Biomarkers to Predict Lethal Radiation Injury to the Rat Lung
Abstract
1. Introduction
2. Results
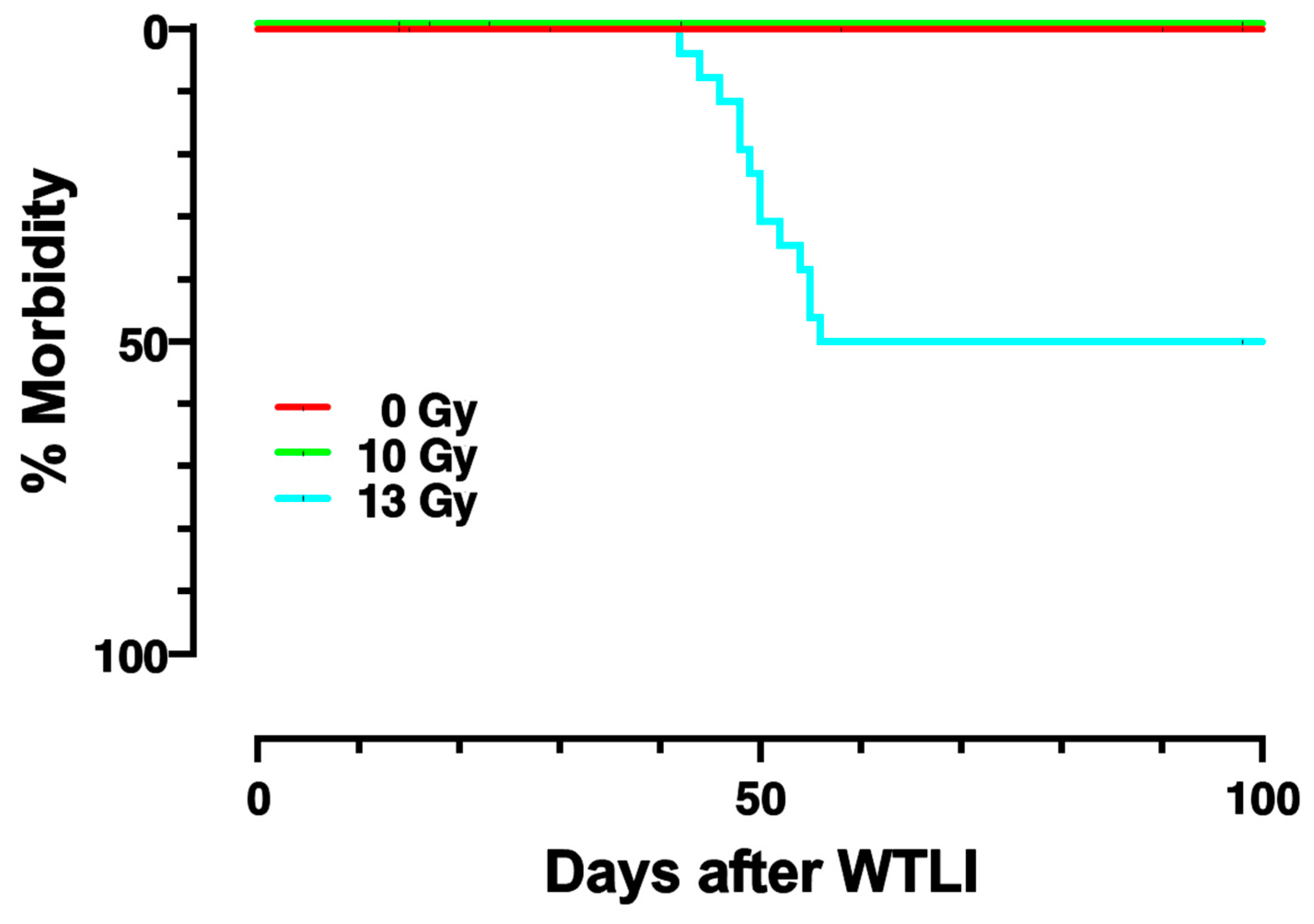
2.1. Dose-Response and Survival after WTLI
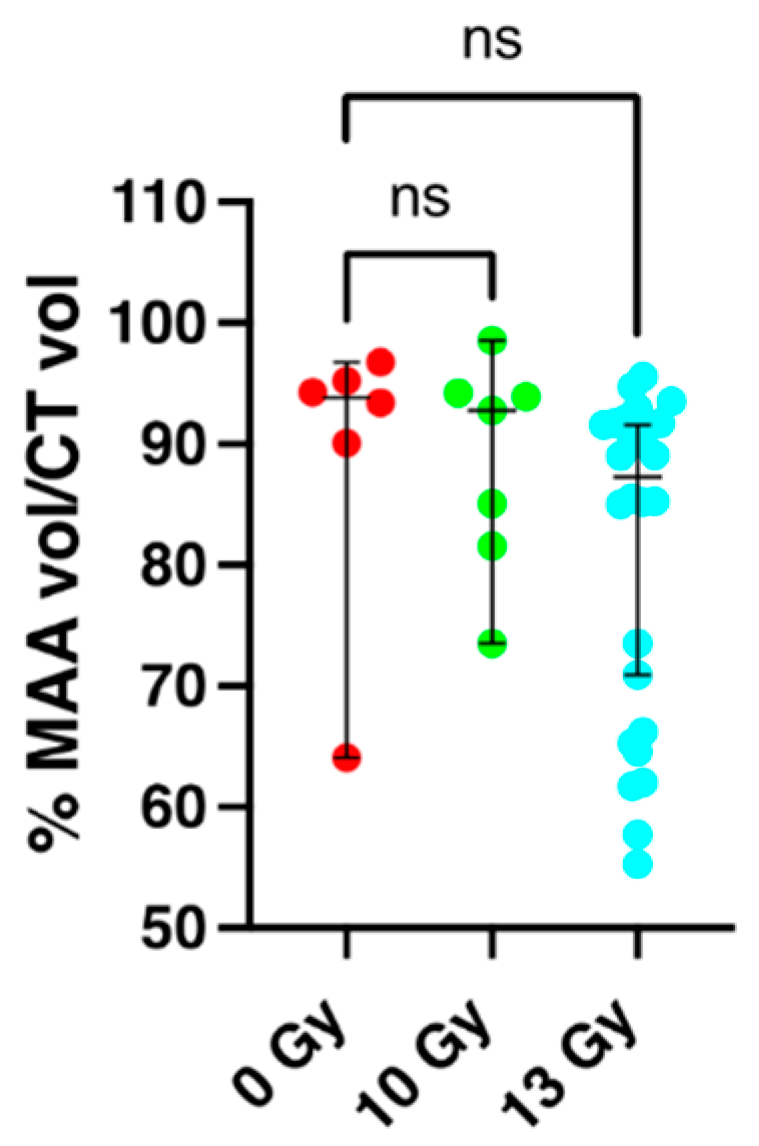
2.2. Perfusion Volume of the Lung and Apoptosis as Measured by SPECT/CT Imaging at 2 Weeks Post-Irradiation
2.3. Changes in Circulating White Blood Cell Counts at 2 Weeks after Irradiation
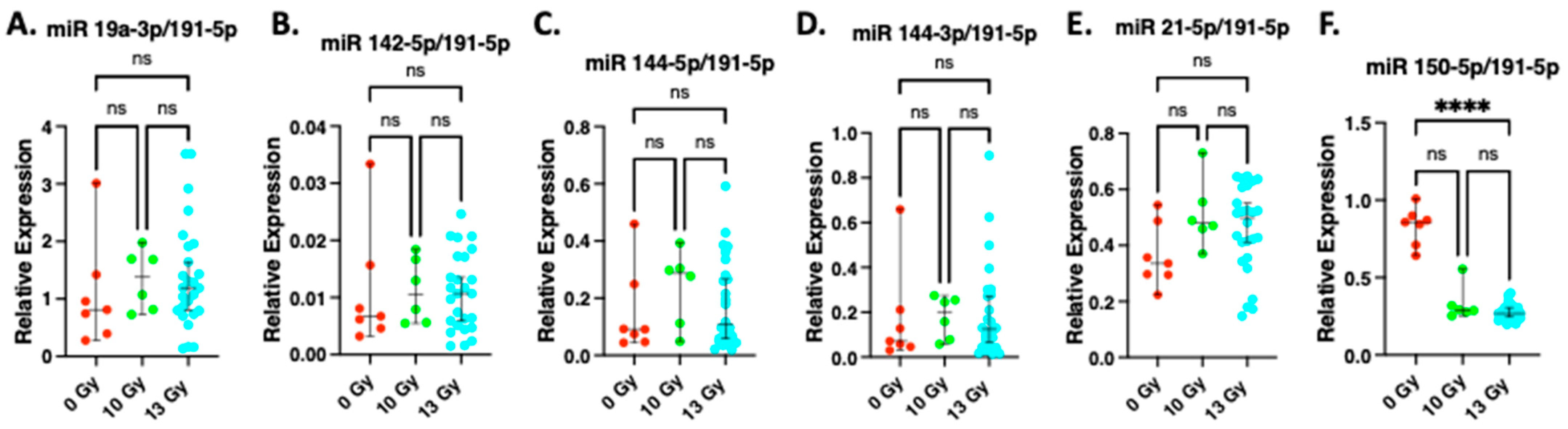
2.4. Expression of miR19a-3p, miR142-5p, miR 144-5p, miR 144-3p, miR 21-5p at 2 Weeks after Irradiation
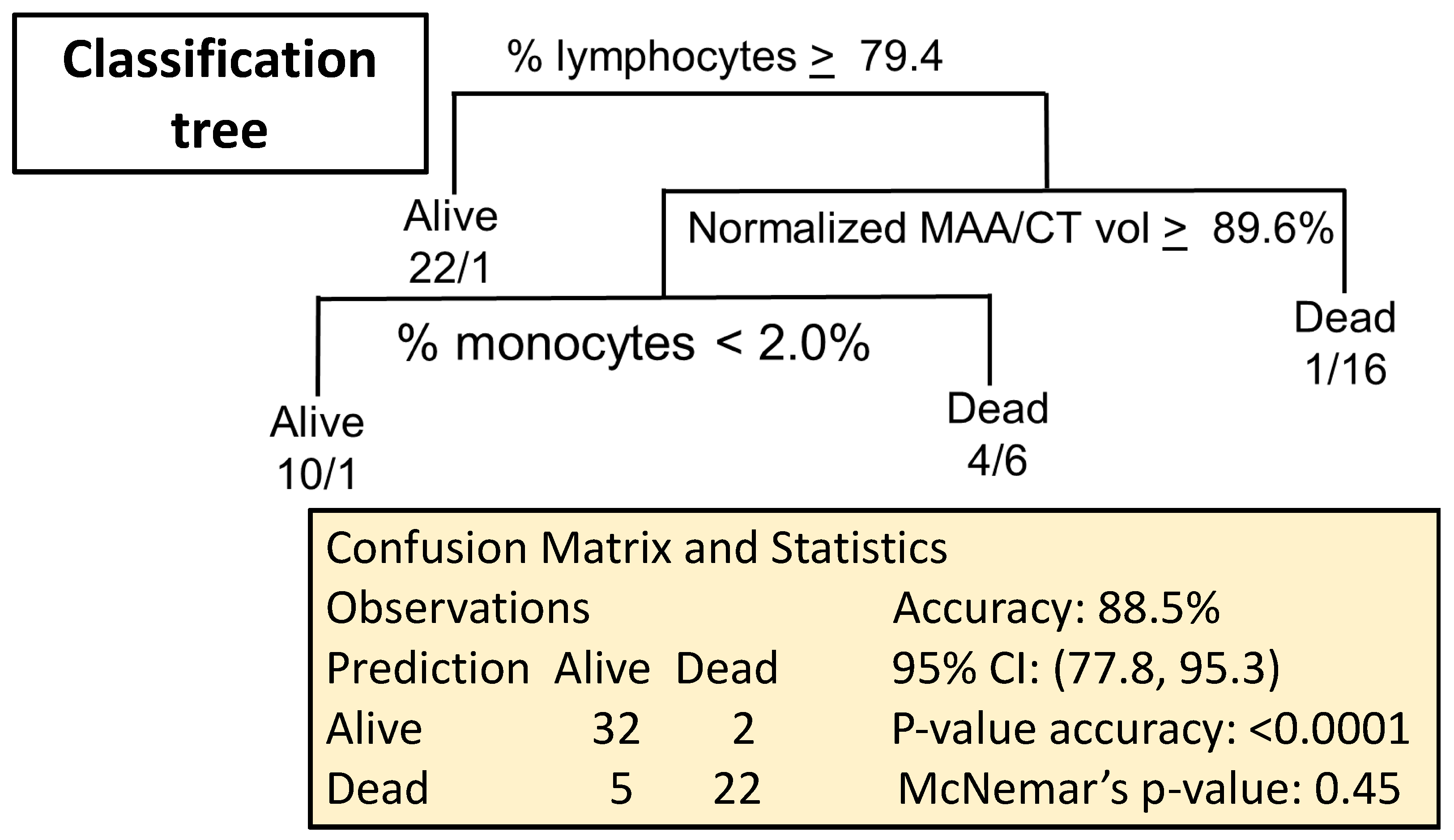
2.5. Combining Biomarkers to Predict Lethal Lung Irradiation
2.5.1. Data Import
2.5.2. Imputation Procedure
2.5.3. Predicting Survival
3. Discussion
4. Materials and Methods
4.1. Animal Care
4.2. Irradiation and Follow Up
4.3. SPECT/CT
4.4. Image Analysis
4.5. Blood Cell Counts
4.6. Measurements of Expression of Circulating microRNA
4.7. Statistical Analyses
4.8. Strategy for Identifying and Testing Biomarkers
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- DiCarlo, A.L.; Homer, M.J.; Coleman, C.N. United States medical preparedness for nuclear and radiological emergencies. J. Radiol. Prot. 2021, 41, 1420–1434. [Google Scholar] [CrossRef]
- Bisserier, M.; Shanmughapriya, S.; Rai, A.K.; Gonzalez, C.; Brojakowska, A.; Garikipati, V.N.S.; Madesh, M.; Mills, P.J.; Walsh, K.; Arakelyan, A.; et al. Cell-Free Mitochondrial DNA as a Potential Biomarker for Astronauts’ Health. J. Am. Heart Assoc. 2021, 10, e022055. [Google Scholar] [CrossRef]
- Zhang, L.; Zhang, M.; Yang, S.; Cao, Y.; Zhang, S.B.; Yin, L.; Tian, Y.; Ma, Y.; Zhang, A.; Okunieff, P.; et al. A new biodosimetric method: Branched DNA-based quantitative detection of B1 DNA in mouse plasma. Br. J. Radiol. 2010, 83, 694–701. [Google Scholar] [CrossRef]
- Aryankalayil, M.J.; Martello, S.; Bylicky, M.A.; Chopra, S.; May, J.M.; Shankardass, A.; MacMillan, L.; Sun, L.; Sanjak, J.; Vanpouille-Box, C.; et al. Analysis of lncRNA-miRNA-mRNA expression pattern in heart tissue after total body radiation in a mouse model. J. Transl. Med. 2021, 19, 336. [Google Scholar] [CrossRef]
- Rogers, C.J.; Kyubwa, E.M.; Lukaszewicz, A.I.; Starbird, M.A.; Nguyen, M.; Copeland, B.T.; Yamada-Hanff, J.; Menon, N. Observation of Unique Circulating miRNA Signatures in Non-Human Primates Exposed to Total-Body vs. Whole Thorax Lung Irradiation. Radiat. Res. 2021, 196, 547–559. [Google Scholar] [CrossRef]
- Yadav, M.; Bhayana, S.; Liu, J.; Lu, L.; Huang, J.; Ma, Y.; Qamri, Z.; Mo, X.; Jacob, D.S.; Parasa, S.T. Two-miRNA–based finger-stick assay for estimation of absorbed ionizing radiation dose. Sci. Transl. Med. 2020, 12, eaaw5831. [Google Scholar] [CrossRef]
- Ghandhi, S.A.; Shuryak, I.; Morton, S.R.; Amundson, S.A.; Brenner, D.J. New approaches for quantitative reconstruction of radiation dose in human blood cells. Sci. Rep. 2019, 9, 18441. [Google Scholar] [CrossRef]
- Groves, A.M.; Williams, J.P.; Hernady, E.; Reed, C.; Fenton, B.; Love, T.; Finkelstein, J.N.; Johnston, C.J. A potential biomarker for predicting the risk of radiation-induced fibrosis in the lung. Radiat. Res. 2018, 190, 513–525. [Google Scholar] [CrossRef]
- Nongrum, S.; Vaiphei, S.T.; Keppen, J.; Ksoo, M.; Kashyap, E.; Sharan, R.N. Identification and preliminary validation of radiation response protein (s) in human blood for a high-throughput molecular biodosimetry technology for the future. Genome Integr. 2017, 8, 5. [Google Scholar] [CrossRef]
- Ostheim, P.; Amundson, S.A.; Badie, C.; Bazyka, D.; Evans, A.C.; Ghandhi, S.A.; Gomolka, M.; López Riego, M.; Rogan, P.K.; Terbrueggen, R. Gene expression for biodosimetry and effect prediction purposes: Promises, pitfalls and future directions–key session ConRad 2021. Int. J. Radiat. Biol. 2022, 98, 843–854. [Google Scholar] [CrossRef]
- Zalesak-Kravec, S.; Huang, W.; Wang, P.; Yu, J.; Liu, T.; Defnet, A.E.; Moise, A.R.; Farese, A.M.; MacVittie, T.J.; Kane, M.A. Multi-omic analysis of non-human primate heart after partial-body radiation with minimal bone marrow sparing. Health Phys. 2021, 121, 352–371. [Google Scholar] [CrossRef]
- Hayes, J.M.; Olson, J.D.; Chino, Y.; Bourland, J.D.; Cline, J.M.; Johnson, T.E. Pseudo Pelger-Huët anomalies as potential biomarkers for acute exposure radiation dose in rhesus macaques (Macaca mulatta). Int. J. Radiat. Biol. 2022, 98, 913–923. [Google Scholar] [CrossRef]
- Fish, B.L.; Gao, F.; Narayanan, J.; Bergom, C.; Jacobs, E.R.; Cohen, E.P.; Moulder, J.E.; Orschell, C.M.; Medhora, M. Combined Hydration and Antibiotics with Lisinopril to Mitigate Acute and Delayed High-dose Radiation Injuries to Multiple Organs. Health Phys. 2016, 111, 410–419. [Google Scholar] [CrossRef]
- Medhora, M.; Gao, F.; Gasperetti, T.; Narayanan, J.; Khan, A.H.; Jacobs, E.R.; Fish, B.L. Delayed Effects of Acute Radiation Exposure (Deare) in Juvenile and Old Rats: Mitigation by Lisinopril. Health Phys. 2019, 116, 529–545. [Google Scholar] [CrossRef]
- Prado, C.; MacVittie, T.; Bennett, A.; Kazi, A.; Farese, A.; Prado, K. Organ doses associated with partial-body irradiation with 2.5% bone marrow sparing of the non-human primate: A retrospective study. Radiat. Res. 2017, 188, 695–705. [Google Scholar] [CrossRef]
- Salem, A.M.; Jackson, I.L.; Gibbs, A.; Poirier, Y.; Newman, D.; Zodda, A.; Vujaskovic, Z.; Kaytor, M.D.; Serebrenik, A.A.; Gobburu, J. Interspecies Comparison and Radiation Effect on Pharmacokinetics of BIO 300, a Nanosuspension of Genistein, after Different Routes of Administration in Mice and Non-Human Primates. Radiat. Res. 2022, 197, 447–458. [Google Scholar] [CrossRef]
- Fish, B.L.; MacVittie, T.J.; Gao, F.; Narayanan, J.; Gasperetti, T.; Scholler, D.; Sheinin, Y.; Himburg, H.A.; Hart, B.; Medhora, M. Rat Models of Partial-body Irradiation with Bone Marrow-sparing (Leg-out PBI) Designed for FDA Approval of Countermeasures for Mitigation of Acute and Delayed Injuries by Radiation. Health Phys. 2021, 121, 419–433. [Google Scholar] [CrossRef]
- Cohen, E.P.; Hankey, K.G.; Bennett, A.W.; Farese, A.M.; Parker, G.A.; MacVittie, T.J. Acute and Chronic Kidney Injury in a Non-Human Primate Model of Partial-Body Irradiation with Bone Marrow Sparing. Radiat. Res. 2017, 188, 661–671. [Google Scholar] [CrossRef]
- Jackson, I.L.; Zodda, A.; Gurung, G.; Pavlovic, R.; Kaytor, M.D.; Kuskowski, M.A.; Vujaskovic, Z. BIO 300, a nanosuspension of genistein, mitigates pneumonitis/fibrosis following high-dose radiation exposure in the C57L/J murine model. Br. J. Pharmacol. 2017, 174, 4738–4750. [Google Scholar] [CrossRef]
- MacVittie, T.J.; Gibbs, A.; Farese, A.M.; Barrow, K.; Bennett, A.; Taylor-Howell, C.; Kazi, A.; Prado, K.; Parker, G.; Jackson, W. AEOL 10150 Mitigates Radiation-Induced Lung Injury in the Nonhuman Primate: Morbidity and Mortality are Administration Schedule-Dependent. Radiat. Res. 2017, 187, 298–318. [Google Scholar] [CrossRef]
- Zhang, R.; Ghosh, S.N.; Zhu, D.; North, P.E.; Fish, B.L.; Morrow, N.V.; Lowry, T.; Nanchal, R.; Jacobs, E.R.; Moulder, J.E. Structural and functional alterations in the rat lung following whole thoracic irradiation with moderate doses: Injury and recovery. Int. J. Radiat. Biol. 2008, 84, 487–497. [Google Scholar] [CrossRef]
- Ghobadi, G.; Van Der Veen, S.; Bartelds, B.; De Boer, R.A.; Dickinson, M.G.; De Jong, J.R.; Faber, H.; Niemantsverdriet, M.; Brandenburg, S.; Berger, R.M. Physiological interaction of heart and lung in thoracic irradiation. Int. J. Radiat. Oncol. Biol. Phys. 2012, 84, e639–e646. [Google Scholar] [CrossRef]
- Medhora, M.; Gao, F.; Glisch, C.; Narayanan, J.; Sharma, A.; Harmann, L.M.; Lawlor, M.; Snyder, L.A.; Fish, B.L.; Down, J.D.; et al. Whole-thorax irradiation induces hypoxic respiratory failure, pleural effusions and cardiac remodeling. J. Radiat. Res. 2015, 56, 248–260. [Google Scholar] [CrossRef]
- Rabender, C.; Mezzaroma, E.; Mauro, A.G.; Mullangi, R.; Abbate, A.; Anscher, M.; Hart, B.; Mikkelsen, R. IPW-5371 Proves Effective as a Radiation Countermeasure by Mitigating Radiation-Induced Late Effects. Radiat. Res. 2016, 186, 478–488. [Google Scholar] [CrossRef]
- Patterson, A.M.; Plett, P.A.; Chua, H.L.; Sampson, C.H.; Fisher, A.; Feng, H.L.; Unthank, J.L.; Miller, S.J.; Katz, B.P.; MacVittie, T.J.; et al. Development of a Model of the Acute and Delayed Effects of High Dose Radiation Exposure in Jackson Diversity Outbred Mice; Comparison to Inbred C57BL/6 Mice. Health Phys. 2020, 119, 633–646. [Google Scholar] [CrossRef]
- Patterson, A.M.; Wu, T.; Chua, H.L.; Sampson, C.H.; Fisher, A.; Singh, P.; Guise, T.A.; Feng, H.; Muldoon, J.; Wright, L. Optimizing and profiling prostaglandin E2 as a medical countermeasure for the hematopoietic acute radiation syndrome. Radiat. Res. 2021, 195, 115–127. [Google Scholar] [CrossRef]
- Wang, Q.; Lee, Y.; Pujol-Canadell, M.; Perrier, J.R.; Smilenov, L.; Harken, A.; Garty, G.; Brenner, D.J.; Ponnaiya, B.; Turner, H.C. Cytogenetic Damage of Human Lymphocytes in Humanized Mice Exposed to Neutrons and X Rays 24 h After Exposure. Cytogenet. Genome Res. 2021, 161, 352–361. [Google Scholar] [CrossRef]
- Blakely, W.F.; Ossetrova, N.I.; Whitnall, M.H.; Sandgren, D.J.; Krivokrysenko, V.I.; Shakhov, A.; Feinstein, E. Multiple parameter radiation injury assessment using a nonhuman primate radiation model—Biodosimetry applications. Health Phys. 2010, 98, 153–159. [Google Scholar] [CrossRef]
- Grace, M.B.; Muderhwa, J.M.; Salter, C.A.; Blakely, W.F. Use of a centrifuge-based automated blood cell counter for radiation dose assessment. Mil. Med. 2006, 171, 908–912. [Google Scholar] [CrossRef]
- Prasanna, P.G.; Blakely, W.F.; Bertho, J.-M.; Chute, J.P.; Cohen, E.P.; Goans, R.E.; Grace, M.B.; Lillis-Hearne, P.K.; Lloyd, D.C.; Lutgens, L.C. Synopsis of partial-body radiation diagnostic biomarkers and medical management of radiation injury workshop. Radiat. Res. 2010, 173, 245–253. [Google Scholar] [CrossRef]
- Bhayana, S.; Song, F.; Jacob, J.; Fadda, P.; Denko, N.C.; Xu-Welliver, M.; Chakravarti, A.; Jacob, N.K. Urinary miRNAs as biomarkers for noninvasive evaluation of radiation-induced renal tubular injury. Radiat. Res. 2017, 188, 706–715. [Google Scholar] [CrossRef]
- Chakraborty, N.; Gautam, A.; Holmes-Hampton, G.P.; Kumar, V.P.; Biswas, S.; Kumar, R.; Hamad, D.; Dimitrov, G.; Olabisi, A.O.; Hammamieh, R. MicroRNA and metabolite signatures linked to early consequences of lethal radiation. Sci. Rep. 2020, 10, 5424. [Google Scholar] [CrossRef] [PubMed]
- Chopra, S.; Moroni, M.; Sanjak, J.; MacMillan, L.; Hritzo, B.; Martello, S.; Bylicky, M.; May, J.; Coleman, C.N.; Aryankalayil, M.J. Whole blood gene expression within days after total-body irradiation predicts long term survival in Gottingen minipigs. Sci. Rep. 2021, 11, 15873. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Wang, P.; Farese, A.M.; MacVittie, T.J.; Kane, M.A. Metabolomics of multiorgan radiation injury in non-human primate model reveals system-wide metabolic perturbations. Health Phys. 2021, 121, 395–405. [Google Scholar] [CrossRef]
- Laiakis, E.C.; Canadell, M.P.; Grilj, V.; Harken, A.D.; Garty, G.Y.; Brenner, D.J.; Smilenov, L.; Fornace, A.J. Small molecule responses to sequential irradiation with neutrons and photons for biodosimetry applications: An initial assessment. Radiat. Res. 2021, 196, 468–477. [Google Scholar] [CrossRef]
- Liu, K.; Singer, E.; Cohn, W.; Micewicz, E.D.; McBride, W.H.; Whitelegge, J.P.; Loo, J.A. Time-Dependent Measurement of Nrf2-Regulated Antioxidant Response to Ionizing Radiation toward Identifying Potential Protein Biomarkers for Acute Radiation Injury. PROTEOMICS Clin. Appl. 2019, 13, 1900035. [Google Scholar] [CrossRef] [PubMed]
- Ossetrova, N.I.; Stanton, P.; Krasnopolsky, K.; Ismail, M.; Doreswamy, A.; Hieber, K.P. Comparison of biodosimetry biomarkers for radiation dose and injury assessment after mixed-field (neutron and gamma) and pure gamma radiation in the mouse total-body irradiation model. Health Phys. 2018, 115, 743–759. [Google Scholar] [CrossRef]
- Bujold, K.; Hauer-Jensen, M.; Donini, O.; Rumage, A.; Hartman, D.; Hendrickson, H.; Stamatopoulos, J.; Naraghi, H.; Pouliot, M.; Ascah, A. Citrulline as a biomarker for gastrointestinal-acute radiation syndrome: Species differences and experimental condition effects. Radiat. Res. 2016, 186, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.W.; Bennett, A.; Carter, C.L.; Tudor, G.; Hankey, K.G.; Farese, A.M.; Booth, C.; MacVittie, T.J.; Kane, M.A. Citrulline as a biomarker in the non-human primate total-and partial-body irradiation models: Correlation of circulating citrulline to acute and prolonged gastrointestinal injury. Health Phys. 2015, 109, 440–451. [Google Scholar] [CrossRef]
- Jones, J.W.; Tudor, G.; Li, F.; Tong, Y.; Katz, B.; Farese, A.M.; MacVittie, T.J.; Booth, C.; Kane, M.A. Citrulline as a biomarker in the murine total-body irradiation model: Correlation of circulating and tissue citrulline to small intestine epithelial histopathology. Health Phys. 2015, 109, 452–465. [Google Scholar] [CrossRef]
- Ainsbury, E.A.; Moquet, J.; Sun, M.; Barnard, S.; Ellender, M.; Lloyd, D. The future of biological dosimetry in mass casualty radiation emergency response, personalized radiation risk estimation and space radiation protection. Int. J. Radiat. Biol. 2022, 98, 421–427. [Google Scholar] [CrossRef]
- Kugathasan, T.; Mothersill, C. Radiobiological and social considerations following a radiological terrorist attack; mechanisms, detection and mitigation: Review of new research developments. Int. J. Radiat. Biol. 2022, 98, 855–864. [Google Scholar] [CrossRef]
- Liu, X.; Shao, C.; Fu, J. Promising Biomarkers of Radiation-Induced Lung Injury: A Review. Biomedicines 2021, 9, 1181. [Google Scholar] [CrossRef]
- Medhora, M.; Haworth, S.; Liu, Y.; Narayanan, J.; Gao, F.; Zhao, M.; Audi, S.; Jacobs, E.R.; Fish, B.L.; Clough, A.V. Biomarkers for radiation pneumonitis using noninvasive molecular imaging. J. Nucl. Med. 2016, 57, 1296–1301. [Google Scholar] [CrossRef]
- Medhora, M.; Gao, F.; Fish, B.L.; Jacobs, E.R.; Moulder, J.E.; Szabo, A. Dose-modifying factor for captopril for mitigation of radiation injury to normal lung. J. Radiat. Res. 2012, 53, 633–640. [Google Scholar] [CrossRef]
- Gao, F.; Liu, P.; Narayanan, J.; Yang, M.; Fish, B.L.; Liu, Y.; Liang, M.; Jacobs, E.R.; Medhora, M. Changes in miRNA in the lung and whole blood after whole thorax irradiation in rats. Sci. Rep. 2017, 7, 44132. [Google Scholar] [CrossRef]
- Kma, L.; Gao, F.; Fish, B.L.; Moulder, J.E.; Jacobs, E.R.; Medhora, M. Angiotensin converting enzyme inhibitors mitigate collagen synthesis induced by a single dose of radiation to the whole thorax. J. Radiat. Res. 2012, 53, 10–17. [Google Scholar] [CrossRef]
- Gao, F.; Fish, B.L.; Moulder, J.E.; Jacobs, E.R.; Medhora, M. Enalapril mitigates radiation-induced pneumonitis and pulmonary fibrosis if started 35 days after whole-thorax irradiation. Radiat. Res. 2013, 180, 546–552. [Google Scholar] [CrossRef]
- Unger, K.; Li, Y.; Yeh, C.; Barac, A.; Srichai, M.B.; Ballew, E.A.; Girgis, M.; Jayatilake, M.; Sridharan, V.; Boerma, M. Plasma metabolite biomarkers predictive of radiation induced cardiotoxicity. Radiother. Oncol. 2020, 152, 133–145. [Google Scholar] [CrossRef]
- Fleckenstein, K.; Zgonjanin, L.; Chen, L.; Rabbani, Z.; Jackson, I.L.; Thrasher, B.; Kirkpatrick, J.; Foster, W.M.; Vujaskovic, Z. Temporal onset of hypoxia and oxidative stress after pulmonary irradiation. Int. J. Radiat. Oncol. Biol. Phys. 2007, 68, 196–204. [Google Scholar] [CrossRef]
- Ghosh, S.; Wu, Q.; Mäder, M.; Fish, B.; Moulder, J.E.; Jacobs, E.R.; Medhora, M.; Molthen, R.C. Vascular injury after whole thoracic x-ray irradiation in the rat. Int. J. Radiat. Oncol. Biol. Phys. 2009, 74, 192–199. [Google Scholar] [CrossRef]
- Mehrvar, S.; Mostaghimi, S.; Camara, A.K.; Foomani, F.H.; Narayanan, J.; Fish, B.; Medhora, M.; Ranji, M. Three-dimensional vascular and metabolic imaging using inverted autofluorescence. J. Biomed. Opt. 2021, 26, 076002. [Google Scholar] [CrossRef]
- Jagtap, J.; Audi, S.; Razeghi-Kondelaji, M.H.; Fish, B.L.; Hansen, C.; Narayan, J.; Gao, F.; Sharma, G.; Parchur, A.K.; Banerjee, A. A rapid dynamic in vivo near-infrared fluorescence imaging assay to track lung vascular permeability after acute radiation injury. Am. J. Physiol. Lung Cell. Mol. Physiol. 2021, 320, L436–L450. [Google Scholar] [CrossRef]
- Kamolz, L.-P.; Andel, H.; Haslik, W.; Donner, A.; Winter, W.; Meissl, G.; Frey, M. Indocyanine green video angiographies help to identify burns requiring operation. Burns 2003, 29, 785–791. [Google Scholar] [CrossRef]
- Seo, J.; An, Y.; Lee, J.; Ku, T.; Kang, Y.; Ahn, C.W.; Choi, C. Principal component analysis of dynamic fluorescence images for diagnosis of diabetic vasculopathy. J. Biomed. Opt. 2016, 21, 046003. [Google Scholar] [CrossRef]
- Speers, C.; Murthy, V.L.; Walker, E.M.; Glide-Hurst, C.K.; Marsh, R.; Tang, M.; Morris, E.L.; Schipper, M.J.; Weinberg, R.L.; Gits, H.C. Cardiac Magnetic Resonance Imaging and Blood Biomarkers for Evaluation of Radiation-Induced Cardiotoxicity in Patients With Breast Cancer: Results of a Phase 2 Clinical Trial. Int. J. Radiat. Oncol. Biol. Phys. 2022, 112, 417–425. [Google Scholar] [CrossRef]
- Fish, B.L.; MacVittie, T.J.; Szabo, A.; Moulder, J.E.; Medhora, M. WAG/RijCmcr rat models for injuries to multiple organs by single high dose ionizing radiation: Similarities to nonhuman primates (NHP). Int. J. Radiat. Biol. 2020, 96, 81–92. [Google Scholar] [CrossRef]
- MacVittie, T.J.; Farese, A.M.; Parker, G.A.; Bennett, A.W.; Jackson III, W.E. Acute radiation-induced lung injury in the non-human primate: A review and comparison of mortality and co-morbidities using models of partial-body irradiation with marginal bone marrow sparing and whole thorax lung irradiation. Health Phys. 2020, 119, 559–587. [Google Scholar] [CrossRef]
- Ghandhi, S.A.; Smilenov, L.; Shuryak, I.; Pujol-Canadell, M.; Amundson, S.A. Discordant gene responses to radiation in humans and mice and the role of hematopoietically humanized mice in the search for radiation biomarkers. Sci. Rep. 2019, 9, 19434. [Google Scholar] [CrossRef]
- Garcia-Barros, M.; Paris, F.; Cordon-Cardo, C.; Lyden, D.; Rafii, S.; Haimovitz-Friedman, A.; Fuks, Z.; Kolesnick, R. Tumor response to radiotherapy regulated by endothelial cell apoptosis. Science 2003, 300, 1155–1159. [Google Scholar] [CrossRef]
- Holcik, M.; Yeh, C.; Korneluk, R.G.; Chow, T. Translational upregulation of X-linked inhibitor of apoptosis (XIAP) increases resistance to radiation induced cell death. Oncogene 2000, 19, 4174–4177. [Google Scholar] [CrossRef] [PubMed]
- Verheij, M.; Bartelink, H. Radiation-induced apoptosis. Cell Tissue Res. 2000, 301, 133–142. [Google Scholar] [CrossRef]
- Johnson, S.E.; Li, Z.; Liu, Y.; Moulder, J.E.; Zhao, M. Whole-body imaging of high-dose ionizing irradiation-induced tissue injuries using 99mTc-duramycin. J. Nucl. Med. 2013, 54, 1397–1403. [Google Scholar] [CrossRef]
- Audi, S.H.; Jacobs, E.R.; Zhao, M.; Roerig, D.L.; Haworth, S.T.; Clough, A.V. In vivo detection of hyperoxia-induced pulmonary endothelial cell death using 99mTc-duramycin. Nucl. Med. Biol. 2015, 42, 46–52. [Google Scholar] [CrossRef]
- Clough, A.V.; Audi, S.H.; Haworth, S.T.; Roerig, D.L. Differential lung uptake of 99mTc-hexamethylpropyleneamine oxime and 99mTc-duramycin in the chronic hyperoxia rat model. J. Nucl. Med. 2012, 53, 1984–1991. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Fish, B.L.; Moulder, J.E.; Medhora, M.; Baker, J.E.; Mader, M.; Cohen, E.P. Safety and blood sample volume and quality of a refined retro-orbital bleeding technique in rats using a lateral approach. Lab Anim. 2014, 43, 63–66. [Google Scholar] [CrossRef] [PubMed]
- Gao, F.; Dong, W.; Liu, P.Y.; Narayanan, J.; Fish, B.L.; Jacobs, E.R.; Medhora, M. Molecular Changes in miRNA in Irradiated Rat Kidneys: Role of miR-34a and its Vascular Targets in the Notch Pathway. Radiat. Res. 2021, 196, 611–622. [Google Scholar] [CrossRef]

| Number | Gy | Notes |
|---|---|---|
| 6 | 0 | Study set |
| 7 | 10 | Study set |
| 26 | 13 | Study set |
| 11 | 0 | Biomarker identification set |
| 11 | 15 | Biomarker identification set |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Medhora, M.; Gao, F.; Gasperetti, T.; Narayanan, J.; Himburg, H.; Jacobs, E.R.; Clough, A.V.; Fish, B.L.; Szabo, A. Biomarkers to Predict Lethal Radiation Injury to the Rat Lung. Int. J. Mol. Sci. 2023, 24, 5627. https://doi.org/10.3390/ijms24065627
Medhora M, Gao F, Gasperetti T, Narayanan J, Himburg H, Jacobs ER, Clough AV, Fish BL, Szabo A. Biomarkers to Predict Lethal Radiation Injury to the Rat Lung. International Journal of Molecular Sciences. 2023; 24(6):5627. https://doi.org/10.3390/ijms24065627
Chicago/Turabian StyleMedhora, Meetha, Feng Gao, Tracy Gasperetti, Jayashree Narayanan, Heather Himburg, Elizabeth R. Jacobs, Anne V. Clough, Brian L. Fish, and Aniko Szabo. 2023. "Biomarkers to Predict Lethal Radiation Injury to the Rat Lung" International Journal of Molecular Sciences 24, no. 6: 5627. https://doi.org/10.3390/ijms24065627
APA StyleMedhora, M., Gao, F., Gasperetti, T., Narayanan, J., Himburg, H., Jacobs, E. R., Clough, A. V., Fish, B. L., & Szabo, A. (2023). Biomarkers to Predict Lethal Radiation Injury to the Rat Lung. International Journal of Molecular Sciences, 24(6), 5627. https://doi.org/10.3390/ijms24065627





