Prediction of Oscillations in Glycolysis in Ethanol-Consuming Erythrocyte-Bioreactors
Abstract
1. Introduction
2. Results and Discussion
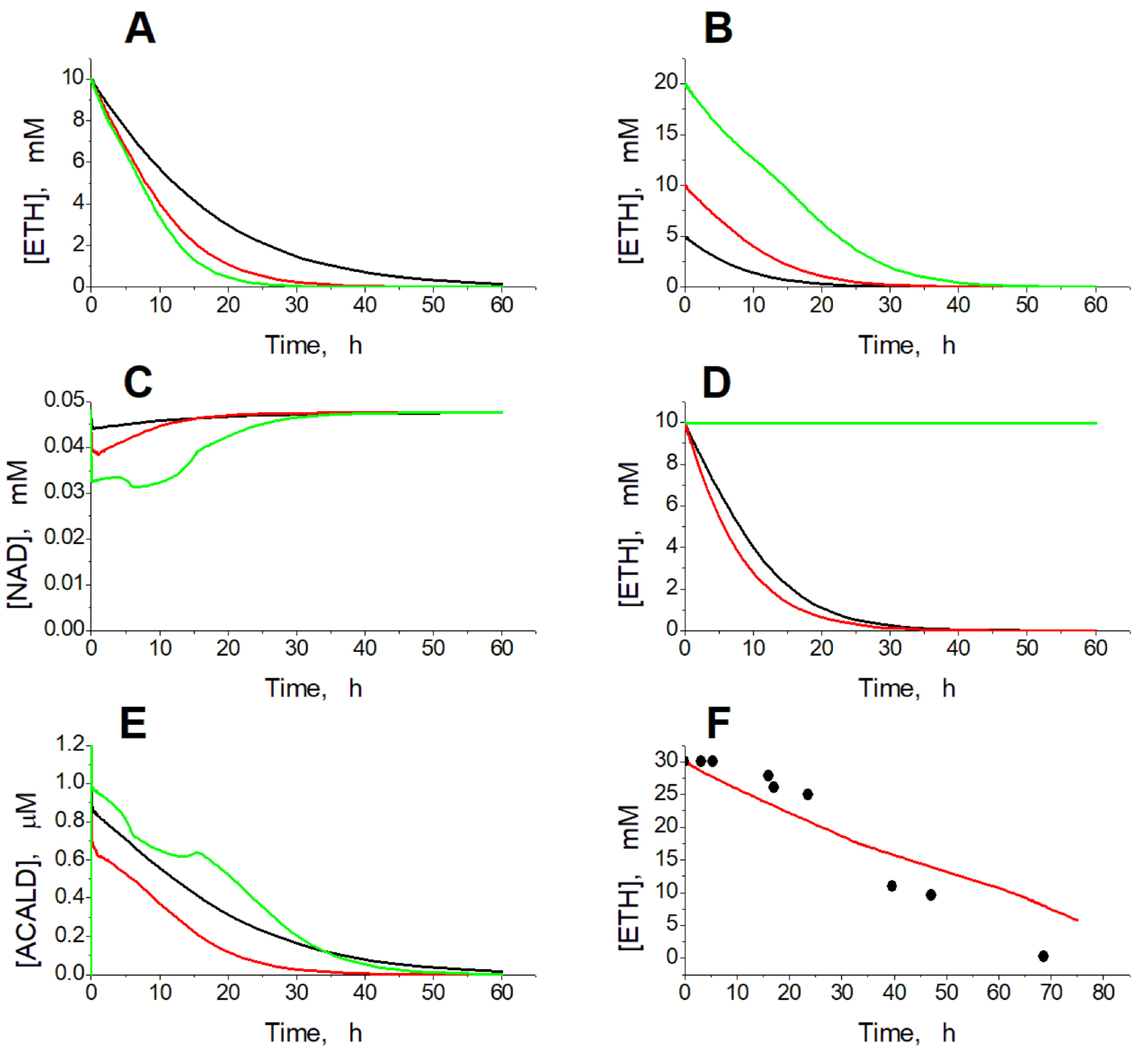
2.1. Kinetics of Ethanol Consumption
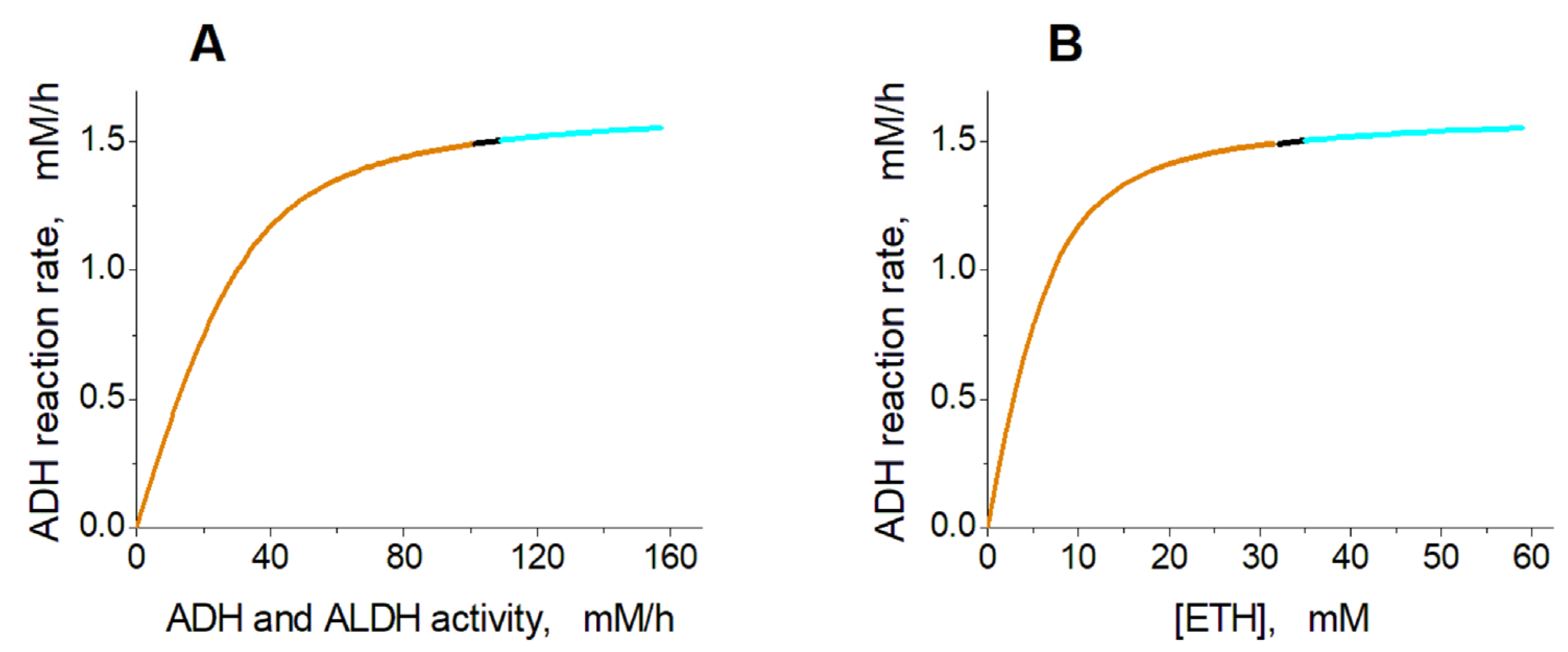
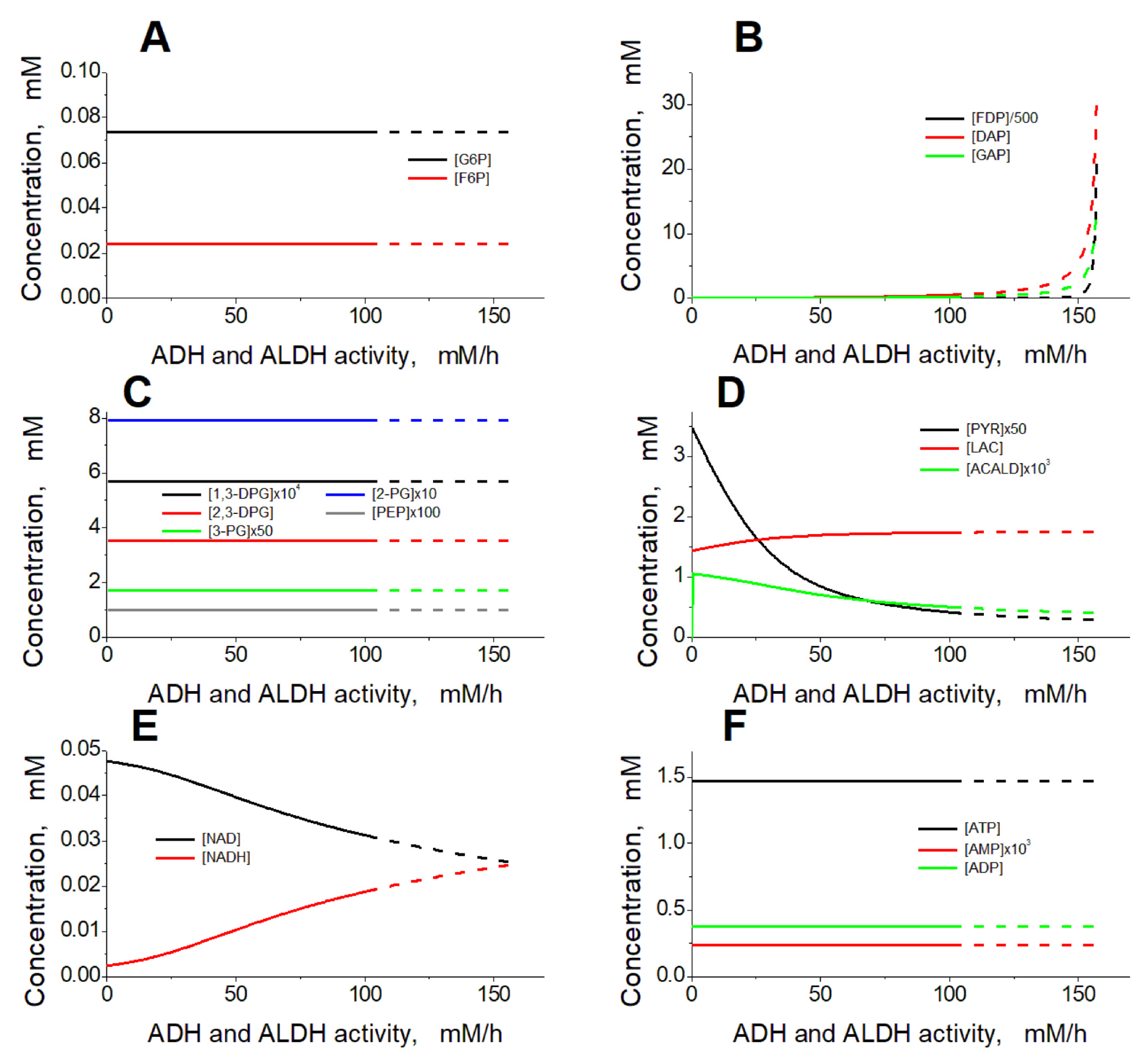
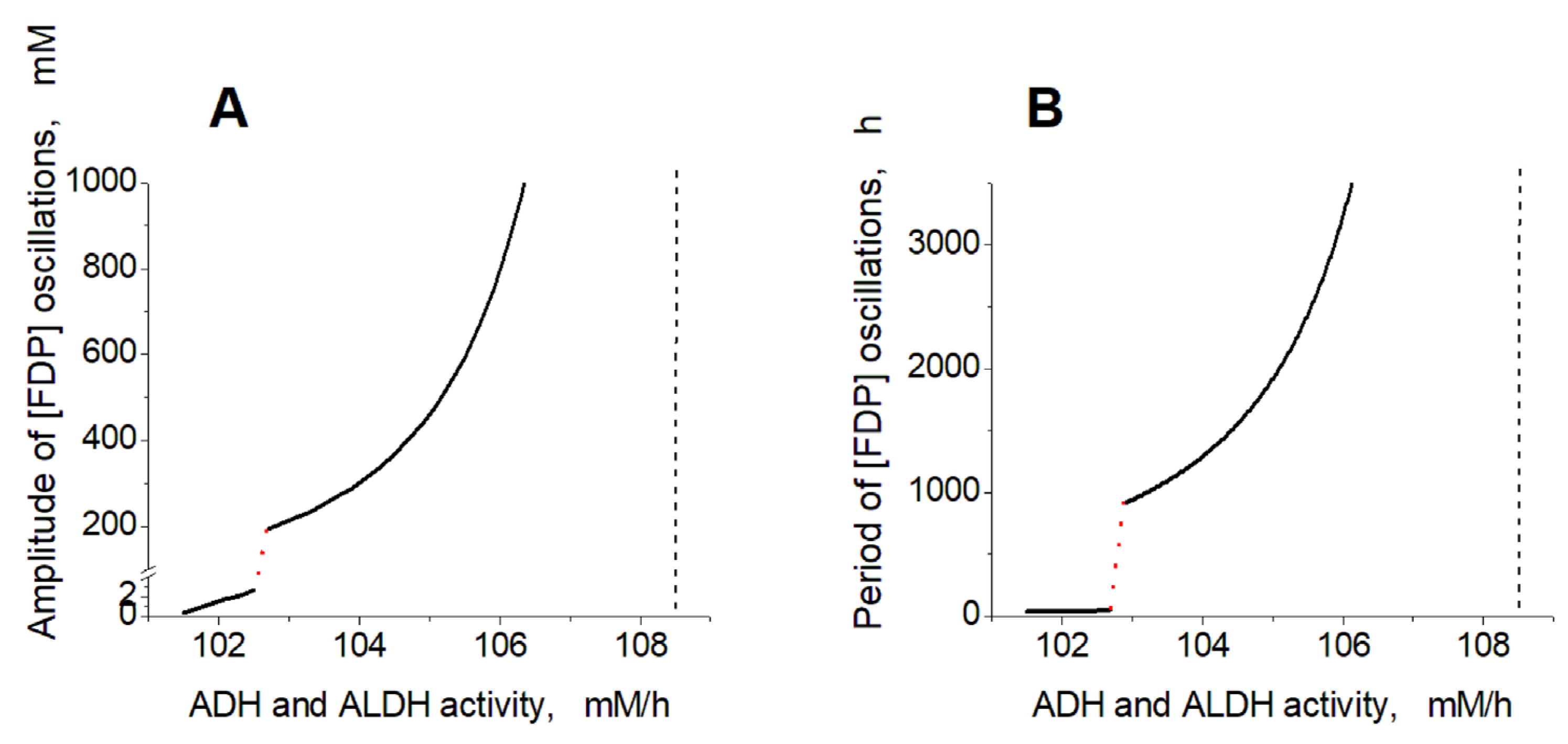
2.2. Effects of ADH and ALDH Activity
2.3. Effects of Ethanol Concentration
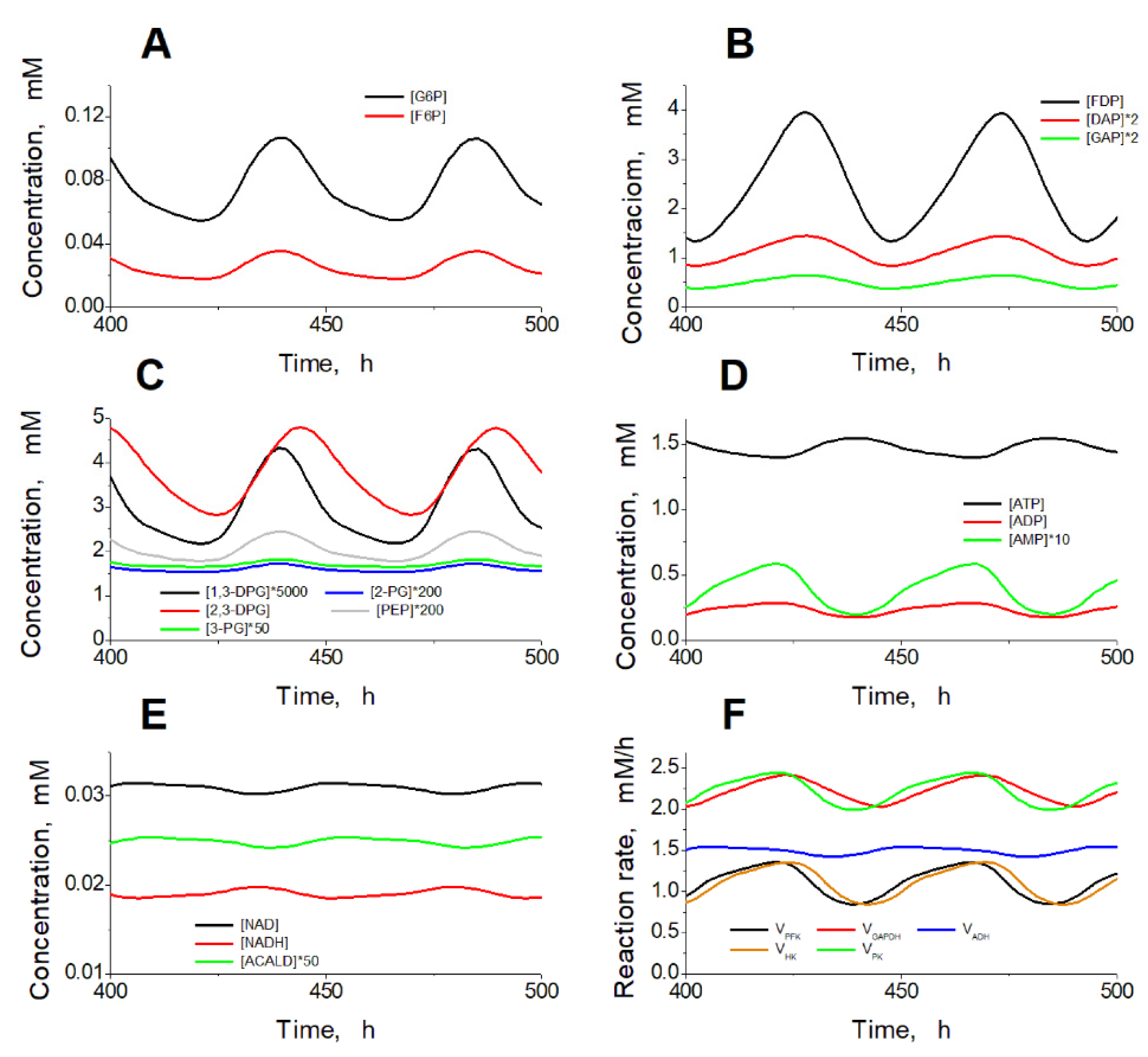
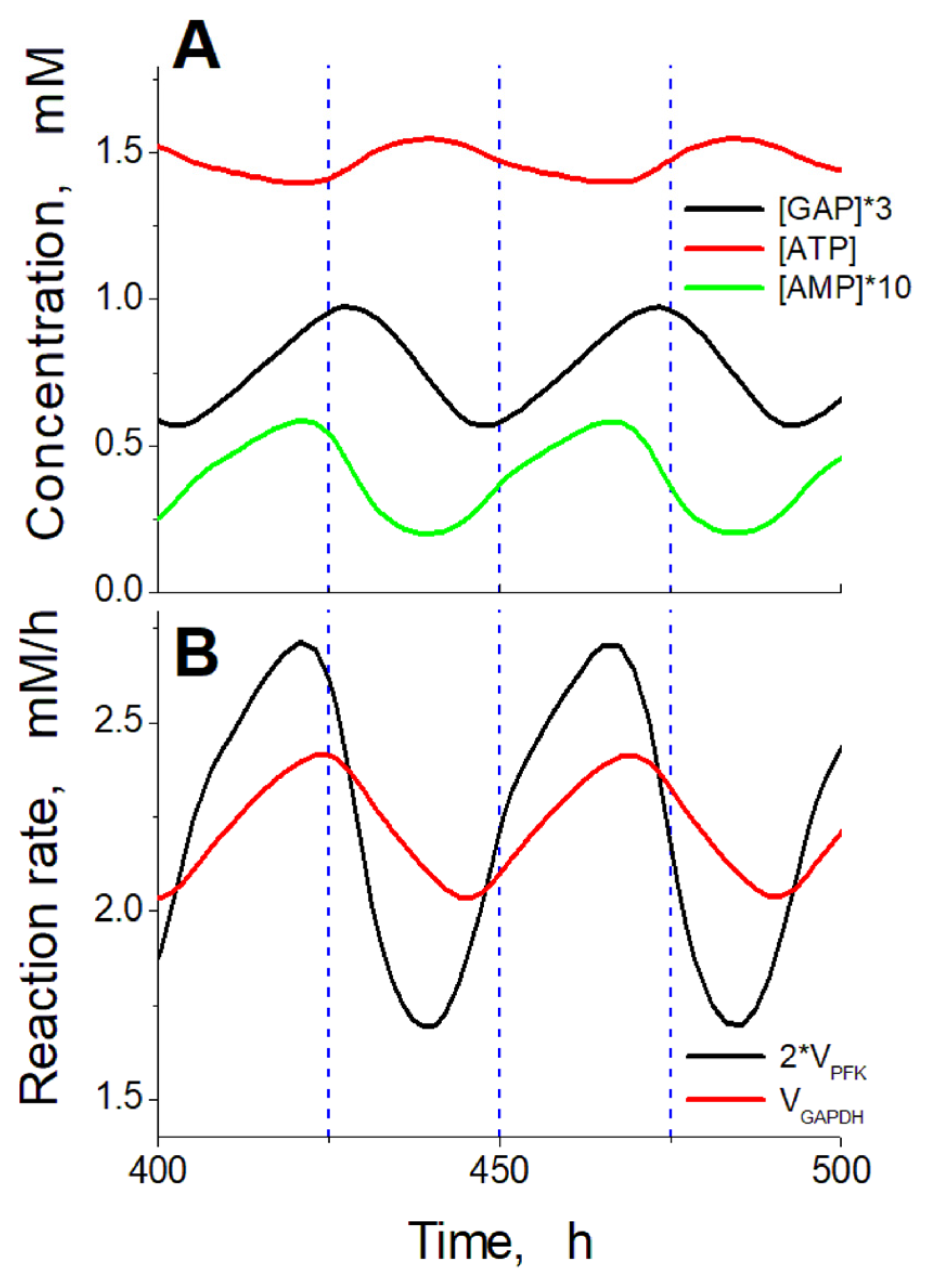
2.4. Mechanism of Oscillations
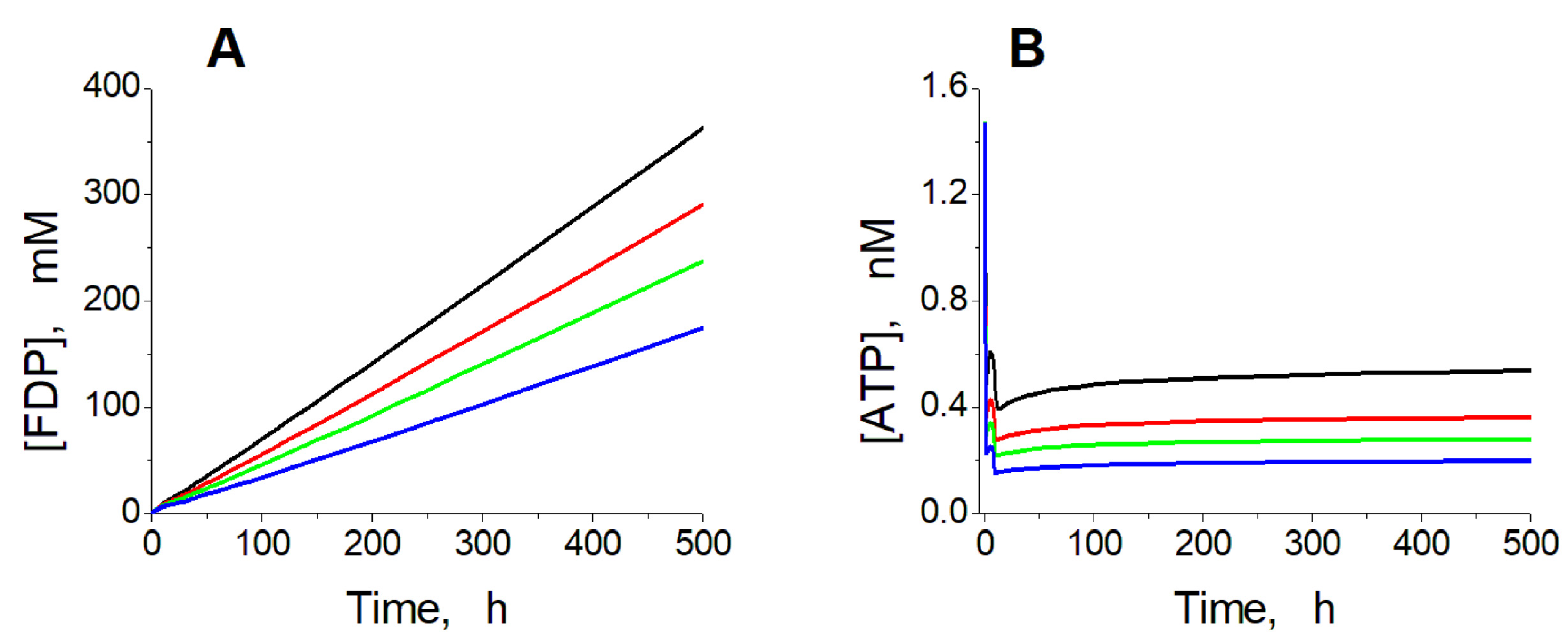
2.5. Accumulation of Glycolysis Intermediates
2.6. Conclusions
3. Mathematical Model
- The rate of the hexokinase reaction does not depend on glucose concentration because the normal physiological glucose concentration in the blood is significantly larger than the value of the hexokinase Michaelis constant for glucose [35].
- The concentration of orthophosphate is constant and equal to 1.0 mM [22].
- The extracellular concentrations of pyruvate ([PYR]ext) and lactate ([LAC]ext) are constant and equal to 0.07 and 1.2 mM, respectively [22].
- The extracellular concentration of acetate is zero.
- Acetaldehyde produced in the EBRs from ethanol does not leave the cells.
- The possible effect of the transmembrane potential on the transport of metabolites was not taken into account.
- The erythrocyte cell volume is constant.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bax, B.E. Erythrocytes as carriers of therapeutic enzymes. Pharmaceutics 2020, 12, 435. [Google Scholar] [CrossRef] [PubMed]
- Koleva, L.; Bovt, E.; Ataullakhanov, F.; Sinauridze, E. Erythrocytes as carriers: From drug delivery to biosensors. Pharmaceutics 2020, 12, 276. [Google Scholar] [CrossRef] [PubMed]
- Thomas, X.; Le Jeune, C. Erythrocyte encapsulated l-asparaginase (GRASPA) in acute leukemia. Int. J. Hematol. Oncol. 2016, 5, 11–25. [Google Scholar] [CrossRef] [PubMed]
- Lynggaard, L.S.; Vaitkeviciene, G.; Langenskiöld, C.; Lehmann, A.K.; Lähteenmäki, P.M.; Lepik, K.; El, H.I.; Schmiegelow, K.; Albertsen, B.K. Asparaginase encapsulated in erythrocytes as second-line treatment in hypersensitive patients with acute lymphoblastic leukaemia. Br. J. Haematol. 2022, 197, 745–754. [Google Scholar] [CrossRef] [PubMed]
- Bax, B.E.; Bain, M.D.; Fairbanks, L.D.; Webster, A.D.; Ind, P.W.; Hershfield, M.S.; Chalmers, R.A. A 9-yr evaluation of carrier erythrocyte encapsulated adenosine deaminase (ADA) therapy in a patient with adult type ADA deficiency. Eur. J. Haematol. 2007, 79, 338–348. [Google Scholar] [CrossRef]
- Levene, M.; Bain, M.D.; Moran, N.F.; Nirmalananthan, N.; Poulton, J.; Scarpelli, M.; Filosto, M.; Mandel, H.; MacKinnon, A.D.; Fairbanks, L.; et al. Safety and efficacy of erythrocyte encapsulated thymidine phosphorylase in mitochondrial neurogastrointestinal encephalomyopathy. J. Clin. Med. 2019, 8, 457. [Google Scholar] [CrossRef]
- Kipper, K.; Hecht, M.; Antunes, N.J.; Fairbanks, L.D.; Levene, M.; Kalkar Uçar, S.; Schaefer, A.; Blakely, E.L.; Bax, B.E. Quantification of plasma and urine thymidine and 2′-deoxyuridine by LC-MS/MS for the pharmacodynamic evaluation of erythrocyte encapsulated thymidine phosphorylase in patients with mitochondrial neurogastrointestinal encephalomyopathy. J. Clin. Med. 2020, 9, 788. [Google Scholar] [CrossRef]
- Sanz, S.; Lizano, C.; Luque, J.; Pinilla, M. In vitro and in vivo study of glutamate dehydrogenase encapsulated into mouse erythrocytes by a hypotonic dialysis procedure. Life Sci. 1999, 65, 2781–2789. [Google Scholar] [CrossRef]
- Kosenko, E.A.; Venediktova, N.I.; Kudryavtsev, A.A.; Ataullakhanov, F.I.; Kaminsky, Y.G.; Felipo, V.; Montoliu, C. Encapsulation of glutamine synthetase in mouse erythrocytes: A new procedure for ammonia detoxification. Biochem. Cell Biol. 2008, 86, 469–476. [Google Scholar] [CrossRef]
- Protasov, E.S.; Borsakova, D.V.; Alexandrovich, Y.G.; Korotkov, A.V.; Kosenko, E.A.; Butylin, A.A.; Ataullakhanov, F.I.; Sinauridze, E.I. Erythrocytes as bioreactors to decrease excess ammonium concentration in blood. Sci. Rep. 2019, 9, 1455. [Google Scholar] [CrossRef]
- Borsakova, D.V.; Koleva, L.D.; Protasov, E.S.; Ataullakhanov, F.I.; Sinauridze, E.I. Ammonium removal by erythrocyte-bioreactors based on glutamate dehydrogenase from Proteus sp. jointly with porcine heart alanine aminotransferase. Sci. Rep. 2022, 12, 5437. [Google Scholar] [CrossRef] [PubMed]
- Ninfali, P.; Rossi, L.; Baronciani, L.; Ropars, C.; Magnani, M. Acetaldehyde oxidation by aldehyde dehydrogenase loaded erythrocytes. Adv. Exp. Med. Biol. 1992, 326, 165–173. [Google Scholar] [CrossRef] [PubMed]
- Ninfali, P.; Rossi, L.; Baronciani, L.; Tirillini, B.; Ropars, C.; Magna, M. Acetaldehyde, ethanol and acetone concentrations in blood of alcohol-treated mice receiving aldehyde dehydrogenase-loaded erythrocytes. Alcohol Alcohol. 1992, 27, 19–23. [Google Scholar]
- Magnani, M.; Laguerre, M.; Rossi, L.; Bianchi, M.; Ninfali, P.; Mangani, F.; Ropars, C. In vivo accelerated acetaldehyde metabolism using acetaldehyde dehydrogenase-loaded erythrocytes. Alcohol Alcohol. 1990, 25, 627–637. [Google Scholar] [CrossRef]
- Lizano, C.; Sanz, S.; Luque, J.; Pinilla, M. In vitro study of alcohol dehydrogenase and acetaldehyde dehydrogenase encapsulated into human erythrocytes by an electroporation procedure. Biochim. Biophys. Acta-Gen. Subj. 1998, 1425, 328–336. [Google Scholar] [CrossRef]
- Lizano, C.; Pérez, M.T.; Pinilla, M. Mouse erythrocytes as carriers for coencapsulated alcohol and aldehyde dehydrogenase obtained by electroporation in vivo survival rate in circulation, organ distribution and ethanol degradation. Life Sci. 2001, 68, 2001–2016. [Google Scholar] [CrossRef]
- Alexandrovich, Y.G.; Kosenko, E.A.; Sinauridze, E.I.; Obydenny, S.I.; Kireev, I.I.; Ataullakhanov, F.I.; Kaminsky, Y.G. Rapid elimination of blood alcohol using erythrocytes: Mathematical modeling and in vitro study. BioMed Res. Int. 2017, 2017, 5849593. [Google Scholar] [CrossRef]
- Protasov, E.; Koleva, L.; Bovt, E.; Ataullakhanov, F.I.; Sinauridze, E. Theoretical analysis of the built-in metabolic pathway effect on the metabolism of erythrocyte-bioreactors that neutralize ammonium. Metabolites 2021, 11, 36. [Google Scholar] [CrossRef] [PubMed]
- Ataullakhanov, F.I.; Buravtsev, V.N.; Zhabotinskii, A.M.; Norina, S.B.; Pichugin, A.V.; Erlikh, L.I. Interaction of Embden-Meyerhof pathway and hexose monophosphate shunt in erythrocytes. Biochemistry 1981, 46, 604–610. (In English) [Google Scholar]
- World Health Organization. Alcohol, 23 May 2022. Available online: https://www.who.int/news-room/fact-sheets/detail/alcohol (accessed on 25 April 2023).
- MacGibbon, A.K.H.; Blackwell, L.F.; Buckley, P.D. Kinetics of sheep-liver cytoplasmic aldehyde dehydrogenase. Eur. J. Biochem. 1977, 77, 93–100. [Google Scholar] [CrossRef]
- Martinov, M.V.; Plotnikov, A.G.; Vitvitsky, V.M.; Ataullakhanov, F.I. Deficiencies of glycolytic enzymes as a possible cause of hemolytic anemia. Biochim. Biophys. Acta-Gen. Subj. 2000, 1474, 75–87. [Google Scholar] [CrossRef] [PubMed]
- Joshi, A.; Palsson, B.O. Metabolic dynamics in the human red cell. Part IV—Data prediction and some model computations. J. Theor. Biol. 1990, 142, 69–85. [Google Scholar] [CrossRef] [PubMed]
- Brüser, A.; Kirchberger, J.; Kloos, M.; Sträter, N.; Schöneberg, T. Functional linkage of adenine nucleotide binding sites in mammalian muscle 6-Phosphofructokinase. J. Biol. Chem. 2012, 287, 17546–17553. [Google Scholar] [CrossRef] [PubMed]
- Ataullakhanov, F.I.; Martinov, M.V.; Shi, Q.; Vitvitsky, V.M. Significance of two transmembrane ion gradients for human erythrocyte volume stabilization. PLoS ONE 2022, 17, e0272675. [Google Scholar] [CrossRef] [PubMed]
- Sel’kov, E.E. Self-oscillations in glycolysis 1. A simple kinetic model. Eur. J. Biochem. 1968, 4, 79–86. [Google Scholar] [CrossRef]
- Andrés, V.; Schultz, V.; Tornheim, K. Oscillatory synthesis of glucose 1,6-bisphosphate and frequency modulation of glycolytic oscillations in skeletal muscle extracts. J. Biol. Chem. 1990, 265, 21441–21447. [Google Scholar] [CrossRef]
- Gustavsson, A.-K.; van Niekerk, D.D.; Adiels, C.B.; du Preez, F.B.; Goksör, M.; Snoep, J.L. Sustained glycolytic oscillations in individual isolated yeast cells. FEBS J. 2012, 279, 2837–2847. [Google Scholar] [CrossRef]
- Weber, A.; Zuschratter, W.; Hauser, M.J.B. Partial synchronisation of glycolytic oscillations in yeast cell populations. Sci. Rep. 2020, 10, 19714. [Google Scholar] [CrossRef]
- Brechmann, P.; Rendall, A.D. Unbounded solutions of models for glycolysis. J. Math. Biol. 2021, 82, 1. [Google Scholar] [CrossRef]
- Halestrap, A.P. Transport of pyruvate and lactate into human erythrocytes. Evidence for the involvement of the chloride carrier and a chloride-independent carrier. Biochem. J. 1975, 156, 193–207. [Google Scholar] [CrossRef]
- Dickinson, F.M.; Monger, G.P. A study of the kinetics and mechanism of yeast alcohol dehydrogenase with a variety of substrates. Biochem. J. 1973, 131, 261–270. [Google Scholar] [CrossRef] [PubMed]
- Ganzhorn, A.J.; Green, D.W.; Hershey, A.D.; Gould, R.M.; Plapp, B.V. Kinetic characterization of yeast alcohol dehydrogenases. Amino acid residue 294 and substrate specificity. J. Biol. Chem. 1987, 262, 3754–3761. [Google Scholar] [CrossRef]
- Vives Corrons, J.L.; Pujades, M.A.; Aguilar i Bascompt, J.L.; Jou, J.M.; Rozman, C.; Ester, A. Pyrimidine 5′nucleotidase and several other red cell enzyme activities in β-thalassaemia trait. Br. J. Haematol. 1984, 56, 483–494. [Google Scholar] [CrossRef] [PubMed]
- Gerber, G.; Preissler, H.; Heinrich, R.; Rapoport, S.M. Hexokinase of human erythrocytes: Purification, kinetic model and its application to the conditions in the cell. Eur. J. Biochem. 1974, 45, 39–52. [Google Scholar] [CrossRef] [PubMed]
- Martinov, M.V.; Vitvitsky, V.M.; Ataullakhanov, F.I. Volume stabilization in human erythrocytes: Combined effects of Ca2+-dependent potassium channels and adenylate metabolism. Biophys. Chem. 1999, 80, 199–215. [Google Scholar] [CrossRef] [PubMed]
- Yeagle, P.L. Membrane Transport. In The Membranes of Cells, 3rd ed.; Elsevier: London, UK, 2016; Chapter 13; pp. 335–378. [Google Scholar] [CrossRef]
- Naccache, P.; Sha’afi, R.I. Patterns of nonelectrolyte permeability in human red blood cell membrane. J. Gen. Physiol. 1973, 62, 714–736. [Google Scholar] [CrossRef]
- Ren, S.; Das, A.; Lien, E. QSAR analysis of membrane permeability to organic compounds. J. Drug Target. 1996, 4, 103–107. [Google Scholar] [CrossRef]
- MATLAB Version: 9.9.0 (R2020b); The MathWorks Inc.: Natick, MA, USA, 2020; Available online: https://www.mathworks.com (accessed on 25 April 2023).

| Metabolite (a) | Concentration, mM | Metabolite (a) | Concentration, mM |
|---|---|---|---|
| G6P | 0.073 | PEP | 0.01 |
| F6P | 0.024 | PYR | 0.07 |
| FDP | 0.0084 | LAC | 1.43 |
| DAP | 0.034 | NAD | 0.048 |
| GAP | 0.015 | NADH | 0.002 |
| 1,3-DPG | 5.7 × 10−4 | ATP | 1.471 |
| 2,3-DPG | 3.5 | ADP | 0.235 |
| 3-PG | 0.034 | AMP | 0.038 |
| 2-PG | 0.008 | ACALD | 0 |
| № | Variable (a) | Equation (b) |
|---|---|---|
| 1 | [G6P] | |
| 2 | [F6P] | |
| 3 | [FDP] | |
| 4 | [DAP] | |
| 5 | [GAP] | |
| 6 | [1,3-DPG] | |
| 7 | [2,3-DPG] | |
| 8 | [3-PG] | |
| 9 | [2-PG] | |
| 10 | [PEP] | |
| 11 | [PYR] | |
| 12 | [LAC] | |
| 13 | [NAD] | |
| 14 | Energy charge (Φ) (c) | |
| 15 | [ETH] | |
| 16 | [ACALD] | |
| 17 | Adenylate pool | [ATP]+[ADP]+[AMP] = P |
| 18 | Adenylate kinase equilibrium | |
| 19 | NAD/NADH pool | [NAD] + [NADH] = N |
| Hexokinase [22] mM/h, mM, mM |
| Glucose-6-phosphate isomerase [22] mM/h, mM, mM, mM |
| Phosphofructokinase [22] mM/h, mM, mM, mM, mM, mM |
| Aldolase [22] mM/h, mM, mM2, mM, mM, mM, mM, mM |
| Triosephosphate isomerase [22,34] mM/h, mM, mM, mM |
| Glyceraldehyde phosphate dehydrogenase [22] mM/h, mM, mM, mM, mM, mM, mM |
| Phosphoglycerate kinase [22,34] mM/h, mM, mM, mM, mM, mM, mM, mM |
| Diphosphoglycerate mutase [22] mM/h, mM, mM |
| Diphosphoglycerate phosphatase [22] mM/h, mM, mM |
| Phosphoglycerate mutase [22] mM/h, mM, mM |
| Enolase [22,34] mM/h, mM, , mM |
| Pyruvate kinase [22] mM/h, mM, mM, mM |
| Lactate dehydrogenase [22] mM/h, mM, mM, , mM, mM, mM, mM |
| Na+/K+-ATPase [22] mM/h, [30] |
| All other ATPases are presented by the following equation [22] mM/h, mM |
| Alcohol dehydrogenase [32,33] ADH activity—this is a varied parameter in the model. |
| Acetaldehyde dehydrogenase [21] —ALDH activity—this is a varied parameter in the model |
| Pyruvate transport [31] ; ; |
| Lactate transport [31] ; ; |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Protasov, E.; Martinov, M.; Sinauridze, E.; Vitvitsky, V.; Ataullakhanov, F. Prediction of Oscillations in Glycolysis in Ethanol-Consuming Erythrocyte-Bioreactors. Int. J. Mol. Sci. 2023, 24, 10124. https://doi.org/10.3390/ijms241210124
Protasov E, Martinov M, Sinauridze E, Vitvitsky V, Ataullakhanov F. Prediction of Oscillations in Glycolysis in Ethanol-Consuming Erythrocyte-Bioreactors. International Journal of Molecular Sciences. 2023; 24(12):10124. https://doi.org/10.3390/ijms241210124
Chicago/Turabian StyleProtasov, Evgeniy, Michael Martinov, Elena Sinauridze, Victor Vitvitsky, and Fazoil Ataullakhanov. 2023. "Prediction of Oscillations in Glycolysis in Ethanol-Consuming Erythrocyte-Bioreactors" International Journal of Molecular Sciences 24, no. 12: 10124. https://doi.org/10.3390/ijms241210124
APA StyleProtasov, E., Martinov, M., Sinauridze, E., Vitvitsky, V., & Ataullakhanov, F. (2023). Prediction of Oscillations in Glycolysis in Ethanol-Consuming Erythrocyte-Bioreactors. International Journal of Molecular Sciences, 24(12), 10124. https://doi.org/10.3390/ijms241210124









