Paving the Way for Motor Imagery-Based Tele-Rehabilitation through a Fully Wearable BCI System †
Abstract
1. Introduction
2. Materials and Methods
2.1. System Overview
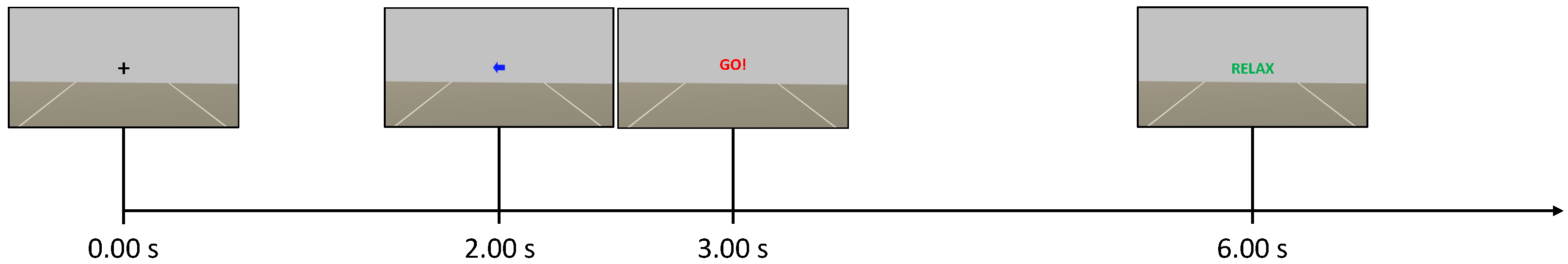
2.2. Experimental Protocol
2.2.1. Control Group
2.2.2. Neurofeedback Group
2.3. EEG Processing
- Time domain filtering by means of a filter bank (FB) with 17 overlapped bandpass Type II Chebyshev filters with order 10 from 4 to 40 ;
- Features extraction using a spatial domain filtering by means of the common spatial patterns (CSPs) algorithm [35];
- Feature selection based on the class-related information content of the features using the mutual-information-based best individual features selector;
- Feature classification exploiting the Bayesian (NBPW) classifier.
2.4. Outcome Measures
- MIQ-3 [41]: this is the most recent version of the movement imagery questionnaire [42] and of the revised movement imagery questionnaire [43]. It is a 12-item questionnaire to assess an individual’s ability to imagine 4 movements using internal visual imagery, external visual imagery, and kinaesthetic imagery. The rating scales range from 1 (very difficult to see/feel) to 7 (very easy to see/feel). The MIQ-3 has good psychometric properties, internal reliability, and predictive validity.
- SUS (system usability scale) [44]: this is one of the most robust and tested psychometric tools for user-perceived usability. The SUS score consists of a value between 0 and 100, with high values indicating better usability. According to Bargor et al. [45], it is possible to adopt a 7-point adjectival scale (from “worst imaginable” to “best imaginable”) for the SUS score. Another variation, proposed in [46], is to consider the score in terms of “acceptable” (value above 70) or ”not acceptable” (value below 50). The range from 50 to 70 is instead “marginally acceptable”.
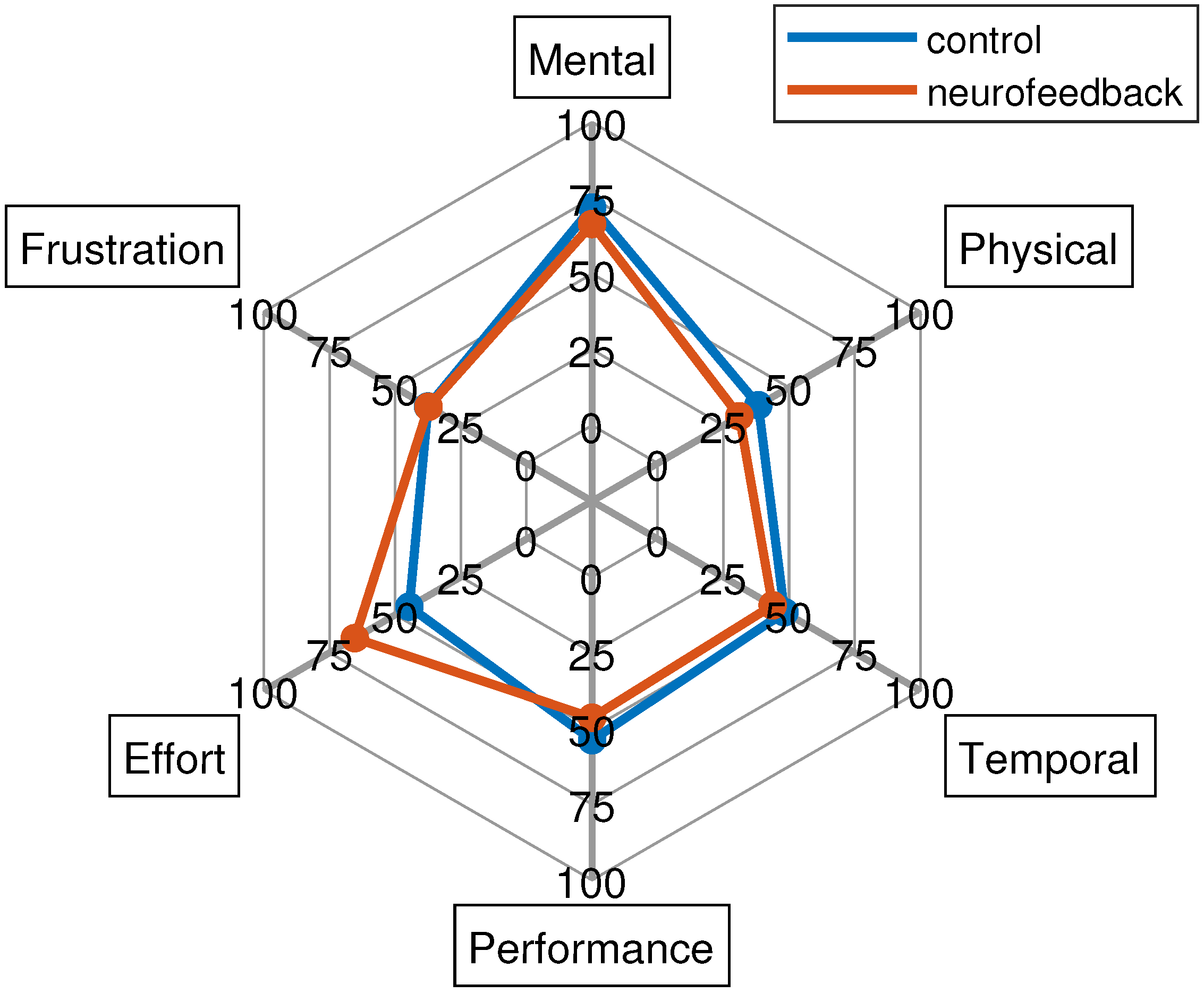
- NASA-TLX (acronym for NASA task load index) [47]: it is a subjective, multidimensional evaluation tool that assesses the perceived workload while performing a task or an activity. The original version also includes a weighting scheme to account for individual differences. However, the most common change made to the questionnaire is the elimination of these weights in order to simplify its application [48]. In this work, it was administered without weights.
- UEQ-S (user experience questionnaire—short form) [49]: a standardized questionnaire to measure the user experience of interactive products. It distinguishes between pragmatic and hedonic quality aspects. The first describes interaction qualities that relate to tasks or goals the user wants to achieve when using the product. The second describes aspects related to pleasure or enjoyment while using the product. Values between −0.8 and +0.8 represent a neutral evaluation of the corresponding scale, values greater than +0.8 represent a positive evaluation, and values lower than −0.8 represent a negative evaluation.
2.5. Statistical Analysis
3. Results
3.1. Participants
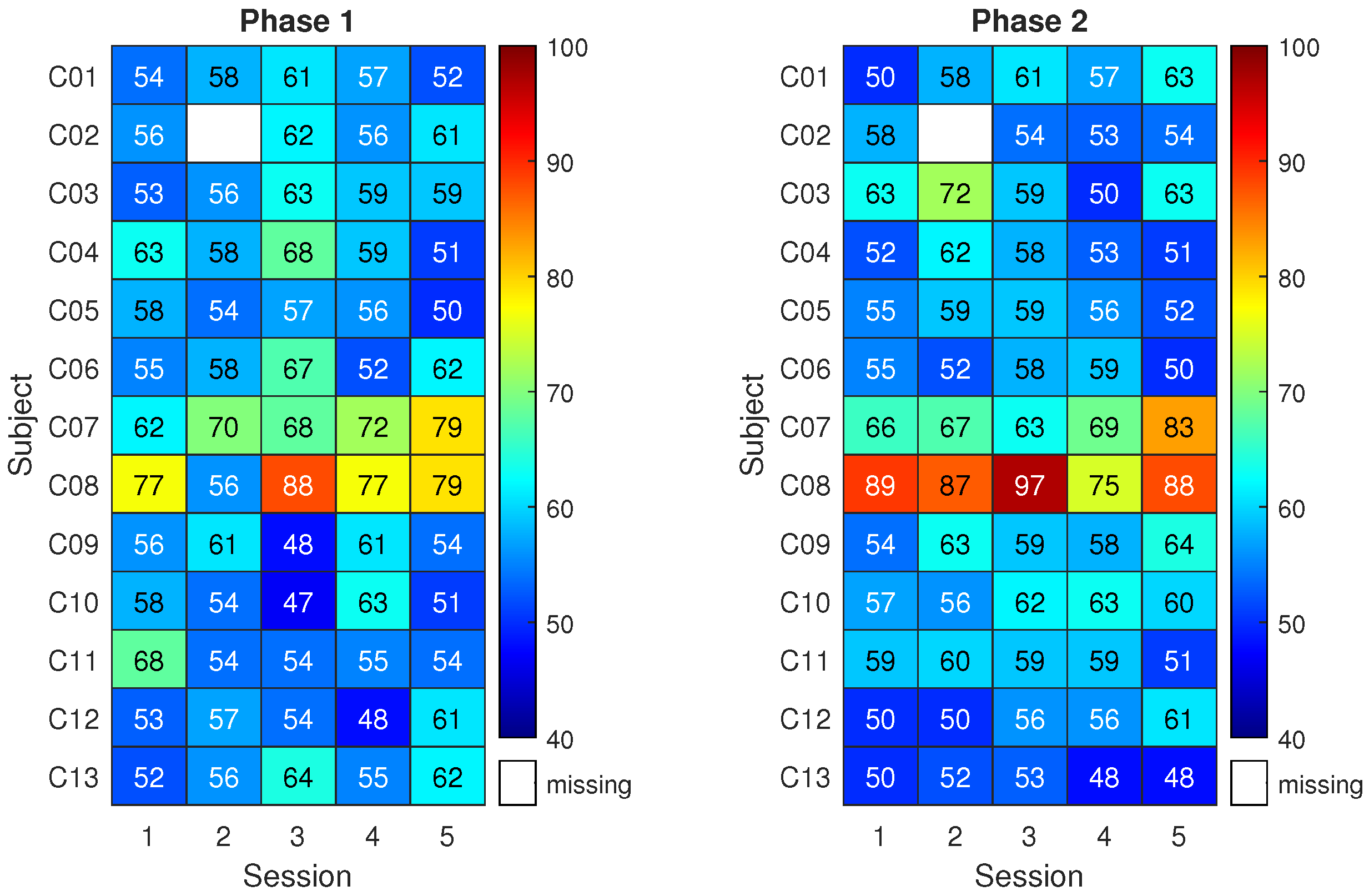
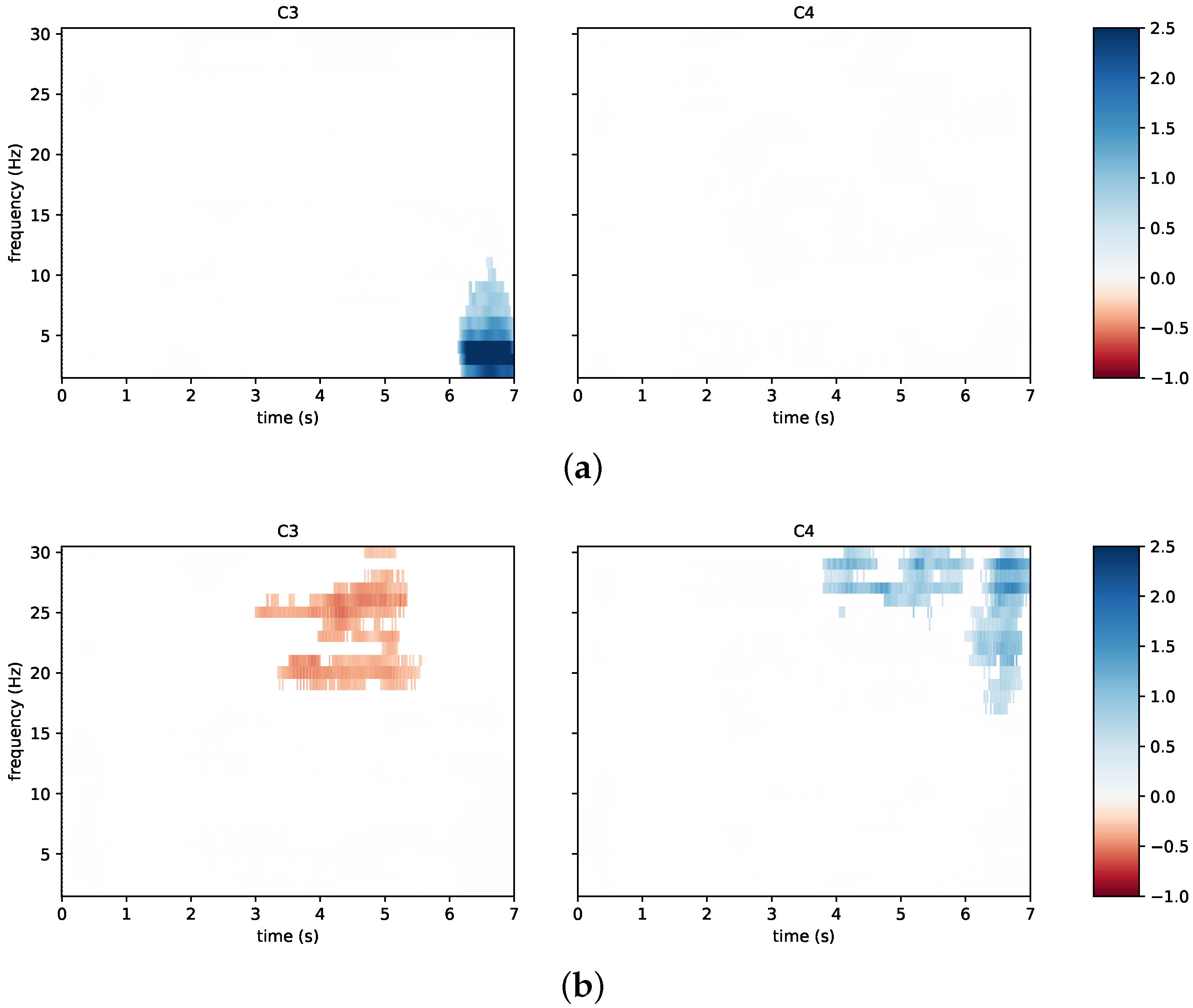
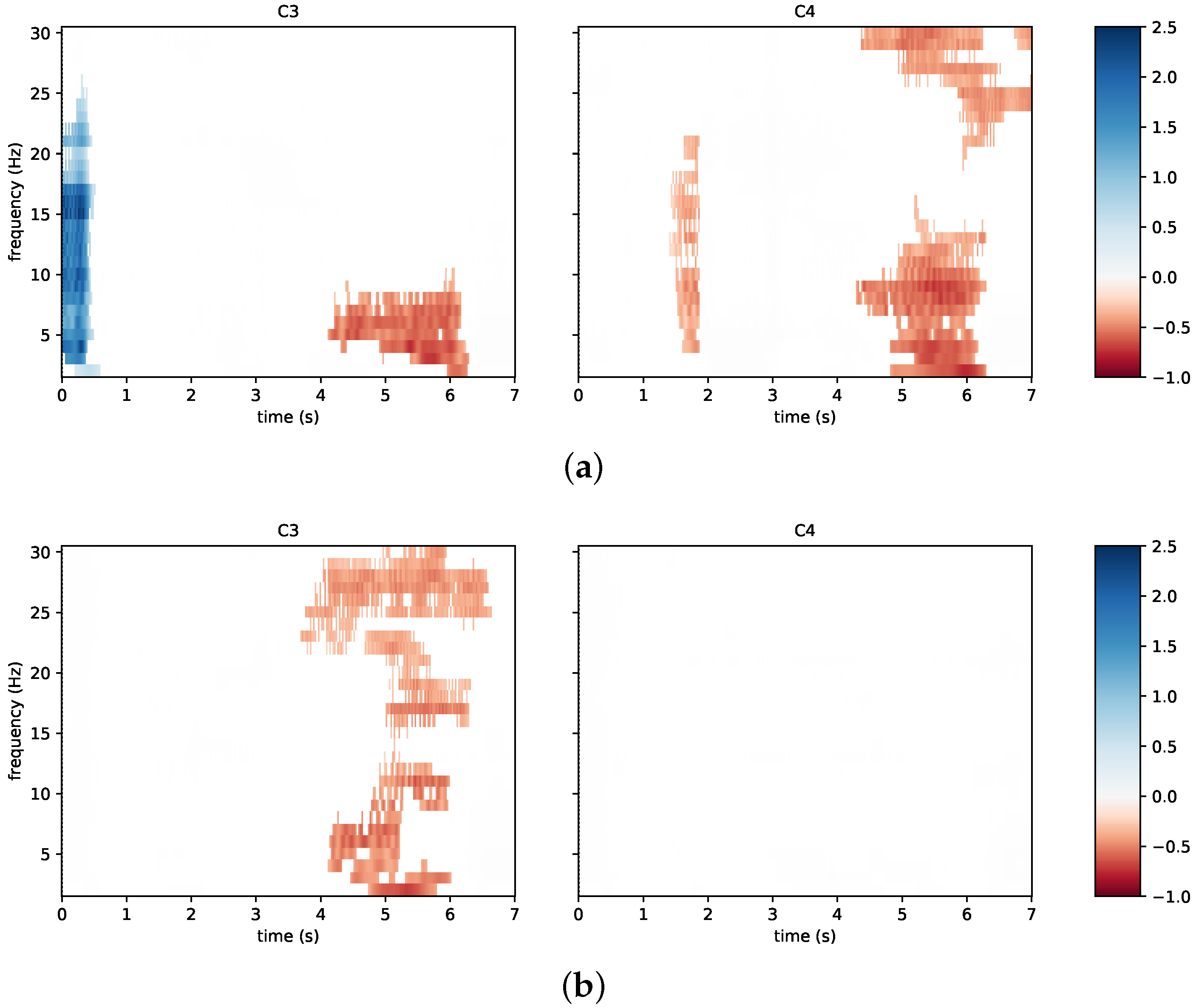
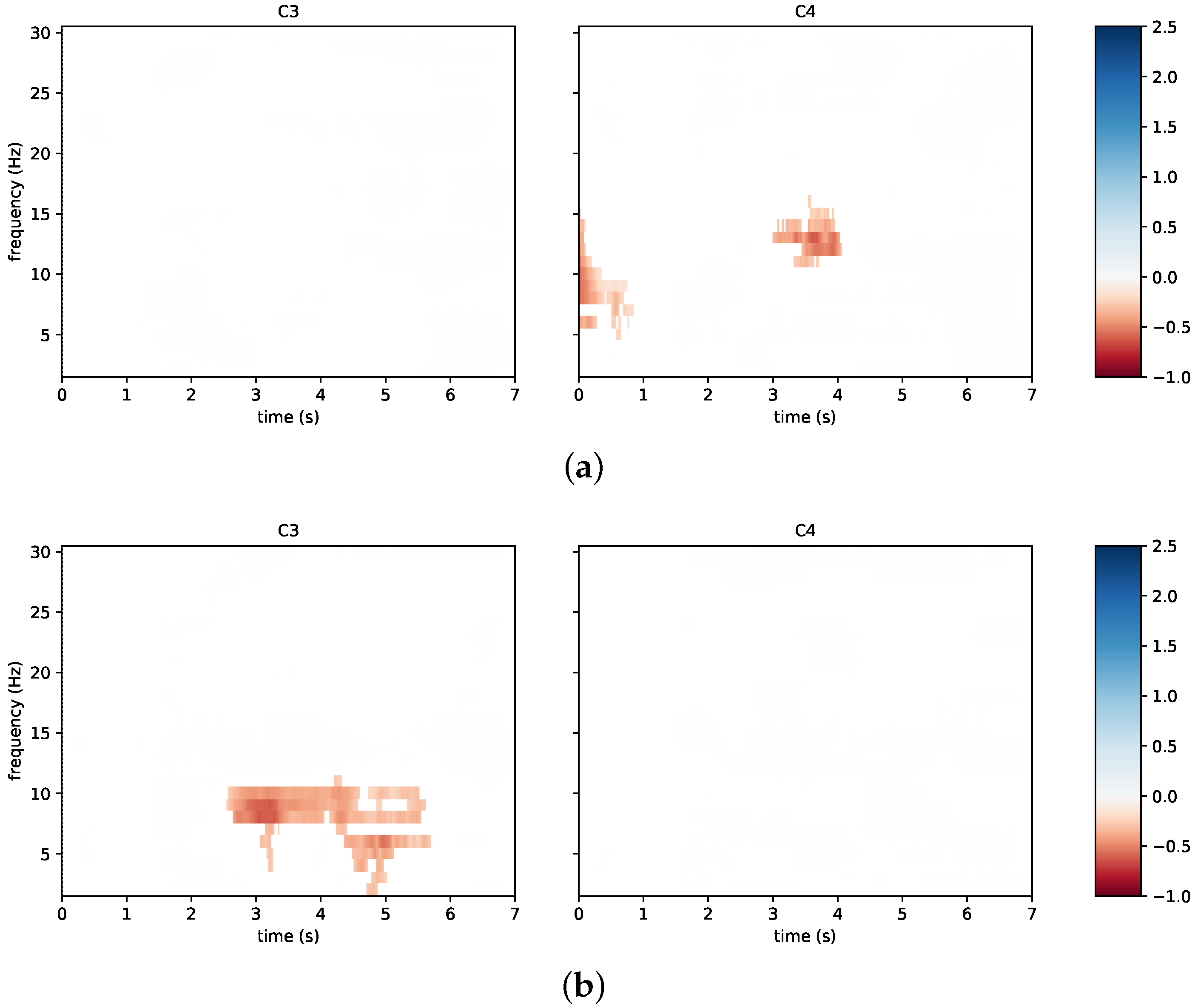
3.2. System Performance
3.3. Questionnaires
4. Discussion
4.1. System Features and Acceptability
4.2. Toward Tele-Rehabilitation
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zampolini, M.; Todeschini, E.; Bernabeu Guitart, M.; Hermens, H.; Ilsbroukx, S.; Macellari, V.; Magni, R.; Rogante, M.; Scattareggia Marchese, S.; Vollenbroek, M.; et al. Tele-rehabilitation: Present and future. Ann. Dell’Istituto Super. Sanita 2008, 44, 125–134. [Google Scholar]
- Piron, L.; Tonin, P.; Trivello, E.; Battistin, L.; Dam, M. Motor tele-rehabilitation in post-stroke patients. Med. Inform. Internet Med. 2004, 29, 119–125. [Google Scholar] [CrossRef]
- Schröder, J.; Van Criekinge, T.; Embrechts, E.; Celis, X.; Van Schuppen, J.; Truijen, S.; Saeys, W. Combining the benefits of tele-rehabilitation and virtual reality-based balance training: A systematic review on feasibility and effectiveness. Disabil. Rehabil. Assist. Technol. 2019, 14, 2–11. [Google Scholar] [CrossRef]
- Coccia, A.; Amitrano, F.; Donisi, L.; Cesarelli, G.; Pagano, G.; Cesarelli, M.; D’Addio, G. Design and validation of an e-textile-based wearable system for remote health monitoring. Acta Imeko 2021, 10, 220–229. [Google Scholar] [CrossRef]
- Cipresso, P.; Serino, S.; Borghesi, F.; Tartarisco, G.; Riva, G.; Pioggia, G.; Gaggioli, A. Continuous measurement of stress levels in naturalistic settings using heart rate variability: An experience-sampling study driving a machine learning approach. ACTA IMEKO 2021, 10, 239–248. [Google Scholar] [CrossRef]
- Mbunge, E.; Muchemwa, B.; Batani, J. Sensors and healthcare 5.0: Transformative shift in virtual care through emerging digital health technologies. Glob. Health J. 2021, 5, 169–177. [Google Scholar] [CrossRef]
- Bulc, V.; Hart, B.; Hannah, M.; Hrovatin, B. Society 5.0 and a Human Centred Health Care. In Medicine-Based Informatics and Engineering; Springer: Berlin/Heidelberg, Germany, 2022; pp. 147–177. [Google Scholar]
- Bacanoiu, M.V.; Danoiu, M. New Strategies to Improve the Quality of Life for Normal Aging versus Pathological Aging. J. Clin. Med. 2022, 11, 4207. [Google Scholar] [CrossRef] [PubMed]
- Truijen, S.; Abdullahi, A.; Bijsterbosch, D.; van Zoest, E.; Conijn, M.; Wang, Y.; Struyf, N.; Saeys, W. Effect of home-based virtual reality training and telerehabilitation on balance in individuals with Parkinson disease, multiple sclerosis, and stroke: A systematic review and meta-analysis. Neurol. Sci. 2022, 43, 2995–3006. [Google Scholar] [CrossRef] [PubMed]
- Belotti, N.; Bonfanti, S.; Locatelli, A.; Rota, L.; Ghidotti, A.; Vitali, A. A Tele-Rehabilitation Platform for Shoulder Motor Function Recovery Using Serious Games and an Azure Kinect Device. In dHealth 2022; IOS Press: Amsterdam, The Netherlands, 2022; pp. 145–152. [Google Scholar]
- Mansour, S.; Ang, K.K.; Nair, K.P.; Phua, K.S.; Arvaneh, M. Efficacy of Brain–Computer Interface and the Impact of Its Design Characteristics on Poststroke Upper-limb Rehabilitation: A Systematic Review and Meta-analysis of Randomized Controlled Trials. Clin. Eeg Neurosci. 2022, 53, 79–90. [Google Scholar] [CrossRef]
- Padfield, N.; Camilleri, K.; Camilleri, T.; Fabri, S.; Bugeja, M. A Comprehensive Review of Endogenous EEG-Based BCIs for Dynamic Device Control. Sensors 2022, 22, 5802. [Google Scholar] [CrossRef]
- Prasad, G.; Herman, P.; Coyle, D.; McDonough, S.; Crosbie, J. Applying a brain-computer interface to support motor imagery practice in people with stroke for upper limb recovery: A feasibility study. J. Neuroeng. Rehabil. 2010, 7, 60. [Google Scholar] [CrossRef]
- Wen, D.; Fan, Y.; Hsu, S.H.; Xu, J.; Zhou, Y.; Tao, J.; Lan, X.; Li, F. Combining brain–computer interface and virtual reality for rehabilitation in neurological diseases: A narrative review. Ann. Phys. Rehabil. Med. 2021, 64, 101404. [Google Scholar] [CrossRef]
- Wang, Z.; Zhou, Y.; Chen, L.; Gu, B.; Liu, S.; Xu, M.; Qi, H.; He, F.; Ming, D. A BCI based visual-haptic neurofeedback training improves cortical activations and classification performance during motor imagery. J. Neural Eng. 2019, 16, 066012. [Google Scholar] [CrossRef] [PubMed]
- Arpaia, P.; Coyle, D.; Donnarumma, F.; Esposito, A.; Natalizio, A.; Parvis, M. Visual and haptic feedback in detecting motor imagery within a wearable brain-computer interface. Measurement 2022, 206, 112304. [Google Scholar] [CrossRef]
- Singh, A.; Hussain, A.A.; Lal, S.; Guesgen, H.W. A comprehensive review on critical issues and possible solutions of motor imagery based electroencephalography brain-computer interface. Sensors 2021, 21, 2173. [Google Scholar] [CrossRef] [PubMed]
- Arpaia, P.; Callegaro, L.; Cultrera, A.; Esposito, A.; Ortolano, M. Metrological characterization of consumer-grade equipment for wearable brain–computer interfaces and extended reality. IEEE Trans. Instrum. Meas. 2021, 71, 1–9. [Google Scholar] [CrossRef]
- Moioli, R.C.; Nardelli, P.H.; Barros, M.T.; Saad, W.; Hekmatmanesh, A.; Silva, P.E.G.; de Sena, A.S.; Dzaferagic, M.; Siljak, H.; Van Leekwijck, W.; et al. Neurosciences and wireless networks: The potential of brain-type communications and their applications. IEEE Commun. Surv. Tutorials 2021, 23, 1599–1621. [Google Scholar] [CrossRef]
- Teplan, M. Fundamentals of EEG measurement. Meas. Sci. Rev. 2002, 2, 1–11. [Google Scholar]
- Ienca, M.; Haselager, P.; Emanuel, E.J. Brain leaks and consumer neurotechnology. Nat. Biotechnol. 2018, 36, 805–810. [Google Scholar] [CrossRef]
- Affanni, A.; Aminosharieh Najafi, T.; Guerci, S. Development of an eeg headband for stress measurement on driving simulators. Sensors 2022, 22, 1785. [Google Scholar] [CrossRef] [PubMed]
- Arpaia, P.; Callegaro, L.; Cultrera, A.; Esposito, A.; Ortolano, M. Metrological characterization of a low-cost electroencephalograph for wearable neural interfaces in industry 4.0 applications. In Proceedings of the 2021 IEEE International Workshop on Metrology for Industry 4.0 & IoT (MetroInd 4.0 & IoT), Rome, Italy, 7–9 June 2021; IEEE: New York, NY, USA, 2021; pp. 1–5. [Google Scholar]
- Jeong, J.H.; Choi, J.H.; Kim, K.T.; Lee, S.J.; Kim, D.J.; Kim, H.M. Multi-domain convolutional neural networks for lower-limb motor imagery using dry vs. wet electrodes. Sensors 2021, 21, 6672. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Li, G.; Chen, J.; Duan, Y.; Zhang, D. Novel semi-dry electrodes for brain–computer interface applications. J. Neural Eng. 2016, 13, 046021. [Google Scholar] [CrossRef] [PubMed]
- Hinrichs, H.; Scholz, M.; Baum, A.K.; Kam, J.W.; Knight, R.T.; Heinze, H.J. Comparison between a wireless dry electrode EEG system with a conventional wired wet electrode EEG system for clinical applications. Sci. Rep. 2020, 10, 5218. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, X.; Sun, H.; Fan, Z.; Zhong, X. Portable brain-computer interface based on novel convolutional neural network. Comput. Biol. Med. 2019, 107, 248–256. [Google Scholar] [CrossRef] [PubMed]
- Lin, B.S.; Pan, J.S.; Chu, T.Y.; Lin, B.S. Development of a wearable motor-imagery-based brain–computer interface. J. Med. Syst. 2016, 40, 1–8. [Google Scholar] [CrossRef]
- Lo, C.C.; Chien, T.Y.; Chen, Y.C.; Tsai, S.H.; Fang, W.C.; Lin, B.S. A wearable channel selection-based brain-computer interface for motor imagery detection. Sensors 2016, 16, 213. [Google Scholar] [CrossRef]
- Lisi, G.; Hamaya, M.; Noda, T.; Morimoto, J. Dry-wireless EEG and asynchronous adaptive feature extraction towards a plug-and-play co-adaptive brain robot interface. In Proceedings of the 2016 IEEE International Conference on Robotics and Automation (ICRA), Stockholm, Sweden, 16–21 May 2016; IEEE: New York, NY, USA, 2016; pp. 959–966. [Google Scholar]
- Lisi, G.; Rivela, D.; Takai, A.; Morimoto, J. Markov switching model for quick detection of event related desynchronization in EEG. Front. Neurosci. 2018, 12, 24. [Google Scholar] [CrossRef]
- Casso, M.I.; Jeunet, C.; Roy, R.N. Heading for motor imagery brain-computer interfaces (MI-BCIs) usable out-of-the-lab: Impact of dry electrode setup on classification accuracy. In Proceedings of the 2021 10th International IEEE/EMBS Conference on Neural Engineering (NER), Virtual, 4–6 May 2021; IEEE: New York, NY, USA, 2021; pp. 690–693. [Google Scholar]
- Simon, C.; Ruddy, K.L. A wireless, wearable Brain-Computer Interface for neurorehabilitation at home; A feasibility study. In Proceedings of the 2022 10th International Winter Conference on Brain-Computer Interface (BCI), Gangwon-do, Republic of Korea, 21–23 February 2022; IEEE: New York, NY, USA, 2022; pp. 1–6. [Google Scholar]
- Arpaia, P.; Coyle, D.; Donnarumma, F.; Esposito, A.; Natalizio, A.; Parvis, M.; Pesola, M.; Vallefuoco, E. Multimodal Feedback in Assisting a Wearable Brain-Computer Interface Based on Motor Imagery. In Proceedings of the 2022 IEEE International Conference on Metrology for Extended Reality, Artificial Intelligence and Neural Engineering (MetroXRAINE), Rome, Italy, 26–28 October 2022; IEEE: New York, NY, USA, 2022; pp. 691–696. [Google Scholar]
- Hekmatmanesh, A.; Nardelli, P.H.; Handroos, H. Review of the state-of-the-art of brain-controlled vehicles. IEEE Access 2021, 9, 110173–110193. [Google Scholar] [CrossRef]
- Ang, K.K.; Chin, Z.Y.; Wang, C.; Guan, C.; Zhang, H. Filter bank common spatial pattern algorithm on BCI competition IV datasets 2a and 2b. Front. Neurosci. 2012, 6, 39. [Google Scholar] [CrossRef]
- Arpaia, P.; Esposito, A.; Natalizio, A.; Parvis, M. How to successfully classify EEG in motor imagery BCI: A metrological analysis of the state of the art. J. Neural Eng. 2022, 19, 031002. [Google Scholar] [CrossRef]
- Mullen, T.R.; Kothe, C.A.; Chi, Y.M.; Ojeda, A.; Kerth, T.; Makeig, S.; Jung, T.P.; Cauwenberghs, G. Real-time neuroimaging and cognitive monitoring using wearable dry EEG. IEEE Trans. Biomed. Eng. 2015, 62, 2553–2567. [Google Scholar] [CrossRef] [PubMed]
- Arpaia, P.; De Bendetto, E.; Esposito, A.; Natalizio, A.; Parvis, M.; Pesola, M. Comparing artifact removal techniques for daily-life electroencephalography with few channels. In Proceedings of the 2022 IEEE International Symposium on Medical Measurements and Applications (MeMeA), Messina, Italy, 22–24 June 2022; IEEE: New York, NY, USA, 2022; pp. 1–6. [Google Scholar]
- Delorme, A.; Makeig, S. EEGLAB: An open source toolbox for analysis of single-trial EEG dynamics including independent component analysis. J. Neurosci. Methods 2004, 134, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Williams, S.E.; Cumming, J.; Ntoumanis, N.; Nordin-Bates, S.M.; Ramsey, R.; Hall, C. Further validation and development of the movement imagery questionnaire. J. Sport Exerc. Psychol. 2012, 34, 621–646. [Google Scholar] [CrossRef] [PubMed]
- Hall, C.R.; Pongrac, J. Movement Imagery: Questionnaire; University of Western Ontario Faculty of Physical Education: London, ON, Canada, 1983. [Google Scholar]
- Hall, C.R.; Martin, K.A. Measuring movement imagery abilities: A revision of the movement imagery questionnaire. J. Ment. Imag. 1997, 21, 143–154. [Google Scholar]
- Brooke, J. SUS-A quick and dirty usability scale. Usability Eval. Ind. 1996, 189, 4–7. [Google Scholar]
- Bangor, A.; Kortum, P.; Miller, J. Determining what individual SUS scores mean: Adding an adjective rating scale. J. Usability Stud. 2009, 4, 114–123. [Google Scholar]
- Bangor, A.; Kortum, P.T.; Miller, J.T. An empirical evaluation of the system usability scale. Int. J.-Hum.-Comput. Interact. 2008, 24, 574–594. [Google Scholar] [CrossRef]
- Hart, S.G.; Staveland, L.E. Development of NASA-TLX (Task Load Index): Results of empirical and theoretical research. In Advances in Psychology; Elsevier: Amsterdam, The Netherlands, 1988; Volume 52, pp. 139–183. [Google Scholar]
- Hart, S.G. NASA-task load index (NASA-TLX); 20 years later. In Proceedings of the Human Factors and Ergonomics Society Annual Meeting; Sage publications: Los Angeles, CA, USA, 2006; Volume 50, pp. 904–908. [Google Scholar]
- Schrepp, M.; Hinderks, A.; Thomaschewski, J. Design and evaluation of a short version of the user experience questionnaire (UEQ-S). Int. J. Interact. Multimed. Artif. Intell. 2017, 4, 103–108. [Google Scholar] [CrossRef]
- Cho, H.; Ahn, M.; Ahn, S.; Kwon, M.; Jun, S.C. EEG datasets for motor imagery brain–computer interface. GigaScience 2017, 6, gix034. [Google Scholar] [CrossRef]
- Rosner, B. Fundamentals of Biostatistics; Cengage learning: Belmont, CA, USA, 2015. [Google Scholar]
- Combrisson, E.; Jerbi, K. Exceeding chance level by chance: The caveat of theoretical chance levels in brain signal classification and statistical assessment of decoding accuracy. J. Neurosci. Methods 2015, 250, 126–136. [Google Scholar] [CrossRef]
- Batula, A.M.; Mark, J.A.; Kim, Y.E.; Ayaz, H. Comparison of brain activation during motor imagery and motor movement using fNIRS. Comput. Intell. Neurosci. 2017, 2017, 5491296. [Google Scholar] [CrossRef] [PubMed]
- Arpaia, P.; Donnarumma, F.; Esposito, A.; Parvis, M. Channel selection for optimal EEG measurement in motor imagery-based brain-computer interfaces. Int. J. Neural Syst. 2021, 31, 2150003. [Google Scholar] [CrossRef] [PubMed]
- López-Larraz, E.; Montesano, L.; Gil-Agudo, Á.; Minguez, J.; Oliviero, A. Evolution of EEG motor rhythms after spinal cord injury: A longitudinal study. PLoS ONE 2015, 10, e0131759. [Google Scholar] [CrossRef]
- Lee, D.; Hwang, S. Motor imagery on upper extremity function for persons with stroke: A systematic review and meta-analysis. Phys. Ther. Rehabil. Sci. 2019, 8, 52–59. [Google Scholar] [CrossRef]
- De Vries, S.; Tepper, M.; Feenstra, W.; Oosterveld, H.; Boonstra, A.M.; Otten, B. Motor imagery ability in stroke patients: The relationship between implicit and explicit motor imagery measures. Front. Hum. Neurosci. 2013, 7, 790. [Google Scholar] [CrossRef] [PubMed]
- Szameitat, A.J.; Shen, S.; Conforto, A.; Sterr, A. Cortical activation during executed, imagined, observed, and passive wrist movements in healthy volunteers and stroke patients. Neuroimage 2012, 62, 266–280. [Google Scholar] [CrossRef]
- Bacanoiu, M.V.; Mititelu, R.R.; Danoiu, M.; Olaru, G.; Buga, A.M. Functional recovery in Parkinson’s Disease: Current state and future perspective. J. Clin. Med. 2020, 9, 3413. [Google Scholar] [CrossRef]
- Caligiore, D.; Mustile, M.; Spalletta, G.; Baldassarre, G. Action observation and motor imagery for rehabilitation in Parkinson’s disease: A systematic review and an integrative hypothesis. Neurosci. Biobehav. Rev. 2017, 72, 210–222. [Google Scholar] [CrossRef]
- Baniqued, P.D.E.; Stanyer, E.C.; Awais, M.; Alazmani, A.; Jackson, A.E.; Mon-Williams, M.A.; Mushtaq, F.; Holt, R.J. Brain–computer interface robotics for hand rehabilitation after stroke: A systematic review. J. Neuroeng. Rehabil. 2021, 18, 15. [Google Scholar] [CrossRef]
- Mladenović, J.; Frey, J.; Pramij, S.; Mattout, J.; Lotte, F. Towards identifying optimal biased feedback for various user states and traits in motor imagery BCI. IEEE Trans. Biomed. Eng. 2021, 69, 1101–1110. [Google Scholar] [CrossRef]
- Tao, L.; Cao, T.; Wang, Q.; Liu, D.; Sun, J. Distribution Adaptation and Classification Framework Based on Multiple Kernel Learning for Motor Imagery BCI Illiteracy. Sensors 2022, 22, 6572. [Google Scholar] [CrossRef] [PubMed]
- Brunner, I.; Lundquist, C.B.; Pedersen, A.R.; Spaich, E.; Dosen, S.; Savic, A. Brain Computer Interface training for patients with severe upper limb paresis after stroke-A randomized controlled pilot trial. J. Neuroeng. Rehabil. 2023. [Google Scholar] [CrossRef]




| Control | Neurofeedback | |
|---|---|---|
| Sex | male: 31%, female: 69% | male: 57%, female: 42% |
| Handedness | right: 85%, left: 15%, both: 0% | right: 79%, left: 14%, both: 7% |
| Practicing sport | yes: 38%, no: 62%, professional: 0% | yes: 64%, no: 36%, professional: 0% |
| BCI experience | no: 38.5%, active: 8%, passive: 15%, | no: 43%, active: 7%, passive: 21%, |
| reactive: 0%, multiple: 38.5% | reactive: 0%, multiple: 29% | |
| NF experience | yes: 46%, no: 54% | yes: 36%, no: 64% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arpaia, P.; Coyle, D.; Esposito, A.; Natalizio, A.; Parvis, M.; Pesola, M.; Vallefuoco, E. Paving the Way for Motor Imagery-Based Tele-Rehabilitation through a Fully Wearable BCI System. Sensors 2023, 23, 5836. https://doi.org/10.3390/s23135836
Arpaia P, Coyle D, Esposito A, Natalizio A, Parvis M, Pesola M, Vallefuoco E. Paving the Way for Motor Imagery-Based Tele-Rehabilitation through a Fully Wearable BCI System. Sensors. 2023; 23(13):5836. https://doi.org/10.3390/s23135836
Chicago/Turabian StyleArpaia, Pasquale, Damien Coyle, Antonio Esposito, Angela Natalizio, Marco Parvis, Marisa Pesola, and Ersilia Vallefuoco. 2023. "Paving the Way for Motor Imagery-Based Tele-Rehabilitation through a Fully Wearable BCI System" Sensors 23, no. 13: 5836. https://doi.org/10.3390/s23135836
APA StyleArpaia, P., Coyle, D., Esposito, A., Natalizio, A., Parvis, M., Pesola, M., & Vallefuoco, E. (2023). Paving the Way for Motor Imagery-Based Tele-Rehabilitation through a Fully Wearable BCI System. Sensors, 23(13), 5836. https://doi.org/10.3390/s23135836









