Phytoplankton Blooms, Red Tides and Mucilaginous Aggregates in the Urban Thessaloniki Bay, Eastern Mediterranean
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling Sites and Sample Collection
2.2. Chl a and Nutrient Measurements
- High ecological water quality: <0.04
- Good: 0.04–0.38
- Moderate: 0.38–0.85
- Poor: 0.85–1.51
- Bad: >1.51
2.3. Microscopic Analysis
2.4. Data Analysis
3. Results
3.1. Environmental Parameters
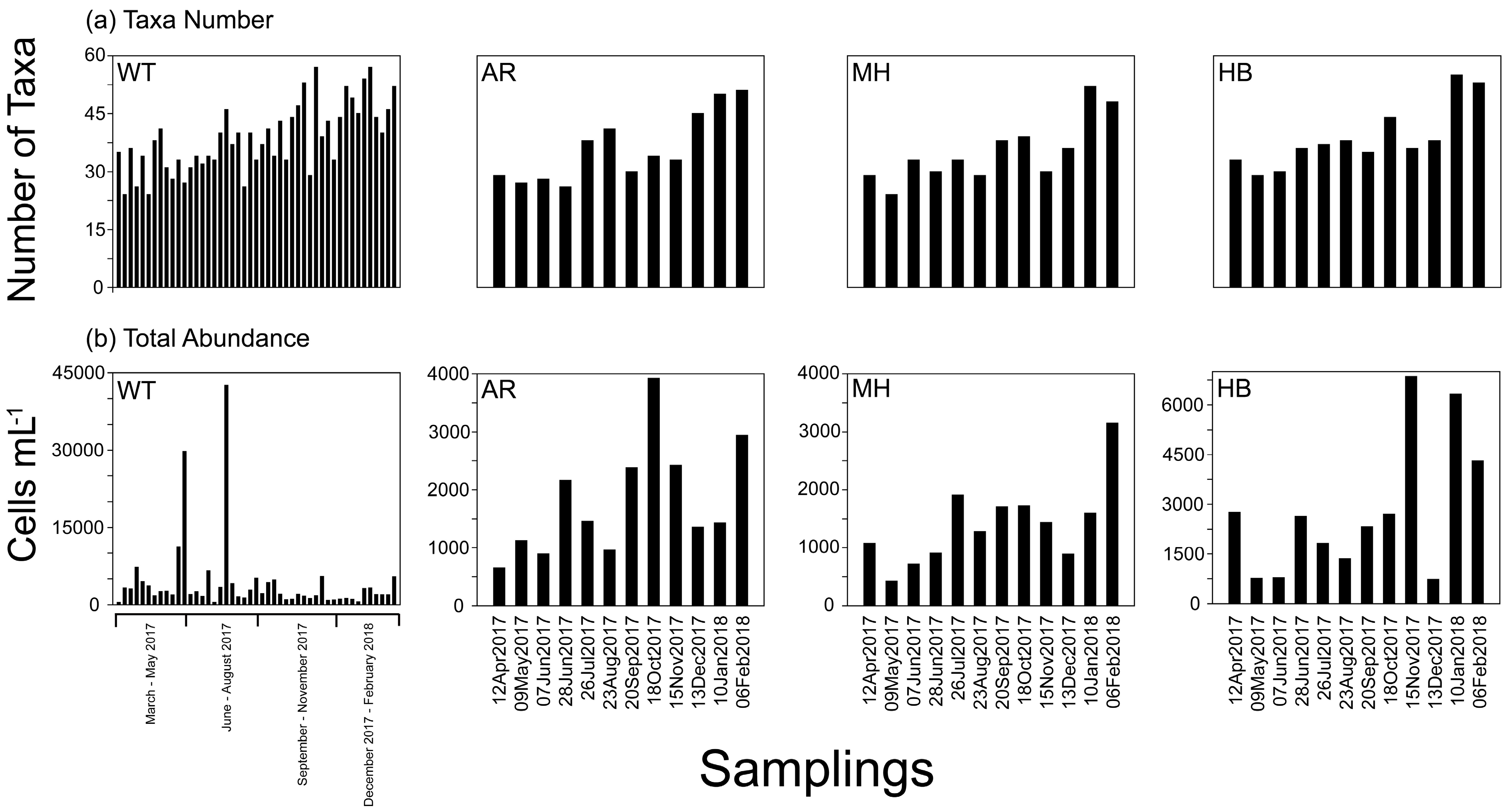
3.2. Plankton Diversity and Abundance
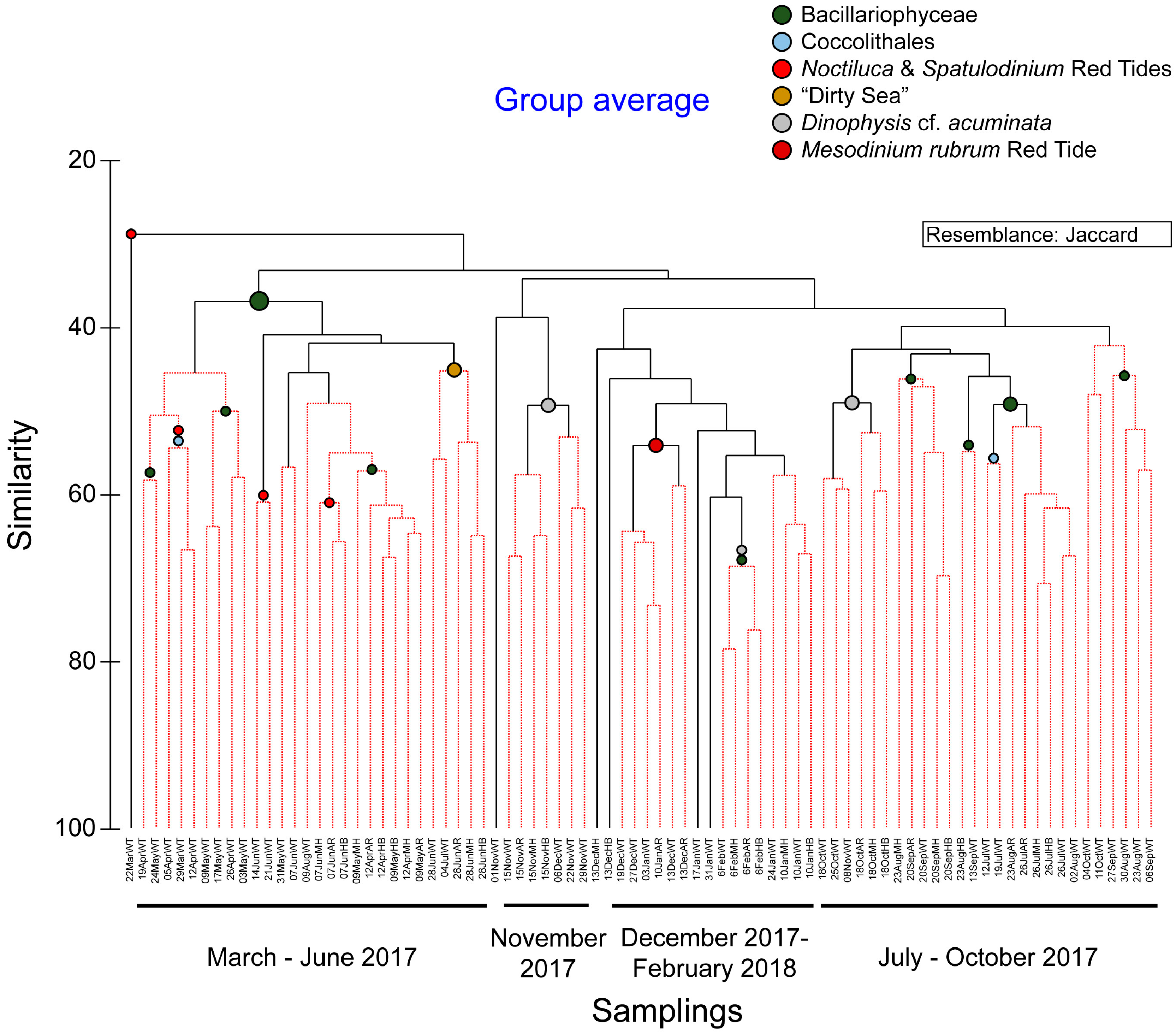
3.3. Phytoplankton Blooms, Red Tides, and a Mucilage Aggregate Phenomenon
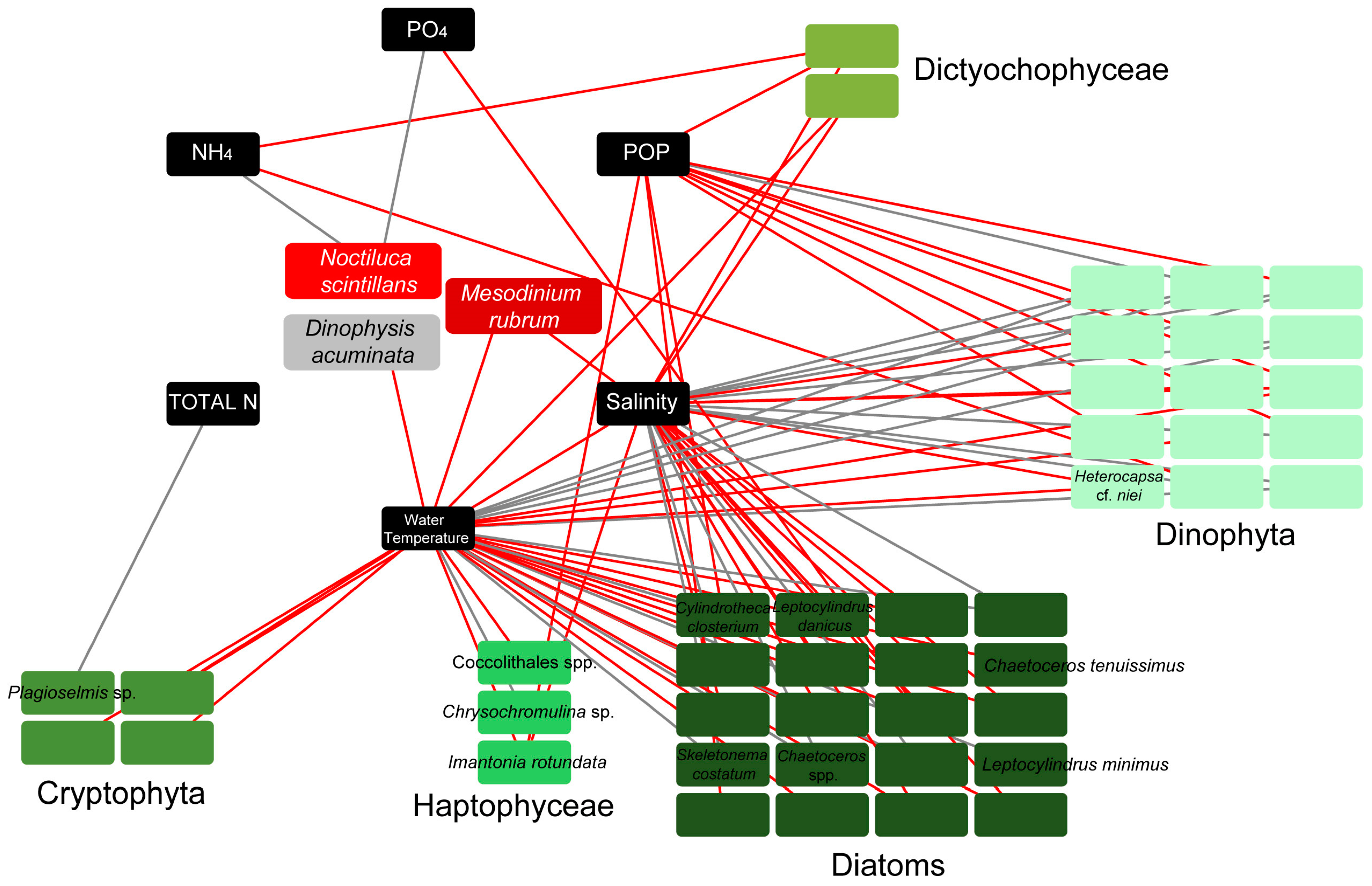
3.4. Links of Environmental Parameters and Plankton Bloom, Red Tide, and Mucilaginous Aggregate-Forming Taxa
4. Discussion
4.1. Environmental Conditions
4.2. Diversity and Composition of the Plankton Community
4.3. Phytoplankton Blooms
4.4. Red Tides
4.5. Mucilage Aggregates
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Rabalais, N.N.; Turner, R.E.; Díaz, R.J.; Justić, D. Global change and eutrophication of coastal waters. ICES J. Mar. Sci. 2009, 66, 1528–1537. [Google Scholar] [CrossRef]
- Garnier, J.; Beusen, A.; Thieu, V.; Billen, G.; Bouwman, L. N:P:Si nutrient export ratios and ecological consequences in coastal seas evaluated by the ICEP approach. Glob. Biogeochem. Cycl. 2010, 24. [Google Scholar] [CrossRef]
- Kroeze, C.; Hofstra, N.; Ivens, W.; Löhr, A.; Strokal, M.; van Wijnen, J. The links between global carbon, water and nutrient cycles in an urbanizing world-the case of coastal eutrophication. Curr. Opin. Environ. Sustain. 2013, 55, 566–572. [Google Scholar] [CrossRef]
- Cardona, Y.; Bracco, A.; Villareal, T.A.; Subramaniam, A.; Weber, S.C.; Montoya, J.P. Highly variable nutrient concentrations in the Northern Gulf of Mexico. Deep-Sea Res. Pt. II 2016, 129, 20–30. [Google Scholar] [CrossRef]
- Smith, V.H. Eutrophication of freshwater and coastal marine ecosystems: A global problem. Environ. Sci. Pollut. Res. 2003, 10, 126–139. [Google Scholar] [CrossRef]
- Turkoglu, M.; Erdogan, Y. Diurnal variations of summer phytoplankton and interactions with some physicochemical characteristics under eutrophication of surface water in the Dardanelles (Çanakkale Strait, Turkey). Turk. J. Biol. 2010, 34, 211–225. [Google Scholar]
- Turkoglu, M. Temporal variations of surface phytoplankton, nutrients and chlorophyll-a in the Dardanelles (Turkish Straits System): A coastal station sample in weekly time intervals. Turk. J. Biol. 2010, 34, 319–333. [Google Scholar]
- Heisler, J.; Glibert, P.M.; Burkholder, J.M.; Anderson, D.M.; Cochlan, W.; Dennison, W.C.; Gobler, C.; Dortch, Q.; Heil, C.; Humphries, E.; et al. Eutrophication and harmful algal blooms: A scientific consensus. Harmful Algae 2008, 8, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Anderson, D.M.; Cembella, A.D.; Hallegraeff, G.M. Progress in understanding harmful algal blooms: Paradigm shifts and new technologies for research, monitoring, and management. Annu. Rev. Mar. Sci. 2012, 4, 143–176. [Google Scholar] [CrossRef]
- Umani, S.F.; Del Negro, P.; Larato, C.; De Vittor, C.; Cabrini, M.; Celio, M.; Bingol, K.; Falconi, C.; Tamberlich, F.; Azam, F. Major inter-annual variations in microbial dynamics in the Gulf of Trieste (northern Adriatic Sea) and their ecosystem implications. Aquat. Microb. Ecol. 2007, 46, 163–175. [Google Scholar] [CrossRef]
- Cibic, T.; Cerino, F.; Karuza, A.; Fornasaro, D.; Comici, C.; Cabrini, M. Structural and functional response of phytoplankton to reduced river inputs and anomalous physical-chemical conditions in the Gulf of Trieste (northern Adriatic Sea). Sci. Total Environ. 2018, 636, 838–853. [Google Scholar] [CrossRef] [PubMed]
- Parsons, M.L.; Dortch, Q. Sedimentological evidence of an increase in Pseudo-nitzschia (Bacillariophyceae) abundance in response to coastal eutrophication. Limnol. Oceanogr. 2002, 47, 551–558. [Google Scholar] [CrossRef]
- Li, J.; Gilbert, P.M.; Zhou, M. Temporal and spatial variability in nitrogen uptake kinetics during harmful dinoflagellate blooms in the East China Sea. Harmful Algae 2010, 9, 531–539. [Google Scholar] [CrossRef]
- Dela-Cruz, J.; Middleton, J.H.; Suthers, I.M. The influence of upwelling, coastal nutrients and water temperature on the distribution of the red tide dinoflagellate, Noctiluca scintillans, along the east coast of Australia. Hydrobiologia 2008, 598, 59–75. [Google Scholar] [CrossRef]
- Harrison, P.J.; Furuya, K.; Glibert, P.M.; Xu, J.; Liu, H.B.; Yin, K.; Lee, J.H.W.; Anderson, D.M.; Gowen, R.; Al-Azri, A.R.; et al. Geographical distribution of red and green Noctiluca scintillans. Chin. J. Oceanol. Limnol. 2011, 29, 807–831. [Google Scholar] [CrossRef]
- Turkoglu, M. Red tides of the dinoflagellate Noctiluca scintillans associated with eutrophication in the Sea of Marmara (the Dardanelles, Turkey). Oceanologia 2013, 55, 709–732. [Google Scholar] [CrossRef]
- Krestenitis, Y.N.; Kombiadou, K.D.; Androulidakis, Y.S. Interannual variability of the physical characteristics of North Thermaikos Gulf (NW Aegean Sea). J. Mar. Syst. 2012, 96, 132–151. [Google Scholar] [CrossRef]
- Nikolaidis, G.; Moustaka-Gouni, M. The structure and dynamics of phytoplankton assemblages from the inner part of the Thermaikos Gulf, Greece. I. Phytoplankton composition and biomass from May 1988 to April 1989. Hegol. Mar. Res. 1990, 44, 487. [Google Scholar] [CrossRef]
- Karageorgis, A.P.; Skourtos, M.S.; Kapsimalis, V.; Kontogianni, A.D.; Skoulikidis, N.T.; Pagou, K.; Nikolaidis, N.P.; Drakopoulou, P.; Zanou, B.; Karamanos, H.; et al. An integrated approach to watershed management within the DPSIR framework: Axios River catchment and Thermaikos Gulf. Reg. Environ. Chang. 2005, 5, 138–160. [Google Scholar] [CrossRef]
- Mihalatou, H.M.; Moustaka-Gouni, M. Pico-, nano-, microplankton abundance and primary productivity in a eutrophic coastal area of the Aegean Sea, Mediterranean. Int. Rev. Hydrobiol. 2002, 87, 439–456. [Google Scholar] [CrossRef]
- Genitsaris, S.; Kormas, K.A.; Moustaka-Gouni, M. Airborne algae and cyanobacteria: Occurrence and related health effects. Front. Biosci. 2011, 3, 772–787. [Google Scholar]
- Friligos, N.; Kondylakis, J.C.; Psyllidou-Giouranovits, R.; Georgakopoulou-Gregoriadou, E. Eutrophication and phytoplankton abundance in the Thermaikos Gulf, Greece. Fresenius Environ. Bull. 1997, 6, 27–31. [Google Scholar]
- Moncheva, S.; Gotsis-Skretas, O.; Pagou, K.; Krastev, A. Phytoplankton blooms in Black Sea and Mediterranean coastal ecosystems subjected to anthropogenic eutrophication: Similarities and differences. Estuar. Coastal Shelf Sci. 2001, 53, 281–295. [Google Scholar] [CrossRef]
- Koukaras, K.; Nikolaidis, G. Dinophysis blooms in Greek coastal waters (Thermaikos Gulf, NW Aegean Sea). J. Plankton Res. 2004, 26, 445–457. [Google Scholar] [CrossRef]
- Reizopoulou, S.; Strogyloudi, E.; Giannakourou, A.; Pagou, K.; Hatzianestis, I.; Pyrgaki, C.; Granéli, E. Okadaic acid accumulation in macrofilter feeders subjected to natural blooms of Dinophysis acuminata. Harmful Algae 2008, 7, 228–234. [Google Scholar] [CrossRef]
- Ferreira, J.G.; Andersen, J.H.; Borja, A.; Bricker, S.B.; Camp, J.; Cardoso da Silva, M.; Garcés, E.; Heiskanen, A.S.; Humborg, C.; Ignatiades, L.; et al. Overview of eutrophication indicators to assess environmental status within the European Marine Strategy Framework Directive. Estuar. Coast. Shelf Sci. 2011, 93, 117–131. [Google Scholar] [CrossRef]
- Weyl, P.K. On the change in electrical conductance of seawater with temperature. Limnol. Oceanogr. 1964, 9, 75–78. [Google Scholar] [CrossRef]
- Jeffrey, S.W.; Humphrey, G.F. New spectrophotometric equation for determining chlorophyll a, b, c1 and c2. Biochem. Physiol. Pflanz. 1975, 167, 194–204. [Google Scholar]
- Hansen, H.P.; Koroleff, F. Determination of nutrients. In Methods of Seawater Analysis, 3rd ed.; Grasshoff, K., Kremling, K., Ehrhardt, M., Eds.; Wiley VCH: Weinheim, Germany, 1999; pp. 159–228. [Google Scholar]
- Primpas, I.; Tsirtsis, G.; Karydis, M.; Kokkoris, G.D. Principal component analysis: Development of a multivariate index for assessing eutrophication according to the European water framework directive. Ecol. Indic. 2010, 10, 178–183. [Google Scholar] [CrossRef]
- Hasle, G.R.; Syvertsen, E.E.; Steidinger, K.A.; Tangen, K.; Tomas, C.R. Identifying Marine Diatoms and Dinoflagellates; Academic Press: Cambridge, MA, USA, 1996; p. 613. [Google Scholar]
- Hasle, G.R.; Syvertsen, E.E.; Throndsen, J.; Steidinger, K.A.; Tangen, K.; Heimdal, B.R. Identifying Marine Phytoplankton; Academic Press: Cambridge, MA, USA, 1997; p. 858. [Google Scholar]
- Gómez, F.; Furuya, K. Kofoidinium, Spatulodinium and other Kofoidiniaceans (Noctilucales, Dinophyceae) in the Pacific Ocean. Eur. J. Protistol. 2007, 43, 115–124. [Google Scholar]
- Utermöhl, H. Zur Vervollkommnung der quantitativen Phytoplankton-Methodik. Mitt. Int. Ver. Theor. Angew. Limnol. 1958, 9, 1–38. [Google Scholar]
- Integrated Monitoring and Assessment Programme of the Mediterranean Sea and Coast and Related Assessment Criteria. Available online: https://wedocs.unep.org/handle/20.500.11822/10947 (accessed on 12 August 2019).
- Hammer, Ø.; Harper, D.A.T.; Ryan, P.D. PAST: Paleontological statistics software package for education and data analysis. Paleontol. Electron. 2001, 4, 9. [Google Scholar]
- Morris, E.K.; Caruso, T.; Buscot, F.; Fischer, M.; Hancock, C.; Maier, T.S.; Meiners, T.; Müller, C.; Obermaier, E.; Prati, D.; et al. Choosing and using diversity indices: Insights for ecological applications from the German Biodiversity Exploratories. Ecol. Evol. 2014, 4, 3514–3524. [Google Scholar] [CrossRef] [PubMed]
- Hillebrand, H.; Bennett, D.M.; Cadotte, M.W. Consequences of dominance: A review of evenness effects on local and regional ecosystem processes. Ecology 2008, 89, 1510–1520. [Google Scholar] [CrossRef] [PubMed]
- Primer v6: User Manual/Tutorial. PRIMER-E Plymouth. Available online: https://www.researchgate.net/publication/285668711_PRIMER_v6_user_manualtutorial_PRIMER-E_Plymouth (accessed on 31 January 2006).
- Reshef, D.N.; Reshef, Y.A.; Finucane, H.K.; Grossman, S.R.; McVean, G.; Turnbaugh, P.J.; Lander, E.S.; Mitzenmacher, M.; Sabeti, P.C. Detecting novel associations in large data sets. Science 2011, 334, 1518–1524. [Google Scholar] [CrossRef] [PubMed]
- Smoot, M.E.; Ono, K.; Ruscheinski, J.; Wang, P.-L.; Ideker, T. Cytoscape 2.8: New features for data integration and network visualization. Bioinformatics 2011, 27, 431–432. [Google Scholar] [CrossRef] [PubMed]
- Kontas, A.; Kucuksezgin, F.; Altay, O.; Uluturhan, E. Monitoring of eutrophication and nutrient limitation in the Izmir Bay (Turkey) before and after Wastewater Treatment Plant. Environ. Int. 2004, 29, 1057–1062. [Google Scholar] [CrossRef]
- Simboura, N.; Pavlidou, A.; Bald, J.; Tsapakis, M.; Pagou, K.; Zeri, C.; Androni, A.; Panayotidis, P. Response of ecological indices to nutrient and chemical contaminant stress factors in Eastern Mediterranean coastal waters. Ecol. Indic. 2016, 70, 89–105. [Google Scholar] [CrossRef]
- Tugrul, S.; Ozhan, K.; Akcay, I. Assessment of trophic status of the northeastern Mediterranean coastal waters: Eutrophication classification tools revisited. Environ. Sci. Pollut. Res. Int. 2018, 26, 14742–14754. [Google Scholar] [CrossRef]
- Pavlidou, A. Nutrient distribution in selected coastal areas of Aegean Sea (East Mediterranean Sea). J. Environ. 2012, 1, 78–88. [Google Scholar]
- Pavlidou, A.; Simboura, N.; Rousselaki, E.; Tsapakis, M.; Pagou, K.; Drakopoulou, P.; Assimakopoulou, G.; Kontoyiannis, H.; Panayotidis, P. Methods of eutrophication assessment in the context of the water framework directive: Examples from the Eastern Mediterranean coastal areas. Cont. Shelf Res. 2015, 108, 156–168. [Google Scholar] [CrossRef]
- Simboura, N.; Tsapakis, M.; Pavlidou, A.; Asimakopoulou, G.; Pagou, K.; Kontoyiannis, H.; Zeri, C.; Krasakopoulou, E.; Rousselaki, E.; Katsiaras, N.; et al. Assessment of the environmental status in the Hellenic coastal waters (Eastern Mediterranean): from the Water Framework Directive to the Marine Strategy Framework Directive. Mediterr. Mar. Sci. 2015, 16, 46–64. [Google Scholar] [CrossRef]
- Karydis, M. Environmental quality assessment based on the analysis of extreme values: A practical approach for evaluating eutrophication. J. Environ. Sci. Heal. 1994, 29, 775–791. [Google Scholar] [CrossRef]
- Xiao, W.; Liu, X.; Irwin, A.J.; Laws, E.A.; Wang, L.; Chen, B.; Zeng, Y.; Huang, B. Warming and eutrophication combine to restructure diatoms and dinoflagellates. Water Res. 2018, 128, 206–216. [Google Scholar] [CrossRef] [PubMed]
- Howarth, R.; Paerl, H.W. Coastal marine eutrophication: Control of both nitrogen and phosphorus is necessary. Proc. Natl. Acad. Sci. USA 2008, 105, E103. [Google Scholar] [CrossRef] [PubMed]
- Howarth, R.W. Coastal nitrogen pollution: A review of sources and trends globally and regionally. Harmful Algae 2008, 8, 14–20. [Google Scholar] [CrossRef]
- Verity, P.G. Effects of temperature, irradiance, and daylength on the marine diatom Leptocylindrus danicus Cleve. II. excretion. J. Exp. Mar. Biol. Ecol. 1981, 55, 159–169. [Google Scholar] [CrossRef]
- Geider, R.J. Light and temperature dependence of the carbon to chlorophyll a ratio in microalgae and cyanobacteria: Implications for physiology and growth of phytoplankton. New Phytol. 1987, 106, 1–34. [Google Scholar] [CrossRef]
- Partel, M.; Zobel, M.; Zobel, K.; Van Der Maarel, E. The species pool and its relation to species richness: Evidence from Estonian plant communities. Oikos 1996, 75, 111–117. [Google Scholar] [CrossRef]
- Sommer, U.; Charalampous, E.; Genitsaris, S.; Moustaka-Gouni, M. Benefits, costs and taxonomic distribution of marine phytoplankton body size. J. Plankton Res. 2017, 39, 494–508. [Google Scholar] [CrossRef]
- Stefanidou, N.; Genitsaris, S.; Lopez-Bautista, J.; Sommer, U.; Moustaka-Gouni, M. Effects of heat shock and salinity changes on coastal Mediterranean phytoplankton in a mesocosm experiment. Mar. Biol. 2018, 165, 154. [Google Scholar] [CrossRef]
- Hillebrand, H.; Matthiessen, B. Biodiversity in a complex world: Consolidation and progress in functional biodiversity research. Ecol. Lett. 2009, 12, 1405–1419. [Google Scholar] [CrossRef] [PubMed]
- Margalef, R. Turbulence and marine life. Sci. Mar. 1997, 61, 109–123. [Google Scholar]
- Passow, U.; Alldredge, A.L. Aggregation of a diatom bloom in a mesocosm: the role of transparent exopolymer particles (TEP). Deep-Sea Res. Pt. II 1995, 42, 99–109. [Google Scholar] [CrossRef]
- Sommer, U. Nutrient competition experiments with periphyton from the Baltic Sea. Mar. Ecol. Prog. Ser. 1996, 140, 161–167. [Google Scholar] [CrossRef]
- Le Moal, M.; Gascuel-Odoux, C.; Ménesguen, A.; Souchon, Y.; Étrillard, C.; Levain, A.; Moatar, F.; Pannard, A.; Souchu, P.; Lefebvre, A.; et al. Eutrophication: A new wine in an old bottle? Sci. Total Environ. 2019, 651, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Smayda, T.J.; Reynolds, C.S. Strategies of marine dinoflagellate survival and some rules of assembly. J. Sea Res. 2003, 49, 95–106. [Google Scholar] [CrossRef]
- Nikolaidis, G.; Koukaras, K.; Aligizaki, K.; Heracleous, A.; Kalopesa, E.; Moschandreou, K.; Tsolaki, E.; Mantoudis, A. Harmful microalgal episodes in Greek coastal waters. J. Biol. Res. 2005, 3, 77–85. [Google Scholar]
- Spatharis, S.; Dolopsakis, N.P.; Economou-Amilli, A.; Tsirtsis, G.; Danielidis, D.B. Dynamics of potentially harmful microalgae in a confined Mediterranean Gulf-Assessing the risk of bloom formation. Harmful Algae 2009, 8, 736–743. [Google Scholar] [CrossRef]
- Giannakourou, A.; Orlova, T.Y.; Assimakopoulou, G.; Pagou, K. Dinoflagellate cysts in recent marine sediments from Thermaikos Gulf, Greece: Effects of resuspension events on vertical cyst distribution. Cont. Shelf Res. 2005, 25, 2585–2596. [Google Scholar] [CrossRef]
- Bodenau, N. Microbial blooms in the Romanian area of the Black Sea and contemporary eutrophication conditions. In Toxic Phytoplankton Blooms in the Sea; Smayda, T.J., Shimizu, Y., Eds.; Elsevier: Amsterdam, The Netherland, 1993; pp. 203–209. [Google Scholar]
- Tsai, S.-F.; Wu, L.-Y.; Chou, W.-C.; Chiang, K.-P. The dynamics of a dominant dinoflagellate, Noctiluca scintillans, in the subtropical coastal waters of the Matsu archipelago. Mar. Pollut. Bull. 2018, 127, 553–558. [Google Scholar] [CrossRef] [PubMed]
- Turkoglu, M.; Koray, T. Phytoplankton species succession and nutrients in the Southern Black Sea (Bay of Sinop). Turk. J. Bot. 2002, 26, 235–252. [Google Scholar]
- Turkoglu, M.; Koray, T. Algal blooms in surface waters of the Sinop Bay in the Black Sea, Turkey. Pak. J. Biol. Sci. 2004, 7, 1577–1585. [Google Scholar]
- Kopuz, U.; Feyzioglu, A.M.; Valente, A. An unusual red-tide event of Noctiluca scintillans (Macartney) in the southeastern Black Sea. Turk. J. Fish. Aquat. Sci. 2014, 14, 261–268. [Google Scholar] [CrossRef]
- Koray, T.; Buyukisik, B.; Parlak, H.; Gokpinar, S. Final reports on research projects dealing with eutrophication and heavy metal accumulation. In Eutrophication Processes and Algal Blooms (Red-Tides) in Izmir Bay; UNEP: Athens, Greece, 1996; Volume 104, pp. 1–26. [Google Scholar]
- Umani, S.F.; Beran, A.; Parlato, S.; Virgilio, D.; Zollet, T.; De Olazabal, A.; Lazzarini, B.; Cabrini, M. Noctiluca scintillans Macartney in the Northern Adriatic Sea: Long-term dynamics, relationships with temperature and eutrophication, and role in the food web. J. Plankton Res. 2004, 26, 545–561. [Google Scholar] [CrossRef]
- Gómez, F. Diversity and distribution of noctilucoid dinoflagellates (Noctilucales, Dinophyceae) in the open Mediterranean Sea. Acta Protozool. 2010, 49, 365–372. [Google Scholar]
- Gárate-Lizárraga, I.; García-Domínguez, F.; Pérez-Cruz, B.; Díaz-Ortiz, J.A. First record of Cochlodinium convoltum and C. helicoides (Gymnodiniales: Dinophyceae) in the Gulf of California. Rev. Biol. Mar. Oceanog. 2011, 46, 495–498. [Google Scholar] [CrossRef]
- Miyaguchi, H.; Fujiki, T.; Kikuchi, T.; Kuwahara, V.S.; Toda, T. Relationship between the bloom of Noctiluca scintillans and environmental factors in the coastal waters of Sagami Bay, Japan. J. Plankton Res. 2006, 28, 313–324. [Google Scholar] [CrossRef]
- Dela-Cruz, J.; Ajani, P.; Lee, R.; Pritchard, T.; Suthers, I. Temporal abundance patterns of the red tide dinoflagellate Noctiluca scintillans along the southeast coast of Australia. Mar. Ecol. Prog. Ser. 2002, 236, 75–88. [Google Scholar] [CrossRef]
- Tiselius, P.; Kiørboe, T. Colonization of diatom aggregates by the dinoflagellate Noctiluca scintillans. Limnol. Oceanogr. 1998, 43, 154–159. [Google Scholar] [CrossRef]
- Zhang, S.; Liu, H.; Ke, Y.; Li, B. Effect of the silica content of diatoms on protozoan grazing. Front. Mar. Sci. 2017, 4, 202. [Google Scholar] [CrossRef]
- Escalera, L.; Pazos, Y.; Morono, A.; Reguera, B. Noctiluca scintillans may act as a vector of toxigenic microalgae. Harmful Algae 2007, 6, 317–320. [Google Scholar] [CrossRef]
- Frangópulos, M.; Spyrakos, E.; Guisande, C. Ingestion and clearance rates of the red Noctiluca scintillans fed on the toxic dinoflagellate Alexandrium minutum (Halim). Harmful Algae 2011, 10, 304–309. [Google Scholar] [CrossRef]
- Vlamis, A.; Katikou, P. Climate influence on Dinophysis spp. spatial and temporal distributions in Greek coastal water. Plankton Benthos Res. 2014, 9, 15–31. [Google Scholar] [CrossRef]
- Monitoring the Marine Environment of Thermaikos Gulf. Available online: https://www.researchgate.net/publication/326292943_Monitoring_the_marine_environment_of_Thermaikos_Gulf/link/5b4441c40f7e9bb59b1b265a/download (accessed on 10 July 2019).
- Mikaelyan, A.S.; Malej, A.; Shiganova, T.A.; Turk, V.; Sivkovitch, A.E.; Musaeva, E.I.; Kogovšek, T.; Lukasheva, T.A. Populations of the red tide forming dinoflagellate Noctiluca scintillans (Macartney): A comparison between the Black Sea and the northern Adriatic Sea. Harmful Algae 2014, 33, 29–40. [Google Scholar] [CrossRef]
- Montani, S.; Pithakpol, S.; Tada, K. Nutrient regeneration in coastal seas by Noctiluca scintillans, a red tide-causing dinoflagellate. J. Mar. Biotechnol. 1998, 6, 224–228. [Google Scholar]
- Schaumann, K.; Gerdes, D.; Hesse, K. Hydrographic and biological characteristics of a Noctiluca scintillans red tide in the German Bight, 1984. Meeresforschung 1988, 32, 77–91. [Google Scholar]
- Baliarsingh, S.K.; Lotliker, A.A.; Trainer, V.L.; Wells, M.L.; Parida, C.; Sahu, B.K.; Srichandan, S.; Sahu, K.C.; Kumar, T.S. Environmental dynamics of red Noctiluca scintillans bloom in tropical coastal waters. Mar. Pollut. Bull. 2016, 111, 277–286. [Google Scholar] [CrossRef]
- Millero, F.J.; Graham, T.B.; Huang, F.; Bustos-Serrano, H.; Pierrot, D. Dissociation constants of carbonic acid in seawater as a function of salinity and temperature. Mar. Chem. 2006, 100, 80–94. [Google Scholar] [CrossRef]
- Kang, N.S.; Lee, K.H.; Jeong, H.J.; Yoo, Y.D.; Seong, K.A.; Potvin, E.; Hwanga, Y.J.; Yoond, E.Y. Red tides in Shiwha Bay, western Korea: A huge dike and tidal power plant established in a semi-enclosed embayment system. Harmful Algae 2013, 30, S114–S130. [Google Scholar] [CrossRef]
- Gustafson, D.E., Jr.; Stoesker, D.K.; Johnson, M.D.; Van Heukelem, W.F.; Sneider, K. Cryptophte algae are robbed of their organelles by the marine Mesodinium rubrum. Nature 2000, 405, 1049–1052. [Google Scholar] [CrossRef] [PubMed]
- Park, M.G.; Kim, S.; Kim, H.S.; Myung, G.; Kang, Y.G.; Yih, W. First successful culture of the marine dinoflagellate Dinophysis acuminata. Aquat. Microb. Ecol. 2006, 45, 101–106. [Google Scholar] [CrossRef]
- Lee, K.H.; Jeong, H.J.; Yoon, E.Y.; Jang, S.H.; Kim, H.S.; Yih, W. Feeding by common heterotrophic dinoflagellates and a ciliate on the red-tide ciliate Mesodinium rubrum. Algae 2014, 29, 153–163. [Google Scholar] [CrossRef]
- Degobbis, D.; Umani, S.F.; Franco, P.; Malej, A.; Precali, R.; Smodlaka, N. Changes in the northern Adriatic ecosystem and the hypertrophic appearance of gelatinous aggregates. Sci. Total Environ. 1995, 165, 43–58. [Google Scholar] [CrossRef]
- Precali, R.; Giani, M.; Marini, M.; Grilli, F.; Ferrari, C.R.; Pečar, O.; Paschini, E. Mucilaginous aggregates in the northern Adriatic in the period 1999–2002: Typology and distribution. Sci. Total Environ. 2005, 353, 10–23. [Google Scholar] [CrossRef] [PubMed]
- Giani, M.; Berto, D.; Zangrando, V.; Castelli, S.; Sist, P.; Urbani, R. Chemical characterization of different typologies of mucilaginous aggregates in the Northern Adriatic Sea. Sci. Total Environ. 2005, 353, 232–246. [Google Scholar] [CrossRef]
- Svetličić, V.; Žutić, V.; Radić, T.M.; Pletikapić, G.; Zimmermann, A.H.; Urbani, R. Polymer networks produced by marine diatoms in the Northern Adriatic Sea. Mar. Drugs 2011, 9, 666–679. [Google Scholar] [CrossRef]
- Pompei, M.; Mazziotti, C.; Guerrini, F.; Cangini, M.; Pigozzi, S.; Benzi, M.; Palamidesi, S.; Boni, L.; Pistocchi, R. Correlation between the presence of Gonyaulax fragilis (Dinophyceae) and the mucilage phenomena of the Emilia-Romagna coast (northern Adriatic Sea). Harmful Algae 2003, 2, 301–316. [Google Scholar] [CrossRef]
- Tüfekçi, V.; Balkis, N.; Beken, C.P.; Ediger, D.; Mantikçi, M. Phytoplankton composition and environmental conditions of a mucilage event in the Sea of Marmara. Turk. J. Biol. 2010, 34, 199–210. [Google Scholar]
- Ignatiades, L.; Gotsis-Skretas, O. A review on toxic and harmful algae in Greek coastal waters (E. Mediterranean Sea). Toxins 2010, 2, 1019–1037. [Google Scholar] [CrossRef]
- De Lazzari, A.; Berto, D.; Cassin, D.; Boldrin, A.; Giani, M. Influence of winds and oceanographic conditions on the mucilage aggregation in the Northern Adriatic Sea in 2003–2006. Mar. Ecol. 2008, 29, 469–482. [Google Scholar] [CrossRef]
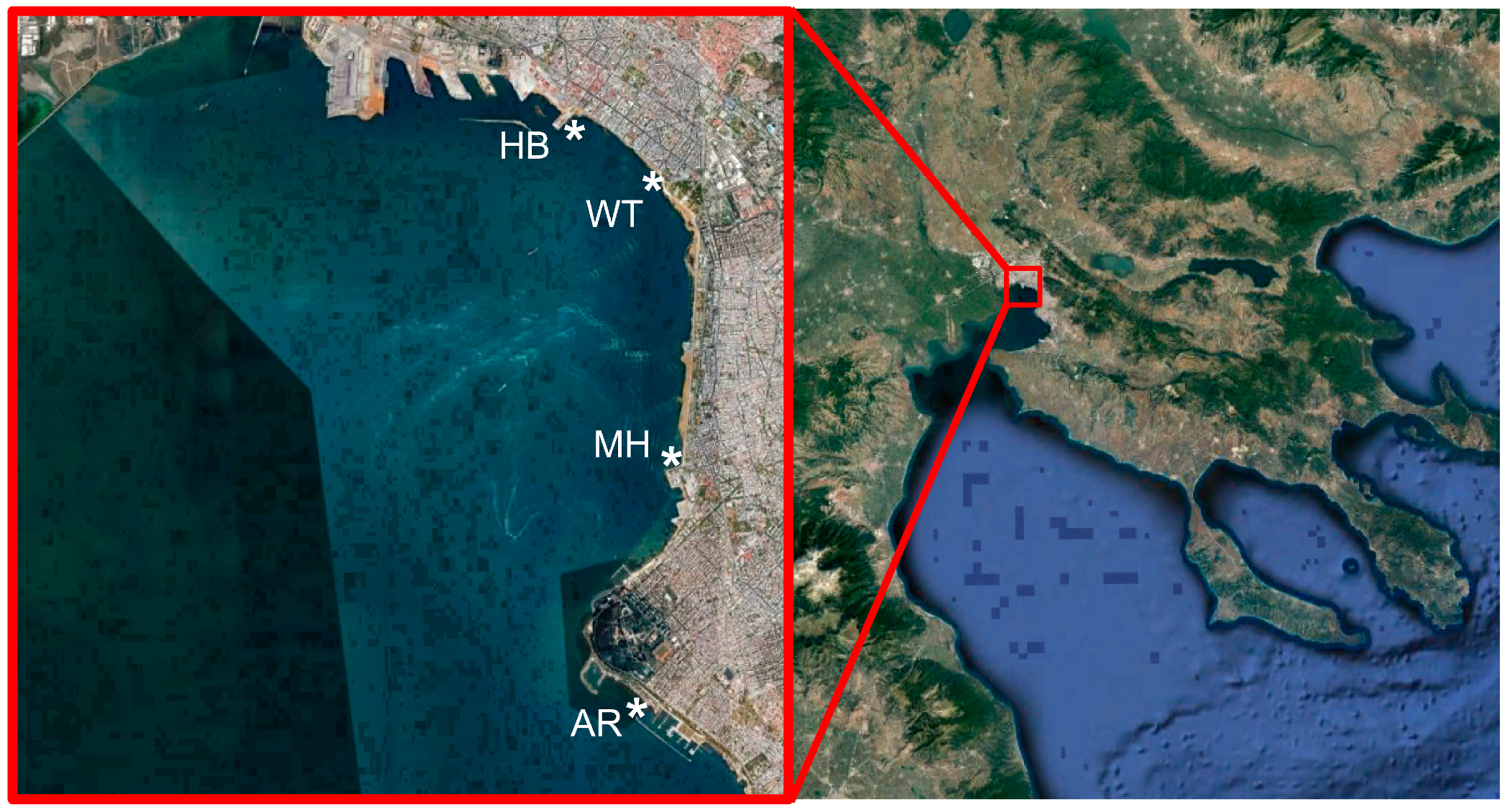
| Sampling Sites | Latitude | Longitude | Number of Samples |
|---|---|---|---|
| White Tower (WT) | 40°37′34 N | 22°56′51 E | 47 |
| Aretsou Beach (AR) | 40°34′29 N | 22°56′38 E | 12 |
| Music Hall coast (MH) | 40°35′57 N | 22°56′53 E | 12 |
| Harbor (HB) | 40°37′55 N | 22°56′09 E | 12 |
| Sample Date and Site | Sample Code | Temperature of Water (°C) | Salinity | SiO4 | PO4 | NO2 | NO3 & NO2 | NH4 | POP | Chl a (μg L−1) |
|---|---|---|---|---|---|---|---|---|---|---|
| 1. 15 March 2017–W. Tower | 15MarWT | 12.1 | 38.8 | 3.85 | 0.75 | 0.37 | 3.59 | 7.59 | 0.20 | 0.60 |
| 2. 22 March 2017–W. Tower | 22MarWT | 14 | 38.6 | 10.17 | 9.54 | 0.77 | 7.53 | 160.3 | 42.1 | 3.54 |
| 3. 29 March 2017–W. Tower | 29MarWT | 14.1 | 37.2 | 4.98 | 0.42 | 0.09 | 1.72 | 3.55 | 0.46 | 1.67 |
| 4. 05 April 2017–W. Tower | 05AprWT | 16.4 | 37.3 | 6.33 | 0.41 | 0.40 | 2.28 | 6.71 | 0.85 | 2.42 |
| 5. 12 April 2017–W. Tower | 12AprWT | 15.5 | 37.7 | 8.49 | 0.78 | 0.08 | 8.36 | 11.2 | 2.81 | 2.76 |
| 6. 12 April 2017–Aretsou | 12AprAR | 16.1 | 37.3 | 3.79 | 0.27 | 0.20 | 2.52 | 4.48 | 0.48 | 1.39 |
| 7. 12 April 2017–Music Hall | 12AprMH | 15.4 | 38.3 | 3.94 | 0.30 | 0.06 | 1.00 | 3.04 | 0.30 | 1.29 |
| 8. 12 April 2017–Harbour | 12AprHB | 15.5 | 38.2 | 4.52 | 0.25 | 0.16 | 1.39 | 3.55 | 0.78 | 2.79 |
| 9. 19 April 2017–W. Tower | 19AprWT | 15.8 | 38 | 2.59 | 0.19 | 0.04 | 0.54 | 1.99 | 0.21 | 0.60 |
| 10. 26 April 2017–W. Tower | 26AprWT | 15.1 | 38.2 | 3.36 | 0.35 | 0.10 | 1.82 | 4.36 | 0.28 | 0.78 |
| 11. 03 May 2017–W. Tower | 03MayWT | 19 | 37.3 | 3.63 | 0.22 | 0.19 | 2.69 | 3.53 | 0.38 | 1.14 |
| 12. 09 May 2017–W. Tower | 09MayWT | 18.6 | 37.8 | 4.76 | 0.20 | 0.15 | 7.36 | 4.61 | 0.25 | 0.95 |
| 13. 09 May 2017–Aretsou | 09MayAR | 19.5 | 38 | 4.10 | 0.18 | 0.21 | 2.26 | 4.63 | 0.22 | 1.10 |
| 14. 09 May 2017–Music Hall | 09MayMH | 19 | 38.1 | 2.37 | 0.30 | 0.13 | 1.71 | 4.45 | 0.14 | 0.48 |
| 15. 09 May 2017–Harbour | 09MayHB | 18.3 | 38.1 | 2.50 | 0.16 | 0.15 | 1.54 | 3.73 | 0.23 | 0.71 |
| 16. 17 May 2017–W. Tower | 17MayWT | 20.1 | 37.1 | 6.34 | 0.49 | 0.19 | 7.08 | 5.32 | 0.97 | 2.77 |
| 17. 24 May 2017–W. Tower | 24MayWT | 22 | 32.8 | 2.95 | 0.15 | 0.18 | 1.97 | 2.67 | 0.49 | 3.63 |
| 18. 31 May 2017–W. Tower | 31MayWT | 22.7 | 37.4 | 2.55 | 0.18 | 0.21 | 5.03 | 2.03 | 0.62 | 4.70 |
| 19. 07 June 2017–W. Tower | 07JunWT | 25.4 | 36.7 | 3.44 | 0.57 | 0.27 | 2.36 | 8.08 | 0.55 | 1.40 |
| 20. 07 June 2017–Aretsou | 07JunAR | 25.7 | 36.3 | 5.93 | 0.20 | 0.14 | 4.70 | 2.96 | 0.51 | 2.17 |
| 21. 07 June 2017–Music Hall | 07JunMH | 24.9 | 36.7 | 4.13 | 0.43 | 0.10 | 0.89 | 4.36 | 0.48 | 1.04 |
| 22. 07 June 2017–Harbour | 07JunHB | 25.9 | 36.5 | 4.87 | 0.48 | 0.21 | 3.11 | 6.01 | 0.49 | 1.14 |
| 23. 14 June 2017–W. Tower | 14JunWT | 25.5 | 36.7 | 6.24 | 0.40 | 0.33 | 3.91 | 7.53 | 0.54 | 1.74 |
| 24. 21 June 2017–W. Tower | 21JunWT | 23.1 | 36.2 | 4.13 | 0.27 | 0.26 | 2.55 | 5.10 | 0.26 | 1.01 |
| 25. 28 June 2017–W. Tower | 28JunWT | 28 | 36.1 | 18.39 | 0.24 | 0.18 | 10.93 | 6.33 | 1.87 | 6.85 |
| 26. 28 June 2017–Aretsou | 28JunAR | 28.4 | 36.2 | 6.99 | 0.18 | 0.14 | 3.72 | 3.34 | 0.70 | 4.10 |
| 27. 28 June 2017–Music Hall | 28JunMH | 27.9 | 36 | 10.04 | 0.44 | 0.21 | 4.46 | 6.99 | 0.38 | 1.11 |
| 28. 28 June 2017–Harbour | 28JunHB | 28.7 | 36.4 | 9.44 | 0.14 | 0.07 | 2.48 | 2.37 | 0.86 | 4.44 |
| 29. 04 July 2017–W. Tower | 04JulWT | 20.5 | 35.7 | 6.79 | 0.49 | 0.16 | 2.43 | 7.68 | 0.23 | 1.21 |
| 30. 12 July 2017–W. Tower | 12JulWT | 28 | 36.7 | 4.69 | 0.19 | 0.07 | 1.98 | 4.24 | 0.41 | 2.56 |
| 31. 19 July 2017–W. Tower | 19JulWT | 23.4 | 35.3 | 5.24 | 0.15 | 0.09 | 1.32 | 2.13 | 0.91 | 9.90 |
| 32. 26 July 2017–W. Tower | 26JulWT | 28.8 | 35.9 | 6.42 | 0.14 | 0.07 | 1.58 | 2.36 | 0.56 | 1.98 |
| 33. 26 July 2017–Aretsou | 26JulAR | 28.1 | 36.4 | 5.47 | 0.12 | 0.20 | 3.34 | 2.47 | 0.58 | 1.75 |
| 34. 26 July 2017–Music Hall | 26JulMH | 29.3 | 36 | 5.25 | 0.13 | 0.10 | 0.72 | 1.93 | 0.79 | 2.14 |
| 35. 26 July 2017–Harbour | 26JulHB | 29 | 35.5 | 6.65 | 0.14 | 0.07 | 0.88 | 4.45 | 0.67 | 1.24 |
| 36. 02 August 2017–W. Tower | 02AugWT | 27.2 | 36.3 | 7.33 | 0.25 | 0.11 | 2.34 | 4.96 | 0.31 | 0.79 |
| 37. 09 August 2017–W. Tower | 09AugWT | 29.7 | 36.6 | 7.91 | 0.30 | 0.15 | 7.21 | 7.26 | 0.66 | 1.13 |
| 38. 23 August 2017–W. Tower | 23AugWT | 24.6 | 37.7 | 6.00 | 0.24 | 0.09 | 1.01 | 3.50 | 0.25 | 0.77 |
| 39. 23 August 2017–Aretsou | 23AugAR | 25 | 36.8 | 7.08 | 0.24 | 0.25 | 2.21 | 3.03 | 0.29 | 1.05 |
| 40. 23 August 2017–Music Hall | 23AugMH | 24.9 | 37.5 | 5.91 | 0.21 | 0.08 | 1.99 | 3.11 | 0.21 | 1.04 |
| 41. 23 August 2017–Harbour | 23AugHB | 24.6 | 37.7 | N/A | N/A | N/A | N/A | N/A | 0.20 | 0.77 |
| 42. 30 August 2017–W. Tower | 30AugWT | 24.9 | 36.3 | 6.03 | 0.63 | 0.40 | 6.29 | 9.63 | 0.74 | 1.86 |
| 43. 06 September 2017–W. Tower | 06SepWT | 24.8 | 37.6 | 8.70 | 0.30 | 0.19 | 3.53 | 6.00 | 0.78 | 1.23 |
| 44. 13 September 2017–W. Tower | 13SepWT | 26.2 | 37.2 | 3.10 | 0.17 | 0.16 | 1.27 | 3.51 | 0.43 | 1.81 |
| 45. 20 September 2017–W. Tower | 20SepWT | 26.6 | 37.3 | 11.83 | 0.25 | 0.19 | 3.80 | 6.64 | 0.84 | 1.66 |
| 46. 20 September 2017–Aretsou | 20SepAR | 26.4 | 37.3 | 10.08 | 0.16 | 0.46 | 5.75 | 5.68 | 0.40 | 2.25 |
| 47. 20 September 2017–Music Hall | 20SepMH | 26.8 | 37.4 | 13.37 | 0.34 | 0.20 | 2.54 | 8.54 | 0.66 | 1.50 |
| 48. 20 September 2017–Harbour | 20SepHB | 26.6 | 37.3 | 13.14 | 0.27 | 0.23 | 4.78 | 7.04 | 0.42 | 1.60 |
| 49. 27 September 2017–W. Tower | 27SepWT | 22.6 | 37.2 | 1.27 | 0.28 | 0.08 | 1.44 | 2.16 | 0.16 | 2.74 |
| 50. 04 October 2017–W. Tower | 04OctWT | 22.2 | 37.4 | 2.78 | 0.38 | 0.14 | 2.16 | 3.18 | 0.16 | 0.66 |
| 51. 11 October 2017–W. Tower | 11OctWT | 20.9 | 37.4 | 2.31 | 0.32 | 0.29 | 2.11 | 4.53 | 0.17 | 0.72 |
| 52. 18 October 2017–W. Tower | 18OctWT | 20.8 | 38.1 | 2.60 | 0.31 | 0.08 | 1.92 | 3.14 | 0.28 | 1.29 |
| 53. 18 October 2017–Aretsou | 18OctAR | 20.5 | 37.4 | 2.07 | 0.22 | 0.09 | 1.80 | 2.49 | 0.21 | 2.24 |
| 54. 18 October 2017–Music Hall | 18OctMH | 20.8 | 37.6 | 1.25 | 0.23 | 0.07 | 0.79 | 2.69 | 0.20 | 1.08 |
| 55. 18 October 2017–Harbour | 18OctHB | 20.8 | 37.7 | 2.66 | 3.71 | 0.19 | 2.03 | 32.86 | 1.12 | 1.50 |
| 56. 25 October 2017–W. Tower | 18OctWT | 19.8 | 36.7 | 2.39 | 0.32 | 0.11 | 4.41 | 3.40 | 0.17 | 0.98 |
| 57. 01 November 2017–W. Tower | 01NovWT | 17.4 | 37.6 | 2.94 | 0.35 | 0.19 | 2.10 | 3.29 | 0.17 | 0.71 |
| 58. 08 November 2017–W. Tower | 08NovWT | 16.7 | 37.6 | 1.35 | 0.30 | 0.10 | 1.02 | 2.92 | 0.39 | 0.94 |
| 59. 15 November 2017–W. Tower | 15NovWT | 16.7 | 37.5 | 2.98 | 0.30 | 0.18 | 3.82 | 6.38 | 0.77 | 6.58 |
| 60. 15 November 2017–Aretsou | 15NovAR | 16.9 | 37.7 | 2.50 | 0.31 | 0.21 | 2.92 | 3.54 | 0.19 | 1.10 |
| 61. 15 November 2017–Music Hall | 15NovMH | 16.8 | 37.8 | 2.77 | 0.23 | 0.11 | 1.85 | 3.09 | 0.08 | 1.34 |
| 62. 15 November 2017–Harbour | 15NovHB | 16.6 | 37.3 | 1.07 | 0.30 | 0.08 | 1.23 | 7.54 | 1.50 | 10.05 |
| 63. 22 November 2017–W. Tower | 22NovWT | 15.2 | 37.8 | 1.32 | 0.27 | 0.10 | 0.72 | 2.42 | 0.15 | 1.02 |
| 64. 29 November 2017–W. Tower | 29NovWT | 13.7 | 37.7 | 5.05 | 0.51 | 0.60 | 2.41 | 4.49 | 0.19 | 1.75 |
| 65. 06 December 2017–W. Tower | 06DecWT | 13.3 | 37.3 | 2.29 | 0.28 | 0.19 | 2.05 | 2.08 | 0.18 | 4.73 |
| 66. 13 December 2017–W. Tower | 13DecWT | 12.5 | 37.8 | 3.87 | 0.37 | 0.10 | 0.54 | 3.20 | 0.49 | 17.28 |
| 67. 13 December 2017–Aretsou | 13DecAR | 12.4 | 37.4 | 5.47 | 0.45 | 0.23 | 3.54 | 2.04 | 1.09 | 16.33 |
| 68. 13 December 2017–Music Hall | 13DecMH | 12.8 | 37.7 | 6.42 | 0.32 | 0.11 | 1.28 | 2.68 | 0.13 | 0.93 |
| 69. 13 December 2017–Harbour | 13DecHB | 12.5 | 37.8 | 6.29 | 0.46 | 0.41 | 2.42 | 4.49 | 0.23 | 1.91 |
| 70. 19 December 2017–W. Tower | 19DecWT | 11.7 | 37.7 | 6.03 | 0.33 | 0.09 | 2.14 | 3.21 | 0.22 | 1.95 |
| 71. 27 December 2017–W. Tower | 27DecWT | 11.1 | 37.9 | 4.84 | 0.37 | 0.11 | 1.69 | 5.75 | 0.08 | 0.27 |
| 72. 03 January 2018–W. Tower | 03JanWT | 11.1 | 38 | 2.63 | 0.22 | 0.06 | 2.38 | 2.08 | 0.20 | 2.52 |
| 73. 10 January 2018–W. Tower | 10JanWT | 10.6 | 36.6 | 9.62 | 1.58 | 0.35 | 21.46 | 17.81 | 1.06 | 1.79 |
| 74. 10 January 2018–Aretsou | 10JanAR | 10.7 | 38.1 | 5.06 | 0.26 | 0.15 | 3.63 | 2.74 | 0.18 | 0.92 |
| 75. 10 January 2018–Music Hall | 10JanMH | 10.7 | 37.8 | 2.72 | 0.54 | 0.16 | 3.81 | 4.58 | 0.25 | 2.95 |
| 76. 10 January 2018–Harbour | 10JanHB | 10.6 | 37.2 | 3.43 | 0.55 | 0.16 | 7.67 | 7.27 | 1.12 | 8.31 |
| 77. 17 January 2018–W. Tower | 17JanWT | 10.4 | 37.8 | 1.46 | 0.26 | 0.04 | 1.01 | 2.24 | 0.12 | 1.02 |
| 78. 24 January 2018–W. Tower | 24JanWT | 9.6 | 37.9 | 2.28 | 0.29 | 0.08 | 2.35 | 2.06 | 0.18 | 1.15 |
| 79. 31 January 2018–W. Tower | 31JanWT | 10.7 | 38.2 | 2.48 | 0.28 | 0.07 | 3.00 | 2.24 | 0.20 | 1.15 |
| 80. 06 February 2018–W. Tower | 06FebWT | 11.9 | 38.4 | 2.27 | 0.32 | 0.09 | 2.78 | 3.22 | 0.28 | 2.82 |
| 81. 06 February 2018–Aretsou | 06FebAR | 11.8 | 38.1 | 3.14 | 0.21 | 0.07 | 2.80 | 5.93 | 0.34 | 2.91 |
| 82. 06 February 2018–Music Hall | 06FebMH | 11.7 | 38.4 | 1.89 | 0.17 | 0.06 | 0.44 | 1.91 | 0.26 | 2.30 |
| 83. 06 February 2018–Harbour | 06FebHB | 11.9 | 38 | 3.96 | 0.29 | 0.08 | 3.97 | 4.02 | 0.60 | 3.36 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Genitsaris, S.; Stefanidou, N.; Sommer, U.; Moustaka-Gouni, M. Phytoplankton Blooms, Red Tides and Mucilaginous Aggregates in the Urban Thessaloniki Bay, Eastern Mediterranean. Diversity 2019, 11, 136. https://doi.org/10.3390/d11080136
Genitsaris S, Stefanidou N, Sommer U, Moustaka-Gouni M. Phytoplankton Blooms, Red Tides and Mucilaginous Aggregates in the Urban Thessaloniki Bay, Eastern Mediterranean. Diversity. 2019; 11(8):136. https://doi.org/10.3390/d11080136
Chicago/Turabian StyleGenitsaris, Savvas, Natassa Stefanidou, Ulrich Sommer, and Maria Moustaka-Gouni. 2019. "Phytoplankton Blooms, Red Tides and Mucilaginous Aggregates in the Urban Thessaloniki Bay, Eastern Mediterranean" Diversity 11, no. 8: 136. https://doi.org/10.3390/d11080136
APA StyleGenitsaris, S., Stefanidou, N., Sommer, U., & Moustaka-Gouni, M. (2019). Phytoplankton Blooms, Red Tides and Mucilaginous Aggregates in the Urban Thessaloniki Bay, Eastern Mediterranean. Diversity, 11(8), 136. https://doi.org/10.3390/d11080136








