Twelve Weeks of High-Intensity Interval Training Alters Adipose Tissue Gene Expression but Not Oxylipin Levels in People with Non-Alcoholic Fatty Liver Disease
Abstract
1. Introduction
2. Results
2.1. Transcriptomic Analysis in Adipose Tissue
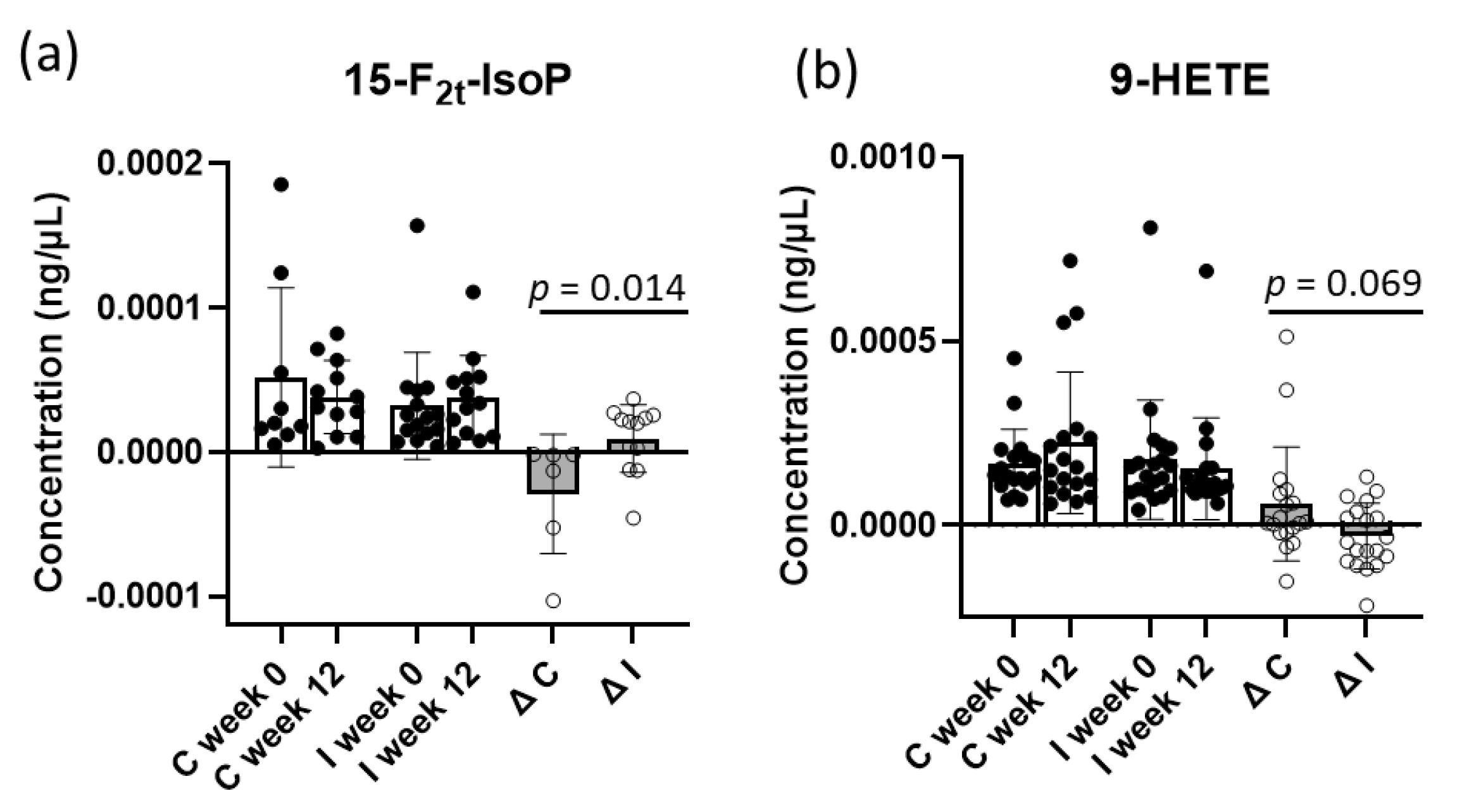
2.2. Oxylipins in Plasma
3. Discussion
3.1. AT Transcriptomics
3.2. Plasma Oxylipins
3.3. Perspectives
4. Materials and Methods
4.1. Transcriptomics in AT
4.2. Lipid Analysis
4.3. Targeted Oxylipins Analysis
4.4. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Riazi, K.; Azhari, H.; Charette, J.H.; Underwood, F.E.; King, J.A.; Afshar, E.E.; Swain, M.G.; Congly, S.E.; Kaplan, G.G.; Shaheen, A.A. The prevalence and incidence of NAFLD worldwide: A systematic review and meta-analysis. Lancet Gastroenterol. Hepatol. 2022, 7, 851–861. [Google Scholar] [CrossRef] [PubMed]
- Powell, E.E.; Wong, V.W.S.; Rinella, M. Non-alcoholic fatty liver disease. Lancet 2021, 397, 2212–2224. [Google Scholar] [CrossRef] [PubMed]
- Mantovani, A.; Petracca, G.; Beatrice, G.; Csermely, A.; Tilg, H.; Byrne, C.D.; Targher, G. Non-alcoholic fatty liver disease and increased risk of incident extrahepatic cancers: A meta-analysis of observational cohort studies. Gut 2022, 71, 778–788. [Google Scholar] [CrossRef]
- Simon, T.G.; Roelstraete, B.; Khalili, H.; Hagström, H.; Ludvigsson, J.F. Mortality in biopsy-confirmed nonalcoholic fatty liver disease: Results from a nationwide cohort. Gut 2021, 70, 1375–1382. [Google Scholar] [CrossRef] [PubMed]
- Pinyopornpanish, K.; Khoudari, G.; Saleh, M.A.; Angkurawaranon, C.; Pinyopornpanish, K.; Mansoor, E.; Dasarathy, S.; McCullough, A. Hepatocellular carcinoma in nonalcoholic fatty liver disease with or without cirrhosis: A population-based study. BMC Gastroenterol. 2021, 21, 394. [Google Scholar] [CrossRef]
- Marchesini, G.; Day, C.P.; Dufour, J.F.; Canbay, A.; Nobili, V.; Ratziu, V.; Tilg, H.; Roden, M.; Gastaldelli, A.; Yki-Järvinen, H.; et al. EASL–EASD–EASO Clinical Practice Guidelines for the management of non-alcoholic fatty liver disease. J. Hepatol. 2016, 64, 1388–1402. [Google Scholar] [CrossRef] [PubMed]
- Cigrovski Berkovic, M.; Bilic-Curcic, I.; Mrzljak, A.; Cigrovski, V. NAFLD and Physical Exercise: Ready, Steady, Go! Front. Nutr. 2021, 8, 734859. Available online: https://www.frontiersin.org/articles/10.3389/fnut.2021.734859 (accessed on 12 January 2023). [CrossRef]
- Babu, A.F.; Csader, S.; Lok, J.; Gómez-Gallego, C.; Hanhineva, K.; El-Nezami, H.; Schwab, U. Positive Effects of Exercise Intervention without Weight Loss and Dietary Changes in NAFLD-Related Clinical Parameters: A Systematic Review and Meta-Analysis. Nutrients 2021, 13, 3135. [Google Scholar] [CrossRef]
- Wang, Y.; Xu, D. Effects of aerobic exercise on lipids and lipoproteins. Lipids Health Dis. 2017, 16, 132. [Google Scholar] [CrossRef]
- Mann, S.; Beedie, C.; Jimenez, A. Differential effects of aerobic exercise, resistance training and combined exercise modalities on cholesterol and the lipid profile: Review, synthesis and recommendations. Sport. Med. 2014, 44, 211–221. [Google Scholar] [CrossRef]
- Malin, S.K.; Rynders, C.A.; Weltman, J.Y.; Barrett, E.J.; Weltman, A. Exercise Intensity Modulates Glucose-Stimulated Insulin Secretion when Adjusted for Adipose, Liver and Skeletal Muscle Insulin Resistance. PLoS ONE 2016, 11, e0154063. [Google Scholar] [CrossRef] [PubMed]
- Min, S.Y.; Learnard, H.; Kant, S.; Gealikman, O.; Rojas-Rodriguez, R.; DeSouza, T.; Desai, A.; Keaney, J.F., Jr.; Corvera, S.; Craige, S.M. Exercise Rescues Gene Pathways Involved in Vascular Expansion and Promotes Functional Angiogenesis in Subcutaneous White Adipose Tissue. Int. J. Mol. Sci. 2019, 20, 2046. [Google Scholar] [CrossRef] [PubMed]
- Ahn, C.; Ryan, B.J.; Schleh, M.W.; Varshney, P.; Ludzki, A.C.; Gillen, J.B.; Van Pelt, D.W.; Pitchford, L.M.; Howton, S.M.; Rode, T.; et al. Exercise training remodels subcutaneous adipose tissue in adults with obesity even without weight loss. J. Physiol. 2022, 600, 2127–2146. [Google Scholar] [CrossRef]
- Kure, T.; Mawatari, S.; Imamura, Y.; Oda, K.; Kumagai, K.; Hiramine, Y.; Miyahara, H.; Kanmura, S.; Moriuchi, A.; Uto, H.; et al. Nonalcoholic fatty liver disease is associated with both subcutaneous and visceral adiposity: A cross-sectional study. Medicine 2019, 98, e17879. [Google Scholar] [CrossRef]
- Azzu, V.; Vacca, M.; Virtue, S.; Allison, M.; Vidal-Puig, A. Adipose Tissue-Liver Cross Talk in the Control of Whole-Body Metabolism: Implications in Nonalcoholic Fatty Liver Disease. Gastroenterology 2020, 158, 1899–1912. [Google Scholar] [CrossRef] [PubMed]
- Cusi, K. Role of Obesity and Lipotoxicity in the Development of Nonalcoholic Steatohepatitis: Pathophysiology and Clinical Implications. Gastroenterology 2012, 142, 711–725. [Google Scholar] [CrossRef]
- Loomba, R.; Friedman, S.L.; Shulman, G.I. Mechanisms and disease consequences of nonalcoholic fatty liver disease. Cell 2021, 184, 2537–2564. [Google Scholar] [CrossRef]
- Tans, R.; Bande, R.; van Rooij, A.; Molloy, B.J.; Stienstra, R.; Tack, C.J.; Wevers, R.A.; Wessels, H.J.; Gloerich, J.; van Gool, A.J. Evaluation of cyclooxygenase oxylipins as potential biomarker for obesity-associated adipose tissue inflammation and type 2 diabetes using targeted multiple reaction monitoring mass spectrometry. Prostaglandins Leukot. Essent. Fat. Acids 2020, 160, 102157. [Google Scholar] [CrossRef]
- Feldstein, A.E.; Lopez, R.; Tamimi, T.A.R.; Yerian, L.; Chung, Y.M.; Berk, M.; Zhang, R.; McIntyre, T.M.; Hazen, S.L. Mass spectrometric profiling of oxidized lipid products in human nonalcoholic fatty liver disease and nonalcoholic steatohepatitis. J. Lipid Res. 2010, 51, 3046–3054. [Google Scholar] [CrossRef]
- Signini, É.F.; Nieman, D.C.; Silva, C.D.; Sakaguchi, C.A.; Catai, A.M. Oxylipin Response to Acute and Chronic Exercise: A Systematic Review. Metabolites 2020, 10, 264. [Google Scholar] [CrossRef]
- Pickens, C.A.; Sordillo, L.M.; Zhang, C.; Fenton, J.I. Obesity is positively associated with arachidonic acid-derived 5- and 11-hydroxyeicosatetraenoic acid (HETE). Metabolism 2017, 70, 177–191. [Google Scholar] [CrossRef] [PubMed]
- Shearer, G.C.; Walker, R.E. An overview of the biologic effects of omega-6 oxylipins in humans. Prostaglandins Leukot. Essent. Fat. Acids 2018, 137, 26–38. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Rempel, J.D.; Ball, T.B.; Aukema, H.; Minuk, G.Y. Plasma Oxylipins Levels in Nonalcoholic Fatty Liver Disease. Dig. Dis. Sci. 2020, 65, 3605–3613. [Google Scholar] [CrossRef]
- Nieman, D.C.; Pence, B.D. Exercise immunology: Future directions. J. Sport Health Sci. 2020, 9, 432–445. [Google Scholar] [CrossRef] [PubMed]
- Stanford, K.I.; Lynes, M.D.; Takahashi, H.; Baer, L.A.; Arts, P.J.; May, F.J.; Lehnig, A.C.; Middelbeek, R.J.; Richard, J.J.; So, K.; et al. 12,13-diHOME: An Exercise-Induced Lipokine that Increases Skeletal Muscle Fatty Acid Uptake. Cell Metab. 2018, 27, 1357. [Google Scholar] [CrossRef]
- Nieman, D.C.; Shanely, R.A.; Luo, B.; Meaney, M.P.; Dew, D.A.; Pappan, K.L. Metabolomics approach to assessing plasma 13- and 9-hydroxy-octadecadienoic acid and linoleic acid metabolite responses to 75-km cycling. Am. J. Physiol.-Regul. Integr. Comp. Physiol. 2014, 307, R68–R74. [Google Scholar] [CrossRef]
- Babu, A.F.; Csader, S.; Männistö, V.; Tauriainen, M.M.; Pentikäinen, H.; Savonen, K.; Klåvus, A.; Koistinen, V.; Hanhineva, K.; Schwab, U. Effects of exercise on NAFLD using non-targeted metabolomics in adipose tissue, plasma, urine, and stool. Sci. Rep. 2022, 12, 6485. [Google Scholar] [CrossRef]
- Pettersson, J.; Hindorf, U.; Persson, P.; Bengtsson, T.; Malmqvist, U.; Werkström, V.; Ekelund, M. Muscular exercise can cause highly pathological liver function tests in healthy men. Br. J. Clin. Pharmacol. 2008, 65, 253–259. [Google Scholar] [CrossRef]
- Khatri, P.; Neupane, A.; Sapkota, S.R.; Bashyal, B.; Sharma, D.; Chhetri, A.; Chirag, K.C.; Banjade, A.; Sapkota, P.; Bhandari, S. Strenuous Exercise-Induced Tremendously Elevated Transaminases Levels in a Healthy Adult: A Diagnostic Dilemma. Case Rep. Hepatol. 2021, 2021, 6653266. [Google Scholar] [CrossRef]
- Lipinski, M.J.; Dewey, F.E.; Biondi-Zoccai, G.G.; Abbate, A.; Vetrovec, G.W.; Froelicher, V.F. Hemoglobin levels predict exercise performance, ST-segment depression, and outcome in patients referred for routine exercise treadmill testing. Clin. Cardiol. 2009, 32, E22–E31. [Google Scholar] [CrossRef]
- da Silva, V.; Corrêa, H.; Neves, R.; Deus, L.; Reis, A.; Souza, M.; Dos Santos, C.; de Castro, D.; Honorato, F.; Simões, H.; et al. Impact of Low Hemoglobin on Body Composition, Strength, and Redox Status of Older Hemodialysis Patients Following Resistance Training. Front. Physiol. 2021, 12, 619054. Available online: https://www.frontiersin.org/articles/10.3389/fphys.2021.619054 (accessed on 2 May 2023). [CrossRef] [PubMed]
- Jannah, K. The effect of jogging exercise to improve hemoglobin levels. J. Phys. Conf. Ser. 2020, 1481, 012028. [Google Scholar]
- Facchini, F.S.; Carantoni, M.; Jeppesen, J.; Reaven, G.M. Hematocrit and hemoglobin are independently related to insulin resistance and compensatory hyperinsulinemia in healthy, non-obese men and women. Metabolism 1998, 47, 831–835. [Google Scholar] [CrossRef]
- Böttiger, L.E.; Carlson, L.A. Relation between Serum Cholesterol and Triglyceride Concentration and Haemoglobin Values in Non-anaemic Healthy Persons. Br. Med. J. 1972, 3, 731. [Google Scholar] [CrossRef] [PubMed]
- Hämäläinen, P.; Saltevo, J.; Kautiainen, H.; Mäntyselkä, P.; Vanhala, M. Erythropoietin, ferritin, haptoglobin, hemoglobin and transferrin receptor in metabolic syndrome: A case control study. Cardiovasc. Diabetol. 2012, 11, 116. [Google Scholar] [CrossRef]
- Koivula, T.; Lempiäinen, S.; Laine, S.; Sjöros, T.; Vähä-Ypyä, H.; Garthwaite, T.; Löyttyniemi, E.; Sievänen, H.; Vasankari, T.; Knuuti, J.; et al. Cross-Sectional Associations of Body Adiposity, Sedentary Behavior, and Physical Activity with Hemoglobin and White Blood Cell Count. Int. J. Environ. Res. Public Health 2022, 19, 14347. [Google Scholar] [CrossRef]
- Tapio, J.; Vähänikkilä, H.; Kesäniemi, Y.A.; Ukkola, O.; Koivunen, P. Higher hemoglobin levels are an independent risk factor for adverse metabolism and higher mortality in a 20-year follow-up. Sci. Rep. 2021, 11, 19936. [Google Scholar] [CrossRef]
- Auvinen, J.; Tapio, J.; Karhunen, V.; Kettunen, J.; Serpi, R.; Dimova, E.Y.; Gill, D.; Soininen, P.; Tammelin, T.; Mykkänen, J.; et al. Systematic evaluation of the association between hemoglobin levels and metabolic profile implicates beneficial effects of hypoxia. Sci. Adv. 2021, 7, eabi4822. [Google Scholar] [CrossRef]
- Thom, C.S.; Wilken, M.B.; Chou, S.T.; Voight, B.F. Body mass index and adipose distribution have opposing genetic impacts on human blood traits. eLife 2022, 11, e75317. [Google Scholar] [CrossRef]
- Shalev, H.; Kapelushnik, J.; Moser, A.; Knobler, H.; Tamary, H. Hypocholesterolemia in chronic anemias with increased erythropoietic activity. Am. J. Hematol. 2007, 82, 199–202. [Google Scholar] [CrossRef]
- Spencer, M.; Yao-Borengasser, A.; Unal, R.; Rasouli, N.; Gurley, C.M.; Zhu, B.; Peterson, C.A.; Kern, P.A. Adipose tissue macrophages in insulin-resistant subjects are associated with collagen VI and fibrosis and demonstrate alternative activation. Am. J. Physiol. Endocrinol. Metab. 2010, 299, E1016–E1027. [Google Scholar] [CrossRef] [PubMed]
- Pasarica, M.; Gowronska-Kozak, B.; Burk, D.; Remedios, I.; Hymel, D.; Gimble, J.; Ravussin, E.; Bray, G.A.; Smith, S.R. Adipose Tissue Collagen VI in Obesity. J. Clin. Endocrinol. Metab. 2009, 94, 5155–5162. [Google Scholar] [CrossRef]
- Li, M.; Liu, L.; Kang, Y.; Huang, S.; Xiao, Y. Circulating thrombospondin 1 (THBS1): A a risk factor for Non-Alcoholic Fatty Liver Disease in obese children. ANM 2022, 79, 16–28. Available online: https://www.karger.com/Article/FullText/527780 (accessed on 5 December 2022).
- Matsuo, Y.; Tanaka, M.; Yamakage, H.; Sasaki, Y.; Muranaka, K.; Hata, H.; Ikai, I.; Shimatsu, A.; Inoue, M.; Chun, T.H.; et al. Thrombospondin 1 as a novel biological marker of obesity and metabolic syndrome. Metabolism 2015, 64, 1490–1499. [Google Scholar] [CrossRef] [PubMed]
- Kiefer, F.W.; Zeyda, M.; Todoric, J.; Huber, J.; Geyeregger, R.; Weichhart, T.; Aszmann, O.; Ludvik, B.; Silberhumer, G.R.; Prager, G.; et al. Osteopontin Expression in Human and Murine Obesity: Extensive Local Up-Regulation in Adipose Tissue but Minimal Systemic Alterations. Endocrinology 2008, 149, 1350–1357. [Google Scholar] [CrossRef]
- Nomiyama, T.; Perez-Tilve, D.; Ogawa, D.; Gizard, F.; Zhao, Y.; Heywood, E.B.; Jones, K.L.; Kawamori, R.; Cassis, L.A.; Tschöp, M.H.; et al. Osteopontin mediates obesity-induced adipose tissue macrophage infiltration and insulin resistance in mice. J. Clin. Investig. 2007, 117, 2877–2888. [Google Scholar] [CrossRef]
- Deng, T.; Lyon, C.J.; Minze, L.J.; Lin, J.; Zou, J.; Liu, J.Z.; Ren, Y.; Yin, Z.; Hamilton, D.J.; Reardon, P.R.; et al. Class II major histocompatibility complex plays an essential role in obesity-induced adipose inflammation. Cell Metab. 2013, 17, 411–422. [Google Scholar] [CrossRef]
- Pessentheiner, A.R.; Ducasa, G.M.; Gordts, P.L.S.M. Proteoglycans in Obesity-Associated Metabolic Dysfunction and Meta-Inflammation. Front. Immunol. 2020, 11, 769. Available online: https://www.frontiersin.org/articles/10.3389/fimmu.2020.00769 (accessed on 6 December 2022). [CrossRef]
- Han, C.Y.; Kang, I.; Harten, I.A.; Gebe, J.A.; Chan, C.K.; Omer, M.; Alonge, K.M.; den Hartigh, L.J.; Kjerulf, D.G.; Goodspeed, L.; et al. Adipocyte-Derived Versican and Macrophage-Derived Biglycan Control Adipose Tissue Inflammation in Obesity. Cell Rep. 2020, 31, 107818. [Google Scholar] [CrossRef]
- Tanimura, R.; Kobayashi, L.; Shirai, T.; Takemasa, T. Effects of exercise intensity on white adipose tissue browning and its regulatory signals in mice. Physiol. Rep. 2022, 10, e15205. [Google Scholar] [CrossRef]
- Mendham, A.E.; Larsen, S.; George, C.; Adams, K.; Hauksson, J.; Olsson, T.; Fortuin-de Smidt, M.C.; Nono Nankam, P.A.; Hakim, O.; Goff, L.M.; et al. Exercise training results in depot-specific adaptations to adipose tissue mitochondrial function. Sci. Rep. 2020, 10, 3785. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, C.; Schneeweiss, P.; Randrianarisoa, E.; Schnauder, G.; Kappler, L.; Machann, J.; Schick, F.; Fritsche, A.; Heni, M.; Birkenfeld, A.; et al. Response of Mitochondrial Respiration in Adipose Tissue and Muscle to 8 Weeks of Endurance Exercise in Obese Subjects. J. Clin. Endocrinol. Metab. 2020, 105, e4023–e4037. [Google Scholar] [CrossRef] [PubMed]
- Pafili, K.; Kahl, S.; Mastrototaro, L.; Strassburger, K.; Pesta, D.; Herder, C.; Pützer, J.; Dewidar, B.; Hendlinger, M.; Granata, C.; et al. Mitochondrial respiration is decreased in visceral but not subcutaneous adipose tissue in obese individuals with fatty liver disease. J. Hepatol. 2022, 77, 1504–1514. [Google Scholar] [CrossRef] [PubMed]
- Böhm, A.; Keuper, M.; Meile, T.; Zdichavsky, M.; Fritsche, A.; Häring, H.U.; de Angelis, M.H.; Staiger, H.; Franko, A. Increased mitochondrial respiration of adipocytes from metabolically unhealthy obese compared to healthy obese individuals. Sci. Rep. 2020, 10, 12407. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Strable, M.S.; Ntambi, J.M. Stearoyl CoA Desaturase 1: Role in Cellular Inflammation and Stress. Adv. Nutr. 2011, 2, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Warensjö, E.; Rosell, M.; Hellenius, M.L.; Vessby, B.; De Faire, U.; Risérus, U. Associations between estimated fatty acid desaturase activities in serum lipids and adipose tissue in humans: Links to obesity and insulin resistance. Lipids Health Dis. 2009, 8, 37. [Google Scholar] [CrossRef]
- Sjögren, P.; Sierra-Johnson, J.; Kallings, L.V.; Cederholm, T.; Kolak, M.; Halldin, M.; Brismar, K.; de Faire, U.; Hellénius, M.L.; Fisher, R.M. Functional changes in adipose tissue in a randomised controlled trial of physical activity. Lipids Health Dis. 2012, 11, 80. [Google Scholar] [CrossRef]
- Nikolaidis, M.G.; Mougios, V. Effects of Exercise on the Fatty-Acid Composition of Blood and Tissue Lipids. Sport. Med. 2004, 34, 1051–1076. [Google Scholar] [CrossRef]
- de Melo, D.G.; Anaruma, C.P.; da Cruz Rodrigues, K.C.; Pereira, R.M.; de Campos, T.D.P.; Canciglieri, R.S.; Ramos, C.O.; Cintra, D.E.; Ropelle, E.R.; da Silva, A.S. Strength training alters the tissue fatty acids profile and slightly improves the thermogenic pathway in the adipose tissue of obese mice. Sci. Rep. 2022, 12, 6913. [Google Scholar] [CrossRef]
- Man, W.C.; Miyazaki, M.; Chu, K.; Ntambi, J. Colocalization of SCD1 and DGAT2: Implying preference for endogenous monounsaturated fatty acids in triglyceride synthesis. J. Lipid Res. 2006, 47, 1928–1939. [Google Scholar] [CrossRef]
- Bhatt-Wessel, B.; Jordan, T.W.; Miller, J.H.; Peng, L. Role of DGAT enzymes in triacylglycerol metabolism. Arch. Biochem. Biophys. 2018, 655, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Palmisano, B.T.; Zhu, L.; Stafford, J.M. Role of Estrogens in the Regulation of Liver Lipid Metabolism. Adv. Exp. Med. Biol. 2017, 1043, 227–256. [Google Scholar] [PubMed]
- Di Cesare, F.; Luchinat, C.; Tenori, L.; Saccenti, E. Age- and Sex-Dependent Changes of Free Circulating Blood Metabolite and Lipid Abundances, Correlations, and Ratios. J. Gerontol. Ser. A 2022, 77, 918–926. [Google Scholar] [CrossRef]
- Palmisano, B.T.; Zhu, L.; Eckel, R.H.; Stafford, J.M. Sex differences in lipid and lipoprotein metabolism. Mol. Metab. 2018, 15, 45–55. [Google Scholar] [CrossRef] [PubMed]
- Magkos, F.; Patterson, B.W.; Mohammed, B.S.; Klein, S.; Mittendorfer, B. Women produce fewer but triglyceride-richer very low-density lipoproteins than men. J. Clin. Endocrinol. Metab. 2007, 92, 1311–1318. [Google Scholar] [CrossRef] [PubMed]
- Côté, I.; Yasari, S.; Pighon, A.; Barsalani, R.; Rabasa-Lhoret, R.; Prud’homme, D.; Lavoie, J.M. Liver fat accumulation may be dissociated from adiposity gain in ovariectomized rats. Climacteric 2012, 15, 594–601. [Google Scholar] [CrossRef]
- Matsuo, K.; Gualtieri, M.R.; Cahoon, S.S.; Jung, C.E.; Paulson, R.J.; Shoupe, D.; Muderspach, L.I.; Wakatsuki, A.; Wright, J.D.; Roman, L.D. Surgical menopause and increased risk of nonalcoholic fatty liver disease in endometrial cancer. Menopause 2016, 23, 189–196. [Google Scholar] [CrossRef]
- DiStefano, J.K. NAFLD and NASH in Postmenopausal Women: Implications for Diagnosis and Treatment. Endocrinology 2020, 161, bqaa134. [Google Scholar] [CrossRef]
- Morrow, J.D.; Hill, K.E.; Burk, R.F.; Nammour, T.M.; Badr, K.F.; Roberts, L.J. A series of prostaglandin F2-like compounds are produced in vivo in humans by a non-cyclooxygenase, free radical-catalyzed mechanism. Proc. Natl. Acad. Sci. USA 1990, 87, 9383–9387. [Google Scholar] [CrossRef]
- Roberts, L.J.; Morrow, J.D. Measurement of F2-isoprostanes as an index of oxidative stress in vivo. Free. Radic. Biol. Med. 2000, 28, 505–513. [Google Scholar] [CrossRef]
- Milne, G.L.; Dai, Q.; Roberts, L.J. The isoprostanes—25 years later. Biochim. Et Biophys. Acta (BBA)—Mol. Cell Biol. Lipids 2015, 1851, 433–445. [Google Scholar] [CrossRef] [PubMed]
- Alejandra García-Flores, L.; Medina, S.; Gómez, C.; Wheelock, C.E.; Cejuela, R.; Miguel Martínez-Sanz, J.; Oger, C.; Galano, J.M.; Durand, T.; Hernández-Sáez, Á.; et al. Aronia—Citrus juice (polyphenol-rich juice) intake and elite triathlon training: A lipidomic approach using representative oxylipins in urine. Food Funct. 2018, 9, 463–475. [Google Scholar] [CrossRef] [PubMed]
- Nikolaidis, M.G.; Kyparos, A.; Vrabas, I.S. F2-isoprostane formation, measurement and interpretation: The role of exercise. Prog. Lipid Res. 2011, 50, 89–103. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B.; Lee, C.Y.J. Using isoprostanes as biomarkers of oxidative stress: Some rarely considered issues. Antioxid. Redox Signal. 2010, 13, 145–156. [Google Scholar] [CrossRef] [PubMed]
- Barr, J.; Caballería, J.; Martínez-Arranz, I.; Domínguez-Díez, A.; Alonso, C.; Muntané, J.; Pérez-Cormenzana, M.; García-Monzón, C.; Mayo, R.; Martín-Duce, A.; et al. Obesity dependent metabolic signatures associated with nonalcoholic fatty liver disease progression. J. Proteome Res. 2012, 11, 2521–2532. [Google Scholar] [CrossRef] [PubMed]
- Gollasch, B.; Dogan, I.; Rothe, M.; Gollasch, M.; Luft, F.C. Maximal exercise and plasma cytochrome P450 and lipoxygenase mediators: A lipidomics study. Physiol. Rep. 2019, 7, e14165. [Google Scholar] [CrossRef]
- Nieman, D.; Gillitt, N.; Chen, G.Y.; Zhang, G.; Sha, W.; Kay, C.; Chandra, P.; Kay, K.; Lila, M.A. Blueberry and Banana Consumption Mitigate Arachidonic, Cytochrome P450 Oxylipin Generation During Recovery from 75-Km Cycling. Curr. Dev. Nutr. 2020, 4 (Suppl. 2). [Google Scholar] [CrossRef]
- Nieman, D.C.; Gillitt, N.D.; Chen, G.Y.; Zhang, Q.; Sakaguchi, C.A.; Stephan, E.H. Carbohydrate intake attenuates post-exercise plasma levels of cytochrome P450-generated oxylipins. PLoS ONE 2019, 14, e0213676. [Google Scholar] [CrossRef]
- Koch, E.; Mainka, M.; Dalle, C.; Ostermann, A.I.; Rund, K.M.; Kutzner, L.; Froehlich, L.F.; Bertrand-Michel, J.; Gladine, C.; Schebb, N.H. Stability of oxylipins during plasma generation and long-term storage. Talanta 2020, 217, 121074. [Google Scholar] [CrossRef]
- Piercy, K.L.; Troiano, R.P.; Ballard, R.M.; Carlson, S.A.; Fulton, J.E.; Galuska, D.A.; George, S.M.; Olson, R.D. The Physical Activity Guidelines for Americans. JAMA 2018, 320, 2020–2028. [Google Scholar] [CrossRef]
- Macosko, E.Z.; Basu, A.; Satija, R.; Nemesh, J.; Shekhar, K.; Goldman, M.; Tirosh, I.; Bialas, A.R.; Kamitaki, N.; Martersteck, E.M.; et al. Highly Parallel Genome-wide Expression Profiling of Individual Cells Using Nanoliter Droplets. Cell 2015, 161, 1202–1214. [Google Scholar] [CrossRef]
- Robinson, M.D.; McCarthy, D.J.; Smyth, G.K. edgeR: A Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 2010, 26, 139–140. [Google Scholar] [CrossRef] [PubMed]
- Ritchie, M.E.; Phipson, B.; Wu, D.; Hu, Y.; Law, C.W.; Shi, W.; Smyth, G.K. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 2015, 43, e47. [Google Scholar] [CrossRef] [PubMed]
- Law, C.W.; Alhamdoosh, M.; Su, S.; Dong, X.; Tian, L.; Smyth, G.K.; Ritchie, M.E. RNA-seq analysis is easy as 1-2-3 with limma, Glimma and edgeR. F1000Research 2018, 5, 1408. [Google Scholar] [CrossRef]
- Yu, G.; Wang, L.G.; Han, Y.; He, Q.Y. clusterProfiler: An R Package for Comparing Biological Themes Among Gene Clusters. OMICS 2012, 16, 284–287. [Google Scholar] [CrossRef]
- Liberzon, A.; Subramanian, A.; Pinchback, R.; Thorvaldsdóttir, H.; Tamayo, P.; Mesirov, J.P. Molecular signatures database (MSigDB) 3.0. Bioinformatics 2011, 27, 1739–1740. [Google Scholar] [CrossRef] [PubMed]
- Yu, G.; Wang, L.G.; Yan, G.R.; He, Q.Y. DOSE: An R/Bioconductor package for disease ontology semantic and enrichment analysis. Bioinformatics 2015, 31, 608–609. [Google Scholar] [CrossRef] [PubMed]
- Hänzelmann, S.; Castelo, R.; Guinney, J. GSVA: Gene set variation analysis for microarray and RNA-Seq data. BMC Bioinform. 2013, 14, 7. [Google Scholar] [CrossRef]
- Ng, A.L.K.; Leung, H.H.; Kawasaki, R.; Ho, W.L.; Chow, L.L.W.; Chow, S.S.W.; Lee, J.C.-Y.; Wong, I.Y. Dietary Habits, Fatty Acids and Carotenoid Levels Are Associated with Neovascular Age-Related Macular Degeneration in Chinese. Nutrients 2019, 11, 1720. [Google Scholar] [CrossRef]
- Quehenberger, O.; Armando, A.M.; Dennis, E.A. High sensitivity quantitative lipidomics analysis of fatty acids in biological samples by gas chromatography-mass spectrometry. Biochim. Biophys. Acta 2011, 1811, 648–656. [Google Scholar] [CrossRef]
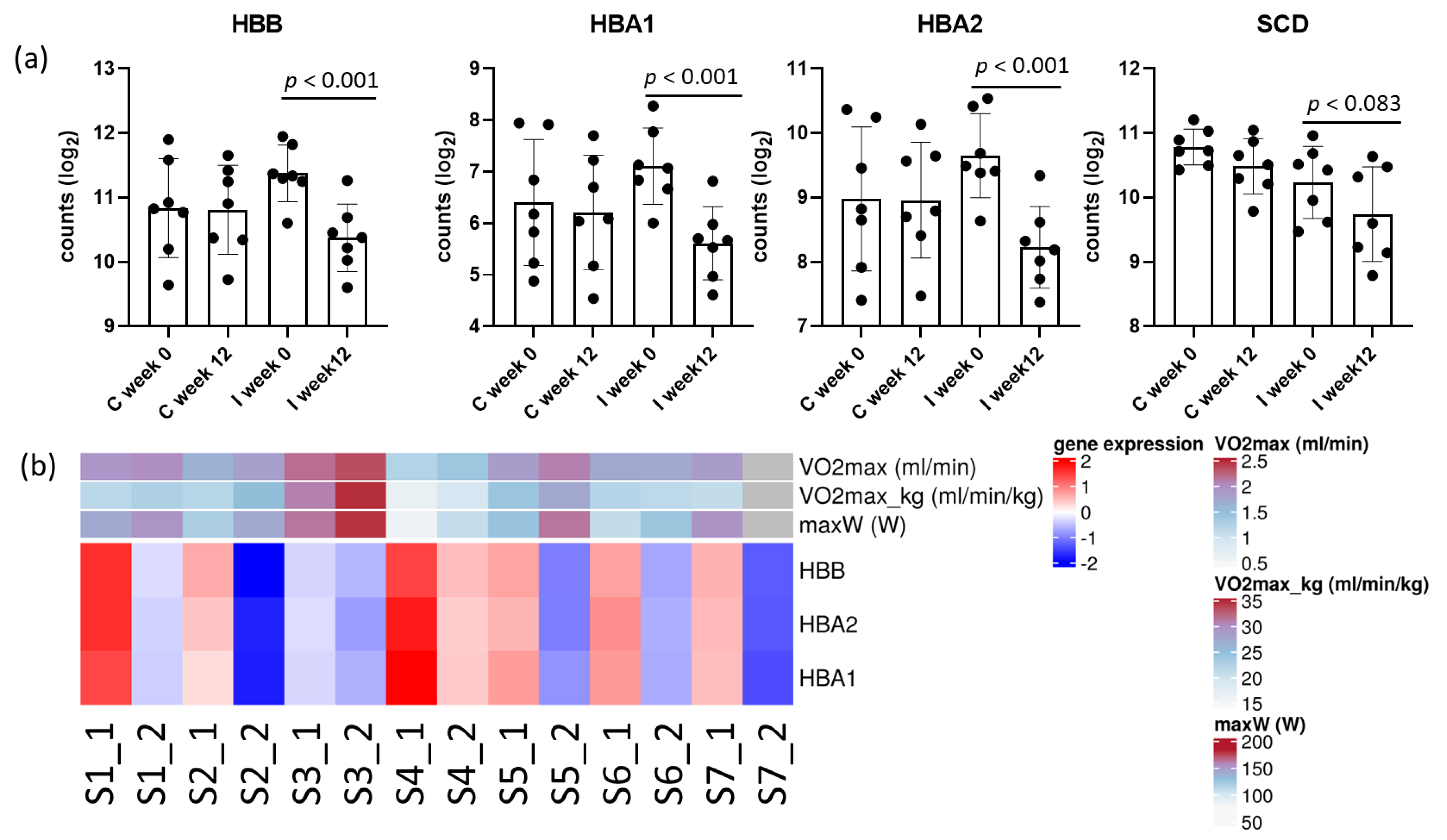


| Intervention (n = 7) | p-Value 1 | Control (n = 7) | p-Value 1 | p-Value 2 | |||
|---|---|---|---|---|---|---|---|
| W0 | W12 | W0 | W12 | ||||
| T2D | 1 | 0 | |||||
| Age, years | 56.9 ± 12.2 | 61.3 ± 7.1 | |||||
| BMI, kg/m2 | 29.3 ± 1.3 | 29.0 ± 1.6 | 0.310 | 31.1 ± 3.5 | 31.3 ± 3.4 | 0.310 | 0.225 |
| Fat mFass, kg | 29.4 ± 4.6 | 29.3 ± 5.2 | 0.553 | 34.9 ± 7.3 | 35.9 ± 7.2 | 0.034 | 0.085 |
| Fat mass, % | 37.5 ± 3.1 | 37.7 ± 3.6 | 0.885 | 40.8 ± 6.0 | 41.7 ± 6.1 | 0.046 | 0.159 |
| Visceral fat area, cm2 | 145 ± 29 | 145 ± 32 | 0.310 | 177 ± 43 | 181 ± 45 | 0.176 | 0.338 |
| IHL, % | 11.24 ± 9.93 | 11.38 ± 8.81 | 0.735 | 17.59 ± 12.69 | 17.66 ± 11.11 | 0.866 | 0.886 |
| ALT, U/L | 44.14 ± 20.47 | 52.14 ± 17.06 | 0.248 | 55.00 ± 25.27 | 46.29 ± 18.92 | 0.028 | 0.025 |
| AST, U/L | 31.71 ± 7.18 | 35.29 ± 11.77 | 0.350 | 38.43 ± 12.93 | 35.29 ± 14.29 | 0.051 | 0.062 |
| ALP, U/L | 80.29 ± 28.38 | 87.71 ± 36.40 | 0.307 | 78.57 ± 21.17 | 76.14 ± 21.98 | 0.446 | 0.178 |
| GGT, U/L | 105.00 ± 91.11 | 133.71 ± 144.26 | 0.237 | 104.14 ± 118.31 | 97.43 ± 130.90 | 0.237 | 0.125 |
| Cholesterol, mmol/L | 5.34 ± 0.93 | 5.37 ± 1.18 | 0.916 | 4.91 ± 0.57 | 4.66 ± 0.56 | 0.108 | 0.199 |
| HDL-C, mmol/L | 1.48 ± 0.29 | 1.51 ± 0.46 | 0.397 | 1.64 ± 0.44 | 1.54 ± 0.45 | 0.051 | 0.141 |
| LDL-C, mmol/L | 3.64 ± 0.92 | 3.51 ± 0.99 | 0.408 | 3.06 ± 0.74 | 2.79 ± 0.65 | 0.058 | 0.438 |
| TG, mmol/L | 1.73 ± 0.57 | 1.67 ± 0.38 | 0.612 | 1.55 ± 0.53 | 1.56 ± 0.53 | 0.999 | 0.654 |
| Gluc, mmol/L | 6.0 ± 0.6 | 6.0 ± 0.5 | 0.595 | 6.0 ± 0.7 | 6.1 ± 0.6 | 0.167 | 0.248 |
| Insulin, mU/L | 13.57 ± 5.2 | 14.73 ± 5.63 | 0.235 | 23.26 ± 13.22 | 24.46 ± 13.20 | 0.735 | 0.654 |
| HbA1c, mmol/mol | 37.0 ± 5.7 | 38.1 ± 4.8 | 0.223 | 37.7 ± 3.0 | 38.9 ± 2.0 | 0.302 | 0.999 |
| hs-CRP, mg/L | 1.36 ± 0.75 | 1.75 ± 1.05 | 0.206 | 2.63 ± 1.14 | 2.33 ± 1.67 | 0.599 | 0.305 |
| VO2max, mL/min | 1.76 ± 0.31 | 1.88 ± 0.31 | 0.046 | 1.86 ± 0.23 | 1.85 ± 0.24 | 0.735 | 0.031 |
| VO2max, mL/min/kg | 23.02 ± 4.43 | 25.14 ± 5.31 | 0.046 | 22.09 ± 3.68 | 21.61 ± 4.01 | 0.866 | 0.032 |
| maxW, watt | 124.50 ± 25.38 | 144.17 ± 23.88 | 0.027 | 127.57 ± 25.16 | 124.57 ± 25.95 | 0.343 | 0.004 |
| Intervention (n = 20) Week 0 | Intervention (n = 20) Week 12 | Control (n = 18) Week 0 | Control (n = 18) Week 12 | p-Value 1 | |
|---|---|---|---|---|---|
| Derived from arachidonic acid | |||||
| 5-F2t-IsoP | 56.2 (±82.5) | 39.2 (±37.3) | 44.2 (±40.8) | 63.3 (±40.8) | 0.175 |
| 15-F2t-IsoP | 31.8 (±37.1) | 37.7 (±29.1) | 51.7 (±62.1) | 38 (±25.2) | 0.014 |
| PGF2a | 109.1 (±128.7) | 83.6 (±94.7) | 112.2 (±122.6) | 118.3 (±86.3) | 0.217 |
| 5-HETE | 179.9 (±158.8) | 189.4 (±124.1) | 197.4 (±130.8) | 234.2 (±114.2) | 0.675 |
| 8-HETE | 67.6 (±71.3) | 51.9 (±57.1) | 74.8 (±46.3) | 64.1 (±36.3) | 0.734 |
| 9-HETE | 178.3 (±161.8) | 153.3 (±138.7) | 166.3 (±95.0) | 222.4 (±192.2) | 0.069 |
| 11-HETE | 96.5 (±86.8) | 69.4 (±101.2) | 131.1 (±98.6) | 142.4 (±126.4) | 0.119 |
| 12-HETE | 112 (±52.9) | 129.2 (±103.3) | 150.5 (±64.1) | 141.9 (±52.9) | 0.303 |
| 15-HETE | 139 (±106.8) | 143.8 (±95.3) | 153.5 (±77.3) | 170 (±84.1) | 0.696 |
| 20-HETE | 337.1 (±240.6) | 263.5 (±147.9) | 375.7 (±218.2) | 299.8 (±178.3) | 0.588 |
| Derived from docosahexaenoic acid | |||||
| 4-F4-NeuroP | 3890 (±3880) | 4785 (±4415) | 5388 (±5202) | 4701 (±4322) | 0.339 |
| 4-HDHA | 349 (±246.8) | 462.2 (±512.8) | 368.9 (±390.0) | 374.1 (±345.4) | 0.696 |
| 7-HDHA | 245.6 (±226.5) | 303.9 (±288.8) | 262.7 (±300.1) | 279.6 (±403.6) | 0.812 |
| 11-HDHA | 704.6 (±938.6) | 950.6 (±1625.0) | 576.7 (±458.6) | 743.1 (±666.3) | 0.217 |
| 14-HDHA | 4155 (±5985) | 6220 (±12320) | 4644 (±6696) | 5349 (±6390) | 0.800 |
| 17-HDHA | 2205 (±1289) | 3355 (±3557) | 2734 (±2452) | 2470 (±1918) | 0.426 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Csader, S.; Ismaiah, M.J.; Kuningas, T.; Heinäniemi, M.; Suhonen, J.; Männistö, V.; Pentikäinen, H.; Savonen, K.; Tauriainen, M.-M.; Galano, J.-M.; et al. Twelve Weeks of High-Intensity Interval Training Alters Adipose Tissue Gene Expression but Not Oxylipin Levels in People with Non-Alcoholic Fatty Liver Disease. Int. J. Mol. Sci. 2023, 24, 8509. https://doi.org/10.3390/ijms24108509
Csader S, Ismaiah MJ, Kuningas T, Heinäniemi M, Suhonen J, Männistö V, Pentikäinen H, Savonen K, Tauriainen M-M, Galano J-M, et al. Twelve Weeks of High-Intensity Interval Training Alters Adipose Tissue Gene Expression but Not Oxylipin Levels in People with Non-Alcoholic Fatty Liver Disease. International Journal of Molecular Sciences. 2023; 24(10):8509. https://doi.org/10.3390/ijms24108509
Chicago/Turabian StyleCsader, Susanne, Marsena Jasiel Ismaiah, Tiina Kuningas, Merja Heinäniemi, Janne Suhonen, Ville Männistö, Heikki Pentikäinen, Kai Savonen, Milla-Maria Tauriainen, Jean-Marie Galano, and et al. 2023. "Twelve Weeks of High-Intensity Interval Training Alters Adipose Tissue Gene Expression but Not Oxylipin Levels in People with Non-Alcoholic Fatty Liver Disease" International Journal of Molecular Sciences 24, no. 10: 8509. https://doi.org/10.3390/ijms24108509
APA StyleCsader, S., Ismaiah, M. J., Kuningas, T., Heinäniemi, M., Suhonen, J., Männistö, V., Pentikäinen, H., Savonen, K., Tauriainen, M.-M., Galano, J.-M., Lee, J. C.-Y., Rintamäki, R., Karisola, P., El-Nezami, H., & Schwab, U. (2023). Twelve Weeks of High-Intensity Interval Training Alters Adipose Tissue Gene Expression but Not Oxylipin Levels in People with Non-Alcoholic Fatty Liver Disease. International Journal of Molecular Sciences, 24(10), 8509. https://doi.org/10.3390/ijms24108509








