Tafenoquine: A 2018 Novel FDA-Approved Prodrug for the Radical Cure of Plasmodium vivax Malaria and Prophylaxis of Malaria
Abstract
1. Introduction
2. Tafenoquine
2.1. Names
2.2. Uses
2.3. Mechanisms of Action
2.4. Clinical Studies
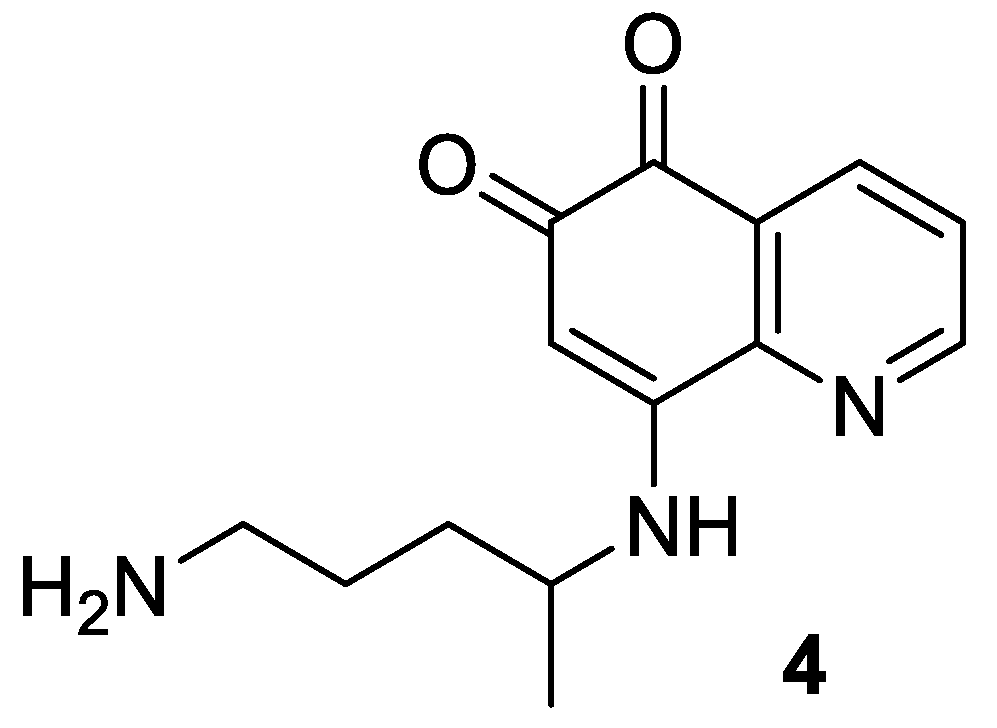
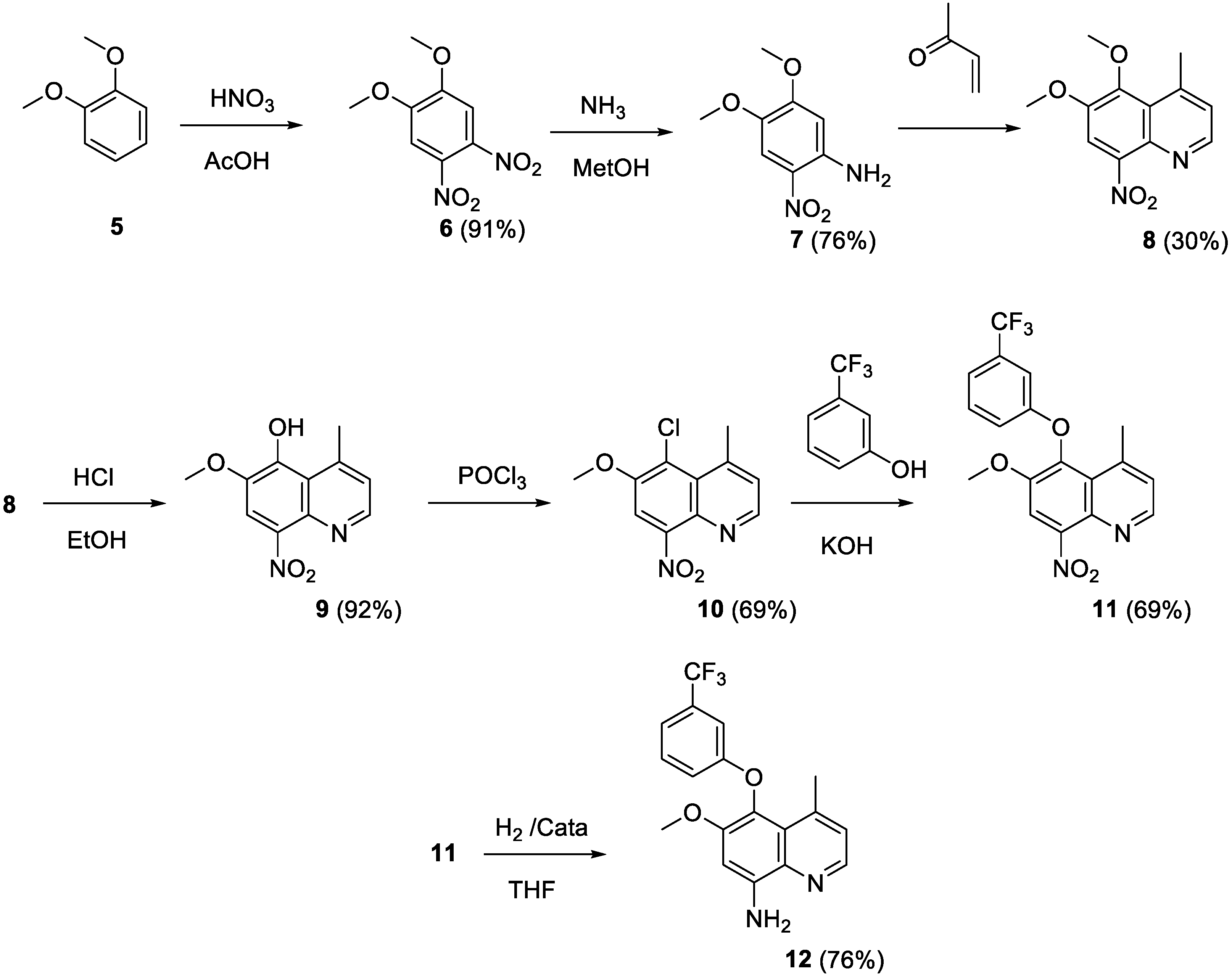
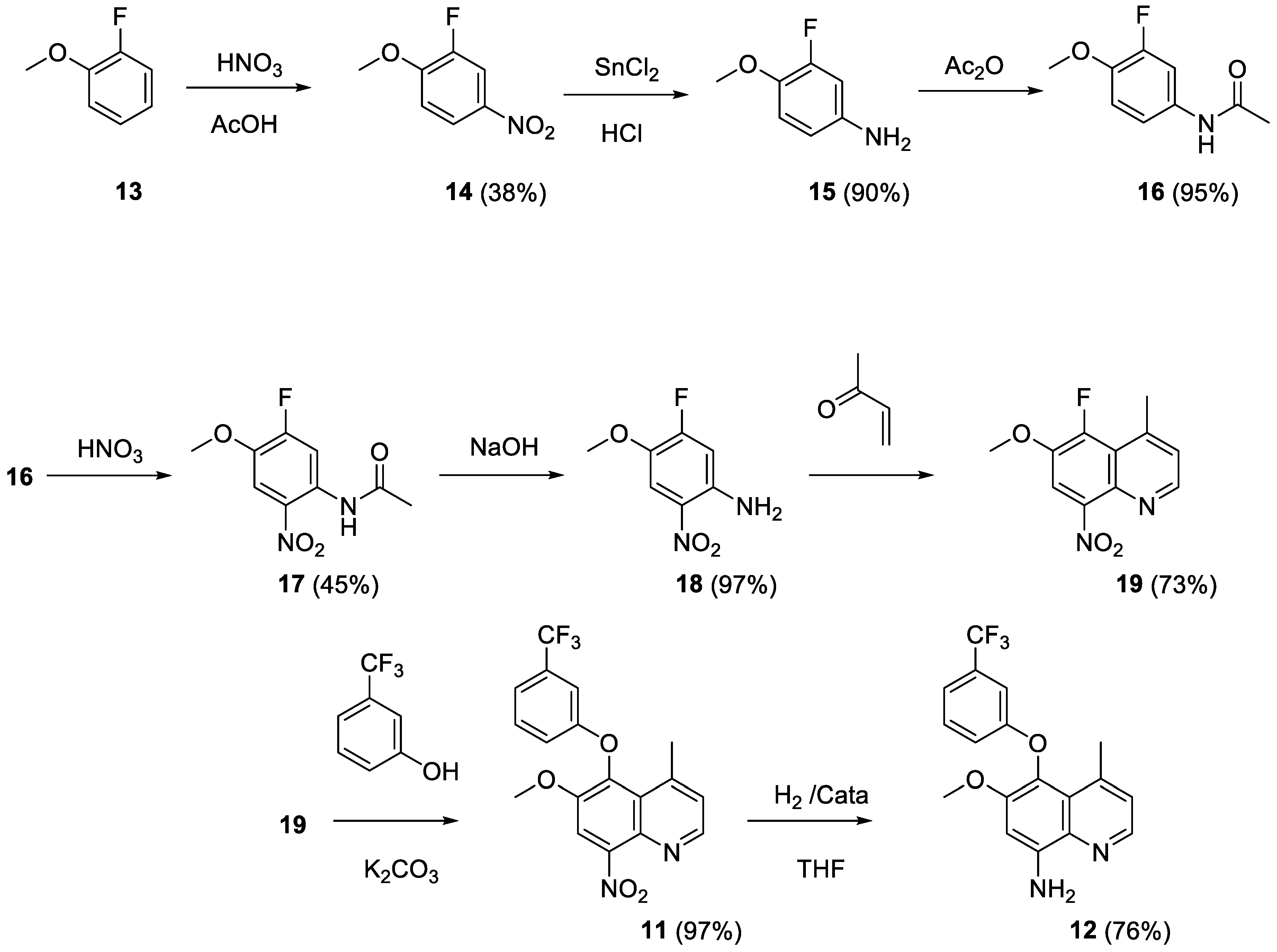
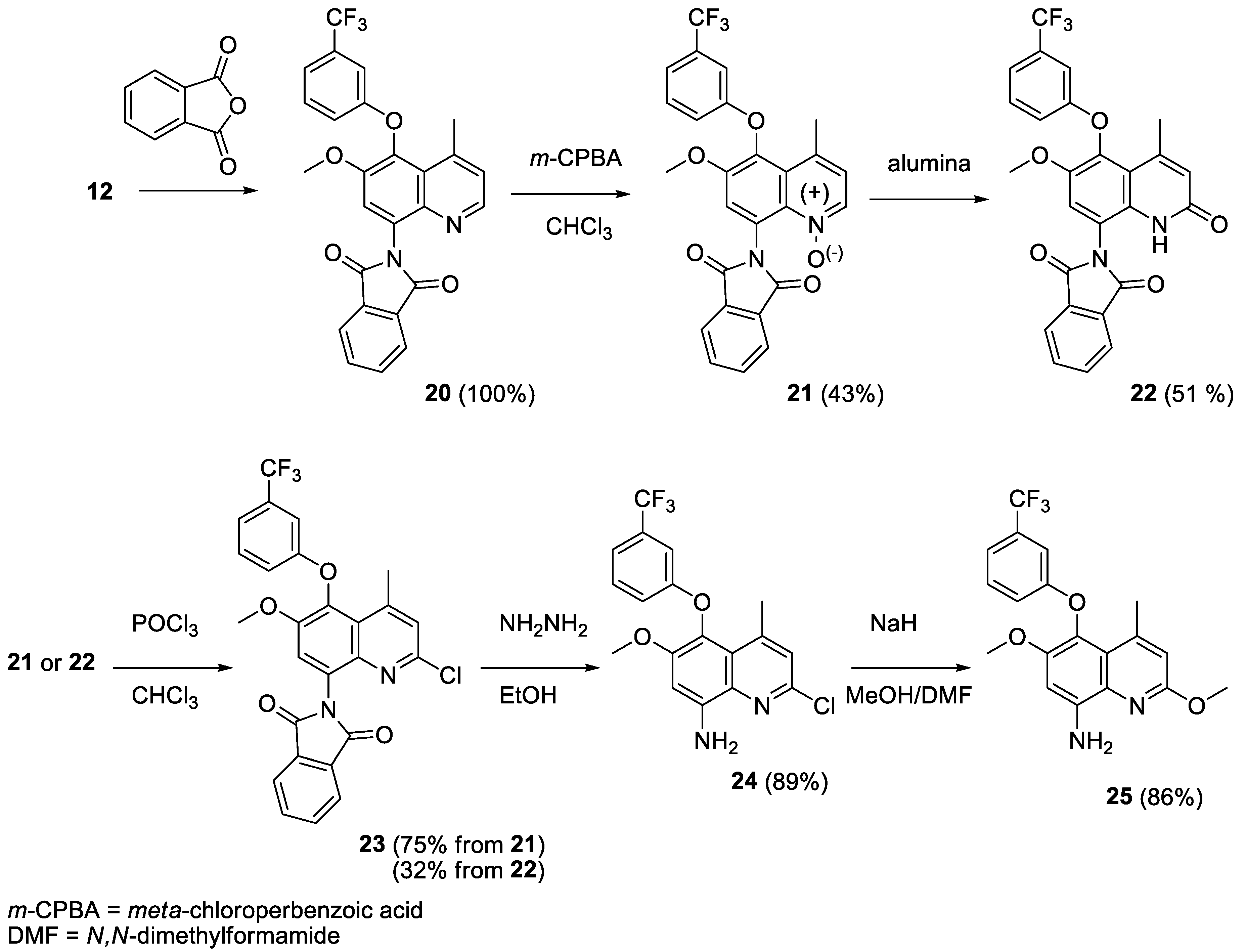
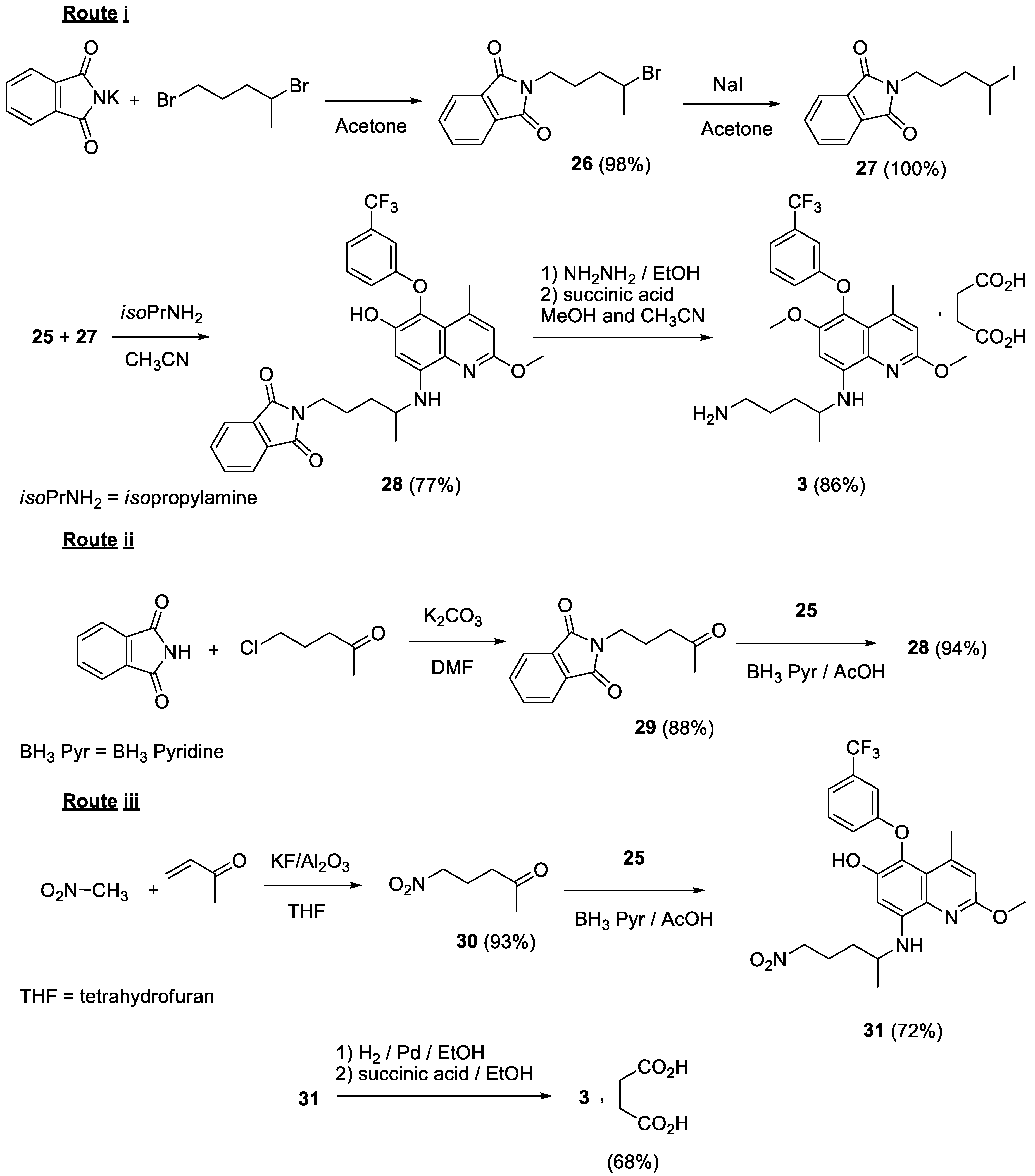
2.5. Syntheses
3. Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- World Malaria Report 2018. Available online: https://www.who.int/malaria/publications/world-malaria-report-2018/report/en/ (accessed on 19 November 2018).
- Campo, B.; Vandal, O.; Wesche, D.L.; Burrows, J.N. Killing the Hypnozoite—Drug Discovery Approaches to Prevent Relapse in Plasmodium vivax. Pathog. Glob. Health 2015, 109, 107–122. [Google Scholar] [CrossRef]
- World Health Organization. Guidelines for the Treatment of Malaria, 3rd ed.; World Health Organization: Geneva, Switzerland, 2015; Volume 9, pp. 93–95. [Google Scholar]
- Allgower, A.; Taylor, W.R.; Chappuis, F.; Eperon, G. Plasmodium vivax, un Parasite qui Sort de l’Ombre. Rev. Med. Suisse 2016, 12, 876–881. [Google Scholar] [PubMed]
- Krintafel (Tafenoquine)—FDA. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/210795s000lbl.pdf (accessed on 16 July 2019).
- Arakoda (Tafenoquine) Tablets—FDA. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/210607lbl.pdf (accessed on 16 July 2019).
- Marcsisin, S.R.; Reichard, G.; Pybus, B.S. Primaquine Pharmacology in the Context of CYP 2D6 Pharmacogenomics: Current State of the Art. Pharmacol. Ther. 2016, 161, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Marcsisin, S.R.; Sousa, J.C.; Reichard, G.A.; Caridha, D.; Zeng, Q.; Roncal, N.; McNulty, R.; Careagabarja, J.; Sciotti, R.J.; Bennett, J.W.; et al. Tafenoquine and NPC-1161B require CYP 2D metabolism for anti-malarial activity: implications for the 8-aminoquinoline class of anti-malarial compounds. Malaria J. 2014, 13, 2. [Google Scholar] [CrossRef] [PubMed]
- Delves, M.; Plouffe, D.; Scheurer, C.; Meister, S.; Wittlin, S.; Winzeler, E.A.; Sinden, R.E.; Leroy, D. The Activities of Current Antimalarial Drugs on the Life Cycle Stages of Plasmodium: A Comparative Study with Human and Rodent Parasites. PLoS Med. 2012, 9, e1001169. [Google Scholar] [CrossRef] [PubMed]
- Vuong, C.; Xie, L.H.; Potter, B.M.J.; Zhang, J.; Zhang, P.; Duan, D.; Nolan, C.K.; Sciotti, R.J.; Zottig, V.E.; Nanayakkara, N.P.D.; et al. Differential Cytochrome P450 2D Metabolism Alters Tafenoquine Pharmacokinetics. Antimicrob. Agents Chemother. 2015, 59, 3864–3869. [Google Scholar] [CrossRef] [PubMed]
- Watson, J.; Taylor, W.R.J.; Bancone, G.; Chu, C.S.; Jittamala, P.; White, N.J. Implications of Current Therapeutic Restrictions for Primaquine and Tafenoquine in the Radical Cure of vivax Malaria. PLoS Negl. Trop. Dis. 2018, 12, e0006440. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; O’Neil, M.; Xie, L.; Caridha, D.; Zeng, Q.; Zhang, J.; Pybus, B.; Hickman, M.; Melendez, V. Assessment of the Prophylactic Activity and Pharmacokinetic Profile of Oral Tafenoquine Compared to Primaquine for Inhibition of Liver Stage Malaria Infections. Malaria J. 2014, 13, 141. [Google Scholar] [CrossRef] [PubMed]
- Brueckner, R.P.; Lasseter, R.P.; Lin, E.T.; Schuste, B.G. First-Time-in-Humans Safety and Pharmacokinetics of WR 238605, a New Antimalarial. Am. J. Trop. Med. Hyg. 1998, 58, 645–649. [Google Scholar] [CrossRef] [PubMed]
- NIH US National Library of Medicine. Available online: https://www.clinicaltrials.gov/ct2/results?term=tafenoquine&age_v=&gndr=&type=&rslt=&Search=Apply (accessed on 16 July 2019).
- Miller, A.K.; Harrell, E.; Ye, L.; Baptiste-Brown, S.; Kleim, J.P.; Ohrt, C.; Duparc, S.; Möhrle, J.J.; Webster, A.; Stinnett, S.; et al. Pharmacokinetic Interactions and Safety Evaluations of Coadministered Tafenoquine and Chloroquine in Healthy Subjects. Br. J. Clin. Pharmacol. 2013, 76, 858–867. [Google Scholar] [CrossRef] [PubMed]
- Llanos-Cuentas, A.; Lacerda, M.V.G.; Hien, T.T.; Vélez, I.D.; Namaik-larp, C.; Chu, C.S.; Villegas, M.F.; Val, F.; Monteiro, W.M.; Brito, M.A.M.; et al. Tafenoquine versus Primaquine to Prevent Relapse of Plasmodium vivax Malaria. N. Eng. J. Med. 2019, 380, 229–241. [Google Scholar] [CrossRef] [PubMed]
- 4-Methyl-5-(unsubstituted and substituted phenoxy)-6-methoxy-8-(aminoalkylamino)quinolines. Available online: https://patents.google.com/patent/US4431807A/en (accessed on 29 July 2019).
- 4-Methyl-5-(unsubstituted and substituted phenoxy)-2,6-dimethoxy-8-(aminoalkylamino)quinolines. Available online: https://patents.google.com/patent/US4617394A/en (accessed on 29 July 2019).
- Process for the Preparation of Anti-malarial Drugs. Available online: https://patents.google.com/patent/WO1997013753A1/en (accessed on 29 July 2019).
- Process for the Preparation of Anti-malarial Drugs. Available online: https://patents.google.com/patent/US6479660B1/en (accessed on 29 July 2019).
- Feline pancreatic lipase composition and method of preparing and using such composition. Available online: https://patents.google.com/patent/WO2003093232A2/en?oq=WO+03%2f093232+A2 (accessed on 29 July 2019).
- Vennerstrom, J.L.; Nuzum, E.O.; Miller, R.E.; Dorn, A.; Gerena, L.; Dande, P.A.; Ellis, W.Y.; Ridley, R.G.; Milhous, W.K. 8-Aminoquinolines Active against Blood Stage Plasmodium falciparum in vitro Inhibit Hematin Polymerization. Antimicrob. Agents Chemother. 1999, 43, 598–602. [Google Scholar] [CrossRef]
- Novitt-Moreno, A.; Ransom, J.; Dow, G.; Smith, B.; Read, L.T.; Toovey, S. Tafenoquine for Malaria Prophylaxis in Adults: An Integrated Safety Analaysis. Travel Med. Infect. Dis. 2017, 17, 19–27. [Google Scholar] [CrossRef] [PubMed]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mayence, A.; Vanden Eynde, J.J. Tafenoquine: A 2018 Novel FDA-Approved Prodrug for the Radical Cure of Plasmodium vivax Malaria and Prophylaxis of Malaria. Pharmaceuticals 2019, 12, 115. https://doi.org/10.3390/ph12030115
Mayence A, Vanden Eynde JJ. Tafenoquine: A 2018 Novel FDA-Approved Prodrug for the Radical Cure of Plasmodium vivax Malaria and Prophylaxis of Malaria. Pharmaceuticals. 2019; 12(3):115. https://doi.org/10.3390/ph12030115
Chicago/Turabian StyleMayence, Annie, and Jean Jacques Vanden Eynde. 2019. "Tafenoquine: A 2018 Novel FDA-Approved Prodrug for the Radical Cure of Plasmodium vivax Malaria and Prophylaxis of Malaria" Pharmaceuticals 12, no. 3: 115. https://doi.org/10.3390/ph12030115
APA StyleMayence, A., & Vanden Eynde, J. J. (2019). Tafenoquine: A 2018 Novel FDA-Approved Prodrug for the Radical Cure of Plasmodium vivax Malaria and Prophylaxis of Malaria. Pharmaceuticals, 12(3), 115. https://doi.org/10.3390/ph12030115





