A Novel Cuticular Protein-like Cpr21L Is Essential for Nymph Survival and Male Fecundity in the Brown Planthopper
Abstract
1. Introduction
2. Results
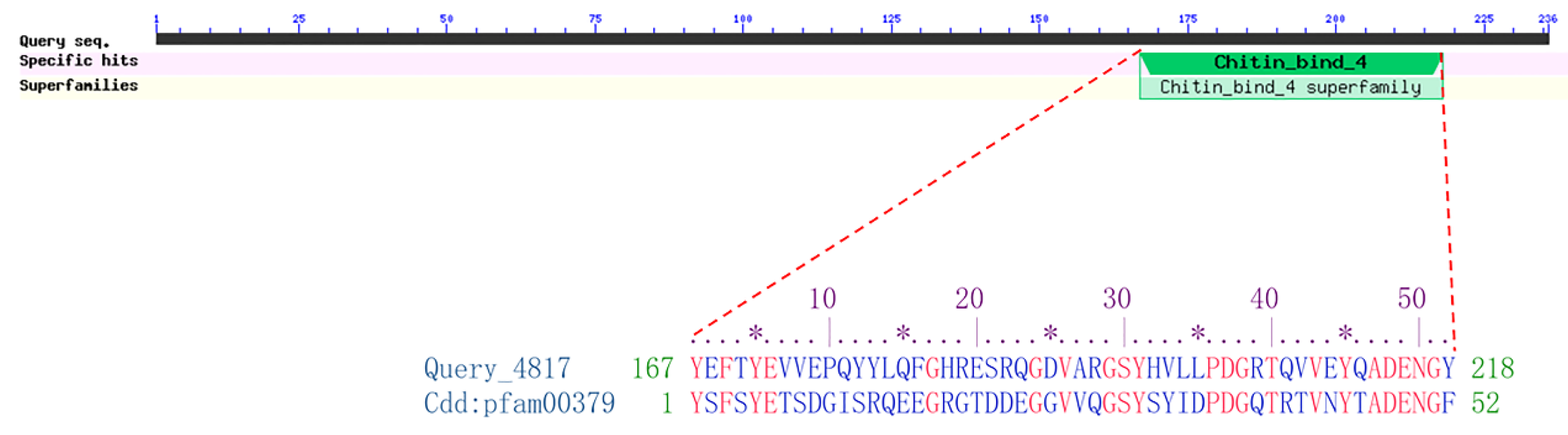
2.1. Sequence and Phylogenetic Analysis
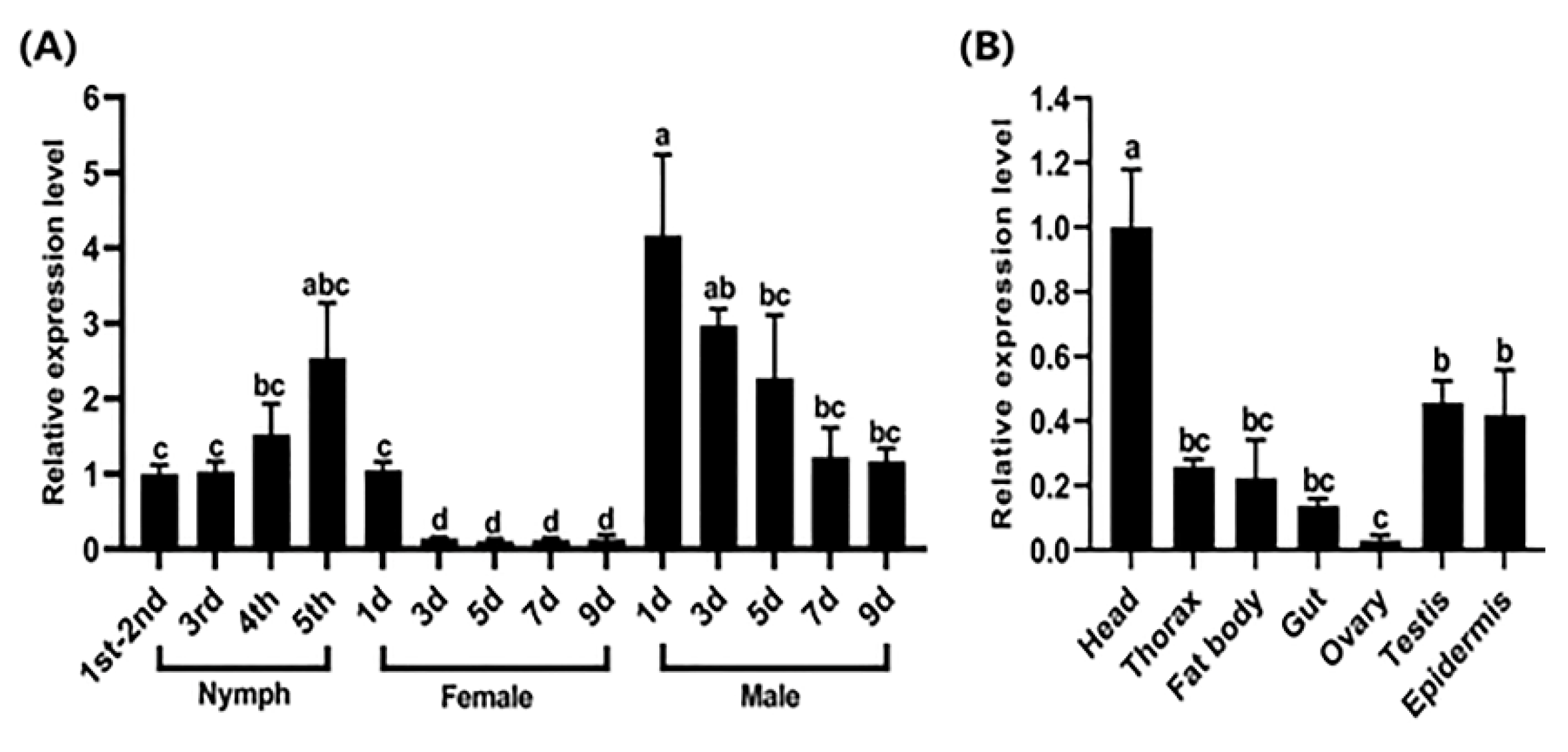
2.2. Spatiotemporal Expression Patterns of the Cpr21L Gene
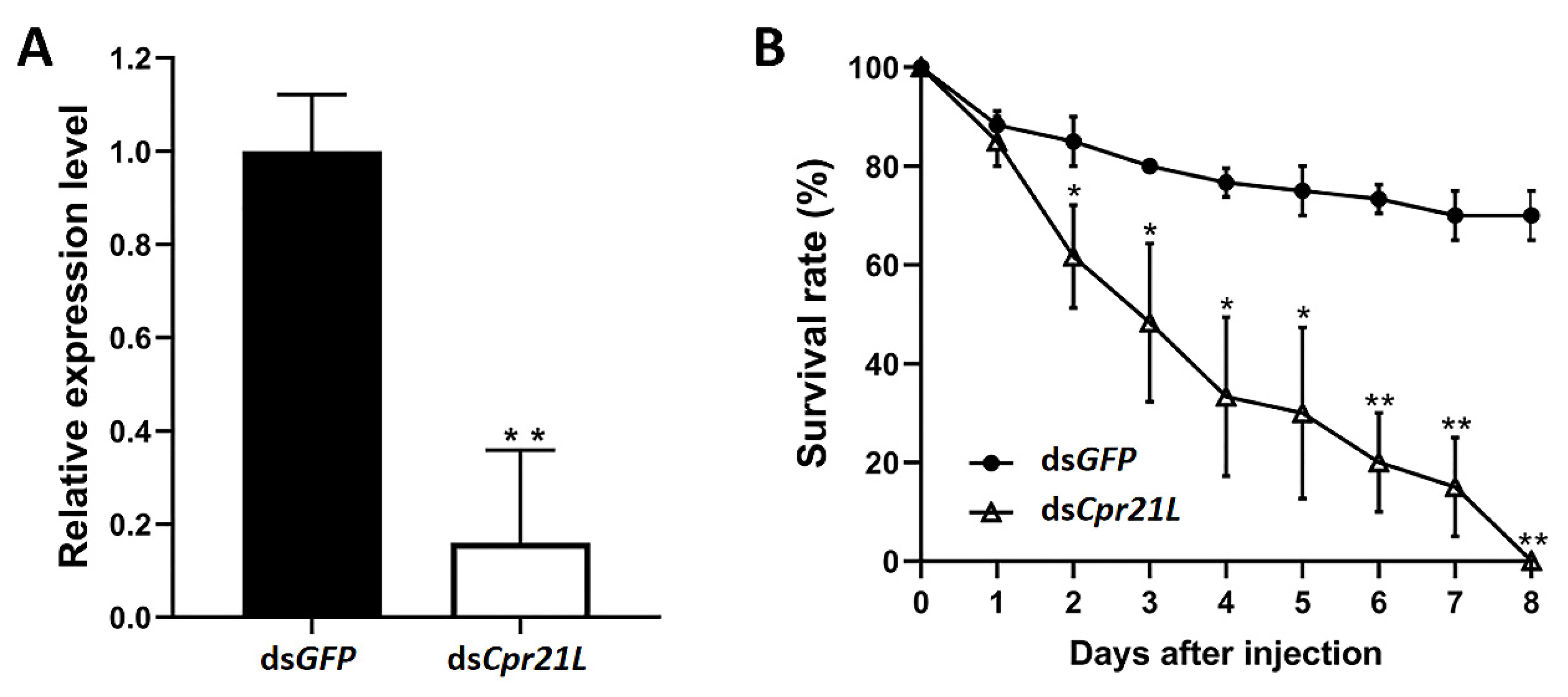
2.3. Effects of Cpr21L Knockdown on the Survival and Development of BPH
2.4. Effects of Cpr21L Knockdown on the Structure of the Epidermis and Cuticle
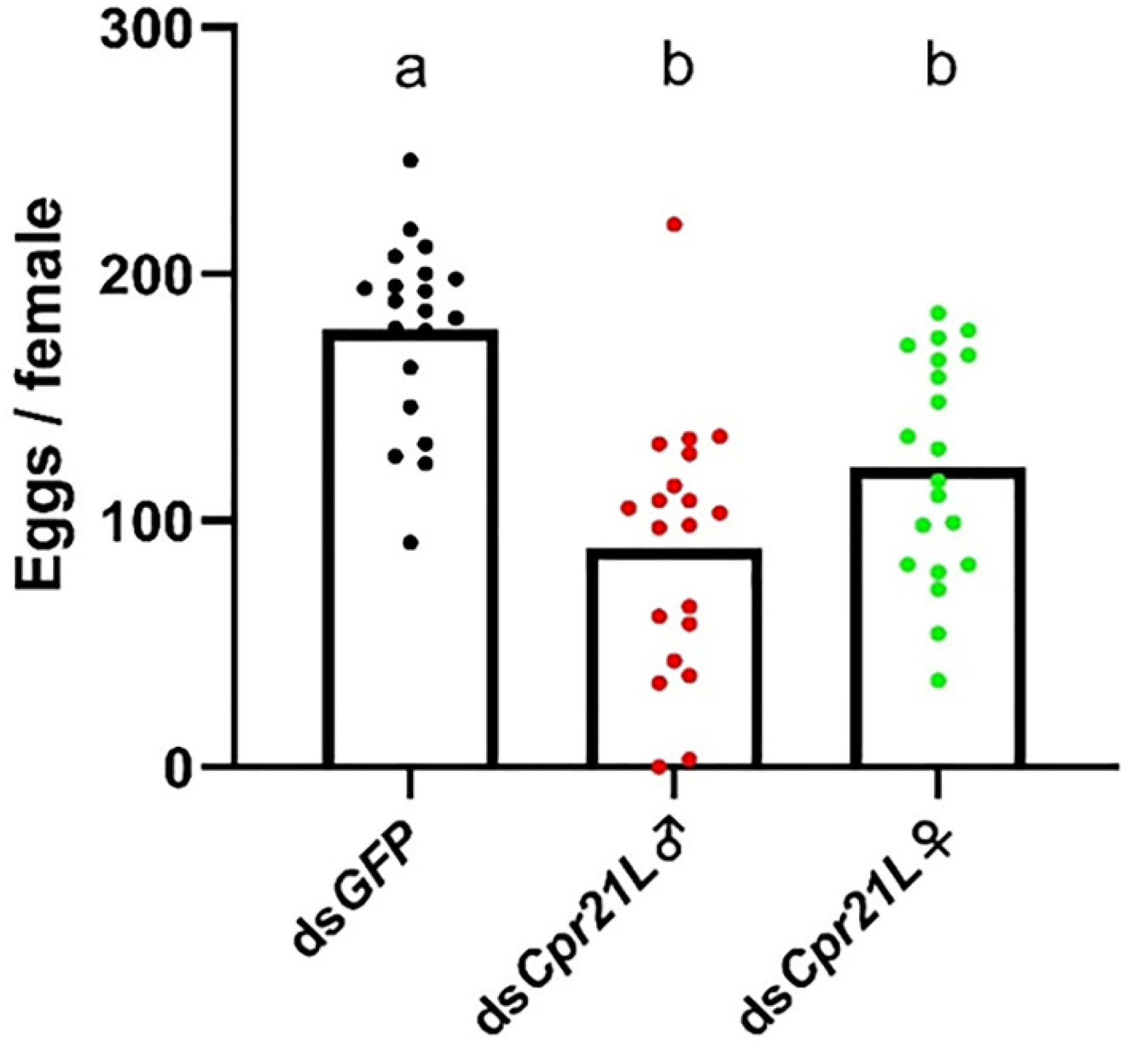
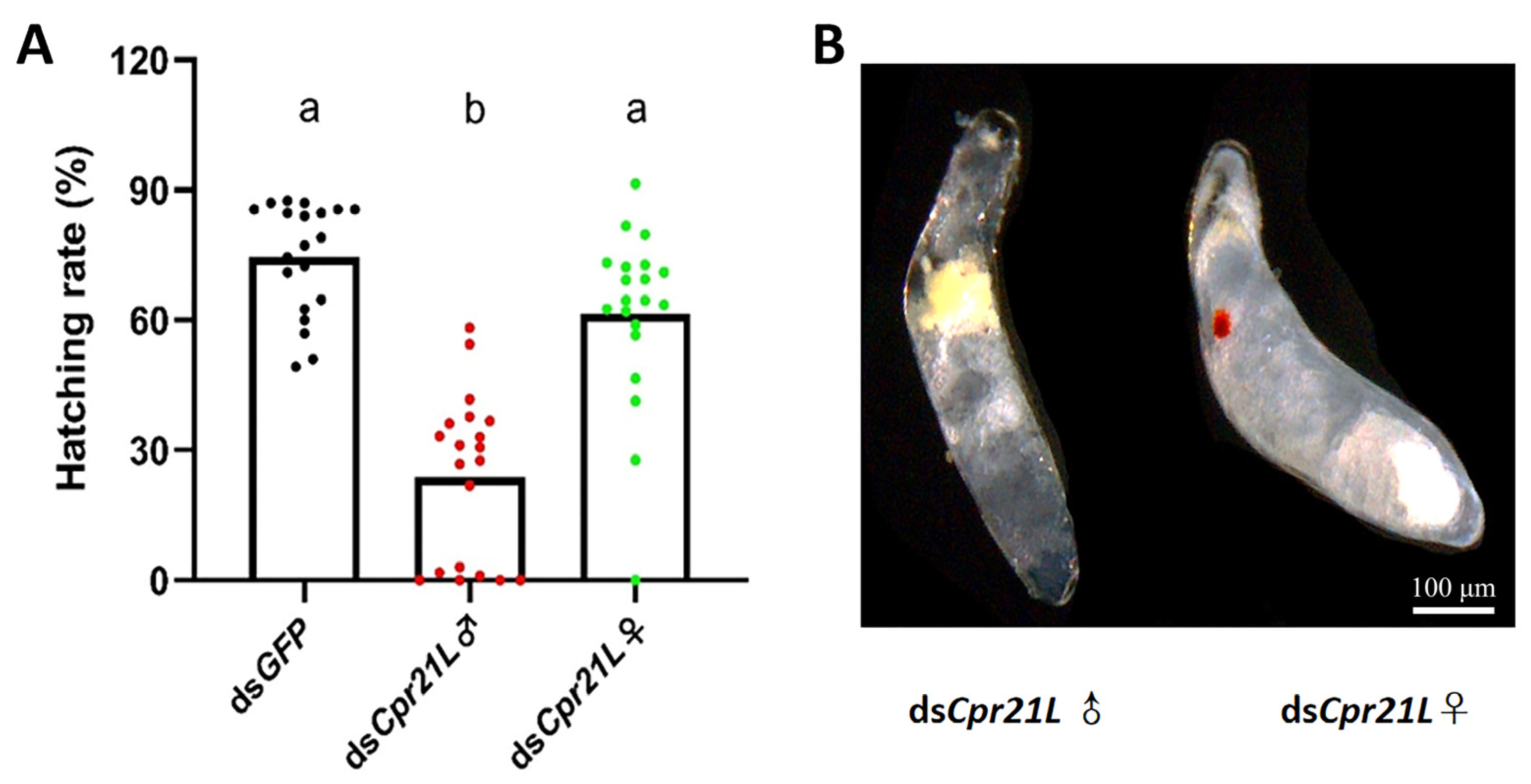
2.5. RNAi of Cpr21L Impacted the Fecundity of Adults
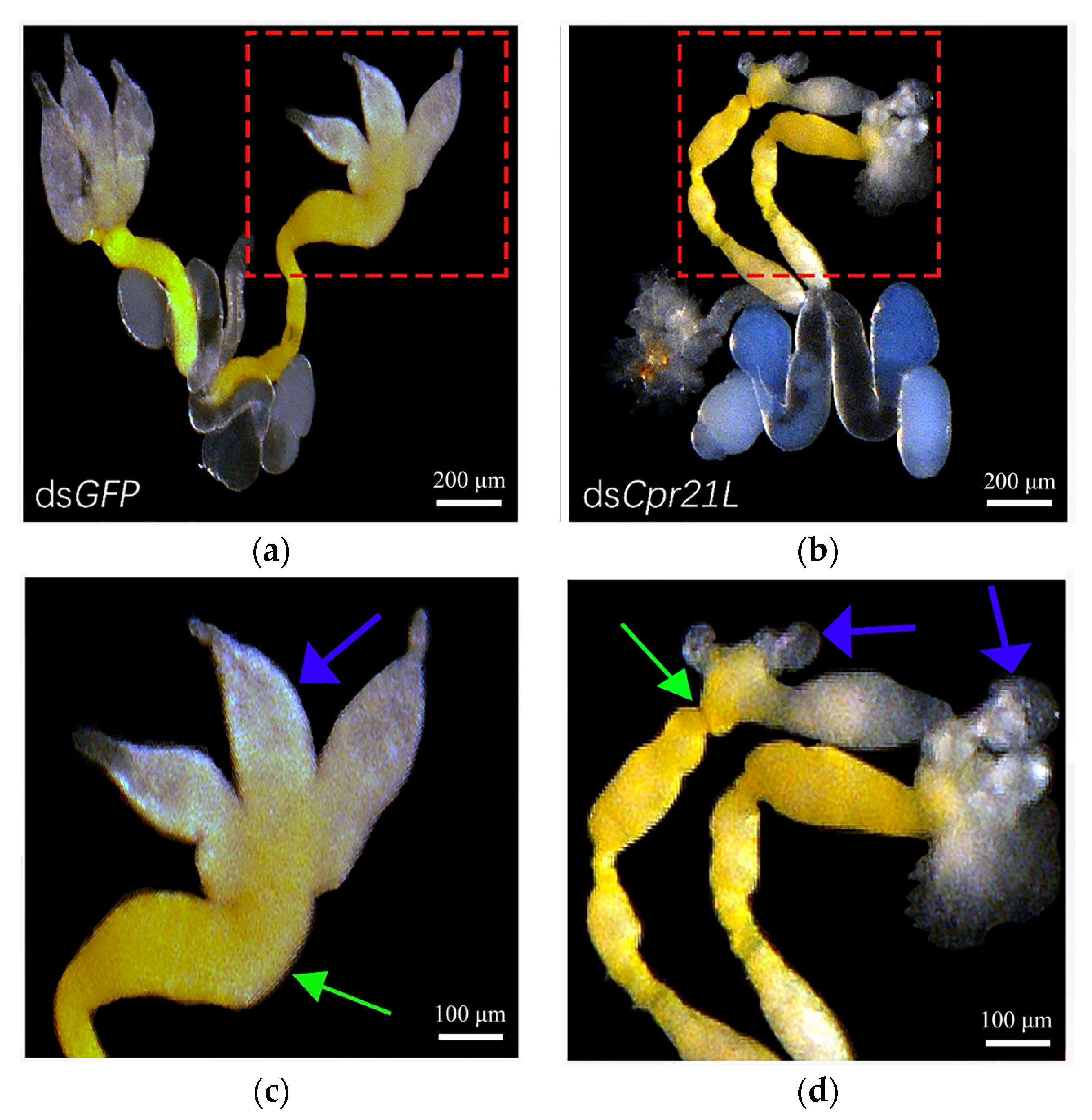
2.6. RNAi of Cpr21L Resulted in Malformed Internal Reproductive Organ of Male Adult
3. Discussion
4. Materials and Methods
4.1. Insects
4.2. RNA Isolation
4.3. cDNA Synthesis
4.4. Sequence and Phylogenetic Analysis of the Cpr21L in BPH
4.5. Spatiotemporal Expression Patterns of Cpr21L
4.6. Double-Stranded RNA Synthesis and Microinjection
4.7. Effects of RNAi against Cpr21L on the Survival and Development of BPH
4.8. Transmission Electron Microscopy (TEM) of dsRNA-Treated BPHs
4.9. Effect of RNAi on Insect Fecundity
4.10. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Xue, J.; Zhou, X.; Zhang, C.-X.; Yu, L.; Fan, H.-W.; Wang, Z.; Xu, H.-J.; Xi, Y.; Zhu, Z.-R.; Zhou, W.-W.; et al. Genomes of the rice pest brown planthopper and its endosymbionts reveal complex complementary contributions for host adaptation. Genome Biol. 2014, 15, 521–540. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.B.; Wang, S.N.; Ren, P.P.; He, F.; Li, Q.; Chen, J.P.; Li, J.M.; Zhang, C.A.X. RNAi-mediated silencing of an egg-specific gene Nllet1 results in hatch failure in the brown planthopper. Pest. Manag. Sci. 2022, 13, 415–427. [Google Scholar] [CrossRef] [PubMed]
- Pan, P.L.; Ye, Y.X.; Lou, Y.H.; Lu, J.B.; Cheng, C.; Shen, Y.; Moussian, B.; Zhang, C.X. A comprehensive omics analysis and functional survey of cuticular proteins in the brown planthopper. Proc. Natl. Acad. Sci. USA 2018, 115, 5175–5180. [Google Scholar] [CrossRef] [PubMed]
- Sirasoonthorn, P.; Kamiya, K.; Miura, K. Antifungal roles of adult-specific cuticular protein genes of the red flour beetle, Tribolium castaneum. J. Invertebr. Pathol. 2021, 186, 107674. [Google Scholar] [CrossRef] [PubMed]
- Vincent, J.F.V.; Wegst, U.G.K. Design and mechanical properties of insect cuticle. Arthropod. Struct. Dev. 2004, 33, 187–199. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Chang, T.; Zhao, K.; Sun, X.; Qiao, H.; Yan, C.; Wang, Y. Genome-wide annotation of cuticular protein genes in non-biting midge Propsilocerus akamusi and transcriptome analysis of their response to heavy metal pollution. Int. J. Biol. Macromol. 2022, 223, 555–566. [Google Scholar] [CrossRef]
- Futahashi, R.; Okamoto, S.; Kawasaki, H.; Zhong, Y.-S.; Iwanaga, M.; Mita, K.; Fujiwara, H. Genome-wide identification of cuticular protein genes in the silkworm, Bombyx mori. Insect Biochem. Mol. Biol. 2008, 38, 1138–1146. [Google Scholar] [CrossRef] [PubMed]
- Cornman, R.S.; Togawa, T.; Dunn, W.A.; He, N.; Emmons, A.C.; Willis, J.H. Annotation and analysis of a large cuticular protein family with the R&R Consensus in Anopheles gambiae. BMC Genom. 2008, 9, 22. [Google Scholar] [CrossRef]
- Appel, E.; Heepe, L.; Lin, C.-P.; Gorb, S.N. Ultrastructure of dragonfly wing veins: Composite structure of fibrous material supplemented by resilin. J. Anat. 2015, 227, 561–582. [Google Scholar] [CrossRef]
- Rapoport, T.A.; Li, L.; Park, E. Structural and Mechanistic Insights into Protein Translocation. Annu. Rev. Cell Dev. Biol. 2017, 33, 369–390. [Google Scholar] [CrossRef]
- Shan, S.-o.; Walter, P. Molecular crosstalk between the nucleotide specificity determinant of the SRP GTPase and the SRP receptor. Biochemistry 2005, 44, 6214–6222. [Google Scholar] [CrossRef]
- Voorhees, R.M.; Hegde, R.S. Toward a structural understanding of co-translational protein translocation. Curr. Opin. Cell Biol. 2016, 41, 91–99. [Google Scholar] [CrossRef]
- Pantazopoulou, A.; Glick, B.S. A kinetic view of membrane traffic pathways can transcend the classical view of golgi compartments. Front. Cell Dev. Biol. 2019, 7, 153–165. [Google Scholar] [CrossRef]
- Zhang, M.; Liu, L.; Lin, X.; Wang, Y.; Li, Y.; Guo, Q.; Li, S.; Sun, Y.; Tao, X.; Zhang, D.; et al. A translocation pathway for vesicle-mediated unconventional protein secretion. Cell 2020, 181, 637–652.e15. [Google Scholar] [CrossRef]
- Zhao, X.-M.; Yang, J.-P.; Gou, X.; Liu, W.-M.; Zhang, J.-Z. Cuticular protein gene LmACP8 is involved in wing morphogenesis in the migratory locust, Locusta migratoria. J. Integr. Agric. 2021, 20, 1596–1606. [Google Scholar] [CrossRef]
- Ham, J.; You, S.; Lim, W.; Song, G.J.E.P. Etoxazole induces testicular malfunction in mice by dysregulating mitochondrial function and calcium homeostasis. Env. Pollut. 2020, 263, 114573. [Google Scholar] [CrossRef] [PubMed]
- Michels, J.; Appel, E.; Gorb, S.N. Functional diversity of resilin in Arthropoda. Beilstein. J. Nanotech. 2016, 7, 1241–1259. [Google Scholar] [CrossRef]
- Qin, G.K.; Hu, X.; Cebe, P.; Kaplan, D.L. Mechanism of resilin elasticity. Nat. Commun. 2012, 3, 1–9. [Google Scholar] [CrossRef]
- Jaloszynski, P.; Ruta, R. Discovery of a complex sclerotized sperm pump in Monotomidae (Coleoptera): Functional morphology and phylogenetic value. J. Morphol. 2021, 282, 1208–1218. [Google Scholar] [CrossRef]
- Rebora, M.; Salerno, G.; Piersanti, S.; Kovalev, A.; Gorb, S. Cuticular modified air sacs underlie white coloration in the olive fruit fly, Bactrocera oleae. Commun. Biol. 2021, 4, 881. [Google Scholar] [CrossRef] [PubMed]
- Yuan, M.; Lu, Y.; Xun, Z.; Hu, W.; Muhammad, S.; Sha, Z.; Jin, B.R.; Li, J.; Wang, X.W.J.P.O. Selection and evaluation of potential reference genes for gene expression analysis in the brown planthopper, Nilaparvata lugens (Hemiptera: Delphacidae) using reverse-transcription quantitative PCR. PLoS ONE 2014, 9, e86503. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.J.; Bao, Y.Y.; Lao, S.H.; Huang, X.H.; Ye, Y.Z.; Wu, J.X.; Xu, H.J.; Zhou, X.P.; Zhang, C.X. Rice ragged stunt virus-induced apoptosis affects virus transmission from its insect vector, the brown planthopper to the rice plant. Sci. Rep. 2015, 5, 11413. [Google Scholar] [CrossRef]



| Primer Name | Sequence (5’→3’) |
|---|---|
| for ORF cloning | |
| Cpr21L-F | CAAGTCCACGGTGTGTTGTG |
| Cpr21L-R | TTAATATCGATTATCGTAGGC |
| for qPCR | |
| Cpr21L-qF | GAGATGTGGCGCGTGGAT |
| Cpr21L-qR | GCTGGCTGAGCTTTTTGC |
| NlRPS11-qF | CCGATCGTGTGGCGTTGAAGGG |
| NlRPS11-qR | ATGGCCGACATTCTTCCAGGTCC |
| for dsRNA synthesis | |
| dsCpr21L-F | GGATCCTAATACGACTCACTATAGGGTGTGTTGTGAACTTGTGAAA |
| dsCpr21L-R | GGATCCTAATACGACTCACTATAGGGCTCCCTGTAAGAATTGTCTG |
| dsGFP-F | GGATCCTAATACGACTCACTATAGGGAAGGGCGAGGAGCTGTTCACCG |
| dsGFP-R | GGATCCTAATACGACTCACTATAGGGCAGCAGGACCATGTGATCGCGC |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, T.; Jiao, Q.; Ye, C.; Wu, J.; Zheng, Y.; Sun, C.; Hao, P.; Yu, X. A Novel Cuticular Protein-like Cpr21L Is Essential for Nymph Survival and Male Fecundity in the Brown Planthopper. Int. J. Mol. Sci. 2023, 24, 2163. https://doi.org/10.3390/ijms24032163
Chen T, Jiao Q, Ye C, Wu J, Zheng Y, Sun C, Hao P, Yu X. A Novel Cuticular Protein-like Cpr21L Is Essential for Nymph Survival and Male Fecundity in the Brown Planthopper. International Journal of Molecular Sciences. 2023; 24(3):2163. https://doi.org/10.3390/ijms24032163
Chicago/Turabian StyleChen, Tongtong, Qiqi Jiao, Chenglong Ye, Jiangen Wu, Yuanyuan Zheng, Chuanxin Sun, Peiying Hao, and Xiaoping Yu. 2023. "A Novel Cuticular Protein-like Cpr21L Is Essential for Nymph Survival and Male Fecundity in the Brown Planthopper" International Journal of Molecular Sciences 24, no. 3: 2163. https://doi.org/10.3390/ijms24032163
APA StyleChen, T., Jiao, Q., Ye, C., Wu, J., Zheng, Y., Sun, C., Hao, P., & Yu, X. (2023). A Novel Cuticular Protein-like Cpr21L Is Essential for Nymph Survival and Male Fecundity in the Brown Planthopper. International Journal of Molecular Sciences, 24(3), 2163. https://doi.org/10.3390/ijms24032163




