Marker-Based Movement Analysis of Human Body Parts in Therapeutic Procedure
Abstract
1. Introduction
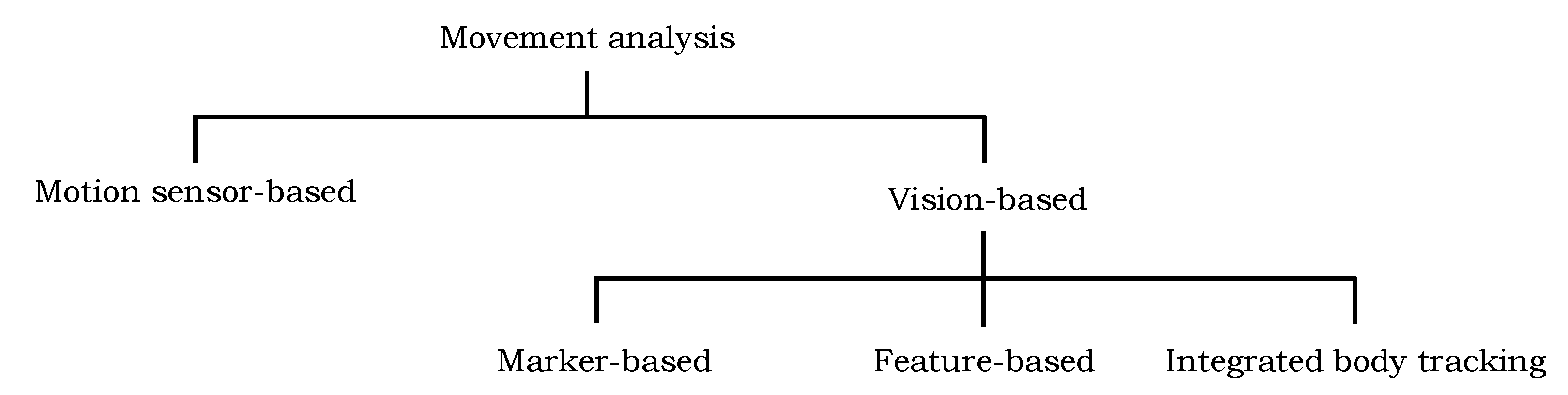
2. Background
2.1. Vision-Based Algorithms
2.1.1. Marker-Based Techniques
2.1.2. Feature-Based Techniques
2.1.3. Integrated Body Tracking Functionality
2.2. Motion Sensor-Based Algorithms
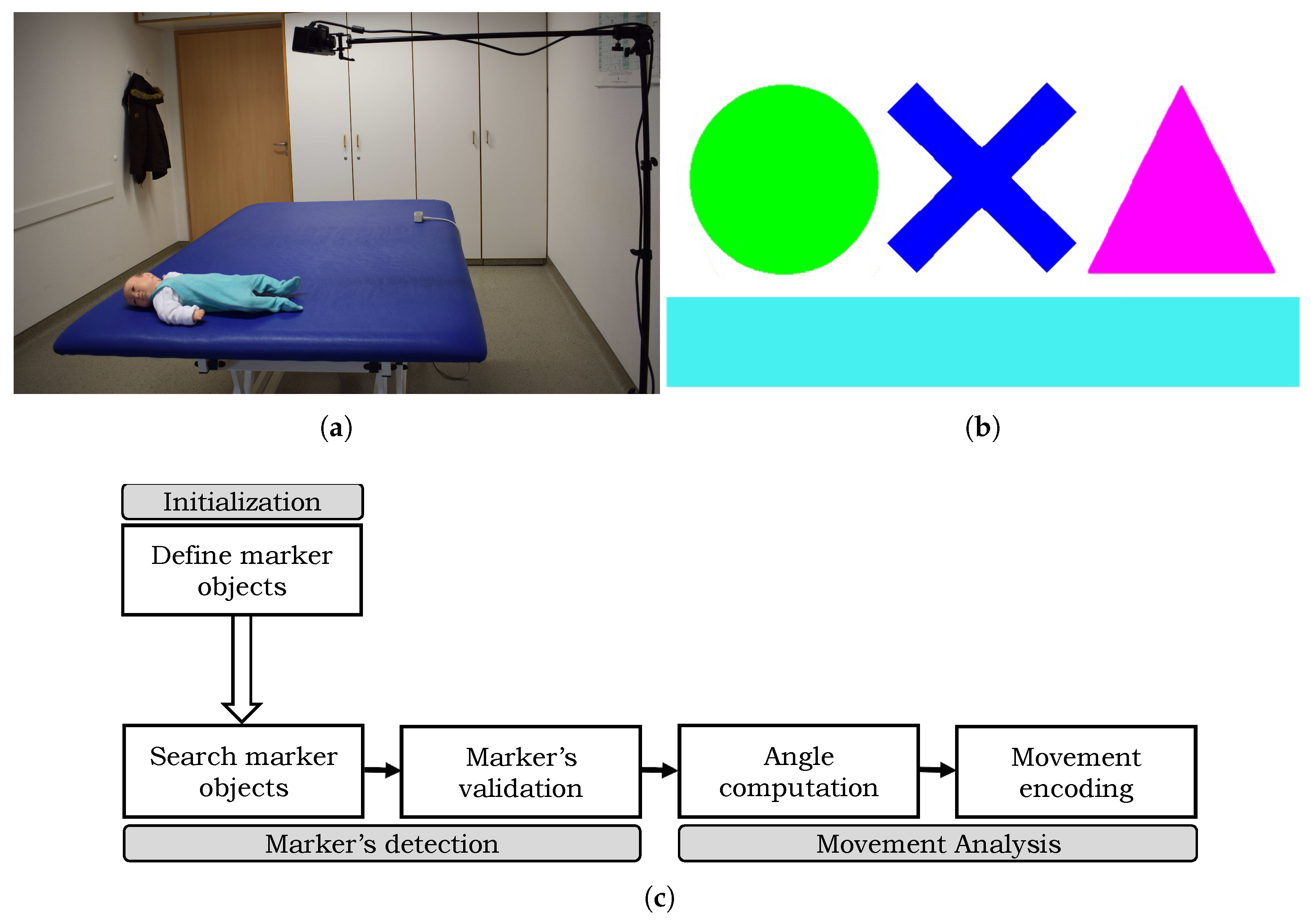
3. Data Acquisition
4. Proposed Movement Analysis Method
4.1. Initialization
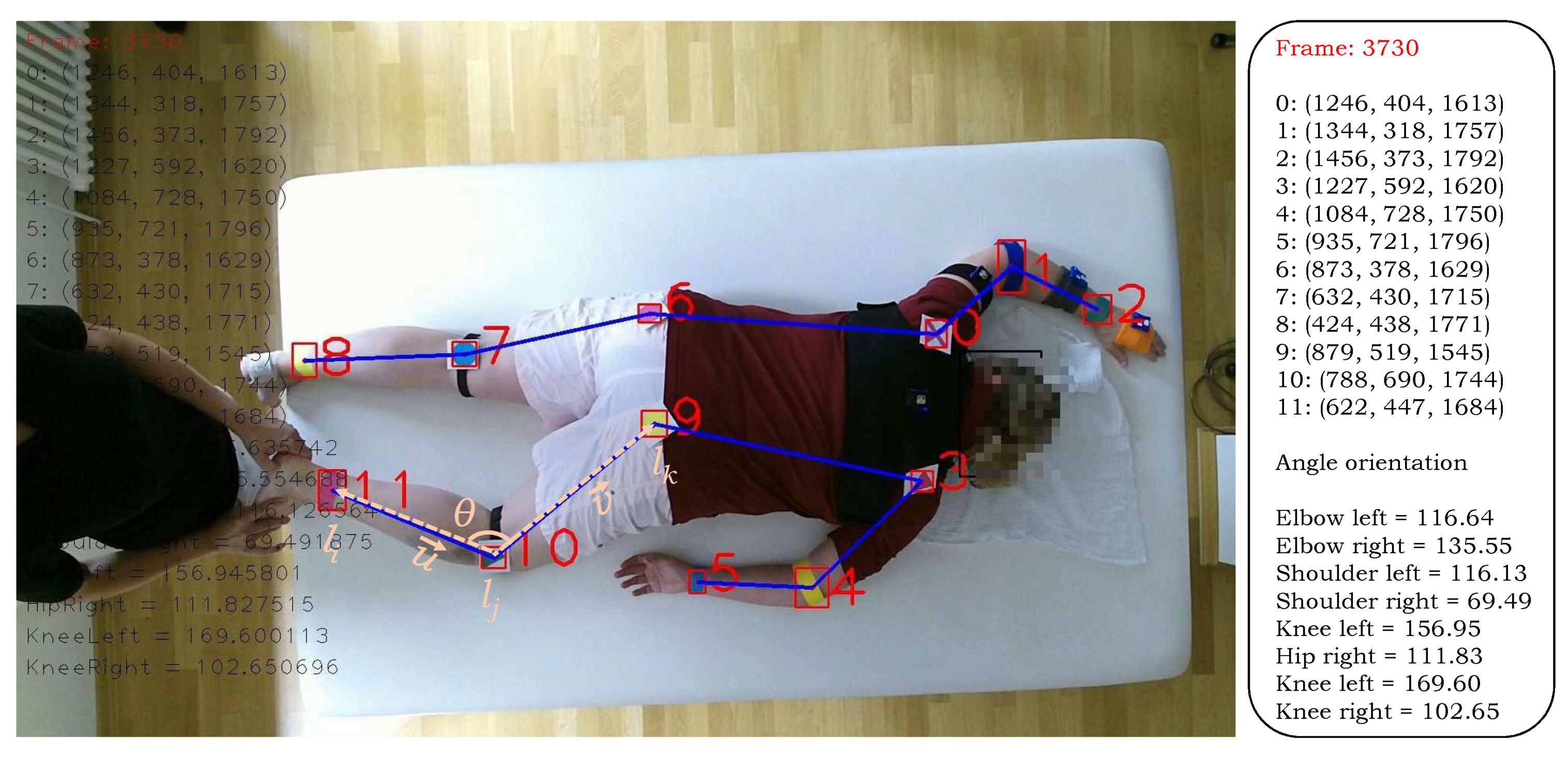
4.2. Marker Detection
4.3. Movement Analysis
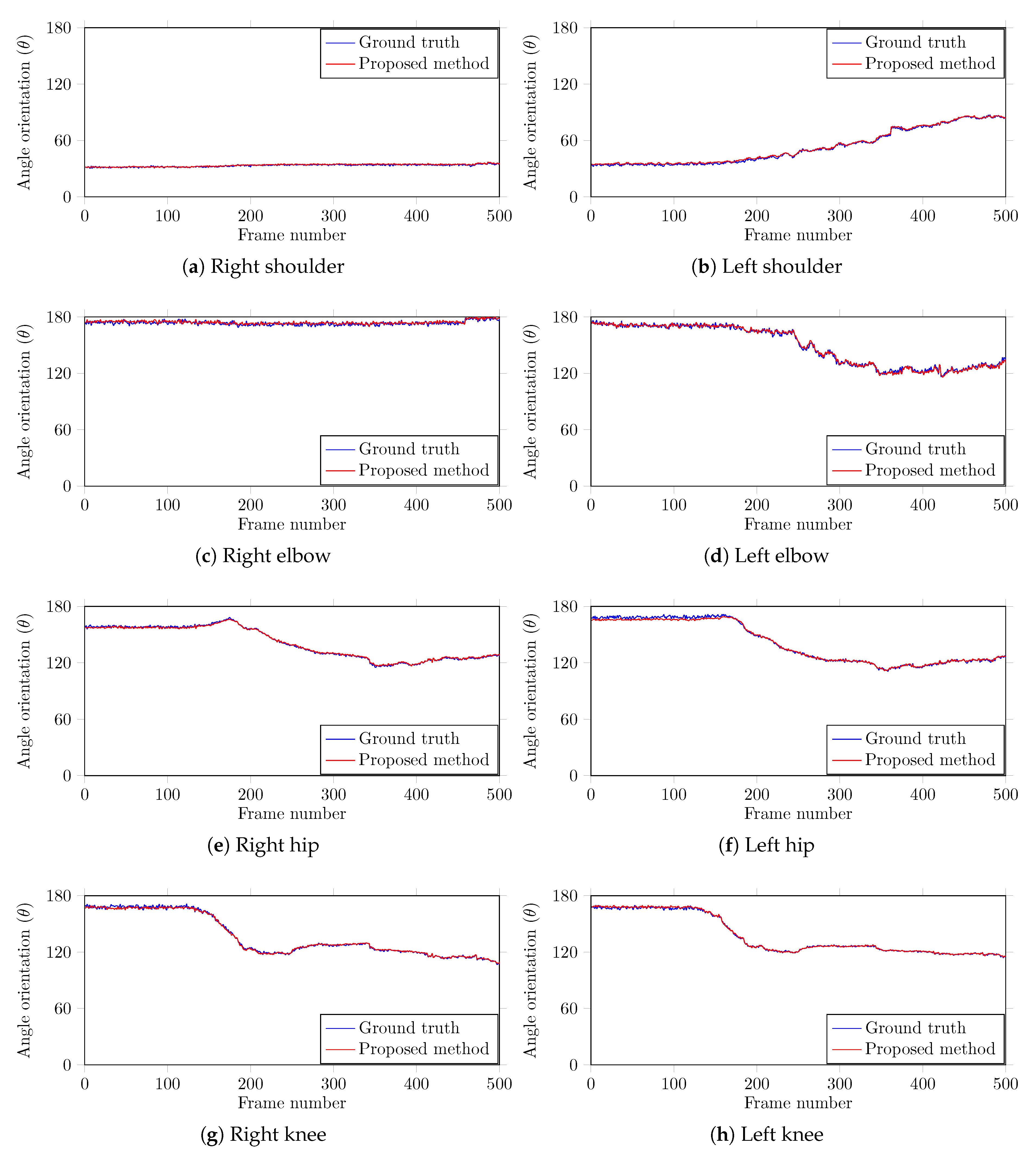
5. Experiments and Results
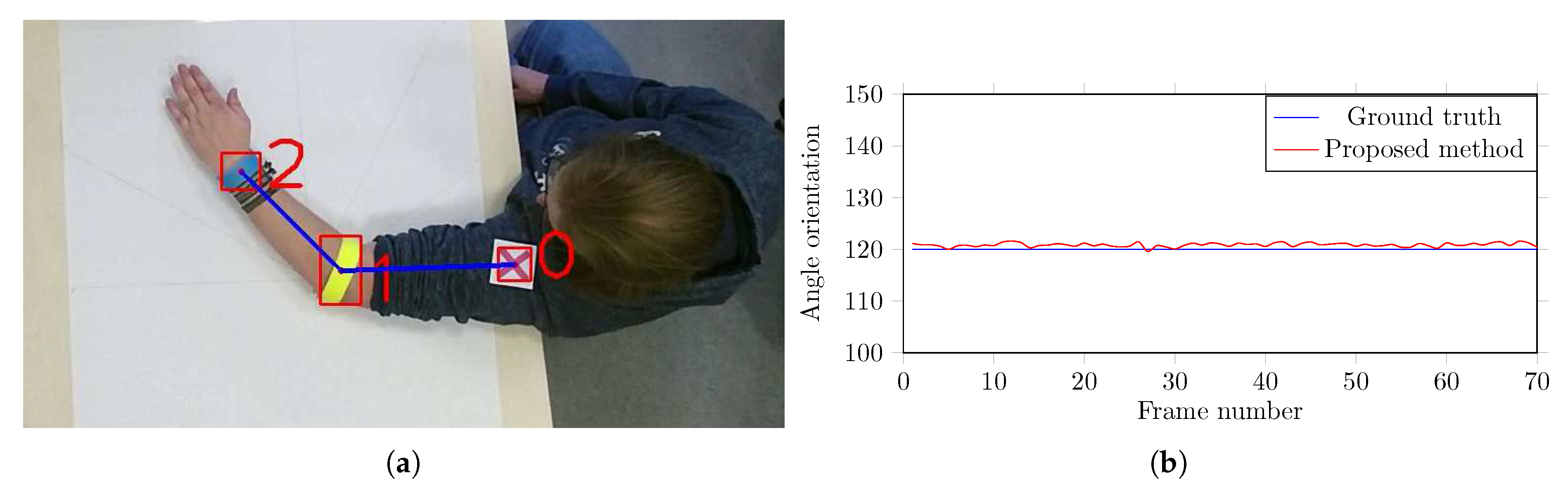
5.1. Performance Evaluation and Comparison
5.2. Computational Complexity Analysis
5.3. Challenges, Shortcomings, and Future Research Directions
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Vojta, V. The basic elements of treatment according to Vojta. Manag. Mot. Disord. Child. Cereb. Palsy 1984, 75, 75–85. [Google Scholar]
- Vojta, V.; Peters, A. Das Vojta-Prinzip: Muskelspiele in Reflexfortbewegung und Motorischer Ontogenese (English Title: The Vojta Principle: Muscular Games in Reflex Movement and Motor Ontogenesis); Springer: Berlin, Germany, 2007. [Google Scholar]
- Bobath, B. Die Hemiplegie Erwachsener: Befundaufnahme, Beurteilung und Behandlung (English Title: The Hemiplegia of Adults: Findings, Assessment and Treatment); Georg Thieme Verlag: New York, NY, USA, 1998. [Google Scholar]
- Luke, C.; Dodd, K.J.; Brock, K. Outcomes of the Bobath concept on upper limb recovery following stroke. Clin. Rehabil. 2004, 18, 888–898. [Google Scholar] [CrossRef] [PubMed]
- Imamura, S.; Sakuma, K.; Takahashi, T. Follow-up study of children with cerebral coordination disturbance (CCD, Vojta). Brain Dev. 1983, 5, 311–314. [Google Scholar] [CrossRef]
- Lim, H.W. The effect of vojta therapy on gross motor function measure and selective voluntary motor control in children with spastic diplegia. J. Korean Soc. Phys. Med. 2012, 7, 213–221. [Google Scholar] [CrossRef]
- Da Gama, A.; Chaves, T.; Figueiredo, L.; Teichrieb, V. Guidance and movement correction based on therapeutics movements for motor rehabilitation support systems. In Proceedings of the 14th Symposium on Virtual and Augmented Reality, Rio Janiero, Brazil, 28–31 May 2012; IEEE: Piscataway, NJ, USA, 2012; pp. 191–200. [Google Scholar]
- Wen, B.; Ma, L.; Weng, C. The impact of constraint induced movement therapy on brain activation in chronic stroke patients with upper extremity paralysis: An fMRI study. Int. J. Imaging Syst. Technol. 2014, 24, 270–275. [Google Scholar] [CrossRef]
- Kim, S.G.; Jennings, J.E.; Strupp, J.P.; Andersen, P.; Uǧurbil, K. Functional MRI of human motor cortices during overt and imagined finger movements. Int. J. Imaging Syst. Technol. 1995, 6, 271–279. [Google Scholar] [CrossRef]
- Khan, M.; Schneider, M.; Farid, M.; Grzegorzek, M. Detection of Infantile Movement Disorders in Video Data Using Deformable Part-Based Model. Sensors 2018, 18, 3202. [Google Scholar] [CrossRef]
- Saygılı, A.; Albayrak, S. An efficient and fast computer-aided method for fully automated diagnosis of meniscal tears from magnetic resonance images. Artif. Intell. Med. 2019, 97, 118–130. [Google Scholar] [CrossRef]
- Burke, J.; Morrow, P.; McNeill, M.; McDonough, S.; Charles, D. Vision based games for upper-limb stroke rehabilitation. In Proceedings of the 2008 International Machine Vision and Image Processing Conference (IMVIP), Portrush, Ireland, 3–5 September 2008; IEEE: Piscataway, NJ, USA, 2008; pp. 159–164. [Google Scholar]
- Tao, Y.; Hu, H. Colour based human motion tracking for home-based rehabilitation. In Proceedings of the International Conference on Systems, Man and Cybernetics, The Hague, The Netherlands, 10–13 October 2004; IEEE: Piscataway, NJ, USA, 2004; Volume 1, pp. 773–778. [Google Scholar]
- Meinecke, L.; Breitbach-Faller, N.; Bartz, C.; Damen, R.; Rau, G.; Disselhorst-Klug, C. Movement analysis in the early detection of newborns at risk for developing spasticity due to infantile cerebral palsy. Hum. Mov. Sci. 2006, 25, 125–144. [Google Scholar] [CrossRef]
- Rado, D.; Sankaran, A.; Plasek, J.; Nuckley, D.; Keefe, D.F. A Real-Time Physical Therapy Visualization Strategy to Improve Unsupervised Patient Rehabilitation; IEEE Visualization: Piscataway, NJ, USA, 2009. [Google Scholar]
- Olsen, M.D.; Herskind, A.; Nielsen, J.B.; Paulsen, R.R. Model-based motion tracking of infants. In Proceedings of the European Conference on Computer Vision, Zurich, Switzerland, 6–12 September 2014; Springer: Berlin, Germany, 2014; pp. 673–685. [Google Scholar]
- Khan, M.H.; Helsper, J.; Farid, M.S.; Grzegorzek, M. A computer vision-based system for monitoring Vojta therapy. Int. J. Med. Inform. 2018, 113, 85–95. [Google Scholar] [CrossRef]
- Penelle, B.; Debeir, O. Human motion tracking for rehabilitation using depth images and particle filter optimization. In Proceedings of the 2nd International Conference on Advances in Biomedical Engineering, Tripoli, Lebanon, 11–13 September 2013; IEEE: Piscataway, NJ, USA, 2013; pp. 211–214. [Google Scholar]
- Zhang, Y.; Lu, X. Measurement method for human body anteflexion angle based on image processing. Int. J. Imaging Syst. Technol. 2019, 29, 518–530. [Google Scholar] [CrossRef]
- Chang, Y.J.; Chen, S.F.; Huang, J.D. A Kinect-based system for physical rehabilitation: A pilot study for young adults with motor disabilities. Res. Dev. Disabil. 2011, 32, 2566–2570. [Google Scholar] [CrossRef] [PubMed]
- Exell, T.; Freeman, C.; Meadmore, K.; Kutlu, M.; Rogers, E.; Hughes, A.M.; Hallewell, E.; Burridge, J. Goal orientated stroke rehabilitation utilising electrical stimulation, iterative learning and microsoft kinect. In Proceedings of the IEEE 13th International Conference on Rehabilitation Robotics (ICORR), Seattle, WA, USA, 24–26 June 2013; IEEE: Piscataway, NJ, USA, 2013; pp. 1–6. [Google Scholar]
- Yao, L.; Xu, H.; Li, A. Kinect-based rehabilitation exercises system: Therapist involved approach. Biomed. Mater. Eng. 2014, 24, 2611–2618. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.H.; Helsper, J.; Yang, C.; Grzegorzek, M. An automatic vision-based monitoring system for accurate Vojta-therapy. In Proceedings of the IEEE/ACIS 15th International Conference on Computer and Information Science (ICIS), Okayama, Japan, 26–29 June 2016; IEEE: Piscataway, NJ, USA, 2016; pp. 1–6. [Google Scholar]
- Khan, M.H.; Shirahama, K.; Farid, M.S.; Grzegorzek, M. Multiple human detection in depth images. In Proceedings of the IEEE 18th International Workshop on Multimedia Signal Processing (MMSP), Montreal, QC, Canada, 21–23 September 2016; IEEE: Piscataway, NJ, USA, 2016; pp. 1–6. [Google Scholar]
- Zhang, W.; Tomizuka, M.; Byl, N. A wireless human motion monitoring system for smart rehabilitation. J. Dyn. Syst. Meas. Control 2016, 138, 111004. [Google Scholar] [CrossRef]
- Qi, B.; Banerjee, S. GonioSense: A wearable-based range of motion sensing and measurement system for body joints: Poster. In Proceedings of the 22nd Annual International Conference on Mobile Computing and Networking, New York, NY, USA, 3–7 October 2016. [Google Scholar]
- Rivolta, M.W.; Aktaruzzaman, M.; Rizzo, G.; Lafortuna, C.L.; Ferrarin, M.; Bovi, G.; Bonardi, D.R.; Caspani, A.; Sassi, R. Evaluation of the Tinetti score and fall risk assessment via accelerometry-based movement analysis. Artif. Intell. Med. 2019, 95, 38–47. [Google Scholar] [CrossRef] [PubMed]
- Paolini, G.; Peruzzi, A.; Mirelman, A.; Cereatti, A.; Gaukrodger, S.; Hausdorff, J.M.; Della Croce, U. Validation of a method for real time foot position and orientation tracking with Microsoft Kinect technology for use in virtual reality and treadmill based gait training programs. IEEE Trans. Neural Syst. Rehabil. Eng. 2014, 22, 997–1002. [Google Scholar] [CrossRef]
- Leder, R.S.; Azcarate, G.; Savage, R.; Savage, S.; Sucar, L.E.; Reinkensmeyer, D.; Toxtli, C.; Roth, E.; Molina, A. Nintendo Wii remote for computer simulated arm and wrist therapy in stroke survivors with upper extremity hemipariesis. In Proceedings of the Virtual Rehabilitation, Vancouver, BC, Canada, 25–27 August 2008; IEEE: Piscataway, NJ, USA, 2008; p. 74. [Google Scholar]
- Chen, C.C.; Liu, C.Y.; Ciou, S.H.; Chen, S.C.; Chen, Y.L. Digitized Hand Skateboard Based on IR-Camera for Upper Limb Rehabilitation. J. Med. Syst. 2017, 41, 36. [Google Scholar] [CrossRef]
- Colyer, S.L.; Evans, M.; Cosker, D.P.; Salo, A.I. A review of the evolution of vision-based motion analysis and the integration of advanced computer vision methods towards developing a markerless system. Sports Med. Open 2018, 4, 24. [Google Scholar] [CrossRef]
- Bernardina, G.R.D.; Monnet, T.; Pinto, H.T.; de Barros, R.M.L.; Cerveri, P.; Silvatti, A.P. Are Action Sport Cameras Accurate Enough for 3D Motion Analysis? A Comparison With a Commercial Motion Capture System. J. Appl. Biomech. 2019, 35, 80–86. [Google Scholar] [CrossRef]
- Barris, S.; Button, C. A Review of Vision-Based Motion Analysis in Sport. Sport. Med. 2008, 38, 1025–1043. [Google Scholar] [CrossRef]
- Moeslund, T.B.; Hilton, A.; Kruger, V. A survey of advances in vision-based human motion capture and analysis. Comput. Vis. Image Underst. 2006, 104, 90–126, Special Issue on Modeling People: Vision-based understanding of a person’s shape, appearance, movement and behaviour. [Google Scholar] [CrossRef]
- Khan, M.H.; Grzegorzek, M. Vojta-Therapy: A Vision-Based Framework to Recognize the Movement Patterns. Int. J. Softw. Innovat. 2017, 5, 18–32. [Google Scholar] [CrossRef]
- Xiao, J.; Moriyama, T.; Kanade, T.; Cohn, J.F. Robust full-motion recovery of head by dynamic templates and re-registration techniques. Int. J. Imaging Syst. Technol. 2003, 13, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.H.; Helsper, J.; Boukhers, Z.; Grzegorzek, M. Automatic recognition of movement patterns in the vojta-therapy using RGB-D data. In Proceedings of the IEEE International Conference on Image Processing (ICIP), Phoenix, AZ, USA, 25–28 September 2016; IEEE: Piscataway, NJ, USA, 2016; pp. 1235–1239. [Google Scholar]
- Shotton, J.; Fitzgibbon, A.; Cook, M.; Sharp, T.; Finocchio, M.; Moore, R.; Kipman, A.; Blake, A. Real-time human pose recognition in parts from single depth images. Commun. ACM 2013, 56, 116–124. [Google Scholar] [CrossRef]
- Hesse, N.; Stachowiak, G.; Breuer, T.; Arens, M. Estimating Body Pose of Infants in Depth Images Using Random Ferns. In Proceedings of the IEEE International Conference on Computer Vision Workshop (ICCVW), Santiago, Chile, 7–13 December 2015. [Google Scholar]
- Evett, L.; Burton, A.; Battersby, S.; Brown, D.; Sherkat, N.; Ford, G.; Liu, H.; Standen, P. Dual Camera Motion Capture for Serious Games in Stroke Rehabilitation. In Proceedings of the IEEE 1st International Conference on Serious Games and Applications for Health (SeGAH), Braga, Portugal, 16–18 November 2011; IEEE Computer Society: Piscataway, NJ, USA, 2011; pp. 1–4. [Google Scholar]
- Stahl, A.; Schellewald, C.; Stavdahl, Ø.; Aamo, O.M.; Adde, L.; Kirkerod, H. An optical flow-based method to predict infantile cerebral palsy. IEEE Trans. Neural Syst. Rehabil. Eng. 2012, 20, 605–614. [Google Scholar] [CrossRef]
- Khan, M.H.; Farid, M.S.; Grzegorzek, M. A generic codebook based approach for gait recognition. Multimed. Tools Appl. 2019, 78, 35689–35712. [Google Scholar] [CrossRef]
- Khan, M.H.; Farid, M.S.; Grzegorzek, M. Spatiotemporal features of human motion for gait recognition. Multimed. Tools Appl. 2019, 13, 369–377. [Google Scholar] [CrossRef]
- Wu, K. Using Human Skeleton to Recognizing Human Exercise by Kinect’s Camera. Master’s Thesis, Department of Computer Science and Information Engineering, National Taipei University of Technology, Taipei, Taiwan, 2011. [Google Scholar]
- Guerrero, C.; Uribe-Quevedo, A. Kinect-based posture tracking for correcting positions during exercise. Stud. Health Technol. Inform. 2013, 184, 158–160. [Google Scholar]
- Luna-Oliva, L.; Ortiz-Gutiérrez, R.M.; Cano-de la Cuerda, R.; Piédrola, R.M.; Alguacil-Diego, I.M.; Sánchez-Camarero, C.; Culebras, M.; del Carmen, M. Kinect Xbox 360 as a therapeutic modality for children with cerebral palsy in a school environment: A preliminary study. NeuroRehab 2013, 33, 513–521. [Google Scholar] [CrossRef]
- Acosta, I.P. Upper Limb Rehabilitation of Stroke Patients Using Kinect and Computer Games. Ph.D. Thesis, School of Computing, University of Utah, Salt Lake City, UT, USA, 2012. [Google Scholar]
- Chang, C.Y.; Lange, B.; Zhang, M.; Koenig, S.; Requejo, P.; Somboon, N.; Sawchuk, A.A.; Rizzo, A.A. Towards pervasive physical rehabilitation using Microsoft Kinect. In Proceedings of the 6th International Conference on Pervasive Computing Technologies for Healthcare (PervasiveHealth) and Workshops, San Diego, CA, USA, 21–24 May 2012; pp. 159–162. [Google Scholar]
- Da Gama, A.; Fallavollita, P.; Teichrieb, V.; Navab, N. Motor rehabilitation using Kinect: A systematic review. Games Health J. 2015, 4, 123–135. [Google Scholar] [CrossRef]
- Mousavi Hondori, H.; Khademi, M. A Review on Technical and Clinical Impact of Microsoft Kinect on Physical Therapy and Rehabilitation. J. Med. Eng. 2014, 2014, 846514. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.H. Human Activity Analysis in Visual Surveillance and Healthcare; Logos Verlag Berlin GmbH: Berlin, Germany, 2018; Volume 45. [Google Scholar]
- Wang, Q.; Markopoulos, P.; Yu, B.; Chen, W.; Timmermans, A. Interactive wearable systems for upper body rehabilitation: A systematic review. J. Neuroeng. Rehabil. 2017, 14, 20. [Google Scholar] [CrossRef] [PubMed]
- Fenu, G.; Steri, G. IMU based post-traumatic rehabilitation assessment. In Proceedings of the 2010 3rd International Symposium on Applied Sciences in Biomedical and Communication Technologies (ISABEL 2010), Rome, Italy, 7–10 November 2010; IEEE: Piscataway, NJ, USA, 2010; pp. 1–5. [Google Scholar]
- Luo, Z.; Lim, C.K.; Yang, W.; Tee, K.Y.; Li, K.; Gu, C.; Nguen, K.D.; Chen, I.M.; Yeo, S.H. An interactive therapy system for arm and hand rehabilitation. In Proceedings of the 2010 IEEE Conference on Robotics, Automation and Mechatronics, Singapore, 28–30 June 2010; IEEE: Piscataway, NJ, USA, 2010; pp. 9–14. [Google Scholar]
- Hamdi, M.M.; Awad, M.I.; Abdelhameed, M.M.; Tolbah, F.A. Lower limb motion tracking using IMU sensor network. In Proceedings of the 2014 Cairo International Biomedical Engineering Conference (CIBEC), Giza, Egypt, 11–13 December 2014; IEEE: Piscataway, NJ, USA, 2014; pp. 28–33. [Google Scholar]
- Heinze, F.; Hesels, K.; Breitbach-Faller, N.; Schmitz-Rode, T.; Disselhorst-Klug, C. Movement analysis by accelerometry of newborns and infants for the early detection of movement disorders due to infantile cerebral palsy. Med. Biol. Eng. Comput. 2010, 48, 765–772. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.H.; Chen, P.C.; Liu, K.C.; Chan, C.T. Wearable sensor-based rehabilitation exercise assessment for knee osteoarthritis. Sensors 2015, 15, 4193–4211. [Google Scholar] [CrossRef]
- Tseng, Y.C.; Wu, C.H.; Wu, F.J.; Huang, C.F.; King, C.T.; Lin, C.Y.; Sheu, J.P.; Chen, C.Y.; Lo, C.Y.; Yang, C.W.; et al. A wireless human motion capturing system for home rehabilitation. In Proceedings of the International Conference on Mobile Data Management: Systems, Services and Middleware (MDM), Taipei, Taiwan, 18–20 May 2009; IEEE: Piscataway, NJ, USA, 2009; pp. 359–360. [Google Scholar]
- Chen, B.R.; Patel, S.; Buckley, T.; Rednic, R.; McClure, D.J.; Shih, L.; Tarsy, D.; Welsh, M.; Bonato, P. A Web-Based System for Home Monitoring of Patients with Parkinsonś Disease Using Wearable Sensors. IEEE Trans. Biomed. Eng. 2011, 58, 831–836. [Google Scholar] [CrossRef]
- Hester, T.; Hughes, R.; Sherrill, D.M.; Knorr, B.; Akay, M.; Stein, J.; Bonato, P. Using wearable sensors to measure motor abilities following stroke. In Proceedings of the International Workshop on Wearable and Implantable Body Sensor Networks, Cambridge, MA, USA, 3–5 April 2006; IEEE: Piscataway, NJ, USA, 2006; p. 4. [Google Scholar]
- Wang, Q.; Chen, W.; Timmermans, A.A.; Karachristos, C.; Martens, J.B.; Markopoulos, P. Smart Rehabilitation Garment for posture monitoring. In Proceedings of the 37th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Milan, Italy, 25–29 August 2015; IEEE: Piscataway, NJ, USA, 2015; pp. 5736–5739. [Google Scholar]
- Bleser, G.; Steffen, D.; Weber, M.; Hendeby, G.; Stricker, D.; Fradet, L.; Marin, F.; Ville, N.; Carré, F. A personalized exercise trainer for the elderly. J. Ambient Intell. Smart Environ. 2013, 5, 547–562. [Google Scholar] [CrossRef]
- Ajdaroski, M.; Tadakala, R.; Nichols, L.; Esquivel, A. Validation of a Device to Measure Knee Joint Angles for a Dynamic Movement. Sensors 2020, 20, 1747. [Google Scholar] [CrossRef]
- Allseits, E.; Kim, K.J.; Bennett, C.; Gailey, R.; Gaunaurd, I.; Agrawal, V. A novel method for estimating knee angle using two leg-mounted gyroscopes for continuous monitoring with mobile health devices. Sensors 2018, 18, 2759. [Google Scholar] [CrossRef]
- Red Cross Children’s Hospital, Siegen, Germany. Available online: http://www.drk-kinderklinik.de (accessed on 1 December 2018).
- Research Group for Pattern Recognition, University of Siegen, Germany. Available online: http://www.ivg.informatik.uni-siegen.de/ (accessed on 5 June 2020).
- Garrido-Jurado, S.; Muñoz-Salinas, R.; Madrid-Cuevas, F.; Marín-Jiménez, M. Automatic generation and detection of highly reliable fiducial markers under occlusion. Pattern Recognit. 2014, 47, 2280–2292. [Google Scholar] [CrossRef]
- Patro, S.; Sahu, K.K. Normalization: A preprocessing stage. arXiv 2015, arXiv:1503.06462. [Google Scholar] [CrossRef]
- Reddi, S. Radial and angular moment invariants for image identification. IEEE Trans. Pattern Anal. Mach. Intell. 1981, 2, 240–242. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.K. Visual pattern recognition by moment invariants. IRE Trans. Inf. Theory 1962, 8, 179–187. [Google Scholar]
- Grewal, M.S. Kalman Filtering; Springer: Berlin, Germany, 2011. [Google Scholar]
- Fox, D.; Hightower, J.; Lao, L.; Schulz, D.; Borriello, G. Bayesian Filters for Location Estimation; Technical Report; University of Washington, Dept. of Computer Science and Engineering: Seattle, WA, USA, 2003. [Google Scholar]
- Alshehri, Y. Gait Analysis Post Anterior Cruciate Ligament Reconstruction Using Inertial Sensors: A Longitudinal Study. Ph.D. Thesis, University of Kansas, Lawrence, KS, USA, 2019. [Google Scholar]
- Lamine, H.; Bennour, S.; Laribi, M.; Romdhane, L.; Zaghloul, S. Evaluation of calibrated kinect gait kinematics using a vicon motion capture system. Comput. Methods Biomech. Biomed. Eng. 2017, 20, 111–112. [Google Scholar] [CrossRef] [PubMed]
- Kang, G.E.; Gross, M.M. Concurrent validation of magnetic and inertial measurement units in estimating upper body posture during gait. Measurement 2016, 82, 240–245. [Google Scholar] [CrossRef]
- Bersamira, J.N.; De Chavez, R.J.A.; Salgado, D.D.S.; Sumilang, M.M.C.; Valles, E.R.; Roxas, E.A.; dela Cruz, A.R. Human Gait Kinematic Estimation based on Joint Data Acquisition and Analysis from IMU and Depth-Sensing Camera. In Proceedings of the 2019 IEEE 11th International Conference on Humanoid, Nanotechnology, Information Technology, Communication and Control, Environment, and Management (HNICEM), Laoag, Philippines, 29 November–1 December 2019; pp. 1–6. [Google Scholar]

| Method | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hesse [39] | 33.0 | 27.0 | 73.0 | 24.0 | 20.0 | 44.0 | 149.0 | 12.0 | 45.0 | 49.0 | 28.0 | 30.0 | 41.0 |
| Khan [10] | 11.9 | 11.0 | 11.4 | 11.2 | 12.4 | 11.9 | 14.4 | 11.2 | 11.9 | 11.7 | 14.0 | 12.8 | 12.7 |
| Proposed | 1.7 | 1.9 | 2.0 | 2.4 | 3.3 | 3.0 | 1.8 | 3.9 | 1.8 | 2.9 | 4.0 | 3.0 | 2.7 |
| 0.63 | 0.58 | 1.10 | 1.16 | 0.85 | 0.65 | 0.64 | 0.71 | 0.79 |
| Activities in the Proposed Algorithm | Computational Complexity |
|---|---|
| Recording of synchronized RGB and depth streams | 25 fps |
| Average processing time per frame | 59.84 ms |
| Number of processed frames per second | 16.71 fps |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khan, M.H.; Zöller, M.; Farid, M.S.; Grzegorzek, M. Marker-Based Movement Analysis of Human Body Parts in Therapeutic Procedure. Sensors 2020, 20, 3312. https://doi.org/10.3390/s20113312
Khan MH, Zöller M, Farid MS, Grzegorzek M. Marker-Based Movement Analysis of Human Body Parts in Therapeutic Procedure. Sensors. 2020; 20(11):3312. https://doi.org/10.3390/s20113312
Chicago/Turabian StyleKhan, Muhammad Hassan, Martin Zöller, Muhammad Shahid Farid, and Marcin Grzegorzek. 2020. "Marker-Based Movement Analysis of Human Body Parts in Therapeutic Procedure" Sensors 20, no. 11: 3312. https://doi.org/10.3390/s20113312
APA StyleKhan, M. H., Zöller, M., Farid, M. S., & Grzegorzek, M. (2020). Marker-Based Movement Analysis of Human Body Parts in Therapeutic Procedure. Sensors, 20(11), 3312. https://doi.org/10.3390/s20113312






