Role of Klotho and AGE/RAGE-Wnt/β-Catenin Signalling Pathway on the Development of Cardiac and Renal Fibrosis in Diabetes
Abstract
1. Introduction
2. Results
2.1. General, Glycaemic, Renal, and Cardiac Biochemical Parameters
2.2. Diabetes-Related Kidney Changes towards Fibrosis
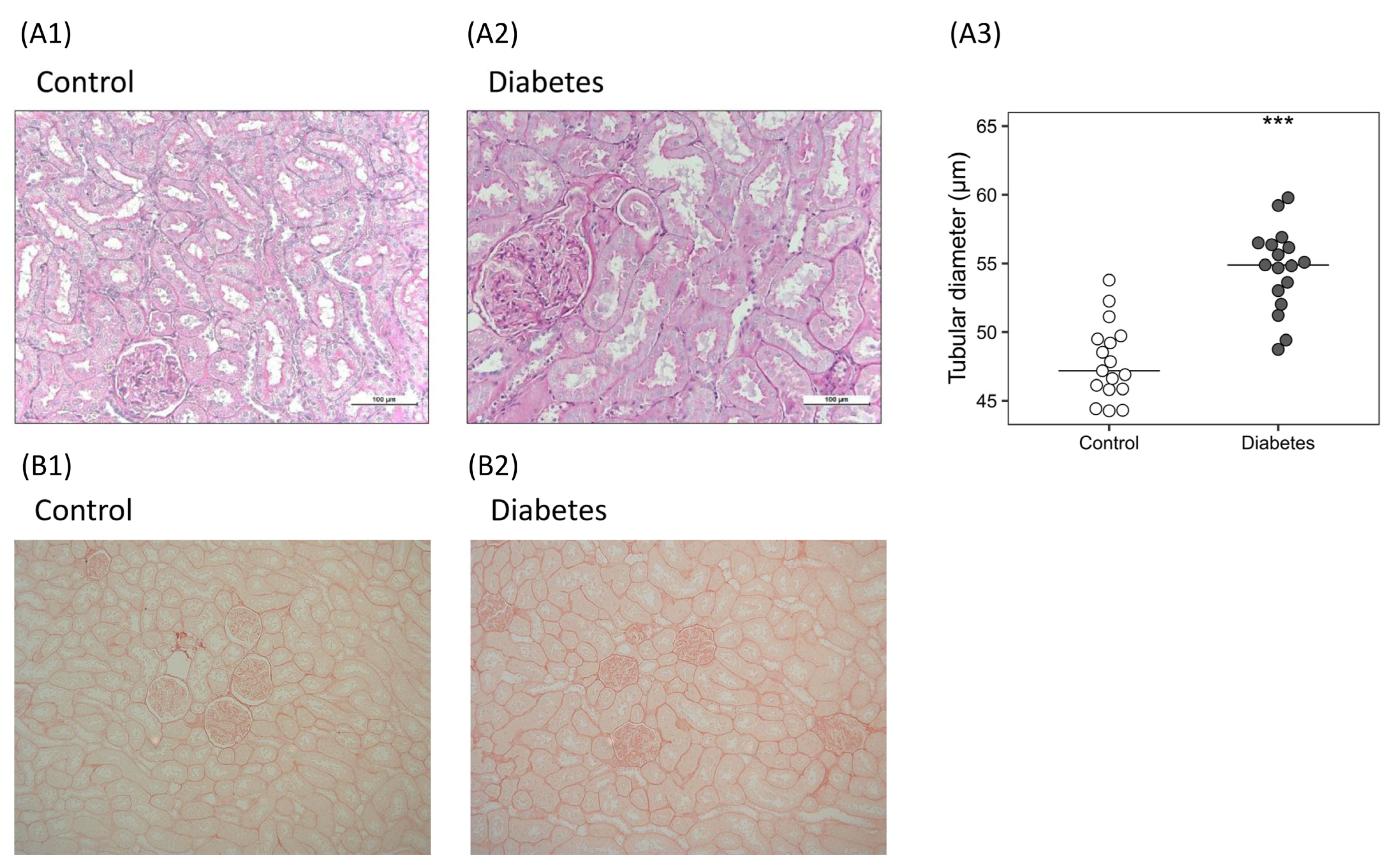
2.2.1. Histological Analysis
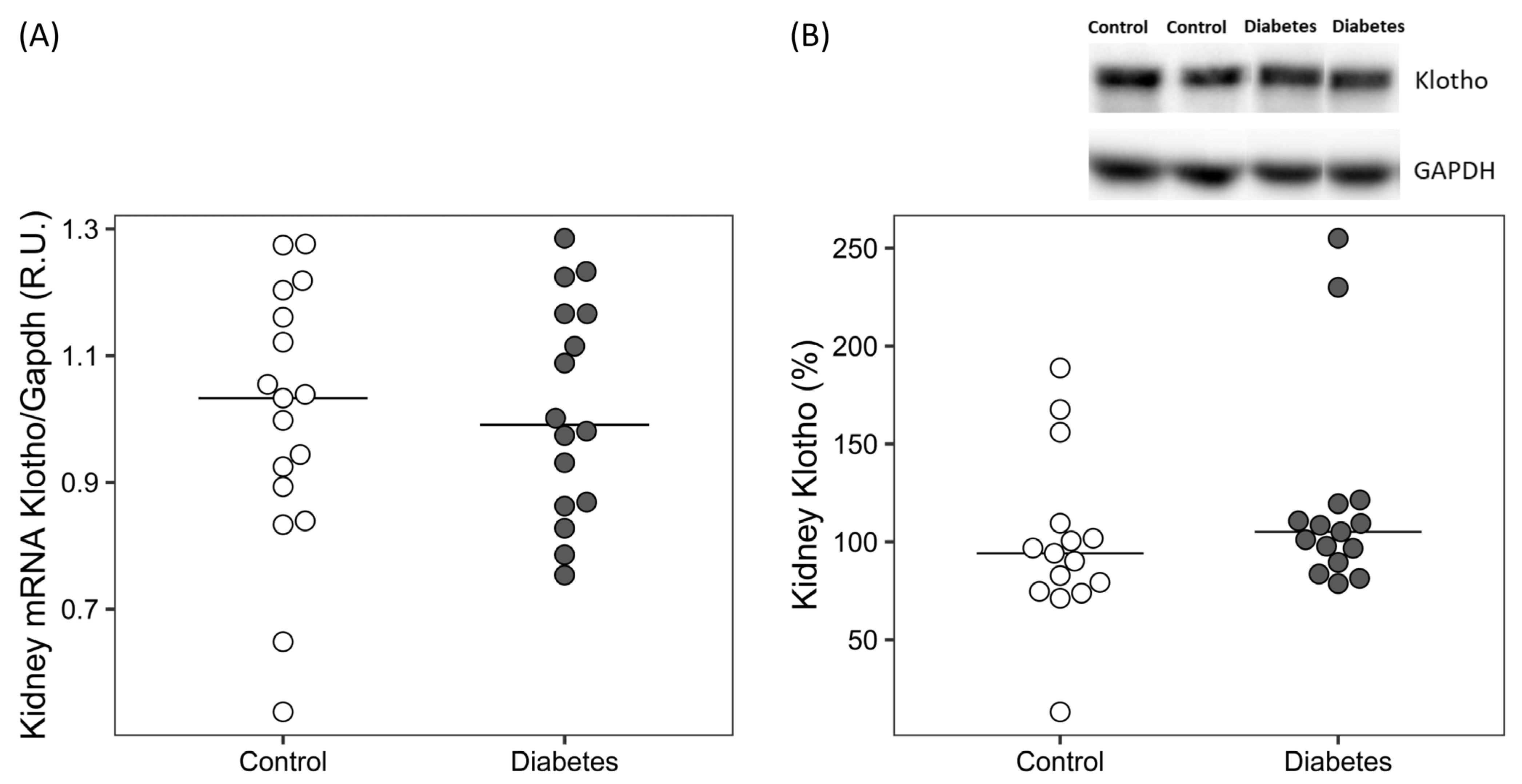
2.2.2. Molecular Markers of Fibrosis, Wnt/β-Catenin, RAGE and ADAM10
2.3. Diabetes-Related Cardiac Changes towards Fibrosis
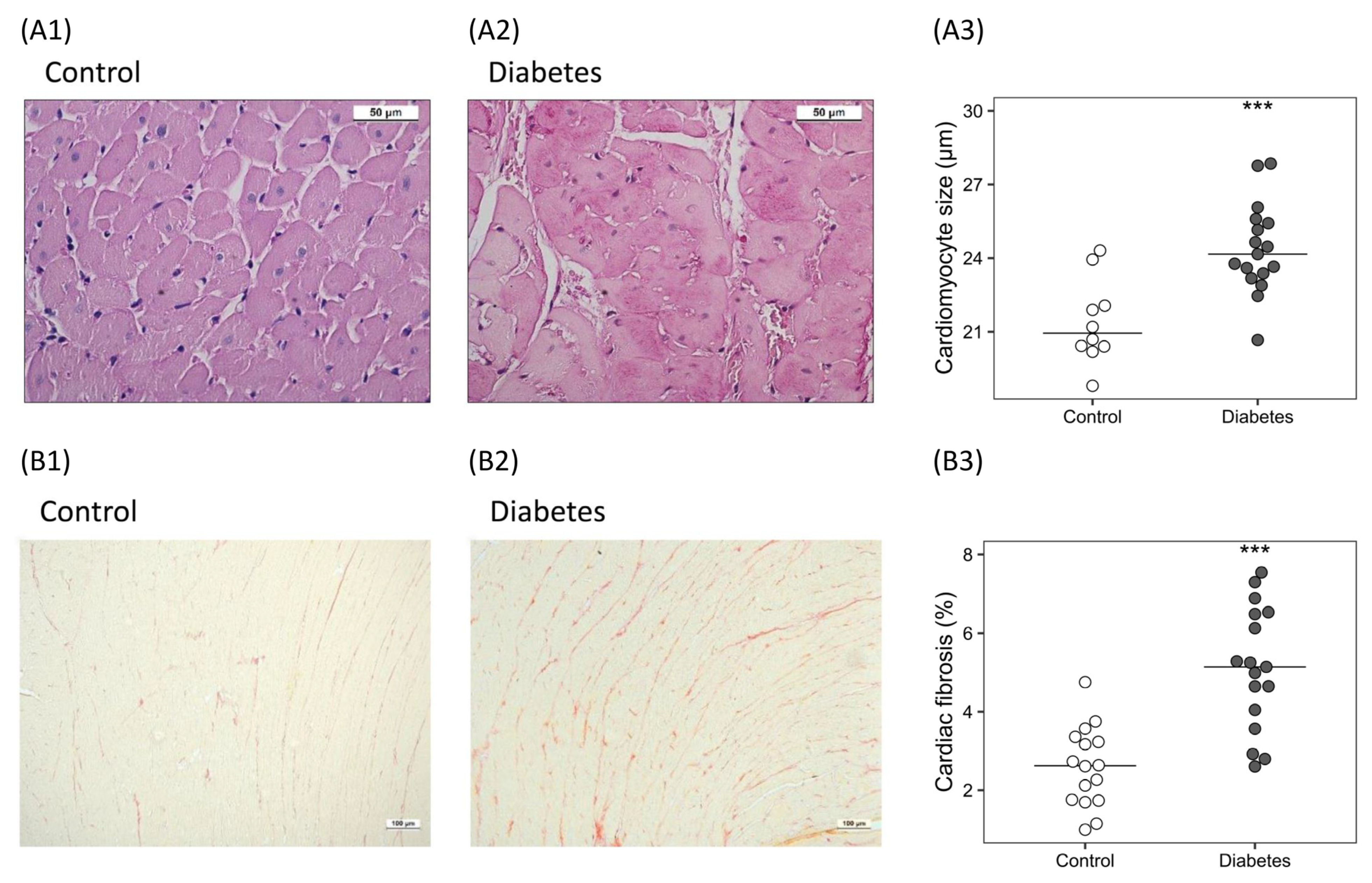
2.3.1. Histological Analysis
2.3.2. Molecular Markers of Fibrosis, Wnt/β-Catenin RAGE and ADAM10
3. Discussion
4. Materials and Methods
4.1. Experimental Model
4.2. Biochemical Analyses
4.3. Histological Analyses
4.4. RNA Extraction and Quantitative Real-Time PCR
4.5. Immunoblotting
4.6. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- International Diabetes Federation IDF Diabetes Atlas. Available online: https://www.diabetesatlas.org/en/ (accessed on 19 August 2022).
- Birkeland, K.I.; Bodegard, J.; Eriksson, J.W.; Norhammar, A.; Haller, H.; Linssen, G.C.M.; Banerjee, A.; Thuresson, M.; Okami, S.; Garal-Pantaler, E.; et al. Heart failure and chronic kidney disease manifestation and mortality risk associations in type 2 diabetes: A large multinational cohort study. Diabetes Obes. Metab. 2020, 22, 1607–1618. [Google Scholar] [CrossRef] [PubMed]
- Kristofi, R.; Bodegard, J.; Norhammar, A.; Thuresson, M.; Nathanson, D.; Nystrom, T.; Birkeland, K.I.; Eriksson, J.W. Cardiovascular and Renal Disease Burden in Type 1 Compared With Type 2 Diabetes: A Two-Country Nationwide Observational Study. Diabetes Care 2021, 44, 1211–1218. [Google Scholar] [CrossRef] [PubMed]
- Salvatore, T.; Pafundi, P.C.; Galiero, R.; Albanese, G.; Di Martino, A.; Caturano, A.; Vetrano, E.; Rinaldi, L.; Sasso, F.C. The Diabetic Cardiomyopathy: The Contributing Pathophysiological Mechanisms. Front. Med. 2021, 8, 695792. [Google Scholar] [CrossRef] [PubMed]
- Zeng, L.F.; Xiao, Y.; Sun, L. A Glimpse of the Mechanisms Related to Renal Fibrosis in Diabetic Nephropathy. Adv. Exp. Med. Biol. 2019, 1165, 49–79. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Randive, R.; Stewart, J.A. Molecular mechanisms of AGE/RAGE-mediated fibrosis in the diabetic heart. World J. Diabetes 2014, 5, 860–867. [Google Scholar] [CrossRef]
- Matsui, T.; Higashimoto, Y.; Nishino, Y.; Nakamura, N.; Fukami, K.; Yamagishi, S.I. RAGE-Aptamer Blocks the Development and Progression of Experimental Diabetic Nephropathy. Diabetes 2017, 66, 1683–1695. [Google Scholar] [CrossRef]
- Yang, R.; Jia, Q.; Ma, S.F.; Wang, Y.; Mehmood, S.; Chen, Y. Exogenous H2S mitigates myocardial fibrosis in diabetic rats through suppression of the canonical Wnt pathway. Int. J. Mol. Med. 2019, 44, 549–558. [Google Scholar] [CrossRef]
- Rooney, B.; O′Donovan, H.; Gaffney, A.; Browne, M.; Faherty, N.; Curran, S.P.; Sadlier, D.; Godson, C.; Brazil, D.P.; Crean, J. CTGF/CCN2 activates canonical Wnt signalling in mesangial cells through LRP6: Implications for the pathogenesis of diabetic nephropathy. FEBS Lett. 2011, 585, 531–538. [Google Scholar] [CrossRef]
- Hu, H.H.; Cao, G.; Wu, X.Q.; Vaziri, N.D.; Zhao, Y.Y. Wnt signaling pathway in aging-related tissue fibrosis and therapies. Ageing Res. Rev. 2020, 60, 101063. [Google Scholar] [CrossRef]
- Lindberg, K.; Amin, R.; Moe, O.W.; Hu, M.C.; Erben, R.G.; Ostman Wernerson, A.; Lanske, B.; Olauson, H.; Larsson, T.E. The kidney is the principal organ mediating klotho effects. J. Am. Soc. Nephrol. JASN 2014, 25, 2169–2175. [Google Scholar] [CrossRef]
- Kuro, O.M. The Klotho proteins in health and disease. Nat. Rev. Nephrol. 2019, 15, 27–44. [Google Scholar] [CrossRef]
- Hu, M.C.; Kuro-o, M.; Moe, O.W. Renal and extrarenal actions of Klotho. Semin. Nephrol. 2013, 33, 118–129. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.C.; Shi, M.; Gillings, N.; Flores, B.; Takahashi, M.; Kuro, O.M.; Moe, O.W. Recombinant alpha-Klotho may be prophylactic and therapeutic for acute to chronic kidney disease progression and uremic cardiomyopathy. Kidney Int. 2017, 91, 1104–1114. [Google Scholar] [CrossRef]
- Liu, H.; Fergusson, M.M.; Castilho, R.M.; Liu, J.; Cao, L.; Chen, J.; Malide, D.; Rovira, I.I.; Schimel, D.; Kuo, C.J.; et al. Augmented Wnt signaling in a mammalian model of accelerated aging. Science 2007, 317, 803–806. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Wang, C.; Hong, X.; Miao, J.; Liao, Y.; Hou, F.F.; Zhou, L.; Liu, Y. Wnt/beta-catenin signaling mediates both heart and kidney injury in type 2 cardiorenal syndrome. Kidney Int. 2019, 95, 815–829. [Google Scholar] [CrossRef] [PubMed]
- Donate-Correa, J.; Martin-Nunez, E.; Mora-Fernandez, C.; Muros-de-Fuentes, M.; Perez-Delgado, N.; Navarro-Gonzalez, J.F. Klotho in cardiovascular disease: Current and future perspectives. World J. Biol. Chem. 2015, 6, 351–357. [Google Scholar] [CrossRef]
- Bi, X.; Yang, K.; Zhang, B.; Zhao, J. The Protective Role of Klotho in CKD-Associated Cardiovascular Disease. Kidney Dis. 2020, 6, 395–406. [Google Scholar] [CrossRef]
- Oh, H.J.; Nam, B.Y.; Wu, M.; Kim, S.; Park, J.; Kang, S.; Park, J.T.; Yoo, T.H.; Kang, S.W.; Han, S.H. Klotho plays a protective role against glomerular hypertrophy in a cell cycle-dependent manner in diabetic nephropathy. Am. J. Physiol. Ren. Physiol. 2018, 315, F791–F805. [Google Scholar] [CrossRef]
- Wu, C.; Wang, Q.; Lv, C.; Qin, N.; Lei, S.; Yuan, Q.; Wang, G. The changes of serum sKlotho and NGAL levels and their correlation in type 2 diabetes mellitus patients with different stages of urinary albumin. Diabetes Res. Clin. Pr. 2014, 106, 343–350. [Google Scholar] [CrossRef]
- Inci, A.; Sari, F.; Coban, M.; Olmaz, R.; Dolu, S.; Sarikaya, M.; Yilmaz, N. Soluble Klotho and fibroblast growth factor 23 levels in diabetic nephropathy with different stages of albuminuria. J. Investig. Med. 2016, 64, 1128–1133. [Google Scholar] [CrossRef]
- Kacso, I.M.; Bondor, C.I.; Kacso, G. Soluble serum Klotho in diabetic nephropathy: Relationship to VEGF-A. Clin. Biochem. 2012, 45, 1415–1420. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.X.; Li, S.S.; Sha, M.Y.; Kong, J.W.; Ye, J.M.; Liu, Q.F. The controversy of klotho as a potential biomarker in chronic kidney disease. Front. Pharmacol. 2022, 13, 931746. [Google Scholar] [CrossRef] [PubMed]
- Typiak, M.; Kulesza, T.; Rachubik, P.; Rogacka, D.; Audzeyenka, I.; Angielski, S.; Saleem, M.A.; Piwkowska, A. Role of Klotho in Hyperglycemia: Its Levels and Effects on Fibroblast Growth Factor Receptors, Glycolysis, and Glomerular Filtration. Int. J. Mol. Sci. 2021, 22, 7867. [Google Scholar] [CrossRef] [PubMed]
- Tervaert, T.W.; Mooyaart, A.L.; Amann, K.; Cohen, A.H.; Cook, H.T.; Drachenberg, C.B.; Ferrario, F.; Fogo, A.B.; Haas, M.; de Heer, E.; et al. Pathologic classification of diabetic nephropathy. J. Am. Soc. Nephrol. JASN 2010, 21, 556–563. [Google Scholar] [CrossRef] [PubMed]
- Sakan, H.; Nakatani, K.; Asai, O.; Imura, A.; Tanaka, T.; Yoshimoto, S.; Iwamoto, N.; Kurumatani, N.; Iwano, M.; Nabeshima, Y.; et al. Reduced renal alpha-Klotho expression in CKD patients and its effect on renal phosphate handling and vitamin D metabolism. PLoS ONE 2014, 9, e86301. [Google Scholar] [CrossRef]
- Navarro-Gonzalez, J.F.; Sanchez-Nino, M.D.; Donate-Correa, J.; Martin-Nunez, E.; Ferri, C.; Perez-Delgado, N.; Gorriz, J.L.; Martinez-Castelao, A.; Ortiz, A.; Mora-Fernandez, C. Effects of Pentoxifylline on Soluble Klotho Concentrations and Renal Tubular Cell Expression in Diabetic Kidney Disease. Diabetes Care 2018, 41, 1817–1820. [Google Scholar] [CrossRef]
- Hu, M.C.; Shi, M.; Zhang, J.; Addo, T.; Cho, H.J.; Barker, S.L.; Ravikumar, P.; Gillings, N.; Bian, A.; Sidhu, S.S.; et al. Renal Production, Uptake, and Handling of Circulating alphaKlotho. J. Am. Soc. Nephrol. JASN 2016, 27, 79–90. [Google Scholar] [CrossRef]
- Qian, Y.; Guo, X.; Che, L.; Guan, X.; Wu, B.; Lu, R.; Zhu, M.; Pang, H.; Yan, Y.; Ni, Z.; et al. Klotho Reduces Necroptosis by Targeting Oxidative Stress Involved in Renal Ischemic-Reperfusion Injury. Cell. Physiol. Biochem. 2018, 45, 2268–2282. [Google Scholar] [CrossRef]
- Zubkiewicz-Kucharska, A.; Wikiera, B.; Noczynska, A. Soluble Klotho Is Decreased in Children With Type 1 Diabetes and Correlated With Metabolic Control. Front. Endocrinol. (Lausanne) 2021, 12, 709564. [Google Scholar] [CrossRef]
- Mengstie, M.A.; Chekol Abebe, E.; Behaile Teklemariam, A.; Tilahun Mulu, A.; Agidew, M.M.; Teshome Azezew, M.; Zewde, E.A.; Agegnehu Teshome, A. Endogenous advanced glycation end products in the pathogenesis of chronic diabetic complications. Front. Mol. Biosci. 2022, 9, 1002710. [Google Scholar] [CrossRef]
- Erusalimsky, J.D. The use of the soluble receptor for advanced glycation-end products (sRAGE) as a potential biomarker of disease risk and adverse outcomes. Redox Biol. 2021, 42, 101958. [Google Scholar] [CrossRef]
- Prasad, K. Is there any evidence that AGE/sRAGE is a universal biomarker/risk marker for diseases? Mol. Cell. Biochem. 2019, 451, 139–144. [Google Scholar] [CrossRef]
- Tan, K.C.; Shiu, S.W.; Chow, W.S.; Leng, L.; Bucala, R.; Betteridge, D.J. Association between serum levels of soluble receptor for advanced glycation end products and circulating advanced glycation end products in type 2 diabetes. Diabetologia 2006, 49, 2756–2762. [Google Scholar] [CrossRef]
- Tsoporis, J.N.; Hatziagelaki, E.; Gupta, S.; Izhar, S.; Salpeas, V.; Tsiavou, A.; Rigopoulos, A.G.; Triantafyllis, A.S.; Marshall, J.C.; Parker, T.G.; et al. Circulating Ligands of the Receptor for Advanced Glycation End Products and the Soluble Form of the Receptor Modulate Cardiovascular Cell Apoptosis in Diabetes. Molecules 2020, 25, 5235. [Google Scholar] [CrossRef] [PubMed]
- Katakami, N.; Matsuhisa, M.; Kaneto, H.; Matsuoka, T.A.; Sakamoto, K.; Nakatani, Y.; Ohtoshi, K.; Hayaishi-Okano, R.; Kosugi, K.; Hori, M.; et al. Decreased endogenous secretory advanced glycation end product receptor in type 1 diabetic patients: Its possible association with diabetic vascular complications. Diabetes Care 2005, 28, 2716–2721. [Google Scholar] [CrossRef]
- Antonelli, A.; Di Maggio, S.; Rejman, J.; Sanvito, F.; Rossi, A.; Catucci, A.; Gorzanelli, A.; Bragonzi, A.; Bianchi, M.E.; Raucci, A. The shedding-derived soluble receptor for advanced glycation endproducts sustains inflammation during acute Pseudomonas aeruginosa lung infection. Biochim. Biophys. Acta–Gen. Subj. 2017, 1861, 354–364. [Google Scholar] [CrossRef]
- Akhmetshina, A.; Palumbo, K.; Dees, C.; Bergmann, C.; Venalis, P.; Zerr, P.; Horn, A.; Kireva, T.; Beyer, C.; Zwerina, J.; et al. Activation of canonical Wnt signalling is required for TGF-beta-mediated fibrosis. Nat. Commun. 2012, 3, 735. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Angeli, M.; Chung, K.J.; Ejimadu, C.; Rosa, A.R.; Lee, T. sFRP2 activates Wnt/beta-catenin signaling in cardiac fibroblasts: Differential roles in cell growth, energy metabolism, and extracellular matrix remodeling. Am. J. Physiol. Cell Physiol. 2016, 311, C710–C719. [Google Scholar] [CrossRef] [PubMed]
- Malachias, M.V.B.; Jhund, P.S.; Claggett, B.L.; Wijkman, M.O.; Bentley-Lewis, R.; Chaturvedi, N.; Desai, A.S.; Haffner, S.M.; Parving, H.H.; Prescott, M.F.; et al. NT-proBNP by Itself Predicts Death and Cardiovascular Events in High-Risk Patients With Type 2 Diabetes Mellitus. J. Am. Heart Assoc. 2020, 9, e017462. [Google Scholar] [CrossRef]
- Zhou, L.; Li, Y.; Zhou, D.; Tan, R.J.; Liu, Y. Loss of Klotho contributes to kidney injury by derepression of Wnt/beta-catenin signaling. J. Am. Soc. Nephrol. JASN 2013, 24, 771–785. [Google Scholar] [CrossRef]
- Chen, K.; Wang, S.; Sun, Q.W.; Zhang, B.; Ullah, M.; Sun, Z. Klotho Deficiency Causes Heart Aging via Impairing the Nrf2-GR Pathway. Circ. Res. 2021, 128, 492–507. [Google Scholar] [CrossRef]
- Memmos, E.; Sarafidis, P.; Pateinakis, P.; Tsiantoulas, A.; Faitatzidou, D.; Giamalis, P.; Vasilikos, V.; Papagianni, A. Soluble Klotho is associated with mortality and cardiovascular events in hemodialysis. BMC Nephrol. 2019, 20, 217. [Google Scholar] [CrossRef]
- Perrone, A.; Giovino, A.; Benny, J.; Martinelli, F. Advanced Glycation End Products (AGEs): Biochemistry, Signaling, Analytical Methods, and Epigenetic Effects. Oxidative Med. Cell. Longev. 2020, 2020, 3818196. [Google Scholar] [CrossRef] [PubMed]
- Barker, S.L.; Pastor, J.; Carranza, D.; Quinones, H.; Griffith, C.; Goetz, R.; Mohammadi, M.; Ye, J.; Zhang, J.; Hu, M.C.; et al. The demonstration of alphaKlotho deficiency in human chronic kidney disease with a novel synthetic antibody. Nephrol. Dial. Transplant. Off. Publ. Eur. Dial. Transpl. Assoc. Eur. Ren. Assoc. 2015, 30, 223–233. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
| Control (n = 17) | Diabetes (n = 17) | p-Value | |
|---|---|---|---|
| Body weight (g) | 541 [474, 580] | 384 [362, 405] | <0.001 |
| Urine volume (mL) | 8 [7, 13.5] | 150 [129, 172] | <0.001 |
| Glycaemic parameters | |||
| Serum Glucose (mg/dL) | 193 [182.2, 204.8] | 714.4 [682.8, 731.8] | <0.001 |
| HbA1c (%) | 5.0 [4.7, 5.0] | >13 * | <0.001 |
| Plasma AGEs (µg/mL) | 155.8 [99.9, 530.0] | 983.3 [761.6, 1115.5] | <0.001 |
| Plasma sRAGE (ng/mL) | 8.3 [7.2, 9.7] | 5.2 [4.9, 7.0] | 0.003 |
| Urinary Glucose (mg/24 h) | 4.5 [3.42, 6.46] | 15,596.7 [13,680.0, 17077.5] | <0.001 |
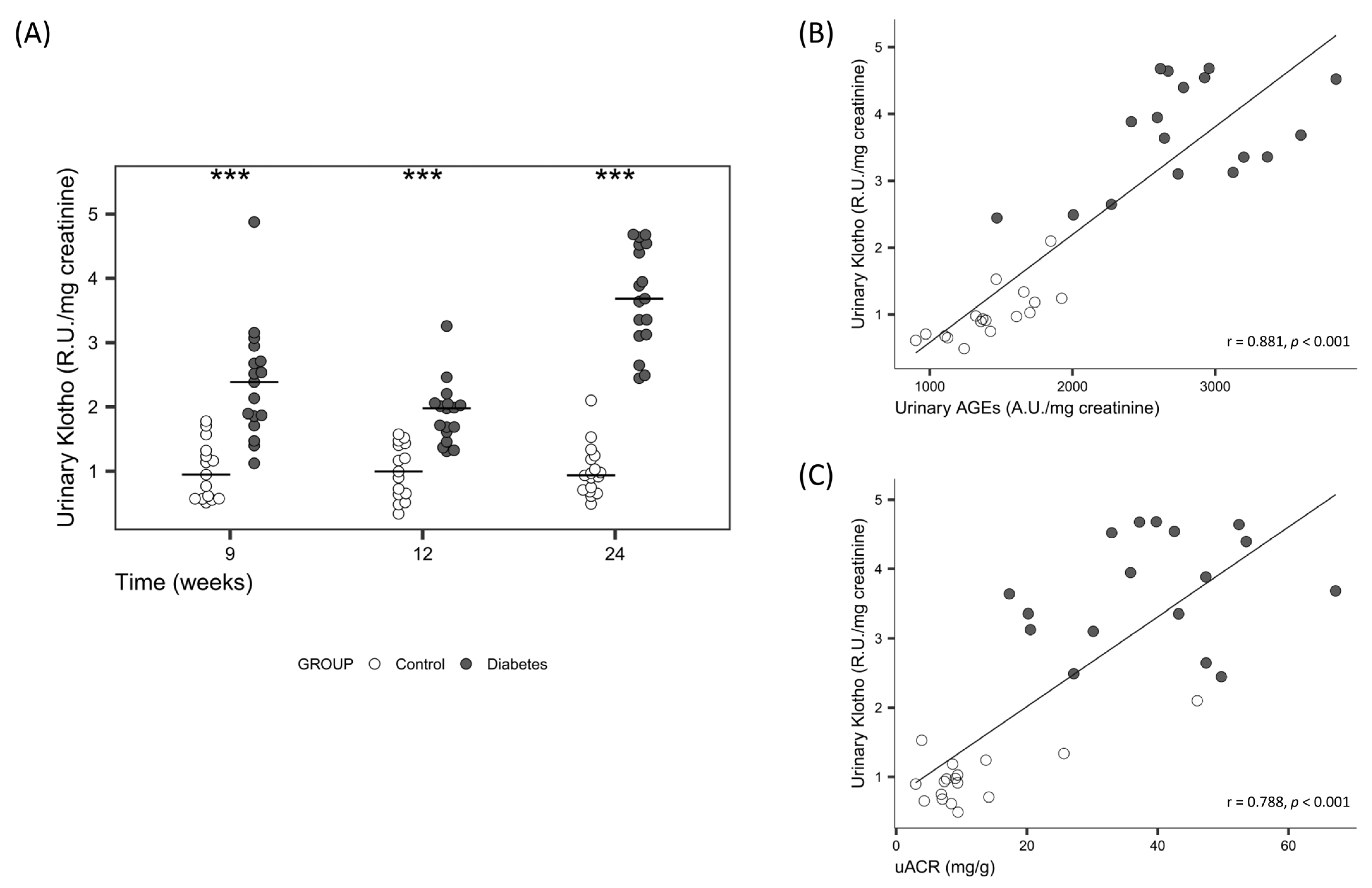
| Urinary AGEs (R.U./mg creatinine) | 1393.2 [1241.4, 1659.2] | 2739.7 [2594.3, 3125.0] | <0.001 |
| Urinary sRAGE (ng/mg creatinine) | 2.9 [2.2, 3.7] | 43.88 [36.6, 50.0] | <0.001 |
| Renal parameters | |||
| Kidney weight (mg/100 g BW) | |||
| Right | 248 [237, 257] | 427 [391, 451] | <0.001 |
| Left | 235 [218, 262] | 406 [376, 440] | <0.001 |
| Serum Proteins (g/L) | 61.5 [59.0, 64.8] | 55.5 [54.4, 57.6] | <0.001 |
| Serum Albumin (g/L) | 42 [39.8, 44.1] | 35 [33.6, 36] | <0.001 |
| Serum Creatinine (mg/dL) | 0.3 [0.2, 0.3] | 0.3 [0.3, 0.4] | 0.009 |
| Serum Urea (mg/dL) | 25.9 [23.4, 27.6] | 34.3 [31.3, 36.1] | <0.001 |
| Serum Calcium (mg/dL) | 10.1 [9.9, 10.2] | 10.1 [9.7, 10.3] | 0.654 |
| Serum sKlotho (pM) | 364.9 [328.9, 444.2] | 321.3 [292.7, 352.3] | 0.024 |
| Urinary proteins (mg/mg creatinine) | 0.7 [0.7, 0.8] | 1.0 [1.0, 1.2] | <0.001 |
| uACR (mg/g) | 8.6 [7.1, 9.4] | 39.7 [30.1, 47.4] | <0.001 |
| Creatinine clearance (mL/min) | 3.1 [2.6, 3.4] | 3.0 [2.7, 3.8] | 0.480 |
| Urinary calcium (mg/mg creatine) | 0.2 [0.22–0.28] | 1.9 [1.6–2.37] | <0.001 |
| Urinary sKlotho (R.U./mg creatinine) | 0.9 [0.7, 1.1] | 3.6 [3.1, 4.5] | <0.001 |
| Cardiac parameters | |||
| Heart weight (mg/100 g BW) | 226 [210, 233] | 242 [230, 255] | 0.006 |
| NT-proBNP (µg/mL) | 269.7 [209.6, 296.1] | 302.1 [286, 334.1] | 0.017 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martín-Carro, B.; Martín-Vírgala, J.; Fernández-Villabrille, S.; Fernández-Fernández, A.; Pérez-Basterrechea, M.; Navarro-González, J.F.; Donate-Correa, J.; Mora-Fernández, C.; Dusso, A.S.; Carrillo-López, N.; et al. Role of Klotho and AGE/RAGE-Wnt/β-Catenin Signalling Pathway on the Development of Cardiac and Renal Fibrosis in Diabetes. Int. J. Mol. Sci. 2023, 24, 5241. https://doi.org/10.3390/ijms24065241
Martín-Carro B, Martín-Vírgala J, Fernández-Villabrille S, Fernández-Fernández A, Pérez-Basterrechea M, Navarro-González JF, Donate-Correa J, Mora-Fernández C, Dusso AS, Carrillo-López N, et al. Role of Klotho and AGE/RAGE-Wnt/β-Catenin Signalling Pathway on the Development of Cardiac and Renal Fibrosis in Diabetes. International Journal of Molecular Sciences. 2023; 24(6):5241. https://doi.org/10.3390/ijms24065241
Chicago/Turabian StyleMartín-Carro, Beatriz, Julia Martín-Vírgala, Sara Fernández-Villabrille, Alejandra Fernández-Fernández, Marcos Pérez-Basterrechea, Juan F. Navarro-González, Javier Donate-Correa, Carmen Mora-Fernández, Adriana S. Dusso, Natalia Carrillo-López, and et al. 2023. "Role of Klotho and AGE/RAGE-Wnt/β-Catenin Signalling Pathway on the Development of Cardiac and Renal Fibrosis in Diabetes" International Journal of Molecular Sciences 24, no. 6: 5241. https://doi.org/10.3390/ijms24065241
APA StyleMartín-Carro, B., Martín-Vírgala, J., Fernández-Villabrille, S., Fernández-Fernández, A., Pérez-Basterrechea, M., Navarro-González, J. F., Donate-Correa, J., Mora-Fernández, C., Dusso, A. S., Carrillo-López, N., Panizo, S., Naves-Díaz, M., Fernández-Martín, J. L., Cannata-Andía, J. B., & Alonso-Montes, C. (2023). Role of Klotho and AGE/RAGE-Wnt/β-Catenin Signalling Pathway on the Development of Cardiac and Renal Fibrosis in Diabetes. International Journal of Molecular Sciences, 24(6), 5241. https://doi.org/10.3390/ijms24065241





