Sporolithon franciscanum sp. nov. (Sporolithales, Rhodophyta), a New Rhodolith-Forming Species from Northeast Brazil
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling
2.2. Morpho-Anatomical Analyses
2.3. Molecular Analysis
3. Results
3.1. Sporolithon franciscanum L.A.S. Leão & Bahia sp. nov.
3.2. Habit and Vegetative Anatomy
3.3. Reproductive Anatomy
3.4. Diagnosis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Silva, P.C.; Johansen, H.W. A reappraisal of the order Corallinales (Rhodophyceae). Br. Phycol. J. 1986, 21, 245–254. [Google Scholar] [CrossRef]
- Borowitza, M.A. Calcification in algae: Mechanisms and the role of metabolism. Crit. Rev. Plant Sci. 1987, 6, 1–45. [Google Scholar] [CrossRef]
- Nash, M.C.; Troitzsch, U.; Opdyke, B.N.; Trafford, J.M.; Russel, B.D.; Kline, D.I. First discovery of dolomite and magnesite in living coralline algae and its geobiological implications. Biogeosciences 2011, 8, 3331–3340. [Google Scholar] [CrossRef]
- Krayesky-Self, S.; Richards, J.L.; Rahmatian, M.; Fredericq, S. Aragonite infill in overgrown conceptacles of coralline Lithothamnion spp. (hapalidiaceae, hapalidiales, rhodophyta): New Insights in biomineralization and phylomineralogy. J. Phycol. 2016, 52, 161–173. [Google Scholar] [CrossRef] [PubMed]
- Woelkerling, W.M.J. The Coralline Red Algae: An Analysis of the Genera and Sub-Families of Nongeniculate Corallinaceae; British Museum Natural History and Oxford University Press: London, UK, 1988; p. 280. [Google Scholar]
- Irvine, L.M.; Chamberlain, Y.M. Seaweeds of the Bristish Isles. Volume I Rhodophyta. Part 2B Corallinales, Hildenbrandiales; Her Majesty’s Stationary Office: London, UK, 1994; p. 276. [Google Scholar]
- Woelkerling, W.J.; Lamy, D. Non-Geniculate Coralline Red Algae and the Paris Museum: Systematics and Scientific History; Publications Scientifiques du Muséum/ADAC: Paris, France, 1998; pp. 1–767. [Google Scholar]
- Maneveldt, G.; Chamberlain, Y.M.; Keats, D.W. A catalogue with keys to the non-geniculate coralline algae (Corallinales, Rhodophyta) of South Africa. S. Afr. J. Bot. 2008, 555–566. [Google Scholar] [CrossRef]
- Harvey, A.S.; Woelkerling, W.J. A guide to nongeniculate coralline red algal (Corallinales, Rhodophyta) rhodolith identification. Cienc. Mar. 2007, 33, 411–426. [Google Scholar] [CrossRef]
- Amado-Filho, G.M.; Maneveldt, G.; Pereira-Filho, G.H.; Manso, R.C.; Bahia, R.G.; Barreto, M.B.B.; Guimaraes, S.M.P.B. Seaweed diversity associated with a Brazilian tropical rhodolith bed. Cienc. Mar. 2010, 36, 371–391. [Google Scholar]
- Pereira-Filho, G.H.; Amado Filho, G.M.; Guimarães, S.M.P.B.; Moura, R.L.; Sumida, P.Y.G.; Abrantes, D.P.; Bahia, R.G.; Guth, A.Z.; Jorge, R.; Francini-Filho, R.B. Reef Fish and Benthic assemblages of the Trindade and Martin Vaz island group, Southwestern Atlantic. Braz. J. Oceanogr. 2011, 59, 201–212. [Google Scholar] [CrossRef]
- Bailey, J.C.; Chapman, R.L. Evolutionary relationships among coralline red algae (Corallinaceae, Rhodophyta) determined by 18S rDNA gene sequence analysis. In Cytology, Genetics and Molecular Biology of Algae; Chaudhary, B.R., Agrawal, S.B., Eds.; SPB Academic Publishing: Amsterdam, The Netherlands, 1996; pp. 363–376. [Google Scholar]
- Broom, J.E.S.; Hart, D.R.; Farr, T.J.; Nelson, W.A.; Neill, K.F.; Harvey, A.S.; Woelkerling, W.J. Utility of psbA and nSSU for phylogenetics reconstruction in the Corallinales based on New Zealand taxa. Mol. Phylogenet. Evol. 2008, 46, 958–973. [Google Scholar] [CrossRef]
- Nelson, W.A.; Sutherland, J.E.; Farr, T.J.; Hart, D.R.; Neill, K.F.; Kim, H.J.; Yoon, H.S. Multi-gene phylogenetic analyses of New Zealand coralline algae: Corallinapetra novaezelandiae gen. et and recognition of the Hapalidiales ord. nov. J. Phycol. 2015, 51, 454–468. [Google Scholar] [CrossRef]
- Richards, J.L.; Sauvage, T.; Schmidt, W.E.; Fredericq, S.; Huguey, J.R.; Gabrielson, P. The coralline genera Sporolithon and Heydrichia (Sporolithales, Rhodophyta) clarified by sequencing type material of their generitypes and other species. J. Phycol. 2017, 53, 1044–1059. [Google Scholar] [CrossRef] [PubMed]
- Hind, K.R.; Gabrielson, P.W.; Saunders, G.W. Molecular-assisted alpha taxonomy reveals pseudocryptic diversity among species of Bossiella (Corallinales, Rhodophyta) in the eastern Pacific Ocean. Phycologia 2014, 53, 443–456. [Google Scholar] [CrossRef]
- Caragnano, A.; Foetisch, A.; Maneveldt, G.; Millet, L.; Liu, L.C.; Lin, S.M.; Rodondi, G.; Payri, C.E. Revision of Corallinaceae (Corallinales, Rhodophyta): Recognizing Dawsoniolithon, gen. nov., Parvicellularium gen. nov. and Chamberlainioideae subfam. nov. containing Chamberlainium gen. nov. and Pneophyllum. J. Phycol. 2018, 54, 391–409. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.C.; Lin, S.M.; Caragnano, A.; Payri, C. Species diversity and molecular phylogeny of non-geniculate coralline algae (Corallinophycidae, Rhodophyta) from Taoyuan algal reefs in northern Taiwan, including Crustaphytum gen. nov. and three new species. J. Appl. Phycol. 2018, 30, 3455–3469. [Google Scholar] [CrossRef]
- Aki, K.; Masasuke, B. Distribution of Lithophyllum kuroshioense sp. nov., Lithophyllum subtile and L. kaiseri (Corallinales, Rhodophyta), but not L. kotschyanum, in the northwestern Pacific Ocean. Phycologia 2019. [Google Scholar] [CrossRef]
- Steneck, R.S. The ecology of coralline algal crusts: Convergent patterns and adaptative strategies. Annu. Rev. Ecol. Systemat. 1986, 17, 273–303. [Google Scholar] [CrossRef]
- Foster, M.S. Rhodoliths: Between rocks and soft places—Minireview. J. Phycol. 2001, 37, 659–667. [Google Scholar] [CrossRef]
- Foster, M.S.; Amado-Filho, G.M.; Kamenos, N.A.; Riosmena-Rodríguez, R.; Steller, D.L. Rhodoliths and Rhodoliths beds. In Research and Discoveries: The Revolution of Science through SCUBA; Lang, M.A., Marinelli, R.L., Roberts, S.J., Taylor, P.R., Eds.; Smithsonian Institution Scholarly Press: Washington, DC, USA, 2013; pp. 143–155. [Google Scholar]
- Maneveldt, G.W.; Gabrielson, P.; Kangwe, J. Sporolithon indopacificum sp. nov. (Sporolithales, Rhodophyta) from tropical western Indian and western Pacific oceans: First report, confirmed by DNA sequence data, of a widely distributed species of Sporolithon. Phytotaxa 2017, 326, 115–128. [Google Scholar] [CrossRef]
- Richards, J.L.; Fredericq, S. Sporolithon sinusmexicanum sp. nov. (Sporolithales, Rhodophyta): A new rhodolith-forming species from deepwater rhodolith beds in the Gulf of Mexico. Phytotaxa 2018, 350, 135–146. [Google Scholar] [CrossRef]
- Richards, J.L.; Gabrielson, P.; Schneider, C.W. Sporolithon mesophoticum sp. nov. (Sporolithales, Rhodophyta) from Plantagenet Bank off Bermuda at a depth of 178 m. Phytotaxa 2018, 385, 67–76. [Google Scholar] [CrossRef]
- Richards, J.L.; Bahia, R.G.; Jesionek, M.B.; Fredericq, S. Sporolithon amadoi sp. nov. (Sporolithales, Rhodophyta), a new rhodolith-forming non-geniculate coralline alga from offshore the northwestern Gulf of Mexico and Brazil. Phytotaxa 2019, 423, 49–67. [Google Scholar] [CrossRef]
- Amado-Filho, G.M.; Bahia, R.G.; Pereira-Filho, G.H.; Longo, L.L. South Atlantic Rhodolith Beds: Latitudinal distribution, species composition, structure and ecosystem functions, threats and conservation status. In Rhodolith/Maerl Beds: A Global Perspective; Riosmena-Rodrigues, R., Nelson, W., Aguirre, J., Eds.; Springer: New York, NY, USA, 2017; Volume 15, pp. 229–318. [Google Scholar]
- Verheij, E. The genus Sporolithon (Sporolithaceae fame nov., Corallinales, Rhodophyta) from the Spermonde Archipelago, Indonesia. Phycologia 1993, 32, 184–196. [Google Scholar] [CrossRef]
- Le Gall, L.; Payri, C.E.; Bittner, L.; Saunders, G.W. Multigene phylogenetic analyses support recognition of the Sporolithales ord. nov. Mol. Phylogenet. Evol. 2010, 54, 302–305. [Google Scholar] [CrossRef] [PubMed]
- Bahia, R.G.; Riosmena-Rodriguez, R.; Maneveldt, G.W.; Amado-Filho, G.M. First report of Sporolithon ptychoides (Sporolithales, Corallinophycidae, Rhodophyta) for the Atlantic Ocean. Phycol. Res. 2011, 59, 64–69. [Google Scholar] [CrossRef]
- Bahia, R.G.; Amado-Filho, G.M.; Maneveldt, G.W. Sporolithon molle (Heydrich) Heydrich (Sporolithales, Corallinophycidae, Rhodophyta): An addition to the Atlantic flora found on a remote oceanic island. Cryptogam. Algol. 2014, 35, 7–14. [Google Scholar] [CrossRef]
- Bahia, R.G.; Amado-Filho, G.M.; Maneveldt, G.W.; Adey, W.H.; Johnson, G.; Marins, B.V.; Longo, L.L. Sporolithon tenue sp. nov. (Sporolithales, Corallinophycidae, Rhodophyta): A new rhodolith-forming species from the tropical southwestern Atlantic. Phycol. Res. 2014, 62, 44–54. [Google Scholar] [CrossRef]
- Bahia, R.G.; Amado-Filho, G.M.; Maneveldt, G.W.; Adey, W.H.; Johnson, G.; Jesionek, M.B.; Longo, L.L. Sporolithon yoneshigueae sp. nov. (Sporolithales, Corallinophycidae, Rhodophyta), a new rhodolith-forming coralline alga from the southwest Atlantic. Phytotaxa 2015, 224, 140–158. [Google Scholar] [CrossRef]
- Henriques, M.C.; Coutinho, L.M.; Riosmena-Rodriguez, R.; Barros-Barreto, M.B.; Khader, S.; Figueiredo, M.A. Three deep water species of Sporolithon (Sporolithales, Rhodophyta) from the Brazilian continental shelf, with the description of Sporolithon elevatum sp. nov. Phytotaxa 2014, 190, 320–330. [Google Scholar] [CrossRef]
- Jesionek, M.B.; Bahia, R.G.; Hernández-Kantún, J.; Adey, W.H.; Yoneshigue-Valentin, Y.; Longo, L.L.; Amado-Filho, G.M. A taxonomic account of non-geniculate coralline algae (Corallinophycidae, Rhodophyta) from shallow reefs of the Abrolhos Bank, Brazil. Algae 2016, 31, 317–340. [Google Scholar] [CrossRef]
- Guiry, M.D.; Guiry, G.M. AlgaeBase. World-Wide Electronic Publication. National University of Ireland, Galway. Available online: http://www.algaebase.org (accessed on 26 April 2020).
- Tomita, N.Y. Contribuição ao conhecimento do gênero Sporolithon (Corallinaceae, Cryptonemiales) no Brasil. Ph.D. Thesis, Universidade de São Paulo, São Paulo, Brazil, 1976; p. 138. [Google Scholar]
- Wynne, M.J. A checklist of benthic marine algae of the tropical and subtropical Western Atlantic: Third revision; J. Cramer in der Gebr. Borntraeger Verlagsbuchhandlung: Stuttgart, Germany, 2011; p. 166. [Google Scholar]
- Costa, I.O.; Horta, P.A.; Bergstrom, E.R.; Nunes, J.M.C. Taxonomic study of crustose coralline algae off the northeastern Brazilian coast. Phytotaxa 2014, 190, 130–161. [Google Scholar] [CrossRef]
- Bahia, R.G.; Maneveldt, G.W.; Amado-Filho, G.M.; Yoneshigue-Valentin, Y. New diagnostic characters for the order Sporolithales (Corallinophycideae, Rhodophyta). J. Phycol. 2015, 51, 1137–1146. [Google Scholar] [CrossRef] [PubMed]
- Rosler, A.; Perfectti, F.; Peña, V.; Braga, J. Phylogenetic relationships of Corallinaceae (Corallinales, Rhodophyta): Taxonomic implications for reef-building corallines. J. Phycol. 2016, 52, 412–431. [Google Scholar] [CrossRef] [PubMed]
- Pezzolesi, L.; Peña, V.; Le Gall, L.; Gabrielson, P.W.; Kaleb, S.; Huguey, J.R.; Rodondi, G.; Hernandez-Kantún, J.J.; Falace, A.; Basso, D.; et al. Mediterranean lithophyllum stictiforme (corallinales, rhodophyta) is a Genetically diverse species complex: Implications for species circumscription, biogeography and conservation of coralligenous habitats. J. Phycol. 2019, 55, 473–492. [Google Scholar] [CrossRef] [PubMed]
- Hall-Spencer, J.M.; Grall, J.; Moore, P.G.; Atkinson, R.J.A. Bivalve fishing and maerl-bed conservation in France and the UK—Retrospect and prospect. Aquat. Conserv. 2003, 13, S33–S41. [Google Scholar] [CrossRef]
- Amado-Filho, G.M.; Maneveldt, G.; Manso, R.C.C.; Marins-Rosa, B.V.; Pacheco, M.R.; Guimarães, S.M.P.B. Structure of rhodolith beds from 4 to 55 meters deep along the southern coast of Espírito Santo State, Brazil. Cienc. Mar. 2007, 33, 399–410. [Google Scholar] [CrossRef]
- Nelson, W.A.; Neill, K.; Farr, T.; Barr, N.; D’Archino, R.; Miller, S.; Stewart, R. Rhodolith Beds in Northern New Zealand: Characterisation of Associated Biodiversity and Vulnerability to Environmental Stressors. N. Z. Aquat. Environ. Biodivers. Rep. 2012, 99, 102. [Google Scholar]
- Foster, M.S.; Riosmena-Rodríguez, R.; Steller, D.; Woelkerling, W.M.J. Living rhodolith beds in the Gulf of California and their significance for paleoenvironmental interpretation. In Pliocene Carbonates and Related Facies Flanking the Gulf of California, Baja California, Mexico; Special Paper Number 31; Johnson, M.E., Ledesma-Vazquez, J., Eds.; Geological Society of America: Boulder, CO, USA, 1997; pp. 127–139. [Google Scholar]
- Steller, D.L.; Riosmena-Rodríguez, R.; Foster, M.S.; Roberts, C. Rhodolith bed diversity in the Gulf of California: The importance of rhodolith structure and consequences of anthropogenic disturbances. Aquat. Conserv. Mar. Freshw. Ecosyst. 2003, 13, 5–20. [Google Scholar] [CrossRef]
- Littler, M.M.; Littler, D.S. Coralline algal rhodoliths form extensive benthic communities in the gulf of Chiriqui, Pacific Panama. Coral Reefs 2008, 27, 553. [Google Scholar] [CrossRef]
- Martin, S.; Hall-Spencer, J.M. Effects of Ocean Warming and Acidification on Rhodolith/Maërl Beds. In Rhodolith/Maerl Beds: A Global Perspective; Riosmena-Rodrigues, R., Nelson, W., Aguirre, J., Eds.; Coastal Research Library, Springer: New York, NY, USA, 2017; Volume 15, pp. 55–85. [Google Scholar]
- McCoy, S.J.; Kamenos, N.A. Coralline algae (Rhodophyta) in a changing world: Integrating ecological, physiological, and geochemical responses to global change. J. Phycol. 2015. [Google Scholar] [CrossRef]
- Kamenos, N.; Cusack, M.; Moore, P. Coralline algae are global palaeothermometers with biweekly resolution. Geochim. Cosmochim. Acta. 2008, 72, 771–779. [Google Scholar] [CrossRef]
- Nelson, W. Calcifi ed macroalgae–critical to coastal ecosystems and vulnerable to change: A review. Mar. Freshw. Res. 2009, 60, 787–801. [Google Scholar] [CrossRef]
- Amado-Filho, G.M.; Moura, R.L.; Bastos, A.C.; Salgado, L.T.; Sumida, P.Y.G.; Guth, A.Z.; Francini-Filho, R.B.; Pereira-Filho, G.H.; Abrantes, D.P.; Brasileiro, P.S.; et al. Rhodolith beds are major CaCO3 bio-factories in the Tropical South West Atlantic. PLoS ONE 2012, 7, e35171. [Google Scholar] [CrossRef]
- Amado-Filho, G.M.; Pereira-Filho, G.H.; Bahia, R.G.; Abrantes, D.P.; Veras, P.C.; Matheus, Z. Occurrence and distribution of rhodolith beds on the Fernando de Noronha Archipelago of Brazil. Aquat. Bot. 2012, 101, 41–45. [Google Scholar] [CrossRef]
- Amado-Filho, G.M.; Bahia, R.G.; Mariath, R.; Jesionek, M.B.; Moura, R.L.; Bastos, A.C.; Pereira-Filho, G.; Francini-Filho, R.B. Spatial and temporal dynamics of the abundance of crustose calcareous algae on the southernmost coral reefs of the western Atlantic (Abrolhos Bank, Brazil). Algae 2018, 33, 85–99. [Google Scholar] [CrossRef]
- Holz, V.L.; Bahia, R.G.; Karez, C.S.; Vieira, F.V.; Moraes, F.C.; Vale, N.F.; Sudatti, D.B.; Salgado, L.T.; Moura, R.L.; Amado-Filho, G.M.; et al. Structure of Rhodolith Beds and Surrounding Habitats at the Doce River Shelf (Brazil). Diversity 2020, 12, 75. [Google Scholar] [CrossRef]
- Morse, D.E.; Morse, A.N.C. Enzymatic characterization of the morphogen recognized by Agaricia humilis (scleractinian coral) larvae. Biol. Bull. 1991, 181, 104–122. [Google Scholar] [CrossRef]
- Harrington, L.; Fabricius, K.; De’ath, G.; Negri, A. Recognition and selection of settlement substrata determine post-settlement survival in corals. Ecology 2004, 85, 3428–3437. [Google Scholar] [CrossRef]
- Roberts, R.D.; Kaspar, H.F.; Barker, R.J. Settlement of abalone (Haliotis iris) larvae in response to five species of coralline algae. Shellfish. Res. 2004, 23, 975–987. [Google Scholar]
- Knoppers, B.; Medeiros, P.R.P.; Souza, W.F.L.; Jennerjahn, T. The São Francisco Estuary, Brazil. In The Handbook of Environmental Chemistry; Wangersky, P.J., Ed.; Springer: Berlin/Heidelberg, Germany, 2005; Volume 5, pp. 51–70. [Google Scholar]
- Medeiros, P.R.P.; Knoppers, B.; Souza, W.F.L.; Oliveira, E.N. Contribution of suspended matter in the lower São Francisco River (SE/AL), across diferent hydrologic conditions. Braz. J. Aquat. Sci. Technol. 2011, 15, 42–53. [Google Scholar] [CrossRef]
- Fontes, L.C.S.; Santos, J.R.; Santos, L.A.; Mendonça, J.B.S.; Santos, M.S. Sedimentos superficiais da plataforma continental de Sergipe-Alagoas. In Geologia e Geomorfologia da Bacia de Sergipe-Alagoas; Carneiro, M.E.R., Ed.; Editora UFS; Coleção Projeto Marseal: São Cristovão, Brazil, 2017; Volume 1, pp. 64–96. [Google Scholar]
- Adey, W.H.; Hernández-Kantún, J.; Johnson, G.; Gabrielson, P.W. Dna sequencing, anatomy, and calcification patterns support a monophyletic, subarctic, carbonate reef-forming Clathromorphum (Hapalidiaceae, Corallinales, Rhodophyta). J. Phycol. 2015, 51, 189–203. [Google Scholar] [CrossRef]
- Thiers, B. Index Herbariorum: A Global Directory of Public Herbaria and Associated Staff. New York Botanical Garden’s Virtual Herbarium. Available online: http://sweetgum.nybg.org/ih/ (accessed on 16 September 2019).
- Maneveldt, G.W.; Van der Merwe, E. Heydrichia cerasina sp. nov. (Sporolithales, Corallinophycidae, Rhodophyta) from the southernmost tip of Africa. Phycologia 2012, 51, 11–21. [Google Scholar] [CrossRef]
- Bahia, R.G.; Abrantes, D.P.; Brasileiro, P.S.; Pereira-Filho, G.H.; Amado-Filho, G.M. Rhodolith bed structure along a depth gradient on the northern coast of Bahia State, Brazil. Braz. J. Oceanogr. 2010, 58, 323–337. [Google Scholar] [CrossRef]
- Chamberlain, Y.M. The genus Leptophytum (Rhodophyta, Corallinaceae) in the British Isles with descriptions of Leptophytum bornetii, L. elatum sp. nov. and L. laeve. Br. Phycol. J. 1990, 25, 179–199. [Google Scholar] [CrossRef]
- Woelkerling, W.J.; Irvine, L.M.; Harvey, A. Growth-forms in non-geniculate coralline red algae (Corallinales, Rhodophyta). Aust. Syst. Bot. 1993, 6, 277–293. [Google Scholar] [CrossRef]
- Kaewsuralikhit, C.; Maneekat, S.; Noiraska, T.; Patarajinda, S.; Baba, M. First record of Sporolithon ptychoides Heydrich (Sporolithales, Corallinophycidae, Rhodophyta) from Thailand. Cryptogam. Algol. 2012, 33, 265–276. [Google Scholar] [CrossRef]
- Jesionek, M.B.; Bahia, R.G.; Lyra, M.B.; Leão, L.A.B.; Amado-Filho, G.M. Newly discovered coralline algae in Southeast Brazil: Tectolithon fluminense gen. et sp. nov. and Crustaphytum atlanticum sp. nov. (Hapalidiales, Rhodophyta). Phycologia 2020, 2020, 1–15. [Google Scholar] [CrossRef]
- Yoon, H.S.; Hackett, J.D.; Bhattacharya, D. A single origin of the peridinin and fucoxanthin containing plastids in dinoflagellates through tertiary endosymbiosis. Proc. Natl. Acad. Sci. USA 2002, 99, 11724–11729. [Google Scholar] [CrossRef]
- Hughey, J.R.; Silva, P.C.; Hommersand, M.H. Solving taxonomic and nomenclatural problems in Pacific Gigartinaceae (Rhodophyta) using DNA from type material. J. Phycol. 2001, 37, 1091–1109. [Google Scholar] [CrossRef]
- Freshwater, D.W.; Rueness, J. Phylogenetic relationships of some European Gelidium (Geliadiales, Rhodophyta) species, Based on rbcL nucleotide sequence analysis. Phycologia 1994, 33, 187–194. [Google Scholar] [CrossRef]
- Tamura, K.; Stecher, G.; Peterson, D.; Filipski, A.; Kumar, S. MEGA6: Molecular evolutionary genetics analysis version 6.0. Mol. Biol. Evol. 2013, 30, 2725–2729. [Google Scholar] [CrossRef]
- Thompson, J.D.; Higgins, D.G.; Gibson, T.J. CLUSTAL W: Improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 1994, 22, 4673–4680. [Google Scholar] [CrossRef] [PubMed]
- Larkin, M.A.; Blackshields, G.; Brown, N.P.; Chenna, R.; McGettigan, P.A.; McWilliam, H.; Valentin, F.; Wallace, I.M.; Wilm, A.; Lopez, R.; et al. Clustal W and Clustal X version 2.0. Bioinformatics 2007, 23, 2947–2948. [Google Scholar] [CrossRef] [PubMed]
- Stamatakis, A. RAxML version 8. 2014: A tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 2014, 30, 1312–1313. [Google Scholar] [CrossRef] [PubMed]
- Felsenstein, J. Confidence limits on phylogenies: An approach using the bootstrap. Evolution 1985, 39, 783–791. [Google Scholar] [CrossRef] [PubMed]
- Huelsenbeck, J.P.; Ronquist, F. MRBAYES: Bayesian inference of phylogeny trees. Bioinformatics 2001, 17, 754–755. [Google Scholar] [CrossRef]
- Keats, D.W.; Chamberlain, Y.M. Sporolithon ptychoides Heydrich and S. episporum (Howe) Dawson: Two coralline red algae (Corallinales, Sporolithaceae). S. Afr. J. Bot. 1993, 59, 541–550. [Google Scholar] [CrossRef]
- Bahia, R.G. Algas Coralináceas Formadoras de Rodolitos da Plataforma Continental Tropical e Ilhas Oceânicas do Brasil: Levantamento Florístico e Taxonomia. Ph.D. Thesis, Escola Nacional de Botânica Tropical (ENBT), Rio de Janeiro, Brazil, 2014; p. 231. [Google Scholar]
- Chelaru, R.; Sãsãran, E.; Tãmas, T.; Bãlc, R.; Bucur, I.I.; Ples, G. Middle Miocene carbonate facies with rhodoliths from the NW Transylvanian Basin (Vălenii Șomcutei Cave, Romania). Facies 2019, 65, 1–16. [Google Scholar] [CrossRef]
- Agência Nacional do Petróleo. Available online: http://webmaps.anp.gov.br/mapas/Lists/DSPAppPages/MapasBrasil.aspx (accessed on 20 June 2019).
- EPBR. Available online: https://epbr.com.br/oferta-permanente-de-areas-de-exploracao-sobe-para-600-blocos/ (accessed on 20 June 2019).
- Figueiredo, M.A.O.; Coutinho, R.; Villas-Bôas, A.B.; Tâmega, F.T.S.; Mariath, R. Deep-water rhodolith productivity and growth in the southwestern Atlantic. J. Appl. Phycol. 2012, 24, 487–493. [Google Scholar] [CrossRef]
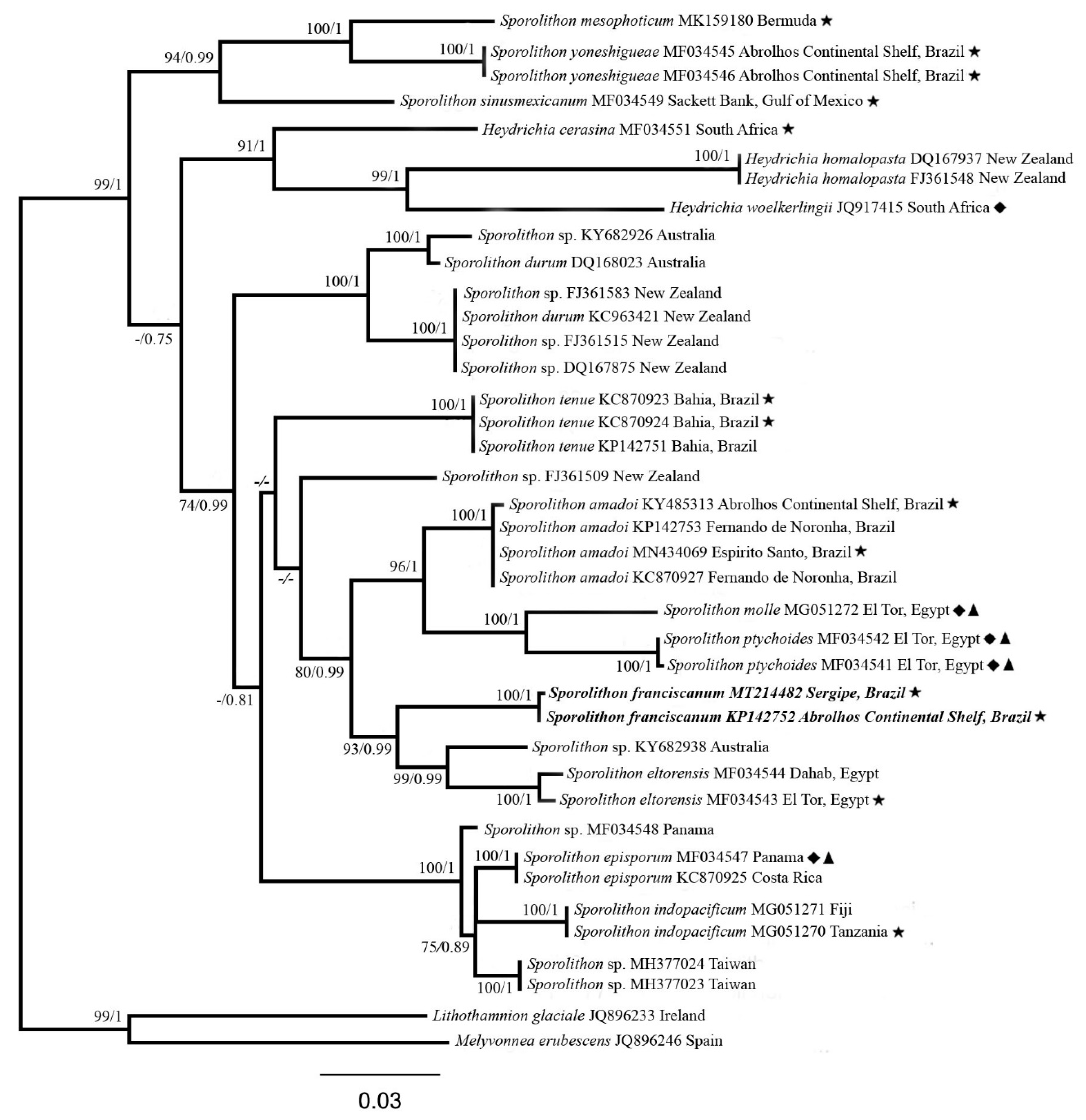
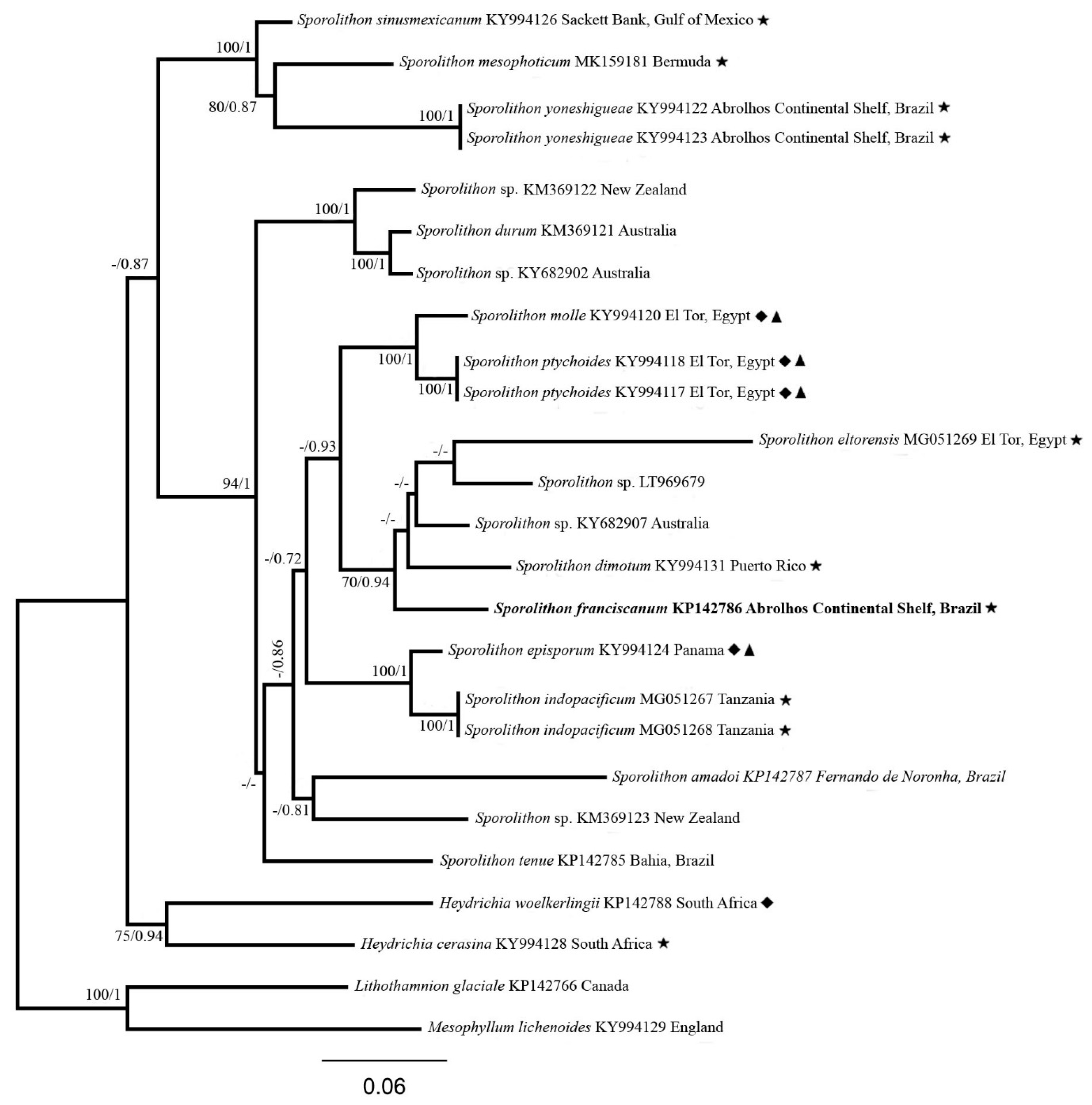

| Sporolithon Species | psbA | rbcL |
|---|---|---|
| S. franciscanum vs Sporolithon sp. (Australia) | 6.5% | 6% |
| S. franciscanum vs S. eltorensis | 6.1% | 12.3% |
| S. franciscanum vs S. dimotum | − | 6.8% |
| S. franciscanum vs Sporolithon sp. | − | 6.8% |
| References and Algal Characteristics | Species | ||||||
|---|---|---|---|---|---|---|---|
| S. episporum | S. franciscanum | S. mesophoticum | S. amadoi | S. sinuxmexicanum | S. tenue | S. yoneshigueae | |
| References | Keats & Chamberlain [80] | This study | Richards et al. [15] | Richards et al. [26] | Richards & Fredericq [24] | Bahia et al. [32] | Bahia et al. [33] |
| Locality | Point Toro, near Colon, Canal Zone (Panama) | Southwestern Atlantic (Aracaju, Sergipe, and Northeast region (Brazil) | Plantagenet (Argus) Bank (Southwest of Bermuda) | Recifes Esquecidos, (Espírito Santo, Brazil) | Sackett Bank, NWGMx (USA) | Southwestern Atlantic (Brazil) | Abrolhos continental shelf (Bahia, Brazil) |
| Growth form | Flat to lumpy | Encrusting | Encrusting | Encrusting to Warty to lumpy | Encrusting | Encrusting to warty to fruticose | Encrusting to warty to lumpy to fruticose |
| Habit | Epilithic or free living as rhodolith | Epilithic or free living as rhodolith | Epilithic, epizoic, or free living as rhodolith | Epilithic, epizoic, or free living as rhodolith | Free living as rhodolith | Free living as rhodolith | Free living as rhodolith |
| Relative abundance of secondary pit connections versus cell fusions (proportion) | Primarily by secondary pit connections; cell fusions rare | Cell fusions predominate by a ratio of 2:1 | Cell fusions (Secondary pit-connections not observed) | Secondary pit connections predominate by a ratio of 2:1; | Abundant cell fusions (secondary pit connections not observed) | Secondary pit connections predominate at a ratio of 2–3:1 | Cell fusions predominate by a ratio of 2–3:1 |
| Vegetative thallus thickness | ND | >250 μm (500–1000 μm) | 120–150 μm | ND (>20 cell layers) | >250 μm (at least 800 μm) ** | 45–250 μm (less than 20 cell layers) | >250 μm (1000–2500 μm) |
| Tetrasporangia length | up to 50 μm | 60–90 μm | ND | ND | ND | 50–75 μm | 90–140 μm |
| Tetrasporangia diameter | ca. 30 μm | 30–60 μm | ND | ND | ND | 30–40 μm | 80–100 μm |
| Tetrasporangial chamber length | 50–70 μm | 70–100 μm | ND | 65–108 μm | 83 μm | 70–95 μm | 140–210 μm |
| Tetrasporangial chamber diameter | 25–40 μm | 30–60 μm | ND | 41–64 μm | 59 μm | 35–55 μm | 70–130 μm |
| Tetrasporangial chamber pore diameter | up to 30 μm | 10–16 μm | 16–23 μm* | 8.5–16.5 μm | 16–22 μm ** | 9–14 μm | 35–43 μm |
| Number of rosette cells surrounding tetraporangial chamber pore | 12–16 *** | 11–13 | 10–14 | 9–13 | 10–12 | 8–12 | 19–24 |
| Layer of elongated cells at the base of tetrasporangial chamber | Present | Present | ND | Present | Absent | Absent | Present |
| Buried tetrasporangial chambers | Absent | Present | Absent | Present | Absent | Absent | Present |
| Position of tetrasporangial chambers relative to the surrounding vegetative surface (number of cells) | Slightly raised (3–4 cells) | Raised, 3–5 cells (incl. epithallial cell) | Flush to slightly raised (ND for number of cells layers) | Raised (ND for number of cells layers) | Slightly raised (ND for number of cells layers) ** | Raised (5–7 cells) | Raised 5–8 cells |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leão, L.A.S.; Bahia, R.G.; Jesionek, M.B.; Adey, W.H.; Johnson, G.; Salgado, L.T.; Pereira, R.C. Sporolithon franciscanum sp. nov. (Sporolithales, Rhodophyta), a New Rhodolith-Forming Species from Northeast Brazil. Diversity 2020, 12, 199. https://doi.org/10.3390/d12050199
Leão LAS, Bahia RG, Jesionek MB, Adey WH, Johnson G, Salgado LT, Pereira RC. Sporolithon franciscanum sp. nov. (Sporolithales, Rhodophyta), a New Rhodolith-Forming Species from Northeast Brazil. Diversity. 2020; 12(5):199. https://doi.org/10.3390/d12050199
Chicago/Turabian StyleLeão, Luis Alfredo S., Ricardo G. Bahia, Michel B. Jesionek, Walter H. Adey, Gabriel Johnson, Leonardo T. Salgado, and Renato C. Pereira. 2020. "Sporolithon franciscanum sp. nov. (Sporolithales, Rhodophyta), a New Rhodolith-Forming Species from Northeast Brazil" Diversity 12, no. 5: 199. https://doi.org/10.3390/d12050199
APA StyleLeão, L. A. S., Bahia, R. G., Jesionek, M. B., Adey, W. H., Johnson, G., Salgado, L. T., & Pereira, R. C. (2020). Sporolithon franciscanum sp. nov. (Sporolithales, Rhodophyta), a New Rhodolith-Forming Species from Northeast Brazil. Diversity, 12(5), 199. https://doi.org/10.3390/d12050199







