An Overview of Recent Developments in the Management of Burn Injuries
Abstract
:1. Introduction
2. The Aim of the Review and Search Strategy
3. Initial Assessment
3.1. The Total Body Surface Area (TBSA)
3.2. Fluid Resuscitation
3.3. Thermoregulation
4. Wound Healing
4.1. Escharotomy
4.2. Debridement
4.3. Topical Treatment
5. Dressing
5.1. Dressings Made of Natural Polymers
5.2. Dressings Made of Inorganic Materials
5.3. Dressings Made of Nanomaterials (NMs)
6. Excision
7. Permanent Wound Coverage
7.1. Autografts
7.1.1. Split-Thickness Skin Graft (STSG)
7.1.2. Full-Thickness Skin Graft (FTSG)
7.1.3. Cultured Epithelial Autograft (CEA)
7.2. Flaps
8. Temporary Wound Coverage
8.1. Allografts and Xenografts
8.2. Human Amnion
8.3. Artificial Skin (Biosynthetic Dressings)
8.4. Fish Skin Grafts (FSG)
9. Additional Actions
9.1. Maggot Debridement Therapy (MDT)
9.2. Negative Pressure Wound Therapy (NPWT)
9.3. Hyperbaric Oxygen Therapy (HBOT)
9.4. Platelet-Rich Plasma (PRP)
9.5. Mesenchymal Stem Cells
9.6. Growth Factor Therapy (GFT)
10. Nutrition/Hypermetabolism
11. Pain Management
12. Psychological Advisory
13. Rehabilitation and Scar Treatment during and after the Acute Phase
14. Systemic Antibiotic Therapy
15. Conclusions and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Johnson, C. Management of burns. Surgery 2018, 36, 435–440. [Google Scholar] [CrossRef]
- Mądry, R.; Strużyna, J. Zastosowanie opatrunków srebrowych w leczeniu oparzeń i odmrożeń, zespołu Lyella oraz ran przewlekłych. Plast. Surg. Burn. 2020, 8, 115–122. [Google Scholar] [CrossRef]
- Przekora, A. A Concise Review on Tissue Engineered Artificial Skin Grafts for Chronic Wound Treatment: Can We Reconstruct Functional Skin Tissue In Vitro? Cells 2020, 9, 1622. [Google Scholar] [CrossRef]
- Winiarska, A.; Korzeniowski, T.; Strużyna, J. Leczenie bólu jako ważny element terapii w oddziale oparzeniowym—przegląd literatury. Plast. Surg. Burn. 2021, 9, 93–100. [Google Scholar] [CrossRef]
- Chrzanowska-Wąsik, M.; Chemperek, E.; Sokołowski, D.; Goniewicz, M.; Bednarz, K.; Rzońca, P. Analiza oparzeń osób dorosłych hospitalizowanych we Wschodnim Centrum Leczenia Oparzeń i Chirurgii Rekonstrukcyjnej w Łęcznej. J. Educ. 2017, 7, 2391–8306. [Google Scholar] [CrossRef]
- Żwierełło, W.; Piorun, K.; Skórka-Majewicz, M.; Maruszewska, A.; Antoniewski, J.; Gutowska, I. Burns: Classification, Pathophysiology, and Treatment: A Review. Int. J. Mol. Sci. 2023, 24, 3749. [Google Scholar] [CrossRef]
- Jeschke, M.G.; Kamolz, L.P.; Shahrokhi, S. Burn Care and Treatment; Springer: Vienna, Austria, 2013. [Google Scholar]
- Yakupu, A.; Zhang, J.; Dong, W.; Song, F.; Dong, J.; Lu, S. The epidemiological characteristic and trends of burns globally. BMC Public Health 2022, 22, 1596. [Google Scholar] [CrossRef]
- GBD 2019 Diseases and Injuries Collaborators. Global burden of 369 diseases and injuries in 204 countries and territories, 1990-2019: A systematic analysis for the Global Burden of Disease Study 2019. Lancet 2020, 396, 1204–1222. [Google Scholar] [CrossRef]
- Greenhalgh, D.G. Management of Burns. N. Engl. J. Med. 2019, 380, 2349–2359. [Google Scholar] [CrossRef]
- Marion, J.W.; Lee, J.; Rosenblum, J.S.; Buckley, T.J. Assessment of temperature and ultraviolet radiation effects on sunburn incidence at an inland U.S. Beach: A cohort study. Environ. Res. 2018, 161, 479–484. [Google Scholar] [CrossRef]
- Guy, G.P., Jr.; Berkowitz, Z.; Watson, M. Estimated Cost of Sunburn-Associated Visits to US Hospital Emergency Departments. JAMA Dermatol. 2017, 153, 90–92. [Google Scholar] [CrossRef] [PubMed]
- Nunez Lopez, O.; Cambiaso-Daniel, J.; Branski, L.; Norbury, W.; Herndon, D. Predicting and managing sepsis in burn patients: Current perspectives. Ther. Clin. Risk Manag. 2017, 13, 1107–1117. [Google Scholar] [CrossRef] [PubMed]
- Vivó, C.; Galeiras, R.; del Caz, M.D.P. Initial evaluation and management of the critical burn patient. Med. Intensiva 2016, 40, 49–59. [Google Scholar] [CrossRef]
- Van Loey, N.E.E.; Van Son, M.J.M. Psychopathology and Psychological Problems in Patients with Burn Scars. Am. J. Clin. Dermatol. 2003, 4, 245–272. [Google Scholar] [CrossRef] [PubMed]
- Jeffrey, J.E. Essentials of Plastic Surgery, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2014. [Google Scholar]
- Hettiaratchy, S.; Dziewulski, P. ABC of burns: Pathophysiology and types of burns. BMJ 2004, 328, 1427–1429. [Google Scholar] [CrossRef]
- Rowan, M.P.; Cancio, L.C.; Elster, E.A.; Burmeister, D.M.; Rose, L.F.; Natesan, S.; Chan, R.K.; Christy, R.J.; Chung, K.K. Burn wound healing and treatment: Review and advancements. Crit. Care 2015, 19, 243. [Google Scholar] [CrossRef]
- Song, D.H.; Neligan, P.C. Plastic Surgery, 4th ed.; Trunk and Lower Extremity; Elsevier: Amsterdam, The Netherlands, 2017. [Google Scholar]
- Szymański, K.; Waś, J. The potential for healing and regeneration of skin in extensive burn wounds using skin regenerating substitutes. Leczenie Ran 2014, 11, 11–20. [Google Scholar] [CrossRef]
- Larouche, J.; Sheoran, S.; Maruyama, K.; Martino, M.M. Immune Regulation of Skin Wound Healing: Mechanisms and Novel Therapeutic Targets. Adv. Wound Care 2018, 7, 209–231. [Google Scholar] [CrossRef]
- Frykberg, R.G.; Banks, J. Challenges in the Treatment of Chronic Wounds. Adv. Wound Care 2015, 4, 560–582. [Google Scholar] [CrossRef]
- Hermans, M.H.E. An Introduction to Burn Care. Adv. Skin Wound Care 2019, 32, 9–18. [Google Scholar] [CrossRef]
- Johnson, R.M.; Richard, R. Partial-Thickness Burns: Identification and Management. Adv. Skin Wound Care 2003, 16, 178–187. [Google Scholar] [CrossRef] [PubMed]
- Markiewicz-Gospodarek, A.; Kozioł, M.; Tobiasz, M.; Baj, J.; Radzikowska-Büchner, E.; Przekora, A. Burn Wound Healing: Clinical Complications, Medical Care, Treatment, and Dressing Types: The Current State of Knowledge for Clinical Practice. Int. J. Environ. Res. Public Health 2022, 19, 1338. [Google Scholar] [CrossRef] [PubMed]
- Thorne, C.H.; Chung, K.C.; Gosain, A.K.; Gurtner, G.C.; Mehrara, B.J.; Rubin, J.P.; Spear, S.L. Grabb and Smith’s Plastic Surgery, 7th ed.; Wolters Kluwer Health Adis (ESP)/Lippincott Williams & Wilkins Health: Philadelphia, PA, USA, 2013. [Google Scholar]
- European Burn Practice Guidelines, 4th ed.; European Burns Association: BC’s-Hertogenbosch, The Netherlands, 2017; Available online: https://www.euroburn.org/wp-content/uploads/EBA-Guidelines-Version-4-2017.pdf (accessed on 7 October 2007).
- Cartotto, R.; Johnson, L.; Rood, J.M.; Lorello, D.; Matherly, A.; Parry, I.; Romanowski, K.; Wiechman, S.; Bettencourt, A.; Carson, J.S.; et al. Clinical Practice Guideline: Early Mobilization and Rehabilitation of Critically Ill Burn Patients. J. Burn Care Res. 2023, 44, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Leclerc, T.; Sjöberg, F.; Jennes, S.; Martinez-Mendez, J.R.; van der Vlies, C.H.; Battistutta, A.; Lozano-Basanta, J.A.; Moiemen, N.; Almeland, S.K. European Burns Association guidelines for the management of burn mass casualty incidents within a European response plan. Burns 2023, 49, 275–303. [Google Scholar] [CrossRef] [PubMed]
- Żwierełło, W.; Styburski, D.; Maruszewska, A.; Piorun, K.; Skórka-Majewicz, M.; Czerwińska, M.; Maciejewska, D.; Baranowska-Bosiacka, I.; Krajewski, A.; Gutowska, I. Bioelements in the treatment of burn injuries—The complex review of metabolism and supplementation (copper, selenium, zinc, iron, manganese, chromium and magnesium). J. Trace Elem. Med. Biol. 2020, 62, 126616. [Google Scholar] [CrossRef]
- Evers, L.H.; Bhavsar, D.; Mailänder, P. The biology of burn injury. Exp. Dermatol. 2010, 19, 777–783. [Google Scholar] [CrossRef]
- Barrow, R.E.; Jeschke, M.G.; Herndon, D.N. Early fluid resuscitation improves outcomes in severely burned children. Resuscitation 2000, 45, 91–96. [Google Scholar] [CrossRef]
- Mehta, M.; Tudor, G.J. Parkland Formula. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2023. Available online: https://www.ncbi.nlm.nih.gov/books/NBK537190/ (accessed on 19 June 2023).
- Haberal, M.; Sakallioglu Abali, A.E.; Karakayali, H. Fluid management in major burn injuries. Indian J. Plast. Surg. 2010, 43, S29–S36. [Google Scholar] [CrossRef]
- Mitchell, K.B.; Khalil, E.; Brennan, A.; Shao, H.; Rabbitts, A.; Leahy, N.E.; Yurt, R.W.; Gallagher, J.J. New management strategy for fluid resuscitation: Quantifying volume in the first 48 hours after burn injury. J. Burn Care Res. 2013, 34, 196–202. [Google Scholar] [CrossRef]
- Coca, S.G.; Bauling, P.; Schifftner, T.; Howard, C.S.; Teitelbaum, I.; Parikh, C.R. Contribution of acute kidney injury toward morbidity and mortality in burns: A contemporary analysis. Am. J. Kidney Dis. 2007, 49, 517–523. [Google Scholar] [CrossRef]
- Dries, D.J.; Waxman, K. Adequate resuscitation of burn patients may not be measured by urine output and vital signs. Crit. Care Med. 1991, 19, 327–329. [Google Scholar] [CrossRef] [PubMed]
- Cancio, L.C. Initial Assessment and Fluid Resuscitation of Burn Patients. Surg. Clin. N. Am. 2014, 94, 741–754. [Google Scholar] [CrossRef] [PubMed]
- Yoshino, Y.; Ohtsuka, M.; Kawaguchi, M.; Sakai, K.; Hashimoto, A.; Hayashi, M.; Madokoro, N.; Asano, Y.; Abe, M.; Ishii, T.; et al. The wound/burn guidelines—6: Guidelines for the management of burns. J. Dermatol. 2016, 43, 989–1010. [Google Scholar] [CrossRef] [PubMed]
- Regan, A.; Hotwagner, D.T. Burn Fluid Management. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2023. Available online: https://www.ncbi.nlm.nih.gov/books/NBK534227/ (accessed on 20 June 2023).
- Blumetti, J.; Hunt, J.L.; Arnoldo, B.D.; Parks, J.K.; Purdue, G.F. The Parkland formula under fire: Is the criticism justified? J. Burn Care Res. 2008, 29, 180–186. [Google Scholar] [CrossRef]
- Daniels, M.; Fuchs, P.C.; Lefering, R.; Grigutsch, D.; Seyhan, H.; Limper, U.; The German Burn Registry; Schiefer, J.L. Is the Parkland formula still the best method for determining the fluid resuscitation volume in adults for the first 24 hours after injury?—A retrospective analysis of burn patients in Germany. Burns 2021, 47, 914–921. [Google Scholar] [CrossRef]
- Greenhalgh, D.G.; Cartotto, R.; Taylor, S.L.; Fine, J.R.; Lewis, G.M.; Smith, D.J., Jr.; Marano, M.A.; Gibson, A.; Wibbenmeyer, L.A.; Holmes, J.H.; et al. Burn Resuscitation Practices in North America: Results of the Acute Burn Resuscitation Multicenter Prospective Trial (ABRUPT). Ann. Surg. 2023, 277, 512–519. [Google Scholar] [CrossRef]
- Chung, K.K.; Blackbourne, L.H.; Wolf, S.E.; White, C.E.; Renz, E.M.; Cancio, L.C.; Holcomb, J.B.; Barillo, D.J. Evolution of burn resuscitation in operation Iraqi freedom. J. Burn Care Res. 2006, 27, 606–611. [Google Scholar] [CrossRef]
- Strobel, A.M.; Fey, R. Emergency Care of Pediatric Burns. Emerg. Med. Clin. N. Am. 2018, 36, 441–458. [Google Scholar] [CrossRef]
- Romanowski, K.S.; Palmieri, T.L. Pediatric burn resuscitation: Past, present, and future. Burn Trauma 2017, 5, 26. [Google Scholar] [CrossRef]
- Demir, S.; Oztorun, C.I.; Erturk, A.; Guney, D.; Ertoy, A.; Doruk, H.; Tanriverdi, F.; Azili, M.N.; Senel, E. Approaches of Emergency Department Physicians to Pediatric Burns: A Survey Assessment. J. Burn Care Res. 2022, 43, 115–120. [Google Scholar] [CrossRef]
- Cartotto, R.C.; Innes, M.; Musgrave, M.A.; Gomez, M.; Cooper, A.B. How well does the Parkland formula estimate actual fluid resuscitation volumes? J. Burn Care Rehabil. 2002, 23, 258–265. [Google Scholar] [CrossRef]
- Engrav, L.H.; Colescott, P.L.; Kemalyan, N.; Heimbach, D.M.; Gibran, N.S.; Solem, L.D.; Dimick, A.R.; Gamelli, R.L.; Lentz, C.W. A biopsy of the use of the Baxter formula to resuscitate burns or do we do it like Charlie did it? J. Burn Care Rehabil. 2000, 21, 91–95. [Google Scholar] [CrossRef] [PubMed]
- Lindahl, L.; Oksanen, T.; Lindford, A.; Varpula, T. Initial fluid resuscitation guided by the Parkland formula leads to high fluid volumes in the first 72 h, increasing mortality and the risk for kidney injury. Burn. Open 2023, 7, 51–58. [Google Scholar] [CrossRef]
- Myburgh, J. Patient-Centered Outcomes and Resuscitation Fluids. N. Engl. J. Med. 2018, 378, 862–863. [Google Scholar] [CrossRef] [PubMed]
- Cappuyns, L.; Tridente, A.; Stubbington, Y.; Dempsey-Hibbert, N.C.; Shokrollahi, K. Review of Burn Resuscitation: Is Plasmalyte® a Comparable Alternative to Ringer’s Lactate? J. Burn Care Res. 2023, 44, 81–86. [Google Scholar] [CrossRef]
- Peeters, Y.; Vandervelden, S.; Wise, R.; Malbrain, M.L. An overview on fluid resuscitation and resuscitation endpoints in burns: Past, present and future. Part 1—historical background, resuscitation fluid and adjunctive treatment. Anaesthesiol. Intensive Ther. 2015, 47, 6–14. [Google Scholar] [CrossRef] [PubMed]
- Peeters, Y.; Lebeer, M.; Wise, R.; Malbrain, M.L. An overview on fluid resuscitation and resuscitation endpoints in burns: Past, present and future. Part 2—avoiding complications by using the right endpoints with a new personalized protocolized approach. Anaesthesiol. Intensive Ther. 2015, 47, 15–26. [Google Scholar] [CrossRef]
- Saffle, J.I. The phenomenon of “fluid creep” in acute burn resuscitation. J. Burn Care Res. 2007, 28, 382–395. [Google Scholar] [CrossRef]
- Marik, P.E. Iatrogenic salt water drowning and the hazards of a high central venous pressure. Ann. Intensive Care 2014, 4, 21. [Google Scholar] [CrossRef]
- Sullivan, S.R.; Friedrich, J.B.; Engrav, L.H.; Round, K.A.; Heimbach, D.M.; Heckbert, S.R.; Carrougher, G.J.; Lezotte, D.C.; Wiechman, S.A.; Honari, S.; et al. “Opioid creep” is real and may be the cause of “fluid creep”. Burns 2004, 30, 583–590. [Google Scholar] [CrossRef]
- Kahn, S.A.; Beers, R.J.; Lentz, C.W. Resuscitation after severe burn injury using high-dose ascorbic acid: A retrospective review. J. Burn Care Res. 2011, 32, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Walker, T.L.; Rodriguez, D.U.; Coy, K.; Hollén, L.I.; Greenwood, R.; Young, A.E. Impact of reduced resuscitation fluid on outcomes of children with 10–20% body surface area scalds. Burns 2014, 40, 1581–1586. [Google Scholar] [CrossRef] [PubMed]
- Klein, M.B.; Edwards, J.A.; Kramer, C.B.; Nester, T.; Heimbach, D.M.; Gibran, N.S. The beneficial effects of plasma exchange after severe burn injury. J. Burn Care Res. 2009, 30, 243–248. [Google Scholar] [CrossRef] [PubMed]
- Osilla, E.V.; Marsidi, J.L.; Shumway, K.R.; Sharma, S. Physiology, Temperature Regulation. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2023. [Google Scholar]
- Rizzo, J.A.; Rowan, M.P.; Driscoll, I.R.; Chan, R.K.; Chung, K.K. Perioperative Temperature Management During Burn Care. J. Burn Care Res. 2017, 38, e277–e283. [Google Scholar] [CrossRef] [PubMed]
- Bindu, B.; Bindra, A.; Rath, G. Temperature management under general anesthesia: Compulsion or option. J. Anaesthesiol. Clin. Pharmacol. 2017, 33, 306–316. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, B.; Kenngott, T.; Fischer, S.; Hundeshagen, G.; Hartmann, B.; Horter, J.; Münzberg, M.; Kneser, U.; Hirche, C. Early hypothermia as risk factor in severely burned patients: A retrospective outcome study. Burns 2019, 45, 1895–1900. [Google Scholar] [CrossRef]
- Driver, J.; Fielding, A.; Mullhi, R.; Chipp, E.; Torlinski, T. Temperature management of adult burn patients in intensive care: Findings from a retrospective cohort study in a tertiary center in the United Kingdom. Anaesthesiol. Intensive Ther. 2022, 54, 226–233. [Google Scholar] [CrossRef]
- Alonso-Fernández, J.M.; Lorente-González, P.; Pérez-Munguía, L.; Cartón-Manrique, A.M.; Peñas-Raigoso, M.C.; Martín-Ferreira, T. Analysis of hypothermia through the acute phase in major burns patients: Nursing care. Enferm Intensiva. 2019, 31, 120–130. [Google Scholar] [CrossRef]
- Page, D.; Trembley, A.L. II Are We Keeping Our Burn Patients Warm Enough? JEMS 2014, 9, 15. Available online: https://www.jems.com/patient-care/are-we-keeping-our-burn-patients-warm-en/ (accessed on 27 April 2023).
- Jeschke, M.G.; van Baar, M.E.; Choudhry, M.A.; Chung, K.K.; Gibran, N.S.; Logsetty, S. Burn injury. Nat. Rev. Dis. Primers. 2020, 6, 11. [Google Scholar] [CrossRef]
- Porter, C.; Tompkins, R.G.; Finnerty, C.C.; Sidossis, L.S.; Suman, O.E.; Herndon, D.N. The metabolic stress response to burn trauma: Current understanding and therapies. Lancet 2016, 388, 1417–1426. [Google Scholar] [CrossRef]
- Williams, F.N.; Herndon, D.N.; Jeschke, M.G. The hypermetabolic response to burn injury and interventions to modify this response. Clin. Plast. Surg. 2009, 36, 583–596. [Google Scholar] [CrossRef] [PubMed]
- Wilmore, D.W.; Mason, A.D., Jr.; Johnson, D.W.; Pruitt, B.A., Jr. Effect of ambient temperature on heat production and heat loss in burn patients. J. Appl. Physiol. 1975, 38, 593–597. [Google Scholar] [CrossRef] [PubMed]
- Ahuja, R.B. ISBI Practice Guidelines for Burn Care: Editorial. Burns 2016, 42, 951–952. [Google Scholar] [CrossRef] [PubMed]
- Mullhi, R.; Ewington, I.; Chipp, E.; Torlinski, T. A descriptive survey of operating theater and intensive care unit temperature management of burn patients in the United Kingdom. Int. J. Burn. Trauma 2021, 11, 136–144. [Google Scholar]
- Pruskowski, K.A.; Rizzo, J.A.; Shields, B.A.; Chan, R.K.; Driscoll, I.R.; Rowan, M.P.; Chung, K.K. A Survey of Temperature Management Practices Among Burn Centers in North America. J. Burn Care Res. 2018, 39, 612–617. [Google Scholar] [CrossRef]
- Lang, D.H.; Ba, T.; Cao, S.J.; Li, F.; Dong, H.; Li, J.L.; Wang, L.F. Research advances on signaling pathways affecting sweat gland development and their involvement in the reconstitution of sweat adenoid cells in vitro. Zhonghua Shao Shang Za Zhi 2022, 38, 195–200. [Google Scholar] [CrossRef]
- Zhang, J.J.; Wang, M.Y.; Zhao, J.; Jiang, D.Y. Research advances on the application of stem cells in sweat gland regeneration. Zhonghua Shao Shang Za Zhi 2022, 38, 296–300. [Google Scholar] [CrossRef]
- Zeng, Y.N.; Kang, Y.B.; Xu, Y.A. Research advances on skin sweat gland regeneration induced by stem cells and tissue engineering. Zhonghua Shao Shang Za Zhi 2021, 37, 900–904. [Google Scholar] [CrossRef]
- Rezvani Ghomi, E.; Khalili, S.; Nouri Khorasani, S.; Esmaeely Neisiany, R.; Ramakrishna, S. Wound dressings: Current advances and future directions. J. Appl. Polym. Sci. 2019, 136, 47738. [Google Scholar] [CrossRef]
- Gacto-Sanchez, P. Surgical treatment and management of the severely burn patient: Review and update. Med. Intensiva 2017, 41, 356–364. [Google Scholar] [CrossRef] [PubMed]
- Butts, C.C.; Holmes, J.H.; Carter, J.E. Surgical Escharotomy and Decompressive Therapies in Burns. J. Burn Care Res. 2020, 41, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Wise, R.J.; Strużyna, J.; Antonov, S.; Korzeniowski, T.; Bugaj, M.; Kuczyński, M.; Chrapusta, A.; Krajewski, A.; Charytonowicz, M. Abdominal compartment syndrome as a complication of the burn shock treatment—definition, prevention, treatment. Plast. Surg. Burn. 2014, 2, 85–90. [Google Scholar] [CrossRef]
- Surowiecka, A.; Korzeniowski, T.; Strużyna, J. Early burn wound excision in mass casualty events. Mil. Med. Res. 2022, 9, 42. [Google Scholar] [CrossRef]
- Almeland, S.K.; Hughes, A.; Leclerc, T.; Ogura, T.; Hayashi, M.; Mills, J.A.; Norton, I.; Potokar, T. Reply: Letter to the Editor on recommendations for burns care in mass casualty incidents: WHO Emergency Medical Teams Technical Working Group on Burns (WHO TWGB) 2017–2020. Burns 2022, 48, 482–484. [Google Scholar] [CrossRef]
- Daugherty, T.H.F.; Ross, A.; Neumeister, M.W. Surgical Excision of Burn Wounds: Best Practices Using Evidence-Based Medicine. Clin. Plast. Surg. 2017, 44, 619–625. [Google Scholar] [CrossRef] [PubMed]
- Williams, F.N.; Lee, J.O. Pediatric Burn Infection. Surg. Infect. 2021, 22, 54–57. [Google Scholar] [CrossRef]
- Korzeniowski, T.; Strużyna, J.; Chrapusta, A.M.; Krajewski, A.; Kucharzewski, M.; Piorun, K.; Nowakowski, J.; Surowiecka, A.; Kozicka, M.; Torres, K. A questionnaire-based study to obtain a consensus from 5 Polish burns centers on eschar removal by bromelain-based enzymatic debridement (Nexobrid®) in burns following the 2020 updated European consensus guidelines. Med. Sci. Monit. 2022, 28, e935632. [Google Scholar] [CrossRef]
- Griffin, B.; Bairagi, A.; Jones, L.; Dettrick, Z.; Holbert, M.; Kimble, R. Early non-excisional debridement of paediatric burns under general anaesthesia reduces time to re-epithelialisation and risk of skin graft. Sci. Rep. 2021, 11, 23753. [Google Scholar] [CrossRef]
- Zacharevskij, E.; Baranauskas, G.; Varkalys, K.; Rimdeika, R.; Kubilius, D. Comparison of non-surgical methods for the treatment of deep partial thickness skin burns of the hand. Burns 2018, 44, 445–452. [Google Scholar] [CrossRef]
- Krieger, Y.; Rubin, G.; Schulz, A.; Rosenberg, N.; Levi, A.; Singer, A.J.; Rosenberg, L.; Shoham, Y. Bromelain-based enzymatic debridement and minimal invasive modality (mim) care of deeply burned hands. Ann. Burn. Fire Disasters 2017, 30, 198–204. [Google Scholar]
- Klasen, H.J. A review on the nonoperative removal of necrotic tissue from burn wounds. Burns 2000, 26, 207–222. [Google Scholar] [CrossRef] [PubMed]
- Loo, Y.L.; Goh, B.K.L.; Jeffery, S. An Overview of the Use of Bromelain-Based Enzymatic Debridement (Nexobrid®) in Deep Partial and Full Thickness Burns: Appraising the Evidence. J. Burn Care Res. 2018, 39, 932–938. [Google Scholar] [CrossRef] [PubMed]
- Korzeniowski, T.; Grywalska, E.; Strużyna, J.; Bugaj-Tobiasz, M.; Surowiecka, A.; Korona-Głowniak, I.; Staśkiewicz, M.; Torres, K. Preliminary Single-Center Experience of Bromelain-Based Eschar Removal in Children with Mixed Deep Dermal and Full Thickness Burns. J. Clin. Med. 2022, 11, 4800. [Google Scholar] [CrossRef] [PubMed]
- Strużyna, J.; Surowiecka, A.; Korzeniowski, T. Letter to the Editor on Recommendations for burns care in mass casualty incidents: WHO Emergency Medical Teams Technical Working Group on Burns (WHO TWGB) 2017–2020. Burns 2021, 47, 1929–1930. [Google Scholar] [CrossRef]
- Ziegler, B.; Hirche, C.; Horter, J.; Kiefer, J.; Grützner, P.A.; Kremer, T.; Kneser, U.; Münzberg, M. In view of standardization Part 2: Management of challenges in the initial treatment of burn patients in Burn Centers in Germany, Austria and Switzerland. Burns 2017, 43, 318–325. [Google Scholar] [CrossRef]
- Negut, I.; Grumezescu, V.; Grumezescu, A.M. Treatment Strategies for Infected Wounds. Molecules 2018, 23, 2392. [Google Scholar] [CrossRef]
- Mihai, M.M.; Holban, A.M.; Giurcaneanu, C.; Popa, L.G.; Oanea, R.M.; Lazar, V.; Chifiriuc, M.C.; Popa, M.; Popa, M.I. Microbial biofilms: Impact on the pathogenesis of periodontitis, cystic fibrosis, chronic wounds and medical device-related infections. Curr. Top. Med. Chem. 2015, 15, 1552–1576. [Google Scholar] [CrossRef]
- Mihai, M.M.; Preda, M.; Lungu, I.; Gestal, M.C.; Popa, M.I.; Holban, A.M. Nanocoatings for Chronic Wound Repair—Modulation of Microbial Colonization and Biofilm Formation. Int. J. Mol. Sci. 2018, 19, 1179. [Google Scholar] [CrossRef]
- Norman, G.; Christie, J.; Liu, Z.; Westby, M.J.; Jefferies, J.M.; Hudson, T.; Edwards, J.; Mohapatra, D.P.; Hassan, I.A.; Dumville, J.C. Antiseptics for burns. Cochrane Database Syst. Rev. 2017, 7, CD011821. [Google Scholar] [CrossRef]
- Fox, C.L.; Monafo, W.W.; Ayvazian, V.H.; Skinner, A.M.; Modak, S.; Stanford, J.; Condict, C. Topical chemotherapy for burns using cerium salts and silver sulfadiazine. Surg. Gynecol. Obstet. 1977, 144, 668. [Google Scholar] [PubMed]
- Tenenhaus, M.; Rennekampff, H.O. Topical Agents and Dressings for Local Burn Wound Care. UpToDate. Available online: https://www.uptodate.com/contents/topical-agents-and-dressings-for-local-burn-wound-care (accessed on 3 February 2023).
- De Francesco, F.; Riccio, M.; Jimi, S. Contribution of Topical Agents such as Hyaluronic Acid and Silver Sulfadiazine to Wound Healing and Management of Bacterial Biofilm. Medicina 2022, 58, 835. [Google Scholar] [CrossRef] [PubMed]
- Wattanaploy, S.; Chinaroonchai, K.; Namviriyachote, N.; Muangman, P. Randomized Controlled Trial of Polyhexanide/Betaine Gel Versus Silver Sulfadiazine for Partial-Thickness Burn Treatment. Int. J. Low Extreme Wounds 2017, 16, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Guiomar, A.J.; Urbano, A.M. Polyhexanide-Releasing Membranes for Antimicrobial Wound Dressings: A Critical Review. Membranes 2022, 12, 1281. [Google Scholar] [CrossRef]
- Jeschke, M.; McCallum, C.; Baron, D.; Godleski, M.; Knighton, J.; Shahrokhi, S. Best practice recommendations for the prevention and management of burns. In Foundations of Best Practice for Skin and Wound Management; A Supplement of Wound Care Canada; The Canadian Association of Wound Care: Sudbury, ON, Canada, 2018; Wounds Canada; Available online: www.woundscanada.ca/docman/public/health-careprofessional/bpr-workshop/1308-bpr-for-the-prevention-and-management-of-burns/file (accessed on 4 March 2021).
- Saeidinia, A.; Keihanian, F.; Lashkari, A.P.; Lahiji, H.G.; Mobayyen, M.; Heidarzade, A.; Golchai, J. Partial-thickness burn wounds healing by topical treatment. Medicine 2017, 96, e6168. [Google Scholar] [CrossRef]
- Fang, Z.-Y.; Sheng, Z.-Y.; Li, N.; Ge, S.D. Modern Treatment of Severe Burns; Springer: Berlin/Heidelberg, Germany, 1992. [Google Scholar]
- Skowrońska, W.; Bazylko, A. The Potential of Medicinal Plants and Natural Products in the Treatment of Burns and Sunburn—A Review. Pharmaceutics 2023, 15, 633. [Google Scholar] [CrossRef]
- Shahzad, M.N.; Ahmed, N. Effectiveness of Aloe Vera gel compared with 1% silver sulphadiazine cream as burn wound dressing in second degree burns. J. Pak. Med. Assoc. 2013, 63, 225–230. [Google Scholar]
- Sharma, S.; Alfonso, A.R.; Gordon, A.J.; Kwong, J.; Lin, L.J.; Chiu, E.S. Second-Degree Burns and Aloe Vera: A Meta-Analysis and Systematic Review. Adv. Skin Wound Care 2022, 35, 1–9. [Google Scholar] [CrossRef]
- Khorasani, G.; Hosseinimehr, S.J.; Azadbakht, M.; Zamani, A.; Mahdavi, M.R. Aloe versus silver sulfadiazine creams for se-cond-degree burns: A randomized controlled study. Surg. Today 2009, 39, 587–591. [Google Scholar] [CrossRef]
- Nasiri, E.; Hosseinimehr, S.J.; Zaghi Hosseinzadeh, A.; Azadbakht, M.; Akbari, J.; Azadbakht, M. The effects of Arnebia euchroma ointment on second-degree burn wounds: A randomized clinical trial. J. Ethnopharmacol. 2016, 189, 107–116. [Google Scholar] [CrossRef]
- Asgarirad, H.; Chabra, A.; Rahimnejad, M.; Zaghi Hosseinzadeh, A.; Davoodi, A.; Azadbakht, M. Comparison of Albizia Julibressin and Silver Sulfadiazine in Healing of Second and Third Degree Burns. World J. Plast. Surg. 2018, 7, 34–44. [Google Scholar]
- Frew, Q.; Rennekampff, H.O.; Dziewulski, P.; Moiemen, N.; Zahn, T.; Hartmann, B. Betulin wound gel accelerated healing of super-ficial partial thickness burns: Results of a randomized, intra-individually controlled, phase III trial with 12-months follow-up. Burns 2019, 45, 876–890. [Google Scholar] [CrossRef] [PubMed]
- Abdullahzadeh, M.; Shafiee, S. To compare the effect of sea buckthorn and silver sulfadiazine dressing on period of wound healing in patients with second-degree burns: A randomized triple-blind clinical trial. Wound Repair Regen. 2021, 29, 732–740. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Deng, X.; Qiang, L.; Yao, M.; Guan, L.; Xie, N.; Zhao, D.; Ma, J.; Ma, L.; Wu, Y.; et al. Investigating the effects of walnut ointment on non-healing burn wounds. Burns 2021, 47, 455–465. [Google Scholar] [CrossRef] [PubMed]
- Jones, V.; Grey, J.E.; Harding, K.G. Wound dressings. BMJ 2006, 332, 777–780. [Google Scholar] [CrossRef]
- Hussain, Z.; Thu, H.E.; Shuid, A.N.; Katas, H.; Hussain, F. Recent Advances in Polymer-based Wound Dressings for the Treatment of Diabetic Foot Ulcer: An Overview of State-of-the-art. Curr. Drug Targets 2018, 19, 527–550. [Google Scholar] [CrossRef]
- Dong, Y.; Cui, M.; Qu, J.; Wang, X.; Hyung, S.; Barrera, J.; Elvassore, N.; Gurtner, G.C. Conformable hyaluronic acid hydrogel delivers adipose-derived stem cells and promotes regeneration of burn injury. Acta Biomater. 2020, 108, 56–66. [Google Scholar] [CrossRef]
- Türe, H. Characterization of hydroxyapatite-containing alginate—Gelatin composite films as a potential wound dressing. Int. J. Biol. Macromol. 2019, 123, 878–888. [Google Scholar] [CrossRef]
- El-Aassar, M.R.; Ibrahim, O.M.; Fouda, M.M.G.; El-Beheri, N.G.; Agwa, M.M. Wound healing of nanofiber comprising Polygalacturonic/Hyaluronic acid embedded silver nanoparticles: In-vitro and in-vivo studies. Carbohydr. Polym. 2020, 238, 116175. [Google Scholar] [CrossRef]
- Labovitiadi, O.; Driscoll, N.H.O.; Lamb, A.J.; Matthews, K.H. Rheological properties of gamma-irradiated antimicrobial wafers and in vitro efficacy against Pseudomonas aeruginosa. Int. J. Pharm. 2013, 453, 462–472. [Google Scholar] [CrossRef]
- Kataria, K.; Gupta, A.; Rath, G.; Mathur, R.B.; Dhakate, S.R. In vivo wound healing performance of drug loaded electrospun composite nanofibers transdermal patch. Int. J. Pharm. 2014, 469, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Mohandas, A.; Anisha, B.S.; Chennazhi, K.P.; Jayakumar, R. Chitosan—Hyaluronic acid/VEGF loaded fibrin nanoparticles composite sponges for enhancing angiogenesis in wounds. Coll. Surf. B Biointerfaces 2015, 127, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Kuddushi, M.; Patel, N.K.; Gawali, S.L.; Mata, J.P.; Montes-campos, H.; Varela, L.M.; Hassan, P.A.; Malek, N.I. Thermo-switchable de novo ionogel as metal absorbing and curcumin loaded smart bandage material. J. Mol. Liq. 2020, 306, 112922. [Google Scholar] [CrossRef]
- PrescQIPP. Wound Care: Silver Dressings. Bulletin 53, March 2014, v 2.0. (Creation Date: 5 June 2013). Available online: https://www.prescqipp.info/umbraco/surface/authorisedmediasurface/index?url=%2fmedia%2f1762%2fb53-wound-care-silver-dressings-201.pdf (accessed on 5 June 2013).
- Aramwit, P.; Muangman, P.; Namviriyachote, N.; Srichana, T. In vitro evaluation of the antimicrobial effectiveness and moisture binding properties of wound dressings. Int. J. Mol. Sci. 2010, 11, 2864–2874. [Google Scholar] [CrossRef]
- Jeschke, M.G.; Shahrokhi, S.; Finnerty, C.C.; Branski, L.K.; Dibildox, M. ABA Organization & Delivery of Burn Care Committee. Wound Coverage Technologies in Burn Care: Established Techniques. J. Burn Care Res. 2018, 39, 313–318. [Google Scholar] [CrossRef] [PubMed]
- Greenwood, J.E. A Randomized, Prospective Study of the Treatment of Superficial Partial-Thickness Burns: AWBAT-S Versus Biobrane. Eplasty 2011, 11, e10. [Google Scholar]
- Wasiak, J.; Cleland, H.; Campbell, F.; Spinks, A. Dressings for superficial and partial thickness burns. Cochrane Database Syst. Rev. 2013, 2013, CD002106. [Google Scholar] [CrossRef]
- Abdul Khalil, H.P.S.; Lai, T.K.; Tye, Y.Y.; Rizal, S.; Chong, E.W.N.; Yap, S.W.; Hamzah, A.A.; Fazita, M.R.N.; Paridah, M.T. A review of extractions of seaweed hydrocolloids: Properties and applications. Express Polym. Lett. 2018, 12, 296–317. [Google Scholar] [CrossRef]
- Li, C.; Wang, Y.; Hu, F.; Gong, H. Effect of hydrocolloid dressing combined with low molecular weight heparin and calcium on scar hyperplasia in burn patients with venous thromboembolism. Int. Wound J. 2023, 20, 2981–2988. [Google Scholar] [CrossRef]
- Martin, F.T.; O’Sullivan, J.B.; Regan, P.J.; McCann, J.; Kelly, J.L. Hydrocolloid dressing in pediatric burns may decrease operative intervention rates. J. Pediatr. Surg. 2010, 45, 600–605. [Google Scholar] [CrossRef]
- Witkowski, W. Wykorzystanie opatrunków hydrożelowych w leczeniu wojennych ran oparzeniowych. Chir. Plast. I Oparzenia Plast. Surg. Burn. 2019, 7, 37–41. [Google Scholar] [CrossRef]
- Cassano, R.; Trombino, S. Trehalose-based hydrogel potentially useful for the skin burn treatment. J. Appl. Polym. Sci. 2017, 134, 44755. [Google Scholar] [CrossRef]
- Kamoun, E.A.; Kenawy, E.R.S.; Chen, X. A review on polymeric hydrogel membranes for wound dressing applications: PVA-based hydrogel dressings. J. Adv. Res. 2017, 8, 217–233. [Google Scholar] [CrossRef] [PubMed]
- Goodwin, N.S.; Spinks, A.; Wasiak, J. The efficacy of hydrogel dressings as a first aid measure for burn wound management in the pre-hospital setting: A systematic review of the literature. Int. Wound J. 2016, 13, 519–525. [Google Scholar] [CrossRef] [PubMed]
- Schwarze, H.; Küntscher, M.; Uhlig, C.; Hierlemann, H.; Prantl, L.; Noack, N.; Hartmann, B. Suprathel®, a new skin substitute, in the management of donor sites of split-thickness skin grafts: Results of a clinical study. Burns 2007, 33, 850–854. [Google Scholar] [CrossRef] [PubMed]
- Eteraf-Oskouei, T.; Najafi, M. Traditional and modern uses of natural honey in human diseases: A review. Iran J. Basic Med. Sci. 2013, 16, 731–742. [Google Scholar]
- Febriyenti, F.; Lucida, H.; Almahdy, A.; Alfikriyah, I.; Hanif, M. Wound-Healing Effect of Honey Gel and Film. J. Pharm. Bioallied. Sci. 2019, 11, 176–180. [Google Scholar] [CrossRef]
- El-Kased, R.F.; Amer, R.I.; Attia, D.; Elmazar, M.M. Honey-based hydrogel: In vitro and comparative In vivo evaluation for burn wound healing. Sci. Rep. 2017, 7, 9692. [Google Scholar] [CrossRef]
- Alven, S.; Aderibigbe, B.A. Chitosan and Cellulose-Based Hydrogels for Wound Management. Int. J. Mol. Sci. 2020, 21, 9656. [Google Scholar] [CrossRef]
- Gheorghiță, D.; Moldovan, H.; Robu, A.; Bița, A.-I.; Grosu, E.; Antoniac, A.; Corneschi, I.; Antoniac, I.; Bodog, A.D.; Băcilă, C.I. Chitosan-Based Biomaterials for Hemostatic Applications: A Review of Recent Advances. Int. J. Mol. Sci. 2023, 24, 10540. [Google Scholar] [CrossRef]
- Moreno, J.A.S.; Mendes, A.C.; Stephansen, K.; Engwer, C.; Goycoolea, F.M.; Boisen, A.; Nielsen, L.H.; Chronakis, I.S. Development of electrosprayed mucoadhesive chitosan microparticles. Carbohydr. Polym. 2018, 190, 240–247. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Frauenlob, M.; Shibata, Y.; Wang, L.; Nakajima, T.; Nonoyama, T.; Tsuda, M.; Tanaka, S.; Kurokawa, T.; Gong, J.P. Chitin-Based Double-Network Hydrogel as Potential Superficial Soft-Tissue-Repairing Materials. Biomacromolecules 2020, 21, 4220–4230. [Google Scholar] [CrossRef] [PubMed]
- Mezzana, P. Clinical efficacy of a new chitin nanofibrils-based gel in wound healing. Acta Chir. Plast. 2008, 50, 81–84. [Google Scholar]
- Singh, R.; Shitiz, K.; Singh, A. Chitin and chitosan: Biopolymers for wound management. Int. Wound J. 2017, 14, 1276–1289. [Google Scholar] [CrossRef] [PubMed]
- Valachova, K.; Svik, K.; Biro, C.; Soltes, L. Skin wound healing with composite biomembranes loaded by tiopronin or captopril. J. Biotechnol. 2020, 310, 49–53. [Google Scholar] [CrossRef]
- Aliakbar Ahovan, Z.; Khosravimelal, S.; Eftekhari, B.S.; Mehrabi, S.; Hashemi, A.; Eftekhari, S.; Brouki Milan, P.; Mobaraki, M.; Seifalian, A.M.; Gholipourmalekabadi, M. Thermo-responsive chitosan hydrogel for healing of full-thickness wounds infected with XDR bacteria isolated from burn patients: In vitro and in vivo animal model. Int. J. Biol. Macromol. 2020, 164, 4475–4486. [Google Scholar] [CrossRef]
- Rasool, A.; Ata, S.; Islam, A. Stimuli responsive biopolymer (chitosan) based blend hydrogels for wound healing application. Carbohydr. Polym. 2019, 203, 423–429. [Google Scholar] [CrossRef]
- Mazurek, Ł.; Szudzik, M.; Rybka, M.; Konop, M. Silk Fibroin Biomaterials and Their Beneficial Role in Skin Wound Healing. Biomolecules 2022, 12, 1852. [Google Scholar] [CrossRef]
- Sultan, M.T.; Lee, O.J.; Kim, S.H.; Ju, H.W.; Park, C.H. Silk Fibroin in Wound Healing Process. Adv. Exp. Med. Biol. 2018, 1077, 115–126. [Google Scholar] [CrossRef]
- Melke, J.; Midha, S.; Ghosh, S.; Ito, K.; Hofmann, S. Silk fibroin as biomaterial for bone tissue engineering. Acta Biomater. 2016, 31, 1–16. [Google Scholar] [CrossRef]
- Aramwit, P.; Kanokpanont, S.; De-Eknamkul, W.; Srichana, T. Monitoring of inflammatory mediators induced by silk sericin. J. Biosci. Bioeng. 2009, 107, 556–561. [Google Scholar] [CrossRef] [PubMed]
- Ersel, M.; Uyanikgil, Y.; Karbek Akarca, F.; Ozcete, E.; Altunci, Y.A.; Karabey, F.; Cavusoglu, T.; Meral, A.; Yigitturk, G.; Oyku Cetin, E. Effects of Silk Sericin on Incision Wound Healing in a Dorsal Skin Flap Wound Healing Rat Model. Med. Sci. Monit. 2016, 22, 1064–1078. [Google Scholar] [CrossRef] [PubMed]
- Jiao, Z.; Song, Y.; Jin, Y.; Zhang, C.; Peng, D.; Chen, Z.; Chang, P.; Kundu, S.C.; Wang, G.; Wang, Z.; et al. In Vivo Characterizations of the Immune Properties of Sericin: An Ancient Material with Emerging Value in Biomedical Applications. Macromol. Biosci. 2017, 17, 1700229. [Google Scholar] [CrossRef] [PubMed]
- Ju, H.W.; Lee, O.J.; Lee, J.M.; Moon, B.M.; Park, H.J.; Park, Y.R.; Lee, M.C.; Kim, S.H.; Chao, J.R.; Ki, C.S.; et al. Wound healing effect of electrospun silk fibroin nanomatrix in burn-model. Int. J. Biol. Macromol. 2016, 85, 29–39. [Google Scholar] [CrossRef]
- Guan, Y.; Sun, F.; Zhang, X.; Peng, Z.; Jiang, B.; Liang, M.; Wang, Y. Silk fibroin hydrogel promote burn wound healing through regulating TLN1 expression and affecting cell adhesion and migration. J. Mater. Sci. Mater. Med. 2020, 31, 48. [Google Scholar] [CrossRef]
- Chouhan, D.; Lohe, T.U.; Samudrala, P.K.; Mandal, B.B. In Situ Forming Injectable Silk Fibroin Hydrogel Promotes Skin Regeneration in Full Thickness Burn Wounds. Adv. Healthc. Mater. 2018, 7, e1801092. [Google Scholar] [CrossRef]
- Zheng, Y.; Wu, J.; Zhu, Y.; Wu, C. Inorganic-Based Biomaterials for Rapid Hemostasis and Wound Healing. Chem. Sci. 2022, 14, 29–53. [Google Scholar] [CrossRef]
- Hissae Yassue-Cordeiro, P.; Henrique Zandonai, C.; Pereira Genesi, B.; Santos Lopes, P.; Sanchez-Lopez, E.; Luisa Garcia, M.; Regina Camargo Fernandes-Machado, N.; Severino, P.; Souto, E.B.; Ferreira da Silva, C. Development of Chitosan/Silver Sulfadiazine/Zeolite Composite Films for Wound Dressing. Pharmaceutics 2019, 11, 535. [Google Scholar] [CrossRef]
- Vijayakumar, V.; Samal, S.K.; Mohanty, S.; Nayak, S.K. Recent advancements in biopolymer and metal nanoparticle-based materials in diabetic wound healing management. Int. J. Biol. Macromol. 2019, 122, 137–148. [Google Scholar] [CrossRef]
- Mihai, M.M.; Dima, M.B.; Dima, B.; Holban, A.M. Nanomaterials for Wound Healing and Infection Control. Materials 2019, 12, 2176. [Google Scholar] [CrossRef]
- Huang, R.; Hu, J.; Qian, W.; Chen, L.; Zhang, D. Recent advances in nanotherapeutics for the treatment of burn wounds. Burn. Trauma 2021, 9, tkab026. [Google Scholar] [CrossRef] [PubMed]
- Arunadevi, N.; Jone Kirubavathy, S. 12—Three-dimensional approaches based on nanotechnology towards wound management. In Nanotechnological Aspects for Next-Generation Wound Management; Solanki, P.R., Kumar, A., Singh, R.P., Singh, J., Singh, K.R.B., Eds.; Academic Press: Cambridge, MA, USA, 2024; pp. 245–280. [Google Scholar]
- Zhou, Y.; Chen, R.; He, T.; Xu, K.; Du, D.; Zhao, N.; Cheng, X.; Yang, J.; Shi, H.; Lin, Y. Biomedical potential of ultrafine ag/AgCl nanoparticles coated on graphene with special reference to antimicrobial performances and burn wound healing. ACS Appl. Mater. Interfaces. 2016, 8, 15067–15075. [Google Scholar] [CrossRef] [PubMed]
- Jiji, S.; Udhayakumar, S.; Maharajan, K.; Rose, C.; Muralidharan, C.; Kadirvelu, K. Bacterial cellulose matrix with in situ impregnation of silver nanoparticles via catecholic redox chemistry for third degree burn wound healing. Carbohydr. Polym. 2020, 245, 116573. [Google Scholar] [CrossRef]
- Dai, T.; Tanaka, M.; Huang, Y.Y.; Hamblin, M.R. Chitosan preparations for wounds and burns: Antimicrobial and wound-healing effects. Expert Rev. Anti. Infect. Ther. 2011, 9, 857–879. [Google Scholar] [CrossRef] [PubMed]
- Li, D.D.; Guo, Q.Q.; Ding, L.M.; Zhang, W.; Cheng, L.; Wang, Y.Q.; Xu, Z.B.; Wang, H.H.; Gao, L.Z. Bimetallic CuCo2S4 Nanozymes with Enhanced Peroxidase Activity at Neutral pH for Combating Burn Infections. ChemBioChem 2020, 21, 2620–2627. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Zheng, T.; Sheng, B.; Zhou, T.; Zhang, Q.; Wu, F.; Zhou, N.; Shen, J.; Zhang, M.; Sun, Y. Functionalization of polyvinyl alcohol composite film wrapped in a-ZnO@CuO@Au nanoparticles for antibacterial application and wound healing. Appl. Mater. Today 2019, 17, 36–44. [Google Scholar] [CrossRef]
- Ye, L.; He, X.; Obeng, E.; Wang, D.; Zheng, D.; Shen, T.; Shen, J.; Hu, R.; Deng, H. The CuO and AgO co-modified ZnO nanocomposites for promoting wound healing in Staphylococcus aureus infection. Mater. Today Bio 2023, 18, 100552. [Google Scholar] [CrossRef]
- Raszewska-Famielec, M.; Flieger, J. Nanoparticles for Topical Application in the Treatment of Skin Dysfunctions-An Overview of Dermo-Cosmetic and Dermatological Products. Int. J. Mol. Sci. 2022, 23, 15980. [Google Scholar] [CrossRef]
- Wang, W.; Lu, K.J.; Yu, C.H.; Huang, Q.L.; Du, Y.Z. Nano-drug delivery systems in wound treatment and skin regeneration. J. Nanobiotechnol. 2019, 17, 82. [Google Scholar] [CrossRef]
- Bellu, E.; Medici, S.; Coradduzza, D.; Cruciani, S.; Amler, E.; Maioli, M. Nanomaterials in Skin Regeneration and Rejuvenation. Int. J. Mol. Sci. 2021, 22, 7095. [Google Scholar] [CrossRef]
- Pryjmaková, J.; Kaimlová, M.; Hubáček, T.; Švorčík, V.; Siegel, J. Nanostructured Materials for Artificial Tissue Replacements. Int. J. Mol. Sci. 2020, 21, 2521. [Google Scholar] [CrossRef] [PubMed]
- Pierre, M.B.R. Current Applications and Benefits of Polymeric Nanocarriers for the Management of Skin Disorders. Curr. Med. Chem. 2022, 29, 5949–5964. [Google Scholar] [CrossRef] [PubMed]
- Bai, Q.; Zheng, C.; Chen, W.; Sun, N.; Gao, Q.; Liu, J.; Hu, F.; Pimpi, S.; Yan, X.; Zhang, Y.; et al. Current challenges and future applications of antibacterial nanomaterials and chitosan hydrogel in burn wound healing. Mater. Adv. 2022, 3, 6707. [Google Scholar] [CrossRef]
- Li, X.; Wei, Z.; Li, B.; Li, J.; Lv, H.; Wu, L.; Zhang, H.; Yang, B.; Zhu, M.; Jiang, J. In vivo migration of Fe3O4@polydopamine nanoparticle-labeled mesenchymal stem cells to burn injury sites and their therapeutic effects in a rat model. Biomater. Sci. 2019, 7, 2861–2872. [Google Scholar] [CrossRef]
- Stoica, A.E.; Albuleț, D.; Bîrcă, A.C.; Iordache, F.; Ficai, A.; Grumezescu, A.M.; Vasile, B.Ș.; Andronescu, E.; Marinescu, F.; Holban, A.M. Electrospun Nanofibrous Mesh Based on PVA, Chitosan, and Usnic Acid for Applications in Wound Healing. Int. J. Mol. Sci. 2023, 24, 11037. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Ma, W.; Ma, H.; Qin, C.; Chen, J.; Wu, C. Spindle-Like Zinc Silicate Nanoparticles Accelerating Innervated and Vascularized Skin Burn Wound Healing. Adv. Healthc. Mater. 2022, 11, e2102359. [Google Scholar] [CrossRef]
- Qadir, A.; Jahan, S.; Aqil, M.; Warsi, M.H.; Alhakamy, N.A.; Alfaleh, M.A.; Khan, N.; Ali, A. Phytochemical-Based Nano-Pharmacotherapeutics for Management of Burn Wound Healing. Gels 2021, 7, 209. [Google Scholar] [CrossRef] [PubMed]
- Tang, T.; Yin, L.; Yang, J.; Shan, G. Emodin, an anthraquinone derivative from Rheum officinale Baill, enhances cutaneous wound healing in rats. Eur. J. Pharmacol. 2007, 567, 177–185. [Google Scholar] [CrossRef]
- Dai, X.Y.; Nie, W.; Wang, Y.C.; Shen, Y.; Li, Y.; Gan, S.J. Electrospun emodin polyvinylpyrrolidone blended nanofibrous membrane: A novel medicated biomaterial for drug delivery and accelerated wound healing. J. Mater. Sci. Mater. Med. 2012, 23, 2709–2716. [Google Scholar] [CrossRef]
- Panichpakdee, J.; Pavasant, P.; Supaphol, P. Electrospun cellulose acetate fiber mats containing emodin with potential for use as wound dressing. Chiang Mai J. Sci. 2016, 43, 1249–1259. Available online: https://cmudc.library.cmu.ac.th/frontend/Info/item/dc:47780 (accessed on 30 March 2021).
- Suwantong, O.; Ruktanonchai, U.; Supaphol, P. In vitro biological evaluation of electrospun cellulose acetate fiber mats containing asiaticoside or curcumin. J. Biomed. Mater. Res. Part A 2010, 94, 1216–1225. [Google Scholar] [CrossRef] [PubMed]
- Suwantong, O.; Ruktanonchai, U.; Supaphol, P. Electrospun cellulose acetate fiber mats containing asiaticoside or Centella asiatica crude extract and the release characteristics of asiaticoside. Polymer 2008, 49, 4239–4247. [Google Scholar] [CrossRef]
- Liakos, I.; Rizzello, L.; Hajiali, H.; Brunetti, V.; Carzino, R.; Pompa, P.P.; Athanassiou, A.; Mele, E. Fibrous wound dressings encapsulating essential oils as natural antimicrobial agents. J. Mater. Chem. B 2015, 3, 1583–1589. [Google Scholar] [CrossRef]
- Huang, S.; Fu, X. Naturally derived materials-based cell and drug delivery systems in skin regeneration. J. Control. Release 2010, 142, 149–159. [Google Scholar] [CrossRef] [PubMed]
- Tokuda, M.; Yamane, M.; Thickett, S.C.; Minami, H.; Zetterlund, P.B. Synthesis of polymeric nanoparticles containing reduced graphene oxide nanosheets stabilized by poly(ionic liquid) using miniemulsion polymerization. Soft Matter. 2016, 12, 3955–3962. [Google Scholar] [CrossRef] [PubMed]
- Yun, Y.H.; Goetz, D.J.; Yellen, P.; Chen, W. Hyaluronan microspheres for sustained gene delivery and site-specific targeting. Biomaterials 2004, 25, 147–157. [Google Scholar] [CrossRef]
- Ahn, S.; Ardoña, H.A.M.; Campbell, P.H.; Gonzalez, G.M.; Parker, K.K. Alfalfa Nanofibers for Dermal Wound Healing. ACS Appl. Mater. Interfaces 2019, 11, 33535–33547. [Google Scholar] [CrossRef]
- Karami, Z.; Rezaeian, I.; Zahedi, P.; Abdollahi, M. Preparation and performance evaluations of electrospun poly(ε-caprolactone), poly(lactic acid), and their hybrid (50/50) nanofibrous mats containing thymol as an herbal drug for effective wound healing. J. Appl. Polym. Sci. 2013, 129, 756–766. [Google Scholar] [CrossRef]
- Suganya, S.; Senthil Ram, T.; Lakshmi, B.S.; Giridev, V.R. Herbal drug incorporated antibacterial nanofibrous mat fabricated by electrospinning: An excellent matrix for wound dressings. J. Appl. Polym. Sci. 2011, 121, 2893. [Google Scholar] [CrossRef]
- Kalomiraki, M.; Thermos, K.; Chaniotakis, N.A. Dendrimers as tunable vectors of drug delivery systems and biomedical and ocular applications. Int. J. Nanomed. 2015, 11, 1–12. [Google Scholar] [CrossRef]
- Thanusha, A.V.; Dinda, A.K.; Koul, V. Evaluation of nano hydrogel composite based on gelatin/HA/CS suffused with Asiatic acid/ZnO and CuO nanoparticles for second degree burns. Mater. Sci. Eng. C Mater. Biol. Appl. 2018, 89, 378–386. [Google Scholar] [CrossRef]
- Manconi, M.; Manca, M.L.; Caddeo, C.; Cencetti, C.; di Meo, C.; Zoratto, N.; Nacher, A.; Fadda, A.M.; Matricardi, P. Preparation of gellan-cholesterol nanohydrogels embedding baicalin and evaluation of their wound healing activity. Eur. J. Pharm. Biopharm. 2018, 127, 244–249. [Google Scholar] [CrossRef] [PubMed]
- Manca, M.; Manconi, M.; Meloni, M.; Marongiu, F.; Allaw, M.; Usach, I.; Peris, J.; Escribano-Ferrer, E.; Tuberoso, C.; Gutierrez, G.; et al. Nanotechnology for Natural Medicine: Formulation of Neem Oil Loaded Phospholipid Vesicles Modified with Argan Oil as a Strategy to Protect the Skin from Oxidative Stress and Promote Wound Healing. Antioxidants 2021, 10, 670. [Google Scholar] [CrossRef] [PubMed]
- Allaw, M.; Pleguezuelos-Villa, M.; Manca, M.L.; Caddeo, C.; Aroffu, M.; Nacher, A.; Diez-Sales, O.; Saurí, A.R.; Ferrer, E.E.; Fadda, A.M.; et al. Innovative strategies to treat skin wounds with mangiferin: Fabrication of transfersomes modified with glycols and mucin. Nanomedicine 2020, 15, 1671–1685. [Google Scholar] [CrossRef] [PubMed]
- Saito, A.; Miyazaki, H.; Fujie, T.; Ohtsubo, S.; Kinoshita, M.; Saitoh, D.; Takeoka, S. Therapeutic efficacy of an antibiotic-loaded nanosheet in a murine burn-wound infection model. Acta Biomater. 2012, 8, 2932–2940. [Google Scholar] [CrossRef]
- Chen, H.; Li, B.Y.; Feng, B.; Wang, H.; Yuan, H.H.; Xu, Z.W. Tetracycline hydrochloride loaded citric acid functionalized chitosan hydrogel for wound healing. RSC Adv. 2019, 9, 19523–19530. [Google Scholar] [CrossRef]
- Mohebali, A.; Abdouss, M.; Afshar Taromi, F. Fabrication of biocompatible antibacterial nanowafers based on HNT/PVA nanocomposites loaded with minocycline for burn wound dressing. Mater. Sci. Eng. C Mater. Biol. Appl. 2020, 110, 110685. [Google Scholar] [CrossRef]
- Mohebali, A.; Abdouss, M. Layered biocompatible pH-responsive antibacterial composite film based on HNT/PLGA/chitosan for controlled release of minocycline as burn wound dressing. Int. J. Biol. Macromol. 2020, 164, 4193–4204. [Google Scholar] [CrossRef]
- Kaur, P.; Gondil, V.S.; Chhibber, S. A novel wound dressing consisting of PVA-SA hybrid hydrogel membrane for topical delivery of bacteriophages and antibiotics. Int. J. Pharm. 2019, 572, 118779. [Google Scholar] [CrossRef]
- Asgarirad, H.; Ebrahimnejad, P.; Mahjoub, M.A.; Jalalian, M.; Morad, H.; Ataee, R.; Hosseini, S.S.; Farmoudeh, A. A promising technology for wound healing; in-vitro and in-vivo evaluation of chitosan nano-biocomposite films containing gentamicin. J. Microencapsul. 2021, 38, 100–107. [Google Scholar] [CrossRef]
- Lan, Y.; Li, W.; Jiao, Y.; Guo, R.; Zhang, Y.; Xue, W.; Zhang, Y. Therapeutic efficacy of antibiotic-loaded gelatin microsphere/silk fibroin scaffolds in infected full-thickness burns. Acta Biomater. 2014, 10, 3167–3176. [Google Scholar] [CrossRef] [PubMed]
- Zilberman, M.; Egozi, D.; Shemesh, M.; Keren, A.; Mazor, E.; Baranes-Zeevi, M.; Goldstein, N.; Berdicevsky, I.; Gilhar, A.; Ullmann, Y. Hybrid wound dressings with controlled release of antibiotics: Structure-release profile effects and in vivo study in a guinea pig burn model. Acta Biomater. 2015, 22, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Thakur, K.; Sharma, G.; Singh, B.; Chhibber, S.; Katare, O.P. Nano-engineered lipid-polymer hybrid nanoparticles of fusidic acid: An investigative study on dermatokinetics profile and MRSA-infected burn wound model. Drug Deliv. Transl. Res. 2019, 9, 748–763. [Google Scholar] [CrossRef] [PubMed]
- Tredget, E.E.; Shankowsky, H.A.; Rennie, R.; Burrell, R.E.; Logsetty, S. Pseudomonas infections in the thermally injured patient. Burns 2004, 30, 3–26. [Google Scholar] [CrossRef]
- Björn, C.; Noppa, L.; Näslund Salomonsson, E.; Johansson, A.L.; Nilsson, E.; Mahlapuu, M.; Håkansson, J. Efficacy and safety profile of the novel antimicrobial peptide PXL150 in a mouse model of infected burn wounds. Int. J. Antimicrob. Agents 2015, 45, 519–524. [Google Scholar] [CrossRef]
- Zhong, G.; Cheng, J.; Liang, Z.C.; Xu, L.; Lou, W.; Bao, C.; Ong, Z.Y.; Dong, H.; Yang, Y.Y.; Fan, W. Short Synthetic β-Sheet Antimicrobial Peptides for the Treatment of Multidrug-Resistant Pseudomonas aeruginosa Burn Wound Infections. Adv. Healthc. Mater. 2017, 6, 1601134. [Google Scholar] [CrossRef]
- Ma, Z.; Han, J.; Chang, B.; Gao, L.; Lu, Z.; Lu, F.; Zhao, H.; Zhang, C.; Bie, X. Membrane-Active Amphipathic Peptide WRL3 with in Vitro Antibiofilm Capability and in Vivo Efficacy in Treating Methicillin-Resistant Staphylococcus aureus Burn Wound Infections. ACS Infect. Dis. 2017, 3, 820–832. [Google Scholar] [CrossRef]
- Obuobi, S.; Tay, H.K.; Tram, N.D.T.; Selvarajan, V.; Khara, J.S.; Wang, Y.; Ee, P.L.R. Facile and efficient encapsulation of antimicrobial peptides via crosslinked DNA nanostructures and their application in wound therapy. J. Control Release 2019, 313, 120–130. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, T.; He, J.; Dong, J. Growth factor therapy in patients with partial-thickness burns: A systematic review and meta-analysis. Int. Wound J. 2016, 13, 354–366. [Google Scholar] [CrossRef]
- Jahromi, M.A.M.; Zangabad, P.S.; Basri, S.M.M.; Zangabad, K.S.; Ghamarypour, A.; Aref, A.R.; Karimi, M.; Hamblin, M.R. Nanomedicine and advanced technologies for burns: Preventing infection and facilitating wound healing. Adv. Drug Deliv. Rev. 2018, 123, 33–64. [Google Scholar] [CrossRef]
- Chakrabarti, S.; Islam, J.; Hazarika, H.; Mazumder, B.; Raju, P.S.; Chattopadhyay, P. Safety profile of silver sulfadiazine-bFGF-loaded hydrogel for partial thickness burn wounds. Cutan. Ocul. Toxicol. 2018, 37, 258–266. [Google Scholar] [CrossRef] [PubMed]
- Hamdan, S.; Pastar, I.; Drakulich, S.; Dikici, E.; Tomic-Canic, M.; Deo, S.; Daunert, S. Nanotechnology-Driven Therapeutic Interventions in Wound Healing: Potential Uses and Applications. ACS Cent. Sci. 2017, 3, 163–175. [Google Scholar] [CrossRef] [PubMed]
- Abazari, M.; Ghaffari, A.; Rashidzadeh, H.; Momeni Badeleh, S.; Maleki, Y. Current status and future outlook of nano-based systems for burn wound management. J. Biomed. Mater Res. B 2020, 108, 1934–1952. [Google Scholar] [CrossRef]
- Burke, J.F.; Bondoc, C.C.; Quinby, W.C. Primary burn excision and immediate grafting: A method shortening illness. J. Trauma 1974, 14, 389–395. [Google Scholar] [CrossRef] [PubMed]
- Herndon, D.N.; Barrow, R.E.; Rutan, R.L.; Rutan, T.C.; Desai, M.H.; Abston, S. A comparison of conservative versus early excision. therapies in severely burned patients. Ann. Surg. 1989, 209, 547–552. [Google Scholar] [CrossRef] [PubMed]
- Latenser, B.A. Critical care of the burn patient: The first 48 h. Crit. Care Med. 2009, 37, 2819–2826. [Google Scholar] [CrossRef]
- Thompson, P.; Herndon, D.N.; Abston, S.; Rutan, T. Effect of early excision on patients with major thermal injury. J. Trauma 1987, 27, 205–207. [Google Scholar] [CrossRef]
- Wu, X.-W.; Herndon, D.N.; Spies, M.; Sanford, A.P.; Wolf, S.E. Effects of delayed wound excision and grafting in severely burned children. Arch. Surg. 2002, 137, 1049–1054. [Google Scholar] [CrossRef]
- Hayashi, K.; Sasabuchi, Y.; Matsui, H.; Nakajima, M.; Otawara, M.; Ohbe, H.; Fushimi, K.; Ono, K.; Yasunaga, H. Does early excision or skin grafting of severe burns improve prognosis? A retrospective cohort study. Burns 2023, 49, 554–561. [Google Scholar] [CrossRef]
- Hakimi, N.; Cheng, R.; Leng, L.; Sotoudehfar, M.; Ba, P.Q.; Bakhtyar, N.; Amini-Nik, S.; Jeschke, M.G.; Günther, A. Handheld skin printer: In situ formation of planar biomaterials and tissues. Lab. Chip. 2018, 18, 1440–1451. [Google Scholar] [CrossRef]
- Davison-Kotler, E.; Sharma, V.; Kang, N.V.; García-Gareta, E. A Universal Classification System of Skin Substitutes Inspired by Factorial Design. Tissue Eng. Part B Rev. 2018, 24, 279–288. [Google Scholar] [CrossRef] [PubMed]
- Tavakoli, S.; Klar, A.S. Bioengineered Skin Substitutes: Advances and Future Trends. Appl. Sci. 2021, 11, 1493. [Google Scholar] [CrossRef]
- Shahrokhi, S.; Arno, A.; Jeschke, M.G. The use of dermal substitutes in burn surgery: Acute phase. Wound Repair Regen. 2014, 22, 14–22. [Google Scholar] [CrossRef]
- Snyder, D.L.; Sullivan, N.; Margolis, D.J.; Schoelles, K. Skin Substitutes for Treating Chronic Wounds; Technology Assessment Program Project ID No. WNDT081; Prepared by the ECRI Institute-Penn Medicine Evidence-Based Practice Center under Contract No. HHSA 290-2015-00005-I; Agency for Healthcare Research and Quality: Rockville, MD, USA, 2020. Available online: http://www.ahrq.gov/research/findings/ta/index.html (accessed on 13 November 2019).
- Bay, C.; Chizmar, Z.; Reece, E.M.; Yu, J.Z.; Winocour, J.; Vorstenbosch, J.; Winocour, S. Comparison of Skin Substitutes for Acute and Chronic Wound Management. Semin. Plast. Surg. 2021, 35, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Vig, K.; Chaudhari, A.; Tripathi, S.; Dixit, S.; Sahu, R.; Pillai, S.; Dennis, V.A.; Singh, S.R. Advances in Skin Regeneration Using Tissue Engineering. Int. J. Mol. Sci. 2017, 18, 789. [Google Scholar] [CrossRef]
- Anyanwu, J.A.; Cindass, R. Burn Debridement, Grafting, and Reconstruction. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2023. Available online: https://www.ncbi.nlm.nih.gov/books/NBK551717/ (accessed on 29 May 2023).
- Granick, M.S.; Posnett, J.; Jacoby, M.; Noruthun, S.; Ganchi, P.A.; Datiashvili, R.O. Efficacy and cost-effectiveness of a high-powered parallel waterjet for wound debridement. Wound Repair Regen. 2006, 14, 394–397. [Google Scholar] [CrossRef]
- Atiyeh, B.S.; Costagliola, M. Cultured epithelial autograft (CEA) in burn treatment: Three decades later. Burns 2007, 33, 405–413. [Google Scholar] [CrossRef]
- Bogdanov, S.B.; Gilevich, I.V.; Melkonyan, K.I.; Sotnichenko, A.; Alekseenko, S.N.; Porhanov, V.A. Total full-thickness skin grafting for treating patients with extensive facial burn injury: A 10-year experience. Burns 2021, 47, 1389–1398. [Google Scholar] [CrossRef]
- Téot, L. Facial Scars Reconstruction. In Textbook on Scar Management: State of the Art Management and Emerging Technologies; Téot, L., Mustoe, T.A., Middelkoop, E., Gauglitz, G.G., Eds.; Springer: Cham, Switzerland, 2020; Chapter 38. [Google Scholar]
- Carsin, H.; Ainaud, P.; Le Bever, H.; Rives, J.; Lakhel, A.; Stephanazzi, J.; Lambert, F.; Perrot, J. Cultured epithelial autografts in extensive burn coverage of severely traumatized patients: A five year single-center experience with 30 patients. Burns 2000, 26, 379–387. [Google Scholar] [CrossRef]
- Halim, A.S.; Khoo, T.L.; Mohd Yussof, S.J. Biologic and synthetic skin substitutes: An overview. Indian J. Plast. Surg. 2010, 43, S23–S28. [Google Scholar] [CrossRef]
- Staud, C.J.; Resch, A.; Christ, A.; Borger, A.; Zaussinger, M.; Teufelsbauer, M.; Worel, N.; Radtke, C. Skin Bank Establishment in Treatment of Severe Burn Injuries: Overview and Experience with Skin Allografts at the Vienna Burn Center. J. Clin. Med. 2023, 12, 4717. [Google Scholar] [CrossRef]
- Keswani, S.M.; Mishra, M.G.; Karnik, S.; Dutta, S.; Mishra, M.; Panda, S.; Varghese, R.; Virkar, T.; Upendran, V. Skin banking at a regional burns centre-The way forward. Burns 2018, 44, 870–876. [Google Scholar] [CrossRef]
- Rezaei, E.; Beiraghi-Toosi, A.; Ahmadabadi, A.; Tavousi, S.H.; Tabrizi, A.A.; Fotuhi, K.; Nooghabi, M.J.; Manafi, A.; Moghadam, S.A. Can Skin Allograft Occasionally Act as a Permanent Coverage in Deep Burns? A Pilot Study. World J. Plast. Surg. 2017, 6, 94–99. [Google Scholar]
- Rogers, A.D.; Allorto, N.L.; Adams, S.; Adams, K.G.; Hudson, D.A.; Rode, H. Isn’t it time for a cadaver skin bank in South Africa? Ann. Burn. Fire Disasters 2013, 26, 142–146. [Google Scholar]
- Glik, J.; Kawecki, M.; Kitala, D.; Klama-Baryła, A.; Łabuś, W.; Grabowski, M.; Durdzińska, A.; Nowak, M.; Misiuga, M.; Kasperczyk, A. A new option for definitive burn wound closure—pair matching type of retrospective case–control study of hand burns in the hospitalised patients group in the Dr Stanislaw Sakiel Centre for Burn Treatment between 2009 and 2015. Int. Wound J. 2017, 14, 849–855. [Google Scholar] [CrossRef]
- Nessler, M.; Chrapusta, A. The use of xenogenic skin substitutes in burns treatment—literature review. Leczenie Ran 2013, 10, 47–52. [Google Scholar] [CrossRef]
- Kotkot, A.; Ghabisha, S.; Ahmed, F.; Al-Wageeh, S.; Al-Shami, E.; Al-Hajri, A.; Aljbri, W.; Mohammed, F. Fish skin as a biological dressing for burn injuries. J. Emerg. Med. Trauma Acute Care 2022, 2022, 18. [Google Scholar] [CrossRef]
- Choi, Y.H.; Cho, Y.S.; Lee, J.H.; Choi, Y.; Noh, S.Y.; Park, S.; Sung, C.; Lim, J.K.; Kim, J.; Shin, J.J.; et al. Cadaver skin allograft may improve mortality rate for burns involving over 30% of total body surface area: A propensity score analysis of data from four burn centers. Cell Tissue Bank 2018, 19, 645–651. [Google Scholar] [CrossRef]
- Schmiedova, I.; Dembickaja, A.; Kiselakova, L.; Nowakova, B.; Slama, P. Using of Amniotic Membrane Derivatives for the Treatment of Chronic Wounds. Membranes 2021, 11, 941. [Google Scholar] [CrossRef]
- Stone, R.; Saathoff, E.C.; Larson, D.A.; Wall, J.T.; Wienandt, N.A.; Magnusson, S.; Kjartansson, H.; Natesan, S.; Christy, R.J. Accelerated Wound Closure of Deep Partial Thickness Burns with Acellular Fish Skin Graft. Int. J. Mol. Sci. 2021, 22, 1590. [Google Scholar] [CrossRef]
- Yoon, J.; Yoon, D.; Lee, H.; Lee, J.; Jo, S.; Kym, D.; Yim, H.; Hur, J.; Chun, W.; Kim, G.; et al. Wound healing ability of acellular fish skin and bovine collagen grafts for split-thickness donor sites in burn patients: Characterization of acellular grafts and clinical application. Int. J. Biol. Macromol. 2022, 205, 452–461. [Google Scholar] [CrossRef]
- Alam, K.; Jeffery, S.L.A. Acellular Fish Skin Grafts for Management of Split Thickness Donor Sites and Partial Thickness Burns: A Case Series. Mil. Med. 2019, 184, 16–20. [Google Scholar] [CrossRef] [PubMed]
- Luze, H.; Nischwitz, S.P.; Smolle, C.; Zrim, R.; Kamolz, L.P. The Use of Acellular Fish Skin Grafts in Burn Wound Management—A Systematic Review. Medicina 2022, 58, 912. [Google Scholar] [CrossRef]
- Yang, C.K.; Polanco, T.O.; Ii, J.C.L. A Prospective, Postmarket, Compassionate Clinical Evaluation of a Novel Acellular Fish-skin Graft Which Contains Omega-3 Fatty Acids for the Closure of Hard-to-heal Lower Extremity Chronic Ulcers. Wounds 2016, 28, 112–118. [Google Scholar] [PubMed]
- Verde, M.E.Q.L.; Ferreira-Júnior, A.E.C.; de Barros-Silva, P.G.; Miguel, E.d.C.; Mathor, M.B.; Lima-Júnior, E.M.; de Moraes-Filho, M.O.; Alves, A.P.N.N. Nile tilapia skin (Oreochromis niloticus) for burn treatment: Ultrastructural analysis and quantitative assessment of collagen. Acta Histochem. 2021, 123, 151762. [Google Scholar] [CrossRef] [PubMed]
- Wallner, C.; Holtermann, J.; Drysch, M.; Schmidt, S.; Reinkemeier, F.; Wagner, J.M.; Dadras, M.; Sogorski, A.; Houschyar, K.S.; Becerikli, M.; et al. The Use of Intact Fish Skin as a Novel Treatment Method for Deep Dermal Burns Following Enzymatic Debridement: A Retrospective Case-Control Study. Eur. Burn J. 2022, 3, 43–55. [Google Scholar] [CrossRef]
- Chambers, L.; Woodrow, S.; Brown, A.; Harris, P.D.; Phillips, D.; Hall, M.; Church, J.C.; Pritchard, D.I. Degradation of extracellular matrix components by defined proteinases from the green bottle larva Lucilia sericata used for the clinical debridement of non-healing wounds. Br. J. Dermatol. 2003, 148, 14–23. [Google Scholar] [CrossRef]
- Bazaliński, D.; Karnas, M.; Wołkowicz, M.; Kózka, M.; Więch, P. Zastosowanie larw Lucilia sericata w oczyszczaniu ran przewle-kłych—opis trzech przypadków. Leczenie Ran 2018, 15, 153–159. [Google Scholar] [CrossRef]
- Rosadi Seswandhana, M.; Anzhari, S.; Dachlan, I.; Widodo Wirohadidjojo, Y.; Aryandono, T. A case series of negative pressure wound therapy as a promising treatment in patients with burn injury. Int. J. Surg. Case Rep. 2020, 69, 64–67. [Google Scholar] [CrossRef]
- Ibrahim, Z.M.; Waked, I.S.; Ibrahim, O. Negative pressure wound therapy versus microcurrent electrical stimulation in wound healing in burns. J. Wound Care 2019, 28, 214–219. [Google Scholar] [CrossRef]
- Frear, C.C.; Griffin, B.R.; Cuttle, L.; Kimble, R.M.; McPhail, S.M. Cost-effectiveness of adjunctive negative pressure wound therapy in paediatric burn care: Evidence from the SONATA in C randomised controlled trial. Sci. Rep. 2021, 11, 16650. [Google Scholar] [CrossRef] [PubMed]
- Frear, C.C.; Zang, T.; Griffin, B.R.; McPhail, S.M.; Parker, T.J.; Kimble, R.M.; Cuttle, L. The modulation of the burn wound environment by negative pressure wound therapy: Insights from the proteome. Wound Repair Regen. 2021, 29, 288–297. [Google Scholar] [CrossRef]
- Frear, C.C.; Cuttle, L.; McPhail, S.M.; Chatfield, M.D.; Kimble, R.M.; Griffin, B.R. Randomized clinical trial of negative pressure wound therapy as an adjunctive treatment for small-area thermal burns in children. Br. J. Surg. 2020, 107, 1741–1750. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.P.; Chen, J.; Chen, T.S.; Jiang, Y.N.; Shen, T.; Xiao, S.C.; Hu, X.Y. Preliminary effect observation on the application of micro-negative pressure in children with small-area deep partial-thickness burn. Zhonghua Shao Shang Za Zhi (Chinese) 2019, 35, 720–725. [Google Scholar] [CrossRef]
- Huang, Z.; Wang, P.; Pan, Z.Y.; Dong, L.; Su, J.; Xu, N.W. Randomized controlled trial on application of negative pressure materials of polyvinyl alcohol and polyurethane in full-thickness burn wounds after escharotomy. Zhonghua Shao Shang Za Zhi (Chinese) 2020, 36, 813–820. [Google Scholar] [CrossRef]
- Sun, T.; Ying, W.; Wang, S.; Chen, C.; Sun, P.; Tan, J. Clinical Application of Vacuum Sealing Drainage for the Treatment of Deep Burn Wounds. Am. Surg. 2023, 89, 1018–1023. [Google Scholar] [CrossRef]
- Huang, C.G.; Jia, Z.G.; Gu, Z.Q.; Zhao, P.; Lyu, G.Z. Clinical effects of vacuum sealing drainage in the treatment of alkali burn wounds. Zhonghua Shao Shang Za Zhi (Chinese) 2020, 36, 534–539. [Google Scholar] [CrossRef]
- Bazaliński, D.; Kózka, M.; Karnas, M.; Więch, P. Effectiveness of Chronic Wound Debridement with the Use of Larvae of Lucilia sericata. J. Clin. Med. 2019, 8, 1845. [Google Scholar] [CrossRef]
- Gazi, U.; Taylan-Ozkan, A.; Mumcuoglu, K.Y. The effect of Lucilia sericata larval excretion/secretion (ES) products on cellular responses in wound healing. Med. Vet. Entomol. 2021, 35, 257–266. [Google Scholar] [CrossRef]
- Stadler, F. The maggot therapy supply chain: A review of the literature and practice. Med. Vet. Entomol. 2020, 34, 1–9. [Google Scholar] [CrossRef]
- Morris, D.; Flores, M.; Harris, L.; Gammon, J.; Nigam, Y. Larval Therapy and Larval Excretions/Secretions: A Potential Treatment for Biofilm in Chronic Wounds? A Systematic Review. Microorganisms 2023, 11, 457. [Google Scholar] [CrossRef] [PubMed]
- Shamloul, G.; Khachemoune, A. Reappraisal and updated review of maggot debridement therapy in chronic lower extremity ulcers. Int. J. Dermatol. 2023, 62, 962–968. [Google Scholar] [CrossRef] [PubMed]
- King, C. Changing attitudes toward maggot debridement therapy in wound treatment: A review and discussion. J. Wound Care 2020, 29, S28–S34. [Google Scholar] [CrossRef] [PubMed]
- Nigam, Y.; Morgan, C. Does maggot therapy promote wound healing? The clinical and cellular evidence. J. Eur. Acad. Dermatol. Venereol. 2016, 30, 776–782. [Google Scholar] [CrossRef] [PubMed]
- Watts, R. Evidence summary: Wound management: Larval therapy. Wound Practice Res. 2016, 24, 180–182. Available online: https://search.informit.com.au/documentSummary;res=IELHEA;dn=362540165996394 (accessed on 1 September 2016).
- Sherman, R.A. Mechanisms of maggot-induced wound healing: What do we know, and where do we go from here? Evid. Based. Complement. Alternat. Med. 2014, 2014, 592419. [Google Scholar] [CrossRef]
- Borkataki, S.; Katoch, R.; Goswami, P.; Bhat, A.; Bhardwaj, H.R.; Chakraborty, D.; Chandrawathani, P. Therapeutic use of Lucilia sericata maggot in controlling bacterial bio-burden in Rat wound model. Trop. Biomed. 2018, 35, 627–638. [Google Scholar]
- Van der Plas, M.J.; Jukema, G.N.; Wai, S.W.; Dogterom-Ballering, H.C.; Lagendijk, E.L.; van Gulpen, C.; van Dissel, J.T.; Bloemberg, G.V.; Nibbering, P.H. Maggot excretions/secretions are differentially effective against biofilms of Staphylococcus aureus and Pseudomonas aeruginosa. J. Antimicrob. Chemother. 2008, 61, 117–122. [Google Scholar] [CrossRef]
- Sun, X.; Jiang, K.; Chen, J.; Wu, L.; Lu, H.; Wanga, A.; Wang, J. A systematic review of maggot debridement therapy for chronically infected wounds and ulcers. Int. J. Infect. Dis. 2014, 25, 32–37. [Google Scholar] [CrossRef]
- Linger, R.J.; Belikoff, E.J.; Yan, Y.; Li, F.; Wantuch, H.A.; Fitzsimons, H.L.; Scott, M.J. Towards next generation maggot debridement therapy: Transgenic Lucilia sericata larvae that produce and secrete a human growth factor. BMC Biotechnol. 2016, 16, 30. [Google Scholar] [CrossRef]
- Bian, H.; Yang, Q.; Ma, T.; Li, W.; Duan, J.; Wei, G.; Wu, X.; Mu, F.; Lin, R.; Wen, A.; et al. Beneficial effects of extracts from Lucilia sericata maggots on burn wounds in rats. Mol. Med. Rep. 2017, 16, 7213–7220. [Google Scholar] [CrossRef]
- Hu, Z.H.; Yu, Z.F.; Huang, J.; Chen, X.F.; Huang, D.B. Effects of Coriaria Sinica Maxim’s extract on microcirculation and oxidative stress of wounds in rats with deep second-degree burn. Zhongguo Ying Yong Sheng Li Xue Za Zhi 2018, 34, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Nasoori, A.; Hoomand, R. Maggot debridement therapy for an electrical burn injury with instructions for the use of Lucilia sericata larvae. Wound Care 2017, 26, 734–741. [Google Scholar] [CrossRef]
- Gaffari, J.; Akbarzadeh, K.; Baniardalani, M.; Hosseini, R.; Masoumi, S.; Amiri, Z.S.; Shabani Kordshouli, R.; Rafinejad, J.; Dahmardehei, M. Larval therapy vs conventional silver dressings for full-thickness burns: A randomized controlled trial. BMC Med. 2023, 21, 361. [Google Scholar] [CrossRef] [PubMed]
- Irish, B. Efficacy of Maggot Debridement Therapy on Burn Wounds as an Alternative Treatment Modality. Master’s Thesis of Science in Physician Assistant Studies, School of Physician Assistant Studies, Gainesville, FL, USA, 2016. Paper 580. Available online: https://core.ac.uk/download/pdf/48859769.pdf (accessed on 15 September 2016).
- Kozłowska, E.; Popow, A.; Kwiatkowska, O. The use of vacuum therapy in medicine. Leczenie Ran 2019, 16, 79–83. [Google Scholar] [CrossRef]
- Woda, Ł.; Banaszkiewicz, Z.; Jawień, A. Terapia podciśnieniowa w leczeniu trudno gojących się ran. Leczenie Ran 2012, 9, 141–145. [Google Scholar]
- Pawlica, P.; Całka, M. Application of negative pressure in wound therapy. Nurs. Public Health 2020, 10, 127–132. [Google Scholar] [CrossRef]
- Fischer, S.; Wall, J.; Pomahac, B.; Riviello, R.; Halvorson, E.G. Extra-large negative pressure wound therapy dressings for burns—Initial experience with technique, fluid management, and outcomes. Burns 2016, 42, 457–465. [Google Scholar] [CrossRef]
- Scherer, S.S.; Pietramaggiori, G.; Mathews, J.C.; Prsa, M.J.; Huang, S.; Orgill, D.P. The Mechanism of Action of the Vacuum-Assisted Closure Device. Plast. Reconstr. Surg. 2008, 122, 786–797. [Google Scholar] [CrossRef]
- Teng, S.C. Use of negative pressure wound therapy in burn patients. Int. Wound J. 2016, 13, 15–18. [Google Scholar] [CrossRef]
- Luo, H.; Chi, Y.; Chen, X.; Chai, J. Usage of Negative Pressure Wound Therapy in Pseudoepitheliomatous Hyperplasia Secondary to Burn Injury: A Case Series. J. Burn Care Res. 2022, 43, 492–495. [Google Scholar] [CrossRef]
- Weitgasser, L.; Ihra, G.; Schäfer, B.; Markstaller, K.; Radtke, C. Update on hyperbaric oxygen therapy in burn treatment. Wien Klin Wochenschr. 2021, 133, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Oley, M.H.; Oley, M.C.; Wewengkang, L.A.W.; Kepel, B.J.; Langi, F.L.F.G.; Setiadi, T.; Aling, D.M.R.; Gunawan, D.F.; Tulong, M.T.; Faruk, M. Bactericidal effect of hyperbaric oxygen therapy in burn injuries. Ann. Med. Surg. 2022, 74, 103314. [Google Scholar] [CrossRef] [PubMed]
- Chiang, I.H.; Chen, S.G.; Huang, K.L.; Chou, Y.C.; Dai, N.T.; Peng, C.K. Adjunctive hyperbaric oxygen therapy in severe burns: Experience in Taiwan Formosa Water Park dust explosion disaster. Burns 2017, 43, 852–857. [Google Scholar] [CrossRef]
- Alyafi, T.; Al-Marzouki, A.H.; Al Hassani, A.N. Therapeutic Outcome of Burn Patients Treated with Hyperbaric Oxygen. Cureus 2021, 13, e18671. [Google Scholar] [CrossRef] [PubMed]
- Lindenmann, J.; Smolle, C.; Kamolz, L.-P.; Smolle-Juettner, F.M.; Graier, W.F. Survey of Molecular Mechanisms of Hyperbaric Oxygen in Tissue Repair. Int. J. Mol. Sci. 2021, 22, 11754. [Google Scholar] [CrossRef]
- Sun, L.; Zhao, L.; Li, P.; Liu, X.; Liang, F.; Jiang, Y.; Kang, N.; Gao, C.; Yang, J. Effect of hyperbaric oxygen therapy on HMGB1/NF-kappaB expression and prognosis of acute spinal cord injury: A randomized clinical trial. Neurosci. Lett. 2019, 692, 47–52. [Google Scholar] [CrossRef]
- Dhamodharan, U.; Karan, A.; Sireesh, D.; Vaishnavi, A.; Somasundar, A.; Rajesh, K.; Ramkumar, K.M. Tissue-specific role of Nrf2 in the treatment of diabetic foot ulcers during hyperbaric oxygen therapy. Free Radic. Biol. Med. 2019, 138, 53–62. [Google Scholar] [CrossRef]
- Baiula, M.; Greco, R.; Ferrazzano, L.; Caligiana, A.; Hoxha, K.; Bandini, D.; Longobardi, P.; Spampinato, S.; Tolomelli, A. Integrin-mediated adhesive properties of neutrophils are reduced by hyperbaric oxygen therapy in patients with chronic non-healing wound. PLoS ONE 2020, 15, e0237746. [Google Scholar] [CrossRef]
- Nasole, E.; Nicoletti, C.; Yang, Z.J.; Girelli, A.; Rubini, A.; Giuffreda, F.; Di Tano, A.; Camporesi, E.; Bosco, G. Effects of alpha lipoic acid and its R+ enantiomer supplemented to hyperbaric oxygen therapy on interleukin-6, TNF-α and EGF production in chronic leg wound healing. J. Enzyme Inhib. Med. Chem. 2014, 29, 297–302. [Google Scholar] [CrossRef]
- Huang, G.; Diao, J.; Yi, H.; Xu, L.; Xu, J.; Xu, W. Signaling pathways involved in HSP32 induction by hyperbaric oxygen in rat spinal neurons. Redox Biol. 2016, 10, 108–118. [Google Scholar] [CrossRef]
- Shyu, K.G.; Wang, B.W.; Pan, C.M.; Fang, W.J.; Lin, C.M. Hyperbaric oxygen boosts long noncoding RNA MALAT1 exosome secretion to suppress microRNA-92a expression in therapeutic angiogenesis. Int. J. Cardiol. 2019, 274, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zhou, Y.; Wang, Z.; Yang, J.; Gao, C.; Su, Q. Effect of VEGF and CX43 on the promotion of neurological recovery by hyperbaric oxygen treatment in spinal cord-injured rats. Spine J. 2013, 14, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Sunkari, V.G.; Lind, F.; Botusan, I.R.; Kashif, A.; Liu, Z.-J.; Ylä-Herttuala, S.; Brismar, K.; Velazquez, O.; Catrina, S.-B. Hyperbaric oxygen therapy activates hypoxia-inducible factor 1 (HIF-1), which contributes to improved wound healing in diabetic mice. Wound Repair Regen. 2015, 23, 98–103. [Google Scholar] [CrossRef]
- Smolle, C.; Lindenmann, J.; Kamolz, L.; Smolle-Juettner, F.-M. The History and Development of Hyperbaric Oxygenation (HBO) in Thermal Burn Injury. Medicina 2021, 57, 49. [Google Scholar] [CrossRef]
- Mathieu, D.; Marroni, A.; Kot, J. Tenth European Consensus Conference on Hyperbaric Medicine: Recommendations for acepted and non-accepted clinical indications and practic of hyperbaric oxygten treatment. Diving Hyperb. Med. J. 2017, 47, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Marques, L.F.; Stessuk, T.; Camargo, I.C.C.; Junior, N.S.; Dos Santos, L.; Ribeiro-Paes, J.T. Platelet-rich plasma (PRP): Methodo-logical aspects and clinical applications. Platelets 2015, 26, 101–113. [Google Scholar] [CrossRef]
- Chen, Z.; Wu, Y.; Turxun, N.; Shen, Y.; Zhang, X. Efficacy and safety of platelet-rich plasma in the treatment of severe burns A protocol for systematic review and meta analysis. Medicine 2020, 99, e23001. [Google Scholar] [CrossRef]
- Mansilla, E.; Marín, G.H.; Berges, M.; Scafatti, S.; Rivas, J.; Núñez, A.; Menvielle, M.; Lamonega, R.; Gardiner, C.; Drago, H.; et al. Cadaveric bone marrow mesenchymal stem cells: First experience treating a patient with large severe burns. Burn. Trauma 2015, 3, 17. [Google Scholar] [CrossRef]
- Martin-Piedra, M.A.; Alfonso-Rodriguez, C.A.; Zapater, A.; Durand-Herrera, D.; Chato-Astrain, J.; Campos, F.; Sanchez-Quevedo, M.C.; Alaminos, M.; Garzon, I. Effective use of mesenchymal stem cells in human skin substitutes generated by tissue engineering. Eur. Cell Mater. 2019, 37, 233–249. [Google Scholar] [CrossRef]
- Surowiecka, A.; Stendel, O.; Strużyna, J. Mezenchymalne komórki macierzyste w leczeniu głębokich oparzeń—wizja przyszłości, czy kolejny mit? Plast. Surg. Burn. 2021, 9, 87–91. [Google Scholar] [CrossRef]
- Jo, H.; Brito, S.; Kwak, B.M.; Park, S.; Lee, M.G.; Bin, B.H. Applications of Mesenchymal Stem Cells in Skin Regeneration and Rejuvenation. Int. J. Mol. Sci. 2021, 22, 2410. [Google Scholar] [CrossRef] [PubMed]
- Surowiecka, A.; Chrapusta, A.; Klimeczek-Chrapusta, M.; Korzeniowski, T.; Drukała, J.; Strużyna, J. Mesenchymal Stem Cells in Burn Wound Management. Int. J. Mol. Sci. 2022, 23, 15339. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Xu, X.; Lei, X.; Tan, J.; Xie, H. Mesenchymal stem cell-based therapy for burn wound healing. Burn. Trauma 2021, 9, tkab002. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Nan, X.; Wang, Y.; Guan, L.; Bai, C.; Shi, S.; Yuan, H.; Chen, L.; Liu, D.; Pei, X. Full-thickness tissue engineered skin constructed with autogenic bone marrow mesenchymal stem cells. Sci. China C Life Sci. 2007, 50, 429–437. [Google Scholar] [CrossRef]
- Turner, P.R.; McConnell, M.; Young, S.L.; Cabral, J.D. 3D Living Dressing Improves Healing and Modulates Immune Response in a Thermal Injury Model. Tissue Eng. Part C Methods 2022, 28, 431–439. [Google Scholar] [CrossRef] [PubMed]
- Rasulov, M.F.; Vasilchenkov, A.V.; Onishchenko, N.A.; Krasheninnikov, M.E.; Kravchenko, V.I.; Gorshenin, T.L.; Pidtsan, R.E.; Potapov, I.V. First experience of the use bone marrow mesenchymal stem cells for the treatment of a patient with deep skin burns. Bull. Exp. Biol. Med. 2005, 139, 141–144. [Google Scholar] [CrossRef]
- Xu, Y.; Huang, S.; Fu, X.; Fu, X. Autologous transplantation of bone marrow-derived mesenchymal stem cells: A promising therapeutic strategy for prevention of skin-graft contraction. Clin. Exp. Dermatol. 2012, 37, 497–500. [Google Scholar] [CrossRef]
- Arkoulis, N.; Watson, S.; Weiler-Mithoff, E. Stem cell enriched dermal substitutes for the treatment of late burn contractures in patients with major burns. Burns 2018, 44, 724–726. [Google Scholar] [CrossRef]
- Behm, B.; Babilas, P.; Landthaler, M.; Schreml, S. Cytokines, chemokines and growth factors in wound healing. J. Eur. Acad. Dermatol. Venereol. 2012, 26, 812–820. [Google Scholar] [CrossRef]
- Demidova-Rice, T.N.; Hamblin, M.R.; Herman, I.M. Acute and impaired wound healing: Pathophysiology and current methods for drug delivery, part 2: Role of growth factors in normal and pathological wound healing: Therapeutic potential and methods of delivery. Adv. Skin Wound Care 2012, 25, 349–370. [Google Scholar] [CrossRef]
- Upton, Z.; Wallace, H.J.; Shooter, G.K.; van Lonkhuyzen, D.R.; Yeoh-Ellerton, S.; Rayment, E.A.; Fleming, J.M.; Broszczak, D.; Queen, D.; Sibbald, R.G.; et al. Human pilot studies reveal the potential of a vitronectin: Growth factor complex as a treatment for chronic wounds. Int. Wound J. 2011, 8, 522–532. [Google Scholar] [CrossRef] [PubMed]
- Ching, Y.H.; Sutton, T.L.; Pierpont, Y.N.; Robson, M.C.; Payne, W.G. The use of growth factors and other humoral agents to accelerate and enhance burn wound healing. Eplasty. 2011, 11, e41. [Google Scholar] [PubMed]
- Fernández-Montequín, J.I.; Valenzuela-Silva, C.M.; Díaz, O.G.; Savigne, W.; Sancho-Soutelo, N.; Rivero-Fernández, F.; Sánchez-Penton, P.; Morejón-Vega, L.; Artaza-Sanz, H.; García-Herrera, A.; et al. Cuban Diabetic Foot Study Group. Intralesional injections of recombinant human epidermal growth factor promote granulation and healing in advanced diabetic foot ulcers: Multicenter, randomised, placebo-controlled, double-blind study. Int. Wound J. 2009, 6, 432–443. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.; Silva, E.A.; Mooney, D.J. Growth factor delivery-based tissue engineering: General approaches and a review of recent developments. J. R. Soc Interface 2011, 8, 153–170. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Zhou, F. Effect of recombinant human basic fibroblast growth factor combined with local application of insulin on pain and healing in diabetic patients with deep II degree burns. Am. J. Transl. Res. 2021, 13, 9364–9370. [Google Scholar]
- Zhang, J.B.; Li, H.; Zhang, L.; Wang, J.L. Observation of curative effect of recombinant human basic fibroblast growth factor combined with compound polymyxin B ointment and local application of insulin on wound healing of deep second-degree burn in diabetes mellitus: A randomized study. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 5987–5993. [Google Scholar] [CrossRef]
- Brown, G.L.; Curtsinger, L., 3rd; Brightwell, J.R.; Ackerman, D.M.; Tobin, G.R.; Polk, H.C., Jr.; George-Nascimento, C.; Valenzuela, P.; Schultz, G.S. Enhancement of epidermal regeneration by biosynthetic epidermal growth factor. J. Exp. Med. 1986, 163, 1319–1324. [Google Scholar] [CrossRef]
- Danilenko, D.M.; Ring, B.D.; Tarpley, J.E.; Morris, B.; Van, G.Y.; Morawiecki, A.; Callahan, W.; Goldenberg, M.; Hershenson, S.; Pierce, G.F. Growth factors in porcine full and partial thickness burn repair. Differing targets and effects of keratinocyte growth factor, platelet-derived growth factor-BB, epidermal growth factor, and neu differentiation factor. Am. J. Pathol. 1995, 147, 1261–1277. [Google Scholar]
- Galeano, M.; Deodato, B.; Altavilla, D.; Squadrito, G.; Seminara, P.; Marini, H.; Stagno d’Alcontres, F.; Colonna, M.; Calò, M.; Lo Cascio, P.; et al. Effect of recombinant adeno-associated virus vector-mediated vascular endothelial growth factor gene transfer on wound healing after burn injury. Crit. Care Med. 2003, 31, 1017–1025. [Google Scholar] [CrossRef]
- Xie, J.L.; Bian, H.N.; Qi, S.H.; Chen, H.D.; Li, H.D.; Xu, Y.B.; Li, T.Z.; Liu, X.S.; Liang, H.Z.; Xin, B.R.; et al. Basic fibroblast growth factor (bFGF) alleviates the scar of the rabbit ear model in wound healing. Wound Repair Regen. 2008, 16, 576–581. [Google Scholar] [CrossRef]
- Miricescu, D.; Badoiu, S.C.; Stanescu-Spinu, I.-I.; Totan, A.R.; Stefani, C.; Greabu, M. Growth Factors, Reactive Oxygen Species, and Metformin—Promoters of the Wound Healing Process in Burns? Int. J. Mol. Sci. 2021, 22, 9512. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.L.; Sun, H.; Zhang, Y.X.; Yang, Y.F.; Li, Y.M. More effective strategies for wound treatment using platelet rich plasma and plasma rich in growth factors. Burns 2023, 49, 738–739. [Google Scholar] [CrossRef] [PubMed]
- García-Sánchez, J.M.; Mirabet Lis, V.; Ruiz-Valls, A.; Pérez-Plaza, A.; Sepúlveda Sanchis, P.; Pérez-Del-Caz, M.D. Platelet rich plasma and plasma rich in growth factors for split-thickness skin graft donor site treatment in the burn patient setting: A randomized clinical trial. Burns 2022, 48, 1662–1670. [Google Scholar] [CrossRef]
- Brooks, N.C.; Marshall, A.H.; Qa’aty, N.; Hiyama, Y.; Boehning, D.; Jeschke, M.G. XBP-1s is linked to suppressed gluconeogenesis in the Ebb phase of burn injury. Mol. Med. 2013, 19, 72–78. [Google Scholar] [CrossRef] [PubMed]
- Palackic, A.; Suman, O.E.; Porter, C.; Murton, A.J.; Crandall, C.G.; Rivas, E. Rehabilitative Exercise Training for Burn Injury. Sports Med. 2021, 51, 2469–2482. [Google Scholar] [CrossRef] [PubMed]
- Jeschke, M.G.; Gauglitz, G.G.; Kulp, G.A.; Finnerty, C.C.; Williams, F.N.; Kraft, R.; Suman, O.E.; Mlcak, R.P.; Herndon, D.N. Long-term persistance of the pathophysiologic response to severe burn injury. PLoS ONE 2011, 6, e21245. [Google Scholar] [CrossRef]
- Aslanoglou, D.; Bertera, S.; Sánchez-Soto, M.; Free, R.B.; Lee, J.; Zong, W.; Xue, X.; Shrestha, S.; Brissova, M.; Logan, R.W.; et al. Dopamine regulates pancreatic glucagon and insulin secretion via adrenergic and dopaminergic receptors. Transl. Psychiatry 2021, 11, 59. [Google Scholar] [CrossRef]
- Rani, M.; Schwacha, M.G. Aging and the pathogenic response to burn. Aging Dis. 2012, 3, 171–180. [Google Scholar]
- Sierawska, O.; Małkowska, P.; Taskin, C.; Hrynkiewicz, R.; Mertowska, P.; Grywalska, E.; Korzeniowski, T.; Torres, K.; Surowiecka, A.; Niedźwiedzka-Rystwych, P.; et al. Innate Immune System Response to Burn Damage—Focus on Cytokine Alteration. Int. J. Mol. Sci. 2022, 23, 716. [Google Scholar] [CrossRef]
- Guo, H.; Callaway, J.B.; Ting, J.P.-Y. Inflammasomes: Mechanism of Action, Role in Disease, and Therapeutics. Nat. Med. 2015, 21, 677–687. [Google Scholar] [CrossRef]
- Korkmaz, H.I.; Krijnen, P.A.J.; Ulrich, M.M.W.; de Jong, E.; van Zuijlen, P.P.M.; Niessen, H.W.M. The Role of Complement in the Acute Phase Response after Burns. Burns 2017, 43, 1390–1399. [Google Scholar] [CrossRef] [PubMed]
- Pereira, C.; Murphy, K.; Jeschke, M.; Herndon, D.N. Post burn muscle wasting and the effects of treatments. Int. J. Biochem. Cell Biol. 2005, 37, 1948–1961. [Google Scholar] [CrossRef] [PubMed]
- Berger, M.M.; Pichard, C. Development and Current Use of Parenteral Nutrition in Critical Care—An Opinion Paper. Crit. Care 2014, 18, 478. [Google Scholar] [CrossRef] [PubMed]
- Clark, A.; Imran, J.; Madni, T.; Wolf, S.E. Nutrition and metabolism in burn patients. Burn. Trauma 2017, 5, 11. [Google Scholar] [CrossRef]
- Dickerson, R.N.; Gervasio, J.M.; Riley, M.L.; Murrell, J.E.; Hickerson, W.L.; Kudsk, K.A.; Brown, R.O. Accuracy of predictive methods for estimating resting energy expenditure in thermally injured patients. JPEN J. Parenter. Enteral. Nutr. 2002, 26, 17–29. [Google Scholar] [CrossRef]
- Chan, M.M.; Chan, G.M. Nutritional therapy for burns in children and adults. Nutrition 2009, 25, 261–269. [Google Scholar] [CrossRef]
- Hall, K.L.; Shahrokhi, S.; Jeschke, M.G. Enteral nutrition support in burn care: A review of current recommendations as instituted in the Ross Tilley Burn Centre. Nutrients 2012, 4, 1554–1565. [Google Scholar] [CrossRef]
- Heyland, D.K.; Wibbenmeyer, L.; Pollack, J.A.; Friedman, B.; Turgeon, A.F.; Eshraghi, N.; Jeschke, M.G.; Bélisle, S.; Grau, D.; Mandell, S.; et al. Re-Energize Trial Team. A Randomized Trial of Enteral Glutamine for Treatment of Burn Injuries. N. Engl. J. Med. 2022, 387, 1001–1010. [Google Scholar] [CrossRef]
- Wischmeyer, P.E. The glutamine story: Where are we now? Curr. Opin. Crit. Care 2006, 12, 142–148. [Google Scholar] [CrossRef]
- Lin, J.J.; Chung, X.J.; Yang, C.Y.; Lau, H.L. A meta-analysis of trials using the intention to treat principle for glutamine supplementation in critically ill patients with burn. Burns 2013, 39, 565–570. [Google Scholar] [CrossRef]
- van Zanten, A.R.; Dhaliwal, R.; Garrel, D.; Heyland, D.K. Enteral glutamine supplementation in critically ill patients: A systematic review and meta-analysis. Crit. Care 2015, 19, 294. [Google Scholar] [CrossRef]
- Yan, H.; Peng, X.; Huang, Y.; Zhao, M.; Li, F.; Wang, P. Effects of early enteral arginine supplementation on resuscitation of severe burn patients. Burns 2007, 33, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Marín, M.C.; Osimani, N.E.; Rey, G.E.; de Alaniz, M.J. n-3 Fatty acid supplementation in burned pediatric patients. Acta Paediatr. 2009, 98, 1982–1987. [Google Scholar] [CrossRef] [PubMed]
- Tihista, S.; Echavarría, E. Effect of omega 3 polyunsaturated fatty acids derived from fish oil in major burn patients: A prospective randomized controlled trial pilot. Clin. Nutr. 2018, 37, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Bohr, S.; Patel, S.J.; Sarin, D.; Irimia, D.; Yarmush, M.L.; Berthiaume, F. Resolvin D2 prevents secondary thrombosis and necrosis in a mouse burn wound model. Wound Repair Regen. 2013, 21, 35–43. [Google Scholar] [CrossRef]
- Inoue, Y.; Yu, Y.M.; Kurihara, T.; Vasilyev, A.; Ibrahim, A.; Oklu, R.; Zhao, G.; Nair, A.V.; Brown, D.; Fischman, A.J.; et al. Kidney and Liver Injuries After Major Burns in Rats Are Prevented by Resolvin D2. Crit. Care Med. 2016, 44, e241–e252. [Google Scholar] [CrossRef]
- Patsouris, D.; Qi, P.; Abdullahi, A.; Stanojcic, M.; Chen, P.; Parousis, A.; Amini-Nik, S.; Jeschke, M.G. Burn Induces Browning of the Subcutaneous White Adipose Tissue in Mice and Humans. Cell Rep. 2015, 13, 1538–1544. [Google Scholar] [CrossRef]
- Nguyen, K.D.; Qiu, Y.; Cui, X.; Goh, Y.P.; Mwangi, J.; David, T.; Mukundan, L.; Brombacher, F.; Locksley, R.M.; Chawla, A. Alternatively activated macrophages produce catecholamines to sustain adaptive thermogenesis. Nature 2011, 480, 104–108. [Google Scholar] [CrossRef]
- Williams, F.N.; Jeschke, M.G.; Chinkes, D.L.; Suman, O.E.; Branski, L.K.; Herndon, D.N. Modulation of the hypermetabolic response to trauma: Temperature, nutrition, and drugs. J. Am. Coll. Surg. 2009, 208, 489–502. [Google Scholar] [CrossRef]
- Herndon, D.N.; Rodriguez, N.A.; Diaz, E.C.; Hegde, S.; Jennings, K.; Mlcak, R.P.; Suri, J.S.; Lee, J.O.; Williams, F.N.; Meyer, W.; et al. Long-term propranolol use in severely burned pediatric patients: A randomized controlled study. Ann. Surg. 2012, 256, 402–411. [Google Scholar] [CrossRef]
- Brooks, N.C.; Song, J.; Boehning, D.; Kraft, R.; Finnerty, C.C.; Herndon, D.N.; Jeschke, M.G. Propranolol improves impaired hepatic phosphatidylinositol 3-kinase/akt signaling after burn injury. Mol. Med. 2012, 18, 707–711. [Google Scholar] [CrossRef]
- Jeschke, M.G.; Klein, D.; Herndon, D.N. Insulin treatment improves the systemic inflammatory reaction to severe trauma. Ann. Surg. 2004, 239, 553–560. [Google Scholar] [CrossRef]
- Pham, T.N.; Warren, A.J.; Phan, H.H.; Molitor, F.; Greenhalgh, D.G.; Palmieri, T.L. Impact of tight glycemic control in severely burned children. J. Trauma 2005, 59, 1148–1154. [Google Scholar] [CrossRef] [PubMed]
- Hemmila, M.R.; Taddonio, M.A.; Arbabi, S.; Maggio, P.M.; Wahl, W.L. Intensive insulin therapy is associated with reduced infectious complications in burn patients. Surgery 2008, 144, 629–635. [Google Scholar] [CrossRef] [PubMed]
- Stanojcic, M.; Chen, P.; Harrison, R.A.; Wang, V.; Antonyshyn, J.; Zúñiga-Pflücker, J.C.; Jeschke, M.G. Leukocyte infiltration and activation of the NLRP3 inflammasome in white adipose tissue following thermal injury. Crit. Care Med. 2014, 42, 1357–1364. [Google Scholar] [CrossRef] [PubMed]
- Macrae, D.; Grieve, R.; Allen, E.; Sadique, Z.; Morris, K.; Pappachan, J.; Parslow, R.; Tasker, R.C.; Elbourne, D. CHiP Investigators. A randomized trial of hyperglycemic control in pediatric intensive care. N. Engl. J. Med. 2014, 370, 107–118. [Google Scholar] [CrossRef]
- Jeschke, M.G.; Pinto, R.; Herndon, D.N.; Finnerty, C.C.; Kraft, R. Hypoglycemia is associated with increased postburn morbidity and mortality in pediatric patients. Crit. Care Med. 2014, 42, 1221–1231. [Google Scholar] [CrossRef]
- Greabu, M.; Badoiu, S.C.; Stanescu-Spinu, I.-I.; Miricescu, D.; Totan, A.R.; Badoiu, S.E.; Costagliola, M.; Jinga, V. Drugs Interfering with Insulin Resistance and Their Influence on the Associated Hypermetabolic State in Severe Burns: A Narrative Review. Int. J. Mol. Sci. 2021, 22, 9782. [Google Scholar] [CrossRef] [PubMed]
- Badoiu, S.C.; Miricescu, D.; Stanescu-Spinu, I.-I.; Ripszky Totan, A.; Badoiu, S.E.; Costagliola, M.; Greabu, M. Glucose Metabolism in Burns—What Happens? Int. J. Mol. Sci. 2021, 22, 5159. [Google Scholar] [CrossRef]
- Takala, J.; Ruokonen, E.; Webster, N.R.; Nielsen, M.S.; Zandstra, D.F.; Vundelinckx, G.; Hinds, C.J. Increased mortality associated with growth hormone treatment in critically ill adults. N. Engl. J. Med. 1999, 341, 785–792. [Google Scholar] [CrossRef]
- Spies, M.; Wolf, S.E.; Barrow, R.E.; Jeschke, M.G.; Herndon, D.N. Modulation of types I and II acute phase reactants with insulin-like growth factor-1/binding protein-3 complex in severely burned children. Crit. Care Med. 2002, 30, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Pham, T.N.; Klein, M.B.; Gibran, N.S.; Arnoldo, B.D.; Gamelli, R.L.; Silver, G.M.; Jeschke, M.G.; Finnerty, C.C.; Tompkins, R.G.; Herndon, D.N. Impact of oxandrolone treatment on acute outcomes after severe burn injury. J. Burn. Care Res. 2008, 29, 902–906. [Google Scholar] [CrossRef] [PubMed]
- Hart, D.W.; Wolf, S.E.; Ramzy, P.I.; Chinkes, D.L.; Beauford, R.B.; Ferrando, A.A.; Wolfe, R.R.; Herndon, D.N. Anabolic effects of oxandrolone after severe burn injury. Ann. Surg. 2001, 233, 556–564. [Google Scholar] [CrossRef] [PubMed]
- Ring, J.; Heinelt, M.; Sharma, S.; Letourneau, S.; Jeschke, M.G. Oxandrolone in the Treatment of Burn Injuries: A systematic review and meta-analysis. J. Burn Care Res. 2020, 41, 190–199. [Google Scholar] [CrossRef]
- Li, H.; Guo, Y.; Yang, Z.; Roy, M.; Guo, Q. The efficacy and safety of oxandrolone treatment for patients with severe burns: A systematic review and meta-analysis. Burns 2016, 42, 717–727. [Google Scholar] [CrossRef]
- Jalkh, A.P.C.; Eastmond, A.K.; Shetty, C.; Rizvi, S.M.H.A.; Sharaf, J.; Williams, K.D.; Tariq, M.; Acharekar, M.V.; Guerrero Saldivia, S.E.; Unnikrishnan, S.N.; et al. Oxandrolone Efficacy in Wound Healing in Burn and Decubitus Ulcer Patients: A systematic review. Cureus 2022, 14, e28079. [Google Scholar] [CrossRef]
- Altarrah, K.; Tan, P.; Acharjee, A.; Hazeldine, J.; Torlinska, B.; Wilson, Y.; Torlinski, T.; Moiemen, N.; Lord, J.M. Differential benefits of steroid therapies in adults after major burn injury. J. Plast. Reconstr. Aesthet. Surg. 2022, 75, 2616–2624. [Google Scholar] [CrossRef]
- Przkora, R.; Jeschke, M.G.; Barrow, R.E.; Suman, O.E.; Meyer, W.J.; Finnerty, C.C.; Sanford, A.P.; Lee, J.; Chinkes, D.L.; Mlcak, R.P.; et al. Metabolic and hormonal changes in severely burned children on long-term oxandrolone therapy. Ann. Surg. 2005, 242, 384–389. [Google Scholar] [CrossRef]
- Jeschke, M.G.; Finnerty, C.C.; Suman, O.E.; Kulp, G.; Mlcak, R.P.; Herndon, D.N. The effect of oxandrolone on the endocrine, inflammatory, and hypermetabolic responses during the acute phase postburn. Ann. Surg. 2007, 246, 351–360. [Google Scholar] [CrossRef]
- Bulger, E.M.; Jurkovich, G.J.; Farver, C.L.; Klotz, P.; Maier, R.V. Oxandrolone does not improve outcome of ventilator dependent surgical patients. Ann. Surg. 2004, 240, 472–478. [Google Scholar] [CrossRef]
- Ali, Y.H.; Ali, T. Nandrolone decanoate safely combats catabolism in burn patients: A new potential indication after withdrawal. Burns 2022, 48, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Romanowski, K.S.; Carson, J.; Pape, K.; Bernal, E.; Sharar, S.; Wiechman, S.; Carter, D.; Liu, Y.M.; Nitzschke, S.; Bhalla, P.; et al. American Burn Association Guidelines on the Management of Acute Pain in the Adult Burn Patient: A Review of the Literature, a Compilation of Expert Opinion, and Next Steps. J. Burn Care Res. 2020, 41, 1129–1151. [Google Scholar] [CrossRef] [PubMed]
- Kwaśniewska, O.; Walczak, J.; Włodarczyk, J.; Włodarczyk, M. Pain Management in Burn Patients. Plast. Surg. Burn. 2021, 9, 101–105. [Google Scholar] [CrossRef]
- American Burn Association (ABA). Available online: https://ameriburn.org/wp-content/uploads/2018/12/nbaw2019_statsdataresources_120618-1.pdf (accessed on 3 March 2023).
- Rabbitts, J.A.; Fisher, E.; Rosenbloom, B.N.; Palermo, T.M. Prevalence and Predictors of Chronic Postsurgical Pain in Children: A Systematic Review and Meta-Analysis. J. Pain 2017, 18, 605–614. [Google Scholar] [CrossRef]
- Rosenberg, L.; Rosenberg, M.; Sharp, S.; Thomas, C.R.; Humphries, H.F.; Holzer, C.E., 3rd; Herndon, D.N.; Meyer, W.J., 3rd. Does Acute Propranolol Treatment Prevent Posttraumatic Stress Disorder, Anxiety, and Depression in Children with Burns? J. Child Adolesc. Psychopharmacol. 2018, 28, 117–123. [Google Scholar] [CrossRef]
- Hansen, J.K.; Voss, J.; Ganatra, H.; Langner, T.; Chalise, P.; Stokes, S.; Bhavsar, D.; Kovac, A.L. Sedation and Analgesia During Pediatric Burn Dressing Change: A Survey of American Burn Association Centers. J. Burn. Care Res. 2019, 40, 287–293. [Google Scholar] [CrossRef]
- Lopez, U.; Martin, J.; van Assche, M.; Fleury Schubert, A.; Fournet, M.; Quartier, V.; Habre, W.; Van der Linden, M. Classification of postoperative behavior disturbances in preschool children: A qualitative study. Paediatr. Anaesth. 2019, 29, 712–720. [Google Scholar] [CrossRef]
- Wang, E.; Thomas, J.J.; Rodriguez, S.T.; Kennedy, K.M.; Caruso, T.J. Virtual reality for pediatric periprocedural care. Curr. Opin. Anaesthesiol. 2021, 34, 284–291. [Google Scholar] [CrossRef]
- Caruso, T.J.; O′Connell, C.; Qian, J.J.; Kung, T.; Wang, E.; Kinnebrew, S.; Pearson, M.; Kist, M.; Menendez, M.; Rodriguez, S.T. Retrospective Review of the Safety and Efficacy of Virtual Reality in a Pediatric Hospital. Pediatr. Qual. Saf. 2020, 5, e293. [Google Scholar] [CrossRef]
- Keefe, F.J.; Main, C.J.; George, S.Z. Advancing Psychologically Informed Practice for Patients With Persistent Musculoskeletal Pain: Promise, Pitfalls, and Solutions. Phys. Ther. 2018, 98, 398–407. [Google Scholar] [CrossRef]
- Rohilla, L.; Agnihotri, M.; Trehan, S.K.; Sharma, R.K.; Ghai, S. Effect of Music Therapy on Pain Perception, Anxiety, and Opioid Use During Dressing Change Among Patients with Burns in India: A Quasi-experimental, Cross-over Pilot Study. Ostomy Wound Manag. 2018, 64, 40–46. [Google Scholar] [CrossRef]
- Yildirim, M.; Koroglu, E.; Yucel, C.; Kirlak, S.; Sen, S. The effect of hospital clown nurse on children′s compliance to burn dressing change. Burns 2019, 45, 190–198. [Google Scholar] [CrossRef] [PubMed]
- Hitching, R.; Hoffman, H.G.; Garcia-Palacios, A.; Adamson, M.M.; Madrigal, E.; Alhalabi, W.; Alhudali, A.; Sampaio, M.; Peterson, B.; Fontenot, M.R.; et al. The Emerging Role of Virtual Reality as an Adjunct to Procedural Sedation and Anesthesia: A Narrative Review. J. Clin. Med. 2023, 12, 843. [Google Scholar] [CrossRef] [PubMed]
- Trost, Z.; France, C.; Anam, M.; Shum, C. Virtual reality approaches to pain: Toward a state of the science. Pain 2021, 162, 325–331. [Google Scholar] [CrossRef]
- Alrimy, T.; Alhalabi, W.; Malibari, A.; Alzahrani, F.; Alrajhi, S.; Yamani, A.; Ahmed, H.; Abduljawad, A.; Nasser, E.; ALattar, S.; et al. Desktop Virtual Reality Offers a Novel Approach to Minimize Pain and Anxiety during Burn Wound Clean-ing/Debridement in Infants and Young Children: A Randomized Crossover Pilot Study. J. Clin. Med. 2023, 12, 4985. [Google Scholar] [CrossRef]
- Khadra, C.; Ballard, A.; Dery, J.; Paquin, D.; Fortin, J.S.; Perreault, I.; Labbe, D.R.; Hoffman, H.G.; Bouchard, S.; LeMay, S. Projector-based virtual reality dome environment for procedural pain and anxiety in young children with burn injuries: A pilot study. J. Pain Res. 2018, 11, 343–353. [Google Scholar] [CrossRef]
- Khadra, C.; Ballard, A.; Paquin, D.; Cotes-Turpin, C.; Hoffman, H.G.; Perreault, I.; Fortin, J.S.; Bouchard, S.; Theroux, J.; Le May, S. Effects of a projector-based hybrid virtual reality on pain in young children with burn injuries during hydrotherapy sessions: A within-subject randomized crossover trial. Burns 2020, 46, 1571–1584. [Google Scholar] [CrossRef]
- Hoffman, H.G.; Richards, T.L.; Van Oostrom, T.; Coda, B.A.; Jensen, M.P.; Blough, D.K.; Sharar, S.R. The analgesic effects of opioids and immersive virtual reality distraction: Evidence from subjective and functional brain imaging assessments. Anesth. Analg. 2007, 105, 1776–1783. [Google Scholar] [CrossRef]
- Joo, S.Y.; Park, C.H.; Cho, Y.S.; Seo, C.H.; Ohn, S.H. Plastic Changes in Pain and Motor Network Induced by Chronic Burn Pain. J. Clin. Med. 2021, 10, 2592. [Google Scholar] [CrossRef]
- Bittner, E.A.; Shank, E.; Woodson, L.; Martyn, J.A. Acute and perioperative care of the burn-injured patient. Anesthesiology 2015, 122, 448–464. [Google Scholar] [CrossRef]
- Volkow, N.D.; McLellan, T.A.; Cotto, J.H.; Karithanom, M.; Weiss, S.R. Characteristics of opioid prescriptions in 2009. JAMA 2011, 305, 1299–1301. [Google Scholar] [CrossRef] [PubMed]
- Emery, M.A.; Eitan, S. Drug-specific differences in the ability of opioids to manage burn pain. Burns 2020, 46, 503–513. [Google Scholar] [CrossRef] [PubMed]
- Dowell, D.; Haegerich, T.M.; Chou, R. CDC Guideline for Prescribing Opioids for Chronic Pain—United States, 2016. MMWR Recomm. Rep. 2016, 65, 1–49. [Google Scholar] [CrossRef] [PubMed]
- Schneider, J.C.; Harris, N.L.; El Shami, A.; Sheridan, R.L.; Schulz, J.T., 3rd; Bilodeau, M.L.; Ryan, C.M. A descriptive review of neu-ropathic-like pain after burn injury. J. Burn Care Res. 2006, 27, 524–528. [Google Scholar] [CrossRef]
- Kaul, I.; Amin, A.; Rosenberg, M.; Rosenberg, L.; Meyer, W.J., 3rd. Use of gabapentin and pregabalin for pruritus and neuropathic pain associated with major burn injury: A retrospective chart review. Burns 2018, 44, 414–422. [Google Scholar] [CrossRef]
- Alencar de Castro, R.J.; Leal, P.C.; Sakata, R.K. Pain management in burn patients. Braz. J. Anesthesiol. 2013, 63, 149–153. [Google Scholar] [CrossRef]
- Gamst-Jensen, H.; Vedel, P.N.; Lindberg-Larsen, V.O.; Egerod, I. Acute pain management in burn patients: Appraisal and thematic analysis of four clinical guidelines. Burns 2014, 40, 1463–1469. [Google Scholar] [CrossRef]
- Bayat, A.; Ramaiah, R.; Bhananker, S.M. Analgesia and sedation for children undergoing burn wound care. Expert. Rev. Neurother. 2010, 10, 1747–1759. [Google Scholar] [CrossRef]
- Gavériaux-Ruff, C.; Kieffer, B.L. Delta opioid receptor analgesia: Recent contributions from pharmacology and molecular approaches. Behav. Pharmacol. 2011, 22, 405–414. [Google Scholar] [CrossRef]
- Yi, S.P.; Kong, Q.H.; Li, Y.L.; Pan, C.L.; Yu, J.; Cui, B.Q.; Wang, Y.F.; Wang, G.L.; Zhou, P.L.; Wang, L.L.; et al. The opioid receptor triple agonist DPI-125 produces analgesia with less respiratory depression and reduced abuse liability. Acta Pharmacol. Sin. 2017, 38, 977–989. [Google Scholar] [CrossRef]
- Zhuang, T.; Xiong, J.; Hao, S.; Du, W.; Liu, Z.; Liu, B.; Zhang, G.; Chen, Y. Bifunctional μ opioid and σ1 receptor ligands as novel analgesics with reduced side effects. Eur. J. Med. Chem. 2021, 223, 113658. [Google Scholar] [CrossRef] [PubMed]
- Berecki, G.; Motin, L.; Adams, D.J. Voltage-Gated R-Type Calcium Channel Inhibition via Human μ-, δ-, and κ-opioid Re-ceptors Is Voltage-Independently Mediated by Gβγ Protein Subunits. Mol. Pharmacol. 2016, 89, 187–196. [Google Scholar] [CrossRef]
- Magalhaes, A.C.; Dunn, H.; Ferguson, S.S. Regulation of GPCR activity, trafficking and localization by GPCR-interacting proteins. Br. J. Pharmacol. 2012, 165, 1717–1736. [Google Scholar] [CrossRef] [PubMed]
- Melief, E.J.; Miyatake, M.; Bruchas, M.R.; Chavkin, C. Ligand-directed c-Jun N-terminal kinase activation disrupts opioid receptor signaling. Proc. Natl. Acad. Sci. USA 2010, 107, 11608–11613. [Google Scholar] [CrossRef]
- Smith, J.S.; Lefkowitz, R.J.; Rajagopal, S. Biased signalling: From simple switches to allosteric microprocessors. Nat. Rev. Drug Discov. 2018, 17, 243–260. [Google Scholar] [CrossRef] [PubMed]
- Thompson, G.L.; Kelly, E.; Christopoulos, A.; Canals, M. Novel GPCR paradigms at the μ-opioid receptor. Br. J. Pharmacol. 2015, 172, 287–296. [Google Scholar] [CrossRef]
- DeWire, S.M.; Yamashita, D.S.; Rominger, D.H.; Liu, G.; Cowan, C.L.; Graczyk, T.M.; Chen, X.T.; Pitis, P.M.; Gotchev, D.; Yuan, C.; et al. AG protein-biased ligand at the μ-opioid receptor is potently analgesic with reduced gastrointestinal and respiratory dysfunction compared with morphine. J. Pharmacol. Exp. Ther. 2013, 344, 708–717. [Google Scholar] [CrossRef]
- Thomas, J.; Mustafa, S.; Johnson, J.; Nicotra, L.; Hutchinson, M. The relationship between opioids and immune signalling in the spinal cord. Handb. Exp. Pharmacol. 2015, 227, 207–238. [Google Scholar] [CrossRef]
- Eidson, L.N.; Inoue, K.; Young, L.J.; Tansey, M.G.; Murphy, A.Z. Toll-like Receptor 4 Mediates Morphine-Induced Neuroinflammation and Tolerance via Soluble Tumor Necrosis Factor Signaling. Neuropsychopharmacology 2017, 42, 661–670. [Google Scholar] [CrossRef]
- Jamison, R.N.; Mao, J. Opioid Analgesics. Mayo Clin. Proc. 2015, 90, 957–968. [Google Scholar] [CrossRef]
- McIntyre, M.K.; Clifford, J.L.; Maani, C.V.; Burmeister, D.M. Progress of clinical practice on the management of burn-associated pain: Lessons from animal models. Burns 2016, 42, 1161–1172. [Google Scholar] [CrossRef]
- Pilat, D.; Piotrowska, A.; Rojewska, E.; Jurga, A.; Ślusarczyk, J.; Makuch, W.; Basta-Kaim, A.; Przewlocka, B.; Mika, J. Blockade of IL-18 signaling diminished neuropathic pain and enhanced the efficacy of morphine and buprenorphine. Mol. Cell Neurosci. 2016, 71, 114–124. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.C.; Luo, C.Q.; Li, X. Systemic inflammatory response syndrome following burns is mediated by brain natriuretic pep-tide/natriuretic peptide A receptor-induced shock factor 1 signaling pathway. Clin. Exp. Pharmacol. Physiol. 2016, 43, 921–929. [Google Scholar] [CrossRef] [PubMed]
- Song, L.; Wang, S.; Zuo, Y.; Chen, L.; Martyn, J.A.; Mao, J. Midazolam exacerbates morphine tolerance and morphine-induced hyperactive behaviors in young rats with burn injury. Brain Res. 2014, 1564, 52–61. [Google Scholar] [CrossRef]
- Mittal, N.; Tan, M.; Egbuta, O.; Desai, N.; Crawford, C.; Xie, C.W.; Evans, C.; Walwyn, W. Evidence that behavioral phenotypes of morphine in β-arr2-/- mice are due to the unmasking of JNK signaling. Neuropsychopharmacology 2012, 37, 1953–1962. [Google Scholar] [CrossRef]
- Emery, M.A.; Bates, M.L.; Wellman, P.J.; Eitan, S. Differential Effects of Oxycodone, Hydrocodone, and Morphine on Activation Levels of Signaling Molecules. Pain Med. 2016, 17, 908–914. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Zhang, L.; Ma, Y.; Chen, L.; Tian, Y.; Mao, J.; Martyn, J.J. Nociceptive behavior following hindpaw burn injury in young rats: Response to systemic morphine. Pain Med. 2011, 12, 87–98. [Google Scholar] [CrossRef] [PubMed]
- Emery, M.A.; Bates, M.L.S.; Wellman, P.J.; Eitan, S. Burn injury decreases the antinociceptive effects of opioids. Behav. Pharmacol. 2017, 28, 285–293. [Google Scholar] [CrossRef]
- Yin, K.; Deuis, J.R.; Lewis, R.J.; Vetter, I. Transcriptomic and behavioural characterisation of a mouse model of burn pain identify the cholecystokinin 2 receptor as an analgesic target. Mol. Pain. 2016, 12, 1744806916665366. [Google Scholar] [CrossRef]
- Agarwal, S.; Polydefkis, M.; Block, B.; Haythornthwaite, J.; Raja, S.N. Transdermal fentanyl reduces pain and improves func-tional activity in neuropathic pain states. Pain Med. 2007, 8, 554–562. [Google Scholar] [CrossRef]
- Grace, P.M.; Strand, K.A.; Galer, E.L.; Urban, D.J.; Wang, X.; Baratta, M.V.; Fabisiak, T.J.; Anderson, N.D.; Cheng, K.; Greene, L.I.; et al. Morphine paradoxically prolongs neuropathic pain in rats by amplifying spinal NLRP3 inflammasome activation. Proc. Natl. Acad. Sci. USA 2016, 113, E3441–E3450. [Google Scholar] [CrossRef] [PubMed]
- Emery, M.A.; Shawn Bates, M.L.; Wellman, P.J.; Eitan, S. Hydrocodone is More Effective than Morphine or Oxycodone in Sup-pressing the Development of Burn-Induced Mechanical Allodynia. Pain Med. 2017, 18, 2170–2180. [Google Scholar] [CrossRef] [PubMed]
- Hoogewerf, C.J.; van Baar, M.E.; Middelkoop, E.; van Loey, N.E. Impact of facial burns: Relationship between depressive symptoms, self-esteem and scar severity. Gen. Hosp. Psychiatry 2014, 36, 271–276. [Google Scholar] [CrossRef]
- Bond, S.; Gourlay, C.; Desjardins, A.; Bodson-Clermont, P.; Boucher, M.È. Anxiety, depression and PTSD-related symptoms in spouses and close relatives of burn survivors: When the supporter needs to be supported. Burns 2017, 43, 592–601. [Google Scholar] [CrossRef]
- Klifto, K.M.; Dellon, A.L.; Hultman, C.S. Prevalence and associated predictors for patients developing chronic neuropathic pain following burns. Burn. Trauma 2020, 8, tkaa011. [Google Scholar] [CrossRef] [PubMed]
- Palackic, A.; Franco-Mesa, C.; Beck, I.; Nolte, S.; Tapking, C.; Panayi, A.C.; Stolle, A.; Haug, V.; Hirche, C.; Kneser, U.; et al. The Impact of Facial Burns on Short- and Long-Term Quality of Life and Psychological Distress—A Prospective Matched Cohort Study. J. Clin. Med. 2023, 12, 5057. [Google Scholar] [CrossRef]
- Kundu, K.; Rawat, V.S.; Chattopadhyay, D. Gender differences in quality of life and psychological impact of facial burn scars in a tertiary care center. Burns 2021, 47, 1153–1160. [Google Scholar] [CrossRef]
- Habib, Z.; Saddul, R.; Kamran, F. Perceptions and experiences of female burn survivors with facial disfigurement. Chin. J. Traumatol. 2021, 24, 53–56. [Google Scholar] [CrossRef]
- Abouzeid, C.A.; Wolfe, A.E.; Ni, P.; Carrougher, G.J.; Gibran, N.S.; Hammond, F.M.; Holavanahalli, R.; McMullen, K.A.; Roaten, K.; Suman, O.; et al. Are burns a chronic condition? Examining patient reported outcomes up to 20 years after burn injury-A Burn Model System National Database investigation. J. Trauma Acute Care Surg. 2022, 92, 1066–1074. [Google Scholar] [CrossRef]
- Bhatti, D.S.; Ul Ain, N.; Zulkiffal, R.; Al-Nabulsi, Z.S.; Faraz, A.; Ahmad, R. Anxiety and Depression Among Non-Facial Burn Patients at a Tertiary Care Center in Pakistan. Cureus 2020, 12, e11347. [Google Scholar] [CrossRef]
- Gurbuz, K.; Demir, M.; Basaran, A.; Das, K. Most Prominent Factors Contributing to Burn Injury-Related Amputations: An Analysis of a Referral Burn Center. J. Burn Care Res. 2022, 43, 921–925. [Google Scholar] [CrossRef]
- Gauglitz, G.G.; Korting, H.C.; Pavicic, T.; Ruzicka, T.; Jeschke, M.G. Hypertrophic scarring and keloids: Pathomechanisms and current and emerging treatment strategies. Mol. Med. 2011, 17, 113–125. [Google Scholar] [CrossRef] [PubMed]
- Finnerty, C.C.; Jeschke, M.G.; Branski, L.K.; Barret, J.P.; Dziewulski, P.; Herndon, D.N. Hypertrophic scarring: The greatest unmet challenge after burn injury. Lancet 2016, 388, 1427–1436. [Google Scholar] [CrossRef]
- Vincent, A.S.; Phan, T.T.; Mukhopadhyay, A.; Lim, H.Y.; Halliwell, B.; Wong, K.P. Human skin keloid fibroblasts display bioenergetics of cancer cells. J. Investig. Dermatol. 2008, 128, 702–709. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Jang, Y.J. Recent Understandings of Biology, Prophylaxis and Treatment Strategies for Hypertrophic Scars and Keloids. Int. J. Mol. Sci. 2018, 19, 711. [Google Scholar] [CrossRef]
- Macintyre, L.; Baird, M. Pressure garments for use in the treatment of hypertrophic scars—A review of the problems associated with their use. Burns 2006, 32, 10–15. [Google Scholar] [CrossRef]
- Kelly, A.P. Medical and surgical therapies for keloids. Dermatol. Ther. 2004, 17, 212–218. [Google Scholar] [CrossRef]
- Renò, F.; Sabbatini, M.; Lombardi, F.; Stella, M.; Pezzuto, C.; Magliacani, G.; Cannas, M. In vitro mechanical compression induces apoptosis and regulates cytokines release in hypertrophic scars. Wound Repair Regen. 2003, 11, 331–336. [Google Scholar] [CrossRef] [PubMed]
- Van den Kerckhove, E.; Stappaerts, K.; Fieuws, S.; Laperre, J.; Massage, P.; Flour, M.; Boeckx, W. The assessment of erythema and thickness on burn related scars during pressure garment therapy as a preventive measure for hypertrophic scarring. Burns 2005, 31, 696–702. [Google Scholar] [CrossRef]
- Fulton, J.E. Silicone Gel Sheeting for the Prevention and Management of Evolving Hypertrophic and Keloid Scars. Dermatol. Surg. 1995, 21, 947–951. [Google Scholar] [CrossRef]
- Jalali, M.; Bayat, A. Current use of steroids in management of abnormal raised skin scars. Surgeon 2007, 5, 175–180. [Google Scholar] [CrossRef]
- Seo, B.F.; Jung, S.N. The Immunomodulatory Effects of Mesenchymal Stem Cells in Prevention or Treatment of Excessive Scars. Stem Cells Int. 2016, 2016, 6937976. [Google Scholar] [CrossRef]
- Waterman, R.S.; Tomchuck, S.L.; Henkle, S.L.; Βncourt, A.M. A new mesenchymal stem cell (MSC) paradigm: Polarization into a pro-inflammatory MSC1 or an Immunosuppressive MSC2 phenotype. PLoS ONE 2010, 5, e10088. [Google Scholar] [CrossRef]
- Negenborn, V.L.; Groen, J.W.; Smit, J.M.; Niessen, F.B.; Mullender, M.G. The Use of Autologous Fat Grafting for Treatment of Scar Tissue and Scar-Related Conditions: A Systematic Review. Plast. Reconstr. Surg. 2016, 137, 31e–43e. [Google Scholar] [CrossRef]
- Lee, J.H.; Kim, S.E.; Lee, A.Y. Effects of interferon-α2b on keloid treatment with triamcinolone acetonide intralesional injection. Int. J. Dermatol. 2008, 47, 183–186. [Google Scholar] [CrossRef] [PubMed]
- Elhefnawy, A.M. Assessment of intralesional injection of botulinum toxin type A injection for hypertrophic scars. Indian J. Dermatol. Venereol. Leprol. 2016, 82, 279–283. [Google Scholar] [CrossRef] [PubMed]
- Jones, C.D.; Guiot, L.; Samy, M.; Gorman, M.; Tehrani, H. The Use of Chemotherapeutics for the Treatment of Keloid Scars. Dermatol. Rep. 2015, 7, 5880. [Google Scholar] [CrossRef] [PubMed]
- Occleston, N.L.; O’Kane, S.; Laverty, H.G.; Cooper, M.; Fairlamb, D.; Mason, T.; Bush, J.A.; Ferguson, M.W. Discovery and development of avotermin (recombinant human transforming growth factor β3): A new class of prophylactic therapeutic for the improvement of scarring. Wound Repair Regen. 2011, 19, s38–s48. [Google Scholar] [CrossRef]
- Gurtner, G.C.; Dauskardt, R.H.; Wong, V.W.; Bhatt, K.A.; Wu, K.; Vial, I.N.; Padois, K.; Korman, J.M.; Longaker, M.T. Improving cutaneous scar formation by controlling the mechanical environment: Large animal and phase I studies. Ann. Surg. 2011, 254, 217–225. [Google Scholar] [CrossRef]
- Daya, M.; Nair, V. Traction-assisted dermatogenesis by serial intermittent skin tape application. Plast. Reconstr. Surg. 2008, 122, 1047–1054. [Google Scholar] [CrossRef]
- Beuth, J.; Hunzelmann, N.; van Leendert, R.; Basten, R.; Noehle, M.; Schneider, B. Safety and efficacy of local administration of contractubex to hypertrophic scars in comparison to corticosteroid treatment. Results of a multicenter, comparative epidemiological cohort study in Germany. In Vivo 2006, 20, 277–283. [Google Scholar] [PubMed]
- Atiyeh, B.S. Nonsurgical management of hypertrophic scars: Evidence-based therapies, standard practices, and emerging methods. Aesthet. Plast. Surg. 2007, 31, 468–492. [Google Scholar] [CrossRef] [PubMed]
- Schiefer, J.L.; Andreae, J.; Fuchs, P.C.; Lefering, R.; Heidekrueger, P.I.; Schulz, A.; Bagheri, M. Evaluation of Scar Quality after Treatment of Superficial Burns with Dressilk® and Suprathel®—In an Intraindividual Clinical Setting. J. Clin. Med. 2022, 11, 2857. [Google Scholar] [CrossRef]
- Avni, T.; Levcovich, A.; Ad-El, D.D.; Leibovici, L.; Paul, M. Prophylactic antibiotics for burns patients: Systematic review and me-ta-analysis. BMJ 2010, 340, c241. [Google Scholar] [CrossRef]
- Greenhalgh, D.G. Sepsis in the burn patient: A different problem than sepsis in the general population. Burn Trauma 2017, 5, 23. [Google Scholar] [CrossRef] [PubMed]
- Kuznetsova, T.A.; Andryukov, B.G.; Besednova, N.N. Modern Aspects of Burn Injury Immunopathogenesis and Prognostic Immunobiochemical Markers (Mini-Review). BioTech 2022, 11, 18. [Google Scholar] [CrossRef]
- Chen, P.; Stanojcic, M.; Jeschke, M.G. Septic predictor index: A novel platform to identify thermally injured patients susceptible to sepsis. Surgery 2018, 163, 409–414. [Google Scholar] [CrossRef]
- Finnerty, C.C.; Jeschke, M.G.; Qian, W.J.; Kaushal, A.; Xiao, W.; Liu, T.; Gritsenko, M.A.; Moore, R.J.; Camp, D.G., 2nd; Moldawer, L.L.; et al. Investigators of the Inflammation and the Host Response Glue Grant. Determination of burn patient outcome by large-scale quantitative discovery proteomics. Crit. Care Med. 2013, 41, 1421–1434. [Google Scholar] [CrossRef]
- Tran, N.K.; Albahra, S.; Pham, T.N.; Holmes, J.H., 4th; Greenhalgh, D.; Palmieri, T.L.; Wajda, J.; Rashidi, H.H. Novel application of an automated-machine learning development tool for predicting burn sepsis: Proof of concept. Sci. Rep. 2020, 10, 12354. [Google Scholar] [CrossRef]
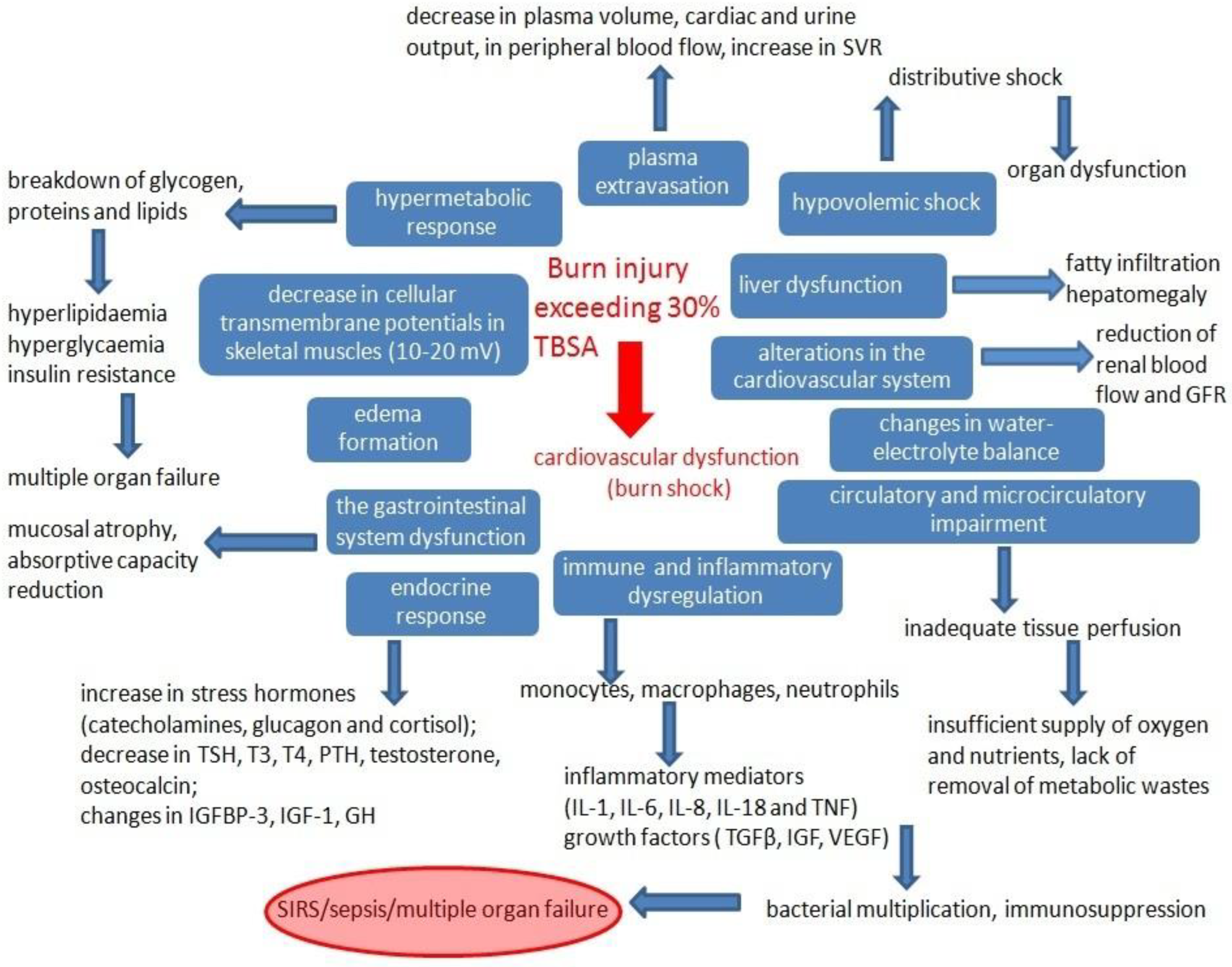
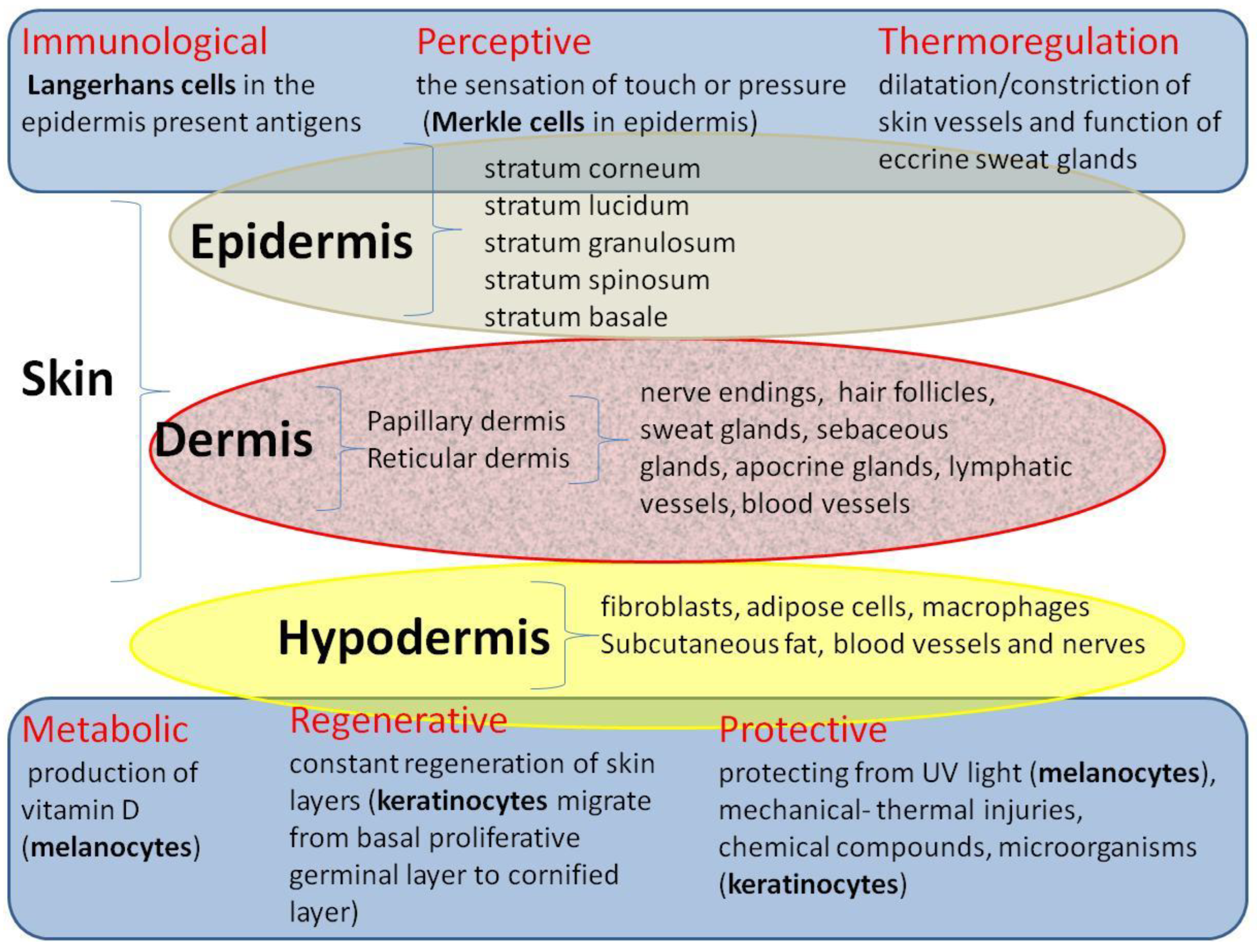
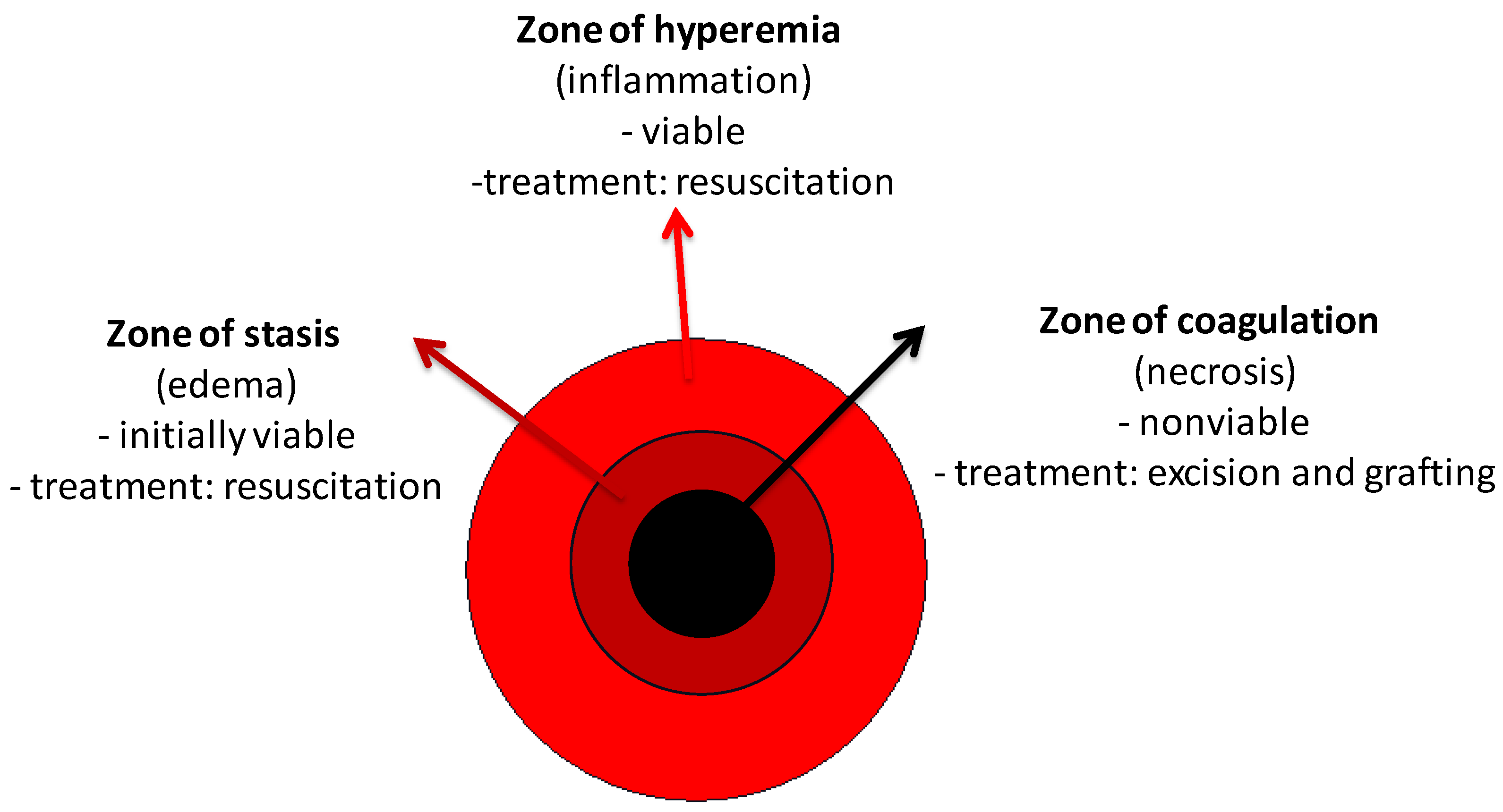
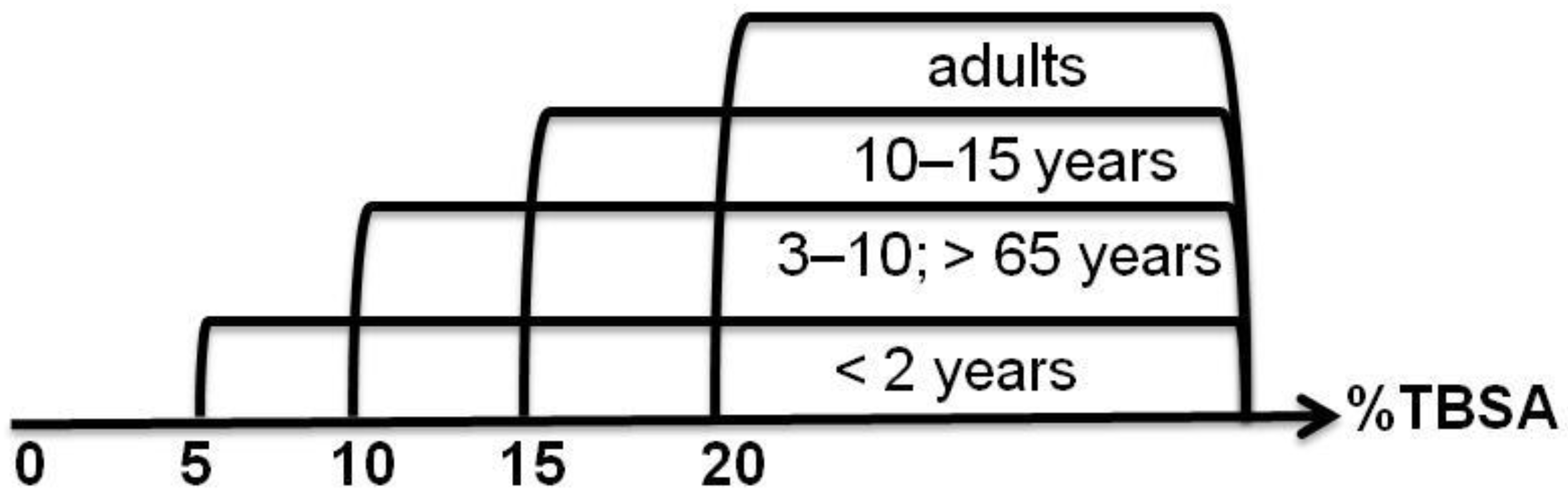

| Feature/ Degree | I Superficial | IIA Superficial Partial Thickness | IIB Deep Partial Thickness | III Full Thickness | IV Full Thickness and Tissues Beneath |
|---|---|---|---|---|---|
| depth of the wound | epidermis only | epidermis + papillary dermis (saving skin appendages) | epidermis + papillary dermis + reticular dermis | epidermis + dermis + hypodermis | epidermis + dermis + hypodermis + tissues beneath (e.g., fascia, bones, tendons) |
| counted in the calculation of TBSA | NO | YES | YES | YES | YES |
| common cause | sunburn | scald (short exposure) | scald, oil (medium time exposure) | scald (long exposure), flame, steam, oil, chemical compounds, electricity | scald (long exposure), flame, steam, oil, chemical compounds, electricity |
| characteristics | swelling, erythema, dryness, blanches with pressure | blisters filled with serum (as a result of the delamination of the epidermis from the basement membrane), blanches with pressure | blisters filled with serum, variety of colors: white/red/grey/yellow, usually excised and grafted | hard, waxy, leathery consistency, color: white/gray/charred, does not bleed when scratched, surgical treatment obligatory | hard, waxy, leathery consistency, color: white/gray/charred, does not bleed when scratched, reaching deeper structures, surgical treatment obligatory |
| sensation | painful | extremely painful | painful or decreased sensation | insensate | insensate |
| healing time | subsides after 72 h, skin peels off after 10 days, no scarring | up to 3 weeks, possible color match defect, hypertrophic scarring (low to moderate risk) | up to 9 weeks, hypertrophic scarring (moderate to high risk) | no healing capacity, without intervention it will separate over the weeks, hypertrophic scarring (high risk) | no healing capacity |
| measures | should heal by itself | should heal by itself | surgical intervention | surgical intervention | surgical intervention |
| Dressings | Category | Dressing Material/Antimicrobial Agents |
|---|---|---|
| Urgosorb® Silver, Urgotul SSD, Urgotul® Silver, Urgotul Duo Silver, Allevyn® Ag Gentle | Antimicrobial silver dressings, soft polymer | polyester mesh impregnated with hydrocolloid, petroleum jelly/silver sulfadiazine (SSD) |
| Bactigras® | Low-adherence wound-dressing pads | white soft paraffin tulle dressing/chlorhexidine acetate BP |
| Acticoat®, Atrauman® Ag | Low-adherence antimicrobial silver dressings | high-density polyethylene mesh of a rayon-polyester with nanocrystalline silver |
| Algosteril, Comfeel Alginate Dressing, Carrasorb H, Kaltostat, Cardinal Health™ Reinforced Gelling Fibers Algisite M | Fiber dressings, alginates | calcium alginate dressing derived from seaweed |
| Askina Calgitrol Ag®, Algicell® Ag, Tegaderm® Aliginate Ag, Sorbsan Silver Flat, Silvercel®, Melgisorb® Ag, Biatain Alginate Ag, Acticoat® Absorbent, Silvercel®, Algisite® Ag, Suprasorb® A + Ag, Actisorb®22 | Antimicrobial silver dressings, alginates | silver alginate wound dressing consisting of an absorbent foam sheet coated with an alginate matrix containing ionic silver and a superabsorbent starch co-polymer |
| OpSite, Tegaderm | Film/membrane dressings | adhesive-coated hydrophilic polyurethane film |
| Aquacel Ag® Physiotulle® Ag | Antimicrobial silver dressings, hydrofibers | sodium carboxymethylcellulose fibers containing 1.2% ionic silver |
| Comfeel, DuoDerm, Comfeel | Hydrocolloid dressings | hydrocolloids (gelatin, pectin and sodium carboxymethylcellulose) in an adhesive polymer matrix |
| Aqua clear, Nu-gel, IntraSite, Aqua-Gel®, Burnaid®, Water-Jel, Hydrosorb | Hydrogel dressings | amorphous hydrogels; high-water-content gels containing insoluble polymers (modified carboxymethylcellulose, hemicellulose, agar, glycerol, alginate, collagen, chitosan and pectin) or synthetic polymers such as polyvinyl alcohol or polyethylene glycol |
| Kerracel™ Gelling Fiber Dressing, AGILE™, AQUACEL® EXTRA™ Hydrofiber® Wound Dressing with Strengthening Fiber | Hydrofiber dressings | sodium carboxymethyl cellulose, strengthening cellulose fibers that form a gel when in contact with exudate |
| Mepitel® | Silicon-coated nylon dressings, low-adherent dressings, foams | safetac layer, made of soft silicone and porous, transparent and flexible polyamide mesh |
| Actisorb® Silver | Odor absorbent antimicrobial silver dressings | activated charcoal dressing with silver, enclosed in a non-adherent nylon sleeve |
| Biobrane, TransCyte, AWBAT-S | Biosynthetic skin substitute dressings | cyanuric chloride and dodecylamine covalently bond the collagen peptide to the silicone–nylon composite |
| Mepilex Border Ag, Mepilex Ag, Allevyn, Biatain | Foam dressings | polyurethane foam dressing with silver and activated carbon with a Safetac silicone contact layer |
| Suprathel | Synthetic antibacterial dressing, polylactic membranes (PLM) | copolymer of DL-lactide (>70%) and ε-caprolactone |
| Silverlon@ Wound Contact, Burn Contact Dressings | Dressing pads | Silverlon knitted nylon material plated with 99% elemental silver and 1% silver oxide |
| PermeaDerm, PermeaDerm B, PermeaDerm CW, PermeaDerm T, PermeaDerm AS | Adherent dressing, a synthetic matrix | a monofilament nylon knitted fabric, bonded to a silicone membrane (B, CW), coated with a mixture of porcine gelatin and a fraction of aloe vera or HMW-native collagen (T) or antiscar coating (AS) |
| PuraPly® Antimicrobial (PuraPly AM) Wound Matrix | Antimicrobial dressing, cross-linked extracellular matrix | a collagen sheet/0.1% polyhexmethylenebiguanide hydrochloride |
| Plant | Active Components | Nanocarrier Type | Nanocarrier Component | Refs. |
|---|---|---|---|---|
| Rheum officinale | emodin (1,3,8-trihydroxy-6-methyl-anthraquinone) | nanofibers | polyvinylpyrrolidone | [181,182] |
| Polygonum cuspidatum | emodin | nanofibers | ultra-fine cellulose acetate | [183] |
| Centella asiatica | asiaticoside | nanofibers | trisachharide triterpene and cellulose acetate | [184] |
| Curcuma longa | 1,7-bis(4-hydroxy-3-methoxyphenyl)-1,6-heptadiene-3,5-dione | nanofibers | cellulose acetate | [185] |
| cinnamon, lemongrass, peppermint | essential oils | nanofibers | cellulose-based nanofibers | [186] |
| Curcuma longa | curcumin | nanofibers | poly(ε-caprolactone)/gum tragacanth (PCL/GT) | [187] |
| Ananas comosus | bromelain | nanofibers | chitosan | [188] |
| Bixa orellana | bixin | nanofibers | polycaprolactone (PCL) | [189] |
| Medicago sativa | genistein | nanofibers | polycaprolactone (PCL) | [190] |
| Thymus vulgaris | thymol and carvacrol (phenolic monoterpenes) | nanofibers | poly(ε-caprolactone) (PCL) and poly(lactic acid) (PLA) | [191] |
| Tecomella undulate | tecomin | nanofibers | polycaprolactone (PCL) and polyvinyl pyrrolidone (PVP) | [192] |
| Syzygium aromaticum | eugenol | magnetic nanospheres | polylactic acid and chitosan | [193] |
| Drosera binata | naphthoquinones (plumbagin) | nanoparticles | silver | [161] |
| Centella Asiatica | pentacyclic triterpenes (asiatic acid) | hydrogel, nanoparticles | gelatin, hyaluronic acid, chondroitin sulfate, zinc oxide and copper oxide | [194] |
| Scutellaria baicalensis | baicalin | nanohydrogel | cholesterol | [195] |
| Azadirachta indica | neem oil | liposomes hyalurosomes | argan | [196] |
| Mangifera indica | mangiferin | transferosomes | propylene glycol and glycerol | [197] |
| Product | Company | Components | Type |
|---|---|---|---|
| CeladermTM | Celadon Science LLC, Brookline, MA, USA | keratinocyte sheets from foreskin | dermal/epidermal, allogeneic |
| PoliActive® | HC Implants BV, Leiden, Netherlands | PEO (polyethylene oxide terephthalate) and PBT (polybuthylene terephthalate) in a porous matrix with cultured keratinocytes and fibroblasts | epidermal, biosynthetic, autogenous |
| Autoderm (Autologous Inferior Dermal Sling), TransDerm, Lyphoderm, Cryoceal | XCELLentis NV, Gent, Belgium | cultured keratinocytes | epidermal, autogenous |
| Laserskin, VivoDerm™ | Fidia Advanced Biopolymers, Aban Terme, Italy; ER Sąuibb and Sons Inc., Princeton, NJ, USA | a patient’s skin section used for in vitro culture of keratinocytes, which are seeded onto a membrane of hardened, esterified hyaluronic acid perforated with a laser | epidermal, autogenous |
| Epicel® | Genzyme Biosurgery Cambridge, MA, USA | keratinocyte-based cultured epidermal autograft | epidermal, autogenous |
| EpiDex® | Modex Therapeutiques, Lausanne, Switzerland | ||
| EPIBASE | Laboratoires Genevrier, Nice, France | ||
| Permacol®, Strattice®, Xenoderm® | Strattice™ Reconstructive Tissue Matrix (LifeCell Corporation, Branchburg, NJ, USA), MBP (Medical Biomaterial Products, Germany), Tissue Science Laboratories PLC, Aldershot, UK | a porcine-derived collagen matrix and its constituent elastin fibers | dermal, acellular, xenogeneic |
| Geistlich Derma-Gide™ | Geistlich Pharma North America Inc., Princeton, NJ, USA | a porcine, porous, resorbable, 3D matrix | dermal, acellular, xenogeneic |
| Helicoll™ | EnColl Corp., Fremont, CA, USA | a collagen matrix derived from bovine sources | dermal, acellular, xenogeneic |
| MatriStem, MicroMatrix® | ACell Inc., Columbia, MD, USA | matrices derived from the porcine urinary bladder matrix | dermal, acellular, xenogeneic |
| E-Z-Derm™ | Brennen Medical Inc., Saint Paul, Minnesota, USA | a collagen matrix made of porcine collagen cross-linked with aldehyde | dermal, acellular, xenogeneic |
| Architect® stabilized Collagen matrix | Harbor MedTech, Inc., Irvine, CA, USA. | decellularized equine pericardial tissue | dermal, acellular, xenogeneic |
| PriMatrix® Dermal Repair Scaffold | TEI Biosciences Inc, Boston, MA, USA | acellular dermal tissue matrix from fetal bovine dermis, rich in type II collagen | dermal, acellular, xenogeneic |
| Cytal® wound matrix | Acell, Inc., Columbia, MD, USA | porcine urinary bladder matrix with an intact epithelial basement membrane | dermal, acellular, xenogeneic |
| Matriderm® | Skin and Health Care AG, Billerbeck, Germany | a multiporous membrane of bovine origin, composed of collagen (types I, III and V) and a hydrolysate of elastin-alpha, treated with gamma rays | dermal, xenogeneic |
| Endoform® Natural Dermal Template | Hollister Wound Care, Libertyville, IL, USA | decellularized tissue extracellular matrix derived from ovine forestomach tissue (e.g., collagen I, III, IV, fibronectin, laminin, elastin, hyaluronic acid, heparin sulfate, GAGs, growth factors and chemokines) | dermal, acellular, xenogeneic |
| Oasis® Wound Matrix | Healthpoint Biotherapeutics, USA; Cook Biotech, Inc., West Lafayette, IN, USA | submucosa of the small intestine of pigs | dermal/epidermal xenogeneic, acellular |
| Myriad Matrix® | Aroa Biosurgery, Auckland, New Zealand | a collagen matrix derived from ovine forestomach | dermal, acellular, xenogeneic |
| Terudermis® | Olympus Terumo Biomaterials Corp., Tokyo, Japan | obtained from heat-denatured bovine collagen, then coated with a silicone film | dermal, xenogeneic |
| Hyalomatrix® Hyalograft 3D®, Hyalomatrix PA® | Anika Therapeutics, former Fidia Advanced Biopolymers, Padua, Italy | a bilayer, hyaluronic acid esterified with a benzyl alcohol (Hyaff) matrix or scaffold with an outer silicone membrane | dermal, synthetic |
| Pelnac Standard Type | Medical Materials Center, Kyoto, Japan | porcine collagen sponge covered with a silicone film | dermal, autologous |
| STRATAGRAFT | Stratatech Corporation, Madison, WI, USA | allogeneic cultured keratinocytes and dermal fibroblasts in murine collagen-dsat | dermal/epidermal allogeneic |
| Transcyte® | Smith & Nephew, Inc., Largo, Florida, USA | neonatal allogeneic fibroblasts (removed by freezing after producing extracellular matrix and growth factors) cultured and multiplied on nylon fibers and placed on a silicone foil | dermal, allogeneic |
| Recell | Avita, Northridge, CA, USA | cell suspension of keratinocytes, fibroblasts, Langerhans cells and melanocytes | dermal/epidermal, autologous |
| OrCei™ | Ortec International, Inc., New York, NY, USA | allogeneic fibroblasts and keratinocytes expanded in vitro and seeded on both sides of a bilayer bovine collagen matrix | dermal/epidermal allogeneic |
| Apligraf | Organogenesis Inc., Canton, Massachusetts, CA, USA; Novartis Pharmaceuticals Corp. East Hanover, NJ, USA | a bilayer composed of a bovine type I collagen lattice with a dermal layer of human fibroblasts and a layer formed from human keratinocytes from cultured cells, from newborn foreskins | dermal/epidermal, cellular, allogeneic and xenogeneic |
| OrCel | Ortec International, Inc., New York, NY, USA; Forticell Bioscience Inc., NY, USA, | composite allograft synthesized by culturing allogeneic neonatal keratinocytes and fibroblasts in a type I bovine collagen porous sponge with nonporous sides | dermal/epidermal allogeneic |
| Tissue Tech Autograft System | Fidia Advanced Biopolymers, Aba-no Terre, Italy | membrane made of hyaluronic acid, enriched with cultured autologous fibroblasts and keratinocytes (Hyalograft 3D—a substitute for the dermis, Laserskin—a substitute for the epidermis) | dermal/epidermal autologous |
| Dermal Regenerative Template DRT, Integra®, Integra Bilayer Matrix Wound Dressing, Integra Omnigraft Regeneration Template | Integra LifeSciences Plainsboro, NJ, USA | a bilayer matrix comprising a dermal layer composed of an acellular matrix consisting of cross-linked bovine collagen and chondroitin-6-sulfate, a type of glycosaminoglycan and an overlying silicone layer acting as the epidermis | dermal/epidermal, natural and synthetic, acellular |
| Dermagraft® | Advanced BioHealing, La Jolla, CA, USA; Organogenesis, Canton, MA, USA | a bio-absorbable polyglactin (vicryl) mesh seeded with cryo-preserved neonatal allogeneic foreskin fibroblasts | dermal, cellular, natural and synthetic, biodegradable, allogeneic |
| Biobrane® | Dow Hickam Pharmaceuticals Inc., Sugar Land, TX, USA; Smith & Nephew, London, UK; Mylan Bertek Pharmaceuticals, Durham, North Carolina, USA | biocomposite dressing; nylon mesh with the addition of porcine collagen and a layer of silicone | dermal/epidermal, acellular |
| TRANSCYTE® Dermagraft-TC ® | Advanced Tissue Sciences Inc. (ATS, La Jolla, CA, USA) | a semi-permeable silicone membrane and an extracellular matrix of newborn human dermal fibroblasts cultured on a porcine collagen-coated nylon mesh | dermal, allogeneic |
| Suprathel® | Polymedics Innovations GmbH, Denkendorf, Germany | polylactide copolymer, trimethylene carbonate and ε-caprolactone (Lacto-capromer) | synthetic, biodegradable |
| NovoSorb® BTM (Biodegradable Temporizing Matrix) | PolyNovo Biomaterials Pty Ltd., Melbourne, Australia | polyurethane bilayer dermal template consisting of a temporary sealing membrane bonded to a 2 mm bioabsorbable open cell matrix | synthetic, biodegradable |
| RestrataTM | Acera Surgical, Inc., St. Louis, MO, USA | electrospun nanofiber matrix | dermal, acellular, synthetic |
| Alloderm® | LifeCell Corporation, Bridgewater, NJ, USA | human cadaver skin that has been chemically treated to remove all cellular material in the dermis | dermal/epidermal, acellular, allogeneic |
| GraftJacket®, GraftJacket RTM | KCI, San Antonio, TX, USA; Wright Medical Group N.V., Memphis, TN, USA | produced from allograft skin | dermal, acellular, allogeneic |
| GLYADERM ® | Euro Skin Bank in the Netherlands | glycerol-preserved acellular dermis | dermal, acellular, allogeneic |
| Karoderm®, SureDerm® | Karocell Tissue Engineering AB, Stockholm, Sweden, Hans Biomed Corp., Seoul, Korea, Wright Medical Technology Inc., Arlington, TN, USA | human skin matrix with preserved basement membrane | dermal, acellular, allogeneic |
| Matrix HD Allograft | RTI Surgical, Alachua, FL, USA | human allograft sterilized using the Tutoplast® Tissue Sterilization process | dermal, acellular, allogeneic |
| Alloskin, AlloSkin RT™, Alloskin™ AC | AlloSource, Centennial, CO, USA | a fresh-frozen, a fresh irradiated (not frozen), or an acellular meshed dermis-only human skin allograft matrix made from cadaveric tissue, extracellular matrix proteins, glycosaminoglycans and cytokines | dermal, acellular, allogeneic |
| TheraSkin® | LifeNet Health, Virginia Beach, VA, USA | biologically active, cryopreserved human skin | dermal/epidermal, cellular, allogeneic |
| SkinTE | PolarityTE, Salt Lake City, UT, USA | donated human dermis or autologous skin sample | dermal/epidermal, cellular |
| DermACELL | LifeNet Health, Virginia Beach, VA, USA | human-derived matrices, sterilized and decellularized to remove immunogenic cellular material | dermal, acellular, allogeneic |
| AlloPatch®, AlloPatch Pliable | Musculoskeletal Transplant Foundation Sports Medicine, Edison, NJ, USA | an open-structure human reticular dermal matrix | dermal, acellular, allogeneic |
| Dermapure® | Tissue Regenix Group, San Antonio, TX, USA | decellularized human dermis | dermal, acellular, allogeneic |
| Grafix, GrafixPL Prime | Osiris Therapeutics, Inc., Columbia, MD, USA | human amnion chorion membrane | dermal, cellular, allogeneic |
| WoundEx® Membrane, WoundEx Flow | Skye Biologics, Inc., El Segundo, CA, USA | a dehydrated amniotic membrane, or a flowable human placental connective tissue matrix | dermal, acellular, allogeneic |
| Xwrap® Amniotic Membrane Derived Allograft | Applied Biologics, Scottsdale, AZ, USA | a chorion-free amniotic membrane wrap, cover, or patch. | dermal, acellular, allogeneic |
| Amnioexcel® | Integra LifeSciences Corp. acquired Derma Sciences, Plainsboro, NJ, USA | a dehydrated human amnion-derived tissue allograft with an intact extracellular matrix | dermal, acellular, allogeneic |
| Amniomatrix Human Amniotic Suspension Allograft, BioDFactor Viable Tissue Matrix, Biodfence, Integra BioFix Amniotic Membrane Allograft, Integra BioFix Flow Placental Tissue Matrix Allograft | Integra LifeSciences Corp., Plainsboro, NJ, USA; Integra LifeSciences, originally BioD, LLC, Plainsboro, NJ, USA | matrices derived from human placental membrane | dermal, acellular, allogeneic |
| EpiFix | MiMedx Group Inc., Marietta, GA | the human placental membranes composed of amnion and chorion, rich in extracellular matrix proteins, growth factors and cytokines | dermal, acellular, allogeneic |
| Affinity® Human Amniotic Allograft | Organogenesis, Inc., Canton, MA, USA | fresh amniotic membrane | dermal, cellular, allogeneic |
| AmnioRepair | Zimmer Biomet, Warsaw, Poland | a freeze-dried epidermal and dermal replacement with epithelial and stromal sides as well as the outer basement membrane | dermal/epidermal, acellular |
| Revita® | StimLabs, LLC, Roswell, GA, USA | intact human placental membrane | dermal, acellular, allogeneic |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Radzikowska-Büchner, E.; Łopuszyńska, I.; Flieger, W.; Tobiasz, M.; Maciejewski, R.; Flieger, J. An Overview of Recent Developments in the Management of Burn Injuries. Int. J. Mol. Sci. 2023, 24, 16357. https://doi.org/10.3390/ijms242216357
Radzikowska-Büchner E, Łopuszyńska I, Flieger W, Tobiasz M, Maciejewski R, Flieger J. An Overview of Recent Developments in the Management of Burn Injuries. International Journal of Molecular Sciences. 2023; 24(22):16357. https://doi.org/10.3390/ijms242216357
Chicago/Turabian StyleRadzikowska-Büchner, Elżbieta, Inga Łopuszyńska, Wojciech Flieger, Michał Tobiasz, Ryszard Maciejewski, and Jolanta Flieger. 2023. "An Overview of Recent Developments in the Management of Burn Injuries" International Journal of Molecular Sciences 24, no. 22: 16357. https://doi.org/10.3390/ijms242216357
APA StyleRadzikowska-Büchner, E., Łopuszyńska, I., Flieger, W., Tobiasz, M., Maciejewski, R., & Flieger, J. (2023). An Overview of Recent Developments in the Management of Burn Injuries. International Journal of Molecular Sciences, 24(22), 16357. https://doi.org/10.3390/ijms242216357






