Assessment of Laying Hens’ Thermal Comfort Using Sound Technology
Abstract
1. Introduction
2. Materials and Methods
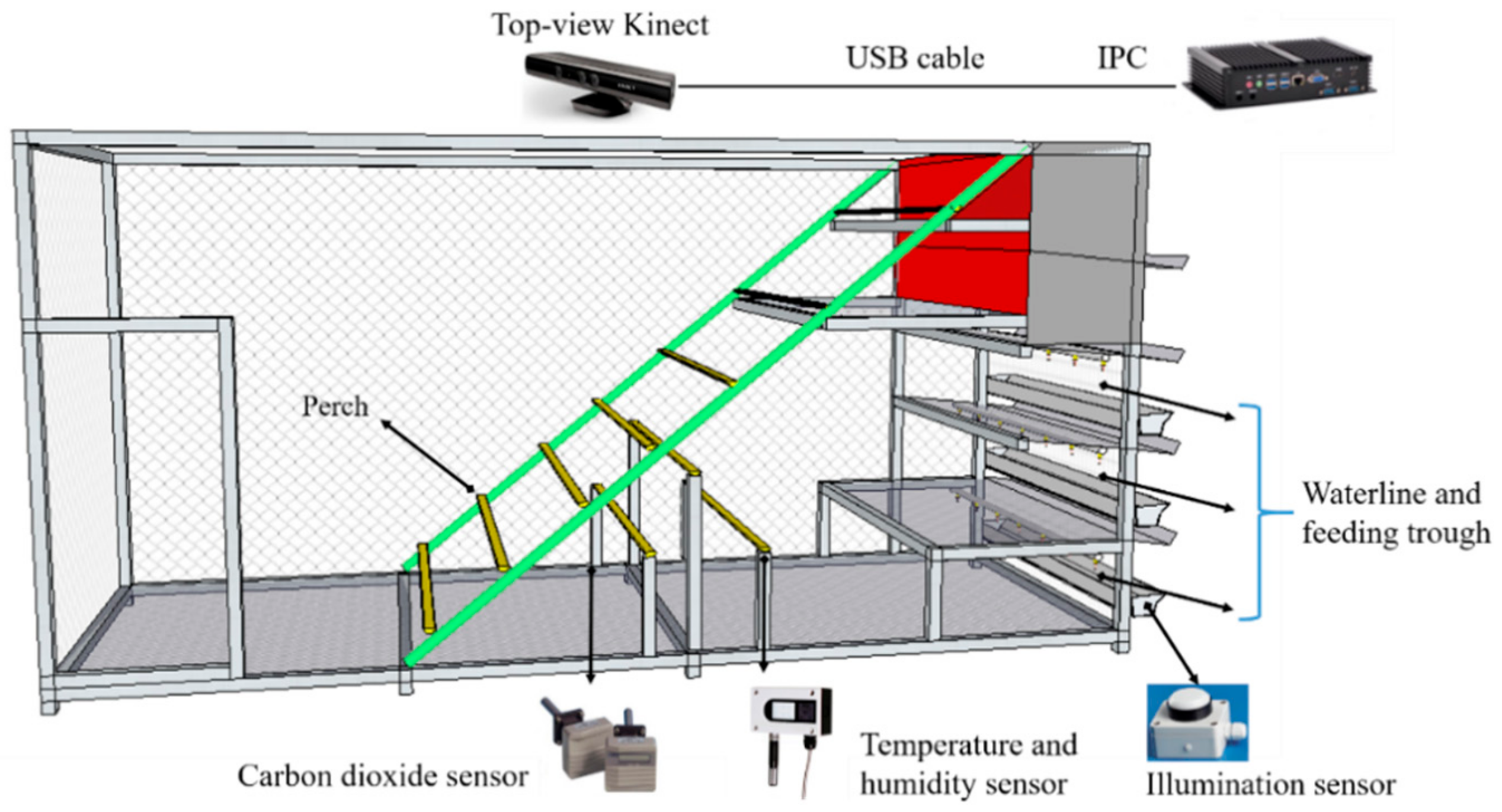
2.1. Animal and House
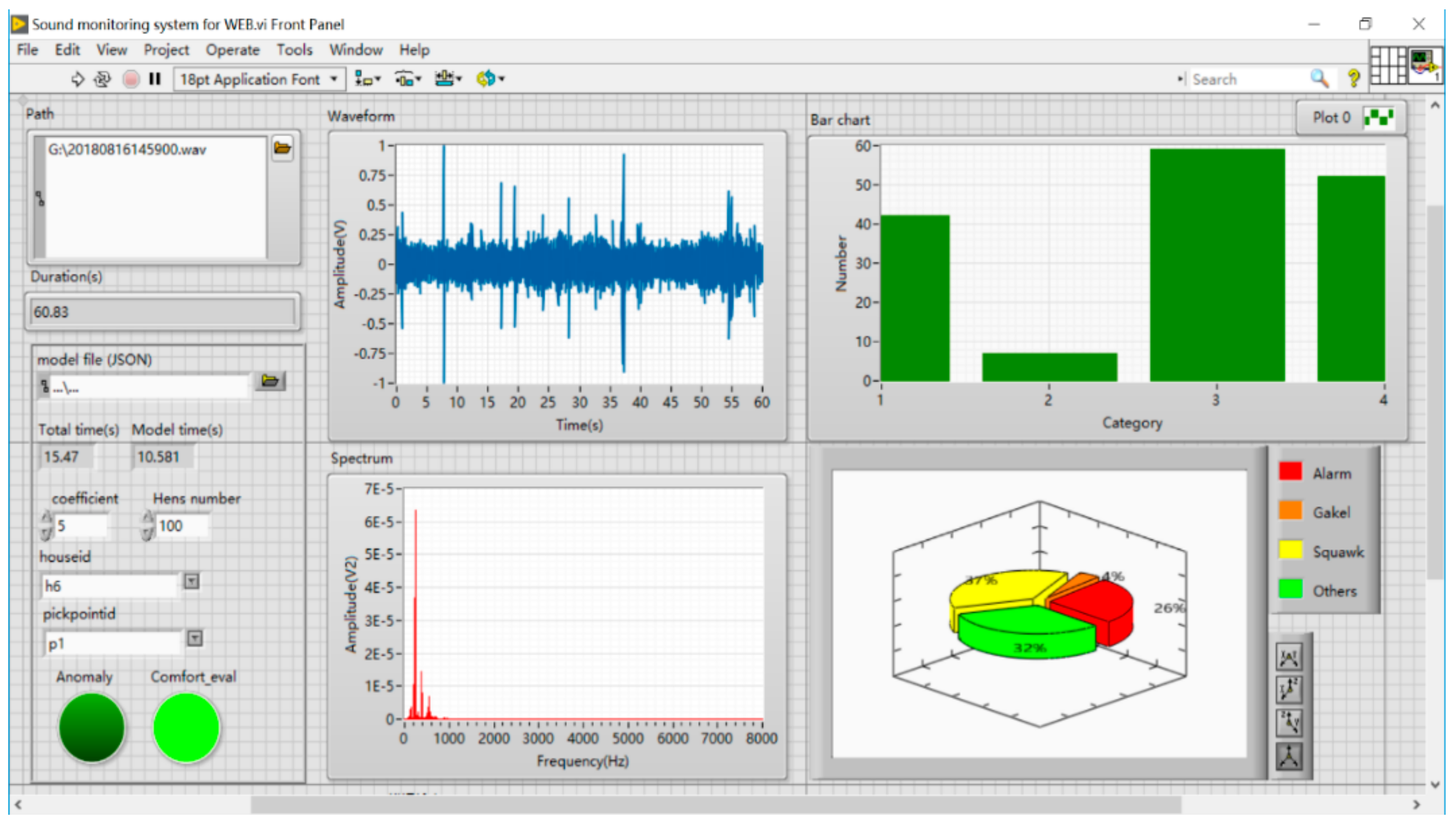
2.2. Data Collection
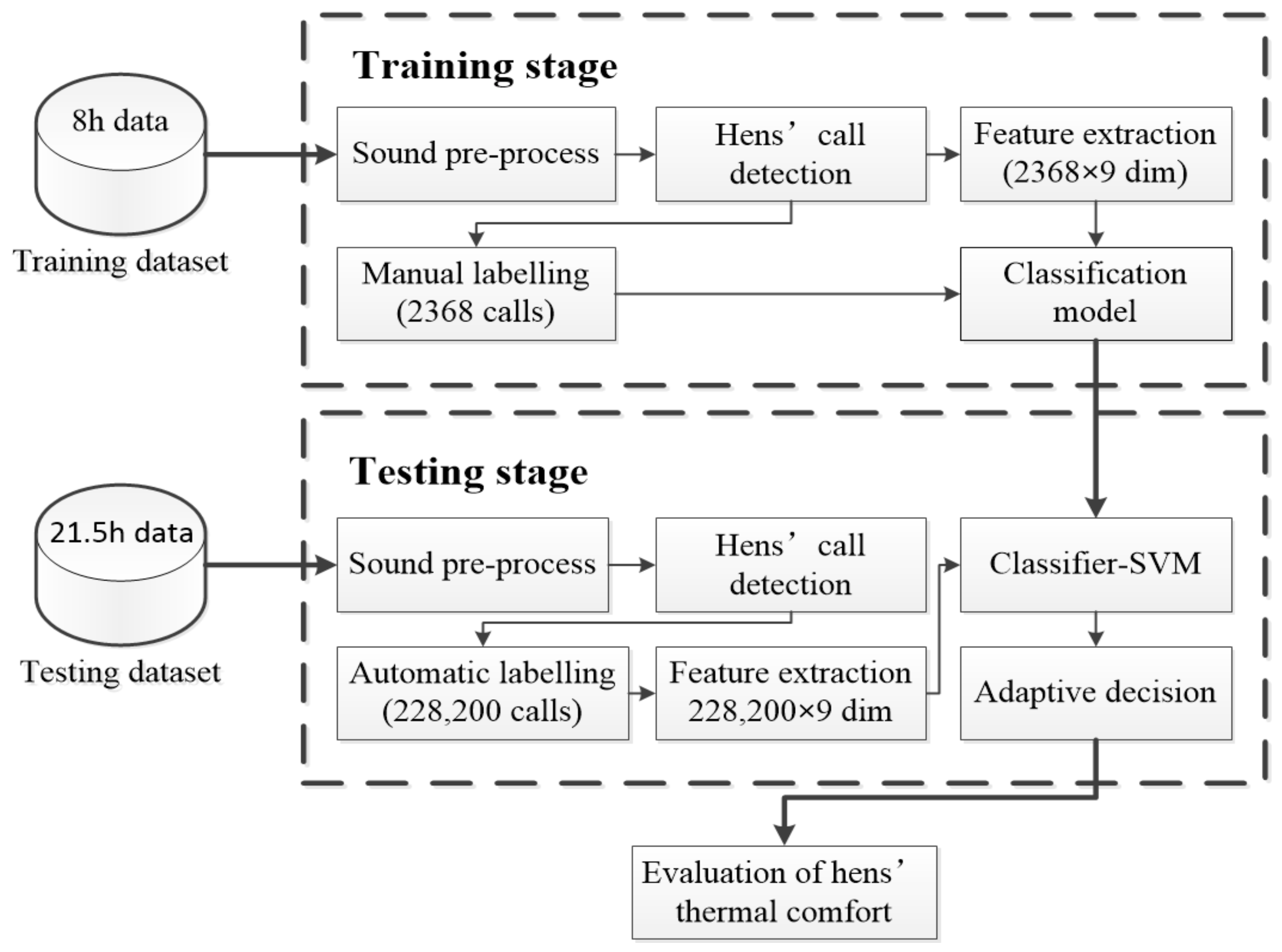
2.3. Sound Signal Pre-Processing and Labelling
2.4. Algorithm for Automatic Hens’ Call Detection
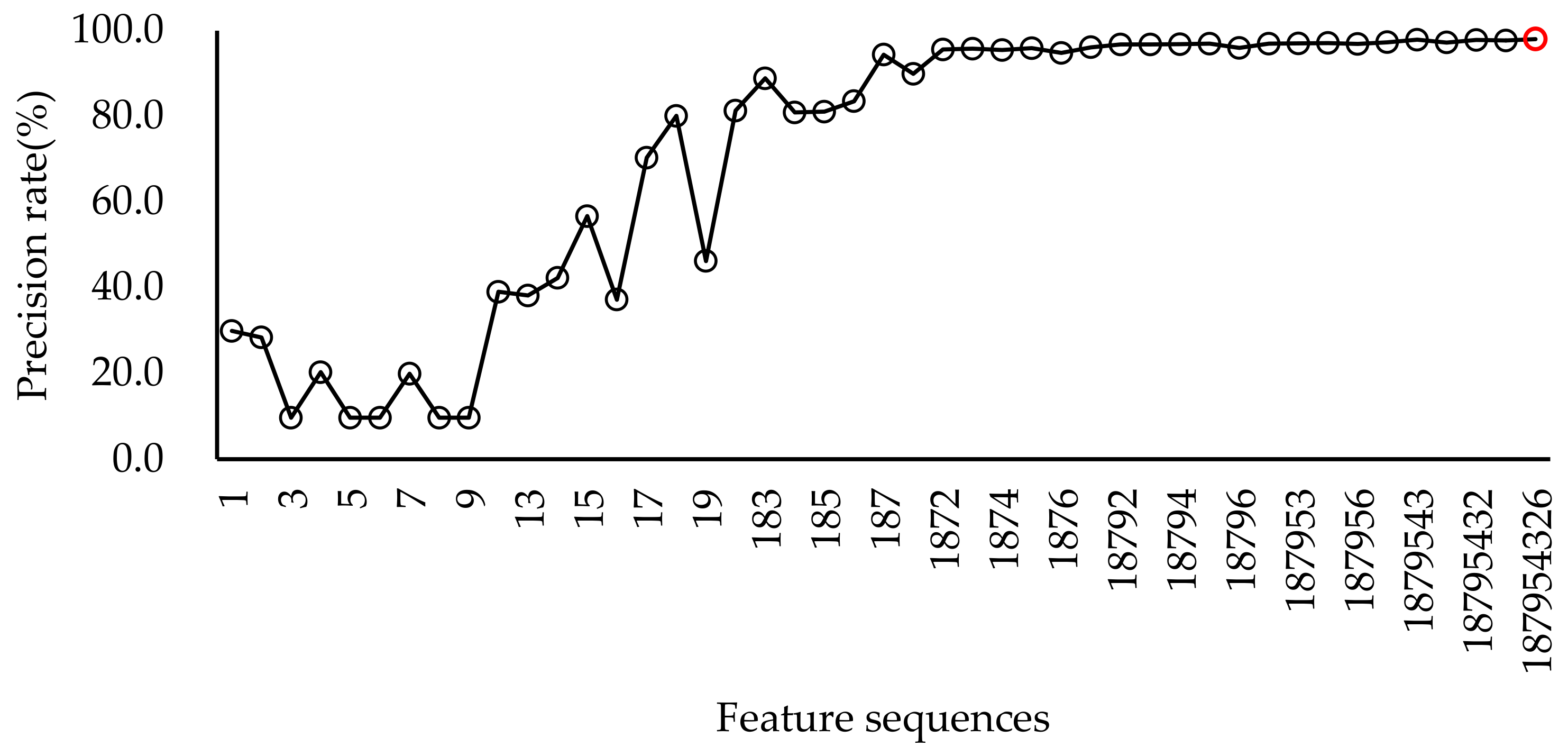
2.4.1. Automatic Sound Event Selection
2.4.2. Feature Extraction
2.4.3. Classification
- Polynomial kernel function:
- Radial basis function with Gaussian kernel:
- Sigmoid function:
2.4.4. Performance Estimation
3. Results and Discussions
3.1. Algorithm Performance
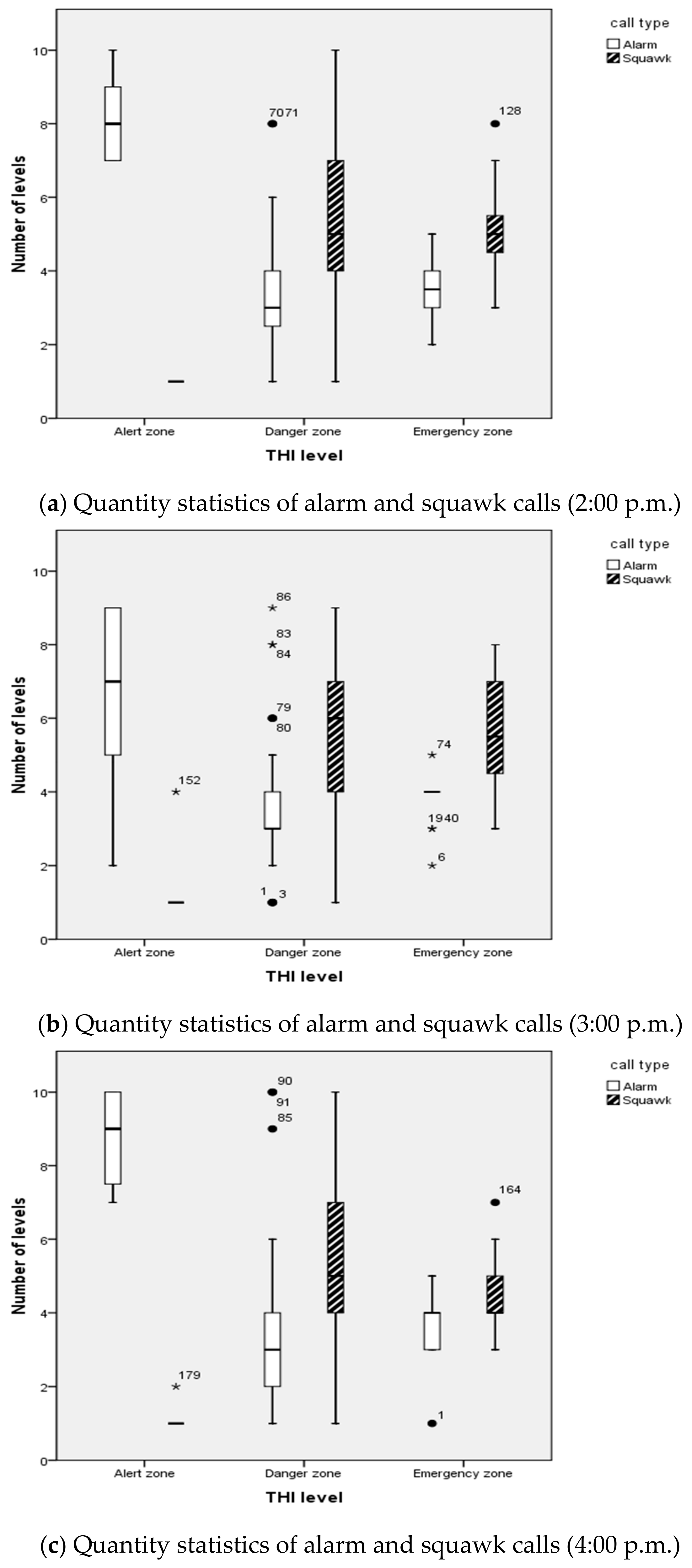
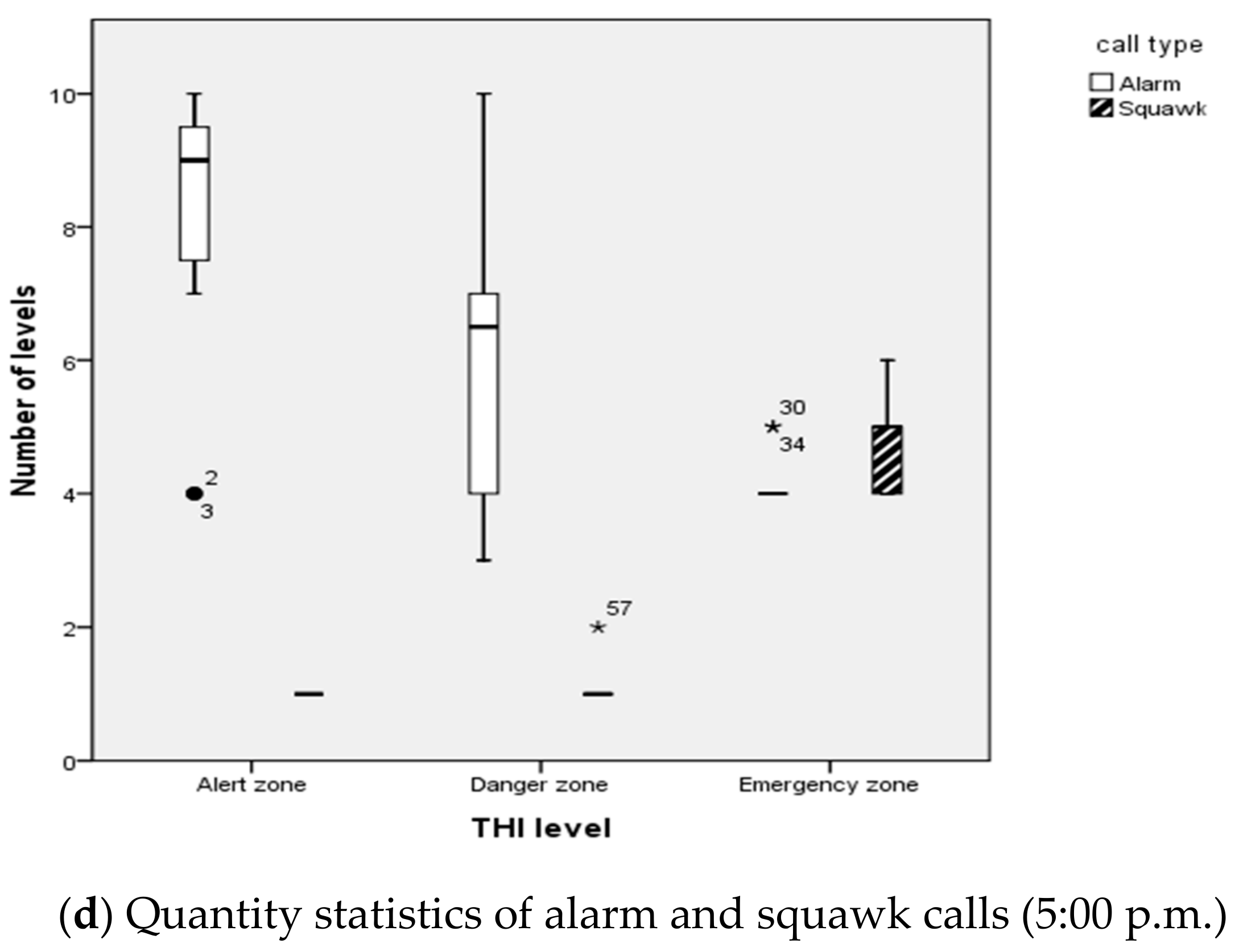
3.2. Thermal Comfort Assessment
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zimmerman, P.H.; Koene, P. The effect of frustrative nonreward on vocalisations and behaviour in the laying hen, Gallus domesticus. Behav. Process. 1998, 44, 73–79. [Google Scholar] [CrossRef]
- Manteuffel, G.; Puppe, B.; Schön, P.C. Vocalization of farm animals as a measure of welfare. Appl. Anim. Behav. Sci. 2004, 88, 163–182. [Google Scholar] [CrossRef]
- Kuhne, F.; Sauerbrey, A.F.C.; Adler, S. The discrimination-learning task determines the kind of frustration-related behaviours in laying hens (Gallus domesticus). Appl. Anim. Behav. 2013, 148, 192–200. [Google Scholar] [CrossRef]
- Nascimento, D.C.N.D.; Dourado, L.R.B.; de Siqueira, J.C.; de Lima, S.B.P.; da Silva, M.C.M.; da Silva, G.G.; Sakomura, N.K.; Ferreira, G.J.B.C.; Biagiotti, D. Productive features of broiler chickens in hot weather: Effects of strain and sex. Semin. Ciências Agrárias 2018, 39, 731–746. [Google Scholar] [CrossRef]
- Lara, L.; Rostagno, M. Impact of heat stress on poultry production. Animals 2013, 3, 356–369. [Google Scholar] [CrossRef]
- Freitas, L.C.S.R.; Tinôco, I.F.F.; Baêta, F.C.; Barbari, M.; Conti, L.; Teles Júnior, C.G.S.; Cândido, M.G.L.; Morais, C.V.; Sousa, F.C. Correlation between egg quality parameters, housing thermal conditions and age of laying hens. Agron. Res. 2017, 15, 687–693. [Google Scholar]
- Mutibvu, T.; Chimonyo, M.; Halimani, T.E. Physiological responses of slow-growing chickens under diurnally cycling temperature in a hot environment. Braz. J. Poult. Sci. 2017, 19, 567–576. [Google Scholar] [CrossRef]
- Vieira, F.M.C.; Groff, P.M.; Silva, I.J.O.; Nazareno, A.C.; Godoy, T.F.; Coutinho, L.L.; Vieira, A.M.C.; Silva-Miranda, K.O. Impact of exposure time to harsh environments on physiology, mortality, and thermal comfort of day-old chickens in a simulated condition of transport. Int. J. Biometeorol. 2019, 63, 777–785. [Google Scholar] [CrossRef]
- Collias, N.E. The vocal repertoire of the red junglefowl-a spectrographic classification and the code of communication. Condor 1987, 89, 510–524. [Google Scholar] [CrossRef]
- Zimmerman, P.H.; Koene, P.; van Hooff, J. Thwarting of behaviour in different contexts and the gakel-call in the laying hen. Appl. Anim. Behav. 2000, 69, 255–264. [Google Scholar] [CrossRef]
- Marx, G.; Leppelt, J.; Ellendorff, F. Vocalisation in chicks (Gallus dom.) during stepwise social isolation. Appl. Anim. Behav. 2001, 75, 61–74. [Google Scholar] [CrossRef]
- Du, X.; Lao, F.; Teng, G. A sound source localisation analytical method for monitoring the abnormal night vocalisations of poultry. Sensors 2018, 18, 2906. [Google Scholar] [CrossRef] [PubMed]
- Bright, A. Vocalisations and acoustic parameters of flock noise from feather pecking and non-feather pecking laying flocks. Br. Poult. Sci. 2008, 49, 241–249. [Google Scholar] [CrossRef] [PubMed]
- Kim, N.Y.; Jang, S.Y.; Kim, S.J.; Jeon, B.T.; Oh, M.R.; Kim, E.K.; Seong, H.J.; Tang, Y.J.; Yun, Y.S.; Moon, S.H. Behavioral and vocal characteristics of laying hens under different housing and feeding conditions. J. Anim. Plant Sci. 2017, 27, 65–74. [Google Scholar]
- Xin, H.; Harmon, J.D. Livestock industry facilities and environment: Heat stress indices for livestock. In Agriculture and Environment Extension Publications; Iowa State University Digital Repository: Ames, IA, USA, 1998; p. 163. [Google Scholar]
- Perera, W.N.U.; Dematawewa, C.M.B. Thermal comfort differences in poultry houses and its influence on growth performance of broiler strains. Acta Hortic. 2017, 1152, 415–420. [Google Scholar] [CrossRef]
- Vandermeulen, J.; Kashiha, M.; Ott, S.; Bahr, C.; Moons, C.P.H.; Tuyttens, F.; Niewold, T.A.; Berckmans, D. Combination of image and sound analysis for behaviour monitoring in pigs. In Proceedings of the 6th european conference on Precision Livestock Farming, Leuven, Belgium, 10–12 September 2013. [Google Scholar]
- Kashiha, M.; Pluk, A.; Bahr, C.; Vranken, E.; Berckmans, D. Development of an early warning system for a broiler house using computer vision. Biosyst. Eng. 2013, 116, 36–45. [Google Scholar] [CrossRef]
- Mielke, A.; Zuberbühler, K. A method for automated individual, species and call type recognition in free-ranging animals. Anim. Behav. 2013, 86, 475–482. [Google Scholar] [CrossRef]
- Favaro, L.; Gamba, M.; Alfieri, C.; Pessani, D.; McElligott, A.G. Vocal individuality cues in the African penguin (Spheniscus demersus): A source-filter theory approach. Sci. Rep.-UK 2015, 5. [Google Scholar] [CrossRef]
- Yeon, S.C.; Jeon, J.H.; Houpt, K.A.; Chang, H.H.; Lee, H.C.; Lee, H.J. Acoustic features of vocalizations of Korean native cows (Bos taurus coreanea) in two different conditions. Appl. Anim. Behav. 2006, 101, 1–9. [Google Scholar] [CrossRef]
- Chuang, M.; Kam, Y.; Bee, M.A. Territorial olive frogs display lower aggression towards neighbours than strangers based on individual vocal signatures. Anim. Behav. 2017, 123, 217–228. [Google Scholar] [CrossRef]
- Dhanalakshmi, P.; Palanivel, S.; Ramalingam, V. Classification of audio signals using SVM and RBFNN. Expert Syst. Appl. 2009, 36, 6069–6075. [Google Scholar] [CrossRef]
- Chen, L.J.; Mao, X.; Xue, Y.L.; Cheng, L.L. Speech emotion recognition: Features and classification models. Digit. Signal Process. 2012, 22, 1154–1160. [Google Scholar] [CrossRef]
- Steen, K.A.; Therkildsen, O.R.; Karstoft, H.; Green, O. A vocal-based analytical method for goose behaviour recognition. Sensors 2012, 12, 3773–3788. [Google Scholar] [CrossRef] [PubMed]
- Cortes, C.; Vapnik, V. Support-vector networks. Mach. Learn. 1995, 20, 273–297. [Google Scholar] [CrossRef]
- Banakar, A.; Sadeghi, M.; Shushtari, A. An intelligent device for diagnosing avian diseases: Newcastle, infectious bronchitis, avian influenza. Comput. Electron. Agr. 2016, 127, 744–753. [Google Scholar] [CrossRef]
- Huang, J.; Wang, W.; Zhang, T. Method for detecting avian influenza disease of chickens based on sound analysis. Biosyst. Eng. 2019, 180, 16–24. [Google Scholar] [CrossRef]
- Bishop, J.C.; Falzon, G.; Trotter, M.; Kwan, P.; Meek, P.D. Livestock vocalisation classification in farm soundscapes. Comput. Electron. Agric. 2019, 162, 531–542. [Google Scholar] [CrossRef]
- Konishi, M. The role of auditory feedback in the vocal behavior of the domestic fowl. Zeitschrift für Tierpsychologie 1963, 20, 349–367. [Google Scholar]
- Cao, Y.F.; Yu, L.G.; Teng, G.H.; Zhao, S.M.; Liu, X.M. Feature extraction and classification of laying hens’ vocalization and mechanical noise. Trans. Chin. Soc. Agric. Eng. 2014, 18, 190–197. [Google Scholar]
- Martin, R. Noise power spectral density estimation based on optimal smoothing and minimum statistics. IEEE Trans. Audio Speech 2001, 9, 504–512. [Google Scholar] [CrossRef]
- Tullo, E.; Fontana, I.; Diana, A.; Norton, T.; Berckmans, D.; Guarino, M. Application note: Labelling, a methodology to develop reliable algorithm in PLF. Comput. Electron. Agric. 2017, 142, 424–428. [Google Scholar] [CrossRef]
- Carpentier, L.; Berckmans, D.; Youssef, A.; Berckmans, D.; van Waterschoot, T.; Johnston, D.; Ferguson, N.; Earley, B.; Fontana, I.; Tullo, E.; et al. Automatic cough detection for bovine respiratory disease in a calf house. Biosyst. Eng. 2018, 173, 45–56. [Google Scholar] [CrossRef]
- Exadaktylos, V.; Silva, M.; Aerts, J.M.; Taylor, C.J.; Berckmans, D. Real-time recognition of sick pig cough sounds. Comput. Electron. Agric. 2008, 63, 207–214. [Google Scholar] [CrossRef]
- Carpentier, L.; Vranken, E.; Berckmans, D.; Paeshuyse, J.; Norton, T. Development of sound-based poultry health monitoring tool for automated sneeze detection. Comput. Electron. Agric. 2019, 162, 573–581. [Google Scholar] [CrossRef]
- Giannakopoulos, T.; Pikrakis, A. Introduction to Audio Analysis: A MATLAB® Approach, 1st ed.; Academic Press: Cambridge, MA, USA, 2014; ISBN 978-0-08-099388-1. [Google Scholar]
- Vapnik, V.N. Statistical Learning Theory; Wiley-Interscience: New York, NY, USA, 1998; pp. 156–160. ISBN 978-0-47-103003-4. [Google Scholar]
- Özbek, M.E.; Özkurt, N.; Savacı, F.A. Wavelet ridges for musical instrument classification. J. Intell. Inf. Syst. 2012, 38, 241–256. [Google Scholar] [CrossRef][Green Version]
- Refaeilzadeh, P.; Tang, L.; Liu, H. Cross-Validation, in Encyclopedia of Database Systems; Springer: Boston, MA, USA, 2009; pp. 532–538. ISBN 9780387355443. [Google Scholar]
- Chen, W.; Chen, S.S.; Lin, C.C.; Chen, Y.Z.; Lin, W.C. Automatic recognition of frog calls using a multi-stage average spectrum. Comput. Math. Appl. 2012, 64, 1270–1281. [Google Scholar] [CrossRef]
- Liu, L.S.; Ni, J.Q.; Li, Y.S.; Erasmus, M.; Stevenson, R.; Shen, M.X. Assessment of heat stress in turkeys using animal vocalization analysis. In Proceedings of the 2018 ASABE Annual International Meeting, Detroit, MI, USA, 29 July–1 August 2018. [Google Scholar] [CrossRef]
- Moura, D.J.d.; Naas, I.d.A.; Alves, E.C.d.S.; Carvalho, T.M.R.d.; Vale, M.M.d.; Lima, K.A.O.d. Noise analysis to evaluate chick thermal comfort. Sci. Agric. 2008, 65, 438–443. [Google Scholar] [CrossRef]
- Tokuda, I.; Riede, T.; Neubauer, J.; Owren, M.J.; Herzel, H. Nonlinear analysis of irregular animal vocalizations. J. Acoust. Soc. Am. 2002, 111, 2908–2919. [Google Scholar] [CrossRef]
- Lee, J.; Noh, B.; Jang, S.; Park, D.; Chung, Y.; Chang, H.H. Stress deection and classification of laying hens by sound analysis. Asian Austral. J. Anim. 2015, 28, 592–598. [Google Scholar] [CrossRef]


| Call Type | Description |
|---|---|
| Gakel | Soft, brief (<0.2 s) repetitive notes generally with a wide frequency range; often emphasize low frequencies (below 2 kHz). Notes with definite, clear harmonic structure [10,30]. |
| Alarm | High pitched sound of duration (<0.2 s) with a distinct harmonic structure, moderately loud (similar to alert calls [9,13,30]). |
| Squawk | Component notes are short (<0.1 s) with an abrupt onset and ending and cover a wide frequency range. This call is a moderately loud sound (similar to distress cries [9,13,30]). |
| Others | Other hens’ vocalisations. Total vocalisation rate is positively correlated with event aversiveness in domestic chickens [13]. |
| Feature Parameters | Description | Order |
|---|---|---|
| Jitter_f0 | Mean absolute difference between frequencies of consecutive f0 periods divided by the mean frequency of f0 (fundamental frequency) [20] | 1 |
| Jitter_F1 | Mean absolute difference between frequencies of consecutive F1 periods divided by the mean frequency of F1 (the first formant) [20] | 2 |
| Jitter_F2 | Mean absolute difference between frequencies of consecutive F2 periods divided by the mean frequency of F2 (the second formant) [20] | 3 |
| Shimmer_F1 | Mean absolute difference between the amplitudes of consecutive F1 periods divided by the mean amplitude of F1 [20] | 4 |
| Shimmer_F3 | Mean absolute difference between the amplitudes of consecutive F3 periods divided by the mean amplitude of F3 (the third formant) [20] | 5 |
| ZCR | The zero-crossing rate (ZCR) of an audio frame is the rate of sign-changes of the signal during the frame [37] | 6 |
| Spectral spread | The spectral spread is the second central moment of the spectrum [36] | 7 |
| Spectral energy | Refer to Equation (1) | 8 |
| Spectral centroid | The spectral centroid is the centre of ‘gravity’ of the spectrum [36] | 9 |
| Real Call Type | Classified by 9 Features | |||||
|---|---|---|---|---|---|---|
| Alarm | Gakel | Squawk | Others | Total | Sensitivity (%) | |
| Alarm | 906 | 0 | 5 | 6 | 917 | 98.8 |
| Gakel | 7 | 96 | 3 | 4 | 110 | 87.3 |
| Squawk | 18 | 0 | 727 | 5 | 750 | 96.9 |
| Others | 8 | 0 | 13 | 570 | 591 | 96.4 |
| Total | 939 | 96 | 748 | 585 | 2368 | - |
| Precision (%) | 96.5 | 100.0 | 97.2 | 97.4 | - | - |
| Call Type | Classification Performance | |
|---|---|---|
| Sensitivity ± SD (%) | Precision ± SD (%) | |
| Alarm | 98.4 ± 0.5 | 95.5 ± 1.4 |
| Gakel | 88.9 ± 1.4 | 100.0 ± 0.0 |
| Squawk | 96.1 ± 1.7 | 96.6 ± 0.4 |
| Others | 97.0 ± 1.3 | 98.1 ± 0.5 |
| Total | 95.1 ± 4.3 | 97.6 ± 1.9 |
| THI | Alarm | ||
|---|---|---|---|
| THI | Pearson Correlation | 1 | −0.414 ** |
| Sig. (2-tailed) | 0.008 | ||
| N | 40 | 40 | |
| Alarm | Pearson Correlation | −0.414 ** | 1 |
| Sig. (2-tailed) | 0.008 | ||
| N | 40 | 40 |
| THI | Squawk | ||
|---|---|---|---|
| THI | Pearson Correlation | 1 | 0.594 ** |
| Sig. (2-tailed) | 0.000 | ||
| N | 40 | 40 | |
| Squawk | Pearson Correlation | 0.594 ** | 1 |
| Sig. (2-tailed) | 0.000 | ||
| N | 40 | 40 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Du, X.; Carpentier, L.; Teng, G.; Liu, M.; Wang, C.; Norton, T. Assessment of Laying Hens’ Thermal Comfort Using Sound Technology. Sensors 2020, 20, 473. https://doi.org/10.3390/s20020473
Du X, Carpentier L, Teng G, Liu M, Wang C, Norton T. Assessment of Laying Hens’ Thermal Comfort Using Sound Technology. Sensors. 2020; 20(2):473. https://doi.org/10.3390/s20020473
Chicago/Turabian StyleDu, Xiaodong, Lenn Carpentier, Guanghui Teng, Mulin Liu, Chaoyuan Wang, and Tomas Norton. 2020. "Assessment of Laying Hens’ Thermal Comfort Using Sound Technology" Sensors 20, no. 2: 473. https://doi.org/10.3390/s20020473
APA StyleDu, X., Carpentier, L., Teng, G., Liu, M., Wang, C., & Norton, T. (2020). Assessment of Laying Hens’ Thermal Comfort Using Sound Technology. Sensors, 20(2), 473. https://doi.org/10.3390/s20020473







