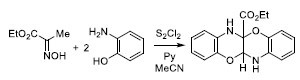
Ethyl 11a,12-Dihydrobenzo[b]benzo[5,6][1,4]oxazino[2,3-e][1,4]oxazine-5a(6H)-carboxylate
Abstract
1. Introduction
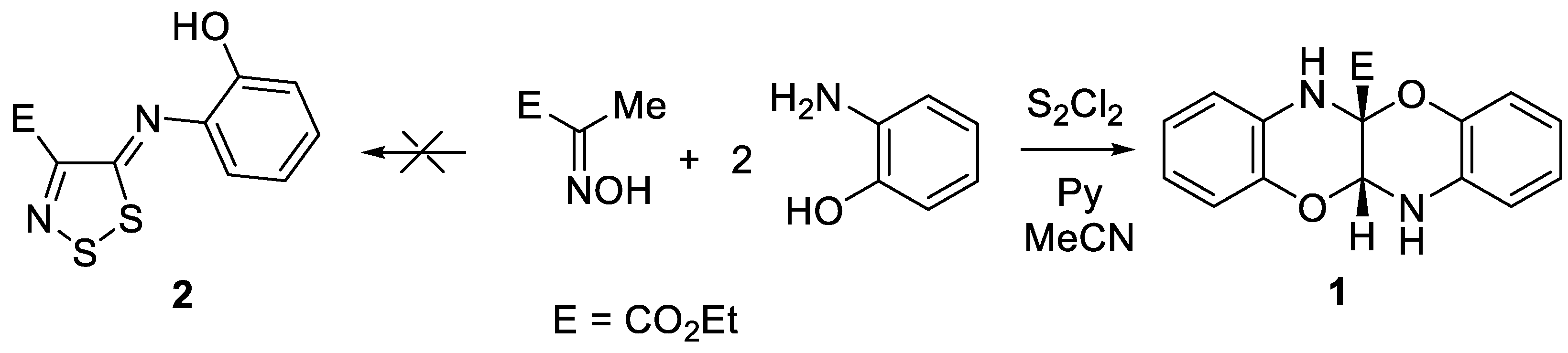
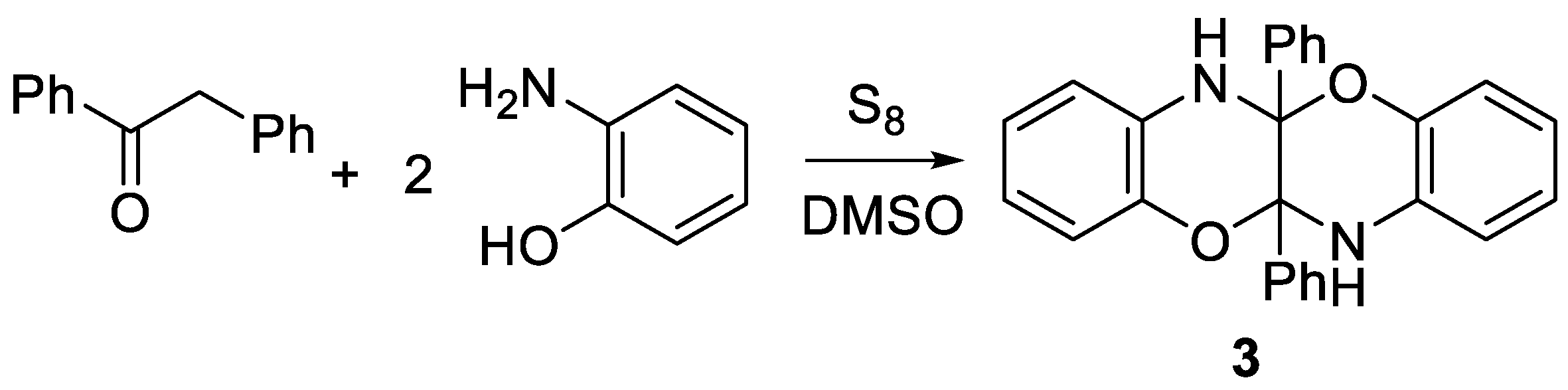
2. Results and Discussion
3. Experimental Section
3.1. General Information
3.2. Synthesis of Ethyl 11a,12-Dihydrobenzo[b]benzo[5,6][1,4]oxazino[2,3-e][1,4]oxazine-5a(6H)-carboxylate 1
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Konstantinova, L.S.; Rakitin, O.A. Sulfur monochloride in organic synthesis. Russ. Chem. Rev. 2014, 83, 225–250. [Google Scholar] [CrossRef]
- Konstantinova, L.S.; Rakitin, O.A. Design of sulfur heterocycles with sulfur monochloride: Retrosynthetic analysis and prospects. Mendeleev Commun. 2009, 19, 55–61. [Google Scholar] [CrossRef]
- Konstantinova, L.S.; Rakitin, O.A. Sulfur monochloride in the synthesis of heterocyclic compounds. Adv. Heterocycl. Chem. 2008, 96, 175–229. [Google Scholar] [CrossRef]
- Konstantinova, L.S.; Bol’shakov, O.I.; Obruchnikova, N.V.; Laborie, H.; Tonga, A.; Sopéna, V.; Lanneluc, I.; Picot, L.; Sablé, s.; Thiéry, V.; et al. One-pot synthesis of 5-phenylimino, 5-thione and 5-one-1,2,3-dithiazoles and evaluation of their antimicrobial and antitumor activity. Bioorg. Med. Chem. Lett. 2009, 19, 136–141. [Google Scholar] [CrossRef] [PubMed]
- Baranovsky, I.V.; Konstantinova, L.S.; Tolmachev, M.A.; Popov, V.V.; Lyssenko, K.A.; Rakitin, O.A. Synthesis of 2-((2-(benzo[d]oxazol-2-yl)-2H-imidazol-4-yl)amino)-phenols from 2-((5H-1,2,3-dithiazol-5-ylidene)amino)phenols through Unprecedented Formation of Imidazole Ring from Two Methanimino Groups, N. D. Zelinsky Institute of Organic Chemistry Russian Academy of Sciences: Moscow, Russia, 2020; unpublished work.
- Nguyen, T.B.; Retailleau, P. Sulfur-catalyzed stereo and regioselective synthesis of heteropropellanes via oxidative condensation of cyclohexanones with 2-aminophenols. Adv. Synth. Catal. 2019, 361, 3588–3592. [Google Scholar] [CrossRef]
- Dilmaç, M.; Spuling, E.; de Meijere, A.; Bräse, S. Propellanes—from a chemical curiosity to “explosive” materials and natural products. Angew. Chem. Int. Ed. 2017, 56, 5684–5718. [Google Scholar] [CrossRef] [PubMed]
- Krause, L.; Herbst-Irmer, R.; Sheldrick, G.M.; Stalke, D. Comparison of silver and molybdenum microfocus X-ray sources for single-crystal structure determination. J. Appl. Cryst. 2015, 48, 3–10. [Google Scholar] [CrossRef] [PubMed]

| Empirical Formula | C17H16N2O4 |
|---|---|
| Formula weight | 312.32 |
| Temperature | 100(2) K |
| Wavelength | 0.71073 Å |
| Crystal system | Orthorhombic |
| Space group | P212121 |
| Unit cell dimensions | a = 5.5972(2) Å b = 15.0183(6) Å c = 16.7717(6) Å |
| Volume | 1409.84(9) Å3 |
| Z | 4 |
| Density (calculated) | 1.471 g/cm3 |
| Absorption coefficient | 0.106 mm−1 |
| F(000) | 656 |
| Crystal size | 0.59 × 0.10 × 0.09 mm3 |
| Theta range for data collection | 2.712 to 33.155° |
| Reflections collected | 34967 |
| Independent reflections | 5372 [R(int) = 0.0477] |
| Completeness to theta = 25.242° | 99.8% |
| Absorption correction | Semi-empirical from equivalents |
| Max. and min. transmission | 0.8499 and 0.8299 |
| Refinement method | Full-matrix least-squares on F2 |
| Data/restraints/parameters | 5372/0/272 |
| Goodness-of-fit on F2 | 1.040 |
| Final R indices [I > 2sigma(I)] | R1 = 0.0345, wR2 = 0.0855 |
| R indices (all data) | R1 = 0.0423, wR2 = 0.0914 |
| Absolute structure parameter | −0.2(3) |
| Largest diff. peak and hole | 0.384 and −0.201 e·Å−3 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Konstantinova, L.S.; Tolmachev, M.A.; Popov, V.V.; Rakitin, O.A. Ethyl 11a,12-Dihydrobenzo[b]benzo[5,6][1,4]oxazino[2,3-e][1,4]oxazine-5a(6H)-carboxylate. Molbank 2020, 2020, M1149. https://doi.org/10.3390/M1149
Konstantinova LS, Tolmachev MA, Popov VV, Rakitin OA. Ethyl 11a,12-Dihydrobenzo[b]benzo[5,6][1,4]oxazino[2,3-e][1,4]oxazine-5a(6H)-carboxylate. Molbank. 2020; 2020(3):M1149. https://doi.org/10.3390/M1149
Chicago/Turabian StyleKonstantinova, Lidia S., Mikhail A. Tolmachev, Vadim V. Popov, and Oleg A. Rakitin. 2020. "Ethyl 11a,12-Dihydrobenzo[b]benzo[5,6][1,4]oxazino[2,3-e][1,4]oxazine-5a(6H)-carboxylate" Molbank 2020, no. 3: M1149. https://doi.org/10.3390/M1149
APA StyleKonstantinova, L. S., Tolmachev, M. A., Popov, V. V., & Rakitin, O. A. (2020). Ethyl 11a,12-Dihydrobenzo[b]benzo[5,6][1,4]oxazino[2,3-e][1,4]oxazine-5a(6H)-carboxylate. Molbank, 2020(3), M1149. https://doi.org/10.3390/M1149