Embedded Ceria Nanoparticles in Crosslinked PVA Electrospun Nanofibers as Optical Sensors for Radicals
Abstract
:1. Introduction
2. Materials and Methods
2.1. Nanoparticle and Polymer Synthesis
2.2. Electrospinning and Crosslinking
2.3. Nanocomposite Characterizations
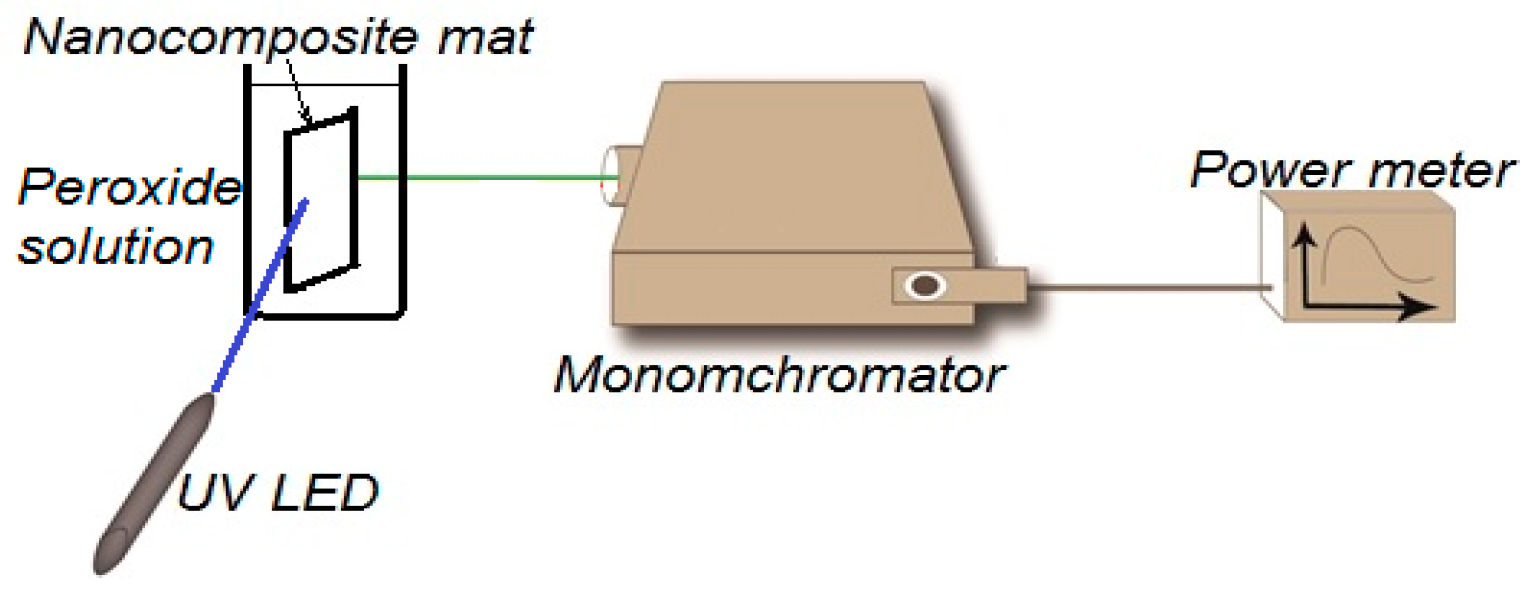
2.4. Fluorescence Quenching Experiment
3. Results
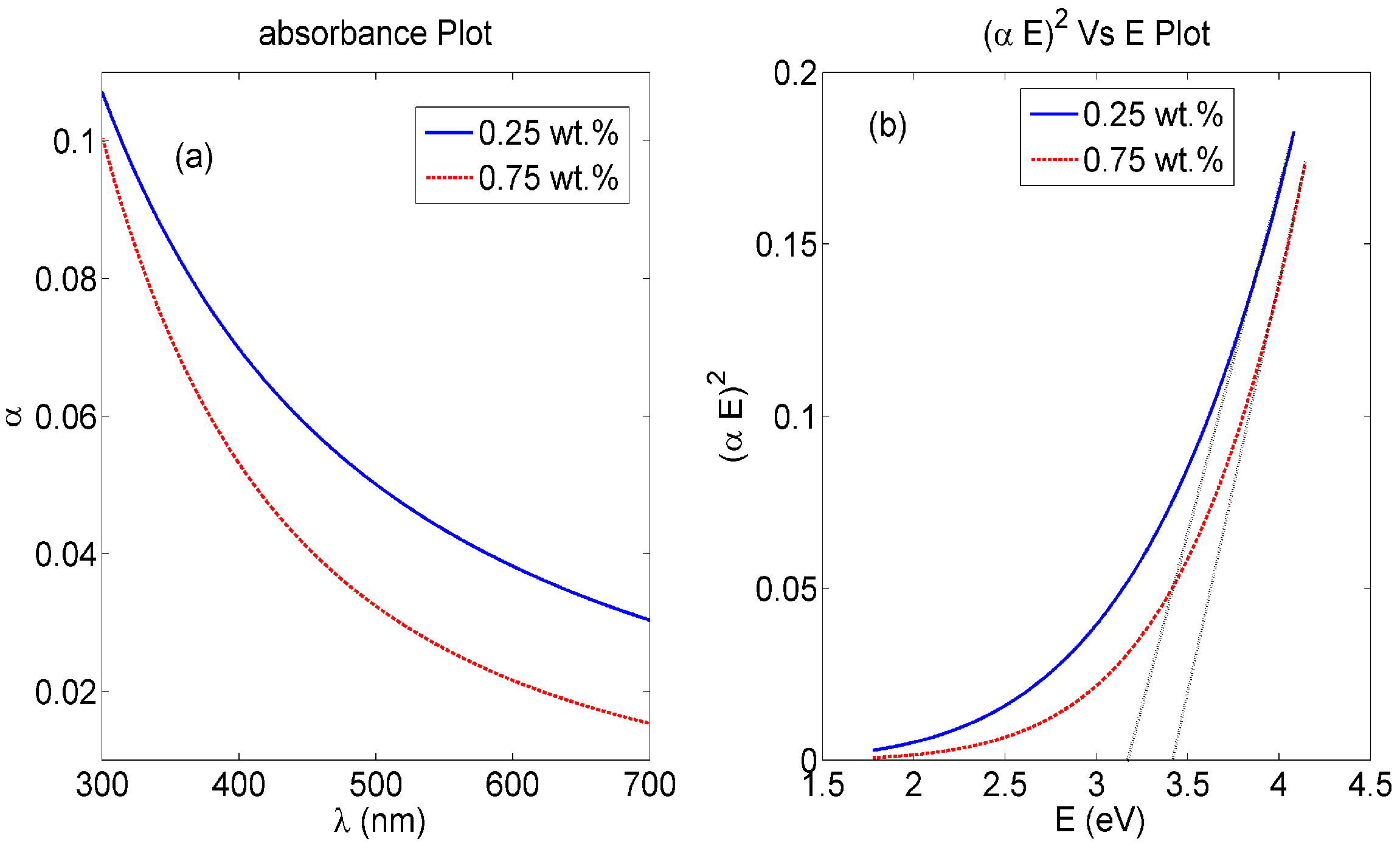
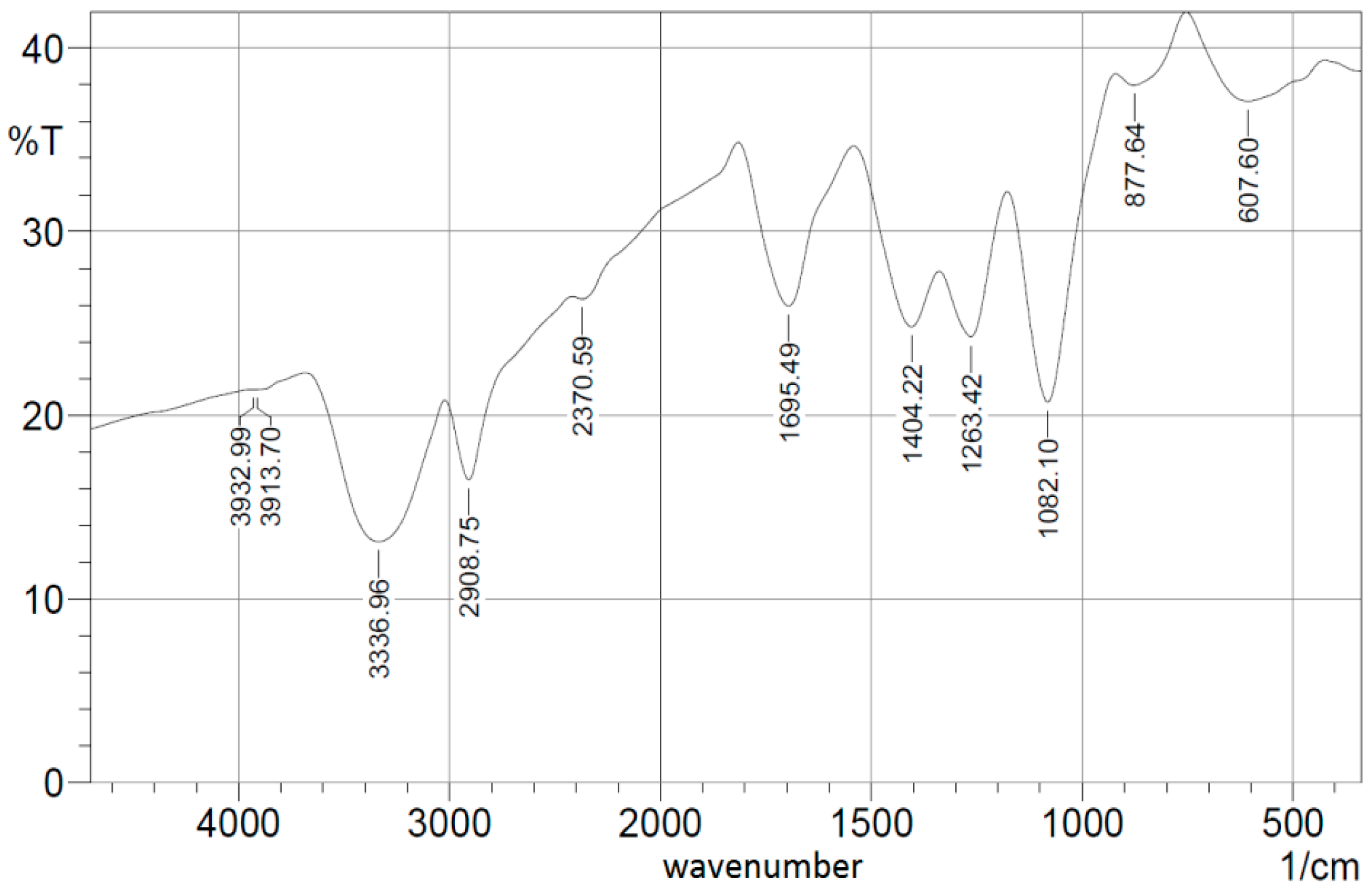
3.1. Nanocomposite Charcterization
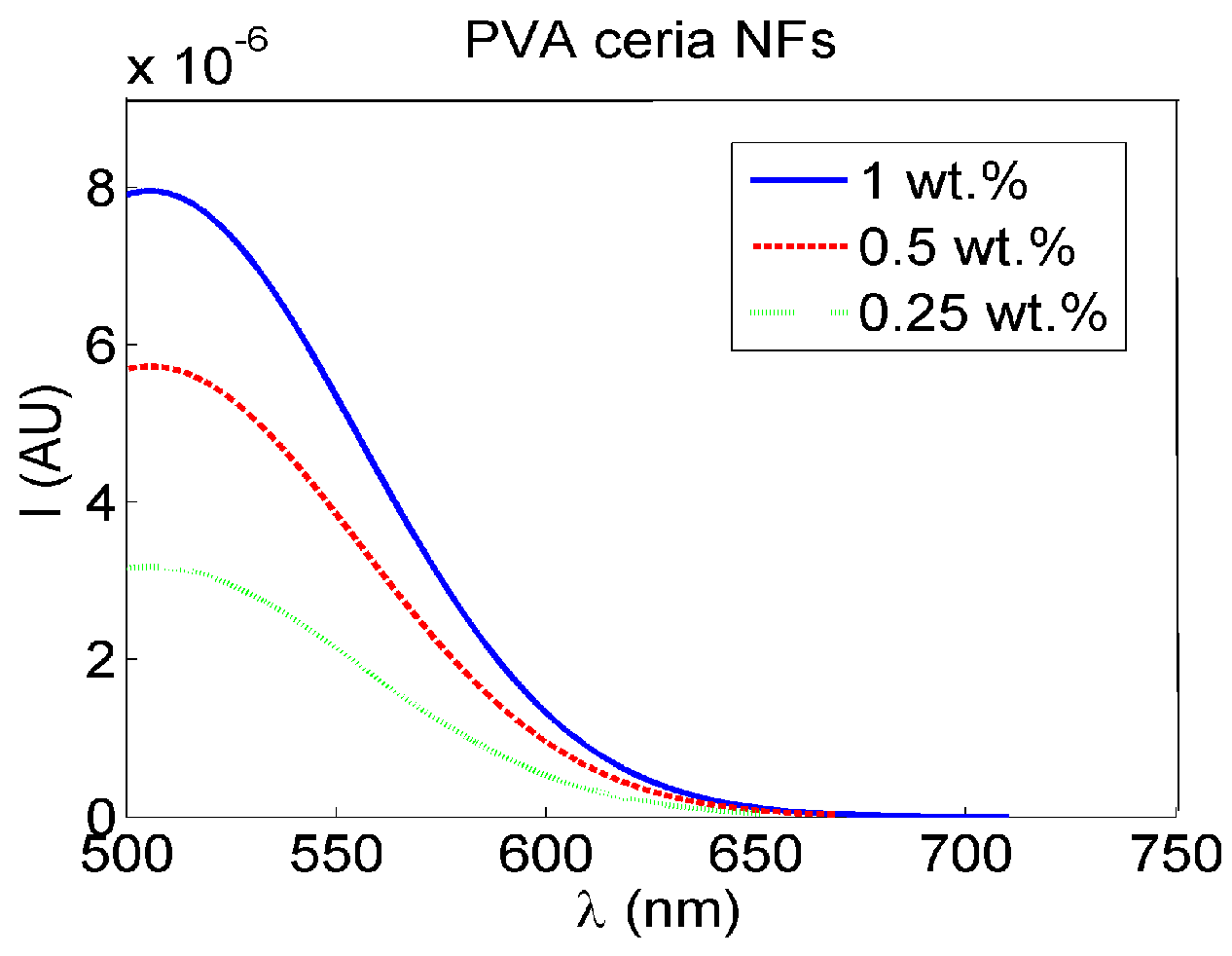
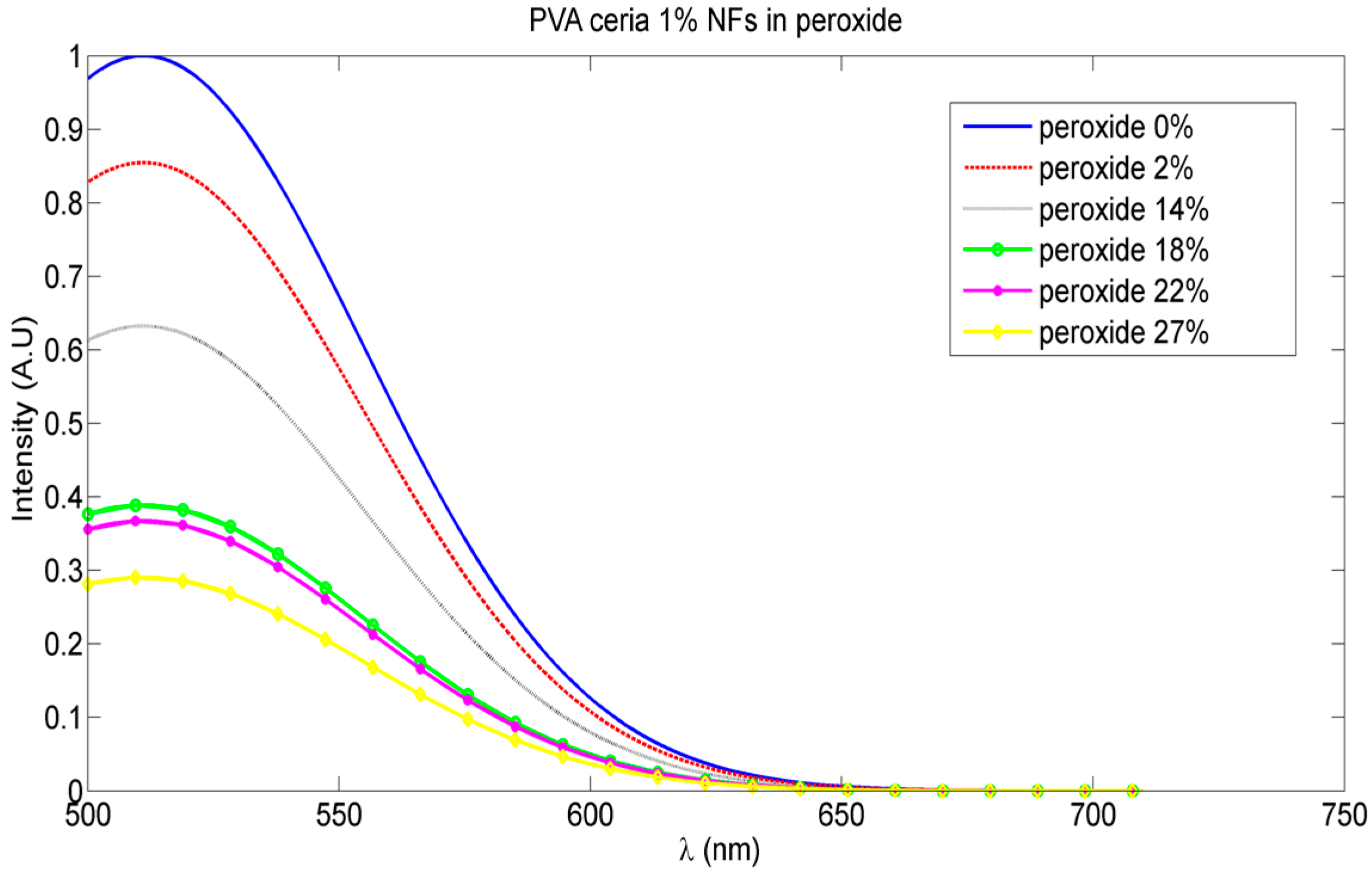
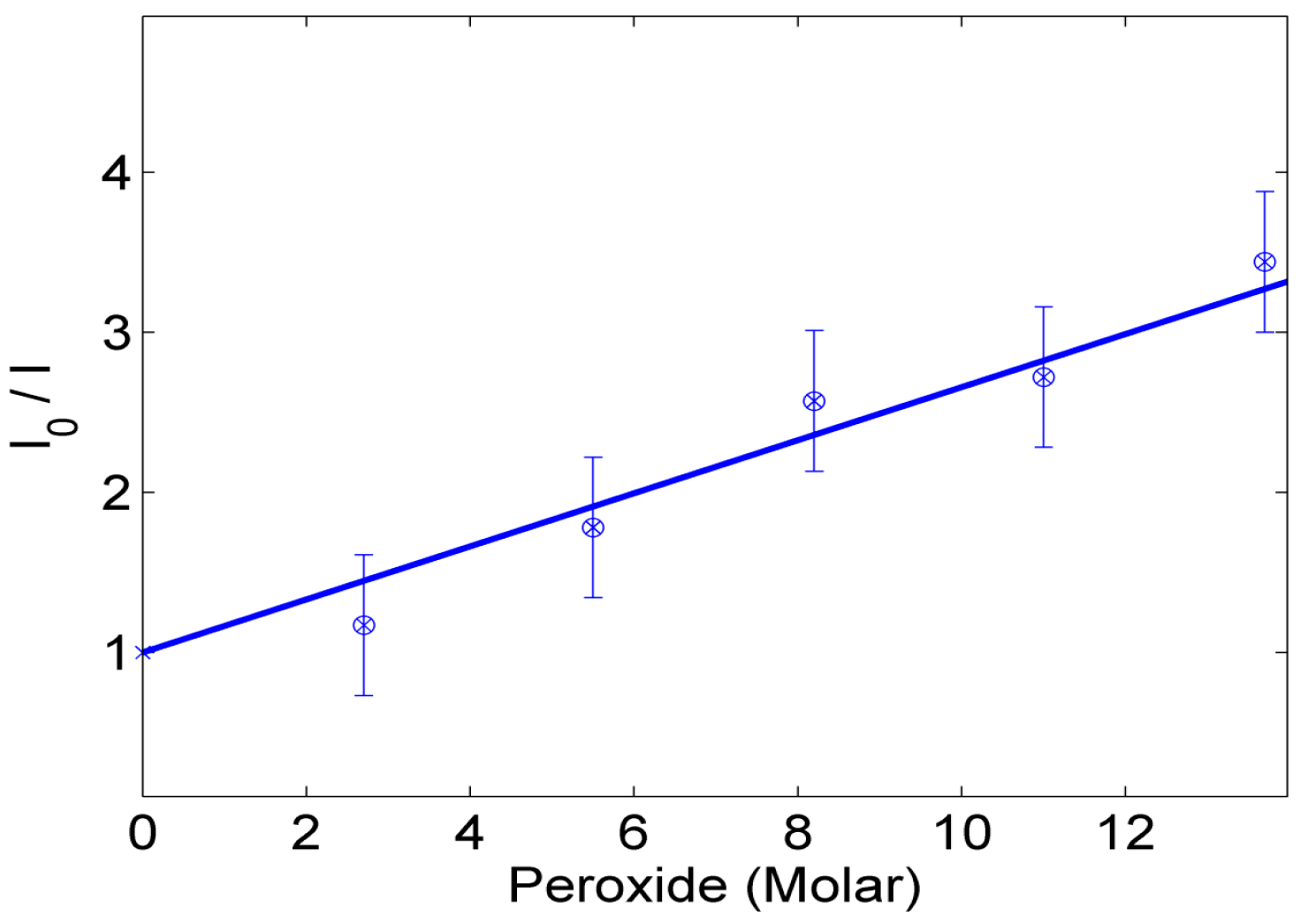
3.2. Optical Sensing of Peroxide Using Fluorescence Quenching Mechanism
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| Ceria NPs | Cerium oxide nanoparticles |
| PVA | Polyvinyl alcohol |
| SEM | Scanning Electron Microscope |
| TEM | Transmission Electron Microscope |
References
- Sarvazyan, A.; Egorov, V.; Sarvazyan, N. Tactile Sensing and Tactile Imaging in Detection of Cancer. In Biosensors and Molecular Technologies for Cancer Diagnostics; Herold, K.E., Rasooly, A., Eds.; CRC Press: Boca Raton, FL, USA, 2012; pp. 339–354. [Google Scholar]
- Sainz, R.; Lombo, F.; Mayo, J. Radical Decisions in Cancer: Redox Control of Cell Growth and Death. Cancers 2012, 4, 442–474. [Google Scholar] [CrossRef] [PubMed]
- Hoigné, J. Inter-calibration of OH radical sources and water quality parameters. Water Sci. Technol. 1997, 35, 1–8. [Google Scholar] [CrossRef]
- Middleburgh, S.; Lagerlof, K.; Grimes, R. Accommodation of Excess Oxygen in Group II Monoxides. J. Am. Ceram. Soc. 2013, 96, 308–311. [Google Scholar] [CrossRef]
- Griffiths, J.; Robinson, S. The Oxy Lite: A fibre-optic oxygen sensor. J. Radiol. 1999, 72, 627–630. [Google Scholar] [CrossRef] [PubMed]
- Braun, R.; Lanzen, J.; Snyder, S.; Dewhirst, M. Comparison of tumor and normal tissue oxygen tension measurements using OxyLite or microelectrodes in rodents. Am. J. Physiol. Heart Circ. Physiol. 2001, 280, H2533–H2544. [Google Scholar] [PubMed]
- Shehata, N.; Meehan, K.; Leber, D. Fluorescence quenching in ceria nanoparticles: A dissolved oxygen molecular probe with a relatively temperature insensitive Stern-Volmer constant up to 50 °C. J. Nanophotonics 2012, 6. [Google Scholar] [CrossRef]
- Feng, N.; Xie, J.; Zhang, D. Synthesis, characterization, photophysical and oxygen-sensing properties of a novel europium(III) complex. Spectrochim. Acta A 2010, 77, 292–296. [Google Scholar] [CrossRef] [PubMed]
- Shehata, N.; Meehan, K.; Hassounah, I.; Hudait, M.; Jain, N.; Clavel, M.; Elhelw, S.; Madi, N. Reduced erbium-doped ceria nanoparticles: One nano-host applicable for simultaneous optical down- and up-conversions. Nanoscale Res. Lett. 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Borisov, S.; Klimant, I. New luminescent oxygen-sensing and temperature-sensing materials based on gadolinium(III) and europium(III) complexes embedded in an acridone/polystyrene conjugate. Anal. Bioanal. Chem. 2012, 404, 2797–2806. [Google Scholar] [CrossRef] [PubMed]
- Quaranta, M.; Borisov, S.; Klimant, I. Indicators for optical oxygen sensors. Bioanal. Rev. 2012, 4, 115–157. [Google Scholar] [CrossRef] [PubMed]
- Matsumura, S.; Shimura, Y.; Terayama, K.; Kiyohara, T. Effects of Molecular Weight and Stereoregularity on Biodegradation of Poly(Vinyl Alcohol) by Alcaligenes Faecalis. Biotechnol. Lett. 1994, 16, 1205–1210. [Google Scholar] [CrossRef]
- Mori, T.; Sakimoto, M.; Kagi, T.; Sakai, T. Isolation and Characterization of a Strain of Bacillus Megaterium that Degrades Poly(vinyl alcohol). Biosci. Biotechnol. Biochem. 1996, 60, 330–332. [Google Scholar] [CrossRef] [PubMed]
- Larrondo, L.; St. John Manley, R. Electrostatic Fiber Spinning from Polymer Melts. I. Experimental Observations on Fiber Formation and Properties. J. Polym. Sci.: Polym. Phys. Ed. 1981, 19, 909–920. [Google Scholar] [CrossRef]
- Shin, Y.M.; Hohman, M.M.; Brenner, M.P.; Rutledge, G.C. Electrospinning: A Whipping Fluid Jet Generates Submicron Polymer Fibers. Appl. Phys. Lett. 2001, 78, 1–3. [Google Scholar] [CrossRef]
- Shehata, N.; Meehan, K.; Hudait, M.; Jain, N. Control of oxygen vacancies and Ce3+ concentrations in doped ceria nanoparticles via the selection of lanthanide. J. Nanoparticle Res. 2012, 14, 1173–1183. [Google Scholar] [CrossRef]
- Shehata, N.; Meehan, K.; Hudait, M.; Jain, N.; Gaballah, S. Studyof Optical and Structural Characteristicsof Ceria Nanoparticles Dopedwith Negative and Positive Association Lanthanide Elements. J. Nanomater. 2014, 2014, 1–7. [Google Scholar] [CrossRef]
- Chen, H.; Chang, H. Homogeneous precipitation of cerium dioxide nanoparticles in alcohol/water mixed solvents. Coll. Surf. A 2014, 242, 61–69. [Google Scholar] [CrossRef]
- Hassounah, I.; Shehata, N.; Hudson, A.; Orler, B.; Meehan, K. Characteristics and 3D formation of PVA and PEO electrospun nanofibers with embedded urea. J. Appl. Polym. Sci. 2014, 131. [Google Scholar] [CrossRef]
- Pankove, J. Optical Processes in Semiconductors, 1st ed.; Dover Publications Inc.: New York, NY, USA, 1971. [Google Scholar]
- Goharshadi, E.K.; Samiee, S.; Nancarrow, P. Fabrication of cerium oxide nanoparticles: Characterization and optical properties. J. ColloidInterface Sci. 2011, 356, 472–480. [Google Scholar] [CrossRef] [PubMed]
- Chu, C.; Lo, Y. A plastic optical fiber sensor for the dual sensing of temperature and oxygen. IEEE Photonics Tech. Lett. 2008, 20, 63–65. [Google Scholar] [CrossRef]

© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shehata, N.; Samir, E.; Gaballah, S.; Hamed, A.; Elrasheedy, A. Embedded Ceria Nanoparticles in Crosslinked PVA Electrospun Nanofibers as Optical Sensors for Radicals. Sensors 2016, 16, 1371. https://doi.org/10.3390/s16091371
Shehata N, Samir E, Gaballah S, Hamed A, Elrasheedy A. Embedded Ceria Nanoparticles in Crosslinked PVA Electrospun Nanofibers as Optical Sensors for Radicals. Sensors. 2016; 16(9):1371. https://doi.org/10.3390/s16091371
Chicago/Turabian StyleShehata, Nader, Effat Samir, Soha Gaballah, Aya Hamed, and Asmaa Elrasheedy. 2016. "Embedded Ceria Nanoparticles in Crosslinked PVA Electrospun Nanofibers as Optical Sensors for Radicals" Sensors 16, no. 9: 1371. https://doi.org/10.3390/s16091371
APA StyleShehata, N., Samir, E., Gaballah, S., Hamed, A., & Elrasheedy, A. (2016). Embedded Ceria Nanoparticles in Crosslinked PVA Electrospun Nanofibers as Optical Sensors for Radicals. Sensors, 16(9), 1371. https://doi.org/10.3390/s16091371




