Machine Learning-Based Classification of Abnormal Liver Tissues Using Relative Permittivity
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Procedures
2.1.1. Animal Experiments
2.1.2. Measurement Setup
2.2. Data
2.3. Classification Experiments
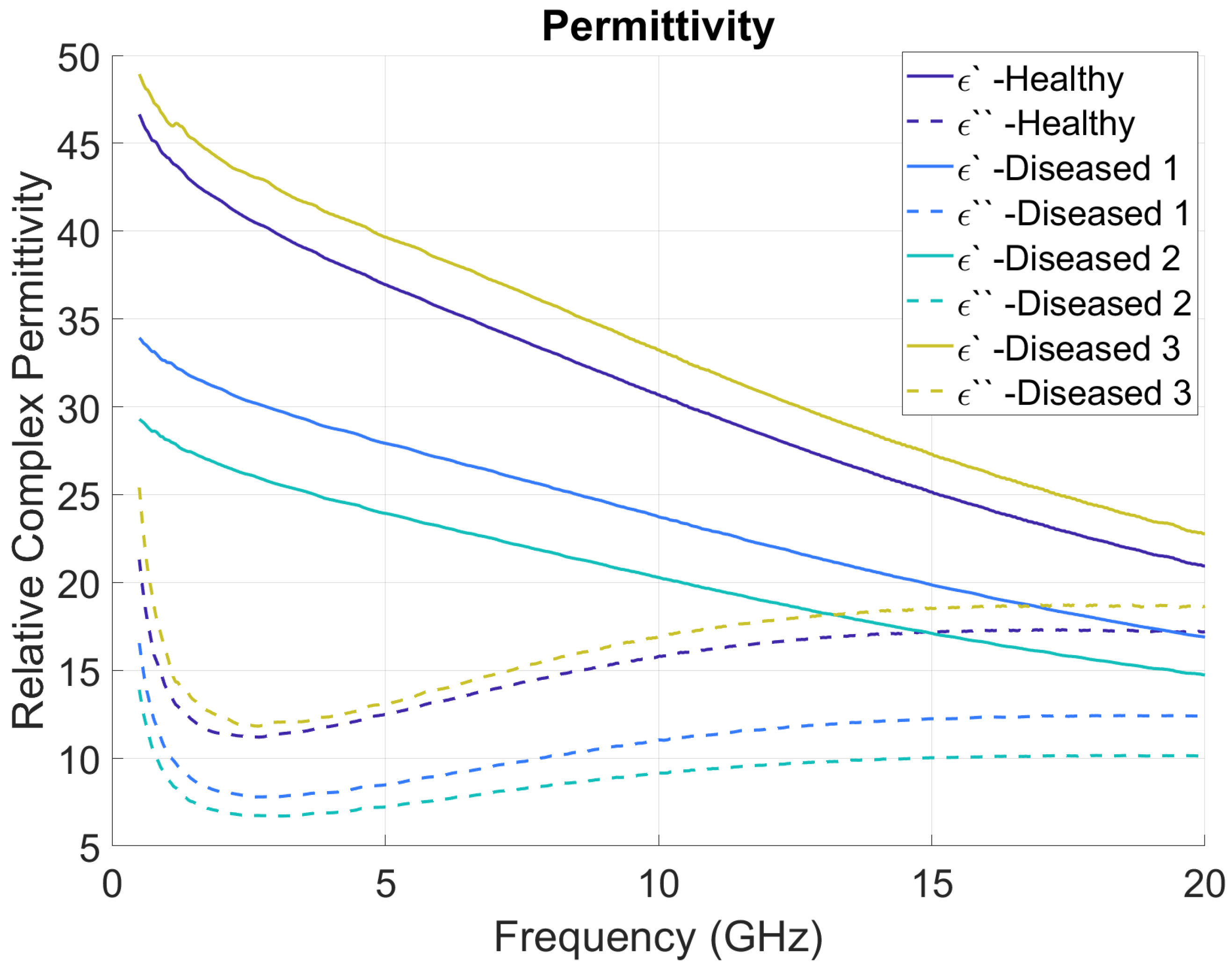
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Gabriel, C.; Gabriel, S.; Corthout, E. The dielectric properties of biological tissues: I. Literature survey. Phys. Med. Biol. 1996, 41, 2231–2249. [Google Scholar] [CrossRef] [PubMed]
- Gabriel, S.; Lau, R.W.; Gabriel, C. The dielectric properties of biological tissues: II. Measurements in the frequency range 10 Hz to 20 GHz. Phys. Med. Biol. 1996, 41, 2251–2269. [Google Scholar] [CrossRef]
- Gabriel, S.; Lau, R.W.; Gabriel, C. The dielectric properties of biological tissues: III. Parametric models for the dielectric spectrum of tissues. Phys. Med. Biol. 1996, 41, 2271–2293. [Google Scholar] [CrossRef] [PubMed]
- Amin, B.; Elahi, M.A.; Shahzad, A.; Porter, E.; McDermott, B.; O’Halloran, M. Dielectric properties of bones for the monitoring of osteoporosis. Med Biol. Eng. Comput. 2018, 57, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Di Meo, S.; Espin-Lopez, P.; Martellosio, A.; Pasian, M.; Bozzi, M.; Perregrini, L.; Mazzanti, A.; Svelto, F.; Summers, P.; Renne, G.; et al. Dielectric properties of breast tissues: Experimental results up to 50 GHz. In Proceedings of the 12th European Conference on Antennas and Propagation (EuCAP 2018), London, UK, 9–13 April 2018; pp. 1–5. [Google Scholar]
- Liewei, S.; Ward, E.R.; Story, B. A review of dielectric properties of normal and malignant breast tissue. In Proceedings of the IEEE SoutheastCon 2002, Columbia, SC, USA, 5–7 April 2002; pp. 457–462. [Google Scholar]
- Surowiec, A.J.; Stuchly, S.S.; Barr, J.R.; Swarup, A. Dielectric properties of breast carcinoma and the surrounding tissues. IEEE Trans. Biomed. Eng. 1988, 35, 257–263. [Google Scholar] [CrossRef]
- Lazebnik, M.; Okoniewski, M.; Booske, J.H.; Hagness, S.C. Highly Accurate Debye Models for Normal and Malignant Breast Tissue Dielectric Properties at Microwave Frequencies. IEEE Microw. Wirel. Components Lett. 2007, 17, 822–882. [Google Scholar] [CrossRef]
- O’Rourke, A.P.; Lazebnik, M.; Bertram, J.M.; Converse, M.C.; Hagness, S.C.; Webster, J.G.; Mahvi, D.M. Dielectric properties of human normal, malignant and cirrhotic liver tissue: In vivo and ex vivo measurements from 0.5 to 20 GHz using a precision open-ended coaxial probe. Phys. Med. Biol. 2007, 52, 4707–4719. [Google Scholar] [CrossRef]
- Yilmaz, T.; Foster, R.; Hao, Y. Towards Accurate Dielectric Property Retrieval of Biological Tissues for Blood Glucose Monitoring. IEEE Trans. Microw. Theory Tech. 2014, 62, 3193–3204. [Google Scholar] [CrossRef]
- Treo, E.; Cervantes, D.; Felice, C.; Tirado, M.; Valentinuzzi, M. Hematocrit measurement by dielectric spectroscopy. IEEE Trans. Biomed. Eng. 2005, 52, 124–127. [Google Scholar] [CrossRef]
- Sacli, B.; Aydinalp, C.; Cansiz, G.; Joof, S.; Yilmaz, T.; Cayoren, M.; Onal, B.; Akduman, I. Microwave dielectric property based classification of renal calculi: Application of a kNN algorithm. Comput. Biol. Med. 2019, 112, 103366. [Google Scholar] [CrossRef]
- Gaddam, S.; Samaddar, P.; Khan, M.; Damani, D.; Shivaram, S.; Roy, S.; Dey, S.; Mitra, D.; Arunachalam, S.P. On the Non-invasive Sensing of Arterial Waveform and Hematocrit using Microwaves. In Proceedings of the IEEE International Symposium on Antennas and Propagation and USNC-URSI Radio Science Meeting (AP-S/URSI), Denver, CO, USA, 10–15 July 2022; pp. 1082–1083. [Google Scholar]
- Samaddar, P.; Steinfort, L.M.N.; Fetzer, J.; Arunachalam, S.P.; Wang, K.K.; Leggett, C.L. Tu1660: The Dielectric Properties of Upper Gastrointestinal Tissue Types—An Ex-Vivo Feasibility Study. Gastroenterology 2022, 162, S-1206. [Google Scholar] [CrossRef]
- Gaddam, S.; Samaddar, P.; Khan, M.; Damani, D.; Shivaram, S.; Roy, S.; Mitra, D.; Dey, S.; Arunachalam, S.P. Towards Non-Invasive Mapping of Blood Flow Velocity using Microwaves. In Proceedings of the IEEE International Symposium on Antennas and Propagation and USNC-URSI Radio Science Meeting (AP-S/URSI), Denver, CO, USA, 10–15 July 2022. [Google Scholar]
- Rahmani, H.; Archang, M.M.; Jamali, B.; Forghani, M.; Ambrus, A.M.; Ramalingam, D.; Sun, Z.; Scumpia, P.O.; Coller, H.A.; Babakhani, A. Towards a Machine-Learning-Assisted Dielectric Sensing Platform for Point-of-Care Wound Monitoring. IEEE Sens. Lett. 2020, 4, 1–4. [Google Scholar] [CrossRef]
- Helwana, A.; Idokob, J.B.; Abiyev, R.H. Machine learning techniques for classification of breast tissue. In Proceedings of the 9th International Conference on Theory and Application of Soft Computing, Computing with Words and Perception, ICSCCW 2017 Procedia Computer Science, Budapest, Hungary, 24–25 August 2017; Volume 120, pp. 402–410. [Google Scholar]
- Yilmaz, T.; Kılıç, M.A.; Erdoğan, M.; Çayören, M.; Tunaoğlu, D.; Kurtoğlu, İ.; Yaslan, Y.; Çayören, H.; Arıkan, A.E.; Teksöz, S.; et al. Machine learning aided diagnosis of hepatic malignancies through in vivo dielectric measurements with microwaves. Phys. Med. Biol. 2016, 61, 5089–5102. [Google Scholar] [CrossRef] [PubMed]
- La Gioia, A.; Porter, E.; Merunka, I.; Shahzad, A.; Salahuddin, S.; Jones, M.; O’Halloran, M. Open-Ended Coaxial Probe Technique for Dielectric Measurement of Biological Tissues: Challenges and Common Practices. Diagnostics 2018, 8, 40. [Google Scholar] [CrossRef] [PubMed]
- Samaddar, P.; Gaddam, S.; K, P.S.; Khan, M.; Mitra, D.; Leggett, C.; Roy, S.; Arunachalam, S.P. On the Effects of the Measured Dielectric Properties at Variable Thickness of Biological Tissue Samples using Open-Ended Coaxial Probe Method. In Proceedings of the IEEE International Symposium on Antennas and Propagation and USNC-URSI Radio Science Meeting (AP-S/URSI), Denver, CO, USA, 10–15 July 2022. [Google Scholar]
- Samaddar, P.; Gaddam, S.; Khan, M.; Roy, S.; Mitra, D.; Arunachalam, S.P. On the Dielectric Characterization of Biological Samples for Microwave Imaging Reconstruction. In Proceedings of the IEEE International Symposium on Antennas and Propagation and USNC-URSI Radio Science Meeting (AP-S/URSI), Denver, CO, USA, 10–15 July 2022; pp. 1082–1083. [Google Scholar]
- Gao, J.; Wei, B.; de Assuncao, T.M.; Liu, Z.; Hu, X.; Ibrahim, S.; Cooper, S.A.; Cao, S.; Shah, V.H.; Kostallari, E. Hepatic stellate cell autophagy inhibits extracellular vesicle release to attenuate liver fibrosis. J. Hepatol. 2020, 73, 1144–1154. [Google Scholar] [CrossRef]
- Kostallari, E.; Hirsova, P.; Prasnicka, A.; Verma, V.K.; Yaqoob, U.; Wongjarupong, N.; Roberts, L.R.; Shah, V.H. Hepatic stellate cell-derived platelet-derived growth factor receptor-alpha-enriched extracellular vesicles promote liver fibrosis in mice through SHP2. Hepatology 2018, 68, 333–348. [Google Scholar] [CrossRef]
- Kostallari, E.; Wei, B.; Sicard, D.; Li, J.; Cooper, S.A.; Gao, J.; Dehankar, M.; Li, Y.; Cao, S.; Yin, M.; et al. Stiffness is associated with hepatic stellate cell heterogeneity during liver fibrosis. Am. J. Physiol. Liver Physiol. 2022, 322, G234–G246. [Google Scholar] [CrossRef]
- Farrugiaa, L.; W, P.S.; Mangiona, L.Z.; Sammut, C.V. Accurate in vivo dielectric properties of liver from 500 MHz to 40 GHz and their correlation to ex vivo measurements. Electromagn. Biol. Med. 2016, 35, 365–373. [Google Scholar] [CrossRef]
- Pollacco, D.A.; Farina, L.; Wismayer, P.S.; Farrugia, L.; Sammut, C.V. Characterization of the dielectric properties of biological tissues and their correlation to tissue hydration. IEEE Trans. Dielectr. Electr. Insul. 2018, 25, 2191–2197. [Google Scholar] [CrossRef]
- N1501A Dielectric Probe Kit, Keysight Technologies. Available online: https://www.keysight.com/us/en/support/N1501A/dielectric-probe-kit.html (accessed on 11 November 2022).
- Cavagnaro, M.; Ruvio, G. Numerical Sensitivity Analysis for Dielectric Characterization of Biological Samples by Open-Ended Probe Technique. Sensors 2020, 20, 3756. [Google Scholar] [CrossRef]
- Porter, E.; la Gioia, A.; Santorelli, A.; O’Halloran, M. Modeling of the Dielectric Properties of Biological Tissues within the Histology Region. IEEE Trans. Dielectr. Electr. Insul. 2017, 24, 3290–3301. [Google Scholar] [CrossRef]
- Andreuccetti, D.; Fossi, R.; Petrucci, C. An Internet Resource for the Calculation of the Dielectric Properties of Body Tissues in the Frequency Range 10 Hz–100 GHz. Available online: http://niremf.ifac.cnr.it/tissprop/ (accessed on 1 December 2022).
- Pedregosa, F.; Varoquaux, G.; Gramfort, A.; Michel, V.; Thirion, B.; Grisel, O.; Blondel, M.; Prettenhofer, P.; Weiss, R.; Dubourg, V.; et al. Scikit-learn: Machine Learning in Python. J. Mach. Learn. Res. 2011, 12, 2825–2830. [Google Scholar]
- King, G.; Zeng, L. Logistic Regression in Rare Events Data. Political Anal. 2001, 9, 137–163. [Google Scholar] [CrossRef]
- Betmouni, S. Diagnostic digital pathology implementation: Learning from the digital health experience. Digit. Health 2021, 7, 20552076211020240. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.Y.; Abel, J.T.; Balis, U.G.; McClintock, D.S.; Pantanowitz, L. Challenges in the Development, Deployment, and Regulation of Artificial Intelligence in Anatomic Pathology. Am. J. Pathol. 2020, 191, 1684–1692. [Google Scholar] [CrossRef]
- Gabriel, C.; Peyman, A. Dielectric Properties of Biological TissuesVariation with Age. In Conn’s Handbook of Models for Human Aging, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 939–952. [Google Scholar]
- Hussein, M.; Awwad, F.; Jithin, D.; El Hasasna, H.; Athamneh, K.; Iratni, R. Breast cancer cells exhibits specific dielectric signature in vitro using the open-ended coaxial probe technique from 200 MHz to 13.6 GHz. Sci. Rep. 2019, 9, 468. [Google Scholar] [CrossRef]




| Tissue Types | Samples | Measurements |
|---|---|---|
| Healthy | 25 | 125 |
| Diseased 1 | 27 | 135 |
| Diseased 2 | 9 | 45 |
| Diseased 3 | 11 | 55 |
| Classifier | Precision | Recall | F1-Score | Accuracy |
|---|---|---|---|---|
| Logistic Regression | 0.81 | 0.80 | 0.80 | 0.80 |
| K-Nearest Neighbors | 0.76 | 0.75 | 0.74 | 0.75 |
| Random Forest | 0.81 | 0.80 | 0.79 | 0.80 |
| Support Vector Machines | 0.90 | 0.90 | 0.90 | 0.90 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Samaddar, P.; Mishra, A.K.; Gaddam, S.; Singh, M.; Modi, V.K.; Gopalakrishnan, K.; Bayer, R.L.; Igreja Sa, I.C.; Khanal, S.; Hirsova, P.; et al. Machine Learning-Based Classification of Abnormal Liver Tissues Using Relative Permittivity. Sensors 2022, 22, 9919. https://doi.org/10.3390/s22249919
Samaddar P, Mishra AK, Gaddam S, Singh M, Modi VK, Gopalakrishnan K, Bayer RL, Igreja Sa IC, Khanal S, Hirsova P, et al. Machine Learning-Based Classification of Abnormal Liver Tissues Using Relative Permittivity. Sensors. 2022; 22(24):9919. https://doi.org/10.3390/s22249919
Chicago/Turabian StyleSamaddar, Poulami, Anup Kumar Mishra, Sunil Gaddam, Mansunderbir Singh, Vaishnavi K. Modi, Keerthy Gopalakrishnan, Rachel L. Bayer, Ivone Cristina Igreja Sa, Shalil Khanal, Petra Hirsova, and et al. 2022. "Machine Learning-Based Classification of Abnormal Liver Tissues Using Relative Permittivity" Sensors 22, no. 24: 9919. https://doi.org/10.3390/s22249919
APA StyleSamaddar, P., Mishra, A. K., Gaddam, S., Singh, M., Modi, V. K., Gopalakrishnan, K., Bayer, R. L., Igreja Sa, I. C., Khanal, S., Hirsova, P., Kostallari, E., Dey, S., Mitra, D., Roy, S., & Arunachalam, S. P. (2022). Machine Learning-Based Classification of Abnormal Liver Tissues Using Relative Permittivity. Sensors, 22(24), 9919. https://doi.org/10.3390/s22249919








