Planar Interdigitated Aptasensor for Flow-Through Detection of Listeria spp. in Hydroponic Lettuce Growth Media
Abstract
:1. Introduction
2. Experimental
2.1. Materials, Reagents and Equipment
2.2. Bacteria Strains and Culture
2.3. Electrochemical Characterization
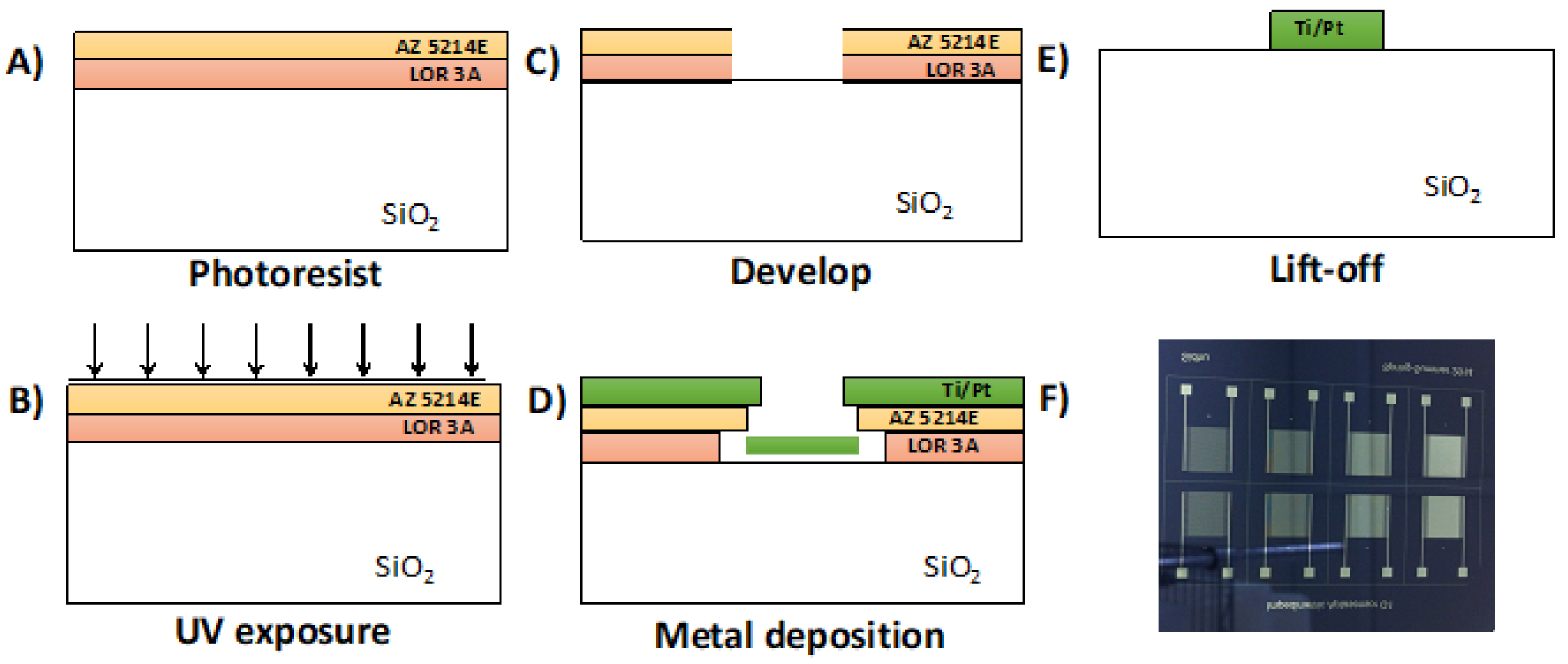
2.4. Pt-IME Fabrication Procedure
2.5. In Silico Model of Pt-IME Capacitance
2.6. Biofunctionalization of Pt-IMEs with Aptamers
2.7. Protocol for Bacteria Detection
2.8. Hysteresis Testing
2.9. Analysis of Hydroponic Water
2.10. Statistical Analysis and Portfolio Analysis
3. Results and Discussion
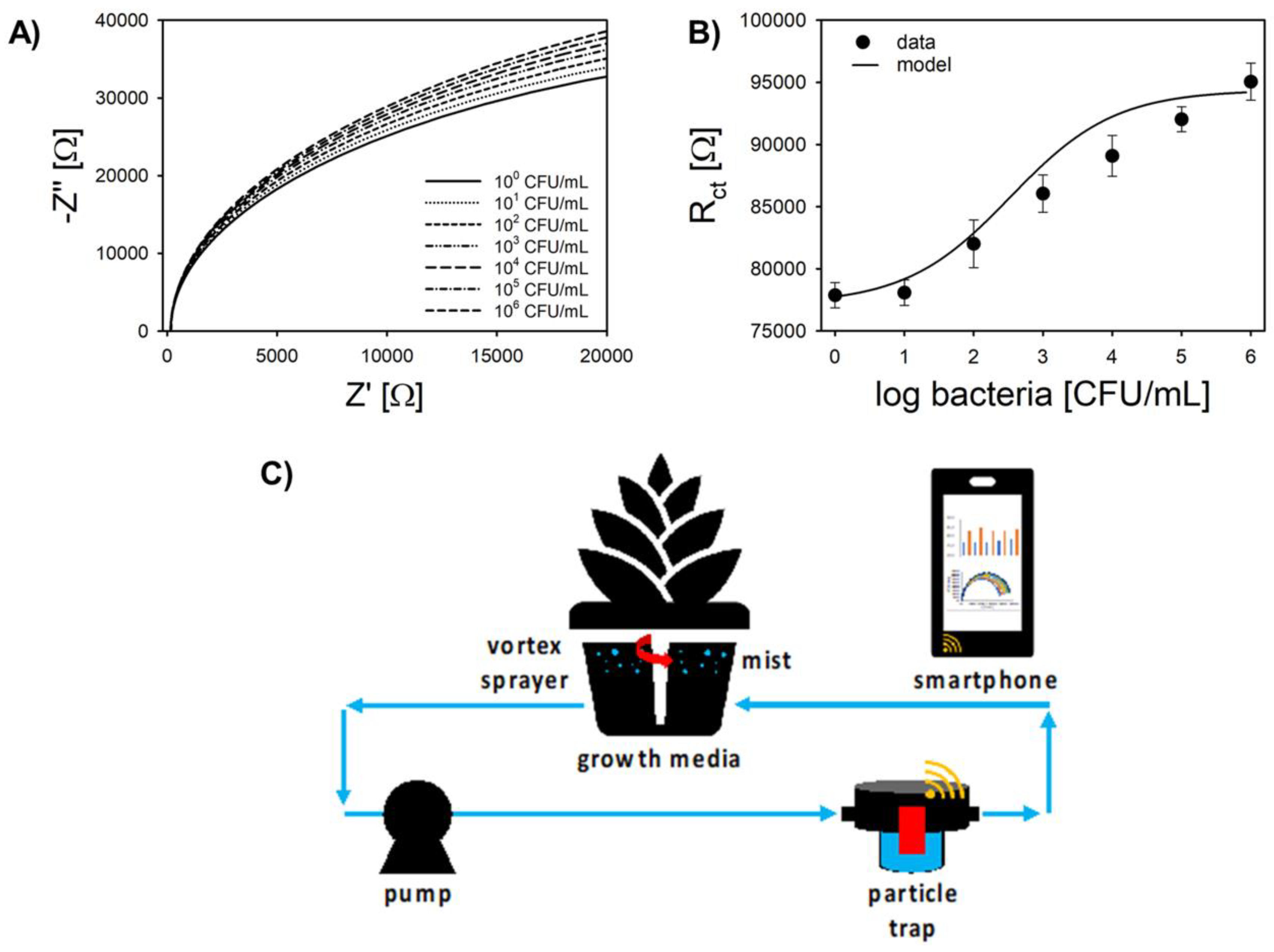
3.1. In Silico Pt-IME Model
3.2. Electrochemical Characterization
3.3. Pt-IME Biofunctionalization
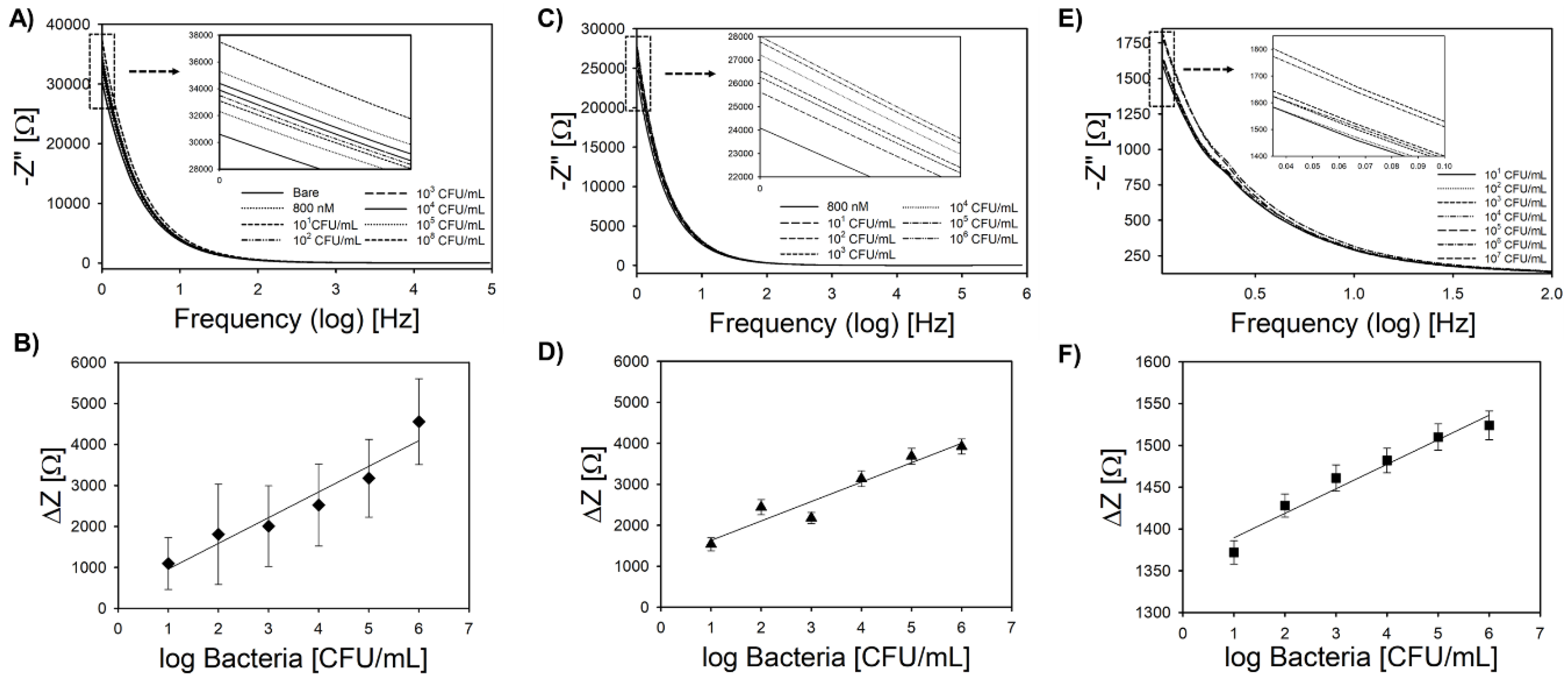
3.4. Bacteria Sensing
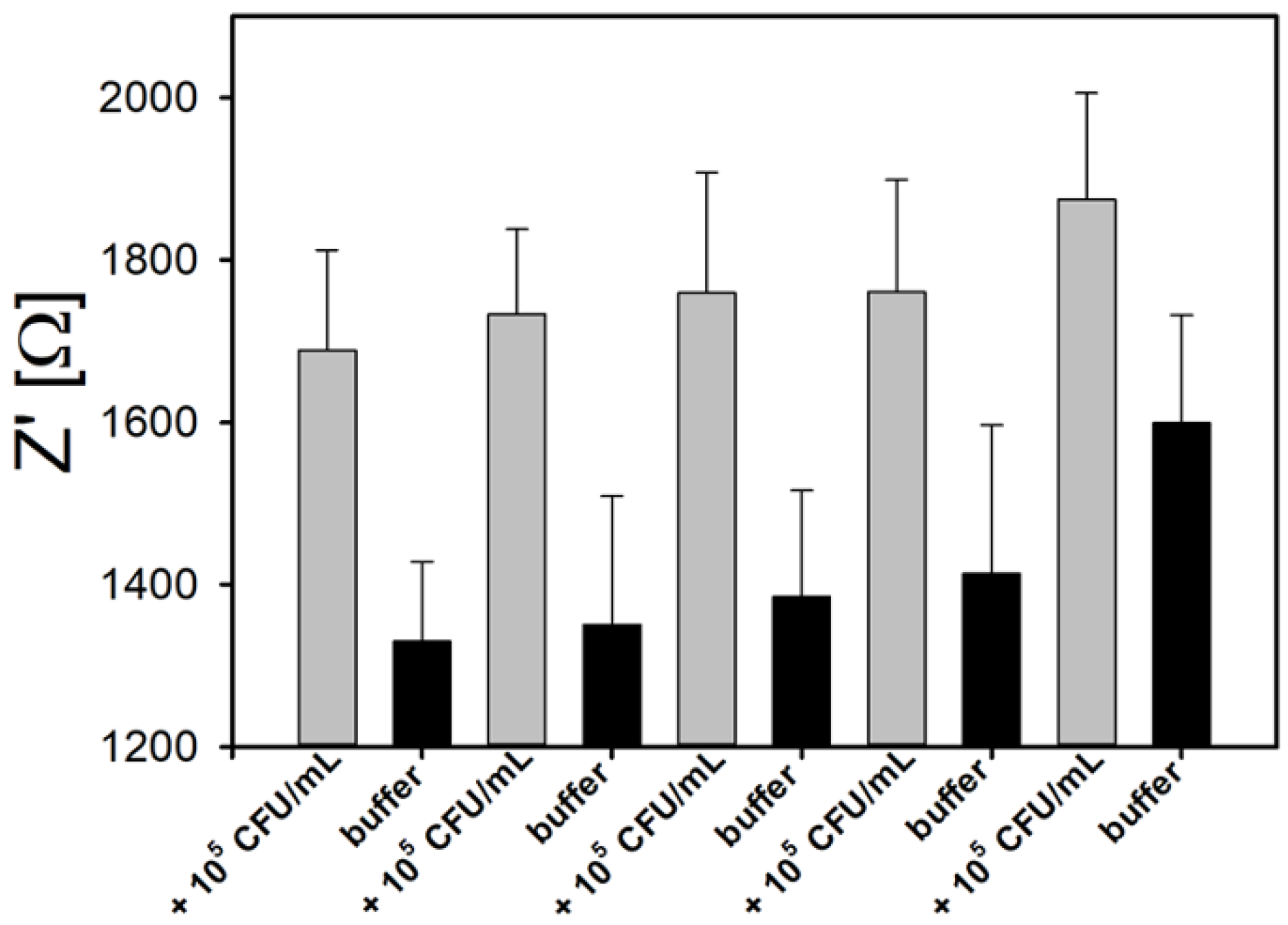
3.5. Hysteresis and Reusability
3.6. Analysis of Hydroponic Water in Particle Trap Filter
3.7. Comparison of Listeria Sensors in the Literature
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Nyachuba, D.G. Foodborne illness: Is it on the rise? Nutr. Rev. 2010, 68, 257–269. [Google Scholar] [CrossRef]
- Hoffmann, S.; Anekwe, T. Making Sense of Recent Cost-of-Foodborne-Illness Estimates; CreateSpace Independent Publishing Platform: Scotts Valley, CA, USA, 2013. [Google Scholar]
- Castell-Perez, E.; Gomes, C.; Tahtouh, J.; Moreira, R.; McLamore, E.S.; Knowles, H.S. Food Processing and Waste Within the Nexus Framework. Curr. Sustain. Renew. Energy Rep. 2017, 4, 99–108. [Google Scholar] [CrossRef]
- Holvoet, K.; De Keuckelaere, A.; Sampers, I.; Van Haute, S.; Stals, A.; Uyttendaele, M. Quantitative study of cross-contamination with Escherichia coli, E. coli O157, MS2 phage and murine norovirus in a simulated fresh-cut lettuce wash process. Food Control 2014, 37, 218–227. [Google Scholar] [CrossRef]
- Jensen, D.A.; Friedrich, L.M.; Harris, L.J.; Danyluk, M.D.; Schaffner, D.W. Cross contamination of Escherichia coli O157:H7 between lettuce and wash water during home-scale washing. Food Microbiol. 2015, 46, 428–433. [Google Scholar] [CrossRef]
- Uyttendaele, M.; Jaykus, L.-A.; Amoah, P.; Chiodini, A.; Cunliffe, D.; Jacxsens, L.; Holvoet, K.; Korsten, L.; Lau, M.; McClure, P.; et al. Microbial Hazards in Irrigation Water: Standards, Norms, and Testing to Manage Use of Water in Fresh Produce Primary Production. Compr. Rev. Food Sci. Food Saf. 2015, 14, 336–356. [Google Scholar] [CrossRef]
- U.S. Environment Protection Agency (EPA). National Water Quality Inventory Report to Congress: EPA 841-R-16-011; EPA: Washington, DC, USA, 2017.
- Marks, A.B. The Risks We Are Willing to Eat: Food Imports and Safety. Harv. Hournal Legis. 2015, 52, 125–172. [Google Scholar]
- U.S. Food and Drug Administration (FDA). FSMA Facts: Background on the FDA Food Safety Modernization Act (FMSA); U.S. Food and Drug Administration: Aurora, IL, USA, 2011; pp. 1–3. Available online: https://www.fda.gov/food/food-safety-modernization-act-fsma/background-fda-food-safety-modernization-act-fsma (accessed on 7 September 2020).
- U.S. Food and Drug Administration (FDA). FSMA Final Rule on Produce Safety; U.S. Food and Drug Administration: Aurora, IL, USA, 2020. Available online: https://www.fda.gov/food/food-safety-modernization-act-fsma/fsma-final-rule-produce-safety (accessed on 21 June 2020).
- Buchanan, R.L.; Gorris, L.G.M.; Hayman, M.M.; Jackson, T.C.; Whiting, R.C. A review of Listeria monocytogenes: An update on outbreaks, virulence, dose-response, ecology, and risk assessments. Food Control 2017, 75, 1–13. [Google Scholar] [CrossRef]
- Kyere, E.O.; Foong, G.; Palmer, J.; Wargent, J.J.; Fletcher, G.C.; Flint, S. Rapid attachment of Listeria monocytogenes to hydroponic and soil grown lettuce leaves. Food Control 2019, 101, 77–80. [Google Scholar] [CrossRef]
- Thakur, M.; Asrani, R.K.; Patial, V. Chapter 6-Listeria monocytogenes: A Food-Borne Pathogen. In Foodborne Diseases; Holban, A.M., Grumezescu, A.M., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 157–192. [Google Scholar] [CrossRef]
- Fox, E.M.; Wall, P.G.; Fanning, S. Control of Listeria species food safety at a poultry food production facility. Food Microbiol. 2015, 51, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Sauders, B.D.; Overdevest, J.; Fortes, E.; Windham, K.; Schukken, Y.; Lembo, A.; Wiedmann, M. Diversity of Listeria species in urban and natural environments. Appl. Environ. Microbiol. 2012, 78, 4420–4433. [Google Scholar] [CrossRef] [Green Version]
- Zhou, H.; Gao, Z.; Luo, G.; Han, L.; Sun, S.; Wang, H. Determination of Listeria monocytogenes in Milk Samples by Signal Amplification Quartz Crystal Microbalance Sensor. Anal. Lett. 2010, 43, 312–322. [Google Scholar] [CrossRef]
- Sidhu, R.; Rong, Y.; Vanegas, D.; Claussen, J.; McLamore, E.; Gomes, C. Impedance biosensor for the rapid detection of Listeria spp. based on aptamer functionalized Pt-interdigitated microelectrodes array. SPIE Commer. Sci. Sens. Imaging 2013, 9863, 98630F. [Google Scholar]
- Geng, T.; Morgan, M.T.; Bhunia, A.K. Detection of low levels of Listeria monocytogenes cells by using a fiber-optic immunosensor. Appl. Environ. Microbiol. 2004, 70, 6138–6146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seo, S.M.; Cho, I.H.; Kim, J.H.; Jeon, J.W.; Oh, E.G.; Yu, H.S.; Shin, S.B.; Lee, H.J.; Paek, S.H. An ELISA-on-a-Chip Biosensor System for Early Screening of Listeria monocytogenes in Contaminated Food Products. Bull. Korean Chem. Soc. 2009, 30, 2993–2998. [Google Scholar] [CrossRef] [Green Version]
- Alhogail, S.; Suaifan, G.A.R.Y.; Zourob, M. Rapid colorimetric sensing platform for the detection of Listeria monocytogenes foodborne pathogen. Biosens. Bioelectron. 2016, 86, 1061–1066. [Google Scholar] [CrossRef]
- Ohk, S.H.; Koo, O.K.; Sen, T.; Yamamoto, C.M.; Bhunia, A.K. Antibody–aptamer functionalized fibre-optic biosensor for specific detection of Listeria monocytogenes from food. J. Appl. Microbiol. 2010, 109, 808–817. [Google Scholar] [CrossRef]
- Li, H.; Bashir, R. Dielectrophoretic separation and manipulation of live and heat-treated cells of Listeria on microfabricated devices with interdigitated electrodes. Sens. Actuators B Chem. 2002, 86, 215–221. [Google Scholar] [CrossRef]
- Sharma, H.; Mutharasan, R. Rapid and sensitive immunodetection of Listeria monocytogenes in milk using a novel piezoelectric cantilever sensor. Biosens. Bioelectron. 2013, 45, 158–162. [Google Scholar] [CrossRef]
- Kyere, E.O.; Palmer, J.; Wargent, J.J.; Fletcher, G.C.; Flint, S. Colonisation of lettuce by Listeria Monocytogenes. Int. J. Food Sci. Technol. 2019, 54, 14–24. [Google Scholar] [CrossRef] [Green Version]
- Self, J.; Conrad, A.; Stroika, S.; Jackson, A.; Whitlock, L.; Jackson, K.; Beal, J.; Wellman, A.; Fatica, M.; Bidol, S.; et al. Multistate Outbreak of Listeriosis Associated with Packaged Leafy Green Salads, United States and Canada, 2015–2016. Emerg. Infect. Dis. J. 2019, 25, 1461. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). Multistate Outbreak of E. coli O157:H7 Infections Linked to Romaine Lettuce. 2018. Available online: https://www.cdc.gov/ecoli/2018/o157h7-04-18/index.html (accessed on 21 June 2020).
- Wells, H.F.; Bond, J.K. Vegetables and Pulses Outlook; United States Department of Agriculture: Washington, DC, USA, 2016.
- Hills, K.D.; Oliveira, D.A.; Cavallaro, N.D.; Gomes, C.L.; McLamore, E.S. Actuation of chitosan-aptamer nanobrush borders for pathogen sensing. Analyst 2018, 143, 1650–1661. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jenkins, D.M.; Lee, B.E.; Jun, S.; Reyes-De-Corcuera, J.; McLamore, E.S. ABE-Stat, a Fully Open-Source and Versatile Wireless Potentiostat Project Including Electrochemical Impedance Spectroscopy. J. Electrochem. Soc. 2019, 166, B3056–B3065. [Google Scholar] [CrossRef]
- Omac, B.; Moreira, R.G.; Castillo, A.; Castell-Perez, E. Growth of Listeria monocytogenes and Listeria innocua on fresh baby spinach leaves: Effect of storage temperature and natural microflora. Postharvest Biol. Technol. 2015, 100, 41–51. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration (FDA). Bioteriological Analytical Manual (BAM); U.S. Food and Drug Administration: Aurora, IL, USA, 2020. Available online: https://www.fda.gov/food/laboratory-methods-food/bacteriological-analytical-manual-bam (accessed on 7 September 2020).
- Vanegas, D.C.; Patiño, L.; Mendez, C.; de Oliveira, D.A.; Torres, A.M.; Gomes, C.L.; McLamore, E.S. Laser Scribed Graphene Biosensor for Detection of Biogenic Amines in Food Samples Using Locally Sourced Materials. Biosensors 2018, 8, 42. [Google Scholar] [CrossRef] [Green Version]
- Burrs, S.L.; Bhargava, M.; Sidhu, R.; Kiernan-Lewis, J.; Gomes, C.; Claussen, J.C.; McLamore, E.S. A paper based graphene-nanocauliflower hybrid composite for point of care biosensing. Biosens. Bioelectron. 2016, 85, 479–487. [Google Scholar] [CrossRef] [Green Version]
- Rong, Y.; Padron, A.V.; Hagerty, K.J.; Nelson, N.; Chi, S.; Keyhani, N.O.; Katz, J.; Datta, S.P.A.; Gomes, C.; McLamore, E.S. Post hoc support vector machine learning for impedimetric biosensors based on weak protein–ligand interactions. Analyst 2018, 143, 2066–2075. [Google Scholar] [CrossRef] [Green Version]
- Vanegas, D.C.; Taguchi, M.; Chaturvedi, P.; Burrs, S.; Tan, M.; Yamaguchi, H.; McLamore, E.S. A comparative study of carbon–platinum hybrid nanostructure architecture for amperometric biosensing. Analyst 2014, 139, 660–667. [Google Scholar] [CrossRef]
- Vanegas, D.C.; Clark, G.; Cannon, A.E.; Roux, S.; Chaturvedi, P.; McLamore, E.S. A self-referencing biosensor for real-time monitoring of physiological ATP transport in plant systems. Biosens. Bioelectron. 2015, 74, 37–44. [Google Scholar] [CrossRef]
- Min, J.; Baeumner, A.J. Characterization and Optimization of Interdigitated Ultramicroelectrode Arrays as Electrochemical Biosensor Transducers. Electroanalysis 2004, 16, 724–729. [Google Scholar] [CrossRef]
- Jun, L.Q.; Djaswadi, G.W.; Hawari, H.F.; Zakariya, M.A.B. Simulation of Interdigitated Electrodes (IDEs) Geometry Using COMSOL Multiphysics. In Proceedings of the 2018 International Conference on Intelligent and Advanced System (ICIAS), Kuala Lumpur, Malaysia, 13–14 August 2018; pp. 1–6. [Google Scholar]
- Oberländer, J.; Jildeh, Z.B.; Kirchner, P.; Wendeler, L.; Bromm, A.; Iken, H.; Wagner, P.; Keusgen, M.; Schöning, M.J. Study of Interdigitated Electrode Arrays Using Experiments and Finite Element Models for the Evaluation of Sterilization Processes. Sensors 2015, 15, 26115–26127. [Google Scholar] [CrossRef] [Green Version]
- GeneLink. Conjugation and Surface Attachment Protocol. 2020. Available online: http://www.genelink.com/oligo_modifications_reference/OMR_mod_category_design.asp?mod_sp_cat_id=2 (accessed on 12 September 2020).
- Migliorini, E.; Weidenhaupt, M.; Picart, C. Practical guide to characterize biomolecule adsorption on solid surfaces (Review). Biointerphases 2018, 13, 06d303. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marhaenanto, B.; Soni, P.; Salakhe, V.M. Development of an internet-based greenhouse control system. Int. Agric. Eng. J. 2013, 22, 72–83. [Google Scholar]
- McLamore, E.S.; Jaroch, D.; Chatni, M.R.; Porterfield, D.M. Self-referencing optrodes for measuring spatially resolved, real-time metabolic oxygen flux in plant systems. Planta 2010, 232, 1087–1099. [Google Scholar] [CrossRef] [PubMed]
- McLamore, E.S.; Palit Austin Datta, S.; Morgan, V.; Cavallaro, N.; Kiker, G.; Jenkins, D.M.; Rong, Y.; Gomes, C.; Claussen, J.; Vanegas, D.; et al. SNAPS: Sensor Analytics Point Solutions for Detection and Decision Support Systems. Sensors 2019, 19, 4935. [Google Scholar] [CrossRef] [Green Version]
- McNaught, A.D.; Wilkinson, A. Compendium of Chemical Terminology: IUPAC Recommendations; Blackwell Science: Hoboken, NJ, USA, 1997. [Google Scholar]
- Tolba, M.; Ahmed, M.U.; Tlili, C.; Eichenseher, F.; Loessner, M.J.; Zourob, M. A bacteriophage endolysin-based electrochemical impedance biosensor for the rapid detection of Listeria cells. Analyst 2012, 137, 5749–5756. [Google Scholar] [CrossRef]
- Goepel, K.D. Implementation of an Online Software Tool for the Analytic Hierarchy Process (AHP-OS). Int. J. Anal. Hierarchy Process 2018, 10, 1–5. [Google Scholar]
- Lai, E.; Lundie, S.; Ashbolt, N.J. Review of multi-criteria decision aid for integrated sustainability assessment of urban water systems. Urban Water J. 2008, 5, 315–327. [Google Scholar] [CrossRef]
- Varshney, M.; Li, Y. Interdigitated array microelectrode based impedance biosensor coupled with magnetic nanoparticle–Antibody conjugates for detection of Escherichia coli O157:H7 in food samples. Biosens. Bioelectron. 2007, 22, 2408–2414. [Google Scholar] [CrossRef]
- Varshney, M.; Li, Y.; Srinivasan, B.; Tung, S. A label-free, microfluidics and interdigitated array microelectrode-based impedance biosensor in combination with nanoparticles immunoseparation for detection of Escherichia coli O157:H7 in food samples. Sens. Actuators B Chem. 2007, 128, 99–107. [Google Scholar] [CrossRef]
- Bäcker, M.; Kramer, F.; Huck, C.; Poghossian, A.; Bratov, A.; Abramova, N.; Schöning, M.J. Planar and 3D interdigitated electrodes for biosensing applications: The impact of a dielectric barrier on the sensor properties. Phys. Status Solidi A 2014, 211, 1357–1363. [Google Scholar] [CrossRef]
- Dimitrov, G.V.; Disselhorst-Klug, C.; Dimitrova, N.A.; Schulte, E.; Rau, G. Simulation analysis of the ability of different types of multi-electrodes to increase selectivity of detection and to reduce cross-talk. J. Electromyogr. Kinesiol. 2003, 13, 125–138. [Google Scholar] [CrossRef]
- Johnson, W.A.; Warne, L.K. Electrophysics of micromechanical comb actuators. J. Microelectromech. Syst. 1995, 4, 49–59. [Google Scholar] [CrossRef]
- Massoud, Y.; Kawa, J.; MacMillen, D.; White, J. Modeling and analysis of differential signaling for minimizing inductive crosstalk. In Proceedings of the 38th Design Automation Conference (IEEE Cat. No.01CH37232), Las Vegas, NV, USA, 22–22 June 2001; pp. 804–809. [Google Scholar]
- Mallén, M.; Díaz-González, M.; Bonilla, D.; Salvador, J.P.; Marco, M.P.; Baldi, A.; Fernández-Sánchez, C. Reusable conductimetric array of interdigitated microelectrodes for the readout of low-density microarrays. Anal. Chim. Acta 2014, 832, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Armitage, N.P.; Briman, M.; Grüner, G. Charge transfer and charge transport on the double helix. Phys. Status Solidi B 2004, 241, 69–75. [Google Scholar] [CrossRef] [Green Version]
- Xu, Y.; Ye, X.; Yang, L.; He, P.; Fang, Y. Impedance DNA Biosensor Using Electropolymerized Polypyrrole/Multiwalled Carbon Nanotubes Modified Electrode. Electroanalysis 2006, 18, 1471–1478. [Google Scholar] [CrossRef]
- Rabe, M.; Verdes, D.; Seeger, S. Understanding protein adsorption phenomena at solid surfaces. Adv. Colloid Interface Sci. 2011, 162, 87–106. [Google Scholar] [CrossRef] [Green Version]
- Vroman, L.; Adams, A.L. Identification of adsorbed protein films by exposure to antisera and water vapor. J. Biomed. Mater. Res. 1969, 3, 669–671. [Google Scholar] [CrossRef]
- Yang, L.; Li, Y.; Erf, G.F. Interdigitated Array Microelectrode-Based Electrochemical Impedance Immunosensor for Detection of Escherichia coli O157:H7. Anal. Chem. 2004, 76, 1107–1113. [Google Scholar] [CrossRef]
- Hu, X.; Mu, L.; Zhou, Q.; Wen, J.; Pawliszyn, J. ssDNA Aptamer-Based Column for Simultaneous Removal of Nanogram Per Liter Level of Illicit and Analgesic Pharmaceuticals in Drinking Water. Environ. Sci. Technol. 2011, 45, 4890–4895. [Google Scholar] [CrossRef]
- Dastider, S.G.; Barizuddin, S.; Dweik, M.; Almasri, M. A micromachined impedance biosensor for accurate and rapid detection of E. coli O157:H7. RSC Adv. 2013, 3, 26297–26306. [Google Scholar] [CrossRef]
- Radke, S.M.; Alocilja, E.C. A high density microelectrode array biosensor for detection of E. coli O157:H7. Biosens. Bioelectron. 2005, 20, 1662–1667. [Google Scholar] [CrossRef] [PubMed]
- McKeague, M.; DeRosa, M.C. Challenges and Opportunities for Small Molecule Aptamer Development. J. Nucleic Acids 2012, 2012, 748913. [Google Scholar] [CrossRef] [PubMed]
- Li, N. Reversible Regulation of Aptamer Activity with Effector-Responsive Hairpin Oligonucleotides. J. Lab. Autom. 2013, 18, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Morgan, V.; Casso-Hartmann, L.; Bahamon-Pinzon, D.; McCourt, K.; Hjort, R.G.; Bahramzadeh, S.; Velez-Torres, I.; McLamore, E.; Gomes, C.; Alocilja, E.C.; et al. Sensor-as-a-Service: Convergence of Sensor Analytic Point Solutions (SNAPS) and Pay-A-Penny-Per-Use (PAPPU) Paradigm as a Catalyst for Democratization of Healthcare in Underserved Communities. Diagnostics 2020, 10, 22. [Google Scholar] [CrossRef] [Green Version]
- Zhao, X.; Lin, C.W.; Wang, J. Advances in Rapid Detection Methods for Foodborne Pathogens. J. Microbiol. Biotechnol. 2014, 24, 297–312. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, S.-H.; Ahn, J.-Y.; Lee, K.-A.; Um, H.-J.; Sekhon, S.S.; Sun Park, T.; Min, J.; Kim, Y.-H. Analytical bioconjugates, aptamers, enable specific quantitative detection of Listeria monocytogenes. Biosens. Bioelectron. 2015, 68, 272–280. [Google Scholar] [CrossRef] [PubMed]
- Boulade, M.; Morlay, A.; Piat, F.; Roupioz, Y.; Livache, T.; Charette, P.G.; Canva, M.; Leroy, L. Early detection of bacteria using SPR imaging and event counting: Experiments with Listeria monocytogenes and Listeria innocua. RSC Adv. 2019, 9, 15554–15560. [Google Scholar] [CrossRef] [Green Version]
- Wang, R.; Ruan, C.; Kanayeva, D.; Lassiter, K.; Li, Y. TiO2 Nanowire Bundle Microelectrode Based Impedance Immunosensor for Rapid and Sensitive Detection of Listeria monocytogenes. Nano Lett. 2008, 8, 2625–2631. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Wang, D.; Cai, G.; Xiong, Y.; Li, Y.; Wang, M.; Huo, H.; Lin, J. Fast and sensitive detection of foodborne pathogen using electrochemical impedance analysis, urease catalysis and microfluidics. Biosens. Bioelectron. 2016, 86, 770–776. [Google Scholar] [CrossRef]
- Bonah, E.; Huang, X.; Yi, R.; Aheto, J.H.; Osae, R.; Golly, M. Electronic nose classification and differentiation of bacterial foodborne pathogens based on support vector machine optimized with particle swarm optimization algorithm. J. Food Process. Eng. 2019, 42, e13236. [Google Scholar] [CrossRef]
- Etayash, H.; Jiang, K.; Thundat, T.; Kaur, K. Impedimetric Detection of Pathogenic Gram-Positive Bacteria Using an Antimicrobial Peptide from Class IIa Bacteriocins. Anal. Chem. 2014, 86, 1693–1700. [Google Scholar] [CrossRef] [PubMed]
- Gómez, R.; Bashir, R.; Bhunia, A.K. Microscale electronic detection of bacterial metabolism. Sens. Actuators B Chem. 2002, 86, 198–208. [Google Scholar] [CrossRef]
- Chen, A.; Yang, S. Replacing antibodies with aptamers in lateral flow immunoassay. Biosens. Bioelectron. 2015, 71, 230–242. [Google Scholar] [CrossRef] [PubMed]
- Hidding, J. A therapeutic Battle: Antibodies vs. Aptamers. Nanosci. Master Program 2017, 1–20. Available online: https://www.rug.nl/research/zernike/education/topmasternanoscience/programme-documents/ns190-papers/ns_190_hidding-atherapeuticbattleantibodiesvs.aptamers.pdf (accessed on 12 September 2020).
- Zhang, Y.; Lai, B.S.; Juhas, M. Recent Advances in Aptamer Discovery and Applications. Molecules 2019, 24, 941. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- U.S. Department of Agriculture Food Safety and Inspection Services (USDA FSIS). Verification Activities for the Listeria monocytogenes (Lm) Regulation and the Ready-to-Eat (RTE) Sampling Program; U.S. Department of Agriculture: Washington, DC, USA, 2014. Available online: https://www.fsis.usda.gov/wps/wcm/connect/b8cd03ed-222c-4cef-ad92-3647e3be6c53/10240.4.pdf?MOD=AJPERES (accessed on 7 September 2020).
- Radhakrishnan, R.; Jahne, M.; Rogers, S.; Suni, I.I. Detection of Listeria Monocytogenes by Electrochemical Impedance Spectroscopy. Electroanalysis 2013, 25, 2231–2237. [Google Scholar] [CrossRef]
- Clime, L.; Hoa, X.D.; Corneau, N.; Morton, K.J.; Luebbert, C.; Mounier, M.; Brassard, D.; Geissler, M.; Bidawid, S.; Farber, J.; et al. Microfluidic filtration and extraction of pathogens from food samples by hydrodynamic focusing and inertial lateral migration. Biomed. Microdevices 2015, 17, 17. [Google Scholar] [CrossRef] [PubMed]
- Kanayeva, D.A.; Wang, R.; Rhoads, D.; Erf, G.F.; Slavik, M.F.; Tung, S.; Li, Y. Efficient Separation and Sensitive Detection of Listeria monocytogenes Using an Impedance Immunosensor Based on Magnetic Nanoparticles, a Microfluidic Chip, and an Interdigitated Microelectrode. J. Food Prot. 2012, 75, 1951–1959. [Google Scholar] [CrossRef]
- Chiriacò, M.S.; Parlangeli, I.; Sirsi, F.; Poltronieri, P.; Primiceri, E. Impedance Sensing Platform for Detection of the Food Pathogen Listeria monocytogenes. Electronics 2018, 7, 347. [Google Scholar] [CrossRef] [Green Version]
- Etayash, H.; Norman, L.; Thundat, T. The detection of Listeria monocytogenes using a surface tethered C-terminal 24 amino acid leucocin A fragment. J. Pept. Sci. 2012, 18, S69. [Google Scholar]
- Davis, D.; Guo, X.; Musavi, L.; Lin, C.S.; Chen, S.H.; Wu, V.C.H. Gold Nanoparticle-Modified Carbon Electrode Biosensor for the Detection of Listeria monocytogenes. Ind. Biotechnol. 2013, 9, 31–36. [Google Scholar] [CrossRef]
- Wang, D.; Chen, Q.; Huo, H.; Bai, S.; Cai, G.; Lai, W.; Lin, J. Efficient separation and quantitative detection of Listeria monocytogenes based on screen-printed interdigitated electrode, urease and magnetic nanoparticles. Food Control 2017, 73, 555–561. [Google Scholar] [CrossRef]
- Harris, L.J.; Farber, J.N.; Beuchat, L.R.; Parish, M.E.; Suslow, T.V.; Garrett, E.H.; Busta, F.F. Outbreaks Associated with Fresh Produce: Incidence, Growth, and Survival of Pathogens in Fresh and Fresh-Cut Produce. Compr. Rev. Food Sci. Food Saf. 2003, 2, 78–141. [Google Scholar] [CrossRef]
- Wilks, S.A.; Michels, H.; Keevil, C.W. The survival of Escherichia coli O157 on a range of metal surfaces. Int. J. Food Microbiol. 2005, 105, 445–454. [Google Scholar] [CrossRef] [PubMed]
- Riggio, G.M.; Jones, S.L.; Gibson, K.E. Risk of Human Pathogen Internalization in Leafy Vegetables during Lab-Scale Hydroponic Cultivation. Horticulturae 2019, 5, 25. [Google Scholar] [CrossRef] [Green Version]


| Added [CFU mL−1] a | Rct; Spiked Sample [kΩ] b | Rct; Control [kΩ] b | Measured [CFU mL−1] | Rec. c [%] |
|---|---|---|---|---|
| 219 | 83.1 | 78.0 | 210 | 96 |
| 230 | 83.1 | 78.1 | 215 | 94 |
| 232 | 83.0 | 78.4 | 198 | 86 |
| 351 | 83.9 | 77.9 | 390 | 89 |
| 217 | 83.0 | 77.9 | 195 | 90 |
| 221 | 83.3 | 78.2 | 246 | 89 |
| Microelectrode (Rec. Element a) | Sample | Time [min] | LOD [CFU mL−1] | Linear Range [CFU mL−1] | Hysteresis [%] | Ref. |
|---|---|---|---|---|---|---|
| TiO2 nanowire bundle (Ab) | buffer | 50 | 470 | 102 to 107 | NR | Wang et al. [70] |
| Screen printed electrode (Ab-NPs b + AuNPs c) | blueberry | 60 | 231 | 103 to 106 | NR | Davis et al. [84] |
| Au-IME with portable potentiostat (Ab) | milk | 60 | 5 | 102 to 103 | NR | Chiriacò et al. [82] |
| Gold screen printed electrode (endolysin) | milk | 30 | 1.1 × 105 | 105 to 109 | NR | Tolba et al. [46] |
| Screen printed IDE | lettuce | 180 | 1.0 × 103 | 103 to 106 | NR | Wang et al. [85] |
| nPt d-rGO e electrode (InlA aptamer) | vegetable broth | 17 | 9.1 | 101 to 107 | NR | Hills et al. [28] |
| Pt-IME with laboratory potentiostat (InlA aptamer) | buffer | 17 | 6 ± 1 | 101 to 106 | 15.6% | This study |
| laboratory potentiostat (In1A aptamer) | vegetable broth | 17 | 7.9 ± 2 | 101 to 106 | NR | This study |
| Pt-IME with smartphone potentiostat (InlA aptamer) | hydroponic media | 27 | 23 ± 4 | 102 to 106 | 15.6% | This study |
| Flow through Pt-IME with smartphone potentiostat (InlA aptamer) | hydroponic media | 27 | 48 ± 12 | 102 to 104 | 24.9% | This study |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sidhu, R.K.; Cavallaro, N.D.; Pola, C.C.; Danyluk, M.D.; McLamore, E.S.; Gomes, C.L. Planar Interdigitated Aptasensor for Flow-Through Detection of Listeria spp. in Hydroponic Lettuce Growth Media. Sensors 2020, 20, 5773. https://doi.org/10.3390/s20205773
Sidhu RK, Cavallaro ND, Pola CC, Danyluk MD, McLamore ES, Gomes CL. Planar Interdigitated Aptasensor for Flow-Through Detection of Listeria spp. in Hydroponic Lettuce Growth Media. Sensors. 2020; 20(20):5773. https://doi.org/10.3390/s20205773
Chicago/Turabian StyleSidhu, Raminderdeep K., Nicholas D. Cavallaro, Cicero C. Pola, Michelle D. Danyluk, Eric S. McLamore, and Carmen L. Gomes. 2020. "Planar Interdigitated Aptasensor for Flow-Through Detection of Listeria spp. in Hydroponic Lettuce Growth Media" Sensors 20, no. 20: 5773. https://doi.org/10.3390/s20205773
APA StyleSidhu, R. K., Cavallaro, N. D., Pola, C. C., Danyluk, M. D., McLamore, E. S., & Gomes, C. L. (2020). Planar Interdigitated Aptasensor for Flow-Through Detection of Listeria spp. in Hydroponic Lettuce Growth Media. Sensors, 20(20), 5773. https://doi.org/10.3390/s20205773







