Effects of Low-Intensity and Long-Term Aerobic Exercise on the Psoas Muscle of mdx Mice: An Experimental Model of Duchenne Muscular Dystrophy
Abstract
1. Introduction
2. Results
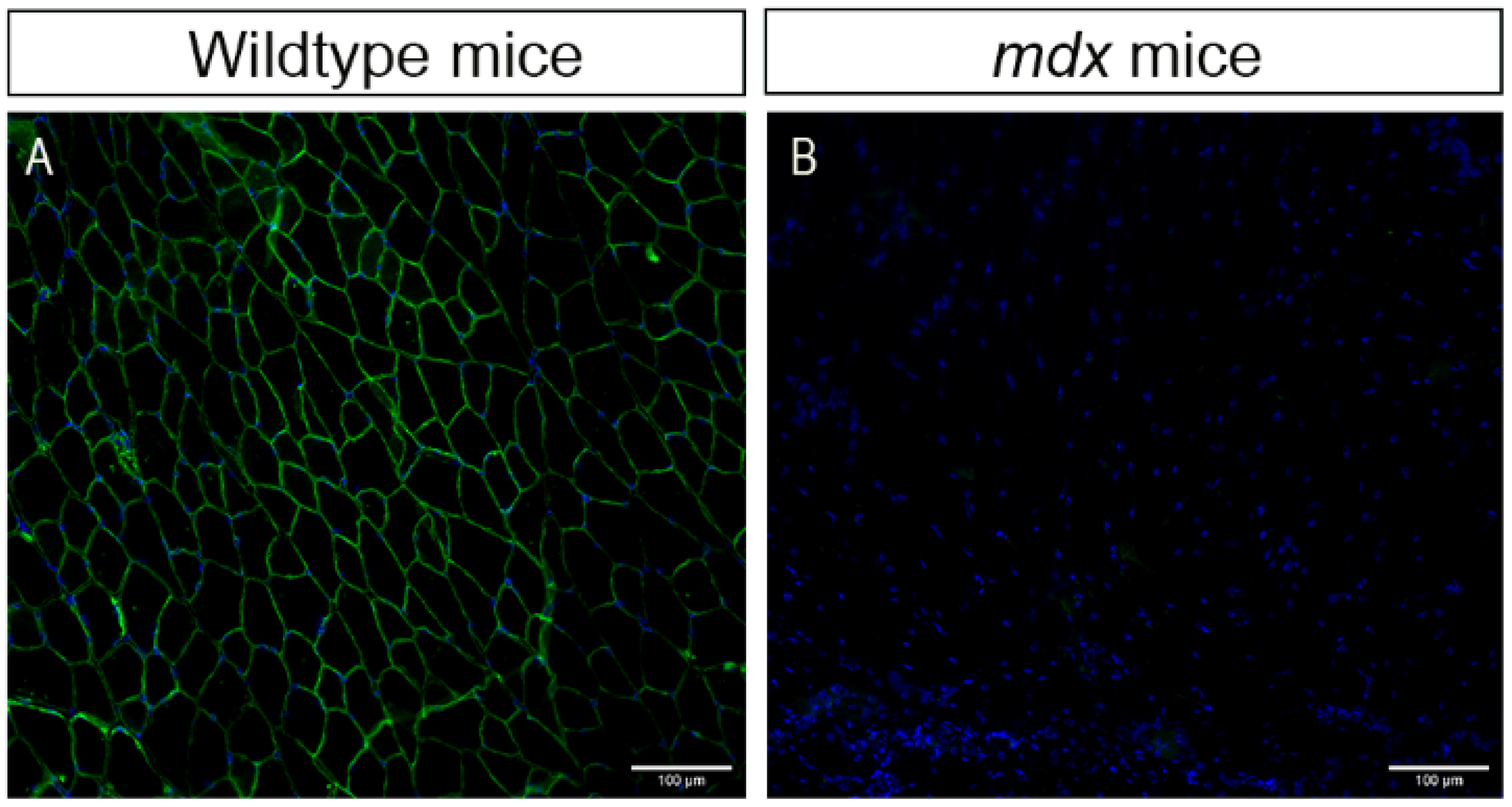
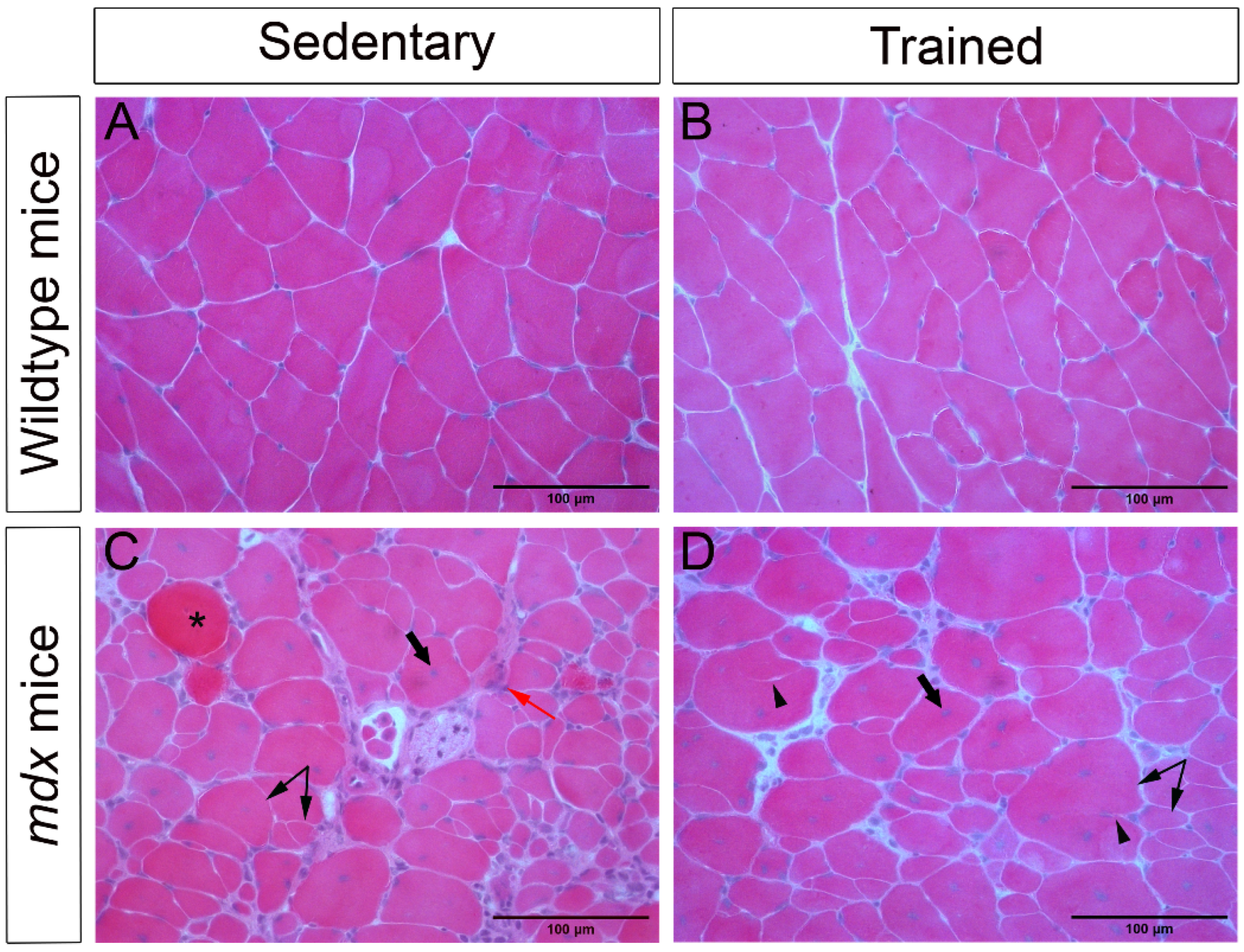
2.1. Morphological Characteristics of the Dystrophic Muscle Improved after Low-Intensity Exercise
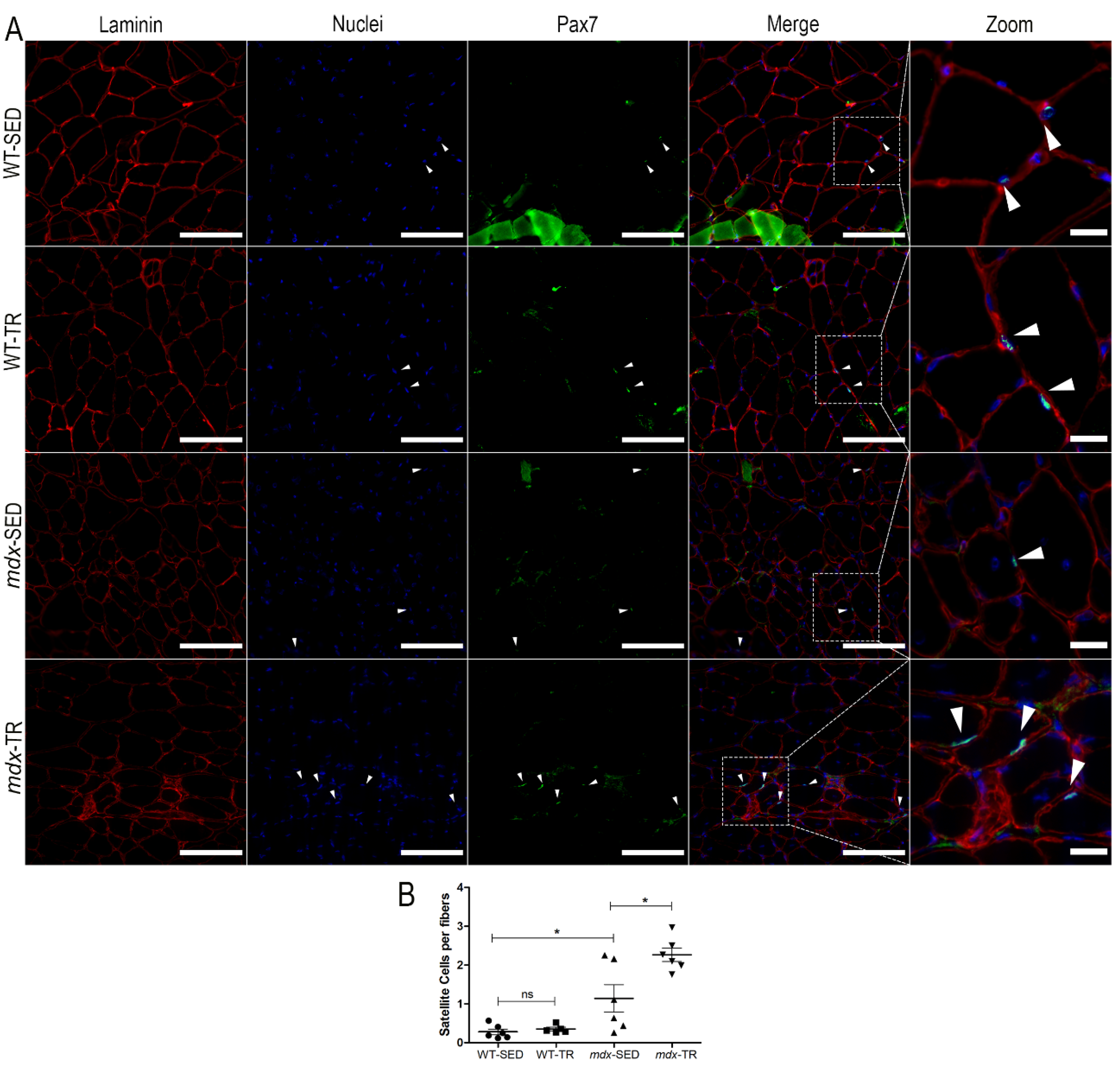
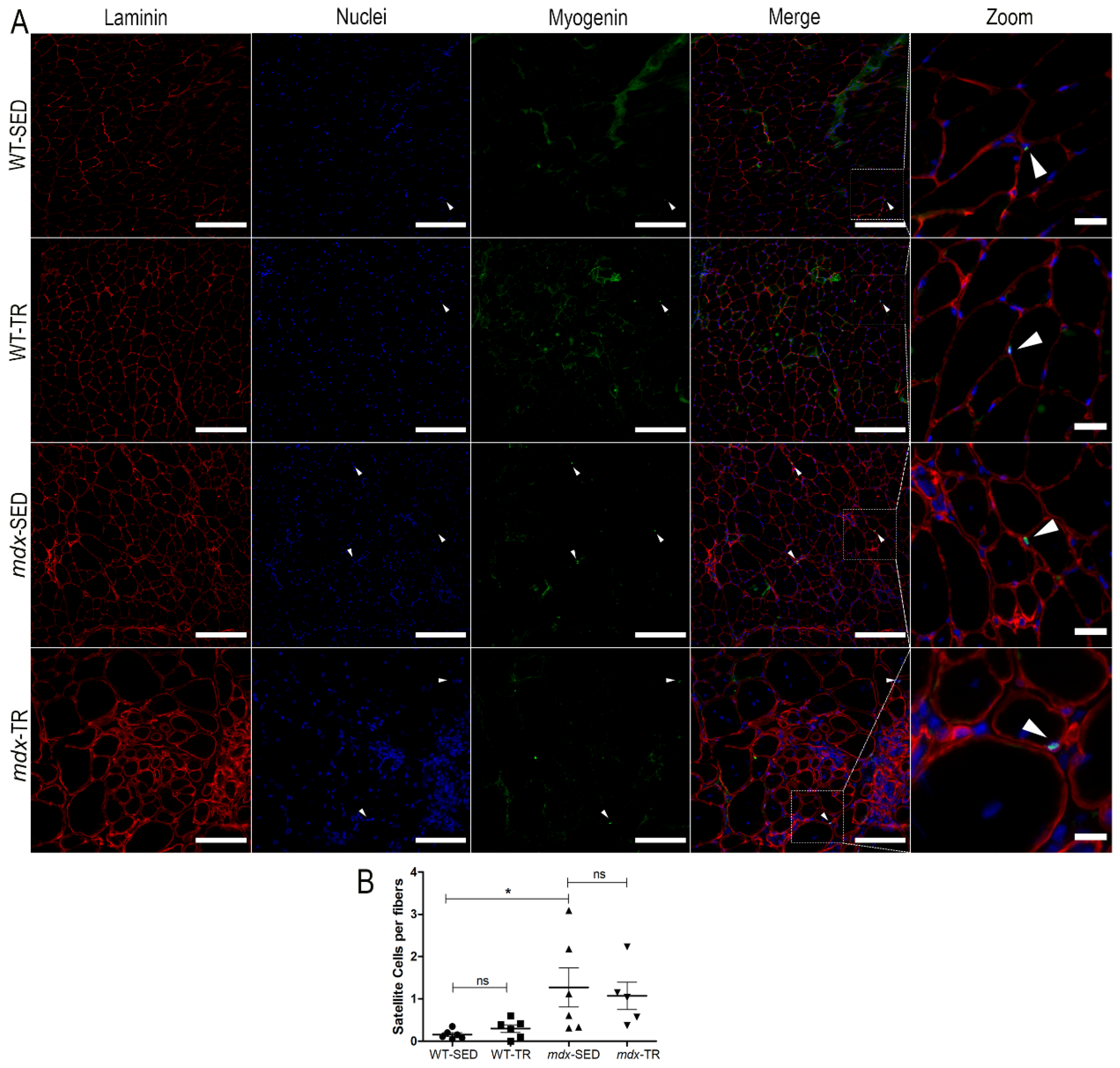
2.2. Low-Intensity Exercise Promotes an Increase in the Number of Satellite Cells in the Dystrophic Muscle
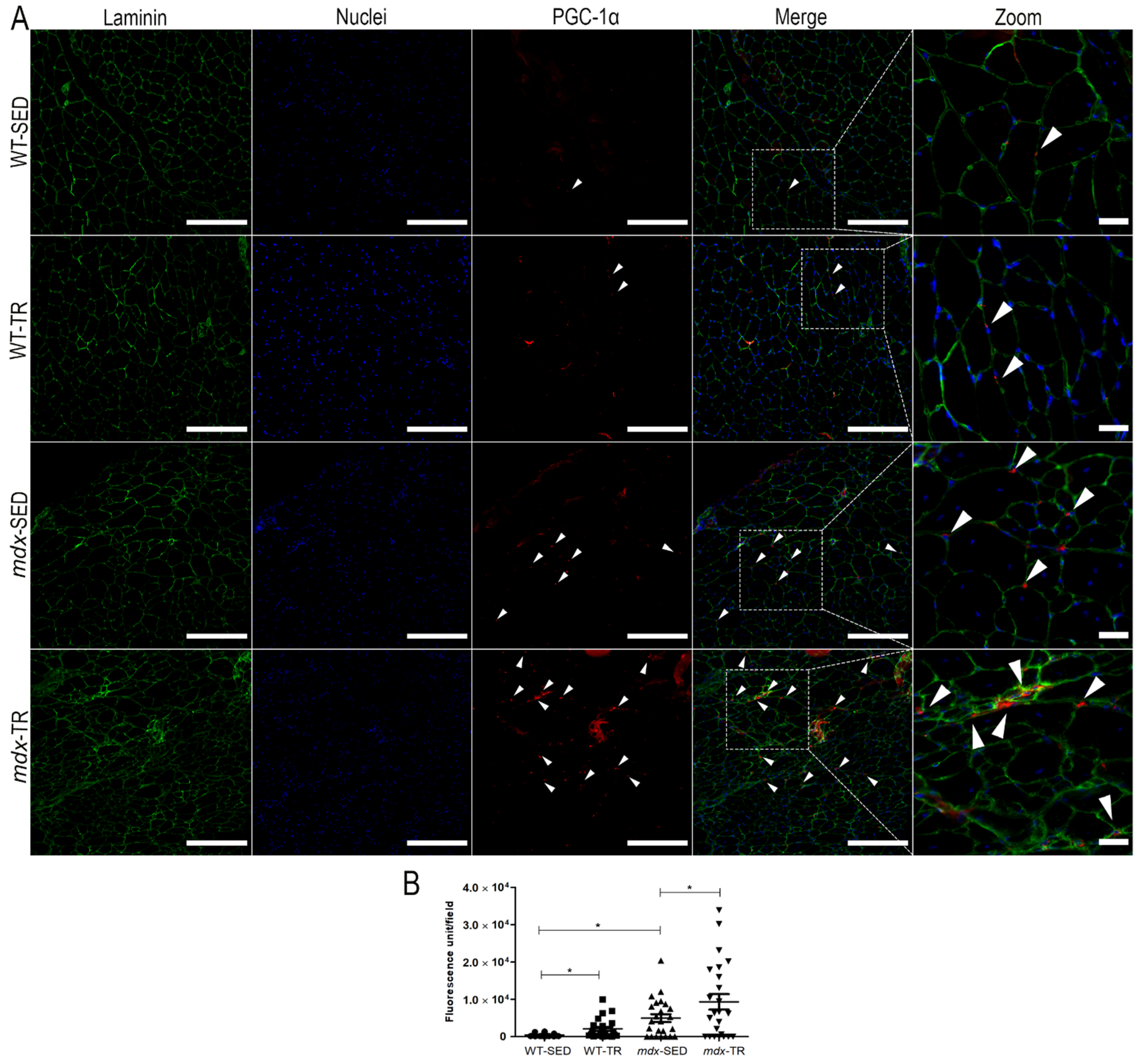
2.3. PGC-1α Is Increased in Dystrophic Muscles
2.4. A Long Period of Low-Intensity Aerobic Exercise Increases the Trophism of Fibers in mdx Mice
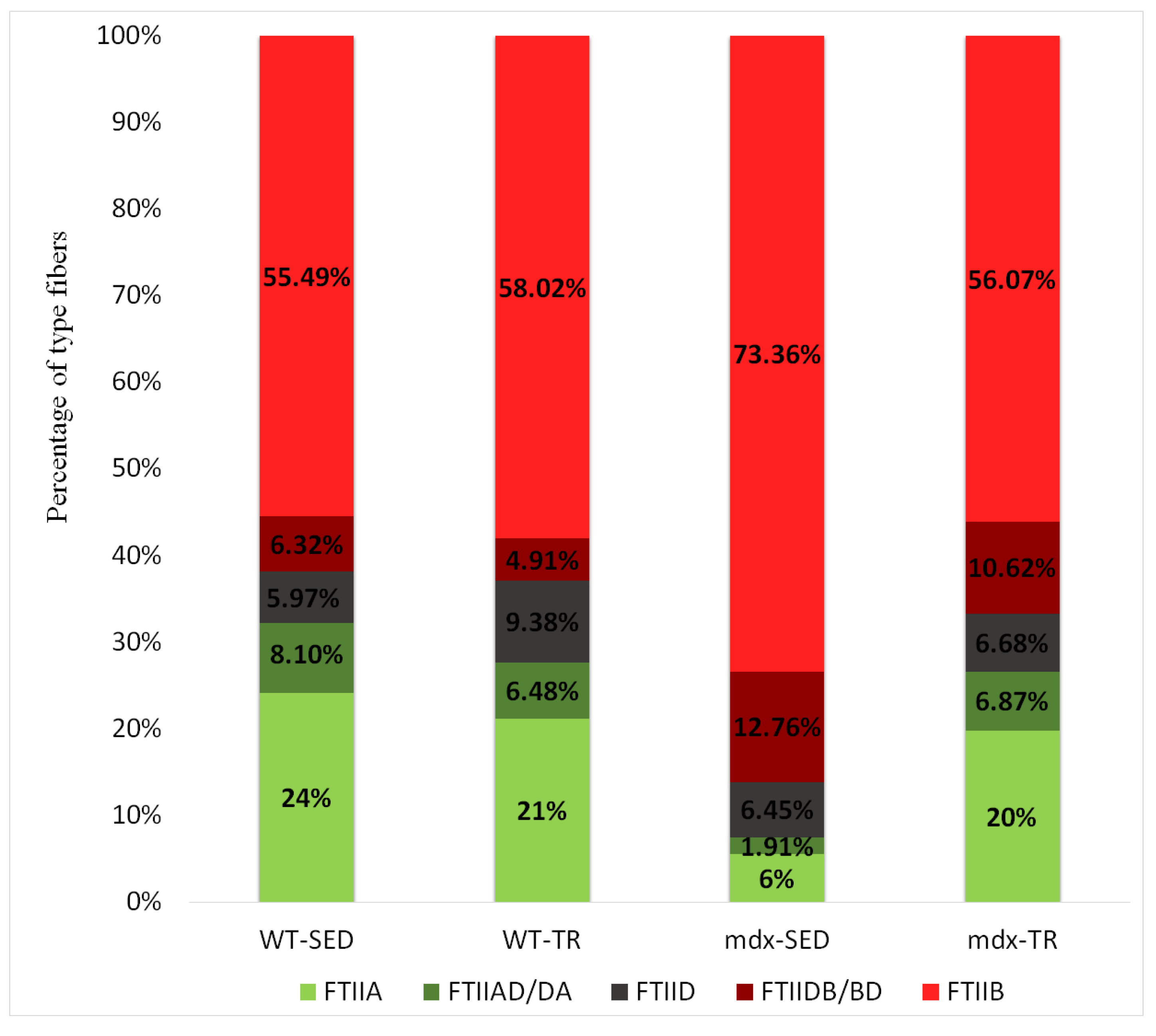
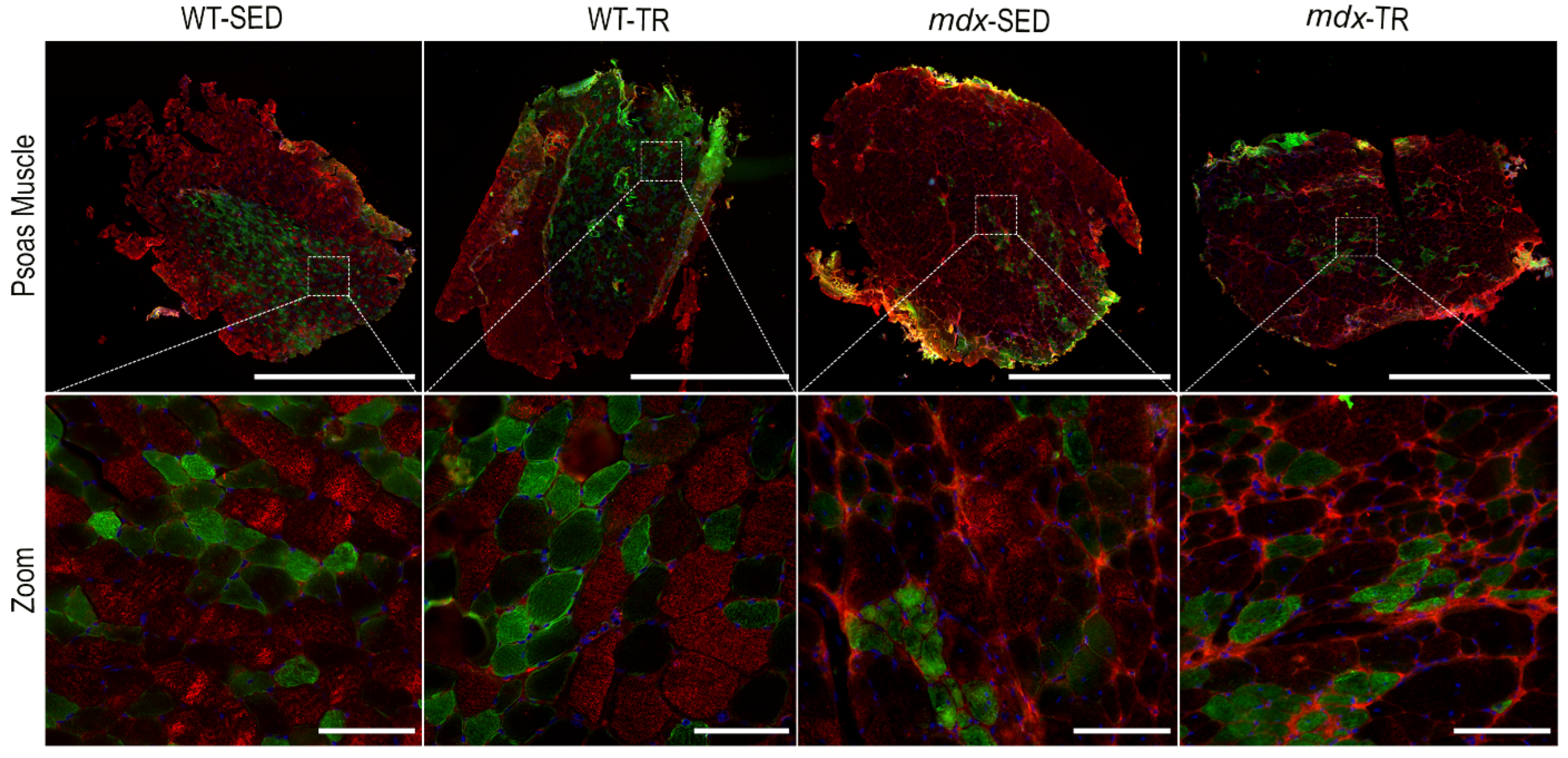
2.5. Low-Intensity Exercise Did Not Affect the Fiber Type Distribution
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Low-Intensity Training
4.3. Histology
4.4. Immunofluorescence
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| DAPI | 4′,6-Diamidino-2-phenylindole |
| DMD | Duchenne muscular dystrophy |
| FTI | Type I fibers |
| FTII | Type II fibers |
| HE | Hematoxylin and eosin |
| IF | Immunofluorescence |
| mdx | Experimental model of Duchenne muscular dystrophy |
| MHC | Myosin heavy chain |
| MOM | Mouse on mouse |
| PBS | Phosphate-buffered saline |
| PGC-1α | Peroxisome proliferator-activated receptor-gamma coactivator-1α |
| SC | Satellite cell |
| WT | Wildtype |
References
- Dietz, A.R.; Connolly, A.; Dori, A.; Zaidman, C.M. Intramuscular blood flow in Duchenne and Becker Muscular Dystrophy: Quantitative power Doppler sonography relates to disease severity. Clin. Neurophysiol. 2020, 131, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Santos, N.M.; Rezende, M.D.M.; Terni, A.; Hayashi, M.C.B.; Fávero, F.M.; Quadros, A.A.J.; Reis, L.I.O.; dos Adissi, M.; Langer, A.L.; Fontes, S.V.; et al. Perfil clínico e funcional dos pacientes com Distrofia Muscular de Duchenne assistidos na Associação Brasileira de Distrofia Muscular (ABDIM) attending the Brazilian Association of muscular dystrophy (ABDIM). Rev. Neurocienc. 2006, 14, 015–022. [Google Scholar] [CrossRef]
- Alderton, J.M.; Steinhardt, R.A. Calcium influx through calcium leak channels is responsible for the elevated levels of calcium-dependent proteolysis in dystrophic myotubes. J. Biol. Chem. 2000, 275, 9452–9460. [Google Scholar] [CrossRef] [PubMed]
- Le Rumeur, E. Dystrophin and the two related genetic diseases, duchenne and becker muscular dystrophies. Bosn. J. Basic Med. Sci. 2015, 15, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Arbanas, J.; Starcevic Klasan, G.; Nikolic, M.; Jerkovic, R.; Miljanovic, I.; Malnar, D. Fibre type composition of the human psoas major muscle with regard to the level of its origin. J. Anat. 2009, 215, 636–641. [Google Scholar] [CrossRef]
- Talbot, J.; Mavez, L. Resistance To Muscle Disease. Wiley Interdiscip. Rev. Dev. Biol. 2016, 5, 518–534. [Google Scholar] [CrossRef]
- Yin, H.; Price, F.; Rudnicki, M.A. Satellite cells and the muscle stem cell niche. Physiol. Rev. 2013, 93, 23–67. [Google Scholar] [CrossRef]
- Handschin, C.; Spiegelman, B.M. The role of exercise and PGC1alpha in inflammation and chronic disease. Nature 2008, 454, 463–469. [Google Scholar] [CrossRef]
- Mourikis, P.; Relaix, F. Activated Muscle Satellite Cells Chase Ghosts. Cell Stem Cell 2016, 18, 160–162. [Google Scholar] [CrossRef]
- Snijders, T.; Nederveen, J.P.; McKay, B.R.; Joanisse, S.; Verdijk, L.B.; van Loon, L.J.C.; Parise, G. Satellite cells in human skeletal muscle plasticity. Front. Physiol. 2015, 6, 1–21. [Google Scholar] [CrossRef]
- Suntar, I.; Sureda, A.; Belwal, T.; Sanches Silva, A.; Vacca, R.A.; Tewari, D.; Sobarzo-Sánchez, E.; Nabavi, S.F.; Shirooie, S.; Dehpour, A.R.; et al. Natural products, PGC-1α, and Duchenne muscular dystrophy. Acta Pharm. Sin. B 2020, 10, 734–745. [Google Scholar] [CrossRef] [PubMed]
- Austin, S.; St-Pierre, J. PGC1α and mitochondrial metabolism—Emerging concepts and relevance in ageing and neurodegenerative disorders. J. Cell Sci. 2012, 125, 4963–4971. [Google Scholar] [CrossRef] [PubMed]
- Mah, J.K. Current and emerging treatment strategies for Duchenne muscular dystrophy. Neuropsychiatr. Dis. Treat. 2016, 12, 1795–1807. [Google Scholar] [CrossRef] [PubMed]
- Mah, M.L.; Cripe, L.; Slawinski, M.K.; Al-Zaidy, S.A.; Camino, E.; Lehman, K.J.; Jackson, J.L.; Iammarino, M.; Miller, N.; Mendell, J.R.; et al. Duchenne and Becker muscular dystrophy carriers: Evidence of cardiomyopathy by exercise and cardiac MRI testing. Int. J. Cardiol. 2020, 316, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Dellorusso, C.; Crawford, R.W.; Chamberlain, J.S.; Brooks, S.V. Tibialis anterior muscles in mdx mice are highly susceptible to contraction-induced injury. J. Muscle Res. Cell Motil. 2001, 22, 467–475. [Google Scholar] [CrossRef]
- Barbin, I.C.C.; Pereira, J.A.; Bersan Rovere, M.; de Oliveira Moreira, D.; Marques, M.J.; Santo Neto, H. Diaphragm degeneration and cardiac structure in mdx mouse: Potential clinical implications for Duchenne muscular dystrophy. J. Anat. 2016, 228, 784–791. [Google Scholar] [CrossRef]
- Kostek, M.C.; Gordon, B. Exercise is an Adjuvant to Contemporary Dystrophy Treatments. Exerc. Sport Sci. Rev. 2018, 46, 34–41. [Google Scholar] [CrossRef]
- Frinchi, M.; Morici, G.; Mudó, G.; Bonsignore, M.R.; Di Liberto, V. Beneficial role of exercise in the modulation of mdx muscle plastic remodeling and oxidative stress. Antioxidants 2021, 10, 558. [Google Scholar] [CrossRef]
- Hayes, A.; Williams, D.A. Contractile function and low-intensity exercise effects of old dystrophic (mdx) mice. Am. J. Physiol. Cell Physiol. 1998, 274, C1138–C1144. [Google Scholar] [CrossRef]
- Shefer, G.; Rauner, G.; Yablonka-Reuveni, Z.; Benayahu, D. Reduced satellite cell numbers and myogenic capacity in aging can be alleviated by endurance exercise. PLoS ONE 2010, 5, e13307. [Google Scholar] [CrossRef]
- Abreu, P.; Mendes, S.V.D.; Ceccatto, V.M.; Hirabara, S.M. Satellite cell activation induced by aerobic muscle adaptation in response to endurance exercise in humans and rodents. Life Sci. 2017, 170, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Gordon, B.S.; Delgado Díaz, D.C.; Kostek, M.C. Resveratrol decreases inflammation and increases utrophin gene expression in the mdx mouse model of duchenne muscular dystrophy. Clin. Nutr. 2013, 32, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Yucel, N.; Chang, A.C.; Day, J.W.; Rosenthal, N.; Blau, H.M. Humanizing the mdx mouse model of DMD: The long and the short of it. Npj Regen. Med. 2018, 3, 4. [Google Scholar] [CrossRef]
- Pedrazzani, P.S.; Araújo, T.O.P.; Sigoli, E.; da Silva, I.R.; da Roza, D.L.; Chesca, D.L.; Rassier, D.E.; Cornachione, A.S. Twenty-one days of low-intensity eccentric training improve morphological characteristics and function of soleus muscles of mdx mice. Sci. Rep. 2021, 11, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Gaiad, T.P.; Oliveira, M.X.; Lobo, A.R.; Libório, L.R.; Pinto, P.A.F.; Fernandes, D.C.; Santos, A.P.; Ambrósio, C.E.; Machado, A.S.D. Low-intensity training provokes adaptive extracellular matrix turnover of a muscular dystrophy model. J. Exerc. Rehabil. 2017, 13, 693–703. [Google Scholar] [CrossRef]
- Meadows, E.; Flynn, J.M.; Klein, W.H. Myogenin regulates exercise capacity but is dispensable for skeletal muscle regeneration in adult mdx mice. PLoS ONE 2011, 6, e16184. [Google Scholar] [CrossRef]
- Jin, Y.; Murakami, N.; Saito, Y.; Goto, Y.I.; Koishi, K.; Nonaka, I. Expression of MyoD and myogenin in dystrophic mice, mdx and dy, during regeneration. Acta Neuropathol. 2000, 99, 619–627. [Google Scholar] [CrossRef]
- Chang, N.C.; Chevalier, F.P.; Rudnicki, M.A. Satellite Cells in Muscular Dystrophy—Lost in Polarity. Trends Mol. Med. 2016, 22, 479–496. [Google Scholar] [CrossRef]
- Ribeiro, A.F.; Souza, L.S.; Almeida, C.F.; Ishiba, R.; Fernandes, S.A.; Guerrieri, D.A.; Santos, A.L.F.; Onofre-Oliveira, P.C.G.; Vainzof, M. Muscle satellite cells and impaired late stage regeneration in different murine models for muscular dystrophies. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef]
- Goodwin, M.L.; Jin, H.; Straessler, K.; Smith-Fry, K.; Zhu, J.-F.; Monument, M.J.; Grossmann, A.; Randall, R.L.; Capecchi, M.R.; Jones, K.B. Modeling alveolar soft part sarcomagenesis in the mouse: A role for lactate in the tumor microenvironment. Cancer Cell 2014, 26, 851–862. [Google Scholar] [CrossRef]
- Joanisse, S.; Snijders, T.; Nederveen, J.P.; Parise, G. The Impact of Aerobic Exercise on the Muscle Stem Cell Response. Exerc. Sport Sci. Rev. 2018, 46, 180–187. [Google Scholar] [CrossRef] [PubMed]
- White, J.D.; Scaffidi, A.; Davies, M.; McGeachie, J.; Rudnicki, M.A.; Grounds, M.D. Myotube formation is delayed but not prevented in MyoD-deficient skeletal muscle: Studies in regenerating whole muscle grafts of adult mice. J. Histochem. Cytochem. 2000, 48, 1531–1543. [Google Scholar] [CrossRef] [PubMed]
- Sabourin, L.A.; Girgis-Gabardo, A.; Scale, P.; Asakura, A.; Rudnicki, M.A. Reduced differentiation potential of primary MyoD-/-myogenic cells derived from adult skeletal muscle. J. Cell Biol. 1999, 144, 631–643. [Google Scholar] [CrossRef]
- Yablonka-Reuveni, Z.; Anderson, J.E. Satellite cells from dystrophic (Mdx) mice display accelerated differentiation in primary cultures and in isolated myofibers. Dev. Dyn. 2006, 235, 203–212. [Google Scholar] [CrossRef] [PubMed]
- Hollinger, K.; Gardan-Salmon, D.; Santana, C.; Rice, D.; Snella, E.; Selsby, J.T. Rescue of dystrophic skeletal muscle by PGC-1α involves restored expression of dystrophin-associated protein complex components and satellite cell signaling. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2013, 305, 13–23. [Google Scholar] [CrossRef]
- Chakkalakal, J.V.; Michel, S.A.; Chin, E.R.; Michel, R.N.; Jasmin, B.J. Targeted inhibition of Ca2+/calmodulin signaling exacerbates the dystrophic phenotype in mdx mouse muscle. Hum. Mol. Genet. 2006, 15, 1423–1435. [Google Scholar] [CrossRef]
- Jahnke, V.E.; Van Der Meulen, J.H.; Johnston, H.K.; Ghimbovschi, S.; Partridge, T.; Hoffman, E.P.; Nagaraju, K. Metabolic remodeling agents show beneficial effects in the dystrophin-deficient mdx mouse model. Skelet. Muscle 2012, 2, 1–11. [Google Scholar] [CrossRef]
- Thirupathi, A.; de Souza, C.T. Multi-regulatory network of ROS: The interconnection of ROS, PGC-1 alpha, and AMPK-SIRT1 during exercise. J. Physiol. Biochem. 2017, 73, 487–494. [Google Scholar] [CrossRef]
- Chan, M.C.; Rowe, G.C.; Raghuram, S.; Patten, I.S.; Farrell, C.; Arany, Z. Post-natal induction of PGC-1α protects against severe muscle dystrophy independently of utrophin. Skelet. Muscle 2014, 4, 1–13. [Google Scholar] [CrossRef]
- Arnold, A.-S. PGC-1α régule la jonction neuromusculaire et améliore la dystrophie musculaire de Duchenne. Méd. Sci. 2007, 23, 1034–1036. [Google Scholar] [CrossRef][Green Version]
- Zelikovich, A.S.; Quattrocelli, M.; Salamone, I.M.; Kuntz, N.L.; McNally, E.M. Moderate exercise improves function and increases adiponectin in the mdx mouse model of muscular dystrophy. Sci. Rep. 2019, 9, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Rodino-Klapac, L.R.; Mendell, J.R.; Sahenk, Z. Update on the treatment of duchenne muscular dystrophy. Curr. Neurol. Neurosci. Rep. 2013, 13, 1–11. [Google Scholar] [CrossRef] [PubMed]
- McGreevy, J.W.; Hakim, C.H.; McIntosh, M.A.; Duan, D. Animal models of Duchenne muscular dystrophy: From basic mechanisms to gene therapy. DMM Dis. Model. Mech. 2015, 8, 195–213. [Google Scholar] [CrossRef] [PubMed]
- Botzenhart, U.U.; Gredes, T.; Gerlach, R.; Zeidler-Rentzsch, I.; Gedrange, T.; Keil, C. Histological features of masticatory muscles after botulinum toxin A injection into the right masseter muscle of dystrophin deficient (mdx-) mice. Ann. Anat. 2020, 229, 151464. [Google Scholar] [CrossRef] [PubMed]
- Sacco, A.; Mourkioti, F.; Tran, R.; Choi, J.; Llewellyn, M.; Kraft, P.; Shkreli, M.; Delp, S.; Pomerantz, J.H.; Artandi, S.E.; et al. Short telemeres and stem cell exhaustion model in mdx mice. Cell 2010, 143, 1059–1071. [Google Scholar] [CrossRef]
- Gehrig, S.M.; Koopman, R.; Naim, T.; Tjoakarfa, C.; Lynch, G.S. Making fast-twitch dystrophic muscles bigger protects them from contraction injury and attenuates the dystrophic pathology. Am. J. Pathol. 2010, 176, 29–33. [Google Scholar] [CrossRef]
- Sandri, M.; Lin, J.; Handschin, C.; Yang, W.; Arany, Z.P.; Lecker, S.H.; Goldberg, A.L.; Spiegelman, B.M. PGC-1α protects skeletal muscle from atrophy by suppressing FoxO3 action and atrophy-specific gene transcription. Proc. Natl. Acad. Sci. USA 2006, 103, 16260–16265. [Google Scholar] [CrossRef]
- Spaulding, H.R.; Ludwig, A.K.; Hollinger, K.; Hudson, M.B.; Selsby, J.T. PGC-1α overexpression increases transcription factor EB nuclear localization and lysosome abundance in dystrophin-deficient skeletal muscle. Physiol. Rep. 2020, 8, 1–9. [Google Scholar] [CrossRef]
- Theilen, N.T.; Kunkel, G.H.; Tyagi, S.C. The Role of Exercise and TFAM in Preventing Skeletal Muscle Atrophy. J. Cell. Physiol. 2017, 232, 2348–2358. [Google Scholar] [CrossRef]
- Sartorius, C.A.; Lu, B.D.; Acakpo-Satchivi, L.; Jacobsen, R.P.; Byrnes, W.C.; Leinwand, L.A. Myosin heavy chains IIa and IId are functionally distinct in the mouse. J. Cell Biol. 1998, 141, 943–953. [Google Scholar] [CrossRef]
- Laure, L.; Suel, L.; Roudaut, C.; Bourg, N.; Ouali, A.; Bartoli, M.; Richard, I.; Danièle, N. Cardiac ankyrin repeat protein is a marker of skeletal muscle pathological remodelling. FEBS J. 2009, 276, 669–684. [Google Scholar] [CrossRef] [PubMed]
- Jansen, G.; Groenen, P.J.; Bächner, D.; Jap, P.H.; Coerwinkel, M.; Oerlemans, F.; Van Den Broek, W.; Gohlsch, B.; Pette, D.; Plomp, J.J.; et al. Abnormal myotonic dystrophy protein kinase levels produce only mild myopathy in mice. Nat. Genet. 1996, 13, 316–324. [Google Scholar] [CrossRef] [PubMed]
- Minamoto, V.B. Classificação e adaptações das fibras musculares: Uma revisão. Fisioter. E Pesqui. 2005, 12, 50–55. [Google Scholar] [CrossRef]
- Webster, C.; Silberstein, L.; Hays, A.P.; Blau, H.M. Fast muscle fibers are preferentially affected in Duchenne muscular dystrophy. Cell 1988, 52, 503–513. [Google Scholar] [CrossRef]
- Al-Rewashdy, H.; Ljubicic, V.; Lin, W.; Renaud, J.M.; Jasmin, B.J. Utrophin A is essential in mediating the functional adaptations of mdx mouse muscle following chronic AMPK activation. Hum. Mol. Genet. 2015, 24, 1243–1255. [Google Scholar] [CrossRef]
- Lindsay, A.; Southern, W.M.; McCourt, P.M.; Larson, A.A.; Hodges, J.S.; Lowe, D.A.; Ervasti, J.M. Variable cytoplasmic actin expression impacts the sensitivity of different dystrophin-deficient mdx skeletal muscles to eccentric contraction. FEBS J. 2019, 286, 2562–2576. [Google Scholar] [CrossRef]
- Selsby, J.T.; Morine, K.J.; Pendrak, K.; Barton, E.R.; Sweeney, H.L. Rescue of dystrophic skeletal muscle by PGC-1α involves a fast to slow fiber type shift in the mdx mouse. PLoS ONE 2012, 7, 1–10. [Google Scholar] [CrossRef]
- Campos, G.E.R.; Luecke, T.J.; Wendeln, H.K.; Toma, K.; Hagerman, F.C.; Murray, T.F.; Ragg, K.E.; Ratamess, N.A.; Kraemer, W.J.; Staron, R.S. Muscular adaptations in response to three different resistance-training regimens: Specificity of repetition maximum training zones. Eur. J. Appl. Physiol. 2002, 88, 50–60. [Google Scholar] [CrossRef]
- Landisch, R.M.; Kosir, A.M.; Nelson, S.A.; Baltgalvis, K.A.; Lowe, D.A. Adaptive and nonadaptive responses to voluntary wheel running by mdx mice. Muscle Nerve 2008, 38, 1290–1293. [Google Scholar] [CrossRef]
- Baltgalvis, K.A.; Call, J.A.; Cochrane, G.D.; Laker, R.C.; Yan, Z.; Lowe, D.A. Exercise training improves plantar flexor muscle function in mdx Mice. Med. Sci. Sports Exerc. 2012, 44, 1671–1679. [Google Scholar] [CrossRef]
- Cheng, X.; Song, L.; Lan, M.; Shi, B.; Li, J. Morphological and molecular comparisons between tibialis anterior muscle and levator veli palatini muscle: A preliminary study on their augmentation potential. Exp. Med. 2018, 15, 247–253. [Google Scholar] [CrossRef] [PubMed]
- Schill, K.E.; Altenberger, A.R.; Lowe, J.; Periasamy, M.; Villamena, F.A.; Rafael-Fortney, J.A.; Devor, S.T. Muscle damage, metabolism, and oxidative stress in mdx mice: Impact of aerobic running. Muscle Nerve 2016, 54, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]
- Curran-Everett, D. Explorations in statistics: The log transformation. Adv. Physiol. Educ. 2018, 42, 343–347. [Google Scholar] [CrossRef] [PubMed]
| Pathological Changes/Groups | WT-SED (%) | WT-TR (%) | mdx-SED (%) | mdx-TR (%) |
|---|---|---|---|---|
| Nuclear centralization | 50 * | 83 * | 100 | 100 |
| Splitting | 16 * | 66 * | 100 | 100 |
| Variation in size | - | 66 | 100 | 100 |
| Basophilic fibers | - | - | 83 * | 66 * |
| Necrosis | - | - | 100 * | 100 * |
| Increased connective tissue | - | 16 * | 33 * | 50 * |
| WT-SED | WT-TR | mdx-SED | mdx-TR | |
|---|---|---|---|---|
| FTIIA | 24.95 * | 24.36 | 20.00 | 18.96 |
| 24.40–25.50 | 23.72–25.01 | 18.67–21.32 | 18.08–19.84 | |
| FTIIAD/DA | 29.63 | 29.93 | 25.07 | 21.81 |
| 28.46–30.80 | 28.44–31.42 | 21.30–28.84 | 20.36–23.25 | |
| FTIID | 27.12 * | 25.33 | 21.34 | 25.76 * |
| 25.87–28.37 | 24.38–26.28 | 19.82–22.85 | 24.21–27.30 | |
| FTIIDB/BD | 29.96 | 25.94 † | 27.09 | 29.63 |
| 28.50–31.42 | 24.23–27.45 | 25.63–28.55 | 27.82–31.45 | |
| FTIIB | 35.51 * | 33.46 † | 32.06 | 34.29 * |
| 34.92–36.11 | 32.90–34.02 | 31.36–32.75 | 33.42–35.15 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sigoli, E.; Antão, R.A.; Guerreiro, M.P.; de Araújo, T.O.P.; Santos, P.K.d.; da Roza, D.L.; Rassier, D.E.; Cornachione, A.S. Effects of Low-Intensity and Long-Term Aerobic Exercise on the Psoas Muscle of mdx Mice: An Experimental Model of Duchenne Muscular Dystrophy. Int. J. Mol. Sci. 2022, 23, 4483. https://doi.org/10.3390/ijms23094483
Sigoli E, Antão RA, Guerreiro MP, de Araújo TOP, Santos PKd, da Roza DL, Rassier DE, Cornachione AS. Effects of Low-Intensity and Long-Term Aerobic Exercise on the Psoas Muscle of mdx Mice: An Experimental Model of Duchenne Muscular Dystrophy. International Journal of Molecular Sciences. 2022; 23(9):4483. https://doi.org/10.3390/ijms23094483
Chicago/Turabian StyleSigoli, Emilly, Rosangela Aline Antão, Maria Paula Guerreiro, Tatiana Oliveira Passos de Araújo, Patty Karina dos Santos, Daiane Leite da Roza, Dilson E. Rassier, and Anabelle Silva Cornachione. 2022. "Effects of Low-Intensity and Long-Term Aerobic Exercise on the Psoas Muscle of mdx Mice: An Experimental Model of Duchenne Muscular Dystrophy" International Journal of Molecular Sciences 23, no. 9: 4483. https://doi.org/10.3390/ijms23094483
APA StyleSigoli, E., Antão, R. A., Guerreiro, M. P., de Araújo, T. O. P., Santos, P. K. d., da Roza, D. L., Rassier, D. E., & Cornachione, A. S. (2022). Effects of Low-Intensity and Long-Term Aerobic Exercise on the Psoas Muscle of mdx Mice: An Experimental Model of Duchenne Muscular Dystrophy. International Journal of Molecular Sciences, 23(9), 4483. https://doi.org/10.3390/ijms23094483






