Selenium, Iodine and Iron–Essential Trace Elements for Thyroid Hormone Synthesis and Metabolism
Abstract
1. Introduction
1.1. Thyroid Hormones and Trace Elements
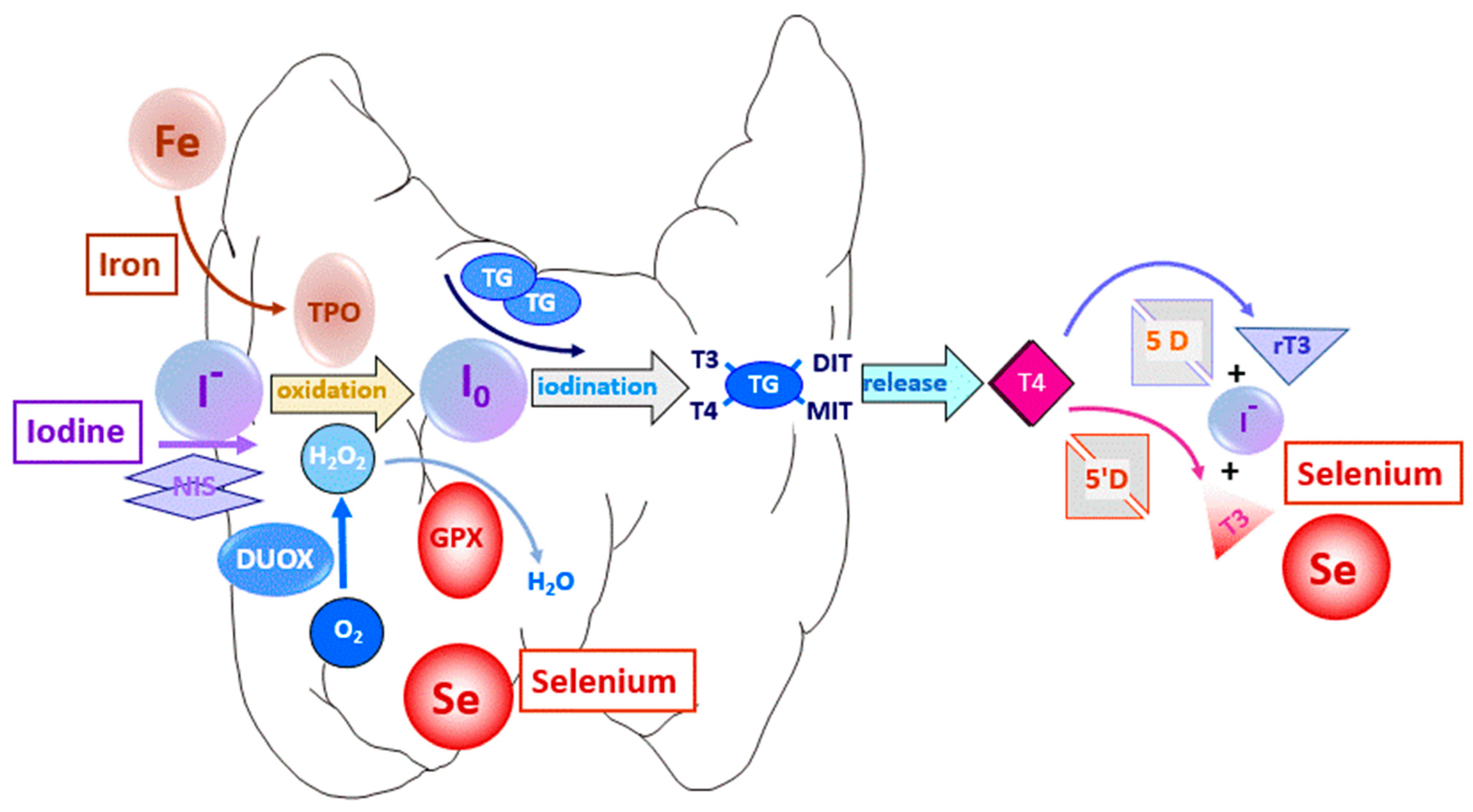
1.2. Trace Element Uptake by Thyrocytes
1.3. Gene Variants Affecting the Thyroid Hormone System
2. Thyroid Function and Iodine Status
Iodine Deficiency
3. Thyroid Function and Selenium Status
3.1. Selenium Deficiency
3.2. Se Status and Thyroid Hormone Concentrations
3.3. Selenium Deficiency and Congenital Hypothyroidism
4. Thyroid Function and Iron Status
Iron Deficiency and Excess
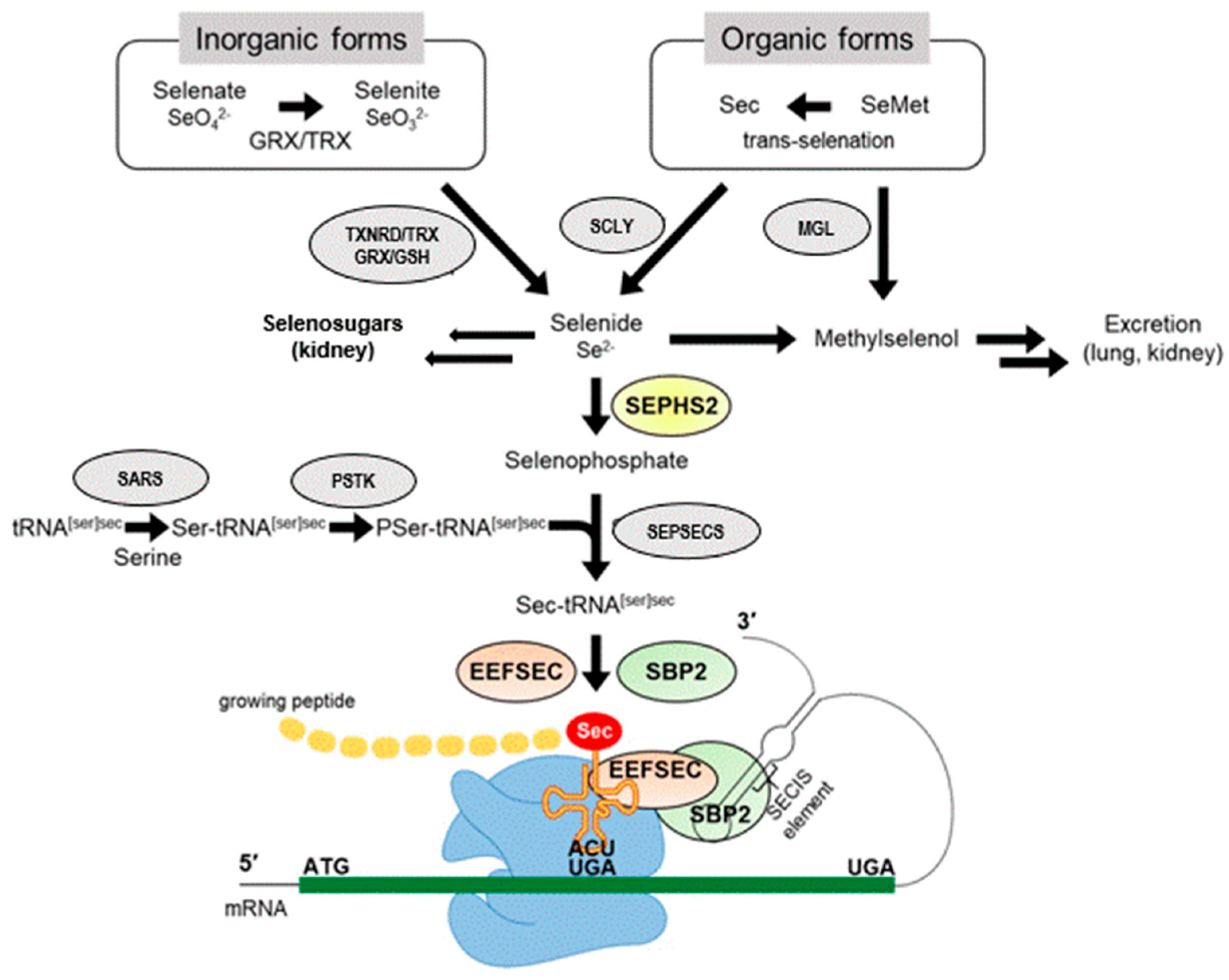
5. Selenoproteins
5.1. Selenoproteins of the Thyroid
5.2. Genetic Models Manipulating Cellular and Systemic Selenium Availability via Selenoprotein Biosynthesis
5.3. Selenoproteins Relevant to the THS
6. Bio-Availability and Bioactivity of Se and Se-Containing Compounds
7. Conclusions and Outlook
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jonklaas, J.; DeSale, S. Levothyroxine prescriptions trends may indicate a downtrend in prescribing. Ther. Adv. Endocrinol. Metab. 2020, 11, 2042018820920551. [Google Scholar] [CrossRef] [PubMed]
- Shobab, L.; Burman, K.D.; Wartofsky, L. Sex Differences in Differentiated Thyroid Cancer. Thyroid 2022, 32, 224–235. [Google Scholar] [CrossRef] [PubMed]
- Gärtner, R. Jod. Ein essenzielles Spurenelement in der Dauerkritik. Ernährungs Umsch. 2021, 68, M702–M711. [Google Scholar]
- Zimmermann, M.B.; Andersson, M. GLOBAL ENDOCRINOLOGY: Global perspectives in endocrinology: Coverage of iodized salt programs and iodine status in 2020. Eur. J. Endocrinol. 2021, 185, R13–R21. [Google Scholar] [CrossRef]
- Kryukov, G.V.; Castellano, S.; Novoselov, S.V.; Lobanov, A.V.; Zehtab, O.; Guigo, R.; Gladyshev, V.N. Characterization of mammalian selenoproteomes. Science 2003, 300, 1439–1443. [Google Scholar] [CrossRef]
- Schmutzler, C.; Mentrup, B.; Schomburg, L.; Hoang-Vu, C.; Herzog, V.; Kohrle, J. Selenoproteins of the thyroid gland: Expression, localization and possible function of glutathione peroxidase 3. Biol. Chem. 2007, 388, 1053–1059. [Google Scholar] [CrossRef]
- Kohrle, J. Local activation and inactivation of thyroid hormones: The deiodinase family. Mol. Cell. Endocrinol. 1999, 151, 103–119. [Google Scholar] [CrossRef]
- Flohé, L.; Toppo, S.; Orian, L. The glutathione peroxidase family: Discoveries and mechanism. Free Radic. Biol. Med. 2022, 187, 113–122. [Google Scholar] [CrossRef]
- Bjørklund, G.; Zou, L.; Wang, J.; Chasapis, C.T.; Peana, M. Thioredoxin reductase as a pharmacological target. Pharmacol. Res. 2021, 174, 105854. [Google Scholar] [CrossRef]
- Köhrle, J.; Frädrich, C. Deiodinases control local cellular and systemic thyroid hormone availability. Free Radic. Biol. Med. 2022, 193 Pt 1, 59–79. [Google Scholar] [CrossRef]
- Winther, K.H.; Rayman, M.P.; Bonnema, S.J.; Hegedüs, L. Selenium in thyroid disorders—Essential knowledge for clinicians. Nat. Rev. Endocrinol. 2020, 16, 165–176. [Google Scholar] [CrossRef]
- Zimmermann, M.B.; Kohrle, J. The impact of iron and selenium deficiencies on iodine and thyroid metabolism: Biochemistry and relevance to public health. Thyroid 2002, 12, 867–878. [Google Scholar] [CrossRef]
- Köhrle, J. Selenium in Endocrinology-Selenoprotein-Related Diseases, Population Studies, and Epidemiological Evidence. Endocrinology 2021, 162, bqaa228. [Google Scholar] [CrossRef]
- Portulano, C.; Paroder-Belenitsky, M.; Carrasco, N. The Na+/I- symporter (NIS): Mechanism and medical impact. Endocr. Rev. 2014, 35, 106–149. [Google Scholar] [CrossRef]
- Ravera, S.; Nicola, J.P.; Salazar-De Simone, G.; Sigworth, F.J.; Karakas, E.; Amzel, L.M.; Bianchet, M.A.; Carrasco, N. Structural insights into the mechanism of the sodium/iodide symporter. Nature 2022, 612, 795–801. [Google Scholar] [CrossRef]
- Wolffram, S.; Ardüser, F.; Scharrer, E. In vivo intestinal absorption of selenate and selenite by rats. J. Nutr. 1985, 115, 454–459. [Google Scholar] [CrossRef]
- Vendeland, S.C.; Deagen, J.T.; Butler, J.A.; Whanger, P.D. Uptake of selenite, selenomethionine and selenate by brush border membrane vesicles isolated from rat small intestine. Biometals 1994, 7, 305–312. [Google Scholar] [CrossRef]
- Weekley, C.M.; Harris, H.H. Which form is that? The importance of selenium speciation and metabolism in the prevention and treatment of disease. Chem. Soc. Rev. 2013, 42, 8870–8894. [Google Scholar] [CrossRef]
- Targovnik, H.M.; Citterio, C.E.; Rivolta, C.M. Iodide handling disorders (NIS, TPO, TG, IYD). Best Pract. Res. Clin. Endocrinol. Metab. 2017, 31, 195–212. [Google Scholar] [CrossRef]
- Lee, K.W.; Shin, Y.; Lee, S.; Lee, S. Inherited Disorders of Thyroid Hormone Metabolism Defect Caused by the Dysregulation of Selenoprotein Expression. Front. Endocrinol. 2021, 12, 803024. [Google Scholar] [CrossRef]
- De Sanctis, V.; Soliman, A.T.; Canatan, D.; Yassin, M.A.; Daar, S.; Elsedfy, H.; Di Maio, S.; Raiola, G.; Corrons, J.V.; Kattamis, C. Thyroid Disorders in Homozygous β-Thalassemia: Current Knowledge, Emerging Issues and Open Problems. Mediterr. J. Hematol. Infect. Dis. 2019, 11, e2019029. [Google Scholar] [CrossRef] [PubMed]
- Orgel, M.N. Thyroid Dysfunction in Hemochromatosis. Endocrinology 1936, 20, 839–845. [Google Scholar] [CrossRef]
- Holzer, G.; Roux, N.; Laudet, V. Evolution of ligands, receptors and metabolizing enzymes of thyroid signaling. Mol. Cell. Endocrinol. 2017, 459, 5–13. [Google Scholar] [CrossRef] [PubMed]
- Hatch-McChesney, A.; Lieberman, H.R. Iodine and Iodine Deficiency: A Comprehensive Review of a Re-Emerging Issue. Nutrients 2022, 14, 3474. [Google Scholar] [CrossRef] [PubMed]
- Dennison, J.O.; Oxnard, C.; Obendorf, P. Historical. In Endemic Cretinism; Dennison, J.O., Oxnard, C., Obendorf, P., Eds.; Springer: New York, NY, USA, 2012. [Google Scholar] [CrossRef]
- Völzke, H.; Caron, P.; Dahl, L.; de Castro, J.J.; Erlund, I.; Gaberšček, S.; Gunnarsdottir, I.; Hubalewska-Dydejczyk, A.; Ittermann, T.; Ivanova, L.; et al. Ensuring Effective Prevention of Iodine Deficiency Disorders. Thyroid 2016, 26, 189–196. [Google Scholar] [CrossRef]
- Ittermann, T.; Albrecht, D.; Arohonka, P.; Bilek, R.; de Castro, J.J.; Dahl, L.; Filipsson Nystrom, H.; Gaberscek, S.; Garcia-Fuentes, E.; Gheorghiu, M.L.; et al. Standardized Map of Iodine Status in Europe. Thyroid 2020, 30, 1346–1354. [Google Scholar] [CrossRef]
- Zimmermann, M.B. The remarkable impact of iodisation programmes on global public health. Proc. Nutr. Soc. 2022, 82, 1–7. [Google Scholar] [CrossRef]
- Levie, D.; Korevaar, T.I.M.; Bath, S.C.; Murcia, M.; Dineva, M.; Llop, S.; Espada, M.; van Herwaarden, A.E.; de Rijke, Y.B.; Ibarluzea, J.M.; et al. Association of Maternal Iodine Status With Child IQ: A Meta-Analysis of Individual Participant Data. J. Clin. Endocrinol. Metab. 2019, 104, 5957–5967. [Google Scholar] [CrossRef]
- Gorstein, J.L.; Bagriansky, J.; Pearce, E.N.; Kupka, R.; Zimmermann, M.B. Estimating the Health and Economic Benefits of Universal Salt Iodization Programs to Correct Iodine Deficiency Disorders. Thyroid 2020, 30, 1802–1809. [Google Scholar] [CrossRef]
- Santi, C.; Bagnoli, L. Celebrating Two Centuries of Research in Selenium Chemistry: State of the Art and New Prospective. Molecules 2017, 22, 2124. [Google Scholar] [CrossRef]
- Corvilain, B.; Contempré, B.; Longombé, A.O.; Goyens, P.; Gervy-Decoster, C.; Lamy, F.; Vanderpas, J.B.; Dumont, J.E. Selenium and the thyroid: How the relationship was established. Am. J. Clin. Nutr. 1993, 57 (Suppl. 2), 244S–248S. [Google Scholar] [CrossRef]
- Boyages, S.C.; Halpern, J.P. Endemic cretinism: Toward a unifying hypothesis. Thyroid 1993, 3, 59–69. [Google Scholar] [CrossRef]
- Schweizer, U.; Chiu, J.; Köhrle, J. Peroxides and peroxide-degrading enzymes in the thyroid. Antioxid. Redox. Signal 2008, 10, 1577–1592. [Google Scholar] [CrossRef]
- Mammen, J.S.R.; Cappola, A.R. Autoimmune Thyroid Disease in Women. JAMA 2021, 325, 2392–2393. [Google Scholar] [CrossRef]
- Kohrle, J.; Gartner, R. Selenium and thyroid. Best Pract. Res. Clin. Endocrinol. Metab. 2009, 23, 815–827. [Google Scholar] [CrossRef]
- Liu, F.; Wang, K.; Nie, J.; Feng, Q.; Li, X.; Yang, Y.; Deng, M.G.; Zhou, H.; Wang, S. Relationship between dietary selenium intake and serum thyroid function measures in U.S. adults: Data from NHANES 2007–2012. Front. Nutr. 2022, 9, 1002489. [Google Scholar] [CrossRef]
- Andrade, G.R.G.; Gorgulho, B.; Lotufo, P.A.; Bensenor, I.M.; Marchioni, D.M. Dietary Selenium Intake and Subclinical Hypothyroidism: A Cross-Sectional Analysis of the ELSA-Brasil Study. Nutrients 2018, 10, 693. [Google Scholar] [CrossRef]
- Winther, K.H.; Bonnema, S.J.; Cold, F.; Debrabant, B.; Nybo, M.; Cold, S.; Hegedüs, L. Does selenium supplementation affect thyroid function? Results from a randomized, controlled, double-blinded trial in a Danish population. Eur. J. Endocrinol. 2015, 172, 657–667. [Google Scholar] [CrossRef]
- Wu, Q.; Wang, Y.; Chen, P.; Wei, J.; Lv., H.; Wang., S.; Wu, Y.; Zhao, X.; Peng, X.; Rijntjes, E.; et al. Increased incidence of Hashimoto’s thyroiditis in selenium deficiency: A prospective six-year cohort study. J. Clin. Endocrinol. Metab. 2022, 107, e3603–e3611. [Google Scholar] [CrossRef]
- Wu, Q.; Rayman, M.P.; Lv, H.; Schomburg, L.; Cui, B.; Gao, C.; Chen, P.; Zhuang, G.; Zhang, Z.; Peng, X.; et al. Low Population Selenium Status Is Associated With Increased Prevalence of Thyroid Disease. J. Clin. Endocrinol. Metab. 2015, 100, 4037–4047. [Google Scholar] [CrossRef]
- Zheng, G.; Cai, Y.; Guo, Y.; Song, F.; Hu, Y.; Li, L.; Zhu, L. The association between dietary selenium intake and Hashimoto’s thyroiditis among US adults: National Health and Nutrition Examination Survey (NHANES), 2007-2012. J. Endocrinol. Investig. 2022; ahead of print. [Google Scholar] [CrossRef]
- Contempre, B.; Duale, N.L.; Dumont, J.E.; Ngo, B.; Diplock, A.T.; Vanderpas, J. Effect of selenium supplementation on thyroid hormone metabolism in an iodine and selenium deficient population. Clin. Endocrinol. 1992, 36, 579–583. [Google Scholar] [CrossRef] [PubMed]
- Contempre, B.; Le Moine, O.; Dumont, J.E.; Denef, J.F.; Many, M.C. Selenium deficiency and thyroid fibrosis. A key role for macrophages and transforming growth factor beta (TGF-beta). Mol. Cell. Endocrinol. 1996, 124, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Contempre, B.; Dumont, J.E.; Denef, J.F.; Many, M.C. Effects of selenium deficiency on thyroid necrosis, fibrosis and proliferation: A possible role in myxoedematous cretinism. Eur. J. Endocrinol. 1995, 133, 99–109. [Google Scholar] [CrossRef] [PubMed]
- Lossow, K.; Renko, K.; Schwarz, M.; Schomburg, L.; Schwerdtle, T.; Kipp, A.P. The Nutritional Supply of Iodine and Selenium Affects Thyroid Hormone Axis Related Endpoints in Mice. Nutrients 2021, 13, 373. [Google Scholar] [CrossRef] [PubMed]
- Godlewska, M.; Banga, P.J. Thyroid peroxidase as a dual active site enzyme: Focus on biosynthesis, hormonogenesis and thyroid disorders of autoimmunity and cancer. Biochimie 2019, 160, 34–45. [Google Scholar] [CrossRef]
- De Deken, X.; Miot, F. DUOX Defects and Their Roles in Congenital Hypothyroidism. Methods Mol. Biol. 2019, 1982, 667–693. [Google Scholar]
- Song, Y.; Driessens, N.; Costa, M.; De Deken, X.; Detours, V.; Corvilain, B.; Maenhaut, C.; Miot, F.; Van Sande, J.; Many, M.C.; et al. Roles of hydrogen peroxide in thyroid physiology and disease. J. Clin. Endocrinol. Metab. 2007, 92, 3764–3773. [Google Scholar] [CrossRef]
- Vassart, G.; Dumont, J.E. The thyrotropin receptor and the regulation of thyrocyte function and growth. Endocr. Rev. 1992, 13, 596–611. [Google Scholar]
- Luo, J.; Wang, X.; Yuan, L.; Guo, L. Iron Deficiency, a Risk Factor of Thyroid Disorders in Reproductive-Age and Pregnant Women: A Systematic Review and Meta-Analysis. Front. Endocrinol. 2021, 12, 629831. [Google Scholar] [CrossRef]
- Zimmermann, M.B.; Burgi, H.; Hurrell, R.F. Iron deficiency predicts poor maternal thyroid status during pregnancy. J. Clin. Endocrinol. Metab. 2007, 92, 3436–3440. [Google Scholar] [CrossRef]
- Yu, X.; Shan, Z.; Li, C.; Mao, J.; Wang, W.; Xie, X.; Liu, A.; Teng, X.; Zhou, W.; Li, C.; et al. Iron deficiency, an independent risk factor for isolated hypothyroxinemia in pregnant and nonpregnant women of childbearing age in China. J. Clin. Endocrinol. Metab. 2015, 100, 1594–1601. [Google Scholar] [CrossRef]
- van Gucht, A.L.M.; Meima, M.E.; Moran, C.; Agostini, M.; Tylki-Szymanska, A.; Krajewska, M.W.; Chrzanowska, K.; Efthymiadou, A.; Chrysis, D.; Demir, K.; et al. Anemia in Patients with Resistance to Thyroid Hormone α: A Role for Thyroid Hormone Receptor α in Human Erythropoiesis. J. Clin. Endocrinol. Metab. 2017, 102, 3517–3525. [Google Scholar] [CrossRef]
- Pastori, V.; Pozzi, S.; Labedz, A.; Ahmed, S.; Ronchi, A.E. Role of Nuclear Receptors in Controlling Erythropoiesis. Int. J. Mol. Sci. 2022, 23, 2800. [Google Scholar] [CrossRef]
- Rayman, M.P. Multiple nutritional factors and thyroid disease, with particular reference to autoimmune thyroid disease. Proc. Nutr. Soc. 2019, 78, 34–44. [Google Scholar] [CrossRef]
- Ravanbod, M.; Asadipooya, K.; Kalantarhormozi, M.; Nabipour, I.; Omrani, G.R. Treatment of iron-deficiency anemia in patients with subclinical hypothyroidism. Am. J. Med. 2013, 126, 420–424. [Google Scholar] [CrossRef]
- Cinemre, H.; Bilir, C.; Gokosmanoglu, F.; Bahcebasi, T. Hematologic effects of levothyroxine in iron-deficient subclinical hypothyroid patients: A randomized, double-blind, controlled study. J. Clin. Endocrinol. Metab. 2009, 94, 151–156. [Google Scholar] [CrossRef]
- Labunskyy, V.M.; Hatfield, D.L.; Gladyshev, V.N. Selenoproteins: Molecular pathways and physiological roles. Physiol. Rev. 2014, 94, 739–777. [Google Scholar] [CrossRef]
- Schweizer, U.; Fradejas-Villar, N. Why 21? The significance of selenoproteins for human health revealed by inborn errors of metabolism. FASEB J. 2016, 30, 3669–3681. [Google Scholar] [CrossRef]
- Schoenmakers, E.; Chatterjee, K. Human Genetic Disorders Resulting in Systemic Selenoprotein Deficiency. Int. J. Mol. Sci. 2021, 22, 12927. [Google Scholar] [CrossRef]
- Patterson, B.H.; Combs, G.F., Jr.; Taylor, P.R.; Patterson, K.Y.; Moler, J.E.; Wastney, M.E. Selenium Kinetics in Humans Change Following 2 Years of Supplementation with Selenomethionine. Front. Endocrinol. 2021, 12, 621687. [Google Scholar] [CrossRef]
- Seale, L.A. Selenocysteine β-Lyase: Biochemistry, Regulation and Physiological Role of the Selenocysteine Decomposition Enzyme. Antioxidants 2019, 8, 357. [Google Scholar] [CrossRef] [PubMed]
- Le, D.T.; Liang, X.; Fomenko, D.E.; Raza, A.S.; Chong, C.K.; Carlson, B.A.; Hatfield, D.L.; Gladyshev, V.N. Analysis of methionine/selenomethionine oxidation and methionine sulfoxide reductase function using methionine-rich proteins and antibodies against their oxidized forms. Biochemistry 2008, 47, 6685–6694. [Google Scholar] [CrossRef] [PubMed]
- Nasim, M.J.; Zuraik, M.M.; Abdin, A.Y.; Ney, Y.; Jacob, C. Selenomethionine: A Pink Trojan Redox Horse with Implications in Aging and Various Age-Related Diseases. Antioxidants 2021, 10, 882. [Google Scholar] [CrossRef] [PubMed]
- Tarrago, L.; Kaya, A.; Kim, H.Y.; Manta, B.; Lee, B.C.; Gladyshev, V.N. The selenoprotein methionine sulfoxide reductase B1 (MSRB1). Free Radic. Biol. Med. 2022, 191, 228–240. [Google Scholar] [CrossRef] [PubMed]
- Rayman, M.P. Selenium intake, status, and health: A complex relationship. Hormones 2020, 19, 9–14. [Google Scholar] [CrossRef]
- Toh, P.; Nicholson, J.L.; Vetter, A.M.; Berry, M.J.; Torres, D.J. Selenium in Bodily Homeostasis: Hypothalamus, Hormones, and Highways of Communication. Int. J. Mol. Sci. 2022, 23, 15445. [Google Scholar] [CrossRef]
- Kang, D.; Lee, J.; Wu, C.; Guo, X.; Lee, B.J.; Chun, J.S.; Kim, J.H. The role of selenium metabolism and selenoproteins in cartilage homeostasis and arthropathies. Exp. Mol. Med. 2020, 52, 1198–1208. [Google Scholar] [CrossRef]
- Dickson, R.C.; Tomlinson, R.H. Selenium in blood and human tissues. Clin. Chim. Acta. 1967, 16, 311–321. [Google Scholar] [CrossRef]
- Ralston, N.V. Effects of soft electrophiles on selenium physiology. Free Radic. Biol. Med. 2018, 127, 134–144. [Google Scholar] [CrossRef]
- Spiller, H.A. Rethinking mercury: The role of selenium in the pathophysiology of mercury toxicity. Clin. Toxicol. 2018, 56, 313–326. [Google Scholar] [CrossRef]
- Ito, M.; Toyoda, N.; Nomura, E.; Takamura, Y.; Amino, N.; Iwasaka, T.; Takamatsu, J.; Miyauchi, A.; Nishikawa, M. Type 1 and type 2 iodothyronine deiodinases in the thyroid gland of patients with 3,5,3′-triiodothyronine-predominant Graves’ disease. Eur. J. Endocrinol. 2011, 164, 95–100. [Google Scholar] [CrossRef]
- Ishii, H.; Inada, M.; Tanaka, K.; Mashio, Y.; Naito, K.; Nishikawa, M.; Matsuzuka, F.; Kuma, K.; Imura, H. Induction of outer and inner ring monodeiodinases in human thyroid gland by thyrotropin. J. Clin. Endocrinol. Metab. 1983, 57, 500–505. [Google Scholar] [CrossRef]
- Citterio, C.E.; Targovnik, H.M.; Arvan, P. The role of thyroglobulin in thyroid hormonogenesis. Nat. Rev. Endocrinol. 2019, 15, 323–338. [Google Scholar] [CrossRef]
- Jehan, C.; Cartier, D.; Bucharles, C.; Anouar, Y.; Lihrmann, I. Emerging roles of ER-resident selenoproteins in brain physiology and physiopathology. Redox Biol. 2022, 55, 102412. [Google Scholar] [CrossRef]
- Rua, R.M.; Nogales, F.; Carreras, O.; Ojeda, M.L. Selenium, selenoproteins and cancer of the thyroid. J. Trace Elem. Med. Biol. 2022, 76, 127115. [Google Scholar] [CrossRef]
- Conrad, M.; Schweizer, U. Mouse Models that Target Individual Selenoproteins. In Selenium; Hatfield, D., Schweizer, U., Tsuji, P., Gladyshev, V., Eds.; Springer: Cham, Switzerland, 2016. [Google Scholar]
- Kasaikina, M.V.; Hatfield, D.L.; Gladyshev, V.N. Understanding selenoprotein function and regulation through the use of rodent models. Biochim. Biophys. Acta 2012, 1823, 1633–1642. [Google Scholar] [CrossRef]
- Moustafa, M.E.; Kumaraswamy, E.; Zhong, N.; Rao, M.; Carlson, B.A.; Hatfield, D.L. Models for assessing the role of selenoproteins in health. J. Nutr. 2003, 133 (Suppl. 7), 2494S–2496S. [Google Scholar] [CrossRef]
- Seale, L.A.; Hashimoto, A.C.; Kurokawa, S.; Gilman, C.L.; Seyedali, A.; Bellinger, F.P.; Raman, A.V.; Berry, M.J. Disruption of the selenocysteine lyase-mediated selenium recycling pathway leads to metabolic syndrome in mice. Mol. Cell. Biol. 2012, 32, 4141–4154. [Google Scholar] [CrossRef]
- Fujisawa, H.; Korwutthikulrangsri, M.; Fu, J.; Liao, X.H.; Dumitrescu, A.M. Role of the Thyroid Gland in Expression of the Thyroid Phenotype of Sbp2-Deficient Mice. Endocrinology 2020, 161, bqz032. [Google Scholar] [CrossRef]
- Fradejas-Villar, N. Consequences of mutations and inborn errors of selenoprotein biosynthesis and functions. Free Radic. Biol. Med. 2018, 127, 206–214. [Google Scholar] [CrossRef]
- Chiu-Ugalde, J.; Wirth, E.K.; Klein, M.O.; Sapin, R.; Fradejas-Villar, N.; Renko, K.; Schomburg, L.; Köhrle, J.; Schweizer, U. Thyroid function is maintained despite increased oxidative stress in mice lacking selenoprotein biosynthesis in thyroid epithelial cells. Antioxid. Redox Signal 2012, 17, 902–913. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Stefan-Lifshitz, M.; Li, C.W.; Tomer, Y. Genetics and epigenetics of autoimmune thyroid diseases: Translational implications. Best Pract. Res. Clin. Endocrinol. Metab. 2022, 101661. [Google Scholar] [CrossRef] [PubMed]
- Krassas, G.E.; Markou, K.B. The impact of thyroid diseases starting from birth on reproductive function. Hormones 2019, 18, 365–381. [Google Scholar] [CrossRef] [PubMed]
- Schomburg, L.; Riese, C.; Michaelis, M.; Griebert, E.; Klein, M.O.; Sapin, R.; Schweizer, U.; Köhrle, J. Synthesis and metabolism of thyroid hormones is preferentially maintained in selenium-deficient transgenic mice. Endocrinology 2006, 147, 1306–1313. [Google Scholar] [CrossRef]
- Behne, D.; Hilmert, H.; Scheid, S.; Gessner, H.; Elger, W. Evidence for specific selenium target tissues and new biologically important selenoproteins. Biochim. Biophys. Acta 1988, 966, 12–21. [Google Scholar] [CrossRef]
- Bermano, G.; Nicol, F.; Dyer, J.A.; Sunde, R.A.; Beckett, G.J.; Arthur, J.R.; Hesketh, J.E. Tissue-specific regulation of selenoenzyme gene expression during selenium deficiency in rats. Biochem. J. 1995, 311, 425–430. [Google Scholar] [CrossRef]
- Jongejan, R.M.S.; Meima, M.E.; Visser, W.E.; Korevaar, T.I.M.; van den Berg, S.A.A.; Peeters, R.P.; de Rijke, Y.B. Binding Characteristics of Thyroid Hormone Distributor Proteins to Thyroid Hormone Metabolites. Thyroid 2022, 32, 990–999. [Google Scholar] [CrossRef]
- Yamauchi, K. The interaction of zinc with the multi-functional plasma thyroid hormone distributor protein, transthyretin: Evolutionary and cross-species comparative aspects. Biometals 2021, 34, 423–437. [Google Scholar] [CrossRef]
- Papp, L.V.; Lu, J.; Holmgren, A.; Khanna, K.K. From selenium to selenoproteins: Synthesis, identity, and their role in human health. Antioxid. Redox Signal 2007, 9, 775–806. [Google Scholar] [CrossRef]
- Schomburg, L.; Schweizer, U. Hierarchical regulation of selenoprotein expression and sex-specific effects of selenium. Biochim. Biophys. Acta 2009, 1790, 1453–1462. [Google Scholar] [CrossRef]
- Chanoine, J.P.; Safran, M.; Farwell, A.P.; Dubord, S.; Alex, S.; Stone, S.; Arthur, J.R.; Braverman, L.E.; Leonard, J.L. Effects of selenium deficiency on thyroid hormone economy in rats. Endocrinology 1992, 131, 1787–1792. [Google Scholar] [CrossRef]
- Wu, S.Y.; Huang, W.S.; Chopra, I.J.; Jordan, M.; Alvarez, D.; Santini, F. Sulfation pathway of thyroid hormone metabolism in selenium-deficient male rats. Am. J. Physiol. 1995, 268 Pt 1, E572–E579. [Google Scholar] [CrossRef]
- Buitendijk, M.; Galton, V.A. Is the kidney a major storage site for thyroxine as thyroxine glucuronide? Thyroid 2012, 22, 187–191. [Google Scholar] [CrossRef]
- Flamant, F.; Gauthier, K. Thyroid hormone receptors: The challenge of elucidating isotype-specific functions and cell-specific response. Biochim. Biophys. Acta 2013, 1830, 3900–3907. [Google Scholar] [CrossRef]
- Videla, L.A.; Valenzuela, R. Perspectives in liver redox imbalance: Toxicological and pharmacological aspects underlying iron overloading, nonalcoholic fatty liver disease, and thyroid hormone action. Biofactors 2022, 48, 400–415. [Google Scholar] [CrossRef]
- Köhrle, J.; Oertel, M.; Gross, M. Selenium supply regulates thyroid function, thyroid hormone synthesis and metabolism by altering the expression of the selenoenzymes Type I 5’-deiodinase and glutathione peroxidase. Thyroidology 1992, 4, 17–21. [Google Scholar]
- Köhrle, J. Selenium and the control of thyroid hormone metabolism. Thyroid 2005, 15, 841–853. [Google Scholar] [CrossRef]
- Santesmasses, D.; Gladyshev, V.N. Pathogenic Variants in Selenoproteins and Selenocysteine Biosynthesis Machinery. Int. J. Mol. Sci. 2021, 22, 11593. [Google Scholar] [CrossRef]
- Schomburg, L. The other view: The trace element selenium as a micronutrient in thyroid disease, diabetes, and beyond. Hormones 2020, 19, 15–24. [Google Scholar] [CrossRef]
- Constantinescu-Aruxandei, D.; Frîncu, R.M.; Capră, L.; Oancea, F. Selenium Analysis and Speciation in Dietary Supplements Based on Next-Generation Selenium Ingredients. Nutrients 2018, 10, 1466. [Google Scholar] [CrossRef]
- Hosnedlova, B.; Kepinska, M.; Skalickova, S.; Fernandez, C.; Ruttkay-Nedecky, B.; Peng, Q.; Baron, M.; Melcova, M.; Opatrilova, R.; Zidkova, J.; et al. Nano-selenium and its nanomedicine applications: A critical review. Int. J. Nanomed. 2018, 13, 2107–2128. [Google Scholar] [CrossRef] [PubMed]
- Sherif, N.A.E.; El-Banna, A.; Abdel-Moneim, R.A.; Sobh, Z.K.; Balah, M.I.F. The possible thyroid disruptive effect of di-(2-ethyl hexyl) phthalate and the potential protective role of selenium and curcumin nanoparticles: A toxicological and histological study. Toxicol. Res. 2022, 11, 108–121. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.; Zhang, J.; Xu, J.F.; Pi, J. The Advancing of Selenium Nanoparticles Against Infectious Diseases. Front. Pharmacol. 2021, 12, 682284. [Google Scholar] [CrossRef] [PubMed]
- Turovsky, E.A.; Mal’tseva, V.N.; Sarimov, R.M.; Simakin, A.V.; Gudkov, S.V.; Plotnikov, E.Y. Features of the cytoprotective effect of selenium nanoparticles on primary cortical neurons and astrocytes during oxygen-glucose deprivation and reoxygenation. Sci. Rep. 2022, 12, 1710. [Google Scholar] [CrossRef]
- Khurana, A.; Tekula, S.; Saifi, M.A.; Venkatesh, P.; Godugu, C. Therapeutic applications of selenium nanoparticles. Biomed. Pharmacother. 2019, 111, 802–812. [Google Scholar] [CrossRef]
- Raymond, L.J.; Ralston, N.V.C. Mercury: Selenium interactions and health implications. Neurotoxicology 2020, 81, 294–299. [Google Scholar] [CrossRef]
- Ralston, N.V.C. Concomitant selenoenzyme inhibitor exposures as etiologic contributors to disease: Implications for preventative medicine. Arch. Biochem. Biophys. 2023, 733, 109469. [Google Scholar] [CrossRef]
- Schaffner, M.; Mühlberger, N.; Conrads-Frank, A.; Qerimi Rushaj, V.; Sroczynski, G.; Koukkou, E.; Heinsbaek Thuesen, B.; Völzke, H.; Oberaigner, W.; Siebert, U.; et al. Benefits and Harms of a Prevention Program for Iodine Deficiency Disorders: Predictions of the Decision-Analytic EUthyroid Model. Thyroid 2021, 31, 494–508. [Google Scholar] [CrossRef]
- Chaker, L.; Razvi, S.; Bensenor, I.M.; Azizi, F.; Pearce, E.N.; Peeters, R.P. Hypothyroidism. Nat. Rev. Dis. Primers. 2022, 8, 30. [Google Scholar] [CrossRef]
- Dijck-Brouwer, D.A.J.; Muskiet, F.A.J.; Verheesen, R.H.; Schaafsma, G.; Schaafsma, A.; Geurts, J.M.W. Thyroidal and Extrathyroidal Requirements for Iodine and Selenium: A Combined Evolutionary and (Patho)Physiological Approach. Nutrients 2022, 14, 3886. [Google Scholar] [CrossRef]
- Zhou, Q.; Xue, S.; Zhang, L.; Chen, G. Trace elements and the thyroid. Front. Endocrinol. 2022, 13, 904889. [Google Scholar] [CrossRef]
- Schomburg, L. Selenoprotein P - Selenium transport protein, enzyme and biomarker of selenium status. Free Radic. Biol. Med. 2022, 191, 150–163. [Google Scholar] [CrossRef]
- Ogun, A.S.; Adeyinka, A. Biochemistry, Transferrin. In StatPearls; StatPearls Publishing LLC.: Treasure Island, FL, USA, 2022. [Google Scholar]
- Szklarz, M.; Gontarz-Nowak, K.; Matuszewski, W.; Bandurska-Stankiewicz, E. Iron: Not Just a Passive Bystander in AITD. Nutrients 2022, 14, 4682. [Google Scholar] [CrossRef]
- Brauer, V.F.; Schweizer, U.; Köhrle, J.; Paschke, R. Selenium and goiter prevalence in borderline iodine sufficiency. Eur. J. Endocrinol. 2006, 155, 807–812. [Google Scholar] [CrossRef][Green Version]
- Vanderpas, J.B.; Contempré, B.; Duale, N.L.; Deckx, H.; Bebe, N.; Longombé, A.O.; Thilly, C.H.; Diplock, A.T.; Dumont, J.E. Selenium deficiency mitigates hypothyroxinemia in iodine-deficient subjects. Am. J. Clin. Nutr. 1993, 57 (Suppl. 2), 271S–275S. [Google Scholar] [CrossRef]
- Arthur, J.R.; Beckett, G.J.; Mitchell, J.H. The interactions between selenium and iodine deficiencies in man and animals. Nutr. Res. Rev. 1999, 12, 55–73. [Google Scholar] [CrossRef]
- Dumont, J.E.; Corvilain, B.; Contempre, B. The biochemistry of endemic cretinism: Roles of iodine and selenium deficiency and goitrogens. Mol. Cell. Endocrinol. 1994, 100, 163–166. [Google Scholar] [CrossRef]
- Köhrle, J.; Jakob, F.; Contempré, B.; Dumont, J.E. Selenium, the thyroid, and the endocrine system. Endocr. Rev. 2005, 26, 944–984. [Google Scholar] [CrossRef]
- Tsatsoulis, A. The role of iodine vs selenium on the rising trend of autoimmune thyroiditis in iodine sufficient countries. Endocrinol. Metab. Int. J. 2018, 6, 412–414. [Google Scholar] [CrossRef]
- Contempré, B.; Morreale de Escobar, G.M.; Denef, J.F.; Dumont, J.E.; Many, M.C. Thiocyanate induces cell necrosis and fibrosis in selenium- and iodine-deficient rat thyroids: A potential experimental model for myxedematous endemic cretinism in central Africa. Endocrinology 2004, 145, 994–1002. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Köhrle, J. Selenium, Iodine and Iron–Essential Trace Elements for Thyroid Hormone Synthesis and Metabolism. Int. J. Mol. Sci. 2023, 24, 3393. https://doi.org/10.3390/ijms24043393
Köhrle J. Selenium, Iodine and Iron–Essential Trace Elements for Thyroid Hormone Synthesis and Metabolism. International Journal of Molecular Sciences. 2023; 24(4):3393. https://doi.org/10.3390/ijms24043393
Chicago/Turabian StyleKöhrle, Josef. 2023. "Selenium, Iodine and Iron–Essential Trace Elements for Thyroid Hormone Synthesis and Metabolism" International Journal of Molecular Sciences 24, no. 4: 3393. https://doi.org/10.3390/ijms24043393
APA StyleKöhrle, J. (2023). Selenium, Iodine and Iron–Essential Trace Elements for Thyroid Hormone Synthesis and Metabolism. International Journal of Molecular Sciences, 24(4), 3393. https://doi.org/10.3390/ijms24043393





