Sedimentary Organic Matter, Prokaryotes, and Meiofauna across a River-Lagoon-Sea Gradient
Abstract
1. Introduction
2. Materials and Methods
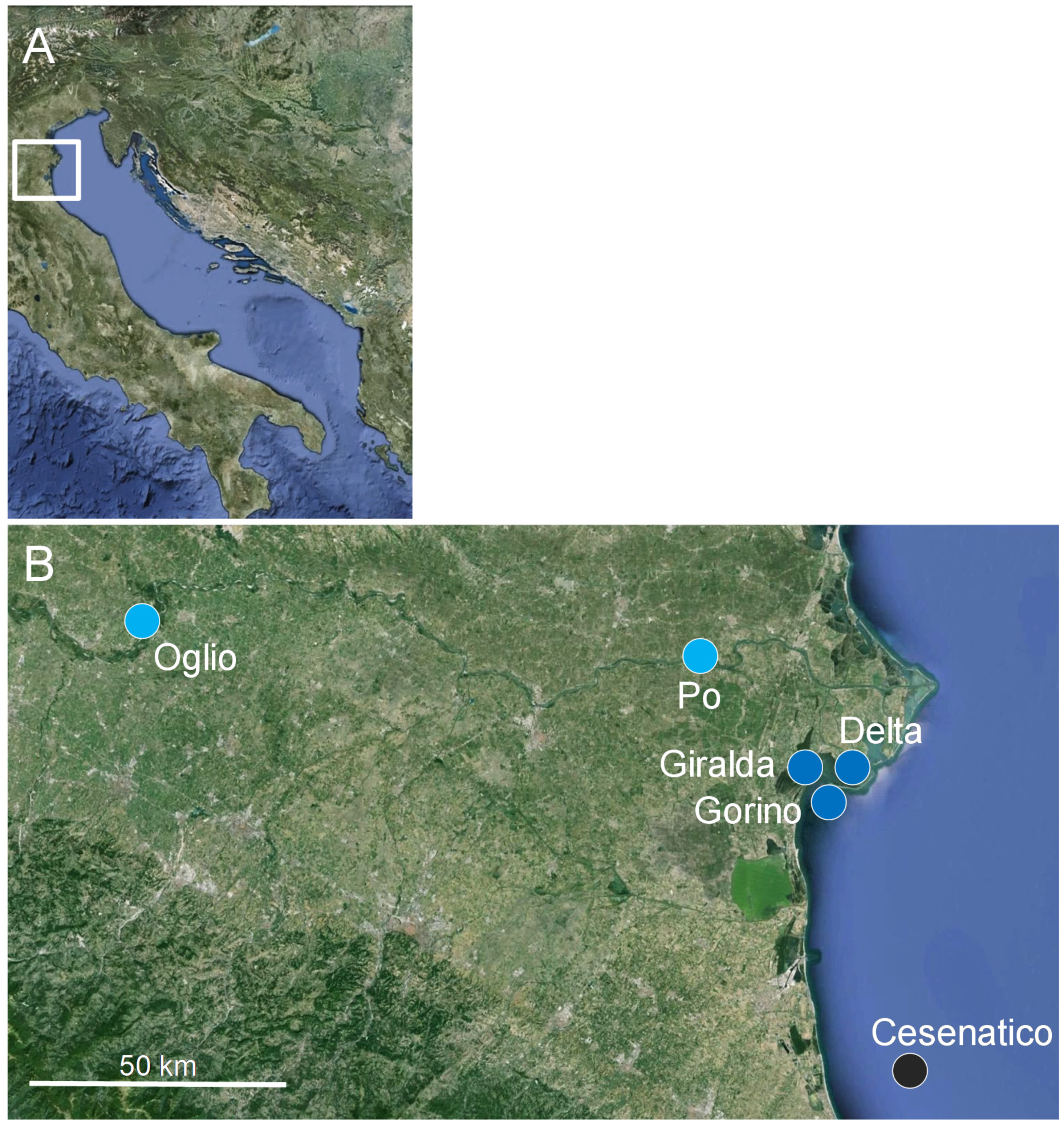
2.1. Study Area
2.2. Sampling
2.3. Sedimentary Organic Matter Quantity, Nutritional Quality, and Degradation Rates
2.4. Prokaryotic Abundance, Biomass, and Diversity
2.5. Meiofauna
2.6. Statistical Analyses
3. Results
3.1. Environmental Parameters
3.2. Content, Biochemical Composition, Nutritional Quality, and Degradation Rates of Organic Matter
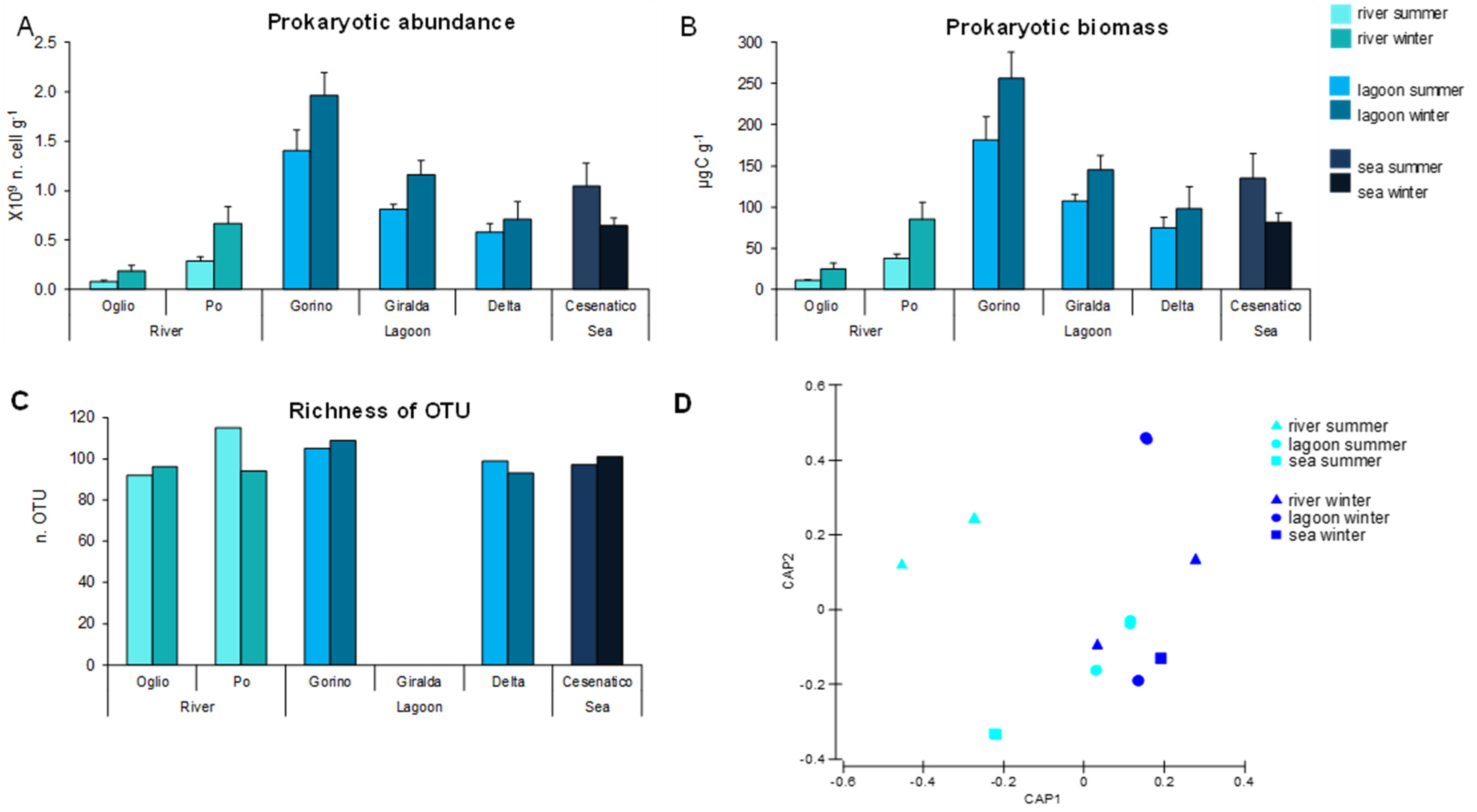
3.3. Prokaryotic Abundance, Biomass, and Diversity
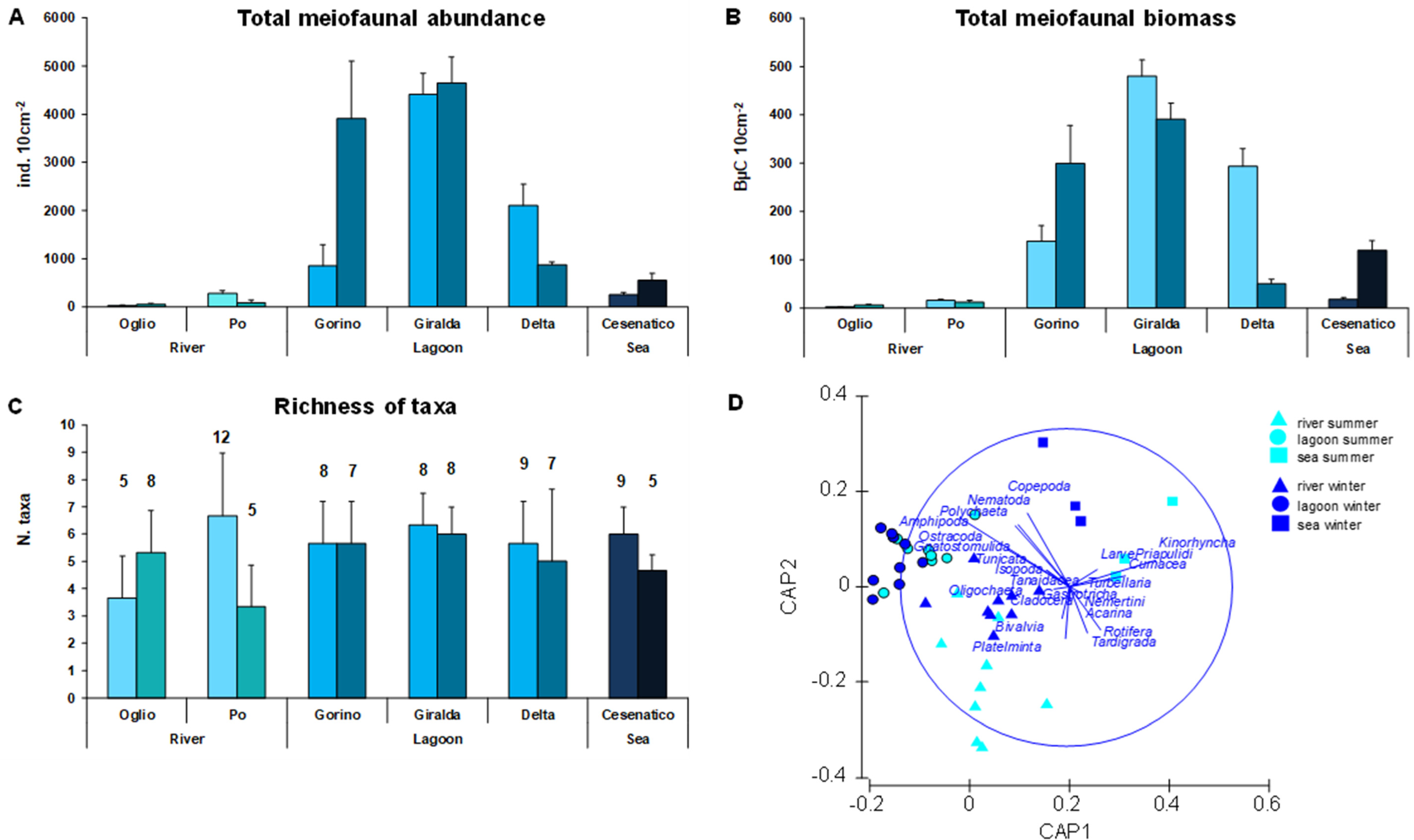
3.4. Meiofaunal Abundance, Richness of Taxa, and Community Structure
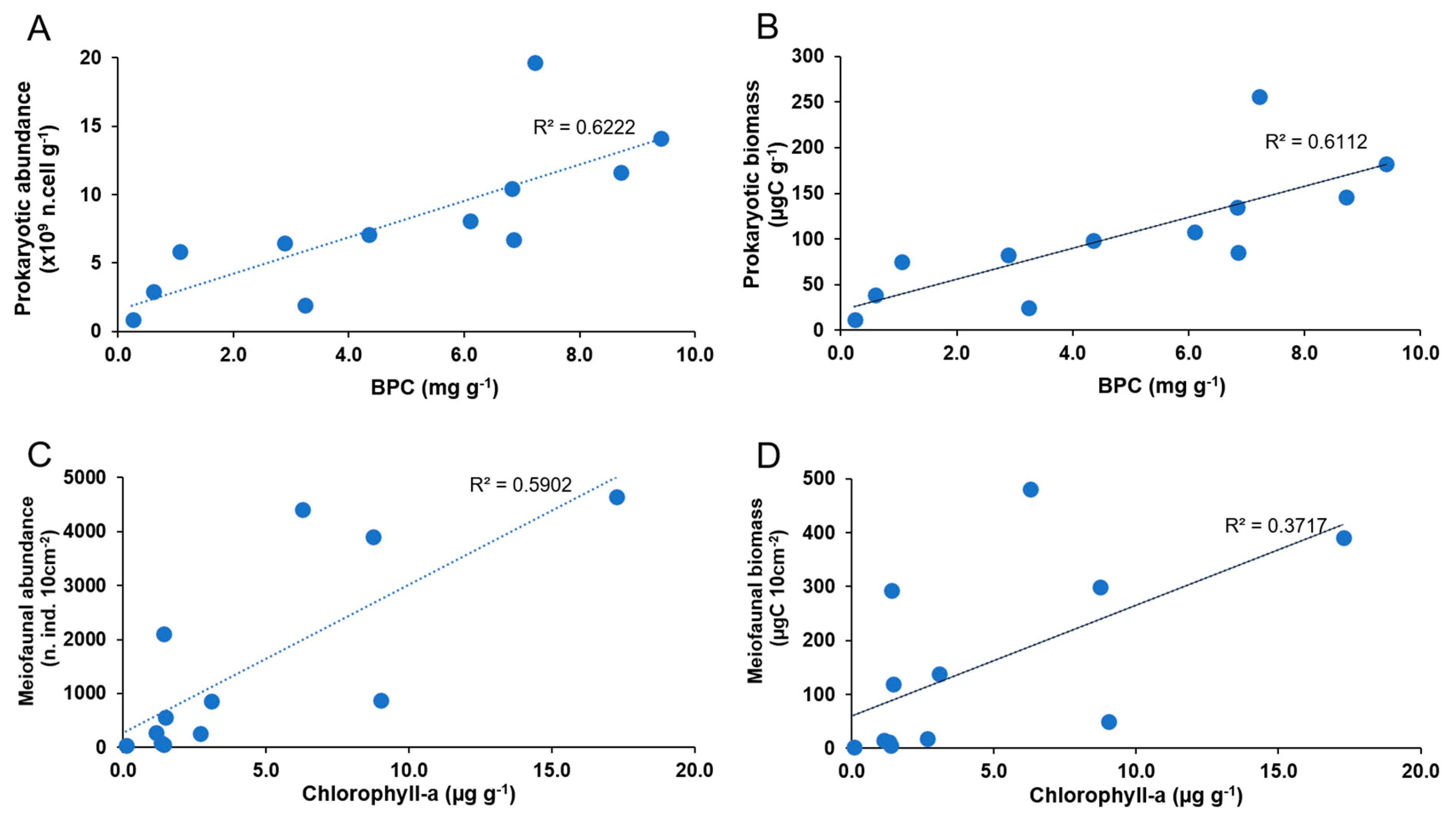
3.5. Relationships between Prokaryotes, Meiofauna, Organic Matter, and Environmental Characteristics
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cebrián, J.; Williams, M.; McClelland, J.; Valiela, I. The dependence of heterotrophic consumption and C accumulation on autotrophic nutrient concentration in ecosystems. Ecol. Lett. 1998, 1, 165–170. [Google Scholar] [CrossRef]
- Huxel, G.R. On the influence of food quality in consumer-resource interactions. Ecol. Lett. 1999, 2, 256–261. [Google Scholar] [CrossRef]
- Pusceddu, A.; Dell’Anno, A.; Fabiano, M.; Danovaro, R. Quantity, biochemical composition and bioavailability of sediment organic matter as complementary signatures of benthic trophic status. Mar. Ecol. Prog. Ser. 2009, 375, 41–52. [Google Scholar] [CrossRef]
- Nixon, S.W. Coastal marine eutrophication: A definition, social causes, and future concerns. Ophelia 1995, 41, 199–219. [Google Scholar] [CrossRef]
- Grall, J.; Chauvaud, L. Marine eutrophication and benthos: The need for new approaches and concepts. Global Chang. Biol. 2002, 8, 813–830. [Google Scholar] [CrossRef]
- Torres, A.I.; Rivera Hernández, J.R.; Giarratano, E.; Faleschini, M.; Green Ruiz, C.R.; Gil, M.N. Potentially toxic elements and biochemical components in surface sediments of NW Mexico: An assessment of contamination and trophic status. Mar. Pollut. Bull. 2019, 149, 110633. [Google Scholar] [CrossRef]
- Cloern, J.E. Our evolving conceptual model of the coastal eutrophication problem. Mar. Ecol. Prog. Ser. 2001, 210, 223–253. [Google Scholar] [CrossRef]
- Fabiano, M.; Danovaro, R.; Fraschetti, S. A three-year time series of elemental and biochemical composition of organic matter in subtidal sandy sediments of the Ligurian Sea (northwestern Mediterranean). Cont. Shelf Res. 1995, 15, 1453–1469. [Google Scholar] [CrossRef]
- Dell’Anno, A.; Pusceddu, A.; Langone, L.; Danovaro, R. Biochemical composition and early diagenesis of organic matter in coastal sediments of the NW Adriatic Sea influenced by riverine inputs. Chem. Ecol. 2008, 24, 75–85. [Google Scholar] [CrossRef]
- Welsh, D.T. It’s a dirty job but someone has to do it: The role of marine benthic macrofauna in organic matter turnover and nutrient recycling to the water column. Chem. Ecol. 2003, 19, 321–342. [Google Scholar] [CrossRef]
- Danovaro, R.; Gambi, C.; Manini, E.; Fabiano, M. Meiofauna response to a dynamic river plume front. Mar. Biol. 2000, 137, 359–370. [Google Scholar] [CrossRef]
- Pusceddu, A.; Gambi, C.; Manini, E.; Danovaro, R. Trophic state, ecosystem efficiency and biodiversity of transitional aquatic ecosystems: Analysis of environmental quality based on different benthic indicators. Chem. Ecol. 2007, 23, 505–515. [Google Scholar] [CrossRef]
- Pusceddu, A.; Danovaro, R. Exergy, ecosystem functioning and efficiency in a coastal lagoon: The role of auxiliary energy. Estuar. Coast. Shelf Sci. 2009, 84, 227–236. [Google Scholar] [CrossRef]
- Magri, M.; Benelli, S.; Bondavalli, C.; Bartoli, M.; Christian, R.R.; Bodini, A. Benthic N pathways in illuminated and bioturbated sediments studied with network analysis. Limnol. Oceanogr. 2018, 63, S68–S84. [Google Scholar] [CrossRef]
- Politi, T.; Zilius, M.; Castaldelli, G.; Bartoli, M.; Daunys, D. Estuarine macrofauna affects benthic biogeochemistry in a hypertrophic lagoon. Water 2019, 11, 1186. [Google Scholar] [CrossRef]
- Pusceddu, A.; Bianchelli, S.; Gambi, C.; Danovaro, R. Assessment of benthic trophic status of marine coastal ecosystems: Significance of meiofaunal rare taxa. Estuar. Coast. Shelf Sci. 2011, 93, 420–430. [Google Scholar] [CrossRef]
- Foti, A.; Fenzi, G.A.; Di Pippo, F.; Gravina, M.F.; Magni, P. Testing the saprobity hypothesis in a Mediterranean lagoon: Effects of confinement and organic enrichment on benthic communities. Mar. Environ. Res. 2014, 99, 85–94. [Google Scholar] [CrossRef]
- Zobrist, E.C.; Coull, B.C. Meiobenthic interactions with microbenthic larvae and juveniles: An experimental assessment of the meiofaunal bottleneck. Mar. Ecol. Prog. Ser. 1992, 88, 1–8. [Google Scholar] [CrossRef]
- Danovaro, R.; Pusceddu, A. Biodiversity and ecosystem functioning in coastal lagoons: Does microbial diversity play any role? Estuar. Coast. Shelf Sci. 2007, 75, 4–12. [Google Scholar] [CrossRef]
- Cibic, T.; Franzo, A.; Celussi, M.; Fabbro, C.; Del Negro, P. Benthic ecosystem functioning in hydrocarbon and heavy-metal contaminated sediments of an Adriatic lagoon. Mar. Ecol. Prog. Ser. 2012, 458, 69–87. [Google Scholar] [CrossRef]
- Heip, C.; Vincx, M.; Vranken, G. The ecology of marine nematodes. Oceanogr. Mar. Biol. Ann. Rev. 1985, 23, 399–489. [Google Scholar]
- Semprucci, F.; Losi, V.; Moreno, M. A review of Italian research on free-living marine nematodes and the future perspectives on their use as Ecological Indicators (EcoInds). Mediterr. Mar. Sci. 2015, 16, 352–365. [Google Scholar] [CrossRef]
- Magliozzi, C.; Usseglio-Polatera, P.; Meyer, A.; Grabowski, R.C. Functional traits of hyporheic and benthic invertebrates reveal importance of wood-driven geomorphological processes in rivers. Funct. Ecol. 2019, 33, 1758–1770. [Google Scholar] [CrossRef]
- Tedesco, L.; Socal, G.; Acri, F.; Veneri, D.; Vichi, M. NW Adriatic Sea biogeochemical variability in the last 20 years (1986-2005). Biogeosciences 2007, 4, 673–687. [Google Scholar] [CrossRef]
- Wang, X.H.; Pinardi, N. Modelling the dynamics of sediment transport and resuspension in the northern Adriatic Sea. J. Geophys. Res. Atmos. 2002, 107, 3225. [Google Scholar] [CrossRef]
- Ursella, L.; Poulain, P.M.; Signell, R.P. Surface drifter derived circulation in the northern and middle Adriatic Sea: Response to wind regime and season. J. Geophys. Res. Oceans 2006, 112, C03S04. [Google Scholar] [CrossRef]
- Bartoli, M.; Nizzoli, D.; Viaroli, P.; Turolla, E.; Castaldelli, G.; Fano, E.A.; Rossi, R. Impact of Tapes philippinarum farming on nutrient dynamics and benthic respiration in the Sacca di Goro. Hydrobiologia 2001, 455, 203–212. [Google Scholar] [CrossRef]
- Viaroli, P.; Azzoni, R.; Bartoli, M.; Giordani, G.; Tajé, L. Evolution of the trophic conditions and dystrophic outbreaks in the Sacca di Goro lagoon (Northern Adriatic Sea). In Mediterranean Ecosystems; Springer: Milano, Italy, 2001; pp. 467–475. [Google Scholar]
- Viaroli, P.; Bartoli, M.; Azzoni, R.; Giordani, G.; Mucchino, C.; Naldi, M.; Tajé, L. Nutrient and iron limitation to Ulva blooms in a eutrophic coastal lagoon (Sacca di Goro, Italy). Hydrobiologia 2005, 550, 57–71. [Google Scholar] [CrossRef]
- Nizzoli, D.; Bartoli, M.; Viaroli, P. Nitrogen and phosphorous budgets during a farming cycle of the Manila clam Ruditapes philippinarum: An in situ experiment. Aquaculture 2006, 261, 98–108. [Google Scholar] [CrossRef]
- Zilius, M.; Giordani, G.; Petkuviene, J.; Lubiene, I.; Ruginis, T.; Bartoli, M. Phosphorus mobility under short-term anoxic conditions in two shallow eutrophic coastal systems (Curonian and Sacca di Goro lagoons). Estuar. Coast. Shelf Sci. 2016, 164, 134–146. [Google Scholar] [CrossRef]
- Naldi, M.; Viaroli, P. Nitrate uptake and storage in the seaweed Ulva rigida C. Agardh in relation to nitrate availability and thallus nitrate content in a eutrophic coastal lagoon (Sacca di Goro, Po River Delta, Italy). J. Exp. Mar. Biol. Ecol. 2002, 269, 65–83. [Google Scholar] [CrossRef]
- Fiordelmondo, C.; Manini, E.; Gambi, C.; Pusceddu, A. Short-term impact of clam harvesting on sediment chemistry, benthic microbes and meiofauna in the Goro lagoon (Italy). Chem. Ecol. 2003, 19, 173–187. [Google Scholar] [CrossRef]
- Mistri, M.; Rossi, R.; Fano, E.A. Structure and secondary production of a soft bottom macrobenthic community in a brackish lagoon (Sacca di Goro, north-eastern Italy). Estuar. Coast. Shelf Sci. 2001, 52, 605–616. [Google Scholar] [CrossRef]
- Ludovisi, A.; Castaldelli, G.; Fano, E.A. Multi-scale spatio-temporal patchiness of macrozoobenthos in the Sacca di Goro lagoon (Po river delta, Italy). Transit. Waters Bull. 2013, 7, 233–244. [Google Scholar] [CrossRef]
- Marinov, D.; Zaldívar, J.M.; Norro, A.; Giordani, G.; Viaroli, P. Integrated modelling in coastal lagoons: Sacca di Goro case study. Hydrobiologia 2008, 611, 147–165. [Google Scholar] [CrossRef]
- Bartoli, M.; Castaldelli, G.; Nizzoli, D.; Viaroli, P. Benthic primary production and bacterial denitrification in a Mediterranean eutrophic coastal lagoon. J. Exp. Mar. Biol. Ecol. 2012, 438, 41–51. [Google Scholar] [CrossRef]
- Dell’Anno, A.; Incera, M.; Mei, M.L.; Pusceddu, A. Mesoscale variability of organic matter composition in NW Adriatic sediments. Chem. Ecol. 2003, 19, 33–45. [Google Scholar] [CrossRef]
- Tesi, T.; Miserocchi, S.; Langone, L.; Boni, L.; Guerrini, F. Sources, fate and distribution of organic matter on the Western Adriatic Continental Shelf, Italy. Water Air Soil Pollut. Focus 2006, 6, 593–603. [Google Scholar] [CrossRef]
- Tesi, T.; Langone, L.; Goni, M.A.; Miserocchi, S.; Bertasi, F. Changes in the composition of organic matter from prodeltaic sediments after a large flood event (Po River, Italy). Geochim. Cosmochim. Acta 2008, 72, 2100–2114. [Google Scholar] [CrossRef]
- Bianchelli, S.; Pusceddu, A.; Buschi, E.; Danovaro, R. Trophic status and meiofauna biodiversity in the Northern Adriatic Sea: Insights for the assessment of good environmental status. Mar. Environ. Res. 2016, 113, 18–30. [Google Scholar] [CrossRef]
- Bianchelli, S.; Pusceddu, A.; Buschi, E.; Danovaro, R. Nematode biodiversity and benthic trophic state are simple tools for the assessment of the environmental quality in coastal marine ecosystems. Ecol. Indic. 2018, 95, 270–287. [Google Scholar] [CrossRef]
- Tahey, T.M.; Duineveld, G.C.A.; de Wilde, P.A.W.J.; Berghuis, E.M.; Kok, A. Sediment O2 demand, density and biomass of the benthos and phytopigments along the northwestern Adriatic coast: The extent of Po enrichment. Oceanol. Acta 1996, 19, 117–130. [Google Scholar]
- Moodley, L.; Heip, C.H.R.; Middelburg, J.J. Benthic activity in sediments of the northwestern Adriatic Sea: Sediment oxygen consumption, macro- and meiofauna dynamics. J. Sea Res. 1998, 40, 263–280. [Google Scholar] [CrossRef]
- Alvisi, F.; Giani, M.; Ravaioli, M.; Giordano, P. Role of sedimentary environment in the development of hypoxia and anoxia in the NW Adriatic shelf (Italy). Estuar. Coast. Shelf Sci. 2013, 128, 9–21. [Google Scholar] [CrossRef]
- Danovaro, R. Organic inputs and ecosystem efficiency in the deep Mediterranean Sea. Chem. Ecol. 2003, 19, 391–398. [Google Scholar] [CrossRef]
- Danovaro, R.; Fonda Umani, S.; Pusceddu, A. Climate change and the potential spreading of marine mucilage and microbial pathogens in the Mediterranean Sea. PLoS ONE 2009, 4, e7006. [Google Scholar] [CrossRef] [PubMed]
- Lorenzen, C.J.; Jeffrey, S.W. Determination of chlorophyll and phaeopigments spectrophotometric equations. Limnol. Oceanogr. 1980, 12, 343–346. [Google Scholar] [CrossRef]
- Danovaro, R. Methods for the Study of Deep-Sea Sediments, Their Functioning and Biodiversity; CRC Press, Taylor & Francis Group: Boca Raton, FL, USA, 2010; pp. 1–428. [Google Scholar]
- Pusceddu, A.; Dell’Anno, A.; Fabiano, M. Organic matter composition in coastal sediments at Terra Nova Bay (Ross Sea) during summer 1995. Polar Biol. 2000, 23, 288–293. [Google Scholar] [CrossRef]
- Pusceddu, A.; Bianchelli, S.; Canals, M.; Sanchez-Vidal, A.; De Madron, X.D.; Heussner, S.; Lykousis, V.; de Stigter, H.; Trincardi, F.; Danovaro, R. Organic matter in sediments of canyons and open slopes of the Portuguese, Catalan, Southern Adriatic and Cretan Sea margins. Deep Sea Res. I Oceanogr. Res. Pap. 2010, 57, 441–457. [Google Scholar] [CrossRef]
- Luna, G.M.; Corinaldesi, C.; Rastelli, E.; Danovaro, R. Patterns and drivers of bacterial α-and β-diversity across vertical profiles from surface to subsurface sediments. Environ. Microbiol. Rep. 2013, 201, 731–739. [Google Scholar] [CrossRef]
- Fuhrman, J.A.; Cram, J.A.; Needham, D.M. Marine microbial community dynamics and their ecological interpretation. Nat. Rev. Microbiol. 2015, 13, 133–146. [Google Scholar] [CrossRef] [PubMed]
- Rastelli, E.; Corinaldesi, C.; Dell’Anno, A.; Tangherlini, M.; Lo Martire, M.; Nishizawa, M.; Nomaki, H.; Nunoura, T.; Danovaro, R. Drivers of bacterial α-and β-diversity and functioning in subsurface hadal sediments. Front. Microbiol. 2019, 10, 2609. [Google Scholar] [CrossRef] [PubMed]
- Niederberger, T.; Bottos, E.M.; Sohm, J.A.; Gunderson, T.E.; Parker, A.E.; Coyne, K.J.; Capone, D.G.; Carpenter, E.J.; Cary, S.C. Rapid microbial dynamics in response to an induced wetting event in antarctic dry valley soils. Front. Microbiol. 2019, 10, 621. [Google Scholar] [CrossRef] [PubMed]
- Andrassy, I. The determination of volume and weight of nematodes. Acta Zool. 1956, 2, 1–15. [Google Scholar]
- Feller, R.J.; Warwick, R.M. Energetics. In Introduction to the Study of Meiofauna; Higgins, R.P., Thiel, H., Eds.; Smithsonian Institute Press: London, UK, 1988; pp. 181–196. [Google Scholar]
- Wieser, W. Benthic studies in Buzzards Bay. II The meiofauna. Limnol. Oceanogr. 1960, 5, 121–137. [Google Scholar] [CrossRef]
- Anderson, M.J. Permutation tests for univariate or multivariate analysis of variance and regression. Can. J. Fish. Aquat. Sci. 2001, 58, 626–639. [Google Scholar] [CrossRef]
- McArdle, B.H.; Anderson, M.J. Fitting multivariate models to community data: A comment on distance-based redundancy analysis. Ecology 2001, 82, 290–297. [Google Scholar] [CrossRef]
- Anderson, M.J.; Willis, T.J. Canonical analysis of principal coordinates: A useful method of constrained ordination for ecology. Ecology 2003, 84, 511–525. [Google Scholar] [CrossRef]
- Clarke, K.R.; Gorley, R.N. PRIMER v6: User Manual/Tutorial. PRIMER-E, Plymouth, 2006, p. 190. Available online: https://www.primer-e.com/ (accessed on 8 May 2020).
- Viaroli, P.; Nizzoli, D.; Pinardi, M.; Soana, E.; Bartoli, M. Eutrophication of the Mediterranean Sea: A watershed—Cascading aquatic filter approach. Rend. Fis. Acc. Lincei 2015, 26, 13–23. [Google Scholar] [CrossRef]
- Zamparas, M.; Zacharias, I. Restoration of eutrophic freshwater by managing internal nutrient loads. A review. Sci. Total Environ. 2014, 496, 551–562. [Google Scholar] [CrossRef]
- Volf, G.; Atanasova, N.; Kompare, B.; Ožanić, N. Modeling nutrient loads to the northern Adriatic. J. Hydrol. 2013, 504, 182–193. [Google Scholar] [CrossRef]
- Degobbis, D.; Precali, R.; Ivančić, I.; Smodlaka, N.; Fuks, D.; Kveder, S. Long-term changes in the northern Adriatic ecosystem related to anthropogenic eutrophication. Int. J. Environ. Pollut. 2000, 13, 495–533. [Google Scholar] [CrossRef]
- Alcamo, J.; Moreno, J.M.; Nováky, B.; Bindi, M.; Corobov, R.; Devoy, R.J.N.; Giannakopoulos, C.; Martin, E.; Olesen, J.E.; Shvidenko, A. Impacts, Adaptation and Vulnerability. Contribution of Working Group II to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change. In Climate Change 2007; Parry, M.L., Canziani, O.F., Palutikof, J.P., van der Linden, P.J., Hanson, C.E., Eds.; Cambridge University Press: Cambridge, UK, 2007; pp. 541–580. [Google Scholar]
- Mozetič, P.; Solidoro, C.; Cossarini, G.; Socal, G.; Precali, R.; Francé, J.; Bianchi, F.; De Vittor, C.; Smodlaka, N.; Fonda Umani, S. Recent trends towards oligotrophication of the Northern Adriatic: Evidence from chlorophyll a time series. Estuar. Coasts 2010, 33, 362–375. [Google Scholar] [CrossRef]
- Cozzi, S.; Giani, M. River water and nutrient discharges in the Northern Adriatic Sea: Current importance and long-term changes. Cont. Shelf Res. 2011, 31, 1881–1893. [Google Scholar] [CrossRef]
- Dell’Anno, A.; Mei, M.L.; Pusceddu, A.; Danovaro, R. Assessing the trophic state and eutrophication of coastal marine systems: A new approach based on the biochemical composition of sediment organic matter. Mar. Pollut. Bull. 2002, 44, 611–622. [Google Scholar] [CrossRef]
- Sabbatini, A.; Bonatto, S.; Bianchelli, S.; Pusceddu, A.; Danovaro, R.; Negri, A. Foraminiferal assemblages and trophic state in coastal sediments of the Adriatic Sea. J. Mar. Syst. 2012, 105, 163–174. [Google Scholar] [CrossRef]
- Conversi, A.; Fonda Umani, S.; Peluso, T.; Molinero, J.C.; Santojanni, A.; Edwards, M. The mediterranean sea regime shift at the end of the 1980s, and intriguing parallelisms with other european basins. PLoS ONE 2010, 5, e10633. [Google Scholar] [CrossRef]
- Gašparović, B. Decreased production of surface-active organic substances as a consequence of the oligotrophication in the northern Adriatic Sea. Estuar. Coast. Shelf Sci. 2012, 115, 33–39. [Google Scholar] [CrossRef]
- Giani, M.; Djakovac, T.; Degobbis, D.; Cozzi, S.; Solidoro, C.; Fonda Umani, S. Recent changes in the marine ecosystems of the northern Adriatic Sea. Estuar. Coast. Shelf Sci. 2012, 115, 1–13. [Google Scholar] [CrossRef]
- Kumar, K.S.S.; Nair, S.M.; Salas, P.M.; Cheriyan, E. Distribution and sources of sedimentary organic matter in Chitrapuzha, a tropical tidal river, southwest coast of India. Environ. Forensics 2017, 18, 135–146. [Google Scholar] [CrossRef]
- Delconte, C.A.; Sacchi, E.; Racchetti, E.; Bartoli, M.; Mas-Pla, J.; Re, V. Nitrogen inputs to a river course in a heavily impacted watershed: A combined hydrochemical and isotopic evaluation (Oglio River Basin, N Italy). Sci. Total Environ. 2014, 466–467, 924–938. [Google Scholar] [CrossRef]
- McLusky, D.S.; Elliott, M. Transitional waters: A new approach, semantics or just muddying the waters? Estuar. Coast. Shelf Sci. 2007, 71, 359–363. [Google Scholar] [CrossRef]
- Pusceddu, A.; Dell’Anno, A.; Danovaro, R.; Manini, E.; Sarà, G.; Fabiano, M. Enzymatically hydrolyzable protein and carbohydrate sedimentary pools as indicators of the trophic state of detritus sink systems: A case study in a Mediterranean coastal lagoon. Estuaries 2003, 26, 641–650. [Google Scholar] [CrossRef]
- Viaroli, P.; Christian, R.R. Description of trophic status, hyperautotrophy and dystrophy of a coastal lagoon through a potential oxygen production and consumption index—TOSI: Trophic Oxygen Status Index. Ecol. Indic. 2004, 3, 237–250. [Google Scholar] [CrossRef]
- Duarte, C.M.; Cebrian, J. The fate of marine autotrophic production. Limnol. Oceanogr. 1996, 41, 1758–1766. [Google Scholar] [CrossRef]
- Fazi, S.; Baldassarre, L.; Cassin, D.; Quero, G.M.; Pizzetti, I.; Cibic, T.; Luna, G.M.; Zonta, R.; Del Negro, P. Prokaryotic community composition and distribution in coastal sediments following a Po river flood event (northern Adriatic Sea, Italy). Estuar. Coast. Shelf Sci. 2020, 233, 106547. [Google Scholar] [CrossRef]
- Gambi, C.; Bianchelli, S.; Pérez, M.; Invers, O.; Ruiz, J.M.; Danovaro, R. Biodiversity response to experimental induced hypoxic-anoxic conditions in seagrass sediments. Biodivers. Conserv. 2009, 18, 33–54. [Google Scholar] [CrossRef]
- Mirto, S.; Bianchelli, S.; Gambi, C.; Krzelj, M.; Pusceddu, A.; Scopa, M.; Holmer, M.; Danovaro, R. Fish-farm impact on metazoan meiofauna in the Mediterranean Sea: Analysis of regional vs. habitat effects. Mar. Environ. Res. 2010, 69, 38–47. [Google Scholar] [CrossRef]
- Bianchelli, S.; Buschi, E.; Danovaro, R.; Pusceddu, A. Biodiversity loss and turnover in alternative states in the Mediterranean Sea: A case study on meiofauna. Sci. Rep. 2016, 6, 34544. [Google Scholar] [CrossRef]

| (A) | Temperature | Salinity | Chlorophyll-a | Phaeopigment | Total Pigment | Protein | Carbohydrate | Lipid | Biopolymeric C | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sampling season | Sampling station | °C | PSU | μg g−1 | μg g−1 | μg g−1 | mg g−1 | mg g−1 | mg g−1 | mg g−1 | ||||||||
| avg | sd | avg | sd | avg | sd | avg | sd | avg | sd | avg | sd | avg | sd | |||||
| Summer | River | Oglio | 25 | 0 | 0.08 | 0.00 | 0.30 | 0.02 | 0.35 | 0.05 | 0.30 | 0.02 | 0.17 | 0.00 | 0.03 | 0.01 | 0.24 | 0.02 |
| River | Po | 23 | 0 | 1.12 | 0.09 | 4.00 | 0.53 | 5.12 | 0.68 | 0.71 | 0.08 | 0.38 | 0.10 | 0.14 | 0.05 | 0.60 | 0.12 | |
| Lagoon | Gorino | 23 | 28 | 3.07 | 0.59 | 68.59 | 19.01 | 71.66 | 19.99 | 14.19 | 1.60 | 3.32 | 0.20 | 1.51 | 0.09 | 9.41 | 0.93 | |
| Lagoon | Giralda | 23 | 5 | 6.29 | 1.03 | 91.37 | 9.93 | 98.66 | 10.50 | 8.18 | 0.92 | 4.05 | 0.09 | 0.64 | 0.06 | 6.10 | 0.53 | |
| Lagoon | Delta | 23 | 28 | 1.40 | 0.08 | 14.03 | 0.74 | 15.43 | 0.88 | 1.48 | 0.14 | 0.44 | 0.02 | 0.19 | 0.02 | 1.05 | 0.09 | |
| Sea | Cesenatico | 17 | 35 | 2.67 | 1.00 | 36.45 | 10.31 | 39.12 | 11.31 | 10.50 | 0.47 | 2.55 | 0.33 | 0.88 | 0.04 | 6.83 | 0.39 | |
| Winter | River | Oglio | 8 | 0 | 1.38 | 0.00 | 19.35 | 6.24 | 20.72 | 3.61 | 2.45 | 0.23 | 3.71 | 0.99 | 0.74 | 0.19 | 3.24 | 0.65 |
| River | Po | 10 | 0 | 1.31 | 0.15 | 8.73 | 0.17 | 10.04 | 0.02 | 5.80 | 0.11 | 7.44 | 0.27 | 1.37 | 0.31 | 6.85 | 0.39 | |
| Lagoon | Gorino | 8 | 28 | 8.74 | 1.58 | 125.33 | 3.10 | 134.07 | 4.57 | 4.60 | 0.27 | 7.43 | 0.60 | 2.66 | 0.31 | 7.22 | 0.61 | |
| Lagoon | Giralda | 8 | 9 | 17.28 | 0.55 | 93.74 | 6.60 | 111.03 | 7.15 | 10.34 | 0.25 | 6.03 | 0.68 | 1.65 | 0.13 | 8.72 | 0.49 | |
| Lagoon | Delta | 13 | 28 | 9.02 | 2.36 | 38.95 | 10.21 | 47.97 | 12.57 | 4.82 | 0.82 | 3.26 | 0.37 | 0.91 | 0.23 | 4.35 | 0.72 | |
| Sea | Cesenatico | 10 | 35 | 1.46 | 0.15 | 16.03 | 1.46 | 17.00 | 0.77 | 5.02 | 0.62 | 0.49 | 0.02 | 0.32 | 0.06 | 2.89 | 0.35 | |
| (B) | Phytopigment: Biopolymeric C | Protein: Biopolymeric C | Protein: Carbohydrate | Aminopeptidase | β-Glucosidase | Alkaline-Phosphatase | ||||||||||||
| Sampling season | Sampling station | % | % | μg C g−1 h−1 | μg C g−1 h−1 | μg P g−1 h−1 | ||||||||||||
| avg | sd | avg | sd | avg | sd | avg | sd | avg | sd | avg | sd | |||||||
| Summer | River | Oglio | 1.32 | 0.03 | 61.30 | 0.78 | 1.25 | 0.02 | 14.20 | 4.38 | 0.49 | 0.24 | 0.67 | 0.25 | ||||
| River | Po | 7.56 | 0.91 | 58.12 | 4.69 | 1.19 | 0.10 | 20.48 | 4.81 | 1.76 | 0.12 | 4.82 | 0.91 | |||||
| Lagoon | Gorino | 1.30 | 0.12 | 73.79 | 1.04 | 1.51 | 0.02 | 13.21 | 5.06 | 0.90 | 0.19 | 3.23 | 0.09 | |||||
| Lagoon | Giralda | 4.10 | 0.33 | 65.56 | 1.72 | 1.34 | 0.04 | 29.82 | 5.11 | 3.39 | 1.27 | 6.53 | 3.70 | |||||
| Lagoon | Delta | 5.37 | 0.15 | 69.35 | 0.60 | 1.42 | 0.01 | 8.59 | 2.39 | 0.68 | 0.19 | 0.92 | 0.31 | |||||
| Sea | Cesenatico | 1.55 | 0.50 | 75.41 | 0.95 | 1.54 | 0.02 | 14.40 | 3.47 | 2.01 | 0.13 | 5.30 | 1.29 | |||||
| Winter | River | Oglio | 1.75 | 0.35 | 37.67 | 4.19 | 0.77 | 0.09 | 16.60 | 10.94 | 3.56 | 1.14 | 1.70 | 0.62 | ||||
| River | Po | 0.76 | 0.04 | 41.55 | 1.57 | 0.85 | 0.03 | 3.51 | 0.83 | 3.85 | 0.77 | 1.46 | 0.46 | |||||
| Lagoon | Gorino | 4.82 | 0.47 | 31.25 | 0.78 | 0.64 | 0.02 | 23.31 | 16.39 | 1.55 | 0.52 | 3.31 | 0.39 | |||||
| Lagoon | Giralda | 7.94 | 0.20 | 58.20 | 1.87 | 1.19 | 0.04 | 24.91 | 2.44 | 3.04 | 0.16 | 5.23 | 1.37 | |||||
| Lagoon | Delta | 8.20 | 0.83 | 54.30 | 0.20 | 1.11 | 0.004 | 20.75 | 9.63 | 4.07 | 1.25 | 2.01 | 0.31 | |||||
| Sea | Cesenatico | 2.02 | 0.04 | 84.91 | 0.11 | 1.73 | 0.002 | 8.47 | 1.49 | 1.02 | 0.24 | 1.98 | 0.26 | |||||
| % Dissimilarity | Responsible Taxa | Presence/Absence % Dissimilarity | Responsible Taxa | ||
|---|---|---|---|---|---|
| In summer | river vs. lagoon | 75.3 | Nematoda | 41.3 | Amphipoda, Rotifera, Ostracoda, Oligochaeta, Tardigrada, Acarina, Polychaeta, Nemertini, Bivalvia, Platelminta |
| lagoon vs. sea | 76.1 | Nematoda | 36.2 | Kinorhyncha, Ostracoda, Rotifera, Priapulida larvae, Polychaeta, Acarina, Cumacea, Oligochaeta, Amphipoda, Nemertea | |
| river vs. sea | 50.0 | Nematoda, Copepoda | 49.4 | Kinorhyncha, Amphipoda, Rotifera, Ostracoda, Polychaeta, Oligochaeta, Tardigrada, Priapulida larvae, Cumacea, Bivalvia | |
| In winter | river vs. lagoon | 80.0 | Nematoda | 41.8 | Amphipoda, Rotifera, Ostracoda, Tardigrada, Polychaeta, Oligochaeta, Acarina, Copepoda |
| lagoon vs. sea | 63.7 | Nematoda | 22.0 | Oligochaeta, Ostracoda, Tardigrada, Acarina, Rotifera, Polychaeta, Amphipoda | |
| river vs. sea | 65.3 | Nematoda, Copepoda | 40.7 | Amphipoda, Rotifera, Oligochaeta, Tardigrada, Polychaeta, Acarina, Copepoda, | |
| In river system | summer vs. winter | 63.4 | Nematoda, Copepoda, Acarina | 37.9 | Tardigrada, Polychaeta, Oligochaeta, Rotifera, Ostracoda, Acarina, Copepoda, Amphipoda, Bivalvia, Platelminta |
| In lagoon system | summer vs. winter | 39.3 | Nematoda | 28.3 | Ostracoda, Oligochaeta, Acarina, Amphipoda, Tardigrada, Rotifera, Nemertea, Polychaeta, Bivalvia, Isopoda, Kinorhyncha |
| In sea system | summer vs. winter | 37.0 | Nematoda, Copepoda | 31.3 | Kinorhyncha, Oligochaeta, Polychaeta, Rotifera, Priapulida larvae, Ostracoda |
| Variable | SS | F | P | % Variance | % Cumulative | |
|---|---|---|---|---|---|---|
| (A) | Temperature | 5505.6 | 2.8388 | *** | 13.6 | 13.6 |
| prokaryotic OTU composition | Phaeopigment | 4690.3 | 3.1907 | ** | 11.6 | 25.2 |
| Salinity | 4165.5 | 2.3033 | * | 10.3 | 35.5 | |
| Chlorophyll-a | 4003.7 | 2.3956 | ** | 9.9 | 45.4 | |
| Chlorophyll-a/Biopolymeric C | 3940.8 | 5.3008 | ** | 9.8 | 55.2 | |
| Lipid | 3410 | 3.3767 | * | 8.4 | 63.6 | |
| Protein | 3293.7 | 2.4585 | * | 8.1 | 71.8 | |
| Carbohydrate | 3227.5 | 2.702 | * | 8.0 | 79.8 | |
| Protein/Biopolymeric C | 2512.2 | 4.4342 | * | 6.2 | 86.0 | |
| Protein/Carbohydrate | 801.55 | 1.4831 | ns | 2.0 | 88.0 | |
| (B) | Phaeopigment | 27,987 | 14.951 | *** | 30.5 | 30.5 |
| meiofaunal taxonomic composition | Salinity | 15,291 | 10.435 | ** | 16.7 | 47.2 |
| Chlorophyll-a/Biopolymeric C | 8447.1 | 6.7732 | *** | 9.2 | 56.4 | |
| Protein | 4192.5 | 3.639 | ** | 4.6 | 61.0 | |
| Carbohydrate | 3872.8 | 4.4542 | ** | 4.2 | 65.2 | |
| Protein/Carbohydrate | 3817.2 | 3.59 | ** | 4.2 | 69.4 | |
| Chlorophyll-a | 3680.3 | 3.7822 | * | 4.0 | 73.4 | |
| Temperature | 2270.2 | 2.7766 | * | 2.5 | 75.9 | |
| Protein/Biopolymeric C | 1294.4 | 1.6557 | ns | 1.4 | 77.3 | |
| Lipid | 1235.1 | 1.5408 | ns | 1.3 | 78.7 | |
| (C) | Chlorophyll-a | 9912.3 | 4.7556 | ** | 22.9 | 22.9 |
| meiofaunal taxonomic composition in summer | Temperature | 8016.6 | 4.7468 | ** | 18.5 | 41.4 |
| Lipid | 7348.9 | 19.458 | ** | 17.0 | 58.4 | |
| Salinity | 7045.1 | 5.3933 | ** | 16.3 | 74.7 | |
| Chlorophyll-a/Biopolymeric C | 6406.7 | 7.0101 | ** | 14.8 | 89.5 | |
| Protein | 824.86 | 2.8042 | * | 1.9 | 91.4 | |
| Phaeopigment | 765.74 | 2.2364 | ns | 1.8 | 93.2 | |
| Protein/Carbohydrate | 351.13 | 1.22 | ns | 0.8 | 94.0 | |
| Protein/Biopolymeric C | 342.36 | 1.2184 | ns | 0.8 | 94.8 | |
| Carbohydrate | 110.93 | 0.36334 | ns | 0.3 | 95.1 | |
| (D) | Phaeopigment | 20,442 | 12.098 | *** | 43.1 | 43.1 |
| meiofaunal taxonomic composition in winter | Salinity | 13,045 | 13.987 | *** | 27.5 | 70.5 |
| Protein | 4212.2 | 6.0307 | *** | 8.9 | 79.4 | |
| Chlorophyll-a | 2762.1 | 5.1176 | *** | 5.8 | 85.2 | |
| Carbohydrate | 2283.7 | 5.7903 | ** | 4.8 | 90.0 | |
| Protein/Biopolymeric C | 785.55 | 2.0692 | ns | 1.7 | 91.7 | |
| Protein/Carbohydrate | 466.83 | 1.266 | ns | 1.0 | 92.7 | |
| Lipid | 364.15 | 0.91691 | ns | 0.8 | 93.4 | |
| Temperature | 323.36 | 0.86176 | ns | 0.7 | 94.1 | |
| Chlorophyll-a/Biopolymeric C | 166.23 | 0.39556 | ns | 0.4 | 94.5 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bianchelli, S.; Nizzoli, D.; Bartoli, M.; Viaroli, P.; Rastelli, E.; Pusceddu, A. Sedimentary Organic Matter, Prokaryotes, and Meiofauna across a River-Lagoon-Sea Gradient. Diversity 2020, 12, 189. https://doi.org/10.3390/d12050189
Bianchelli S, Nizzoli D, Bartoli M, Viaroli P, Rastelli E, Pusceddu A. Sedimentary Organic Matter, Prokaryotes, and Meiofauna across a River-Lagoon-Sea Gradient. Diversity. 2020; 12(5):189. https://doi.org/10.3390/d12050189
Chicago/Turabian StyleBianchelli, Silvia, Daniele Nizzoli, Marco Bartoli, Pierluigi Viaroli, Eugenio Rastelli, and Antonio Pusceddu. 2020. "Sedimentary Organic Matter, Prokaryotes, and Meiofauna across a River-Lagoon-Sea Gradient" Diversity 12, no. 5: 189. https://doi.org/10.3390/d12050189
APA StyleBianchelli, S., Nizzoli, D., Bartoli, M., Viaroli, P., Rastelli, E., & Pusceddu, A. (2020). Sedimentary Organic Matter, Prokaryotes, and Meiofauna across a River-Lagoon-Sea Gradient. Diversity, 12(5), 189. https://doi.org/10.3390/d12050189







