MTAP Deletion as a Therapeutic Vulnerability in Cancer: From Molecular Mechanism to Clinical Targeting
Abstract
1. Introduction
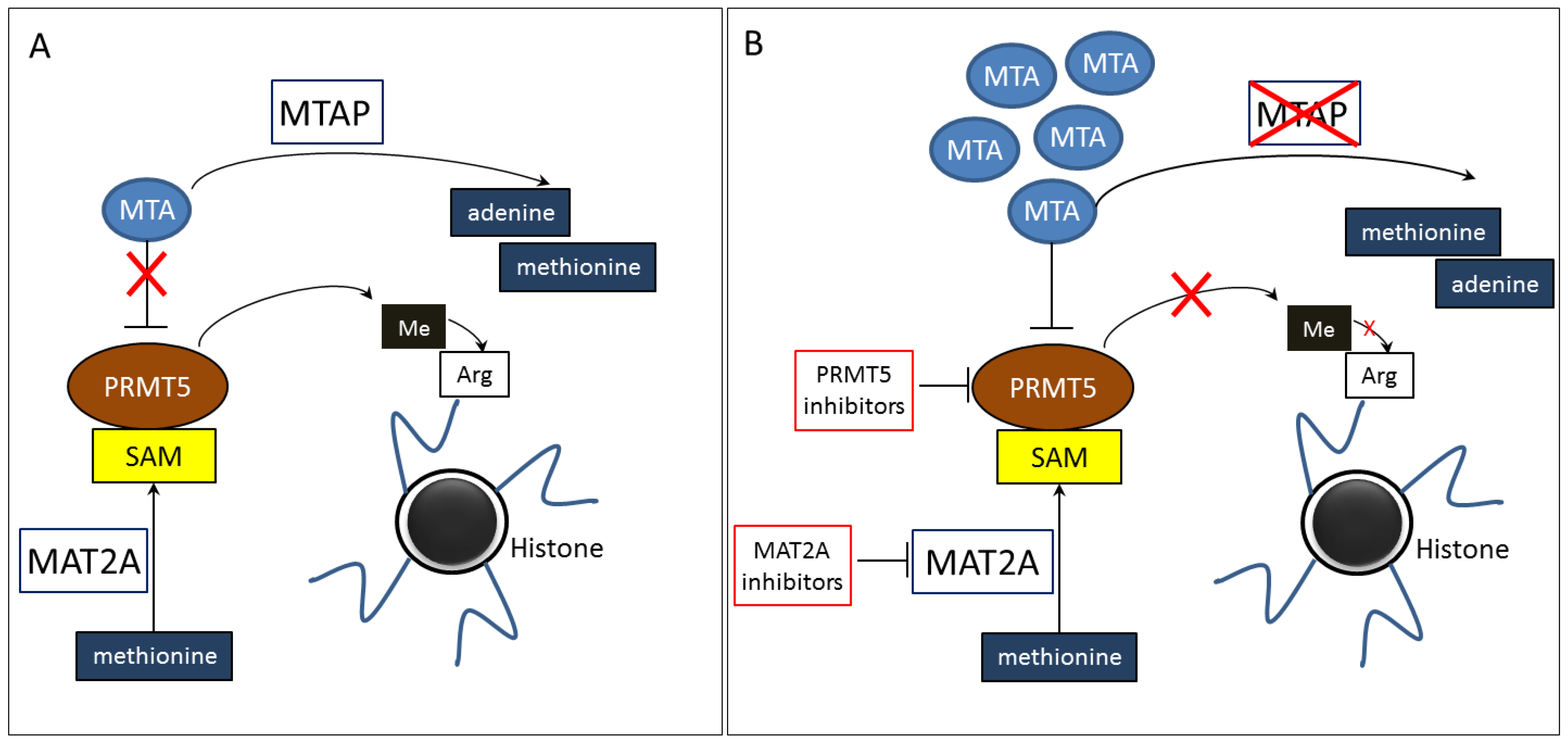
1.1. The Molecular Framework of MTAP-Associated Regulation
1.2. Abnormalities in MTAP Gene
2. Diagnosis of MTAP Gene Deletion
3. Molecularly Targeted Therapies for Cancer Patients with MTAP Gene Deletion
4. PRMT5 Inhibitors in Clinical Trials
5. MAT2A Inhibitors in Clinical Trials
6. Summary
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Backlund, P.S.; Smith, R.A. Methionine synthesis from 5′-methylthioadenosine in rat liver. J. Biol. Chem. 1981, 256, 1533–1535. [Google Scholar] [CrossRef]
- Kirovski, G.; Stevens, A.P.; Czech, B.; Dettmer, K.; Weiss, T.S.; Wild, P.; Hartmann, A.; Bosserhoff, A.K.; Oefner, P.J.; Hellerbrand, C. Down-regulation of methylthioadenosine phosphorylase (MTAP) induces progression of hepatocellular carcinoma via accumulation of 5′-deoxy-5′-methylthioadenosine (MTA). Am. J. Pathol. 2011, 178, 1145–1152. [Google Scholar] [CrossRef]
- Mavrakis, K.J.; McDonald, E.R.; Schlabach, M.R.; Billy, E.; Hoffman, G.R.; Deweck, A.; Ruddy, D.A.; Venkatesan, K.; Yu, J.; McAllister, G.; et al. Disordered methionine metabolism in MTAP/CDKN2A-deleted cancers leads to dependence on PRMT5. Science 2016, 351, 1208–1213. [Google Scholar] [CrossRef]
- Brune, M.M.; Prince, S.S.; Vlajnic, T.; Chijioke, O.; Roma, L.; König, D.; Bubendorf, L. MTAP as an emerging biomarker in thoracic malignancies. Lung Cancer 2024, 197, 107963. [Google Scholar] [CrossRef]
- Bistulfi, G.; Affronti, H.C.; Foster, B.A.; Karasik, E.; Gillard, B.; Morrison, C.; Mohler, J.; Phillips, J.G.; Smiraglia, D.J. The essential role of methylthioadenosine phosphorylase in prostate cancer. Oncotarget 2016, 7, 14380–14393. [Google Scholar] [CrossRef]
- Pawan, C.P.K.; Biswas, N.; Pingle, S.C.; Lin, F.; Anekoji, M.; Jones, L.D.; Kesari, S.; Wang, F.; Ashili, S. MTAP loss: A possible therapeutic approach for glioblastoma. J. Transl. Med. 2022, 20, 620. [Google Scholar] [CrossRef]
- Li, C.; Gui, G.; Zhang, L.; Qin, A.; Zhou, C.; Zha, X. Overview of methionine adenosyltransferase 2A (MAT2A) as an anticancer target: Structure, function, and inhibitors. J. Med. Chem. 2022, 65, 9531–9547. [Google Scholar] [CrossRef]
- Ikushima, H.; Watanabe, K.; Shinozaki-Ushiku, A.; Oda, K.; Kage, H. Pan-cancer clinical and molecular landscape of MTAP deletion in nationwide and international comprehensive genomic data. ESMO Open 2025, 10, 104535. [Google Scholar] [CrossRef]
- Kryukov, G.V.; Wilson, F.H.; Ruth, J.R.; Paulk, J.; Tsherniak, A.; Marlow, S.E.; Vazquez, F.; Weir, B.A.; Fitzgerald, M.E.; Tanaka, M.; et al. MTAP deletion confers enhanced dependency on the arginine methyltransferase PRMT5 in human cancer cells. Science 2016, 351, 1214–1218. [Google Scholar] [CrossRef]
- Mauri, G.; Patelli, G.; Roazzi, L.; Valtorta, E.; Amatu, A.; Marrapese, G.; Bonazzina, E.; Tosi, F.; Bencardino, K.; Ciarlo, G.; et al. Clinicopathological characterization of MTAP alterations in gastrointestinal cancers. J. Clin. Pathol. 2025, 78, 195–201. [Google Scholar] [CrossRef]
- Clouser, M.C.; Suh, M.; Movva, N.; Hildebrand, J.S.; Pastula, S.T.; Schoehl, M.; Hindoyan, A.; Balasubramanian, A.; Fryzek, J.P.; Yang, S.-R. A systematic literature review of MTAP deletions in solid and hematologic cancers. Cancer Treat. Res. Commun. 2025, 44, 100966. [Google Scholar] [CrossRef]
- Brune, M.M.; Roma, L.; Chijioke, O.; Alborelli, I.; Zacharias, M.; Bubendorf, L.; Vlajnic, T.; Deigendesch, N.; Pollinger, J.; Hirschmann, P.; et al. MTAP expression by immunohistochemistry: A novel biomarker in NSCLC. J. Thorac. Oncol. 2025. [Google Scholar] [CrossRef]
- Bray, C.; Ballcells, C.; McNeish, I.A.; Keun, H.C. The potential and challenges of targeting MTAP-negative cancers beyond synthetic lethality. Front. Oncol. 2023, 13, 1264785. [Google Scholar] [CrossRef]
- Chang, W.; Hsu, S.; Zhang, J.; Li, J.; Yang, D.D.; Chu, C.; Yoo, E.E.; Zhang, W.; Yu, S.; Chen, C. MTAP deficiency contributes to immune landscape remodeling and tumor evasion. Immunology 2022, 168, 331–345. [Google Scholar] [CrossRef]
- Gjuka, D.; Adib, E.; Garrison, K.; Chen, J.; Zhang, Y.; Li, W.; Boutz, D.; Lamb, C.; Tanno, Y.; Nassar, A.; et al. Enzyme-mediated depletion of methylthioadenosine restores T cell function in MTAP-deficient tumors and reverses immunotherapy resistance. Cancer Cell 2023, 41, 1774–1787. [Google Scholar] [CrossRef]
- Dono, A.; Pichardo-Rojas, D.; Mora, L.M.; Pichardo-Rojas, P.S.; Marin-Castañeda, L.A.; Carrillo, A.; Medrano, A.C.; Esquenazi, Y.; Ballester, L.Y. MTAP immunohistochemistry as a surrogate marker of CDKN2A loss in brain tumors: A meta-analysis and literature review Open Access. J. Neuropathol. Exp. Neurol. 2025, 84, 600–610. [Google Scholar] [CrossRef]
- Vlajnic, T.; Chijioke, O.; Roma, L.; Prince, S.S.; Zellweger, T.; Rentsch, C.A.; Bubendorf, L. Loss of MTAP expression by immunohistochemistry is a surrogate marker for homozygous 9p21.3 deletion in urothelial carcinoma. Mod. Pathol. 2024, 37, 100495. [Google Scholar] [CrossRef]
- Gorbokon, N.; Teljuk, K.; Reiswich, V.; Lennartz, M.; Minner, S. Deficiency of MTAP Is frequent and mostly homogeneous in pancreatic ductal adenocarcinomas. Cancers 2025, 17, 1205. [Google Scholar] [CrossRef]
- Febres-Aldana, C.A.; Chang, J.C.; Jungbluth, A.A.; Adusumilli, P.S.; Bodd, F.M.; Frosina, D.; Geronimo, J.A.; Hernandez, E.; Irawan, H.; Offin, M.D.; et al. Comparison of immunohistochemistry, next-generation sequencing and fluorescence in situ hybridization for detection of MTAP loss in pleural mesothelioma. Modern. Pathol. 2024, 37, 100420. [Google Scholar] [CrossRef]
- Vrugt, B.; Kirschner, M.B.; Meerang, M.; Oehl, K.; Wagner, U.; Soltermann, A.; Moch, H.; Opitz, I.; Wild, P.J. Deletions of CDKN2A and MTAP detected by copy-number variation array are associated with loss of p16 and MTAP protein in pleural mesothelioma. Cancers 2023, 15, 4978. [Google Scholar] [CrossRef]
- Cheng, X.-Y.; Liu, Z.; Shang, L.; Cai, H.-Q.; Zhang, Y.; Cai, Y.; Xu, X.; Hao, J.-J.; Wang, M.-R. Deletion and downregulation of MTAP contribute to the motility of esophageal squamous carcinoma cells. OncoTargets Ther. 2017, 10, 5855–5862. [Google Scholar] [CrossRef]
- Gast, A.; Scherer, D.; Chen, B.; Bloethner, S.; Melchert, S.; Sucker, A.; Hemminki, K.; Schadendorf, D.; Kumar, R. Somatic alterations in the melanoma genome: A high-resolution array-based comparative genomic hybridization study. Genes Chromosom. Cancer 2010, 49, 733–745. [Google Scholar] [CrossRef]
- M’SOka, T.; Nishioka, J.; Taga, A.; Kato, K.; Kawasaki, H.; Yamada, Y.; Yu, A.; Komada, Y.; Nobori, T. Detection of methylthioadenosine phosphorylase (MTAP) and p16 gene deletion in T cell acute lymphoblastic leukemia by real-time quantitative PCR assay. Leukemia 2000, 14, 935–940. [Google Scholar] [CrossRef]
- Kumari, S.; Singh, J.; Arora, M.; Ali, M.S.; Pandey, A.K.; Benjamin, M.; Palanichamy, J.K.; Bakhshi, S.; Qamar, I.; Chopra, A. Copy number alterations in CDKN2A/2B and MTAP genes are associated with low MEF2C expression in T-cell acute lymphoblastic leukemia. Cureus 2022, 14, e32151. [Google Scholar] [CrossRef]
- Satomi, K.; Ohno, M.; Matsushita, Y.; Takahashi, M.; Miyakita, Y.; Narita, Y.; Ichimura, K.; Yoshida, A. Utility of methylthioadenosine phosphorylase immunohistochemical deficiency as a surrogate for CDKN2A homozygous deletion in the assessment of adult-type infiltrating astrocytoma. Modern Pathol. 2021, 34, 688–700. [Google Scholar] [CrossRef]
- Meng, L.; Zhang, Y.; Du, X.; Feng, S.; Sheng, H.; Xing, Y.; Cheng, Y.; Guan, Y.; Yi, X. The performance of MTAP homozygous deletion detection based on next-generation sequencing technology. J. Clin. Oncol. 2025, 43, 14571. [Google Scholar] [CrossRef]
- Bertino, J.R.; Waud, W.R.; Parker, W.B.; Lubin, M. Targeting tumors that lack methylthioadenosine phosphorylase (MTAP) activity: Current strategies. Cancer Biol. Ther. 2011, 11, 627–632. [Google Scholar] [CrossRef]
- Marjon, K.; Cameron, M.J.; Quang, P.; Clasquin, M.F.; Mandley, E.; Kunii, K.; McVay, M.; Choe, S.; Kernytsky, A.; Gross, S.; et al. MTAP deletions in cancer create vulnerability to targeting of the MAT2A/PRMT5/RIOK1 axis. Cell Rep. 2016, 15, 574–587. [Google Scholar] [CrossRef]
- Fan, N.; Zhang, Y.; Zou, S. Methylthioadenosine phosphorylase deficiency in tumors: A compelling therapeutic target. Front. Cell Dev. Biol. 2023, 11, 1173356. [Google Scholar] [CrossRef]
- Feustel, K.; Falchook, G.S. Protein arginine methyltransferase 5 (PRMT5) inhibitors in oncology clinical trials: A review. J. Immunother. Precis. Oncol. 2022, 5, 58–67. [Google Scholar] [CrossRef]
- Hu, M.; Chen, X. A review of the known MTA-cooperative PRMT5 inhibitors. RSC Adv. 2024, 14, 39653–39691. [Google Scholar] [CrossRef]
- Shen, Z.; Li, C. Targeting PRMT5: Current Inhibitors and Emerging Strategies for Therapeutic Intervention. Processes 2025, 13, 2878. [Google Scholar] [CrossRef]
- Watts, J.; Minden, M.D.; Bachiashvili, K.; Brunner, A.M.; Abedin, S.; Crossman, T.; Zajac, M.; Moroz, V.; Egger, J.L.; Tarkar, A.; et al. Phase I/II study of the clinical activity and safety of GSK3326595 in patients with myeloid neoplasms. Ther. Adv. Hematol. 2024, 14, 20406207241275376. [Google Scholar] [CrossRef]
- Siu, L.; Rasco, D.; Vinay, S.P.; Romano, P.M.; Menis, J.; Opdam, F.; Heinhuis, K.; Egger, J.; Gorman, S.; Parasrampuria, R.; et al. METEOR-1: A phase I study of GSK3326595, a first-in-class protein arginine methyltransferase 5 (PRMT5) inhibitor, in advanced solid tumours. Ann. Oncol. 2019, 30, v159. [Google Scholar] [CrossRef]
- Vieito, M.; Moreno, V.; Spreafico, A.; Brana, I.; Wang, J.S.; Preis, M.; Hernández, T.; Genta, S.; Hansen, A.R.; Doger, B.; et al. Phase 1 Study of JNJ-64619178, a Protein Arginine Methyltransferase 5 Inhibitor, in Advanced Solid Tumors. Clin. Cancer Res. 2023, 29, 3592–3602. [Google Scholar] [CrossRef]
- Monga, V.; Johanns, T.M.; Stupp, R.; Chandra, S.; Falchook, G.S.; Giglio, P.; Philipovskiy, A.; Alnahhas, I.; Babbar, N.; Sun, W.; et al. A phase 1 study of the protein arginine methyltransferase 5 (PRMT5) brain-penetrant inhibitor PRT811 in patients (pts) with recurrent high-grade glioma or uveal melanoma (UM). J. Clin. Oncol. 2023, 41, 3008. [Google Scholar] [CrossRef]
- Jiang, Z.; Li, X.; Xiao, Z.; Gan, W.; Zhang, X.; Zhang, Y.; Wang, W.E.Q.; Huang, Y.; Shi, Q.; Tang, Y. Combined inhibition by PRMT5 and MAT2A demonstrates a strong synthetic lethality in MTAP homozygous-deficient glioma models. Cell Death Discov. 2025, 11, 261. [Google Scholar] [CrossRef] [PubMed]
- Engstrom, L.D.; Aranda, R.; Waters, L.; Moya, K.; Bowcut, V.; Vegar, L.; Trinh, D.; Hebbert, A.; Smith, C.R.; Kulyk, S.; et al. MRTX1719 Is an MTA-Cooperative PRMT5 Inhibitor That Exhibits Synthetic Lethality in Preclinical Models and Patients with MTAP-Deleted Cancer. Cancer Discov. 2023, 13, 2412–2431. [Google Scholar] [CrossRef] [PubMed]
- Hai, J.; Xavier, S.; George, S.; Engstrom, L.; Olson, P.; Simpson, T.; Lei, M.; Chen, B. MTA-cooperative PRMT5 inhibition (BMS-986504) induces MTAP del tumor cell-intrinsic immune evasion and regulation. Cancer Immunol. Res. 2024, 12, B011. [Google Scholar] [CrossRef]
- Belmontes, B.; Slemmons, K.K.; Su, C.; Liu, S.; Policheni, A.N.; Moriguchi, J.; Tan, H.; Xie, F.; Aiello, D.A.; Yang, Y.; et al. AMG 193, a Clinical Stage MTA-Cooperative PRMT5 Inhibitor, Drives Antitumor Activity Preclinically and in Patients with MTAP-Deleted Cancers. Cancer Discov. 2025, 15, 139–161. [Google Scholar] [CrossRef]
- Rodon, J.; Prenen, H.; Sacher, A.; Villalona-Calero, M.; Penel, N.; El Helali, A.; Rottey, S.; Yamamoto, N.; Ghiringhelli, F.; Goebeler, M.; et al. First-in-human study of AMG 193, an MTA-cooperative PRMT5 inhibitor, in patients with MTAP-deleted solid tumors: Results from phase I dose exploration. Ann. Oncol. 2024, 35, 1138–1147. [Google Scholar] [CrossRef]
- Briggs, K.J.; Cottrell, K.M.; Tonini, M.R.; Tsai, A.; Zhang, M.; Whittington, D.A.; Zhang, W.; Lombardo, S.A.; Yoda, S.; Wilker, E.W.; et al. TNG908 is a brain-penetrant, MTA-cooperative PRMT5 inhibitor developed for the treatment of MTAP-deleted cancers. Transl. Oncol. 2025, 52, 102264. [Google Scholar] [CrossRef]
- Zhang, M.; Tsai, A.; Cottrell, K.M.; Haines, B.B.; Huang, A.; Andersen, J.N.; Crystal, A.S.; Briggs, K.J. TNG462, an MTA-cooperative PRMT5 inhibitor, demonstrates strong efficacy in combination with clinically relevant targeted therapies in MTAP-null preclinical models. In Proceedings of the American Association for Cancer Research Annual Meeting 2025, Part 1 (Regular Abstracts), Chicago, IL, USA, 25–30 April 2025; p. 2996. [Google Scholar]
- Cottrell, K.M.; Briggs, K.J.; Tsai, A.; Tonini, M.R.; Whittington, D.A.; Gong, S.; Liang, C.; McCarren, P.; Zhang, M.; Zhang, W.; et al. Discovery of TNG462: A Highly Potent and Selective MTA-Cooperative PRMT5 Inhibitor to Target Cancers with MTAP Deletion. J. Med. Chem. 2025, 68, 5097–5119. [Google Scholar] [CrossRef]
- Konteatis, Z.; Travins, J.; Gross, S.; Marjon, K.; Barnett, A.; Mandley, E.; Nicolay, B.; Nagaraja, R.; Chen, Y.; Sun, Y.; et al. Discovery of AG-270, a First-in-Class Oral MAT2A Inhibitor for the Treatment of Tumors with Homozygous MTAP Deletion. J. Med. Chem. 2021, 64, 4430–4449. [Google Scholar] [CrossRef] [PubMed]
- Gounder, M.; Johnson, M.; Heist, R.S.; Shapiro, G.I.; Postel-Vinay, S.; Wilson, F.H.; Garralda, E.; Wulf, G.; Almon, C.; Nabhan, S.; et al. MAT2A inhibitor AG-270/S095033 in patients with advanced malignancies: A phase I trial. Nat. Commun. 2025, 16, 423. [Google Scholar] [CrossRef] [PubMed]
- Herzberg, B. Phase 1 expansion results of IDE397, a first-in-class, oral, MAT2A inhibitor (MAT2Ai) in MTAP deleted(del) non-small cell lung cancer (NSCLC) and urothelial cancer (UC). In Proceedings of the LBA Session EORT NCI AACR, Barcelona, Spain, 23–25 October 2024. Abstract No. 501. [Google Scholar]
- Mantia, C.; Kim, C.G.; Ahnert, J.R.; Alhalabi, O.; Wei, A.Z.; Stein, M.N.; Johnson, M.L.; Garmezy, B.; Tewari, A.; Devarakonda, S.; et al. Phase 1 dose escalation (DEs) expansion (DEx) study to evaluate the safety efficacy of IDE397 plus sacituzumab-govitecan in patients with advanced urothelial carcinoma (UC) with MTAP deletion (MTAPdel). J. Clin. Oncol. 2025, 43, TPS892. [Google Scholar] [CrossRef]
- Fischer, M.M.; Gerrick, K.; Belmontes, B.; Slemmons, K.; Freyman, Y.; Jain, J.; Federowicz, S.; Bishof, I.; Rao, A.A.; Fleury, M.; et al. Dual inhibition of MAT2A and PRMT5 delivers synergistic anti-tumor responses in preclinical models of MTAP-deleted cancer. Cancer Res. 2023, 83, 1644. [Google Scholar] [CrossRef]
- Zhang, G.; Li, J.; Li, J.J. Targeting MTAP-deleted tumor via synthetic lethality with a combination of a PRMT5i and an MAT2A. Cancer Res. 2024, 84, 7603. [Google Scholar] [CrossRef]
- Heist, R.S.; Gounder, M.M.; Postel-Vinay, S.; Wilson, F.; Garralda, E.; Do, K.; Shapiro, G.I.; Martin-Romano, P.; Wulf, G.; Cooper, M.; et al. A phase 1 trial of AG-270 in patients with advanced solid tumors or lymphoma with homozygous MTAP deletion. Mol. Cancer Ther. 2019, 18, PR03. [Google Scholar] [CrossRef]
| No | NCT Number | Study Title | Drug Name and Targets | Disease | Line of Treatment | Study Status and Phase |
|---|---|---|---|---|---|---|
| 1 | NCT03361358 | Pre-Screening Study to Identify MTAP Loss in Advanced Solid Tumors or Lymphoma | No | Advanced solid tumor 9 other than CNS malignancy) or lymphoma | – | Completed, Study type: observational |
| PRMT5 inhibitors in clinical trials | ||||||
| 2 | NCT05094336 | A Study of AMG 193 in Participants with Advanced MTAP-null Solid Tumors (MTAPESTRY 101) | AMG 193 (PRMT5 inhibitor) monotherapy or in combination with docetaxel | Locally advanced or metastatic solid tumors | II and subsequent | Recruiting, Phase I/II |
| 3 | NCT06593522 | A Phase 2 Study of AMG 193 in Participants with MTAP-deleted Advanced NSCLC (MTAPESTRY 201) | AMG 193 (PRMT5 inhibitor) | Advanced NSCLC | II and subsequent | Recruiting, Phase II |
| 4 | NCT06333951 | Alone or in Combination with Other Therapies in Subjects with Advanced Thoracic Tumors with Homozygous MTAP-deletion (Master Protocol) (MTAPESTRY 104) | AMG 193 (PRMT5 inhibitor) monotherapy or in combination with carboplatin, paclitaxel and pembrolizumab or carboplatin, pemetrexed and pembrolizumab or pembrolizumab alone or sotorasib | Advanced or metastatic NSCLC | I, II or subsequent (patients with CNS metastases) | Recruiting, Phase I |
| 5 | NCT06360354 | A Study Evaluating AMG 193 in Combination with Other Therapies in Participants with Advanced Gastrointestinal, Biliary Tract, or Pancreatic Cancers with Homozygous Methylthioadenosine Phosphorylase (MTAP) | AMG 193 (PRMT5 inhibitor) in combination with gemcitabine and nab-paclitaxel or with modified FOLFIRINOX | Advanced gastrointestinal, biliary tract and pancreatic cancers | I and subsequent | Recriuting, Phase I |
| 6 | NCT05275478 | Safety and Tolerability of TNG908 in Patients with MTAP-deleted Solid Tumors | TNG908 (ralometostat) (PRMT5 inhibitor) | Advanced or metastatic solid tumors | No standard treatment possible | Active, not recruiting, Phase I/II |
| 7 | NCT05732831 | Safety and Tolerability of TNG462 in Patients with MTAP-deleted Solid Tumors | TNG462 (PRMT5 inhibitor) monotherapy or in combination with pembrolizumab | Locally advanced solid tumor | No standard treatment possible | Recruiting, Phase I |
| 8 | NCT05245500 | Phase 1 Study of MRTX1719 in Solid Tumors with MTAP Deletion | BMS-986504 (MRTX1719) (PRMT5 inhibitor). In phase II in combination with standard of care therapy in selected solid tumor malignancies | Mesothelioma, NSCLC, malignant peripheral nerve sheath tumors, pancreatic adenocarcinoma, advanced solid tumors | II and subsequent | Recruiting, Phase I/II |
| 9 | NCT06855771 | A Study of BMS-986504 in Participants with Pre-treated Advanced or Metastatic Non-small Cell Lung Cancer (NSCLC) with Homozygous MTAP Deletion | BMS-986504 (MRTX1719) (PRMT5 inhibitor) | Advanced or metastatic NSCLC | No standard treatment possible | Recruiting, Phase II |
| 10 | NCT06883747 | Clinical Trial of BMS-986504 in Recurrent GBM Patients | BMS-986504 (MRTX1719) (PRMT5 inhibitor) | Advanced glioblastoma | No standard treatment possible | Recruiting, Early phase I |
| 11 | NCT06672523 | A Study to Evaluate the Mass Balance, Metabolism, Elimination, and Drug Levels of [14C]-BMS-986504 (MRTX1719) in Participants with Advanced Solid Tumors With Homozygous Methylthioadenosine Phosphorylase Deletion | BMS-986504 (MRTX1719) (PRMT5 inhibitor) | Advanced or metastatic solid tumors | No standard treatment possible | Recruiting, Phase I |
| 12 | NCT06973863 | A Study of PEP08 in Patients with MTAP-Del Advanced or Metastatic Solid Tumors | PEP08 (PRMT5 inhibitor) monotherapy or in combination with standard of care therapy in selected solid tumor malignancies | Advanced or metastatic solid tumors | No standard treatment possible | Not yet recruiting Phase I |
| 13 | NCT06914128 | A First-in-human Study to Learn How Safe BAY 3713372 is and How it Works in Participants with MTAP-deleted Solid Tumors | BAY 3713372 (PRMT5 inhibitor) | Advanced or metastatic solid tumors | No standard treatment possible | Recruiting Phase I |
| 14 | NCT06796699 | A Phase Ia/Ib Clinical Study of GH56 Capsules in Subjects with MTAP-Deleted Advanced Solid Tumors | GH56 (PRMT5 inhibitor) | Advanced or metastatic solid tumors | No standard treatment possible | Recruiting, Phase I |
| 15 | NCT06968572 | Phase I Study of HSK41959 in Solid Tumors with MTAP Deletion | HSK41959 (PRMT5 inhibitor) | Advanced or metastatic solid tumors | No standard treatment possible | Recruiting, Phase I |
| MAT2A inhibitors in clinical trials | ||||||
| 16 | NCT04794699 | Study of IDE397 in Participants with Solid Tumors Harboring MTAP Deletion | IDE397 (MAT2A inhibitor) in combination with docetaxel or paclitaxel or sacitzumab govitecan | Advanced or metastatic solid tumors | II and subsequent | Recruiting, Phase I |
| 17 | NCT06568614 | An Investigational Study of BG-89894 Tablets in Participants with Advanced Solid Tumors | BG-89894 (SYH2039) (MAT2A inhibitor) | Advanced or metastatic solid tumors | No standard treatment possible | Recruiting, Phase I |
| 18 | NCT03435250 | Study of AG-270 in Participants with Advanced Solid Tumors or Lymphoma with MTAP Loss | AG-270 (MAT2A inhibitor) monotherapy or in combination with gemcitabine or nab-paclitaxel and gemcitabine | Advanced Solid Tumors or Lymphoma | II and subsequent | Terminated, Phase I |
| 19 | NCT06414460 | Study of ISM3412 in Participants with Locally Advanced/Metastatic Solid Tumors | ISM3412 (MAT2A inhibitor) | Locally advanced or metastatic Solid Tumors | No standard treatment possible | Recriuting, Phase I |
| Combined use of PRMT5 and MAT2A inhibitors in clinical trials | ||||||
| 20 | NCT06188702 | S095035 as a Single Agent and in Combination in Adult Participants with Advanced or Metastatic Solid Tumors with Deletion of MTAP | AG 270 (MAT2A inhibitor), TNG462 (PRMT5 inhibitor) | Advanced or metastatic solid tumors | II and subsequent | Recruiting Phase I/II |
| 21 | NCT05975073 | A Phase 1/2 Study of AMG 193 in Combination with IDE397 in Participants with Advanced Methylthioadenosine Phosphorylase (MTAP)-Null Solid Tumors | AMG 193 (PRMT5 inhibitor), IDE397 (MAT2A inhibitor) | Advanced or metastatic solid tumors, advanced NSCLC | II and subsequent (in NSCLC patients), No standard treatment possible (in other patients) | Active, not recruiting Phase I/II |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krawczyk, P.; Wojas-Krawczyk, K. MTAP Deletion as a Therapeutic Vulnerability in Cancer: From Molecular Mechanism to Clinical Targeting. Int. J. Mol. Sci. 2025, 26, 11956. https://doi.org/10.3390/ijms262411956
Krawczyk P, Wojas-Krawczyk K. MTAP Deletion as a Therapeutic Vulnerability in Cancer: From Molecular Mechanism to Clinical Targeting. International Journal of Molecular Sciences. 2025; 26(24):11956. https://doi.org/10.3390/ijms262411956
Chicago/Turabian StyleKrawczyk, Paweł, and Kamila Wojas-Krawczyk. 2025. "MTAP Deletion as a Therapeutic Vulnerability in Cancer: From Molecular Mechanism to Clinical Targeting" International Journal of Molecular Sciences 26, no. 24: 11956. https://doi.org/10.3390/ijms262411956
APA StyleKrawczyk, P., & Wojas-Krawczyk, K. (2025). MTAP Deletion as a Therapeutic Vulnerability in Cancer: From Molecular Mechanism to Clinical Targeting. International Journal of Molecular Sciences, 26(24), 11956. https://doi.org/10.3390/ijms262411956





