1. Introduction
Understanding how biodiversity responds to environmental change constitutes a central question in community ecology and conservation biology [
1,
2]. Wetlands, as critically important ecosystems, provide vital services to society [
3,
4,
5], including carbon sequestration, nutrient cycling, flood and drought mitigation, water purification, and the maintenance of biodiversity [
6,
7,
8,
9,
10]. Characterized by high productivity, diverse habitats, and abundant water resources, wetlands sustain unique and rich biodiversity, which in turn constitute key elements in sustaining wetland ecosystem functioning [
11]. For example, some studies have shown that differences in ecosystem function among different life forms due to plant phylogeny or plant adaptations to the environment, and the complex interactions between species of different functional groups in natural ecosystems can have significant effects on the structure and function of ecosystems [
12,
13]. However, wetlands are also among the ecosystems most heavily impacted by human activities and are experiencing unprecedented degradation and loss [
14,
15]. China experienced a net loss of 60.9 × 10
3 km
2 in wetland area from 1980 to 2020 [
16]. In this context, constructed and newly formed wetlands have gained increasing attention as complementary or alternative systems to natural wetlands, given their ecological functions and conservation values [
17,
18,
19].
Coal mining subsidence areas are depressions formed by vertical ground subsidence resulting from stress redistribution in overlying strata after underground coal extraction [
20,
21]. These topographically low-lying areas readily accumulate precipitation and groundwater, forming waterlogged zones that gradually develop into the newly formed wetlands through hydrological accumulation and ecological succession, characterized by inundated environments, hydric soils, and wetland vegetation communities [
22,
23]. For example, several end-pit lakes in the Rocky Mountain foothills of west-central Alberta, Canada [
24], coal-mining ponds in southern Brazil [
25], and coal mining basins within the Czech Republic [
22] are all formed in this way. Notably, mining subsidence wetlands (MSWs) differ substantially from natural wetlands. First, the spatiotemporal heterogeneity of mining activities leads to significant variations in subsidence age, developmental stage, and environmental conditions [
26,
27]. Second, wetland stability varies with the time since subsidence: initial-stage wetlands exhibit hydrological fluctuations and unstable substrate structures, whereas late-stage wetlands demonstrate community stability comparable to natural wetlands, attributable to sediment compaction and organic matter accumulation that create stable microhabitats [
28,
29]. This unique formation mechanism likely results in biological communities with distinct characteristics from those in natural wetlands [
27]. Critically, coal mining subsidence acts as a key driver shaping macrozoobenthos assemblages in these newly formed wetlands. The pronounced spatiotemporal heterogeneity in environmental features and biological communities across different subsidence chronosequences provides a natural laboratory for studying community responses to environmental changes [
27].
Functional traits and functional diversity provide powerful analytical tools for elucidating how environmental changes drive community responses and associated ecosystem processes [
30,
31]. Ecological traits (e.g., feeding habitats, habit) regulate species’ adaptive capacity to habitat environmental changes [
32,
33], while life-history traits (e.g., voltinism, growth rate) reflect species-specific strategies shaped by long-term environmental adaptation, offering critical insights into habitat filtering and species interactions during community assembly [
34]. In wetland ecosystems, macrozoobenthos functional diversity often exhibits greater resistance to disturbances than taxonomic diversity [
35]. For instance, under eutrophication stress, functional redundancy enables pollution-tolerant species (e.g., chironomid larvae, tubificid worms) to maintain organic matter decomposition through similar filter-feeding strategies, thereby stabilizing ecosystem functioning during initial species loss [
36,
37,
38]. However, when environmental pressures directly target key functional traits (e.g., drought eliminating gill-breathing bivalves), functional diversity responds more rapidly, serving as a better early-warning indicator of ecosystem degradation than species diversity [
39]. This demonstrates that functional diversity simultaneously reflects ecosystem buffering capacity and sensitivity to specific stressors [
39,
40]. Despite these advantages, current assessments of community succession still predominantly rely on traditional taxonomic metrics, with limited application of trait-based approaches [
41,
42].
Macrozoobenthos constitute a vital component of freshwater wetland ecosystems. Their widespread distribution, ease of collection, and sensitivity to environmental changes make them excellent bioindicators for monitoring aquatic ecosystem health [
43,
44,
45,
46]. Occupying intermediate trophic levels, macrozoobenthos play crucial roles in energy flow and nutrient cycling, thereby maintaining critical wetland ecosystem functions [
47]. The functional trait combinations of wetland macrozoobenthos typically reflect adaptations to stagnant, low-oxygen environments [
48]. Body size demonstrates clear environmental filtering: small-bodied taxa (e.g., chironomid larvae) dominate hydrologically unstable temporary habitats, while large species prevail in stable water bodies [
49]. Respiratory types vary from cutaneous respiration (e.g., oligochaetes, chironomids) to specialized gills or spiracles that enhance oxygen uptake under hypoxic conditions. Macrozoobenthos feeding habits can affect secondary productivity and the nutrient cycle [
50]. These functional trait patterns not only reveal environmental filtering mechanisms but also provide novel quantitative metrics for wetland health assessment [
49,
51].
Wetland ecosystems in temperate climate zones exhibit pronounced seasonal dynamics [
52]. This study investigates the temporal patterns of macrozoobenthos functional diversity during ecosystem development in MSWs of temperate regions through multi-seasonal sampling. Considering the habitat heterogeneity and water parameter variations caused by different subsidence ages, this study proposes three hypotheses: (1) habitat complexity and stability increase with the time since subsidence, leading to higher taxonomic and functional diversity in older subsidence wetlands [
37,
53]; (2) functional diversity indices will show greater sensitivity to subsidence age than taxonomic diversity, as functional traits directly reflect species’ environmental adaptation strategies; (3) younger subsidence wetlands (initial stage) exhibit stronger seasonal fluctuations in species composition, taxonomic diversity, and functional diversity compared to mature wetlands, owing to their greater environmental instability. This study not only analyzes taxonomic diversity, but also explores the response patterns of functional diversity of macrozoobenthos in temperate coal mining subsidence wetlands along the subsidence age gradient through multi-season functional trait analysis. This not only provides new insights into the theory of wetland ecological succession but also makes significant contributions to both fundamental research and practical applications in wetland ecology.
2. Materials and Methods
2.1. Study Area and Sampling Design
Based on field surveys and remote sensing interpretation, we selected 13 MSWs (1–18 years post-subsidence) in Jining City (35°40′80″~35°52′78″ N, 116°80′82″~116°88′34″ E;
Figure 1;
Table A1) for macrozoobenthos surveys during spring (May), summer (July), autumn (September), and winter (December) of 2024. Located in the temperate climate zone of China, Jining experiences hot rainy summers and cold dry winters, with mean annual precipitation of 600 mm and average temperatures of 13.5–15 °C [
54]. These wetlands were all formed by coal mining-induced subsidence and were originally farmland prior to subsidence [
27]. They exhibited significant spatial heterogeneity in area (0.0287–1.42 km
2) and hydrological connectivity. Water sources primarily included precipitation and groundwater, with some wetlands connected to adjacent water systems via artificial channels. Dominant vegetation comprised emergent plants (
Phragmites australis,
Typha angustifolia), accompanied by floating species (
Trapa bispinosa,
Lemna minor). Surrounding landscapes were predominantly agricultural (corn–wheat rotation systems), with scattered villages and plantation forests [
27].
Due to spatiotemporal variations in underground coal mining activities, surface subsidence in Jining’s coal mining areas occurred during different stages. Based on field conditions and subsidence age, we classified these MSWs into three distinct successional stages:
Initial stage (IS, five wetlands with a subsidence history of 1–4 years): Characterized by small wetland areas and fragile ecosystems, these wetlands exhibit strong hydrological dependence on precipitation and may experience complete drying during prolonged droughts. The aquatic vegetation is dominated by Phragmites australis and Typha angustifolia, forming relatively simple plant communities.
Middle stage (MS, four wetlands with a subsidence history of 5–10 years): These wetlands represent a transitional stage of dynamic development, where continued subsidence leads to gradual expansion of wetland area and improved environmental stability. While Phragmites australis and Typha angustifolia remain dominant, the vegetation begins to show increased structural complexity compared to IS.
Late stage (LS, four wetlands with a subsidence history of more than 15 years post-subsidence): Subsidence processes are largely complete, resulting in well-developed “subsidence lakes” with extensive shoreline vegetation. Distinct zonation patterns emerge, dominated by bands of Phragmites australis community and Typha angustifolia community. Some sites develop submerged macrophytes including Stuckenia pectinata and Potamogeton crispus. However, these wetlands often display reduced community complexity, tending toward homogeneous, single-dominant plant communities.
2.2. Sample Collection and Processing
Three sampling sites were randomly established in each newly formed wetland. At each site, three replicate samples were collected using a 1/40 Peterson grab sampler (Wildlife Supply Company, Yulee, USA) (following a shallow-to-deep gradient) and subsequently pooled into a single composite sample. The collected samples were sieved through a 250-μm mesh sieve, transferred to sealed bags, and preserved in 75% ethanol. Macrozoobenthos identification was conducted following relevant taxonomic keys and literature [
55,
56,
57,
58] in the laboratory. Morphological identification was primarily performed using stereomicroscopes and compound microscopes. Specifically, aquatic oligochaetes and chironomid larvae were identified under a compound microscope, while hirudineans, gastropods, and most aquatic insects were examined using a SOPTOP SZN stereomicroscope (SOPTOP, Ningbo, China). Specimens were classified into the lowest feasible taxonomic level (usually at genus level). Furthermore, all benthic organisms from each sampling site were enumerated, with counts recorded for each taxonomic unit. For fragmented specimens, only heads were counted while tails were disregarded. For data analysis, densities were standardized to individuals per square meter (ind./m
2).
2.3. Measurement of Environmental Variables
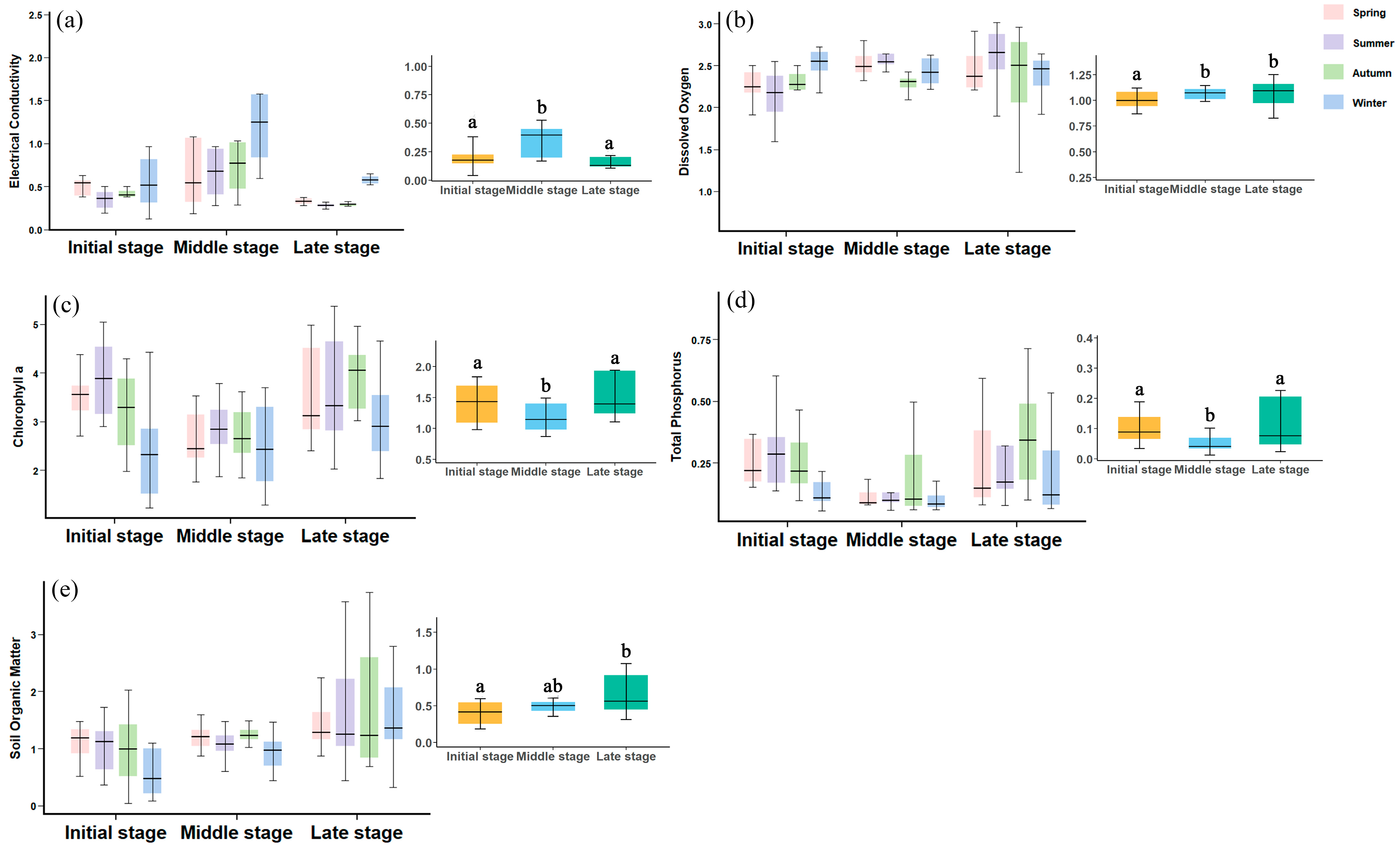
A total of seven environmental variables were measured, including physical and water chemistry variables. Physical habitat variables comprised soil organic matter (SOM), while water physicochemical variables included pH, dissolved oxygen (DO), electrical conductivity (Cond), total nitrogen (TN), total phosphorus (TP), and chlorophyll-
a (
Chl-a). In situ measurements of Cond, pH, DO were conducted using a DZB-718 portable multiparameter water quality analyzer (Shanghai Yidian Scientific Instrument Co., Ltd., Shanghai, China). Prior to biological sampling, 1 L water samples were collected for laboratory analysis of TN, TP, and
Chl-a, which were measured according to APHA (2012) [
59].
The formation time of newly formed wetlands was determined through a visual interpretation of satellite imagery (2006–2024, sourced from the Geospatial Data Cloud platform) based on established interpretation keys using ArcGIS 10.8 software. Additionally, the surface area of subsidence wetlands in 2024 was measured using the area calculation tool in ArcGIS.
2.4. Functional Traits
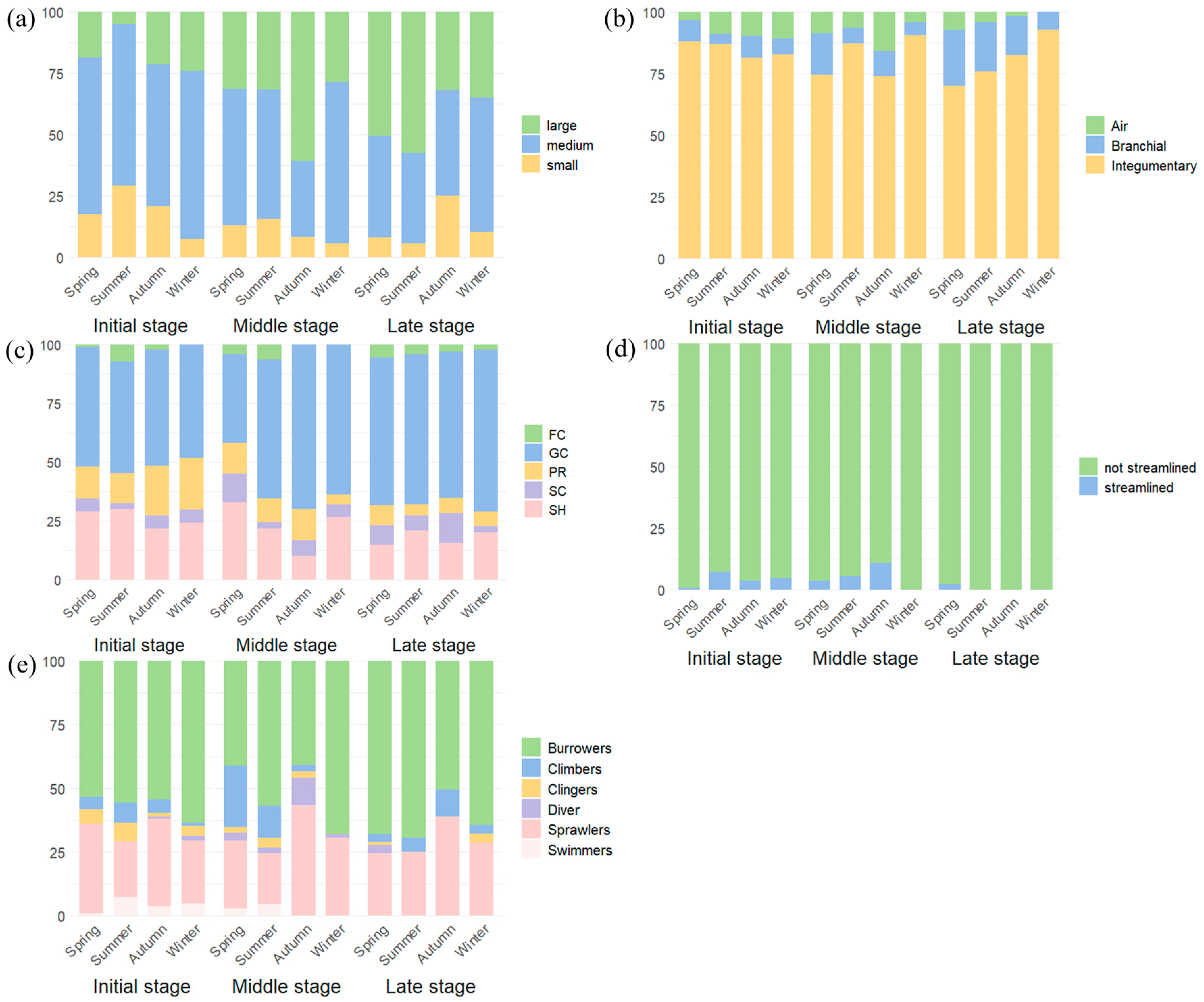
A total of five functional traits with 19 trait categories were selected, and were known to be sensitive to environmental changes: body size, respiration type, functional feeding groups (FFG), shape, and habit (
Table 1). Following the approach of Colzani et al. [
60], each trait category was assigned a binary score (0, 1, 2,…). Trait classification and scoring were primarily derived from published literature [
58,
61].
2.5. Data Analysis
2.5.1. Analysis of Taxonomic and Functional Composition
The functional trait composition of macrozoobenthos was characterized using the Community-Weighted Mean (CWM). CWM was calculated based on two data matrices: a community composition matrix (sites × species) and a functional trait matrix (species × traits), implemented via the “dbFD” function in the R package.
An analysis of similarities (ANOSIM) was applied to examine differences in taxonomic and functional composition among wetlands at three subsidence stages. The Global R statistic indicates the magnitude of dissimilarity, quantifying the ratio between among-group differences and within-group differences. Global R ≈ 1 signifies that among-group differences significantly exceed within-group differences, indicating a strong influence of the grouping factor on community structure. Conversely, Global R ≈ 0 suggests no significant distinction between among-group and within-group differences, implying negligible grouping effects. Permutation tests were used to assess whether among-group differences were significantly greater than within-group differences. p ≤ 0.05 indicates that the grouping factor significantly influenced community structure, while p > 0.05 suggests non-significant grouping effects. Prior to analysis, both community data and trait data (CWM) were log(x + 1)-transformed. These analyses were performed using the “anosim” function in the R (version 4.4.1) package vegan.
2.5.2. Analysis of Taxonomic and Functional Diversity
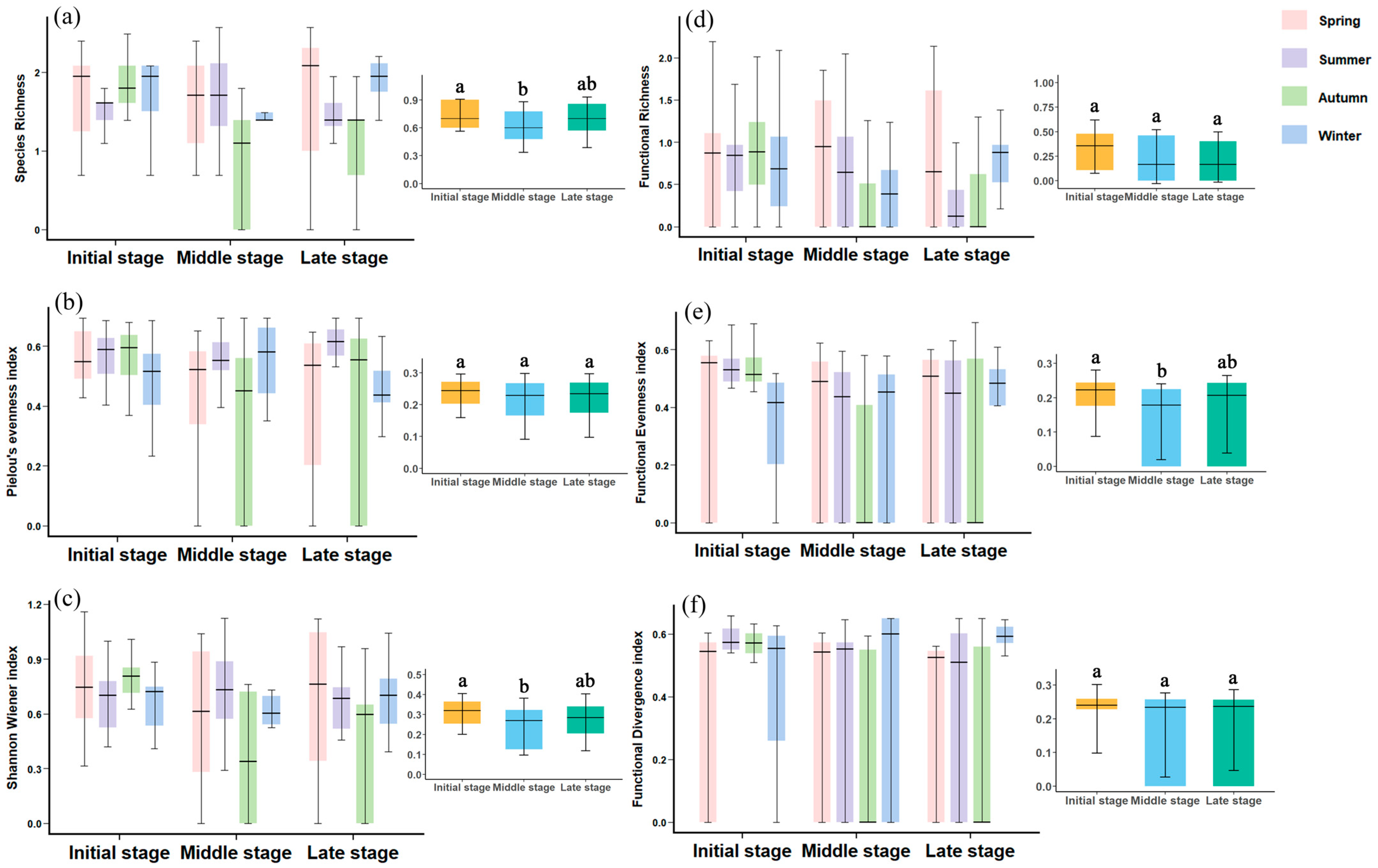
Taxonomic diversity of macrozoobenthos in MSWs was assessed using species richness, Pielou’s evenness index (J) [
62], Shannon–Wiener index (H′) [
63], and Simpson index. Species with relative abundance exceeding 10% were classified as dominant. These analyses were conducted in Primer6.
Functional diversity was characterized using functional richness (FRic), functional evenness index (FEve), and functional dispersion index (FDis). The CWM values represented the mean percentage contribution of each trait within a given trait category. These analyses were implemented via the “dbFD” function in the R package.
Following the framework for studying functional diversity and redundancy proposed by Ricotta et al. [
64,
65], we calculated the functional redundancy index. Quadratic Entropy Index (RaoQ) and Simpson index were used to measure functional diversity (DF) and taxonomic diversity (DS) within individual communities, respectively. The functional redundancy index (FRI) was computed as: FRI = 1 − DF/DS. RaoQ was implemented via the “dbFD” function in the R package and other analyses were conducted using the “gdm” package in the R package.
2.5.3. Statistical Analysis of Key Drivers
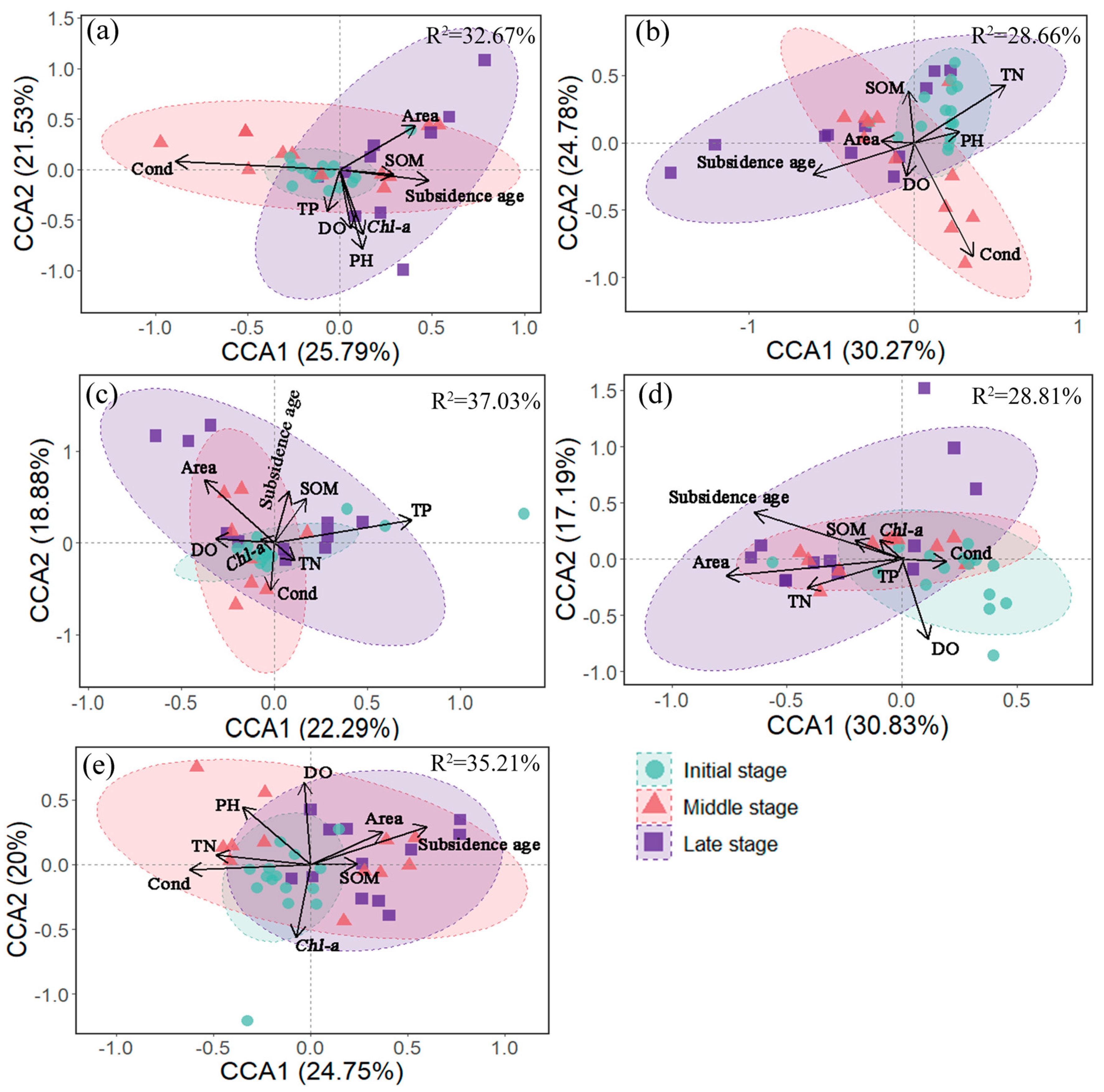
Constrained ordination analysis (CCA) was employed to examine the relationships between macrozoobenthos community taxonomic and functional composition, and environmental variables, respectively. First, to improve data normality and homoscedasticity, environmental variables (except pH), community data, and trait data were log(x + 1)-transformed. Highly correlated environmental variables (|r| > 0.70) were excluded. Correlation analyses were performed using the “Hmisc” package in the R package.
Subsequently, detrended correspondence analysis (DCA) was applied to macrozoobenthos community and trait data to determine the maximum axis length. If the maximum axis length exceeded 4, CCA was selected; if it was less than 3, redundancy analysis (RDA) was chosen; and if it fell between 3 and 4, either method was appropriate [
66]. DCA was conducted using the “decorana” function, while CCA and RDA were performed using the “cca” and “rda” functions, respectively, in the R package (version 4.4.1) vegan (
https://www.r-project.org/ accessed on 1 June 2024).
4. Discussion
Functional traits reflect species’ responses to environmental changes [
31,
67]. To understand how functional traits mediate species’ responses to these changes is crucial for predicting ecosystem dynamics and guiding conservation efforts. Therefore, this study compared macrozoobenthos community diversity across different subsidence stages of MSWs from functional traits and diversity perspectives, while investigating key influencing factors. Our findings revealed the following: (1) As the subsidence age increased, the species richness and FRic of the MSWs showed a declining trend, with biodiversity in LS being lower than IS. (2) Taxonomic diversity metrics were more sensitive to the subsidence age than functional diversity metrics. (3) Wetlands in IS exhibited significant environmental fluctuations, with seasonal variations in species composition and functional diversity being markedly greater than in wetlands in LS. This outcome validated the research hypothesis. The study elucidates the temporal evolution patterns of functional traits in macrozoobenthos in MSWs, providing a new evaluation dimension—functional diversity—for the ecological assessment of such wetlands. It holds significant theoretical value for understanding the succession mechanisms of wetland ecosystems under human-induced disturbances.
Our study reveals distinct patterns in the functional trait composition of macrozoobenthos across three subsidence stages of MSWs. The community was dominated by medium-sized organisms (9–16 mm), though the proportion of large-bodied taxa (>16 mm) increased significantly with subsidence age. Since large species typically exhibit higher individual biomass, this shift suggests enhanced secondary productivity and potential impacts on nutrient cycling through altered excretion patterns, indicating progressive ecosystem stabilization during succession [
49,
68,
69]. Respiratory adaptations showed a clear temporal trend: while integumentary respiration remained predominant overall, branchial respirers increased markedly in LS, likely reflecting improved water quality and dissolved oxygen conditions [
69]. Functional feeding groups varied significantly, with collector-gatherers peaking in MS, while shredders and predators were relatively more abundant in IS. Morphologically, non-streamlined body shapes dominated across all stages and burrowing was the prevalent life habit, probably due to stable sedimentary environments with high substrate heterogeneity [
50]. Notably, the dominant functional trait combination (medium size, integumentary respiration, collector-gatherer feeding, non-streamlined shape, and burrowing habit) consistently matched the biological characteristics of Chironomidae larvae, the taxonomically dominant group across all subsidence stages. This finding aligns with the conclusion of Zhang et al. [
27] regarding the persistent dominance of chironomids throughout wetlands’ development in MSWs.
Contrary to our first hypothesis, we found that both species richness and FRic of macrozoobenthos exhibited declining trends with wetland maturation (
Figure 3), despite increasing ecosystem stability. This counterintuitive pattern can be attributed to several ecological processes: Chironomid larvae, the dominant taxa across all subsidence stages, possess unique life history strategies that facilitate cross-ecosystem dispersal and rapid colonization of newly formed aquatic habitats [
70,
71,
72,
73]. Functional trait analysis revealed that IS were characterized by medium-sized organisms (63.9% of community composition), dominance of integumentary respiration (84.7%—an adaptation to fluctuating water conditions), and relatively high proportions of shredders (26.1%) and predators (17.3%), likely supported by abundant organic matter inputs from surrounding agricultural lands and elevated nitrogen/conductivity levels. The high FRic (
Figure 3) in IS probably resulted from both elevated habitat heterogeneity creating diverse microhabitats and resource availability supporting multiple feeding guilds. In contrast, LS exhibited environmental homogenization (stable conditions with low conductivity and high dissolved oxygen) and monodominant vegetation stands (primarily
Phragmites australis and
Typha angustifolia), leading to strong habitat filtering that was selected for only a few tolerant trait combinations (e.g., medium body size, burrowing habit). This successional trajectory toward reduced functional diversity despite ecosystem stabilization aligns with established theories of environmental filtering in anthropogenic wetlands [
27,
28,
29,
37], demonstrating how heterogeneous habitats can override expected biodiversity increases during ecosystem development.
Taxonomic diversity metrics exhibited greater sensitivity to subsidence age than functional diversity indices. CCA revealed that macrozoobenthos community composition was significantly influenced (
p < 0.01) by multiple environmental parameters (TN,
Chl-a, pH, Cond, DO, and subsidence age) (
Figure 6 and
Figure 7,
Table 3), with their synergistic effects driving pronounced compositional changes over time. In contrast, functional diversity showed delayed responses to environmental changes, likely due to buffering effects from functional redundancy within ecosystems (
Figure 5,
Table A5) [
38,
39,
74]. Although species richness declined across successional stages, dominant taxa (particularly
Chironomus and
Glyptotendipes) consistently maintained core functional traits including medium body size, integumentary respiration, collector-gatherer feeding and burrowing habits (
Figure 4,
Table A4). This functional conservatism suggests that key ecological processes may persist despite taxonomic turnover [
75], demonstrating how functional redundancy buffers against short-term impacts of species loss [
38]. Notably, LS showed increased proportions of large-bodied (>16 mm) and branchial-respiring taxa (
Table A4), whose traits may functionally compensate for diversity declines [
76,
77], thereby maintaining stable FDiv. These findings collectively suggest that while taxonomic diversity reflects direct environmental filtering effects in newly formed wetlands, the relative stability of functional diversity underscores ecosystem resilience through dual mechanisms of functional redundancy and trait compensation [
38,
39].
Our results confirm the hypothesis that IS exhibit greater seasonal fluctuations in taxonomic diversity and functional diversity compared to mature systems (
Figure 3). The initial high FRic in the newly formed wetlands was sustained by nutrient inputs from agricultural surroundings and diverse microhabitats supporting multiple functional groups. However, progressive heterogeneous habitats (e.g., Phragmites-dominated monocultures) and intensified environmental filtering (e.g., trait selection under stable low Cond/high DO conditions) gradually reduced functional space volume over time [
27,
28]. The elevated FEve observed in IS (
Figure 3e) suggests equitable trait distribution among pioneer species under resource-abundant conditions, while its subsequent decline reflects competitive exclusion-induced functional convergence, followed by slight recovery indicating potential niche differentiation. The consistently stable FDiv (
Figure 3f) likely stems from the persistence of core functional traits (medium body size, burrowing habit) in dominant taxa throughout succession, complemented by LS additions of large-bodied and branchial-respiring species [
75]. IS ecosystems, characterized by unstable hydrology, pronounced microtopographic variation, and low functional redundancy, showed amplified seasonal responses due to their transient microhabitats and limited buffering capacity. In contrast, mature wetlands demonstrated dampened seasonal variability through stabilized environmental conditions, enhanced community resistance, and developed biotic interactions [
11,
78]. This developmental trajectory from highly dynamic pioneer systems to stable mature ecosystems illustrates how anthropogenic wetlands gradually acquire natural-like stability over decadal timescales, with the attenuation of seasonal biodiversity fluctuations serving as a key indicator of ecosystem maturation in post-subsidence wetlands.