Discrimination between Complete versus Non-Complete Pathologic Response to Neoadjuvant Therapy Using Ultrasensitive Mutation Analysis: A Proof-of-Concept Study in BRCA1-Driven Breast Cancer Patients
Abstract
1. Introduction
2. Results
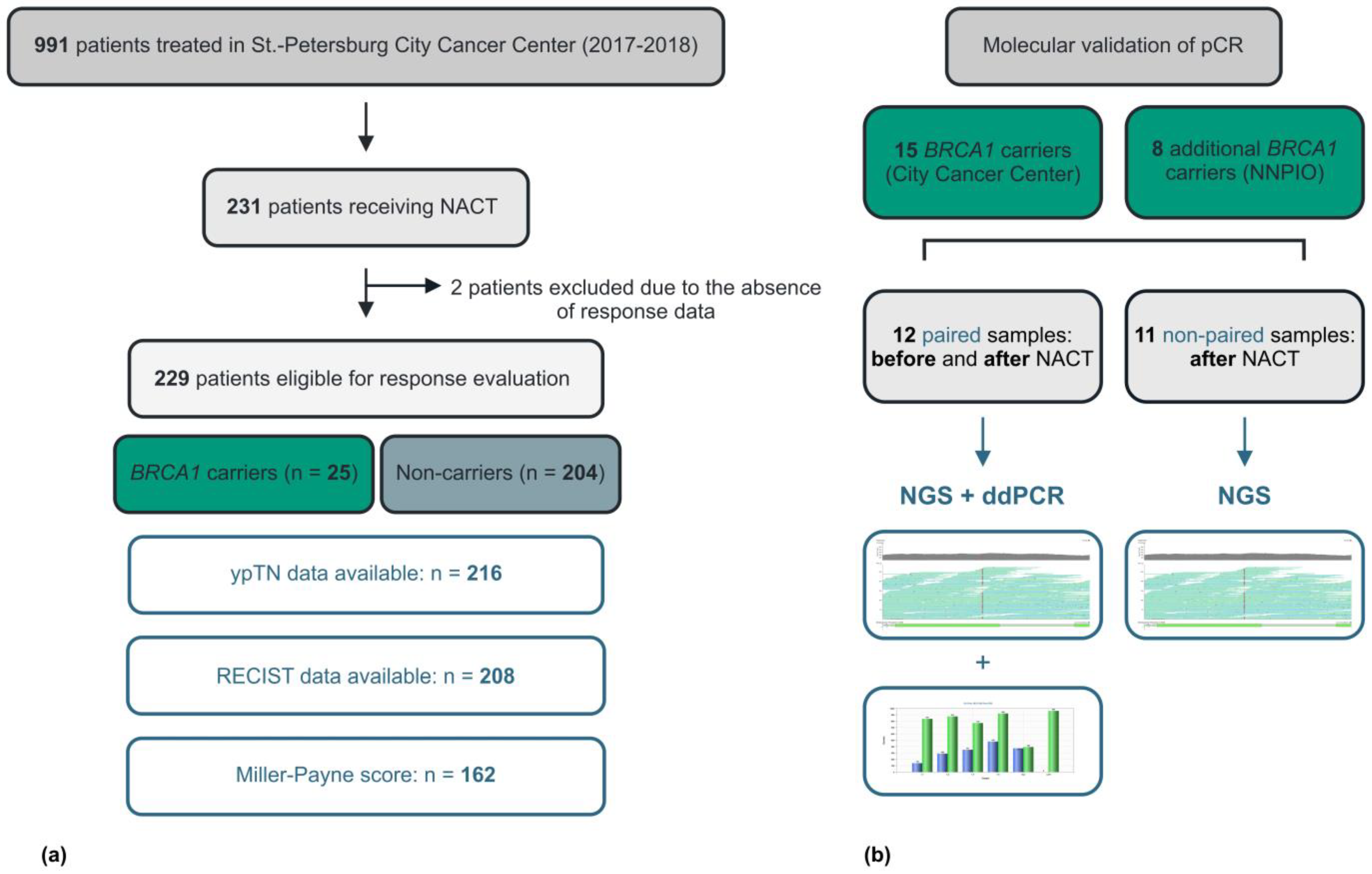
2.1. Clinical and Pathologic Response to NACT
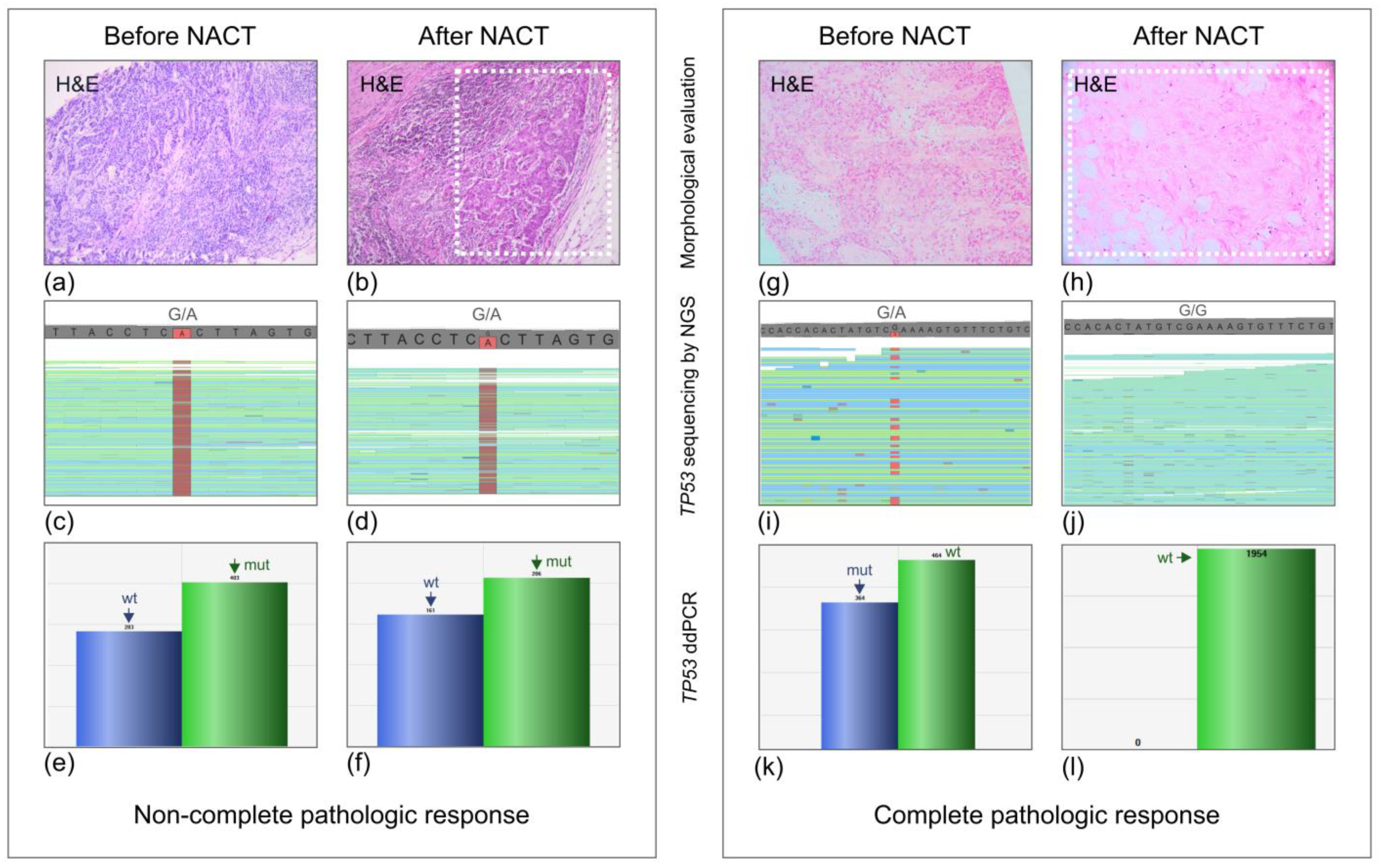
2.2. TP53 Mutation Analysis
3. Discussion
4. Materials and Methods
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Asaoka, M.; Gandhi, S.; Ishikawa, T.; Takabe, K. Neoadjuvant Chemotherapy for Breast Cancer: Past, Present, and Future. Breast Cancer Basic Clin. Res. 2020, 14, 1178223420980377. [Google Scholar] [CrossRef] [PubMed]
- Bardia, A.; Baselga, J. Neoadjuvant Therapy as a Platform for Drug Development and Approval in Breast Cancer. Clin. Cancer Res. 2013, 19, 6360–6370. [Google Scholar] [CrossRef] [PubMed]
- Bonnefoi, H.; Litière, S.; Piccart, M.; MacGrogan, G.; Fumoleau, P.; Brain, E.; Petit, T.; Rouanet, P.; Jassem, J.; Moldovan, C.; et al. Pathological complete response after neoadjuvant chemotherapy is an independent predictive factor irrespective of simplified breast cancer intrinsic subtypes: A landmark and two-step approach analyses from the EORTC 10994/BIG 1-00 phase III trial. Ann. Oncol. 2014, 25, 1128–1136. [Google Scholar] [CrossRef] [PubMed]
- Cortazar, P.; Zhang, L.; Untch, M.; Mehta, K.; Costantino, J.P.; Wolmark, N.; Bonnefoi, H.; Cameron, D.; Gianni, L.; Valagussa, P.; et al. Pathological complete response and long-term clinical benefit in breast cancer: The CTNeoBC pooled analysis. Lancet 2014, 384, 164–172. [Google Scholar] [CrossRef] [PubMed]
- Spring, L.M.; Fell, G.; Arfe, A.; Sharma, C.; Greenup, R.A.; Reynolds, K.L.; Smith, B.L.; Alexander, B.M.; Moy, B.; Isakoff, S.J.; et al. Pathologic Complete Response after Neoadjuvant Chemotherapy and Impact on Breast Cancer Recurrence and Survival: A Comprehensive Meta-analysis. Clin. Cancer Res. 2020, 26, 2838–2848. [Google Scholar] [CrossRef]
- Imyanitov, E.N. Cytotoxic and targeted therapy for BRCA1/2-driven cancers. Hered. Cancer Clin. Pract. 2021, 19, 36. [Google Scholar] [CrossRef]
- Imyanitov, E.N.; Iyevleva, A.G. Molecular tests for prediction of tumor sensitivity to cytotoxic drugs. Cancer Lett. 2022, 526, 41–52. [Google Scholar] [CrossRef]
- Byrski, T.; Gronwald, J.; Huzarski, T.; Grzybowska, E.; Budryk, M.; Stawicka, M.; Mierzwa, T.; Szwiec, M.; Wiśniowski, R.; Siolek, M.; et al. Pathologic Complete Response Rates in Young Women with BRCA1-Positive Breast Cancers After Neoadjuvant Chemotherapy. J. Clin. Oncol. 2010, 28, 375–379. [Google Scholar] [CrossRef]
- Paluch-Shimon, S.; Friedman, E.; Berger, R.; Papa, M.; Dadiani, M.; Friedman, N.; Shabtai, M.; Zippel, D.; Gutman, M.; Golan, T.; et al. Neo-adjuvant doxorubicin and cyclophosphamide followed by paclitaxel in triple-negative breast cancer among BRCA1 mutation carriers and non-carriers. Breast Cancer Res. Treat. 2016, 157, 157–165. [Google Scholar] [CrossRef]
- Tutt, A.; Tovey, H.; Cheang, M.C.U.; Kernaghan, S.; Kilburn, L.; Gazinska, P.; Owen, J.; Abraham, J.; Barrett, S.; Barrett-Lee, P.; et al. Carboplatin in BRCA1/2-mutated and triple-negative breast cancer BRCAness subgroups: The TNT Trial. Nat. Med. 2018, 24, 628–637. [Google Scholar] [CrossRef]
- Bignon, L.; Fricker, J.P.; Nogues, C.; Mouret-Fourme, E.; Stoppa-Lyonnet, D.; Caron, O.; Lortholary, A.; Faivre, L.; Lasset, C.; Mari, V.; et al. Efficacy of anthracycline/taxane-based neo-adjuvant chemotherapy on triple-negaive breast cancer in BRCA1/BRCA2 mutation carriers. Breast J. 2018, 24, 269–277. [Google Scholar] [CrossRef]
- Hahnen, E.; Lederer, B.; Hauke, J.; Loibl, S.; Kröber, S.; Schneeweiss, A.; Denkert, C.; Fasching, P.A.; Blohmer, J.U.; Jackisch, C.; et al. Germline Mutation Status, Pathological Complete Response, and Disease-Free Survival in Triple-Negative Breast Cancer: Secondary Analysis of the GeparSixto Randomized Clinical Trial. JAMA Oncol. 2017, 3, 1378–1385. [Google Scholar] [CrossRef]
- Fasching, P.A.; Loibl, S.; Hu, C.; Hart, S.N.; Shimelis, H.; Moore, R.; Schem, C.; Tesch, H.; Untch, M.; Hilfrich, J.; et al. BRCA1/2 Mutations and Bevacizumab in the Neoadjuvant Treatment of Breast Cancer: Response and Prognosis Results in Patients with Triple-Negative Breast Cancer from the GeparQuinto Study. J. Clin. Oncol. 2018, 36, 2281–2287. [Google Scholar] [CrossRef]
- Pohl-Rescigno, E.; Hauke, J.; Loibl, S.; Möbus, V.; Denkert, C.; Fasching, P.A.; Kayali, M.; Ernst, C.; Weber-Lassalle, N.; Hanusch, C.; et al. Association of Germline Variant Status with Therapy Response in High-risk Early-Stage Breast Cancer: A Secondary Analysis of the GeparOcto Randomized Clinical Trial. JAMA Oncol. 2020, 6, 744–748. [Google Scholar] [CrossRef]
- Wunderle, M.; Gass, P.; Häberle, L.; Flesch, V.M.; Rauh, C.; Bani, M.R.; Hack, C.C.; Schrauder, M.G.; Jud, S.M.; Emons, J.; et al. BRCA mutations and their influence on pathological complete response and prognosis in a clinical cohort of neoadjuvantly treated breast cancer patients. Breast Cancer Res. Treat. 2018, 171, 85–94. [Google Scholar] [CrossRef]
- Tung, N.; Arun, B.; Hacker, M.R.; Hofstatter, E.; Toppmeyer, D.L.; Isakoff, S.J.; Borges, V.; Legare, R.D.; Isaacs, C.; Wolff, A.C.; et al. TBCRC 031: Randomized Phase II Study of Neoadjuvant Cisplatin Versus Doxorubicin-Cyclophosphamide in Germline BRCA Carriers with HER2-Negative Breast Cancer (the INFORM trial). J. Clin. Oncol. 2020, 38, 1539–1548. [Google Scholar] [CrossRef]
- Mavaddat, N.; Barrowdale, D.; Andrulis, I.L.; Domchek, S.M.; Eccles, D.; Nevanlinna, H.; Ramus, S.J.; Spurdle, A.; Robson, M.; Sherman, M.; et al. Pathology of breast and ovarian cancers among BRCA1 and BRCA2 mutation carriers: Results from the Consortium of Investigators of Modifiers of BRCA1/2 (CIMBA). Cancer Epidemiol. Biomarkers Prev. 2012, 21, 134–147. [Google Scholar] [CrossRef]
- Arun, B.; Bayraktar, S.; Liu, D.D.; Gutierrez Barrera, A.M.; Atchley, D.; Pusztai, L.; Litton, J.K.; Valero, V.; Meric-Bernstam, F.; Hortobagyi, G.N.; et al. Response to neoadjuvant systemic Breast Cancer Res Treat 123 therapy for breast cancer in BRCA mutation carriers and noncarriers: A single-institution experience. J. Clin. Oncol. 2011, 29, 3739–3746. [Google Scholar] [CrossRef]
- Pfeifer, W.; Sokolenko, A.P.; Potapova, O.N.; Bessonov, A.A.; Ivantsov, A.O.; Laptiev, S.A.; Zaitseva, O.A.; Yatsuk, O.S.; Matsko, D.E.; Semiglazova, T.; et al. Breast cancer sensitivity to neoadjuvant therapy in BRCA1 and CHEK2 mutation carriers and non-carriers. Breast Cancer Res. Treat. 2014, 148, 675–683. [Google Scholar] [CrossRef]
- Martins, F.C.; De, S.; Almendro, V.; Gönen, M.; Park, S.Y.; Blum, J.L.; Herlihy, W.; Ethington, G.; Schnitt, S.J.; Tung, N.; et al. Evolutionary Pathways in BRCA1-Associated Breast Tumors. Cancer Discov. 2012, 2, 503–511. [Google Scholar] [CrossRef]
- Sokolenko, A.P.; Savonevich, E.L.; Ivantsov, A.O.; Raskin, G.A.; Kuligina, E.S.; Gorodnova, T.V.; Preobrazhenskaya, E.V.; Kleshchov, M.A.; Tiurin, V.I.; Mukhina, M.S.; et al. Rapid selection of BRCA1-proficient tumor cells during neoadjuvant therapy for ovarian cancer in BRCA1 mutation carriers. Cancer Lett. 2017, 397, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Ogston, K.N.; Miller, I.D.; Payne, S.; Hutcheon, A.W.; Sarkar, T.K.; Smith, I.; Schofield, A.; Heys, S.D. A new histological grading system to assess response of breast cancers to primary chemotherapy: Prognostic significance and survival. Breast 2003, 12, 320–327. [Google Scholar] [CrossRef] [PubMed]
- Sokolenko, A.P.; Gorodnova, T.V.; Bizin, I.V.; Kuligina, E.S.; Kotiv, K.B.; Romanko, A.A.; Ermachenkova, T.I.; Ivantsov, A.O.; Preobrazhenskaya, E.V.; Sokolova, T.N.; et al. Molecular predictors of the outcome of paclitaxel plus carboplatin neoadjuvant therapy in high-grade serous ovarian cancer patients. Cancer Chemother. Pharmacol. 2021, 88, 439–450. [Google Scholar] [CrossRef] [PubMed]
- Kuligina, E.S.; Meerovich, R.; Zagorodnev, K.A.; Kholmatov, M.M.; Sokolova, T.N.; Laidus, T.A.; Romanko, A.A.; Martianov, A.S.; Anisimova, M.O.; Zaitseva, O.A.; et al. Content of circulating tumor DNA depends on the tumor type and the dynamics of tumor size, but is not influenced significantly by physical exercise, time of the day or recent meal. Cancer Genet. 2021, 256-257, 165–178. [Google Scholar] [CrossRef]
| All Cases (n = 208) | |||
|---|---|---|---|
| BRCA1-carriers (n = 19) | Non-carriers (n = 189) | p-value | |
| All regimens | 16/19 (84%) | 117/189 (62%) | 0.08 |
| Platinum-containing (n = 33) | 12/14 (86%) | 8/19 (42%) | 0.02 |
| Without platinum (n = 175) | 4/5 (80%) | 109/170 (64%) | 0.66 |
| Triple-negative carcinomas (n = 57) | |||
| BRCA1-carriers (n = 18) | Non-carriers (n = 39) | ||
| All regimens | 15/18 (83%) | 24/39 (62%) | 0.13 |
| Platinum-containing (n = 28) | 11/13 (85%) | 6/15 (40%) | 0.02 |
| Without platinum (n = 29) | 4/5 (80%) | 18/24 (75%) | 1.0 |
| All Cases (n = 216) | |||
|---|---|---|---|
| BRCA1-carriers (n = 24) | Non-carriers (n = 192) | p-value | |
| All regimens | 13/24 (54%) | 29/192 (15%) | <0.0001 |
| Platinum-containing (n = 37) | 9/18 (50%) | 3/19 (16%) | 0.04 |
| Without platinum (n = 179) | 4/6 (67%) | 26/173 (15%) | 0.008 |
| Triple-negative carcinomas (n = 59) | |||
| BRCA1-carriers (n = 20) | Non-carriers (n = 39) | ||
| All regimens | 12/20 (60%) | 6/39 (15%) | 0.001 |
| Platinum-containing (n = 30) | 8/15 (53%) | 3/15 (20%) | 0.128 |
| Without platinum (n = 29) | 4/5 (80%) | 3/24 (13%) | 0.007 |
| ID | BRCA1 Mutation | Subtype | Ki-67, Grade | NACT (No. of Cycles) | Miller–Payne Score | TP53 Mutation | TP53 Detected after NACT | Samples Available for Analysis (Method) |
|---|---|---|---|---|---|---|---|---|
| NBC1 | c.5266dupC | Triple-negative | 17%, G2 | P (2) | 2 | P151R | Yes | Pre-NACT (NGS) and post-NACT (NGS) |
| NBC3 | c.5266dupC | Luminal B | 69%, G3 | AC (4) | 3 | Ex8 55-bp del | Yes | Post-NACT (NGS) |
| NBC4 | c.5266dupC | Triple-negative | 75%, G3 | TP (4) | 4 | R213X | Yes | Pre-NACT (NGS) and post-NACT (ddPCR) |
| NBC6 | c.5266dupC | Triple-negative | 33%, G3 | P (6) | 5 (pCR) | R213X | No | Pre-NACT (NGS) and post-NACT (ddPCR) |
| NBC8 | c.5266dupC | Triple-negative | 90%, G3 | P (4) | 3 | Y163C | Yes | Pre-NACT (NGS) and post-NACT (NGS) |
| NBC9 | c.5266dupC | Triple-negative | 49%, G3 | FAC (3) | 3 | I255N | Yes | Pre-NACT (NGS) and post-NACT (ddPCR) |
| NBC10 | c.5266dupC | Triple-negative | 29%, G3 | P (4) | 5 (pCR) | - | No | Post-NACT (NGS) |
| NBC11 | c.5266dupC | Triple-negative | 64%, G3 | TP (4) | 5 (pCR) | Y234C | No | Pre-NACT (NGS) and post-NACT (ddPCR) |
| NBC12 | c.68_69delAG | Triple-negative | 36%, G3 | P (6) | 1 | R306X | Yes | Pre- NACT (NGS) and post-NACT (NGS) |
| NBC13 | c.68_69delAG | Triple-negative | Nd | TP (6) | 3 | M237I | Yes | Pre-NACT (NGS) and post-NACT (ddPCR) |
| NBC14 | c.5266dupC | Triple-negative | 85%, G3 | FAC (6) | Nd * (RCB II) | c.755delT | Yes | Post-NACT (NGS) |
| NBC15 | c.5266dupC | Triple-negative | 75%, G3 | TP (2) | 5 (pCR) | - | No | Post-NACT (NGS) |
| NBC16 | c.5266dupC | Triple-negative | 60%, G3 | TP (4) | 5 (pCR) | - | No | Post-NACT (NGS) |
| NBC18 | c.5266dupC | Triple-negative | 65%, G3 | AC (4) | 5 (pCR) | - | No | Post-NACT (NGS) |
| NBC19 | c.5266dupC | Triple-negative | 75%, G3 | TP (5) | 5 (pCR) | Y220C | No | Pre-NACT (NGS) and post-NACT (ddPCR) |
| NBC21 | c.5266dupC | Triple-negative | 90%, G3 | AC (4), T (12) | Nd * (RCB II) | c.473insTCCG | Yes | Post-NACT (NGS) |
| NBC23 | c.5266dupC | Triple-negative | 80%, G3 | FAC (4), T (12) | 5 (pCR) | - | No | Post-NACT (NGS) |
| NBC24 | c.5266dupC | Luminal B | 30%, G2 | FAC (4), P (9) | 4 | R273L | Yes | Pre-NACT (NGS) and post-NACT (ddPCR) |
| NBC25 | c.5266dupC | Triple-negative | 80%, G3 | FAC (4), P (12) | 5 (pCR) | c.445delT | No | Pre-NACT (NGS) and post-NACT (ddPCR) |
| NBC26 | c.5266dupC | Triple-negative | 90%, G3 | FAC (4), P (12) | 5 (pCR) | R273H | No | Pre-NACT (NGS) and post-NACT (ddPCR) |
| NBC29 | c.5266dupC | Triple-negative | 90%, G3 | AC (4), T (12) | 2 | Ex7 17-bp del | Yes | Post-NACT (NGS) |
| NBC31 | c.4034delA | Triple-negative | 90%, G3 | AC (4), P (6) | 1 | C275Y | Yes | Post-NACT (NGS) |
| NBC32 | c.4034delA | Triple-negative | 90%, G3 | AC (4), P (2) | 2 | R273C | Yes | Post-NACT (NGS) |
| Study | No. of BRCA1/2 Carriers | NACT Composition | pCR Rate |
|---|---|---|---|
| Arun et al. [18] | 57 BRCA1 and 23 BRCA2 carriers | Three treatment groups: anthracycline-taxane (AT) therapy, anthracycline-based regimens without a taxane, or single-agent taxane | The highest pCR rates were seen in AT group in all patient categories: 46% in BRCA1 carriers, 13% in BRCA2 carriers, and 22% in non-carriers |
| Byrski et al. [8] | 107 BRCA1 carriers | Cisplatin 75 mg/m2 every 3 weeks for 4 cycles | 65/107 patients (61%) |
| Pfeifer et al. [19] | 19 BRCA1 carriers | Anthracycline-based schemes without taxanes and taxane-containing regimens | The overall pCR rate was 32%; pCR was achieved in 56% of patients receiving anthracycline-based regimens without taxanes |
| Paluch-Shimon et al. [9] | 34 BRCA1 carriers | 4 cycles of dose-dense doxorubicin (60 mg/m2) and cyclophosphamide (600 mg/m2) every 2 weeks followed by dose-dense paclitaxel (175 mg/m2) every 2 weeks for 4 cycles or 12 weekly cycles of paclitaxel (80/m2) | 68% in BRCA1 carriers compared to 37% in non-carriers (p = 0.01) |
| Bignon et al. [11] | 46 BRCA1 carriers, 6 BRCA2 carriers, and one BRCA1/BRCA2 double heterozygote; all triple-negative BCs | Anthracycline-based schemes either with taxanes or without taxanes | The overall pCR rate in BRCA1 mutation carriers was 38%; it was statistically non-significantly higher in patients receiving NACT with taxanes compared to patients who didn’t receive taxanes (12/25 (43%) vs. 5/18 (28%), respectively) |
| Wunderle et al. [15] | 43 BRCA1 carriers, 16 BRCA2 carriers | Either anthracycline-based therapy (4 cycles of epirubicin (80–90 mg/m2) and cyclophosphamide (600 mg/m2) every 3 weeks, followed by 12 cycles of paclitaxel (80–90 mg/m2) weekly) or platinum-based therapy (6 cycles of carboplatin AUC5 on day 1 and paclitaxel (80–90 mg/m2) on days 1, 8, and 15, every 3 weeks) | pCR rate was 58% for BRCA1 mutation carriers, 44% for BRCA2 carriers, and 23% for sporadic carcinomas. In BRCA1 carriers, pCR rate was 11/25 (44%) among patients receiving anthracycline-based therapy and 14/18 (78%) in patients receiving platinum-based therapy (p = 0.03) |
| Fasching et al. [13] | 74 BRCA1 carriers and 16 BRCA2 carriers, all triple-negative BCs | 4 cycles of epirubicin/cyclophosphamide followed by 4 cycles of docetaxel; or 4 cycles of epirubicin/cyclophosphamide/bevacizumab followed by 4 cycles of docetaxel/bevacizumab | pCR rate was 49% in BRCA1 carriers and 56% in BRCA2 carriers |
| Tung et al. [16] | 81 BRCA1 carriers, 35 BRCA2 carriers, and two double heterozygotes BRCA1/BRCA2 | Cisplatin 75 mg/m2, 4 cycles of AC (doxorubicin 60 mg/m2 and cyclophosphamide 600 mg/m2), 4 cycles every 2 (dose-dense) or 3 weeks | pCR in the cisplatin arm: BRCA1: 20%; BRCA2: 13%; pCR in the AC arm: BRCA1: 28%; BRCA2: 25% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sokolenko, A.P.; Moiseyenko, F.V.; Iyevleva, A.G.; Ivantsov, A.O.; Dolmatov, G.D.; Shelekhova, K.V.; Gulo, E.V.; Topal, A.X.; Artemieva, E.V.; Abduloeva, N.H.; et al. Discrimination between Complete versus Non-Complete Pathologic Response to Neoadjuvant Therapy Using Ultrasensitive Mutation Analysis: A Proof-of-Concept Study in BRCA1-Driven Breast Cancer Patients. Int. J. Mol. Sci. 2023, 24, 1870. https://doi.org/10.3390/ijms24031870
Sokolenko AP, Moiseyenko FV, Iyevleva AG, Ivantsov AO, Dolmatov GD, Shelekhova KV, Gulo EV, Topal AX, Artemieva EV, Abduloeva NH, et al. Discrimination between Complete versus Non-Complete Pathologic Response to Neoadjuvant Therapy Using Ultrasensitive Mutation Analysis: A Proof-of-Concept Study in BRCA1-Driven Breast Cancer Patients. International Journal of Molecular Sciences. 2023; 24(3):1870. https://doi.org/10.3390/ijms24031870
Chicago/Turabian StyleSokolenko, Anna P., Fedor V. Moiseyenko, Aglaya G. Iyevleva, Alexandr O. Ivantsov, Georgiy D. Dolmatov, Ksenia V. Shelekhova, Elizaveta V. Gulo, Anastasya X. Topal, Elizaveta V. Artemieva, Nuriniso H. Abduloeva, and et al. 2023. "Discrimination between Complete versus Non-Complete Pathologic Response to Neoadjuvant Therapy Using Ultrasensitive Mutation Analysis: A Proof-of-Concept Study in BRCA1-Driven Breast Cancer Patients" International Journal of Molecular Sciences 24, no. 3: 1870. https://doi.org/10.3390/ijms24031870
APA StyleSokolenko, A. P., Moiseyenko, F. V., Iyevleva, A. G., Ivantsov, A. O., Dolmatov, G. D., Shelekhova, K. V., Gulo, E. V., Topal, A. X., Artemieva, E. V., Abduloeva, N. H., Rysev, N. A., Barsova, D. A., Levchenko, N. V., Volkov, N. M., Egorenkov, V. V., Moiseyenko, V. M., & Imyanitov, E. N. (2023). Discrimination between Complete versus Non-Complete Pathologic Response to Neoadjuvant Therapy Using Ultrasensitive Mutation Analysis: A Proof-of-Concept Study in BRCA1-Driven Breast Cancer Patients. International Journal of Molecular Sciences, 24(3), 1870. https://doi.org/10.3390/ijms24031870






