Hemorrhagic Transformation of Ischemic Strokes
Abstract
1. Introduction
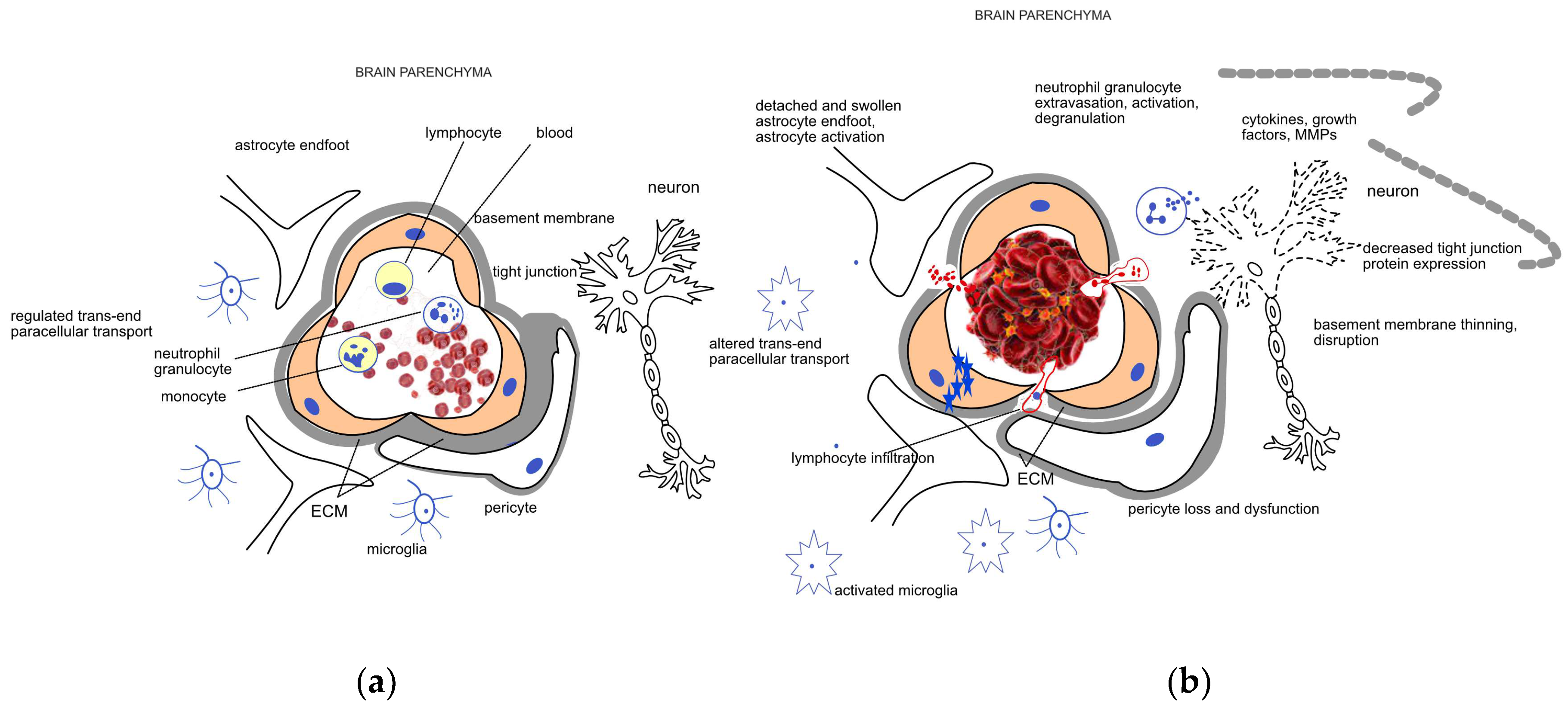
2. Pathophysiology of HT
3. Clinical Risk Factors of HT
3.1. Role of Hypertension
3.2. Role of Early Signs of Ischemia, Vessel Calcification, and Hyperdense Middle Cerebral Artery Sign on Cranial CT and Collateral Score on CTA
3.3. Role of Recombinant Tissue Plasminogen Activator (rtPA) and Mechanical Thrombectomy
3.4. Role of Hyperglycemia
3.5. Role of Admission NIHSS Points
3.6. Role of Advanced Age
3.7. Role of Timing
3.8. Role of Low Platelet Count and Size
3.9. Role of Cardioembolism
3.10. Role of Antithrombotic Treatment
3.11. Role of Further Clinical Factors
3.11.1. Liver Fibrosis
3.11.2. Iron Homeostasis
3.12. Role of Gender
4. Biomarkers of HT
4.1. Matrix Metalloproteinase-9 (MMP-9)
4.2. Cellular Fibronectin (c-Fn)
4.3. Ferritin
4.4. Neutrophil-to-Lymphocyte Ratio (NLR)
4.5. S100 Calcium-Binding Protein B (S100B)
4.6. Soluble ICAM-1 (sICAM-1)
4.7. Vascular Adhesion Protein-1 (VAP-1) and Semicarbazide-Sensitive Amine Oxidase (SSAO)
5. Role of the Gut Microbiome—Promising Experimental Stroke Models
6. Therapeutic Approaches
6.1. Minocycline
6.2. Fingolimod
6.3. Tacrolimus
6.4. Statins
6.5. Others
7. Discussion
8. Take Home Messages
9. Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Katan, M.; Luft, A. Global Burden of Stroke. Semin. Neurol. 2018, 38, 208–211. [Google Scholar] [CrossRef] [PubMed]
- Jickling, G.C.; Liu, D.; Stamova, B.; Ander, B.P.; Zhan, X.; Lu, A.; Sharp, F.R. Hemorrhagic transformation after ischemic stroke in animals and humans. J. Cereb. Blood Flow Metab. 2014, 34, 185–199. [Google Scholar] [CrossRef] [PubMed]
- Spronk, E.; Sykes, G.; Falcione, S.; Munsterman, D.; Joy, T.; Kamtchum-Tatuene, J.; Jickling, G.C. Hemorrhagic Transformation in Ischemic Stroke and the Role of Inflammation. Front. Neurol. 2021, 12, 661955. [Google Scholar] [CrossRef]
- Fiorelli, M.; Bastianello, S.; von Kummer, R.; del Zoppo, G.J.; Larrue, V.; Lesaffre, E.; Ringleb, A.P.; Lorenzano, S.; Manelfe, C.; Bozzao, L. Hemorrhagic transformation within 36 hours of a cerebral infarct: Relationships with early clinical deterioration and 3-month outcome in the European Cooperative Acute Stroke Study I (ECASS I) cohort. Stroke 1999, 30, 2280–2284. [Google Scholar] [CrossRef] [PubMed]
- The NINDS t-PA Stroke Study Group. Intracerebral hemorrhage after intravenous t-PA therapy for ischemic stroke. Stroke 1997, 28, 2109–2118. [Google Scholar] [CrossRef]
- Külkens, S.; Hacke, W. Thrombolysis with alteplase for acute ischemic stroke: Review of SITS-MOST and other Phase IV studies. Expert Rev. Neurother. 2007, 7, 783–788. [Google Scholar] [CrossRef]
- Sandoval, K.E.; Witt, K.A. Blood-brain barrier tight junction permeability and ischemic stroke. Neurobiol. Dis. 2008, 32, 200–219. [Google Scholar] [CrossRef]
- Sulimai, N.; Brown, J.; Lominadze, D. Vascular Effects on Cerebrovascular Permeability and Neurodegeneration. Biomolecules 2023, 13, 648. [Google Scholar] [CrossRef]
- Bernardo-Castro, S.; Sousa, J.A.; Brás, A.; Cecília, C.; Rodrigues, B.; Almendra, L.; Machado, C.; Santo, G.; Silva, F.; Ferreira, L.; et al. Pathophysiology of Blood-Brain Barrier Permeability Throughout the Different Stages of Ischemic Stroke and Its Implication on Hemorrhagic Transformation and Recovery. Front. Neurol. 2020, 11, 594672. [Google Scholar] [CrossRef]
- Iadecola, C.; Anrather, J. The immunology of stroke: From mechanisms to translation. Nat. Med. 2011, 17, 796–808. [Google Scholar] [CrossRef]
- Kim, J.; Song, T.J.; Park, J.H.; Lee, H.S.; Nam, C.M.; Nam, H.S.; Kim, Y.D.; Heo, J.H. Different prognostic value of white blood cell subtypes in patients with acute cerebral infarction. Atherosclerosis 2012, 222, 464–467. [Google Scholar] [CrossRef] [PubMed]
- Castellanos, M.; Sobrino, T.; Millán, M.; García, M.; Arenillas, J.; Nombela, F.; Brea, D.; Perez de la Ossa, N.; Serena, J.; Vivancos, J.; et al. Serum cellular fibronectin and matrix metalloproteinase-9 as screening biomarkers for the prediction of parenchymal hematoma after thrombolytic therapy in acute ischemic stroke: A multicenter confirmatory study. Stroke 2007, 38, 1855–1859, Erratum in Stroke 2007, 38, e76. [Google Scholar] [CrossRef] [PubMed]
- Courties, G.; Herisson, F.; Sager, H.B.; Heidt, T.; Ye, Y.; Wei, Y.; Sun, Y.; Severe, N.; Dutta, P.; Scharff, J.; et al. Ischemic stroke activates hematopoietic bone marrow stem cells. Circ. Res. 2015, 116, 407–417. [Google Scholar] [CrossRef] [PubMed]
- Paulson, O.B.; Strandgaard, S.; Edvinsson, L. Cerebral autoregulation. Cerebrovasc. Brain Metab. Rev. 1990, 2, 161–192. [Google Scholar]
- Xiong, L.; Liu, X.; Shang, T.; Smielewski, P.; Donnelly, J.; Guo, Z.N.; Yang, Y.; Leung, T.; Czosnyka, M.; Zhang, R.; et al. Impaired cerebral autoregulation: Measurement and application to stroke. J. Neurol. Neurosurg. Psychiatry 2017, 88, 520–531. [Google Scholar] [CrossRef]
- Wang, A.; Ortega-Gutierrez, S.; Petersen, N.H. Autoregulation in the Neuro ICU. Curr. Treat. Options Neurol. 2018, 20, 20. [Google Scholar] [CrossRef]
- Whiteley, W.N.; Slot, K.B.; Fernandes, P.; Sandercock, P.; Wardlaw, J. Risk factors for intracranial hemorrhage in acute ischemic stroke patients treated with recombinant tissue plasminogen activator: A systematic review and meta-analysis of 55 studies. Stroke 2012, 43, 2904–2909. [Google Scholar] [CrossRef]
- Jensen, M.; Schlemm, E.; Cheng, B.; Lettow, I.; Quandt, F.; Boutitie, F.; Ebinger, M.; Endres, M.; Fiebach, J.B.; Fiehler, J.; et al. Clinical Characteristics and Outcome of Patients With Hemorrhagic Transformation After Intravenous Thrombolysis in the WAKE-UP Trial. Front. Neurol. 2020, 11, 957. [Google Scholar] [CrossRef]
- Pires, P.W.; Dams Ramos, C.M.; Matin, N.; Dorrance, A.M. The effects of hypertension on the cerebral circulation. Am. J. Physiol. Heart Circ. Physiol. 2013, 304, H1598–H1614. [Google Scholar] [CrossRef]
- Powers, W.J.; Rabinstein, A.A.; Ackerson, T.; Adeoye, O.M.; Bambakidis, N.C.; Becker, K.; Biller, J.; Brown, M.; Demaerschalk, B.M.; Hoh, B.; et al. Guidelines for the Early Management of Patients With Acute Ischemic Stroke: 2019 Update to the 2018 Guidelines for the Early Management of Acute Ischemic Stroke: A Guideline for Healthcare Professionals From the American Heart Association/American Stroke Association. Stroke 2019, 50, e344–e418, Erratum in Stroke 2019, 50, e440–e441. [Google Scholar] [CrossRef]
- Goyal, M.; Demchuk, A.M.; Menon, B.K.; Eesa, M.; Rempel, J.L.; Thornton, J.; Roy, D.; Jovin, T.G.; Willinsky, R.A.; Sapkota, B.L.; et al. Randomized assessment of rapid endovascular treatment of ischemic stroke. N. Engl. J. Med. 2015, 372, 1019–1030. [Google Scholar] [CrossRef]
- Jovin, T.G.; Saver, J.L.; Ribo, M.; Pereira, V.; Furlan, A.; Bonafe, A.; Baxter, B.; Gupta, R.; Lopes, D.; Jansen, O.; et al. Diffusion-weighted imaging or computerized tomography perfusion assessment with clinical mismatch in the triage of wake up and late presenting strokes undergoing neurointervention with Trevo (DAWN) trial methods. Int. J. Stroke 2017, 12, 641–652. [Google Scholar] [CrossRef] [PubMed]
- Goyal, N.; Tsivgoulis, G.; Pandhi, A.; Dillard, K.; Alsbrook, D.; Chang, J.J.; Krishnaiah, B.; Nickele, C.; Hoit, D.; Alsherbini, K.; et al. Blood pressure levels post mechanical thrombectomy and outcomes in non-recanalized large vessel occlusion patients. J. Neurointerv. Surg. 2018, 10, 925–931. [Google Scholar] [CrossRef] [PubMed]
- Mistry, E.A.; Mayer, S.A.; Khatri, P. Blood Pressure Management after Mechanical Thrombectomy for Acute Ischemic Stroke: A Survey of the StrokeNet Sites. J. Stroke Cerebrovasc. Dis. 2018, 27, 2474–2478. [Google Scholar] [CrossRef] [PubMed]
- Arba, F.; Piccardi, B.; Palumbo, V.; Biagini, S.; Galmozzi, F.; Iovene, V.; Giannini, A.; Testa, G.D.; Sodero, A.; Nesi, M.; et al. Blood-brain barrier leakage and hemorrhagic transformation: The Reperfusion Injury in Ischemic StroKe (RISK) study. Eur. J. Neurol. 2021, 28, 3147–3154. [Google Scholar] [CrossRef]
- Langel, C.; Popovic, K.S. Infarct-core CT perfusion parameters in predicting post-thrombolysis hemorrhagic transformation of acute ischemic stroke. Radiol. Oncol. 2018, 53, 25–30. [Google Scholar] [CrossRef]
- Yu, Y.; Zhang, F.L.; Qu, Y.M.; Zhang, P.; Zhou, H.W.; Luo, Y.; Wang, Y.; Liu, J.; Qin, H.Q.; Guo, Z.N.; et al. Intracranial Calcification is Predictive for Hemorrhagic Transformation and Prognosis After Intravenous Thrombolysis in Non-Cardioembolic Stroke Patients. J. Atheroscler. Thromb. 2021, 28, 356–364. [Google Scholar] [CrossRef] [PubMed]
- Strbian, D.; Engelter, S.; Michel, P.; Meretoja, A.; Sekoranja, L.; Ahlhelm, F.J.; Mustanoja, S.; Kuzmanovic, I.; Sairanen, T.; Forss, N.; et al. Symptomatic intracranial hemorrhage after stroke thrombolysis: The SEDAN score. Ann. Neurol. 2012, 71, 634–641. [Google Scholar] [CrossRef] [PubMed]
- Zou, M.; Churilov, L.; He, A.; Campbell, B.; Davis, S.M.; Yan, B. Hyperdense middle cerebral artery sign is associated with increased risk of hemorrhagic transformation after intravenous thrombolysis for patients with acute ischaemic stroke. J. Clin. Neurosci. 2013, 20, 984–987. [Google Scholar] [CrossRef]
- Tian, B.; Tian, X.; Shi, Z.; Peng, W.; Zhang, X.; Yang, P.; Li, Z.; Zhang, X.; Lou, M.; Yin, C.; et al. Clinical and Imaging Indicators of Hemorrhagic Transformation in Acute Ischemic Stroke After Endovascular Thrombectomy. Stroke 2022, 53, 1674–1681. [Google Scholar] [CrossRef]
- Hao, Y.; Yang, D.; Wang, H.; Zi, W.; Zhang, M.; Geng, Y.; Zhou, Z.; Wang, W.; Xu, H.; Tian, X.; et al. Predictors for Symptomatic Intracranial Hemorrhage After Endovascular Treatment of Acute Ischemic Stroke. Stroke 2017, 48, 1203–1209. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Wang, X.; Yu, Z. Ischemia-reperfusion Injury in the Brain: Mechanisms and Potential Therapeutic Strategies. Biochem. Pharmacol. 2016, 5, 213. [Google Scholar] [CrossRef]
- Khatri, P.; Wechsler, L.R.; Broderick, J.P. Intracranial hemorrhage associated with revascularization therapies. Stroke 2007, 38, 431–440. [Google Scholar] [CrossRef] [PubMed]
- Yepes, M.; Sandkvist, M.; Moore, E.G.; Bugge, T.H.; Strickland, D.K.; Lawrence, D.A. Tissue-type plasminogen activator induces opening of the blood-brain barrier via the LDL receptor-related protein. J. Clin. Investig. 2003, 112, 1533–1540. [Google Scholar] [CrossRef]
- Simão, F.; Ustunkaya, T.; Clermont, A.C.; Feener, E.P. Plasma kallikrein mediates brain hemorrhage and edema caused by tissue plasminogen activator therapy in mice after stroke. Blood 2017, 129, 2280–2290. [Google Scholar] [CrossRef] [PubMed]
- Su, E.J.; Fredriksson, L.; Geyer, M.; Folestad, E.; Cale, J.; Andrae, J.; Gao, Y.; Pietras, K.; Mann, K.; Yepes, M.; et al. Activation of PDGF-CC by tissue plasminogen activator impairs blood-brain barrier integrity during ischemic stroke. Nat. Med. 2008, 14, 731–737. [Google Scholar] [CrossRef]
- Shi, K.; Zou, M.; Jia, D.M.; Shi, S.; Yang, X.; Liu, Q.; Dong, J.F.; Sheth, K.N.; Wang, X.; Shi, F.D. tPA Mobilizes Immune Cells That Exacerbate Hemorrhagic Transformation in Stroke. Circ. Res. 2021, 128, 62–75. [Google Scholar] [CrossRef]
- Teng, D.; Pannell, J.S.; Rennert, R.C.; Li, J.; Li, Y.S.; Wong, V.W.; Chien, S.; Khalessi, A.A. Endothelial trauma from mechanical thrombectomy in acute stroke: In vitro live-cell platform with animal validation. Stroke 2015, 46, 1099–1106. [Google Scholar] [CrossRef]
- Berkhemer, O.A.; Fransen, P.S.; Beumer, D.; van den Berg, L.A.; Lingsma, H.F.; Yoo, A.J.; Schonewille, W.J.; Vos, J.A.; Nederkoorn, P.J.; Wermer, M.J.; et al. A randomized trial of intraarterial treatment for acute ischemic stroke. N. Engl. J. Med. 2015, 372, 11–20, Erratum in N. Engl. J. Med. 2015, 372, 394. [Google Scholar] [CrossRef]
- Saver, J.L.; Goyal, M.; Bonafe, A.; Diener, H.C.; Levy, E.I.; Pereira, V.M.; Albers, G.W.; Cognard, C.; Cohen, D.J.; Hacke, W.; et al. Stent-retriever thrombectomy after intravenous t-PA vs. t-PA alone in stroke. N. Engl. J. Med. 2015, 372, 2285–2295. [Google Scholar] [CrossRef]
- Campbell, B.C.; Mitchell, P.J.; Kleinig, T.J.; Dewey, H.M.; Churilov, L.; Yassi, N.; Yan, B.; Dowling, R.J.; Parsons, M.W.; Oxley, T.J.; et al. Endovascular therapy for ischemic stroke with perfusion-imaging selection. N. Engl. J. Med. 2015, 372, 1009–1018. [Google Scholar] [CrossRef] [PubMed]
- Jovin, T.G.; Chamorro, A.; Cobo, E.; de Miquel, M.A.; Molina, C.A.; Rovira, A.; San Román, L.; Serena, J.; Abilleira, S.; Ribó, M.; et al. Thrombectomy within 8 hours after symptom onset in ischemic stroke. N. Engl. J. Med. 2015, 372, 2296–2306. [Google Scholar] [CrossRef]
- Albers, G.W.; Marks, M.P.; Kemp, S.; Christensen, S.; Tsai, J.P.; Ortega-Gutierrez, S.; McTaggart, R.A.; Torbey, M.T.; Kim-Tenser, M.; Leslie-Mazwi, T.; et al. Thrombectomy for Stroke at 6 to 16 Hours with Selection by Perfusion Imaging. N. Engl. J. Med. 2018, 378, 708–718. [Google Scholar] [CrossRef]
- Nogueira, R.G.; Jadhav, A.P.; Haussen, D.C.; Bonafe, A.; Budzik, R.F.; Bhuva, P.; Yavagal, D.R.; Ribo, M.; Cognard, C.; Hanel, R.A.; et al. Thrombectomy 6 to 24 Hours after Stroke with a Mismatch between Deficit and Infarct. N. Engl. J. Med. 2018, 378, 11–21. [Google Scholar] [CrossRef]
- Goyal, M.; Menon, B.K.; van Zwam, W.H.; Dippel, D.W.; Mitchell, P.J.; Demchuk, A.M.; Dávalos, A.; Majoie, C.B.; van der Lugt, A.; de Miquel, M.A.; et al. Endovascular thrombectomy after large-vessel ischaemic stroke: A meta-analysis of individual patient data from five randomised trials. Lancet 2016, 387, 1723–1731. [Google Scholar] [CrossRef]
- Desilles, J.P.; Meseguer, E.; Labreuche, J.; Lapergue, B.; Sirimarco, G.; Gonzalez-Valcarcel, J.; Lavallée, P.; Cabrejo, L.; Guidoux, C.; Klein, I.; et al. Diabetes mellitus, admission glucose, and outcomes after stroke thrombolysis: A registry and systematic review. Stroke 2013, 44, 1915–1923. [Google Scholar] [CrossRef]
- Johnston, K.C.; Bruno, A.; Pauls, Q.; Hall, C.E.; Barrett, K.M.; Barsan, W.; Fansler, A.; Van de Bruinhorst, K.; Janis, S.; Durkalski-Mauldin, V.L.; et al. Intensive vs Standard Treatment of Hyperglycemia and Functional Outcome in Patients With Acute Ischemic Stroke: The SHINE Randomized Clinical Trial. JAMA 2019, 322, 326–335, Erratum in JAMA 2019, 322, 1718. [Google Scholar] [CrossRef]
- Gray, C.S.; Hildreth, A.J.; Sandercock, P.A.; O’Connell, J.E.; Johnston, D.E.; Cartlidge, N.E.; Bamford, J.M.; James, O.F.; Alberti, K.G.; GIST Trialists Collaboration. Glucose-potassium-insulin infusions in the management of post-stroke hyperglycaemia: The UK Glucose Insulin in Stroke Trial (GIST-UK). Lancet Neurol. 2007, 6, 397–406. [Google Scholar] [CrossRef]
- Kerenyi, L.; Kardos, L.; Szász, J.; Szatmári, S.; Bereczki, D.; Hegedüs, K.; Csiba, L. Factors influencing hemorrhagic transformation in ischemic stroke: A clinicopathological comparison. Eur. J. Neurol. 2006, 13, 1251–1255. [Google Scholar] [CrossRef]
- Kidwell, C.S.; Saver, J.L.; Carneado, J.; Sayre, J.; Starkman, S.; Duckwiler, G.; Gobin, Y.P.; Jahan, R.; Vespa, P.; Villablanca, J.P.; et al. Predictors of hemorrhagic transformation in patients receiving intra-arterial thrombolysis. Stroke 2002, 33, 717–724. [Google Scholar] [CrossRef]
- Qian, Y.; Qian, Z.T.; Huang, C.H.; Wang, H.Y.; Lu, X.; Cao, K.; Sun, J.Y.; Li, Q.Y. Predictive Factors and Nomogram to Evaluate the Risk of Symptomatic Intracerebral Hemorrhage for Stroke Patients Receiving Thrombectomy. World Neurosurg. 2020, 144, e466–e474. [Google Scholar] [CrossRef]
- Sohrabji, F.; Bake, S.; Lewis, D.K. Age-related changes in brain support cells: Implications for stroke severity. Neurochem. Int. 2013, 63, 291–301. [Google Scholar] [CrossRef]
- Weiskopf, D.; Weinberger, B.; Grubeck-Loebenstein, B. The aging of the immune system. Transpl. Int. 2009, 22, 1041–1050. [Google Scholar] [CrossRef]
- Héja, M.; Fekete, I.; Horváth, L.; Márton, S.; Fekete, K.E. Experiences With Intravenous Thrombolysis in Acute Ischemic Stroke by Elderly Patients-A “Real World Scenario”. Front. Neurol. 2021, 12, 721337. [Google Scholar] [CrossRef]
- Raychev, R.; Saver, J.L.; Jahan, R.; Nogueira, R.G.; Goyal, M.; Pereira, V.M.; Gralla, J.; Levy, E.I.; Yavagal, D.R.; Cognard, C.; et al. The impact of general anesthesia, baseline ASPECTS, time to treatment, and IV tPA on intracranial hemorrhage after neurothrombectomy: Pooled analysis of the SWIFT PRIME, SWIFT, and STAR trials. J. Neurointerv. Surg. 2020, 12, 2–6, Erratum in J. Neurointerv. Surg. 2021, 13, e21. [Google Scholar] [CrossRef]
- Domingo, R.A.; Tripathi, S.; Perez-Vega, C.; Martinez, J.; Suarez Meade, P.; Ramos-Fresnedo, A.; English, S.W., Jr.; Huynh, T.; Lin, M.P.; Fox, W.C.; et al. Influence of Platelet Count on Procedure-Related Outcomes After Mechanical Thrombectomy for Large Vessel Occlusion: A Systematic Review and Meta-Analysis. World Neurosurg. 2022, 157, 187–192. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.R.; Chen, Y.B.; Zeng, Y.Y.; Ruan, Y.T.; Yuan, C.X.; Cheng, Q.Q.; Chen, H.J.; Luan, X.Q.; Huang, G.Q.; He, J.C. Hemostasis functions are associated with hemorrhagic transformation in non-atrial fibrillation patients: A case-control study. BMC Neurol. 2021, 21, 36. [Google Scholar] [CrossRef]
- Hornig, C.R.; Bauer, T.; Simon, C.; Trittmacher, S.; Dorndorf, W. Hemorrhagic transformation in cardioembolic cerebral infarction. Stroke 1993, 24, 465–468. [Google Scholar] [CrossRef]
- Molina, C.A.; Montaner, J.; Abilleira, S.; Ibarra, B.; Romero, F.; Arenillas, J.F.; Alvarez-Sabín, J. Timing of spontaneous recanalization and risk of hemorrhagic transformation in acute cardioembolic stroke. Stroke 2001, 32, 1079–1084. [Google Scholar] [CrossRef] [PubMed]
- Tornyos, D.; Bálint, A.; Kupó, P.; El Abdallaoui, O.E.A.; Komócsi, A. Antithrombotic Therapy for Secondary Prevention in Patients with Non-Cardioembolic Stroke or Transient Ischemic Attack: A Systematic Review. Life 2021, 11, 447. [Google Scholar] [CrossRef] [PubMed]
- Berge, E.; Whiteley, W.; Audebert, H.; De Marchis, G.M.; Fonseca, A.C.; Padiglioni, C.; de la Ossa, N.P.; Strbian, D.; Tsivgoulis, G.; Turc, G. European Stroke Organisation (ESO) guidelines on intravenous thrombolysis for acute ischaemic stroke. Eur. Stroke J. 2021, 6, I–LXII. [Google Scholar] [CrossRef] [PubMed]
- Sussman, E.S.; Jin, M.; Pendharkar, A.V.; Pulli, B.; Feng, A.; Heit, J.J.; Telischak, N.A. Dual antiplatelet therapy after carotid artery stenting: Trends and outcomes in a large national database. J. Neurointerv. Surg. 2021, 13, 8–13. [Google Scholar] [CrossRef]
- Dawson, J.; Béjot, Y.; Christensen, L.M.; De Marchis, G.M.; Dichgans, M.; Hagberg, G.; Heldner, M.R.; Milionis, H.; Li, L.; Pezzella, F.R.; et al. European Stroke Organisation (ESO) guideline on pharmacological interventions for long-term secondary prevention after ischaemic stroke or transient ischaemic attack. Eur. Stroke J. 2022, 7, I–XLI. [Google Scholar] [CrossRef]
- Seiffge, D.J.; Werring, D.J.; Paciaroni, M.; Dawson, J.; Warach, S.; Milling, T.J.; Engelter, S.T.; Fischer, U.; Norrving, B. Timing of anticoagulation after recent ischaemic stroke in patients with atrial fibrillation. Lancet Neurol. 2019, 18, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Abbas, M.; Malicke, D.T.; Schramski, J.T. Stroke Anticoagulation. [Updated 2023 Apr 3]. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2023. Available online: https://www.ncbi.nlm.nih.gov/books/NBK549826/ (accessed on 1 January 2023).
- Otto, C.M.; Nishimura, R.A.; Bonow, R.O.; Carabello, B.A.; Erwin, J.P., 3rd; Gentile, F.; Jneid, H.; Krieger, E.V.; Mack, M.; McLeod, C.; et al. 2020 ACC/AHA Guideline for the Management of Patients With Valvular Heart Disease: Executive Summary: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation 2021, 143, e35–e71. [Google Scholar] [CrossRef] [PubMed]
- Powers, W.J. Strokelore: Antithrombotic therapy and hemorrhagic infarction. J. Stroke Cerebrovasc. Dis. 2022, 31, 106395. [Google Scholar] [CrossRef] [PubMed]
- Parikh, N.S.; Navi, B.B.; Schneider, Y.; Jesudian, A.; Kamel, H. Association Between Cirrhosis and Stroke in a Nationally Representative Cohort. JAMA Neurol. 2017, 74, 927–932. [Google Scholar] [CrossRef]
- Baik, M.; Kim, S.U.; Kang, S.; Park, H.J.; Nam, H.S.; Heo, J.H.; Kim, B.K.; Park, J.Y.; Kim, D.Y.; Ahn, S.H.; et al. Liver Fibrosis, Not Steatosis, Associates with Long-Term Outcomes in Ischaemic Stroke Patients. Cerebrovasc. Dis. 2019, 47, 32–39. [Google Scholar] [CrossRef]
- Yuan, C.X.; Ruan, Y.T.; Zeng, Y.Y.; Cheng, H.R.; Cheng, Q.Q.; Chen, Y.B.; He, W.L.; Huang, G.Q.; He, J.C. Liver Fibrosis Is Associated With Hemorrhagic Transformation in Patients With Acute Ischemic Stroke. Front. Neurol. 2020, 11, 867. [Google Scholar] [CrossRef]
- Jagavelu, K.; Routray, C.; Shergill, U.; O’Hara, S.P.; Faubion, W.; Shah, V.H. Endothelial cell toll-like receptor 4 regulates fibrosis-associated angiogenesis in the liver. Hepatology 2010, 52, 590–601. [Google Scholar] [CrossRef]
- Seki, E.; Schwabe, R.F. Hepatic inflammation and fibrosis: Functional links and key pathways. Hepatology 2015, 61, 1066–1079. [Google Scholar] [CrossRef]
- Polimeni, L.; Del Ben, M.; Baratta, F.; Perri, L.; Albanese, F.; Pastori, D.; Violi, F.; Angelico, F. Oxidative stress: New insights on the association of non-alcoholic fatty liver disease and atherosclerosis. World J. Hepatol. 2015, 7, 1325–1336. [Google Scholar] [CrossRef]
- Tripodi, A.; Mannucci, P.M. The coagulopathy of chronic liver disease. N. Engl. J. Med. 2011, 365, 147–156. [Google Scholar] [CrossRef]
- MacKenzie, E.L.; Iwasaki, K.; Tsuji, Y. Intracellular iron transport and storage: From molecular mechanisms to health implications. Antioxid. Redox Signal. 2008, 10, 997–1030. [Google Scholar] [CrossRef]
- Russo, N.; Edwards, M.; Andrews, T.; O’Brien, M.; Bhatia, K.P. Hereditary haemochromatosis is unlikely to cause movement disorders–A critical review. J. Neurol. 2004, 251, 849–852. [Google Scholar] [CrossRef]
- Won, S.M.; Lee, J.H.; Park, U.J.; Gwag, J.; Gwag, B.J.; Lee, Y.B. Iron mediates endothelial cell damage and blood-brain barrier opening in the hippocampus after transient forebrain ischemia in rats. Exp. Mol. Med. 2011, 43, 121–128. [Google Scholar] [CrossRef]
- García-Yébenes, I.; García-Culebras, A.; Peña-Martínez, C.; Fernández-López, D.; Díaz-Guzmán, J.; Negredo, P.; Avendaño, C.; Castellanos, M.; Gasull, T.; Dávalos, A.; et al. Iron Overload Exacerbates the Risk of Hemorrhagic Transformation After tPA (Tissue-Type Plasminogen Activator) Administration in Thromboembolic Stroke Mice. Stroke 2018, 49, 2163–2172. [Google Scholar] [CrossRef]
- Won, Y.D.; Kim, J.M.; Cheong, J.H.; Ryu, J.I.; Koh, S.H.; Han, M.H. Effect of Possible Osteoporosis on Parenchymal-Type Hemorrhagic Transformation in Patients with Cardioembolic Stroke. J. Clin. Med. 2021, 10, 2526. [Google Scholar] [CrossRef]
- Howard, V.J.; Madsen, T.E.; Kleindorfer, D.O.; Judd, S.E.; Rhodes, J.D.; Soliman, E.Z.; Kissela, B.M.; Safford, M.M.; Moy, C.S.; McClure, L.A.; et al. Sex and Race Differences in the Association of Incident Ischemic Stroke With Risk Factors. JAMA Neurol. 2019, 76, 179–186. [Google Scholar] [CrossRef]
- WHO International Programme on Chemical Safety Biomarkers in Risk Assessment: Validity and Validation. Available online: http://www.inchem.org/documents/ehc/ehc/ehc222.htm (accessed on 11 September 2023).
- Montaner, J.; Molina, C.A.; Monasterio, J.; Abilleira, S.; Arenillas, J.F.; Ribó, M.; Quintana, M.; Alvarez-Sabín, J. Matrix metalloproteinase-9 pretreatment level predicts intracranial hemorrhagic complications after thrombolysis in human stroke. Circulation 2003, 107, 598–603. [Google Scholar] [CrossRef]
- Krishnamoorthy, S.; Singh, G.; Jose, K.J.; Soman, B.; Foerch, C.; Kimberly, W.T.; Millán, M.; Świtońska, M.; Maestrini, I.; Bordet, R.; et al. Biomarkers in the Prediction of Hemorrhagic Transformation in Acute Stroke: A Systematic Review and Meta-Analysis. Cerebrovasc. Dis. 2022, 51, 235–247. [Google Scholar] [CrossRef] [PubMed]
- Lapchak, P.A.; Chapman, D.F.; Zivin, J.A. Metalloproteinase inhibition reduces thrombolytic (tissue plasminogen activator)-induced hemorrhage after thromboembolic stroke. Stroke 2000, 31, 3034–3040. [Google Scholar] [CrossRef] [PubMed]
- Sumii, T.; Lo, E.H. Involvement of matrix metalloproteinase in thrombolysis-associated hemorrhagic transformation after embolic focal ischemia in rats. Stroke 2002, 33, 831–836. [Google Scholar] [CrossRef]
- Pankov, R.; Yamada, K.M. Fibronectin at a glance. J. Cell Sci. 2002, 115 Pt 20, 3861–3863. [Google Scholar] [CrossRef]
- Wang, W.; Knovich, M.A.; Coffman, L.G.; Torti, F.M.; Torti, S.V. Serum ferritin: Past, present and future. Biochim. Biophys. Acta 2010, 1800, 760–769. [Google Scholar] [CrossRef] [PubMed]
- Rosell, A.; Cuadrado, E.; Ortega-Aznar, A.; Hernández-Guillamon, M.; Lo, E.H.; Montaner, J. MMP-9-positive neutrophil infiltration is associated to blood-brain barrier breakdown and basal lamina type IV collagen degradation during hemorrhagic transformation after human ischemic stroke. Stroke 2008, 39, 1121–1126. [Google Scholar] [CrossRef] [PubMed]
- Acanfora, D.; Gheorghiade, M.; Trojano, L.; Furgi, G.; Pasini, E.; Picone, C.; Papa, A.; Iannuzzi, G.L.; Bonow, R.O.; Rengo, F. Relative lymphocyte count: A prognostic indicator of mortality in elderly patients with congestive heart failure. Am. Heart J. 2001, 142, 167–173. [Google Scholar] [CrossRef]
- Zhang, R.; Wu, X.; Hu, W.; Zhao, L.; Zhao, S.; Zhang, J.; Chu, Z.; Xu, Y. Neutrophil-to-lymphocyte ratio predicts hemorrhagic transformation in ischemic stroke: A meta-analysis. Brain Behav. 2019, 9, e01382. [Google Scholar] [CrossRef]
- Wang, D.D.; Bordey, A. The astrocyte odyssey. Prog. Neurobiol. 2008, 86, 342–367. [Google Scholar] [CrossRef]
- Roebuck, K.A.; Finnegan, A. Regulation of intercellular adhesion molecule-1 (CD54) gene expression. J. Leukoc. Biol. 1999, 66, 876–888. [Google Scholar] [CrossRef]
- Wu, B.N.; Wu, J.; Hao, D.L.; Mao, L.L.; Zhang, J.; Huang, T.T. High serum sICAM-1 is correlated with cerebral microbleeds and hemorrhagic transformation in ischemic stroke patients. Br. J. Neurosurg. 2018, 32, 631–636. [Google Scholar] [CrossRef] [PubMed]
- Stolen, C.M.; Yegutkin, G.G.; Kurkijärvi, R.; Bono, P.; Alitalo, K.; Jalkanen, S. Origins of serum semicarbazide-sensitive amine oxidase. Circ. Res. 2004, 95, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Guillamon, M.; Garcia-Bonilla, L.; Solé, M.; Sosti, V.; Parés, M.; Campos, M.; Ortega-Aznar, A.; Domínguez, C.; Rubiera, M.; Ribó, M.; et al. Plasma VAP-1/SSAO activity predicts intracranial hemorrhages and adverse neurological outcome after tissue plasminogen activator treatment in stroke. Stroke 2010, 41, 1528–1535. [Google Scholar] [CrossRef] [PubMed]
- Cryan, J.F.; O’Riordan, K.J.; Sandhu, K.; Peterson, V.; Dinan, T.G. The gut microbiome in neurological disorders. Lancet Neurol. 2020, 19, 179–194. [Google Scholar] [CrossRef]
- Huang, Q.; Di, L.; Yu, F.; Feng, X.; Liu, Z.; Wei, M.; Luo, Y.; Xia, J. Alterations in the gut microbiome with hemorrhagic transformation in experimental stroke. CNS Neurosci. Ther. 2022, 28, 77–91. [Google Scholar] [CrossRef]
- Yin, J.; Liao, S.X.; He, Y.; Wang, S.; Xia, G.H.; Liu, F.T.; Zhu, J.J.; You, C.; Chen, Q.; Zhou, L.; et al. Dysbiosis of Gut Microbiota With Reduced Trimethylamine-N-Oxide Level in Patients With Large-Artery Atherosclerotic Stroke or Transient Ischemic Attack. J. Am. Heart Assoc. 2015, 4, e002699. [Google Scholar] [CrossRef]
- Kurita, N.; Yamashiro, K.; Kuroki, T.; Tanaka, R.; Urabe, T.; Ueno, Y.; Miyamoto, N.; Takanashi, M.; Shimura, H.; Inaba, T.; et al. Metabolic endotoxemia promotes neuroinflammation after focal cerebral ischemia. J. Cereb. Blood Flow Metab. 2020, 40, 2505–2520. [Google Scholar] [CrossRef]
- Singh, V.; Roth, S.; Llovera, G.; Sadler, R.; Garzetti, D.; Stecher, B.; Dichgans, M.; Liesz, A. Microbiota Dysbiosis Controls the Neuroinflammatory Response after Stroke. J. Neurosci. 2016, 36, 7428–7440. [Google Scholar] [CrossRef]
- Cirillo, C.; Brihmat, N.; Castel-Lacanal, E.; Le Friec, A.; Barbieux-Guillot, M.; Raposo, N.; Pariente, J.; Viguier, A.; Simonetta-Moreau, M.; Albucher, J.F.; et al. Post-stroke remodeling processes in animal models and humans. J. Cereb. Blood Flow Metab. 2020, 40, 3–22. [Google Scholar] [CrossRef]
- Chen, R.; Wu, P.; Cai, Z.; Fang, Y.; Zhou, H.; Lasanajak, Y.; Tang, L.; Ye, L.; Hou, C.; Zhao, J. Puerariae Lobatae Radix with chuanxiong Rhizoma for treatment of cerebral ischemic stroke by remodeling gut microbiota to regulate the brain-gut barriers. J. Nutr. Biochem. 2019, 65, 101–114. [Google Scholar] [CrossRef]
- Sadler, R.; Cramer, J.V.; Heindl, S.; Kostidis, S.; Betz, D.; Zuurbier, K.R.; Northoff, B.H.; Heijink, M.; Goldberg, M.P.; Plautz, E.J.; et al. Short-Chain Fatty Acids Improve Poststroke Recovery via Immunological Mechanisms. J. Neurosci. 2020, 40, 1162–1173. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Won, S.J.; Xu, Y.; Swanson, R.A. Targeting microglial activation in stroke therapy: Pharmacological tools and gender effects. Curr. Med. Chem. 2014, 21, 2146–2155. [Google Scholar] [CrossRef] [PubMed]
- Switzer, J.A.; Hess, D.C.; Ergul, A.; Waller, J.L.; Machado, L.S.; Portik-Dobos, V.; Pettigrew, L.C.; Clark, W.M.; Fagan, S.C. Matrix metalloproteinase-9 in an exploratory trial of intravenous minocycline for acute ischemic stroke. Stroke 2011, 42, 2633–2635. [Google Scholar] [CrossRef] [PubMed]
- Knecht, T.; Story, J.; Liu, J.; Davis, W.; Borlongan, C.V.; Dela Peña, I.C. Adjunctive Therapy Approaches for Ischemic Stroke: Innovations to Expand Time Window of Treatment. Int. J. Mol. Sci. 2017, 18, 2756. [Google Scholar] [CrossRef]
- Tian, D.C.; Shi, K.; Zhu, Z.; Yao, J.; Yang, X.; Su, L.; Zhang, S.; Zhang, M.; Gonzales, R.J.; Liu, Q.; et al. Fingolimod enhances the efficacy of delayed alteplase administration in acute ischemic stroke by promoting anterograde reperfusion and retrograde collateral flow. Ann. Neurol. 2018, 84, 717–728. [Google Scholar] [CrossRef]
- Salas-Perdomo, A.; Miró-Mur, F.; Gallizioli, M.; Brait, V.H.; Justicia, C.; Meissner, A.; Urra, X.; Chamorro, A.; Planas, A.M. Role of the S1P pathway and inhibition by fingolimod in preventing hemorrhagic transformation after stroke. Sci. Rep. 2019, 9, 8309. [Google Scholar] [CrossRef]
- Zhu, Z.; Fu, Y.; Tian, D.; Sun, N.; Han, W.; Chang, G.; Dong, Y.; Xu, X.; Liu, Q.; Huang, D.; et al. Combination of the Immune Modulator Fingolimod With Alteplase in Acute Ischemic Stroke: A Pilot Trial. Circulation 2015, 132, 1104–1112. [Google Scholar] [CrossRef]
- Beller, E.; Reuter, L.; Kluge, A.; Preibisch, C.; Lindauer, U.; Bogdanov, A.; Lämmer, F.; Delbridge, C.; Matiasek, K.; Schwaiger, B.J.; et al. Pilot study to assess visualization and therapy of inflammatory mechanisms after vessel reopening in a mouse stroke model. Sci. Rep. 2018, 8, 745. [Google Scholar] [CrossRef]
- Maeda, M.; Furuichi, Y.; Noto, T.; Matsuoka, N.; Mutoh, S.; Yoneda, Y. Tacrolimus (FK506) suppresses rt-PA-induced hemorrhagic transformation in a rat thrombotic ischemia stroke model. Brain Res. 2009, 1254, 99–108. [Google Scholar] [CrossRef]
- Zhang, L.; Chopp, M.; Jia, L.; Cui, Y.; Lu, M.; Zhang, Z.G. Atorvastatin extends the therapeutic window for tPA to 6 h after the onset of embolic stroke in rats. J. Cereb. Blood Flow Metab. 2009, 29, 1816–1824. [Google Scholar] [CrossRef]
- Lu, D.; Liu, Y.; Mai, H.; Zang, J.; Shen, L.; Zhang, Y.; Xu, A. Rosuvastatin Reduces Neuroinflammation in the Hemorrhagic Transformation After rt-PA Treatment in a Mouse Model of Experimental Stroke. Front. Cell. Neurosci. 2018, 12, 225. [Google Scholar] [CrossRef] [PubMed]
- Yin, B.; Li, D.D.; Xu, S.Y.; Huang, H.; Lin, J.; Sheng, H.S.; Fang, J.H.; Song, J.N.; Zhang, M. Simvastatin pretreatment ameliorates t-PA-induced hemorrhage transformation and MMP-9/TIMP-1 imbalance in thromboembolic cerebral ischemic rats. Neuropsychiatr. Dis. Treat. 2019, 15, 1993–2002. [Google Scholar] [CrossRef] [PubMed]
- Cuartero, M.I.; Ballesteros, I.; Moraga, A.; Nombela, F.; Vivancos, J.; Hamilton, J.A.; Corbí, Á.L.; Lizasoain, I.; Moro, M.A. N2 neutrophils, novel players in brain inflammation after stroke: Modulation by the PPARγ agonist rosiglitazone. Stroke 2013, 44, 3498–3508. [Google Scholar] [CrossRef] [PubMed]
- Fujiwara, N.; Som, A.T.; Pham, L.D.; Lee, B.J.; Mandeville, E.T.; Lo, E.H.; Arai, K. A free radical scavenger edaravone suppresses systemic inflammatory responses in a rat transient focal ischemia model. Neurosci. Lett. 2016, 633, 7–13. [Google Scholar] [CrossRef]
- Lapergue, B.; Dang, B.Q.; Desilles, J.P.; Ortiz-Munoz, G.; Delbosc, S.; Loyau, S.; Louedec, L.; Couraud, P.O.; Mazighi, M.; Michel, J.B.; et al. High-density lipoprotein-based therapy reduces the hemorrhagic complications associated with tissue plasminogen activator treatment in experimental stroke. Stroke 2013, 44, 699–707. [Google Scholar] [CrossRef]
- Csiba, L. New findings provided by body and brain autopsy of 700 stroke patients with and without thrombolysis. In Proceedings of the Europen Academy of Stroke (EAN), Budapest, Hungary, 1 July 2023. [Google Scholar]
- Hudák, L.; Nagy, A.C.; Molnár, S.; Méhes, G.; Nagy, K.E.; Oláh, L.; Csiba, L. Discrepancies between clinical and autopsy findings in patients who had an acute stroke. Stroke Vasc. Neurol. 2022, 7, 215–221. [Google Scholar] [CrossRef]
- Szepesi, R.; Csokonay, Á.; Murnyák, B.; Kouhsari, M.C.; Hofgárt, G.; Csiba, L.; Hortobágyi, T. Haemorrhagic transformation in ischaemic stroke is more frequent than clinically suspected—A neuropathological study. J. Neurol. Sci. 2016, 368, 4–10. [Google Scholar] [CrossRef]
| Ischemic Phase | Revascularization Phase | |||
|---|---|---|---|---|
| cerebral blood flow | occlusion, decreased cerebral blood flow | acute elevation of cerebral blood flow = hyperemia | decreased blood flow = hypoperfusion | |
| permeability | transcellular increase | initial reperfusional permeability | first phase of biphasic permeability | second phase of biphasic permeability |
| onset | immediately | upon spontaneous opening or thrombolysis or MET (minutes, hours) | 3–8 (5) h | 18–96 (72) h |
| cause | failed intracellular homeostasis, mitochondrial dysfunction | acute opening of the BBB, loss of cerebral autoregulation | cerebral metabolic depletion, microvascular obstruction, insufficient nutritional support | increased inflammatory activity, angiogenesis |
| underlying mechanisms | ATP depletion, excitotoxic glutamate efflux from neurons | disassembly of tight junctions | inflammatory and oxidative stress on the BBB, ECM degradation | imperfect tight junction reassembly, new assembly fails to reach the original paracellular impermeability |
| type of edema | cytotoxic | vasogenic | ||
| Pre-Stroke Risk Factors | Potential Resolution |
|---|---|
| • hypertension | individualized treatment |
| • hyperglycemia | individualized treatment |
| • advanced age | primary prevention |
| • liver fibrosis | primary prevention, supportive treatment |
| • altered iron homeostasis | use of neuroprotective substances |
| • antithrombotic treatment | individualized treatment |
| • low platelet count and mean platelet volume | individualized treatment |
| Cranial CT, CTA Signs | |
| • early ischemic sign | shortening time till reperfusion therapy, potent edema decrease, individualized treatment |
| • hyperdense middle cerebral artery sign | |
| • calcification of cerebral vessels | |
| • poor collateral circulation | |
| Stroke Factors | |
| • severe form of ischemic stroke | careful patient selection for reperfusion therapy |
| • reperfusion therapy | careful observation, optimal management |
| Biomarker | Abbreviation | Physiological Role | Predictive Value |
|---|---|---|---|
| matrix metalloproteinase 9 | MMP9 | proteolytic enzyme | sensitive and specific marker |
| cellular fibronectin | c-Fn | major component of the ECM | sensitive and specific marker |
| ferritin | NA | acute phase protein | neither specific nor sensitive enough |
| neutrophil-to-lymphocyte ratio | NLR | blood cells | low discriminative ability |
| S100 calcium-binding protein B | S100B | glial-specific protein | sensitive but not specific |
| soluble ICAM-1 | sICAM-1 | adhesive protein involved in inflammatory responses | sensitive but not specific |
| vascular adhesion protein-1 | VAP-1 | adhesion molecule | sensitive but not specific |
| semicarbazide-sensitive amine oxidase | SSAO | enzyme |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kovács, K.B.; Bencs, V.; Hudák, L.; Oláh, L.; Csiba, L. Hemorrhagic Transformation of Ischemic Strokes. Int. J. Mol. Sci. 2023, 24, 14067. https://doi.org/10.3390/ijms241814067
Kovács KB, Bencs V, Hudák L, Oláh L, Csiba L. Hemorrhagic Transformation of Ischemic Strokes. International Journal of Molecular Sciences. 2023; 24(18):14067. https://doi.org/10.3390/ijms241814067
Chicago/Turabian StyleKovács, Kitti Bernadett, Viktor Bencs, Lilla Hudák, László Oláh, and László Csiba. 2023. "Hemorrhagic Transformation of Ischemic Strokes" International Journal of Molecular Sciences 24, no. 18: 14067. https://doi.org/10.3390/ijms241814067
APA StyleKovács, K. B., Bencs, V., Hudák, L., Oláh, L., & Csiba, L. (2023). Hemorrhagic Transformation of Ischemic Strokes. International Journal of Molecular Sciences, 24(18), 14067. https://doi.org/10.3390/ijms241814067





