CCL5’s Role in Periodontal Disease: A Narrative Review
Abstract
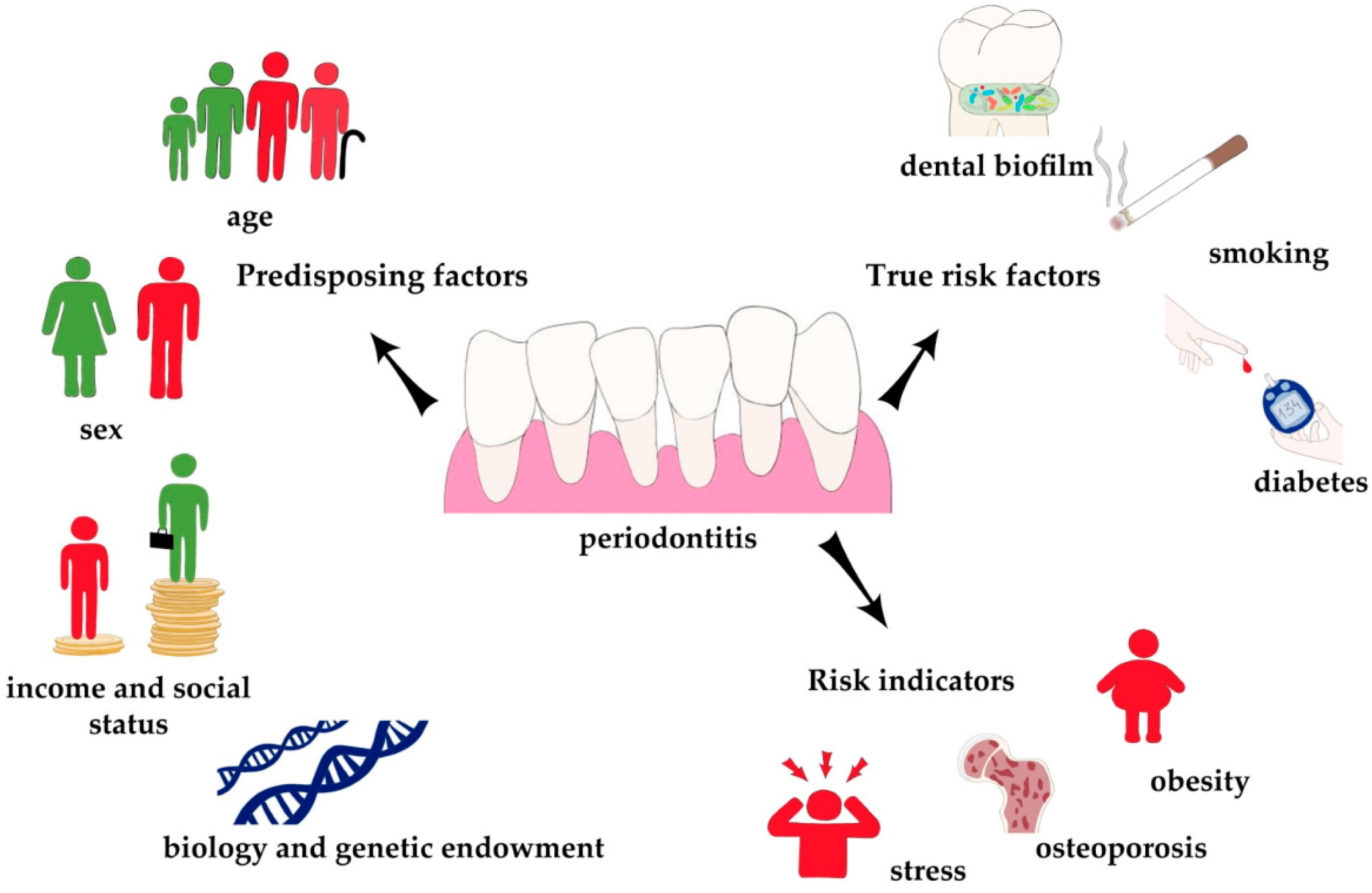
:1. Introduction
2. Results
2.1. CCL5 in Periodontitis
2.1.1. CCL5 in the Gingival Crevicular Fluid and Gingiva of Patients with Periodontitis
2.1.2. CCL5 in the Blood of Patients with Periodontitis
2.1.3. CCL5 in Periodontitis in the Elderly
2.1.4. CCL5 in Periodontitis and Obesity
2.1.5. CCL5 in Periodontitis and Gender
2.1.6. CCL5 in Periodontitis and Cigarette Smoking
2.1.7. CCL5 in Periodontitis and Diabetes
2.1.8. CCL5 in Periodontitis and Rheumatoid Arthritis
2.1.9. CCL5 in Periodontitis and Vitamin D Deficiency
2.2. Source of CCL5
2.2.1. Cells Responsible for Increased Level of CCL5
2.2.2. Bacteria as a Factor Responsible for Increased Level of CCL5
3. Discussion
3.1. CCL5 in Disease Mechanisms of Periodontitis
3.1.1. CCL5-CCR5 Axis Gene Polymorphism as an Indicator of CCL5 Significance in the Pathogenesis of Periodontitis
3.1.2. Involvement of CCL5 in Gingival Infiltration by Immune Cells in Periodontitis
3.1.3. CCL5 as a Factor in Alveolar Bone Destruction
3.1.4. Effect of CCL5 on Periodontal Ligament Stem Cells
3.1.5. Link between Periodontitis and Cancer Progression: Impact on Lymph Node Metastasis
4. Materials and Methods
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Slots, J. Periodontitis: Facts, fallacies and the future. Periodontol. 2000 2017, 75, 7–23. [Google Scholar] [CrossRef]
- Tonetti, M.S.; Jepsen, S.; Jin, L.; Otomo-Corgel, J. Impact of the global burden of periodontal diseases on health, nutrition and wellbeing of mankind: A call for global action. J. Clin. Periodontol. 2017, 44, 456–462. [Google Scholar] [CrossRef]
- Hegde, R.; Awan, K.H. Effects of periodontal disease on systemic health. Dis. Mon. 2019, 65, 185–192. [Google Scholar] [CrossRef]
- Schenkein, H.A.; Loos, B.G. Inflammatory mechanisms linking periodontal diseases to cardiovascular diseases. J. Periodontol. 2013, 84 (Suppl. S4), S51–S69. [Google Scholar] [CrossRef]
- Reyes, L.; Herrera, D.; Kozarov, E.; Roldá, S.; Progulske-Fox, A. Periodontal bacterial invasion and infection: Contribution to atherosclerotic pathology. J. Periodontol. 2013, 84 (Suppl. S4), S30–S50. [Google Scholar] [CrossRef]
- Machado, V.; Botelho, J.; Escalda, C.; Hussain, S.B.; Luthra, S.; Mascarenhas, P.; Orlandi, M.; Mendes, J.J.; D’Aiuto, F. Serum C-Reactive Protein and Periodontitis: A Systematic Review and Meta-Analysis. Front. Immunol. 2021, 12, 706432. [Google Scholar] [CrossRef]
- Carrizales-Sepúlveda, E.F.; Ordaz-Farías, A.; Vera-Pineda, R.; Flores-Ramírez, R. Periodontal Disease, Systemic Inflammation and the Risk of Cardiovascular Disease. Heart Lung Circ. 2018, 27, 1327–1334. [Google Scholar] [CrossRef]
- Shankar, S.; Manath, S.; Alqahtani, S.M.; Ganji, K.K.; Nagate, R.R.; Ghokale, S.T.; Nagarajappa, A.K.; Javali, M.A.; Tikare, S.; Khader, M.A. Variations of Serum CRP Levels in Periodontal Health and Diseases: A Clinico-Biochemical Study. Diagnostics 2023, 13, 2483. [Google Scholar] [CrossRef]
- GBD 2016 Disease and Injury Incidence and Prevalence Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2017, 390, 1211–1259. [Google Scholar] [CrossRef]
- Nazir, M.; Al-Ansari, A.; Al-Khalifa, K.; Alhareky, M.; Gaffar, B.; Almas, K. Global Prevalence of Periodontal Disease and Lack of Its Surveillance. Sci. World J. 2020, 2020, 2146160, Erratum in Lancet 2017, 390, e38. [Google Scholar] [CrossRef]
- Bouchard, P.; Carra, M.C.; Boillot, A.; Mora, F.; Rangé, H. Risk factors in periodontology: A conceptual framework. J. Clin. Periodontol. 2017, 44, 125–131. [Google Scholar] [CrossRef]
- Garcia, R.I.; Compton, R.; Dietrich, T. Risk assessment and periodontal prevention in primary care. Periodontol. 2000 2016, 71, 10–21. [Google Scholar] [CrossRef]
- Genco, R.J.; Borgnakke, W.S. Risk factors for periodontal disease. Periodontol. 2000 2013, 62, 59–94. [Google Scholar] [CrossRef]
- Curtis, M.A.; Diaz, P.I.; Van Dyke, T.E. The role of the microbiota in periodontal disease. Periodontol. 2000 2020, 83, 14–25. [Google Scholar] [CrossRef]
- Butera, A.; Pascadopoli, M.; Pellegrini, M.; Gallo, S.; Zampetti, P.; Scribante, A. Oral Microbiota in Patients with Peri-Implant Disease: A Narrative Review. Appl. Sci. 2022, 12, 3250. [Google Scholar] [CrossRef]
- Lamont, R.J.; Koo, H.; Hajishengallis, G. The oral microbiota: Dynamic communities and host interactions. Nat. Rev. Microbiol. 2018, 16, 745–759. [Google Scholar] [CrossRef]
- Van Dyke, T.E.; Bartold, P.M.; Reynolds, E.C. The Nexus between Periodontal Inflammation and Dysbiosis. Front. Immunol. 2020, 11, 511. [Google Scholar] [CrossRef]
- Lee, C.T.; Teles, R.; Kantarci, A.; Chen, T.; McCafferty, J.; Starr, J.R.; Brito, L.C.; Paster, B.J.; Van Dyke, T.E. Resolvin E1 Reverses Experimental Periodontitis and Dysbiosis. J. Immunol. 2016, 197, 2796–2806. [Google Scholar] [CrossRef]
- Butera, A.; Gallo, S.; Maiorani, C.; Molino, D.; Chiesa, A.; Preda, C.; Esposito, F.; Scribante, A. Probiotic Alternative to Chlorhexidine in Periodontal Therapy: Evaluation of Clinical and Microbiological Parameters. Microorganisms 2020, 9, 69. [Google Scholar] [CrossRef]
- Butera, A.; Gallo, S.; Pascadopoli, M.; Maiorani, C.; Milone, A.; Alovisi, M.; Scribante, A. Parobiotics in Non-Surgical Periodontal Therapy: Clinical and Microbiological Aspects in a 6-Month Follow-Up Domiciliary Protocol for Oral Hygiene. Microorganisms 2022, 10, 337. [Google Scholar] [CrossRef]
- Butera, A.; Gallo, S.; Pascadopoli, M.; Taccardi, D.; Scribante, A. Home Oral Care of Periodontal Patients Using Antimicrobial Gel with Postbiotics, Lactoferrin, and Aloe Barbadensis Leaf Juice Powder vs. Conventional Chlorhexidine Gel: A Split-Mouth Randomized Clinical Trial. Antibiotics 2022, 11, 118. [Google Scholar] [CrossRef]
- Colombo, M.; Gallo, S.; Garofoli, A.; Poggio, C.; Arciola, C.R.; Scribante, A. Ozone Gel in Chronic Periodontal Disease: A Randomized Clinical Trial on the Anti-Inflammatory Effects of Ozone Application. Biology 2021, 10, 625. [Google Scholar] [CrossRef]
- Mombelli, A. Microbial colonization of the periodontal pocket and its significance for periodontal therapy. Periodontol. 2000 2018, 76, 85–96. [Google Scholar] [CrossRef]
- Wang, Y.; Wu, Y.; Wang, Y.; Xu, H.; Mei, X.; Yu, D.; Wang, Y.; Li, W. Antioxidant Properties of Probiotic Bacteria. Nutrients 2017, 9, 521. [Google Scholar] [CrossRef]
- Butera, A.; Gallo, S.; Pascadopoli, M.; Luraghi, G.; Scribante, A. Ozonized Water Administration in Peri-Implant Mucositis Sites: A Randomized Clinical Trial. Appl. Sci. 2021, 11, 7812. [Google Scholar] [CrossRef]
- Żółkiewicz, J.; Marzec, A.; Ruszczyński, M.; Feleszko, W. Postbiotics-A Step Beyond Pre- and Probiotics. Nutrients 2020, 12, 2189. [Google Scholar] [CrossRef]
- Butera, A.; Pascadopoli, M.; Pellegrini, M.; Gallo, S.; Zampetti, P.; Cuggia, G.; Scribante, A. Domiciliary Use of Chlorhexidine vs Postbiotic Gels in Patients with Peri-Implant Mucositis: A Split-Mouth Randomized Clinical Trial. Appl. Sci. 2022, 12, 2800. [Google Scholar] [CrossRef]
- Usui, M.; Onizuka, S.; Sato, T.; Kokabu, S.; Ariyoshi, W.; Nakashima, K. Mechanism of alveolar bone destruction in periodontitis—Periodontal bacteria and inflammation. Jpn. Dent. Sci. Rev. 2021, 57, 201–208. [Google Scholar] [CrossRef]
- Tsukasaki, M. RANKL and osteoimmunology in periodontitis. J. Bone Miner. Metab. 2021, 39, 82–90. [Google Scholar] [CrossRef]
- Hughes, C.E.; Nibbs, R.J.B. A guide to chemokines and their receptors. FEBS J. 2018, 285, 2944–2971. [Google Scholar] [CrossRef]
- Schall, T.J.; Jongstra, J.; Dyer, B.J.; Jorgensen, J.; Clayberger, C.; Davis, M.M.; Krensky, A.M. A human T cell-specific molecule is a member of a new gene family. J. Immunol. 1988, 141, 1018–1025. [Google Scholar] [CrossRef]
- Combadiere, C.; Ahuja, S.K.; Tiffany, H.L.; Murphy, P.M. Cloning and functional expression of CC CKR5, a human monocyte CC chemokine receptor selective for MIP-1(alpha), MIP-1(beta), and RANTES. J. Leukoc. Biol. 1996, 60, 147–152. [Google Scholar] [CrossRef]
- Raport, C.J.; Gosling, J.; Schweickart, V.L.; Gray, P.W.; Charo, I.F. Molecular cloning and functional characterization of a el human CC chemokine receptor (CCR5) for RANTES, MIP-1beta, and MIP-1alpha. J. Biol. Chem. 1996, 271, 17161–17166. [Google Scholar] [CrossRef]
- Neote, K.; DiGregorio, D.; Mak, J.Y.; Horuk, R.; Schall, T.J. Molecular cloning, functional expression, and signaling characteristics of a C-C chemokine receptor. Cell 1993, 72, 415–425. [Google Scholar] [CrossRef]
- Juremalm, M.; Olsson, N.; Nilsson, G. Selective CCL5/RANTES-induced mast cell migration through interactions with chemokine receptors CCR1 and CCR4. Biochem. Biophys. Res. Commun. 2002, 297, 480–485. [Google Scholar] [CrossRef]
- Combadiere, C.; Ahuja, S.K.; Murphy, P.M. Cloning and functional expression of a human eosinophil CC chemokine receptor. J. Biol. Chem. 1995, 270, 16491–16494. [Google Scholar] [CrossRef]
- Mortier, A.; Van Damme, J.; Proost, P. Regulation of chemokine activity by posttranslational modification. Pharmacol. Ther. 2008, 120, 197–217. [Google Scholar] [CrossRef]
- Volin, M.V.; Shah, M.R.; Tokuhira, M.; Haines, G.K.; Woods, J.M.; Koch, A.E. RANTES expression and contribution to monocyte chemotaxis in arthritis. Clin. Immunol. Immunopathol. 1998, 89, 44–53. [Google Scholar] [CrossRef]
- Fischer, F.R.; Luo, Y.; Luo, M.; Santambrogio, L.; Dorf, M.E. RANTES-induced chemokine cascade in dendritic cells. J. Immunol. 2001, 167, 1637–1643. [Google Scholar] [CrossRef]
- Loetscher, P.; Seitz, M.; Clark-Lewis, I.; Baggiolini, M.; Moser, B. Activation of NK cells by CC chemokines. Chemotaxis, Ca2+ mobilization, and enzyme release. J. Immunol. 1996, 156, 322–327. [Google Scholar] [CrossRef]
- Alam, R.; Stafford, S.; Forsythe, P.; Harrison, R.; Faubion, D.; Lett-Brown, M.A.; Grant, J.A. RANTES is a chemotactic and activating factor for human eosinophils. J. Immunol. 1993, 150 Pt 1, 3442–3448. [Google Scholar] [CrossRef] [PubMed]
- Bischoff, S.C.; Krieger, M.; Brunner, T.; Rot, A.; von Tscharner, V.; Baggiolini, M.; Dahinden, C.A. RANTES and related chemokines activate human basophil granulocytes through different G protein-coupled receptors. Eur. J. Immunol. 1993, 23, 761–767. [Google Scholar] [CrossRef] [PubMed]
- Murooka, T.T.; Rahbar, R.; Platanias, L.C.; Fish, E.N. CCL5-mediated T-cell chemotaxis involves the initiation of mRNA translation through mTOR/4E-BP1. Blood 2008, 111, 4892–4901. [Google Scholar] [CrossRef] [PubMed]
- Zhao, N.; Zhang, C.; Wu, Y.; Ding, J.; Wang, F.; Cheng, W.; Li, H.; Zhu, R. ROS-CCL5 axis recruits CD8+ T lymphocytes promoting the apoptosis of granulosa cells in diminished ovary reserve. J. Reprod. Immunol. 2023, 155, 103789. [Google Scholar] [CrossRef]
- Kimata, H.; Yoshida, A.; Ishioka, C.; Fujimoto, M.; Lindley, I.; Furusho, K. RANTES and macrophage inflammatory protein 1 alpha selectively enhance immunoglobulin (IgE) and IgG4 production by human B cells. J. Exp. Med. 1996, 183, 2397–2402. [Google Scholar] [CrossRef]
- Gamonal, J.; Bascones, A.; Jorge, O.; Silva, A. Chemokine RANTES in gingival crevicular fluid of adult patients with periodontitis. J. Clin. Periodontol. 2000, 27, 675–681. [Google Scholar] [CrossRef]
- Gamonal, J.; Acevedo, A.; Bascones, A.; Jorge, O.; Silva, A. Characterization of cellular infiltrate, detection of chemokine receptor CCR5 and interleukin-8 and RANTES chemokines in adult periodontitis. J. Periodontal Res. 2001, 36, 194–203. [Google Scholar] [CrossRef]
- Tymkiw, K.D.; Thunell, D.H.; Johnson, G.K.; Joly, S.; Burnell, K.K.; Cavanaugh, J.E.; Brogden, K.A.; Guthmiller, J.M. Influence of smoking on gingival crevicular fluid cytokines in severe chronic periodontitis. J. Clin. Periodontol. 2011, 38, 219–228. [Google Scholar] [CrossRef]
- Emingil, G.; Atilla, G.; Hüseyi, A. Gingival crevicular fluid monocyte chemoattractant protein-1 and RANTES levels in patients with generalized aggressive periodontitis. J. Clin. Periodontol. 2004, 31, 829–834. [Google Scholar] [CrossRef]
- Zein Elabdeen, H.R.; Mustafa, M.; Ali, R.; Bolstad, A.I. Cytokine profile in gingival crevicular fluid and plasma of patients with aggressive periodontitis. Acta Odontol. Scand. 2017, 75, 616–622. [Google Scholar] [CrossRef]
- Thunell, D.H.; Tymkiw, K.D.; Johnson, G.K.; Joly, S.; Burnell, K.K.; Cavanaugh, J.E.; Brogden, K.A.; Guthmiller, J.M. A multiplex immunoassay demonstrates reductions in gingival crevicular fluid cytokines following initial periodontal therapy. J. Periodontal Res. 2010, 45, 148–152. [Google Scholar] [CrossRef] [PubMed]
- Söderlin, M.K.; Persson, G.R.; Renvert, S.; Sanmartin Berglund, J. Cytokines in gingival crevicular fluid in elderly rheumatoid arthritis patients in a population-based cross-sectional study: RANTES was associated with periodontitis. J. Periodontal Res. 2021, 56, 907–916. [Google Scholar] [CrossRef]
- Shimada, Y.; Tabeta, K.; Sugita, N.; Yoshie, H. Profiling biomarkers in gingival crevicular fluid using multiplex bead immunoassay. Arch. Oral. Biol. 2013, 58, 724–730. [Google Scholar] [CrossRef] [PubMed]
- Haytural, O.; Yaman, D.; Ural, E.C.; Kantarci, A.; Demirel, K. Impact of periodontitis on chemokines in smokers. Clin. Oral. Investig. 2015, 19, 979–986. [Google Scholar] [CrossRef]
- Gamonal, J.; Acevedo, A.; Bascones, A.; Jorge, O.; Silva, A. Levels of interleukin-1 beta, -8, and -10 and RANTES in gingival crevicular fluid and cell populations in adult periodontitis patients and the effect of periodontal treatment. J. Periodontol. 2000, 71, 1535–1545. [Google Scholar] [CrossRef] [PubMed]
- Rath-Deschner, B.; Memmert, S.; Damanaki, A.; Nokhbehsaim, M.; Eick, S.; Cirelli, J.A.; Götz, W.; Deschner, J.; Jäger, A.; Nogueira, A.V.B. CXCL1, CCL2, and CCL5 modulation by microbial and biomechanical signals in periodontal cells and tissues-in vitro and in vivo studies. Clin. Oral. Investig. 2020, 24, 3661–3670. [Google Scholar] [CrossRef] [PubMed]
- de Queiroz, A.C.; Taba MJr O’Connell, P.A.; da Nóbrega, P.B.; Costa, P.P.; Kawata, V.K.; Trevisan, G.L.; aes ABJr de Souza, S.L.; Palioto, D.B.; Grisi, M.F. Inflammation markers in healthy and periodontitis patients: A preliminary data screening. Braz. Dent. J. 2008, 19, 3–8. [Google Scholar] [CrossRef]
- Souto, G.R.; Queiroz-ior, C.M.; Costa, F.O.; Mesquita, R.A. Smoking effect on chemokines of the human chronic periodontitis. Immunobiology 2014, 219, 633–636. [Google Scholar] [CrossRef]
- Kim, H.K.; Park, H.R.; Sul, K.H.; Chung, H.Y.; Chung, J. Induction of RANTES and CCR5 through NF-kappaB activation via MAPK pathway in aged rat gingival tissues. Biotechnol. Lett. 2006, 28, 17–23. [Google Scholar] [CrossRef]
- Bourgeois, D.; Bouchard, P.; Mattout, C. Epidemiology of periodontal status in dentate adults in France, 2002–2003. J. Periodontal Res. 2007, 42, 219–227. [Google Scholar] [CrossRef]
- Gürkan, A.; Eren, G.; Çetinkalp, Ş.; Akçay, Y.D.; Emingil, G.; Atilla, G. Monocyte chemotactic protein-1, RANTES and macrophage migration inhibitory factor levels in gingival crevicular fluid of metabolic syndrome patients with gingivitis. Arch. Oral. Biol. 2016, 69, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, H.G.; Idris, S.B.; Ahmed, M.F.; Åstrøm, A.N.; Mustafa, K.; Ibrahim, S.O.; Mustafa, M. Influence of type 2 diabetes on local production of inflammatory molecules in adults with and without chronic periodontitis: A cross-sectional study. BMC Oral. Health 2015, 15, 86. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.Z.; Yuan, Y.H.; Liu, H.H.; Li, S.S.; Zhang, B.W.; Chen, W.; An, Z.J.; Chen, S.Y.; Wu, Y.Z.; Han, B.; et al. Epidemiologic relationship between periodontitis and type 2 diabetes mellitus. BMC Oral. Health 2020, 20, 204. [Google Scholar] [CrossRef] [PubMed]
- Jönsson, D.; Aggarwal, P.; Nilsson, B.O.; Demmer, R.T. Beneficial effects of hormone replacement therapy on periodontitis are vitamin D associated. J. Periodontol. 2013, 84, 1048–1057. [Google Scholar] [CrossRef] [PubMed]
- Cheng, R.; Billet, S.; Liu, C.; Haldar, S.; Choudhury, D.; Tripathi, M.; Hav, M.; Merchant, A.; Hu, T.; Huang, H.; et al. Periodontal inflammation recruits distant metastatic breast cancer cells by increasing myeloid-derived suppressor cells. Oncogene 2020, 39, 1543–1556. [Google Scholar] [CrossRef] [PubMed]
- Mustafa, M.; Wondimu, B.; Bakhiet, M.; Modéer, T. Production of Rantes/CCL5 in human gingival fibroblasts challenged with tumor necrosis factor alpha. Eur. J. Oral. Sci. 2001, 109, 44–49. [Google Scholar] [CrossRef] [PubMed]
- Fageeh, H.N.; Fageeh, H.I.; Khan, S.S.; Maganur, P.C.; Vyas, N.; Patil, V.R.; Raj, A.T.; Baeshen, H.A.; Patil, S. Gingival crevicular fluid infiltrating CD14+ monocytes promote inflammation in periodontitis. Saudi, J. Biol. Sci. 2021, 28, 3069–3075. [Google Scholar] [CrossRef] [PubMed]
- Bodet, C.; Chandad, F.; Grenier, D. Inflammatory responses of a macrophage/epithelial cell co-culture model to mono and mixed infections with Porphyromonas gingivalis, Treponema denticola, and Tannerella forsythia. Microbes Infect. 2006, 8, 27–35. [Google Scholar] [CrossRef]
- Bodet, C.; Chandad, F.; Grenier, D. Porphyromonas gingivalis-induced inflammatory mediator profile in an ex vivo human whole blood model. Clin. Exp. Immunol. 2006, 143, 50–57. [Google Scholar] [CrossRef]
- Shaik-Dasthagirisaheb, Y.B.; Kantarci, A.; Gibson, F.C., 3rd. Immune response of macrophages from young and aged mice to the oral pathogenic bacterium Porphyromonas gingivalis. Immun. Ageing 2010, 7, 15. [Google Scholar] [CrossRef]
- Ohno, T.; Okahashi, N.; Morisaki, I.; Amano, A. Signaling pathways in osteoblast proinflammatory responses to infection by Porphyromonas gingivalis. Oral. Microbiol. Immunol. 2008, 23, 96–104. [Google Scholar] [CrossRef]
- Bekić, M.; Radaić, M.; Đokić, J.; Tomić, S.; Eraković, M.; Radojević, D.; Duka, M.; Marković, D.; Marković, M.; Ismaili, B.; et al. Mesenchymal Stromal Cells from Healthy and Inflamed Human Gingiva Respond Differently to Porphyromonas gingivalis. Int. J. Mol. Sci. 2022, 23, 3510. [Google Scholar] [CrossRef] [PubMed]
- Abhyankar, V.P.; Bates, A.M.; Fischer, C.L.; Johnson, G.K.; Guthmiller, J.M.; Progulske-Fox, A.; Brogden, K.A. Dataset on the chemokine and cytokine responses of multi-cell cultures treated with Porphyromonas gingivalis hemagglutinin B. Data Brief 2018, 22, 964–970. [Google Scholar] [CrossRef] [PubMed]
- Ansai, T.; Yamamoto, E.; Awano, S.; Yu, W.; Turner, A.J.; Takehara, T. Effects of periodontopathic bacteria on the expression of endothelin-1 in gingival epithelial cells in adult periodontitis. Clin. Sci. 2002, 103 (Suppl. S48), 327S–331S. [Google Scholar] [CrossRef] [PubMed]
- Palioto, D.B.; Finoti, L.S.; Kinane, D.F.; Benakanakere, M. Epigenetic and inflammatory events in experimental periodontitis following systemic microbial challenge. J. Clin. Periodontol. 2019, 46, 819–829. [Google Scholar] [CrossRef] [PubMed]
- Repeke, C.E.; Ferreira SBJr Claudino, M.; Silveira, E.M.; de Assis, G.F.; Avila-Campos, M.J.; Silva, J.S.; Garlet, G.P. Evidences of the cooperative role of the chemokines CCL3, CCL4 and CCL5 and its receptors CCR1+ and CCR5+ in RANKL+ cell migration throughout experimental periodontitis in mice. Bone 2010, 46, 1122–1130. [Google Scholar] [CrossRef]
- Chung, J.; Choi, M.J.; Jeong, S.Y.; Oh, J.S.; Kim, H.K. Chemokines gene expression of RAW 264.7 cells by Actinobacillus actinomycetemcomitans lipopolysaccharide using microarray and RT-PCR analysis. Mol. Cells 2009, 27, 257–261. [Google Scholar] [CrossRef]
- Alvarez, C.; Benítez, A.; Rojas, L.; Pujol, M.; Carvajal, P.; Díaz-Zúñiga, J.; Vernal, R. Differential expression of CC chemokines (CCLs) and receptors (CCRs) by human T lymphocytes in response to different Aggregatibacter actinomycetemcomitans serotypes. J. Appl. Oral. Sci. 2015, 23, 536–546. [Google Scholar] [CrossRef]
- Garlet, G.P.; Avila-Campos, M.J.; Milanezi, C.M.; Ferreira, B.R.; Silva, J.S. Actinobacillus actinomycetemcomitans-induced periodontal disease in mice: Patterns of cytokine, chemokine, and chemokine receptor expression and leukocyte migration. Microbes Infect. 2005, 7, 738–747. [Google Scholar] [CrossRef]
- Bodet, C.; Chandad, F.; Grenier, D. Modulation of cytokine production by Porphyromonas gingivalis in a macrophage and epithelial cell co-culture model. Microbes Infect. 2005, 7, 448–456. [Google Scholar] [CrossRef]
- Shih, Y.S.; Fu, E.; Fu, M.M.; Lin, F.G.; Chiu, H.C.; Shen, E.C.; Chiang, C.Y. Association of CCL5 and CCR5 gene polymorphisms with periodontitis in Taiwanese. J. Periodontol. 2014, 85, 1596–1602. [Google Scholar] [CrossRef] [PubMed]
- Nickel, R.G.; Casolaro, V.; Wahn, U.; Beyer, K.; Barnes, K.C.; Plunkett, B.S.; Freidhoff, L.R.; Sengler, C.; Plitt, J.R.; Schleimer, R.P.; et al. Atopic dermatitis is associated with a functional mutation in the promoter of the C-C chemokine RANTES. J. Immunol. 2000, 164, 1612–1616. [Google Scholar] [CrossRef] [PubMed]
- Savarrio, L.; Donati, M.; Carr, C.; Kinane, D.F.; Berglundh, T. Interleukin-24, RANTES and CCR5 gene polymorphisms are not associated with chronic adult periodontitis. J. Periodontal Res. 2007, 42, 152–158. [Google Scholar] [CrossRef] [PubMed]
- Kabashima, H.; Yoneda, M.; Nagata, K.; Hirofuji, T.; Maeda, K. The presence of chemokine (MCP-1, MIP-1alpha, MIP-1beta, IP-10, RANTES)-positive cells and chemokine receptor (CCR5, CXCR3)-positive cells in inflamed human gingival tissues. Cytokine 2002, 20, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Votta, B.J.; White, J.R.; Dodds, R.A.; James, I.E.; Connor, J.R.; Lee-Rykaczewski, E.; Eichman, C.F.; Kumar, S.; Lark, M.W.; Gowen, M. CKbeta-8 [CCL23], a el CC chemokine, is chemotactic for human osteoclast precursors and is expressed in bone tissues. J. Cell Physiol. 2000, 183, 196–207. [Google Scholar] [CrossRef]
- Yu, X.; Huang, Y.; Collin-Osdoby, P.; Osdoby, P. CCR1 chemokines promote the chemotactic recruitment, RANKL development, and motility of osteoclasts and are induced by inflammatory cytokines in osteoblasts. J. Bone Miner. Res. 2004, 19, 2065–2077. [Google Scholar] [CrossRef] [PubMed]
- Niwa, T.; Mizukoshi, K.; Azuma, Y.; Kashimata, M.; Shibutani, T. Fundamental study of osteoclast chemotaxis toward chemoattractants expressed in periodontitis. J. Periodontal Res. 2013, 48, 773–780. [Google Scholar] [CrossRef]
- Yano, S.; Mentaverri, R.; Kanuparthi, D.; Bandyopadhyay, S.; Rivera, A.; Brown, E.M.; Chattopadhyay, N. Functional expression of beta-chemokine receptors in osteoblasts: Role of regulated upon activation, normal T cell expressed and secreted (RANTES) in osteoblasts and regulation of its secretion by osteoblasts and osteoclasts. Endocrinology 2005, 146, 2324–2335. [Google Scholar] [CrossRef]
- Andrade, I., Jr.; Taddei, S.R.; Garlet, G.P.; Garlet, T.P.; Teixeira, A.L.; Silva, T.A.; Teixeira, M.M. CCR5 down-regulates osteoclast function in orthodontic tooth movement. J. Dent. Res. 2009, 88, 1037–1041. [Google Scholar] [CrossRef]
- Lee, J.S.; Lee, J.B.; Cha, J.K.; Choi, E.Y.; Park, S.Y.; Cho, K.S.; Kim, C.S. Chemokine in inflamed periodontal tissues activates healthy periodontal-ligament stem cell migration. J. Clin. Periodontol. 2017, 44, 530–539. [Google Scholar] [CrossRef]
- Nagata, M.; Iwasaki, K.; Akazawa, K.; Komaki, M.; Yokoyama, N.; Izumi, Y.; Morita, I. Conditioned Medium from Periodontal Ligament Stem Cells Enhances Periodontal Regeneration. Tissue Eng. Part. A 2017, 23, 367–377. [Google Scholar] [CrossRef] [PubMed]
- Fokkema, S.J.; Loos, B.G.; van der Velden, U. Monocyte-derived RANTES is intrinsically elevated in periodontal disease while MCP-1 levels are related to inflammation and are inversely correlated with IL-12 levels. Clin. Exp. Immunol. 2003, 131, 477–483. [Google Scholar] [CrossRef] [PubMed]

| Source | Result of the Analysis | Number of Patients/Number of Healthy People in the Study | Method of Analysis/Type of Sample |
|---|---|---|---|
| Gamonal et al., 2000 [46] | Higher concentration and total CCL5 in GCF in patients with periodontitis. CCL5 concentration is higher in GCF at sites with <3 mm probing depth than in >6 mm, but total CCL5 is at the same level regardless of probing depth. | 12 people with periodontitis; 12 healthy subjects | ELISA; GCF |
| Gamonal et al., 2001 [47] | Higher CCL5 concentrations in patients with periodontitis. Quantity and concentration are not significantly associated with probing depth. | 12 people with periodontitis; 6 healthy subjects | ELISA; GCF |
| Tymkiw et al., 2011 [48] | CCL5 concentration is higher in sites affected by periodontitis than in sites not affected by periodontitis. CCL5 concentration is higher in sites covered by periodontitis compared to samples from healthy subjects. In smokers, reduced CCL5 concentration in sites covered and not covered by periodontitis. | 40 patients with periodontitis (20 smokers, 20 non-smokers); 12 healthy subjects | multiplexed fluorescent bead-based immunoassay; GCF |
| Emingil et al., 2004 [49] | Higher total CCL5 per sample in patients with generalized aggressive periodontitis than in healthy subjects; concentrations did not differ between patients and healthy subjects. The amount of CCL5 positively correlated with probing depth and clinical attachment loss. | 20 people with periodontitis; 10 healthy subjects | Enzyme immunoassay; GCF |
| Zein Elabdeen et al., 2017 [50] | CCL5 levels are reduced in patients with periodontitis compared to healthy subjects. CCL5 levels in GCF do not differ between patients with periodontitis and healthy subjects. | 19 people with aggressive periodontitis; 19 healthy subjects | multiplex fluorescent bead-based immunoassays; plasma |
| Thunell et al., 2010 [51] | The concentration of CCL5 in sites affected by periodontitis does not differ from non-periodontitis sites. After therapy, CCL5 concentration decreases. | 6 patients with generalized severe chronic periodontitis | multiplex fluorescent bead-based immunoassay; GCF |
| Söderlin et al., 2021 [52] | CCL5 concentration is higher but not statistically significant (p = 0.087) in patients with periodontitis and with rheumatoid arthritis compared to patients with rheumatoid arthritis but without periodontitis. CCL5 concentration positively correlated with patient age and smoking but not with gender and BMI. | 80 people with periodontitis and rheumatoid arthritis; 52 people with rheumatoid arthritis but without periodontitis | xMap technology (bead-based multiplexed immunoassay); GCF |
| Shimada et al., 2013 [53] | Higher total CCL5 in periodontitis sites than in non-periodontitis sites. | 11 patients with generalized chronic periodontitis | multiplexed fluorescent bead-based immunoassay; GCF |
| Haytural et al., 2015 [54] | Total CCL5 in GCF is elevated in patients with periodontitis relative to healthy subjects. Smoking does not affect the total CCL5 in GCF. | 30 patients with chronic periodontitis (20 smokers) and 20 healthy subjects (10 smokers) | multiplex immunoassay; GCF |
| Haytural et al., 2015 [54] | Serum CCL5 concentrations in patients with periodontitis do not differ from healthy subjects. Smoking does not affect serum CCL5 concentrations. | 30 patients with chronic periodontitis (20 smokers) and 20 healthy subjects (10 smokers) | multiplex immunoassay; serum |
| Gamonal et al., 2000 [55] | CCL5 concentration is higher in GCF at sites with <3 mm probing depth than in >6 mm, but total CCL5 is at the same level regardless of probing depth. | 12 people with periodontitis; 12 healthy subjects | ELISA; GCF |
| Rath-Deschner et al., 2020 [56] | Higher expression of CCL5 at the mRNA level in people with periodontitis. | 6 people with periodontitis; 6 samples from healthy sites | qRT-PCR; gingival biopsies |
| de Queiroz et al. 2008 [57] | CCL5 levels are reduced in patients with periodontitis relative to healthy subjects. | 17 people with chronic periodontitis; 8 healthy subjects | Inflammatory flow cytometry multiplex assays; serum |
| Souto et al., 2014 [58] | Higher CCL5 concentrations in smokers than in non-smokers. | 43 patients with chronic periodontitis (23 smokers, 20 non-smokers) | ELISA; gingival tissues |
| Description | Characteristic |
|---|---|
| Inflammatory responses are increased in older adults. In subjects with rheumatoid arthritis and periodontitis, CCL5 levels correlated positively with age [52]. Other studies have not confirmed this [46]. | Age |
| CCL5 levels are higher in obese patients with gingivitis than in lean patients with and without gingivitis [61]. Other studies have not confirmed this [52]. | Obesity |
| CCL5 levels not related to gender in periodontitis patients. | Sex |
| Higher CCL5 levels in smokers with periodontitis than in non-smokers with periodontitis [52,58]. The opposite has also been observed [48]. | Cigarette smoking |
| Patients with diabetes and periodontitis have lower CCL5 levels than patients with diabetes alone [62]. | Diabetes |
| Unrelated. | Rheumatoid arthritis |
| Description | Mechanism |
|---|---|
| Potential recruitment of Th1 cells and macrophages by CCL5 | Immune cell infiltration |
| Increased osteoclast activity (stimulation of osteoclast precursors to fuse, induction of osteoclast chemotaxis). | Alveolar bone destruction |
| CCL5 induces chemotaxis of PDLSCs and, thus, regenerative effects on lesions caused by periodontitis. | PDLSCs |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barczak, K.; Droździk, A.; Bosiacki, M.; Łagocka, R.; Cenariu, D.; Uriciuc, W.A.; Baranowska-Bosiacka, I. CCL5’s Role in Periodontal Disease: A Narrative Review. Int. J. Mol. Sci. 2023, 24, 17332. https://doi.org/10.3390/ijms242417332
Barczak K, Droździk A, Bosiacki M, Łagocka R, Cenariu D, Uriciuc WA, Baranowska-Bosiacka I. CCL5’s Role in Periodontal Disease: A Narrative Review. International Journal of Molecular Sciences. 2023; 24(24):17332. https://doi.org/10.3390/ijms242417332
Chicago/Turabian StyleBarczak, Katarzyna, Agnieszka Droździk, Mateusz Bosiacki, Ryta Łagocka, Diana Cenariu, Willi Andrei Uriciuc, and Irena Baranowska-Bosiacka. 2023. "CCL5’s Role in Periodontal Disease: A Narrative Review" International Journal of Molecular Sciences 24, no. 24: 17332. https://doi.org/10.3390/ijms242417332
APA StyleBarczak, K., Droździk, A., Bosiacki, M., Łagocka, R., Cenariu, D., Uriciuc, W. A., & Baranowska-Bosiacka, I. (2023). CCL5’s Role in Periodontal Disease: A Narrative Review. International Journal of Molecular Sciences, 24(24), 17332. https://doi.org/10.3390/ijms242417332






