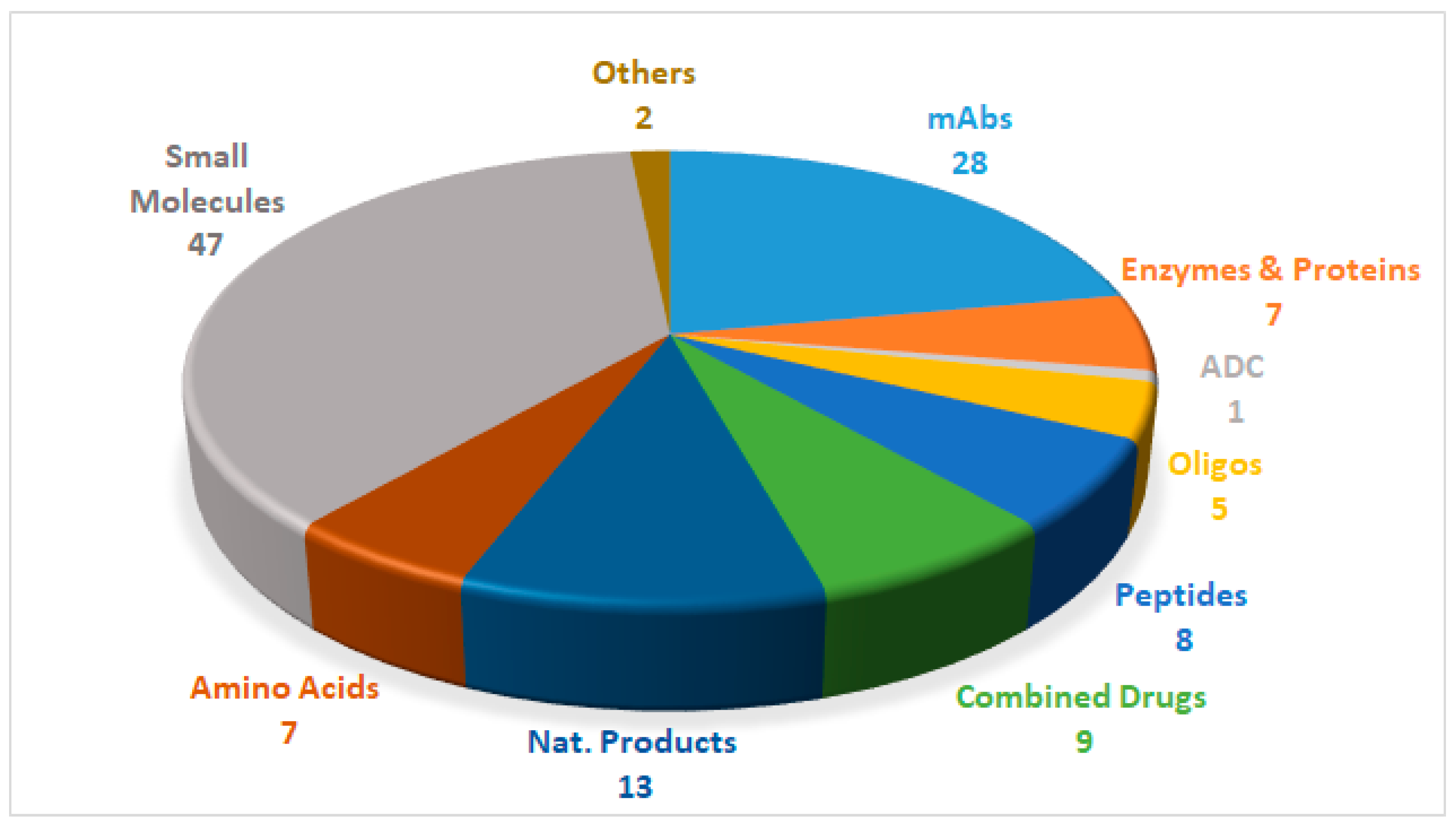
2018 FDA Tides Harvest
Abstract
1. Introduction
2. Oligonucleotides
2.1. Patisiran (OnpattroTM)
2.2. Inotersen (TegsediTM)
2.3. Comments
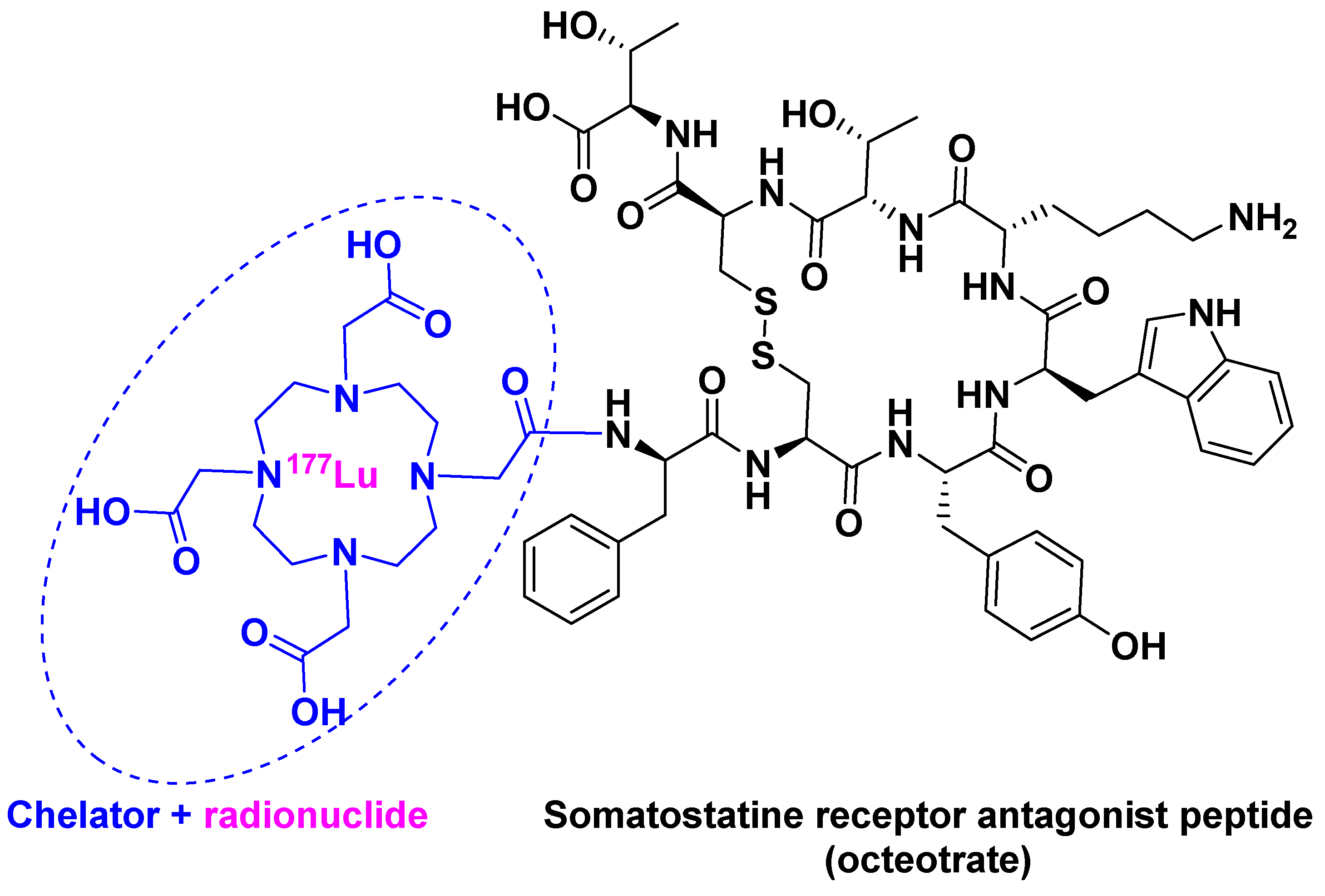
3. Lutathera®
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mullard, A. 2016 FDA drug approvals. Nat. Rev. Drug Discov. 2017, 16, 73–76. [Google Scholar] [CrossRef] [PubMed]
- Torre, B.G.; Albericio, F. The pharmaceutical industry in 2016. An analysis of FDA drug approvals from a perspective of the molecule type. Molecules 2017, 22. [Google Scholar] [CrossRef] [PubMed]
- Mullard, A. 2017 FDA drug approvals. Nat. Rev. Drug Discov. 2018, 17, 81–85. [Google Scholar] [CrossRef] [PubMed]
- De la Torre, B.G.; Albericio, F. The pharmaceutical industry in 2017. An analysis of FDA drug approvals from the perspective of molecules. Molecules 2018, 23. [Google Scholar] [CrossRef] [PubMed]
- Mullard, A. 2018 FDA drug approvals. Nat. Rev. Drug Discov. 2019, 18, 85–89. [Google Scholar] [CrossRef] [PubMed]
- De la Torre, B.G.; Albericio, F. The pharmaceutical industry in 2018. An analysis of FDA drug approvals from the perspective of molecules. Molecules 2019, 24. [Google Scholar] [CrossRef] [PubMed]
- Al Musaimi, O.; Al Shaer, D.; de la Torre, B.G.; Albericio, F. 2017 FDA peptide harvest. Pharmaceuticals 2018, 11. [Google Scholar] [CrossRef] [PubMed]
- Butler, J.S.; Chan, A.; Costelha, S.; Fishman, S.; Willoughby, J.L.; Borland, T.D.; Milstein, S.; Foster, D.J.; Goncalves, P.; Chen, Q.; et al. Preclinical evaluation of rnai as a treatment for transthyretin-mediated amyloidosis. Amyloid 2016, 23, 109–118. [Google Scholar] [CrossRef] [PubMed]
- Rowczenio, D.M.; Noor, I.; Gillmore, J.D.; Lachmann, H.J.; Whelan, C.; Hawkins, P.N.; Obici, L.; Westermark, P.; Grateau, G.; Wechalekar, A.D. Online registry for mutations in hereditary amyloidosis including nomenclature recommendations. Hum. Mutat. 2014, 35, E2403–E2412. [Google Scholar] [CrossRef] [PubMed]
- Chakradhar, S. A protein puzzle. Nat. Med. 2017, 23, 266–269. [Google Scholar] [CrossRef] [PubMed]
- Adams, D.; Suhr, O.B.; Dyck, P.J.; Litchy, W.J.; Leahy, R.G.; Chen, J.; Gollob, J.; Coelho, T. Trial design and rationale for apollo, a phase 3, placebo-controlled study of patisiran in patients with hereditary attr amyloidosis with polyneuropathy. BMC Neurol. 2017, 17. [Google Scholar] [CrossRef] [PubMed]
- Vieira Simoes, C.J.; Lourenco de Almeida, Z.C.; Vasconcelos Dias de Pinho EMelo, T.M.; Pontes Meireles Ferreira de Brito, R.M.; Silva Costa, D.C.; Cabral Cardoso Lopes, A.L. Bis-Furan Derivatives as Transthyretin (TTR) Stabilizers and Amyloid Inhibitors for the Treatment of Familial Amyloid Polyneuropathy (FAP). 2018. Available online: https://patents.google.com/patent/WO2016203402A1/ko (accessed on 22 March 2019).
- Hawkins, P.N.; Ando, Y.; Dispenzeri, A.; Gonzalez-Duarte, A.; Adams, D.; Suhr, O.B. Evolving landscape in the management of transthyretin amyloidosis. Ann. Med. 2015, 47, 625–638. [Google Scholar] [CrossRef] [PubMed]
- Yang, J. Patisiran for the treatment of hereditary transthyretin-mediated amyloidosis. Exp. Rev. Clin. Pharmacol. 2019, 12, 95–99. [Google Scholar] [CrossRef] [PubMed]
- Fire, A.; Xu, S.; Montgomery, M.K.; Kostas, S.A.; Driver, S.E.; Mello, C.C. Potent and specific genetic interference by double-stranded rna in caenorhabditis elegans. Nature 1998, 391, 806–811. [Google Scholar] [CrossRef] [PubMed]
- FDA Approval Letter of Patisiran 2018. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2018/210922Orig1s000ltr.pdf (accessed on 22 February 2019).
- FDA Label of Patisiran 2018. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/210922s000lbl.pdf (accessed on 22 February 2019).
- Jarvis, L.M. The new drugs of 2018. Chem. Eng. News 2019, 97, 33–37. [Google Scholar] [CrossRef]
- European Medicine Agency: Tegsedi Assessment Report 2018. Available online: https://www.ema.europa.eu/documents/assessment-report/tegsedi-epar-public-assessment-report_en.pdf (accessed on 22 February 2019).
- European Medicine Agency: Tegsedi Label. Available online: https://s3-us-west-2.amazonaws.com/drugbank/cite_this/attachments/files/000/001/938/original/tegsedi-epar-product-information_en.pdf?1539980614 (accessed on 22 February 2019).
- FDA Approval Letter of Tegsedi 2018. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/nda/2018/211172Orig1s000Approv.pdf (accessed on 22 February 2019).
- Fidler, B. FDA Oks Akcea Rare Disease Drug, Setting Up Market Clash with Alnylam 2018. Available online: https://xconomy.com/boston/2018/10/05/fda-oks-akcea-rare-disease-drug-setting-up-market-clash-with-alnylam/ (accessed on 22 February 2019).
- Kam, B.L.; Teunissen, J.J.; Krenning, E.P.; de Herder, W.W.; Khan, S.; van Vliet, E.I.; Kwekkeboom, D.J. Lutetium-labelled peptides for therapy of neuroendocrine tumours. Eur. J. Nucl. Med. Mol. Imaging 2012, 39, S103–S112. [Google Scholar] [CrossRef] [PubMed]
- Lutathera FDA Label 2018. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/208700s000lbl.pdf (accessed on 22 February 2019).
- Banerjee, S.; Pillai, M.R.; Knapp, F.F. Lutetium-177 therapeutic radiopharmaceuticals: Linking chemistry, radiochemistry, and practical applications. Chem. Rev. 2015, 115, 2934–2974. [Google Scholar] [CrossRef] [PubMed]
- FDA Approval Letter of Lutathera 2018. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2018/208700Orig1s000ltr.pdf (accessed on 22 February 2019).

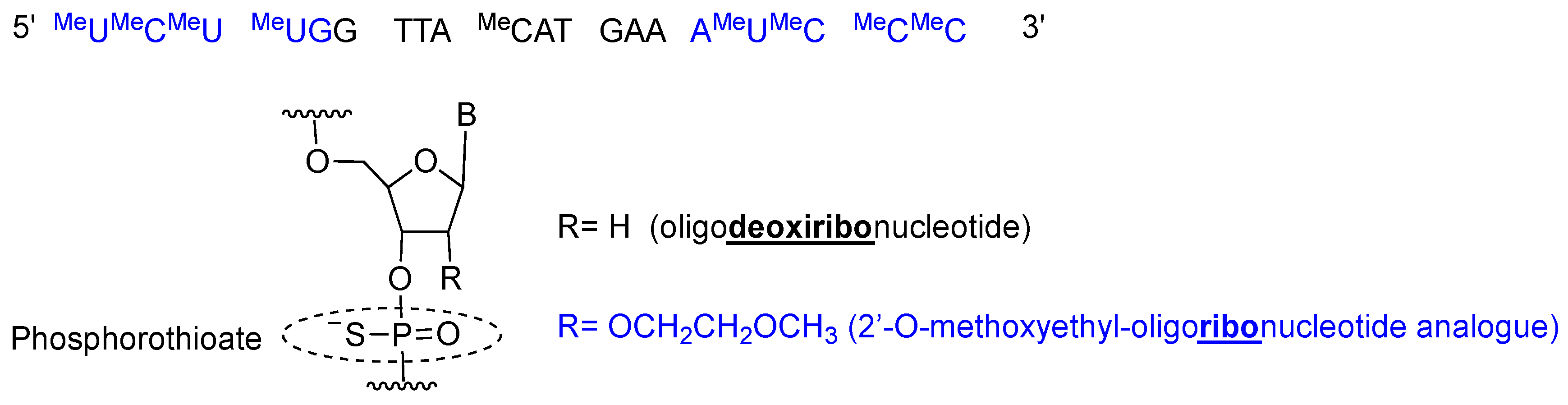
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al Shaer, D.; Al Musaimi, O.; Albericio, F.; de la Torre, B.G. 2018 FDA Tides Harvest. Pharmaceuticals 2019, 12, 52. https://doi.org/10.3390/ph12020052
Al Shaer D, Al Musaimi O, Albericio F, de la Torre BG. 2018 FDA Tides Harvest. Pharmaceuticals. 2019; 12(2):52. https://doi.org/10.3390/ph12020052
Chicago/Turabian StyleAl Shaer, Danah, Othman Al Musaimi, Fernando Albericio, and Beatriz G. de la Torre. 2019. "2018 FDA Tides Harvest" Pharmaceuticals 12, no. 2: 52. https://doi.org/10.3390/ph12020052
APA StyleAl Shaer, D., Al Musaimi, O., Albericio, F., & de la Torre, B. G. (2019). 2018 FDA Tides Harvest. Pharmaceuticals, 12(2), 52. https://doi.org/10.3390/ph12020052






