NMR Study of Water-Soluble Carotenoid Crocin: Formation of Mixed Micelles, Interaction with Lipid Membrane and Antioxidant Activity
Abstract
1. Introduction
2. Results and Discussion
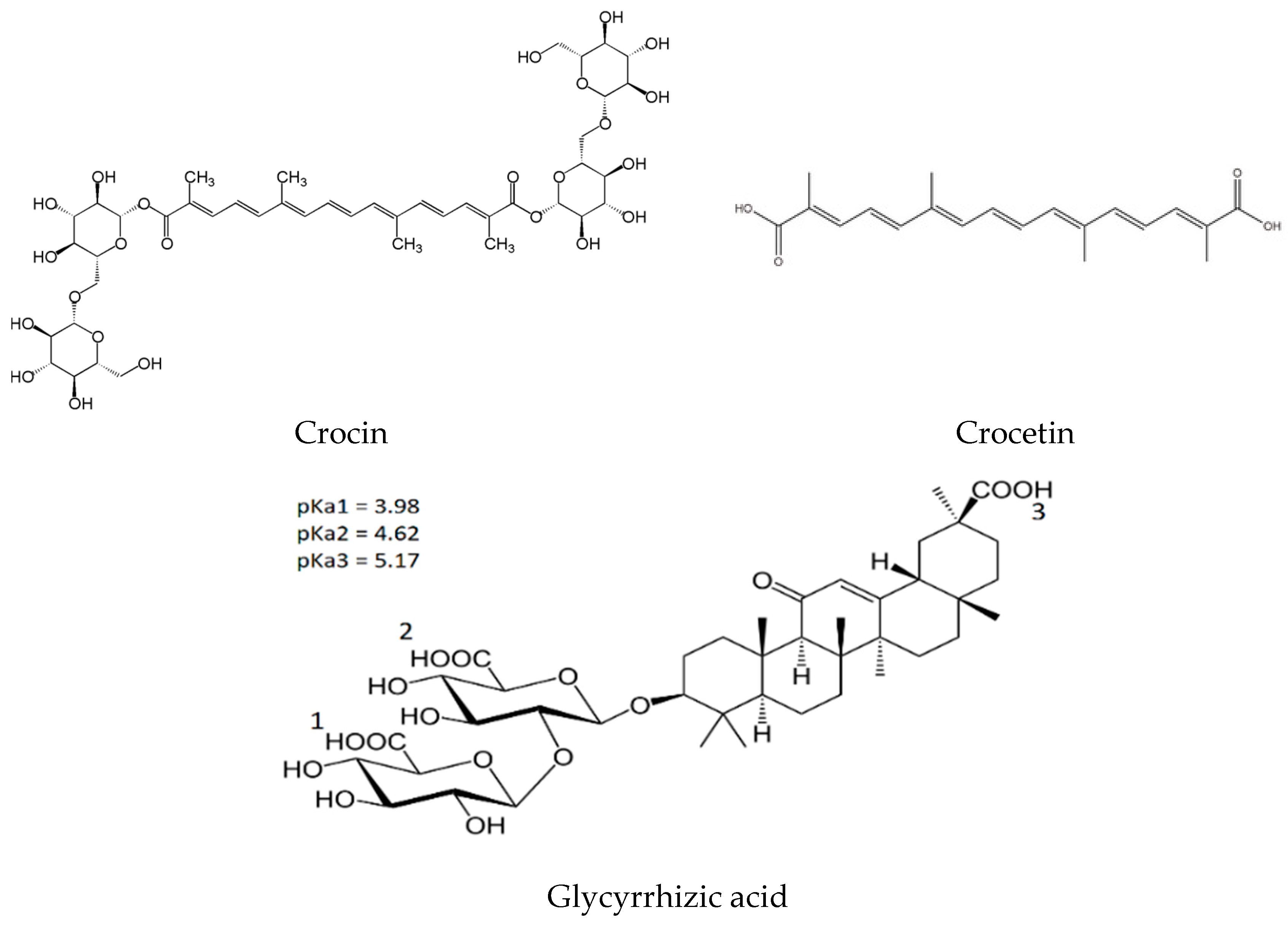
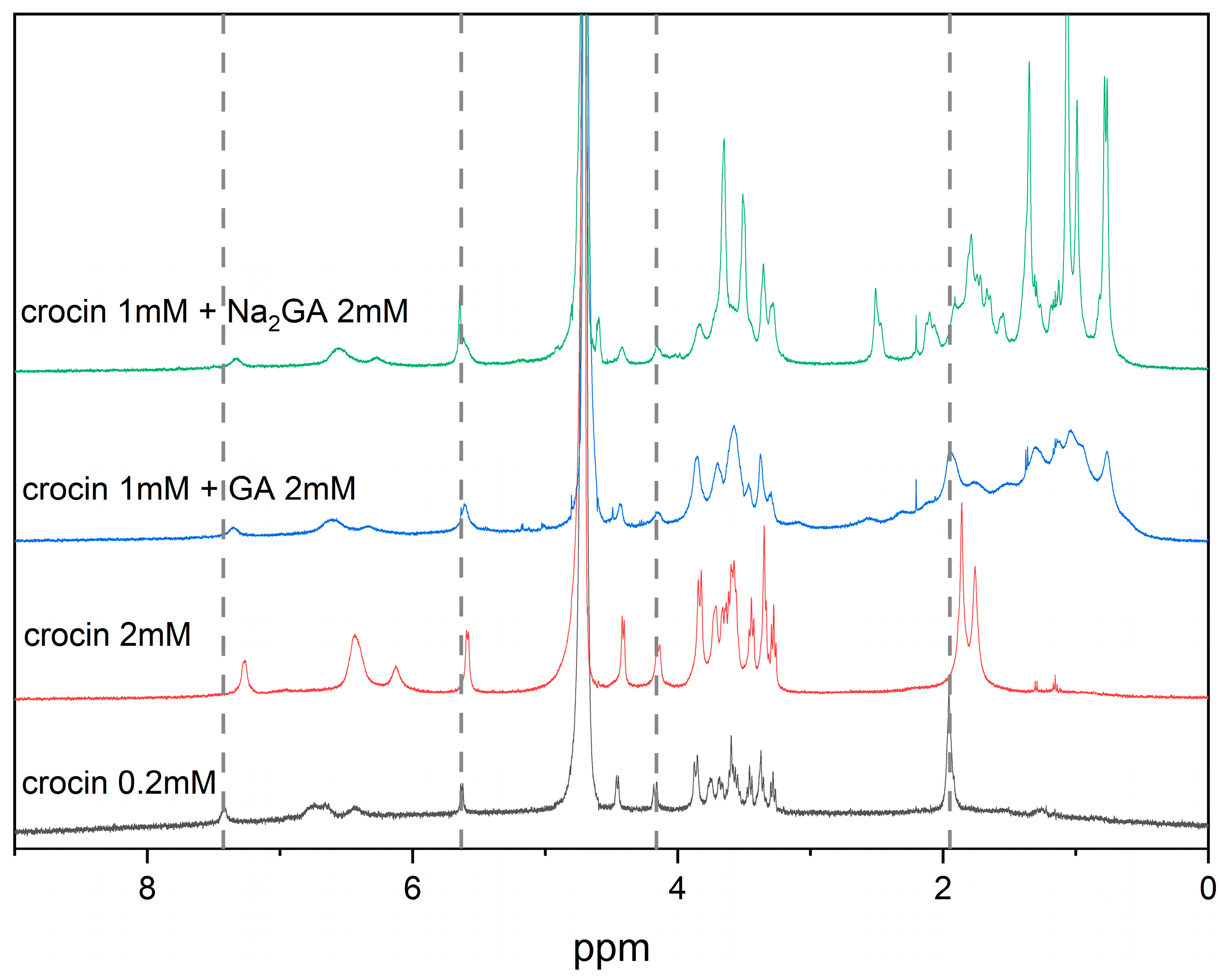
2.1. NMR Study of Crocin/GA Mixed Micelles in Water Solution
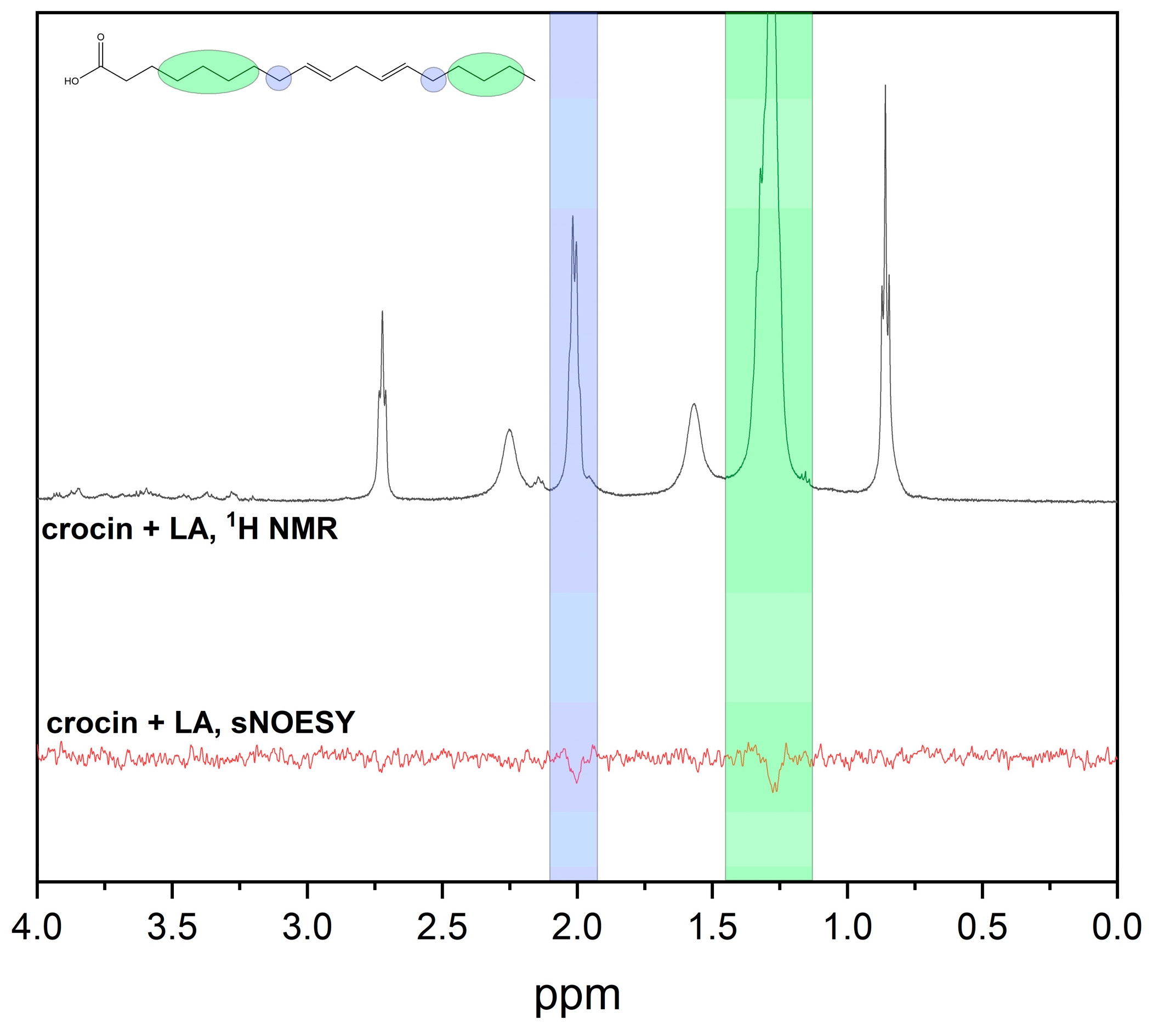
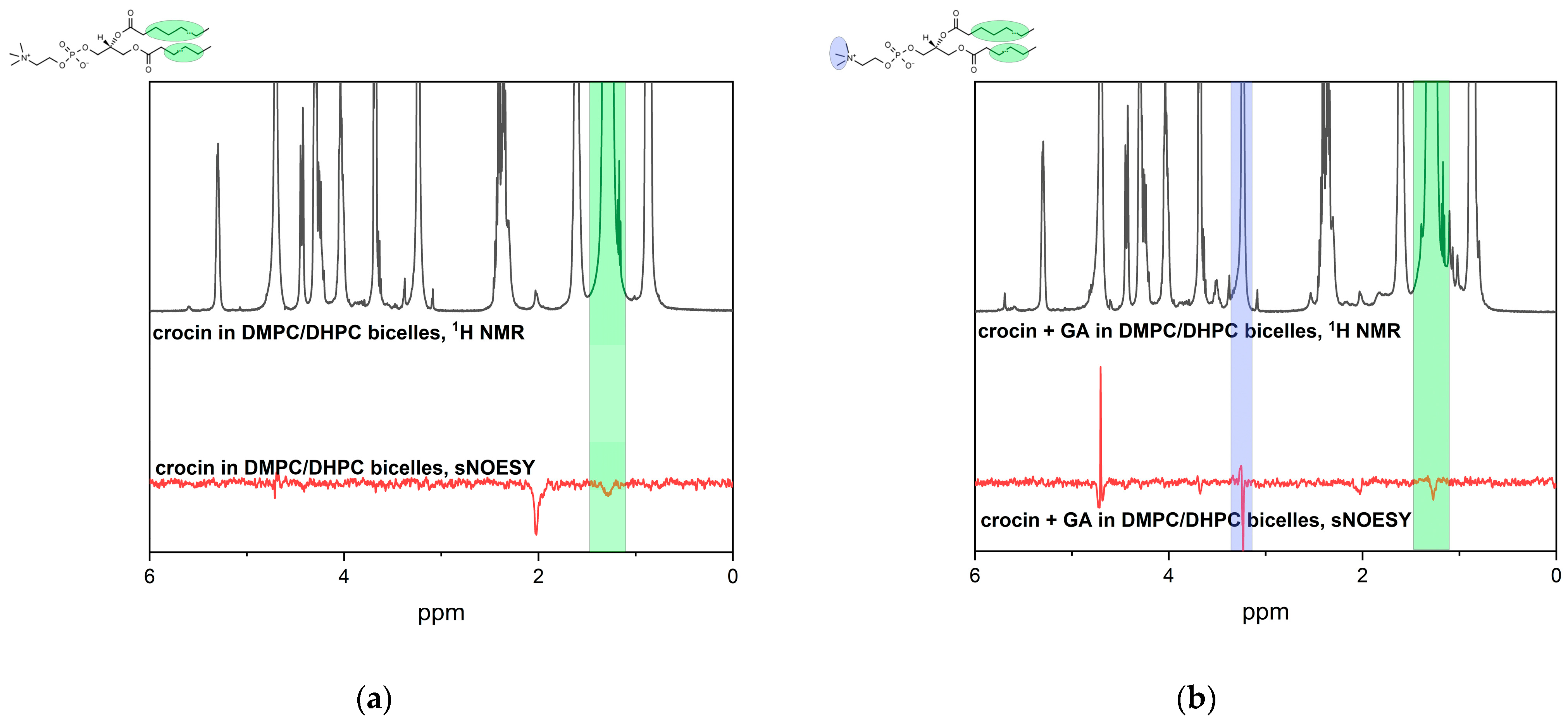
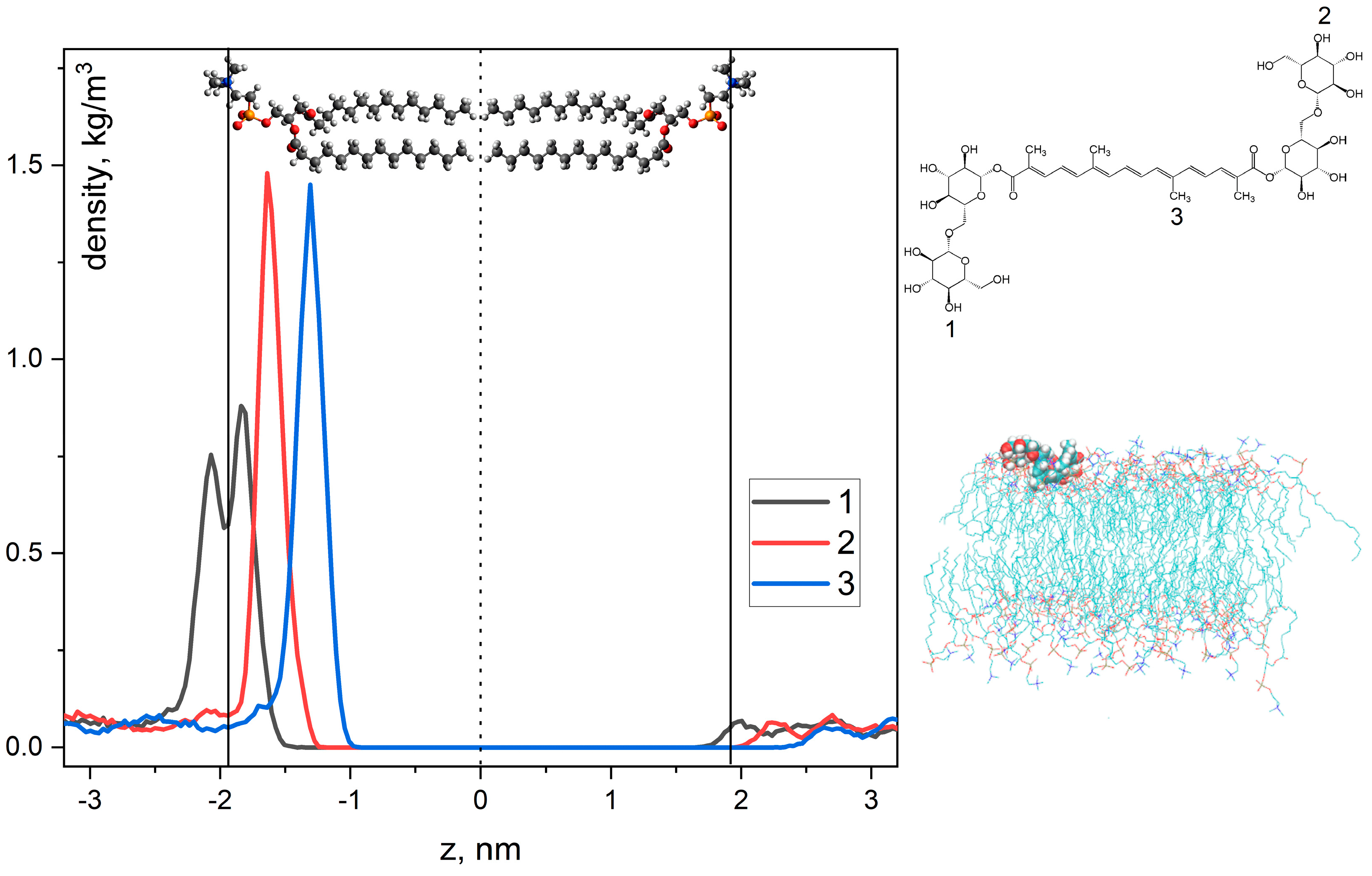
2.2. NMR and MD Study of Crocin Interaction with LA Micelles and DMPC/DHPC Bicelles
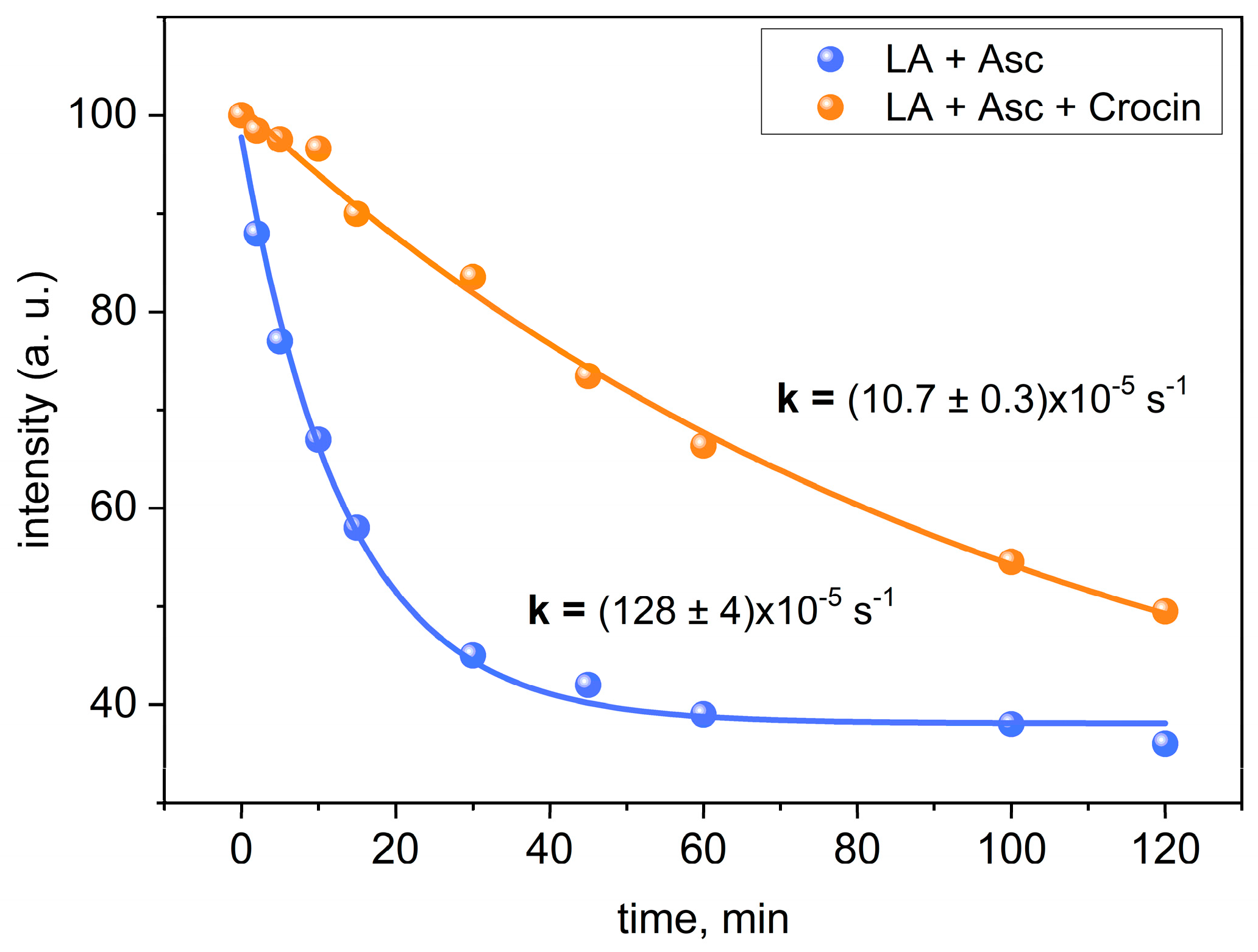
2.3. Peroxidation of Linoleic Acid Micelles in the Presence of Crocin
LOO + LOO → LOOL + O2
3. Materials and Methods
3.1. Materials
3.2. NMR and Selective NOESY Measurements
3.3. Sample Preparation for Lipid Peroxidation Studies
3.4. Sample Preparation for NMR Study of Crocin Interaction with Bicells
3.5. Molecular Dynamics Simulations
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bathaie, S.Z.; Mousavi, S.Z. New Applications and Mechanisms of Action of Saffron and its Important Ingredients. Crit. Rev. Food Sci. Nutr. 2010, 50, 761–786. [Google Scholar] [CrossRef]
- Rahaiee, S.; Moini, S.; Hashemi, M.; Shojaosadati, S.A. Evaluation of antioxidant activities of bioactive compounds and various extracts obtained from saffron (Crocus sativus L.): A review. J. Food Sci. Technol. 2015, 52, 1881–1888. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, S.A.; Kazemi, A.; Sabahi, M.; Razipour, S.; Salehipour, A.; Ghiasian, M.; Ghasemi, H.; Ranjbar, A. Probable antioxidant therapy of Saffron Crocin in patients with multiple sclerosis: A randomized controlled trial. Biomedicine 2020, 40, 516–521. [Google Scholar] [CrossRef]
- Siddiqui, S.A.; Redha, A.A.; Snoeck, E.R.; Singh, S.; Simal-Gandara, J.; Ibrahim, S.A.; Jafari, S.M. Anti-Depressant Properties of Crocin Molecules in Saffron. Molecules 2022, 27, 2076. [Google Scholar] [CrossRef] [PubMed]
- Ghasemnejad-Berenji, M. Immunomodulatory and anti-inflammatory potential of crocin in COVID-19 treatment. J. Food Biochem. 2021, 45, e13718. [Google Scholar] [CrossRef]
- Bao, X.; Hu, J.; Zhao, Y.; Jia, R.; Zhang, H.; Xia, L. Advances on the anti-tumor mechanisms of the carotenoid Crocin. PeerJ 2023, 11, e15535. [Google Scholar] [CrossRef]
- Chen, Y.; Cai, L.; Zhao, C.; Xu, H.C.; Cao, C.Y.; Liu, Y.; Jia, L.; Yin, H.X.; Chen, C.; Zhang, H. Spectroscopic, stability and radical-scavenging properties of a novel pigment from gardenia. Food Chem. 2008, 109, 269–277. [Google Scholar] [CrossRef] [PubMed]
- White, L.J.; Boles, J.E.; Hilton, K.L.F.; Ellaby, R.J.; Hiscock, J.R. Towards the Application of Supramolecular Self-Associating Amphiphiles as Next-Generation Delivery Vehicles. Molecules 2020, 25, 4126. [Google Scholar] [CrossRef]
- Rutkauskaite, A.; White, L.J.; Hilton, K.L.F.; Picci, G.; Croucher, L.; Caltagirone, C.; Hiscock, J.R. Supramolecular self-associating amphiphiles: Determination of molecular self-association properties and calculation of critical micelle concentration using a high-throughput, optical density based methodology. Org. Biomol. Chem. 2022, 20, 5999–6006. [Google Scholar] [CrossRef]
- Dora, N.O.; Blackburn, E.; Boles, J.E.; Williams, G.T.; White, L.J.; Turner, S.E.G.; Hothersall, J.D.; Askwith, T.; Doolan, J.A.; Mulvihill, D.P.; et al. Supramolecular self-associating amphiphiles (SSAs) as nanoscale enhancers of cisplatin anticancer activity. RSC Adv. 2021, 11, 14213–14217. [Google Scholar] [CrossRef]
- Kuperkar, K.; Patel, D.; Atanase, L.I.; Bahadur, P. Amphiphilic Block Copolymers: Their Structures, and Self-Assembly to Polymeric Micelles and Polymersomes as Drug Delivery Vehicles. Polymers 2022, 14, 4702. [Google Scholar] [CrossRef]
- Petrova, S.S.; Schlotgauer, A.A.; Kruppa, A.I.; Leshina, T.V. Self-Association of Glycyrrhizic Acid. NMR Study. Z. Für Phys. Chem. 2017, 231, 839–855. [Google Scholar] [CrossRef]
- Khan, Z.; Malik, M.A.; Al-Thabaiti, S.A.; Bashir, O.; Khan, T.A. Natural dye bolaform sugar-based surfactant: Self aggregation and mixed micellization with ionic surfactants. Dye. Pigment 2016, 131, 168–176. [Google Scholar] [CrossRef]
- Zaheer, Z.; Kosa, S.A.; Akram, M. Solubilization of Congo red into non-ionic bolaform sugar based surfactant: A multi spectroscopic approach. J. Saudi Chem. Soc. 2021, 25, 101257. [Google Scholar] [CrossRef]
- Khan, Z.; Al-thabaiti, S. Micellization and aggregation behavior of crocin with bovine serum albumin: A biophysical study. J. Mol. Liq. 2018, 264, 17–23. [Google Scholar] [CrossRef]
- Kanakis, C.D.; Tarantilis, P.A.; Tajmir-Riahi, H.A.; Polissiou, M.G. Crocetin, dimethylcrocetin, and safranal bind human serum albumin: Stability and antioxidative properties. J. Agric. Food Chem. 2007, 55, 970–977. [Google Scholar] [CrossRef] [PubMed]
- Maggi, L.; Carmona, M.; Zalacain, A.; Tomé, M.M.; Murcia, M.A.; Alonso, G.L. Parabens as Agents for Improving Crocetin Esters’ Shelf-Life in Aqueous Saffron Extracts. Molecules 2009, 14, 1160. [Google Scholar] [CrossRef] [PubMed]
- Focsan, A.L.; Polyakov, N.E.; Kispert, L.D. Carotenoids: Importance in Daily Life-Insight Gained from EPR and ENDOR. Appl. Magn. Reson. 2021, 52, 1093–1112. [Google Scholar] [CrossRef] [PubMed]
- Carmona, M.; Zalacain, A.; Pardo, J.E.; López, E.; Alvarruiz, A.; Alonso, G.L. Influence of different drying and aging conditions on saffron constituents. J. Agric. Food Chem. 2005, 53, 3974–3979. [Google Scholar] [CrossRef] [PubMed]
- Tsimidou, M.; Biliaderis, C.G. Kinetic Studies of Saffron (Crocus sativus L.) Quality Deterioration. J. Agric. Food Chem. 1997, 45, 2890–2898. [Google Scholar] [CrossRef]
- Ligia Focsan, A.; Polyakov, N.E.; Kispert, L.D. Supramolecular Carotenoid Complexes of Enhanced Solubility and Stability—The Way of Bioavailability Improvement. Molecules 2019, 24, 3947. [Google Scholar] [CrossRef] [PubMed]
- Polyakov, N.E.; Focsan, A.L.; Gao, Y.; Kispert, L.D. The Endless World of Carotenoids—Structural, Chemical and Biological Aspects of Some Rare Carotenoids. Int. J. Mol. Sci. 2023, 24, 9885. [Google Scholar] [CrossRef] [PubMed]
- Bera, S.; Mitra, R.; Singh, J. Recent advancement in protected delivery methods for carotenoid: A smart choice in modern nutraceutical formulation concept. Biotechnol. Genet. Eng. Rev. 2023, 1–57. [Google Scholar] [CrossRef]
- Bodbodak, S.; Nejatian, M.; Ghandehari Yazdi, A.P.; Kamali Rousta, L.; Rafiee, Z.; Jalali-Jivan, M.; Kharazmi, M.S.; Jafari, S.M. Improving the thermal stability of natural bioactive ingredients via encapsulation technology. Crit. Rev. Food Sci. Nutr. 2022. [Google Scholar] [CrossRef] [PubMed]
- Apanasenko, I.E.; Selyutina, O.Y.; Polyakov, N.E.; Suntsova, L.P.; Meteleva, E.S.; Dushkin, A.V.; Vachali, P.; Bernstein, P.S. Solubilization and stabilization of macular carotenoids by water soluble oligosaccharides and polysaccharides. Arch. Biochem. Biophys. 2015, 572, 58–65. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, Y. Construction of lipid-biomacromolecular compounds for loading and delivery of carotenoids: Preparation methods, structural properties, and absorption-enhancing mechanisms. Crit. Rev. Food Sci. Nutr. 2024. [Google Scholar] [CrossRef]
- Akhavan, S.; Assadpour, E.; Katouzian, I.; Jafari, S.H. Lipid nano scale cargos for the protection and delivery of food bioactive ingredients and nutraceuticals. Trends Food Sci. Technol. 2018, 74, 132–146. [Google Scholar] [CrossRef]
- Liao, Y.; Li, Z.; Zhou, Q.; Sheng, M.; Qu, Q.; Shi, Y.; Yang, J.; Lv, L.; Dai, X.; Shi, X. Saponin surfactants used in drug delivery systems: A new application for natural medicine components. Int. J. Pharm. 2021, 603, 120709. [Google Scholar] [CrossRef]
- Selyutina, O.Y.; Polyakov, N.E. Glycyrrhizic acid as a multifunctional drug carrier—From physicochemical properties to biomedical applications: A modern insight on the ancient drug. Int. J. Pharm. 2019, 559, 271. [Google Scholar] [CrossRef]
- Ghafari, F.; Sadeghian, Z.; Oftadeh Harsin, A.; Khodabandelo, S.; Ranjbar, A. Anti-oxidative properties of nanocrocin in Zearalenone induced toxicity on Hek293 cell; The novel formulation and cellular assessment. Hum. Exp. Toxicol. 2023, 42, 09603271231169911. [Google Scholar] [CrossRef]
- Mirhadi, E.; Nassirli, H.; Malaekeh-Nikouei, B. An updated review on therapeutic effects of nanoparticle-based formulations of saffron components (safranal, crocin, and crocetin). J. Pharm. Investig. 2020, 50, 47–58. [Google Scholar] [CrossRef]
- Shibata, S. A Drug over the Millennia: Pharmacognosy, Chemistry, and Pharmacology of Licorice. Yakugaku Zasshi 2000, 120, 849–862. [Google Scholar] [CrossRef]
- Fiore, C.; Eisenhut, M.; Ragazzi, E.; Zanchin, G.; Armanini, D. A history of the therapeutic use of liquorice in Europe. J. Ethnopharmacol. 2005, 99, 317–324. [Google Scholar] [CrossRef]
- Ming, L.J.; Yin, A.C.Y. Therapeutic Effects of Glycyrrhizic Acid. Nat. Prod. Commun. 2013, 8, 415–418. [Google Scholar] [CrossRef]
- Hasan, M.K.; Ara, I.; Mondal, M.S.A.; Kabir, Y. Phytochemistry, pharmacological activity, and potential health benefits of Glycyrrhiza glabra. Heliyon 2021, 7, e07240. [Google Scholar] [CrossRef]
- Andersen, F.A. Final Report on the Safety Assessment of Glycyrrhetinic Acid, Potassium Glycyrrhetinate, Disodium Succinoyl Glycyrrhetinate, Glyceryl Glycyrrhetinate, Glycyrrhetinyl Stearate, Stearyl Glycyrrhetinate, Glycyrrhizic Acid, Ammonium Glycyrrhizate, Dipotassium Glycyrrhizate, Disodium Glycyrrhizate, Trisodium Glycyrrhizate, Methyl Glycyrrhizate, and Potassium Glycyrrhizinate. Int. J. Toxicol. 2007, 26 (Suppl. 2), 79–112. [Google Scholar] [CrossRef]
- Bi, X.; Yang, L.; Lin, Y.; Deng, W.; Jiang, T.; Zhang, L.; Lu, Y.; Yi, W.; Xie, Y.; Li, M. Efficacy and Safety of Glycyrrhizic Acid in Treatment of Autoimmune Hepatitis. Am. J. Chin. Med. 2023, 51, 391–405. [Google Scholar] [CrossRef]
- Li, M.; Xiang, L.; Li, Y. Efficacy and safety of compound glycyrrhizin in the patients with vitiligo: A systematic review and meta-analysis. Expert Rev. Clin. Pharmacol. 2023, 16, 601–611. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Sheng, Z.; Xiao, J.; Li, Y.; Huang, J.; Jia, J.; Zeng, X.; Li, L. Advances in the roles of glycyrrhizic acid in cancer therapy. Front. Pharmacol. 2023, 14, 1265172. [Google Scholar] [CrossRef] [PubMed]
- Harikrishnan, R.; Devi, G.; Van Doan, H.; Jawahar, S.; Balasundaram, C.; Saravanan, K.; Arockiaraj, J.; Soltani, M.; Jaturasitha, S. Study on antioxidant potential, immunological response, and inflammatory cytokines induction of glycyrrhizic acid (GA) in silver carp against vibriosis. Fish Shellfish. Immunol. 2021, 119, 193–208. [Google Scholar] [CrossRef]
- Takayama, F.; Egashira, T.; Yamanaka, Y. Effects of glycyrrhizin on lipid peroxidation induced by lipopolysaccharide. Jpn. J. Pharmacol. 1995, 67, 104. [Google Scholar] [CrossRef]
- Polyakov, N.E.; Leshina, T.V.; Salakhutdinov, N.F.; Kispert, L.D. Host-guest complexes of carotenoids with β-glycyrrhizic acid. J. Phys. Chem. B 2006, 110, 6991–6998. [Google Scholar] [CrossRef] [PubMed]
- Polyakov, N.E.; Leshina, T.V.; Salakhutdinov, N.F.; Konovalova, T.A.; Kispert, L.D. Antioxidant and redox properties of supramolecular complexes of carotenoids with β-glycyrrhizic acid. Free Rad. Biol. Med. 2006, 40, 1804–1809. [Google Scholar]
- Popova, M.V.; Tchernyshev, Y.S.; Michel, D. NMR investigation of the short-chain ionic surfactant-water systems. Langmuir 2004, 20, 632–636. [Google Scholar] [CrossRef] [PubMed]
- Haouas, M.; Falaise, C.; Leclerc, N.; Floquet, S.; Cadot, E. NMR spectroscopy to study cyclodextrin-based host–guest assemblies with polynuclear clusters. Dalt. Trans. 2023, 52, 13467–13481. [Google Scholar] [CrossRef] [PubMed]
- Dewanjee, S.; Bhattacharjee, N.; Chakraborty, P.; Bhattacharjee, S. Carotenoids as Antioxidants. In Carotenoids: Structure and Function in the Human Body; Zia-Ul-Haq, M., Dewanjee, S., Riaz, M., Eds.; Springer: Cham, Switzerland, 2021. [Google Scholar] [CrossRef]
- Mihaljević, B.; Tartaro, I.; Ferreri, C.; Chatgilialoglu, C. Linoleic acid peroxidation vs. isomerization: A biomimetic model of free radical reactivity in the presence of thiols. Org. Biomol. Chem. 2011, 9, 3541–3548. [Google Scholar] [CrossRef]
- Laguerre, M.; Tenon, M.; Bily, A.; Birti, S.; Laguerre, M.; Tenon, M.; Bily, A.; Birti, S. Toward a Spatiotemporal Model of Oxidation in Lipid Dispersions: A Hypothesis-Driven Review. Eur. J. Lipid Sci. Technol. 2020, 122, 1900209. [Google Scholar] [CrossRef]
- Timoshnikov, V.A.; Kichigina, L.A.; Selyutina, O.Y.; Polyakov, N.E.; Kontoghiorghes, G.J. Antioxidant Activity of Deferasirox and Its Metal Complexes in Model Systems of Oxidative Damage: Comparison with Deferiprone. Molecules 2021, 26, 5064. [Google Scholar] [CrossRef]
- Cotticelli, M.G.; Crabbe, A.M.; Wilson, R.B.; Shchepinov, M.S. Insights into the role of oxidative stress in the pathology of Friedreich ataxia using peroxidation resistant polyunsaturated fatty acids. Redox Biol. 2013, 1, 398–404. [Google Scholar] [CrossRef]
- Pham, A.N.; Xing, G.; Miller, C.J.; Waite, T.D. Fenton-like copper redox chemistry revisited: Hydrogen peroxide and superoxide mediation of copper-catalyzed oxidant production. J. Catal. 2013, 301, 54–64. [Google Scholar] [CrossRef]
- Schneider, C. An update on products and mechanisms of lipid peroxidation. Mol. Nutr. Food Res. 2009, 53, 315–321. [Google Scholar] [CrossRef] [PubMed]
- Spiteller, G. Linoleic acid peroxidation—The dominant lipid peroxidation process in low density lipoprotein—And its relationship to chronic diseases. Chem. Phys. Lipids 1998, 95, 105–162. [Google Scholar] [CrossRef]
- Zhu, T.F.; Budin, I.; Szostak, J.W. Preparation of Fatty Acid Micelles. Methods Enzymol. 2013, 533, 283–288. [Google Scholar] [CrossRef]
- Stroet, M.; Caron, B.; Visscher, K.M.; Geerke, D.P.; Malde, A.K.; Mark, A.E. Automated Topology Builder Version 3.0: Prediction of Solvation Free Enthalpies in Water and Hexane. J. Chem. Theory Comput. 2018, 14, 5834–5845. [Google Scholar] [CrossRef] [PubMed]
- Poger, D.; Mark, A.E. On the validation of molecular dynamics simulations of saturated and cis-monounsaturated phosphatidylcholine lipid bilayers: A comparison with experiment. J. Chem. Theory Comput. 2010, 6, 325–336. [Google Scholar] [CrossRef] [PubMed]
- Parrinello, M.; Rahman, A. Polymorphic transitions in single crystals: A new molecular dynamics method. J. Appl. Phys. 1981, 52, 7182–7190. [Google Scholar] [CrossRef]
- Hoover, W.G. Canonical dynamics: Equilibrium phase-space distributions. Phys. Rev. A 1985, 31, 1695–1697. [Google Scholar] [CrossRef]
- Selyutina, O.Y.; Apanasenko, I.E.; Shilov, A.G.; Khalikov, S.S.; Polyakov, N.E. Effect of natural polysaccharides and oligosaccharides on the permeability of cell membranes. Russ. Chem. Bull. 2017, 66, 129–135. [Google Scholar] [CrossRef]
- Hoseini, M.; Sharifi, S.; Sazgarnia, A. The Influence of Anionic, Cationic Surfactant and AOT/Water/Heptane Reverse Micelle on Photophysical Properties of Crocin: Compare with RPMI Effect. J. Fluoresc. 2020, 30, 665–677. [Google Scholar] [CrossRef]
- Selyutina, O.Y.; Kononova, P.A.; Koshman, V.E.; Shelepova, E.A.; Azad, M.G.; Afroz, R.; Dharmasivam, M.; Bernhardt, P.V.; Polyakov, N.E.; Richardson, D.R. Ascorbate-and iron-driven redox activity of Dp44mT and Emodin facilitates peroxidation of micelles and bicelles. Biochim. Biophys. Acta—Gen. Subj. 2022, 1866, 130078. [Google Scholar] [CrossRef]




| System | Rate Constant, in 10−5 s−1 |
|---|---|
| LA | 17 ± 1 |
| LA + 1 mM crocin | 2.9 ± 0.2 |
| LA + 1 mM Na2GA | 1.8 ± 0.2 |
| LA + 0.1 mM crocin + 1 mM Na2GA | 1.8 ± 0.2 |
| LA + 2.5 mM ascorbate | 128 ± 4 |
| LA + 1 mM crocin + ascorbate | 10.7 ± 0.3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Su, W.; Mastova, A.V.; Ul’yanova, M.A.; Kononova, P.A.; Selyutina, O.Y.; Evseenko, V.I.; Meteleva, E.S.; Dushkin, A.V.; Su, W.; Polyakov, N.E. NMR Study of Water-Soluble Carotenoid Crocin: Formation of Mixed Micelles, Interaction with Lipid Membrane and Antioxidant Activity. Int. J. Mol. Sci. 2024, 25, 3194. https://doi.org/10.3390/ijms25063194
Su W, Mastova AV, Ul’yanova MA, Kononova PA, Selyutina OY, Evseenko VI, Meteleva ES, Dushkin AV, Su W, Polyakov NE. NMR Study of Water-Soluble Carotenoid Crocin: Formation of Mixed Micelles, Interaction with Lipid Membrane and Antioxidant Activity. International Journal of Molecular Sciences. 2024; 25(6):3194. https://doi.org/10.3390/ijms25063194
Chicago/Turabian StyleSu, Wenjing, Anna V. Mastova, Maya A. Ul’yanova, Polina A. Kononova, Olga Yu. Selyutina, Veronika I. Evseenko, Elizaveta S. Meteleva, Alexander V. Dushkin, Weike Su, and Nikolay E. Polyakov. 2024. "NMR Study of Water-Soluble Carotenoid Crocin: Formation of Mixed Micelles, Interaction with Lipid Membrane and Antioxidant Activity" International Journal of Molecular Sciences 25, no. 6: 3194. https://doi.org/10.3390/ijms25063194
APA StyleSu, W., Mastova, A. V., Ul’yanova, M. A., Kononova, P. A., Selyutina, O. Y., Evseenko, V. I., Meteleva, E. S., Dushkin, A. V., Su, W., & Polyakov, N. E. (2024). NMR Study of Water-Soluble Carotenoid Crocin: Formation of Mixed Micelles, Interaction with Lipid Membrane and Antioxidant Activity. International Journal of Molecular Sciences, 25(6), 3194. https://doi.org/10.3390/ijms25063194







