Constant Load Pedaling Exercise Combined with Electrical Muscle Stimulation Leads to an Early Increase in Sweat Lactate Levels
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Experimental Protocol
2.3. Sweat Lactate Threshold Measurements
2.4. Statistical Analysis
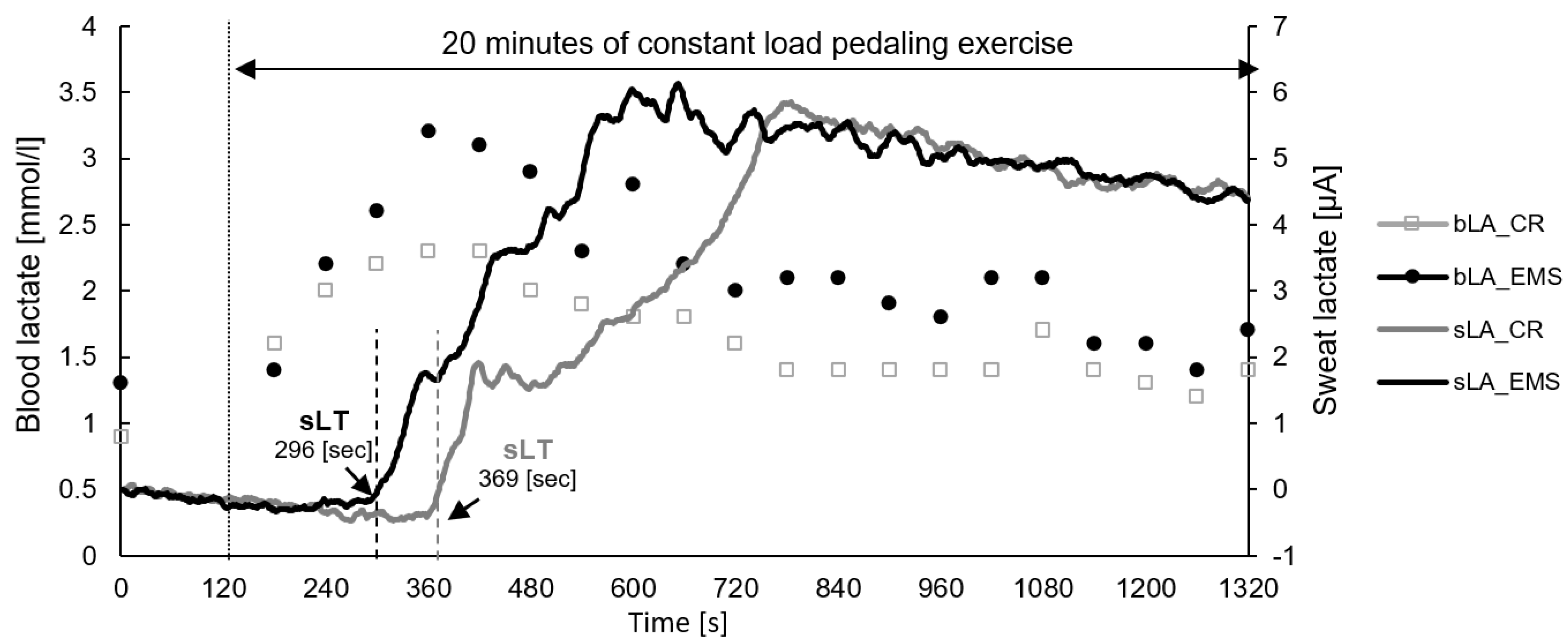
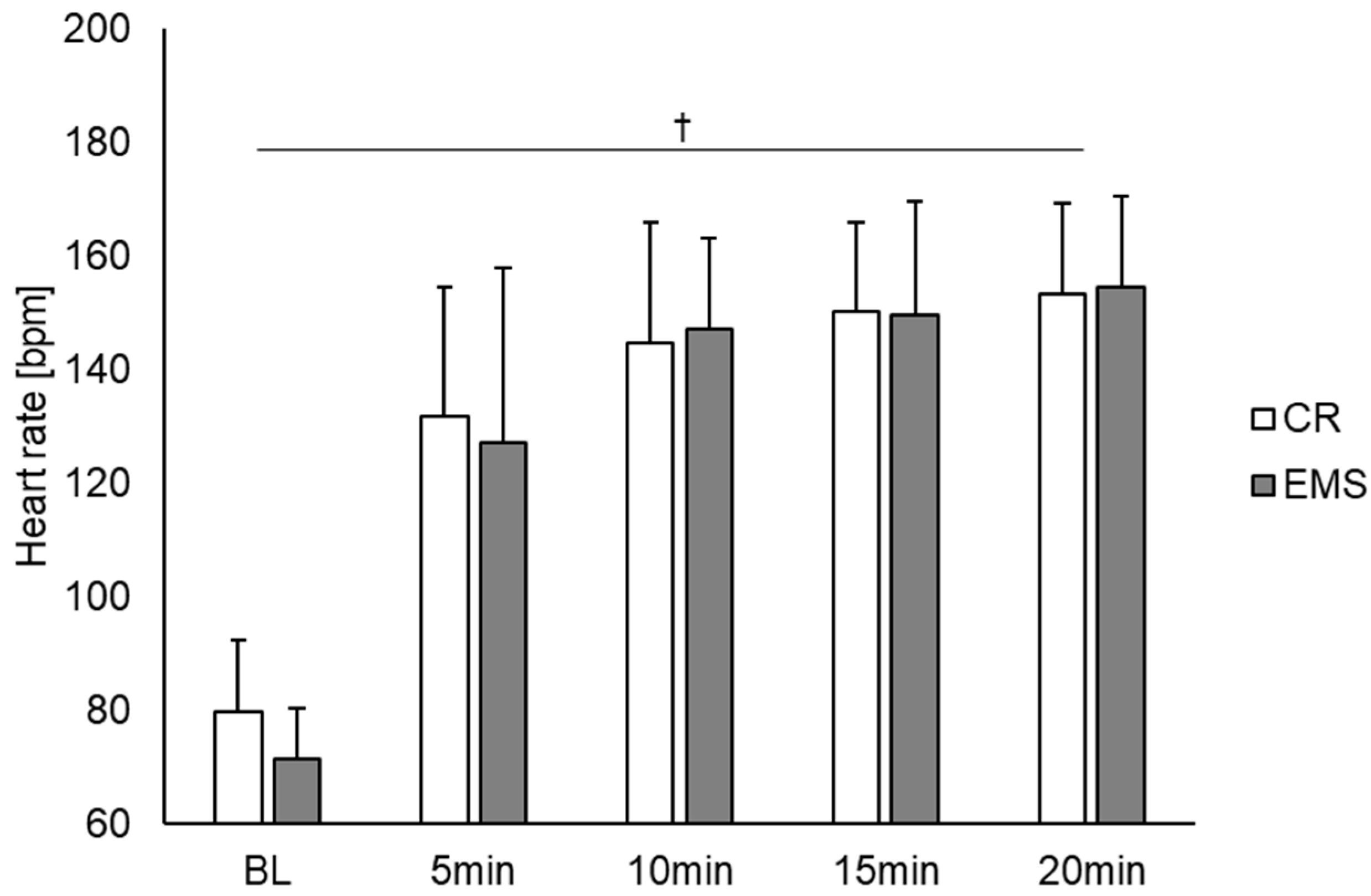
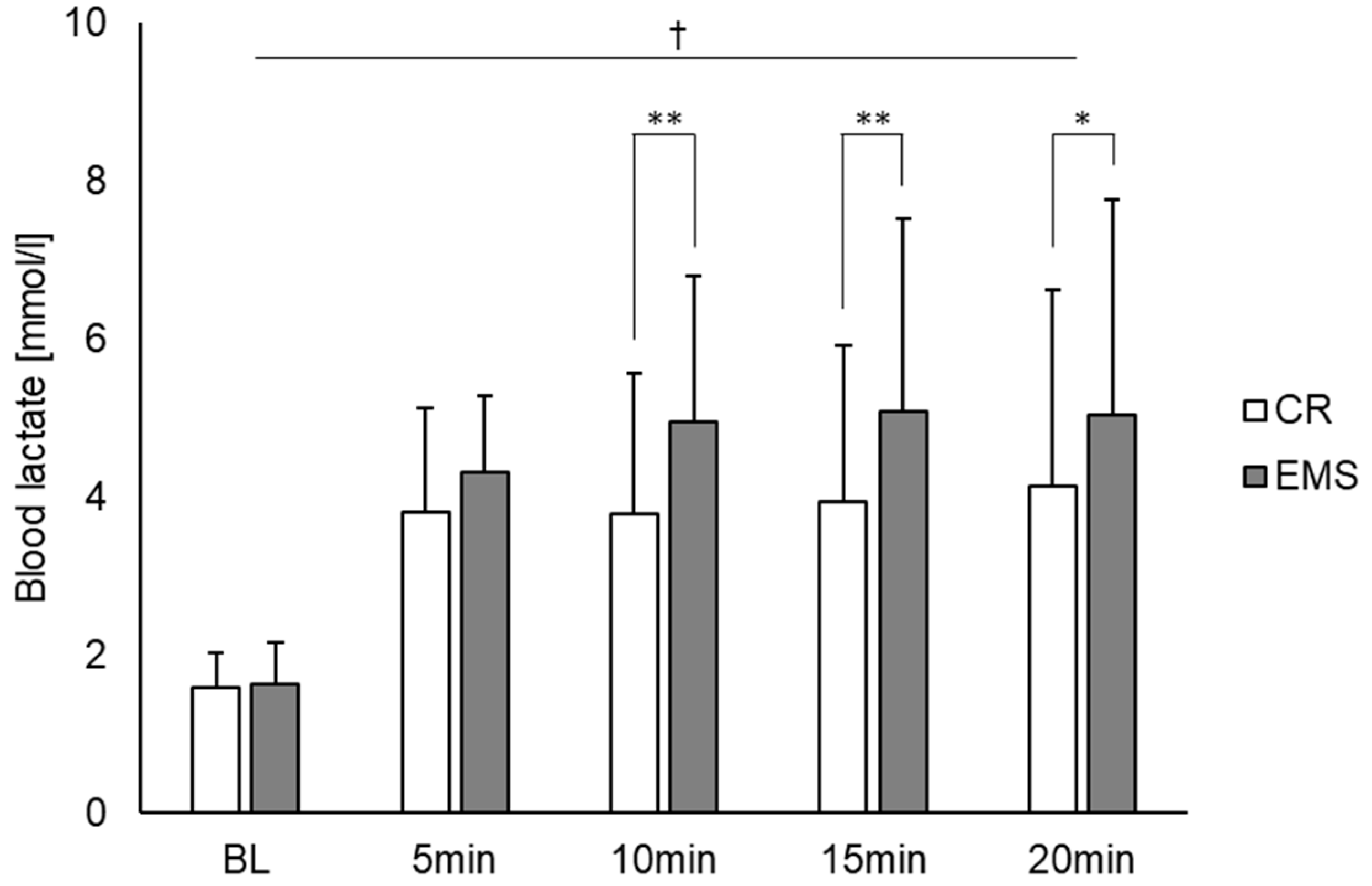
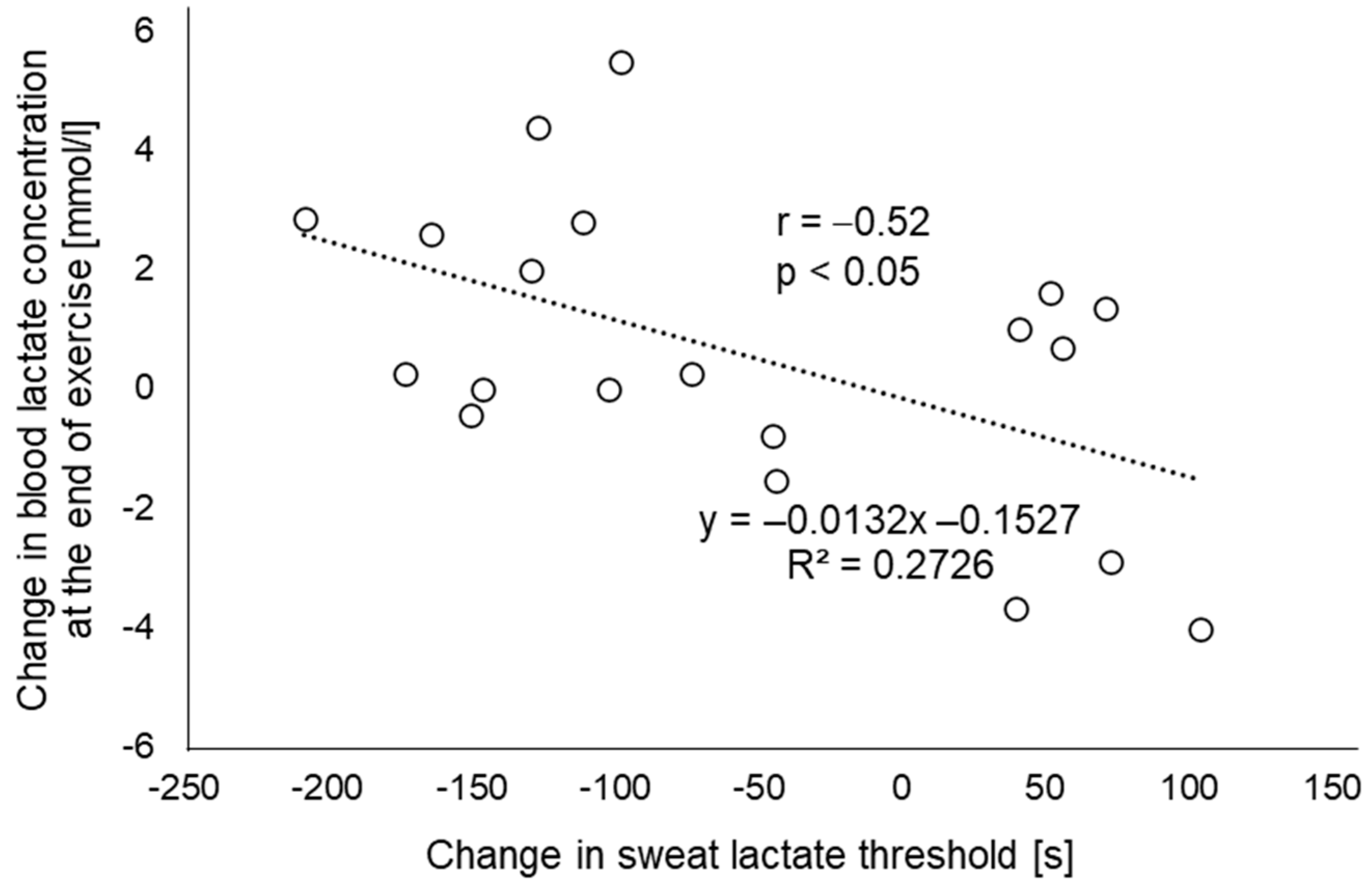
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Malone, J.K.; Blake, C.; Caulfield, B.M. Neuromuscular electrical stimulation during recovery from exercise: A systematic review. J. Strength Cond. Res. 2014, 28, 2478–2506. [Google Scholar] [CrossRef] [PubMed]
- Paillard, T.; Noe, F.; Passelergue, P.; Dupui, P. Electrical Stimulation Superimposed onto Voluntary Muscular Contraction. Sports Med. 2005, 35, 951–966. [Google Scholar] [CrossRef] [PubMed]
- Maffiuletti, N.A. Physiological and methodological considerations for the use of neuromuscular electrical stimulation. Eur. J. Appl. Physiol. 2010, 110, 223–234. [Google Scholar] [CrossRef] [PubMed]
- Fuentes, J.P.; Olivo, S.A.; Magee, D.J.; Gross, D.P. Effectiveness of Interferential Current Therapy in the Management of Musculoskeletal Pain: A Systematic Review and Meta-Analysis. Phys. Ther. 2010, 90, 1219–1238. [Google Scholar] [CrossRef]
- Kristensen, M.G.H.; Busk, H.; Wienecke, T. Neuromuscular Electrical Stimulation Improves Activities of Daily Living Post Stroke: A Systematic Review and Meta-analysis. Arch. Rehabilitation Res. Clin. Transl. 2021, 4, 100167. [Google Scholar] [CrossRef]
- Wang, H.-Y.; Chen, Y.-H.; Kuan, Y.-C.; Huang, S.-W.; Lin, L.-F.; Chen, H.-C. The effectiveness of functional electrical stimulation of the legs in patients with heart failure: A systematic review and meta-analysis of randomized controlled trials. Clin. Rehabilitation 2021, 36, 303–316. [Google Scholar] [CrossRef]
- Sánchez, B.R.; Puche, P.P.; González-Badillo, J.J. Percutaneous Electrical Stimulation in Strength Training: An Update. J. Strength Cond. Res. 2005, 19, 438–448. [Google Scholar] [CrossRef]
- Paillard, T. Combined Application of Neuromuscular Electrical Stimulation and Voluntary Muscular Contractions. Sports Med. 2008, 38, 161–177. [Google Scholar] [CrossRef]
- Watanabe, K.; Taniguchi, Y.; Moritani, T. Metabolic and cardiovascular responses during voluntary pedaling exercise with electrical muscle stimulation. Eur. J. App. Physiol. 2014, 114, 1801–1807. [Google Scholar] [CrossRef]
- Watanabe, K.; Yoshida, T.; Ishikawa, T.; Kawade, S.; Moritani, T. Effect of the Combination of Whole-Body Neuromuscular Electrical Stimulation and Voluntary Exercise on Metabolic Responses in Human. Front. Physiol. 2019, 10, 291. [Google Scholar] [CrossRef]
- Watanabe, K.; Takada, T.; Kawade, S.; Moritani, T. Effect of exercise intensity on metabolic responses on combined application of electrical stimulation and voluntary exercise. Physiol. Rep. 2021, 9, e14758. [Google Scholar] [CrossRef]
- Hui, S.; Ghergurovich, J.M.; Morscher, R.J.; Jang, C.; Teng, X.; Lu, W.; Esparza, L.A.; Reya, T.; Zhan, L.; Guo, J.Y.; et al. Glucose feeds the TCA cycle via circulating lactate. Nature 2017, 551, 115–118. [Google Scholar] [CrossRef]
- Pilegaard, H.; Saltin, B.; Neufer, P.D. Exercise induces transient transcriptional activation of the PGC-1α gene in human skeletal muscle. J. Physiol. 2003, 546 Pt 3, 851–858. [Google Scholar] [CrossRef]
- Hashimoto, T.; Hussien, R.; Oommen, S.; Gohil, K.; Brooks, G.A. Lactate sensitive transcription factor network in L6 cells: Activation of MCT1 and mitochondrial biogenesis. FASEB J. 2007, 21, 2602–2612. [Google Scholar] [CrossRef]
- Perry, C.G.R.; Lally, J.; Holloway, G.P.; Heigenhauser, G.J.F.; Bonen, A.; Spriet, L.L. Repeated transient mRNA bursts precede increases in transcriptional and mitochondrial proteins during training in human skeletal muscle. J. Physiol. 2010, 588 Pt 23, 4795–4810. [Google Scholar] [CrossRef] [PubMed]
- Kitaoka, Y.; Takeda, K.; Tamura, Y.; Hatta, H. Lactate administration increases mRNA expression of PGC-1α and UCP3 in mouse skeletal muscle. Appl. Physiol. Nutr. Metab. 2016, 41, 695–698. [Google Scholar] [CrossRef] [PubMed]
- Fiorenza, M.; Gunnarsson, T.P.; Hostrup, M.; Iaia, F.M.; Schena, F.; Pilegaard, H.; Bangsbo, J. Metabolic stress-dependent regulation of the mitochondrial biogenic molecular response to high-intensity exercise in human skeletal muscle. J. Physiol. 2018, 596, 2823–2840. [Google Scholar] [CrossRef] [PubMed]
- Chodari, L.; Aytemir, M.D.; Vahedi, P.; Alipour, M.; Vahed, S.Z.; Khatibi, S.M.H.; Ahmadian, E.; Ardalan, M.; Eftekhari, A. Targeting Mitochondrial Biogenesis with Polyphenol Compounds. Oxidative Med. Cell Longev. 2021, 2021, 4946711. [Google Scholar] [CrossRef]
- Oliveira, A.N.; Hood, D.A. Exercise is mitochondrial medicine for muscle. Sports Med. Health Sci. 2019, 1, 11–18. [Google Scholar] [CrossRef]
- Yan, Z.; Lira, V.A.; Greene, N.P. Exercise Training-Induced Regulation of Mitochondrial Quality. Exerc. Sport Sci. Rev. 2012, 40, 159–164. [Google Scholar] [CrossRef]
- Lundby, C.; Jacobs, R.A. Adaptations of skeletal muscle mitochondria to exercise training. Exp. Physiol. 2016, 101, 17–22. [Google Scholar] [CrossRef]
- Tobina, T.; Yoshioka, K.; Hirata, A.; Mori, S.; Kiyonaga, A.; Tanaka, H. Peroxisomal proliferator-activated receptor gamma co-activator-1 alpha gene expression increases above the lactate threshold in human skeletal muscle. J. Sports Med. Phys. Fit. 2011, 51, 683–688. [Google Scholar]
- Van Hoovels, K.; Xuan, X.; Cuartero, M.; Gijssel, M.; Swarén, M.; Crespo, G.A. Can Wearable Sweat Lactate Sensors Contribute to Sports Physiology? ACS Sens. 2021, 6, 3496–3508. [Google Scholar] [CrossRef] [PubMed]
- Jia, W.; Bandodkar, A.J.; Valdés-Ramírez, G.; Windmiller, J.R.; Yang, Z.; Ramírez, J.; Chan, G.; Wang, J. Electrochemical Tattoo Biosensors for Real-Time Noninvasive Lactate Monitoring in Human Perspiration. Anal. Chem. 2013, 85, 6553–6560. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.; Emaminejad, S.; Nyein, H.Y.Y.; Challa, S.; Chen, K.; Peck, A.; Fahad, H.M.; Ota, H.; Shiraki, H.; Kiriya, D.; et al. Fully integrated wearable sensor arrays for multiplexed in situ perspiration analysis. Nature 2016, 529, 509–514. [Google Scholar] [CrossRef] [PubMed]
- Imani, S.; Bandodkar, A.J.; Mohan, A.M.V.; Kumar, R.; Yu, S.; Wang, J.; Mercier, P.P. A wearable chemical–electrophysiological hybrid biosensing system for real-time health and fitness monitoring. Nat. Commun. 2016, 7, 11650. [Google Scholar] [CrossRef]
- Seki, Y.; Nakashima, D.; Shiraishi, Y.; Ryuzaki, T.; Ikura, H.; Miura, K.; Suzuki, M.; Watanabe, T.; Nagura, T.; Matsumato, M.; et al. A novel device for detecting anaerobic threshold using sweat lactate during exercise. Sci. Rep. 2021, 11, 4929. [Google Scholar] [CrossRef]
- Gaskill, S.E.; Ruby, B.C.; Walker, A.J.; Sanchez, O.A.; Serfass, R.C.; Leon, A.S. Validity and reliability of combining three methods to determine ventilatory threshold. Med. Sci. Sports Exerc. 2001, 33, 1841–1848. [Google Scholar] [CrossRef]
- Hamada, T.; Kimura, T.; Moritani, T. Selective fatigue of fast motor units after electrically elicited muscle contractions. J. Electromyogr. Kinesiol. 2004, 14, 531–538. [Google Scholar] [CrossRef]
- Gregory, C.M.; Bickel, C.S. Recruitment Patterns in Human Skeletal Muscle During Electrical Stimulation. Phys. Ther. 2005, 85, 358–364. [Google Scholar] [CrossRef]
- Jubeau, M.; Gondin, J.; Martin, A.; Sartorio, A.; Maffiuletti, N.A. Random Motor Unit Activation by Electrostimulation. Endoscopy 2007, 28, 901–904. [Google Scholar] [CrossRef]
- Bickel, C.S.; Gregory, C.M.; Dean, J. Motor unit recruitment during neuromuscular electrical stimulation: A critical appraisal. Eur. J. Appl. Physiol. 2011, 111, 2399–2407. [Google Scholar] [CrossRef]
- Brooks, G.A. Anaerobic threshold: Review of the concept and directions for future research. Med. Sci. Sports Exerc. 1985, 17, 22–34. [Google Scholar] [CrossRef]
- Lamont, L.S. Sweat lactate secretion during exercise in relation to women’s aerobic capacity. J. Appl. Physiol. 1987, 62, 194–198. [Google Scholar] [CrossRef]
- Green, J.M.; Bishop, P.A.; Muir, I.H.; McLester, J.R., Jr.; Heath, H.E. Effects of High and Low Blood Lactate Concentrations on Sweat Lactate Response. Int. J. Sports Med. 2000, 21, 556–560. [Google Scholar] [CrossRef]
- Buono, M.J.; Lee, N.V.L.; Miller, P.W. The relationship between exercise intensity and the sweat lactate excretion rate. J. Physiol. Sci. 2009, 60, 103–107. [Google Scholar] [CrossRef] [PubMed]
- Alvear-Ordenes, I.; García-López, D.; De Paz, J.A.; González-Gallego, J. Sweat Lactate, Ammonia, and Urea in Rugby Players. Endoscopy 2005, 26, 632–637. [Google Scholar] [CrossRef]
- Shiraishi, Y.; Katsumata, Y.; Sadahiro, T.; Azuma, K.; Akita, K.; Isobe, S.; Yashima, F.; Miyamoto, K.; Nishiyama, T.; Tamura, Y.; et al. Real-Time Analysis of the Heart Rate Variability During Incremental Exercise for the Detection of the Ventilatory Threshold. J. Am. Heart Assoc. 2018, 7, e006612. [Google Scholar] [CrossRef] [PubMed]
- Komkova, M.A.; Eliseev, A.A.; Poyarkov, A.A.; Daboss, E.V.; Evdokimov, P.V.; Eliseev, A.A.; Karyakin, A.A. Simultaneous monitoring of sweat lactate content and sweat secretion rate by wearable remote biosensors. Biosens. Bioelectron. 2022, 202, 113970. [Google Scholar] [CrossRef] [PubMed]
- Yüzer, E.; Doğan, V.; Kılıç, V.; Şen, M. Smartphone embedded deep learning approach for highly accurate and automated colorimetric lactate analysis in sweat. Sensors Actuators B Chem. 2022, 371, 132489. [Google Scholar] [CrossRef]
- Spriet, L.L.; Söderlund, K.; Bergström, M.; Hultman, E. Anaerobic energy release in skeletal muscle during electrical stimulation in men. J. Appl. Physiol. 1987, 62, 611–615. [Google Scholar] [CrossRef] [PubMed]
- Bergstrom, M.; Hultman, E. Energy cost and fatigue during intermittent electrical stimulation of human skeletal muscle. J. Appl. Physiol. 1988, 65, 1500–1505. [Google Scholar] [CrossRef] [PubMed]
- Greenhaff, P.; Söderlund, K.; Ren, J.M.; Hultman, E. Energy metabolism in single human muscle fibres during intermittent contraction with occluded circulation. J. Physiol. 1993, 460, 443–453. [Google Scholar] [CrossRef] [PubMed]
- Vanderthommen, M.; Duteil, S.; Wary, C.; Raynaud, J.S.; Leroy-Willig, A.; Crielaard, J.M.; Carlier, P.G. A comparison of voluntary and electrically induced contractions by interleaved 1H- and 31P-NMRS in humans. J. Appl. Physiol. 2003, 94, 1012–1024. [Google Scholar] [CrossRef] [PubMed]
- Morresi, N.; Casaccia, S.; Sorcinelli, M.; Arnesano, M.; Revel, G.M. Analysing performances of Heart Rate Variability measurement through a smartwatch. In Proceedings of the 2020 IEEE International Symposium on Medical Measurements and Applications (MeMeA), Bari, Italy, 1 June–1 July 2020; IEEE: Piscataway, NJ, USA, 2020; pp. 1–6. [Google Scholar]
- Morresi, N.; Casaccia, S.; Revel, G.M. Metrological characterization and signal processing of a wearable sensor for the measurement of heart rate variability. In Proceedings of the 2021 IEEE International Symposium on Medical Measurements and Applications (MeMeA), Lausanne, Switzerland, 23–25 June 2021; IEEE: Piscataway, NJ, USA, 2021; pp. 1–6. [Google Scholar]

| Mean (SD) | Range | |
|---|---|---|
| Age (years) | 20.7 (0.8) | 19–22 |
| Height (cm) | 174.0 (5.4) | 161.0–184.0 |
| Weight (kg) | 66.8 (9.0) | 46.8–91.5 |
| BMI (kg/m2) | 22.0 (2.2) | 18.1–27.0 |
| Body fat ratio (%) | 15.4 (4.3) | 8.6–23.8 |
| Fat mass (kg) | 10.5 (4.0) | 4.0–20.1 |
| Lean body mass (kg) | 56.4 (6.2) | 42.8–71.4 |
| Muscle mass (kg) | 53.4 (5.9) | 40.5–67.7 |
| Total body water (kg) | 40.5 (6.5) | 30.3–57.6 |
| Body water (%) | 59.7 (4.7) | 49.6–65.9 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sawada, T.; Okawara, H.; Nakashima, D.; Ikeda, K.; Nagahara, J.; Fujitsuka, H.; Hoshino, S.; Maeda, Y.; Katsumata, Y.; Nakamura, M.; et al. Constant Load Pedaling Exercise Combined with Electrical Muscle Stimulation Leads to an Early Increase in Sweat Lactate Levels. Sensors 2022, 22, 9585. https://doi.org/10.3390/s22249585
Sawada T, Okawara H, Nakashima D, Ikeda K, Nagahara J, Fujitsuka H, Hoshino S, Maeda Y, Katsumata Y, Nakamura M, et al. Constant Load Pedaling Exercise Combined with Electrical Muscle Stimulation Leads to an Early Increase in Sweat Lactate Levels. Sensors. 2022; 22(24):9585. https://doi.org/10.3390/s22249585
Chicago/Turabian StyleSawada, Tomonori, Hiroki Okawara, Daisuke Nakashima, Kaito Ikeda, Joji Nagahara, Haruki Fujitsuka, Sosuke Hoshino, Yuta Maeda, Yoshinori Katsumata, Masaya Nakamura, and et al. 2022. "Constant Load Pedaling Exercise Combined with Electrical Muscle Stimulation Leads to an Early Increase in Sweat Lactate Levels" Sensors 22, no. 24: 9585. https://doi.org/10.3390/s22249585
APA StyleSawada, T., Okawara, H., Nakashima, D., Ikeda, K., Nagahara, J., Fujitsuka, H., Hoshino, S., Maeda, Y., Katsumata, Y., Nakamura, M., & Nagura, T. (2022). Constant Load Pedaling Exercise Combined with Electrical Muscle Stimulation Leads to an Early Increase in Sweat Lactate Levels. Sensors, 22(24), 9585. https://doi.org/10.3390/s22249585







