Identification of Differential Proteins in Thrombi of Cardioembolic and Atherothrombotic Etiology in Patients with Ischemic Stroke
Abstract
1. Introduction
2. Results
2.1. Patient Characteristics
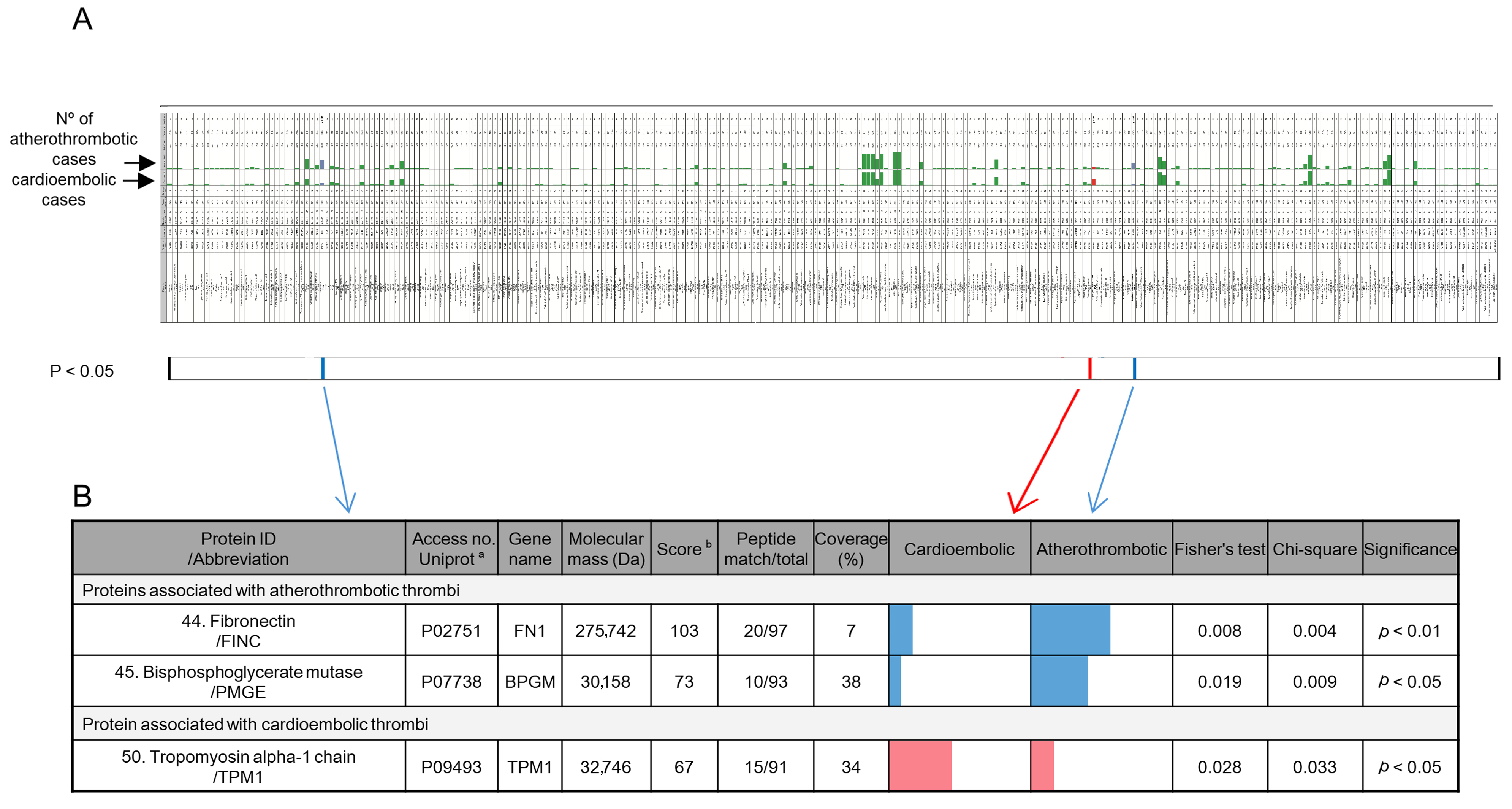
2.2. Biomarker Identification
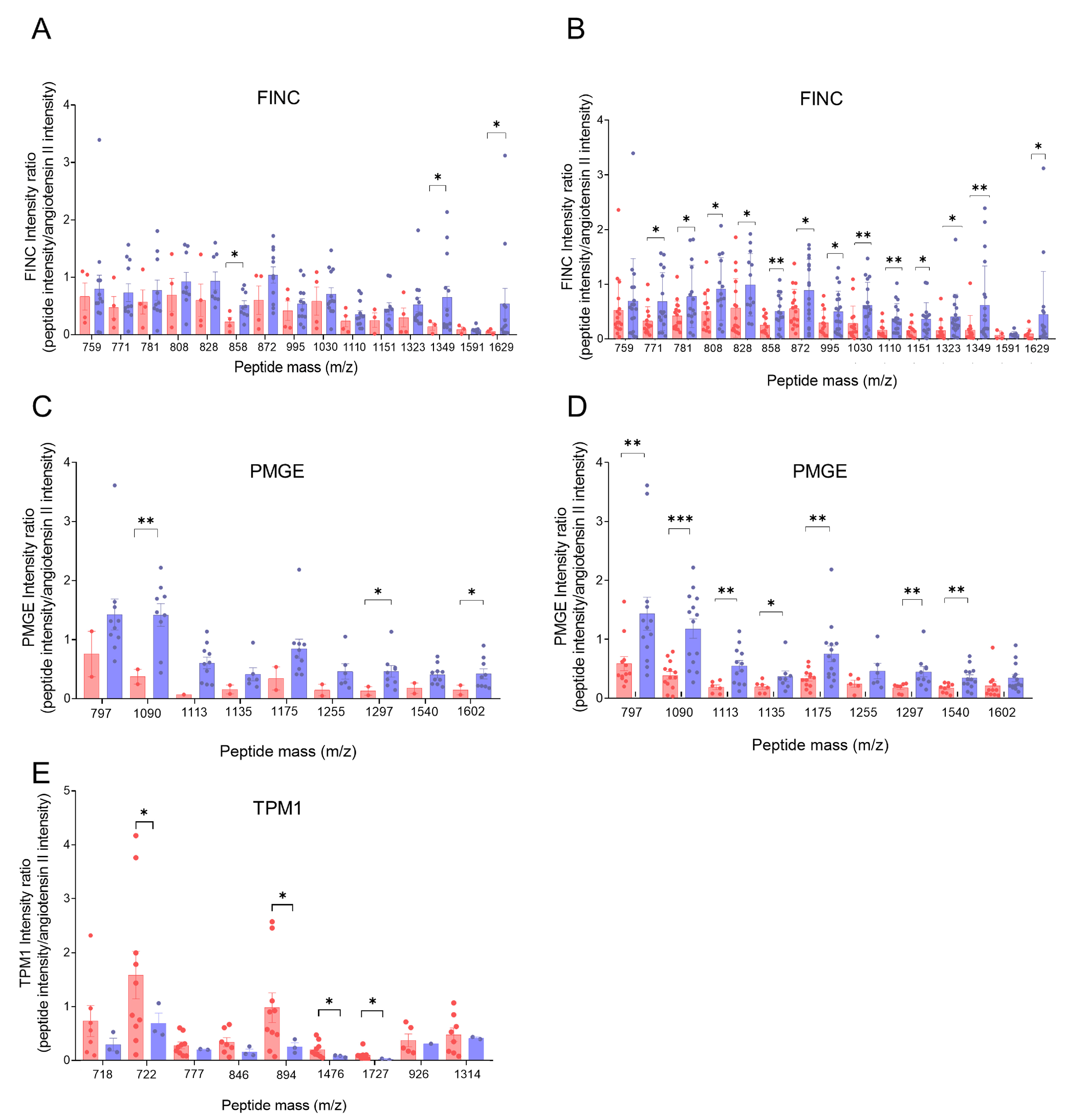
2.3. Quantification of Biomarkers
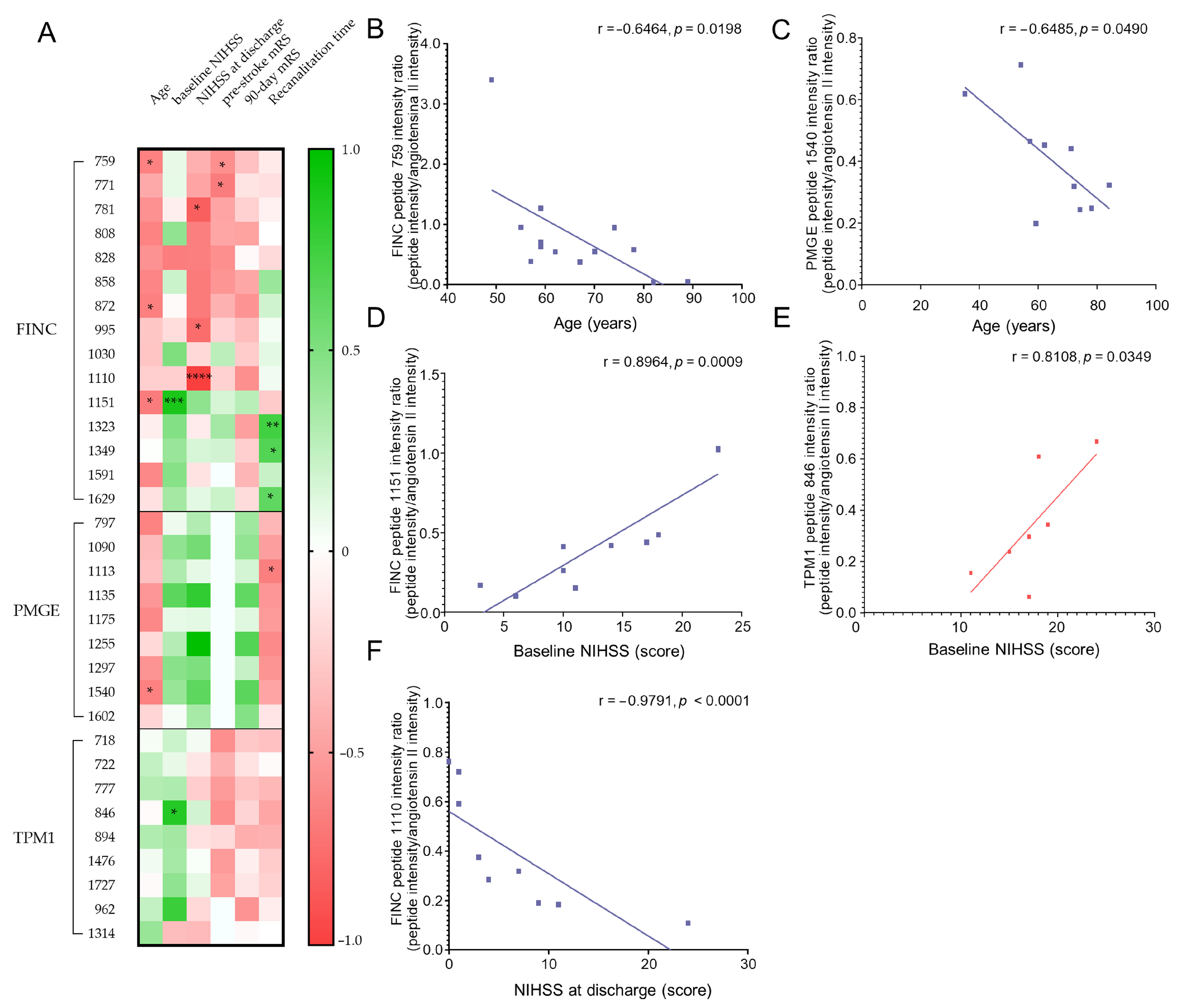
2.4. Correlation of Biomarkers with Clinical Outcomes in Thrombus Analysis
2.5. Sensitivity, Specificity, and Predictive Value of Biomarkers
3. Discussion
4. Materials and Methods
4.1. Subjects
4.2. Thrombi Processing
4.3. Protein Extraction
4.4. Gel Electrophoresis
4.5. In-Gel Protein Digestion and Protein Identification by Mass Spectrometry
4.6. Quantification of Peptides by Mass Spectrometry
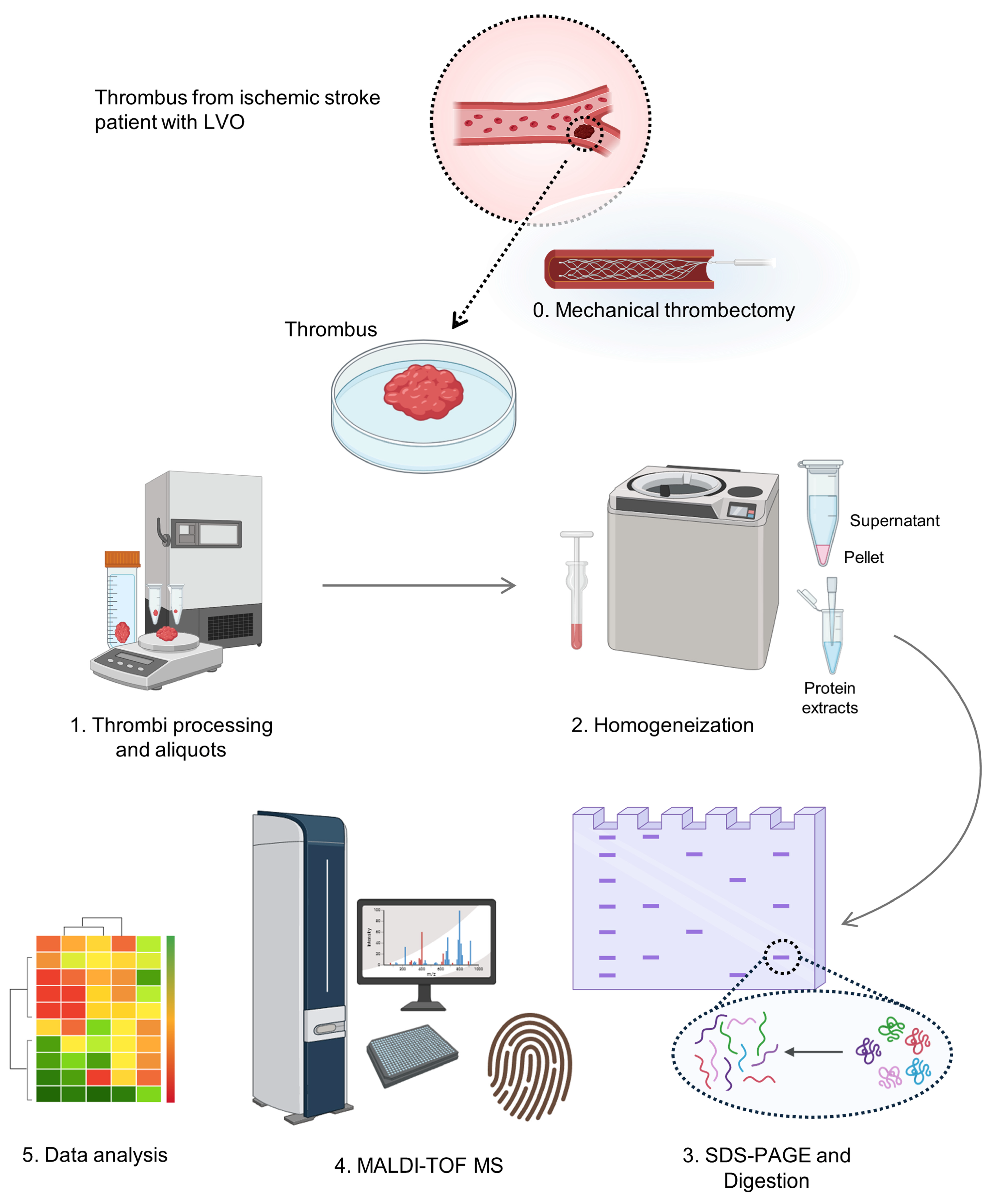
4.7. Data Collection and Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Correction Statement
References
- Feigin, V.L.; Brainin, M.; Norrving, B.; Martins, S.; Sacco, R.L.; Hacke, W.; Fisher, M.; Pandian, J.; Lindsay, P. World Stroke Organization (WSO): Global stroke fact sheet 2022. Int. J. Stroke 2022, 17, 18–19, Erratum in Int. J. Stroke 2022, 17, 478. [Google Scholar] [CrossRef]
- Mortality and Global Health Estimates. Available online: https://www.who.int/data/gho/data/themes/mortality-and-global-health-estimates (accessed on 10 April 2025).
- Katan, M.; Luft, A. Global burden of stroke. Semin. Neurol. 2018, 38, 208–211. [Google Scholar] [CrossRef] [PubMed]
- Sacco, R.L.; Kasner, S.E.; Broderick, J.P.; Caplan, L.R.; Connors, J.J.; Culebras, A.; Elkind, M.S.; George, M.G.; Hamdan, A.D.; Higashida, R.T.; et al. An updated definition of stroke for the 21st century. Stroke 2013, 44, 2064–2089, Correction in Stroke 2013, 50, e239. [Google Scholar] [CrossRef] [PubMed]
- Kuriakose, D.; Xiao, Z. Pathophysiology and treatment of stroke: Present status and future perspectives. Int. J. Mol. Sci. 2020, 21, 7609. [Google Scholar] [CrossRef]
- Hankey, G.J. Potential new risk factors for ischemic stroke: What is their potential? Stroke 2006, 37, 2181–2188. [Google Scholar] [CrossRef]
- Smith, W.S.; Lev, M.H.; English, J.D.; Camargo, E.C.; Chou, M.; Johnston, S.C.; Gonzalez, G.; Schaefer, P.W.; Dillon, W.P.; Koroshetz, W.J.; et al. Significance of large vessel intracranial occlusion causing acute ischemic stroke and TIA. Stroke J. Cereb. Circ. 2009, 40, 3834–3840. [Google Scholar] [CrossRef]
- Qin, C.; Yang, S.; Chu, Y.-H.; Zhang, H.; Pang, X.-W.; Chen, L.; Zhou, L.-Q.; Chen, M.; Tian, D.-S.; Wang, W. Signaling pathways involved in ischemic stroke: Molecular mechanisms and therapeutic interventions. Signal Transduct. Target. Ther. 2022, 7, 215, Correction in Signal Transduct. Target. Ther. 2022, 7, 278. [Google Scholar]
- Zhao, Y.; Zhang, X.; Chen, X.; Wei, Y. Neuronal injuries in cerebral infarction and ischemic stroke: From mechanisms to treatment (Review). Int. J. Mol. Med. 2022, 49, 15. [Google Scholar] [CrossRef]
- Knight-Greenfield, A.; Nario, J.J.Q.; Gupta, A. Causes of acute stroke. Radiol. Clin. N. Am. 2019, 57, 1093–1108. [Google Scholar] [CrossRef]
- Adams, H.P., Jr.; Bendixen, B.H.; Kappelle, L.J.; Biller, J.; Love, B.B.; Gordon, D.L.; Marsh, E.E., 3rd. Classification of subtype of acute ischemic stroke. Definitions for use in a multicenter clinical trial. TOAST. Trial of Org 10172 in Acute Stroke Treatment. Stroke 1993, 24, 35–41. [Google Scholar] [CrossRef]
- Ay, H.; Furie, K.L.; Singhal, A.; Smith, W.S.; Sorensen, A.G.; Koroshetz, W.J. An evidence-based causative classification system for acute ischemic stroke. Ann. Neurol. 2005, 58, 688–697. [Google Scholar] [CrossRef]
- Herpich, F.; Rincon, F. Management of acute ischemic stroke. Crit. Care Med. 2020, 48, 1654–1663. [Google Scholar] [CrossRef]
- Mosconi, M.G.; Paciaroni, M. Treatments in ischemic stroke: Current and future. Eur. Neurol. 2022, 85, 349–366. [Google Scholar] [CrossRef]
- Lee, M.H.; Im, S.H.; Jo, K.W.; Yoo, D.S. Recanalization rate and clinical outcomes of intravenous tissue plasminogen activator administration for large vessel occlusion stroke patients. J. Korean Neurosurg. Soc. 2023, 66, 144–154. [Google Scholar] [CrossRef] [PubMed]
- Berkhemer, O.A.; Fransen, P.S.S.; Beumer, D.; Berg, L.A.V.D.; Lingsma, H.F.; Yoo, A.J.; Schonewille, W.J.; Vos, J.A.; Nederkoorn, P.J.; Wermer, M.J.H.; et al. A randomized trial of intraarterial treatment for acute ischemic stroke. N. Engl. J. Med. 2015, 372, 11–20, Correction in N. Engl. J. Med. 2015, 372, 394. [Google Scholar] [CrossRef] [PubMed]
- Goyal, M.; Demchuk, A.M.; Menon, B.K.; Eesa, M.; Rempel, J.L.; Thornton, J.; Roy, D.; Jovin, T.G.; Willinsky, R.A.; Sapkota, B.L.; et al. Randomized assessment of rapid endovascular treatment of ischemic stroke. N. Engl. J. Med. 2015, 372, 1019–1030. [Google Scholar] [CrossRef] [PubMed]
- Campbell, B.C.; Mitchell, P.J.; Kleinig, T.J.; Dewey, H.M.; Churilov, L.; Yassi, N.; Yan, B.; Dowling, R.J.; Parsons, M.W.; Oxley, T.J.; et al. Endovascular therapy for ischemic stroke with perfusion-imaging selection. N. Engl. J. Med. 2015, 372, 1009–1018. [Google Scholar] [CrossRef]
- Saver, J.L.; Goyal, M.; Bonafe, A.; Diener, H.-C.; Levy, E.I.; Pereira, V.M.; Albers, G.W.; Cognard, C.; Cohen, D.J.; Hacke, W.; et al. Stent-retriever thrombectomy after intravenous t-PA vs. t-PA alone in stroke. N. Engl. J. Med. 2015, 372, 2285–2295. [Google Scholar] [CrossRef]
- Jovin, T.G.; Chamorro, A.; Cobo, E.; De Miquel, M.A.; Molina, C.A.; Rovira, A.; Román, L.S.; Serena, J.; Abilleira, S.; Ribo, M.; et al. Thrombectomy within 8 hours after symptom onset in ischemic stroke. N. Engl. J. Med. 2015, 372, 2296–2306. [Google Scholar] [CrossRef]
- Oliveira, A.J.F.; Viana, S.M.N.; Santos, A.S. Mechanical thrombectomy for acute ischemic stroke: Systematic review and meta-analysis. Einstein 2022, 20, eRW6642. [Google Scholar] [CrossRef]
- Fitzgerald, S.; Mereuta, O.M.; Doyle, K.M.; Dai, D.; Kadirvel, R.; Kallmes, D.F.; Brinjikji, W. Correlation of imaging and histopathology of thrombi in acute ischemic stroke with etiology and outcome. J. Neurosurg. Surg. 2017, 9, 529–534. [Google Scholar] [CrossRef]
- Gunning, G.M.; McArdle, K.; Mirza, M.; Duffy, S.; Gilvarry, M.; Brouwer, P.A. Clot friction variation with fibrin content; implications for resistance to thrombectomy. J. Neurosurg. Surg. 2018, 10, 34–38. [Google Scholar] [CrossRef] [PubMed]
- Maekawa, K.; Shibata, M.; Nakajima, H.; Mizutani, A.; Kitano, Y.; Seguchi, M.; Yamasaki, M.; Kobayashi, K.; Sano, T.; Mori, G.; et al. Erythrocyte-rich thrombus is associated with reduced number of maneuvers and procedure time in patients with acute ischemic stroke undergoing mechanical thrombectomy. Cerebrovasc. Dis. Extra 2018, 8, 39–49. [Google Scholar] [CrossRef] [PubMed]
- Sporns, P.B.; Jeibmann, A.; Minnerup, J.; Broocks, G.; Nawabi, J.; Schön, G.; Fiehler, J.; Wildgruber, M.; Heindel, W.; Kemmling, A.; et al. Histological clot composition is associated with preinterventional clot migration in acute stroke patients. Stroke 2019, 50, 2065–2071. [Google Scholar] [CrossRef] [PubMed]
- BBoeckh-Behrens, T.; Kleine, J.F.; Zimmer, C.; Neff, F.; Scheipl, F.; Pelisek, J.; Schirmer, L.; Nguyen, K.; Karatas, D.; Poppert, H. Thrombus histology suggests cardioembolic cause in cryptogenic stroke. Stroke 2016, 47, 1864–1871. [Google Scholar] [CrossRef]
- Sporns, P.B.; Hanning, U.; Schwindt, W.; Velasco, A.; Minnerup, J.; Zoubi, T.; Heindel, W.; Jeibmann, A.; Niederstadt, T.U. Ischemic stroke: What does the histological composition tell us about the origin of the thrombus? Stroke 2017, 48, 2206–2210. [Google Scholar] [CrossRef]
- Niesten, J.M.; van der Schaaf, I.C.; van Dam, L.; Vink, A.; Vos, J.A.; Schonewille, W.J.; de Bruin, P.C.; Mali, W.P.T.M.; Velthuis, B.K.; Baron, J.-C. Histopathologic composition of cerebral thrombi of acute stroke patients is correlated with stroke subtype and thrombus attenuation. PLoS ONE 2014, 9, e88882. [Google Scholar] [CrossRef]
- Powers, W.J.; Rabinstein, A.A.; Ackerson, T.; Adeoye, O.M.; Bambakidis, N.C.; Becker, K.; Biller, J.; Brown, M.; Demaerschalk, B.M.; Hoh, B.; et al. 2018 Guidelines for the early management of patients with acute ischemic stroke: A guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2018, 49, e46–e110. [Google Scholar] [CrossRef]
- Kapil, N.; Datta, Y.H.; Alakbarova, N.; Bershad, E.; Selim, M.; Liebeskind, D.S.; Bachour, O.; Rao, G.H.R.; Divani, A. A Antiplatelet and anticoagulant therapies for prevention of ischemic stroke. Clin. Appl. Thromb. 2017, 23, 301–318. [Google Scholar] [CrossRef]
- Kolominsky-Rabas, P.L.; Weber, M.; Gefeller, O.; Neundoerfer, B.; Heuschmann, P.U. Epidemiology of ischemic stroke subtypes according to toast criteria. Stroke 2001, 32, 2735–2740. [Google Scholar] [CrossRef]
- Dagonnier, M.; Donnan, G.A.; Davis, S.M.; Dewey, H.M.; Howells, D.W. Acute stroke biomarkers: Are we there yet? Front. Neurol. 2021, 12, 619721. [Google Scholar] [CrossRef]
- Muñoz, R.; Santamaría, E.; Rubio, I.; Ausín, K.; Ostolaza, A.; Labarga, A.; Roldán, M.; Zandio, B.; Mayor, S.; Bermejo, R.; et al. Mass spectrometry-based proteomic profiling of thrombotic material obtained by endovascular thrombectomy in patients with ischemic stroke. Int. J. Mol. Sci. 2018, 19, 498. [Google Scholar] [CrossRef]
- Dargazanli, C.; Zub, E.; Deverdun, J.; Decourcelle, M.; de Bock, F.; Labreuche, J.; Lefèvre, P.-H.; Gascou, G.; Derraz, I.; Bareiro, C.R.; et al. Machine learning analysis of the cerebrovascular thrombi proteome in human ischemic stroke: An exploratory study. Front. Neurol. 2020, 11, 575376. [Google Scholar] [CrossRef]
- Rossi, R.; Mereuta, O.M.; e Silva, M.B.; Gil, S.M.; Douglas, A.; Pandit, A.; Gilvarry, M.; McCarthy, R.; O’COnnell, S.; Tierney, C.; et al. Potential biomarkers of acute ischemic stroke etiology revealed by mass spectrometry-based proteomic characterization of formalin-fixed paraffin-embedded blood clots. Front. Neurol. 2022, 13, 854846. [Google Scholar] [CrossRef]
- Abbasi, M.; Fitzgerald, S.; Ayers-Ringler, J.; Espina, V.; Mueller, C.; Rucker, S.; Kadirvel, R.; Kallmes, D.; Brinjikji, W. Proteomic analysis of cardioembolic and large artery atherosclerotic clots using reverse phase protein array technology reveals key cellular interactions within clot microenvironments. Cureus 2021, 13, e13499. [Google Scholar] [CrossRef]
- Lopez-Pedrera, C.; Oteros, R.; Ibáñez-Costa, A.; Luque-Tévar, M.; Muñoz-Barrera, L.; Barbarroja, N.; Chicano-Gálvez, E.; Marta-Enguita, J.; Orbe, J.; Velasco, F.; et al. The thrombus proteome in stroke reveals a key role of the innate immune system and new insights associated with its etiology, severity, and prognosis. J. Thromb. Haemost. 2023, 21, 2894–2907. [Google Scholar] [CrossRef] [PubMed]
- Jagatheesan, G.; Rajan, S.; Ahmed, R.P.H.; Petrashevskaya, N.; Boivin, G.; Arteaga, G.M.; Tae, H.-J.; Liggett, S.B.; Solaro, R.J.; Wieczorek, D.F. Striated muscle tropomyosin isoforms differentially regulate cardiac performance and myofilament calcium sensitivity. J. Muscle Res. Cell Motil. 2010, 31, 227–239. [Google Scholar] [CrossRef] [PubMed]
- Kopylova, G.V.; Shchepkin, D.V.; Nabiev, S.R.; Matyushenko, A.M.; Koubassova, N.A.; Levitsky, D.I.; Bershitsky, S.Y. Cardiomyopathy-associated mutations in tropomyosin differently affect actin-myosin interaction at single-molecule and ensemble levels. J. Muscle Res. Cell Motil. 2019, 40, 299–308. [Google Scholar] [CrossRef]
- Yaghi, S.; Chang, A.D.; Ricci, B.A.; Jayaraman, M.V.; McTaggart, R.A.; Hemendinger, M.; Narwal, P.; Dakay, K.; Mac Grory, B.; Cutting, S.M.; et al. Early elevated troponin levels after ischemic stroke suggests a cardioembolic source. Stroke 2018, 49, 121–126, Correction in Stroke 2018, 49, e24. [Google Scholar] [CrossRef]
- Wang, Y.; Reheman, A.; Spring, C.M.; Kalantari, J.; Marshall, A.H.; Wolberg, A.S.; Gross, P.L.; Weitz, J.I.; Rand, M.L.; Mosher, D.F.; et al. Plasma fibronectin supports hemostasis and regulates thrombosis. J. Clin. Investig. 2014, 124, 4281–4293. [Google Scholar] [CrossRef]
- To, W.S.; Midwood, K.S. Plasma and cellular fibronectin: Distinct and independent functions during tissue repair. Fibrogenes. Tissue Repair. 2011, 4, 21. [Google Scholar] [CrossRef]
- Wang, L.; Ren, C.; Li, Y.; Gao, C.; Li, N.; Li, H.; Wu, D.; He, X.; Xia, C.; Ji, X. Remote ischemic conditioning enhances oxygen supply to ischemic brain tissue in a mouse model of stroke: Role of elevated 2,3-biphosphoglycerate in erythrocytes. J. Cereb. Blood Flow Metab. 2021, 41, 1277–1290. [Google Scholar] [CrossRef]
- Wang, L.; Deng, L.; Yuan, R.; Liu, J.; Li, Y.; Liu, M. Association of matrix metalloproteinase 9 and cellular fibronectin and outcome in acute ischemic stroke: A systematic review and meta-analysis. Front. Neurol. 2020, 11, 523506. [Google Scholar] [CrossRef] [PubMed]
- Uchida, Y.; Kan, H.; Inoue, H.; Oomura, M.; Shibata, H.; Kano, Y.; Kuno, T.; Usami, T.; Takada, K.; Yamada, K.; et al. Penumbra detection with oxygen extraction fraction using magnetic susceptibility in patients with acute ischemic stroke. Front. Neurol. 2022, 13, 752450. [Google Scholar] [CrossRef] [PubMed]
- Shevchenko, A.; Tomas, H.; Havlis, J.; Olsen, J.V.; Mann, M. In-gel digestion for mass spectrometric characterization of proteins and proteomes. Nat. Protoc. 2006, 1, 2856–2860. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Alonso, E.; Guerra-Pérez, N.; Escobar-Peso, A.; Regidor, I.; Masjuan, J.; Alcázar, A. Differential association of 4e-bp2-interacting proteins is related to selective delayed neuronal death after ischemia. Int. J. Mol. Sci. 2021, 22, 10327. [Google Scholar] [CrossRef]
- Cid, C.; Garcia-Bonilla, L.; Camafeita, E.; Burda, J.; Salinas, M.; Alcazar, A. Proteomic characterization of protein phosphatase 1 complexes in ischemia-reperfusion and ischemic tolerance. Proteomics 2007, 7, 3207–3218. [Google Scholar] [CrossRef]
- Martínez-Alonso, E.; Alcázar, P.; Camafeita, E.; Fernández-Lucas, M.; Ruíz-Roso, G.; Alcázar, A. Proteomic analysis of plasma proteins of high-flux haemodialysis and on-line haemodiafiltration patients reveals differences in transthyretin levels related with anaemia. Sci. Rep. 2020, 10, 16029. [Google Scholar] [CrossRef]
| Cardioembolic Etiology (range/percentage) | Atherothrombotic Etiology (range/percentage) | p-Value; Result 1 | |
| Demographic data | |||
| Age (years) | 78 (41–90) | 64 (35–89) | 0.002; ** |
| Gender, female | 15 (55.6%) | 8 (29.6%) | 0.098; ns |
| Risk Factors | |||
| Hypertension | 21 (77.8%) | 15 (55.6%) | 0.148; ns |
| Diabetes mellitus | 6 (22.2%) | 8 (29.6%) | 0.757; ns |
| Hyperlipidemia | 21 (77.8%) | 22 (81.5%) | 0.999; ns |
| Smoking habit | 2 (7.4%) | 12 (44.4%) | 0.004; ** |
| Drinking habit | 1 (3.7%) | 4 (14.8%) | 0.351; ns |
| Atrial fibrillation | 24 (88.9%) | 0 (0.0%) | 0.000; **** |
| Ischemic heart disease | 3 (11.1%) | 4 (14.8%) | 0.999; ns |
| Previous stroke | 4 (14.8%) | 5 (18.5%) | 0.999; ns |
| LVO localization | |||
| MCA | 15 (55.6%) | 6 (22.2%) | 0.024; * |
| Basilar artery | 4 (14.8%) | 4 (14.8%) | 0.999; ns |
| MCA + carotid artery | 8 (29.6%) | 17 (63.0%) | 0.028; * |
| In-hospital work-up | |||
| Secondary transfer | 10 (37.0%) | 13 (48.2%) | 0.583; ns |
| Onset-to-recanalization time (minutes) ± SD | 112.8 ± 13.0 | 143.3 ± 27.8 | 0.331; ns |
| Pre-stroke mRS | 0 (0–3) | 0 (0–3) | 0.999; ns |
| Baseline NIHSS | 18 (9–26) | 15 (3–23) | 0.004; ** |
| NIHSS at discharge | 4 (0–26) | 4 (0–30) | 0.460; ns |
| 90-day mRS | 2 (0–6) | 2 (0–6) | 0.576; ns |
| Death in hospital | 2 (7.4%) | 5 (19.2%) | 0.250; ns |
| Stent | 0 (0.0%) | 21 (77.8%) | 0.000; **** |
| rtPA administration | 11 (40.7%) | 17 (63.0%) | 0.173; ns |
| Sensitivity | Specificity | PPV 1 | NPV 2 | |
|---|---|---|---|---|
| Biomarker panel associated with atherothrombotic thrombi | ||||
| FINC + | 0.52 | 0.85 | 0.78 | 0.64 |
| PMGE + | 0.37 | 0.93 | 0.83 | 0.60 |
| FINC + or PMGE +, TPM1 − | 0.59 | 1.0 | 1.0 | 0.71 |
| Biomarker panel associated with cardioembolic thrombi | ||||
| TPM1 + | 0.41 | 0.89 | 0.79 | 0.60 |
| FINC −, PMGE −, TPM1 + | 0.19 | 1.0 | 1.0 | 0.55 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peracho, L.; Martínez-Alonso, E.; Bermúdez, I.; Cruz-Culebras, A.; De Felipe, A.; Fandiño, E.; García-Madrona, S.; Matute-Lozano, M.C.; Méndez-Cendón, J.C.; Vera-Lechuga, R.; et al. Identification of Differential Proteins in Thrombi of Cardioembolic and Atherothrombotic Etiology in Patients with Ischemic Stroke. Int. J. Mol. Sci. 2025, 26, 8333. https://doi.org/10.3390/ijms26178333
Peracho L, Martínez-Alonso E, Bermúdez I, Cruz-Culebras A, De Felipe A, Fandiño E, García-Madrona S, Matute-Lozano MC, Méndez-Cendón JC, Vera-Lechuga R, et al. Identification of Differential Proteins in Thrombi of Cardioembolic and Atherothrombotic Etiology in Patients with Ischemic Stroke. International Journal of Molecular Sciences. 2025; 26(17):8333. https://doi.org/10.3390/ijms26178333
Chicago/Turabian StylePeracho, Lorena, Emma Martínez-Alonso, Isabel Bermúdez, Antonio Cruz-Culebras, Alicia De Felipe, Eduardo Fandiño, Sebastián García-Madrona, María Consuelo Matute-Lozano, Jose Carlos Méndez-Cendón, Rocío Vera-Lechuga, and et al. 2025. "Identification of Differential Proteins in Thrombi of Cardioembolic and Atherothrombotic Etiology in Patients with Ischemic Stroke" International Journal of Molecular Sciences 26, no. 17: 8333. https://doi.org/10.3390/ijms26178333
APA StylePeracho, L., Martínez-Alonso, E., Bermúdez, I., Cruz-Culebras, A., De Felipe, A., Fandiño, E., García-Madrona, S., Matute-Lozano, M. C., Méndez-Cendón, J. C., Vera-Lechuga, R., Masjuan, J., & Alcázar, A. (2025). Identification of Differential Proteins in Thrombi of Cardioembolic and Atherothrombotic Etiology in Patients with Ischemic Stroke. International Journal of Molecular Sciences, 26(17), 8333. https://doi.org/10.3390/ijms26178333





