Protective Effect of Cashew Gum (Anacardium occidentale L.) on 5-Fluorouracil-Induced Intestinal Mucositis
Abstract
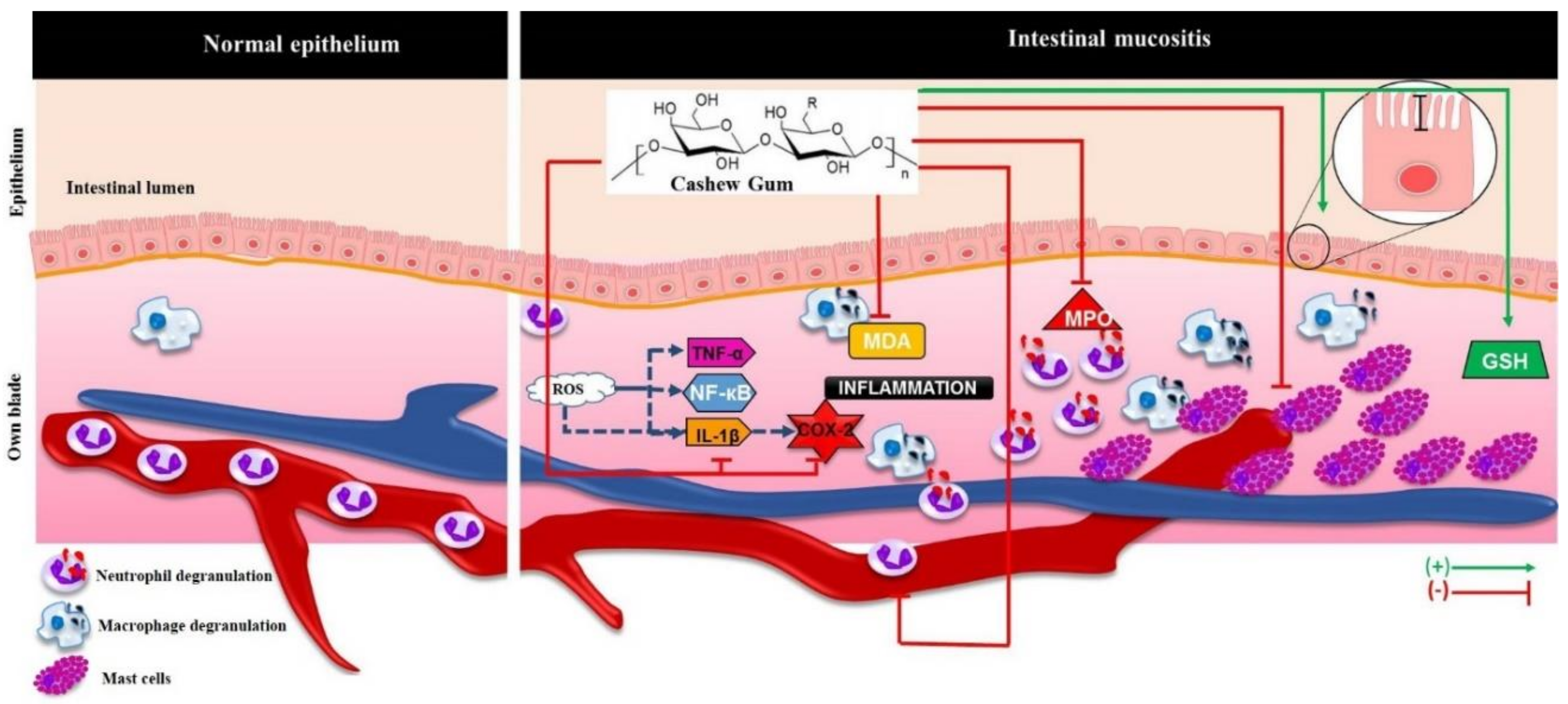
:1. Introduction
2. Results
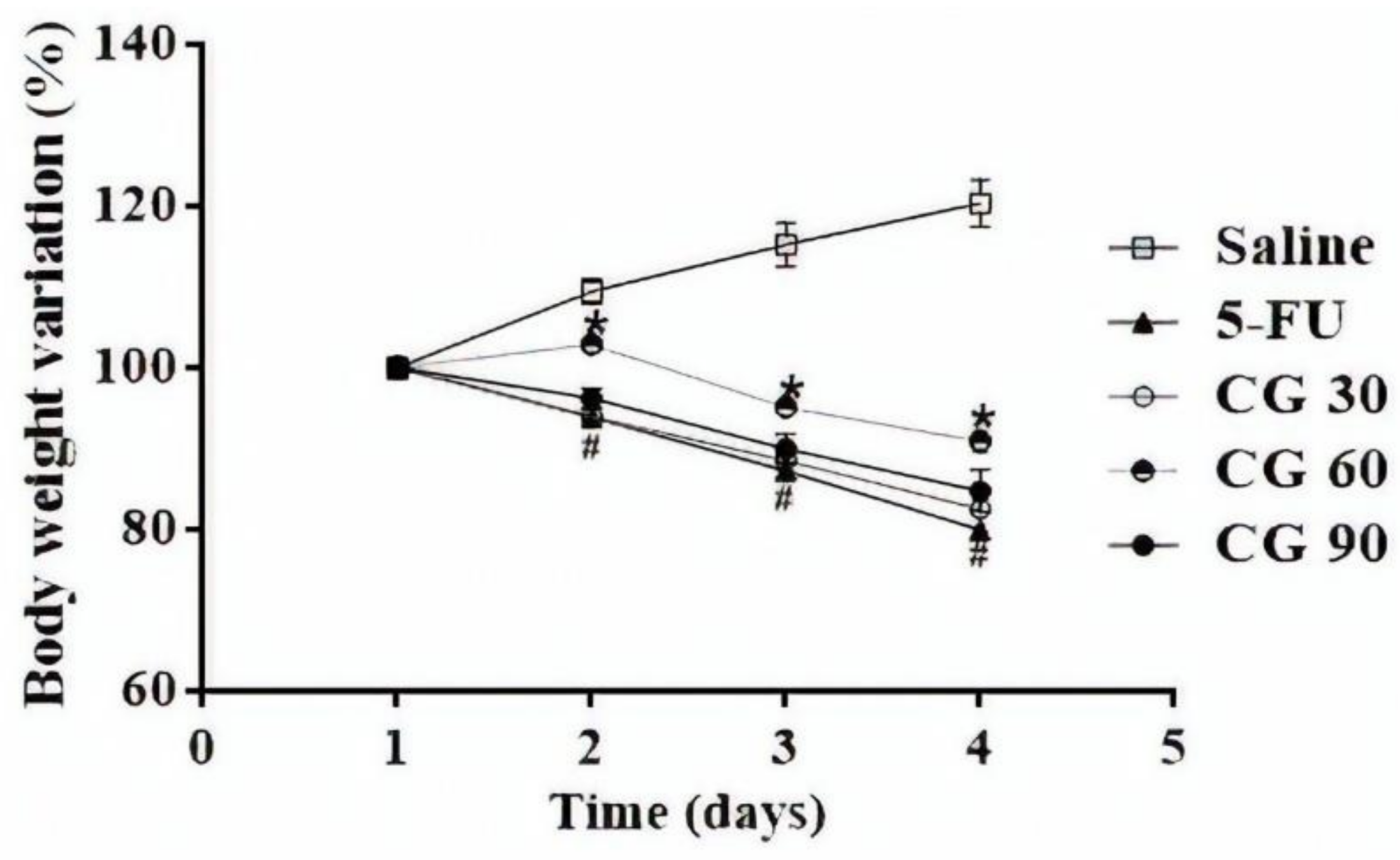
2.1. Weight Analysis
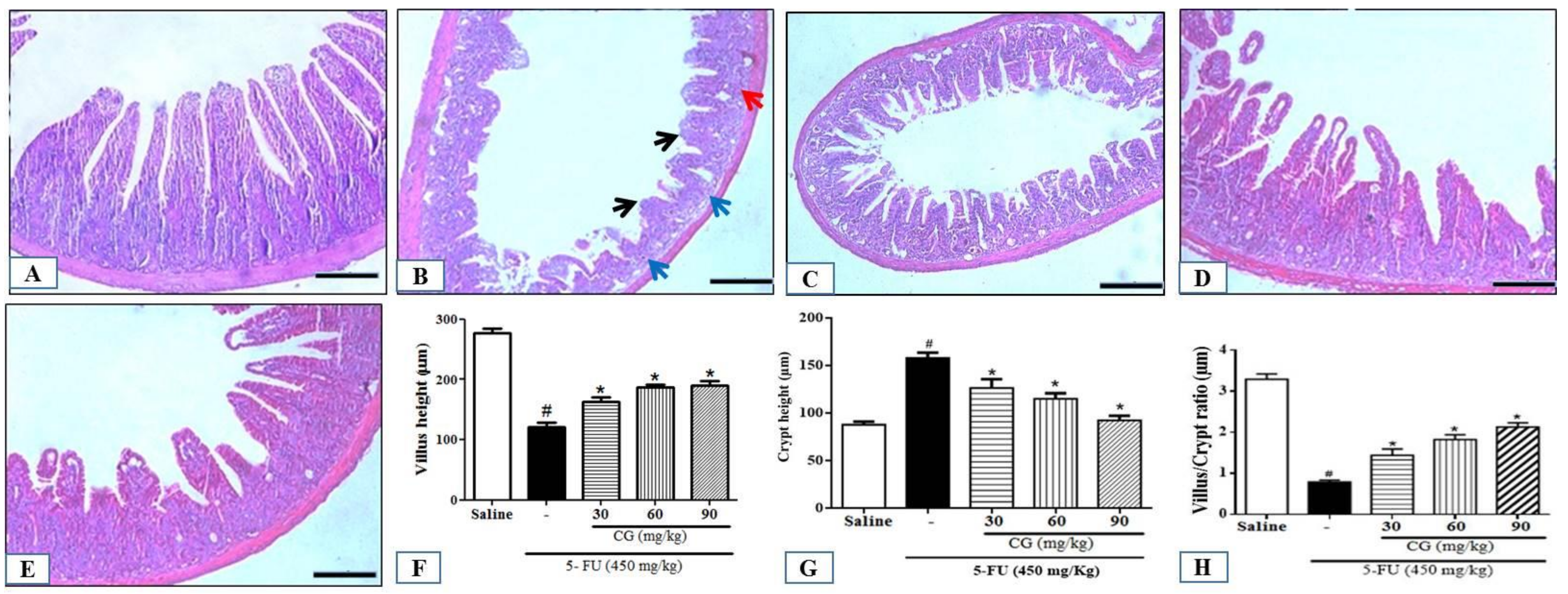
2.2. Histopathological and Morphometric Analysis
2.3. Leukocyte Count
2.4. Myeloperoxidase Assay (MPO)
2.5. Malondialdehyde (MDA) and Glutathione (GSH) Levels
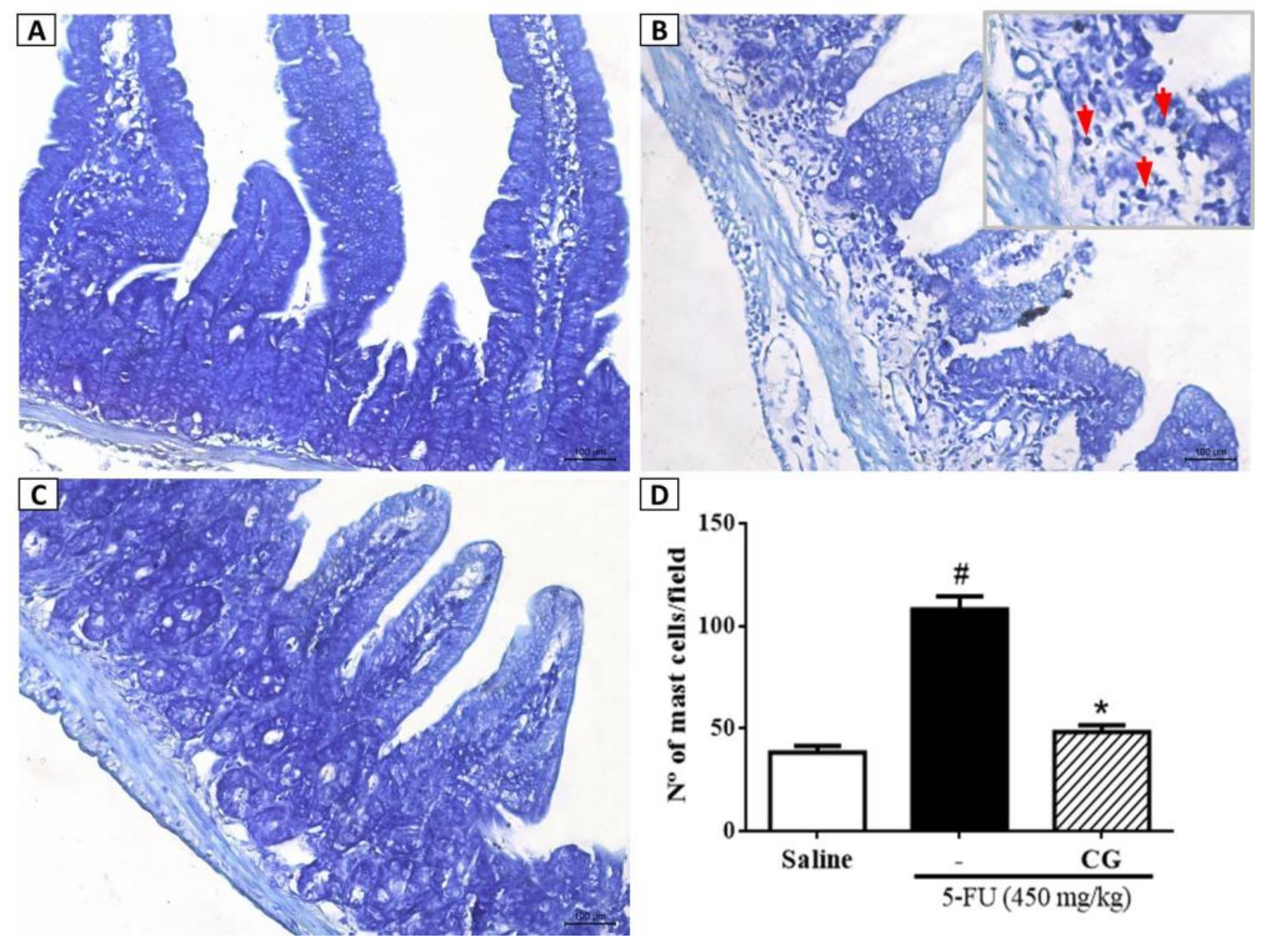
2.6. Mast Cell Concentration Analysis
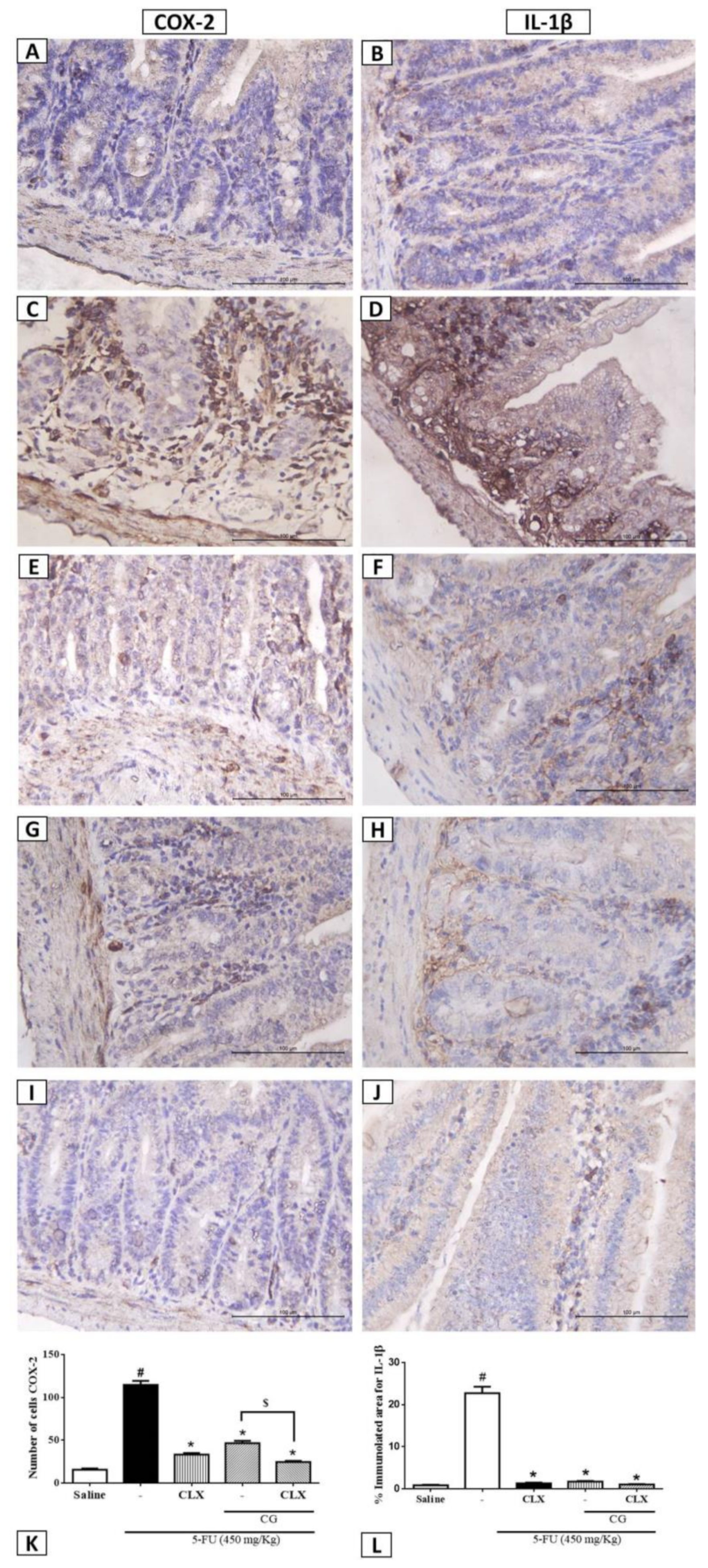
2.7. Effect of CG on Cyclooxygenase-2 Pathway in Histopathological and Morphometric Analyses
2.8. Immunohistochemistry for the Detection of COX-2 and IL-1β
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Drugs and Plant Materials
4.3. Extraction and Purification of Cashew Gum
4.4. Induction of Experimental Intestinal Mucositis
4.5. Histopathological and Morphometric Analysis
4.6. Leukocyte Count
4.7. Dosage of Malondialdehyde (MDA)
4.8. Concentration of Glutathione (GSH)
4.9. Myeloperoxidase Assay (MPO)
4.10. Mast Cell Analysis
4.11. Immunohistochemistry for the Detection of COX-2 and IL-1β
4.12. Statistical Analysis
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Kumar, V.; Abbas, A.K.; Aster, J.C. Robbins Basic Pathology E-Book, 9th ed.; Elsevier Health Sciences: Rio de Janeiro, Brazil, 2013. [Google Scholar]
- Ferreira, J.E.V.; de Figueiredo, A.F.; Barbosa, J.P.; Pinheiro, J.C. Chemometric study on molecules with anticancer properties. In Chemometrics in Practical Applications; IntechOpen: London, UK, 2012; pp. 185–186. [Google Scholar]
- Organization, W.H. Cancer. Available online: http://www.who.int/cancer/en/ (accessed on 17 August 2018).
- Chang, C.-T.; Ho, T.-Y.; Lin, H.; Liang, J.-A.; Huang, H.-C.; Li, C.-C.; Lo, H.-Y.; Wu, S.-L.; Huang, Y.-F.; Hsiang, C.-Y. 5-Fluorouracil induced intestinal mucositis via nuclear factor-κB activation by transcriptomic analysis and in vivo bioluminescence imaging. PLoS ONE 2012, 7, e31808. [Google Scholar] [CrossRef]
- Udofot, O.; Affram, K.; Bridg’ette Israel, E.A. Cytotoxicity of 5-fluorouracil-loaded pH-sensitive liposomal nanoparticles in colorectal cancer cell lines. Integr. Cancer Sci. Ther. 2015, 2, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Wilhelm, M.; Mueller, L.; Miller, M.C.; Link, K.; Holdenrieder, S.; Bertsch, T.; Kunzmann, V.; Stoetzer, O.J.; Suttmann, I.; Braess, J. Prospective, multicenter study of 5-fluorouracil therapeutic drug monitoring in metastatic colorectal cancer treated in routine clinical practice. Clin. Colorectal Cancer 2016, 15, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Kawashima, R.; Fujimaki, M.; Ikenoue, Y.; Danjo, K.; Koizumi, W.; Ichikawa, T. Influence of an elemental diet on 5-fluorouracil-induced morphological changes in the mouse salivary gland and colon. Support. Care Cancer 2016, 24, 1609–1616. [Google Scholar] [CrossRef] [PubMed]
- Kawashima, R.; Kawakami, F.; Maekawa, T.; Yamamoto, H.; Koizumi, W.; Ichikawa, T. Elemental diet moderates 5-fluorouracil-induced gastrointestinal mucositis through mucus barrier alteration. Cancer Chemother. Pharmacol. 2015, 76, 269–277. [Google Scholar] [CrossRef]
- Kobuchi, S.; Ito, Y.; Sakaeda, T. Population Pharmacokinetic–Pharmacodynamic Modeling of 5-Fluorouracil for Toxicities in Rats. Eur. J. Drug Metab. Pharmacokinet. 2017, 42, 707–718. [Google Scholar] [CrossRef]
- Peterson, D.; Bensadoun, R.-J.; Roila, F.; Group, E.G.W. Management of oral and gastrointestinal mucositis: ESMO Clinical Practice Guidelines. Ann. Oncol. 2011, 22, 78–84. [Google Scholar] [CrossRef]
- Kim, H.J.; Kim, J.H.; Moon, W.; Park, J.; Park, S.J.; Am Song, G.; Han, S.H.; Lee, J.H. Rebamipide attenuates 5-fluorouracil-induced small intestinal mucositis in a mouse model. Biol. Pharm. Bull. 2015, 38, 179–183. [Google Scholar] [CrossRef]
- Ofori-Kwakye, K.; Amekyeh, H.; El-Duah, M.; Kipo, S.L. Mechanical and tablet coating properties of cashew tree (Anacardium occidentale l) gum-based films. Asian J. Pharm. Clin. Res. 2012, 5, 62–68. [Google Scholar]
- Olusola, A.; Toluwalope, G.; Olutayo, O. Carboxymethylation of Anacardium occidentale L. exudate gum: Synthesis and characterization. Sch. Acad. J. Pharm. 2014, 3, 213–216. [Google Scholar]
- Schirato, G.V.; Monteiro, F.M.F.; Silva, F.d.O.; Luís, d.L.F.J.; Leão, A.M.d.A.C.; Porto, A.L.F. O polissacarídeo do Anacardium occidentale L. na fase inflamatória do processo cicatricial de lesões cutâneas. Ciência Rural 2006, 36, 149–154. [Google Scholar] [CrossRef]
- Kumar, A.; Moin, A.; Ahmed, A.; Shivakumar, H.G. Cashew gum a versatile hydrophyllic polymer: A review. Curr. Drug Ther. 2012, 7, 2–12. [Google Scholar] [CrossRef]
- Quelemes, P.V.; Araruna, F.B.; de Faria, B.E.; Kuckelhaus, S.A.; da Silva, D.A.; Mendonça, R.Z.; Eiras, C.; dos S Soares, M.J.; Leite, J.R.S. Development and antibacterial activity of cashew gum-based silver nanoparticles. Int. J. Mol. Sci. 2013, 14, 4969–4981. [Google Scholar] [CrossRef]
- Araújo, T.S.; Costa, D.S.; Sousa, N.A.; Souza, L.K.; de Araújo, S.; Oliveira, A.P.; Sousa, F.B.M.; Silva, D.A.; Barbosa, A.L.; Leite, J.R.S. Antidiarrheal activity of cashew GUM, a complex heteropolysaccharide extracted from exudate of Anacardium occidentale L. in rodents. J. Ethnopharmacol. 2015, 174, 299–307. [Google Scholar] [CrossRef] [PubMed]
- AlRashdi, A.S.; Salama, S.M.; Alkiyumi, S.S.; Abdulla, M.A.; Hadi, A.H.A.; Abdelwahab, S.I.; Taha, M.M.; Hussiani, J.; Asykin, N. Mechanisms of gastroprotective effects of ethanolic leaf extract of Jasminum sambac against HCl/ethanol-induced gastric mucosal injury in rats. Evid.Based Complement. Altern. Med. 2012, 2012, 786426. [Google Scholar] [CrossRef]
- Bonacorsi, C.; Da Fonseca, L.M.; Raddi, M.S.G.; Kitagawa, R.R.; Vilegas, W. Comparison of Brazilian plants used to treat gastritis on the oxidative burst of Helicobacter pylori-stimulated neutrophil. Evid. Based Complement. Altern. Med. 2013, 2013, 851621. [Google Scholar] [CrossRef] [PubMed]
- Reddy, S.C.; Shetty, B.V.; Rao, G.M. Oral Ingestion of Spondias pinnata Bark Extract Trim Down Severity of Small Intestinal Mucositis in Etoposide Treated Rats. Cancer Sci. Ther. 2015, 7, 030–033. [Google Scholar] [CrossRef]
- Chikezie, P.C. Sodium metabisulfite–induced polymerization of sickle cell hemoglobin incubated in the extracts of three medicinal plants (Anacardium occidentale, Psidium guajava, and Terminalia catappa). Pharmacogn. Mag. 2011, 7, 126–132. [Google Scholar] [CrossRef]
- Okpashi, V.E.; Bayim, B.P.-R.; Obi-Abang, M. Comparative effects of some medicinal plants: Anacardium occidentale, Eucalyptus globulus, Psidium guajava, and Xylopia aethiopica extracts in alloxan-induced diabetic male Wistar albino rats. Biochem. Res. Int. 2014, 2014. [Google Scholar] [CrossRef]
- Jaiswal, Y.; Tatke, P.; Gabhe, S.; Vaidya, A. Antidiabetic activity of extracts of Anacardium occidentale Linn. leaves on n-streptozotocin diabetic rats. J. Tradit. Complement. Med. 2017, 7, 421–427. [Google Scholar] [CrossRef]
- Souza, N.C.; de Oliveira, J.M.; Morrone, M.d.S.; Albanus, R.D.O.; Amarante, M.d.S.M.; Camillo, C.d.S.; Langassner, S.M.Z.; Gelain, D.P.; Moreira, J.C.F.; Dalmolin, R.J.S. Antioxidant and anti-inflammatory properties of Anacardium occidentale leaf extract. Evid. Based Complement. Altern. Med. 2017, 2017, 2787308. [Google Scholar] [CrossRef] [PubMed]
- Silva, J.G.d.; Souza, I.A.; Higino, J.S.; Siqueira-Junior, J.P.; Pereira, J.V.; Pereira, M.d.S.V. Atividade antimicrobiana do extrato de Anacardium occidentale Linn. em amostras multiresistentes de Staphylococcus aureus. Rev. Bras. Farmacogn. 2007, 17, 572–577. [Google Scholar] [CrossRef]
- Tchikaya, F.O.; Bantsielé, G.B.; Kouakou-Siransy, G.; Datté, J.Y.; Datté, P.; Zirihi, N.G.; Offoumou, M.A. Anacardium occidentale Linn.(Anacardiaceae) stem bark extract induces hypotensive and cardio-inhibitory effects in experimental animal models. Afr. J. Tradit. Complement. Altern. Med. 2011, 8, 452–461. [Google Scholar] [CrossRef]
- Cardoso Palheta, I.; Caldeira Tavares-Martins, A.C.; Araujo Lucas, F.C.; Goncalves Jardim, M.A. Ethnobotanical study of medicinal plants in urban home gardens in the city of Abaetetuba, Pará state, Brazil. Boletín Latinoamericano y del Caribe de Plantas Medicinales y Aromáticas 2017, 16, 206–262. [Google Scholar]
- Vasconcelos, M.d.S.; Rochette, N.F.G.; de Oliveira, M.L.M.; Nunes-Pinheiro, D.C.S.; Tomé, A.R.; Maia de Sousa, F.Y.; Pinheiro, F.G.M.; Moura, C.F.H.; Miranda, M.R.A.; Mota, E.F. Anti-inflammatory and wound healing potential of cashew apple juice (Anacardium occidentale L.) in mice. Exp. Biol. Med. 2015, 240, 1648–1655. [Google Scholar] [CrossRef]
- Yeung, C.-Y.; Chan, W.-T.; Jiang, C.-B.; Cheng, M.-L.; Liu, C.-Y.; Chang, S.-W.; Chiau, J.-S.C.; Lee, H.-C. Amelioration of chemotherapy-induced intestinal mucositis by orally administered probiotics in a mouse model. PLoS ONE 2015, 10, e0138746. [Google Scholar] [CrossRef]
- Carvalho, R.D.; Breyner, N.; Menezes-Garcia, Z.; Rodrigues, N.M.; Lemos, L.; Maioli, T.U.; Souza, D.G.; Carmona, D.; Faria, A.M.; Langella, P. Secretion of biologically active pancreatitis-associated protein I (PAP) by genetically modified dairy Lactococcus lactis NZ9000 in the prevention of intestinal mucositis. Microb. Cell Factories 2017, 16, 27. [Google Scholar] [CrossRef]
- Li, Y.; Liu, M.; Zuo, Z.; Liu, J.; Yu, X.; Guan, Y.; Zhan, R.; Han, Q.; Zhang, J.; Zhou, R. TLR9 Regulates the NF-κB–NLRP3–IL-1β Pathway Negatively in Salmonella-Induced NKG2D-Mediated Intestinal Inflammation. J. Immunol. 2017, 199, 761–773. [Google Scholar] [CrossRef]
- Gerhard, D.; Sousa, F.J.D.S.S.; Andraus, R.A.C.; Pardo, P.E.; Nai, G.A.; Neto, H.B.; Messora, M.R.; Maia, L.P. Probiotic therapy reduces inflammation and improves intestinal morphology in rats with induced oral mucositis. Braz. Oral Res. 2017, 31, 1–11. [Google Scholar] [CrossRef]
- De Araújo, A.A.; Borba, P.B.; de Souza, F.H.D.; Nogueira, A.C.; Saldanha, T.S.; Araújo, T.E.F.; Silva, A.I.; de Araújo Júnior, R.F. In a methotrexate-induced model of intestinal mucositis, olmesartan reduced inflammation and induced enteropathy characterized by severe diarrhea, weight loss, and reduced sucrose activity. Biol. Pharm. Bull. 2015, 38, 746–752. [Google Scholar] [CrossRef]
- De Barros, P.A.V.; Andrade, M.E.R.; de Vasconcelos Generoso, S.; Miranda, S.E.M.; dos Reis, D.C.; Leocádio, P.C.L.; e Souza, É.L.d.S.; dos Santos Martins, F.; da Gama, M.A.S.; Cassali, G.D. Conjugated linoleic acid prevents damage caused by intestinal mucositis induced by 5-fluorouracil in an experimental model. Biomed. Pharmacother. 2018, 103, 1567–1576. [Google Scholar] [CrossRef] [PubMed]
- Al-Asmari, A.K.; Khan, A.Q.; Al-Asmari, S.A.; Al-Rawi, A.; Al-Omani, S. Alleviation of 5-fluorouracil-induced intestinal mucositis in rats by vitamin E via targeting oxidative stress and inflammatory markers. J. Complement. Integr. Med. 2016, 13, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Wu, Z.; Di, J.; Pan, Y.; Zhang, H.; Du, Y.; Cheng, Z.; Jin, Z.; Wang, Z.; Zheng, Q. CXCL9 attenuated chemotherapy-induced intestinal mucositis by inhibiting proliferation and reducing apoptosis. Biomed. Pharmacother. 2011, 65, 547–554. [Google Scholar] [CrossRef]
- Zhang, S.; Liu, Y.; Xiang, D.; Yang, J.; Liu, D.; Ren, X.; Zhang, C. Assessment of dose-response relationship of 5-fluorouracil to murine intestinal injury. Biomed. Pharmacother. 2018, 106, 910–916. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Aninat, C.; Michaux, G.; Morel, F. Anticancer drug 5-fluorouracil induces reproductive and developmental defects in Caenorhabditis elegans. Reprod. Toxicol. 2010, 29, 415–420. [Google Scholar] [CrossRef] [PubMed]
- Soares, P.M.; Mota, J.M.S.; Gomes, A.S.; Oliveira, R.B.; Assreuy, A.M.S.; Brito, G.A.C.; Santos, A.A.; Ribeiro, R.A.; Souza, M.H. Gastrointestinal dysmotility in 5-fluorouracil-induced intestinal mucositis outlasts inflammatory process resolution. Cancer Chemother. Pharmacol. 2008, 63, 91–98. [Google Scholar] [CrossRef]
- Quaresma, M.P. Lactobacillus spp. e Bifidobacterium sp. Attenuate Experimental Intestinal Mucositis Induced by 5-Fluorouracil in Mice; Federal University of Ceará: Fortaleza, Brazil, 2016. [Google Scholar]
- Sonis, S.T. A biological approach to mucositis. J. Support. Oncol. 2004, 2, 21–32. [Google Scholar]
- Guabiraba, R.; Besnard, A.; Menezes, G.; Secher, T.; Jabir, M.; Amaral, S.; Braun, H.; Lima-Junior, R.C.; Ribeiro, R.; Cunha, F. IL-33 targeting attenuates intestinal mucositis and enhances effective tumor chemotherapy in mice. Mucosal Immunol. 2014, 7, 1079–1093. [Google Scholar] [CrossRef]
- Bastos, C.C.C.; de Ávila, P.H.M.; dos Santos Filho, E.X.; de Ávila, R.I.; Batista, A.C.; Fonseca, S.G.; Lima, E.M.; Marreto, R.N.; de Mendonca, E.F.; Valadares, M.C. Use of Bidens pilosa L.(Asteraceae) and Curcuma longa L.(Zingiberaceae) to treat intestinal mucositis in mice: Toxico-pharmacological evaluations. Toxicol. Rep. 2016, 3, 279–287. [Google Scholar] [CrossRef] [PubMed]
- Justino, P.F.; Melo, L.F.; Nogueira, A.F.; Costa, J.V.; Silva, L.M.; Santos, C.M.; Mendes, W.O.; Costa, M.R.; Franco, A.X.; Lima, A.A.; et al. Treatment with Saccharomyces boulardii reduces the inflammation and dysfunction of the gastrointestinal tract in 5-fluorouracil-induced intestinal mucositis in mice. Br. J. Nutr. 2014, 111, 1611–1621. [Google Scholar] [CrossRef] [PubMed]
- De Ávila, P.H.M.; de Ávila, R.I.; dos Santos Filho, E.X.; Bastos, C.C.C.; Batista, A.C.; Mendonca, E.F.; Serpa, R.C.; Marreto, R.N.; da Cruz, A.F.; Lima, E.M. Mucoadhesive formulation of Bidens pilosa L.(Asteraceae) reduces intestinal injury from 5-fluorouracil-induced mucositis in mice. Toxicol. Rep. 2015, 2, 563–573. [Google Scholar] [CrossRef]
- Al-Asmari, A.; Al-Zahrani, A.; Khan, A.; Al-Shahrani, H.; Ali Al Amri, M. Taurine ameliorates 5-flourouracil-induced intestinal mucositis, hepatorenal and reproductive organ damage in Wistar rats: A biochemical and histological study. Hum. Exp. Toxicol. 2016, 35, 10–20. [Google Scholar] [CrossRef] [PubMed]
- Heber, D. Oxidative Stress Markers and Inflammation: The Role of Spices and Herbs. Nutr. Today 2014, 49, S4–S5. [Google Scholar] [CrossRef]
- Nogueira, L.T.; Costa, D.V.; Gomes, A.S.; Martins, C.S.; Silva, A.M.; Coelho-Aguiar, J.M.; Castelucci, P.; Lima-Júnior, R.C.; Leitão, R.F.; Moura-Neto, V.; et al. The involvement of mast cells in the irinotecan-induced enteric neurons loss and reactive gliosis. J. Neuroinflamm. 2017, 14, 79. [Google Scholar] [CrossRef]
- Hamilton, M.J.; Frei, S.M.; Stevens, R.L. The multifaceted mast cell in inflammatory bowel disease. Inflamm. Bowel Dis. 2014, 20, 2364–2378. [Google Scholar] [CrossRef] [PubMed]
- De Winter, B.Y.; van den Wijngaard, R.M.; de Jonge, W.J. Intestinal mast cells in gut inflammation and motility disturbances. Biochim. Biophys. Acta Mol. Basis Dis. 2012, 1822, 66–73. [Google Scholar] [CrossRef]
- Theoharides, T.C.; Alysandratos, K.-D.; Angelidou, A.; Delivanis, D.-A.; Sismanopoulos, N.; Zhang, B.; Asadi, S.; Vasiadi, M.; Weng, Z.; Miniati, A.; et al. Mast cells and inflammation. Biochim. Biophys. Acta Mol. Basis Dis. 2012, 1822, 21–33. [Google Scholar] [CrossRef]
- Ramsay, D.B.; Stephen, S.; Borum, M.; Voltaggio, L.; Doman, D.B. Mast cells in gastrointestinal disease. Gastroenterol. Hepatol. 2010, 6, 772–777. [Google Scholar]
- Bischoff, S.C. Mast cells in gastrointestinal disorders. Eur. J. Pharmacol. 2016, 778, 139–145. [Google Scholar] [CrossRef]
- Short, S.S.; Wang, J.; Castle, S.L.; Fernandez, G.E.; Smiley, N.; Zobel, M.; Pontarelli, E.M.; Papillon, S.C.; Grishin, A.V.; Ford, H.R. Low doses of celecoxib attenuate gut barrier failure during experimental peritonitis. Lab. Investig. 2013, 93, 1265–1275. [Google Scholar] [CrossRef] [PubMed]
- Javle, M.M.; Cao, S.; Durrani, F.A.; Pendyala, L.; Lawrence, D.D.; Smith, P.F.; Creaven, P.J.; Noel, D.C.; Iyer, R.V.; Rustum, Y.M. Celecoxib and mucosal protection: Translation from an animal model to a phase I clinical trial of celecoxib, irinotecan, and 5-fluorouracil. Clin. Cancer Res. 2007, 13, 965–971. [Google Scholar] [CrossRef] [PubMed]
- Verri Jr, W.A.; Cunha, T.M.; Parada, C.A.; Poole, S.; Cunha, F.Q.; Ferreira, S.H. Hypernociceptive role of cytokines and chemokines: Targets for analgesic drug development? Pharmacol. Ther. 2006, 112, 116–138. [Google Scholar] [CrossRef] [PubMed]
- Al-Azri, A.R.; Gibson, R.J.; Bowen, J.M.; Stringer, A.M.; Keefe, D.M.; Logan, R.M. Involvement of matrix metalloproteinases (MMP-3 and MMP-9) in the pathogenesis of irinotecan-induced oral mucositis. J. Oral Pathol. Med. 2015, 44, 459–467. [Google Scholar] [CrossRef]
- Ribeiro, R.A.; Wanderley, C.W.; Wong, D.V.; Mota, J.M.S.; Leite, C.A.; Souza, M.H.; Cunha, F.Q.; Lima-Junior, R.C. Irinotecan-and 5-fluorouracil-induced intestinal mucositis: Insights into pathogenesis and therapeutic perspectives. Cancer Chemother. Pharmacol. 2016, 78, 881–893. [Google Scholar] [CrossRef]
- Al-Dasooqi, N.; Sonis, S.T.; Bowen, J.M.; Bateman, E.; Blijlevens, N.; Gibson, R.J.; Logan, R.M.; Nair, R.G.; Stringer, A.M.; Yazbeck, R.; et al. Emerging evidence on the pathobiology of mucositis. Support. Care Cancer 2013, 21, 2075–2083. [Google Scholar] [CrossRef]
- De Paula, R.; Santana, S.; Rodrigues, J. Composition and rheological properties of Albizia lebbeck gum exudate. Carbohydr. Polym. 2001, 44, 133–139. [Google Scholar] [CrossRef]
- Silva, D.A.; Feitosa, J.P.; Maciel, J.S.; Paula, H.C.; de Paula, R.C. Characterization of crosslinked cashew gum derivatives. Carbohydr. Polym. 2006, 66, 16–26. [Google Scholar] [CrossRef]
- Da Silva, D.A.; Feitosa, J.P.; Paula, H.C.; de Paula, R.C. Synthesis and characterization of cashew gum/acrylic acid nanoparticles. Mater. Sci. Eng. C 2009, 29, 437–441. [Google Scholar] [CrossRef]
- Soares, P.M.; Mota, J.M.S.; Souza, E.P.; Justino, P.F.; Franco, A.X.; Cunha, F.Q.; Ribeiro, R.A.; Souza, M.H. Inflammatory intestinal damage induced by 5-fluorouracil requires IL-4. Cytokine 2013, 61, 46–49. [Google Scholar] [CrossRef]
- Dos Santos Filho, E.X.; Ávila, P.H.M.; Bastos, C.C.C.; Batista, A.C.; Naves, L.N.; Marreto, R.N.; Lima, E.M.; Mendonca, E.F.; Valadares, M.C. Curcuminoids from Curcuma longaL. reduced intestinal mucositis induced by 5-fluorouracil in mice: Bioadhesive, proliferative, anti-inflammatory and antioxidant effects. Toxicol. Rep. 2016, 3, 55–62. [Google Scholar] [CrossRef]
- MacPherson, B.; Pfeiffer, C. Experimental production of diffuse colitis in rats. Digestion 1978, 17, 135–150. [Google Scholar] [CrossRef] [PubMed]
- Moura, R.; Wada, C.; Purchio, A.; Almeida, T. Studies of the Figurative Elements of Blood, 3rd ed.; Atheneu: São Paulo, Brazil, 1998. [Google Scholar]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Ohkawa, H.; Ohishi, N.; Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 1979, 95, 351–358. [Google Scholar] [CrossRef]
- Sedlak, J.; Lindsay, R.H. Estimation of total, protein-bound, and nonprotein sulfhydryl groups in tissue with Ellman’s reagent. Anal. Biochem. 1968, 25, 192–205. [Google Scholar] [CrossRef]
- Bradley, P.P.; Priebat, D.A.; Christensen, R.D.; Rothstein, G. Measurement of cutaneous inflammation: Estimation of neutrophil content with an enzyme marker. J. Investig. Dermatol. 1982, 78, 206–209. [Google Scholar] [CrossRef] [PubMed]
- Michalany, J. Histological Technique Pathological Anatomy: With Instructions for the Surgeon, Nurse, Cytotechnician, 3rd ed.; Michalany: São Paulo, Brazil, 2008. [Google Scholar]



| Groups | Scores |
|---|---|
| Saline | 0 (0-0) |
| 5-FU | 2 (1-3) # |
| CG 30 | 3 (3-3) |
| CG 60 | 1 (1-2) |
| CG 90 | 1 (0-1) * |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miranda, J.A.L.d.; Barreto, J.E.F.; Martins, D.S.; Pimentel, P.V.d.S.; Costa, D.V.d.S.; Silva, R.R.e.; Souza, L.K.M.d.; Lima, C.N.d.C.; Rocha, J.A.; Freitas, A.P.F.d.; et al. Protective Effect of Cashew Gum (Anacardium occidentale L.) on 5-Fluorouracil-Induced Intestinal Mucositis. Pharmaceuticals 2019, 12, 51. https://doi.org/10.3390/ph12020051
Miranda JALd, Barreto JEF, Martins DS, Pimentel PVdS, Costa DVdS, Silva RRe, Souza LKMd, Lima CNdC, Rocha JA, Freitas APFd, et al. Protective Effect of Cashew Gum (Anacardium occidentale L.) on 5-Fluorouracil-Induced Intestinal Mucositis. Pharmaceuticals. 2019; 12(2):51. https://doi.org/10.3390/ph12020051
Chicago/Turabian StyleMiranda, João Antônio Leal de, João Erivan Façanha Barreto, Dainesy Santos Martins, Paulo Vitor de Souza Pimentel, Deiziane Viana da Silva Costa, Reyca Rodrigues e Silva, Luan Kelves Miranda de Souza, Camila Nayane de Carvalho Lima, Jefferson Almeida Rocha, Ana Paula Fragoso de Freitas, and et al. 2019. "Protective Effect of Cashew Gum (Anacardium occidentale L.) on 5-Fluorouracil-Induced Intestinal Mucositis" Pharmaceuticals 12, no. 2: 51. https://doi.org/10.3390/ph12020051
APA StyleMiranda, J. A. L. d., Barreto, J. E. F., Martins, D. S., Pimentel, P. V. d. S., Costa, D. V. d. S., Silva, R. R. e., Souza, L. K. M. d., Lima, C. N. d. C., Rocha, J. A., Freitas, A. P. F. d., Silva, D. A. d., Scafuri, A. G., Leitão, R. F. d. C., Brito, G. A. d. C., Medeiros, J. V. R., & Cerqueira, G. S. (2019). Protective Effect of Cashew Gum (Anacardium occidentale L.) on 5-Fluorouracil-Induced Intestinal Mucositis. Pharmaceuticals, 12(2), 51. https://doi.org/10.3390/ph12020051





