Abstract
Understanding the hierarchy of populations from the scale of metapopulations to mesopopulations and member local populations is fundamental to understanding the population dynamics of any species. Jellyfish by definition are planktonic and it would be assumed that connectivity would be high among local populations, and that populations would minimally vary in both ecological and genetic clade-level differences over broad spatial scales (i.e., hundreds to thousands of km). Although data exists on the connectivity of scyphozoan jellyfish, there are few data on cubozoans. Cubozoans are capable swimmers and have more complex and sophisticated visual abilities than scyphozoans. We predict, therefore, that cubozoans have the potential to have finer spatial scale differences in population structure than their relatives, the scyphozoans. Here we review the data available on the population structures of scyphozoans and what is known about cubozoans. The evidence from realized connectivity and estimates of potential connectivity for scyphozoans indicates the following. Some jellyfish taxa have a large metapopulation and very large stocks (>1000 s of km), while others have clade-level differences on the scale of tens of km. Data on distributions, genetics of medusa and polyps, statolith shape, elemental chemistry of statoliths and biophysical modelling of connectivity suggest that some of the ~50 species of cubozoans have populations of surprisingly small spatial scales and low levels of connectivity. Despite their classification as plankton, therefore, some scyphozoans and cubozoans have stocks of small spatial scales. Causal factors that influence the population structure in many taxa include the distribution of polyps, behavior of medusa, local geomorphology and hydrodynamics. Finally, the resolution of patterns of connectivity and population structures will be greatest when multiple methods are used.
1. Introduction
Jellyfish are ecologically important as predators [1], prey [2,3,4] and structures in the pelagic environment [5,6]. The abundance of jellyfish can vary greatly in space and time and increases in abundance can have detrimental effects on ecosystems and human livelihoods. For example, predation on small planktonic food and specific groups (e.g., fish larvae) by jellyfish can result in important changes in marine food chains. These changes can in turn result in phase shifts that fundamentally alter trophic pathways, where in some cases baitfish may be largely replaced by jellyfish [7]. Jellyfish blooms can also affect human infrastructure [8] such as aquaculture facilities, power plants that utilize seawater and nets used to catch boney fishes [9]. Jellyfish have also been introduced in ballast water, affecting native assemblages [10]. Further, jellyfish are venomous and when humans are stung the affects can range from nuisance-level irritations to severe responses and even death; consequently, the presence of jellyfish affects tourist industries at multiple latitudes globally [11]. In contrast, jellyfish are also an exploitable resource. They are targeted by fisheries [12], and annual reported catches are about 0.9 Mt [13]. The magnitude of species influences on ecosystem functions and human activities is inherently linked to their population dynamics, and this is particularly true for particular jellyfish species which have boom and bust abundance cycles. Accordingly, there are multiple reasons why knowledge is required on the sources, sinks, connectivity and related population structure of jellyfish.
There are about 200 morpho-species of “true jellyfish” (Class Scyphozoa [14]), with true (genetic) diversity likely being double [15]. In contrast only ~50 species of box jellyfish (Class Cubozoa) have been described [16]. Our knowledge of biology and population ecology by class, based on number of publications, is as asymmetrical as species richness [17]. Accordingly, many general paradigms on jellyfish are based on scyphozoans. For this reason, we have combined the knowledge available on population structures of both classes of jellyfish to assess their commonality and differences.
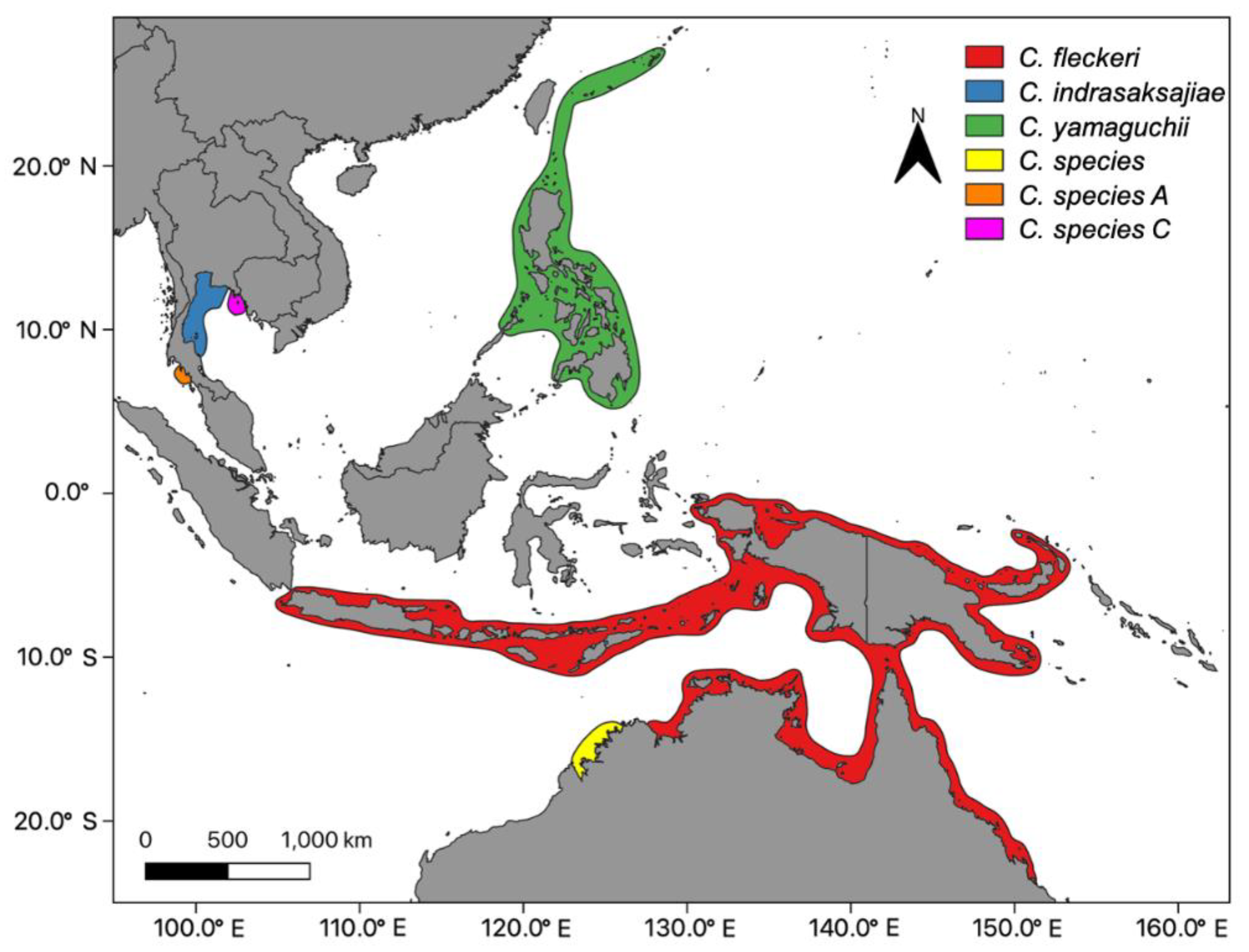
Population structures have generally been described according to three levels. Metapopulations, mesopopulations and local populations [18]. Metapopulations are made up of a number of mesopopulations, or in a fisheries context these are coined “stocks.” A metapopulation often corresponds with the biogeographic range of a species. For example, the biogeographic range of the three described species of Chironex are likely to be composed of multiple stocks (Figure 1). In some cases, there may be anti-tropical distributions of a species where metapopulations are found in both hemispheres, but are quite separate [19]. This scenario is likely for widespread taxa of jellyfish such as Carybdea rastoni and Copula sivickisi found in northern and southern hemispheres and multiple seas [17]. Separate metapopulations, or in some cases more than one, likely correspond to the Large Marine Ecosystems (LME) coined by Abboud et al. [20].
Figure 1.
The geographic range of all know species of Chironex. Chironex fleckeri Southcott, 1956; Chironex yamaguchii [21]; Chironex indrasaksajiae [22], and unnamed species in northern Australia [23] and Thailand [24]; Taxonomy [25]).
Population connectivity is the exchange of individuals among geographically separated population units [26]. Within metapopulations, a robust biological stock should have little to no connectivity with other stocks. Accordingly, successful immigration and emigration, involving any life history stage, should be very low or zero [27]. It is possible that an incipient stock is largely separate ecologically, but there is sufficient genetic exchange that they do not constitute separate clades. Where separation has been sufficient enough to result in significant clade-level genetic differences, there is the potential for incipient speciation. Within a stock, local populations can be identified that may be limited to a small geographic area such as a bay, estuary or island. Further, ecological interactions are likely among individuals from different local populations through the movement of adults or early life history stages, so genetic connectivity therefore would be assumed. It is also possible that local populations with little or zero connectivity could contribute to a mosaic of genetic diversity within a stock, and under the right circumstances could become a separate stock.
Plankton largely drifts or wanders by definition [28]. Jellyfish have historically been classified as plankton. Accordingly, it would be assumed that connectivity would be high among local jellyfish populations and that populations would vary little in both ecological and genetic clade-level differences over broad spatial scales (i.e., hundreds to thousands of km). However, findings on the behavior and swim speeds of jellyfish suggest it may be more appropriate to classify them as actively swimming nekton. Nekton can swim to either maximize dispersal or minimize it by remaining close to natal areas and these strategies can spatially broaden or restrict species population structures, respectively.
Scyphozoans and cubozoans have a number of attributes that assist in restricting the dispersal of medusa from localized areas. Both classes are active swimmers. Scyphozoans cover a large size range, from just a couple of centimeters (e.g., Linuche unguiculata, the thimble jellyfish) to the lion’s mane jellyfish (Cyanea capillata), which can grow to over 2 m bell diameter. Jellyfish in general are highly efficient swimmers [29] and small scyphozoans can reach surprising speeds. For example, Larson [30] recorded Linuche unguiculata which ranged in size from 0.5 to 2.2 cm bell diameter swimming at average speeds ranging from 3.5 to 7.2 cm s−1. The larger scyphozoan taxa are even more mobile. Stomolophus meleagris, which can grow to 25 cm bell diameter, have been recorded swimming at speeds of 15 cm s−1 [31].
Although cubozoans are generally small, they are generally highly mobile. The fifty known species of cubozoans range in size from those with a bell diameter of a few cm (e.g., Copula sivickisi) to those of about 20–25 cm (e.g., Chironex fleckeri). All of the species that have been observed swim well. For example, relatively small Copula sivickisi (size range: 0.4 to 1.1 cm Inter Pedalial Distance) have been recorded swimming at maximum speeds of 12 cm s−1 in swim trials [32], while the larger Chironex fleckeri (size range: 4 to 12 cm Inter Pedalial Distance) have been measured to swim at speeds of up to 16.6 cm s−1 in the field [33].
Scyphozoans and cubozoans have excellent sensory abilities. The sensory systems of both classes include ocelli, specialized structures to sense light; however, cubozoans also possess image-forming eyes similar in structure to the eyes of vertebrates and cephalopods [34]. Jellyfish can migrate vertically and have some ability to move and orientate horizontally (scyphozoans, Aurelia, Mastigias, Stomolophus, Pelagia [8,26,35,36]). More complex behaviors include responding to objects, conspecifics, currents, diel cycles, shadow and light, bioluminescent plankton, habitat type and small-scale geography (Table 1). Some cubozoans have even been observed to rest (C. rastoni, [37]) or attach to the substratum (Copula sivickisi [38]). Accordingly, predictions for population structures of jellyfish based on passive dispersal are likely to be highly inaccurate.

Table 1.
Behaviors involving interactions with environmental factors documented in medusae from different scyphozoan and cubozoan species. The methods used to determine the behaviors are indicated. Mating behavior is not included.
Further, the different life histories of scyphozoans and cubozoans could have great ramifications for species population structures. Jellyfish, including those in the classes Scyphozoa and Cubozoa, generally have bipartite lifecycles, the visible medusae which have been discussed produce free swimming larvae which settle and develop into sessile polyps. Scyphozoan polyps successively strobilate larval medusae called ephyrae, which are poor swimmers. In contrast, cubozoan polyps typically metamorphose into juvenile medusae which are nearly fully formed and have some swimming ability. The poor swimming ephyrae of scyphozoans, therefore, have greater potential to disperse from source locations compared to juvenile cubozoans. Notably, some scyphozoan jellyfishes lack a polyp phase and are holoplanktonic (e.g., Pelagia noctiluca and Periphylla spp.). Their entire lifecycle is completed in the plankton, where their planula larvae develop into ephyrae which develop into medusae. The lack of a polyp phase could also have a great effect on population structures. Indeed, the polypoid and podocyst phases of scyphozoans and cubozoans [4,17] provide an additional mechanism to reduce dispersal and related emigration from a population unit. Where taxa already have a restricted distribution, as medusa, the benthic stages are likely to contribute to that pattern. The potential, therefore, for complex and highly differentiated population structures is great, especially where the local geomorphology is convoluted and could facilitate retention, such as estuaries, bays and fjords.
Multiple methods have been used to determine levels of connectivity and to differentiate populations of marine organisms at different spatial scales, and some of the methods demonstrate the potential for connectivity while for others it is realized connectivity (sensu [48]), as follows. Realized connectivity can be demonstrated though population genetics [49], tagging [50] and intergenerational tags [51], while methods that demonstrate the potential for connectivity include: morphometrics of body shape and body parts (e.g., [52,53]), elemental chemistry [54] and biophysical modelling, where predictions are made on physical oceanography and mobility of different life history stages [55]. Where the information is spatially comprehensive in time and space, known patterns of distribution can also contribute to predictions on the potential for population connectivity [56]. Of course, the most compelling cases for the determination of stock structure comes from the use of multiple techniques and related corroborative data [56,57,58].
The objective of this study was to review the evidence for levels of connectivity and related differences in population structure for the Scyphozoa and Cubozoa. The specific aims of this review were to provide examples of: (1) metapopulations and spatially disjunct ecological patterns with evidence for allopatry; (2) finer scale stock and local population patterns, as informed by genetics data, statolith shape, elemental chemistry and biophysical modelling; and (3) the contribution of polyps to restricting distributions and dispersal. We conclude our synthesis by identifying multiple scyphozoan and cubozoan species that have stocks on scales of km to tens of km, which has strong inferences for the nature of speciation.
2. Evidence from Scyphozoan and Cubozoan Populations
2.1. Metapopulations and Spatially Disjunct Ecological Patterns
The biogeographic ranges of many jellyfish taxa have been determined from collections and in some cases from more detailed ecological studies. The data for some scyphozoans align with the traditional belief that the structure of zooplankton populations should be well mixed over broad spatial scales. For example, the scyphozoan Aurelia aurita is a cosmopolitan species that is found in the Pacific, Atlantic and the Red Sea [25]. Although mostly found in the northern hemisphere, it is also found in the southern hemisphere around New Zealand. For this species, therefore, it would be expected that the range is made up of multiple metapopulations, most likely corresponding to parts of ocean basins. Within these metapopulations there is evidence of small-scale potential stocks (Table 2). The cubozoan, Copula sivickizi, also has a very broad distribution, where it is found over most of the Indo-east Pacific, has been recorded in the Indian and Atlantic Oceans and even extends to temperate areas of Japan and New Zealand [25].

Table 2.
Examples of the spatial scales of populations of jellyfish by class. Methods used to detect differences among populations: museum collections and distribution patterns, genetics; elemental chemistry (LAICP-MS), ecological data on the timing of reproduction, recruitment and movements; statolith morphology and biophysical modelling. Spatial scale is the scale at which differences were detected according to the method. Range is the maximum linear distance between detections, calculated as the distance along a coastline where applicable. WoRMS [25] has been used as a major source for the biogeography (range) of species. Evidence based on * realized connectivity. # potential for connectivity.
Many taxa are not found in multiple oceans/seas, but their biogeographic ranges extend for over 1000 km (Table 2). For example, the four known taxa of Chironex are spatially discrete in tropical latitudes of the Indo-Pacific (Figure 1. Chironex fleckeri is only found along the east and northern coastlines of Australia, Papua New Guinea and large parts of Indonesia. Chironex yamaguchii also has a broad range from tropical islands of Japan to the Philippines. Although Chironex indrasaksajiae and an unnamed species from Western Australia appear to have very restricted distributions, this may in part be due to their recent discovery and description. Further, a study using PCR primers has detected another two incipient species in the waters of Thailand [24].
There are other examples of species where they are well known to have a very restricted distribution and therefore a small metapopulation. In an extreme case, incipient species of Mastigias papua have been identified in isolated marine lakes in Palau and the lakes are only separated by tens of km [62,72]. The chirodopid Chiropsella bronzie is recorded from the east coast of tropical Australia [17], but is only found at mainland locations between Mission Beach (17°57.088 E, 146°05.799 S) and Cooktown (15°2.035, 145°15.051; J. Seymour pers. com.). The carybdeid, Carybdea arborifera, is only recorded in Hawaii [17]. Although some taxa such as Aurelia aurita, Catostylus mosaicus, Chironex fleckeri and Copula sivickizi have broad distributions, there is an increasing body of evidence for small stocks and local populations that are likely to facilitate incipient speciation (Table 2).
For example, the rhizostome Catostylus mosaicus is found in tropical to temperate latitudes around all of Australia, and this potentially forms one metapopulation. This species is generally found in estuaries, drowned river valleys, bays and coastal lakes. Detailed studies on the ecology of this species on the East coast of Australia have demonstrated that the species is highly mobile and is capable of maintaining position in coastal waters. Furthermore, patterns of recruitment and reproduction vary among locations that are only separated by tens to hundreds of km [56].
An added complexity to understanding patterns of distribution is introductions from shipping, be that in ballast water or via fouling. Perhaps one of the best documented cases is for Phyllorhiza punctata, which was historically only found in the western hemisphere, but was introduced to North America through the Panama Canal, and the same species has also been detected outside of its normal range in the Mediterranean [73]. Although introductions are problematic, rapid progress in the sophistication of techniques in genetics will allow more rapid determination of source populations.
2.2. Population Genetics
Population genetics have been examined for scyphozoans, and to a lesser extent, cubozoans. This has allowed a fine-grain understanding of the presence of stocks within metapopulations. There is strong evidence for scyphozoans that at least some taxa have identifiable stocks at spatial scales of tens of km to hundreds of km (Table 2). However, even within the same genus, there are contrasting patterns. For example, Ben Faleh et al. [59] concluded that there is high gene flow of Aurelia in the Mediterranean at scales of over 100 km. In contrast, there is evidence from China [60] that medium-scale genetic structuring of Aurelia is found at scales of hundreds of km.
The data on genetics for some scyphozoans align with the traditional belief that the structure of zooplankton populations should be well mixed over broad spatial scales. For example, the holoplanktonic semaeostome, Pelagia, has a life history with no scyphistoma stage, unlike many other scyphozoan taxa. Pelagia noctiluca was found to not have genetically or geographically distinct populations within the Mediterranean and East Atlantic despite two clades being identified [65]. Miller et al. [64] also reported little variation among locations for P. noctiluca off Southern Africa. However, genetic differences were found to exist between populations in the northern and southern Atlantic, where at these spatial scales (thousands of km) their status is likely to be at the level of metapopulations.
Rhizostoma octopus, which commonly occurs in large blooms in the north-eastern Atlantic Ocean, particularly within the Irish Sea, has “stocks.” Multiple studies have reported the species as having multiple distinct “stocks” at spatial scales of hundreds of km [62,63]. Lee et al. [62] reported three genetically distinct populations through examination of the mitochondrial gene cytochrome oxidase subunit 1 (CO1) and the nuclear calmodulin gene (CaM). The mitochondrial gene highlighted greater variation than that of the nuclear gene due to a faster mutation rate [74], however both underlined the genetic variation between locations. Examination into the potential processes behind the identified genetic variation revealed physical oceanographic processes to be key. Locations with genetically distinct individuals were found to be relatively isolated by currents, hence explaining the genetic difference and supporting the separate “stocks,” while those locations with similarities were physically connected and so may allow migration/population connectivity between said locations. A comparable study by Glynn et al. [63] reported similar findings, and both studies concluded that this jellyfish has genetically distinct, self-sustaining populations (stocks) at scales of as little as a few 100 kms.
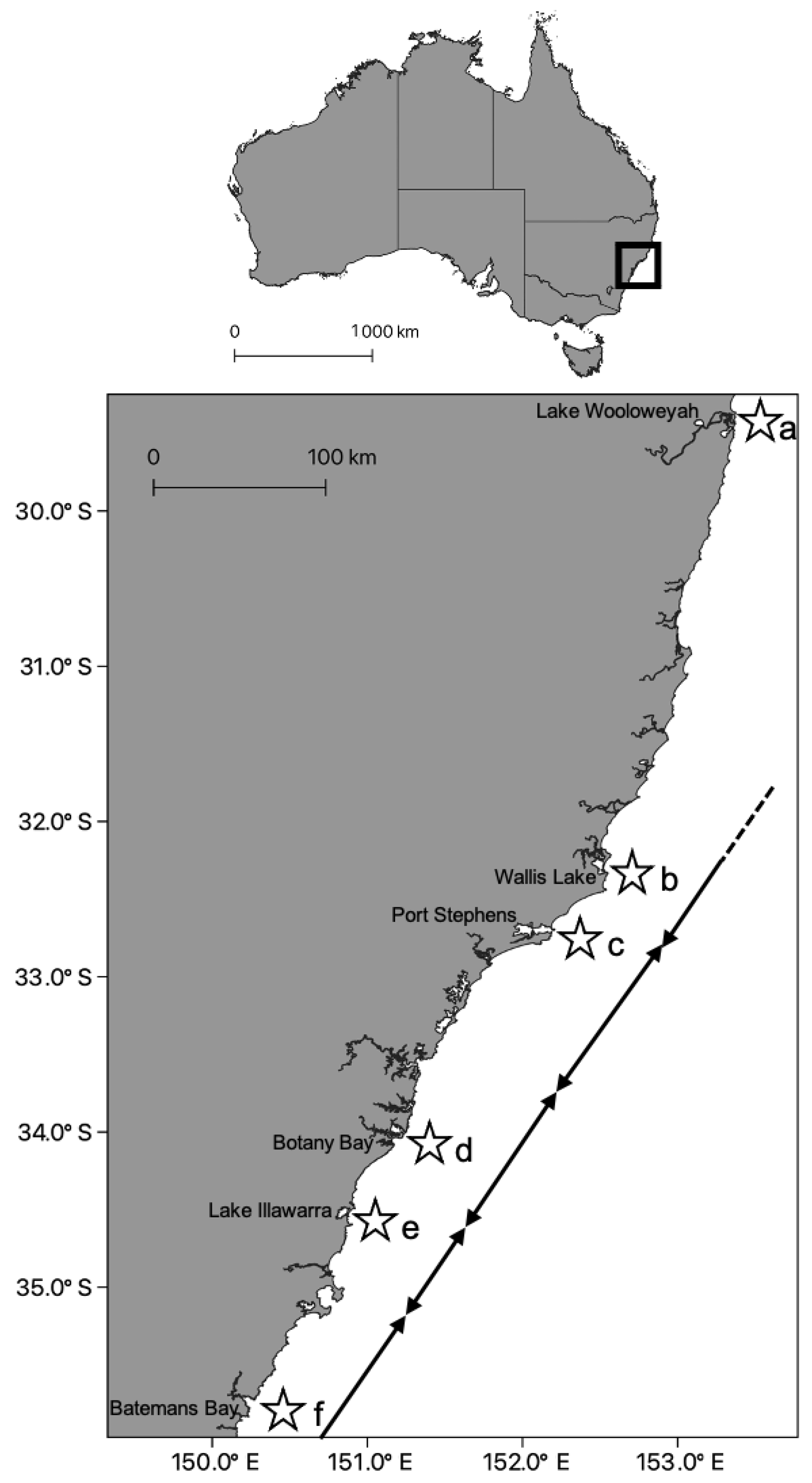
Another rhizostome that has been found to contain a hierarchical population structure is Catostylus mosaicus. This jellyfish is characterized by local populations within a variety of coastal environments including estuaries, bays and coastal lakes. In line with ecological and behavioural evidence (Table 2), the species was found to have molecular variation at small spatial scales, ranging from tens of kilometres [57]. Dawson [57,58] examined the CO1 gene and internal transcribed spacer 1 (ITS1) of specimens collected along the east Australian coast and found there to be two distinct clades at spatial scales of hundreds of km. Upon further examination of these clades, Dawson found there was significant genetic variation within and among collection sites within each clade, suggesting a finer-scale phylogeographic population structure. This genetic variation concurred with ecological differences reported by Pitt and Kingsford [56], and thus populations within closely spaced individual estuaries and lakes should be considered as distinct, self-sustaining populations (Figure 2). These studies highlight the growing evidence of some scyphozoan taxa having distinct population units that inhabit relatively small, often geographically constrained areas.
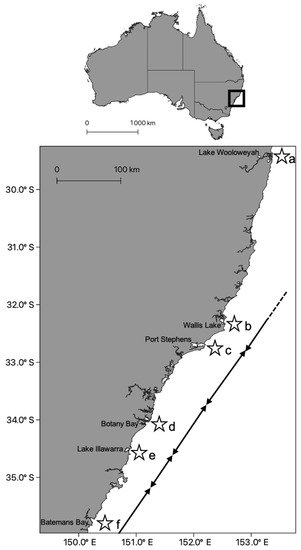
Figure 2.
Incipient speciation of Catostylus mosaicus (Scyphozoa, Rhizostomeae, Catostylidae) [57]. Populations of C. mosaicus were restricted to bays, coastal lakes and drowned river valley systems along the East coast of Australia in the state of New South Wales (Upper). (Lower) Ecological differences were found among sites (a–f) by Pitt and Kingsford [56]; these findings aligned with a high degree of genetic differentiation among sites [57]. Where black arrows meet demarcates high genetic variation between sites. The dashed arrow indicates the direction of Dawson’s next site in Southern Queensland, which differed from site a.
Abboud et al. [20] focused on macromedusae of the hydrozoa and scyphozoa and asked two questions for what they considered Large Marine Ecosystems (LMEs); probably the equivalent of what we have termed metapopulations. (1) Do congeneric individuals found within the same LME constitute individual species? (2) Do congeneric individuals from different LMEs constitute different species? Overall, the scale of mismatch among LMEs and genetic structure was >70%. LMEs did not match species boundaries for ~24% of comparisons. This may not be surprising given the spatial population structures of some taxa. However, over 19% of within LME comparisons detected cryptic species and 67% showed significant intraspecific phylogeographic structure.
Despite the obvious need, few studies have examined the genetics of cubozoan taxa. Of the references we could find, two were concerned with the development of microsatellites for C. fleckeri [75] and Carukia barnesi [76] to facilitate the undertaking of studies on population genetics. The phylogeny of cubozoan species has also been investigated [69,77]. The development of primers and probes allowed for the application of eDNA to detect cubozoans [78]. Further, genetic sequencing used to develop species-specific PCR primers for Chironex indrasaksajiae revealed two incipient species in Thailand (Figure 1). Although there is a strong suggestion that these taxa may have small metapopulations, each species has only been detected at one or two sites [24]. This review highlights the need for population genetic studies to be undertaken on cubozoan taxa, as this would develop a more in-depth understanding of the ecology of these cryptic and elusive taxa, thereby assisting in the management of these potentially deadly organisms [11].
We predict that clade-level differences in the population structures of most cubozoans will be greater than that demonstrated for scyphozoans as they show greater complexity in behavior and they have greater sensory capabilities [17]. Furthermore, there is strong evidence of differences among stocks at small spatial scales (tens to hundreds of km) from biophysical modelling, elemental chemistry and statolith morphometrics. However, there will be exceptions. For example, Lawley [69] suggested that populations of Alatina alata in the Pacific are not genetically distinct and must have connectivity over thousands of km, or the less favoured argument of human vector-based introductions. It is also worthy to note that Alatina alata is generally found in oceanic waters rather than shallow coastal waters.
Contrasting genetic patterns within and among taxa are likely due to biological and behavioral attributes such as swimming ability and orientation, as well as coastal versus oceanic distributions and local differences in geography and physical oceanography. There are also potential methodological issues with the genetics methodology used. Techniques used to examine genetic variation within and between populations have varying sensitivities/resolutions. For example, with Aurelia aurita, allozyme electrophoresis was utilized by Ben Faleh et al. [59] to investigate the genetic variation and population structure of the species. However, it is known that the technique has a low resolution as it only examines protein coding regions of DNA [79]. In contrast, techniques such as microsatellites and mitochondrial markers (utilized by Dong et al. [60]) have a higher sensitivity to reveal genetic variation, hence potentially resulting in the contrast of reported findings for Aurelia. Modern techniques will allow greater resolution of hierarchies of population structures and cryptic species [80].
2.3. Morphometrics and Elemental Chemistry
Morphometrics of both soft tissue and carbonate statoliths have been used to differentiate populations and potential clades and to identify the likely connectivity. Differences in morphometrics and more recently elemental chemistry of the statoliths have been found among population units of jellyfish at scales that probably correspond to the separation of metapopulations (thousands of km) to populations at stock or local population levels separated by hundreds of km or less. For example, in addition to evidence from genetics, morphological differences in clades of the rhizostome Catostylus mosaicus have been found between locations separated by hundreds of km on the East coast of Australia. The clades differed in color and the dimensions of their papillae, oral disk and bell depth. The differences were sufficiently great to be considered incipient species [57,58].
Bolton and Graham [81] tested a hypothesis on the source of Phyllorhiza punctata that had invaded to the Gulf of Mexico by describing the morphology of specimens. They postulated that the invasion of the Gulf of Mexico by P. punctata represented a distribution shift from an invasive hub in the Caribbean. Further, that this could be detected through measurements of variation in morphological metrics that included the following: morphometrics of the bell, color and characteristics of different sections of anatomy, the shape of spots, and the presence of bumps and symbiotic zooxanthellae. Their model was that populations in the Gulf of Mexico and Caribbean would be more morphological similar than specimens from more distant locations such as Australia and the East coast of the US. The model was rejected, as it turned out that populations in the Gulf of Mexico and Caribbean were morphologically dissimilar, implying separate invasions from distant sources such as Australia and the east Coast of North America. The sources, therefore, were separated by thousands of km and likely represent separate metapopulations.
Elemental fingerprints of hard structures have been used to elucidate spatial patterns of fish stocks [82] and other taxa; recently the technique was applied to cubozoans. Mooney and Kingsford [83] discovered that the morphometrics of the statoliths of cubozoans (hard structures made of a sulphate called “basanite” [84] that are components of medusae sensory systems) could be used to differentiate taxa. Furthermore, they also detected within-species variation. Discrimination among populations separated by hundreds of km was detected for Chironex fleckeri and Copula sivickisi. At some locations, variation in shape was found at spatial scales of km to tens of km. For example, the elemental chemistry of C. fleckeri was different at Magnetic Island when compared to the mainland which was only ~20 km away [71].
2.4. Biophysical Modelling
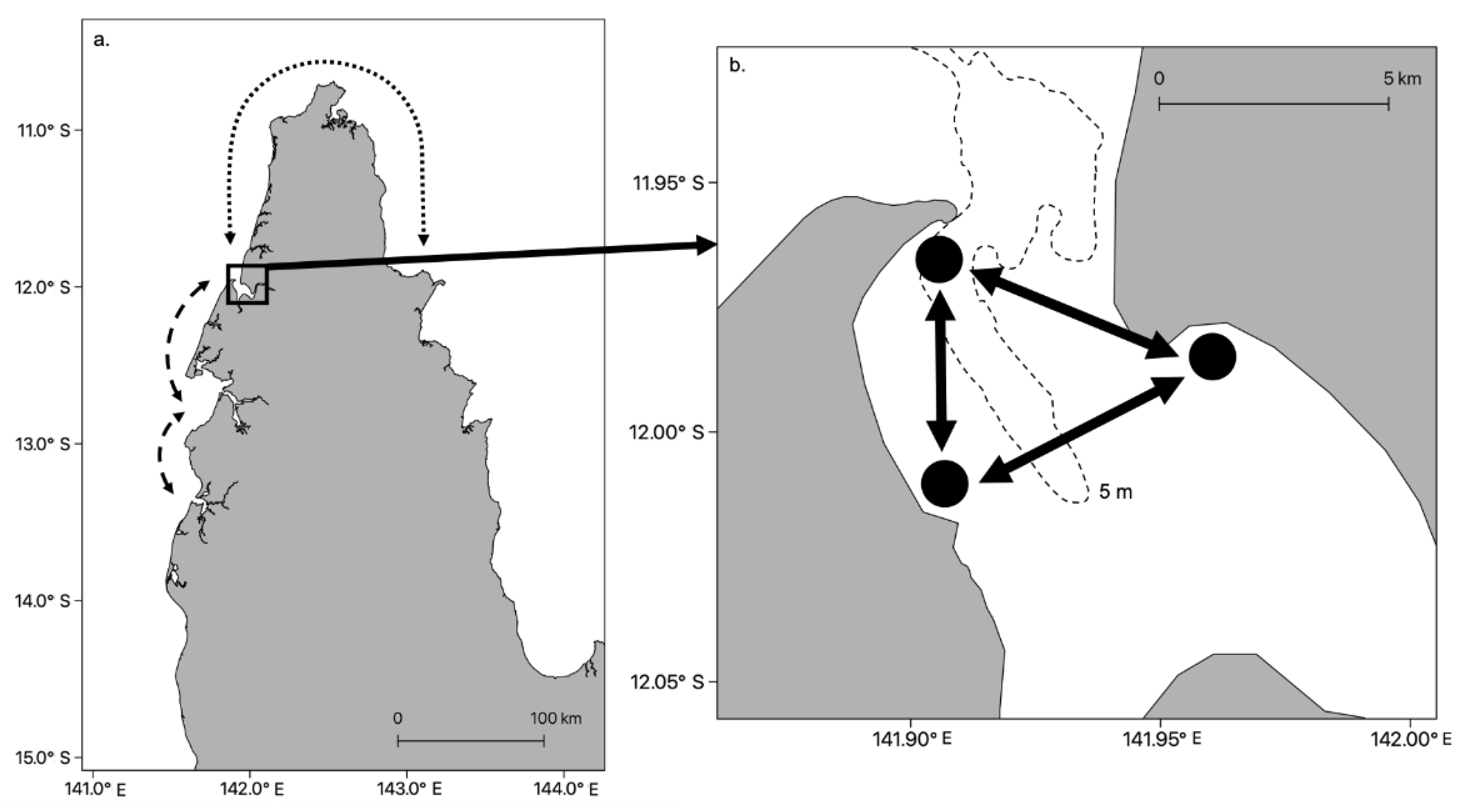
Advances in our knowledge of jellyfish behavior and improvements in the sophistication of oceanographic models has allowed biophysical models of jellyfish dispersion to be developed. The models can be used to estimate dispersal from polyp beds [85,86], potential connectivity between local populations and make predictions on likely stock boundaries [32,33]. For example, in a biophysical modelling study, Fossette et al. [87] programmed medusae of the scyphozoan Rhizostoma octopus to swim counter-current, and demonstrated that this behavior was integral to the maintenance of local blooms covering tens of km over temporal scales of several months. Schlaefer et al. [33] studied the behavior of the large cubozoan Chironex fleckeri in a semi-enclosed Bay in northern Australia (Port Musgrave, Figure 3). An oceanographic model that did not include jellyfish behavior indicated there was a high probability that jellyfish could remain inside the bay even if they behaved as passive particles. When active swimming, obstacle avoidance and directional behavior were included in the model, it demonstrated that not only was emigration out of the bay and to other bays unlikely, but that local populations within the bay were surprisingly robust (Figure 3).
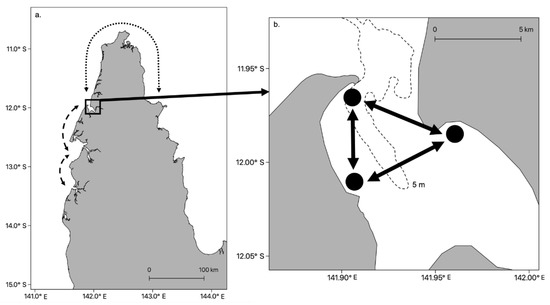
Figure 3.
Level of connectivity among incipient stocks of Chironex fleckeri [33]. (a) Cape York in northern Australia showing low-to-zero connectivity between bays and estuaries at scales of tens of km on the west coast (dashed line) and a potential rare event to the east coast (dotted line). The latter was only possible less than 5–7 K years before present as land stretched from Cape York to Papua New Guinea during the last ice age [88]. (b) Local populations of C. fleckeri in Port Musgrave. Arrows indicate potential connectivity among local populations based on the current, but behavioral studies suggest this would be low. The dashed line indicates the 5 m isopleth.
Copula sivickisi is a small cubozoan that frequents tropical reefs [41]. Schaefer et al. [32] focused on this species at the inner shelf location, Magnetic Island, on the Great Barrier Reef. The species is an active swimmer and is unique in that it can attach to the substratum, especially during the day. It prefers shallow algal beds of Sargassum and the abundance of medusae is highest in <5 m of water. Biophysical modelling determined that a high percentage of medusae were likely to stay within 2 km of the bay, in which they were released as virtual medusae. It was concluded that copulating medusae likely self-seed local populations (hundreds of meters to km wide) and that connectivity with adjacent local populations was surprisingly low. Further, the chances of emigration from Magnetic Island (45 km in circumference) to other locations with suitable habitats was low and the island, therefore, likely corresponds to a stock unit.
2.5. The Contribution of Polyps to Restricting Distributions
Polyps are a key component of the life history of scyphozoan and cubozoan jellyfish and are the source of medusae [4,17]. We hypothesize, therefore, that the polypoid stages are important for a greater understanding of the point sources of dispersal, connectivity and the likelihood of retention in population. Given the abilities of polyps to prevail for long periods of time, generally longer than the medusoid phase, and their abilities to reproduce asexually, polyp beds can be a spatially robust source of medusae. Some field studies have examined the role of polyps in determining the abundance of medusa and, therefore, their role in the population dynamics of scyphozoan populations (e.g., [89]). Distributional studies of polyps have also provided some evidence for insular populations, while others have not. For example, Marques et al. [90] mapped the distribution and habitat preferences of Aurelia sp. polyps in the mostly enclosed Thau lagoon, north-western Mediterranean Sea (France). The polyps were primarily found on artificial structures which “raises the possibility of the potential isolation of this population from the other populations of the Mediterranean Sea.” An estuarine-bound population of Cyanea in the Niantic River, USA, had a strong relationship between the distribution of medusa and the planula and polypoid phases [91]. The estuary was only a few km long and the polypoid phases were most abundant at the upper reaches of the estuary. Similar to the Thau Lagoon example, the estuary was largely enclosed. Toyokawa et al. [92] carried out extensive surveys for polyps and also collected some data on the distribution of ephyrae in Mikawa and Ise Bays. The bays are both tens of km wide and/or deep, but they share a common boarder. They found that polyps were most abundant on artificial structures deep in the bays and that the two bays shared a population of medusae sourced from polyps in the two bays.
Recent findings on the genetics of Aurelia polyps have provided strong evidence for populations of small spatial scale. van Walraven et al. [93] sampled Aurelia polyps in Southern North Sea and Gullmar Fjord (Sweden) and compared haplotypes using 18S mRNA and COI mDNA. Population differentiation in polyps was detected between the Dogger Bank in the English Channel and nearby coastal locations (marinas, wrecks and settlement plates separated by tens of km), indicating extremely low connectivity. However, no differences were found among coastal locations.
The relationship between cubozoan polyps and the abundance of medusae has scarcely been addressed. Hartwick [94] did locate a wild population of C. fleckeri polyps and noted that young medusae occurred nearby; however, no data on the relationship between polyp and medusae abundance were collected. The polyps of most jellyfish are difficult to find, but studies on connectivity may be refined through the detection of polyp beds using eDNA, as has been done for the cubozoan Copula sivickisi [78].
3. Conclusions
It is clear that despite the classification of scyphozoan and cubozoan jellyfish as plankton, many of them are highly mobile and demonstrate orientation behavior that can result in restricted distributions. Moreover, the cubozoans in particular have good eyesight that can further assist in maintaining their position within the boundaries of a population. The biogeographic range of holoplanktonic and oceanic taxa can extend over thousands of km and this, to some extent, has reinforced the view that jellyfish are plankton largely at the mercy of currents. However, evidence from distributional data and more recently information from population genetics, morphometrics/elemental chemistry and biophysical modelling has demonstrated that some taxa have a complex hierarchy of population units, from metapopulations to surprisingly small stocks and local populations, where the latter in some cases may turn out to be stocks. Discrete populations at scale of tens of km or less are likely to be self-sustaining, where causal factors include behaviorally restricted distributions of medusae in space, and in some cases assistance from local geomorphology and currents. Further, sexual reproduction and the subsequent release of planulae by scyphozoans [4] and cubozoans [17] will create localized polyp beds that are likely to make these stocks increasingly robust. The scales of jellyfish stocks are not only relevant to understanding their ecology but to demarcating boundaries for fisheries quotas [12], and for determining the risk of envenomation from dangerous jellyfish such as cubozoans [11]. Using multiple methods to determine levels of connectivity and stock boundaries is recommended, as a combination of approaches (Table 2) provides greater resolution of the hierarchy of population units for each species.
Author Contributions
Conceptualization, M.J.K., J.A.S. and S.J.M.; writing—original draft preparation, M.J.K.; writing—review and editing, M.J.K., J.A.S. and S.J.M. All authors have read and agreed to the published version of the manuscript.
Funding
The project was partly funded by an ARC Grant to M.J.K.
Acknowledgments
We would like to thank Mike Dawson for his thoughtful comments on connectivity among jellyfish populations.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Moller, H. Reduction of a larval herring population by a jellyfish predator. Science 1984, 224, 621–622. [Google Scholar] [CrossRef]
- Strand, S.W.; Hamner, W.M. Predatory behavior of Phacellophora camtschatica and size-selective predation upon Aurelia aurita (Scyphozoa: Cnidaria) in Saanich Inlet, British Columbia. Mar. Biol. 1988, 99, 409–414. [Google Scholar] [CrossRef]
- Brodeur, R.D.; Merati, N. Predation on walleye pollock (Theragra chalcogramma) eggs in the western Gulf of Alaska: The roles of vertebrate and invertebrate predators. Mar. Biol. 1993, 117, 483–495. [Google Scholar]
- Arai, M.N. A Functional Biology of Scyphozoa; Chapman & Hall: London, UK, 1997; p. 316. [Google Scholar]
- Kingsford, M.J. Biotic and abiotic structure in the pelagic environment: Importance to small fish. Bull. Mar. Sci. 1993, 53, 393–415. [Google Scholar]
- Doyle, T.K.; Hays, G.C.; Harrod, C.; Houghton, J.D.R. Ecological and Societal Benefits of Jellyfish. In Jellyfish Blooms; Pitt, K.A., Lucas, C.H., Eds.; Springer: Dordrecht, The Netherlands, 2014; pp. 105–127. [Google Scholar]
- Lynam, C.P.; Gibbons, M.J.; Axelsen, B.E.; Sparks, C.A.J.; Coetzee, J.; Heywood, B.G.; Brierley, A.S. Jellyfish overtake fish in a heavily fished ecosystem. Curr. Biol. 2006, 16, 492–493. [Google Scholar] [CrossRef] [PubMed]
- Canepa, A.; Fuentes, V.; Sabatés, A.; Piraino, S.; Boero, F.; Gili, J.-M. Pelagia noctiluca in the Mediterranean Sea. In Jellyfish Blooms, 1st ed.; Pitt, K.A., Lucas, C.H., Eds.; Springer: Dordrecht, The Netherlands; Berlin/Heidelberg, Germany; New York, NY, USA, 2014; pp. 237–266. [Google Scholar]
- Uye, S.-I. The Giant Jellyfish Nemopilema nomurai in East Asian marginal Seas. In Jellyfish Blooms; Pitt, K.A., Lucas, C.H., Eds.; Springer: Dordrecht, The Netherlands; Berlin/Heidelberg, Germany; New York, NY, USA, 2014; pp. 185–206. [Google Scholar]
- Graham, W.M.; Martin, D.L.; Felder, D.L.; Asper, V.L. Ecological and economic implications of a tropical jellyfish invader. Biol. Invasions 2003, 5, 53–69. [Google Scholar] [CrossRef]
- Kingsford, M.J.; Becken, J.S.; Bordehore, C.; Fuentes, V.L.; Pitt, K.A.; Yangihara, A.A. Empowering stakeholders to manage stinging jellyfish: A perspective. Coast. Man. 2018, 46, 1–18. [Google Scholar] [CrossRef]
- Kingsford, M.J.; Pitt, K.A.; Gillanders, B.M. Management of jellyfish fisheries, with special reference to the Order Rhizostomeae. Oceanogr. Mar. Biol. Ann. Rev. 2000, 38, 85–156. [Google Scholar]
- Brotz, L. Jellyfish fisheries—A global assessment. In Global Atlas of Marine Fisheries: A Critical Appraisal of Catches and Ecosystem Impacts; Pauly, D., Zeller, D., Eds.; Island Press: Washington, DC, USA, 2016; pp. 110–124. [Google Scholar]
- Daly, M.; Brugler, M.R.; Cartwright, P.; Collins, A.G.; Dawson, M.N.; Fautin, D.G.; France, S.C.; McFadden, C.S.; Opresko, D.M.; Rodriguez, E.; et al. The phylum Cnidaria: A review of phylogenetic patterns and diversity 300 years after Linnaeus. Zootaxa 2007, 1668, 127–182. [Google Scholar] [CrossRef]
- Dawson, M.N. Some implications of molecular phylogenetics for understanding biodiversity in jellyfishes, with emphasis on Scyphozoa. In Coelenterate Biology 2003; Springer: Dordrecht, The Netherlands, 2004; pp. 249–260. [Google Scholar]
- Collins, A.G.; Jarms, G. WoRMS Cubozoa: World list of Cubozoa (version 2018-04-01). In Species 2000 Naturalis & ITIS Catalogue of Life, 2018 Annual Checklist; Roskov, Y., Abucay, L., Orrell, T., Nicolson, D., Bailly, N., Kirk, P.M., Bourgoin, T., DeWalt, R.E., Decock, W., De Wever, A., et al., Eds.; Species 2000: Leiden, The Netherlands, 2018. [Google Scholar]
- Kingsford, M.J.; Mooney, C.M. The Ecology of Box Jellyfishes (Cubozoa). In Jellyfish Blooms; Pitt, K.A., Lucas, C.H., Eds.; Springer: Dordrecht, The Netherlands; Berlin/Heidelberg, Germany, 2014; pp. 267–302. [Google Scholar]
- Waples, R.S.; Gaggiotti, O.E. What is a population? An empirical evaluation of some genetic methods for identifying the number of gene pools and their degree of connectivity. Mol. Ecol. 2006, 15, 1419–1439. [Google Scholar] [CrossRef]
- Hutchings, P.; Kingsford, M.J. Biodiversity. In The Great Barrier Reef: Biology, Environment and Management; Hutchings, P., Kingsford, M.J., Hoegh-Guldberg, O., Eds.; CSIRO: Melbourne, Australia, 2019; pp. 183–189. [Google Scholar]
- Abboud, S.S.; Gómez Daglio, L.; Dawson, M.N. A global estimate of genetic and geographic differentiation in macromedusae—implications for identifying the causes of jellyfish blooms. Mar. Ecol. Prog. Ser. 2018, 591, 199–216. [Google Scholar] [CrossRef]
- Lewis, C.; Bentlage, B. Clarifying the identity of the Japanese Habu-kurage, Chironex yamaguchii, sp nov (Cnidaria: Cubozoa: Chirodropida). Zootaxa 2009, 2030, 59–65. [Google Scholar] [CrossRef]
- Sucharitakul, P.; Chomdej, S.; Achalawitkun, T.; Aongsara, S.; Arsiranant, S.; Paiphongpheaw, P.; Chanachon, K. Chirodropid box jellyfish in the Gulf of Thailand. Mar. Biodiv. 2018, 49, 1247–1252. [Google Scholar] [CrossRef]
- Keesing, J.K.; Strzelecki, J.; Stowar, M.; Wakeford, M.; Miller, K.J.; Gershwin, L.A.; Liu, D.Y. Abundant box jellyfish, Chironex sp (Cnidaria: Cubozoa: Chirodropidae), discovered at depths of over 50 m on western Australian coastal reefs. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef]
- Nuankanya, S.; Kasetsin, K.; Thunyaporn, P.; Mitila, P.; Supaporn, O.; Usawadee, D.; Bungbai, S.; Sam-ang, L.; Worawut, P.; Satariya, T. Rapid and Accurate Species-Specific PCR for the Identification of Lethal Chironex Box Jellyfish in Thailand. Int. J. Environ. Res. Public Health 2021, 18, 219. [Google Scholar] [CrossRef]
- WoRMS. World Register of Marine Species. Available online: http://www.marinespecies.org (accessed on 12 March 2021).
- Pineda, J.; Hare, J.A.; Sponaugle, S. Larval Transport and Dispersal in the Coastal Ocean and Consequences for Population Connectivity. Oceanography 2007, 20, 22–39. [Google Scholar] [CrossRef]
- Begg, G.A.; Friedland, K.D.; Pearce, J.B. Stock identification and its role in stock assessment and fisheries management: An overview. Fish. Res. 1999, 43, 1–8. [Google Scholar] [CrossRef]
- McManus, M.A.; Woodson, C.B. Plankton distribution and dispersal. J. Exp. Biol. 2012, 215, 1008–1016. [Google Scholar] [CrossRef]
- Gemmell, B.J.; Colin, S.P.; Costello, J.H. Widespread utilization of passive energy recapture in swimming medusae. J. Exp. Biol. 2018, 22, 1–5. [Google Scholar] [CrossRef]
- Larson, R.J. Riding Langmuir circulations and swimming in circles: A novel form of clustering behavior by the scyphomedusa Linuche unguiculata. Mar. Biol. 1992, 112, 229–235. [Google Scholar] [CrossRef]
- Shanks, A.L.; Graham, W.M. Orientated swimming in the jellyfish Stomolophus meleagris L. Agassiz (Scyphozoan: Rhizostomida). J. Exp. Mar. Biol. Ecol. 1987, 108, 159–169. [Google Scholar] [CrossRef]
- Schlaefer, J.A.; Wolanski, E.; Yadav, S.; Kingsford, M.J. Behavioural maintenance of highly localised jellyfish (Copula sivickisi, class Cubozoa) populations. Mar. Biol. 2020, 167. [Google Scholar] [CrossRef]
- Schlaefer, J.A.; Wolanski, E.; Kingsford, M.J. Swimming behaviour can maintain localised jellyfish (Chironex fleckeri: Cubozoa) populations. Mar. Ecol. Prog. Ser. 2018, 591, 287–302. [Google Scholar] [CrossRef]
- Nilsson, D.E.; Gislen, L.; Coates, M.M.; Skogh, C.; Garm, A. Advanced optics in a jellyfish eye. Nature 2005, 435, 201–205. [Google Scholar] [CrossRef]
- Hamner, W.M.; Hauri, I.R. Long-distance horizontal migrations of zooplankton (Scyphomedusae: Mastigias). Limnol. Oceanogr. 1981, 26, 414–423. [Google Scholar]
- Hamner, W.M.; Hamner, P.P.; Strand, S.W. Sun-compass migration by Aurelia aurita (Scyphozoa): Population rentention and reproduction in Saanich Inlet, British Columbia. Mar. Biol. 1994, 119, 347–356. [Google Scholar] [CrossRef]
- Matsumoto, G.I. Observations on the anatomy and behaviour of the cubozoan Carybdea rastonii Haacke. Mar. Freshw. Behav. Physiol. 1995, 26, 139–148. [Google Scholar] [CrossRef]
- Hartwick, R.F. Observations on the anatomy, behaviour, reproduction and life cycle of the cubozoan Carybdea sivickisi. Hydrobiologia 1991, 216–217, 171–179. [Google Scholar] [CrossRef]
- Hamner, W.M.; Jones, M.S.; Hamner, P.P. Swimming, feeding, circulation and vision in the Australian box jellyfish, Chironex fleckeri (Cnidaria: Cubozoa). Mar. Freshw. Res. 1995, 46, 985–990. [Google Scholar] [CrossRef]
- Garm, A.; O’Connor, M.; Parkefelt, L.; Nilsson, D.E. Visually guided obstacle avoidance in the box jellyfish Tripedalia cystophora and Chiropsella bronzie. J. Exp. Biol. 2007, 210, 3616–3623. [Google Scholar] [CrossRef]
- Kingsford, M.J.; Seymour, J.E.; O’Callaghan, M.D. Abundance patterns of cubozoans on and near the Great Barrier Reef. Hydrobiologia 2012, 690, 257–268. [Google Scholar] [CrossRef]
- Gordon, M.R.; Seymour, J.E. Quantifying movement of the tropical Australian cubozoan Chironex fleckeri using acoustic telemetry. Hydrobiologia 2009, 616, 87–97. [Google Scholar] [CrossRef]
- Buskey, E. Behavioral adaptations of the cubozoan medusa Tripedalia cystophora for feeding on copepod (Dioithona oculata) swarms. Mar. Biol. 2003, 142, 225–232. [Google Scholar] [CrossRef]
- Garm, A.; Bielecki, J.; Petie, R.; Nilsson, D.E. Opposite Patterns of Diurnal Activity in the Box Jellyfish Tripedalia cystophora and Copula sivickisi. Biol. Bull. 2012, 222, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Stewart, S.E. Field behavior of Tripedalia cystophora (class Cubozoa). Mar. Freshw. Behav. Physiol. 1996, 27, 175–188. [Google Scholar] [CrossRef]
- Garm, A.; Bielecki, J. Swim pacemakers in box jellyfish are modulated by the visual input. J. Comp. Physiol. A 2008, 194, 641–651. [Google Scholar] [CrossRef] [PubMed]
- Garm, A.; Oskarsson, M.; Nilsson, D.E. Box Jellyfish Use Terrestrial Visual Cues for Navigation. Curr. Biol. 2011, 21, 798–803. [Google Scholar] [CrossRef]
- Olson, R.R.; McPherson, R. Potential vs. realized larval dispersal: Fish predation on larvae of the ascidian Lissoclinum patella (Gottschaldt). J. Exp. Mar. Biol. Ecol. 1987, 110, 245–246. [Google Scholar] [CrossRef]
- Gerlach, G.; Atema, J.; Raupach, M.J.; Deister, F.; Muller, A.; Kingsford, M.J. Cryptic species of cardinalfish with evidence for old and new divergence. Coral Reefs 2016, 35, 437–450. [Google Scholar] [CrossRef]
- Brill, R.W.; Block, B.A.; Boggs, C.H.; Bigelow, K.A.; Freund, E.V.; Marcinek, D.J. Horizontal movements and depth distribution of large adult yellowfin tuna (Thunnus albacares) near the Hawaiian Islands, recorded using ultrasonic telemetry: Implications for the physiology and ecology of the species. Mar. Biol. 1999, 133, 385–408. [Google Scholar] [CrossRef]
- Almany, G.R.; Berumen, M.L.; Thorrold, S.R.; Planes, S.; Jones, G.P. Local replenishment of coral reef fish populations in a marine reserve. Science 2007, 316, 742–744. [Google Scholar] [CrossRef] [PubMed]
- Haddon, M.; Willis, T.J. Morphometric and meristic comparison of orange roughy (Hoplstethus atlanticus: Trachichthyidae) from the Puysegur Bank and Lord Howe Rise, New Zealand, and its implication for stock structure. Mar. Biol. 1995, 123, 19–27. [Google Scholar] [CrossRef]
- Small, C.G. The Statistical Theory of Shape; Springer: Berlin/Heidelberg, Germany, 1996. [Google Scholar]
- Fowler, A.J.; Gillanders, B.M.; Hall, K.C. Relationship between elemental concentration and age from otoliths of adult snapper (Pagrus auratus, Sparidae): Implications for movement and stock structure. Mar. Freshw. Res. 2005, 56, 661–676. [Google Scholar] [CrossRef]
- Wolanski, E.; Kingsford, M.J. Oceanographic and behavioural assumptions in models of the fate of coral and coral reef fish larvae. J. Roy. Soc. Int. 2014, 11, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Pitt, K.A.; Kingsford, M.J. Geographic separation of stocks of the edible jellyfish, Catostylus mosaicus (Rhizostomeae) in New South Wales, Australia. Mar. Ecol. Prog. Ser. 2000, 196, 143–155. [Google Scholar] [CrossRef]
- Dawson, M.N. Incipient speciation of Catostylus mosaicus (Scyphozoa, Rhizostomeae, Catostylidae), comparative phylogeography, and biogeography in south-east Australia. J. Biogeogr. 2005, 31, 1–19. [Google Scholar] [CrossRef]
- Dawson, M.N. Morphological and molecular redescription of Catostylus mosaicus (Scyphozoa: Rhizostomeae: Catostylidae) from south-east Australia. J. Mar. Biol. Assoc. 2005, 85, 723–731. [Google Scholar] [CrossRef]
- Ben Faleh, A.R.; Ben Othmen, A.; Deli, T.; Annabi, A.; Said, K. High genetic homogeneity of the moon jelly Aurelia aurita (Scyphozoa, Semaeostomeae) along the Mediterranean coast of Tunisia. Afr. J. Mar. Sci. 2009, 31, 73–80. [Google Scholar] [CrossRef]
- Dong, Z.; Liu, Z.; Liu, D. Genetic characterization of the scyphozoan jellyfish Aurelia spp. in Chinese coastal waters using mitochondrial markers. Biochem. Syst. Ecol. 2015, 60, 15–23. [Google Scholar] [CrossRef]
- De Angelis, S.A.; Stampar, S.N.; Maronna, M.M.; Morandini, A.C. Absence of cryptic species and population structure in Lychnorhiza lucerna (Cnidaria) from southwestern Atlantic Ocean. Genome 2017, 60, 925–926. [Google Scholar]
- Lee, P.L.; Dawson, M.N.; Neill, S.P.; Robins, P.E.; Houghton, J.D.; Doyle, T.K.; Hays, G.C. Identification of genetically and oceanographically distinct blooms of jellyfish. J. R. Soc. Interface 2013, 10, 20120920. [Google Scholar] [CrossRef]
- Glynn, F.; Houghton, J.D.; Provan, J. Population genetic analyses reveal distinct geographical blooms of the jellyfish Rhizostoma octopus (Scyphozoa). Biol. J. Linn. Soc. 2015, 116, 582–592. [Google Scholar] [CrossRef]
- Miller, B.J.; Von der Heyden, S.; Gibbons, M.J. Significant population genetic structuring of the holoplanktic scyphozoan Pelagia noctiluca in the Atlantic Ocean. Afr. J. Mar. Sci. 2012, 34, 425–430. [Google Scholar] [CrossRef]
- Stopar, K.; Ramšak, A.; Trontelj, P.; Malej, A. Lack of genetic structure in the jellyfish Pelagia noctiluca (Cnidaria: Scyphozoa: Semaeostomeae) across European seas. Mol. Phylogenet. Evol. 2010, 57, 417–428. [Google Scholar] [CrossRef]
- Dawson, M.N.; Hamner, W.M. Geographic variation and behavioural evolution in marine plankton: The case of Mastigias (Scyphozoa, Rhizostomeae). Mar. Biol. 2003, 143, 1161–1174. [Google Scholar] [CrossRef]
- Mamet, L.N.G.; Daglio, L.G.; García-De León, F.J. High genetic differentiation in the edible cannonball jellyfish (cnidaria: Scyphozoa: Stomolophus spp.) from the Gulf of California, Mexico. Fish. Res. 2019, 219, 105328. [Google Scholar] [CrossRef]
- Dawson, M.N.; Cieciel, K.; Decker, M.B.; Hays, G.C.; Lucas, C.L. Population-level perspectives on global change: Genetic and demographic analyses indicate various scales, timing, and causes of scyphozoan jellyfish blooms. Biol. Invasions 2015, 17, 851–867. [Google Scholar] [CrossRef]
- Lawley, J.W.; Ames, C.L.; Bentlage, B.; Yanagihara, A.; Goodwill, R.; Kayal, E.; Hurwitz, K.; Collins, A.G. Box jellyfish Alatina alata has a circumtropical distribution. Biol. Bull. 2016, 231, 152–169. [Google Scholar] [CrossRef] [PubMed]
- Mooney, C.J.; Kingsford, M.J. Statolith morphometrics as a tool to distinguish among populations of three cubozoan species. Hydrobiologia 2017, 787, 111–121. [Google Scholar] [CrossRef]
- Mooney, C.J.; Kingsford, M.J. Discriminating populations of medusae (Chironex fleckeri, Cubozoa) using statolith microchemistry. Mar. Freshw. Res. 2016, 68, 1144–1152. [Google Scholar] [CrossRef]
- Dawson, M.N.; Hamner, W.M. Rapid evolutionary radiation of marine zooplankton in peripheral environments. Proc. Natl. Acad. Sci. USA 2005, 102, 9235–9240. [Google Scholar] [CrossRef] [PubMed]
- Galil, B.S.; Spanier, E.; Ferguson, W.W. The scyphomedusae of the mediterrannean coast of Israel, including two lessepsian migrants new to the Mediterranean. Zoo. Mededel. 1990, 64, 95–105. [Google Scholar]
- Brown, W.M.; George, M.; Wilson, A.C. Rapid evolution of animal mitochondrial DNA. Proc. Natl. Acad. Sci. USA 1979, 76, 1967–1971. [Google Scholar] [CrossRef]
- Peplow, L.M.; Kingsford, M.J.; Seymour, J.E.; van Oppen, M.J.H. Eight microsatellite loci for the Irukandji syndrome-causing carybdeid jellyfish, Carukia barnesi (Cubozoa, Cnidaria). Mol. Ecol. Resour. 2009, 9, 670–672. [Google Scholar] [CrossRef] [PubMed]
- Coughlan, J.P.; Seymour, J.; Cross, T.F. Isolation and characterization of seven polymorphic microsatellite loci in the box jellyfish (Chironex fleckeri, Cubozoa, Cnidaria). Mol. Ecol. Notes 2006, 6, 41–43. [Google Scholar] [CrossRef]
- Aungtonya, C.; Xiao, J.; Zhang, X.; Wutthituntisil, N. The Genus Chiropsoides (Chirodropida: Chiropsalmidae) from the Andaman Sea, Thai waters. Acta Oceanol. Sin. 2018, 30, 119–125. [Google Scholar] [CrossRef]
- Bolte, B.; Goldsbury, J.; Huerlimann, R.; Jerry, D.; Kingsford, M.J. Validation of eDNA as a viable method of detection for dangerous cubozoan jellyfish. Environ. DNA 2021. [Google Scholar] [CrossRef]
- Berta, A.; Sumich, J.L.; Kovacs, K.M. Population structure and dynamics. In Marine Mammals—Evolutionary Biology; Berta, A., Sumich, J.L., Kovacs, K.M., Eds.; Academic Press: Burlington, MA, USA, 2015; pp. 416–445. [Google Scholar]
- Holland, B.S.; Dawson, M.N.; Crow, G.L.; Hofmann, D.K. Global phylogeography of Cassiopea (Scyphozoa: Rhizostomeae): Molecular evidence for cryptic species and multiple invasions of the Hawaiian Islands. Mar. Biol. 2004, 146, 1119–1128. [Google Scholar] [CrossRef]
- Bolton, T.F.; Graham, W.M. Morphological variation among populations of an invasive jellyfish. Mar. Ecol. Prog. Ser. 2004, 278, 125–139. [Google Scholar] [CrossRef]
- Campana, S.E.; Chouinard, G.A.; Hanson, J.M.; Frechet, A.; Brattey, J. Otolith elemental fingerprints as biological tracers of fish stocks. Fish. Res. 2000, 46, 343–357. [Google Scholar] [CrossRef]
- Mooney, C.J.; Kingsford, M.J. Statolith Morphometrics Can Discriminate among Taxa of Cubozoan Jellyfishes. PLoS ONE 2016, 11, 16. [Google Scholar] [CrossRef]
- Tiemann, H.; Sotje, I.; Becker, A.; Jarms, G.; Epple, M. Calcium sulfate hemihydrate (bassanite) statoliths in the cubozoan Carybdea sp. Zool. Anz. 2006, 245, 13–17. [Google Scholar] [CrossRef]
- Barz, K.; Hinrichsen, H.H.; Hirche, H.J. Scyphozoa in the Bornholm Basin (central Baltic Sea)-the role of advection. J. Mar. Syst. 2006, 60, 167–176. [Google Scholar] [CrossRef]
- Chen, K.; Ciannelli, L.; Decker, B.B.; Ladd, C.; Cheng, W.; Zhou, Z.; Chan, K.-S. Reconstructing Source-Sink Dynamics in a Population with a Pelagic Dispersal Phase. PLoS ONE 2014, 9, e95316. [Google Scholar] [CrossRef]
- Fossett, S.; Gleiss, A.C.; Chalumeau, J.; Bastian, T. Current-oriented swimming by jellyfish and its role in bloom maintenance. Curr. Biol. 2015, 25, 342–347. [Google Scholar] [CrossRef] [PubMed]
- Hopley, D.; Smithers, S.G.; Parnell, K.E. The Geomorpholgy of the Great Barrier Reef; Cambridge University Press: New York, NY, USA, 2007; p. 532. [Google Scholar]
- Shahrestani, S.; Bi, H. Settlement and survival of Chrysaora chesapeakei polyps: Implications for adult abundance. Mar. Ecol. Progr. Ser. 2018, 601, 139–151. [Google Scholar] [CrossRef]
- Marques, R.; Cantou, M.; Soriano, S.; Molinero, J.C.; Bonnet, D. Mapping distribution and habitats of Aurelia sp. polyps in Thau lagoon, north-western Mediterranean Sea (France). Mar. Biol. 2015, 162, 1441–1449. [Google Scholar] [CrossRef]
- Colin, S.P.; Kremer, P. Population maintenance of the scyphozoan Cyanea sp. settled planulae and the distribution of medusae in the Niantic River, Connecticut, USA. Estuaries 2002, 25, 70–75. [Google Scholar] [CrossRef]
- Toyokawa, M.; Aoki, K.; Yamada, S.; Yasuda, A.; Murata, Y.; Kikuchi, T. Distribution of ephyrae and polyps of jellyfish Aurelia aurita (Linnaeus 1758) sensu lato in Mikawa Bay, Japan. J. Oceanogr. 2011, 67, 209–218. [Google Scholar] [CrossRef]
- Van Walraven, L.; Driessen, F.; van Bleijswijk, J.; Bol, A.; Luttikhuizen, P.C.; Coolen, J.W.; van der Veer, H.W. Where are the polyps? Molecular identification, distribution and population differentiation of Aurelia aurita jellyfish polyps in the southern North Sea area. Mar. Biol. 2016, 163, 1–13. [Google Scholar] [CrossRef]
- Hartwick, R.F. Distributional ecology and behavior of the early life stages of the box-jellyfish Chironex fleckeri. Hydrobiologia 1991, 216–217, 181–188. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).