Straightforward and Efficient Protocol for the Synthesis of Pyrazolo [4,3-b]pyridines and Indazoles
Abstract
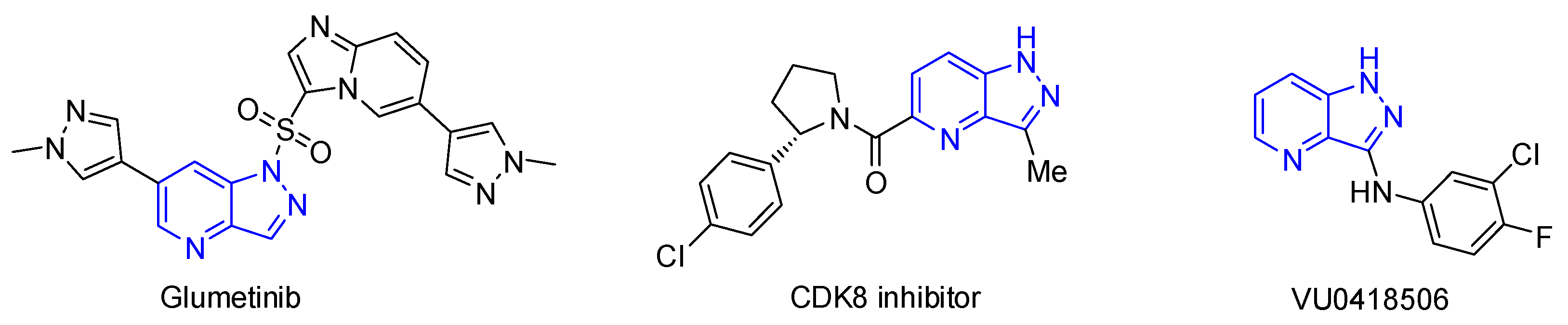
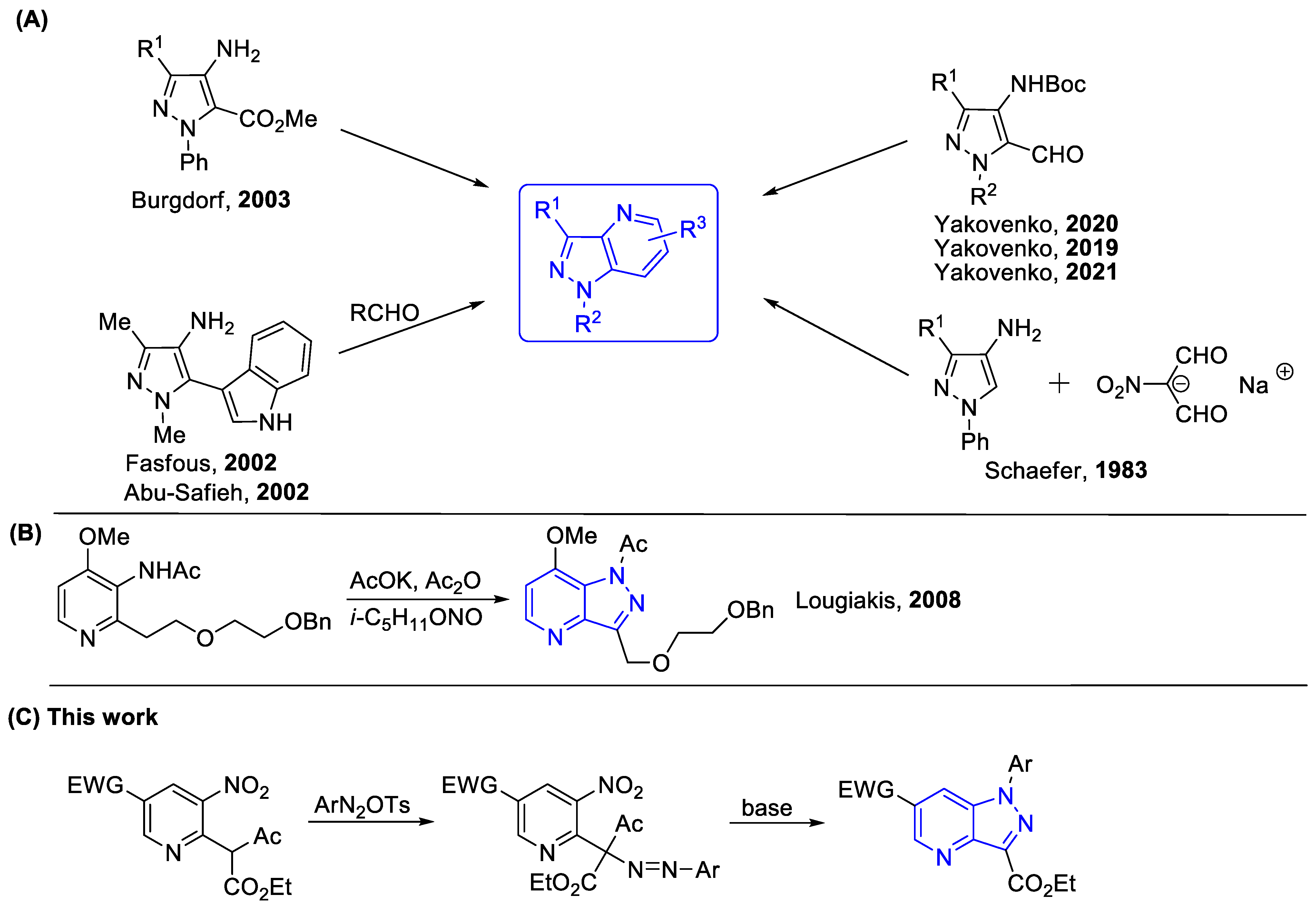
1. Introduction
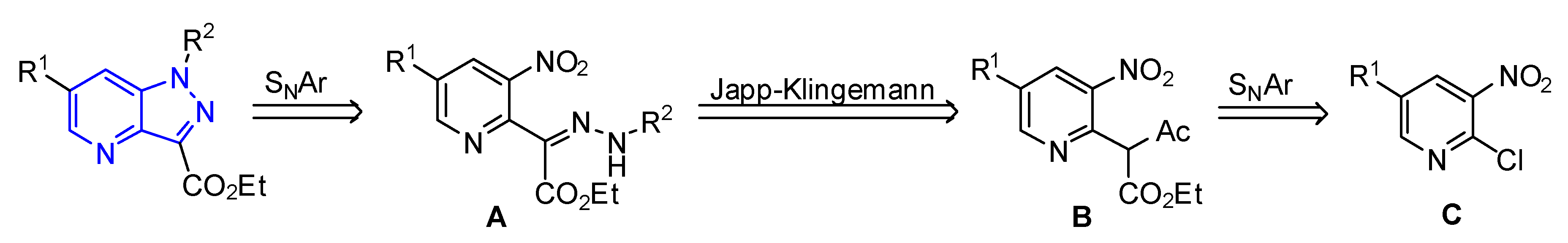
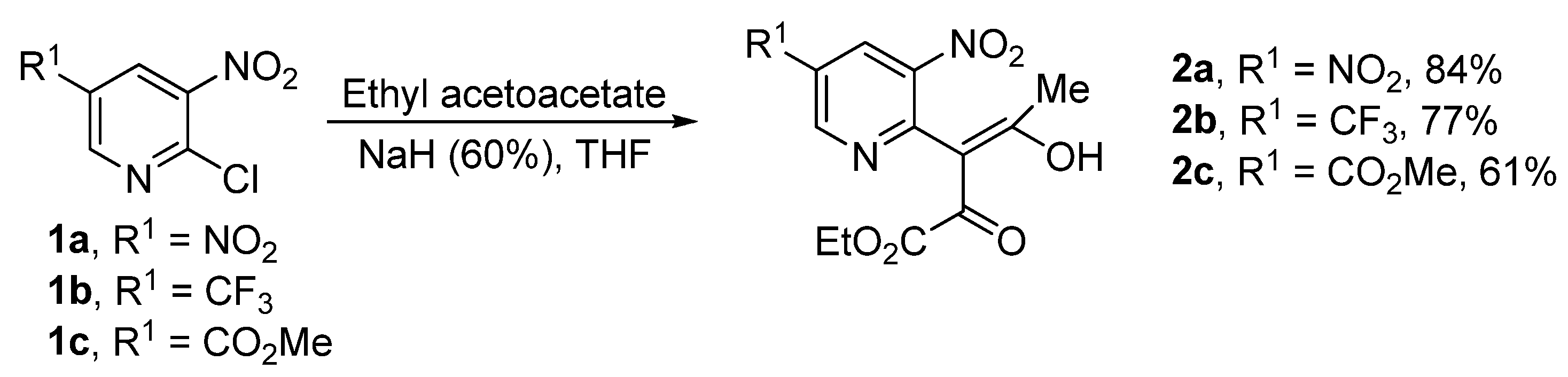
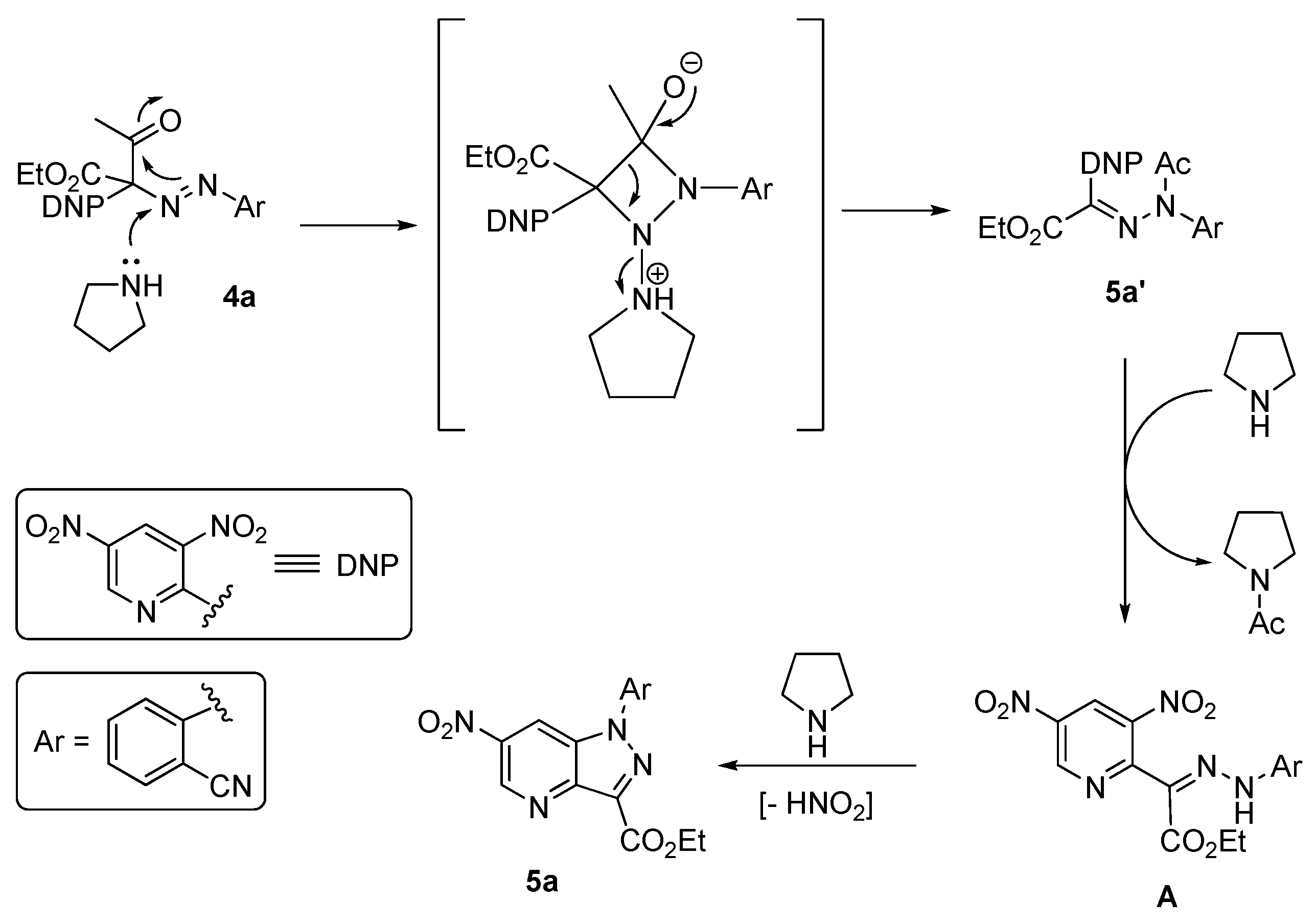
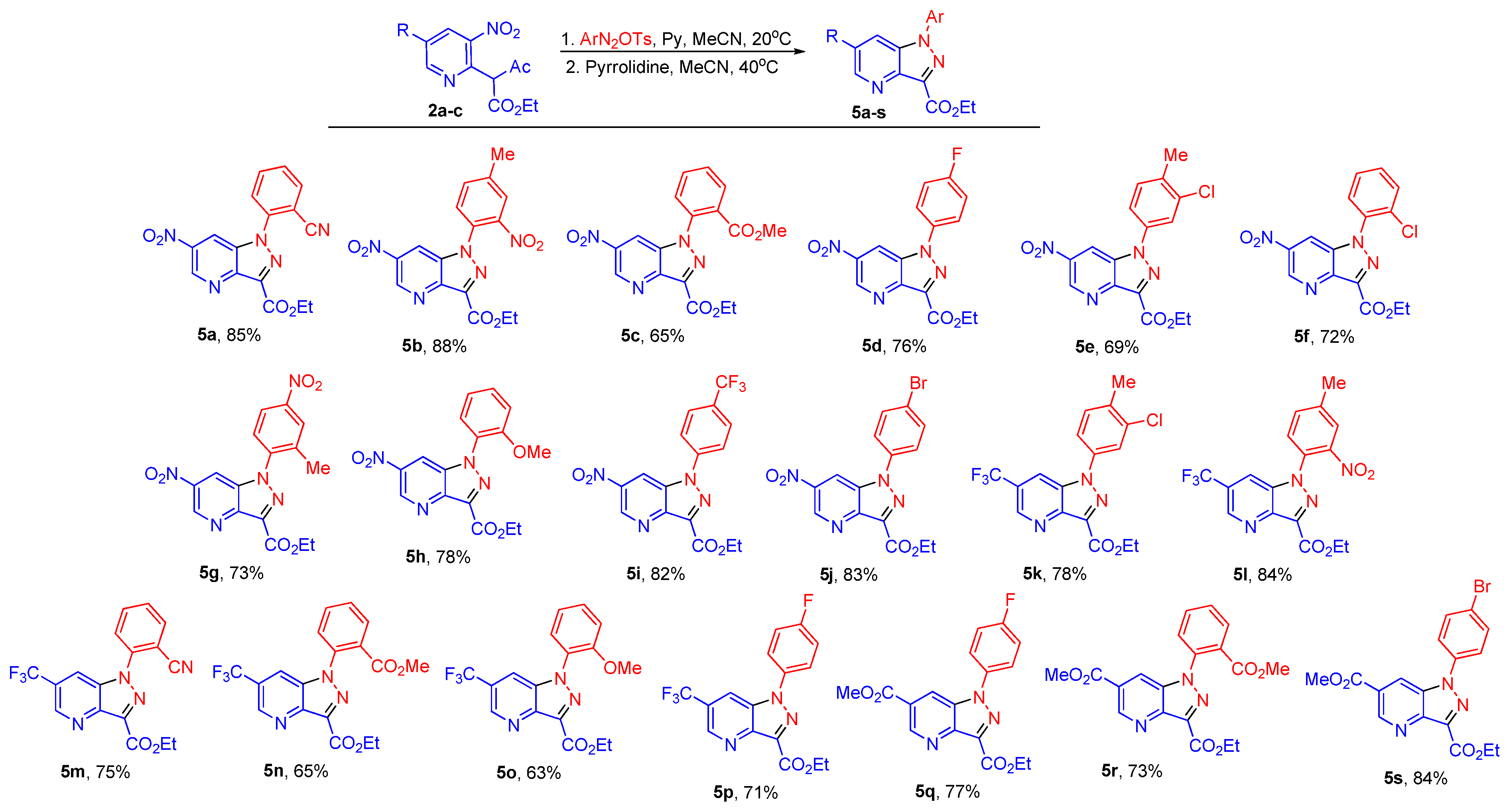
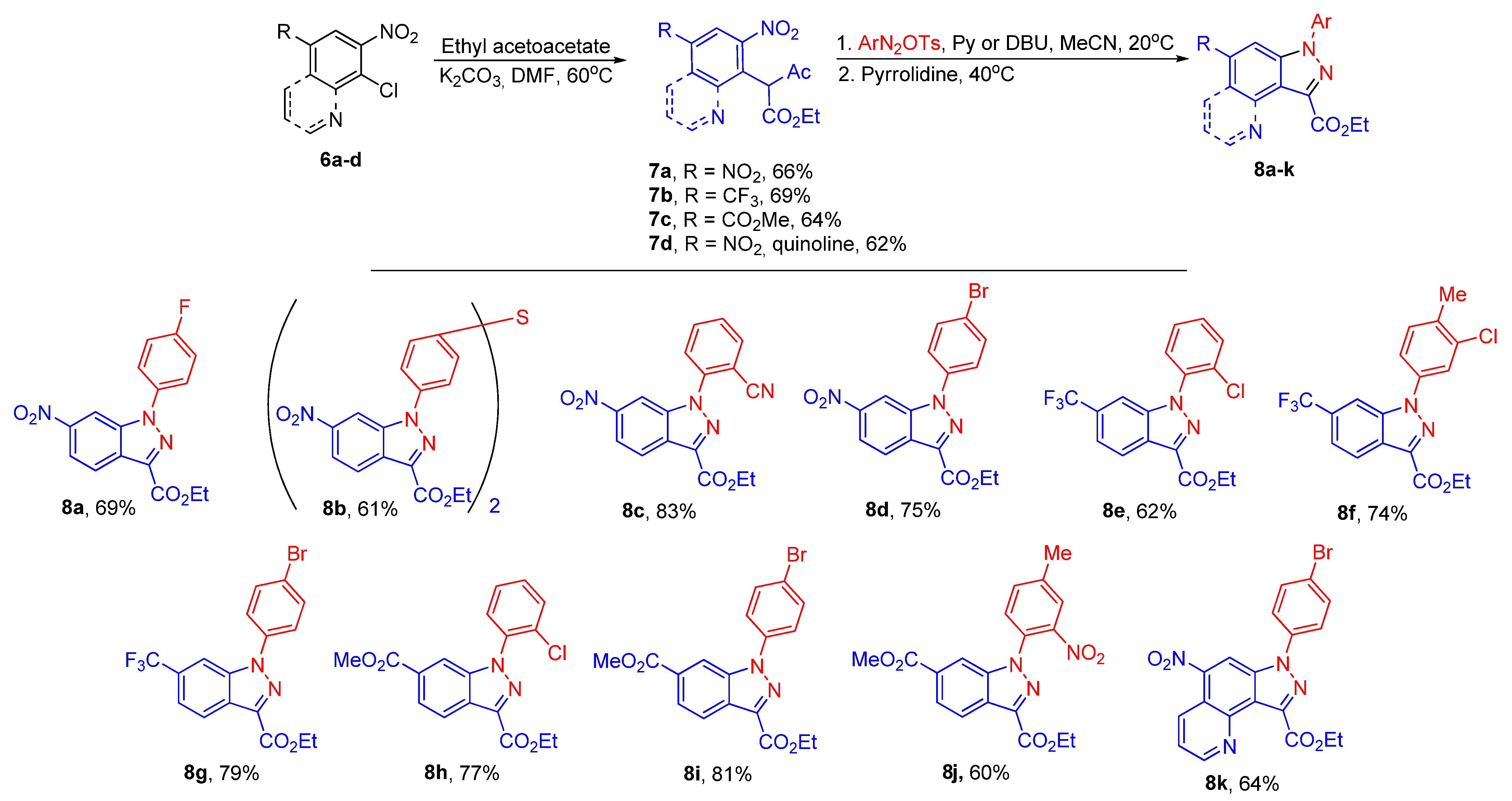
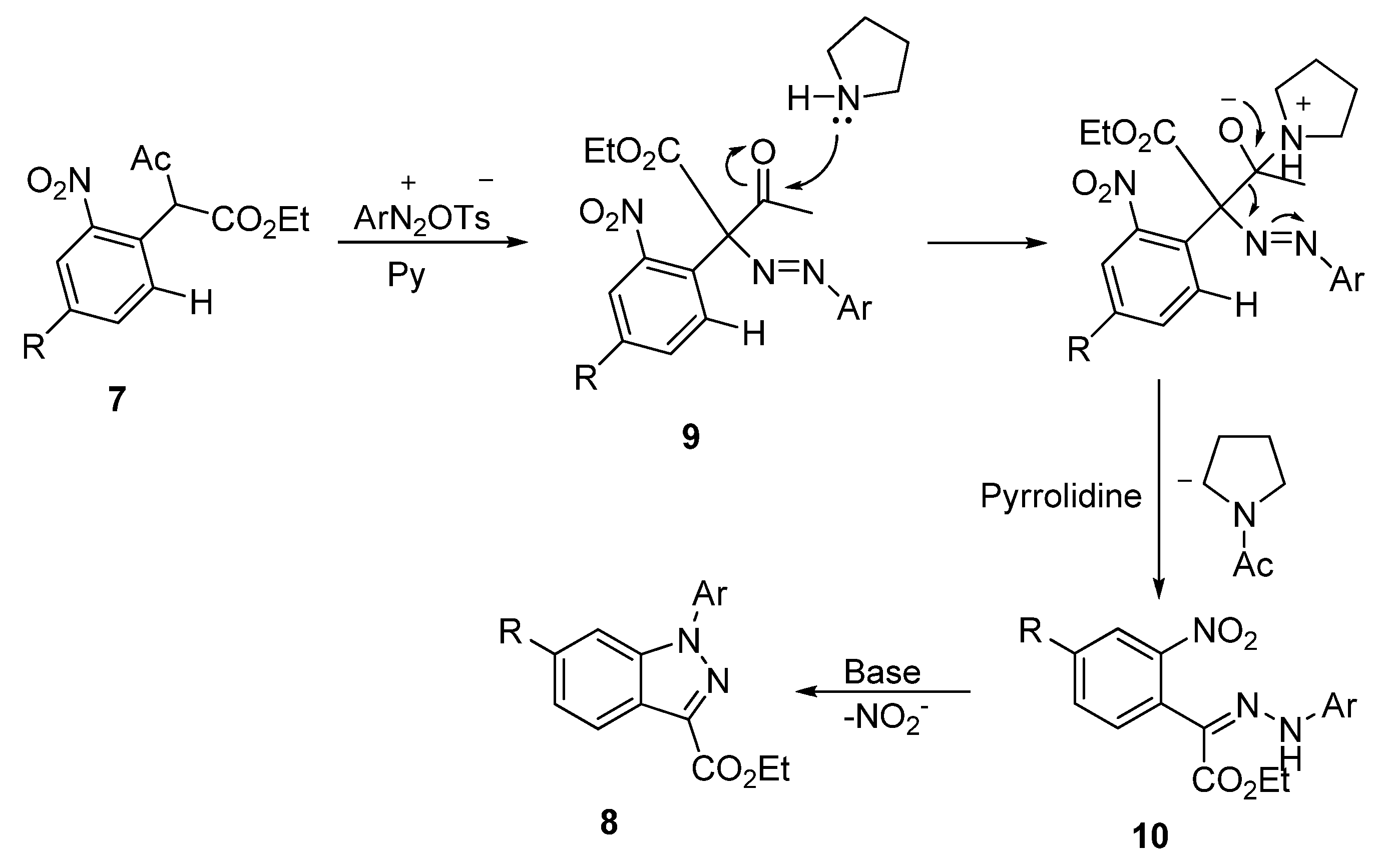
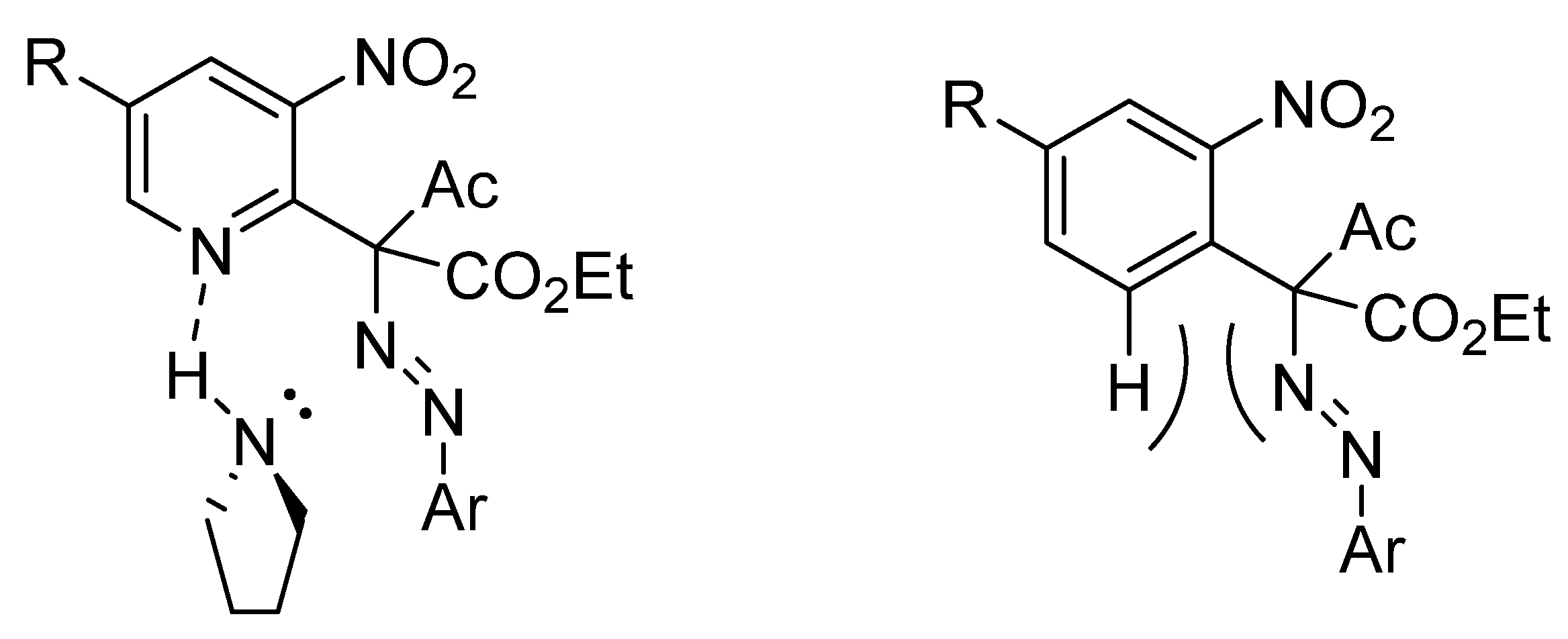
2. Results and Discussion
3. Materials and Methods
3.1. General Information
3.2. General Procedure for the Synthesis of Compounds 2a–c
3.3. General Procedure for the Synthesis of Compounds 7a–d
3.4. General Procedure for the Synthesis of Aryldiazonium Tosylates 3 [26]
3.5. Synthesis of the Intermediates 4a, 5a′ and 5q′
3.6. General Procedure for the Synthesis of Pyrazolo [4,3-b]pyridines 5 and Indazoles 8
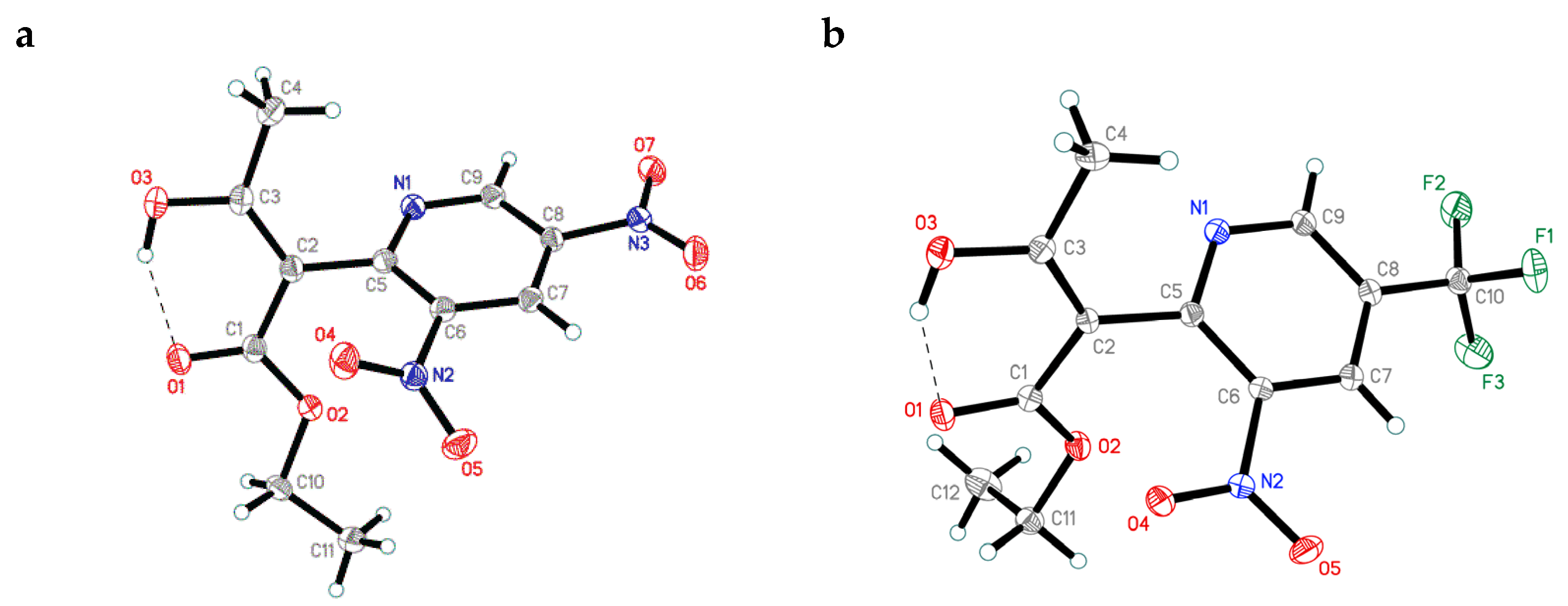
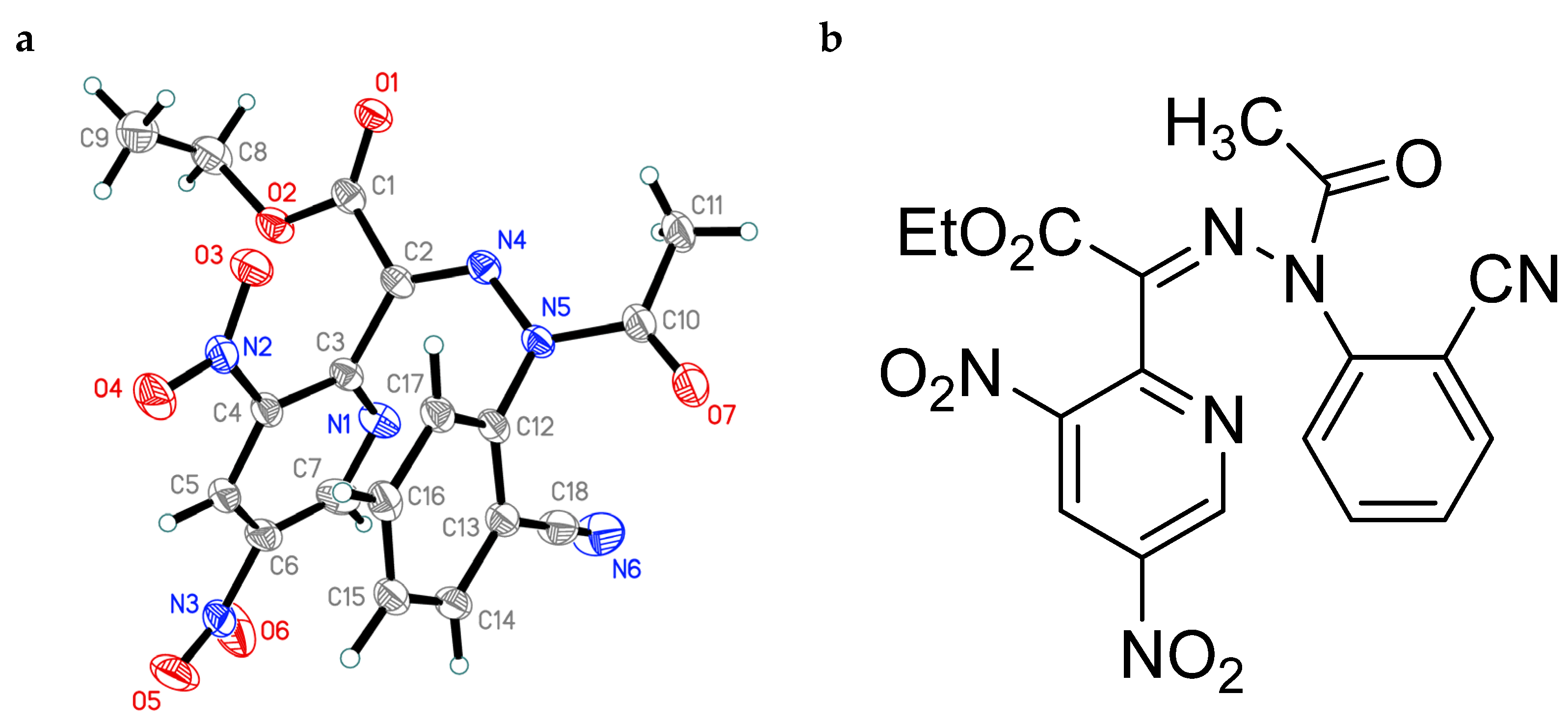
3.7. X-ray Crystallographic Data and Refinement Details
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tang, J.; Yang, H.; Cui, Y.; Cheng, G. Nitrogen-rich tricyclic-based energetic materials. Mater. Chem. Front. 2021, 5, 7108–7118. [Google Scholar] [CrossRef]
- Zlotin, S.G.; Churakov, A.M.; Egorov, M.P.; Fershtat, L.L.; Klenov, M.S.; Kuchurov, I.V.; Makhova, N.N.; Smirnov, G.A.; Tomilov, Y.V.; Tartakovsky, V.A. Advanced energetic materials: Novel strategies and versatile applications. Mendeleev Commun. 2021, 31, 731–749. [Google Scholar] [CrossRef]
- Bystrov, D.M.; Pivkina, A.N.; Fershtat, L.L. An Alliance of polynitrogen heterocycles: Novel energetic tetrazinedioxide-hydroxytetrazole-based materials. Molecules 2022, 27, 5891. [Google Scholar] [CrossRef] [PubMed]
- Dalinger, I.L.; Shkineva, T.K.; Vatsadze, I.A.; Kormanov, A.V.; Kozeev, A.M.; Suponitsky, K.Y.; Pivkina, A.N.; Sheremetev, A.B. Novel energetic CNO oxidizer: Pernitro-substituted pyrazolyl-furazan framework. FirePhysChem 2021, 1, 83–89. [Google Scholar] [CrossRef]
- Parker, W.B. Enzymology of purine and pyrimidine antimetabolites used in the treatment of cancer. Chem. Rev. 2009, 109, 2880–2893. [Google Scholar] [CrossRef] [PubMed]
- Rios, M.-C.; Bravo, N.-F.; Sanchez, C.-C.; Portilla, J. Chemosensors based on N-heterocyclic dyes: Advances in sensing highly toxic ions such as CN- and Hg+. RSC. Adv. 2021, 11, 34206–34234. [Google Scholar] [CrossRef] [PubMed]
- Tucker, T.J.; Sisko, J.T.; Tynebor, R.M.; William, T.M.; Felock, P.J.; Flynn, J.A.; Lai, M.-T.; Liang, Y.; McGaughey, G.; Liu, M.; et al. Discovery of 3-{5-[(6-Amino-1H-pyrazolo[3,4-b]pyridine-3-yl)methoxy]-2-chlorophenoxy}-5-chlorobenzonitrile (MK-4965): A Potent, Orally Bioavailable HIV-1 Non-Nucleoside Reverse Transcriptase Inhibitor with Improved Potency against Key Mutant Viruses. J. Med. Chem. 2008, 51, 6503–6511. [Google Scholar] [CrossRef]
- Herdemann, M.; Heit, I.; Bosch, F.-U.; Quintini, G.; Scheipers, C.; Weber, A. Identification of potent ITK inhibitors through focused compound library design including structural information. Bioorg. Med. Chem. Lett. 2010, 20, 6998–7003. [Google Scholar] [CrossRef]
- Pryde, D.C.; Marron, B.E.; West, C.W.; Reister, S.; Amato, G.; Yoger, K.; Antonio, B.; Padilla, K.; Cox, P.J.; Turner, J.; et al. Discovery of a series of indazole TRPA1 antagonists. ACS Med. Chem. Lett. 2017, 8, 666–671. [Google Scholar] [CrossRef]
- Wilcoxen, K.; Huang, C.Q.; McCarthy, J.R.; Grigoriadis, D.E.; Chen, C. Synthesis of 3-phenylpyrazolo[4,3-b]pyridines via a convenient synthesis of 4-amino-3-arylpyrazoles and SAR of corticotropin-Releasing factor receptor type-1 antagonists. Bioorg. Med. Chem. Lett. 2003, 13, 3367–3370. [Google Scholar] [CrossRef]
- Ai, J.; Chen, Y.; Peng, X.; Ji, Y.; Xi, Y.; Shen, Y.; Yang, X.; Su, Y.; Sun, Y.; Gao, Y.; et al. Preclinical Evaluation of SCC244 (Glumetinib), a Novel, Potent, and Highly Selective Inhibitor of c-Met in MET-dependent Cancer Models. Mol. Cancer Ther. 2018, 17, 751–762. [Google Scholar] [CrossRef] [PubMed]
- Czodrowski, P.; Mallinger, A.; Wienke, D.; Esda, C.; Pöschke, O.; Busch, M.; Rohdich, F.; Eccles, S.A.; Ortiz-Ruiz, M.-J.; Schneider, R.; et al. Structure-Based Optimization of Potent, Selective, and Orally Bioavailable CDK8 Inhibitors Discovered by High-Throughput Screening. J. Med. Chem. 2016, 59, 9337–9349. [Google Scholar] [CrossRef] [PubMed]
- Engers, D.W.; Blobaum, A.L.; Gogliotti, R.D.; Cheung, Y.-Y.; Salovich, J.M.; Garcia-Barrantes, P.M.; Daniels, J.S.; Morrison, R.; Jones, C.K.; Soars, M.G.; et al. Discovery, Synthesis, and Preclinical Characterization of N-(3-Chloro-4-fluorophenyl)-1H-pyrazolo[4,3-b]pyridin-3-amine (VU0418506), a Novel Positive Allosteric Modulator of the Metabotropic Glutamate Receptor 4 (mGlu4). ACS Chem. Neurosci. 2016, 7, 1192–1200. [Google Scholar] [CrossRef] [PubMed]
- Yakovenko, G.G.; Lukianov, O.A.; Yagodkina-Yakovenko, M.S.; Bol’but, A.V.; Vovk, M.V. Synthesis of 5-amino-1H-pyrazolo[4,3-b]pyridine derivatives and annulation of imidazole and pyrimidine rings thereto. Chem. Heterocycl. Compd. 2020, 56, 347–354. [Google Scholar] [CrossRef]
- Yakovenko, G.G.; Lukianov, O.A.; Bol’but, A.V.; Vovk, M.V. N-Boc-4-aminopyrazole-5-carbaldehydes in Friendländer synthesis of pyrazolo[4,3-b]pyridines. Chem. Heterocycl. Compd. 2019, 55, 379–385. [Google Scholar] [CrossRef]
- Yakovenko, G.G.; Saliyeva, L.N.; Rusanov, E.B.; Donchak, I.S.; Vovk, M.V. Synthesis of methyl(ethyl) pyrazolo[4,3-b]pyridine-6-carboxylates and their conversion to tert-butyl 4,5,6,7-tetrahydropyrazolo-[4,3-b]pyridine-6-carboxylates. Chem. Heterocycl. Compd. 2021, 57, 1137–1145. [Google Scholar] [CrossRef]
- Barreiro, E.J.; Camara, C.A.; Verli, H.; Brazil-Más, L.; Castro, N.G.; Cintra, W.M.; Aracava, Y.; Rodrigues, C.R.; Fraga, C.A.M. Design, Synthesis, and Pharmacological Profile of Novel Fused Pyrazolo[4,3-d]pyridine and Pyrazolo[3,4-b][1,8]naphthyridine Isosteres: A New Class of Potent and Selective Acetylcholinesterase Inhibitors. J. Med. Chem. 2003, 46, 1144–1152. [Google Scholar] [CrossRef]
- Fasfous, I.I.; El-Abadelah, M.M.; Sabri, S.S. Pictet-Spengler synthesis of pyrazole-fused β-carbolines. J. Heterocycl. Chem. 2002, 39, 225–227. [Google Scholar] [CrossRef]
- Abu-Safieh, K.A.; El-Abadelah, M.M.; Sabri, S.S.; Voelter, W.; Mössmer, C.M.; Stroebele, M. Bishler-Napieralski synthesis of some new pyrazole-fused β-carbolines. Z. Naturforsch. B 2002, 57B, 1327–1332. [Google Scholar] [CrossRef]
- Schaefer, H.; Gewald, K.; Schmidt, M. 5,6-Condensed 3-nitropyridines from heterocyclic amines and nitromalonaldehyde. Chem. Heterocycl. Compd. 1983, 19, 1163–1166. [Google Scholar] [CrossRef]
- Yakovenko, G.G.; Saliyeva, L.N.; Vovk, M.V. 5-Aminopyrazoles as effective reagents in the synthesis of pyrazolo-annulated pyridines. Chem. Heterocycl. Compd. 2022, 58, 159–177. [Google Scholar] [CrossRef]
- Lougiakis, N.; Marakos, P.; Poul, N.; Balzarini, J. Synthesis and antiviral activity evaluation of some novel acyclic C-nucleosides. Chem. Pharm. Bull. 2008, 56, 775–780. [Google Scholar] [CrossRef] [PubMed]
- Bastrakov, M.A.; Nikol’skiy, V.V.; Starosotnikov, A.M.; Fedyanin, I.V.; Shevelev, S.A.; Knyazev, D.A. Reactions of 3-R-5-nitropyridines with nucleophiles: Nucleophilic substitution vs conjugate addition. Tetrahedron 2019, 75, 130659. [Google Scholar] [CrossRef]
- Nishiwaki, N.; Nishida, D.; Ohnishi, T.; Hidaka, F.; Shimizu, S.; Tamura, M.; Hori, K.; Tohda, Y.; Ariga, M. Transacylation of α-Aryl-β-keto Esters. J. Org. Chem. 2003, 68, 8650–8656. [Google Scholar] [CrossRef] [PubMed]
- Phillips, R.R. Japp-Klingemann reaction. Org. React. 1959, 10, 143–178. [Google Scholar] [CrossRef]
- Filimonov, V.D.; Trusova, M.; Postnikov, P.; Krasnokutskaya, E.A.; Lee, Y.M.; Hawng, H.Y.; Kim, H.; Chi, K.-W. Unusually stable, versatile, and pure arenediazonium tosylates: Their preparation, and synthetic applicability. Org. Lett. 2008, 10, 3961–3964. [Google Scholar] [CrossRef]
- Kutonova, K.V.; Trusova, M.E.; Stankevich, A.V.; Postnikov, P.S.; Filimonov, V.D. Matsuda-Heck reaction with arenediazonium tosylates in water. Beilstein J. Org. Chem. 2015, 11, 358–362. [Google Scholar] [CrossRef]
- Dar’in, D.; Kantin, G.; Bakulina, O.; Žalubovskis, R.; Krasavin, M. Flexible Entry into 3-Arylpent-2-enedioic Acids via Heck–Matsuda Arylation of Dimethyl Glutaconate with Arenediazonium Tosylates. Synthesis 2019, 51, 2230–2236. [Google Scholar] [CrossRef]
- Curtin, D.Y.; Poutsma, M.L. Rearrangements of Benzeneazotribenzoylmethane and its Derivatives. Structural Reassignments. J. Amer. Chem. Soc. 1962, 84, 4887–4892. [Google Scholar] [CrossRef]
- Halasz, I.; Cindro, N.; Dinnebier, R.E.; Vancik, H. An Old Story in New Light: X-Ray Powder Diffraction Provides Novel Insights into a Long-Known Organic Solid-State Rearrangement Reaction. Croat. Chem. Acta 2013, 86, 187–192. [Google Scholar] [CrossRef]
- Starosotnikov, A.M.; Nikol’skiy, V.V.; Borodulya, A.N.; Kachala, V.V.; Bastrakov, M.A.; Solkan, V.N.; Shevelev, S.A. Synthesis and functionalization of 5,7-dinitroquinoline and its N-oxide. Asian J. Org. Chem. 2016, 5, 685–690. [Google Scholar] [CrossRef]
- Rigaku. CrysAlisPro, Version 1.171.42, Rigaku Oxford Diffraction; Rigaku: Tokyo, Japan, 2022.
- Bruker. APEX-III; Bruker AXS Inc.: Madison, WI, USA, 2019. [Google Scholar]
- Krause, L.; Herbst-Irmer, R.; Sheldrick, G.M.; Stalke, D. Comparison of silver and molybdenum microfocus X-ray sources for single-crystal structure determination. J. Appl. Cryst. 2015, 48, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Sheldrick, G.M. SHELXT—Integrated space-group and crystal-structure determination. Acta Cryst. 2015, A71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Cryst. 2015, C71, 3–8. [Google Scholar] [CrossRef]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. OLEX2: A complete structure solution, refinement and analysis program. J. Appl. Cryst. 2009, 42, 339–341. [Google Scholar] [CrossRef]
- Hübschle, C.B.; Sheldrick, G.M.; Dittrich, B. ShelXle: A Qt graphical interface for SHELXL. J. Appl. Cryst. 2011, 44, 1281–1284. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nikol’skiy, V.V.; Minyaev, M.E.; Bastrakov, M.A.; Starosotnikov, A.M. Straightforward and Efficient Protocol for the Synthesis of Pyrazolo [4,3-b]pyridines and Indazoles. Int. J. Mol. Sci. 2023, 24, 1758. https://doi.org/10.3390/ijms24021758
Nikol’skiy VV, Minyaev ME, Bastrakov MA, Starosotnikov AM. Straightforward and Efficient Protocol for the Synthesis of Pyrazolo [4,3-b]pyridines and Indazoles. International Journal of Molecular Sciences. 2023; 24(2):1758. https://doi.org/10.3390/ijms24021758
Chicago/Turabian StyleNikol’skiy, Vladislav V., Mikhail E. Minyaev, Maxim A. Bastrakov, and Alexey M. Starosotnikov. 2023. "Straightforward and Efficient Protocol for the Synthesis of Pyrazolo [4,3-b]pyridines and Indazoles" International Journal of Molecular Sciences 24, no. 2: 1758. https://doi.org/10.3390/ijms24021758
APA StyleNikol’skiy, V. V., Minyaev, M. E., Bastrakov, M. A., & Starosotnikov, A. M. (2023). Straightforward and Efficient Protocol for the Synthesis of Pyrazolo [4,3-b]pyridines and Indazoles. International Journal of Molecular Sciences, 24(2), 1758. https://doi.org/10.3390/ijms24021758






