Bent Fiber Sensor for Preservative Detection in Milk
Abstract
:1. Introduction
2. Materials and Methods
2.1. Theory
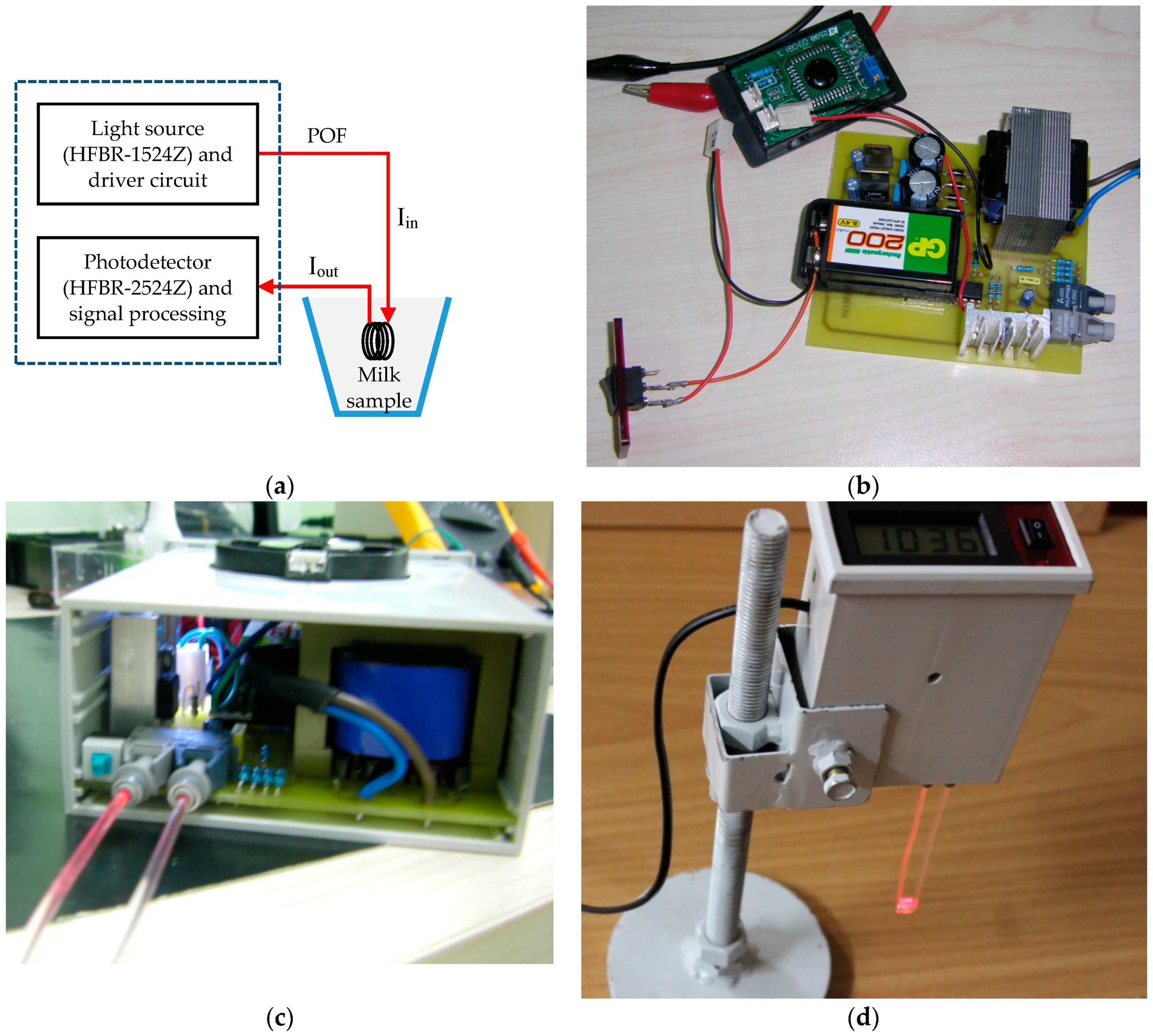
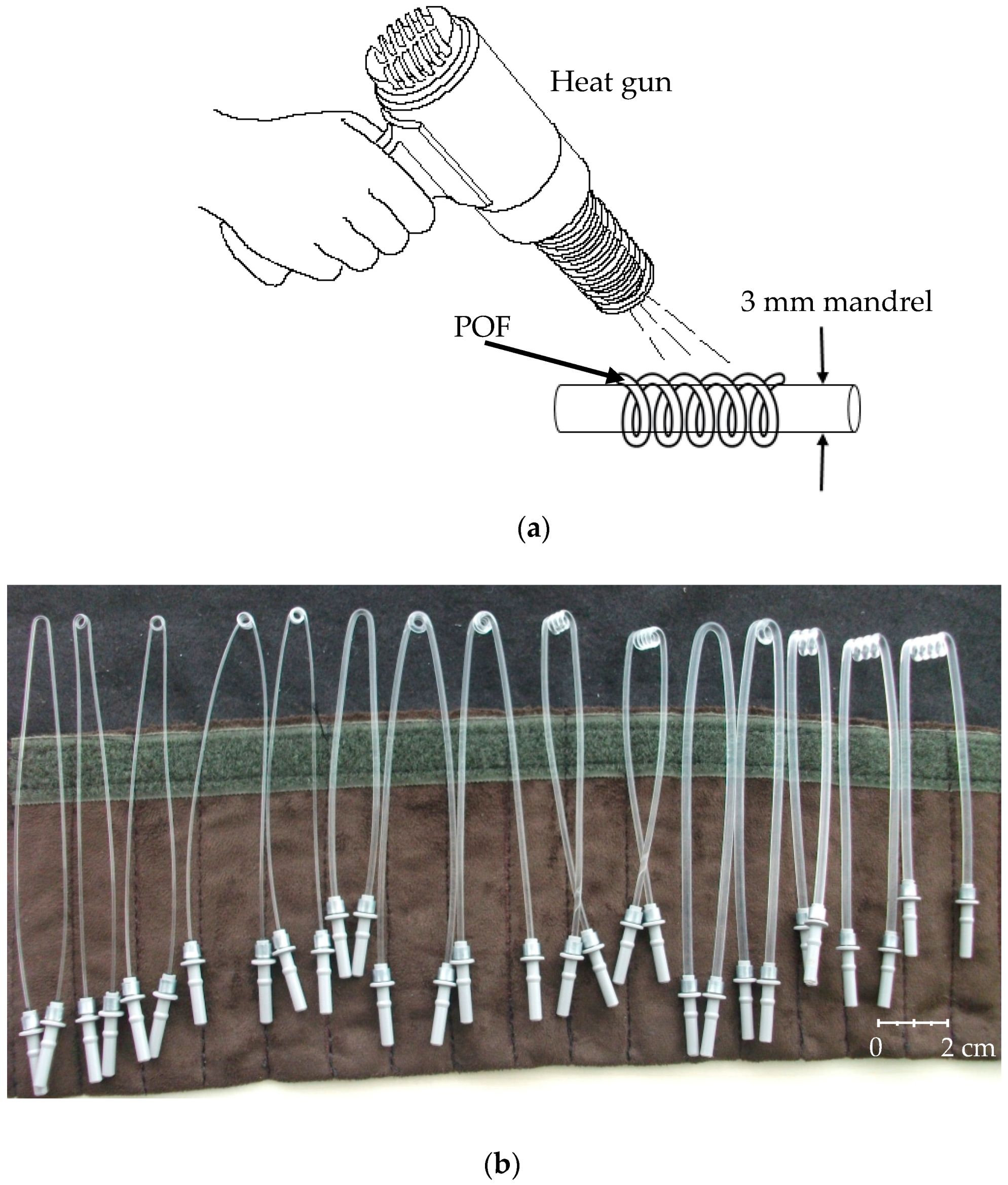
2.2. Sensing Probe
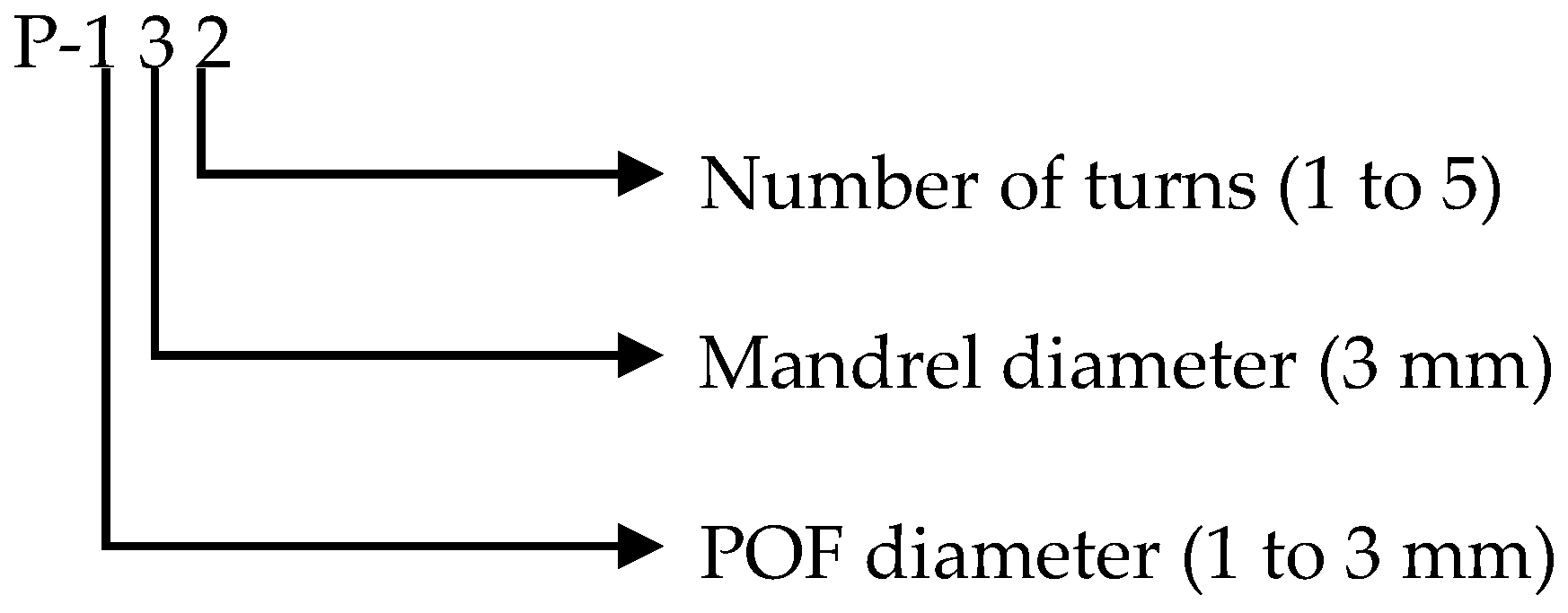
2.3. Probe Selection
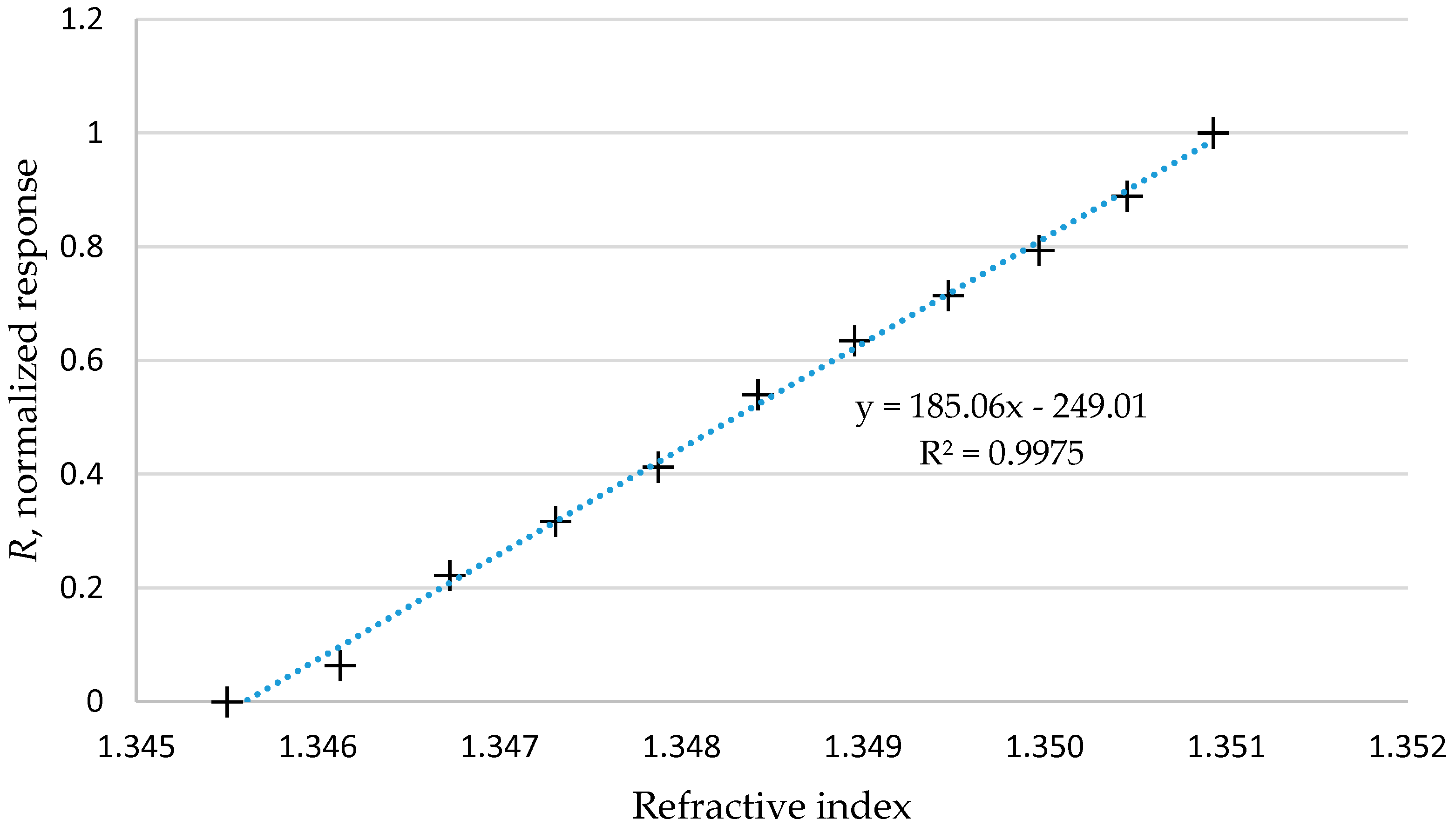
3. Results and Discussion
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Florescu, M.; Hu, W. Evaluation of Si nanowire as biosensing device. J. Optoelectron. Adv. Mater. 2015, 17, 1092–1098. [Google Scholar]
- Van der Spiegel, M.; Sterrenburg, P.; Haasnoot, W.; Van Der Fels-Klerx, H.J. Towards a decision support system for control of multiple food safety hazards in raw milk production. Trends Food Sci. Technol. 2013, 34, 137–145. [Google Scholar] [CrossRef]
- De Souza, G.C.S.; da Silva, P.A.B.; Leotério, D.M.D.S.; Paim, A.P.S.; Lavorante, A.F. A multicommuted flow system for fast screening/sequential spectrophotometric determination of dichromate, salicylic acid, hydrogen peroxide and starch in milk samples. Food Control 2014, 46, 127–135. [Google Scholar] [CrossRef]
- Jain, P.; Sarma, S.E. Light scattering and transmission measurement using digital imaging for online analysis of constituents in milk. In Proceedings of the Optical Measurement Systems for Industrial Inspection IX, Munich, Germany, 22–25 June 2015.
- Liu, J.; Ji, Z.; Tian, M. Near-infrared spectroscopy (NIRS) analysis of major components of milk and the development of analysis instrument. In Proceedings of the Multispectral, Hyperspectral, and Ultraspectral Remote Sensing Technology, Techniques and Applications V, Beijing, China, 13–16 October 2014.
- Homola, J.; Dostalek, J.; Chen, S.; Rasooly, A.; Jiang, S.; Yee, S.S. Spectral surface plasmon resonance biosensor for detection of staphylococcal enterotoxin B in milk. Int. J. Food Microbiol. 2002, 75, 61–69. [Google Scholar] [CrossRef]
- Adrian, J.; Pasche, S.; Diserens, J.M.; Sánchez-Baeza, F.; Gao, H.; Marco, M.P.; Voirin, G. Waveguide interrogated optical immunosensor (WIOS) for detection of sulfonamide antibiotics in milk. Biosens. Bioelectron. 2009, 24, 3340–3346. [Google Scholar] [CrossRef] [PubMed]
- Wandermur, G.; Rodrigues, D.; Allil, R.; Queiroz, V.; Peixoto, R.; Werneck, M.; Miguel, M. Plastic optical fiber-based biosensor platform for rapid cell detection. Biosens. Bioelectron. 2014, 54, 661–666. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Liu, X.; Li, Y.; Ying, Y. A simple and rapid optical biosensor for detection of aflatoxin B1 based on competitive dispersion of gold nanorods. Biosens. Bioelectron. 2013, 47, 361–367. [Google Scholar] [CrossRef] [PubMed]
- Castillo, M.; González, R.; Payne, F.A.; Laencina, J.; López, M.B. Optical monitoring of milk coagulation and inline cutting time prediction in Murcian al Vino cheese. Appl. Eng. Agric. 2005, 21, 465–471. [Google Scholar] [CrossRef]
- Abdelgawad, A.R.; Guamis, B.; Castillo, M. Using a fiber optic sensor for cutting time prediction in cheese manufacture from a mixture of cow, sheep and goat milk. J. Food Eng. 2014, 125, 157–168. [Google Scholar] [CrossRef]
- Arango, O.; Trujillo, A.J.; Castillo, M. Predicting coagulation and syneresis parameters of milk gels when inulin is added as fat substitute using infrared light backscatter. J. Food Eng. 2015, 157, 63–69. [Google Scholar] [CrossRef]
- Revilla, I.; González-Martín, I.; Hernández-Hierro, J.M.; Vivar-Quintana, A.; González-Pérez, C.; Lurueña-Martínez, M.A. Texture evaluation in cheeses by NIRS technology employing a fibre-optic probe. J. Food Eng. 2009, 92, 24–28. [Google Scholar] [CrossRef]
- González-Martín, I.; Hernández-Hierro, J.M.; Vivar-Quintana, A.; Revilla, I.; González-Pérez, C. The application of near infrared spectroscopy technology and a remote reflectance fibre-optic probe for the determination of peptides in cheeses (cow’s, ewe’s and goat’s) with different ripening times. Food Chem. 2009, 114, 1564–1569. [Google Scholar] [CrossRef]
- Ranieri, M.L.; Ivy, R.A.; Mitchell, W.R.; Call, E.; Masiello, S.N.; Wiedmann, M.; Boor, K.J. Real-time PCR detection of Paenibacillus spp. in raw milk to predict shelf life performance of pasteurized fluid milk products. Appl. Environ. Microbiol. 2012, 78, 5855–5863. [Google Scholar] [CrossRef] [PubMed]
- Balabin, R.M.; Smirnov, S.V. Melamine detection by mid-and near-infrared (MIR/NIR) spectroscopy: A quick and sensitive method for dairy products analysis including liquid milk, infant formula, and milk powder. Talanta 2011, 85, 562–568. [Google Scholar] [CrossRef] [PubMed]
- Borecki, M.; Niemiec, T.; Korwin-Pawlowski, M.L.; Kuczyńska, B.; Doroz, P.; Urbańska, K.; Szmidt, M.; Szmidt, J. Optoelectronic and photonic sensors of mastitis in cow milk. In Proceedings of the Electron Technology Conference, Ryn, Poland, 16–20 April 2013.
- Kaşıkçı, G.; Çetin, Ö.; Bingöl, E.B.; Gündüz, M.C. Relations between electrical conductivity, somatic cell count, California mastitis test and some quality parameters in the diagnosis of subclinical mastitis in dairy cows. Turk. J. Vet. Anim. Sci. 2012, 36, 49–55. [Google Scholar]
- Ozhikandathil, J.; Badilescu, S.; Packirisamy, M. Gold nanostructure-integrated silica-on-silicon waveguide for the detection of antibiotics in milk and milk products. In Proceedings of the Photonics North 2012, Montreal, QC, Canada, 6–8 June 2012.
- Hao, X.J.; Zhou, X.H.; Zhang, Y.; Liu, L.H.; Long, F.; Song, L.; Shi, H.C. Melamine detection in dairy products by using a reusable evanescent wave fiber-optic biosensor. Sens. Actuat. B Chem. 2014, 204, 682–687. [Google Scholar] [CrossRef]
- Narsaiah, K.; Jha, S.N.; Bhardwaj, R.; Sharma, R.; Kumar, R. Optical biosensors for food quality and safety assurance—A review. J. Food Sci. Technol. 2012, 49, 383–406. [Google Scholar] [CrossRef] [PubMed]
- Abdelhaseib, M.U.; Singh, A.K.; Bailey, M.; Singh, M.; El-Khateib, T.; Bhunia, A.K. Fiber optic and light scattering sensors: Complimentary approaches to rapid detection of Salmonella enterica in food samples. Food Control 2016, 61, 135–145. [Google Scholar] [CrossRef]
- Preejith, P.V.; Lim, C.S.; Kishen, A.; John, M.S.; Asundi, A. Total protein measurement using a fiber-optic evanescent wave-based biosensor. Biotechnol. Lett. 2003, 25, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Da Silveira, C.R.; Costa, J.C.; RoccoGiraldi, M.T.M.; Jorge, P.; López Barbero, A.P.; Germano, S.B. Bent optical fiber taper for refractive index measurements with tunable sensitivity. Microw. Opt. Technol. Lett. 2015, 57, 921–924. [Google Scholar] [CrossRef]
- Zawawi, M.A.; O’Keeffe, S.; Lewis, E. Plastic Optical Fibre Sensor for Spine Bending Monitoring with Power Fluctuation Compensation. Sensors 2013, 13, 14466–14483. [Google Scholar] [CrossRef] [PubMed]
- Borecki, M. Clad radiation rating in optical polymer fibres. Proc. SPIE 2006, 6159. [Google Scholar] [CrossRef]
- Borecki, M. Light behaviour in polymer optical fibre bend—A new analysis method. Opt. Appl. 2013, 33, 191–204. [Google Scholar]
- Borecki, M. Intelligent fiber optic sensor for estimating the concentration of a mixture-design and working principle. Sensors 2007, 7, 384–399. [Google Scholar] [CrossRef]
- Borecki, M.; Kruszewski, J. Skew radiation in optical fiber: The proposal of share measure. In Proceedings of the Optical Fibers: Applications, Warsaw, Poland, 31 August–2 September 2005.
- Fang, Y.-L.; Wang, C.-T.; Chiang, C.-C. A Small U-Shaped Bending-Induced Interference Optical Fiber Sensor for the Measurement of Glucose Solutions. Sensors 2016, 16, 1460. [Google Scholar] [CrossRef] [PubMed]
- Gupta, B.D.; Dodeja, H.; Tomar, A.K. Fibre-optic evanescent field absorption sensor based on a U-shaped probe. Opt. Quant. Electron. 1996, 28, 1629–1639. [Google Scholar] [CrossRef]
- Gupta, B.D.; Sharma, N.K. Fabrication and characterization of U-shaped fiber-optic pH probes. Sens. Actuat. B Chem. 2002, 82, 89–93. [Google Scholar] [CrossRef]
- HFBR-0500Z Series–Avago Technologies. Available online: http://www.avagotech.com/docs/AV02-1501EN (accessed on 8 December 2016).
- Saraçoglu, Ö.G.; Ozsoy, S. Simple equation to estimate the output power of an evanescent field absorption-based fiber sensor. Opt. Eng. 2002, 41, 598–600. [Google Scholar]
- Gloge, D. Weakly guiding fibers. Appl. Opt. 1971, 10, 2252–2258. [Google Scholar] [CrossRef] [PubMed]
- Cerdán, J.F.; Peris-Tortajada, M.; Puchades, R.; Maquieira, A. Automation of the determination of hydrogen peroxide, dichromate, formaldehyde and bicarbonate in milk by flow injection analysis. Fresenius J. Anal. Chem. 1992, 344, 123–127. [Google Scholar] [CrossRef]
- Snyder, A.W.; Love, J. Optical Waveguide Theory; Kluwer Academic Publishers: London, UK, 2000; pp. 179–188. [Google Scholar]

| Probe | Readouts (in Arbitrary Units) | I1−I0 | |
|---|---|---|---|
| I0 (in Air) | I1 (in Water) | ||
| P-131 | 1000 | 1002 | 2 |
| P-132 | 1000 | 1003 | 3 |
| P-133 | 1000 | 1005 | 5 |
| P-134 | 1000 | 1006 | 6 |
| P-135 | 1003 | 1017 | 14 |
| P-231 | 1002 | 1052 | 50 |
| P-232 | 1004 | 1113 | 109 |
| P-233 | 1005 | 1258 | 253 |
| P-234 | 1022 | NOP 1 | - |
| P-235 | 1043 | NOP | - |
| P-331 | 1012 | 1303 | 291 |
| P-332 | 1048 | NOP | - |
| P-333 | 1090 | NOP | - |
| P-334 | 1298 | NOP | - |
| P-335 | 1401 | NOP | - |
| Probe | Readouts (in Arbitrary Units) | I1−I0 | |
|---|---|---|---|
| I0 (in Air) | I1 (in Water) | ||
| P-131 | 1000 | 1002 | 2 |
| P-231 | 1002 | 1052 | 50 |
| P-331 | 1012 | 1303 | 291 |
| Hydrogen Peroxide-39 Concentration (%) | Refractive Index | I0 (in Air) | I1 (in Milk) | I1−I0 |
|---|---|---|---|---|
| 0.00000 | 1.34550 | 1008 | 1310 | 302 |
| 1.63934 | 1.34566 | 1008 | 1310 | 302 |
| 3.22581 | 1.34581 | 1008 | 1312 | 304 |
| 4.76190 | 1.34595 | 1008 | 1313 | 305 |
| 6.25000 | 1.34609 | 1008 | 1314 | 306 |
| 7.69231 | 1.34623 | 1008 | 1315 | 307 |
| 9.09091 | 1.34636 | 1008 | 1316 | 308 |
| 10.44776 | 1.34649 | 1008 | 1319 | 311 |
| 11.76471 | 1.34662 | 1008 | 1319 | 311 |
| 13.04348 | 1.34674 | 1008 | 1321 | 313 |
| 14.28571 | 1.34686 | 1008 | 1322 | 314 |
| Sodium Carbonate-12.5 Concentration (%) | Refractive Index | I0 (in Air) | I1 (in Milk) | I1−I0 |
|---|---|---|---|---|
| 0.00000 | 1.34550 | 1008 | 1310 | 302 |
| 1.63934 | 1.34571 | 1008 | 1314 | 306 |
| 3.22581 | 1.34591 | 1008 | 1318 | 310 |
| 4.76190 | 1.34611 | 1008 | 1322 | 314 |
| 6.25000 | 1.34630 | 1008 | 1324 | 316 |
| 7.69231 | 1.34648 | 1008 | 1326 | 318 |
| 9.09091 | 1.34666 | 1008 | 1328 | 320 |
| 10.44776 | 1.34684 | 1008 | 1330 | 322 |
| 11.76471 | 1.34701 | 1008 | 1330 | 322 |
| 13.04348 | 1.34717 | 1008 | 1331 | 323 |
| 14.28571 | 1.34733 | 1008 | 1332 | 324 |
| Formaldehyde-37 Concentration (%) | Refractive Index | I0 (in Air) | I1 (in Milk) | I1−I0 |
|---|---|---|---|---|
| 0.00000 | 1.34550 | 1008 | 1310 | 302 |
| 1.63934 | 1.34598 | 1008 | 1312 | 304 |
| 3.22581 | 1.34644 | 1008 | 1318 | 310 |
| 4.76190 | 1.34689 | 1008 | 1320 | 312 |
| 6.25000 | 1.34732 | 1008 | 1323 | 315 |
| 7.69231 | 1.34774 | 1008 | 1329 | 321 |
| 9.09091 | 1.34815 | 1008 | 1334 | 326 |
| 10.44776 | 1.34854 | 1008 | 1336 | 328 |
| 11.76471 | 1.34892 | 1008 | 1339 | 331 |
| 13.04348 | 1.34930 | 1008 | 1343 | 335 |
| 14.28571 | 1.34966 | 1008 | 1346 | 338 |
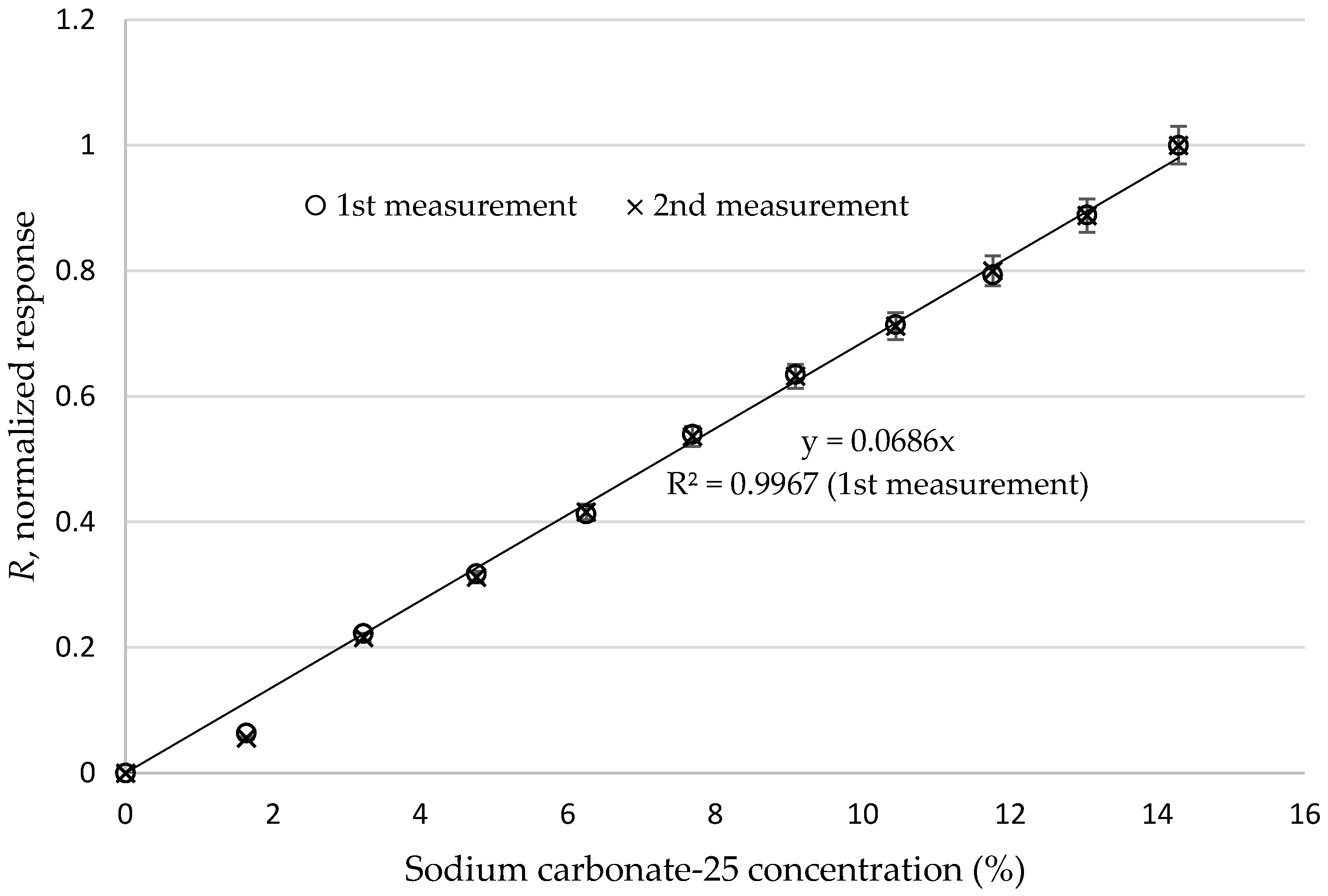
| Sodium Carbonate-25 Concentration (%) | Refractive Index | I0 (in Air) | I1 (in Milk) | I1−I0 |
|---|---|---|---|---|
| 0.00000 | 1.34550 | 1008 | 1310 | 302 |
| 1.63934 | 1.34612 | 1008 | 1314 | 306 |
| 3.22581 | 1.34673 | 1008 | 1324 | 316 |
| 4.76190 | 1.34731 | 1008 | 1330 | 322 |
| 6.25000 | 1.34788 | 1008 | 1336 | 328 |
| 7.69231 | 1.34842 | 1008 | 1344 | 336 |
| 9.09091 | 1.34895 | 1008 | 1350 | 342 |
| 10.44776 | 1.34947 | 1008 | 1355 | 347 |
| 11.76471 | 1.34997 | 1008 | 1360 | 352 |
| 13.04348 | 1.35046 | 1008 | 1366 | 358 |
| 14.28571 | 1.35093 | 1008 | 1373 | 365 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saracoglu, O.G.; Hayber, S.E. Bent Fiber Sensor for Preservative Detection in Milk. Sensors 2016, 16, 2094. https://doi.org/10.3390/s16122094
Saracoglu OG, Hayber SE. Bent Fiber Sensor for Preservative Detection in Milk. Sensors. 2016; 16(12):2094. https://doi.org/10.3390/s16122094
Chicago/Turabian StyleSaracoglu, Omer Galip, and Sekip Esat Hayber. 2016. "Bent Fiber Sensor for Preservative Detection in Milk" Sensors 16, no. 12: 2094. https://doi.org/10.3390/s16122094
APA StyleSaracoglu, O. G., & Hayber, S. E. (2016). Bent Fiber Sensor for Preservative Detection in Milk. Sensors, 16(12), 2094. https://doi.org/10.3390/s16122094







