Soil Biological Fertility and Bacterial Community Response to Land Use Intensity: A Case Study in the Mediterranean Area
Abstract
1. Introduction
2. Materials and Methods
2.1. Measurements
2.2. Soil Microbial Biomass
2.3. Microbial Respiration
2.4. Biological Fertility Index
2.5. Soil DNA Extraction
2.6. Real-Time Quantitative PCR Analysis (qPCR)
2.7. Analyses of 16S rRNA Gene Sequence, Species Richness and Diversity
2.8. Statistical Analysis
3. Results
3.1. Chemical and Microbial Parameters
3.2. Soil Biological Fertility Index
3.3. Nucleic Acid Concentration
3.4. Bacterial Quantification
3.5. Correlation Coefficients among Parameters
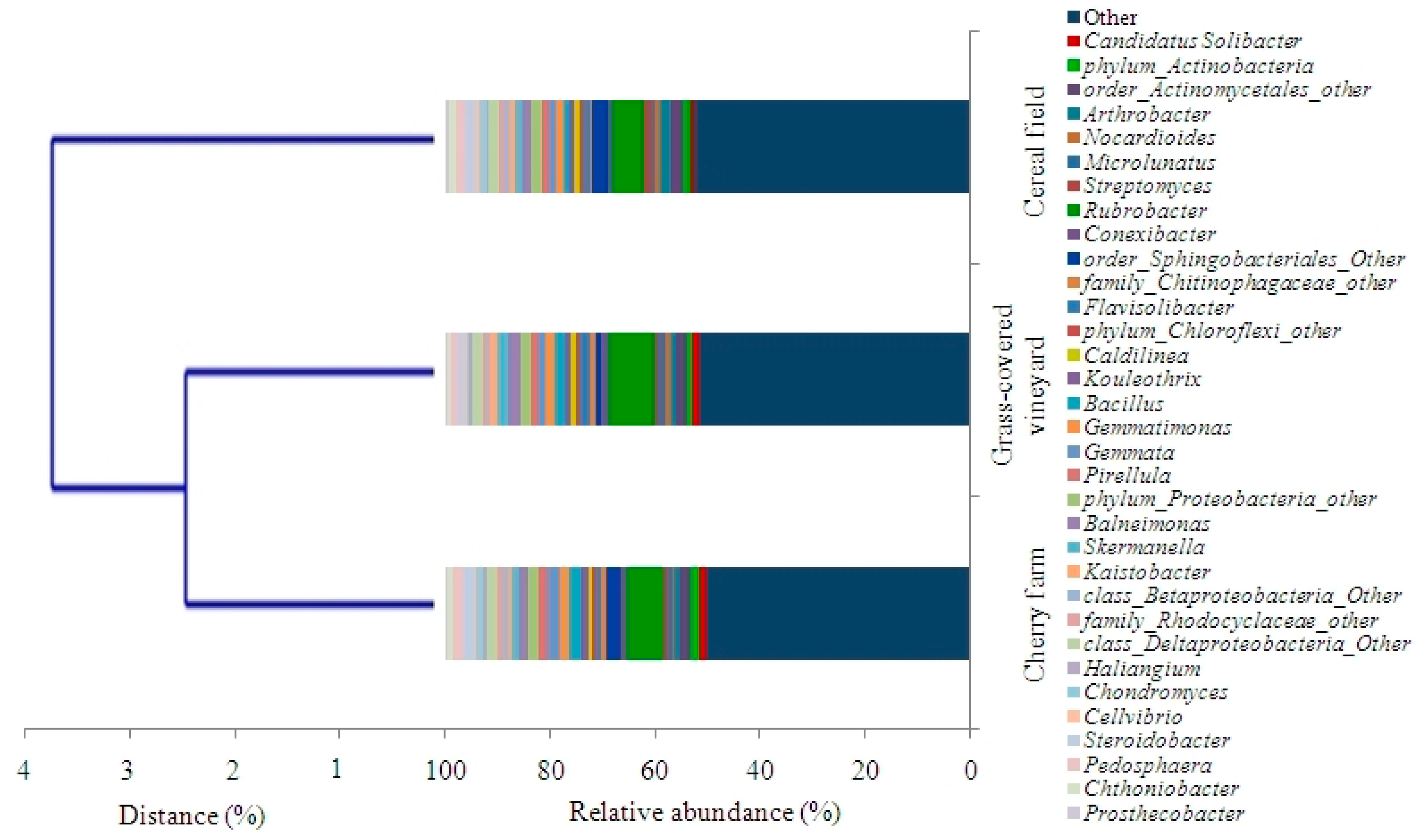
3.6. Bacterial α Diversity and Community Composition
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cerdà, A.; Hooke, J.; Romero-Diaz, A.; Montanarella, L.; Lavee, H. Soil erosion on Mediterranean type-ecosystems. Land Degrad. Dev. 2010, 21, 71–74. [Google Scholar] [CrossRef]
- Shelef, O.; Stavi, I.; Zdruli, P.; Rachmilevitch, S. Land use change, a case study from southern Italy: General implications for agricultural subsidy policies. Land Degrad. Dev. 2014, 27, 868–870. [Google Scholar] [CrossRef]
- Zdruli, P.; Calabrese, J.; Ladisa, G.A.O. Impacts of land cover change on soil quality of manmade soils cultivated with table grapes in the Apulia Region of south-eastern Italy. Catena 2014, 121, 13–21. [Google Scholar] [CrossRef]
- Bai, X.Y.; Wang, S.J.; Xiong, K.N. Assessing spatial-temporal evolution processes of karst rocky desertification land: Indications for restoration strategies. Land Degrad. Dev. 2013, 24, 47–56. [Google Scholar] [CrossRef]
- Bajocco, S.; Smiraglia, D.; Scaglione, M.; Raparelli, E.; Salvati, L. Exploring the role of land degradation on agricultural land use change dynamics. Sci. Total Environ. 2018, 636, 1373–1381. [Google Scholar] [CrossRef]
- Masciopinto, C.; De Giglio, O.; Scrascia, M.; Fortunato, F.; La Rosa, G.; Suffredini, E.; Pazzani, C.; Prato, R.; Montagna, M.T. Human health risk assessment for the occurrence of enteric viruses in drinking water from wells: Role of flood runoff injections. Sci. Total Environ. 2019, 666, 559–571. [Google Scholar] [CrossRef]
- Zdruli, P. Land resources of the Mediterranean: Status, pressures, trends and impacts on future regional development. Land Degrad. Dev. 2014, 25, 373–384. [Google Scholar] [CrossRef]
- Lindtner, P.; Gömöryová, E.; Gömöry, D.; Stašiov, S.; Kubovčík, V. Development of physico-chemical and biological soil properties on the European ground squirrel mounds. Geoderma 2019, 339, 85–93. [Google Scholar] [CrossRef]
- Pepper, I.L.; Brusseau, M.L. Physical-chemical characteristics of soils and the subsurface. Environ. Pollut. Sci. 2019, 9–22. [Google Scholar] [CrossRef]
- Juhos, K.; Czigany, S.; Madarasz, B.; Ladanyi, M. Interpretation of soil quality indicators for land suitability assessment–A multivariate approach for Central European arable soils. Ecol. Indic. 2019, 99, 261–272. [Google Scholar] [CrossRef]
- Li, P.; Shi, K.; Wang, Y.; Kong, D.; Liu, T.; Jiao, J.; Liu, M.; Li, H.; Hu, F. Soil quality assessment of wheat-maize cropping system with different productivities in China: Establishing a minimum data set. Soil Tillage Res. 2019, 190, 31–40. [Google Scholar] [CrossRef]
- Francaviglia, R.; Renzi, G.; Ledda, L.; Benedetti, A. Organic carbon pools and soil biological fertility are affected by land use intensity in Mediterranean ecosystems of Sardinia, Italy. Sci. Total Environ. 2017, 599–600, 789–796. [Google Scholar] [CrossRef] [PubMed]
- Renzi, G.; Canfora, L.; Salvati, L.; Benedetti, A. Validation of the soil Biological Fertility Index (BFI) using a multidimensional statistical approach: A country-scale exercise. Catena 2017, 149, 294–299. [Google Scholar] [CrossRef]
- Laudicina, V.A.; Novara, A.; Barbera, V.; Egli, M.; Badalucco, L. Long-term tillage and cropping system effects on chemical and biochemical characteristics of soil organic matter in a Mediterranean semiarid environment. Land Degrad. Dev. 2015, 26, 45–53. [Google Scholar] [CrossRef]
- Soleimani, A.; Hosseini, S.M.; MassahBavani, A.R.; Jafari, M.; Francaviglia, R. Influence of land use and land cover change on soil organic carbon and microbial activity in the forests of northern Iran. Catena 2019, 177, 227–237. [Google Scholar] [CrossRef]
- Ahmed, I.U.; Mengistie, H.K.; Godbold, D.L.; Sanden, H. Soil moisture integrates the influence of land-use and season on soil microbial community composition in the Ethiopian highlands. Appl. Soil Ecol. 2019, 135, 85–90. [Google Scholar] [CrossRef]
- Kome, G.; Enang, R.; Yerima, B.; Lontsi, M. Models relating soil pH measurements in H2O, KCl and CaCl2 for volcanic ash soils of Cameroon. Geoderma Reg. 2018, 14, e00185. [Google Scholar] [CrossRef]
- Soil Survey Staff. Soil Survey Laboratory Information Manual; Soil Survey Investigations Report No. 45, Version 2.0.; USDA-Natural Resources Conservation Service: Washington, DC, USA, 2011.
- Walkley, A.; Black, I.A. An examination of the Degtjareff method for determining soil organic matter, and a proposed modification of the chromic acid titration method. Soil Sci. 1934, 37, 29–38. [Google Scholar] [CrossRef]
- Martínez, J.M.; Galantini, J.; Duval, M.; López, F.; Iglesias, J. Estimating soil organic carbon in Mollisols and its particle-size fractions by loss-on-ignition in the semiarid and semi humid Argentinean Pampas. Geoderma Reg. 2018, 12, 49–55. [Google Scholar] [CrossRef]
- Vance, E.D.; Brookes, P.C.; Jenkinson, D.S. An extraction method for measuring soil microbial biomass C. Soil Biol. Biochem. 1987, 9, 703–707. [Google Scholar] [CrossRef]
- Isermeyer, H. EineeinfacheMethodezurBestimmung der Bodenatmung und der KarbonateimBoden. J. Plant. Nutr. Soil Sci. 1952, 56, 26–38. [Google Scholar]
- Liu, X.; RezaeiRashti, M.; Dougall, A.; Esfandbod, M.; van Zwieten, L.; Chen, C. Subsoil application of compost improved sugarcane yield through enhanced supply and cycling of soil labile organic carbon and nitrogen in an acidic soil at tropical Australia. Soil Tillage Res. 2018, 180, 73–81. [Google Scholar] [CrossRef]
- Mganga, K.Z.; Razavi, B.S.; Kuzyakov, Y. Land use affects soil biochemical properties in Mt. Kilimanjaro region. Catena 2016, 141, 22–29. [Google Scholar] [CrossRef]
- Geyer, K.; Dijkstra, P.; Sinsabaugh, R.; Frey, S. Clarifying the interpretation of carbon use efficiency in soil through methods comparison. Soil Biol. Biochem. 2019, 128, 79–88. [Google Scholar] [CrossRef]
- Herlemann, D.P.R.; Labrenz, M.; Juergens, K.; Bertilsson, S.; Waniek, J.J.; Anderrson, A.F. Transition in bacterial communities along the 2000 km salinity gradient of the Baltic Sea. ISME J. 2011, 5, 1571–1579. [Google Scholar] [CrossRef] [PubMed]
- Pascazio, S.; Crecchio, C.; Scagliola, M.; Mininni, A.N.; Dichio, B.; Xiloyannis, C.; Sofo, A. Microbial-based soil quality indicators in irrigated and rainfed soil portions of Mediterranean olive and peach orchards under sustainable management. Agric. Water Manag. 2018, 195, 172–179. [Google Scholar] [CrossRef]
- Martin-Didonet, C.C.G.; Chubatsu, L.S.; Souza, E.M.; Kleina, M.; Rego, F.G.M.; Rigo, L.U.; Yates, M.G.; Pedrosa, F.O. Genome structure of the Genus Azospirillum. J. Bacteriol. 2000, 182, 4113–4116. [Google Scholar] [CrossRef]
- Wallenstein, M.D.; Vilgalys, R.J. Quantitative analyses of nitrogen cycling genes in soils. Pedobiologia 2005, 49, 665–672. [Google Scholar] [CrossRef]
- Kuczynski, J.; Stombaugh, J.; Walters, W.A.; González, A.; Caporaso, J.G.; Knight, R. Using QIIME to analyze 16S rRNA gene sequences from microbial communities. Curr. Protoc. Biol. 2011, 27. [Google Scholar] [CrossRef]
- Jat, H.; Datta, A.; Choudhary, M.; Yadav, A.; Choudhary, V.; Sharma, P.; Gathala, M.; Jat, M.; McDonald, A. Effects of tillage, crop establishment and diversification on soil organic carbon, aggregation, aggregate associated carbon and productivity in cereal systems of semi-arid Northwest India. Soil Tillage Res. 2019, 190, 128–138. [Google Scholar] [CrossRef]
- Qiu, Q.; Wu, L.; Li, B. Crop residue-derived dissolved organic matter accelerates the decomposition of native soil organic carbon in a temperate agricultural ecosystem. Acta Ecol.Sinica. 2019, 39, 69–76. [Google Scholar] [CrossRef]
- Novara, A.; Minacapilli, M.; Santoro, A.; Rodrigo-Comino, J.; Carrubba, A.; Sarno, M.; Venezia, G.; Gristina, L. Real cover crops contribution to soil organic carbon sequestration in sloping vineyard. Sci. Total Environ. 2019, 652, 300–306. [Google Scholar] [CrossRef] [PubMed]
- Ramesh, T.; Bolan, N.; Kirkham, M.; Wijesekara, H.; Kanchikerimath, M.; Srinivasa Rao, C.; Sandeep, S.; Rinklebe, J.; Ok, Y.; Choudhury, B.; et al. Soil organic carbon dynamics: Impact of land use changes and management practices: A review. Adv. Agron. 2019. [Google Scholar] [CrossRef]
- Safaei, M.; Bashari, H.; Mosaddeghi, M.; Jafari, R. Assessing the impacts of land use and land cover changes on soil functions using landscape function analysis and soil quality indicators in semi-arid natural ecosystems. Catena 2019, 177, 260–271. [Google Scholar] [CrossRef]
- Fujita, K.; Miyabara, Y.; Kunito, T. Microbial biomass and ecoenzymaticstoichiometries vary in response to nutrient availability in an arable soil. Eur. J. Soil Biol. 2019, 91, 1–8. [Google Scholar] [CrossRef]
- Denardin, L.; Carmona, F.; Veloso, M.; Martins, A.; Freitas, T.; Carlos, F.; Marcolin, É.; Camargo, F.; Anghinoni, I. No-tillage increases irrigated rice yield through soil quality improvement along time. Soil Tillage Res. 2019, 186, 64–69. [Google Scholar] [CrossRef]
- Hao, M.; Hu, H.; Liu, Z.; Dong, Q.; Sun, K.; Feng, Y.; Li, G.; Ning, T. Shifts in microbial community and carbon sequestration in farmland soil under long-term conservation tillage and straw returning. Appl. Soil Ecol. 2019, 136, 43–54. [Google Scholar] [CrossRef]
- González-Ubierna, S.; Lai, R. Modelling the effects of climate factors on soil respiration across Mediterranean ecosystems. J. Arid Environ. 2019. [Google Scholar] [CrossRef]
- Malik, A.A.; Puissant, J.; Buckeridge, K.M.; Goodall, T.; Jehmlich, N.; Chowdhuty, S.; Gweon, H.S.; Peyton, J.M.; Mason, K.E.; Agtmaal, M.V.; et al. Land use driven change in soil pH affects microbial carbon cycling processes. Nat. Commun. 2018, 9, 3591. [Google Scholar] [CrossRef]
- Van Wesemael, B.; Chartin, C.; Wiesmeier, M.; von Lützow, M.; Hobley, E.; Carnol, M.; Krüger, I.; Campion, M.; Roisin, C.; Hennart, S.; et al. An indicator for organic matter dynamics in temperate agricultural soils. Agric. Ecosyst. Environ. 2019, 274, 62–75. [Google Scholar] [CrossRef]
- Nannipieri, P.; Ascher, J.; Ceccherini, M.; Landi, L.; Pietramellara, G.; Renella, G. Microbial diversity and soil functions. Eur.J. Soil Sci. 2003, 54, 655–670. [Google Scholar] [CrossRef]
- Nsabimana, D.; Haynes, R.J.; Wallis, F.M. Size, activity and catabolic diversity of the soil microbial biomass as affected by land use. Appl. Soil Ecol. 2004, 26, 81–92. [Google Scholar] [CrossRef]
- Mocali, S.; Paffetti, D.; Emiliani, G.; Benedetti, A.; Fani, R. Diversity of heterotrophic aerobic cultivable microbial communities of soils treated with fumigants and dynamics of metabolic, microbial, and mineralization quotients. Biol. Fertil. Soils. 2008, 44, 557–569. [Google Scholar] [CrossRef]
- Birge, H.E. What Happens During Soil Incubation? Exploring Microbial Biomass, Carbon AVAILABILITY and temperature Constraints on Soil Respiration. Master’s Thesis, Colorado State University, Fort Collins, CO, USA, 2013. [Google Scholar]
- Dungait, J.A.; Hopkins, D.W.; Gregory, A.S.; Whitmore, A.P. Soil organic matter turnover is governed by accessibility not recalcitrance. Glob. Chang. Biol. 2012, 18, 1781–1796. [Google Scholar] [CrossRef]
- Basso, F.; Bove, E.; Dumontet, S.; Ferrara, A.; Pisante, M.; Quaranta, G.; Taberner, M. Evaluating environmental sensitivity at the basin scale through the use of geographic information systems and remote sensed data: An example covering the Agri basin (southern Italy). Catena 2000, 40, 19–35. [Google Scholar] [CrossRef]
- Willy, D.; Muyanga, M.; Mbuvi, J.; Jayne, T. The effect of land use change on soil fertility parameters in densely populated areas of Kenya. Geoderma 2019, 343, 254–262. [Google Scholar] [CrossRef] [PubMed]
- Ghimire, B.; Ghimire, R.; VanLeeuwen, D.; Mesbah, A. Cover crop residue amount and quality effects on soil organic carbon mineralization. Sustainability 2017, 9, 2316. [Google Scholar] [CrossRef]
- Papini, R.; Valboa, G.; Favilli, F.; L’Abate, G. Influence of land use on organic carbon pool and chemical properties of VerticCambisols in central and southern Italy. Agric. Ecosyst. Environ. 2011, 140, 68–79. [Google Scholar] [CrossRef]
- Wang, J.; Liu, G.; Zhang, C.; Wang, G.; Fang, L.; Cui, Y. Higher temporal turnover of soil fungi than bacteria during long-term secondary succession in a semiarid abandoned farmland. Soil Tillage Res. 2019, 194, 104305. [Google Scholar] [CrossRef]
- Sainju, U.M.; Lenssen, A.W.; Allen, B.L.; Stevens, W.B.; Jabro, J.D. Nitrogen balance in response to dryland crop rotations and cultural practices. Agric. Ecosyst. Environ. 2016, 233, 25–32. [Google Scholar] [CrossRef]
- Agnieszka, W.; Zofia, S.; Aleksandra, B.; Artur, B. Evaluation of factors influencing the biomass of soil microorganisms and DNA content. Open J. Soil Sci. 2012, 2, 64–69. [Google Scholar] [CrossRef]
- Kallenbach, C.M.; Frey, S.D.; Grandy, A.S. Direct evidence for microbialderived soil organic matter formation and its ecophysiological controls. Nat. Commun. 2016, 7, 13630. [Google Scholar] [CrossRef] [PubMed]
- Männistö, M.; Ganzert, L.; Tiirola, M.; Haggblom, M.M.; Stark, S. Do shifts in life strategies explain microbial community responses to increasing nitrogen in tundra soil? Soil Biol. Biochem. 2016, 96, 216–228. [Google Scholar] [CrossRef]
- Fierer, N.; Bradford, M.A.; Jackson, R.B. Toward an ecological classification of soil bacteria. Ecology 2007, 88, 354–1364. [Google Scholar] [CrossRef]
- Fontaine, S.; Mariotti, A.; Abbadie, L. The priming effect of organic matter: A question of microbial competition? Soil Biol. Biochem. 2003, 35, 837–843. [Google Scholar] [CrossRef]
- Jiang, X.; Wright, A.L.; Wang, X.; Liang, F. Tillage-induced changes in fungal and bacterial biomass associated with soil aggregates: A long-term field study in a subtropical rice soil in China. Appl. Soil Ecol. 2011, 48, 168–173. [Google Scholar] [CrossRef]
- Nazaries, L.; Tottey, W.; Robinson, L.; Khachane, A.; Abu-al-Soud, W.; Sorensen, S.; Singh, B.K. Shifts in the microbial community structure explain the response of soil respiration to land-use change but not to climate warming. Soil Biol. Biochem. 2015, 89, 123–134. [Google Scholar] [CrossRef]
- Navarro-Noya, Y.E.; Gómez-Acata, S.; Montoya-Ciriaco, N.; Rojas-Valdez, A.; Suárez-Arriaga, M.C.; Valenzuela-Encinas, S.; Jiménez-Bueno, N.; Verhulst, N.; Govaerts, B.; Dendooven, L. Relative impacts of tillage, residue management and crop-rotation on soil bacterial communities in a semi-arid agroecosystem. Soil Biol. Biochem. 2013, 65, 86–95. [Google Scholar] [CrossRef]
- Rincon-Florez, V.A.; Clement, N.G.; Dang, Y.; Schenk, P.M.; Carvalhais, L.C. Short-term impact of an occasional tillage on microbial communities in a Vertosol after 43 years of no-tillage or conventional tillage. Eur. J. Soil Biol. 2016, 74, 32–38. [Google Scholar] [CrossRef]
- Li, J.; Wu, X.; Gebremikael, M.T.; Wu, H.; Cai, D. Response of soil organic carbon fractions, microbial community composition and carbon mineralization to high-input fertilizer practices under an intensive agricultural system. PLoS ONE 2018, 13, e0195144. [Google Scholar] [CrossRef]
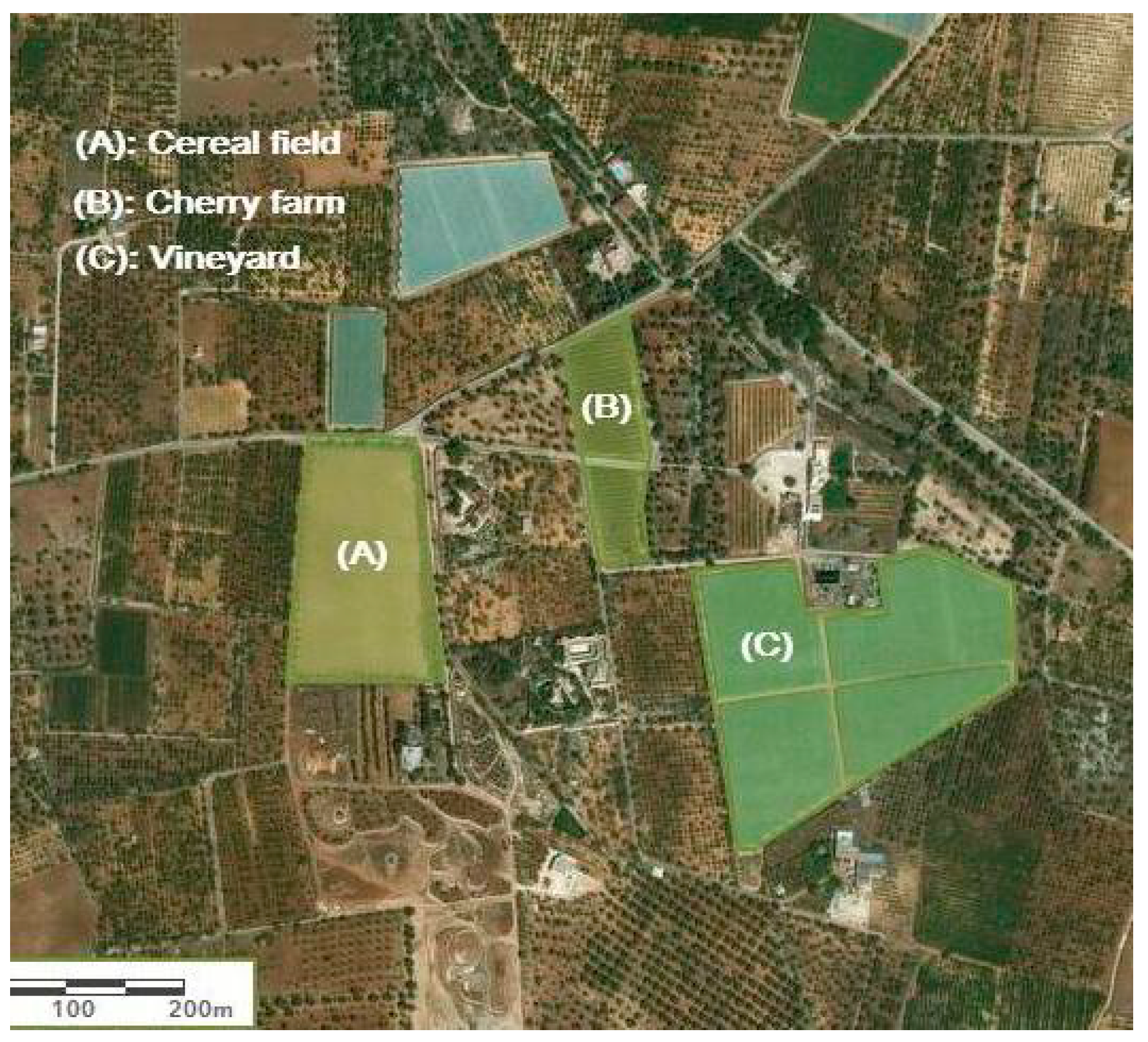
| Land Use | Area (Ha) | Cultivation History (Year) | Management | Irrigation and Fertilization |
|---|---|---|---|---|
| Cereal field | 2.4 | 7 | Soil tillage (40 cm), residues removal from the field, integrated pest management, | No irrigation (dryland farming), chemical fertilization before seeding |
| Grass-covered vineyard | 4.9 | 10 | No tillage, residues left on the field, integrated pest management, | Drip irrigation with continuous mixed fertilization (chemical–organic) |
| Cherry farm | 1.2 | 10 | Soil tillage (40 cm), residues removal from the farm, integrated pest management | Drip irrigation with continuous chemical fertilization |
| Parameters | Scores | ||||
|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | |
| Soil organic matter, SOM (%) | <1.0 | ≥1.0 | >1.5 | >2.0 | >3.0 |
| ≤1.5 | ≤2.0 | ≤3.0 | |||
| Basal soil respiration, BSR (mg CO2–C kg−1 soil d−1) | <5 | ≥5 | >10 | >15 | >20 |
| ≤10 | ≤15 | ≤20 | |||
| Cumulative soil respiration, CSR (mg CO2–C kg−1 soil) | <100 | ≥100 | >250 | >400 | >600 |
| ≤250 | ≤400 | ≤600 | |||
| Microbial biomass carbon, MBC (mg C kg−1 soil) | <100 | ≥100 | >200 | >300 | >400 |
| ≤200 | ≤300 | ≤400 | |||
| Metabolic quotient, qCO2 (mg CO2–C 10−2 h−1 mg MBC −1) | ≥0.4 | ≥0.4 | <0.3 | <0.2 | <0.1 |
| ≤0.3 | ≥0.2 | ≥0.1 | |||
| Mineralization quotient, qM (%) | <1.0 | ≥1 | >2 | >3 | >4 |
| ≤2 | ≤3 | ≤4 | |||
| Classes of the Biological Fertility Index (BFI) | |||||
| Fertility class | I | II | III | IV | V |
| Stress | Pre-stress (alarm) | Medium | Good | High | |
| BFI scores sum | 6 | 7–12 | 13–18 | 19–24 | 25–30 |
| Parameter | Unit | Land Use | ||
|---|---|---|---|---|
| Cereal Field | Grass-Covered Vineyard | Cherry Farm | ||
| Soil pH | 7.95 ± 0.04 | 7.95 ± 0.01 | 7.98 ± 0.04 | |
| Soil texture | Sandy Clay Loam | |||
| SOC | % | 1.69 ± 0.17 a | 1.82 ± 0.13 a | 1.36 ± 0.11 b |
| SOM | % | 2.91 ± 0.29 a | 3.13 ± 0.22 a | 2.35 ± 0.19 b |
| MBC | μg C g−1 soil | 197.08 ± 18.76 a | 201.92 ± 20.81 a | 186.45 ± 14.19 b |
| CUE | μg C g−1 soil | 117.06 ± 1.14 b | 110.53 b ± 4.96 | 138.08 a ± 12.98 |
| BSR | mg CO2–C kg−1 soil d−1 | 9.73 ± 0.87 b | 11.84 ± 0.66 a | 10.51 ± 1.11 ab |
| CSR | mg CO2–C kg−1 soil | 153.91 ± 10.77 b | 214.49 ± 13.69 a | 226.90 ± 18.88 a |
| qCO2 | mg CO2–C 10−2 h−1 mg MBC−1 | 0.050 ± 0.004 b | 0.058 ± 0.005 a | 0.056 a ± 0.004 |
| qM | % | 0.91 ± 0.08 c | 1.17 ± 0.10 b | 1.66 ± 0.15 a |
| Nucleic acid concentration | ngµL−1 0.5 g−1 | 154.8 ± 6.01 b | 160.6 ± 5.49 a | 143.7 ±5.94 c |
| 16S rRNA- CN | g−1 DS | 8.06 ± 1.28 × 107 a | 1.82 ± 0.51 × 107 c | 5.18± 0.32 × 107 b |
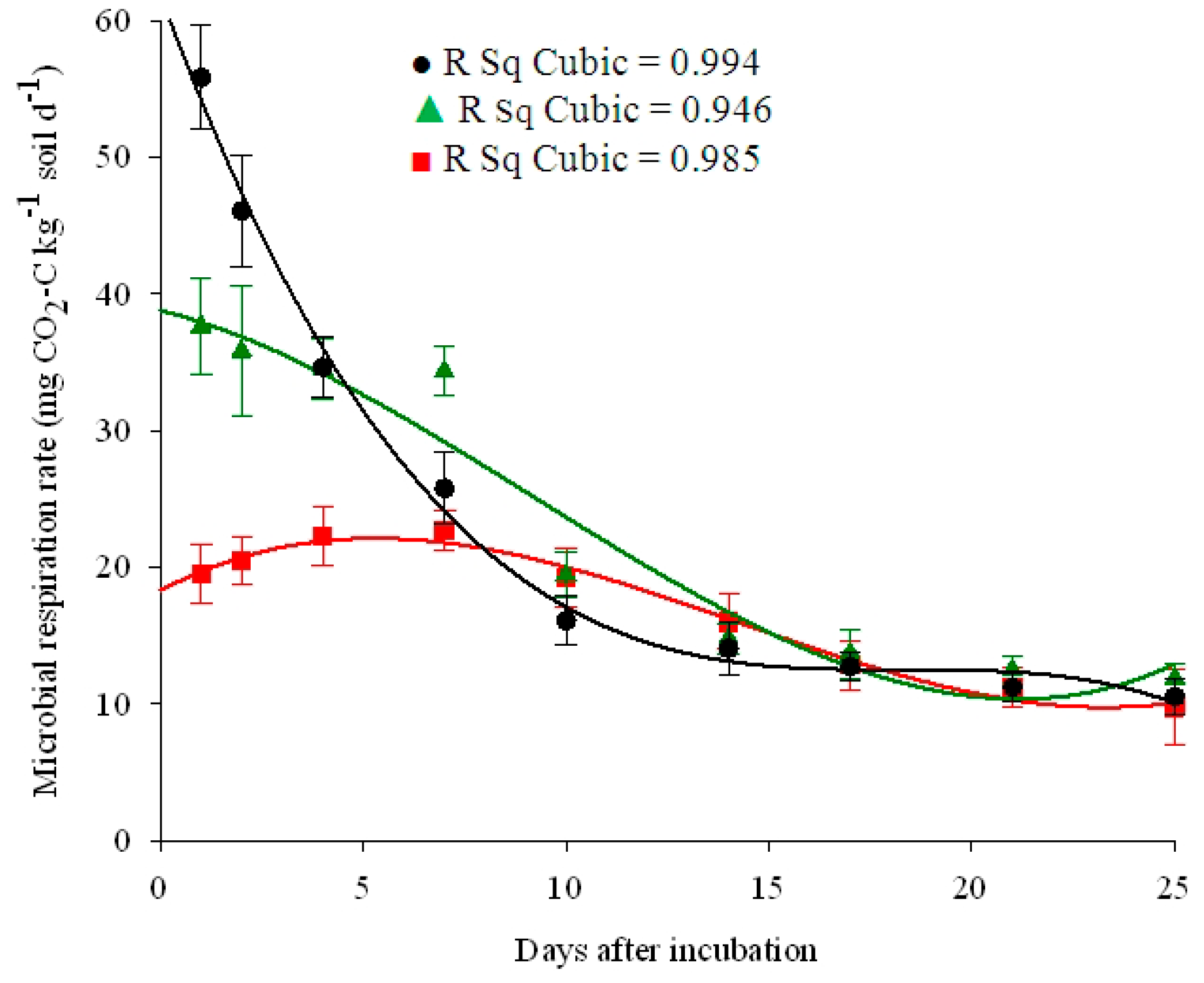
| Land Use | Equation | R Square | Constant | b1 | b2 | b3 |
|---|---|---|---|---|---|---|
| Cereal field | Cubic | 0.98 ** | 18.28 | 1.57 | −0.18 | 0.004 |
| Grass-covered vineyard | Cubic | 0.95 ** | 38.79 | −0.75 | −0.12 | 0.004 |
| Cherry farm | Cubic | 0.99 ** | 61.92 | −8.11 | 0.44 | −0.008 |
| Land Use | SOM | BSR | CSR | MBC | qCO2 | qM | BFI Score | BFI Class |
|---|---|---|---|---|---|---|---|---|
| Cereal field | 4 | 2 | 2 | 2 | 5 | 1 | 16 | III (medium) |
| Grass-covered vineyard | 5 | 3 | 2 | 3 | 5 | 2 | 20 | IV (good) |
| Cherry farm | 4 | 3 | 2 | 2 | 5 | 2 | 18 | III (medium) |
| Model | Independent Variables | Standardized Coefficients (Beta) | t | Significance Level | |
|---|---|---|---|---|---|
| 1 | (Constant) | 1.059 | 0.401 | ||
| SOM | 1.119 | 0.932 | 0.450 | ||
| BSR | 0.501 | 0.603 | 0.608 | ||
| CSR | 1.230 | 1.352 | 0.309 | ||
| MBC | −1.411 | −0.550 | 0.638 | ||
| qCO2 | −1.448 | −0.625 | 0.596 | ||
| qM | −0.466 | −0.980 | 0.430 | ||
| 2 | (Constant) | 3.756 | 0.033 | ||
| SOM | 0.519 | 1.181 | 0.323 | ||
| BSR | 0.208 | 0.372 | 0.735 | ||
| CSR | 0.792 | 2.061 | 0.131 | ||
| qCO2 | −0.206 | −0.454 | 0.680 | ||
| qM | −0.426 | −1.035 | 0.377 | ||
| 3 | (Constant) | 4.827 | 0.008 | ||
| SOM | 0.650 | 2.782 | 0.050 | ||
| CSR | 0.896 | 3.835 | 0.019 | ||
| qCO2 | −0.046 | −0.363 | 0.735 | ||
| qM | −0.445 | −1.229 | 0.286 | ||
| 4 | (Constant) | 5.318 | 0.003 | ||
| SOM | 0.641 | 3.035 | 0.029 | ||
| CSR | 0.903 | 4.269 | 0.008 | ||
| qM | −0.484 | −1.544 | 0.183 | ||
| 5 | (Constant) | 5.703 | 0.001 | ||
| SOM | 0.931 | 8.765 | 0.000 | ||
| CSR | 0.612 | 5.761 | 0.001 | ||
| Model Summary | |||||
| Model | R | R2 | Adjusted R2 | Std. Error of the Estimate | |
| 1 | 0.983 | 0.966 | 0.863 | 0.46969 | |
| 2 | 0.980 | 0.961 | 0.895 | 0.41147 | |
| 3 | 0.979 | 0.959 | 0.918 | 0.36447 | |
| 4 | 0.978 | 0.957 | 0.932 | 0.33132 | |
| 5 | 0.968 | 0.937 | 0.916 | 0.36752 | |
| Parameters | SOM | BSR | CSR | MBC | qCO2 | qM | Nucleic Acid Concentration | 16S rRNA- CN |
|---|---|---|---|---|---|---|---|---|
| SOM | 1 | |||||||
| BSR | 0.25 ns | 1 | ||||||
| CSR | −0.27 ns | 0.60 ns | 1 | |||||
| MBC | 0.78 * | −0.10 ns | −0.09 ns | 1 | ||||
| qCO2 | −0.41 ns | 0.68 * | 0.44 ns | −0.79 * | 1 | |||
| qM | −0.76 * | 0.27 ns | 0.76 * | −0.58 ns | 0.59 ns | 1 | ||
| Nucleic acid concentration | 0.91 ** | 0.31 ns | −0.21 ns | 0.68 * | −0.33 ns | −0.69 * | 1 | |
| 16S rRNA- CN | −0.15 ns | −0.71 * | −0.68 * | −0.00 ns | −0.45 ns | −0.25 ns | −0.27 ns | 1 |
| Land Use | CHAO1 | Simpson | Shannon | Goods-Coverage |
|---|---|---|---|---|
| Cherry farm | 4882.3 ± 108.95 a | 0.85 ± 0.00 a | 9.93 ± 0.8 a | 0.99 ± 0.00 a |
| Cereal field | 4558.8 ± 97.04 a | 0.87 ± 0.00 a | 9.84 ± 0.05 a | 0.99 ± 0.00 a |
| Grass-covered vineyard | 4973.2 ± 61.94 a | 0.85 ± 0.00 a | 10.14 ± 0.4 a | 0.99 ± 0.00 a |
| (A) | Land Use | |||
| Phylum | Cherry Farm (%) | Cereal Field (%) | Grass-Covered Vineyard (%) | |
| Proteobacteria | 26.38 ± 0.58 b | 29.29 ± 0.79 a | 26.43 ± 0.23 b | |
| Actinobacteria | 22.88 ± 0.25 b | 23.71 ± 1.05 b | 25.51 ± 1.41 a | |
| Bacteroidetes | 7.58 ± 0.99 a | 8.82 ± 0.46 a | 6.88 ± 0.35 a | |
| Planctomycetes | 5.03 ± 0.19 a | 4.13 ± 0.21 ab | 3.98 ± 0.28 b | |
| Verrucomicrobia | 4.67 ± 0.11 ab | 5.28 ± 0.51 a | 3.09 ± 0.11 b | |
| Chloroflexi | 4.33 ± 0.66 a | 3.85 ± 0.27 a | 3.89 ± 0.23 a | |
| Firmicutes | 3.25 ± 0.39 ab | 2.32 ± 0.20 b | 3.45 ± 0.14 a | |
| Gemmatimonadetes | 2.00 ± 0.04 a | 1.31 ± 0.05 c | 1.64 ± 0.10 b | |
| Acidobacteria | 1.23 ± 0.07 b | 0.95 ± 0.03 c | 1.38 ± 0.02 a | |
| (B) | ||||
| Phylum | Family | Cherry Farm (%) | Cereal Field (%) | Grass-Covered Vineyard (%) |
| Actinobacteria | Rubrobacteraceae | 7.20 ± 0.35 b | 6.09 ± 0.27 b | 8.68 ± 0.28 a |
| Proteobacteria | Bradyrhizobiaceae | 2.33 ± 0.29 a | 2.56 ± 0.08 a | 2.78 ± 0.13 a |
| Firmicutes | Bacillaceae | 2.10 ± 0.31 a | 1.23 ± 0.14 b | 2.02 ± 0.16 a |
| Actinobacteria | Solirubrobacteraceae | 2.56 ± 0.58 a | 1.87 ± 0.02 b | 2.42 ± 0.19 a |
| Bacteroidetes | Chitinophagaceae | 2.75 ± 0.24 b | 2.81 ± 0.02 b | 3.87 ± 0.32 a |
| Planctomycetes | Gemmataceae | 2.55 ± 0.04 a | 1.26 ± 0.10 b | 1.61 ± 0.13 b |
| Gemmatimonadetes | Gemmatimonadaceae | 2.00 ± 0.04 a | 1.31 ± 0.05 c | 1.64 ± 0.10 b |
| Proteobacteria | Rhodospirillaceae | 1.66 ± 0.08 b | 1.97 ± 0.04 ab | 2.59 ± 0.17 a |
| Proteobacteria | Sinobacteraceae | 2.04 ± 0.08 a | 2.11 ± 0.22 a | 1.37 ± 0.12 b |
| Verrucomicrobia | Pedosphaeraceae | 2.31 ± 0.33 a | 1.65 ± 0.05 b | 8.68 ± 0.28 a |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yaghoubi Khanghahi, M.; Murgese, P.; Strafella, S.; Crecchio, C. Soil Biological Fertility and Bacterial Community Response to Land Use Intensity: A Case Study in the Mediterranean Area. Diversity 2019, 11, 211. https://doi.org/10.3390/d11110211
Yaghoubi Khanghahi M, Murgese P, Strafella S, Crecchio C. Soil Biological Fertility and Bacterial Community Response to Land Use Intensity: A Case Study in the Mediterranean Area. Diversity. 2019; 11(11):211. https://doi.org/10.3390/d11110211
Chicago/Turabian StyleYaghoubi Khanghahi, Mohammad, Pasqua Murgese, Sabrina Strafella, and Carmine Crecchio. 2019. "Soil Biological Fertility and Bacterial Community Response to Land Use Intensity: A Case Study in the Mediterranean Area" Diversity 11, no. 11: 211. https://doi.org/10.3390/d11110211
APA StyleYaghoubi Khanghahi, M., Murgese, P., Strafella, S., & Crecchio, C. (2019). Soil Biological Fertility and Bacterial Community Response to Land Use Intensity: A Case Study in the Mediterranean Area. Diversity, 11(11), 211. https://doi.org/10.3390/d11110211





