Role of Plant-Derived Compounds in the Molecular Pathways Related to Inflammation
Abstract
1. Introduction
2. Molecular Pathways of Inflammation
2.1. Pattern Recognition Receptors
2.2. Nuclear Factor-κB
2.3. Transcription Factor Nrf2
2.4. Mitogen-Activated Protein Kinases
3. Flavonoids
3.1. Flavones
| Compounds | Model | Mode of Action | Reference |
|---|---|---|---|
| Flavones | |||
| Apigenin | TGF-β1-induced nasal fibroblasts | ↓ MAPK, NF-κB | [57] |
| human melanocytes (PIG3V) | ↑ Nrf2 | [58] | |
| murine microglial cell line (BV2) | ↑ Nrf2, ↓ NF-κB, TNF-α, IL-1β, IL-6 | [59] | |
| LPS-induced acute lung injury in mice | ↓ NF-κB, TNF-α, IL-1β, IL-6, COX-2 | [60] | |
| coculture of adipocytes (3T3-L1) and macrophages (RAW 264.7) | ↓ NF-κB, MAPK | [73] | |
| Diosmetin | S. pneumoniae-induced meningitis in rats | ↓ NF-κB, TNF-α, IL-1β, IL-6 | [74] |
| LPS-induced acute lung injury in mice | ↑ Nrf2, ↓ TNF-α, IL-1β, IL-6 | [61] | |
| Diosmin | alloxan-induced nephropathy in rats | ↓ NF-κB, TNF-α, IL-1β, IL-6 | [75] |
| doxorubicin-induced hepatotoxicity in rats | ↓ NF-κB, MAPK, TNF-α, IL-6, IL-1β | [76] | |
| Luteolin | monolayer of colon adenocarcinoma cell line (Caco-2) | ↑ Nrf2, ↓ NF-κB, MAPK | [77] |
| S. aureus-induced mastitis in mice | ↓ NF-κB, TNF-α, IL-1β, IL-6, MMP-2, MMP-9 | [64] | |
| LPS-induced ulcerative colitis in rats | ↓ NF-κB, IL-17, IL-23 | [78] | |
| Vitexin | rat insulinoma cell line (INS-1) | ↑ Nrf2, ↓ NF-κB | [79] |
| LPS-induced acute lung injury in mice | ↑ Nrf2 | [62] | |
| S. aureus-induced mastitis in mice | ↓ NF-κB, MAPK, TNF-α, IL-1β, IL-6 | [63] | |
| Flavonols | |||
| Avicularin | IL-1β-induced rat and human chondrocytes | ↓ MAPK, iNOS, COX-2, MMP3, MMP-13, TRAF-6 | [80] |
| IL-1β-induced murine chondrocytes | ↑ Nrf2, ↓ NF-κB, COX-2 | [81] | |
| Hyperoside | TNF-α-induced intervertebral disc degeneration in human nucleus pulposus cells | ↑ Nrf2, ↓ NF-κB, IL-6, iNOS, COX-2, TNF-α, IL-1β, MMP-3, MMP-13 | [82] |
| human lung carcinoma cell line (A549) | ↓MAPK | [83] | |
| OVA *-induced asthma model in mice | ↑ Nrf2, ↓ NF-κB, IL-4, IL-5, IL-13 | [84] | |
| Isoquercitrin | human liver cancer cell line (HepG2) | ↓ MAPK | [85] |
| acute myocardial infraction model in rats | ↓ NF-κB, TNF-α, IL-6, NO | [86] | |
| middle cerebral artery occlusion in rats | ↑ Nrf2, ↓ NF-κB | [87] | |
| Isorhamnetin | bronchial epithelial cell line (BEAS-2B) | ↓ MAPK, NF-κB, IL-1β, IL-8, IL-6 | [88] |
| LPS-induced chronic obstructive pulmonary disease in mice | ↑ Nrf2, ↓ IL-6 | [89] | |
| LPS-induced lung injury in mice | ↓ NF-κB, IL-6, IL-1β, TNF-α | [90] | |
| Kaempferol | LPS- & ATP-induced cardiac fibroblasts | ↓ NF-κB, TNF-α, IL-1β, IL-6, IL-18 | [91] |
| I/R **-induced lung injury in rats | ↓ NF-κB, TNF-α, IL-6, HMGB1 | [92] | |
| cadmium-chloride-induced nephropathy in rats | ↑ Nrf2, ↓ NF-κB, ROS, IL-6, TNF-α | [93] | |
| murine microglial cell line (BV2) spinal cord injury model in rats | ↓ MAPK, IL-1β, IL-18, Nox4, | [94] | |
| Quercetin | IL-1β-induced nucleus pulposus cells | ↑ Nrf2, ↓ NF-κB, IL-6, IL-8, MMP-3, MMP13 | [95] |
| leptin-induced human umbilical vein endothelial cell line (HUVEC) | ↓ NF-κB, TNF-α | [96] | |
| chronic prostatitis/chronic pelvic pain syndrome model in rats | ↓MAPK, NF-κB, IL-1β, IL-2, IL-6, IL-17A, TNF-α, MCP-1 | [97] | |
| cardiomyopathic rats induced by doxorubicin | ↑ Nrf2 | [98] | |
| CAD *** patients | ↓ NF-κB, TNF-α, IL-1β | [99] | |
| Rutin | LPS-induced murine macrophages (RAW 264.7) | ↓ NF-κB | [100] |
| H2O2-induced oxidative stress in HUVEC | ↑ Nrf2, ↓ NF-κB | [101] | |
| HMGB1-induced HUVEC | ↓ NF-κB, VCAM-1, TNF-α, IL-6 | [102] | |
| LPS-induced acute lung injury model in mice | ↓MAPK, TNF-α, IL-6, IL-1β | [103] | |
| chronic colitis model in mice | ↓ NF-κB, TNF-α, IL-1β, IL-6 | [104] | |
| Spiraeoside | human cardiomyocytes (AC16) | ↑ Nrf2 | [105] |
| Tiliroside | murine microglial cell line (BV2) | ↑ Nrf2, ↓ NF-κB, IL-6, TNF-α, IL-1β | [106] |
| bone-marrow-derived macrophage (BMM) | ↓ NF-κB, MAPK, ROS | [107] | |
| Flavanones | |||
| Eriodictyol | human osteoarthritis chondrocytes | ↑ Nrf2, ↓ NF-κB, IL-6, TNF-α, MMP-3, MMP-13 | [108] |
| LPS-induced microglial cell line (BV2) | ↑ Nrf2, ↓ MAPK, COX-2 | [109] | |
| Hesperidin | chronic obstructive pulmonary disease model in mice | ↓ NF-κB, IL-6, IL-8 | [110] |
| As2O3-induced cardiotoxicity in mice | ↑ Nrf2, ↓ IL-6, ROS, TNF-α | [111] | |
| human neuroblastoma cell line (SH-SY5Y) | ↓ MAPK | [112] | |
| Naringenin | cardiomyocytes (H9C2) | ↑ Nrf2, ↓ IL-6, IL-1β, TNF-α, MPO | [113] |
| LPS-induced normal human bronchial epithelial cell line (NHBE) | ↓ NF-κB, IL-6, TNF-α, MPO, NO | [114] | |
| Flavanonols | |||
| Taxifolin | acute alcohol-induced liver injury in mice | ↓ NF-κB, IL-1β, IL-6, TNF-α | [115] |
| benzo[a]pyrene-induced lung injury in mice | ↑ Nrf2, ↓ NF-κB, TNF-α, COX-2 | [116] | |
| Iron-overload-induced hepatocellular injury in rats | ↓ MAPK, IL-1β, IL-6, TNF-α | [117] | |
| Flavan-3-ols | |||
| Catechin | Porphyromonas gingivalis-stimulated human monocytes (THP-1) | ↓ NF-κB, MAPK, pro-IL-1β, TLR2, TLR4, NLRP3 | [118] |
| LPS-induced murine macrophages (RAW 264.7) | ↓ NF-κB, MAPK, TNF-α, COX-2, iNOS | [119] | |
| Epicatechin | high-glucose-induced human monocytes (THP-1) | ↓ NF-κB, TNF-α | [120] |
| LPS-induced acute lung injury model in mice | ↓ MAPK, TNF-α, IL-6 | [121] | |
| hemoglobin toxicity in primary astrocytes from wild type and Nrf2−/− mice | ↑ Nrf2, ↓ ROS | [122] | |
| Isoflavones | |||
| Formononetin | human neuroblastoma cell line (SH-SY5Y) | ↑ Nrf2, ↓ MAPK, ROS | [123] |
| gastric ulcer model in rats | ↓ NF-κB, IL-6, IL-1β, TNF-α, MPO | [124] | |
| Genistein | homocysteine-induced inflammatory injury in endothelial cell (ECV-304) | ↓ NF-κB, IL-6 | [125] |
| liver granuloma and fibrosis model in mice | ↓ NF-κB, IL-1β, TNF-α, IL-4, IL-10 | [126] | |
| diabetic nephropathy model in rats | ↓ NF-κB, MAPK | [127] | |
| Chalcones | |||
| Isosalipurposide | oxidative injury in human hepatocarcinoma cell line (HepG2) | ↑ Nrf2 | [128] |
| Xanthohumol | LPS-induced acute lung injury in mice | ↑ Nrf2, ↓ NF-κB, IL-6, IL-1β, TNF-α | [129] |
| LPS-induced depressive-like symptoms in mice | ↑ Nrf2, ↓ NF-κB | [130] | |
| Flavonolignans | |||
| Silibinin | LPS-induced murine macrophages (RAW 264.7) | ↓ MAPK | [131] |
| human colorectal cell lines (SW480, HT-29, LoVo) | ↓ NF-κB, COX-2, MMP-9 | [132] | |
| neuroblastoma cell line (SH-SY5Y) | ↑ Nrf2 | [133] | |
| Anthocyanins | |||
| Cyanidin | palmitate-induced lipotoxicity in human colon adenoracinoma cell line (Caco-2) | ↑ Nrf2, ↓ NF-κB, IL-6, IL-8, COX-2 | [134] |
| human colon cancer cell lines (HCT116, HT29, SW620) | ↑ Nrf2, ↓ NF-κB | [135] | |
| human nucleus pulposus cells | ↑ Nrf2 | [136] | |
| pulmonary artery hypertension model in rats | ↓ MAPK, IL-6, TNF-α | [137] | |
| Delphinidin | murine embryonic stem cell lines (ES-E14TG2a, ES-R1) | ↓ NF-κB | [138] |
| H2O2-induced human retinal pigment epithelial cell line (ARPE-19) | ↑ Nrf2 | [139] | |
| Malvidin | osteoarthritis in rats, primary rat chondrocytes | ↓ NF-κB, TNF-α, IL-1β, IL-6, MMP-3, MMP-9, MMP-13 | [140] |
| sepsis-related acute liver injury model in mice | ↑ Nrf2, TNF-α, IL-1β, IL-6 | [141] | |
| Pelargonidin | LPS-induced murine macrophage cell line (J774) | ↓ NF-κB, NO | [142] |
| middle cerebral artery occlusion (MCAO) in rats | ↑ Nrf2, ↓ TNF-α, IL-6 | [143] | |
3.2. Flavonols
3.3. Flavanones
3.4. Flavanonols
3.5. Flavan-3-ols
3.6. Isoflavones
3.7. Chalcones
3.8. Flavonolignans
3.9. Anthocyanins
4. Lignans
5. Tannins
5.1. Gallotannins and Ellagitannins
5.2. Procyanidins
| Compounds | Model | Mode of Action | Reference |
|---|---|---|---|
| Lignans | |||
| Nordihydroguaiaretic acid | human SH-SY5Y neuroblastoma cells | ↓ ERK1/2 | [173] |
| I/R * model in rats | ↑ Nrf2 | [172] | |
| Tannins | |||
| Gallotannins and ellagitannins | |||
| Castalagin | osteoclasts differentiated from bone marrow-derived macrophages (BMM) | ↓ NF-κB, MAPK | [188] |
| Epicatechin gallate | cisplatin-induced nephrotoxicity in rats | ↓ MAPK, IL-6, TNF-α | [194] |
| cerebral edema in mice | ↓ NF-κB, TNF-α, IL-1β | [190] | |
| atherosclerosis model in mice | ↑ Nrf2, ↓ NF-κB, IL-6, TNF-α | [193] | |
| Epigallocatechin gallate | RANKL-induced osteoclast differentiation in macrophage cell line (RAW 264.7) | ↓ NF-κB, MAPK | [189] |
| CoCl2-induced murine microglial cell line (BV2) | ↑ Nrf2 ↓ NF-κB, IL-6, COX-2 | [191] | |
| acute lung injury model in mice | ↓ NF-κB, IL-6, TNF-α, IL-1β | [195] | |
| Pentagalloylglucose | UVB-induced human dermal fibroblasts | ↓ NF-κB, MAPK | [197] |
| AGE **-induced mouse mesangial cells (MES) | ↑ Nrf2, ↓ TNF-α, IL-1β | [196] | |
| Procyanidins | |||
| Procyanidin A1 | LPS-induced murine macrophage cell line (RAW 264.7) | ↑ Nrf2 ↓ NF-κB, MAPK, IL-6, TNF-α, NO | [198] |
| LPS-induced human umbilical vein endothelial cells (HUVECs) | ↓ NF-κB, IL-6, TNF-α, IL-1β | [200] | |
| Procyanidin B2 | LPS-induced human monocyte cell line (THP-1) | ↓ NF-κB, MAPK, COX-2 | [199] |
| acute kidney injury model in mice | ↑ Nrf2 | [201] | |
| Phenolic glycosides | |||
| Salicin | LPS-induced murine macrophages RAW 264.7 | ↓ NF-κB, MAPK, IL-6, TNF-α, IL-1β | [202] |
| rheumatoid arthritis fibroblast-like synoviocytes (RA-FLSs) | ↑ Nrf2, ↓ NF-κB, IL-1β, TNF-α, MMP-1, MMP-3 | [203] | |
| Phenolic acids and depsides | |||
| Caffeic acid | LPS endothelial cell line (YPEN-1) | ↓ NF-κB, COX-2 | [204] |
| LPS-induced primary bovine mammary epithelial cells | ↓ NF-κB, MAPK, IL-1β, IL-6, TNF-α, IL-8 | [205] | |
| t-BHP ***-induced HepG2 cells | ↑ Nrf2 | [206] | |
| Chlorogenic acid | porcine jejunal epithelial cell line (IPEC-J2) | ↓ NF-κB, TNF-α | [207] |
| endometriosis model in mice | ↑ Nrf2, ↓ NF-κB, IL-6, TNF-α, IL-1β | [208] | |
| DSS ****-induced ulcerative colitis in mice | ↓ MAPK, IL-6, TNF-α, IL-1β | [209] | |
| Cichoric acid | neuroinflammation model in mice | ↓ NF-κB, MAPK, COX-2, TNF-α, IL-1β | [210] |
| LPS-induced murine microglial cell line (BV2) and mice brain | ↑ Nrf2, ↓ NF-κB, MAPK | [211] | |
| p-Coumaric acid | doxorubicin-treated cardiomyoblast cell line (H9C2) | ↑ Nrf2 | [212] |
| human lens epithelial cell line (HLE SRA 01/04) | ↓ MAPK | [213] | |
| Curcumin | LPS-induced murine microglial cell line (BV2) | ↓ NF-κB, IL-6, IL-1β | [214] |
| H2O2-induced murine macrophage cell line (RAW 264.7) | ↓ Nrf2 | [215] | |
| influenza A virus infection in lung cancer cell line (A549) | ↑ Nrf2,↓ NF-κB, MAPK, MMP-2, MMP-9 | [216] | |
| Ellagic acid | CCl4-induced pancreas damage in rats | ↑ Nrf2, ↓ NF-κB, TNF-α | [217] |
| rotenone-induced neurotoxicity in mice, neuronal cell lines (MN9D, BV2, C6) | ↑ Nrf2 | [218] | |
| TNF-α/IFN-γ-induced keratinocyte cell line (HaCaT) | ↓ MAPK, TNF-α, IL-6 | [219] | |
| Ferulic acid | LPS-induced bovine endometrial epithelial cell line (BEEC) | ↓ NF-κB, MAPK, IL-6, TNF-α, IL-1β, IL-8 | [220] |
| LPS-induced primary bovine mammary epithelial cells | ↑ Nrf2, ↓ NF-κB | [221] | |
| acute lung injury model in mice | ↓ NF-κB, IL-6, TNF-α, IL-1β | [222] | |
| Gallic acid | elastase-induced emphysema in rats | ↑ Nrf2, ↓ NF-кB | [223] |
| ethanol-induced gastric ulcer in rats | ↑ Nrf2 ↓ IL-1β, IL-6 | [224] | |
| diabetic nephropathy model in rats | ↓MAPK, IL-1β, IL-6, TNF-α | [225] | |
| Rosmarinic acid | human hepatoma cell line (HepG2) | ↓ NF-κB, MMP-2, MMP-9 | [226] |
| acute liver injury model in mice | ↑ Nrf2, ↓ NF-κB, MAPK | [227] | |
| spinal cord injury model in rats | ↑ Nrf2, ↓ NF-κB, IL-6, TNF-α, IL-1β | [228] | |
| Phenylpropanoids | |||
| Echinacoside | IL-1β -induced rat chondrocytes | ↑ Nrf2 | [229] |
| Verbascoside | LPS-induced murine microglial cell line (BV2) | ↓ NF-κB, IL-6, IL-1β | [230] |
| Stilbenoids | |||
| Resveratrol | porcine jejunal epithelial cell line (IPEC-J2) | ↑ Nrf2 | [231] |
| fibrosis model in mice | ↓ NF-κB | [232] | |
| IL-1β-stimulated rat synovial cells (RSC-364) | ↓ MAPK | [233] | |
6. Phenolic Glycosides
7. Phenolic Acids and Depsides
8. Phenylpropanoids
9. Stilbenoids
10. Other Compounds Related to Chronic Inflammation
11. Plant Materials and Network Pharmacology
12. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- GBD 2017 Causes of Death Collaborators. Global, regional, and national age-sex-specific mortality for 282 causes of death in 195 countries and territories, 1980–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1736–1788. [Google Scholar] [CrossRef]
- Serhan, C.N.; Ward, P.A.; Gilroy, D.W. (Eds.) Fundamentals of Inflammation; Cambridge University Press: New York, NY, USA, 2010. [Google Scholar]
- Guo, H.; Callaway, J.B.; Ting, J.P.Y. Inflammasomes: Mechanism of action, role in disease, and therapeutics. Nat. Med. 2015, 21, 677–687. [Google Scholar] [CrossRef]
- Mogensen, T.H. Pathogen recognition and inflammatory signaling in innate immune defenses. Clin. Microbiol. Rev. 2009, 22, 240–273. [Google Scholar] [CrossRef]
- Zheng, D.; Liwinski, T.; Elinav, E. Inflammasome activation and regulation: Toward a better understanding of complex mechanisms. Cell Discov. 2020, 6, 36. [Google Scholar] [CrossRef]
- Mariathasan, S.; Newton, K.; Monack, D.M.; Vucic, D.; French, D.M.; Lee, W.P.; Roose-Girma, M.; Erickson, S.; Dixit, V.M. Differential activation of the inflammasome by caspase-1 adaptors ASC and Ipaf. Nature 2004, 430, 213–218. [Google Scholar] [CrossRef]
- Furman, D.; Campisi, J.; Verdin, E.; Carrera-Bastos, P.; Targ, S.; Franceschi, C.; Ferrucci, L.; Gilroy, D.W.; Fasano, A.; Miller, G.W.; et al. Chronic inflammation in the etiology of disease across the life span. Nat. Med. 2019, 25, 1822–1832. [Google Scholar] [CrossRef]
- Serhan, C.N.; de la Rosa, X.; Jouvene, C. Novel mediators and mechanisms in the resolution of infectious inflammation: Evidence for vagus regulation. J. Intern. Med. 2019, 286, 240–258. [Google Scholar] [CrossRef] [PubMed]
- Pirault, J.; Bäck, M. Lipoxin and resolvin receptors transducing the resolution of inflammation in cardiovascular disease. Front. Pharmacol. 2018, 9, 1273. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.Y.; Nuñez, G. Sterile inflammation: Sensing and reacting to damage. Nat. Rev. Immunol. 2010, 10, 826–837. [Google Scholar] [CrossRef] [PubMed]
- Sameer, A.S.; Nissar, S. Toll-like receptors (TLRs): Structure, functions, signaling, and role of their polymorphisms in colorectal cancer susceptibility. Biomed. Res. Int. 2021, 2021, 1157023. [Google Scholar] [CrossRef] [PubMed]
- Borsoi, F.T.; Neri-Numa, I.A.; de Oliveira, W.Q.; de Araújo, F.F.; Pastore, G.M. Dietary polyphenols and their relationship to the modulation of non-communicable chronic diseases and epigenetic mechanisms: A mini-review. Food Chem. Mol. Sci. 2023, 6, 100155. [Google Scholar] [CrossRef] [PubMed]
- Ramaroson, M.-L.; Koutouan, C.; Helesbeux, J.-J.; Le Clerc, V.; Hamama, L.; Geoffriau, E.; Briard, M. Role of phenylpropanoids and flavonoids in plant resistance to pests and diseases. Molecules 2022, 27, 8371. [Google Scholar] [CrossRef] [PubMed]
- Roh, J.S.; Sohn, D.H. Damage-associated molecular patterns in inflammatory diseases. Immune Netw. 2018, 18, e27. [Google Scholar] [CrossRef] [PubMed]
- Tang, D.; Kang, R.; Coyne, C.B.; Zeh, H.J.; Lotze, M.T. PAMPs and DAMPs: Signal 0s that spur autophagy and immunity. Immunol. Rev. 2012, 249, 158–175. [Google Scholar] [CrossRef]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.C. NF-κB signaling in inflammation. Signal Transduct. Target. Ther. 2017, 2, 17023. [Google Scholar] [CrossRef]
- Kawasaki, T.; Kawai, T. Toll-like receptor signaling pathways. Front. Immunol. 2014, 5, 461. [Google Scholar] [CrossRef]
- Stewart, C.R.; Stuart, L.M.; Wilkinson, K.; van Gils, J.M.; Deng, J.; Halle, A.; Rayner, K.J.; Boyer, L.; Zhong, R.; Frazier, W.A.; et al. CD36 ligands promote sterile inflammation through assembly of a Toll-like receptor 4 and 6 heterodimer. Nat. Immunol. 2010, 11, 155–161. [Google Scholar] [CrossRef]
- Kagan, J.C.; Medzhitov, R. Phosphoinositide-mediated adaptor recruitment controls Toll-like receptor signaling. Cell 2006, 125, 943–955. [Google Scholar] [CrossRef]
- Zanoni, I.; Ostuni, R.; Marek, L.R.; Barresi, S.; Barbalat, R.; Barton, G.M.; Granucci, F.; Kagan, J.C. CD14 controls the LPS-induced endocytosis of Toll-like receptor 4. Cell 2011, 147, 868–880. [Google Scholar] [CrossRef]
- Shu, H.; Peng, Y.; Hang, W.; Nie, J.; Zhou, N.; Wang, D.W. The role of CD36 in cardiovascular disease. Cardiovasc. Res. 2020, 118, 115–129. [Google Scholar] [CrossRef]
- Ullah, M.O.; Sweet, M.J.; Mansell, A.; Kellie, S.; Kobe, B. TRIF-dependent TLR signaling, its functions in host defense and inflammation, and its potential as a therapeutic target. J. Leukoc. Biol. 2016, 100, 27–45. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T.; Asano, N.; Kudo, M.; Strober, W. Nucleotide-binding oligomerization domain 1 and gastrointestinal disorders. Proc. Jpn. Acad. Ser. B Phys. Biol. Sci. 2017, 93, 578–599. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, N.; Lill, J.R.; Phung, Q.; Jiang, Z.; Bakalarski, C.; de Mazière, A.; Klumperman, J.; Schlatter, M.; Delamarre, L.; Mellman, I. Endosomes are specialized platforms for bacterial sensing and NOD2 signalling. Nature 2014, 509, 240–244. [Google Scholar] [CrossRef] [PubMed]
- Burberry, A.; Zeng, M.Y.; Ding, L.; Wicks, I.; Inohara, N.; Morrison, S.J.; Núñez, G. Infection mobilizes hematopoietic stem cells through cooperative NOD-like receptor and Toll-like receptor signaling. Cell Host Microbe 2014, 15, 779–791. [Google Scholar] [CrossRef]
- Caruso, R.; Warner, N.; Inohara, N.; Núñez, G. NOD1 and NOD2: Signaling, host defense, and inflammatory disease. Immunity 2014, 41, 898–908. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, H.; Su, Y.; Zhang, B. Application and prospect of targeting innate immune sensors in the treatment of autoimmune diseases. Cell Biosci. 2022, 12, 68. [Google Scholar] [CrossRef]
- Ogura, Y.; Inohara, N.; Benito, A.; Chen, F.F.; Yamaoka, S.; Nunez, G. Nod2, a Nod1/Apaf-1 family member that is restricted to monocytes and activates NF-kappaB. J. Biol. Chem. 2001, 276, 4812–4818. [Google Scholar] [CrossRef]
- Park, J.H.; Kim, Y.G.; Shaw, M.; Kanneganti, T.D.; Fujimoto, Y.; Fukase, K.; Inohara, N.; Núñez, G. Nod1/RICK and TLR signaling regulate chemokine and antimicrobial innate immune responses in mesothelial cells. J. Immunol. 2007, 179, 514–521. [Google Scholar] [CrossRef]
- Ogura, Y.; Lala, S.; Xin, W.; Smith, E.; Dowds, T.A.; Chen, F.F.; Zimmermann, E.; Tretiakova, M.; Cho, J.H.; Hart, J.; et al. Expression of NOD2 in Paneth cells: A possible link to Crohn’s ileitis. Gut 2003, 52, 1591–1597. [Google Scholar] [CrossRef]
- Su, X.; Liu, B.; Wang, S.; Wang, Y.; Zhang, Z.; Zhou, H.; Li, F. NLRP3 inflammasome: A potential therapeutic target to minimize renal ischemia/reperfusion injury during transplantation. Transpl. Immunol. 2022, 75, 101718. [Google Scholar] [CrossRef]
- Sun, S.C. Non-canonical NF-κB signaling pathway. Cell Res. 2011, 21, 71–85. [Google Scholar] [CrossRef] [PubMed]
- Karin, M.; Delhase, M. The I kappa B kinase (IKK) and NF-kappa B: Key elements of proinflammatory signalling. Semin. Immunol. 2000, 12, 85–98. [Google Scholar] [CrossRef]
- Israël, A. The IKK complex, a central regulator of NF-kappaB activation. Cold Spring Harb. Perspect. Biol. 2010, 2, a000158. [Google Scholar] [CrossRef]
- Dutta, J.; Fan, Y.; Gupta, N.; Fan, G.; Gélinas, C. Current insights into the regulation of programmed cell death by NF-kappaB. Oncogene 2006, 25, 6800–6816. [Google Scholar] [CrossRef] [PubMed]
- Tucureanu, M.M.; Rebleanu, D.; Constantinescu, C.A.; Deleanu, M.; Voicu, G.; Butoi, E.; Calin, M.; Manduteanu, I. Lipopolysaccharide-induced inflammation in monocytes/macrophages is blocked by liposomal delivery of G(i)-protein inhibitor. Int. J. Nanomed. 2018, 13, 63–76. [Google Scholar] [CrossRef]
- Wang, N.; Liang, H.; Zen, K. Molecular mechanisms that influence the macrophage M1-M2 polarization balance. Front. Immunol. 2014, 5, 614. [Google Scholar] [CrossRef] [PubMed]
- Canning, P.; Sorrell, F.J.; Bullock, A.N. Structural basis of Keap1 interactions with Nrf2. Free Radic. Biol. Med. 2015, 88, 101–107. [Google Scholar] [CrossRef]
- He, F.; Ru, X.; Wen, T. NRF2, a transcription factor for stress response and beyond. Int. J. Mol. Sci. 2020, 21, 4777. [Google Scholar] [CrossRef]
- Robledinos-Antón, N.; Fernández-Ginés, R.; Manda, G.; Cuadrado, A. Activators and inhibitors of Nrf2: A review of their potential for clinical development. Oxidative Med. Cell. Longev. 2019, 2019, 9372182. [Google Scholar] [CrossRef]
- Sturm, C.; Wagner, A.E. Brassica-derivedplant bioactives as modulators of chemopreventive and inflammatory signaling pathways. Int. J. Mol. Sci. 2017, 18, 1890. [Google Scholar] [CrossRef]
- Li, W.; Yu, S.; Liu, T.; Kim, J.-H.; Blank, V.; Li, H.; Kong, A.N.T. Heterodimerization with small Maf proteins enhances nuclear retention of Nrf2 via masking the NESzip motif. Biochim. Biophys. Acta (BBA) Mol. Cell Res. 2008, 1783, 1847–1856. [Google Scholar] [CrossRef]
- Mackenzie, P.I.; Hu, D.G.; Gardner-Stephen, D.A. The regulation of UDP-glucuronosyltransferase genes by tissue-specific and ligand-activated transcription factors. Drug Metab. Rev. 2010, 42, 99–109. [Google Scholar] [CrossRef] [PubMed]
- Liby, K.T.; Sporn, M.B. Synthetic oleanane triterpenoids: Multifunctional drugs with a broad range of applications for prevention and treatment of chronic disease. Pharmacol. Rev. 2012, 64, 972–1003. [Google Scholar] [CrossRef] [PubMed]
- Reisman, S.A.; Aleksunes, L.M.; Klaassen, C.D. Oleanolic acid activates Nrf2 and protects from acetaminophen hepatotoxicity via Nrf2-dependent and Nrf2-independent processes. Biochem. Pharmacol. 2009, 77, 1273–1282. [Google Scholar] [CrossRef]
- Olszewska, M.A.; Granica, S.; Kolodziejczyk-Czepas, J.; Magiera, A.; Czerwińska, M.E.; Nowak, P.; Rutkowska, M.; Wasiński, P.; Owczarek, A. Variability of sinapic acid derivatives during germination and their contribution to antioxidant and anti-inflammatory effects of broccoli sprouts on human plasma and human peripheral blood mononuclear cells. Food Funct. 2020, 11, 7231–7244. [Google Scholar] [CrossRef]
- Cargnello, M.; Roux, P.P. Activation and function of the MAPKs and their substrates, the MAPK-activated protein kinases. Microbiol. Mol. Biol. Rev. 2011, 75, 50–83. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.; Pei, J.; Wang, A.; Shuai, W.; Feng, L.; Bu, F.; Zhu, Y.; Zhang, L.; Wang, G.; Ouyang, L. Development of small molecule extracellular signal-regulated kinases (ERKs) inhibitors for cancer therapy. Acta Pharm. Sin. B 2022, 12, 2171–2192. [Google Scholar] [CrossRef]
- Kassouf, T.; Sumara, G. Impact of conventional and atypical MAPKs on the development of metabolic diseases. Biomolecules 2020, 10, 1256. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.K.; Choi, E.J. Pathological roles of MAPK signaling pathways in human diseases. Biochim. Biophys. Acta 2010, 1802, 396–405. [Google Scholar] [CrossRef]
- Vial, E.; Pouysségur, J. Regulation of Tumor Cell Motility by ERK Mitogen-Activated Protein Kinases. Ann. N. Y. Acad. Sci. 2004, 1030, 208–218. [Google Scholar] [CrossRef] [PubMed]
- Perry, R.J.; Camporez, J.G.; Kursawe, R.; Titchenell, P.M.; Zhang, D.; Perry, C.J.; Jurczak, M.J.; Abudukadier, A.; Han, M.S.; Zhang, X.M.; et al. Hepatic acetyl CoA links adipose tissue inflammation to hepatic insulin resistance and type 2 diabetes. Cell 2015, 160, 745–758. [Google Scholar] [CrossRef]
- Spencer, J.P. The interactions of flavonoids within neuronal signalling pathways. Genes Nutr. 2007, 2, 257–273. [Google Scholar] [CrossRef]
- Behl, T.; Upadhyay, T.; Singh, S.; Chigurupati, S.; Alsubayiel, A.M.; Mani, V.; Vargas-De-La-Cruz, C.; Uivarosan, D.; Bustea, C.; Sava, C.; et al. Polyphenols targeting MAPK mediated oxidative stress and inflammation in rheumatoid arthritis. Molecules 2021, 26, 6570. [Google Scholar] [CrossRef]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, e47. [Google Scholar] [CrossRef]
- Hostetler, G.L.; Ralston, R.A.; Schwartz, S.J. Flavones: Food sources, bioavailability, metabolism, and bioactivity. Adv. Nutr. 2017, 8, 423–435. [Google Scholar] [CrossRef]
- Yang, H.-W.; Kim, H.-J.; Park, J.-H.; Shin, J.-M.; Lee, H.-M. Apigenin alleviates TGF-β1-induced nasal mucosa remodeling by inhibiting MAPK/NF-kB signaling pathways in chronic rhinosinusitis. PLoS ONE 2018, 13, e0201595. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Wang, J.; Zhao, G.; Lin, M.; Lang, Y.; Zhang, D.; Feng, D.; Tu, C. Apigenin protects human melanocytes against oxidative damage by activation of the Nrf2 pathway. Cell Stress Chaperones 2020, 25, 277–285. [Google Scholar] [CrossRef]
- Chen, P.; Huo, X.; Liu, W.; Li, K.; Sun, Z.; Tian, J. Apigenin exhibits anti-inflammatory effects in LPS-stimulated BV2 microglia through activating GSK3β/Nrf2 signaling pathway. Immunopharmacol. Immunotoxicol. 2020, 42, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Liu, Y.T.; Xiao, L.; Zhu, L.; Wang, Q.; Yan, T. Anti-inflammatory effects of apigenin in lipopolysaccharide-induced inflammatory in acute lung injury by suppressing COX-2 and NF-kB pathway. Inflammation 2014, 37, 2085–2090. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Ci, X.; Wen, Z.; Peng, L. Diosmetin alleviates lipopolysaccharide-induced acute lung injury through activating the Nrf2 pathway and inhibiting the NLRP3 inflammasome. Biomol. Ther. 2018, 26, 157–166. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Yu, T.; Liu, J.; Gu, L. Vitexin attenuates lipopolysaccharide-induced acute lung injury by controlling the Nrf2 pathway. PLoS ONE 2018, 13, e0196405. [Google Scholar] [CrossRef]
- Chen, Y.; Yang, J.; Huang, Z.; Yin, B.; Umar, T.; Yang, C.; Zhang, X.; Jing, H.; Guo, S.; Guo, M.; et al. Vitexin mitigates Staphylococcus aureus-induced mastitis via regulation of ROS/ER stress/NF-κB/MAPK pathway. Oxidative Med. Cell. Longev. 2022, 2022, 7977433. [Google Scholar] [CrossRef]
- Guo, Y.F.; Xu, N.N.; Sun, W.; Zhao, Y.; Li, C.Y.; Guo, M.Y. Luteolin reduces inflammation in Staphylococcus aureus-induced mastitis by inhibiting NF-kB activation and MMPs expression. Oncotarget 2017, 8, 28481–28493. [Google Scholar] [CrossRef]
- Savineau, J.P.; Marthan, R. Diosmin-induced increase in sensitivity to Ca2+ of the smooth muscle contractile apparatus in the rat isolated femoral vein. Br. J. Pharmacol. 1994, 111, 978–980. [Google Scholar] [CrossRef]
- Feldo, M.; Wójciak, M.; Ziemlewska, A.; Dresler, S.; Sowa, I. Modulatory effect of diosmin and diosmetin on metalloproteinase activity and inflammatory mediators in human skin fibroblasts treated with lipopolysaccharide. Molecules 2022, 27, 4264. [Google Scholar] [CrossRef] [PubMed]
- Kirienko, A.; Radak, D. Clinical acceptability study of once-daily versus twice-daily micronized purified flavonoid fraction in patients with symptomatic chronic venous disease: A randomized controlled trial. Int. Angiol. 2016, 35, 399–405. [Google Scholar] [PubMed]
- Shelygin, Y.; Krivokapic, Z.; Frolov, S.A.; Kostarev, I.V.; Astashov, V.L.; Vasiliev, S.V.; Lakhin, A.V.; Rodoman, G.V.; Soloviev, A.O.; Stoyko, Y.M.; et al. Clinical acceptability study of micronized purified flavonoid fraction 1000 mg tablets versus 500 mg tablets in patients suffering acute hemorrhoidal disease. Curr. Med. Res. Opin. 2016, 32, 1821–1826. [Google Scholar] [CrossRef]
- Cazaubon, M.; Benigni, J.P.; Steinbruch, M.; Jabbour, V.; Gouhier-Kodas, C. Is there a difference in the clinical efficacy of diosmin and micronized purified flavonoid fraction for the treatment of chronic venous disorders? Review of available evidence. Vasc. Health Risk Manag. 2021, 17, 591–600. [Google Scholar] [CrossRef]
- Freag, M.S.; Elnaggar, Y.S.; Abdallah, O.Y. Lyophilized phytosomal nanocarriers as platforms for enhanced diosmin delivery: Optimization and ex vivo permeation. Int. J. Nanomed. 2013, 8, 2385–2397. [Google Scholar] [CrossRef]
- Silvestro, L.; Tarcomnicu, I.; Dulea, C.; Attili, N.R.; Ciuca, V.; Peru, D.; Rizea Savu, S. Confirmation of diosmetin 3-O-glucuronide as major metabolite of diosmin in humans, using micro-liquid-chromatography-mass spectrometry and ion mobility mass spectrometry. Anal. Bioanal. Chem. 2013, 405, 8295–8310. [Google Scholar] [CrossRef] [PubMed]
- Olędzka, A.; Cichocka, K.; Woliński, K.; Melzig, M.F.; Czerwińska, M.E. Potentially bio-accessible metabolites from an extract of Cornus mas fruit after gastrointestinal digestion in vitro and gut microbiota ex vivo treatment. Nutrients 2022, 14, 2287. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Sung, J.; Kim, Y.; Jeong, H.S.; Lee, J. Inhibitory effect of diosmetin on inflammation and lipolysis in coculture of adipocytes and macrophages. J. Food Biochem. 2020, 44, e13261. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Jiang, Y.; Lu, D. Diosmetin suppresses neuronal apoptosis and inflammation by modulating the phosphoinositide 3-kinase (PI3K)/AKT/nuclear factor-κB (NF-κB) signaling pathway in a rat model of pneumococcal meningitis. Med. Sci. Monit. 2019, 25, 2238–2245. [Google Scholar] [CrossRef]
- Ahmed, S.; Mundhe, N.; Borgohain, M.; Chowdhury, L.; Kwatra, M.; Bolshette, N.; Ahmed, A.; Lahkar, M. Diosmin modulates the NF-kB signal transduction pathways and downregulation of various oxidative stress markers in alloxan-induced diabetic nephropathy. Inflammation 2016, 39, 1783–1797. [Google Scholar] [CrossRef] [PubMed]
- AlAsmari, A.F.; Alharbi, M.; Alqahtani, F.; Alasmari, F.; AlSwayyed, M.; Alzarea, S.I.; Al-Alallah, I.A.; Alghamdi, A.; Hakami, H.M.; Alyousef, M.K.; et al. Diosmin alleviates doxorubicin-induced liver injury via modulation of oxidative stress-mediated hepatic inflammation and apoptosis via NfkB and MAPK pathway: A preclinical study. Antioxidants 2021, 10, 1998. [Google Scholar] [CrossRef]
- Yuan, J.; Che, S.; Zhang, L.; Ruan, Z. Reparative effects of ethanol-induced intestinal barrier injury by flavonoid luteolin via MAPK/NF-κB/MLCK and Nrf2 signaling pathways. J. Agric. Food Chem. 2021, 69, 4101–4110. [Google Scholar] [CrossRef]
- Li, B.; Du, P.; Du, Y.; Zhao, D.; Cai, Y.; Yang, Q.; Guo, Z. Luteolin alleviates inflammation and modulates gut microbiota in ulcerative colitis rats. Life Sci. 2021, 269, 119008. [Google Scholar] [CrossRef]
- Ganesan, K.; Ramkumar, K.M.; Xu, B. Vitexin restores pancreatic β-cell function and insulin signaling through Nrf2 and NF-κB signaling pathways. Eur. J. Pharmacol. 2020, 888, 173606. [Google Scholar] [CrossRef]
- Zou, Z.L.; Sun, M.H.; Yin, W.F.; Yang, L.; Kong, L.Y. Avicularin suppresses cartilage extracellular matrix degradation and inflammation via TRAF6/MAPK activation. Phytomedicine 2021, 91, 153657. [Google Scholar] [CrossRef]
- Sun, K.; Luo, J.; Jing, X.; Xiang, W.; Guo, J.; Yao, X.; Liang, S.; Guo, F.; Xu, T. Hyperoside ameliorates the progression of osteoarthritis: An in vitro and in vivo study. Phytomedicine 2021, 80, 153387. [Google Scholar] [CrossRef]
- Xie, T.; Yuan, J.; Mei, L.; Li, P.; Pan, R. Hyperoside ameliorates TNF-α-induced inflammation, ECM degradation and ER stress-mediated apoptosis via the SIRT1/NF-κB and Nrf2/ARE signaling pathways in vitro. Mol. Med. Rep. 2022, 26, 260. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Sun, Y.; Guo, X.; Zhong, C.; Li, Z. Hyperoside inhibited the migration and invasion of lung cancer cells through the upregulation of PI3K/AKT and p38 MAPK pathways. Int. J. Clin. Exp. Pathol. 2017, 10, 9382–9390. [Google Scholar] [PubMed]
- Ye, P.; Yang, X.-L.; Chen, X.; Shi, C. Hyperoside attenuates OVA-induced allergic airway inflammation by activating Nrf2. Int. Immunopharmacol. 2017, 44, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.; Tang, B.; Tang, K.; Dong, X.; Deng, J.; Liao, L.; Liao, Z.; Yang, H.; He, S. Isoquercitrin inhibits the progression of liver cancer in vivo and in vitro via the MAPK signalling pathway. Oncol. Rep. 2014, 31, 2377–2384. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; Jiang, Y.; Zhang, X.; Chen, X.; Liu, Z.; Tian, X. Isoquercetin ameliorates myocardial infarction through anti-inflammation and anti-apoptosis factor and regulating TLR4-NF-κB signal pathway. Mol. Med. Rep. 2018, 17, 6675–6680. [Google Scholar] [CrossRef]
- Dai, Y.; Zhang, H.; Zhang, J.; Yan, M. Isoquercetin attenuates oxidative stress and neuronal apoptosis after ischemia/reperfusion injury via Nrf2-mediated inhibition of the NOX4/ROS/NF-κB pathway. Chem. Biol. Interact. 2018, 284, 32–40. [Google Scholar] [CrossRef]
- Ren, X.; Han, L.; Li, Y.; Zhao, H.; Zhang, Z.; Zhuang, Y.; Zhong, M.; Wang, Q.; Ma, W.; Wang, Y. Isorhamnetin attenuates TNF-α-induced inflammation, proliferation, and migration in human bronchial epithelial cells via MAPK and NF-κB pathways. Anat. Rec. 2021, 304, 901–913. [Google Scholar] [CrossRef]
- Xu, Y.; Li, J.; Lin, Z.; Liang, W.; Qin, L.; Ding, J.; Chen, S.; Zhou, L. Isorhamnetin alleviates airway inflammation by regulating the Nrf2/Keap1 pathway in a mouse model of COPD. Front. Pharmacol. 2022, 13, 860362. [Google Scholar] [CrossRef]
- Li, Y.; Chi, G.; Shen, B.; Tian, Y.; Feng, H. Isorhamnetin ameliorates LPS-induced inflammatory response through downregulation of NF-κB signaling. Inflammation 2016, 39, 1291–1301. [Google Scholar] [CrossRef]
- Tang, X.L.; Liu, J.X.; Dong, W.; Li, P.; Li, L.; Hou, J.C.; Zheng, Y.Q.; Lin, C.R.; Ren, J.G. Protective effect of kaempferol on LPS plus ATP-induced inflammatory response in cardiac fibroblasts. Inflammation 2015, 38, 94–101. [Google Scholar] [CrossRef]
- Yang, C.; Yang, W.; He, Z.; He, H.; Yang, X.; Lu, Y.; Li, H. Kaempferol improves lung ischemia-reperfusion injury via antiinflammation and antioxidative stress regulated by SIRT1/HMGB1/NF-κB axis. Front. Pharmacol. 2019, 10, 1635. [Google Scholar] [CrossRef]
- Alshehri, A.S.; El-Kott, A.F.; El-Kenawy, A.E.; Zaki, M.S.A.; Morsy, K.; Ghanem, R.A.; Salem, E.T.; Ebealy, E.R.; Khalifa, H.S.; Altyar, A.E.; et al. The ameliorative effect of kaempferol against CdCl2-mediated renal damage entails activation of Nrf2 and inhibition of NF-kB. Environ. Sci. Pollut. Res. Int. 2022, 29, 57591–57602. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Yao, X.; Sun, B.; Jiang, W.; Liao, C.; Dai, X.; Chen, Y.; Chen, J.; Ding, R. Pretreatment with kaempferol attenuates microglia-mediate neuroinflammation by inhibiting MAPKs-NF-κB signaling pathway and pyroptosis after secondary spinal cord injury. Free Radic. Biol. Med. 2021, 168, 142–154. [Google Scholar] [CrossRef] [PubMed]
- Shao, Z.; Wang, B.; Shi, Y.; Xie, C.; Huang, C.; Chen, B.; Zhang, H.; Zeng, G.; Liang, H.; Wu, Y.; et al. Senolytic agent quercetin ameliorates intervertebral disc degeneration via the Nrf2/NF-κB axis. Osteoarthr. Cartil. 2021, 29, 413–422. [Google Scholar] [CrossRef]
- Indra, M.R.; Karyono, S.; Ratnawati, R.; Malik, S.G. Quercetin suppresses inflammation by reducing ERK1/2 phosphorylation and NF kappa B activation in Leptin-induced Human Umbilical Vein Endothelial Cells (HUVECs). BMC Res. Notes 2013, 6, 275. [Google Scholar] [CrossRef]
- Meng, L.Q.; Yang, F.Y.; Wang, M.S.; Shi, B.K.; Chen, D.X.; Chen, D.; Zhou, Q.; He, Q.B.; Ma, L.X.; Cheng, W.L.; et al. Quercetin protects against chronic prostatitis in rat model through NF-κB and MAPK signaling pathways. Prostate 2018, 78, 790–800. [Google Scholar] [CrossRef]
- Sharma, A.; Parikh, M.; Shah, H.; Gandhi, T. Modulation of Nrf2 by quercetin in doxorubicin-treated rats. Heliyon 2020, 6, e03803. [Google Scholar] [CrossRef] [PubMed]
- Chekalina, N.; Burmak, Y.; Petrov, Y.; Borisova, Z.; Manusha, Y.; Kazakov, Y.; Kaidashev, I. Quercetin reduces the transcriptional activity of NF-kB in stable coronary artery disease. Indian Heart J. 2018, 70, 593–597. [Google Scholar] [CrossRef]
- Tian, C.; Liu, X.; Chang, Y.; Wang, R.; Yang, M.; Liu, M. Rutin prevents inflammation induced by lipopolysaccharide in RAW 264.7 cells via conquering the TLR4-MyD88-TRAF6-NF-κB signalling pathway. J. Pharm. Pharmacol. 2021, 73, 110–117. [Google Scholar] [CrossRef]
- Sthijns, M.M.J.P.E.; Schiffers, P.M.; Janssen, G.M.; Lemmens, K.J.A.; Ides, B.; Vangrieken, P.; Bouwman, F.G.; Mariman, E.C.; Pader, I.; Arnér, E.S.J.; et al. Rutin protects against H2O2-triggered impaired relaxation of placental arterioles and induces Nrf2-mediated adaptation in Human Umbilical Vein Endothelial Cells exposed to oxidative stress. Biochim. Biophys. Acta Gen. Subj. 2017, 1861, 1177–1189. [Google Scholar] [CrossRef]
- Yoo, H.; Ku, S.K.; Baek, Y.D.; Bae, J.S. Anti-inflammatory effects of rutin on HMGB1-induced inflammatory responses in vitro and in vivo. Inflamm. Res. 2014, 63, 197–206. [Google Scholar] [CrossRef]
- Yeh, C.H.; Yang, J.J.; Yang, M.L.; Li, Y.C.; Kuan, Y.H. Rutin decreases lipopolysaccharide-induced acute lung injury via inhibition of oxidative stress and the MAPK-NF-κB pathway. Free Radic. Biol. Med. 2014, 69, 249–257. [Google Scholar] [CrossRef]
- Sharma, A.; Tirpude, N.V.; Kumari, M.; Padwad, Y. Rutin prevents inflammation-associated colon damage via inhibiting the p38/MAPKAPK2 and PI3K/Akt/GSK3β/NF-κB signalling axes and enhancing splenic Tregs in DSS-induced murine chronic colitis. Food Funct. 2021, 12, 8492–8506. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Zhang, Z.; Zhang, L.; Yao, X.; Zhong, X.; Cheng, G.; Wang, L.; Wan, Q. Spiraeoside protects human cardiomyocytes against high glucose-induced injury, oxidative stress, and apoptosis by activation of PI3K/Akt/Nrf2 pathway. J. Biochem. Mol. Toxicol. 2020, 34, e22548. [Google Scholar] [CrossRef]
- Velagapudi, R.; El-Bakoush, A.; Olajide, O.A. Activation of Nrf2 pathway contributes to neuroprotection by the dietary flavonoid tiliroside. Mol. Neurobiol. 2018, 55, 8103–8123. [Google Scholar] [CrossRef]
- Li, K.; Xiao, Y.; Wang, Z.; Fu, F.; Shao, S.; Song, F.; Zhao, J.; Lin, X.; Liu, Q.; Xu, J. Tiliroside is a new potential therapeutic drug for osteoporosis in mice. J. Cell. Physiol. 2019, 234, 16263–16274. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, Y.; Chen, Y.; Zhou, B.; Shan, X.; Yang, G. Eriodictyol inhibits IL-1β-induced inflammatory response in human osteoarthritis chondrocytes. Biomed. Pharmacother. 2018, 107, 1128–1134. [Google Scholar] [CrossRef]
- He, P.; Yan, S.; Wen, X.; Zhang, S.; Liu, Z.; Liu, X.; Xiao, C. Eriodictyol alleviates lipopolysaccharide-triggered oxidative stress and synaptic dysfunctions in BV-2 microglial cells and mouse brain. J. Cell. Biochem. 2019, 120, 14756–14770. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; He, N.; Xing, H.; Sun, Y.; Ding, J.; Liu, L. Function of hesperidin alleviating inflammation and oxidative stress responses in COPD mice might be related to SIRT1/PGC-1α/NF-κB signaling axis. J. Recept. Signal Transduct. Res. 2020, 40, 388–394. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.; Li, J.; Liu, P.; Si, M.; Jin, Y.; Wang, H.; Ma, D.; Chu, L. Based on activation of p62-Keap1-Nrf2 pathway, hesperidin protects arsenic-trioxide-induced cardiotoxicity in mice. Front. Pharmacol. 2021, 12, 758670. [Google Scholar] [CrossRef] [PubMed]
- Lim, C.; Zhen, A.X.; Ok, S.; Fernando, P.D.S.M.; Herath, H.M.U.L.; Piao, M.J.; Kang, K.A.; Hyun, J.W. Hesperidin protects SH-SY5Y neuronal cells against high glucose-induced apoptosis via regulation of MAPK signaling. Antioxidants 2022, 11, 1707. [Google Scholar] [CrossRef]
- Xu, S.; Wu, B.; Zhong, B.; Lin, L.; Ding, Y.; Jin, X.; Huang, Z.; Lin, M.; Wu, H.; Xu, D. Naringenin alleviates myocardial ischemia/reperfusion injury by regulating the nuclear factor-erythroid factor 2-related factor 2 (Nrf2)/System xc-/glutathione peroxidase 4 (GPX4) axis to inhibit ferroptosis. Bioengineered 2021, 12, 10924–10934. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.H.; Ma, C.H.; Yue, Z.Q.; Yao, X.; Mao, C.M. Protective effect of naringenin against lipopolysaccharide-induced injury in normal human bronchial epithelium via suppression of MAPK signaling. Inflammation 2015, 38, 195–204. [Google Scholar] [CrossRef]
- Ding, C.; Zhao, Y.; Chen, X.; Zheng, Y.; Liu, W.; Liu, X. Taxifolin, a novel food, attenuates acute alcohol-induced liver injury in mice through regulating the NF-κB-mediated inflammation and PI3K/Akt signalling pathways. Pharm. Biol. 2021, 59, 868–879. [Google Scholar] [CrossRef] [PubMed]
- Islam, J.; Shree, A.; Vafa, A.; Afzal, S.M.; Sultana, S. Taxifolin ameliorates Benzo[a]pyrene-induced lung injury possibly via stimulating the Nrf2 signalling pathway. Int. Immunopharmacol. 2021, 96, 107566. [Google Scholar] [CrossRef] [PubMed]
- Salama, S.A.; Kabel, A.M. Taxifolin ameliorates iron overload-induced hepatocellular injury: Modulating PI3K/AKT and p38 MAPK signaling, inflammatory response, and hepatocellular regeneration. Chem. Biol. Interact. 2020, 330, 109230. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.A.; Song, Y.R.; Park, M.H.; Chung, H.Y.; Na, H.S.; Chung, J. Catechin ameliorates Porphyromonas gingivalis-induced inflammation via the regulation of TLR2/4 and inflammasome signaling. J. Periodontol. 2020, 91, 661–670. [Google Scholar] [CrossRef]
- Sunil, M.A.; Sunitha, V.S.; Santhakumaran, P.; Mohan, M.C.; Jose, M.S.; Radhakrishnan, E.K.; Mathew, J. Protective effect of (+)-catechin against lipopolysaccharide-induced inflammatory response in RAW 264.7 cells through downregulation of NF-κB and p38 MAPK. Inflammopharmacology 2021, 29, 1139–1155. [Google Scholar] [CrossRef]
- Cordero-Herrera, I.; Chen, X.; Ramos, S.; Devaraj, S. (−)-Epicatechin attenuates high-glucose-induced inflammation by epigenetic modulation in human monocytes. Eur. J. Nutr. 2017, 56, 1369–1373. [Google Scholar] [CrossRef] [PubMed]
- Xing, J.; Yu, Z.; Zhang, X.; Li, W.; Gao, D.; Wang, J.; Ma, X.; Nie, X.; Wang, W. Epicatechin alleviates inflammation in lipopolysaccharide-induced acute lung injury in mice by inhibiting the p38 MAPK signaling pathway. Int. Immunopharmacol. 2019, 66, 146–153. [Google Scholar] [CrossRef] [PubMed]
- Lan, X.; Han, X.; Li, Q.; Wang, J. (−)-Epicatechin, a natural flavonoid compound, protects astrocytes against hemoglobin toxicity via Nrf2 and AP-1 signaling pathways. Mol. Neurobiol. 2017, 54, 7898–7907. [Google Scholar] [CrossRef] [PubMed]
- Sugimoto, M.; Ko, R.; Goshima, H.; Koike, A.; Shibano, M.; Fujimori, K. Formononetin attenuates H2O2-induced cell death through decreasing ROS level by PI3K/Akt-Nrf2-activated antioxidant gene expression and suppressing MAPK-regulated apoptosis in neuronal SH-SY5Y cells. Neurotoxicology 2021, 85, 186–200. [Google Scholar] [CrossRef] [PubMed]
- Yi, L.; Lu, Y.; Yu, S.; Cheng, Q.; Yi, L. Formononetin inhibits inflammation and promotes gastric mucosal angiogenesis in gastric ulcer rats through regulating NF-κB signaling pathway. J. Recept. Signal Transduct. Res. 2022, 42, 16–22. [Google Scholar] [CrossRef]
- Han, S.; Wu, H.; Li, W.; Gao, P. Protective effects of genistein in homocysteine-induced endothelial cell inflammatory injury. Mol. Cell. Biochem. 2015, 403, 43–49. [Google Scholar] [CrossRef]
- Wan, C.; Jin, F.; Du, Y.; Yang, K.; Yao, L.; Mei, Z.; Huang, W. Genistein improves schistosomiasis liver granuloma and fibrosis via dampening NF-kB signaling in mice. Parasitol. Res. 2017, 116, 1165–1174. [Google Scholar] [CrossRef]
- Li, Y.; Ou, S.; Liu, Q.; Gan, L.; Zhang, L.; Wang, Y.; Qin, J.; Liu, J.; Wu, W. Genistein improves mitochondrial function and inflammatory in rats with diabetic nephropathy via inhibiting MAPK/NF-κB pathway. Acta Cir. Bras. 2022, 37, e370601. [Google Scholar] [CrossRef]
- Han, J.Y.; Cho, S.S.; Yang, J.H.; Kim, K.M.; Jang, C.H.; Park, D.E.; Bang, J.S.; Jung, Y.S.; Ki, S.H. The chalcone compound isosalipurposide (ISPP) exerts a cytoprotective effect against oxidative injury via Nrf2 activation. Toxicol. Appl. Pharmacol. 2015, 287, 77–85. [Google Scholar] [CrossRef]
- Lv, H.; Liu, Q.; Wen, Z.; Feng, H.; Deng, X.; Ci, X. Xanthohumol ameliorates lipopolysaccharide (LPS)-induced acute lung injury via induction of AMPK/GSK3β-Nrf2 signal axis. Redox Biol. 2017, 12, 311–324. [Google Scholar] [CrossRef]
- Rahman, S.U.; Ali, T.; Hao, Q.; He, K.; Li, W.; Ullah, N.; Zhang, Z.; Jiang, Y.; Li, S. Xanthohumol attenuates lipopolysaccharide-induced depressive like behavior in mice: Involvement of NF-κB/Nrf2 signaling pathways. Neurochem. Res. 2021, 46, 3135–3148. [Google Scholar] [CrossRef]
- Youn, C.K.; Park, S.J.; Lee, M.Y.; Cha, M.J.; Kim, O.H.; You, H.J.; Chang, I.Y.; Yoon, S.P.; Jeon, Y.J. Silibinin inhibits LPS-induced macrophage activation by blocking p38 MAPK in RAW 264.7 cells. Biomol. Ther. 2013, 21, 258–263. [Google Scholar] [CrossRef]
- Raina, K.; Agarwal, C.; Agarwal, R. Effect of silibinin in human colorectal cancer cells: Targeting the activation of NF-κB signaling. Mol. Carcinog. 2013, 52, 195–206. [Google Scholar] [CrossRef] [PubMed]
- Tie, F.; Fu, Y.; Hu, N.; Wang, H. Silibinin protects against H2O2-induced oxidative damage in SH-SY5Y cells by improving mitochondrial function. Antioxidants 2022, 11, 1101. [Google Scholar] [CrossRef] [PubMed]
- Bashllari, R.; Molonia, M.S.; Muscarà, C.; Speciale, A.; Wilde, P.J.; Saija, A.; Cimino, F. Cyanidin-3-O-glucoside protects intestinal epithelial cells from palmitate-induced lipotoxicity. Arch. Physiol. Biochem. 2020, 6, 1–8. [Google Scholar] [CrossRef]
- Lee, D.Y.; Yun, S.M.; Song, M.Y.; Jung, K.; Kim, E.H. Cyanidin chloride induces apoptosis by inhibiting NF-κB signaling through activation of Nrf2 in colorectal cancer cells. Antioxidants 2020, 9, 285. [Google Scholar] [CrossRef] [PubMed]
- Bai, X.; Lian, Y.; Hu, C.; Yang, S.; Pei, B.; Yao, M.; Zhu, X.; Shang, L.; Li, Z. Cyanidin-3-glucoside protects against high glucose-induced injury in human nucleus pulposus cells by regulating the Nrf2/HO-1 signaling. J. Appl. Toxicol. 2022, 42, 1137–1145. [Google Scholar] [CrossRef]
- Ouyang, S.; Chen, W.; Gaofeng, Z.; Changcheng, L.; Guoping, T.; Minyan, Z.; Yang, L.; Min, Y.; Luo, J. Cyanidin-3-O-β-glucoside protects against pulmonary artery hypertension induced by monocrotaline via the TGF-β1/p38 MAPK/CREB signaling pathway. Mol. Med. Rep. 2021, 23, 338. [Google Scholar] [CrossRef]
- Seo, B.N.; Ryu, J.M.; Yun, S.P.; Jeon, J.H.; Park, S.S.; Oh, K.B.; Park, J.K.; Han, H.J. Delphinidin prevents hypoxia-induced mouse embryonic stem cell apoptosis through reduction of intracellular reactive oxygen species-mediated activation of JNK and NF-κB, and Akt inhibition. Apoptosis 2013, 18, 811–824. [Google Scholar] [CrossRef] [PubMed]
- Ni, T.; Yang, W.; Xing, Y. Protective effects of delphinidin against H(2)O(2)-induced oxidative injuries in human retinal pigment epithelial cells. Biosci. Rep. 2019, 39, BSR20190689. [Google Scholar] [CrossRef]
- Dai, T.; Shi, K.; Chen, G.; Shen, Y.; Pan, T. Malvidin attenuates pain and inflammation in rats with osteoarthritis by suppressing NF-κB signaling pathway. Inflamm. Res. 2017, 66, 1075–1084. [Google Scholar] [CrossRef]
- Fan, H.; Cui, J.; Liu, F.; Zhang, W.; Yang, H.; He, N.; Dong, Z.; Dong, J. Malvidin protects against lipopolysaccharide-induced acute liver injury in mice via regulating Nrf2 and NLRP3 pathways and suppressing apoptosis and autophagy. Eur. J. Pharmacol. 2022, 933, 175252. [Google Scholar] [CrossRef]
- Hämäläinen, M.; Nieminen, R.; Vuorela, P.; Heinonen, M.; Moilanen, E. Anti-inflammatory effects of flavonoids: Genistein, kaempferol, quercetin, and daidzein inhibit STAT-1 and NF-kappaB activations, whereas flavone, isorhamnetin, naringenin, and pelargonidin inhibit only NF-kappaB activation along with their inhibitory effect on iNOS expression and NO production in activated macrophages. Mediat. Inflamm. 2007, 2007, 45673. [Google Scholar] [CrossRef]
- Fu, K.; Chen, M.; Zheng, H.; Li, C.; Yang, F.; Niu, Q. Pelargonidin ameliorates MCAO-induced cerebral ischemia/reperfusion injury in rats by the action on the Nrf2/HO-1 pathway. Transl. Neurosci. 2021, 12, 20–31. [Google Scholar] [CrossRef]
- Teshika, J.D.; Zakariyyah, A.M.; Zaynab, T.; Zengin, G.; Rengasamy, K.R.; Pandian, S.K.; Fawzi, M.M. Traditional and modern uses of onion bulb (Allium cepa L.): A systematic review. Crit. Rev. Food Sci. Nutr. 2019, 59, S39–S70. [Google Scholar] [CrossRef] [PubMed]
- Jafarinia, M.; Sadat Hosseini, M.; Kasiri, N.; Fazel, N.; Fathi, F.; Ganjalikhani Hakemi, M.; Eskandari, N. Quercetin with the potential effect on allergic diseases. Allergy Asthma Clin. Immunol. 2020, 16, 36. [Google Scholar] [CrossRef]
- Singh, N.; Baby, D.; Rajguru, J.P.; Patil, P.B.; Thakkannavar, S.S.; Pujari, V.B. Inflammation and cancer. Ann. Afr. Med. 2019, 18, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Bi, R.; Hao, J.; Wang, S.; Huo, Y.; Demoz, R.M.; Banda, R.; Tian, S.; Xin, C.; Fu, M.; et al. A study on the protective effects of taxifolin on human umbilical vein endothelial cells and THP-1 cells damaged by hexavalent chromium: A probable mechanism for preventing cardiovascular disease induced by heavy metals. Food Funct. 2020, 11, 3851–3859. [Google Scholar] [CrossRef] [PubMed]
- Alanezi, A.A.; Almuqati, A.F.; Alfwuaires, M.A.; Alasmari, F.; Namazi, N.I.; Althunibat, O.Y.; Mahmoud, A.M. Taxifolin prevents cisplatin nephrotoxicity by modulating Nrf2/HO-1 pathway and mitigating oxidative stress and inflammation in mice. Pharmaceuticals 2022, 15, 1310. [Google Scholar] [CrossRef] [PubMed]
- Crozier, A.; Jaganath, I.B.; Clifford, M.N. Phenols, polyphenols and tannins: An overview. In Plant Secondary Metabolites; John Wiley & Sons: Hoboken, NJ, USA, 2006; Chapter 1; pp. 1–24. [Google Scholar] [CrossRef]
- Chen, L.R.; Ko, N.Y.; Chen, K.H. Isoflavone supplements for menopausal women: A systematic review. Nutrients 2019, 11, 2649. [Google Scholar] [CrossRef]
- Cykowiak, M.; Krajka-Kuźniak, V.; Kleszcz, R.; Kucińska, M.; Szaefer, H.; Piotrowska-Kempisty, H.; Plewiński, A.; Murias, M.; Baer-Dubowska, W. Comparison of the impact of xanthohumol and phenethyl isothiocyanate and their combination on Nrf2 and NF-κB pathways in HepG2 cells in vitro and tumor burden in vivo. Nutrients 2021, 13, 3000. [Google Scholar] [CrossRef]
- Wang, W.; Chen, Z.; Zheng, T.; Zhang, M. Xanthohumol alleviates T2DM-induced liver steatosis and fibrosis by mediating the NRF2/RAGE/NF-κB signaling pathway. Future Med. Chem. 2021, 13, 2069–2081. [Google Scholar] [CrossRef]
- Yadav, V.R.; Prasad, S.; Sung, B.; Aggarwal, B.B. The role of chalcones in suppression of NF-κB-mediated inflammation and cancer. Int. Immunopharmacol. 2011, 11, 295–309. [Google Scholar] [CrossRef] [PubMed]
- Ezhilarasan, D.; Lakshmi, T. A molecular insight into the role of antioxidants in nonalcoholic fatty liver diseases. Oxidative Med. Cell. Longev. 2022, 2022, 9233650. [Google Scholar] [CrossRef]
- Opinion of the HMPC on a European Union Herbal Monograph on Silybum marianum (L.) Gaertn., Fructus; European Medicine Agency: Amsterdam, The Netherlands, 2018.
- Boigk, G.; Stroedter, L.; Herbst, H.; Waldschmidt, J.; Riecken, E.O.; Schuppan, D. Silymarin retards collagen accumulation in early and advanced biliary fibrosis secondary to complete bile duct obliteration in rats. Hepatology 1997, 26, 643–649. [Google Scholar] [CrossRef]
- Davis-Searles, P.R.; Nakanishi, Y.; Kim, N.-C.; Graf, T.N.; Oberlies, N.H.; Wani, M.C.; Wall, M.E.; Agarwal, R.; Kroll, D.J. Milk thistle and prostate cancer: Differential effects of pure flavonolignans from Silybum marianum on antiproliferative end points in human prostate carcinoma cells. Cancer Res. 2005, 65, 4448–4457. [Google Scholar] [CrossRef]
- Kim, B.R.; Seo, H.S.; Ku, J.M.; Kim, G.J.; Jeon, C.Y.; Park, J.H.; Jang, B.H.; Park, S.J.; Shin, Y.C.; Ko, S.G. Silibinin inhibits the production of pro-inflammatory cytokines through inhibition of NF-κB signaling pathway in HMC-1 human mast cells. Inflamm. Res. 2013, 62, 941–950. [Google Scholar] [CrossRef] [PubMed]
- Kavitha, C.V.; Deep, G.; Gangar, S.C.; Jain, A.K.; Agarwal, C.; Agarwal, R. Silibinin inhibits prostate cancer cells- and RANKL-induced osteoclastogenesis by targeting NFATc1, NF-κB, and AP-1 activation in RAW264.7 cells. Mol. Carcinog. 2014, 53, 169–180. [Google Scholar] [CrossRef]
- Tian, L.; Li, W.; Wang, T. Therapeutic effects of silibinin on LPS-induced acute lung injury by inhibiting NLRP3 and NF-κB signaling pathways. Microb. Pathog. 2017, 108, 104–108. [Google Scholar] [CrossRef]
- Xie, Y.; Feng, S.L.; Mai, C.T.; Zheng, Y.F.; Wang, H.; Liu, Z.Q.; Zhou, H.; Liu, L. Suppression of up-regulated LXRα by silybin ameliorates experimental rheumatoid arthritis and abnormal lipid metabolism. Phytomedicine 2021, 80, 153339. [Google Scholar] [CrossRef]
- Matias, M.L.; Gomes, V.J.; Romao-Veiga, M.; Ribeiro, V.R.; Nunes, P.R.; Romagnoli, G.G.; Peracoli, J.C.; Peracoli, M.T.S. Silibinin downregulates the NF-κB pathway and NLRP1/NLRP3 inflammasomes in monocytes from pregnant women with preeclampsia. Molecules 2019, 24, 1548. [Google Scholar] [CrossRef]
- Wei, P.; Li, X.; Wang, S.; Dong, Y.; Yin, H.; Gu, Z.; Na, X.; Wei, X.; Yuan, J.; Cao, J.; et al. Silibinin ameliorates formaldehyde-induced cognitive impairment by inhibiting oxidative stress. Oxidative Med. Cell. Longev. 2022, 2022, 5981353. [Google Scholar] [CrossRef]
- Jaakola, L.; Määttä, K.; Pirttilä, A.M.; Törrönen, R.; Kärenlampi, S.; Hohtola, A. Expression of genes involved in anthocyanin biosynthesis in relation to anthocyanin, proanthocyanidin, and flavonol levels during bilberry fruit development. Plant Physiol. 2002, 130, 729–739. [Google Scholar] [CrossRef]
- Waller, C.W.; Gisvold, O. A phytochemical Investigation of Larrea divaricata Cav. J. Am. Pharm. Assoc.-Sci. Ed. 1945, 34, 78–81. [Google Scholar] [CrossRef]
- Brennan, P.; O’Neill, L.A. Inhibition of nuclear factor kappaB by direct modification in whole cells-mechanism of action of nordihydroguaiaritic acid, curcumin and thiol modifiers. Biochem. Pharmacol. 1998, 55, 965–973. [Google Scholar] [CrossRef]
- Pupe, A.; Degreef, H.; Garmyn, M. Induction of tumor necrosis factor-alpha by UVB: A role for reactive oxygen intermediates and eicosanoids. Photochem. Photobiol. 2003, 78, 68–74. [Google Scholar] [CrossRef]
- van Puijenbroek, A.A.; Wissink, S.; van der Saag, P.T.; Peppelenbosch, M.P. Phospholipase A2 inhibitors and leukotriene synthesis inhibitors block TNF-induced NF-kappaB activation. Cytokine 1999, 11, 104–110. [Google Scholar] [CrossRef]
- Won, J.S.; Im, Y.B.; Khan, M.; Singh, A.K.; Singh, I. Involvement of phospholipase A2 and lipoxygenase in lipopolysaccharide-induced inducible nitric oxide synthase expression in glial cells. Glia 2005, 51, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Mahajan, U.M.; Gupta, C.; Wagh, P.R.; Karpe, P.A.; Tikoo, K. Alteration in inflammatory/apoptotic pathway and histone modifications by nordihydroguaiaretic acid prevents acute pancreatitis in swiss albino mice. Apoptosis 2011, 16, 1138–1149. [Google Scholar] [CrossRef]
- Kim, H.Y.; Goo, J.H.; Joo, Y.A.; Lee, H.Y.; Lee, S.M.; Oh, C.T.; Ahn, S.M.; Kim, N.H.; Hwang, J.S. Impact on inflammation and recovery of skin barrier by nordihydroguaiaretic Acid as a protease-activated receptor 2 antagonist. Biomol. Ther. 2012, 20, 463–469. [Google Scholar] [CrossRef]
- Zúñiga-Toalá, A.; Zatarain-Barrón, Z.L.; Hernández-Pando, R.; Negrette-Guzmán, M.; Huerta-Yepez, S.; Torres, I.; Pinzón, E.; Tapia, E.; Pedraza-Chaverri, J. Nordihydroguaiaretic acid induces Nrf2 nuclear translocation in vivo and attenuates renal damage and apoptosis in the ischemia and reperfusion model. Phytomedicine 2013, 20, 775–779. [Google Scholar] [CrossRef] [PubMed]
- Meyer, G.E.; Chesler, L.; Liu, D.; Gable, K.; Maddux, B.A.; Goldenberg, D.D.; Youngren, J.F.; Goldfine, I.D.; Weiss, W.A.; Matthay, K.K.; et al. Nordihydroguaiaretic acid inhibits insulin-like growth factor signaling, growth, and survival in human neuroblastoma cells. J. Cell. Biochem. 2007, 102, 1529–1541. [Google Scholar] [CrossRef]
- Puga, A.; Barnes, S.J.; Chang, C.; Zhu, H.; Nephew, K.P.; Khan, S.A.; Shertzer, H.G. Activation of transcription factors activator protein-1 and nuclear factor-kappaB by 2,3,7,8-tetrachlorodibenzo-p-dioxin. Biochem. Pharmacol. 2000, 59, 997–1005. [Google Scholar] [CrossRef]
- Lorenzo, J.M.; Munekata, P.E.; Putnik, P.; Kovačević, D.B.; Muchenje, V.; Barba, F.J. Chapter 6—Sources, chemistry, and biological potential of ellagitannins and ellagic acid derivatives. In Studies in Natural Products Chemistry; Atta ur, R., Ed.; Elsevier: Amsterdam, The Netherlands, 2019; Volume 60, pp. 189–221. [Google Scholar]
- Al-khawalde, A.A.-M.A.; Abukhalil, M.H.; Jghef, M.M.; Alfwuaires, M.A.; Alaryani, F.S.; Aladaileh, S.H.; Algefare, A.I.; Karimulla, S.; Alasmari, F.; Aldal’in, H.K.; et al. Punicalagin protects against the development of methotrexate-Induced hepatotoxicity in Mice via activating Nrf2 signaling and decreasing oxidative stress, inflammation, and cell death. Int. J. Mol. Sci. 2022, 23, 12334. [Google Scholar] [CrossRef]
- Ge, G.; Bai, J.; Wang, Q.; Liang, X.; Tao, H.; Chen, H.; Wei, M.; Niu, J.; Yang, H.; Xu, Y.; et al. Punicalagin ameliorates collagen-induced arthritis by downregulating M1 macrophage and pyroptosis via NF-κB signaling pathway. Sci. China Life Sci. 2022, 65, 588–603. [Google Scholar] [CrossRef]
- Tang, L.; Li, T.; Zhang, B.; Zhang, Z.; Sun, X.; Zhu, Y.; Feng, B.; Su, Z.; Yang, L.; Li, H.; et al. Punicalagin alleviates psoriasis by inhibiting NF-κB-mediated IL-1β transcription and caspase-1-regulated IL-1β secretion. Front. Pharmacol. 2022, 13, 817526. [Google Scholar] [CrossRef]
- Aladaileh, S.H.; Al-Swailmi, F.K.; Abukhalil, M.H.; Ahmeda, A.F.; Mahmoud, A.M. Punicalagin prevents cisplatin-induced nephrotoxicity by attenuating oxidative stress, inflammatory response, and apoptosis in rats. Life Sci. 2021, 286, 120071. [Google Scholar] [CrossRef]
- Huang, M.; Wu, K.; Zeng, S.; Liu, W.; Cui, T.; Chen, Z.; Lin, L.; Chen, D.; Ouyang, H. Punicalagin inhibited inflammation and migration of fibroblast-Like synoviocytes through NF-κB pathway in the experimental study of rheumatoid arthritis. J. Inflamm. Res. 2021, 14, 1901–1913. [Google Scholar] [CrossRef]
- Uddin, M.S.; Hasana, S.; Ahmad, J.; Hossain, M.F.; Rahman, M.M.; Behl, T.; Rauf, A.; Ahmad, A.; Hafeez, A.; Perveen, A.; et al. Anti-neuroinflammatory potential of polyphenols by inhibiting NF-κB to halt Alzheimer’s disease. Curr. Pharm. Des. 2021, 27, 402–414. [Google Scholar] [CrossRef]
- Abdelazeem, K.N.M.; Kalo, M.Z.; Beer-Hammer, S.; Lang, F. The gut microbiota metabolite urolithin A inhibits NF-κB activation in LPS stimulated BMDMs. Sci. Rep. 2021, 11, 7117. [Google Scholar] [CrossRef]
- Tao, H.; Li, W.; Zhang, W.; Yang, C.; Zhang, C.; Liang, X.; Yin, J.; Bai, J.; Ge, G.; Zhang, H.; et al. Urolithin A suppresses RANKL-induced osteoclastogenesis and postmenopausal osteoporosis by, suppresses inflammation and downstream NF-κB activated pyroptosis pathways. Pharmacol. Res. 2021, 174, 105967. [Google Scholar] [CrossRef]
- Wei, W.; Peng, C.; Gu, R.; Yan, X.; Ye, J.; Xu, Z.; Sheng, X.; Huang, G.; Guo, Y. Urolithin A attenuates RANKL-induced osteoclastogenesis by co-regulating the p38 MAPK and Nrf2 signaling pathway. Eur. J. Pharmacol. 2022, 921, 174865. [Google Scholar] [CrossRef]
- Huang, W.C.; Liou, C.J.; Shen, S.C.; Hu, S.; Chao, J.C.; Hsiao, C.Y.; Wu, S.J. Urolithin A inactivation of TLR3/TRIF signaling to block the NF-κB/STAT1 axis reduces inflammation and enhances antioxidant defense in poly(I:C)-induced RAW264.7 cells. Int. J. Mol. Sci. 2022, 23, 4697. [Google Scholar] [CrossRef] [PubMed]
- Qu, Z.; An, H.; Feng, M.; Huang, W.; Wang, D.; Zhang, Z.; Yan, L. Urolithin B suppresses osteoclastogenesis via inhibiting RANKL-induced signalling pathways and attenuating ROS activities. J. Cell. Mol. Med. 2022, 26, 4428–4439. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhuang, Q.; Tao, L.; Zheng, K.; Chen, S.; Yang, Y.; Feng, C.; Wang, Z.; Shi, H.; Shi, J.; et al. Urolithin B suppressed osteoclast activation and reduced bone loss of osteoporosis via inhibiting ERK/NF-κB pathway. Cell Prolif. 2022, 55, e13291. [Google Scholar] [CrossRef] [PubMed]
- Iwatake, M.; Okamoto, K.; Tanaka, T.; Tsukuba, T. Castalagin exerts inhibitory effects on osteoclastogenesis through blocking a broad range of signaling pathways with low cytotoxicity. Phytother. Res. 2015, 29, 917–924. [Google Scholar] [CrossRef]
- Xu, H.; Liu, T.; Jia, Y.; Li, J.; Jiang, L.; Hu, C.; Wang, X.; Sheng, J. (−)-Epigallocatechin-3-gallate inhibits osteoclastogenesis by blocking RANKL-RANK interaction and suppressing NF-κB and MAPK signaling pathways. Int. Immunopharmacol. 2021, 95, 107464. [Google Scholar] [CrossRef]
- Chen, G.; Cheng, K.; Niu, Y.; Zhu, L.; Wang, X. (−)-Epicatechin gallate prevents inflammatory response in hypoxia-activated microglia and cerebral edema by inhibiting NF-κB signaling. Arch. Biochem. Biophys. 2022, 729, 109393. [Google Scholar] [CrossRef]
- Kim, S.R.; Seong, K.J.; Kim, W.J.; Jung, J.Y. Epigallocatechin gallate protects against hypoxia-induced inflammation in microglia via NF-κB suppression and Nrf-2/HO-1 activation. Int. J. Mol. Sci. 2022, 23, 4004. [Google Scholar] [CrossRef]
- Li, W.; Yu, J.; Xiao, X.; Li, W.; Zang, L.; Han, T.; Zhang, D.; Niu, X. The inhibitory effect of (−)-epicatechin gallate on the proliferation and migration of vascular smooth muscle cells weakens and stabilizes atherosclerosis. Eur. J. Pharmacol. 2021, 891, 173761. [Google Scholar] [CrossRef]
- Yu, J.; Li, W.; Xiao, X.; Huang, Q.; Yu, J.; Yang, Y.; Han, T.; Zhang, D.; Niu, X. (−)-Epicatechin gallate blocks the development of atherosclerosis by regulating oxidative stress in vivo and in vitro. Food Funct. 2021, 12, 8715–8727. [Google Scholar] [CrossRef]
- Malik, S.; Suchal, K.; Bhatia, J.; Gamad, N.; Dinda, A.K.; Gupta, Y.K.; Arya, D.S. Molecular mechanisms underlying attenuation of cisplatin-induced acute kidney injury by epicatechin gallate. Lab. Investig. 2016, 96, 853–861. [Google Scholar] [CrossRef]
- Wang, J.; Fan, S.M.; Zhang, J. Epigallocatechin-3-gallate ameliorates lipopolysaccharide-induced acute lung injury by suppression of TLR4/NF-κB signaling activation. Braz. J. Med. Biol. Res. 2019, 52, e8092. [Google Scholar] [CrossRef] [PubMed]
- Tong, J.; Fang, J.; Zhu, T.; Xiang, P.; Shang, J.; Chen, L.; Zhao, J.; Wang, Y.; Tong, L.; Sun, M. Pentagalloylglucose reduces AGE-induced inflammation by activating Nrf2/HO-1 and inhibiting the JAK2/STAT3 pathway in mesangial cells. J. Pharmacol. Sci. 2021, 147, 305–314. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.H.; Choi, M.S.; Lee, H.G.; Lee, S.H.; Noh, K.H.; Kwon, S.; Jeong, A.J.; Lee, H.; Yi, E.H.; Park, J.Y.; et al. Photoprotective potential of penta-O-galloyl-β-D-glucose by targeting NF-κB and MAPK signaling in UVB radiation-induced human dermal fibroblasts and mouse skin. Mol. Cells 2015, 38, 982–990. [Google Scholar] [CrossRef] [PubMed]
- Han, S.; Gao, H.; Chen, S.; Wang, Q.; Li, X.; Du, L.J.; Li, J.; Luo, Y.Y.; Li, J.X.; Zhao, L.C.; et al. Procyanidin A1 alleviates inflammatory response induced by LPS through NF-κB, MAPK, and Nrf2/HO-1 pathways in RAW264.7 cells. Sci. Rep. 2019, 9, 15087. [Google Scholar] [CrossRef]
- Zhang, W.Y.; Liu, H.Q.; Xie, K.Q.; Yin, L.L.; Li, Y.; Kwik-Uribe, C.L.; Zhu, X.Z. Procyanidin dimer B2 [epicatechin-(4beta-8)-epicatechin] suppresses the expression of cyclooxygenase-2 in endotoxin-treated monocytic cells. Biochem. Biophys. Res. Commun. 2006, 345, 508–515. [Google Scholar] [CrossRef]
- Song, D.Q.; Liu, J.; Wang, F.; Li, X.F.; Liu, M.H.; Zhang, Z.; Cao, S.S.; Jiang, X. Procyanidin B2 inhibits lipopolysaccharide-induced apoptosis by suppressing the Bcl-2/Bax and NF-κB signalling pathways in human umbilical vein endothelial cells. Mol. Med. Rep. 2021, 23, 1. [Google Scholar] [CrossRef]
- Liu, J.X.; Yang, C.; Liu, Z.J.; Su, H.Y.; Zhang, W.H.; Pan, Q.; Liu, H.F. Protection of procyanidin B2 on mitochondrial dynamics in sepsis associated acute kidney injury via promoting Nrf2 nuclear translocation. Aging 2020, 12, 15638–15655. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wu, Q.; Deng, Y.; Lv, H.; Qiu, J.; Chi, G.; Feng, H. D(−)-Salicin inhibits the LPS-induced inflammation in RAW264.7 cells and mouse models. Int. Immunopharmacol. 2015, 26, 286–294. [Google Scholar] [CrossRef]
- Zhai, K.F.; Duan, H.; Khan, G.J.; Xu, H.; Han, F.K.; Cao, W.G.; Gao, G.Z.; Shan, L.L.; Wei, Z.J. Salicin from Alangium chinense ameliorates rheumatoid arthritis by modulating the Nrf2-HO-1-ROS pathways. J. Agric. Food Chem. 2018, 66, 6073–6082. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.R.; Jung, Y.R.; Kim, D.H.; An, H.J.; Kim, M.K.; Kim, N.D.; Chung, H.Y. Caffeic acid regulates LPS-induced NF-κB activation through NIK/IKK and c-Src/ERK signaling pathways in endothelial cells. Arch. Pharm. Res. 2014, 37, 539–547. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Fang, G.; Yin, S.; Zhao, X.; Zhang, C.; Li, J.; Liu, Z. Caffeic acid prevented LPS-induced injury of primary bovine mammary epithelial cells through inhibiting NF-κB and MAPK activation. Mediat. Inflamm. 2019, 2019, 1897820. [Google Scholar] [CrossRef]
- Yang, S.Y.; Pyo, M.C.; Nam, M.H.; Lee, K.W. ERK/Nrf2 pathway activation by caffeic acid in HepG2 cells alleviates its hepatocellular damage caused by t-butylhydroperoxide-induced oxidative stress. BMC Complement. Altern. Med. 2019, 19, 139. [Google Scholar] [CrossRef]
- Chen, J.; Luo, Y.; Li, Y.; Chen, D.; Yu, B.; He, J. Chlorogenic acid attenuates oxidative stress-induced intestinal epithelium injury by co-regulating the PI3K/Akt and IκBα/NF-κB signaling. Antioxidants 2021, 10, 1915. [Google Scholar] [CrossRef]
- Gao, F.; Fu, K.; Li, H.; Feng, Y.; Tian, W.; Cao, R. Chlorogenic acid ameliorates mice clinical endometritis by activating Keap1/Nrf2 and inhibiting NFκB signalling pathway. J. Pharm. Pharmacol. 2021, 73, 785–795. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.; Wang, C.; Yu, L.; Sheng, T.; Wu, Z.; Wang, X.; Zhang, D.; Lin, Y.; Gong, Y. Chlorogenic acid attenuates dextran sodium sulfate-induced ulcerative colitis in mice through MAPK/ERK/JNK pathway. BioMed Res. Int. 2019, 2019, 6769789. [Google Scholar] [CrossRef]
- Liu, Q.; Chen, Y.; Shen, C.; Xiao, Y.; Wang, Y.; Liu, Z.; Liu, X. Chicoric acid supplementation prevents systemic inflammation-induced memory impairment and amyloidogenesis via inhibition of NF-κB. FASEB J. 2017, 31, 1494–1507. [Google Scholar] [CrossRef]
- Liu, Q.; Hu, Y.; Cao, Y.; Song, G.; Liu, Z.; Liu, X. Chicoric acid ameliorates lipopolysaccharide-induced oxidative stress via promoting the Keap1/Nrf2 transcriptional signaling pathway in BV-2 microglial cells and mouse brain. J. Agric. Food Chem. 2017, 65, 338–347. [Google Scholar] [CrossRef]
- Sunitha, M.C.; Dhanyakrishnan, R.; PrakashKumar, B.; Nevin, K.G. p-Coumaric acid mediated protection of H9c2 cells from Doxorubicin-induced cardiotoxicity: Involvement of augmented Nrf2 and autophagy. Biomed. Pharmacother. 2018, 102, 823–832. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.; Zheng, T.T.; Liang, Y.; Duan, L.F.; Zhang, Y.D.; Wang, L.J.; He, G.M.; Xiao, H.T. p-Coumaric acid protects human lens epithelial cells against oxidative stress-induced apoptosis by MAPK signaling. Oxidative Med. Cell. Longev. 2018, 2018, 8549052. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zheng, Y.; Luo, Y.; Du, Y.; Zhang, X.; Fu, J. Curcumin inhibits LPS-induced neuroinflammation by promoting microglial M2 polarization via TREM2/TLR4/NF-κB pathways in BV2 cells. Mol. Immunol. 2019, 116, 29–37. [Google Scholar] [CrossRef]
- Lin, X.; Bai, D.; Wei, Z.; Zhang, Y.; Huang, Y.; Deng, H.; Huang, X. Curcumin attenuates oxidative stress in RAW264.7 cells by increasing the activity of antioxidant enzymes and activating the Nrf2-Keap1 pathway. PLoS ONE 2019, 14, e0216711. [Google Scholar] [CrossRef] [PubMed]
- Dai, J.; Gu, L.; Su, Y.; Wang, Q.; Zhao, Y.; Chen, X.; Deng, H.; Li, W.; Wang, G.; Li, K. Inhibition of curcumin on influenza A virus infection and influenzal pneumonia via oxidative stress, TLR2/4, p38/JNK MAPK and NF-κB pathways. Int. Immunopharmacol. 2018, 54, 177–187. [Google Scholar] [CrossRef]
- Aslan, A.; Beyaz, S.; Gok, O.; Can, M.I.; Erman, F.; Erman, O. The impact of ellagic acid on some apoptotic gene expressions: A new perspective for the regulation of pancreatic Nrf-2/NF-κB and Akt/VEGF signaling in CCl(4)-induced pancreas damage in rats. Immunopharmacol. Immunotoxicol. 2021, 43, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.Z.; Zhu, G.F.; Zheng, C.Q.; Li, J.J.; Sheng, S.; Li, D.D.; Wang, G.Q.; Zhang, F. Ellagic acid protects dopamine neurons from rotenone-induced neurotoxicity via activation of Nrf2 signalling. J. Cell. Mol. Med. 2020, 24, 9446–9456. [Google Scholar] [CrossRef]
- Gil, T.Y.; Hong, C.H.; An, H.J. Anti-inflammatory effects of ellagic acid on keratinocytes via MAPK and STAT pathways. Int. J. Mol. Sci. 2021, 22, 1277. [Google Scholar] [CrossRef] [PubMed]
- Yin, P.; Zhang, Z.; Li, J.; Shi, Y.; Jin, N.; Zou, W.; Gao, Q.; Wang, W.; Liu, F. Ferulic acid inhibits bovine endometrial epithelial cells against LPS-induced inflammation via suppressing NK-κB and MAPK pathway. Res. Vet. Sci. 2019, 126, 164–169. [Google Scholar] [CrossRef]
- Liu, M.; Zhang, C.; Xu, X.; Zhao, X.; Han, Z.; Liu, D.; Bo, R.; Li, J.; Liu, Z. Ferulic acid inhibits LPS-induced apoptosis in bovine mammary epithelial cells by regulating the NF-κB and Nrf2 signalling pathways to restore mitochondrial dynamics and ROS generation. Vet. Res. 2021, 52, 104. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Lin, L.; Wu, H. Ferulic acid alleviates lipopolysaccharide-induced acute lung injury through inhibiting TLR4/NF-κB signaling pathway. J. Biochem. Mol. Toxicol. 2021, 35, e22664. [Google Scholar] [CrossRef]
- Sohrabi, F.; Dianat, M.; Badavi, M.; Radan, M.; Mard, S.A. Gallic acid suppresses inflammation and oxidative stress through modulating Nrf2-HO-1-NF-κB signaling pathways in elastase-induced emphysema in rats. Environ. Sci. Pollut. Res. Int. 2021, 28, 56822–56834. [Google Scholar] [CrossRef]
- Zhou, D.; Yang, Q.; Tian, T.; Chang, Y.; Li, Y.; Duan, L.R.; Li, H.; Wang, S.W. Gastroprotective effect of gallic acid against ethanol-induced gastric ulcer in rats: Involvement of the Nrf2/HO-1 signaling and anti-apoptosis role. Biomed. Pharmacother. 2020, 126, 110075. [Google Scholar] [CrossRef]
- Ahad, A.; Ahsan, H.; Mujeeb, M.; Siddiqui, W.A. Gallic acid ameliorates renal functions by inhibiting the activation of p38 MAPK in experimentally induced type 2 diabetic rats and cultured rat proximal tubular epithelial cells. Chem. Biol. Interact. 2015, 240, 292–303. [Google Scholar] [CrossRef] [PubMed]
- An, Y.; Zhao, J.; Zhang, Y.; Wu, W.; Hu, J.; Hao, H.; Qiao, Y.; Tao, Y.; An, L. Rosmarinic acid induces proliferation suppression of hepatoma cells associated with NF-κB signaling pathway. Asian Pac. J. Cancer Prev. 2021, 22, 1623–1632. [Google Scholar] [CrossRef]
- Li, Z.; Feng, H.; Wang, Y.; Shen, B.; Tian, Y.; Wu, L.; Zhang, Q.; Jin, M.; Liu, G. Rosmarinic acid protects mice from lipopolysaccharide/d-galactosamine-induced acute liver injury by inhibiting MAPKs/NF-κB and activating Nrf2/HO-1 signaling pathways. Int. Immunopharmacol. 2019, 67, 465–472. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Lu, Y.; Yang, F.; Li, S.; He, X.; Gao, Y.; Zhang, G.; Ren, E.; Wang, Y.; Kang, X. Rosmarinic acid exerts a neuroprotective effect on spinal cord injury by suppressing oxidative stress and inflammation via modulating the Nrf2/HO-1 and TLR4/NF-κB pathways. Toxicol. Appl. Pharmacol. 2020, 397, 115014. [Google Scholar] [CrossRef] [PubMed]
- Tan, Z.; Zhang, B. Echinacoside alleviates osteoarthritis in rats by activating the Nrf2-HO-1 signaling pathway. Immunopharmacol. Immunotoxicol. 2022, 44, 850–859. [Google Scholar] [CrossRef]
- Chen, S.; Liu, H.; Wang, S.; Jiang, H.; Gao, L.; Wang, L.; Teng, L.; Wang, C.; Wang, D. The neuroprotection of verbascoside in Alzheimer’s disease mediated through mitigation of neuroinflammation via blocking NF-κB-p65 signaling. Nutrients 2022, 14, 1417. [Google Scholar] [CrossRef]
- Zhuang, Y.; Wu, H.; Wang, X.; He, J.; He, S.; Yin, Y. Resveratrol attenuates oxidative stress-induced intestinal barrier injury through PI3K/Akt-mediated Nrf2 signaling pathway. Oxidative Med. Cell. Longev. 2019, 2019, 7591840. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Li, X.; Chen, H.; Han, L.; Ji, X.; Wang, Q.; Wei, L.; Miu, Y.; Wang, J.; Mao, J.; et al. Resveratrol alleviates bleomycin-induced pulmonary fibrosis via suppressing HIF-1α and NF-κB expression. Aging 2021, 13, 4605–4616. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Chang, C.C.; Yang, Y.; Yuan, L.; Xu, L.; Ho, C.T.; Li, S. Resveratrol alleviates rheumatoid arthritis via reducing ROS and inflammation, inhibiting MAPK signaling pathways, and suppressing angiogenesis. J. Agric. Food Chem. 2018, 66, 12953–12960. [Google Scholar] [CrossRef]
- Gao, F.; Zhang, S. Salicin inhibits AGE-induced degradation of type II collagen and aggrecan in human SW1353 chondrocytes: Therapeutic potential in osteoarthritis. Artif. Cells Nanomed. Biotechnol. 2019, 47, 1043–1049. [Google Scholar] [CrossRef]
- Xiao, Y.; Xian, Y.; Hu, X.; Qi, Z. D(−)-salicin inhibits RANKL-induced osteoclast differentiation and function in vitro. Fitoterapia 2022, 157, 104981. [Google Scholar] [CrossRef]
- Song, Y.; Tian, X.; Wang, X.; Feng, H. Vascular protection of salicin on IL-1β-induced endothelial inflammatory response and damages in retinal endothelial cells. Artif. Cells Nanomed. Biotechnol. 2019, 47, 1995–2002. [Google Scholar] [CrossRef]
- Kong, C.S.; Kim, K.H.; Choi, J.S.; Kim, J.E.; Park, C.; Jeong, J.W. Salicin, an extract from white willow bark, inhibits angiogenesis by blocking the ROS-ERK pathways. Phytother. Res. 2014, 28, 1246–1251. [Google Scholar] [CrossRef]
- Arceusz, A.; Wesolowski, M.; Konieczynski, P. Methods for extraction and determination of phenolic acids in medicinal plants: A review. Nat. Prod. Commun. 2013, 8. [Google Scholar] [CrossRef]
- Šudomová, M.; Hassan, S.T.S. Nutraceutical curcumin with promising protection against herpesvirus infections and their associated inflammation: Mechanisms and pathways. Microorganisms 2021, 9, 292. [Google Scholar] [CrossRef]
- Khan, M.A.; Rabbani, G.; Kumari, M.; Khan, M.J. Ellagic acid protects type II collagen induced arthritis in rat via diminution of IKB phosphorylation and suppression IKB-NF-kB complex activation: In vivo and in silico study. Inflammopharmacology 2022, 30, 1729–1743. [Google Scholar] [CrossRef]
- Biała, W.; Jasiński, M. The phenylpropanoid case—It is transport that matters. Front. Plant Sci. 2018, 9, 1610. [Google Scholar] [CrossRef]
- Agar, O.T.; Cankaya, I.I.T. Chapter 5—Analysis of phenylethanoids and their glycosidic derivatives. In Recent Advances in Natural Products Analysis; Sanches Silva, A., Nabavi, S.F., Saeedi, M., Nabavi, S.M., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 221–254. [Google Scholar] [CrossRef]
- Zhang, Y.; Wu, Q.; Zhong, L.; Wang, L.; Gong, D. Echinacoside promotes the proliferation of human renal tubular epithelial cells by blocking the HBX/TREM2-mediated NF-κB signalling pathway. Mol. Med. Rep. 2020, 22, 1137–1144. [Google Scholar] [CrossRef]
- Qiu, H.; Liu, X. Echinacoside improves cognitive impairment by inhibiting Aβ deposition through the PI3K/AKT/Nrf2/PPARγ signaling pathways in APP/PS1 mice. Mol. Neurobiol. 2022, 59, 4987–4999. [Google Scholar] [CrossRef]
- Yang, X.; Yv, Q.; Ye, F.; Chen, S.; He, Z.; Li, W.; Dong, F. Echinacoside protects dopaminergic neurons through regulating IL-6/JAK2/STAT3 pathway in Parkinson’s disease model. Front. Pharmacol. 2022, 13, 848813. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, Z.; Xiang, J.; Cai, M.; Yu, Z.; Li, X.; Wu, T.; Cai, D. Neuroprotective effects of echinacoside on regulating the stress-Active p38MAPK and NF-κB p52 signals in the mice model of Parkinson’s disease. Neurochem. Res. 2017, 42, 975–985. [Google Scholar] [CrossRef]
- Shi, Y.; Cao, H.; Liu, Z.; Xi, L.; Dong, C. Echinacoside induces mitochondria-mediated pyroptosis through Raf/MEK/ERK signaling in non-small cell lung cancer cells. J. Immunol. Res. 2022, 2022, 3351268. [Google Scholar] [CrossRef] [PubMed]
- Shao, Y.; Liu, Y.; Shen, B.; Zhou, Q.; Wei, Z. Echinacoside ameliorates cyclophosphamide-induced bladder damage in mice. J. Med. Food 2022, 25, 722–731. [Google Scholar] [CrossRef] [PubMed]
- Khorashadizadeh, N.; Neamati, A.; Moshiri, M.; Etemad, L. Verbascoside inhibits paraquate-induced pulmonary toxicity via modulating oxidative stress, inflammation, apoptosis and DNA damage in A549 cell. Drug Chem. Toxicol. 2022, 45, 2212–2220. [Google Scholar] [CrossRef]
- Arango-De la Pava, L.D.; Zamilpa, A.; Trejo-Espino, J.L.; Domínguez-Mendoza, B.E.; Jiménez-Ferrer, E.; Pérez-Martínez, L.; Trejo-Tapia, G. Synergism and subadditivity of verbascoside-lignans and -iridoids binary mixtures isolated from Castilleja tenuiflora Benth. on NF-κB/AP-1 inhibition activity. Molecules 2021, 26, 547. [Google Scholar] [CrossRef]
- Ma, H.; Qin, S.; Zhao, S. Osteoarthritis is prevented in rats by verbascoside via nuclear factor kappa B (NF-κB) pathway 4downregulation. Med. Sci. Monit. 2020, 26, e921276. [Google Scholar] [CrossRef]
- Kimura, Y.; Okuda, H. Resveratrol isolated from Polygonum cuspidatum root prevents tumor growth and metastasis to lung and tumor-induced neovascularization in Lewis lung carcinoma-bearing mice. J. Nutr. 2001, 131, 1844–1849. [Google Scholar] [CrossRef]
- Stefanachi, A.; Leonetti, F.; Pisani, L.; Catto, M.; Carotti, A. Coumarin: A Natural, Privileged and Versatile Scaffold for Bioactive Compounds. Molecules 2018, 23, 250. [Google Scholar] [CrossRef]
- Assessment Report on Aesculus hippocastanum L., Cortex; European Medicine Agency: Amsterdam, The Netherlands, 2012.
- Yang, X.D.; Chen, Z.; Ye, L.; Chen, J.; Yang, Y.Y. Esculin protects against methionine choline-deficient diet-induced non-alcoholic steatohepatitis by regulating the Sirt1/NF-κB p65 pathway. Pharm. Biol. 2021, 59, 922–932. [Google Scholar] [CrossRef]
- Tian, X.; Peng, Z.; Luo, S.; Zhang, S.; Li, B.; Zhou, C.; Fan, H. Aesculin protects against DSS-Induced colitis though activating PPARγ and inhibiting NF-кB pathway. Eur. J. Pharmacol. 2019, 857, 172453. [Google Scholar] [CrossRef]
- Tianzhu, Z.; Shumin, W. Esculin inhibits the inflammation of LPS-induced acute lung injury in mice via regulation of TLR/NF-κB pathways. Inflammation 2015, 38, 1529–1536. [Google Scholar] [CrossRef]
- Zhao, X.L.; Chen, L.F.; Wang, Z. Aesculin modulates bone metabolism by suppressing receptor activator of NF-κB ligand (RANKL)-induced osteoclastogenesis and transduction signals. Biochem. Biophys. Res. Commun. 2017, 488, 15–21. [Google Scholar] [CrossRef]
- Wang, S.K.; Chen, T.X.; Wang, W.; Xu, L.L.; Zhang, Y.Q.; Jin, Z.; Liu, Y.B.; Tang, Y.Z. Aesculetin exhibited anti-inflammatory activities through inhibiting NF-кB and MAPKs pathway in vitro and in vivo. J. Ethnopharmacol. 2022, 296, 115489. [Google Scholar] [CrossRef]
- Lee, H.C.; Liu, F.C.; Tsai, C.N.; Chou, A.H.; Liao, C.C.; Yu, H.P. Esculetin ameliorates lipopolysaccharide-induced acute lung injury in mice via modulation of the AKT/ERK/NF-κB and RORγt/IL-17 pathways. Inflammation 2020, 43, 962–974. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, Z.; Wu, H.; Wang, J.; Zhang, S. Esculetin alleviates murine lupus nephritis by inhibiting complement activation and enhancing Nrf2 signaling pathway. J. Ethnopharmacol. 2022, 288, 115004. [Google Scholar] [CrossRef] [PubMed]
- Ozal, S.A.; Turkekul, K.; Gurlu, V.; Guclu, H.; Erdogan, S. Esculetin protects human retinal pigment epithelial cells from lipopolysaccharide-induced inflammation and cell death. Curr. Eye Res. 2018, 43, 1169–1176. [Google Scholar] [CrossRef] [PubMed]
- Brezáni, V.; Leláková, V.; Hassan, S.T.S.; Berchová-Bímová, K.; Nový, P.; Klouček, P.; Maršík, P.; Dall’Acqua, S.; Hošek, J.; Šmejkal, K. Anti-infectivity against Herpes simplex virus and selected microbes and anti-inflammatory activities of compounds isolated from Eucalyptus globulus Labill. Viruses 2018, 10, 360. [Google Scholar] [CrossRef]
- Bumrungpert, A.; Kalpravidh, R.W.; Chitchumroonchokchai, C.; Chuang, C.C.; West, T.; Kennedy, A.; McIntosh, M. Xanthones from mangosteen prevent lipopolysaccharide-mediated inflammation and insulin resistance in primary cultures of human adipocytes. J. Nutr. 2009, 139, 1185–1191. [Google Scholar] [CrossRef]
- Bumrungpert, A.; Kalpravidh, R.W.; Chuang, C.C.; Overman, A.; Martinez, K.; Kennedy, A.; McIntosh, M. Xanthones from mangosteen inhibit inflammation in human macrophages and in human adipocytes exposed to macrophage-conditioned media. J. Nutr. 2010, 140, 842–847. [Google Scholar] [CrossRef] [PubMed]
- Feng, Z.; Lu, X.; Gan, L.; Zhang, Q.; Lin, L. Xanthones, a promising anti-inflammatory scaffold: Structure, activity, and drug likeness analysis. Molecules 2020, 25, 598. [Google Scholar] [CrossRef] [PubMed]
- Gunter, N.V.; Teh, S.S.; Lim, Y.M.; Mah, S.H. Natural xanthones and skin inflammatory diseases: Multitargeting mechanisms of action and potential application. Front. Pharmacol. 2020, 11, 594202. [Google Scholar] [CrossRef]
- Hopkins, A.L. Network pharmacology: The next paradigm in drug discovery. Nat. Chem. Biol. 2008, 4, 682–690. [Google Scholar] [CrossRef]
- Zhang, R.; Zhu, X.; Bai, H.; Ning, K. Network pharmacology databases for Traditional Chinese Medicine: Review and assessment. Front. Pharmacol. 2019, 10, 123. [Google Scholar] [CrossRef]
- Yang, Y.; Qian, C.; Wu, R.; Wang, R.; Ou, J.; Liu, S. Exploring the mechanism of the Fructus Mume and Rhizoma Coptidis herb pair intervention in ulcerative colitis from the perspective of inflammation and immunity based on systemic pharmacology. BMC Complement. Med. Ther. 2023, 23, 11. [Google Scholar] [CrossRef] [PubMed]
- Shen, C.; Wang, Y.; Zhang, H.; Li, W.; Chen, W.; Kuang, M.; Song, Y.; Zhong, Z. Exploring the active components and potential mechanisms of Rosa roxburghii Tratt in treating type 2 diabetes mellitus based on UPLC-Q-exactive Orbitrap/MS and network pharmacology. Chin. Med. 2023, 18, 12. [Google Scholar] [CrossRef] [PubMed]
- Deng, H.; Jiang, J.; Zhang, S.; Wu, L.; Zhang, Q.; Sun, W. Network pharmacology and experimental validation to identify the potential mechanism of Hedyotis diffusa Willd against rheumatoid arthritis. Sci. Rep. 2023, 13, 1425. [Google Scholar] [CrossRef]
- Hua, Z.; Dai, S.; Li, S.; Wang, J.; Peng, H.; Rong, Y.; Yu, H.; Liu, M. Deciphering the protective effect of Buzhong Yiqi Decoction on osteoporotic fracture through network pharmacology and experimental validation. J. Orthop. Surg. Res. 2023, 18, 86. [Google Scholar] [CrossRef] [PubMed]
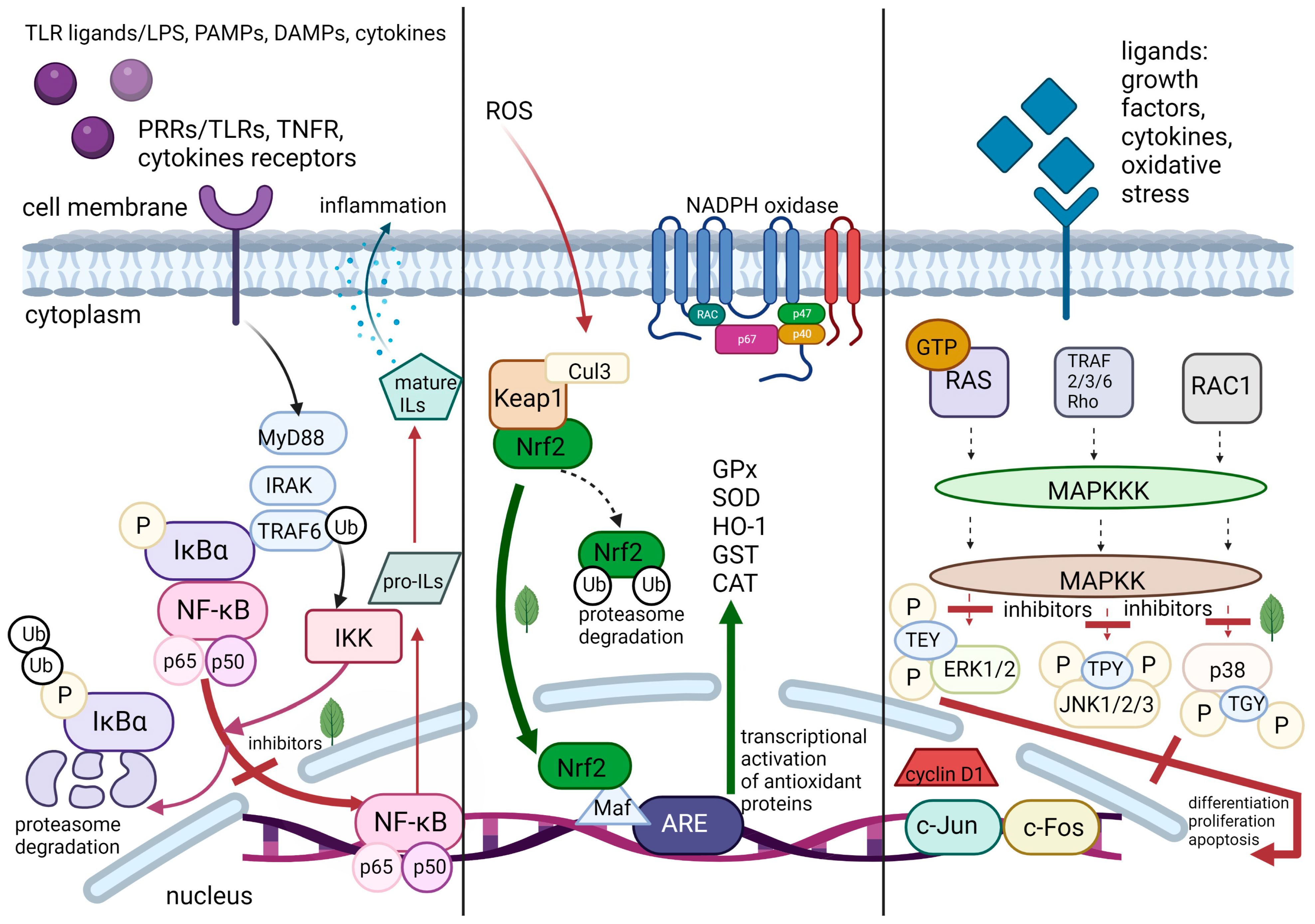
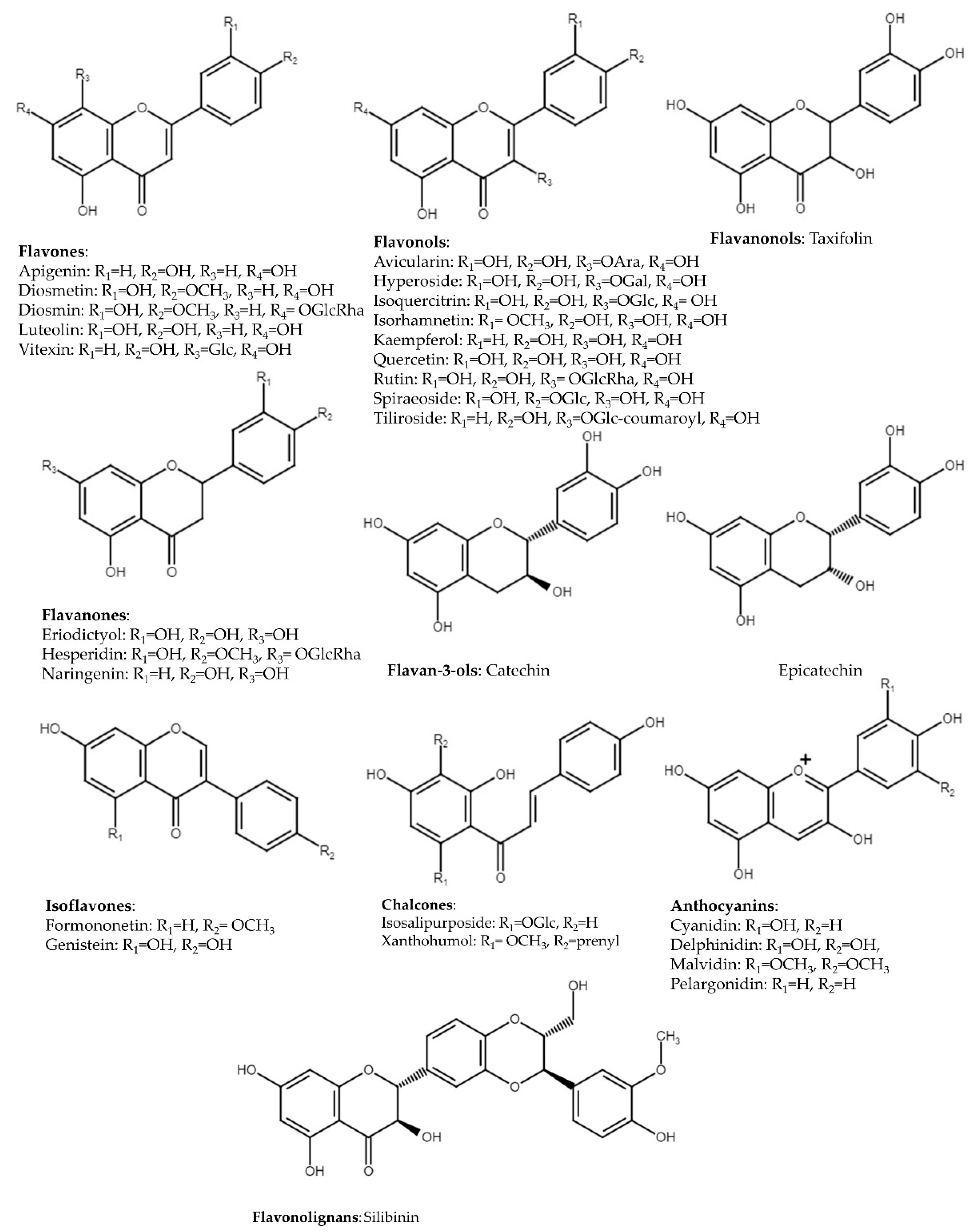
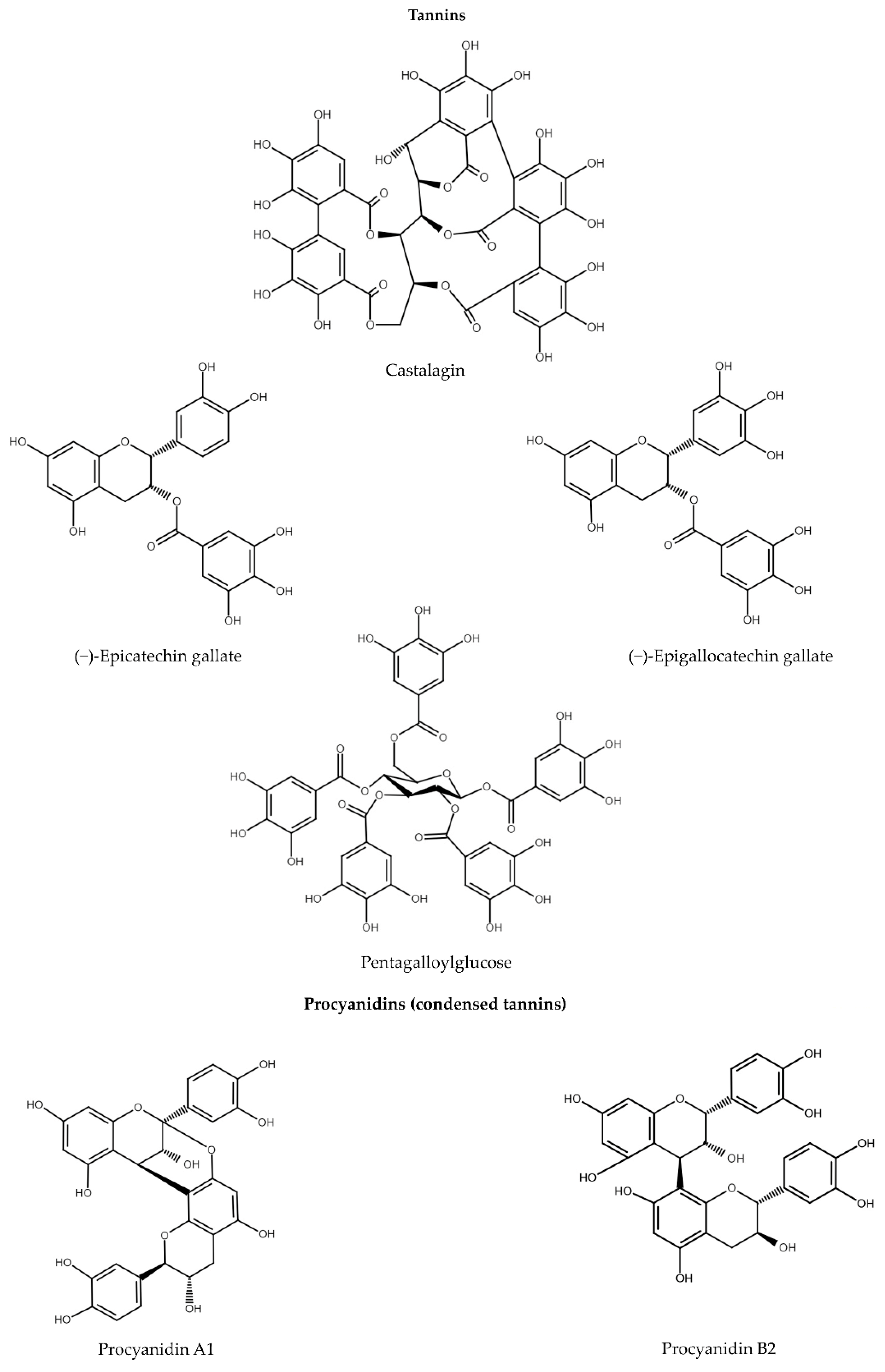
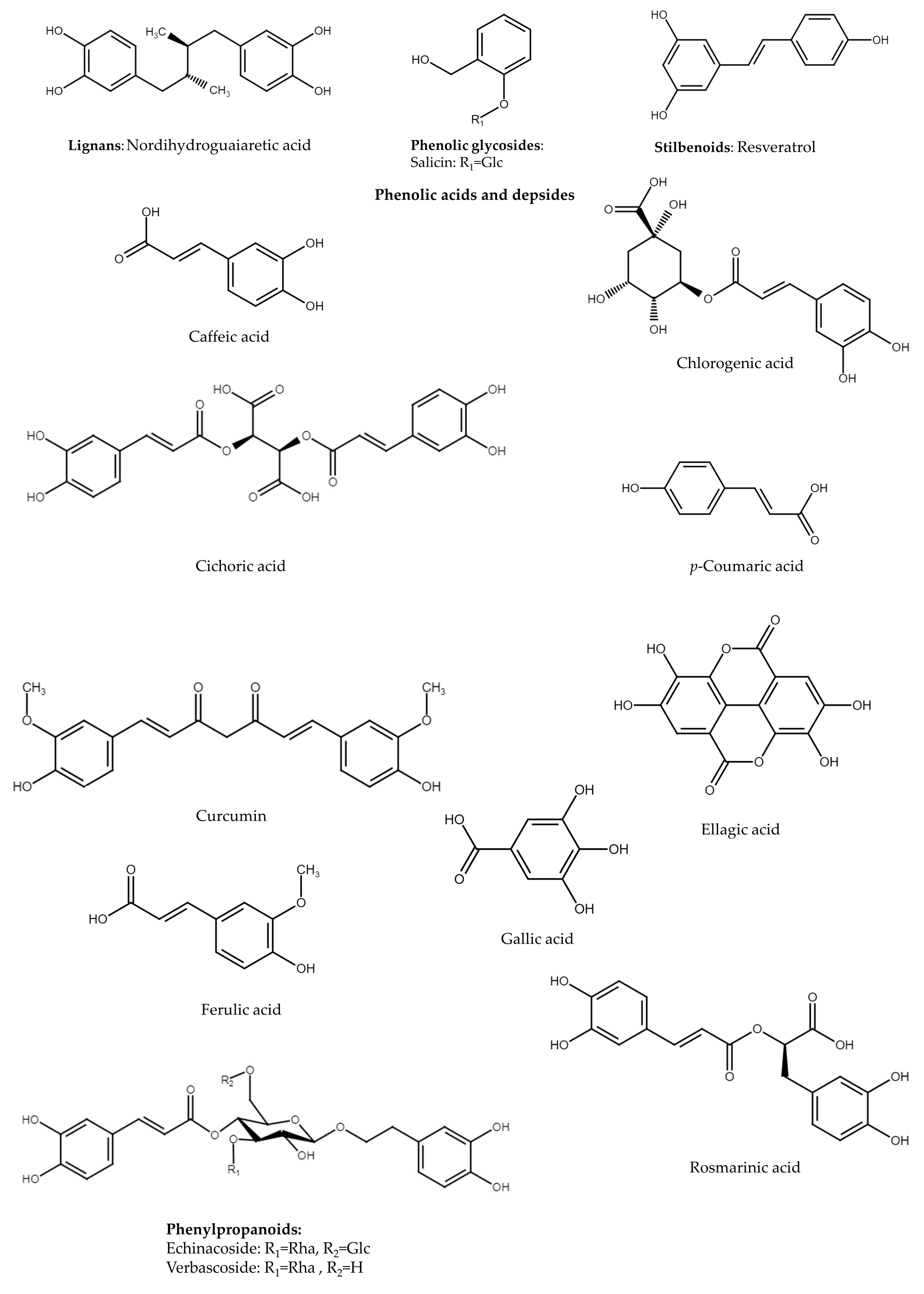
| Compounds | Scopus | PubMed | Medline | ||||||
|---|---|---|---|---|---|---|---|---|---|
| NF-κB | Nrf2 | MAPK | NF-κB | Nrf2 | MAPK | NF-κB | Nrf2 | MAPK | |
| Apigenin | 354 | 213 | 290 | 286 | 88 | 138 | 346 | 113 | 270 |
| Diosmetin | 33 | 24 | 17 | 14 | 14 | 7 | 13 | 13 | 9 |
| Diosmin | 26 | 12 | 16 | 19 | 8 | 5 | 149 | 46 | 123 |
| Luteolin | 375 | 292 | 371 | 305 | 127 | 175 | 350 | 157 | 292 |
| Vitexin | 44 | 35 | 30 | 28 | 15 | 10 | 23 | 12 | 14 |
| Avicularin | 8 | 1 | 3 | 10 | 1 | 2 | 8 | 1 | 2 |
| Hyperoside | 42 | 28 | 23 | 43 | 28 | 20 | 34 | 23 | 21 |
| Isoquercitrin | 37 | 27 | 31 | 24 | 17 | 16 | 18 | 14 | 17 |
| Isorhamnetin | 88 | 64 | 90 | 51 | 21 | 34 | 37 | 14 | 33 |
| Kaempferol | 360 | 213 | 399 | 272 | 72 | 173 | 190 | 54 | 173 |
| Quercetin | 972 | 877 | 885 | 827 | 390 | 417 | 572 | 320 | 435 |
| Rutin | 146 | 86 | 101 | 181 | 83 | 65 | 114 | 58 | 65 |
| Spiraeoside | - | 1 | 1 | - | 1 | 1 | - | 1 | 1 |
| Tiliroside | 5 | 6 | 9 | 5 | 4 | 4 | 5 | 4 | 4 |
| Eriodictyol | 28 | 46 | 34 | 18 | 17 | 14 | 15 | 17 | 13 |
| Hesperidin | 142 | 120 | 108 | 14 | 61 | 44 | 94 | 54 | 47 |
| Naringenin | 169 | 132 | 169 | 119 | 51 | 58 | 47 | 44 | 59 |
| Taxifolin | 60 | 51 | 44 | 40 | 27 | 14 | 31 | 22 | 17 |
| Catechin | 539 | 361 | 407 | 603 | 236 | 282 | 391 | 206 | 310 |
| Epicatechin | 169 | 142 | 142 | 610 | 238 | 292 | 79 | 47 | 64 |
| Formononetin | 75 | 51 | 99 | 43 | 19 | 34 | 34 | 18 | 35 |
| Genistein | 553 | 231 | 668 | 489 | 83 | 484 | 289 | 70 | 614 |
| Isosalipurposide | 3 | 3 | - | 1 | 1 | - | 1 | 1 | 1 |
| Xanthohumol | 53 | 61 | 32 | 52 | 38 | 10 | 33 | 34 | 9 |
| Silibinin | 131 | 77 | 79 | 115 | 33 | 41 | 99 | 36 | 60 |
| Cyanidin | 110 | 84 | 84 | 109 | 47 | 48 | 40 | 24 | 25 |
| Delphinidin | 49 | 25 | 51 | 43 | 8 | 27 | 29 | 7 | 29 |
| Malvidin | 25 | 19 | 24 | 23 | 10 | 10 | 10 | 5 | 8 |
| Pelargonidin | 24 | 18 | 28 | 11 | 7 | 10 | 8 | 5 | 8 |
| Nordihydroguaiaretic acid | 47 | 22 | 34 | 39 | 11 | 25 | 17 | 9 | 30 |
| Castalagin | 3 | 1 | - | 2 | - | - | 2 | - | - |
| Epicatechin gallate | 72 | 57 | 63 | 22 | 10 | 16 | 12 | 7 | 20 |
| Epigallocatechin gallate | 526 | 427 | 399 | 367 | 142 | 191 | 235 | 127 | 213 |
| Pentagalloylglucose | 2 | 2 | 4 | 12 | 5 | 9 | 8 | 4 | 9 |
| Procyanidin A1 | 5 | 1 | 5 | 2 | 1 | 2 | 1 | 1 | 3 |
| Procyanidin B2 | 36 | 27 | 18 | 28 | 16 | 7 | 18 | 15 | 6 |
| Salicin | 7 | 5 | 4 | 10 | 5 | 3 | 7 | 5 | 3 |
| Caffeic acid | 535 | 249 | 248 | 405 | 109 | 96 | 340 | 120 | 137 |
| Chlorogenic acid | 260 | 182 | 164 | 196 | 92 | 70 | 148 | 76 | 77 |
| Cichoric acid | 17 | 20 | 10 | 25 | 12 | 9 | 5 | 1 | 1 |
| Coumaric acid | 169 | 131 | 111 | 153 | 79 | 63 | 116 | 68 | 69 |
| Curcumin | 1797 | 1165 | 855 | 1691 | 499 | 453 | 1192 | 429 | 507 |
| Ellagic acid | 155 | 104 | 88 | 123 | 46 | 33 | 99 | 35 | 39 |
| Ferulic acid | 181 | 182 | 156 | 125 | 81 | 60 | 94 | 74 | 65 |
| Gallic acid | 342 | 223 | 242 | 344 | 114 | 106 | 197 | 78 | 88 |
| Rosmarinic acid | 118 | 104 | 77 | 101 | 47 | 38 | 75 | 44 | 39 |
| Echinacoside | 14 | 10 | 12 | 9 | 9 | 4 | 7 | 5 | 3 |
| Verbascoside | 23 | 3 | 4 | 45 | 12 | 12 | 18 | 1 | 5 |
| Resveratrol | 1108 | 1046 | 676 | 995 | 394 | 346 | 711 | 342 | 361 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olędzka, A.J.; Czerwińska, M.E. Role of Plant-Derived Compounds in the Molecular Pathways Related to Inflammation. Int. J. Mol. Sci. 2023, 24, 4666. https://doi.org/10.3390/ijms24054666
Olędzka AJ, Czerwińska ME. Role of Plant-Derived Compounds in the Molecular Pathways Related to Inflammation. International Journal of Molecular Sciences. 2023; 24(5):4666. https://doi.org/10.3390/ijms24054666
Chicago/Turabian StyleOlędzka, Agata J., and Monika E. Czerwińska. 2023. "Role of Plant-Derived Compounds in the Molecular Pathways Related to Inflammation" International Journal of Molecular Sciences 24, no. 5: 4666. https://doi.org/10.3390/ijms24054666
APA StyleOlędzka, A. J., & Czerwińska, M. E. (2023). Role of Plant-Derived Compounds in the Molecular Pathways Related to Inflammation. International Journal of Molecular Sciences, 24(5), 4666. https://doi.org/10.3390/ijms24054666







