Multiplexed Electrochemical Immunosensors for Clinical Biomarkers
Abstract
:1. Introduction
2. Multiplexed Electrochemical Immunosensors Involving Electrode Arrays
3. Multiplexed Electrochemical Immunosensors Using Barcode Configurations
4. Microfluidic Devices
5. General Conclusions and Future Prospects
Acknowledgments
Conflicts of Interest
References
- Munge, B.S.; Stracensky, T.; Gamez, K.; DiBiase, D.; Rusling, J.F. Multiplex immunosensor arrays for electrochemical detection of cancer biomarker proteins. Electroanalysis 2016, 28, 2644–2658. [Google Scholar] [CrossRef]
- Wang, J. Electrochemical biosensors: Towards point-of-care cancer diagnostics. Biosens. Bioelectron. 2006, 21, 1887–1892. [Google Scholar] [CrossRef] [PubMed]
- Chandra, P. Miniaturized multiplex electrochemical biosensor in clinical bioanalysis. J. Bioanal. Biomed. 2013, 5, e122. [Google Scholar]
- Rusling, J.F. Multiplexed electrochemical protein detection and translation to personalized cancer diagnostics. Anal. Chem. 2013, 85, 5304–5310. [Google Scholar] [CrossRef] [PubMed]
- Gubala, V.; Klein, R.; Templeton, D.M.; Schwenk, M. Immunodiagnosis and immunosensor design. Pure Appl. Chem. 2014, 86, 1539–1571. [Google Scholar] [CrossRef]
- Zhu, Q.; Chai, Y.; Zhuo, Y.; Yuan, R. Ultrasensitive simultaneous detection of four biomarkers based on hybridization chain reaction and biotin–streptavidin signal amplification strategy. Biosens. Bioelectron. 2015, 68, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Wu, J.; Ju, H.; Yan, F. Multiplexed electrochemical immunoassay using streptavidin/nanogold/carbon nanohorn as a signal tag to induce silver deposition. Anal. Chim. Acta 2014, 847, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Du, D.; Wang, J.; Lu, D.; Dohnalkova, A.; Lin, Y. Multiplexed electrochemical immunoassay of phosphorylated proteins based on enzyme-functionalized gold nanorod labels and electric field-driven acceleration. Anal. Chem. 2011, 83, 6580–6585. [Google Scholar] [CrossRef] [PubMed]
- Wan, Y.; Su, Y.; Zhu, X.; Liu, G.; Fan, C. Development of electrochemical immunosensors towards point of care diagnostics. Biosens. Bioelectron. 2013, 47, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Liu, N. Design of immunoprobes for electrochemical multiplexed tumor marker detection. Expert Rev. Mol. Diagn. Early online 2015, 15, 1–9. [Google Scholar]
- Kokkinos, C.; Economou, A.; Prodromidis, M.I. Electrochemical immunosensors: Critical survey of different architectures and transduction strategies. TrAC Trends Anal. Chem. 2016, 79, 88–105. [Google Scholar] [CrossRef]
- Yang, Z.H.; Zhuo, Y.; Chai, Y.Q.; Yuan, R. High throughput immunosenor based on multi-label strategy and a novel array electrode. Sci. Rep. 2014, 4, 4747. [Google Scholar] [CrossRef] [PubMed]
- Tang, D.; Hou, L.; Niessner, R.; Xu, M.; Gao, Z.; Knopp, D. Multiplexed electrochemical immunoassay of biomarkers using metal sulfide quantum dot nanolabels and trifunctionalized magnetic beads. Biosens. Bioelectron. 2013, 46, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Liu, N.; Ma, Z. Platinum porous nanoparticles hybrid with metal ions as probes for simultaneous detection of multiplex cancer biomarkers. Biosens. Bioelectron. 2014, 53, 324–329. [Google Scholar] [CrossRef] [PubMed]
- Delaney, J.L.; Hogan, C.F.; Tian, J.F.; Shen, W. Electrogenerated chemiluminescence detection in paper-based microfluidic sensors. Anal. Chem. 2011, 83, 1300–1306. [Google Scholar] [CrossRef] [PubMed]
- Chikkaveeraiah, B.V.; Mania, V.; Patel, V.; Gutkind, J.S.; Rusling, J.F. Microfluidic electrochemical immunoarray for ultrasensitive detection of two cancer biomarker proteins in serum. Biosens. Bioelectron. 2011, 26, 4477–4483. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Tang, D.; Niessner, R.; Chen, G.; Knopp, D. Magneto-controlled graphene immunosensing platform for simultaneous multiplexed electrochemical immunoassay using distinguishable signal tags. Anal. Chem. 2011, 83, 5407–5414. [Google Scholar] [CrossRef] [PubMed]
- Wan, Y.; Deng, W.; Su, Y.; Zhu, X.; Peng, C.; Hu, H.; Peng, H.; Song, S.; Fan, C. Carbon nanotube-based ultrasensitive multiplexing electrochemical immunosensor for cancer biomarkers. Biosens. Bioelectron. 2011, 30, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Lu, C.-Y.; Zhou, H.; Xu, J.-J.; Chen, H.-Y. Flexible gold electrode array for multiplexed immunoelectrochemical measurement of three protein biomarkers for prostate cancer. ACS Appl. Mater. Interfaces 2014, 6, 20137–20143. [Google Scholar] [CrossRef] [PubMed]
- Cui, Z.; Wu, D.; Zhang, Y.; Ma, H.; Li, H.; Du, B.; Wei, Q.; Ju, H. Ultrasensitive electrochemical immunosensors for multiplexed determination using mesoporous platinum nanoparticles as nonenzymatic labels. Anal. Chim. Acta 2014, 807, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Lai, G.; Wang, L.; Wu, J.; Ju, H.; Yan, F. Electrochemical stripping analysis of nanogold label-induced silver deposition for ultrasensitive multiplexed detection of tumor markers. Anal. Chim. Acta 2012, 721, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Lai, G.; Wu, J.; Ju, H.; Yan, F. Streptavidin-functionalized silver-nanoparticle-enriched carbon nanotube tag for ultrasensitive multiplexed detection of tumor markers. Adv. Funct. Mater. 2011, 21, 2938–2943. [Google Scholar] [CrossRef]
- Ge, S.; Yu, F.; Ge, L.; Yan, M.; Yu, J.; Chen, D. Disposable electrochemical immunosensor for simultaneous assay of a panel of breast cancer tumor markers. Analyst 2012, 137, 4727–4733. [Google Scholar] [CrossRef] [PubMed]
- Eletxigerra, U.; Martinez-Perdiguero, J.; Merino, S.; Barderas, R.; Ruiz-Valdepeñas Montiel, V.; Villalonga, R.; Pingarrón, J.M.; Campuzano, S. Electrochemical magnetoimmunosensor for progesterone receptor determination. Application to the simultaneous detection of estrogen and progesterone breast-cancer related receptors in raw cell lysates. Electroanalysis 2016, 28, 1787–1794. [Google Scholar] [CrossRef]
- Gupta, R.K.; Pandya, R.; Sieffert, T.; Meyyappan, M.; Koehne, J.E. Multiplexed electrochemical immunosensor for label-free detection of cardiac markers using a carbon nanofiber array chip. J. Electroanal. Chem. 2016, 773, 53–62. [Google Scholar] [CrossRef]
- Esteban-Fernández de Ávila, B.; Escamilla-Gómez, V.; Lanzone, V.; Campuzano, S.; Pedrero, M.; Compagnone, D.; Pingarrón, J.M. Multiplexed determination of amino-terminal pro-B type natriuretic peptide and C-reactive protein cardiac biomarkers in human serum at a disposable electrochemical magnetoimmunosensor. Electroanalysis 2014, 26, 254–261. [Google Scholar] [CrossRef]
- Zhang, G.-J.; Luo, Z.H.H.; Huang, M.J.; Ang, J.J.; Kang, T.G.; Ji, H. An integrated chip for rapid, sensitive, and multiplexed detection of cardiacbiomarkers from fingerprick blood. Biosens. Bioelectron. 2011, 28, 459–463. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Tirado, R.; Salvo, C.; González-Cortés, A.; Yáñez-Sedeño, P.; Langa, F.; Pingarrón, J.M. Electrochemical immunosensor for simultaneous determination of interleukin-1 beta and tumor necrosis factor alpha in serum and salivausing dual screen printed electrodes modified with functionalized double-walled carbon nanotubes. Anal. Chim. Acta 2017, 959, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Baraket, A.; Lee, M.; Zine, N.; Trivella, M.G.; Zabala, M.; Bausells, J.; Sigaud, M.; Jaffrezic-Renault, N.; Errachid, A. A fully integrated electrochemical BioMEMS fabrication process for cytokine detection: Application for heart failure. Proc. Eng. 2014, 87, 377–379. [Google Scholar] [CrossRef]
- Martínez-García, G.; Agüí, L.; Yáñez-Sedeño, P.; Pingarrón, J.M. Multiplexed electrochemical immunosensing of obesity-related hormones at grafted graphene-modified electrodes. Electrochim. Acta 2016, 202, 209–215. [Google Scholar] [CrossRef]
- Moreno-Guzmán, M.; González-Cortés, A.; Yáñez-Sedeño, P.; Pingarrón, J.M. Multiplexed ultrasensitive determination of adreno-corticotropinand cortisol hormones at a dual electrochemical immunosensor. Electroanalysis 2012, 24, 1100–1108. [Google Scholar] [CrossRef]
- Neves, M.M.P.S.; González-García, M.B.; Delerue-Matos, C.; Costa-García, A. Multiplexed electrochemical immunosensor for detection of celiac disease serological markers. Sens. Actuators B Chem. 2013, 187, 33–39. [Google Scholar] [CrossRef]
- Chen, X.; Jia, X.; Han, J.; Ma, J.; Man, Z. Electrochemical immunosensor for simultaneous detection of multiplex cancer biomarkers based on graphene nanocomposites. Biosens. Bioelectron. 2013, 50, 356–361. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Ma, Z. Multiplexed electrochemical immunoassay of biomarkers using chitosan nanocomposites. Biosens. Bioelectron. 2014, 5, 343–349. [Google Scholar] [CrossRef] [PubMed]
- Feng, D.; Li, L.; Han, X.; Fang, X.; Li, X; Zhang, Y. Simultaneous electrochemical detection of multiple tumor markers using functionalized graphene nanocomposites as non-enzymatic labels. Sens. Actuators B Chem. 2014, 201, 360–368. [Google Scholar] [CrossRef]
- Lai, W.Q.; Zhuang, J.Y.; Tang, J.; Chen., G.N.; Tang, D.P. One-step electrochemical immunosensing for simultaneous detection of two biomarkers using thionine and ferrocene as distinguishable signal tags. Microchim. Acta 2012, 178, 357–365. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, L.; Lu, J.; Ge, L.; Ge, S.; Yan, M.; Song, X.; Yu, J. Triple catalysis amplification strategy for simultaneous multiplexed electrochemical immunoassays based on cactus-like MnO2 functionalized nanoporous gold. Sens. Actuators B Chem. 2013, 186, 545–549. [Google Scholar] [CrossRef]
- Zhang, X.; Ren, X.; Cao, W.; Li, Y.; Du, B.; Wei, Q. Simultaneous electrochemical immunosensor based on water-soluble polythiophene derivative and functionalized magnetic material. Anal. Chim. Acta 2014, 845, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.F.; Tang, D.P.; Lou, F.M.; Yang, X.M.; Chen, G.N. Simultaneous electrochemical multiplexed immunoassay of biomarkers based on multifunctionalized graphene nanotags. Chem. Electro. Chem. 2014, 1, 441–447. [Google Scholar] [CrossRef]
- Jia, X.; Liu, Z.; Liu, N.; Ma, Z. A label-free immunosensor based on graphene nanocomposites for simultaneous multiplexed electrochemical determination of tumor markers. Biosens. Bioelectron. 2014, 53, 160–166. [Google Scholar] [CrossRef] [PubMed]
- Lu, W.; Tao, L.; Wang, Y.; Cao, Z.; Ge, J.; Dong, J.; Qian, W. An electrochemical immunosensor for simultaneous multiplexed detection of two lung cancer biomarkers using Au nanoparticles coated resin microspheres composed of L-tryptophan and caffeic acid. Ionics 2015, 21, 1141–1152. [Google Scholar] [CrossRef]
- Ge, X.; Zhang, A.; Lin, Y.; Du, D. Simultaneous immunoassay of phosphorylated proteins based on apoferritin templated metallic phosphates as voltammetrically distinguishable signal reporters. Biosens. Bioelectron. 2016, 80, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Gan, N.; Zhang, H.; Li, T.; Qiao, L.; Cao, Y.; Su, X.; Jiang, S. Simultaneous electrochemical immunoassay using graphene–Au grafted recombinant apoferritin-encoded metallic labels as signal tags and dual-template magnetic molecular imprinted polymer as capture probes. Biosens. Bioelectron. 2015, 65, 78–82. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Wang, Y.; Zhu, J.J. Simultaneous detection of tumor cell apoptosis regulators Bcl-2 and Bax through a dual-signal-marked electrochemical immunosensor. ACS Appl. Mater. Interfaces 2016, 8, 7674–7682. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Huang, X.; Zeng, Q.; Wang, L. Metallic nanocrystallites-incorporated ordered mesoporous carbon as labels for a sensitive simultaneous multianalyte electrochemical immunoassay. Biosens. Bioelectron. 2015, 73, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Gan, N.; Zhou, J.; Xiong, P.; Cao, Y.; Li, T.; Pan, D.; Jiang, S. Signal amplification for multianalyte electrochemical immunoassay with bidirectional stripping voltammetry using metal-enriched polymer nanolabels. Sens. Actuators B Chem. 2014, 197, 244–253. [Google Scholar] [CrossRef]
- Xu, T.; Jia, X.; Chen, X.; Man, Z. Simultaneous electrochemical detection of multiple tumor markers using metal ions tagged immunocolloidal gold. Biosens. Bioelectron. 2014, 56, 174–179. [Google Scholar] [CrossRef] [PubMed]
- Feng, D.; Li, L.; Zhao, J.; Zhang, Y. Simultaneous electrochemical detection of multiple biomarkers using gold nanoparticles decorated multiwall carbon nanotubes as signal enhancers. Anal. Biochem. 2015, 482, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Feng, D.; Zhang, Y. Simultaneous detection of two tumor markers using silver and gold nanoparticles decorated carbon nanospheres as labels. Anal. Biochem. 2016, 505, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Xu, T.; Liu, N.; Yuan, J.; Ma, Z. Triple tumor markers assay based on carbon–gold nanocomposite. Biosens. Bioelectron. 2015, 70, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Qing, Y.; Shan, J.; Jin, F.; Yuan, R.; Wang, D. Cation-exchange antibody labeling for simultaneous electrochemical detection of tumor markers CA15-3 and CA19-9. Microchim. Acta 2013, 180, 651–657. [Google Scholar] [CrossRef]
- Liu, Z.; Rong, Q.; Ma, Z.; Han, H. One-step synthesis of redox-active polymer/Au nanocomposites for electrochemical immunoassay of multiplexed tumor markers. Biosens. Bioelectron. 2015, 65, 307–313. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Shu, B.; Jiang, B.; Ding, L.; Qi, H.; Yang, M.; Qu, F. Ultrasensitive multiplexed protein biomarker detection based on electrochemical tag incorporated polystyrene spheres as label. Sens. Actuators B Chem. 2013, 186, 768–773. [Google Scholar] [CrossRef]
- Zhu, Q.; Chai, Y.; Yuan, R.; Zhuo, Y.; Han, J.; Li, Y.; Liao, N. Amperometric immunosensor for simultaneous detection of three analytes in one interface using dual functionalized graphene sheets integrated with redox-probes as tracer matrixes. Biosens. Bioelectron. 2013, 43, 440–445. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Zhuo, Y.; Chai, Y.; Yuan, R.; Zhang, W.; Zhu, Q. Simultaneous electrochemical detection of multiple tumor markers based on dual catalysis amplification of multi-functionalized onion-like mesoporous graphene sheets. Anal. Chim. Acta 2012, 746, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Z.; Peng, N.; Qing, Y.; Shan, J.; Li, M.; Guan, W.; Dai, N.; Gu, X.; Wang, D. An electrochemical immunosensor for simultaneous multiplexed detection of neuron-specific enolase and pro-gastrin-releasing peptide using liposomes as enhancer. Electrochim. Acta 2011, 56, 5624–5629. [Google Scholar] [CrossRef]
- Wang, Z.; Liu, N.; Feng, F.; Ma, Z. Synthesis of cadmium, lead and copper alginate nanobeads as immunosensing probes for the detection of AFP, CEA and PSA. Biosens. Bioelectron. 2015, 70, 98–105. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Li, T.; Gan, N.; Zhang, H.; Long, N.; Hua, F.; Cao, Y.; Jiang, Q.; Jiang, S. Electrochemical coding for multiplexed immunoassays of biomarkers based on bio-based polymer-nanotags. Electrochim. Acta 2015, 163, 238–245. [Google Scholar] [CrossRef]
- Rong, Q.; Feng, F.; Ma, Z. Metal ions doped chitosan–poly(acrylic acid) nanospheres: Synthesis and their application in simultaneously electrochemical detection of four markers of pancreatic cancer. Biosens. Bioelectron. 2016, 75, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Rama, E.C.; Costa-García, A. Screen-printed electrochemical immunosensors for the detection of cancer and cardiovascular biomarkers. Electroanalysis 2011, 28, 1700–1715. [Google Scholar] [CrossRef]
- Arduini, F.; Micheli, L.; Moscone, D.; Palleschi, G.; Piermarini, S.; Ricci, F.; Volpe, G. Electrochemical biosensors based on nanomodified screen-printed electrodes: Recent applications in clinical analysis. TrAC Trends Anal. Chem. 2016, 79, 114–126. [Google Scholar] [CrossRef]
- Bode, A.M.; Dong, Z. Post-translational modification of p53 in tumorigenesis. Nat. Rev. Cancer 2002, 4, 793–805. [Google Scholar] [CrossRef] [PubMed]
- Sakaguchi, K.; Herrera, J.E.; Saito, S.; Miki, T.; Bustin, M.; Vassilev, A.; Anderson, C.W.; Appela, E. DNA damage activates p53 through a phosphorylation-acetylation cascade. Genes Dev. 2013, 12, 2831–2841. [Google Scholar] [CrossRef]
- Bar, J.; Moskovits, J.; Oren, M. Involvement of stromal p53 in tumor-stroma interactions. Semin. Cell Dev. Biol. 2009, 21, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.S.; Lytton-Jean, A.K.R.; Hurst, S.J.; Mirkin, C.A. Silver nanoparticle-oligonucleotide conjugates based on DNA with triple cyclic disulfide moieties. Nano Lett. 2007, 7, 2112–2115. [Google Scholar] [CrossRef] [PubMed]
- Russo, J.; Russo, I.H. The role of estrogen in the initiation of breast cancer. J. Steroid Biochem. Mol. Biol. 2006, 102, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Katzenellenbogen, B.S.; Katzenellenbogen, J.A. Estrogen receptor transcription and transactivation Estrogen receptor alpha and estrogen receptor beta: Regulation by selective estrogen receptor modulators and importance in breast cancer. Breast Cancer Res. 2000, 2, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Ellmann, S.; Sticht, H.; Thiel, F.; Beckmann, M.W.; Strock, R.; Strissel, P.L. Estrogen and progesterone receptors: from molecular structures to clinical targets. Cell Mol. Life Sci. 2009, 66, 2405–2426. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-J.; Cui, X.; Hilsenbeck, S.G.; Lee, A.V. Progesterone receptor loss correlates with human epidermal growth factor receptor 2 overexpression in estrogen receptor-positive breast cancer. Clin. Cancer Res. 2006, 12, 1013s–1018s. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Zhou, D.M. Cardiac markers and their point-of-care testing for diagnosis of acute myocardial infarction. Clin. Biochem. 2006, 39, 771–780. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Qi, M.; Hutchinson, M.R.; Yang, G.; Goldys, E.M. Recent advances in cytokine detection by immunosensing. Biosens. Bioelectron. 2016, 79, 810–821. [Google Scholar] [CrossRef] [PubMed]
- Stenken, J.A.; Poschenrieder, A.J. Bioanalytical chemistry of cytokines—A review. Anal. Chim. Acta 2015, 853, 95–115. [Google Scholar] [CrossRef] [PubMed]
- Shaftel, S.S.; Griffin, W.S.T.; O’Banion, M.K. The role of interleukin-1 in neuroinflammation and Alzheimer disease: An evolving perspective. J. Neuroinflam. 2008, 5, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Feldmann, M.; Brennan, F.M.; Maini, R.N. Role of cytokines in rheumatoid arthritis. Annu. Rev. Immunol. 1996, 14, 397–440. [Google Scholar] [CrossRef] [PubMed]
- Palladino, M.A.; Bahjat, F.R.; Theodorakis, E.A.; Moldawer, L.L. Anti-TNF-[alpha] therapies: The next generation. Nat. Rev. Drug Discov. 2003, 2, 736–746. [Google Scholar] [CrossRef] [PubMed]
- Krause, C.E.; Otieno, B.A.; Bishop, G.W.; Phadke, G.; Choquette, L.; Lalla, R.V.; Peterson, D.E.; Rusling, J.F. Ultrasensitive microfluidic array for serum proinflammatory cytokines and C-reactive protein to assess oral mucositis risk in cancer patients. Anal. Bioanal. Chem. 2015, 407, 7239–7243. [Google Scholar] [CrossRef] [PubMed]
- Gomes, F.I.F.; Aragao, M.G.B.; Barbosa, F.C.B.; Bezerra, M.M.; Pinto, V.D.P.T.; Chaves, H.V. Inflammatory cytokines interleukin-1β and tumour necrosis factor-α-novel biomarkers for the detection of periodontal diseases: A literature review. J. Oral Maxillofac. Res. 2016, 7, e2. [Google Scholar] [CrossRef] [PubMed]
- Wong, I.P.; Baldock, P.A.; Herzog, H. Gastrointestinal peptides and bone health. Curr. Opin. Endocrinol. Diabetes Obes. 2010, 17, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Hill, B.R.; de Souza, M.J.; Wagstaff, D.A.; Williams, N.I. The impact of weight loss on the 24-h profile of circulating peptide YY and its association with 24-h ghrelin in normal weight premenopausal women. Peptides 2013, 49, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Verma, P.; Maire, P.; Novak, P. Concatenation of electrochemical grafting with chemical or electrochemical modification for preparing electrodes with specific surface functionality. Electrochim. Acta 2011, 56, 3555–3561. [Google Scholar] [CrossRef]
- John, M.; Lila, A.R.; Bandgar, T.; Menon, P.S.; Shah, N.S. Diagnostic efficacy of midnight cortisol and midnight ACTH in the diagnosis and localisation of Cushing’s syndrome. Pituitary 2010, 13, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Abad, J.; Vélez, M.; Santamaría, C.; Guisan, J.; Matheus, P.; Vázquez, L.; Gazaryan, I.; Gorton, L.; Gibson, T.; Fernández, V. Immobilization of peroxidase glycoprotein on gold electrodes modified with mixed epoxy-boronic acid monolayers. J. Am. Chem. Soc. 2002, 124, 12845–12853. [Google Scholar] [CrossRef] [PubMed]
- Ho, J.A.; Hsu, W.A.; Liao, W.C.; Chiu, J.K.; Chen, M.L.; Chang, H.C.; Li, C.C. Ultrasensitive electrochemical detection of biotin using electrically addressable site-oriented antibody immobilization approach via aminophenyl boronic acid. Biosens. Bioelectron. 2011, 26, 1021–1027. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.L.; Lo, M.Y.; Mou, C.Y. PtRu nanoparticles supported on ozone-treated mesoporous carbon thin film as highly active anode materials for direct methanol fuel cells. J. Phys. Chem. C 2009, 113, 16158–16168. [Google Scholar] [CrossRef]
- Du, D.; Zou, Z.X.; Shin, Y.S.; Wang, J.; Wu, H.; Engelhard, M.H.; Liu, J.; Aksay, I.A.; Lin, Y.H. Sensitive immunosensor for cancer biomarker based on dual signal amplification strategy of graphene sheets and multienzyme functionalized carbon nanospheres. Anal. Chem. 2010, 82, 2989–2995. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.; Wang, K.; Wu, L.; Yu, S.H.; Antonietti, M.; Titirici, M.M. Engineering carbon materials from the hydrothermal carbonization process of biomass. Adv. Mater. 2010, 22, 813–828. [Google Scholar] [CrossRef] [PubMed]
- Ensafi, A.A.; Nasr-Esfahani, P.; Heydari-Bafrooei, E.; Rezaei, B. Redox targeting of DNA anchored to MWCNTs and TiO2 nanoparticles dispersed in polydialyldimethylammonium chloride and chitosan. Coll. Surf. B Biointerfaces 2014, 121, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Bavykin, D.V.; Milsom, E.V.; Marken, F.; Kim, D.H.; Marsh, D.H.; Riley, J.; Walsh, F.C.; El-Abiary, K.H.; Lapkin, A.A. A novel cation-binding TiO2 nanotube substrate for electro- and bioelectro-catalysis. Electrochem. Commun. 2005, 7, 1050–1058. [Google Scholar] [CrossRef]
- Suthaus, J.; Adam, N.; Groetzinger, J.; Scheller, J.; Rose-John, S. Viral interleukin-6: Structure, pathophysiology and strategies of neutralization. Eur. J. Cell Biol. 2011, 90, 495–504. [Google Scholar] [CrossRef] [PubMed]
- Ding, H.-S.; Yang, J.; Yang, J.; Ding, J.-W.; Chen, P.; Zhu, P. Interleukin-17 contributes to cardiovascular diseases. Mol. Biol. Rep. 2012, 39, 7473–7478. [Google Scholar] [CrossRef] [PubMed]
- Guthrie, G.J.K.; Roxburgh, C.S.D.; Horgan, P.G.; McMillan, D.C. Does interleukin-6 link explain the link between tumour necrosis, local and systemic inflammatory responses and outcome in patients with colorectal cancer? Cancer Treat. Rev. 2013, 39, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Han, Y.; Fei, G.; Ren, T.; Liu, Z. IL-17 promoted metastasis of non-small-cell lung cancer cells. Immunol. Lett. 2012, 148, 144–150. [Google Scholar] [CrossRef] [PubMed]
- Giri, B.; Pandey, B.; Neupane, B.; Ligler, F.S. Signal amplification strategies for microfluidic immunoassays. TrAC Trends Anal. Chem. 2016, 79, 326–334. [Google Scholar] [CrossRef]
- Bange, A.; Halsall, H.B.; Heinemann, W.R. Microfluidic immunosensor systems. Biosens. Bioelectron. 2005, 20, 2488–2503. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F.; Lu, M.; Wang, W.; Bian, Z.-P.; Zhang, J.-R.; Zhu, J.-J. Electrochemical immunosensor for simultaneous detection of dual cardiac markers based on a poly(dimethylsiloxane)-gold nanoparticles composite microfluidic chip: A proof of principle. Clin. Chem. 2010, 56, 1701–1707. [Google Scholar] [CrossRef] [PubMed]
- Bruzewicz, D.A.; Reches, M.; Whitesides, G.M. Low-cost printing of poly(dimethylsiloxane) barriers to define microchannels in paper. Anal. Chem. 2008, 80, 3387–3392. [Google Scholar] [CrossRef] [PubMed]
- Martinez, W.; Phillips, S.T.; Wiley, B.J.; Gupta, M.; Whitesides, G.M. FLASH: A rapidmethod for prototyping paper-based microfluidic devices. Lab Chip 2008, 8, 2146–2150. [Google Scholar] [CrossRef] [PubMed]
- Hasanzadeh, M.; Shadjouc, N. Electrochemical and photoelectrochemical nano-immunosensing using origami paper based method. Mater. Sci. Eng. C 2016, 61, 979–1001. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Li, L.; Ge, S.; Song, X.; Ge, L.; Yan, M.; Yu, J. Multiplex electrochemical origami immunodevice based on cuboid silver-paper electrode and metal ions tagged nanoporous silver–chitosan. Biosens. Bioelectron. 2014, 56, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Sun, G; Zhang, L; Zhang, Y; Yang, H; Ma, C; Ge, S; Yan, M; Yu, J; Song, X. Multiplexed enzyme-free electrochemical immunosensor based on ZnO nanorods modified reduced graphene oxide-paper electrode and silver deposition-induced signal amplification strategy. Biosens. Bioelectron. 2015, 71, 30–36. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Liu, X. A portable paper-based microfluidic platform for multiplexed electrochemical detection of human immunodeficiency virus and hepatitis C virus antibodies in serum. Biomicrofluidics 2016, 10, 024119. [Google Scholar] [CrossRef] [PubMed]
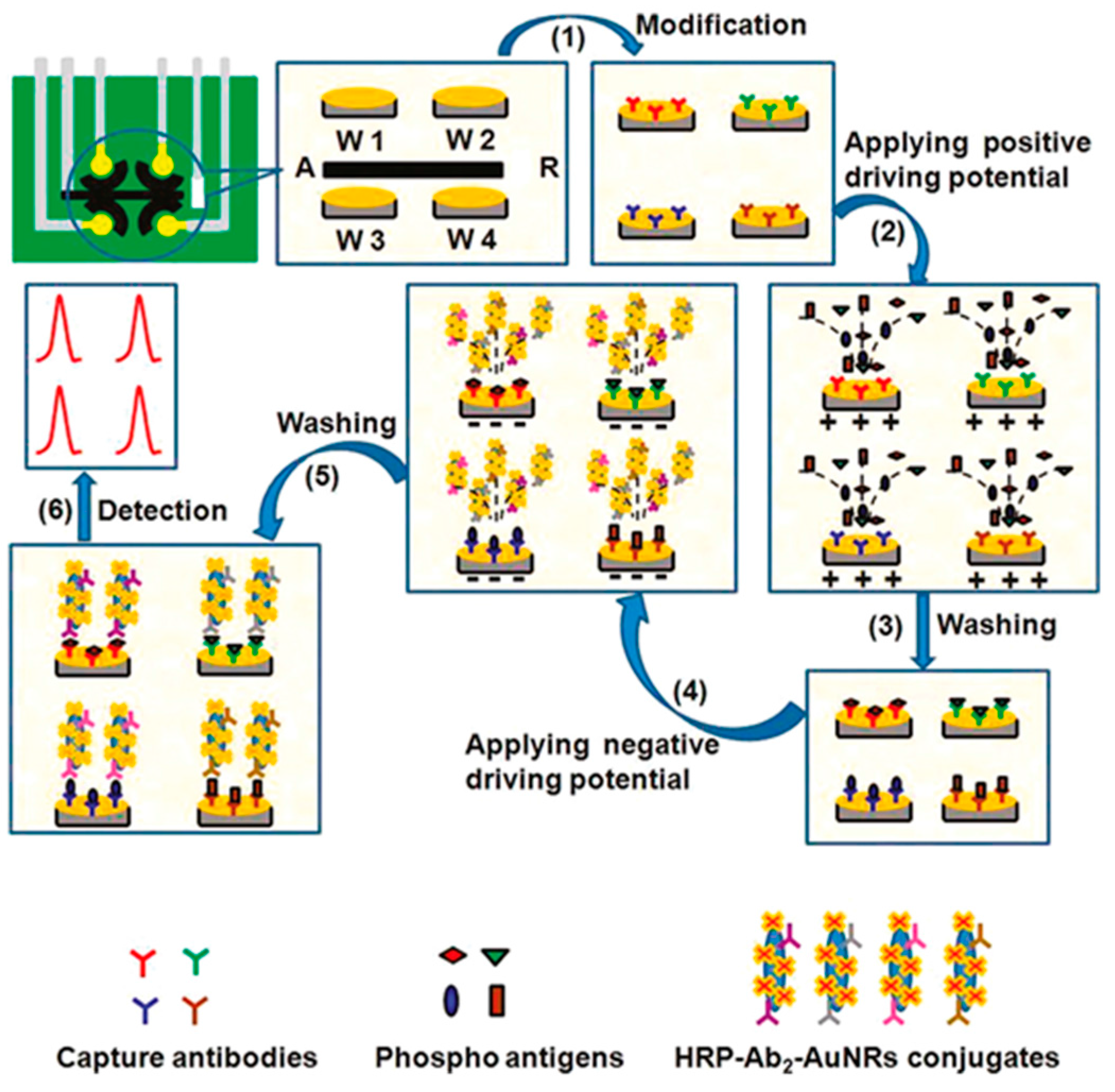
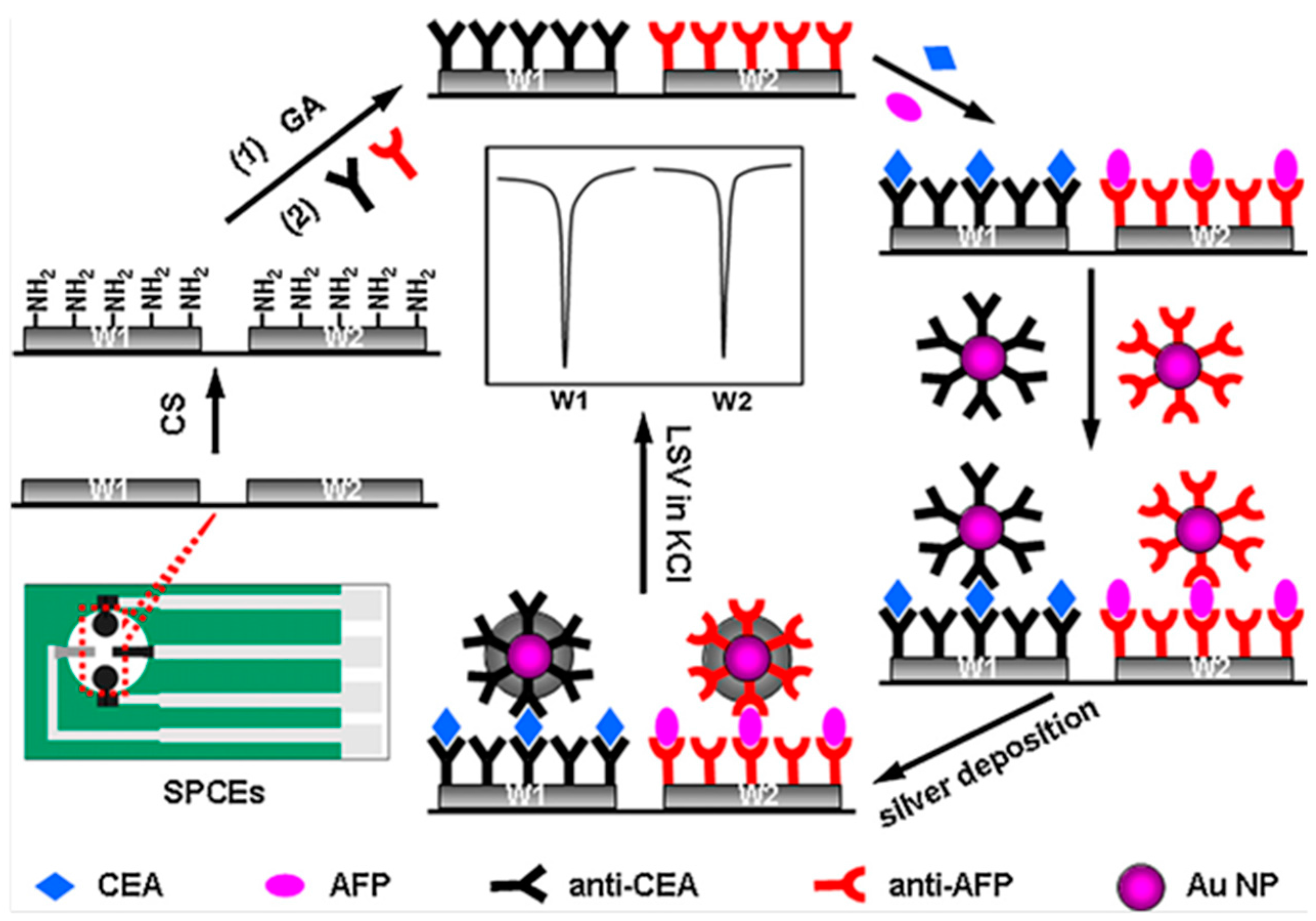
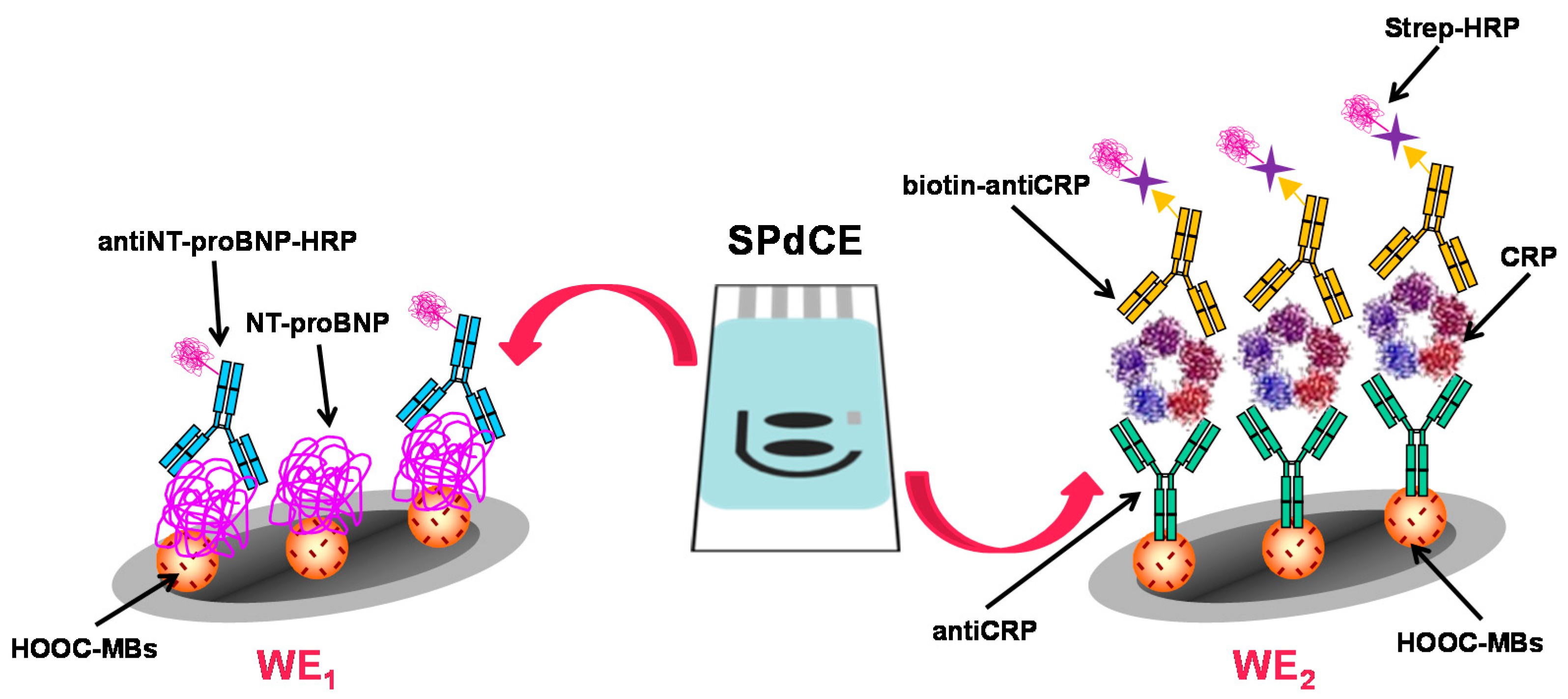
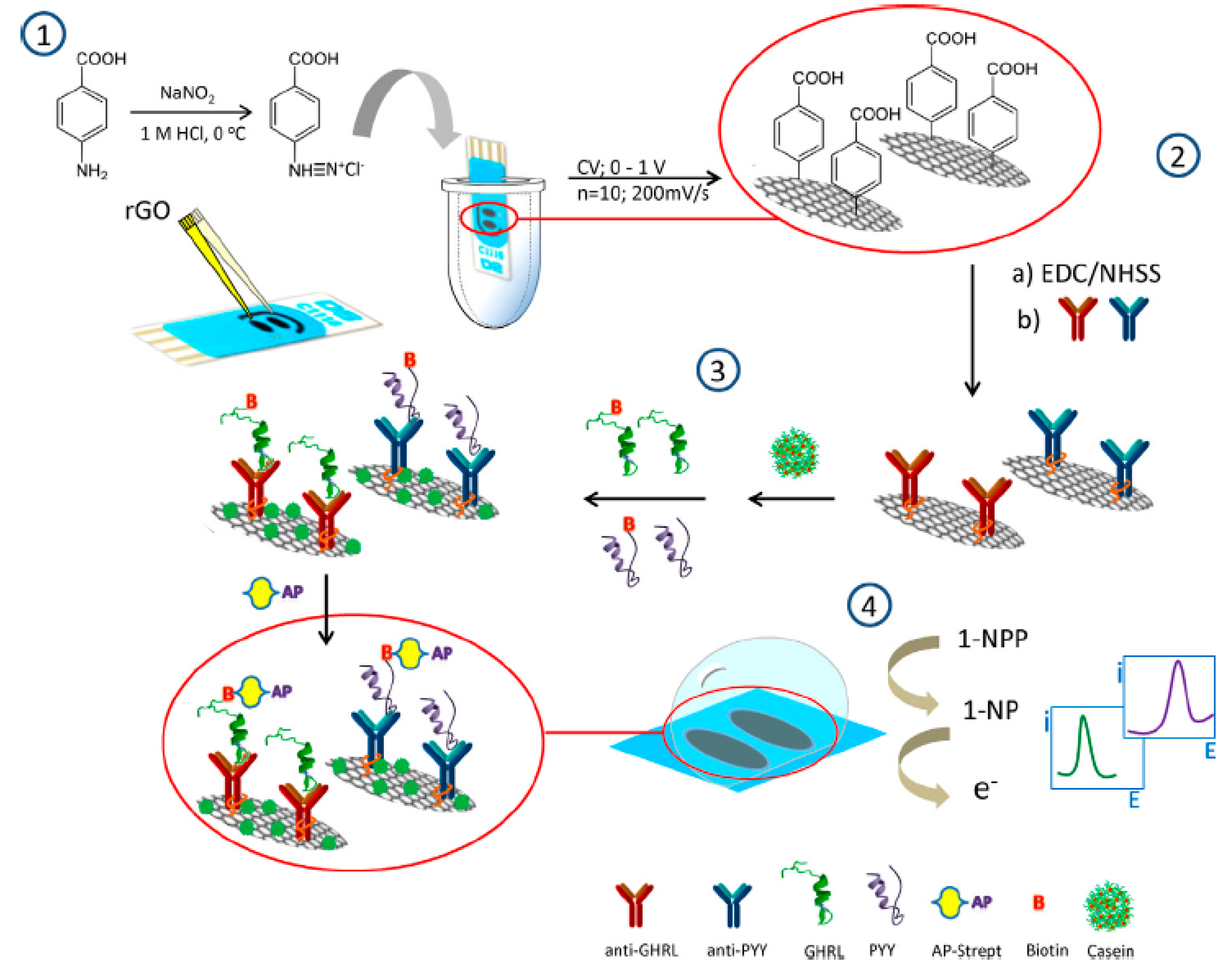
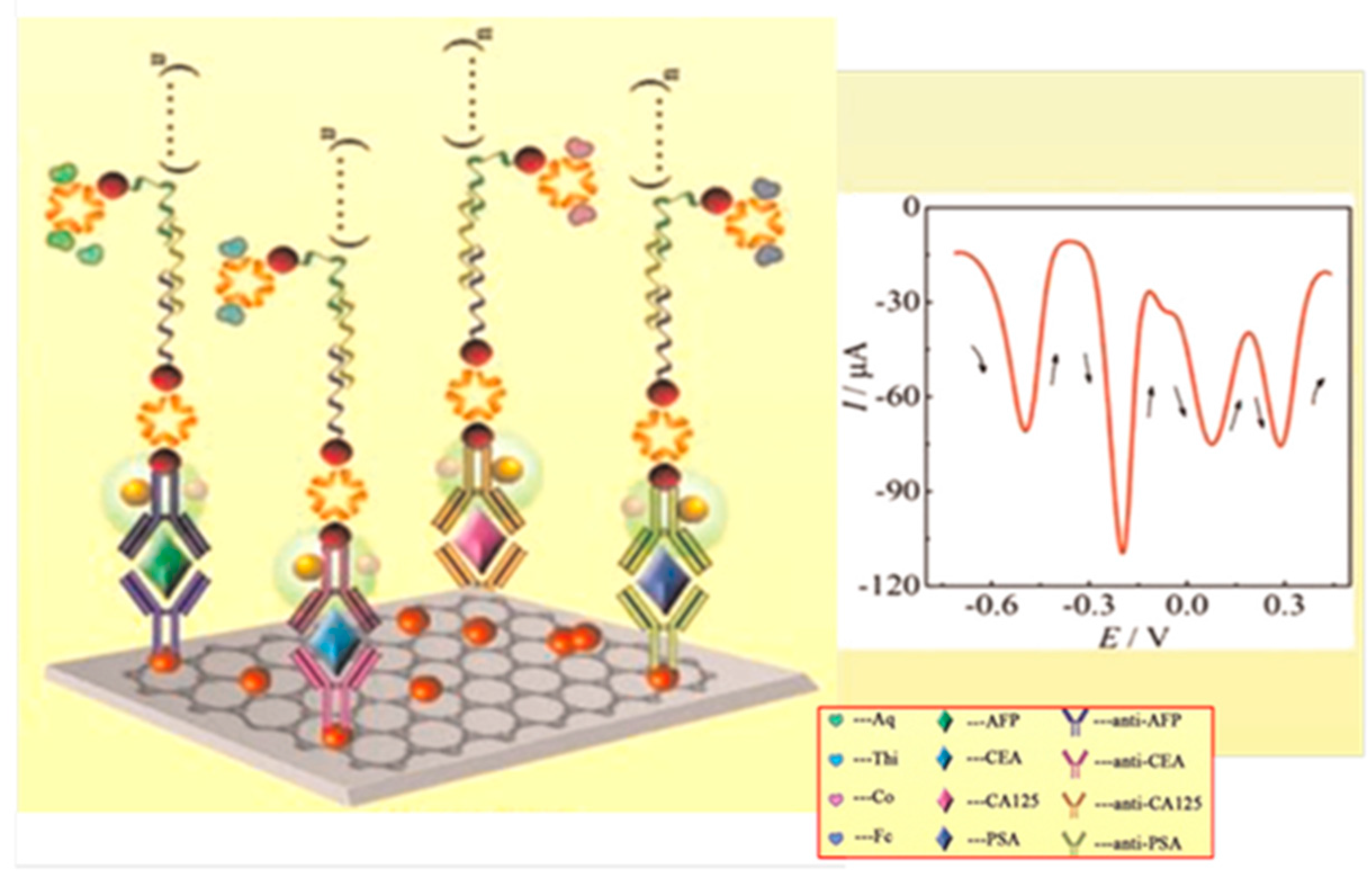
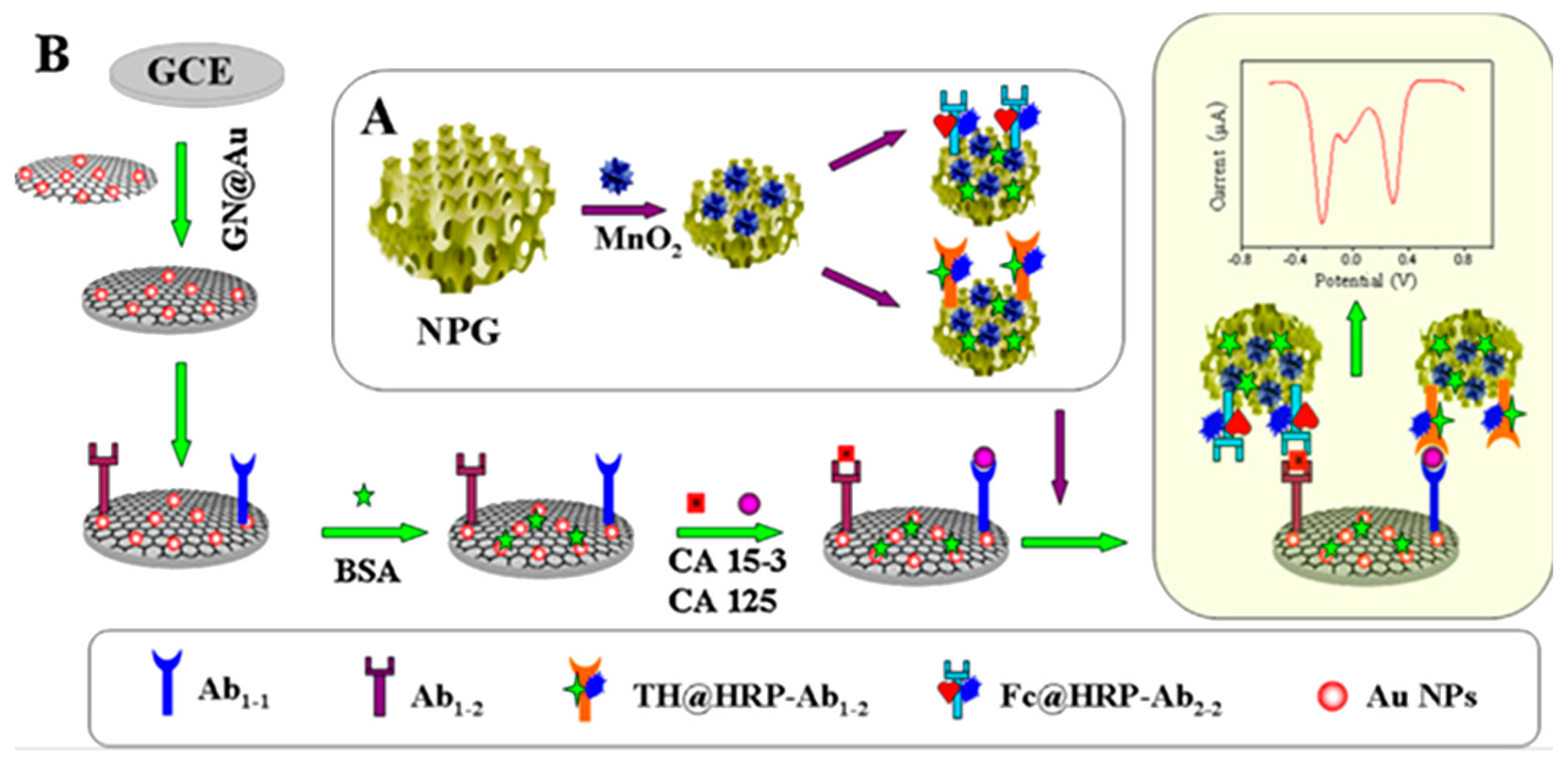
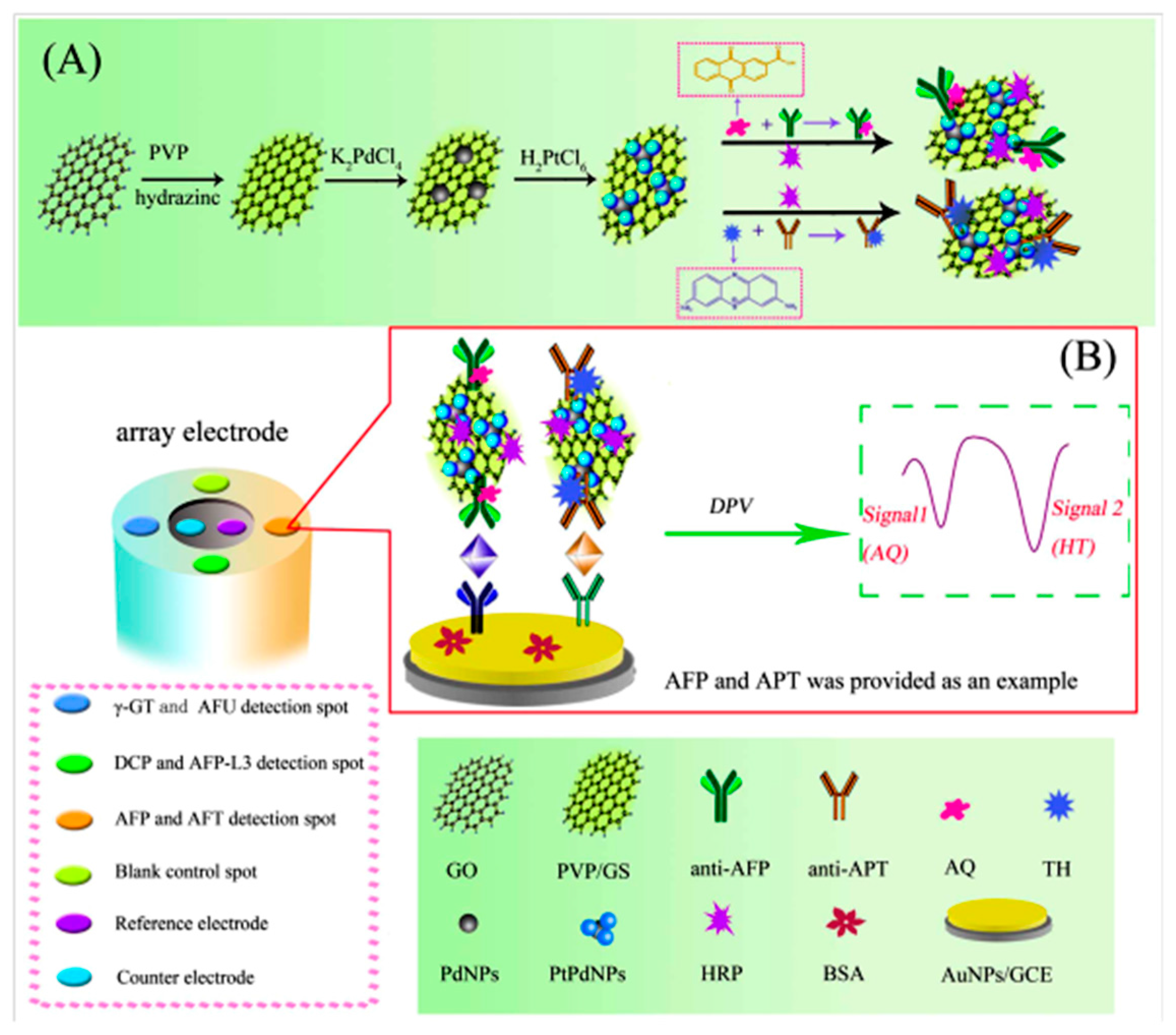
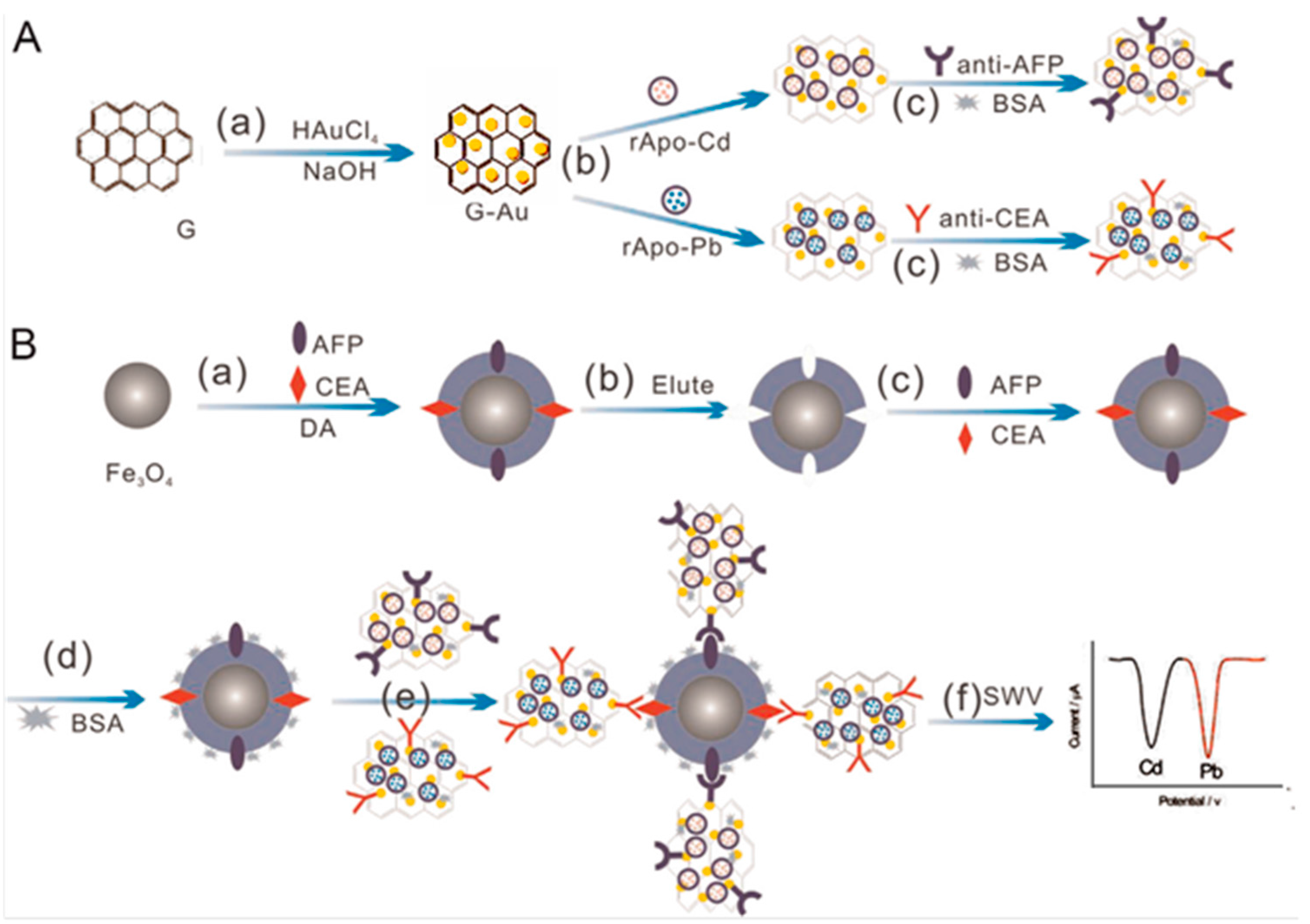

| Electrodes | Label | Analyte | Immunoassay | Technique | Linear Range | LOD | Sample | Ref. |
|---|---|---|---|---|---|---|---|---|
| Cancer biomarkers | ||||||||
| GA/Chit/SPAuEs (2WEs) | Strept/AuNPs/CNHs | AFP CEA | Sandwich-type; Ag deposition | LSASV | 0.1–1000 pg·mL−1 | 0.024 pg·mL−1 AFP 0.032 pg·mL−1 CEA | serum | [7] |
| SPAuEs (4WEs) | AuNRs-HRP | phospho-p53392 phospho-p5315 phospho-p5346 total p53 | Sandwich-type; detection of H2O2/THI | SWV | 0.01–20 nM p53392 0.05–20 nM p5315 0.1–50 nM p5346 0.05–20 nM total p53 | 5 pM p53392 20 pM p5315 30 pM p5346 10 pM total p53 | plasma | [8] |
| carboxylated-SPCEs (16WEs) | HRP-MWCNTs | PSA IL-8 | Sandwich-type; detection of TMB | amperometry | 5–4000 pg·mL−1 PSA 8–1000 pg·mL−1 IL-8 | 5 pg·mL−1 PSA 8 pg·mL−1 IL-8 | serum | [18] |
| AuNPs film/PDMS (8 × 8 WEs) | HRP/AuNRs | PSA; PSMA; IL-6 | Sandwich-type; detection of H2O2 | amperometry | 0.1–10 ng·mL−1 PSA 0.005–1 ng·mL−1 IL-6 0.8–400 PSMA | 0.1 ng·mL−1 PSA 0.005 ng·mL−1 IL-6 0.8 ng·mL−1 PSMA | serum | [19] |
| G/SPCEs (3WEs) | M-PtNPs | CA125, CA153, CEA | Sandwich-type; detection of H2O2 | DPV | 0.05–20 U·mL−1 CA125; 0.008–24 U·mL−1 CA153; 0.02–20 24 U·mL−1 CEA | 0.002 U·mL−1; 0.001 U·mL−1; 7.0 pg·mL−1 | serum | [20] |
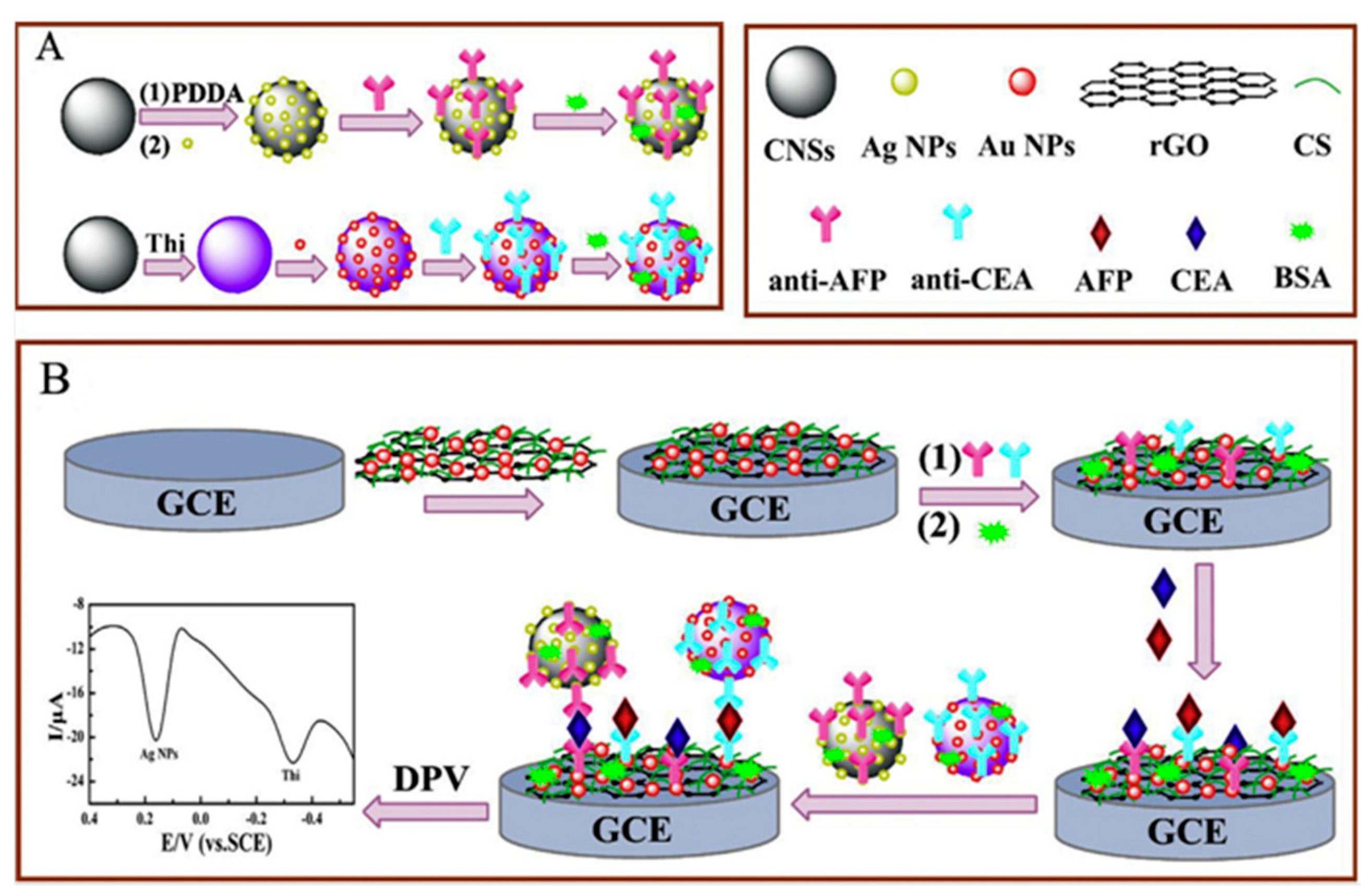
| Chit/SPCEs (2WEs) | AuNPs | CEA AFP | Sandwich-type; Ag deposition | LSASV | 0.005–5.0 ng·mL−1 | 3.5 pg·mL−1 CEA 3.9 pg·mL−1 AFP | serum | [21] |
| Chit/SPCEs | Strept/AgNPs/MWCNTs | CEA AFP | Sandwich-type; Ag deposition | LSASV | 0.1–5000 pg·mL−1 | 0.093 pg·mL−1 CEA 0.061 pg·mL−1AFP | serum | [22] |
| AuNPs/G/SPCEs (3WEs) | AP-AuCs/G | CA153, CA125, CEA | Sandwich-type; addition of 3-IP and Ag deposition | LSASV | 0.005–50 U·mL−1 CA 153 0.001–100 U·mL−1 CA 125 0.004–200 U·mL−1 CEA | 0.0015 U·mL−1 CA 153 0.00034 U·mL−1 CA 125 0.0012 U·mL−1 CEA | serum | [23] |
| SPCEs (2WEs) | Strept-HRP | ERα PR | Sandwich-type magnetoimmuno-sensor; detection of H2O2/HQ | amperometry | 73–1500 pg·mL−1 (PR) | 22 pg·mL−1 (PR) | cell lysates | [24] |
| Cardiac biomarkers | ||||||||
| CNF microE chip (3 × 3) | - | CRP, cTNI, Mb | Direct; detection of Fe(CN)64− | DPV | - | - | serum | [25] |
| HOOC-MBs/SPCEs (2WEs) | HRP | NT-proBNP CRP | Competitive ( NT-pro BNP); sandwich-type (CRP); detection of H2O2 in presence of TMB | amperometry | 2.5–504 ng·mL−1 NT-proBNP 2–100 ng·mL−1 CRP | 0.47 ng·mL−1 | serum | [26] |
| SiNW array | - | cTnT, CK-MM, CK-MB | Direct assay | Resistance changes caused by specific binding | 1 pg·mL−1–10 ng·mL−1 | 1 pg·mL−1 (2 μL blood 45 min) | blood | [27] |
| Other clinical biomarkers | ||||||||
| HOOC-Phe-DWCNTs/SPCEs (2WEs) | poly-HRP-Strept | IL-1β TNF-α | Sandwich-type; detection of H2O2 in the presence of HQ | amperometry | 0.5–100 pg·mL−1 IL-1β 1–200 pg·mL−1 TNF-α | 0.38 pg·mL−1 IL-1β 0.85 pg·mL−1 TNF-α | serum saliva | [28] |
| Au microE chip (8WEs) | - | IL-1β, IL-10, IL-6 | Direct; detection of Fe(CN)63−/4− | EIS | 1–15 pg·mL−1 | - | - | [29] |
| rGO/SPCEs (2WEs) | Strept-AP | GHRL PYY | Sandwich-type; addition of 1-NPP | DPV | 10−3–100 ng·mL−1 GHRL 10−4–10 ng·mL−1 PYY | 1.0 pg·mL−1 GHRL 0.02 pg·mL−1 PYY | serum saliva | [30] |
| SPCEs (2WEs) | Strept-AP | cortisol ACTH | Sandwich-type; addition of 1-NPP | DPV | 0.1–500 ng·mL−1 cortisol 5.0 × 10−5–0.1 ng·mL−1 ACTH | 37 pg·mL−1 cortisol 0.04 pg·mL−1 ACTH | serum | [31] |
| AuNPs/MWCNTs/SPCEs (2WEs) | AP | IgA and IgG type AGA anti-tTG | Sandwich-type; addition of 3-IP and Ag deposition | cyclic-ASV | up to 100 U mL−1 | 2.45 U·mL−1 tTG IgA 2.95 U·mL−1 tTG IgG | serum | [32] |
| Electrode | Label | Analyte | Immunoassay | Technique | Linear Range | LOD | Sample | Ref. |
|---|---|---|---|---|---|---|---|---|
| Cancer biomarkers | ||||||||
| G/Au/GCE | THI (or Co or Fc or AQ)-Strept-Biotin-dsDNA/Strept-Biotin-Ab2/AuNPs/SiO2/Fe3O4 | CEA, CA125, PSA, AFP | Sandwich-type; direct detection after conjugation | DPV | 0.2–600 pg·mL−1 CEA; 0.2–1000 pg·mL−1 CA125; 0.2–800 pg·mL−1 PSA; 0.2–800 pg·mL−1 AFP | 48 fg·mL−1 CEA; 77 fg·mL−1 CA125; 60 fg·mL−1 PSA; 62 fg·mL−1AFP | - | [6] |
| AuNPs/GCE | PVP/GS/PtPdNPs/HRP/AQ PVP/GS/PtPdNPs/HRP/THI | AFP, APT; DCP, AFP-L3, γ-GT, AFU | Sandwich-type; addition of H2O2 | DPV | 0.025–5.0 ng·mL−1 AFP; 0.024–9.6 ng·mL−1 APT; 0.032–3.2 UL−1 DCP; 0.024–2.4 ng·mL−1 AFP-L3; 1.0–9.5UL−1 γ-GT; 1.2–9.0 UL−1 AFU | 0.008 ng·mL−1 AFP; 0.0082 ng·mL−1 APT; 0.01UL−1 DCP; 0.008 ng·mL−1 AFP-L3; 0.33 UL−1 γ-GT; 0.4 UL−1 AFU | serum | [12] |
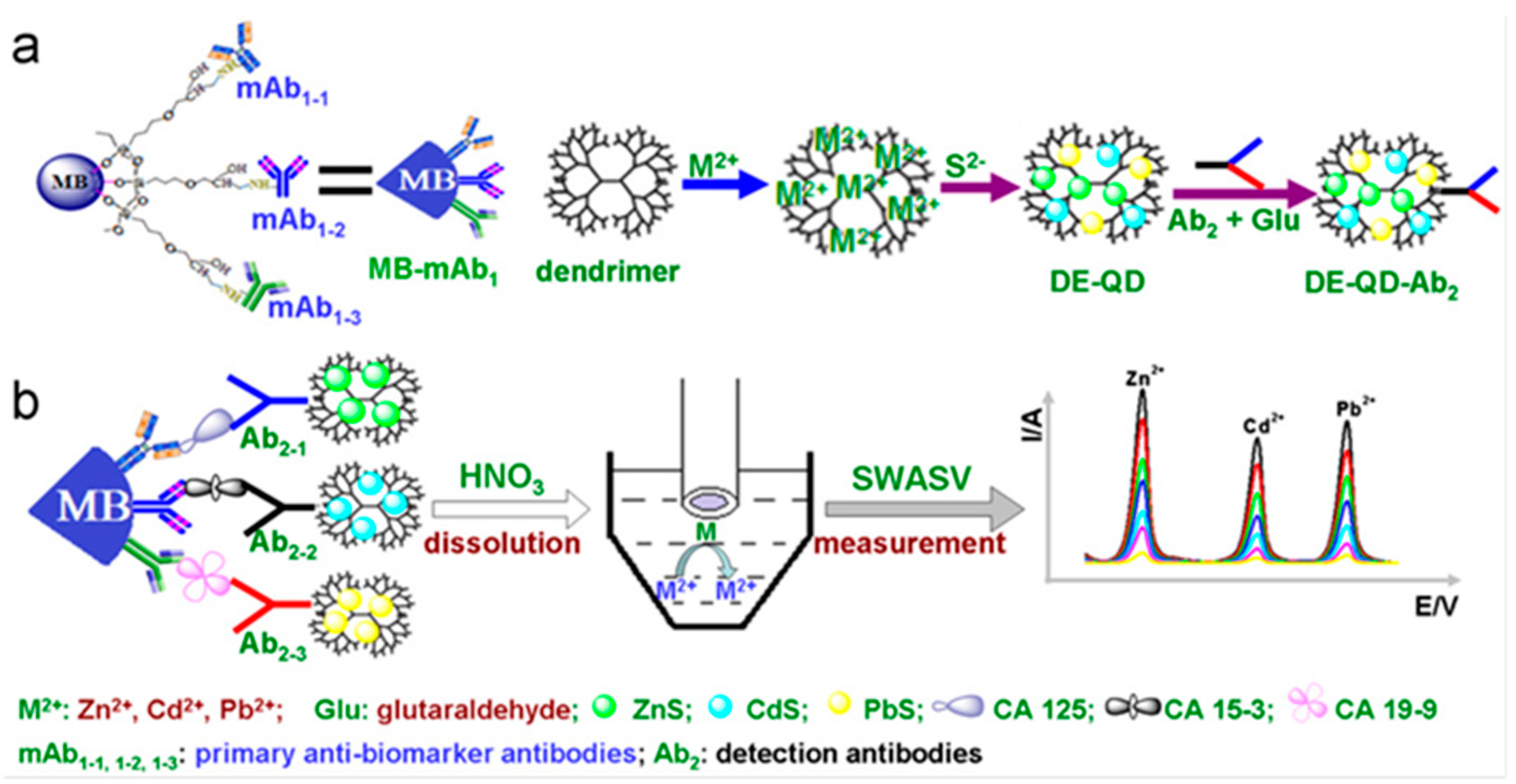
| GCE (Hg) | PAMAM-CdS (or ZnS, or PbS) | CA125 CA15-3 CA19-9 | Sandwich-type; direct detection of Cd, Zn and Ag | SWASV | 0.01–50 U·mL−1 | 0.005 U·mL−1 | serum | [13] |
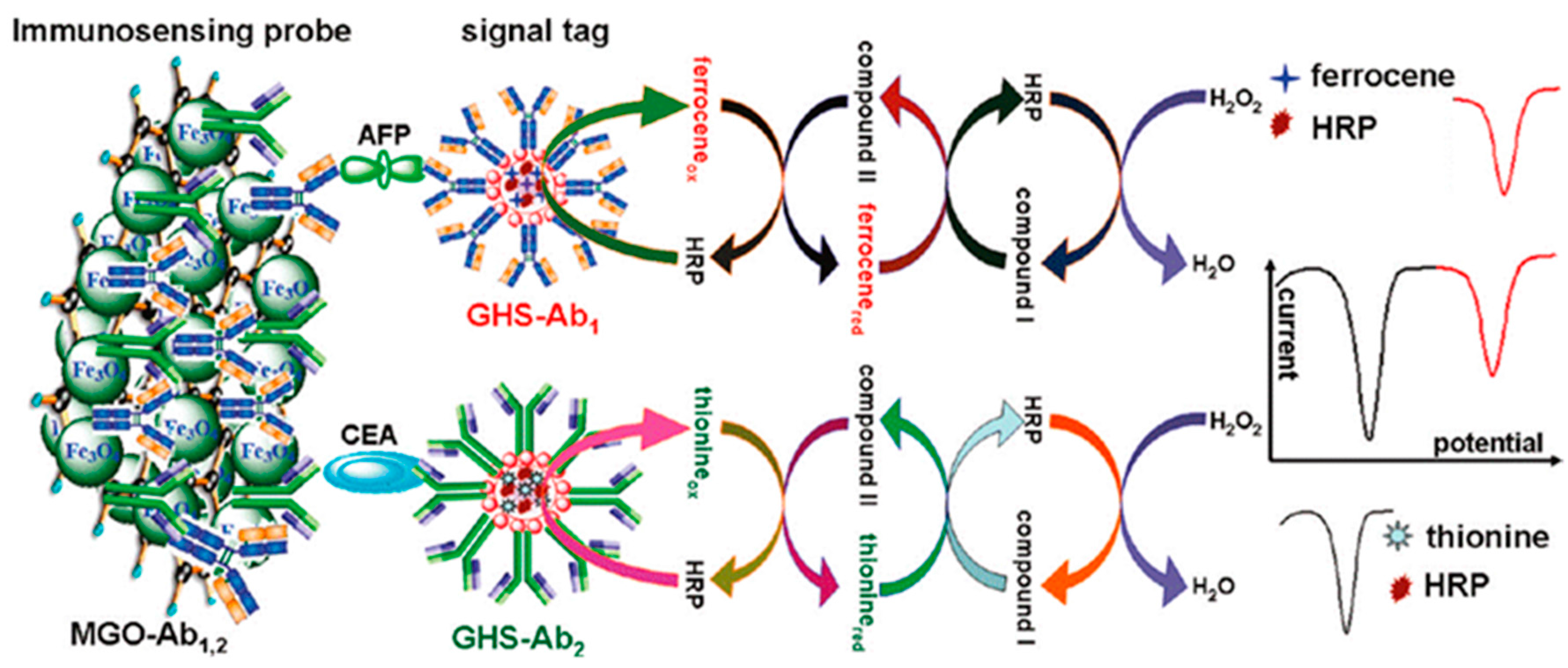
| IL/rGO/GCE | PtNPs-Cu2+ PtNPs-Cd2+ | AFP CEA | Sandwich-type; reduction of metal ions | DPV | 0.05–200 ng·mL−1 | 0.05 ng·mL−1 AFP 0.002 ng·mL−1 CEA | serum | [14] |
| AuNPs/Chit/GCE | CGS-PB; CGS-TB | CEA; AFP | Sandwich-type; direct detection | DPV | 0.5–60 ng·mL−1 | 0.1 ng·mL−1 (CEA) 0.05 ng·mL−1 (AFP) | serum | [33] |
| AuNPs/Chit/GCE | Chit/AuNPs/PB; Chit/AuNPs/Fc | CEA; AFP | Sandwich-type; direct detection | DPV | 0.05–100 ng·mL−1 | 0.02 ng·mL−1 (CEA) 0.03 ng·mL−1 (AFP) | serum | [34] |
| HAG/PANI/rGO/GCE | AuNPs/rGO/PB; AuNPs/rGO/PDDA/THI | CEA; AFP | Sandwich-type; direct detection | DPV | 0.6–80 ng·mL−1 | 0.12 ng·mL−1 (CEA) 0.08 ng·mL−1 (AFP) | serum | [35] |
| AuNPs/GCE | Fc/HRP THI/HRP | CEA AFP | Direct assay after addition of H2O2 | DPV | 0.01–50 ng·mL−1 | 0.01 ng·mL−1 | serum | [36] |
| AuNPs/G/GCE | THI-HRP-NPG-MnO2 Fc-HRP-NPG-MnO2 | CA-15-3 CA-125 | Sandwich-type; addition of H2O2 | SWV | 0.01–50 U·mL−1 | 3.5 mU·mL−1 (CA-153) 4.1 mU·mL−1(CA-125) | serum | [37] |
| PDPMT-Cl-Chit/GCE | THI-Fe3O4NPs Fca-Fe3O4NPs | SCC-Ag CEA | Sandwich; direct detection | DPV | 0.01–10 ng·mL−1 | 4 pg·mL−1 (SCC-Ag) 5 pg·mL−1 (CEA) | serum | [38] |
| CD/G/GCE | CD/Fc/G CD/THI/G | CEA AFP | Sandwich-type with HRP-Ab2 and H2O2 detection | DPV | 0.003–40 ng·mL−1 CEA 0.001–60 ng mL−1 AFP | 0.8 pg·mL−1 CEA 0.5 pg·mL−1 AFP | serum | [39] |
| rGO/THI (or PB)/AuNPs/ITO | - | CEA AFP | Direct detection of of THI or PB | SWV | 0.01–300 ng·mL−1 | 0.650 pg·mL−1 CEA 0.885 pg·mL−1 AFP | serum | [40] |
| TB (orPB)/AuNPs/TCCRMSs/FTO | - | CEA NSE | Direct; detection of TB or PB | SWV | 2–25 ng·mL−1 | 0.11 ng·mL−1 CEA 0.08 ng·mL−1 NSE | serum | [41] |
| HOOC-MBs/SPCE | ACTP/AuNPs ATLP/AuNPs | p5315, p53392 | Sandwich-type; detection of metals | SWV | 1–20 ng·mL−1 (p5315) 0.5–20 ng·mL−1 (p53392) | 0.5 ng·mL−1 (p5315) 0.2 ng·mL−1 (p53392) | serum | [42] |
| GCE (Bi) | G/AuNPs-r-Apo-Cd G/AuNPs-r-Apo-Pb | AFP CEA | Sandwich-type; detection of metals | SWASV | 0.001–5 ng·mL−1 | 0.3 pg·mL−1 AFP; 0.35 pg·mL−1 CEA | serum | [43] |
| rGO | CdSeTe@CdS Ag NCs | Bcl-2 Bax | Sandwich-type; direct detection of Cd and Ag | SWASV | 1–250 ng·mL−1 | 0.5 fmol | leukemiaK562 cells | [44] |
| Chit/MWCNTs/GCE | OMC-Zn OMC-Cd | AFP HER-2 | Sandwich-type; oxidation of metal ions | DPV | 0.001–150 ng·mL−1 | 0.6 pg·mL−1 AFP 0.35 pg·mL−1 HER-2 | serum | [45] |
| GCE | EnvisionTM-CdS (or PbS or AuNPs) | AFP CEA CA19-9 | Sandwich-type; detection of metals | DPASV DPCSV | 0.001–50 ng·mL−1 AFP; CEA; 0.005–100 ng·mL−1 CA19-9 | 0.02 pg·mL−1 AFP; 0.05 pg·mL−1 CEA; 0.3 pg·mL−1 CA19-9 | serum | [46] |
| Glu/Chit/AuNPs/AuE | BSA/AuNPs-Pb2+ BSA/AuNPs-Cd2+ | CEA AFP | Sandwich-type; reduction of metal ions | DPV | 0.01–50 ng·mL−1 | 4.6 pg·mL−1 CEA 3.0 pg·mL−1 AFP | serum | [47] |
| AuNPs/AuE | AuNPs@MWCNTs-Pb2+ AuNPs@MWCNTs-Cd2+ | CEA AFP | Sandwich-type; reduction of metal ions | SWV | 0.01–60 ng·mL−1 | 3.0 pg·mL−1 CEA 4.5 pg·mL−1 AFP | serum | [48] |
| AuNPs/rGO/Chit | AgNPs/THI/CNSs AgNPs/CNSs | CEA AFP | Sandwich-type; direct detection | DPV | 0.01–80 ng·mL−1 | 2.8 pg·mL−1 CEA 3.5 pg·mL−1 AFP | serum | [49] |
| IL/rGO/PSS/GCE | CGN-THI CGN-DAP CGN-Cd2+ | CEA PSA AFP | Sandwich-type; direct detection | SWV | 0.01–100 ng·mL−1 | 2.7 pg·mL−1 CEA 4.8 pg·mL−1 PSA 3.1 pg·mL−1 AFP | serum | [50] |
| AuNPs/AuE | TiO2/Nf- Co(dcbpy)32+ TiO2/Nf- MB | CA15-3 CA19-9 | Sandwich-type; direct detection | DPV | 5–100 U·mL−1 CA15-3 1–100 U·mL−1 CA19-9 | 0.3 UmL−1 CA15-3 1.6 UmL−1 CA19-9 | serum | [51] |
| AuNPs/PAPT/GCE | poly(VFc-ATP) AuNPsPOPD/AuNPs | AFP CEA | Sandwich-type; direct detection | DPV | 0.01–100 ng·mL−1 | 0.003 ng·mL−1 AFP 0.006 ng·mL−1 CEA | serum | [52] |
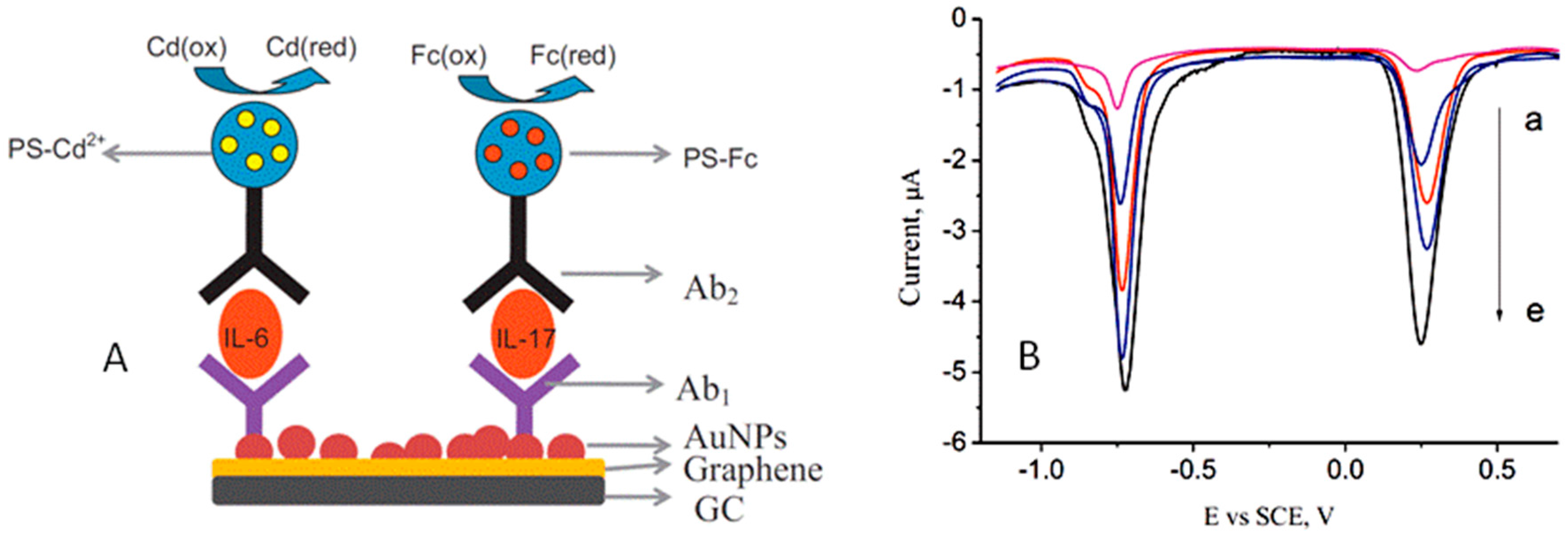
| AuNPs/G/GCE | PS-Cd2+ PS-Fc | IL-6 IL-17 | Sandwich-type; direct detection | SWV | 1–1000 pg·mL−1 IL-6 2–1000 pg·mL−1 IL-17 | 0.5 pg·mL−1 1 pg | serum | [53] |
| Protein A/Nafion/GCE | GS/AuNPs-THI (or Cobpy)33+ or Fc) | AFP CEA SS2 | Sandwich.type; direct detection | DPV | 0.016–50 ng·mL−1 AFP 0.010–50 ng·mL−1CEA 0.012–50 ng·mL−1 AFP | 5.4 pg·mL−1 AFP 2.8 pg·mL−1 CEA 4.2 pg·mL−1 AFP | - | [54] |
| AuNPs/PEI/PTCA/GCE | Au@PBNPs/O-GS-Strept-AP; Au@NiNPs/O-GS-Strept-AP | fPSA PSA | Sandwich-type; AA-P addition | DPV | 0.02–10 ng·mL−1 fPSA 0.01–50 ng·mL−1 PSA | 6.7 pg·mL−1 3.4 pg·mL−1 | serum | [55] |
| MWCNTs/GCE | AA liposome UA liposome | NSE ProGRP | Sandwich-type, direct detection | LSV | 50–1000 pg·mL−1 | 5.0 pg·mL−1 NSE 100 pg·mL−1 ProGRP | serum | [56] |
| AuNPs/IL/rGO/GCE | Cd (or Pb or Cu) AlgNBs | AFP CEA PSA | Sandwich-type; direct detection | DPV | 0.01–100 ng·mL−1 | 0.01 ng·mL−1AFP 0.0086 ng·mL−1CEA 0.0075 ng·mL−1PSA | serum | [57] |
| GCE (Bi) | PLL/AuNPs/Cd-Apo (or Pb-Apo) | AFP CEA | Sandwich-type; direct detection | ASSWV | 0.01–50 ng·mL−1 | 4 pg·mL−1 | serum | [58] |
| Chit/AuNPs/GCE | GA/Chit/pAA NSs-Cu2+ (or Cd2+ or Zn2+) | CEA CA19-9 CA125 CA242 | Sandwich-type; direct detection | SWV | 0.1–100 ng·mL−1 CEA 1–150 U·mL−1 CA 19-9, CA125,CA242 | 0.02 ng·mL−1 CEA 0.4 U·mL−1 CA19-9 0.3 U·mL−1 CA125 0.4 U·mL−1 CA242 | serum | [59] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yáñez-Sedeño, P.; Campuzano, S.; Pingarrón, J.M. Multiplexed Electrochemical Immunosensors for Clinical Biomarkers. Sensors 2017, 17, 965. https://doi.org/10.3390/s17050965
Yáñez-Sedeño P, Campuzano S, Pingarrón JM. Multiplexed Electrochemical Immunosensors for Clinical Biomarkers. Sensors. 2017; 17(5):965. https://doi.org/10.3390/s17050965
Chicago/Turabian StyleYáñez-Sedeño, Paloma, Susana Campuzano, and José M. Pingarrón. 2017. "Multiplexed Electrochemical Immunosensors for Clinical Biomarkers" Sensors 17, no. 5: 965. https://doi.org/10.3390/s17050965
APA StyleYáñez-Sedeño, P., Campuzano, S., & Pingarrón, J. M. (2017). Multiplexed Electrochemical Immunosensors for Clinical Biomarkers. Sensors, 17(5), 965. https://doi.org/10.3390/s17050965







