Aminooxy Click Modification of a Periodate-Oxidized Immunoglobulin G: A General Approach to Antibody–Drug Conjugates with Dye-Mediated Expeditious Stoichiometry Control
Abstract
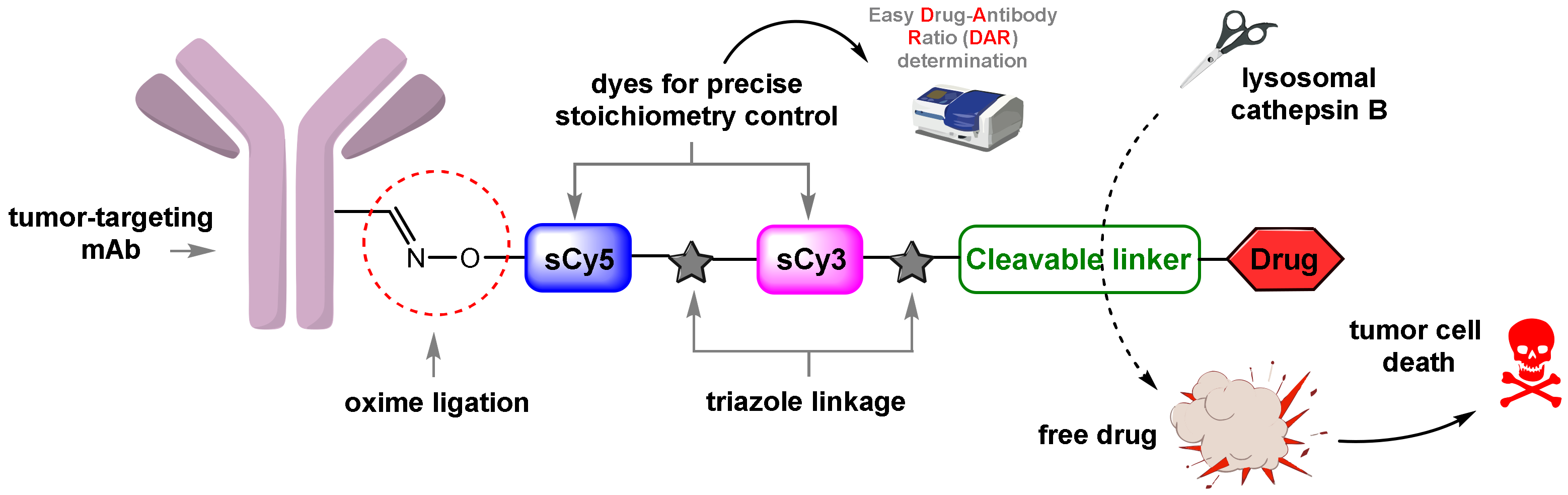
1. Introduction
2. Results and Discussion
2.1. Approaches to the Assembly of ADCs
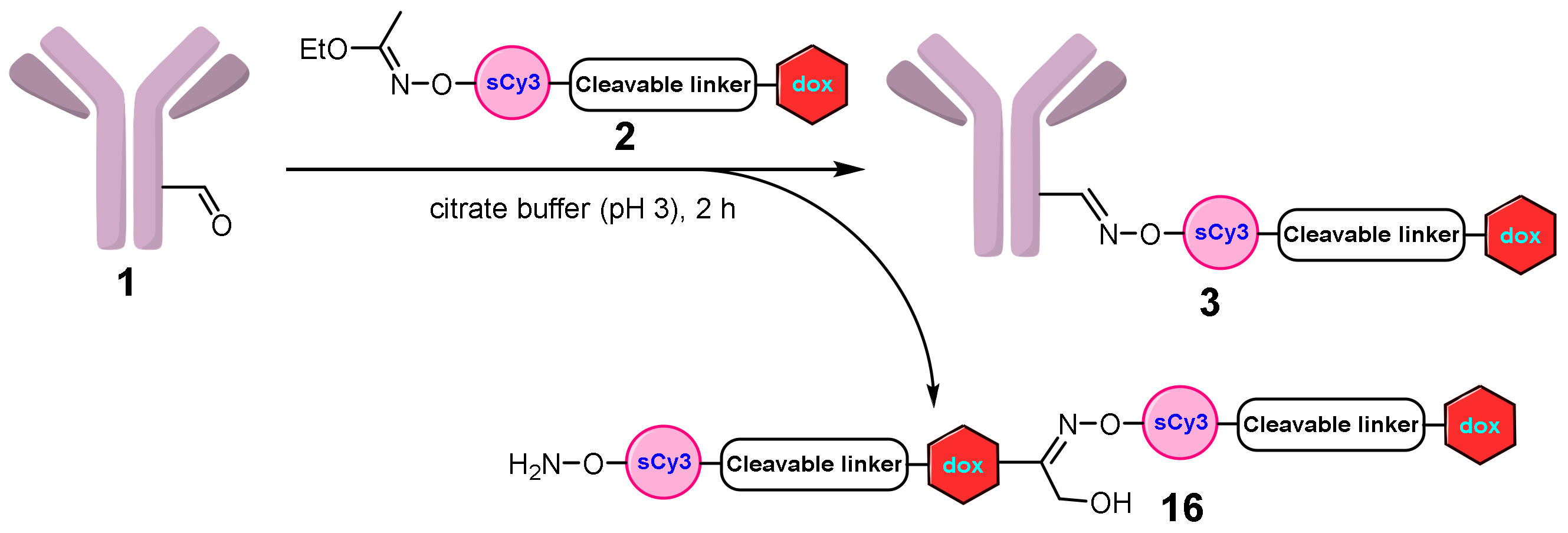
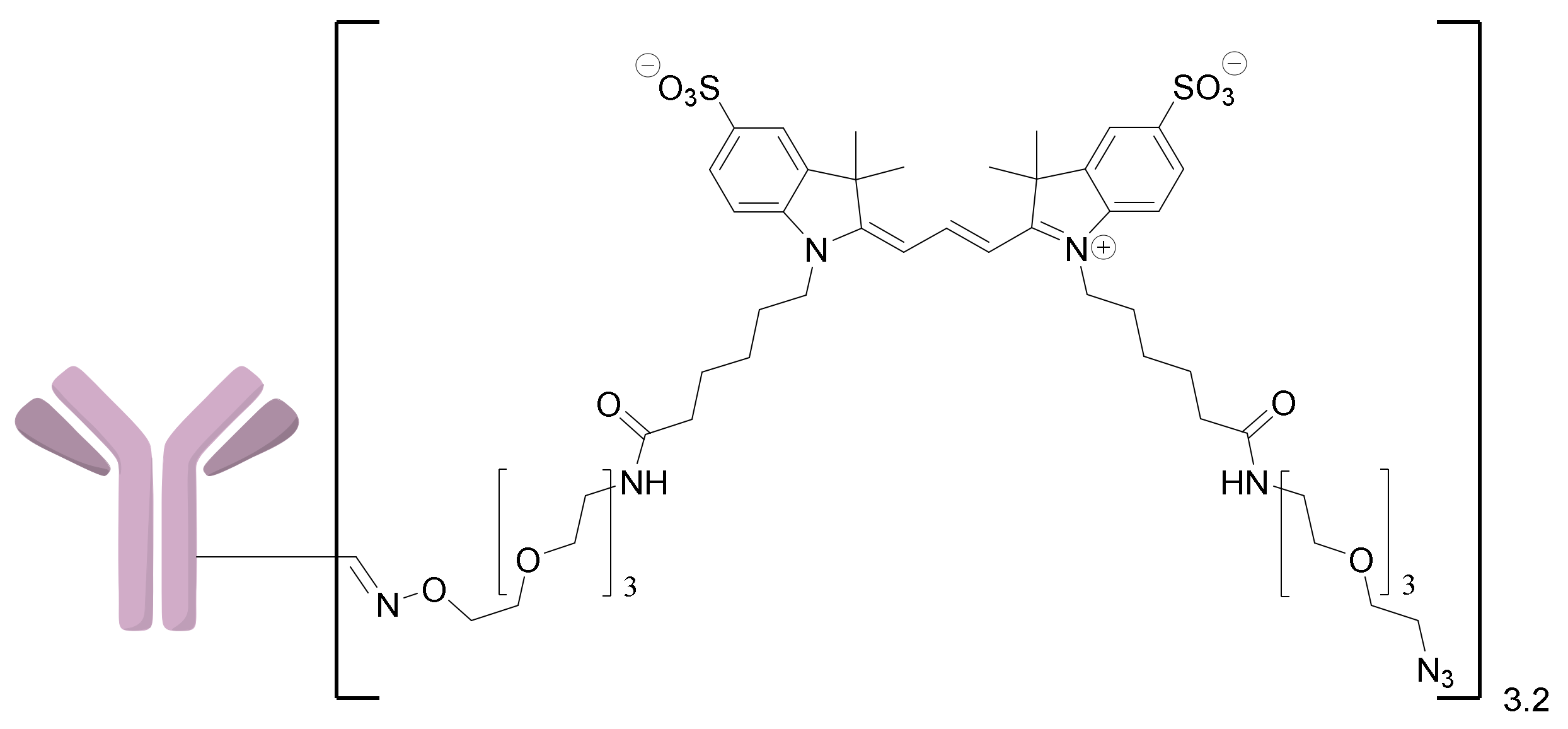
2.1.1. Approach A
2.1.2. Approach B
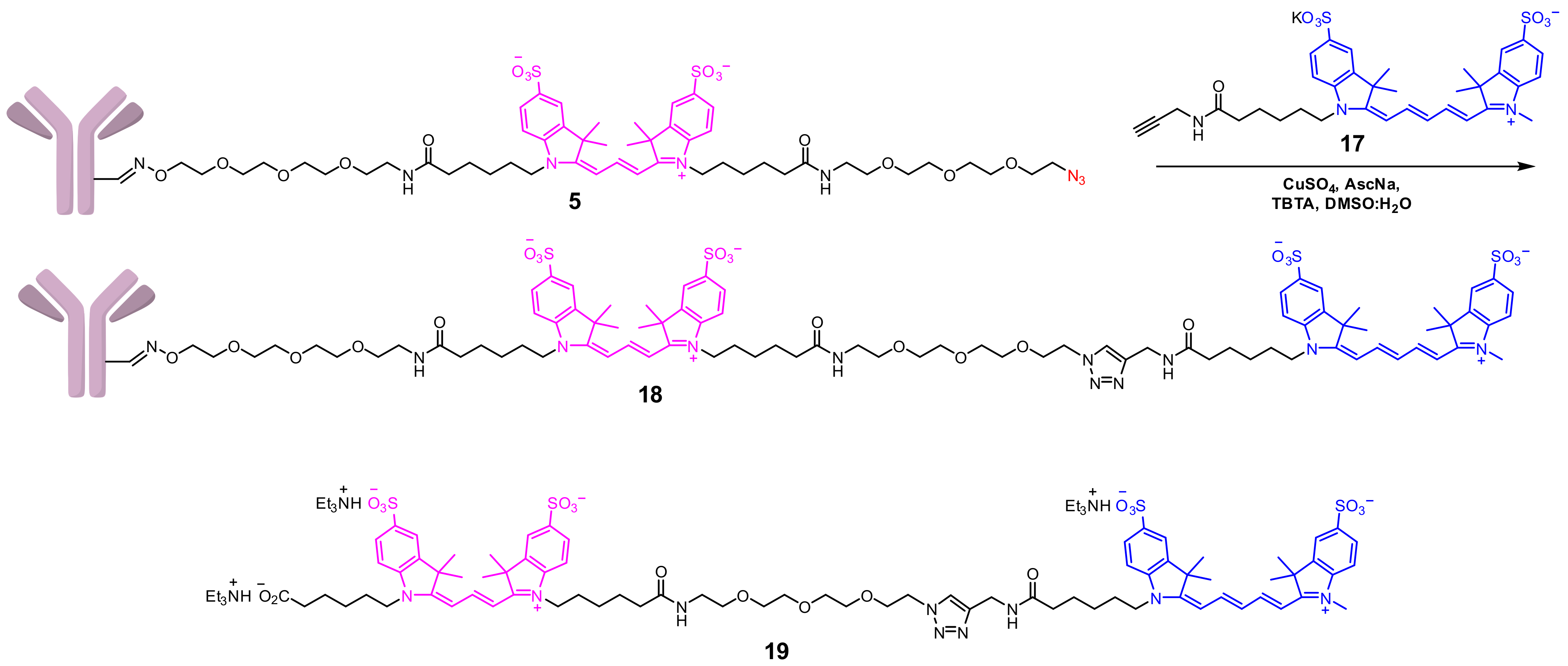
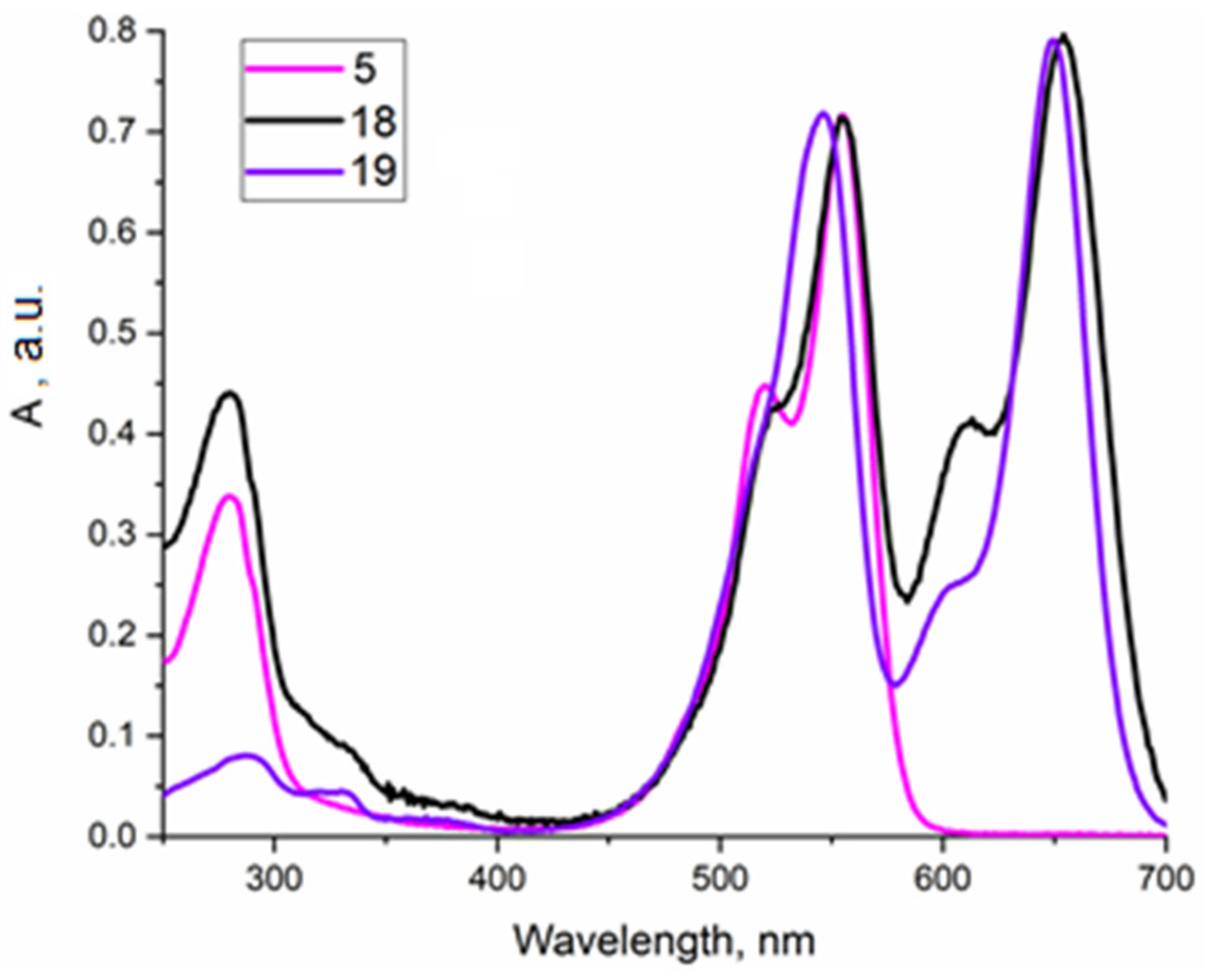
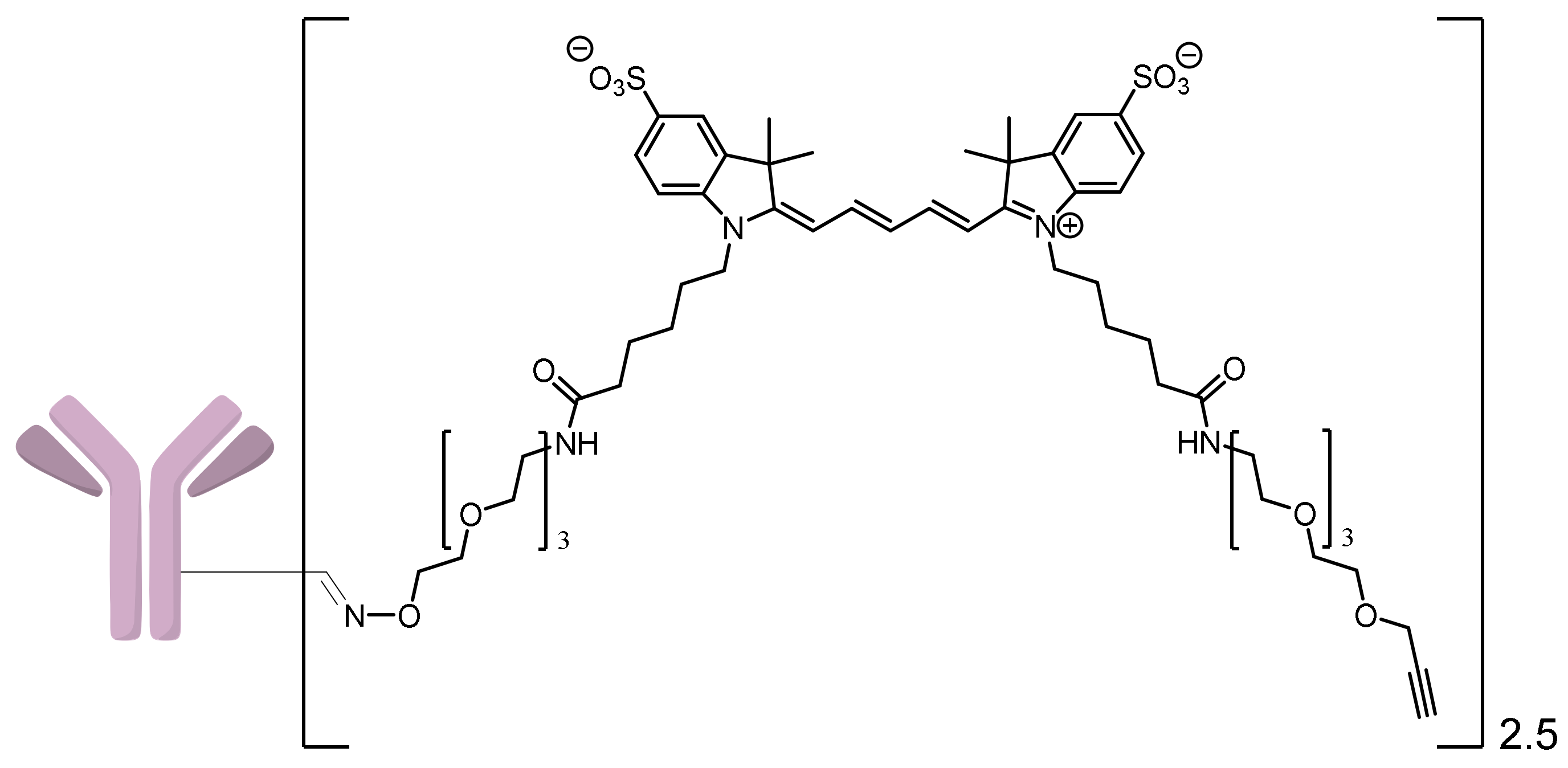
2.1.3. Approach C
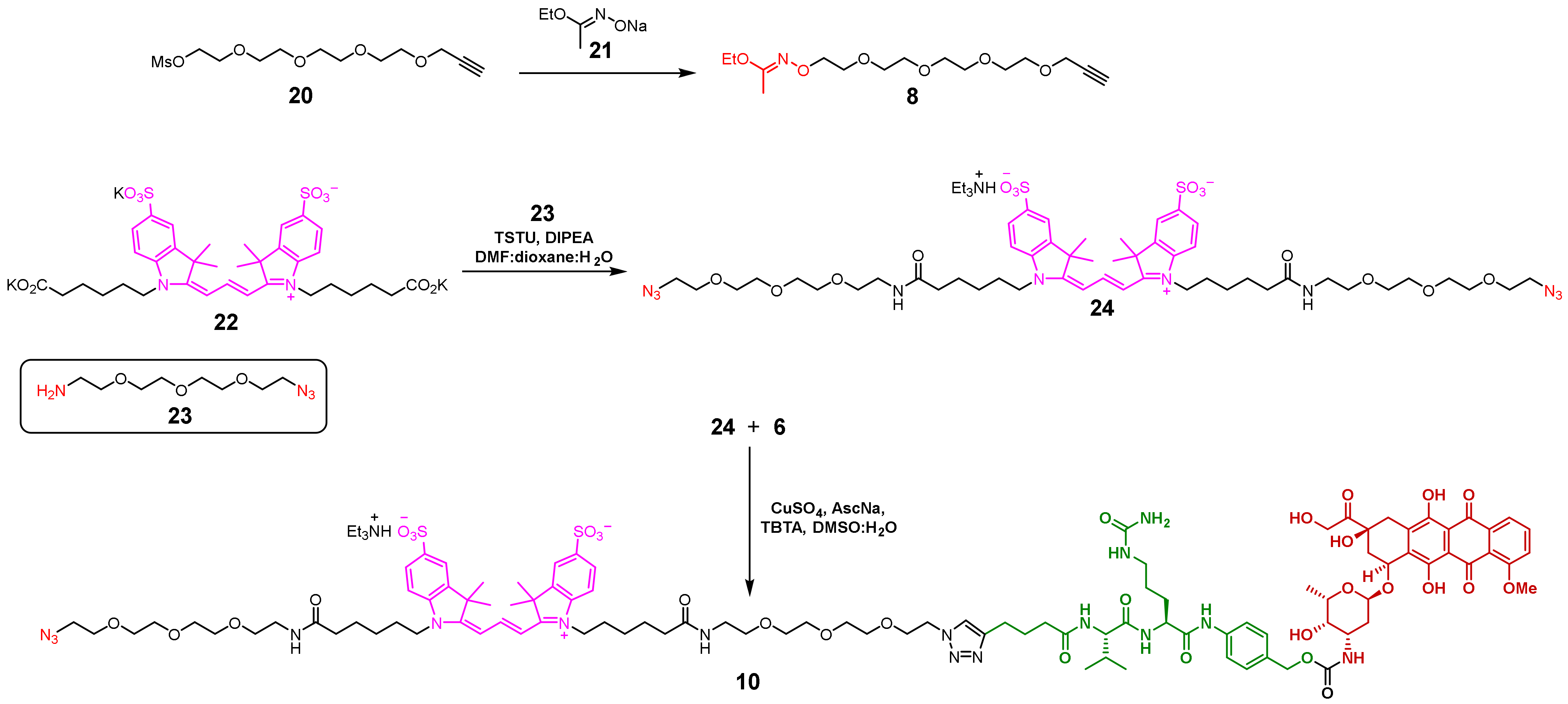
2.1.4. Approach D
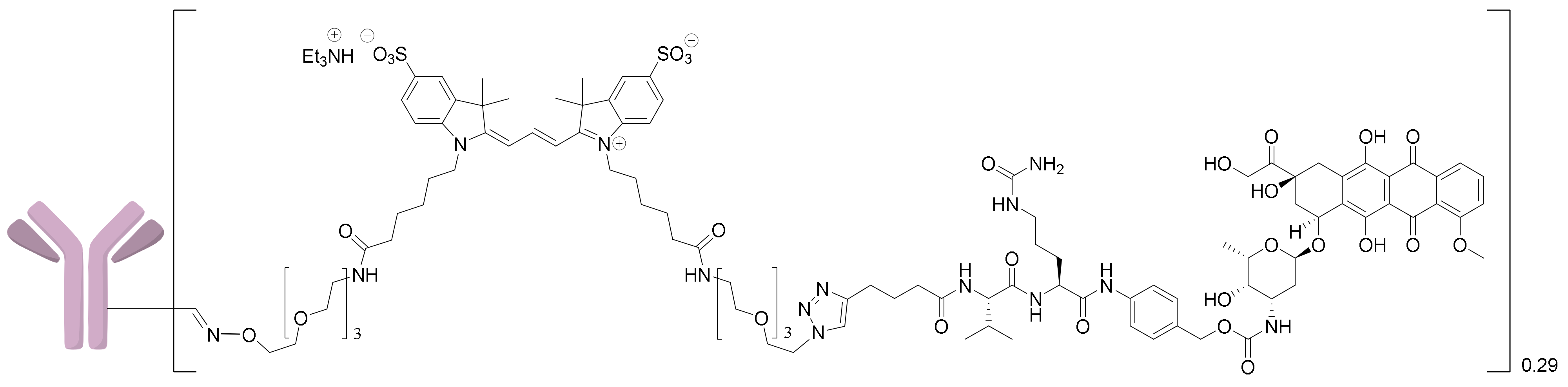
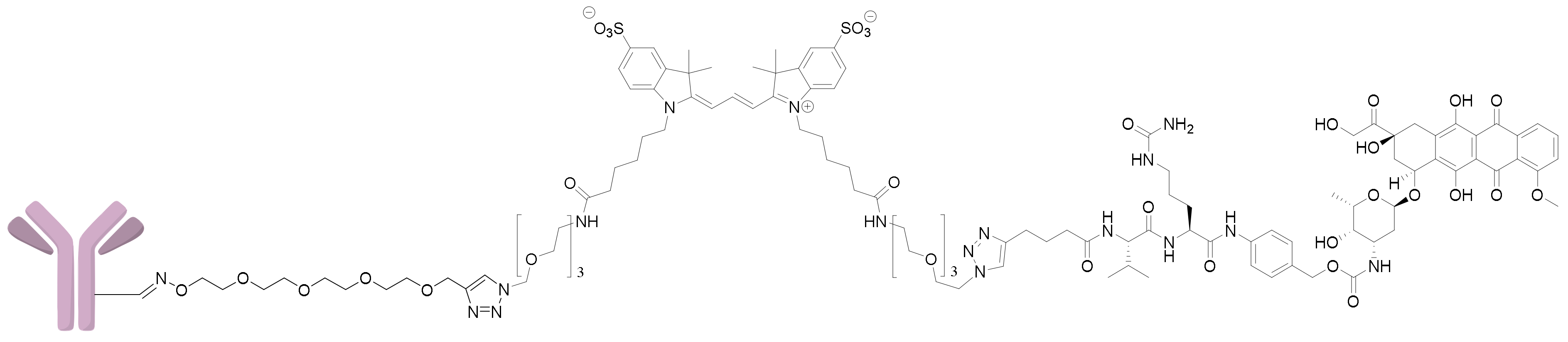
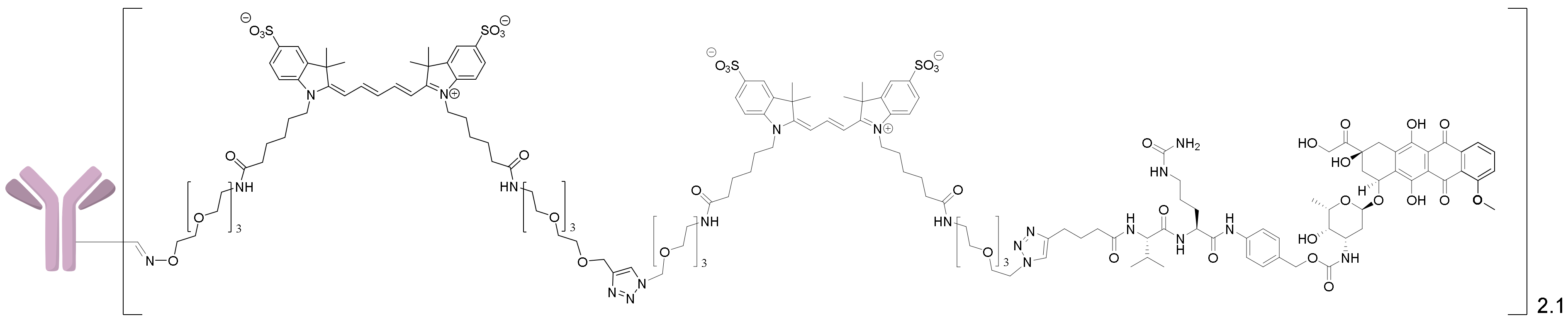
2.1.5. Synthesis of the 6H8-MMAE Conjugate
2.2. Affinity Determination
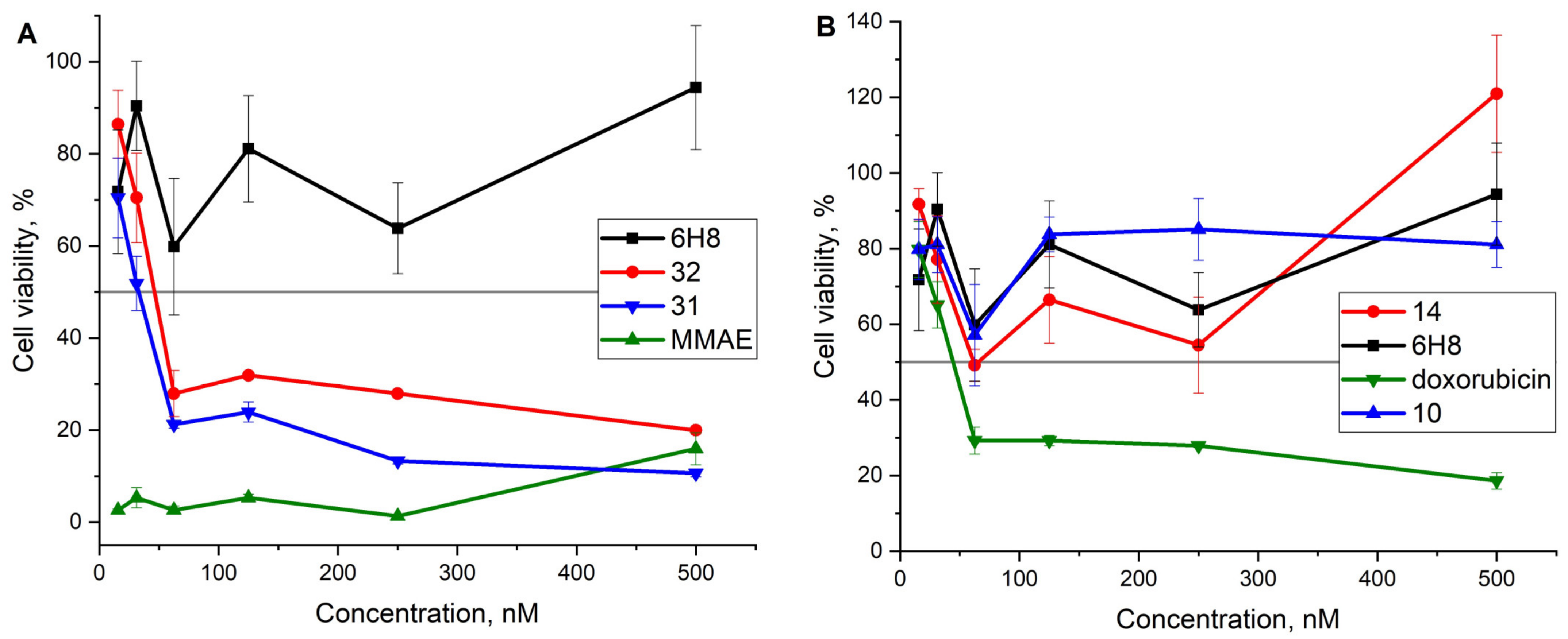
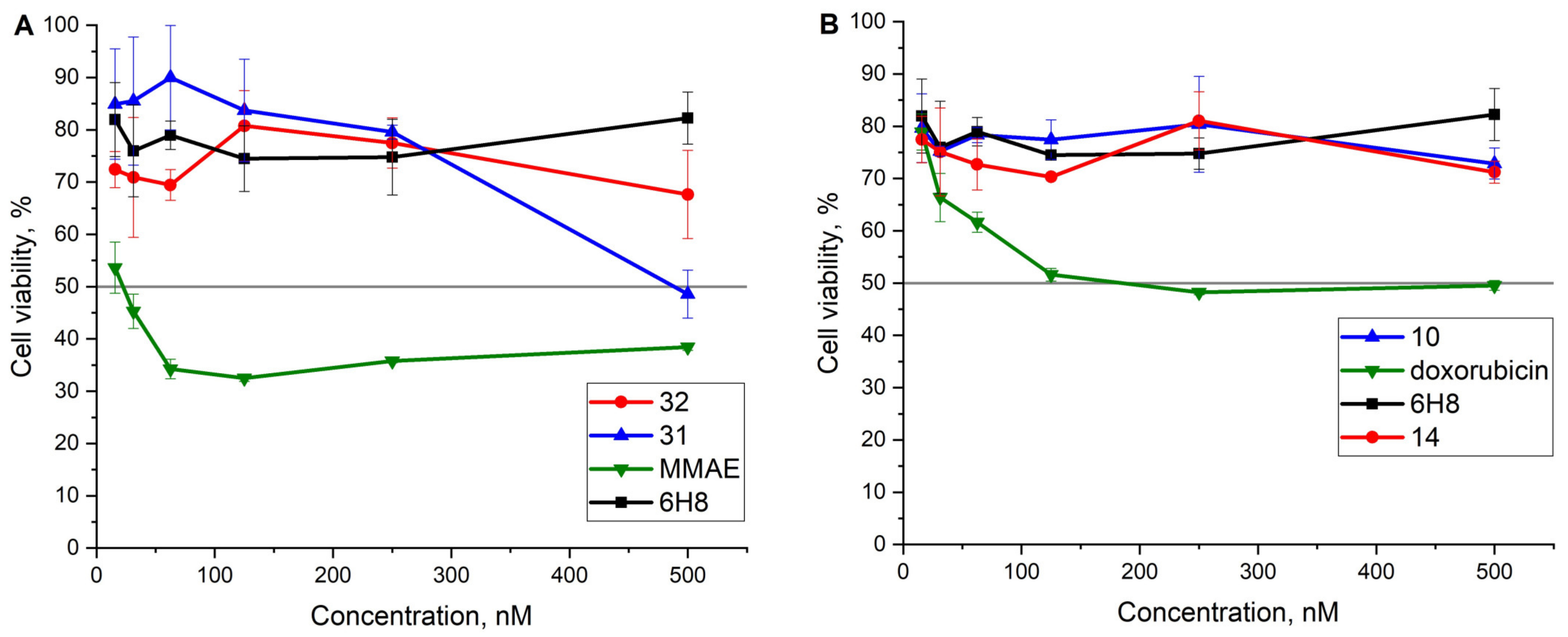
2.3. Cell Viability Assay
3. Materials and Methods
3.1. General Methods
3.2. Synthetic Procedures
3.2.1. Diazide 24
3.2.2. Conjugate 6
3.2.3. Conjugate 2
3.2.4. Conjugate 10
3.2.5. Imidate 8
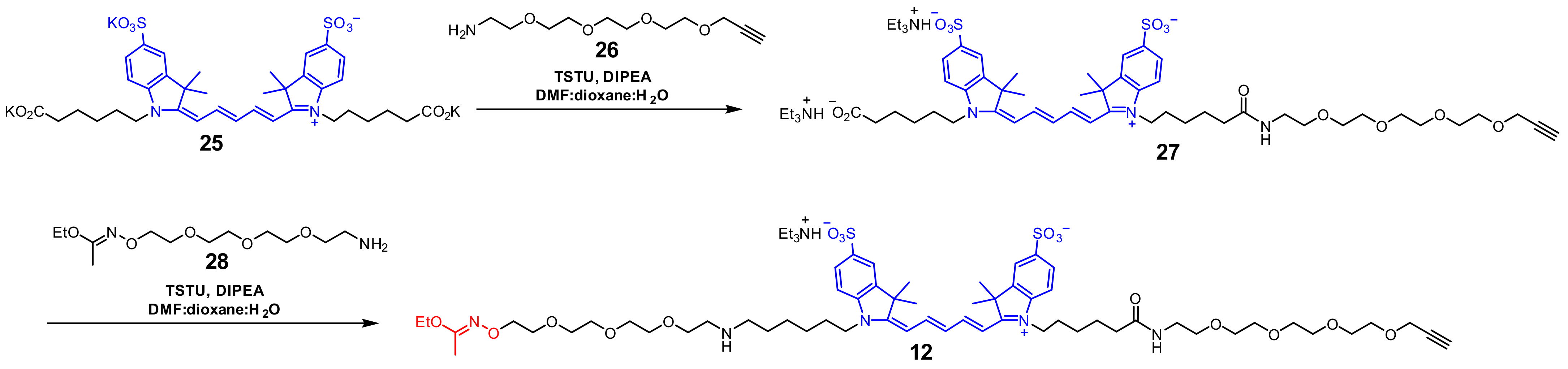
3.2.6. Alkyne 27
3.2.7. Imidate 12
3.2.8. Imidate 30
3.2.9. Conjugate 31
3.3. Synthesis of Antibody Conjugates
3.3.1. General Procedure A
Conjugate 5
Conjugate 9
Conjugate 13
Conjugate 3
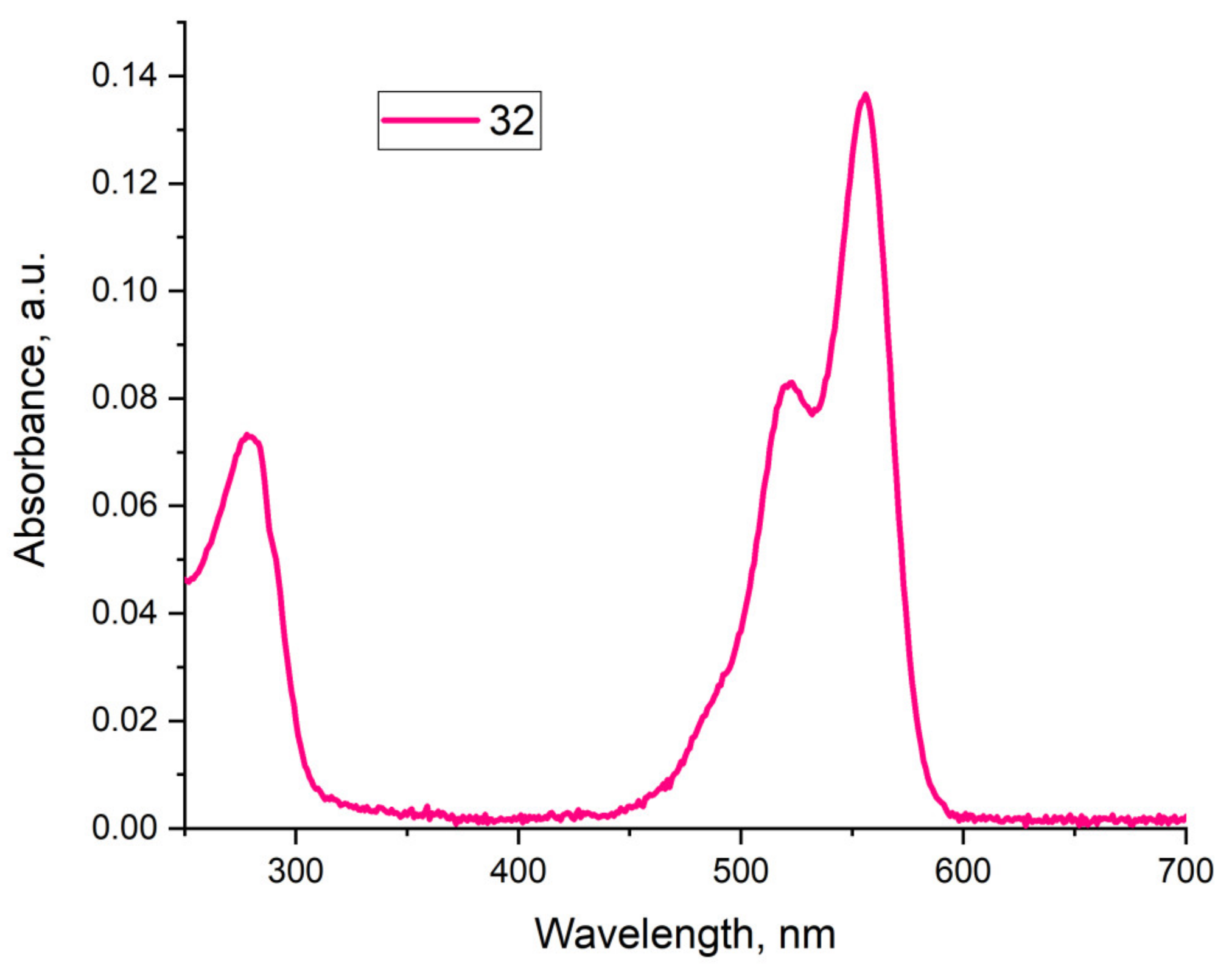
Conjugate 32
3.3.2. General Procedure B
Conjugate 18
Conjugate 11
Conjugate 14
3.4. ELISA Assay
3.5. MTT Assay
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Fu, Z.; Li, S.; Han, S.; Shi, C.; Zhang, Y. Antibody drug conjugate: The “biological missile” for targeted cancer therapy. Signal Transduct. Target. Ther. 2022, 7, 93. [Google Scholar] [CrossRef] [PubMed]
- Jin, S.; Sun, Y.; Liang, X.; Gu, X.; Ning, J.; Xu, Y.; Chen, S.; Pan, L. Emerging new therapeutic antibody derivatives for cancer treatment. Signal Transduct. Target. Ther. 2022, 7, 39. [Google Scholar] [CrossRef]
- Abecassis, P.-Y.; Amara, C. In vivo testing of drug-linker stability. In Antibody−Drug Conjugates; Ducry, L., Ed.; Methods in Molecular Biology; Humana Press: Totowa, NJ, USA, 2013; Volume 1045, pp. 101–116. ISBN 978-1-62703-540-8. [Google Scholar] [CrossRef]
- Anami, Y.; Xiong, W.; Gui, X.; Deng, M.; Zhang, C.C.; Zhang, N.; An, Z.; Tsuchikama, K. Enzymatic conjugation using branched linkers for constructing homogeneous antibody–drug conjugates with high potency. Org. Biomol. Chem. 2017, 15, 5635–5642. [Google Scholar] [CrossRef]
- Jain, N.; Smith, S.W.; Ghone, S.; Tomczuk, B. Current ADC linker chemistry. Pharm. Res. 2015, 32, 3526–3540. [Google Scholar] [CrossRef]
- Tsuchikama, K.; An, Z. Antibody−drug conjugates: Recent advances in conjugation and linker chemistries. Protein Cell 2018, 9, 33–46. [Google Scholar] [CrossRef] [PubMed]
- Bargh, J.D.; Isidro-Llobet, A.; Parker, J.S.; Spring, D.R. Cleavable linkers in antibody–drug conjugates. Chem. Soc. Rev. 2019, 48, 4361–4374. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.S.; Kong, T.W.S.; Khoo, J.Y.X.; Loh, T.-P. Recent developments in chemical conjugation strategies targeting native amino acids in proteins and their applications in antibody–drug conjugates. Chem. Sci. 2021, 12, 13613–13647. [Google Scholar] [CrossRef]
- Walsh, S.J.; Bargh, J.D.; Dannheim, F.M.; Hanby, A.R.; Seki, H.; Counsell, A.J.; Ou, X.; Fowler, E.; Ashman, N.; Takada, Y.; et al. Site-selective modification strategies in antibody–drug conjugates. Chem. Soc. Rev. 2021, 50, 1305–1353. [Google Scholar] [CrossRef]
- Wei, D.; Mao, Y.; Wang, H.; Qu, S.; Chen, J.; Li, J.; Jiang, B.; Chen, H. A mild phenoxysilyl linker for self-immolative release of antibody–drug conjugates. Chin. Chem. Lett. 2023, 108091. [Google Scholar] [CrossRef]
- Von Witting, E.; Hober, S.; Kanje, S. Affinity-based methods for site-specific conjugation of antibodies. Bioconjug. Chem. 2021, 32, 1515–1524. [Google Scholar] [CrossRef]
- Adumeau, P.; Sharma, S.K.; Brent, C.; Zeglis, B.M. Site-specifically labeled immunoconjugates for molecular imaging—Part 1: Cysteine residues and glycans. Mol. Imaging Biol. 2016, 18, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Martin, C.; Brachet, G.; Colas, C.; Allard-Vannier, E.; Kizlik-Masson, C.; Esnault, C.; Respaud, R.; Denevault-Sabourin, C.; Chourpa, I.; Gouilleux-Gruart, V.; et al. In vitro characterization and stability profiles of antibody–fluorophore conjugates derived from interchain cysteine cross-linking or lysine bioconjugation. Pharmaceuticals 2019, 12, 176. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Wang, Z.; Wang, Z.; Luo, F.; Guan, M.; Xu, M.; Li, Y.; Zhang, Y.; Wang, Z.; Wang, W. A simple and efficient method to generate dual site-specific conjugation ADCs with cysteine residue and an unnatural amino acid. Bioconjug. Chem. 2021, 32, 1094–1104. [Google Scholar] [CrossRef] [PubMed]
- Jackson, D.; Atkinson, J.; Guevara, C.I.; Zhang, C.; Kery, V.; Moon, S.-J.; Virata, C.; Yang, P.; Lowe, C.; Pinkstaff, J.; et al. In vitro and in vivo evaluation of cysteine and site specific conjugated herceptin antibody–drug conjugates. PLoS ONE 2014, 9, e83865. [Google Scholar] [CrossRef] [PubMed]
- Levengood, M.R.; Zhang, X.; Hunter, J.H.; Emmerton, K.K.; Miyamoto, J.B.; Lewis, T.S.; Senter, P.D. Orthogonal cysteine protection enables homogeneous multi-drug antibody–drug conjugates. Angew. Chem. Int. Ed. 2017, 56, 733–737. [Google Scholar] [CrossRef]
- Nilchan, N.; Li, X.; Pedzisa, L.; Nanna, A.R.; Roush, W.R.; Rader, C. Dual-mechanistic antibody–drug conjugate via site-specific selenocysteine/cysteine conjugation. Antib. Ther. 2019, 2, 71–78. [Google Scholar] [CrossRef]
- Scales, S.J.; Tsai, S.P.; Zacharias, N.; dela Cruz-Chuh, J.; Bullen, G.; Velasquez, E.; Chang, J.; Bruguera, E.; Kozak, K.R.; Sadowsky, J. Development of a cysteine-conjugatable disulfide FRET probe: Influence of charge on linker cleavage and payload trafficking for an anti-HER2 antibody conjugate. Bioconjug. Chem. 2019, 30, 3046–3056. [Google Scholar] [CrossRef]
- Edelmann, M.R.; Hauri, S. Functional in vitro assessment of modified antibodies: Impact of label on protein properties. PLoS ONE 2021, 16, e0257342. [Google Scholar] [CrossRef]
- Kim, C.H.; Axup, J.Y.; Dubrovska, A.; Kazane, S.A.; Hutchins, B.A.; Wold, E.D.; Smider, V.V.; Schultz, P.G. Synthesis of bispecific antibodies using genetically encoded unnatural amino acids. J. Am. Chem. Soc. 2012, 134, 9918–9921. [Google Scholar] [CrossRef]
- Axup, J.Y.; Bajjuri, K.M.; Ritland, M.; Hutchins, B.M.; Kim, C.H.; Kazane, S.A.; Halder, R.; Forsyth, J.S.; Santidrian, A.F.; Stafin, K.; et al. Synthesis of site-specific antibody–drug conjugates using unnatural amino acids. Proc. Natl. Acad. Sci. USA 2012, 109, 16101–16106. [Google Scholar] [CrossRef]
- Giddens, J.P.; Wang, L.-X. Chemoenzymatic glyco-engineering of monoclonal antibodies. In Glyco-Engineering; Castilho, A., Ed.; Methods in Molecular Biology; Springer: New York, NY, USA, 2015; Volume 1321, pp. 375–387. ISBN 978-1-4939-2759-3. [Google Scholar] [CrossRef]
- Huang, W.; Giddens, J.; Fan, S.-Q.; Toonstra, C.; Wang, L.-X. Chemoenzymatic glycoengineering of intact IgG antibodies for gain of functions. J. Am. Chem. Soc. 2012, 134, 12308–12318. [Google Scholar] [CrossRef] [PubMed]
- Van Berkel, S.S.; van Delft, F.L. Enzymatic strategies for (near) clinical development of antibody–drug conjugates. Drug Discov. Today Technol. 2018, 30, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Castilho, A.; Gruber, C.; Thader, A.; Oostenbrink, C.; Pechlaner, M.; Steinkellner, H.; Altmann, F. Processing of complex N-glycans in IgG Fc-region is affected by core fucosylation. mAbs 2015, 7, 863–870. [Google Scholar] [CrossRef]
- Quast, I.; Peschke, B.; Lünemann, J.D. Regulation of antibody effector functions through IgG Fc N-glycosylation. Cell. Mol. Life Sci. 2017, 74, 837–847. [Google Scholar] [CrossRef] [PubMed]
- Qasba, P.K. Glycans of antibodies as a specific site for drug conjugation using glycosyltransferases. Bioconjug. Chem. 2015, 26, 2170–2175. [Google Scholar] [CrossRef]
- Wolfe, C.A.C.; Hage, D.S. Studies on the rate and control of antibody oxidation by periodate. Anal. Biochem. 1995, 231, 123–130. [Google Scholar] [CrossRef]
- Zuberbühler, K.; Casi, G.; Bernardes, G.J.L.; Neri, D. Fucose-specific conjugation of hydrazide derivatives to a vascular-targeting monoclonal antibody in IgG format. Chem. Commun. 2012, 48, 7100. [Google Scholar] [CrossRef]
- Kölmel, D.K.; Kool, E.T. Oximes and hydrazones in bioconjugation: Mechanism and catalysis. Chem. Rev. 2017, 117, 10358–10376. [Google Scholar] [CrossRef]
- Sapozhnikova, K.A.; Misyurin, V.A.; Ryazantsev, D.Y.; Kokin, E.A.; Finashutina, Y.P.; Alexeeva, A.V.; Ivanov, I.A.; Kocharovskaya, M.V.; Tikhonova, N.A.; Popova, G.P.; et al. Sensitive immunofluorescent detection of the PRAME antigen using a practical antibody conjugation approach. Int. J. Mol. Sci. 2021, 22, 12845. [Google Scholar] [CrossRef]
- Misyurin, V.A.; Finashutina, Y.P.; Turba, A.A.; Larina, M.V.; Solopova, O.N.; Lyzhko, N.A.; Kesaeva, L.A.; Kasatkina, N.N.; Aliev, T.K.; Misyurin, A.V.; et al. Epitope analysis of murine and chimeric monoclonal antibodies recognizing the cancer testis antigen PRAME. Dokl. Biochem. Biophys. 2020, 492, 135–138. [Google Scholar] [CrossRef]
- Finashutina, Y.P.; Misyurin, A.V.; Akhlynina, T.V.; Lyzhko, N.A.; Krutov, A.A.; Aksenova, E.V.; Misyurin, V.A.; Baryshnikov, A.Y. Production of purified human recombinant antigen PRAME and specific monoclonal antibodies. Russ. J. Biother. 2015, 14, 29–36. [Google Scholar] [CrossRef]
- Sapozhnikova, K.A.; Misyurin, A.V.; Pestov, N.B.; Meleshkina, E.G.; Oreshkov, S.D.; Ganzhula, E.P.; Mikhailova, A.S.; Korshun, V.A.; Misyurin, V.A.; Brylev, V.A. Detection of the PRAME protein on the surface of melanoma cells using a fluorescently labeled monoclonal antibody. Russ. J. Bioorg. Chem. 2021, 47, 1077–1085. [Google Scholar] [CrossRef]
- Sapozhnikova, K.A.; Gulyak, E.L.; Misyurin, V.A.; Simonova, M.A.; Ryabukhina, E.V.; Alexeeva, A.V.; Tikhonova, N.A.; Lyzhko, N.A.; Popova, G.P.; Misyurin, A.V.; et al. Branched linkers for site-specific fluorescent labeling of antibodies. Molecules 2023, 28, 425. [Google Scholar] [CrossRef] [PubMed]
- Wadelin, F.; Fulton, J.; McEwan, P.A.; Spriggs, K.A.; Emsley, J.; Heery, D.M. Leucine-rich repeat protein PRAME: Expression, potential functions and clinical implications for leukaemia. Mol. Cancer 2010, 9, 226. [Google Scholar] [CrossRef] [PubMed]
- Kaczorowski, M.; Chłopek, M.; Kruczak, A.; Ryś, J.; Lasota, J.; Miettinen, M. PRAME expression in cancer. A systematic immunohistochemical study of >5800 epithelial and nonepithelial tumors. Am. J. Surg. Pathol. 2022, 46, 1467–1476. [Google Scholar] [CrossRef]
- Ikeda, H.; Lethé, B.; Lehmann, F.; Van Baren, N.; Baurain, J.-F.; De Smet, C.; Chambost, H.; Vitale, M.; Moretta, A.; Boon, T.; et al. Characterization of an antigen that is recognized on a melanoma showing partial HLA loss by CTL expressing an NK inhibitory receptor. Immunity 1997, 6, 199–208. [Google Scholar] [CrossRef]
- Proto-Siqueira, R.; Figueiredo-Pontes, L.L.; Panepucci, R.A.; Garcia, A.B.; Rizzatti, E.G.; Nascimento, F.M.; Ishikawa, H.C.F.; Larson, R.E.; Falcão, R.P.; Simpson, A.J.; et al. PRAME is a membrane and cytoplasmic protein aberrantly expressed in chronic lymphocytic leukemia and mantle cell lymphoma. Leuk. Res. 2006, 30, 1333–1339. [Google Scholar] [CrossRef] [PubMed]
- Penskaya, E.A.; Misyurin, V.A.; Misyurina, A.E.; Kravchenko, S.K.; Gorenkova, L.G.; Plastinina, L.V.; Tikhonova, V.V.; Finashutina, Y.P.; Lyzhko, N.A.; Kasatkina, N.N.; et al. Prognostic value of the PRAME gene expression in T-cell lymphoproliferative disorders. Clin. Oncohematol. 2019, 12, 73–78. [Google Scholar] [CrossRef]
- Misyurin, V.A.; Misyurina, A.E.; Kravchenko, S.K.; Lyzhko, N.A.; Finashutina, Y.P.; Kasatkina, N.N.; Mar’in, D.S.; Nesterova, E.S.; Sharkunov, N.N.; Baryshnikova, M.A.; et al. Prognostic value of PRAME activity in tumor cells of follicular lymphoma. Clin. Oncohematol. 2019, 12, 173–178. [Google Scholar] [CrossRef]
- Misyurin, V.A. Clinical significance of the PRAME gene expression in oncohematological diseases. Clin. Oncohematol. 2018, 11, 26–33. [Google Scholar] [CrossRef]
- Tacar, O.; Sriamornsak, P.; Dass, C.R. Doxorubicin: An update on anticancer molecular action, toxicity and novel drug delivery systems. J. Pharm. Pharmacol. 2012, 65, 157–170. [Google Scholar] [CrossRef] [PubMed]
- Pommier, Y.; Leo, E.; Zhang, H.; Marchand, C. DNA Topoisomerases and their poisoning by anticancer and antibacterial drugs. Chem. Biol. 2010, 17, 421–433. [Google Scholar] [CrossRef]
- Pang, B.; Qiao, X.; Janssen, L.; Velds, A.; Groothuis, T.; Kerkhoven, R.; Nieuwland, M.; Ovaa, H.; Rottenberg, S.; van Tellingen, O.; et al. Drug-induced histone eviction from open chromatin contributes to the chemotherapeutic effects of doxorubicin. Nat. Commun. 2013, 4, 1908. [Google Scholar] [CrossRef] [PubMed]
- Nicolaou, K.C.; Rigol, S. The role of organic synthesis in the emergence and development of antibody–drug conjugates as targeted cancer therapies. Angew. Chem. Int. Ed. 2019, 58, 11206–11241. [Google Scholar] [CrossRef] [PubMed]
- Francisco, J.A.; Cerveny, C.G.; Meyer, D.L.; Mixan, B.J.; Klussman, K.; Chace, D.F.; Rejniak, S.X.; Gordon, K.A.; DeBlanc, R.; Toki, B.E.; et al. CAC10-VcMMAE, an anti-CD30–monomethyl auristatin E conjugate with potent and selective antitumor activity. Blood 2003, 102, 1458–1465. [Google Scholar] [CrossRef]
- Tong, J.T.W.; Harris, P.W.R.; Brimble, M.A.; Kavianinia, I. An insight into FDA approved antibody–drug conjugates for cancer therapy. Molecules 2021, 26, 5847. [Google Scholar] [CrossRef]
- Agard, N.J.; Prescher, J.A.; Bertozzi, C.R. A strain-promoted [3 + 2] azide−alkyne cycloaddition for covalent modification of biomolecules in living systems. J. Am. Chem. Soc. 2004, 126, 15046–15047. [Google Scholar] [CrossRef]
- Cazzato, G.; Cascardi, E.; Colagrande, A.; Belsito, V.; Lospalluti, L.; Foti, C.; Arezzo, F.; Dellino, M.; Casatta, N.; Lupo, C.; et al. PRAME immunoexpression in 275 cutaneous melanocytic lesions: A double institutional experience. Diagnostics 2022, 12, 2197. [Google Scholar] [CrossRef]
- Pankov, D.; Sjöström, L.; Kalidindi, T.; Lee, S.-G.; Sjöström, K.; Gardner, R.; McDevitt, M.R.; O’Reilly, R.; Thorek, D.L.J.; Larson, S.M.; et al. In vivo immuno-targeting of an extracellular epitope of membrane bound preferentially expressed antigen in melanoma (PRAME). Oncotarget 2017, 8, 65917–65931. [Google Scholar] [CrossRef]
- Castle, J.C.; Loewer, M.; Boegel, S.; de Graaf, J.; Bender, C.; Tadmor, A.D.; Boisguerin, V.; Bukur, T.; Sorn, P.; Paret, C.; et al. Immunomic, genomic and transcriptomic characterization of CT26 colorectal carcinoma. BMC Genom. 2014, 15, 190. [Google Scholar] [CrossRef]
- Lezcano, C.; Jungbluth, A.A.; Nehal, K.S.; Hollmann, T.J.; Busam, K.J. PRAME expression in melanocytic tumors. Am. J. Surg. Pathol. 2018, 42, 1456–1465. [Google Scholar] [CrossRef] [PubMed]
- Kalinovsky, D.V.; Kibardin, A.V.; Kholodenko, I.V.; Svirshchevskaya, E.V.; Doronin, I.I.; Konovalova, M.V.; Grechikhina, M.V.; Rozov, F.N.; Larin, S.S.; Deyev, S.M.; et al. Therapeutic efficacy of antibody–drug conjugates targeting GD2-positive tumors. J. Immunother. Cancer 2022, 10, e004646. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, L.; Fan, S.; Xiao, D.; Xie, F.; Li, W.; Zhong, W.; Zhou, X. Antibody–drug conjugate using ionized Cys-linker-MMAE as the potent payload shows optimal therapeutic safety. Cancers 2020, 12, 744. [Google Scholar] [CrossRef]
- Machulkin, A.E.; Uspenskaya, A.A.; Zyk, N.U.; Nimenko, E.A.; Ber, A.P.; Petrov, S.A.; Polshakov, V.I.; Shafikov, R.R.; Skvortsov, D.A.; Plotnikova, E.A.; et al. Synthesis, characterization, and preclinical evaluation of a small-molecule prostate-specific membrane antigen-targeted monomethyl auristatin E conjugate. J. Med. Chem. 2021, 64, 17123–17145. [Google Scholar] [CrossRef]
- Goswami, L.N.; Houston, Z.H.; Sarma, S.J.; Jalisatgi, S.S.; Hawthorne, M.F. Efficient synthesis of diverse heterobifunctionalized clickable oligo(ethylene glycol) linkers: Potential applications in bioconjugation and targeted drug delivery. Org. Biomol. Chem. 2013, 11, 1116. [Google Scholar] [CrossRef]
- Schwabacher, A.W.; Lane, J.W.; Schiesher, M.W.; Leigh, K.M.; Johnson, C.W. Desymmetrization reactions: Efficient preparation of unsymmetrically substituted linker molecules. J. Org. Chem. 1998, 63, 1727–1729. [Google Scholar] [CrossRef]
- Kalinovsky, D.V.; Kholodenko, I.V.; Kibardin, A.V.; Doronin, I.I.; Svirshchevskaya, E.V.; Ryazantsev, D.Y.; Konovalova, M.V.; Rozov, F.N.; Larin, S.S.; Deyev, S.M.; et al. Minibody-based and ScFv-based antibody fragment-drug conjugates selectively eliminate GD2-positive tumor cells. Int. J. Mol. Sci. 2023, 24, 1239. [Google Scholar] [CrossRef] [PubMed]










Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sapozhnikova, K.A.; Gulyak, E.L.; Brylev, V.A.; Misyurin, V.A.; Oreshkov, S.D.; Alexeeva, A.V.; Ryazantsev, D.Y.; Simonova, M.A.; Ryabukhina, E.V.; Popova, G.P.; et al. Aminooxy Click Modification of a Periodate-Oxidized Immunoglobulin G: A General Approach to Antibody–Drug Conjugates with Dye-Mediated Expeditious Stoichiometry Control. Int. J. Mol. Sci. 2023, 24, 5134. https://doi.org/10.3390/ijms24065134
Sapozhnikova KA, Gulyak EL, Brylev VA, Misyurin VA, Oreshkov SD, Alexeeva AV, Ryazantsev DY, Simonova MA, Ryabukhina EV, Popova GP, et al. Aminooxy Click Modification of a Periodate-Oxidized Immunoglobulin G: A General Approach to Antibody–Drug Conjugates with Dye-Mediated Expeditious Stoichiometry Control. International Journal of Molecular Sciences. 2023; 24(6):5134. https://doi.org/10.3390/ijms24065134
Chicago/Turabian StyleSapozhnikova, Ksenia A., Evgeny L. Gulyak, Vladimir A. Brylev, Vsevolod A. Misyurin, Sergey D. Oreshkov, Anastasiya V. Alexeeva, Dmitry Yu. Ryazantsev, Maria A. Simonova, Ekaterina V. Ryabukhina, Galina P. Popova, and et al. 2023. "Aminooxy Click Modification of a Periodate-Oxidized Immunoglobulin G: A General Approach to Antibody–Drug Conjugates with Dye-Mediated Expeditious Stoichiometry Control" International Journal of Molecular Sciences 24, no. 6: 5134. https://doi.org/10.3390/ijms24065134
APA StyleSapozhnikova, K. A., Gulyak, E. L., Brylev, V. A., Misyurin, V. A., Oreshkov, S. D., Alexeeva, A. V., Ryazantsev, D. Y., Simonova, M. A., Ryabukhina, E. V., Popova, G. P., Tikhonova, N. A., Lyzhko, N. A., Barmashov, A. E., Misyurin, A. V., Ustinov, A. V., Alferova, V. A., & Korshun, V. A. (2023). Aminooxy Click Modification of a Periodate-Oxidized Immunoglobulin G: A General Approach to Antibody–Drug Conjugates with Dye-Mediated Expeditious Stoichiometry Control. International Journal of Molecular Sciences, 24(6), 5134. https://doi.org/10.3390/ijms24065134





