Sea Slugs (Mollusca, Gastropoda, Heterobranchia) from the Medes Islands (Costa Brava, NE Spain): Biodiversity and Ecological Study over a Decade
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Sampling
2.3. Photographic Equipment
3. Results
3.1. General Biodiversity
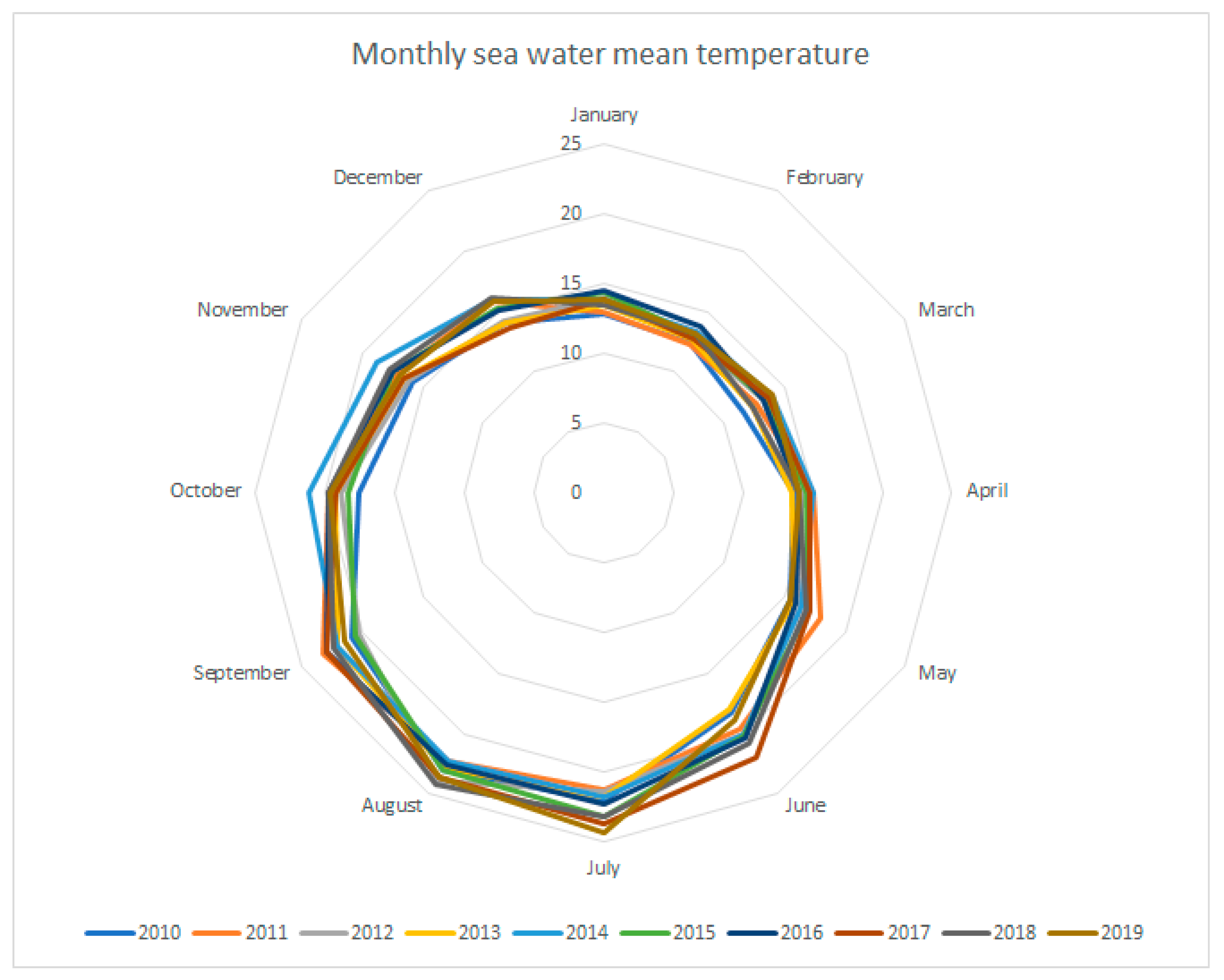
3.2. Abundance by Sampling Stations and Seasons
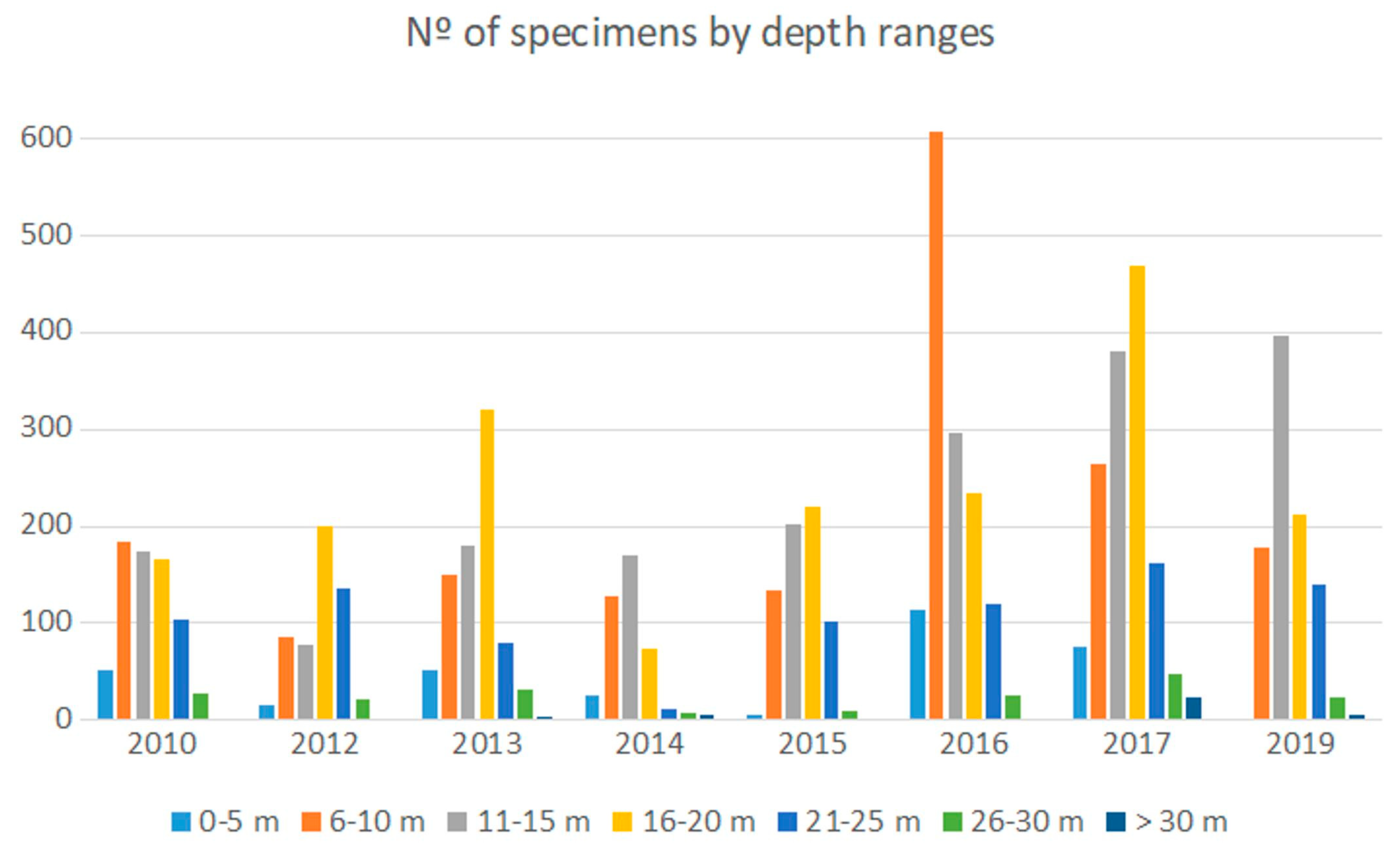
3.3. Abundance by Depth Intervals
3.4. Shannon–Wiener Index
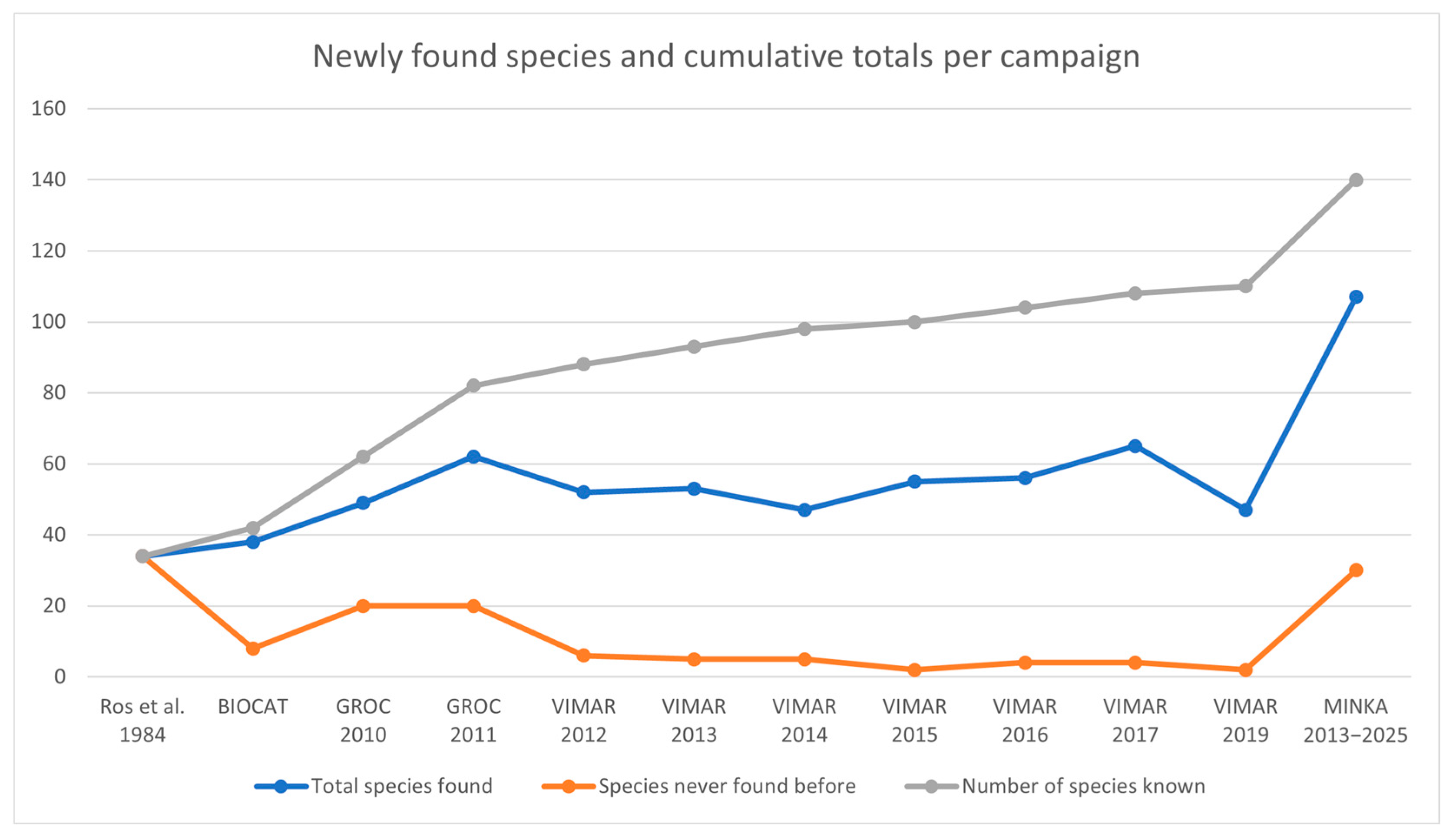
3.5. Later Findings
3.6. Annotated Taxonomic Checklist of Sea Slugs in the PNMMBT
- Class GASTROPODA
- Subclass Heterobranchia
- Subterclass Acteonimorpha
- Acteonidae
- * Acteon tornatilis 1
- Order Aplysiida
- Aplysiidae
- Aplysia depilans
- Aplysia fasciata
- Aplysia punctata 2
- Petalifera petalifera
- Phyllaplysia lafonti
- Order Cephalaspidea
- Aglajidae
- Aglaja tricolorata
- * Camachoaglaja africana 3
- Philinopsis depicta
- Bullidae
- * Bulla striata 4
- Cylichnidae
- * Cylichna cylindracea 5
- Haminoeidae
- Haminoea hydatis
- Haminoea navicula
- Philinidae
- * Hermania scabra 6
- * Philine catena 7
- * Philine quadripartita 8
- Order Nudibranchia
- Aeolidiidae
- Aeolidiella alderi
- Berghia coerulescens
- * Berghia verrucicornis 9
- * Cerberilla bernadettae 10
- * Limenandra nodosa 11
- Spurilla neapolitana
- Arminidae
- Armina neapolitana
- Cadlinidae
- Cadlina laevis
- Calycidorididae
- Diaphorodoris alba
- Diaphorodoris luteocincta 12
- Diaphorodoris papillata
- Chromodorididae
- Felimare bilineata
- Felimare fontandraui
- Felimare orsinii
- Felimare picta
- Felimare tricolor
- Felimare villafranca
- Felimida binza
- Felimida krohni
- Felimida luteorosea
- Felimida purpurea
- Coryphellidae
- Coryphella lineata 13
- Dendrodorididae
- Dendrodoris grandiflora
- Dendrodoris limbata
- * Dendrodoris temarana 14
- Doriopsilla rarispinosa 15
- Dendronotidae
- Scyllaea pelágica 47
- Discodorididae
- Geitodoris planata
- * Geitodoris portmanni 16
- Jorunna tomentosa
- * Paradoris indecora 17
- Peltodoris atromaculata
- Platydoris argo
- * Taringa armata 18
- Tayuva maculosa 19
- Dorididae
- Doris verrucosa
- Dotidae
- * Doto cervicenigra 20
- Doto coronata
- Doto dunnei
- Doto eireana
- Doto floridicola
- Doto koenneckeri
- Doto paulinae
- * Doto pygmaea 21
- Doto rosea
- Eubranchidae
- * Amphorina andra 22, 24
- * Eubranchus exiguus 23
- Eubranchus farrani 24
- Facelinidae
- Caloria elegans
- Caloria quatrefagesi
- Cratena peregrina
- Facelina annulicornis
- Facelina auriculata
- * Facelina dubia 25
- Facelina rubrovittata
- Facelina vicina 26
- Facelinopsis marioni
- Favorinus branchialis
- Fionidae
- * Fiona pinnata 27
- Flabellinidae
- Calmella cavolini
- Calmella gaditana
- Edmundsella pedata
- Flabellina affinis
- Paraflabellina gabinierei
- Paraflabellina ischitana
- Goniodorididae
- * Goniodoridella picoensis 28
- Okenia mediterranea
- Trapania lineata
- Trapania maculata
- Hancockiidae
- Hancockia uncinata
- Janolidae
- Antiopella cristata
- Myrrhinidae
- Nemesignis banyulensis
- Onchidorididae
- Atalodoris pusilla 29
- Atalodoris pictoni 30
- Atalodoris sparsa
- * Idaliadoris depressa 31
- Idaliadoris neapolitana
- Phyllidiidae
- Phyllidia flava
- Piseinotecidae
- Piseinotecus soussi
- Polyceridae
- Crimora papillata
- Limacia cf. iberica 32
- Limacia inesae 33
- * Martadoris mediterranea 34
- Polycera quadrilineata
- Samlidae
- Luisella babai
- Tethydidae
- Tethys fimbria
- Trinchesiidae
- Tenellia albopunctata
- Tenellia caerulea
- * Tenellia cuanensis 35
- * Tenellia foliata 36
- Tenellia genovae
- Tenellia miniostriata
- Tenellia morrowae 37
- Tenellia ocellata
- Tritoniidae
- Candiella lineata
- Candiella manicata
- Candiella odhneri 38
- Candiella striata
- Marionia blainvillea
- Order Pleurobranchida
- Pleurobranchidae
- Berthella aurantiaca 39
- Berthella ocellata
- Berthella perforata 40
- Berthellina edwardsii
- Pleurobranchaea meckeli
- Pleurobranchus testudinarius
- Order Pteropoda
- Creseidae
- * Creseis acicula 41
- Superorder Ringiculimorpha
- Ringiculidae
- * Ringicula conformis 42
- Order Runcinida
- Runcinidae
- Runcina adriatica
- Runcina bahiensis
- Runcina coronata
- * Runcina cf. ornata 43
- Superorder Sacoglossa
- Hermaeidae
- Hermaea variopicta
- Limapontiidae
- Placida cremoniana
- Placida dendritica
- Placida verticilata
- * Placida viridis 44
- Oxynoidae
- * Oxynoe olivacea 45
- Plakobranchidae
- Bosellia mimetica
- Elysia flava
- Elysia gordanae 46
- Elysia timida
- Elysia viridis
- Thuridilla hopei
- Order Umbraculida
- Tylodinidae
- Tylodina perversa
- Umbraculidae
- Umbraculum umbraculum
- Observation and Taxonomical Notes
- (1)
- First report of Acteon tornatilis in the PNMMBT was recorded on 9 January 2021 at Griells by Xavier Salvador (MINKA—https://minka-sdg.org/observations/74189—accessed on 7 July 2025).
- (2)
- A recent study by Golestani et al. [28] concluded that all Mediterranean Sea hares previously identified as Aplysia parvula actually belong to Aplysia punctata. As a result, our records have been synonymized accordingly.
- (3)
- First report of Camachoaglaja africana in the PNMMBT was recorded on 4 June 2023 at the Falaguer dive site by Joan Roig (MINKA—https://minka-sdg.org/observations/130896—accessed on 7 July 2025).
- (4)
- First report of Bulla striata in the PNMMBT was recorded on 29 June 2024 at Cala Montgó by Xavier Salvador (MINKA—https://minka-sdg.org/observations/289396—accessed on 7 July 2025).
- (5)
- First report of Cylichna cylindracea in the PNMMBT was recorded on 9 January 2021 at Griells by Xavier Salvador (MINKA—https://minka-sdg.org/observations/74187—accessed on 7 July 2025).
- (6)
- First report of Hermania scabra in the PNMMBT was recorded on 20 July 2024 at Griells by Xavier Salvador (MINKA—https://minka-sdg.org/observations/306206—accessed on 7 July 2025).
- (7)
- First report of Philine catena in the PNMMBT was recorded on 26 December 2017 at Cala Montgó by Xavier Salvador (MINKA—https://minka-sdg.org/observations/34350—accessed on 7 July 2025).
- (8)
- First report of Philine quadripartita in the PNMMBT was recorded on 14 July 2024 at Cala Montgó by Xavier Salvador (MINKA—https://minka-sdg.org/observations/299032—accessed on 7 July 2025).
- (9)
- First report of Berghia verrucicornis in the PNMMBT was recorded on 14 September 2017 at Cala Montgó by Margot Bosch (MINKA—https://minka-sdg.org/observations/482580—accessed on 7 July 2025).
- (10)
- First report of Cerberilla bernadettae in the PNMMBT was recorded on 18 February 2021 at Griells by Xavier Salvador (MINKA—https://minka-sdg.org/observations/74254—accessed on 7 July 2025).
- (11)
- First report of Limenandra nodosa in the PNMMBT was recorded on 25 July 2016 at Cala Montgó by Guillem Mas (MINKA—https://minka-sdg.org/observations/480229—accessed on 7 July 2025).
- (12)
- Many authors previously considered Diaphorodoris luteocincta var. alba and var. reticulata to be morphological variants of the same species. However, morphological and molecular analyses by Furfaro et al. [29] refute this view, demonstrating that they represented two distinct species: D. alba and D. luteocincta, respectively. Our records of D. luteocincta prior to the taxonomic revision by Furfaro et al. [29] include individuals that are now identified as D. alba.
- (13)
- Flabellina lineata was reassigned to the genus Fjordia by Korshunova et al. [31] following molecular and morphological analyses that redefined the family Flabellinidae and established several new genera within Coryphellidae. However, a subsequent comprehensive revision of the family by Ekimova et al. [32] proposed a more conservative approach, arguing that excessive splitting—especially in allopatric taxa—led to taxonomic instability. As a result, they synonymized several recently erected genera, including Fjordia, under an expanded concept of Coryphella, currently accepted as the valid name in taxonomic databases like WoRMS.
- (14)
- First report of Dendrodoris temarana in the PNMMBT was recorded on 30 June 2025 at Platja de Pals by Marta N.P. (MINKA—https://minka-sdg.org/observations/510533—accessed on 7 July 2025). Galià-Camps et al. [33] have recently reinstated this species, originally described by Pruvot-Fol in 1953 but considered, until now, synonymous with D. grandiflora.
- (15)
- Furfaro et al. [34] revealed that Doriopsilla areolata (sensu lato) represents a Mediterranean cryptic species complex, including the reinstated D. rarispinosa (previously a synonym) and at least one putative lineage (Doriopsilla sp1). True D. areolata (s.s.) is currently confirmed only in the Adriatic Sea, whereas morphologically similar Atlantic and western Mediterranean individuals likely belong to the still-undescribed taxa Doriopsilla sp1. Our records in the PNMMBT correspond strictly to D. rarispinosa.
- (16)
- First report of Geitodoris portmanni in the PNMMBT area of influence was recorded on 16 July 2015 at Cala Aiguafreda by Xavier Salvador (MINKA—https://minka-sdg.org/observations/24820—accessed on 7 July 2025).
- (17)
- First report of Paradoris indecora in the PNMMBT was recorded on 26 December 2017 at Cala Montgó by Xavier Salvador (MINKA—https://minka-sdg.org/observations/34361—accessed on 7 July 2025).
- (18)
- First report of Taringa armata in the PNMMBT was recorded on 18 February 2021 at Griells by Xavier Salvador (MINKA—https://minka-sdg.org/observations/74256—accessed on 7 July 2025).
- (19)
- Dayrat’s [35] revision of Discodorididae consolidated several morphologically similar species into broader taxa, including the synonymization of the Mediterranean Discodoris maculosa under the circumtropical Tayuva lilacina. However, molecular studies [36] revealed T. lilacina to be a cryptic species complex. Mediterranean individuals were reassigned to Tayuva maculosa (Bergh, 1884), while northeastern Atlantic populations were resurrected as Tayuva confusa [37]. The study also identified a close phylogenetic relationship between Tayuva and Peltodoris, noting historical synonymies that warrant further genomic investigation. We adopt the classification of this species as Tayuva maculosa, currently accepted by WoRMS.
- (20)
- First report of Doto cervicenigra in the PNMMBT was recorded on 18 February 2019 in the port of L’Estartit by Xavier Salvador (MINKA—https://minka-sdg.org/observations/40684—accessed on 7 July 2025).
- (21)
- First report of Doto pygmaea in the PNMMBT was recorded on 20 July 2024 at Griells by Xavier Salvador (MINKA—https://minka-sdg.org/observations/306188—accessed on 7 July 2025).
- (22)
- Recent work by Toso et al. [38] proposed the reclassification of Amphorina andra as a junior synonym of Eubranchus viriola, citing occurrences of A. andra in brackish habitats as supporting evidence. However, this conclusion has been met with significant reservations. Martynov (pers. comm., 2025) highlights several critical concerns, including potential misidentification of individuals and the well-documented morphological and epigenetic distinctions of A. viriola, a taxon originally described from the brackish transition zone between the North and Baltic Seas [39]. Notably, A. viriola exhibits consistent phenotypic differences in this unique habitat, reinforcing its separation from A. andra. Given the lack of comprehensive molecular or ecological analysis in the proposed synonymy, we propose to provisionally keep the validity of A. andra until further studies are conducted to demonstrate whether it is Eubranchus viriola, or not. First report of Amphorina andra in the PNMMBT was recorded on 3 February 2021 at Griells by Xavier Salvador (MINKA—https://minka-sdg.org/observations/74245—accessed on 7 July 2025).
- (23)
- First report of Eubranchus exiguus in the PNMMBT was recorded on 2 November 2020 at Griells by Xavier Salvador (MINKA—https://minka-sdg.org/observations/42343—accessed on 7 July 2025).
- (24)
- The current classification of Fionoidea reflects a fundamental dichotomy in systematic philosophy. One perspective advocates for narrowly circumscribed genera based on fine-scale morphological and molecular distinctions, an approach that frequently results in monotypic genera. While this methodology serves to highlight proposed autapomorphies, it has been criticized for promoting taxonomic inflation and instability, particularly when such distinctions are not supported by robust synapomorphic evidence. An alternative framework favors the recognition of broader, more inclusive genera, emphasizing stability and practical utility. This approach is particularly justified in groups where much of the diversity remains undescribed or poorly characterized. By maintaining larger, morphologically cohesive units, this system reduces nomenclatural disruption while still reflecting evolutionary relationships. A prudent course of action would be to adopt a conservative classification that recognizes well-supported clades without unnecessary splitting. Genera should be subdivided only when supported by unambiguous synapomorphies and comprehensive phylogenetic evidence. This balanced approach would maintain scientific rigor while ensuring the classification remains accessible to both specialists and non-specialists alike. This would provide much-needed stability while still allowing for future refinements as our understanding of fionid phylogeny improves. We currently adopt the classification of former Amphorina farrani as Eubranchus farrani, currently accepted by WoRMS.
- (25)
- First report of Facelina dubia in the PNMMBT was recorded on 24 July 2022 at Griells by Xavier Salvador (MINKA—https://minka-sdg.org/observations/73574—accessed on 7 July 2025).
- (26)
- Facelina vicina is often confused with Facelina bostoniensis, an amfiatlantic species initially described on the shores of Massachusetts and Maine (USA), and also present in the Atlantic European shores from Norway to the Iberian Peninsula (Cantabria and Portugal). According to Carmona [40], reports of F. bostoniensis from the Mediterranean Sea correspond to Facelina vicina (Bergh, 1882).
- (27)
- Fiona pinnata is a cosmopolitan species that has been reported from all the world’s oceans. In Catalonia, it has consistently been found associated with floating objects that reach the coast. First report in the PNMMBT influence area was recorded on 11 August 2024 by Xavier Salvador (MINKA—https://minka-sdg.org/observations/326651—accessed on 7 July 2025).
- (28)
- Originally described from Pico Island in the Azores [41], Goniodoridella picoensis has shown an exceptionally rapid spread across the Mediterranean Sea. Since the first Iberian record in La Herradura, southern Spain [42], it has been reported in numerous locations from the Alboran Sea to Israel within months, as detailed by Trainito et al. [43]. It reached Catalonia (Tamariu) by June 2021, the Balearic Islands by early 2022, and by 2023 had become frequent along the Catalan coast, including the PNMMBT.
- (29)
- Onchidoris albonigra was originally described from the Mediterranean, but its poorly defined diagnosis led to confusion. Schmekel & Portmann [44] attempted a redescription based on Gulf of Naples material, yet inconsistencies persisted. Subsequent records, including those by Perrone [45] and Betti et al. [46], likely refer to Atalodoris pictoni or A. pusilla. Catalan individuals identified as O. albonigra (OPK, 2012) have since been confirmed as A. pusilla through both morphology and molecular data. Given the lack of type material and overlapping features—especially in mantle pigmentation and gill/rhinophore structure—Furfaro et al. [47] regarded O. albonigra as a junior synonym of A. pusilla following ICZN guidelines.
- (30)
- Atalodoris pictoni was recently described after being long misidentified as Onchidoris pusilla due to shared external features such as a dark brown dorsum and white rhinophores. However, it differs in having dark gills matching the body color, unlike the white gills of O. pusilla. Hallas & Gosliner [48] restructured the Onchidorididae, merging Onchidoris and Adalaria (with median rachidian tooth) and creating Knoutsodonta for species lacking it. However, they overlooked the earlier genus Atalodoris Iredale & O’Donoghue, 1923—whose type species, Doris pusilla, also lacks a median tooth—rendering Knoutsodonta a junior synonym. Of the fifteen species listed under Knoutsodonta by Furfaro & Trainito [49], six are Mediterranean; Ortea [50] proposes transferring all species to Atalodoris in line with taxonomic priority.
- (31)
- Furfaro et al. [47] introduced a new genus Idaliadoris Furfaro & Trainito, 2022 (type species Idaliadoris neapolitana) grouping eight species with finger-like dorsal papillae, more or less swollen, and with a common egg spawn shape. According to Furfaro et al. [47], the shape of the egg masses was revealed to be a useful diagnostic character allowing distinction between genera in the Onchidorididae systematics. First report of Idaliadoris depressa in the area of influence of the PNMMBT was recorded on 11 September 2015 at Punta de la Creu, Begur by Xavier Salvador (MINKA—https://minka-sdg.org/observations/4013—accessed on 7 July 2025).
- (32)
- Limacia iberica is morphologically very similar to Limacia clavigera, with which it has been probably confused due to the poor description by Müller [51], who in 1776 described it with six words, literally, although the author later expanded the description with further information and illustrations in 1788 and 1806. These species differ in that Limacia clavigera has far fewer lateral papillae (20–24) and, in addition, they are much shorter and with an orange tip (not white as in L. iberica). Limacia clavigera has a tuberculated and much less spiculated dorsum, while in L. iberica there is a single mid-dorsal row of eight tubercles, and the mantle spicules are so long and abundant that they “break” the skin and can be seen with the naked eye. The tail of Limacia clavigera is longer and more stylized and with distinct yellow spots. The rhinophores are shorter and have fewer lamellae (10–14). A specimen with mixed traits between L. iberica and L. clavigera was found at Punta Salines (L’Estartit, Spain) on 19 September 2019 by Glenn Biscop (OPK—https://opistobranquis.info/wp-content/uploads/2019/10/Limacia-cf.-iberica-with-mixed-traits-with-L.clavigera-@-Punta-Salines-LEstartit-19.09.2019-by-Glenn-Biscop-P9190364LR.jpg—accessed on 7 July 2025).
- (33)
- Historically misidentified with Limacia clavigera, according to the study by Toms et al. [52], the species L. clavigera appears to be strictly distributed in Atlantic waters from Norway to the coasts of western Andalusia (Spain), while the individuals of L. inesae, smaller in size, are distributed throughout the Mediterranean, Canary Islands, Madeira, and the Azores.
- (34)
- Martadoris mediterranea closely resembles M. limaciformis, originally described in the Red Sea, with both species exhibiting a deep saffron-red body with scattered pale spots. However, M. limaciformis is distinguished by purple tips on the rhinophores and gills [53,54]. To rule out the hypothesis of a Lessepsian introduction, Domínguez et al. [55] conducted phylogenetic analyses confirming that M. mediterranea is a distinct species. It was subsequently assigned to the newly established genus Martadoris by Willan & Chang [56]. The first record of Martadoris mediterranea in the PNMMBT was recorded by Elena Bretaudeau on 1 July 2024, at a depth of 50 meters (OPK—https://opistobranquis.info/wp-content/uploads/2024/07/image_123650291-1.jpg—accessed on 7 July 2025).
- (35)
- As with the family Eubranchidae, Trinchesiidae illustrates the broader debate between narrowly splitting genera based on fine-scale morphological and molecular differences and maintaining more inclusive, morphologically cohesive groups. In this case, we adopt the terminology proposed by several authors and currently accepted by WoRMS, transferring the Mediterranean species formerly placed in Trinchesia to the genus Tenellia, pending future studies that may resolve their taxonomic position. As previously emphasized, a balanced approach—splitting taxa only when shared, unambiguous traits are clearly supported—maintains scientific rigor while ensuring the classification remains accessible to both specialists and non-specialists. Historically, Tenellia caerulea and T. cuanensis were considered the same species due to their overall morphological and coloration patterns similarities. However, Korshunova et al. [57] demonstrated, through an integrative approach combining molecular and morphological data, that these represent distinct species. Each shows consistent genetic divergence and subtle but stable differences in radular morphology and body coloration, supporting their recognition as separate taxa.
- First report of Tenellia cuanensis in the area of influence of the PNMMBT was recorded (as Cuthona caerulea) on 14 February 2015 at Punta del Romaní, L’Escala by Guillem Mas and Josep Lluís Peralta (MINKA—https://minka-sdg.org/observations/474948—accessed on 7 July 2025).
- (36)
- First report of Tenellia foliata in the area of influence of the PNMMBT was recorded on 2 July 2015 at Punta del Romaní, L’Escala by Guillem Mas (MINKA—https://minka-sdg.org/observations/476475—accessed on 7 July 2025).
- (37)
- Historically, Tenellia morrowae and T. caerulea were considered the same species due to their overall morphological and coloration patterns similarities. However, Korshunova et al. [57] demonstrated, through an integrative approach combining molecular and morphological data, that these represent distinct species. Each shows consistent genetic divergence and subtle but stable differences in radular morphology and body coloration, supporting their recognition as separate taxa.
- (38)
- Candiella odhneri was originally described by J. Tardy [58] as Duvaucelia odhneri, later transferred to Tritonia as Tritonia odhneri (Tardy, 1963). However, this name was preoccupied by Tritonia odhneri Marcus, 1959, from the Chilean Pacific. To resolve the homonymy, Eveline Marcus [59] proposed the replacement name Tritonia nilsodhneri, which remained in use until late 2020. At that time, Korshunova & Martynov [60] reinstated the genus Duvaucelia to accommodate small Tritoniidae species characterized by a non-bilobed oral veil and a moderate number of lateral radular teeth, mainly found in temperate and subtropical European waters. More recently, De Vasconcelos et al. [61] demonstrated that the original description of Duvaucelia gracilis (type species of Duvaucelia) actually referred to a specimen of Marionia blainvillea, not Duvaucelia manicata as previously assumed. Consequently, Duvaucelia is no longer considered a valid genus, and the name Candiella has been reinstated, based on its type species Candiella plebeia.
- (39)
- Berthella aurantiaca has traditionally been confused with Berthellina edwardsii due to their remarkably similar external morphology—so much so that they are virtually indistinguishable by eye alone. Reliable differentiation between the two requires examination of the radula and mandibular structures. In B. edwardsii, the radula bears over 150 finely denticulate teeth per half-row, and the jaws are elongated with smooth mandibular plates. In contrast, Berthella aurantiaca has significantly fewer radular teeth (typically 50–80 per half-row), shorter jaws, and mandibular plates with lateral denticles. Adult individuals may also be distinguished by shell size: in B. edwardsii, the internal shell never exceeds 5.5 mm even in animals up to 60 mm long, as shell growth ceases around 15 mm body length (Jakov Prkić, pers. comm.). In B. aurantiaca, the shell is proportionally larger—Vayssière reported a 15 mm shell in a 30 mm individual—as it continues to grow and encloses the entire visceral mass. We have no reason to question the B. aurantiaca identifications of historical records, and therefore consider the records of this species in the PNMMBT to be valid. However, our observations over the years along the Catalan coast indicate that all examined individuals corresponded to Berthellina edwardsii.
- (40)
- Following the study by Ghanimi et al. [62] on the Berthella stellata species complex, it has been shown that the species previously identified as Berthella plumula (Montagu, 1803) actually comprises two distinct taxa. The name B. plumula is retained for Atlantic populations, while the Mediterranean individuals are now referred to as Berthella perforata (Philippi, 1844).
- (41)
- First report of Creseis acicula in the PNMMBT was recorded on 19 April 2025 at Cala Montgó, L’Escala by Enric Badosa (MINKA—https://minka-sdg.org/observations/441760—accessed on 7 July 2025).
- (42)
- First report of Ringicula conformis in the PNMMBT was recorded on 14 July 2024 at Cala Montgó, L’Escala by Xavier Salvador (MINKA—https://minka-sdg.org/observations/299037—accessed on 7 July 2025).
- (43)
- There is a report of what the observer believes is Runcina ornata in the PNMMBT, recorded on 26 December 2017 at Cala Montgó, L’Escala by Xavier Salvador (MINKA—https://minka-sdg.org/observations/34349—accessed on 7 July 2025). However, there are several authors [63,64,65] that already hinted at the possibility that Mediterranean individuals traditionally assigned to R. ornata might not correspond to the original species described from New Zealand. They acknowledge the lack of consistent diagnostic features and raise the issue of potential cryptic diversity and the need for further taxonomic work.
- (44)
- First report of Placida viridis in the PNMMBT was recorded on 5 February 2021 at Griells by Xavier Salvador (MINKA—https://minka-sdg.org/observations/74253—accessed on 7 July 2025).
- (45)
- First report of Oxynoe olivacea in the PNMMBT was recorded on 2 November 2024 at Cala Montgó, L’Escala by Eduardo Pérez (MINKA—https://minka-sdg.org/observations/397576—accessed on 7 July 2025).
- (46)
- The genus Elysia accounts for approximately one-third of sacoglossan diversity, yet its species composition in the northeastern Atlantic and Mediterranean has remained poorly understood. To address this gap, Martín-Hervás et al. [66] conducted an integrative study combining molecular phylogenetics, morphology, literature review, and species delimitation methods. Their analyses confirmed the presence of five European Elysia species: E. viridis, E. timida, E. flava, E. margaritae, and E. rubeni. Crucially, they revalidated Elysia gordanae as a distinct species, rejecting the synonymy with E. margaritae proposed by Ortea et al. [67], which had been questionable due to stark differences in adult size. Furthermore, they re-examined the status of Elysia hetta, originally described by Perrone [68] based on morphological traits now recognized as falling within the intraspecific variability of E. gordanae. Martín-Hervás et al. concluded that E. hetta is likely a junior synonym of E. gordanae, supported by photographic and descriptive comparisons with both the original material and the subsequent literature.
- (47)
- First report of Scyllaea pelagica in the PNMMBT was recorded on 7 August 2025 at Ponedora de Sípies by Boris Weitzmann (MINKA—https://minka-sdg.org/observations/537411—accessed on 7 July 2025).
4. Discussion and Conclusions
Author Contributions
Funding
Institution Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Carmel, Y.; Kent, R.; Bar-Massada, A.; Blank, L.; Liberzon, J.; Nezer, O.; Sapir, G.; Federman, R. Trends in Ecological Research during the Last Three Decades—A Systematic Review. PLoS ONE 2013, 8, e59813. [Google Scholar] [CrossRef]
- Doney, S.C.; Ruckelshaus, M.; Duffy, J.E.; Barry, J.P.; Chan, F.; English, C.A.; Galindo, H.M.; Grebmeier, J.M.; Hollowed, A.B.; Knowlton, N.; et al. Climate Change Impacts on Marine Ecosystems. Ann. Rev. Mar. Sci. 2012, 4, 11–37. [Google Scholar] [CrossRef] [PubMed]
- M@re Nostrum. Available online: http://marenostrum.org/ (accessed on 5 July 2025).
- Sea Slug Forum. Available online: http://www.seaslugforum.net/ (accessed on 5 July 2025).
- Mediterranean Slug Site. Available online: https://web.archive.org/web/20241203114122/http://www.medslugs.de/ (accessed on 5 July 2025).
- OPK—Opistobranquis. Available online: https://opistobranquis.info/en/ (accessed on 5 July 2025).
- GROC. Grup de Recerca d’Opistobranquis de Catalunya. Available online: https://opistobranquis.org/en/home (accessed on 5 July 2025).
- Ros, J.D. Catálogo provisional de los opistobranquios (Gastropoda: Euthyneura) de las costas ibéricas. Misc. Zool. 1976, 3, 21–51. [Google Scholar]
- Ballesteros, M. Contribución al Conocimiento de los Sacoglosos y Nudibranquios (Mollusca: Opisthobranchia). Estudio Anatómico, Sistemático y Faunístico de las Especies del Mediterráneo Español. Ph.D. Thesis, University of Barcelona, Barcelona, Spain, 1980; 367p. [Google Scholar]
- Ballesteros, M. Lista actualizada de los opistobranquios (Mollusca: Gastropoda: Opisthobranchia) de las costas catalanas. Spira 2007, 2, 163–188. [Google Scholar]
- Ballesteros, M.; Madrenas, E.; Pontes, M. Actualización del catálogo de los moluscos opistobranquios (Gastropoda: Heterobranchia) de las costas catalanas. Spira 2016, 6, 1–28. [Google Scholar]
- Ballesteros Vázquez, M.; Madrenas-Tomàs, E.; Pontes-Garcia, M. Els Nudibranquis del mar Catalá. Guía de camp. Col·lecció Maluquer 3; Brau Edicions: Girona, Spain, 2019; 192p. [Google Scholar]
- Doménech, A.; Ávila, C.; Ballesteros, M. Spatial and Temporal variability of the Opisthobranch molluscs of Port Lligat Bay (Catalonia, NE Spain). J. Mollus. Stud. 2002, 68, 29–37. [Google Scholar] [CrossRef]
- Dacosta, J.M.; Pontes, M.; Ollé, A.; Aguilar, L. Seguiment de mol·luscs opistobranquis a la platja des Caials (Cadaqués, Alt Empordà). Contribució al catàleg del Parc Natural de Cap de Creus. Ann. L’inst. D’estud. Empord. 2009, 40, 107–130. [Google Scholar]
- Parera, A.; Pontes, M.; Salvador, X.; Ballesteros, M. Sea slugs (Mollusca, Gastropoda, Heterobranchia), the other inhabitants of the city of Barcelona (Spain). Butll. Inst. Cat. Hist. Nat. 2020, 84, 75–100. [Google Scholar] [CrossRef]
- Ballesteros, M.; Pontes, M. Heterobranch Sea Slugs from Blanes (Costa Brava, NE Spain). VIMAR-Vida Marina 2024. Available online: https://vidamarina.info/?p=6557 (accessed on 18 March 2024).
- Programa Bentos. 1972–1974. Estudi Ecològic de les Comunitats Bentòniques Marines de Substracte dur, Departament Ecologia, Universitat de Barcelona: Barcelona, Spain, Unpublished.
- Programa Medes I, 1976–1978. Estudi Ecològic de les Comunitats Terrestres i Marínes de les Illes Medes, Departament Ecologia, Universitat de Barcelona: Barcelona, Spain, Unpublished.
- Programa Medes II, 1980–1981. Estudi Ecològic dels Sistemes Bentònics de les Illes Medes, Departament Ecologia, Universitat de Barcelona: Barcelona, Spain, Unpublished.
- Ros, J.D.; Olivella, I.; Gili, J.M. Els Sistemas Naturals de les Illes Medes; Institut d’Estudis Catalans: Barcelona, Spain, 1984; 828p. [Google Scholar]
- Altimira, C.; Huelin, M.F.; Ros, J.D. Mol·luscs bentònics de les illes Medes (Girona). I. Sistemàtica. Butll. Inst. Cat. Hist. Nat. 1981, 47, 69–75. [Google Scholar]
- Huelin, M.F.; Ros, J.D. Els mol.luscs marins de les Illes Medes. In Els Sistemes Naturals de les Illes Medes; Ros, J.D., Olivella, I., Gili, J.M., Eds.; Arxius Secció de Ciències: Barcelona, Spain, 1984; Volume 73, pp. 457–504. [Google Scholar]
- BIOCAT, Biodiversity Data Bank of Catalonia. Available online: http://biodiver.bio.ub.es/biocat/ (accessed on 7 July 2024).
- VIMAR. Seguiment i Actualització del Catàleg dels Opistobranquis del Parc Natural del Montgrí, les Illes Medes i el Baix Ter; Informe 2017; VIMAR: Barcelona, Spain, 2017; 45p. [Google Scholar]
- WoRMS Editorial Board. World Register of Marine Species. 2025. Available online: https://www.marinespecies.org (accessed on 13 August 2025).
- Servei Meteorològic de Catalunya. Available online: https://mediambient.gencat.cat/ca/05_ambits_dactuacio/meteorologia/ (accessed on 4 July 2025).
- MINKA Citizen Observatory; EMBIMOS Research Group; Institut de Ciències del Mar (ICM-CSIC). Project: Llimacs Marins del PNMMBT. VIMAR Vida Marina. 2025. Available online: https://minka-sdg.org/projects/llimacs-marins-del-pnmmbt (accessed on 7 July 2025).
- Golestani, H.; Crocetta, F.; Padula, V.; Camacho-García, Y.; Langeneck, J.; Poursanidis, D.; Pola, M.; Yokeş, M.B.; Cervera, J.L.; Jung, D.-W.; et al. The little Aplysia coming of age: From one species to a complex of species complexes in Aplysia parvula (Mollusca: Gastropoda: Heterobranchia). Zool. J. Linn. Soc. 2019, 187, 279–330. [Google Scholar] [CrossRef]
- Furfaro, G.; Picton, B.; Martynov, A.; Mariottini, P. Diaphorodoris alba Portmann & Sandmeier, 1960 is a valid species: Molecular and morphological comparison with D. luteocincta (M. Sars, 1870) (Gastropoda: Nudibranchia). Zootaxa 2016, 4193, 304–316. [Google Scholar] [CrossRef]
- Furfaro, G.; Mariottini, P. Check-list of the Nudibranchs (Mollusca Gastropoda) from the biodiversity hot spot “Scoglio del Corallo” (Argentario promontory, Tuscany). Biodivers. J. 2016, 7, 67–78. [Google Scholar]
- Korshunova, T.; Martynov, A.; Bakken, T.; Evertsen, J.; Fletcher, K.; Mudianta, W.; Saito, H.; Lundin, K.; Schrödl, M.; Picton, B. Polyphyly of the traditional family Flabellinidae affects a major group of Nudibranchia: Aeolidacean taxonomic reassessment with descriptions of several new families, genera, and species (Mollusca, Gastropoda). ZooKeys 2017, 717, 1–139. [Google Scholar] [CrossRef]
- Ekimova, I.; Valdés, Á.; Malaquias, M.A.E.; Rauch, C.; Chichvarkhin, A.; Mikhlina, A.; Antokhina, T.; Chichvarkhina, O.; Schepetov, D. High-level taxonomic splitting in allopatric taxa causes confusion downstream: A revision of the nudibranch family Coryphellidae. Zool. J. Linn. Soc. 2022, 196, 215–249. [Google Scholar] [CrossRef]
- Galià-Camps, C.; Cervera, J.L.; Valdés, Á.; Ballesteros, M. Attack on crypsis: Molecular and morphological study of Dendrodoris Ehrenberg, 1831 (Mollusca: Gastropoda: Nudibranchia) from the Mediterranean Sea and Northern Atlantic Ocean reinstates Dendrodoris temarana Pruvot-Fol, 1953. Zootaxa 2022, 5133, 383–406. [Google Scholar] [CrossRef] [PubMed]
- Furfaro, G.; Schreier, C.; Trainito, E.; Pontes, M.; Madrenas, E.; Girard, P.; Mariottini, P. The Sea Slug Doriopsilla areolata Bergh, 1880 (Mollusca, Gastropoda) in the Mediterranean Sea: Another Case of Cryptic Diversity. Diversity 2022, 14, 297. [Google Scholar] [CrossRef]
- Dayrat, B. A Monographic Revision of Basal Discodorid Sea Slugs (Mollusca: Gastropoda: Nudibranchia: Doridina). Proc. Calif. Acad. Sci. Fourth Ser. 2010, 61 (Suppl. I), 1–403. [Google Scholar]
- Fernández-Vilert, R.; Arnedo, M.A.; Salvador, X.; Valdés, Á.; Schrödl, M.; Moles, J. Shining disco: Shedding light into the systematics of the family Discodorididae (Gastropoda: Nudibranchia). Zool. J. Linn. Soc. 2025, 203, zlae170. [Google Scholar] [CrossRef]
- Ballesteros, M.; Ortea, J.A.; Llera, E. Revisión de los doridáceos del Atlántico nordeste atribuibles al complejo Maculosa-fragilis. Boll. Malacol. 1984, 20, 227–257. [Google Scholar]
- Toso, Y.; Martini, F.; Riccardi, A.; Furfaro, G. Unraveling the Sea Slug Fauna from an Extremely Variable Environment, The ‘Passetto’ Rocky Tide Pools (North Adriatic Sea). Water 2024, 16, 1687. [Google Scholar] [CrossRef]
- Korshunova, T.; Malmberg, K.; Prkić, J.; Petani, A.; Fletcher, K.; Lundin, K.; Martynov, A. Fine-scale species delimitation: Speciation in process and periodic patterns in nudibranch diversity. ZooKeys 2020, 917, 15–50. [Google Scholar] [CrossRef]
- Carmona, L. Investigating the amphiatlantic status of Facelina bostoniensis (Couthouy, 1838) (Nudibranchia: Aeolidida). J. Moll. Stud. 2020, 86, 64–71. [Google Scholar] [CrossRef]
- Paz-Sedano, S.; Ortigosa, D.; Pola, M. A new Okenia Menke, 1830 from the Azores Islands, Portugal (Mollusca, Nudibranchia, Goniodorididae). Spixiana 2017, 40, 13–22. [Google Scholar]
- Pontes, M.; Madrenas, E. First records of the NE Atlantic nudibranch Okenia picoensis Paz-Sedano, Ortigosa & Pola, 2017 (Gastropoda, Goniodorididae) in the Mediterranean Sea. In New Alien Mediterranean Biodiversity Records (March 2021); Mediterr. Mediterranean Marine Science: Athens, Greece, 2021; Volume 2, pp. 186–187. [Google Scholar]
- Trainito, E.; Migliore, V.; Doneddu, M. How many seas must a nudibranch sail? Okenia picoensis (Mollusca: Nudibranchia: Goniodorididae) conquering the Mediterranean. Stud. Mar. 2022, 35, 15–25. [Google Scholar] [CrossRef]
- Schmekel, L.; Portmann, A. Opisthobranchia des Mittelmeeres, Nudibranchia und Saccoglossa; Springer: Berlin/Heidelberg, Germany, 1982; 410p. [Google Scholar]
- Perrone, A. S-Opistobranchi (Aplysiomorpha, Pleurobrancomorpha, Sacoglossa Nudibranchia) del litorale Salentino (Mare Jonio). Thalass. Salentina 1986, 16, 19–42. [Google Scholar]
- Betti, F.; Bava, S.; Cattaneo-Vietti, R. Composition and seasonality of a heterobranch assemblage in a sublittoral, unconsolidated, wave-disturbed community in the Mediterranean Sea. J. Moll. Stud. 2017, 83, 325–332. [Google Scholar] [CrossRef]
- Furfaro, G.; Trainito, E.; Fantin, M.; D’Elia, M.; Madrenas, E.; Mariottini, P. Mediterranean Matters: Revision of the Family Onchidorididae (Mollusca, Nudibranchia) with the Description of a New Genus and a New Species. Diversity 2022, 15, 38. [Google Scholar] [CrossRef]
- Hallas, J.; Gosliner, T.M. Family matters: The first molecular phylogeny of the Onchidorididae Gray, 1827 (Mollusca, Gastropoda, Nudibranchia). Mol. Phylogenet. Evol. 2015, 88, 16–27. [Google Scholar] [CrossRef]
- Furfaro, G.; Trainito, E. A new species from the Mediterranean Sea and North-Eastern Atlantic Ocean: Knoutsodonta pictoni n. sp. (Gastropoda, Heterobranchia, Nudibranchia). Biodivers. J. 2017, 8, 725–738. [Google Scholar]
- Ortea, J.A. Nueva especie del género Atalodoris Iredale & O’Donoghue, 1923 (Mollusca: Nudibranchia) colectada en la Concha de Artedo, Asturias, dedicada al periodista deportivo José María García. Rev. Acad. Canar. Cienc. 2021, 33, 97–106. [Google Scholar]
- Müller, O.F. Zoologiae Danicae Prodromus, seu Animalium Daniae et Norvegiae Indigenarum Characters, Nomina, et Synonyma Imprimis Popularium; Typis Hallageriis, Havniae: Copenhagen, Denmark, 1776; Volume 32, pp. 1–282. [Google Scholar]
- Toms, J.A.; Pola, M.; Von der Heyden, S.; Gosliner, T.M. Disentangling species of the genus Limacia O.F. Müller, 1781, from southern Africa and Europe using integrative taxonomical methods, with the description of four new species. Mar. Biodivers. 2021, 51, 1. [Google Scholar] [CrossRef]
- Baba, K. The genera Gymnodoris and Nembrotha from Japan (Nudibranchia–Polyceridae). Publ. Seto Mar. Biol. Lab. 1960, 8, 71–75. [Google Scholar] [CrossRef]
- Pola, M.; Cervera, J.L.; Gosliner, T.M. Taxonomic revision and phylogenetic analysis of the genus Tambja Burn, 1962 (Mollusca, Nudibranchia, Polyceridae). Zool. Scr. 2006, 35, 491–530. [Google Scholar] [CrossRef]
- Domínguez, M.; Pola, M.; Ramón, M. A new species of Tambja (Mollusca, Gastropoda, Nudibranchia) from the Mediterranean Sea: Description of the first species of the genus from the Balearic Islands and Malta. Helgol. Mar. Res. 2015, 69, 205–212. [Google Scholar] [CrossRef]
- Willan, R.C.; Chang, Y.-W. Description of three new species of Tambja (Gastropoda, Nudibranchia, Polyceridae) from the western Pacific Ocean reveals morphological characters with taxonomic and phylogenetic significance for traditional Polyceridae and related «phaneorobranch» nudibranchs. Basteria 2017, 81, 1–23. [Google Scholar]
- Korshunova, T.; Picton, B.; Furfaro, G.; Mariottini, P.; Pontes, M.; Prkić, J.; Fletcher, K.; Malmberg, K.; Lundin, K.; Martynov, A. Multilevel fine-scale diversity challenges the ‘cryptic species’ concept. Sci. Rep. 2019, 9, 6732. [Google Scholar] [CrossRef] [PubMed]
- Tardy, J. Description d’une nouvelle espèce de Tritoniidae: Duvaucelia odhneri, récoltée sur la côte atlantique française. Bull. Inst. océanogr. 1963, 60, 1–10. [Google Scholar]
- Marcus, E.d.B.R. The Western Atlantic Tritoniidae. Bol. Zool. 1983, 6, 177–214. [Google Scholar] [CrossRef]
- Korshunova, T.; Martynov, A.V. Consolidated data on the phylogeny and evolution of the family Tritoniidae (Gastropoda: Nudibranchia) contribute to genera reassessment and clarify the taxonomic status of the neuroscience models Tritonia and Tochuina. PLoS ONE 2020, 15, e0242103. [Google Scholar] [CrossRef]
- De Vasconcelos Silva, F.; Pola, M.; Cervera, J.L. A stomach plate to divide them all: A phylogenetic reassessment of the family Tritoniidae (Nudibranchia: Cladobranchia). Zool. J. Linn. Soc. 2023, 199, 445–476. [Google Scholar] [CrossRef]
- Ghanimi, H.; Schrödl, M.; Goddard, J.H.R.; Ballesteros, M.; Gosliner, T.M.; Buske, Y.; Valdés, Á. Stargazing under the sea: Molecular and morphological data reveal a constellation of species in the Berthella stellata (Risso, 1826) species complex (Mollusca, Heterobranchia, Pleurobranchidae). Mar. Biodivers 2020, 50, 11. [Google Scholar] [CrossRef]
- Trainito, E.; Doneddu, M. Contribution to the knowledge of the molluscan fauna in the Marine Protected Area Tavolara-Punta Coda Cavallo: Ordo Nudibranchia. Boll. Malacol. 2015, 51, 54–70. [Google Scholar]
- Cervera, J.L.; Calado, G.; Gavaia, C.; Malaquías, M.A.E.; Templado, J.; Ballesteros, M.; García-Gómez, J.C.; Megina, C. An annotated and updated checklist of the opisthobranchs (Mollusca: Gastropoda) from Spain and Portugal (including islands and archipelagos). Bol. Inst. Esp. Oceanogr. 2004, 20, 5–111. [Google Scholar]
- Moro, L.; Ortea, J.A.; Bacallado, J.J. Nuevas citas y nuevos datos anatómicos de las babosas marinas (Mollusca: Heterobanchia) de las islas Canarias y su entorno. Rev. Acad. Canar. Cienc. 2016, XXVIII, 9–52. [Google Scholar]
- Martín-Hervás, M.R.; Carmona, L.; Jensen, K.R.; Krug, P.J.; Vitale, F.; Cervera, J.L. A global phylogeny of Elysia Risso, 1818 (Gastropoda: Heterobranchia): Molecular systematic insights focusing on European taxa and description of a new species. Zool. J. Linn. Soc. 2024, 200, 670–689. [Google Scholar] [CrossRef]
- Ortea, J.A.; Moro, L.; Bacallado, J.J. Nota sobre dos especies del género Elysia Risso, 1818 (Mollusca: Sacoglossa) en las islas de Cabo Verde. Rev. Acad. Canar. Cienc. 2017, 29, 21–30. [Google Scholar]
- Perrone, A.S. Una nuova specie di Elysiidae, Elysia hetta nov. sp. dal litorale salentino (Mediterraneo, Golfo di Taranto) (Opisthobranchia: Sacoglossa). Atti Soc. Ital. Sci. Nat. Mus. Civ. Stor. Nat. Milano 1990, 130, 249–252. [Google Scholar]
- Goddard, J.H.R.; Goddard, W.M.; Goddard, Z.E. Benthic Heterobranch Sea Slugs (Gastropoda; Heterobranchia) from Santa Barbara County, California. I. Review of the literature and Naples Point, 2002–2019. Proc. Calif. Acad. Sci. Fourth Ser. 2020, 66, 275–298. [Google Scholar]
- Goddard, J.H.R.; Goddard, Z.E.; Goddard, W.M. Benthic Heterobranch Sea Slugs (Gastropoda: Heterobranchia) from Santa Barbara County, California. II. Tar Pits Reef, Carpinteria, 2008–2020. Proc. Calif. Acad. Sci. Fourth Ser. 2021, 67, 1–20. [Google Scholar]
- Vitale, D.; Giacobbe, S.; Spinelli, A.; De Matteo, S.; Cervera, J.L. “Opisthobranch” (mollusks) inventory of the Faro Lake: A Sicilian biodiversity hot spot. Ital. J. Zool. 2016, 83, 524–530. [Google Scholar] [CrossRef]
- Lipej, L.; Fortič, A.; Trkov, D.; Mavrič, B.; Ivajnšič, D. Faunistic, ecological, and zoogeographical survey of heterobranch fauna in the Adriatic Sea: Experiences from Slovenia. Mediterr. Mar. Sci. 2025, 26, 3. [Google Scholar] [CrossRef]
- Linares, C.; Coma, R.; Diaz, D.; Zabala, M.; Hereu, B.; Dantart, L. Immediate and delayed effects of a mass mortality event on gorgonian population dynamics and benthic community structure in the NW Mediterranean Sea. Mar. Ecol. Prog. Ser. 2005, 305, 127–137. [Google Scholar] [CrossRef]
- Garrabou, J.; Coma, R.; Bensoussan, N.; Bally, M.; Chevaldonné, P.; Cigliano, M.; Diaz, D.; Harmelin, J.G.; Gambi, M.C.; Kersting, D.K.; et al. Mass mortality in Northwestern Mediterranean rocky benthic communities: Effects of the 2003 heat wave. Glob. Change Biol. 2009, 15, 1090–1103. [Google Scholar] [CrossRef]
- Linares, C.; Doak, D.F. Forecasting the combined effects of disparate disturbances on the persistence of long-lived gorgonians: A case study of Paramuricea clavata. Mar. Ecol. Prog. Ser. 2010, 402, 59–68. [Google Scholar] [CrossRef]
- Coma, R.; Pola, E.; Ribes, M.; Zabala, M. Long-term assessment of the patterns of mortality of a temperate octocoral in protected and unprotected areas: A contribution to conservation and management needs. Ecol. Appl. 2004, 14, 1466–1478. [Google Scholar] [CrossRef]
- Oehlmann, J.; Schulte-Oehlmann, U. Chapter 17—Molluscs as bioindicators. In Bioindicators & Biom. : Principles, Concepts and Applications; Markert, B.A., Breure, A.M., Zechmeister, H.G., Eds.; Elsevier Science Ltd.: Amsterdam, The Netherlands, 2003; Volume 6, pp. 577–635. [Google Scholar]
- Goddard, J.H.R.; Schaefer, M.C.; Hoover, C.; Valdés, A. Regional extinction of a conspicuous dorid nudibranch (Mollusca: Gastropoda) in California. Mar. Biol. 2013, 160, 1497–1510. [Google Scholar] [CrossRef]
- Chow, L.H.; Yu, V.P.F.; Kho, Z.Y.; See, G.C.L.; Wang, A.; Baker, D.M.; Tsang, L.M. An Updated Checklist of Sea Slugs (Gastropoda, Heterobranchia) from Hong Kong Supported by Citizen Science. Zool. Stud. 2022, 61, 52. [Google Scholar] [CrossRef]
- Mannino, A.M.; Balistreri, P. Citizen science: A successful tool for monitoring invasive alien species (IAS) in Marine Protected Areas. The case study of the Egadi Islands MPA (Tyrrhenian Sea, Italy). Biodiversity 2018, 19, 42–48. [Google Scholar] [CrossRef]
- iNaturalist Community. Available online: https://www.inaturalist.org/ (accessed on 5 July 2025).

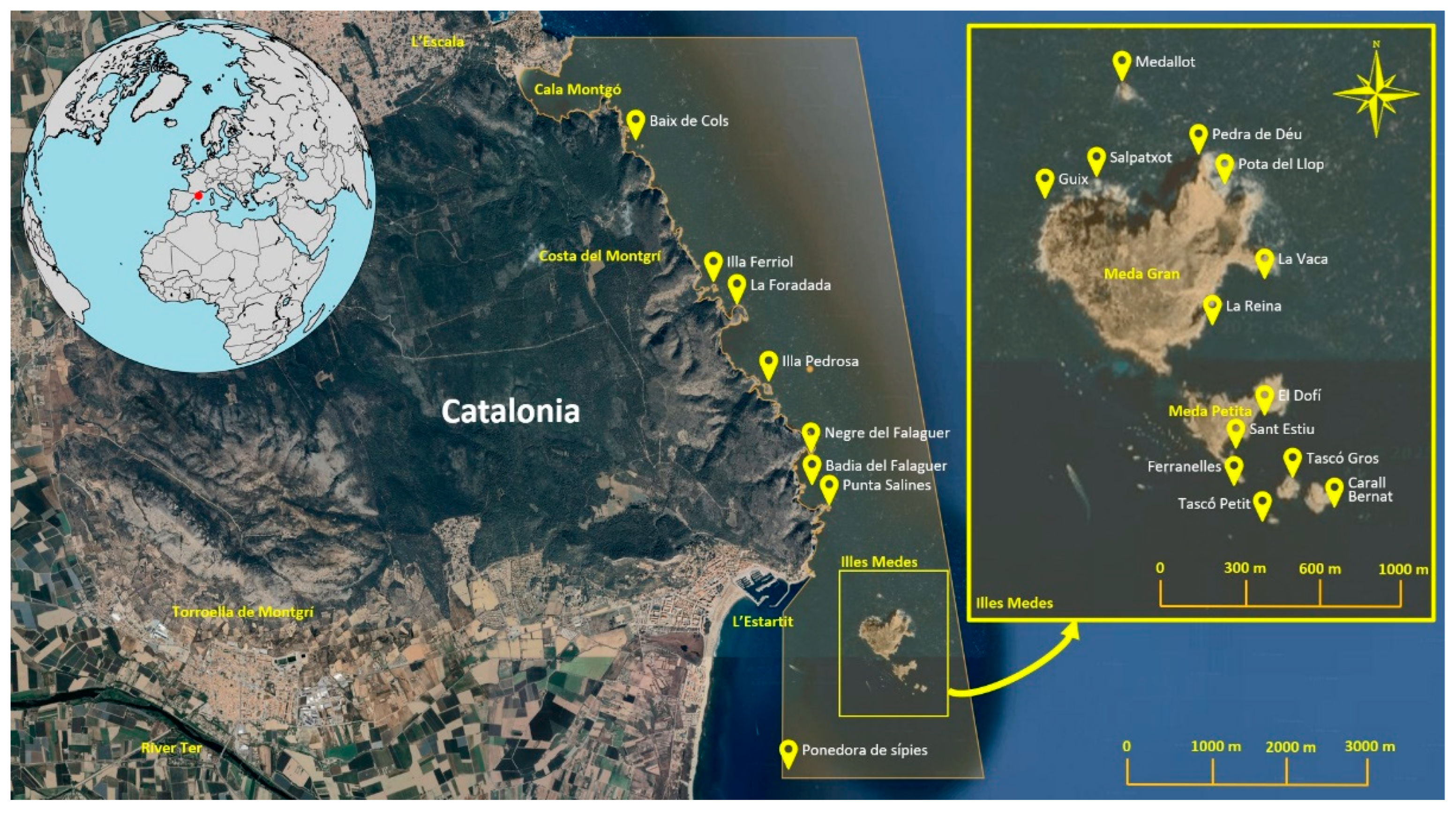




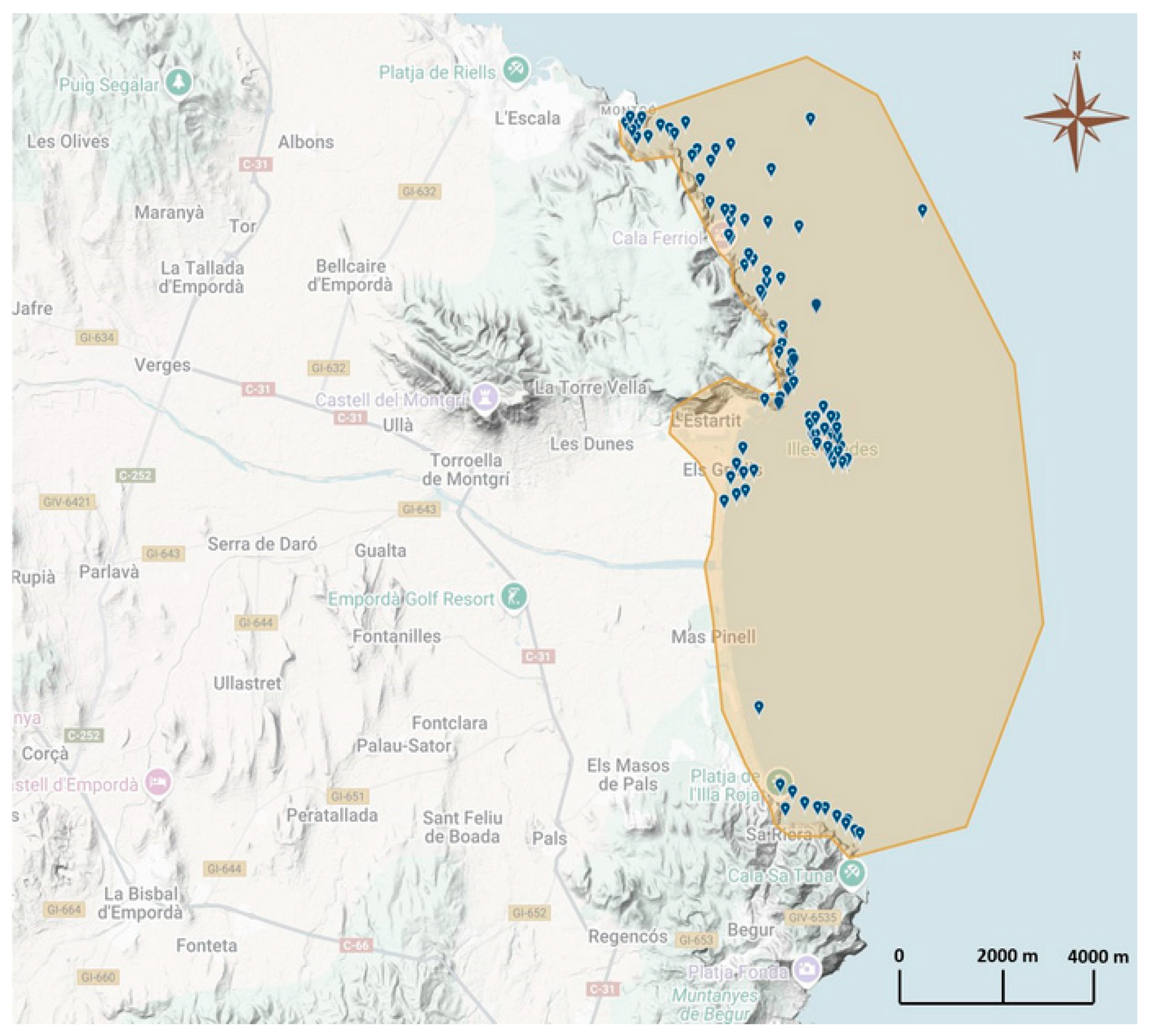
| Zone | Locality | Coordinates | Nº. Surveys | Depth Range |
|---|---|---|---|---|
| Montgrí | Baix de Cols | 42.100353 N, 3.186637 E | 1 | (2.5–18.5 m) |
| Illa Ferriol | 42.084415 N, 3.197003 E | 2 | (7.0–18.0 m) | |
| La Foradada | 42.081541 N, 3.201541 E | 1 | (5.0–14.0 m) | |
| Illa Pedrosa | 42.073283 N, 3.204867 E | 1 | (4.0–17.4 m) | |
| Badia de Falaguer | 42.067198 N, 3.210972 E | 6 | (2.7–26.2 m) | |
| Punta Salines | 42.060978 N, 3.213847 E | 3 | (3.4–25.6 m) | |
| Illes Medes | Medallot | 42.051491 N, 3.221987 E | 3 | (3.7–36.9 m) |
| Guix | 42.049182 N, 3.219273 E | 4 | (5.8–21.6 m) | |
| Salpatxot | 42.049533 N, 3.220824 E | 8 | (3.0–26.0 m) | |
| Pedra de Déu | 42.050033 N, 3.224174 E | 6 | (6.0–26.5 m) | |
| Pota del Llop | 42.049331 N, 3.225443 E | 2 | (4.5–30.8 m) | |
| La Vaca | 42.047388 N, 3.225768 E | 7 | (3.0–31.0 m) | |
| La Reina | 42.046387 N, 3.224656 E | 1 | (5.2–25.0 m) | |
| El Dofí | 42.043866 N, 3.226427 E | 10 | (4.0–32.8 m) | |
| Sant Estiu | 42.043197 N, 3.225954 E | 3 | (6.0–20.4 m) | |
| Ferranelles | 42.042231 N, 3.225505 E | 20 | (0.5–27.0 m) | |
| Tascó Gros | 42.042121 N, 3.226816 E | 5 | (1.8–30.0 m) | |
| Tascó Petit | 42.041124 N, 3.226538 E | 17 | (3.2–27.2 m) | |
| Carall Bernat | 42.041460 N, 3.228149 E | 6 | (5.2–39.3 m) | |
| Baix Ter | Ponedora de Sípies | 42.042833 N, 3.202150 E | 1 | (20.0–26.0 m) |
| Total Surveys | 107 |
| Year | Number of Individuals | Total Species | Diving Hours | Individuals/Hour |
|---|---|---|---|---|
| 2010 | 725 | 48 | 48 | 15 |
| 2011 | 1160 | 62 | 89 | 13 |
| 2012 | 582 | 49 | 40 | 14 |
| 2013 | 788 | 52 | 63 | 12 |
| 2014 | 461 | 36 | 32 | 14 |
| 2015 | 691 | 55 | 54 | 12 |
| 2016 | 1467 | 61 | 53 | 27 |
| 2017 | 1447 | 61 | 62 | 23 |
| 2019 | 968 | 45 | 53 | 18 |
| Total | 8289 | 98 | 494 | Mean: 16 |
| ID | Species | Absolute Abundance | % Relative Abundance |
|---|---|---|---|
| 1 | Cratena peregrina | 1379 | 16.98 |
| 2 | Edmundsella pedata | 687 | 8.46 |
| 3 | Flabellina affinis | 683 | 8.41 |
| 4 | Felimare tricolor | 606 | 7.46 |
| 5 | Peltodoris atromaculata | 544 | 6.70 |
| 6 | Diaphorodoris papillata | 520 | 6.40 |
| 7 | Calmella cavolini | 329 | 4.05 |
| 8 | Thuridilla hopei | 215 | 2.65 |
| 9 | Paraflabellina ischitana | 209 | 2.57 |
| 10 | Felimare orsinii | 190 | 2.34 |
| 11 | Tenellia caerulea | 185 | 2.28 |
| 12 | Diaphorodoris luteocincta | 184 | 2.27 |
| 13 | Placida dendritica | 175 | 2.16 |
| 14 | Candiella striata | 162 | 2.00 |
| 15 | Nemesignis banyulensis | 149 | 1.84 |
| 16 | Doto koenneckeri | 145 | 1.79 |
| 17 | Felimida krohni | 142 | 1.75 |
| 18 | Facelinopsis marioni | 134 | 1.65 |
| 19 | Felimare fontandraui | 116 | 1.43 |
| 20 | Candiella odhneri | 114 | 1.40 |
| 21 | Diaphorodoris alba | 113 | 1.39 |
| 22 | Elysia viridis | 103 | 1.27 |
| 23 | Elysia timida | 88 | 1.08 |
| 24 | Bosellia mimetica | 82 | 1.01 |
| 25 | Phyllidia flava | 63 | 0.78 |
| 26 | Berghia coerulescens | 61 | 0.75 |
| 27 | Crimora papillata | 57 | 0.70 |
| 28 | Caloria elegans | 51 | 0.63 |
| 29 | Luisella babai | 46 | 0.57 |
| 30 | Tenellia ocellata | 44 | 0.54 |
| 31 | Felimare villafranca | 42 | 0.52 |
| 32 | Limacia clavigera | 26 | 0.32 |
| 33 | Tylodina perversa | 25 | 0.31 |
| 34 | Polycera quadrilineata | 25 | 0.31 |
| 35 | Doto floridicola | 24 | 0.30 |
| 36 | Candiella manicata | 24 | 0.30 |
| 37 | Tenellia genovae | 20 | 0.25 |
| 38 | Platydoris argo | 19 | 0.23 |
| 39 | Marionia blainvillea | 17 | 0.21 |
| 40 | Elysia gordanae | 16 | 0.20 |
| 41 | Aplysia parvula | 16 | 0.20 |
| 42 | Berthellina edwardsii | 15 | 0.18 |
| 43 | Felimare picta | 15 | 0.18 |
| 44 | Eubranchus farrani | 15 | 0.18 |
| 45 | Favorinus branchialis | 15 | 0.18 |
| 46 | Facelina auriculata | 14 | 0.17 |
| 47 | Tenellia morrowae | 14 | 0.17 |
| 48 | Coryphella lineata | 13 | 0.16 |
| 49 | Paraflabellina gabinierei | 12 | 0.15 |
| 50 | Felimida luteorosea | 12 | 0.15 |
| 51 | Facelina rubrovittata | 11 | 0.14 |
| 52 | Trapania maculata | 11 | 0.14 |
| 53 | Felimida binza | 10 | 0.12 |
| 54 | Placida verticilata | 9 | 0.11 |
| 55 | Dendrodoris limbata | 8 | 0.10 |
| 56 | Trapania lineata | 7 | 0.09 |
| 57 | Felimida purpurea | 7 | 0.09 |
| 58 | Aplysia fasciata | 6 | 0.07 |
| 59 | Runcina adriatica | 6 | 0.07 |
| 60 | Tenellia miniostriata | 5 | 0.06 |
| 61 | Aglaja tricolorata | 5 | 0.06 |
| 62 | Aplysia punctata | 5 | 0.06 |
| 63 | Spurilla neapolitana | 5 | 0.06 |
| 64 | Calmella gaditana | 4 | 0.05 |
| 65 | Idaliadoris neapolitana | 4 | 0.05 |
| 66 | Candiella lineata | 4 | 0.05 |
| 67 | Doto sp. | 4 | 0.05 |
| 68 | Dendrodoris grandiflora | 3 | 0.04 |
| 69 | Piseinotecus soussi | 3 | 0.04 |
| 70 | Doto coronata | 3 | 0.04 |
| 71 | Placida cremoniana | 3 | 0.04 |
| 72 | Doto paulinae | 3 | 0.04 |
| 73 | Doto dunnei | 3 | 0.04 |
| 74 | Petalifera petalifera | 2 | 0.02 |
| 75 | Facelina annulicornis | 2 | 0.02 |
| 76 | Aeolidiella alderi | 2 | 0.02 |
| 77 | Umbraculum umbraculum | 2 | 0.02 |
| 78 | Atalodoris pusilla | 2 | 0.02 |
| 79 | Hancockia uncinata | 2 | 0.02 |
| 80 | Atalodoris sparsa | 2 | 0.02 |
| 81 | Geitodoris planata | 1 | 0.01 |
| 82 | Tenellia foliata | 1 | 0.01 |
| 83 | Tenellia albopunctata | 1 | 0.01 |
| 84 | Jorunna tomentosa | 1 | 0.01 |
| 85 | Antiopella cristata | 1 | 0.01 |
| 86 | Facelina vicina | 1 | 0.01 |
| 87 | Hermaea variopicta | 1 | 0.01 |
| 88 | Elysia flava | 1 | 0.01 |
| 89 | Berthella perforata | 1 | 0.01 |
| 90 | Caloria quatrefagesi | 1 | 0.01 |
| 91 | Felimare bilineata | 1 | 0.01 |
| 92 | Tayuva maculosa | 1 | 0.01 |
| 93 | Armina neapolitana | 1 | 0.01 |
| 94 | Berthella ocellata | 1 | 0.01 |
| 95 | Aplysia depilans | 1 | 0.01 |
| 96 | Okenia mediterranea | 1 | 0.01 |
| 97 | Doto eireana | 1 | 0.01 |
| 98 | Doriopsilla rarispinosa | 1 | 0.01 |
| Zone and Location | Observed Individuals | Sample Hours | Ratio Individuals/Hour |
|---|---|---|---|
| Montgrí coast | 1411 | 71 | Average 20 |
| Baix de Cols | 183 | 5 | 37 |
| Falaguer | 522 | 27 | 19 |
| Illa Ferriol | 52 | 12 | 4 |
| Illa Pedrosa | 64 | 6 | 11 |
| La Foradada | 228 | 5 | 46 |
| Punta Salines | 361 | 16 | 23 |
| Medes Islands | 6877 | 408 | Average 16 |
| Carall Bernat | 486 | 22 | 23 |
| Dofí Nord | 289 | 27 | 11 |
| Dofí Sud | 416 | 26 | 16 |
| El Guix | 281 | 18 | 16 |
| El Medallot | 239 | 12 | 23 |
| Ferranelles | 1483 | 93 | 16 |
| La Reina | 85 | 6 | 14 |
| La Vaca | 474 | 37 | 13 |
| Pedra de Déu | 385 | 17 | 16 |
| Pota del Llop | 133 | 9 | 15 |
| Racó del Sant Estiu | 137 | 17 | 8 |
| Salpatxot | 470 | 34 | 14 |
| Tascó Gros | 610 | 22 | 28 |
| Tascó Petit | 1389 | 68 | 21 |
| Baix Ter | 1 | 1 | Average 1 |
| Ponedora de Sípies | 1 | 1 | 1 |
| Seawater Temperature | T < 15 °C | T ≥ 15 °C T < 20 °C | T ≥ 20 °C |
|---|---|---|---|
| Shannon Diversity Index | 4.68 | 4.54 | 4.04 |
| Total Species Found | Species Never Found Before | Number of Species Known | |
|---|---|---|---|
| Ros et al. [20] | 34 | 34 | 34 |
| BIOCAT | 38 | 8 | 42 |
| GROC 2010 | 49 | 20 | 62 |
| GROC 2011 | 62 | 20 | 82 |
| VIMAR 2012 | 52 | 6 | 88 |
| VIMAR 2013 | 53 | 5 | 93 |
| VIMAR 2014 | 47 | 5 | 98 |
| VIMAR 2015 | 55 | 2 | 100 |
| VIMAR 2016 | 56 | 4 | 104 |
| VIMAR 2017 | 65 | 4 | 108 |
| VIMAR 2019 | 47 | 2 | 110 |
| MINKA 2013–2025 | 108 | 31 | 141 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pontes, M.; Madrenas, E.; Garcia-Tort, A.; Espada, O.; Ballesteros, M. Sea Slugs (Mollusca, Gastropoda, Heterobranchia) from the Medes Islands (Costa Brava, NE Spain): Biodiversity and Ecological Study over a Decade. Diversity 2025, 17, 606. https://doi.org/10.3390/d17090606
Pontes M, Madrenas E, Garcia-Tort A, Espada O, Ballesteros M. Sea Slugs (Mollusca, Gastropoda, Heterobranchia) from the Medes Islands (Costa Brava, NE Spain): Biodiversity and Ecological Study over a Decade. Diversity. 2025; 17(9):606. https://doi.org/10.3390/d17090606
Chicago/Turabian StylePontes, Miquel, Enric Madrenas, Arnau Garcia-Tort, Olga Espada, and Manuel Ballesteros. 2025. "Sea Slugs (Mollusca, Gastropoda, Heterobranchia) from the Medes Islands (Costa Brava, NE Spain): Biodiversity and Ecological Study over a Decade" Diversity 17, no. 9: 606. https://doi.org/10.3390/d17090606
APA StylePontes, M., Madrenas, E., Garcia-Tort, A., Espada, O., & Ballesteros, M. (2025). Sea Slugs (Mollusca, Gastropoda, Heterobranchia) from the Medes Islands (Costa Brava, NE Spain): Biodiversity and Ecological Study over a Decade. Diversity, 17(9), 606. https://doi.org/10.3390/d17090606







