The Role of NLRP3 Inflammasome Activation and Oxidative Stress in Varicocele-Mediated Male Hypofertility
Abstract
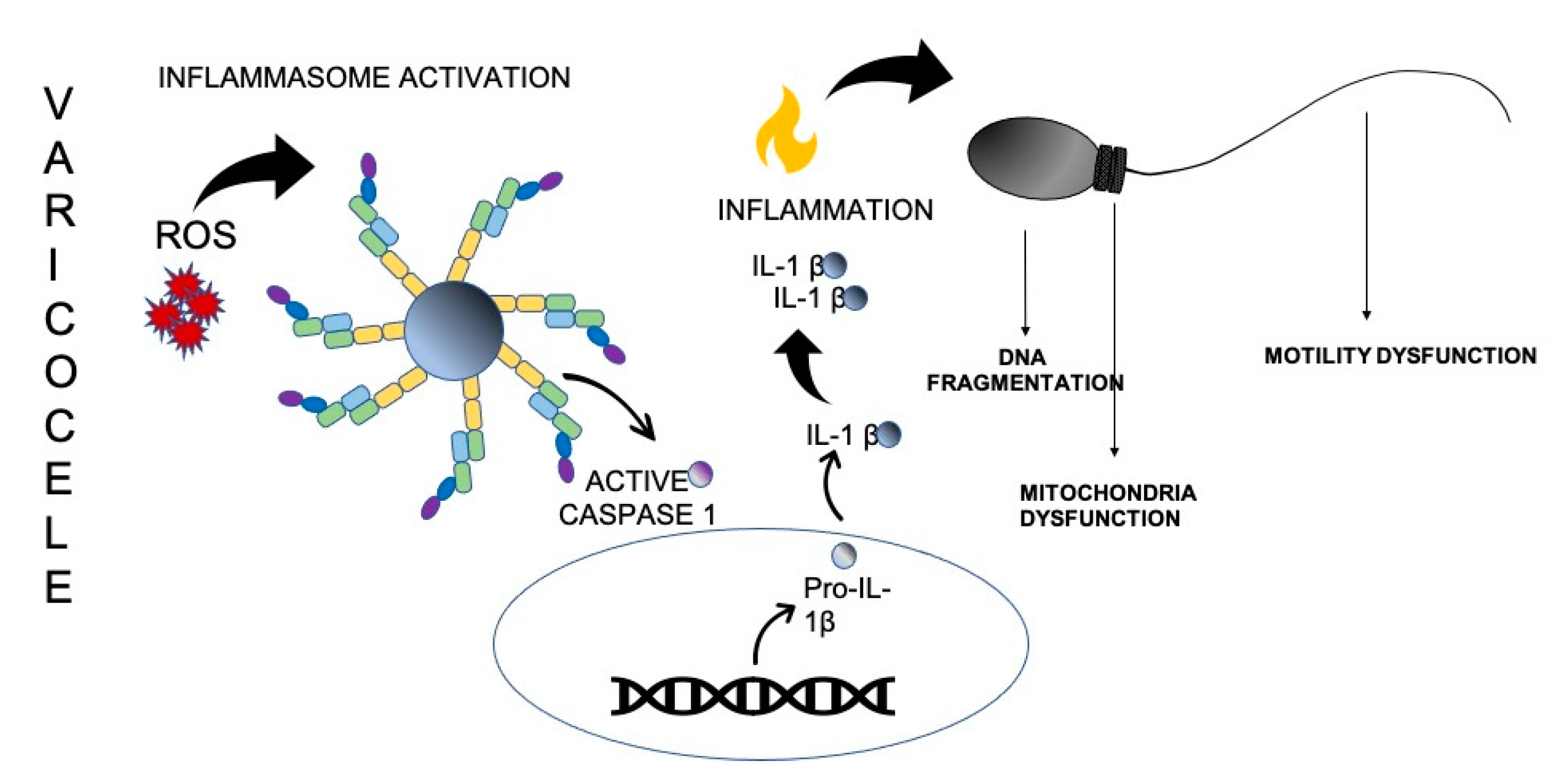
1. Introduction
2. OS and Inflammasome Activation in the Pathogenesis of VC
2.1. Testis Immunity and Inflammatory Response
2.2. OS-Induced Inflammation in VC
2.2.1. Lipid Peroxidation
2.2.2. Mitochondrial Dysfunction
2.2.3. Chromatin Damage, Sperm DNA Fragmentation and Apoptosis
3. Possible Therapeutic Approaches for VC Targeting OS and Inflammation
3.1. Resveratrol
3.2. Vitamins
3.3. Coenzyme Q10
3.4. Lycopene
3.5. Kallikrein Therapy
3.6. Pentoxifylline Therapy
3.7. Cinnoxicam Therapy
| Natural, Non-Enzymatic Antioxidants | |||
| Antioxidants | Natural Source | Main Effects | References |
| Resveratrol | Grapes, mulberry plants | Anti-inflammatory and anti-apoptotic effects; improvement in sperm count, concentration, and motility | [82] [84] |
| Vitamin E | Vegetable oil, fruits, nuts, seeds | Anti-oxidant effect; increases semen parameters; prevention of DNA damage | [88] [89] |
| Vitamin C | Fruits, vegetables | Anti-oxidant and anti-inflammatory effects; restoration of sperm motility and morphology | [89] [90] |
| CoQ10 | Oil fish, organ meats, whole grain | Anti-oxidant effect; improvement in sperm motility, ejaculation volume, and sperm density; restoration of mitocondrial function | [94] [95] [96] |
| Ubiquinol | Oil fish, organ meats, whole grain | Anti-oxidant effect; improvement in sperm motility, ejaculation volume, and sperm density; restoration of mitocondrial function | [97] [98] [99] [100] [101] [102] [103] |
| Lycopene | Vegetables, fruits | Anti-oxidant effect; increases sperm parameters; protection from DNA damage | [104] [105] [106] [107] |
| Therapeutical, non-enzymatic, drugs | |||
| Antioxidants | Chemical products | Main effects | References |
| Kallikrein | Drug | Arterial vasodilator; improvement in semen parameters, testicular testosterone, and sperm maturation | [108] [109] [110] |
| Pentoxifylline | Drug | Potent vasodilator; immunodulatory effect; anti-oxidant activity; improvement in sperm morphology | [111] [112] [113] [114] |
| Cinnoxicam | Drug | Anti-inflammatory effect; improvement in semen parameters | [115] |
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Damsgaard, J.; Joensen, U.N.; Carlsen, E.; Erenpreiss, J.; Blomberg Jensen, M.; Matulevicius, V.; Zilaitiene, B.; Olesen, I.A.; Perheentupa, A.; Punab, M.; et al. Varicocele Is Associated with Impaired Semen Quality and Reproductive Hormone Levels: A Study of 7035 Healthy Young Men from Six European Countries. Eur. Urol. 2016, 70, 1019–1029. [Google Scholar] [CrossRef]
- Adamkovicova, M.; Toman, R.; Martiniakova, M.; Omelka, R.; Babosova, R.; Krajcovicova, V.; Grosskopf, B.; Massanyi, P. Sperm motility and morphology changes in rats exposed to cadmium and diazinon. Reprod. Biol. Endocrinol. 2016, 14, 42. [Google Scholar] [CrossRef] [PubMed]
- Leslie, S.W.; Sajjad, H.; Siref, L.E. Varicocele; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Fainberg, J.; Kashanian, J.A. Recent advances in understanding and managing male infertility. F1000Research 2019, 8, 670. [Google Scholar] [CrossRef] [PubMed]
- Ambar, R.F.; Parekh, N.; Agarwal, A. Recent advances and controversies in diagnosing and treating male infertility. Fac. Rev. 2020, 9, 22. [Google Scholar] [CrossRef] [PubMed]
- Rehman, R.; Amjad, S.; Tariq, H.; Zahid, N.; Akhter, M.; Ashraf, M. Oxidative stress and male infertility: A cross sectional study. J. Pak. Med. Assoc. 2020, 70, 461–466. [Google Scholar] [CrossRef] [PubMed]
- Pizzino, G.; Irrera, N.; Cucinotta, M.; Pallio, G.; Mannino, F.; Arcoraci, V.; Squadrito, F.; Altavilla, D.; Bitto, A. Oxidative Stress: Harms and Benefits for Human Health. Oxidative Med. Cell Longev. 2017, 2017, 8416763. [Google Scholar] [CrossRef] [PubMed]
- Rajendran, P.; Nandakumar, N.; Rengarajan, T.; Palaniswami, R.; Gnanadhas, E.N.; Lakshminarasaiah, U.; Gopas, J.; Nishigaki, I. Antioxidants and human diseases. Clin. Chim. Acta 2014, 436, 332–347. [Google Scholar] [CrossRef]
- Bisht, S.; Faiq, M.; Tolahunase, M.; Dada, R. Oxidative stress and male infertility. Nat. Rev. Urol. 2017, 14, 470–485. [Google Scholar] [CrossRef]
- Sikka, S.C. Relative impact of oxidative stress on male reproductive function. Curr. Med. Chem. 2001, 8, 851–862. [Google Scholar] [CrossRef]
- Aitken, R.J.; Clarkson, J.S. Cellular basis of defective sperm function and its association with the genesis of reactive oxygen species by human spermatozoa. J. Reprod. Fertil. 1987, 81, 459–469. [Google Scholar] [CrossRef]
- Takeshima, T.; Usui, K.; Mori, K.; Asai, T.; Yasuda, K.; Kuroda, S.; Yumura, Y. Oxidative stress and male infertility. Reprod. Med. Biol. 2021, 20, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Baazm, M.; Ghafarizadeh, A.A.; Noshad Kamran, A.R.; Beyer, C.; Zendedel, A. Presence of The NLRP3 Inflammasome Components in Semen of Varicocele Patients. Int. J. Fertil. Steril. 2020, 14, 46–50. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Xu, S.; Chen, H.; He, M.; Deng, Y.; Cao, Z.; Pi, H.; Chen, C.; Li, M.; Ma, Q.; et al. CdSe/ZnS quantum dots induce hepatocyte pyroptosis and liver inflammation via NLRP3 inflammasome activation. Biomaterials 2016, 90, 27–39. [Google Scholar] [CrossRef] [PubMed]
- Kelley, N.; Jeltema, D.; Duan, Y.; He, Y. The NLRP3 Inflammasome: An Overview of Mechanisms of Activation and Regulation. Int. J. Mol. Sci. 2019, 20, 3328. [Google Scholar] [CrossRef] [PubMed]
- Bauernfeind, F.G.; Horvath, G.; Stutz, A.; Alnemri, E.S.; MacDonald, K.; Speert, D.; Fernandes-Alnemri, T.; Wu, J.; Monks, B.G.; Fitzgerald, K.A.; et al. Cutting edge: NF-kappaB activating pattern recognition and cytokine receptors license NLRP3 inflammasome activation by regulating NLRP3 expression. J. Immunol. 2009, 183, 787–791. [Google Scholar] [CrossRef]
- Franchi, L.; Eigenbrod, T.; Nunez, G. Cutting edge: TNF-alpha mediates sensitization to ATP and silica via the NLRP3 inflammasome in the absence of microbial stimulation. J. Immunol. 2009, 183, 792–796. [Google Scholar] [CrossRef]
- Burke, R.M.; Dale, B.L.; Dholakia, S. The NLRP3 Inflammasome: Relevance in Solid Organ Transplantation. Int. J. Mol. Sci. 2021, 22, 10721. [Google Scholar] [CrossRef]
- Lane, T.; Flam, B.; Lockey, R.; Kolliputi, N. TXNIP shuttling: Missing link between oxidative stress and inflammasome activation. Front. Physiol. 2013, 4, 50. [Google Scholar] [CrossRef]
- Carroll, P.A.; Freie, B.W.; Cheng, P.F.; Kasinathan, S.; Gu, H.; Hedrich, T.; Dowdle, J.A.; Venkataramani, V.; Ramani, V.; Wu, X.; et al. The glucose-sensing transcription factor MLX balances metabolism and stress to suppress apoptosis and maintain spermatogenesis. PLoS Biol. 2021, 19, e3001085. [Google Scholar] [CrossRef]
- Lundy, S.D.; Sabanegh, E.S., Jr. Varicocele management for infertility and pain: A systematic review. Arab J. Urol. 2018, 16, 157–170. [Google Scholar] [CrossRef]
- Chen, Q.; Deng, T.; Han, D. Testicular immunoregulation and spermatogenesis. Semin. Cell Dev. Biol. 2016, 59, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Le Tortorec, A.; Denis, H.; Satie, A.P.; Patard, J.J.; Ruffault, A.; Jegou, B.; Dejucq-Rainsford, N. Antiviral responses of human Leydig cells to mumps virus infection or poly I:C stimulation. Hum. Reprod. 2008, 23, 2095–2103. [Google Scholar] [CrossRef] [PubMed]
- Aslani, F.; Schuppe, H.C.; Guazzone, V.A.; Bhushan, S.; Wahle, E.; Lochnit, G.; Lustig, L.; Meinhardt, A.; Fijak, M. Targeting high mobility group box protein 1 ameliorates testicular inflammation in experimental autoimmune orchitis. Hum. Reprod. 2015, 30, 417–431. [Google Scholar] [CrossRef] [PubMed]
- Garcia, T.X.; Hofmann, M.C. Regulation of germ line stem cell homeostasis. Anim. Reprod. 2015, 12, 35–45. [Google Scholar]
- Lu, L.F.; Lind, E.F.; Gondek, D.C.; Bennett, K.A.; Gleeson, M.W.; Pino-Lagos, K.; Scott, Z.A.; Coyle, A.J.; Reed, J.L.; Van Snick, J.; et al. Mast cells are essential intermediaries in regulatory T-cell tolerance. Nature 2006, 442, 997–1002. [Google Scholar] [CrossRef]
- Zhao, S.; Zhu, W.; Xue, S.; Han, D. Testicular defense systems: Immune privilege and innate immunity. Cell Mol. Immunol. 2014, 11, 428–437. [Google Scholar] [CrossRef]
- Hutson, J.C. Physiologic interactions between macrophages and Leydig cells. Exp. Biol. Med. 2006, 231, 1–7. [Google Scholar] [CrossRef]
- Hedger, M.P. Macrophages and the immune responsiveness of the testis. J. Reprod. Immunol. 2002, 57, 19–34. [Google Scholar] [CrossRef]
- Toocheck, C.; Clister, T.; Shupe, J.; Crum, C.; Ravindranathan, P.; Lee, T.K.; Ahn, J.M.; Raj, G.V.; Sukhwani, M.; Orwig, K.E.; et al. Mouse Spermatogenesis Requires Classical and Nonclassical Testosterone Signaling. Biol. Reprod. 2016, 94, 11. [Google Scholar] [CrossRef]
- Walker, W.H. Androgen Actions in the Testis and the Regulation of Spermatogenesis. Adv. Exp. Med. Biol. 2021, 1288, 175–203. [Google Scholar] [CrossRef]
- Fan, W.; Xu, Y.; Liu, Y.; Zhang, Z.; Lu, L.; Ding, Z. Obesity or Overweight, a Chronic Inflammatory Status in Male Reproductive System, Leads to Mice and Human Subfertility. Front. Physiol. 2017, 8, 1117. [Google Scholar] [CrossRef] [PubMed]
- Mayer, C.; Adam, M.; Glashauser, L.; Dietrich, K.; Schwarzer, J.U.; Kohn, F.M.; Strauss, L.; Welter, H.; Poutanen, M.; Mayerhofer, A. Sterile inflammation as a factor in human male infertility: Involvement of Toll like receptor 2, biglycan and peritubular cells. Sci. Rep. 2016, 6, 37128. [Google Scholar] [CrossRef] [PubMed]
- Ozbek, E.; Turkoz, Y.; Gokdeniz, R.; Davarci, M.; Ozugurlu, F. Increased nitric oxide production in the spermatic vein of patients with varicocele. Eur. Urol. 2000, 37, 172–175. [Google Scholar] [CrossRef] [PubMed]
- Klein, B.; Haggeney, T.; Fietz, D.; Indumathy, S.; Loveland, K.L.; Hedger, M.; Kliesch, S.; Weidner, W.; Bergmann, M.; Schuppe, H.C. Specific immune cell and cytokine characteristics of human testicular germ cell neoplasia. Hum. Reprod. 2016, 31, 2192–2202. [Google Scholar] [CrossRef]
- Hedger, M.P.; Meinhardt, A. Cytokines and the immune-testicular axis. J. Reprod. Immunol. 2003, 58, 1–26. [Google Scholar] [CrossRef]
- Su, Y.B.; Kuo, M.J.; Lin, T.Y.; Chien, C.S.; Yang, Y.P.; Chou, S.J.; Leu, H.B. Cardiovascular manifestation and treatment in COVID-19. J. Chin. Med. Assoc. 2020, 83, 704–709. [Google Scholar] [CrossRef]
- Sultana, T.; Svechnikov, K.; Weber, G.; Soder, O. Molecular cloning and expression of a functionally different alternative splice variant of prointerleukin-1alpha from the rat testis. Endocrinology 2000, 141, 4413–4418. [Google Scholar] [CrossRef][Green Version]
- Zeinali, M.; Hadian Amree, A.; Khorramdelazad, H.; Karami, H.; Abedinzadeh, M. Inflammatory and anti-inflammatory cytokines in the seminal plasma of infertile men suffering from varicocele. Andrologia 2017, 49, e12685. [Google Scholar] [CrossRef]
- Allen, J.D.; Gow, A.J. Nitrite, NO and hypoxic vasodilation. Br. J. Pharmacol. 2009, 158, 1653–1654. [Google Scholar] [CrossRef]
- Tafuri, S.; Ciani, F.; Iorio, E.L.; Esposito, L.; Cocchia, N. Reactive oxygen species (ROS) and male fertility. In New Discoveries in Embryology; IntechOpen: London, UK, 2015; pp. 19–33. [Google Scholar]
- Aitken, R.J.; Curry, B.J. Redox regulation of human sperm function: From the physiological control of sperm capacitation to the etiology of infertility and DNA damage in the germ line. Antioxid. Redox Signal. 2011, 14, 367–381. [Google Scholar] [CrossRef]
- Moretti, E.; Collodel, G.; Fiaschi, A.I.; Micheli, L.; Iacoponi, F.; Cerretani, D. Nitric oxide, malondialdheyde and non-enzymatic antioxidants assessed in viable spermatozoa from selected infertile men. Reprod. Biol. 2017, 17, 370–375. [Google Scholar] [CrossRef] [PubMed]
- Moazamian, R.; Polhemus, A.; Connaughton, H.; Fraser, B.; Whiting, S.; Gharagozloo, P.; Aitken, R.J. Oxidative stress and human spermatozoa: Diagnostic and functional significance of aldehydes generated as a result of lipid peroxidation. Mol. Hum. Reprod. 2015, 21, 502–515. [Google Scholar] [CrossRef] [PubMed]
- Wagner, H.; Cheng, J.W.; Ko, E.Y. Role of reactive oxygen species in male infertility: An updated review of literature. Arab J. Urol. 2018, 16, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.H.; Zhao, J.; Xu, J.; Zhu, P.R.; Yu, M.M.; Jiang, W.J.; Zhang, J.; Li, W.W.; Wu, Q.Y.; Li, Z.R.; et al. Correlation of the single nucleotide polymorphism rs662 of PON1 with the risk of male infertility. Nat. J. Androl. 2018, 24, 708–712. [Google Scholar]
- Moscatelli, N.; Spagnolo, B.; Pisanello, M.; Lemma, E.D.; De Vittorio, M.; Zara, V.; Pisanello, F.; Ferramosca, A. Single-cell-based evaluation of sperm progressive motility via fluorescent assessment of mitochondria membrane potential. Sci. Rep. 2017, 7, 17931. [Google Scholar] [CrossRef]
- Bazrafkan, M.; Nikmehr, B.; Shahverdi, A.; Hosseini, S.R.; Hassani, F.; Poorhassan, M.; Mokhtari, T.; Abolhassani, F.; Choobineh, H.; Beyer, C.; et al. Lipid Peroxidation and Its Role in the Expression of NLRP1a and NLRP3 Genes in Testicular Tissue of Male Rats: A Model of Spinal Cord Injury. Iran. Biomed. J. 2018, 22, 151–159. [Google Scholar] [CrossRef]
- Zhang, X.; Ibrahim, E.; de Rivero Vaccari, J.P.; Lotocki, G.; Aballa, T.C.; Dietrich, W.D.; Keane, R.W.; Lynne, C.M.; Brackett, N.L. Involvement of the inflammasome in abnormal semen quality of men with spinal cord injury. Fertil. Steril. 2013, 99, 118–124.e2. [Google Scholar] [CrossRef]
- Ferramosca, A.; Provenzano, S.P.; Coppola, L.; Zara, V. Mitochondrial respiratory efficiency is positively correlated with human sperm motility. Urology 2012, 79, 809–814. [Google Scholar] [CrossRef]
- Amaral, A.; Ramalho-Santos, J.; St John, J.C. The expression of polymerase gamma and mitochondrial transcription factor A and the regulation of mitochondrial DNA content in mature human sperm. Hum. Reprod. 2007, 22, 1585–1596. [Google Scholar] [CrossRef]
- Pelliccione, F.; Micillo, A.; Cordeschi, G.; D’Angeli, A.; Necozione, S.; Gandini, L.; Lenzi, A.; Francavilla, F.; Francavilla, S. Altered ultrastructure of mitochondrial membranes is strongly associated with unexplained asthenozoospermia. Fertil. Steril. 2011, 95, 641–646. [Google Scholar] [CrossRef]
- Samanta, L.; Agarwal, A.; Swain, N.; Sharma, R.; Gopalan, B.; Esteves, S.C.; Durairajanayagam, D.; Sabanegh, E. Proteomic Signatures of Sperm Mitochondria in Varicocele: Clinical Use as Biomarkers of Varicocele Associated Infertility. J. Urol. 2018, 200, 414–422. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Zhang, D.; Hu, D.; Zhou, X.; Zhou, Y. The role of mitochondria in NLRP3 inflammasome activation. Mol. Immunol. 2018, 103, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Bryant, C.; Fitzgerald, K.A. Molecular mechanisms involved in inflammasome activation. Trends Cell Biol. 2009, 19, 455–464. [Google Scholar] [CrossRef] [PubMed]
- Shimada, K.; Crother, T.R.; Karlin, J.; Dagvadorj, J.; Chiba, N.; Chen, S.; Ramanujan, V.K.; Wolf, A.J.; Vergnes, L.; Ojcius, D.M.; et al. Oxidized mitochondrial DNA activates the NLRP3 inflammasome during apoptosis. Immunity 2012, 36, 401–414. [Google Scholar] [CrossRef]
- Bui, A.D.; Sharma, R.; Henkel, R.; Agarwal, A. Reactive oxygen species impact on sperm DNA and its role in male infertility. Andrologia 2018, 50, e13012. [Google Scholar] [CrossRef]
- Spiropoulos, J.; Turnbull, D.M.; Chinnery, P.F. Can mitochondrial DNA mutations cause sperm dysfunction? Mol. Hum. Reprod. 2002, 8, 719–721. [Google Scholar] [CrossRef]
- Kao, S.H.; Chao, H.T.; Wei, Y.H. Multiple deletions of mitochondrial DNA are associated with the decline of motility and fertility of human spermatozoa. Mol. Hum. Reprod. 1998, 4, 657–666. [Google Scholar] [CrossRef]
- St John, J.C.; Sakkas, D.; Barratt, C.L. A role for mitochondrial DNA and sperm survival. J. Androl. 2000, 21, 189–199. [Google Scholar]
- Gashti, N.G.; Salehi, Z.; Madani, A.H.; Dalivandan, S.T. 4977-bp mitochondrial DNA deletion in infertile patients with varicocele. Andrologia 2014, 46, 258–262. [Google Scholar] [CrossRef]
- Santana, V.P.; Miranda-Furtado, C.L.; de Oliveira-Gennaro, F.G.; Dos Reis, R.M. Genetics and epigenetics of varicocele pathophysiology: An overview. J. Assist. Reprod. Genet. 2017, 34, 839–847. [Google Scholar] [CrossRef]
- Agarwal, A.; Majzoub, A.; Baskaran, S.; Panner Selvam, M.K.; Cho, C.L.; Henkel, R.; Finelli, R.; Leisegang, K.; Sengupta, P.; Barbarosie, C.; et al. Sperm DNA Fragmentation: A New Guideline for Clinicians. World J. Men Health 2020, 38, 412–471. [Google Scholar] [CrossRef] [PubMed]
- Esterbauer, H. Cytotoxicity and genotoxicity of lipid-oxidation products. Am. J. Clin. Nutr. 1993, 57, 779S–786S. [Google Scholar] [CrossRef] [PubMed]
- Luczaj, W.; Skrzydlewska, E. DNA damage caused by lipid peroxidation products. Cell Mol. Biol. Lett. 2003, 8, 391–413. [Google Scholar] [PubMed]
- Aitken, R.J.; Gibb, Z.; Baker, M.A.; Drevet, J.; Gharagozloo, P. Causes and consequences of oxidative stress in spermatozoa. Reprod. Fertil. Dev. 2016, 28, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Bauer, N.C.; Corbett, A.H.; Doetsch, P.W. The current state of eukaryotic DNA base damage and repair. Nucleic Acids Res. 2015, 43, 10083–10101. [Google Scholar] [CrossRef] [PubMed]
- Henkel, R.; Kierspel, E.; Stalf, T.; Mehnert, C.; Menkveld, R.; Tinneberg, H.R.; Schill, W.B.; Kruger, T.F. Effect of reactive oxygen species produced by spermatozoa and leukocytes on sperm functions in non-leukocytospermic patients. Fertil. Steril. 2005, 83, 635–642. [Google Scholar] [CrossRef]
- Selvaratnam, J.S.; Robaire, B. Effects of Aging and Oxidative Stress on Spermatozoa of Superoxide-Dismutase 1- and Catalase-Null Mice. Biol. Reprod. 2016, 95, 60. [Google Scholar] [CrossRef]
- Verma, R.; Balakrishnan, L.; Sharma, K.; Khan, A.A.; Advani, J.; Gowda, H.; Tripathy, S.P.; Suar, M.; Pandey, A.; Gandotra, S.; et al. A network map of Interleukin-10 signaling pathway. J. Cell Commun. Signal. 2016, 10, 61–67. [Google Scholar] [CrossRef]
- Guo, H.; Liu, H.; Jian, Z.; Cui, H.; Fang, J.; Zuo, Z.; Deng, J.; Li, Y.; Wang, X.; Zhao, L.; et al. Nickel induces inflammatory activation via NF-kappaB, MAPKs, IRF3 and NLRP3 inflammasome signaling pathways in macrophages. Aging 2019, 11, 11659–11672. [Google Scholar] [CrossRef]
- Walenta, L.; Schmid, N.; Schwarzer, J.U.; Kohn, F.M.; Urbanski, H.F.; Behr, R.; Strauss, L.; Poutanen, M.; Mayerhofer, A. NLRP3 in somatic non-immune cells of rodent and primate testes. Reproduction 2018, 156, 231–238. [Google Scholar] [CrossRef]
- Wang, X.; Sharma, R.K.; Sikka, S.C.; Thomas, A.J., Jr.; Falcone, T.; Agarwal, A. Oxidative stress is associated with increased apoptosis leading to spermatozoa DNA damage in patients with male factor infertility. Fertil. Steril. 2003, 80, 531–535. [Google Scholar] [CrossRef]
- Elmore, S. Apoptosis: A review of programmed cell death. Toxicol. Pathol. 2007, 35, 495–516. [Google Scholar] [CrossRef] [PubMed]
- Koppers, A.J.; Mitchell, L.A.; Wang, P.; Lin, M.; Aitken, R.J. Phosphoinositide 3-kinase signalling pathway involvement in a truncated apoptotic cascade associated with motility loss and oxidative DNA damage in human spermatozoa. Biochem. J. 2011, 436, 687–698. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Su, Y.; Xu, J.; Hu, Z.; Zhao, K.; Liu, C.; Zhang, H. Varicocele-Mediated Male Infertility: From the Perspective of Testicular Immunity and Inflammation. Front. Immunol. 2021, 12, 729539. [Google Scholar] [CrossRef]
- Finelli, R.; Leisegang, K.; Kandil, H.; Agarwal, A. Oxidative Stress: A Comprehensive Review of Biochemical, Molecular, and Genetic Aspects in the Pathogenesis and Management of Varicocele. World J. Men Health 2022, 40, 87–103. [Google Scholar] [CrossRef]
- Agarwal, A.; Prabakaran, S.; Allamaneni, S.S. Relationship between oxidative stress, varicocele and infertility: A meta-analysis. Reprod. Biomed. Online 2006, 12, 630–633. [Google Scholar] [CrossRef]
- Karthikeyan, V.; Manian, R. A Review on Antioxidant Therapy for Varicocele Associated Male Infertility. Eur. J. Mol. Clin. Med. 2020, 7, 2454–2471. [Google Scholar]
- Barati, E.; Nikzad, H.; Karimian, M. Oxidative stress and male infertility: Current knowledge of pathophysiology and role of antioxidant therapy in disease management. Cell Mol. Life Sci. 2020, 77, 93–113. [Google Scholar] [CrossRef]
- Aitken, R.J.; Roman, S.D. Antioxidant systems and oxidative stress in the testes. Oxidative Med. Cell Longev. 2008, 1, 15–24. [Google Scholar] [CrossRef]
- Hajipour, E.; Mashayekhi, F.J.; Mosayebi, G.; Baazm, M.; Zendedel, A. Resveratrol decreases apoptosis and NLRP3 complex expressions in experimental varicocele rat model. Iran. J. Basic Med. Sci. 2018, 21, 225–229. [Google Scholar] [CrossRef]
- Berman, A.Y.; Motechin, R.A.; Wiesenfeld, M.Y.; Holz, M.K. The therapeutic potential of resveratrol: A review of clinical trials. NPJ Precis. Oncol. 2017, 1, 35. [Google Scholar] [CrossRef] [PubMed]
- Illiano, E.; Trama, F.; Zucchi, A.; Iannitti, R.G.; Fioretti, B.; Costantini, E. Resveratrol-Based Multivitamin Supplement Increases Sperm Concentration and Motility in Idiopathic Male Infertility: A Pilot Clinical Study. J. Clin. Med. 2020, 9, 4017. [Google Scholar] [CrossRef] [PubMed]
- Tsampoukas, G.; Gkeka, K.; Dellis, A.; Brown, D.; Katsouri, A.; Alneshawy, A.; Moussa, M.; Papatsoris, A.; Buchholz, N. Vitamins as primary or adjunctive treatment in infertile men with varicocele: A systematic review. Arab J. Urol. 2021, 19, 264–273. [Google Scholar] [CrossRef]
- Busetto, G.M.; Agarwal, A.; Virmani, A.; Antonini, G.; Ragonesi, G.; Del Giudice, F.; Micic, S.; Gentile, V.; De Berardinis, E. Effect of metabolic and antioxidant supplementation on sperm parameters in oligo-astheno-teratozoospermia, with and without varicocele: A double-blind placebo-controlled study. Andrologia 2018, 50, e12927. [Google Scholar] [CrossRef] [PubMed]
- Sohrabipour, S.; Jafari, A.; Kamalinejad, M.; Sarrafnejd, A.; Shahrestany, T.; Sadeghipour, H.R. The role of flaxseed and vitamin E on oxidative stress in prepubertal rats with experimental varicocele: An experimental study. Iran. J. Reprod. Med. 2013, 11, 459–466. [Google Scholar] [PubMed]
- Ener, K.; Aldemir, M.; Isik, E.; Okulu, E.; Ozcan, M.F.; Ugurlu, M.; Tangal, S.; Ozayar, A. The impact of vitamin E supplementation on semen parameters and pregnancy rates after varicocelectomy: A randomised controlled study. Andrologia 2016, 48, 829–834. [Google Scholar] [CrossRef] [PubMed]
- Sang, X.; Wang, H.; Chen, Y.; Guo, Q.; Lu, A.; Zhu, X.; Meng, G. Vitamin C inhibits the activation of the NLRP3 inflammasome by scavenging mitochondrial ROS. Inflammasome 2016, 2, 13–19. [Google Scholar] [CrossRef]
- Cyrus, A.; Kabir, A.; Goodarzi, D.; Moghimi, M. The effect of adjuvant vitamin C after varicocele surgery on sperm quality and quantity in infertile men: A double blind placebo controlled clinical trial. Int. Braz. J. Urol. 2015, 41, 230–238. [Google Scholar] [CrossRef]
- Raizner, A.E. Coenzyme Q10. Methodist Debakey Cardiovasc. J. 2019, 15, 185–191. [Google Scholar] [CrossRef]
- Sanz, A.; Navas, P. Editorial: Coenzyme Q Redox State and Cellular Homeostasis. Front. Physiol. 2018, 9, 912. [Google Scholar] [CrossRef]
- Gvozdjakova, A.; Kucharska, J.; Dubravicky, J.; Mojto, V.; Singh, R.B. Coenzyme Q(1)(0), alpha-tocopherol, and oxidative stress could be important metabolic biomarkers of male infertility. Dis. Markers 2015, 2015, 827941. [Google Scholar] [CrossRef] [PubMed]
- Al-Qadhi, H.I.; Jawad, H.M.; Al-Nasiri, U.S. Coenzyme Q10 Effect on Varicocele Associated Asthenozoospermia. IOSR-JPBS 2016, 11, 69–73. [Google Scholar]
- Festa, R.; Giacchi, E.; Raimondo, S.; Tiano, L.; Zuccarelli, P.; Silvestrini, A.; Meucci, E.; Littarru, G.P.; Mancini, A. Coenzyme Q10 supplementation in infertile men with low-grade varicocele: An open, uncontrolled pilot study. Andrologia 2014, 46, 805–807. [Google Scholar] [CrossRef] [PubMed]
- Chokchaiwong, S.; Kuo, Y.T.; Lin, S.H.; Hsu, Y.C.; Hsu, S.P.; Liu, Y.T.; Chou, A.J.; Kao, S.H. Coenzyme Q10 serves to couple mitochondrial oxidative phosphorylation and fatty acid beta-oxidation, and attenuates NLRP3 inflammasome activation. Free Radic. Res. 2018, 52, 1445–1455. [Google Scholar] [CrossRef]
- Safarinejad, M.R.; Safarinejad, S.; Shafiei, N.; Safarinejad, S. Effects of the reduced form of coenzyme Q10 (ubiquinol) on semen parameters in men with idiopathic infertility: A double-blind, placebo controlled, randomized study. J. Urol. 2012, 188, 526–531. [Google Scholar] [CrossRef]
- Langsjoen, P.H.; Langsjoen, A.M. Comparison study of plasma coenzyme Q10 levels in healthy subjects supplemented with ubiquinol versus ubiquinone. Clin. Pharmacol. Drug Dev. 2014, 3, 13–17. [Google Scholar] [CrossRef]
- Mae, T.; Sakamoto, Y.; Morikawa, S.; Hidaka, T. Pharmaceutical Composition Comprising Coenzyme Q10. U.S. Patent No. 6,184,255, 2001. [Google Scholar]
- Failla, M.L.; Chitchumroonchokchai, C.; Aoki, F. Increased bioavailability of ubiquinol compared to that of ubiquinone is due to more efficient micellarization during digestion and greater GSH-dependent uptake and basolateral secretion by Caco-2 cells. J. Agric. Food Chem. 2014, 62, 7174–7182. [Google Scholar] [CrossRef]
- Miles, M.V.; Horn, P.; Miles, L.; Tang, P.; Steele, P.; DeGrauw, T. Bioequivalence of coenzyme Q10 from over-the-counter supplements. Nutr. Res. 2002, 22, 919–929. [Google Scholar] [CrossRef]
- Alf, D.; Schmidt, M.E.; Siebrecht, S.C. Ubiquinol supplementation enhances peak power production in trained athletes: A double-blind, placebo controlled study. J. Int. Soc. Sports Nutr. 2013, 10, 24. [Google Scholar] [CrossRef]
- Alahmar, A.T. The impact of two doses of coenzyme Q10 on semen parameters and antioxidant status in men with idiopathic oligoasthenoteratozoospermia. Clin. Exp. Reprod. Med. 2019, 46, 112–118. [Google Scholar] [CrossRef]
- Babaei, A.; Asadpour, R.; Mansouri, K.; Sabrivand, A.; Kazemi-Darabadi, S. Lycopene protects sperm from oxidative stress in the experimental varicocele model. Food Sci. Nutr. 2021, 9, 6806–6817. [Google Scholar] [CrossRef] [PubMed]
- Kelkel, M.; Schumacher, M.; Dicato, M.; Diederich, M. Antioxidant and anti-proliferative properties of lycopene. Free Radic. Res. 2011, 45, 925–940. [Google Scholar] [CrossRef] [PubMed]
- Durairajanayagam, D.; Agarwal, A.; Ong, C.; Prashast, P. Lycopene and male infertility. Asian J. Androl. 2014, 16, 420–425. [Google Scholar] [CrossRef] [PubMed]
- Gupta, N.P.; Kumar, R. Lycopene therapy in idiopathic male infertility—A preliminary report. Int. Urol. Nephrol. 2002, 34, 369–372. [Google Scholar] [CrossRef]
- Kalinska, M.; Meyer-Hoffert, U.; Kantyka, T.; Potempa, J. Kallikreins—The melting pot of activity and function. Biochimie 2016, 122, 270–282. [Google Scholar] [CrossRef]
- Garg, H.; Kumar, R. An update on the role of medical treatment including antioxidant therapy in varicocele. Asian J. Androl. 2016, 18, 222–228. [Google Scholar] [CrossRef]
- Micic, S.; Tulic, C.; Dotlic, R. Kallikrein therapy of infertile men with varicocele and impaired sperm motility. Andrologia 1990, 22, 179–183. [Google Scholar] [CrossRef]
- Al Mosawi, A.J. The use of pentoxifylline in male reproduction abnormalities. Prog. Men Health 2021, 1, 21–23. [Google Scholar] [CrossRef]
- Lee, D. Global and local missions of cAMP signaling in neural plasticity, learning, and memory. Front. Pharmacol. 2015, 6, 161. [Google Scholar] [CrossRef]
- Wang, Y.; Kang, Y.; Qi, C.; Zhang, T.; Zhao, H.; Ji, X.; Yan, W.; Huang, Y.; Cui, R.; Zhang, G.; et al. Pentoxifylline enhances antioxidative capability and promotes mitochondrial biogenesis for improving age-related behavioral deficits. Aging 2020, 12, 25487–25504. [Google Scholar] [CrossRef]
- Oliva, A.; Dotta, A.; Multigner, L. Pentoxifylline and antioxidants improve sperm quality in male patients with varicocele. Fertil. Steril. 2009, 91, 1536–1539. [Google Scholar] [CrossRef] [PubMed]
- Cavallini, G.; Biagiotti, G.; Ferraretti, A.P.; Gianaroli, L.; Vitali, G. Medical therapy of oligoasthenospermia associated with left varicocele. BJU Int. 2003, 91, 513–518. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Poli, G.; Fabi, C.; Sugoni, C.; Bellet, M.M.; Costantini, C.; Luca, G.; Brancorsini, S. The Role of NLRP3 Inflammasome Activation and Oxidative Stress in Varicocele-Mediated Male Hypofertility. Int. J. Mol. Sci. 2022, 23, 5233. https://doi.org/10.3390/ijms23095233
Poli G, Fabi C, Sugoni C, Bellet MM, Costantini C, Luca G, Brancorsini S. The Role of NLRP3 Inflammasome Activation and Oxidative Stress in Varicocele-Mediated Male Hypofertility. International Journal of Molecular Sciences. 2022; 23(9):5233. https://doi.org/10.3390/ijms23095233
Chicago/Turabian StylePoli, Giulia, Consuelo Fabi, Chiara Sugoni, Marina Maria Bellet, Claudio Costantini, Giovanni Luca, and Stefano Brancorsini. 2022. "The Role of NLRP3 Inflammasome Activation and Oxidative Stress in Varicocele-Mediated Male Hypofertility" International Journal of Molecular Sciences 23, no. 9: 5233. https://doi.org/10.3390/ijms23095233
APA StylePoli, G., Fabi, C., Sugoni, C., Bellet, M. M., Costantini, C., Luca, G., & Brancorsini, S. (2022). The Role of NLRP3 Inflammasome Activation and Oxidative Stress in Varicocele-Mediated Male Hypofertility. International Journal of Molecular Sciences, 23(9), 5233. https://doi.org/10.3390/ijms23095233







