± 9.8 | 6.5 ± 4.9 | 0.29 * |
| SpO2/FiO2 ratio, mean SD | 327.4 ± 108.7 | 338.4 ± 88.6 | 0.39 * | 330.3 ± 108.6 | 341.8 ± 87.1 | 0.45 * |
| SpO2, mean SD, % | 93.9 ± 3.6 | 94.0 ± 3.9 | 0.81 * | 94.0 ± 3.6 | 94.2 ± 3.3 | 0.62 § |
| Oxygen flow, mean SD, L/min | 4.0 ± 4.8 | 3.1 ± 3.6 | 0.44 § | 3.9 ± 4.6 | 3.0 ± 3.3 | 0.54 § |
| PaO2, mean SD, mmHg | 70.4 ± 24.7 | 68.6 ± 17.5 | 0.54 * | 71.6 ± 28.4 | 68.6 ± 17.8 | 0.46 * |
| Temperature, mean SD, °C | 37.2 ± 1.1 | 37.5 ± 1.2 | 0.06 * | 37.3 ± 1.1 | 37.5 ± 1.2 | 0.22 * |
| C-reactive protein, mean SD, mg/L | 132.1 ± 99.2 | 135.2 ± 88.2 | 0.79 * | 138.2 ± 101.7 | 131.5 ± 84.6 | 0.64 * |
| Lymphocyte count, mean SD, mcL | 1451.9 ± 3320.1 | 1198.6 ± 1095.7 | 0.40 * | 1595.9 ± 4290.1 | 1228,7 ± 1204.0 | 0.46 * |
| Characteristics at Study Inclusion | ||||||
| Delay between first symptoms and inclusion, mean SD, days | 8.4 ± 4.7 | 8.3 ± 4.2 | 0.82 * | 8.4 ± 4,8 | 8.6 ± 4.2 | 0.84 * |
| Delay between admission and inclusion, mean SD, days | 3.0 ± 8.0 | 3.5 ± 14.6 | 0.28 § | 3.2 ± 8.5 | 2,2 ± 3.3 | 0.61 § |
| SpO2/FiO2 ratio, mean SD, | 212.9 ± 41.7 | 199.4 ± 49.8 | 0.02 * | 210.9 ± 41.9 | 206.5 ± 48.7 | 0.53 * |
| SpO2, mean SD, % | 94.5 ± 3.6 | 94.0 ± 3.7 | 0.32 * | 94.3 ± 4.0 | 94.2 ± 3.6 | 0.84 * |
| O2 flow support, mean SD, L/min | 8.4 ± 3.4 | 9.9 ± 5.3 | 0.03 § | 8.6 ± 3.4 | 9,3 ± 4.8 | 0.59 § |
| PaO2, mean SD, mmHg | 74.3 ± 25.4 | 76.8 ± 24.5 | 0.51 * | 75.7 ± 29.0 | 78.3 ± 24.8 | 0.59 * |
| PaCO2, mean SD, mmHg | 36.0 ± 9.1 | 35.9 ± 7.7 | 0.93 * | 36.3 ± 9.0 | 35.6 ± 7.6 | 0.63 * |
| Systolic blood pressure, mean SD, mmHg | 130.7 ± 20.0 | 131.4 ± 20.9 | 0.8 * | 131.3 ± 21.2 | 130.8 ± 20.3 | 0.9 * |
| Temperature, mean SD, °C | 37.3 ± 1.2 | 37.3 ± 1.2 | 0.61 § | 37.3 ± 1.3 | 37.3 ± 1.1 | 0.55 § |
| C-reactive protein, mean SD, mg/L | 144.9 ± 100.7 | 168.0 ± 95.0 | 0.07 * | 150.8 ± 105.1 | 163.7 ± 97.9 | 0.42 * |
| Lymphocyte count, mean SD, mcL | 1224.0 ± 2294.0 | 1128.4 ± 1010.8 | 0.66 § | 1278.8 ± 2907.4 | 1168.8 ± 1108.5 | 0.75 § |
| eGFR, mean SD, mL/min/1.73 m2 | 72.6 ± 34.7 | 152.2 ± 701.9 | 0.25 * | 75.2 ± 34.5 | 171.8 ± 788.2 | 0.27 * |
| Use of non-invasive ventilation or high flow oxygenotherapy after inclusion | 5 (3.6%) | 8 (7.5%) | 0.17 | 4 (4.8%) | 4 (4.8%) | 1.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rossi, B.; Nguyen, L.S.; Zimmermann, P.; Boucenna, F.; Dubret, L.; Baucher, L.; Guillot, H.; Bouldouyre, M.-A.; Allenbach, Y.; Salem, J.-E.; et al. Effect of Tocilizumab in Hospitalized Patients with Severe COVID-19 Pneumonia: A Case-Control Cohort Study. Pharmaceuticals 2020, 13, 317. https://doi.org/10.3390/ph13100317
Rossi B, Nguyen LS, Zimmermann P, Boucenna F, Dubret L, Baucher L, Guillot H, Bouldouyre M-A, Allenbach Y, Salem J-E, et al. Effect of Tocilizumab in Hospitalized Patients with Severe COVID-19 Pneumonia: A Case-Control Cohort Study. Pharmaceuticals. 2020; 13(10):317. https://doi.org/10.3390/ph13100317
Chicago/Turabian StyleRossi, Benjamin, Lee S. Nguyen, Philippe Zimmermann, Faiza Boucenna, Louis Dubret, Louise Baucher, Helene Guillot, Marie-Anne Bouldouyre, Yves Allenbach, Joe-Elie Salem, and et al. 2020. "Effect of Tocilizumab in Hospitalized Patients with Severe COVID-19 Pneumonia: A Case-Control Cohort Study" Pharmaceuticals 13, no. 10: 317. https://doi.org/10.3390/ph13100317
APA StyleRossi, B., Nguyen, L. S., Zimmermann, P., Boucenna, F., Dubret, L., Baucher, L., Guillot, H., Bouldouyre, M.-A., Allenbach, Y., Salem, J.-E., Barsoum, P., Oufella, A., & Gros, H. (2020). Effect of Tocilizumab in Hospitalized Patients with Severe COVID-19 Pneumonia: A Case-Control Cohort Study. Pharmaceuticals, 13(10), 317. https://doi.org/10.3390/ph13100317





